Mobile technologies offer potential uses across the life span, even in the absence of symptoms in at-risk individuals, said Dror Ben-Zeev, professor of psychiatry and behavioral sciences at the University of Washington (see Figure 5-1). For example, parents with severe mental illnesses or substance use disorders are vulnerable themselves, and thus they may contribute to a biological and environmental context that places their children at risk for mental health challenges. Deploying mobile health interventions that focus on mental health promotion, education, or parenting skills can have utility in this situation, said Ben-Zeev. Then, as a child with mental illness gradually starts showing signs of illness, mobile technologies may also play a beneficial role through the use of targeted online forums, analysis of social media content, and clinically relevant or diagnostic gaming, he said. Since the penetration rate of social media and gaming is very high in adolescents, these technologies may enable identification of biobehavioral markers at an early stage, he said.
The first episode of a mental illness can be an overwhelming and confusing time for those affected and their families as they struggle with symptoms, the complexity of the health care system, stigma, etc. As a result, clinical research using mobile health interventions has been relatively limited during this illness stage, but that can change, said Ben-Zeev. However, once symptoms are under control, mobile health technologies can be useful in helping people focus on their illness as well as their strengths to optimize symptom management and to think about vocational and psychosocial rehabilitation.
Lara Mangravite, president of Sage Bionetworks, commented that 5 or 10 years may pass before these digital technologies achieve the goal of transforming clinical care by not only monitoring and managing disease, but also understanding what it takes to be healthy in real-world settings through a combination of pharmaceutical interventions, lifestyle and environmental changes, and an overall focus on wellness.
Mangravite described one example of the kind of technology currently being studied at Sage—a smartphone-based application called mPower1—which she said has the potential to transition a traditional clinic visit from a place where acute testing is done to a place where the clinician and patient together review longitudinal data that have been collected over time. Alternatively, the data could be evaluated remotely and form the basis for
___________________
1 To find out more about mPower, see https://parkinsonmpower.org (accessed July 29, 2018).
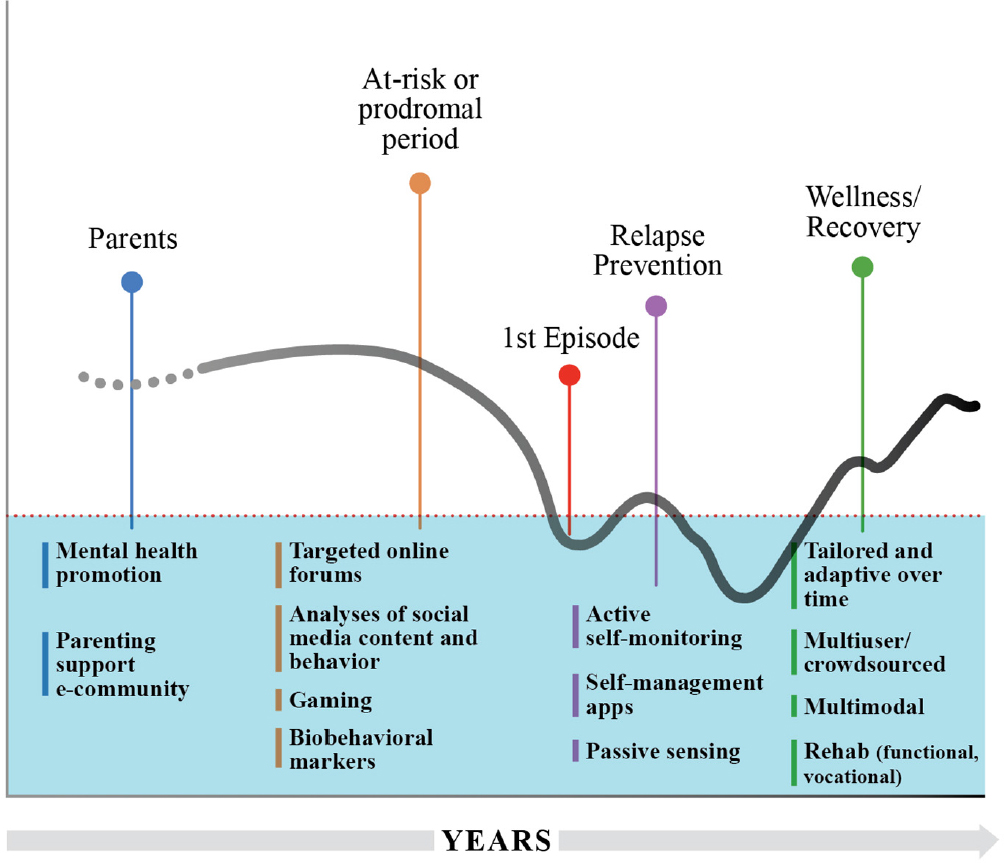
SOURCE: Adapted from materials presented by Ben-Zeev, June 6, 2018.
determining whether a clinic visit is necessary or even enable the clinician to remotely administer treatments or promote self-management.
Ben-Zeev and colleagues have conducted a randomized controlled trial of FOCUS, a smartphone-based intervention application for self-management of schizophrenia, versus a patient (peer) group intervention called the Wellness Recovery Plan. Both interventions improved clinical outcomes like psychiatric symptoms and recovery, in a comparable manner. Both interventions produced high patient satisfaction ratings. But, a significantly greater percent of patients who were randomized to the mHealth intervention actually commenced treatment (90 percent) compared to those assigned to the clinic intervention (58 percent) (Ben-Zeev et al., 2018). These data suggest that remote management of health care
can bring together the patient, caregiver, community, and clinicians over the course of a treatment plan, according to Mangravite.
Vaibhav Narayan suggested that there might be lower barriers to conducting this type of research in other parts of the world. Husseini Manji said Janssen has considered conducting work in parts of Germany and the United Kingdom that have more closed health care systems. Implementing and debugging these types of technologies may be possible before bringing them to the United States, he said. However, Ben-Zeev noted that different regulatory standards, clinical practices, and technological infrastructure may pose difficulties in terms of how much can be learned and applied to the U.S. context.
CLINICIAN USE OF MOBILE HEALTH TECHNOLOGIES
Clinicians vary in their acceptance, comfort, and willingness to use mobile health interventions in practice. The ideal but clearly non-existent hypothetical clinician, conjured up by Ben-Zeev for demonstration purposes, is a digital native who is already familiar with mobile technology; has formal mobile health training; keeps up with the latest interventional research; and is highly motivated, incentivized by his or her organization to deploy mobile health technologies, excited about multidisciplinary teamwork, a critical thinker, and an avid user of data. Such clinicians also have the time, patience, and capacity to learn, grow, and expand their clinical expertise to improve patient outcomes in a deep, meaningful, and sustainable manner.
In the real world, however, Ben-Zeev and colleagues have found that clinicians are often too busy to explore the use of mobile technologies. They want to know if digital data can be found in the electronic health records they are already required to integrate into their practices. They may resent having to incorporate the new technologies into their care plans because of the increased requirements and documentation. They have concerns about being sued, new training and certification requirements, and lack of experience with statistical data, said Ben-Zeev. Furthermore, they want to know if the technology will improve patient outcomes.
Steven Hyman raised the additional concern that reliance on computationally derived phenotyping could reduce face-to-face interactions between physicians and patients. Husseini Manji, however, suggested that if these technologies are implemented correctly, they could improve the quality of patient interaction with the clinical team because people could
come to their appointments armed with data on how they have been doing, thus enabling the conversation to move more quickly to more substantive issues. Moreover, said William Marks, in the future if physicians are able to monitor their patients remotely, they could focus their attention on those patients who are most in need, eliminating the trip to the clinic.
On top of clinician and clinical staff resistance to using mobile technologies, there is a gap between what researchers think is good for a subject and what the subjects may actually want, noted Daniela Brunner. One way to overcome this, she said, is to focus on educating people about what is available and then having them request it from their clinicians. Mangravite agreed that in the short term, people asking their physicians for these devices and apps is the main way they will penetrate the market. Indeed, said Ben-Zeev, there has been a pendulum swing in psychiatry toward more patient-focused care and shared decision making. In the digital health world, this has led to products specifically designed to inform patients on how to interact with their clinicians to ensure that their voices are heard. In the development of tools, this means integrating people with lived experience and their family members right from the beginning of the process, he said. For example, he recalled that in developing a mobile health intervention for people with schizophrenia, a topic that came up in focus groups with patients and family members was content focusing on sleep. Ben-Zeev said if patients and family members had not called this topic to their attention, he would never have thought of integrating it into a mobile health intervention for schizophrenia because it is not a core symptom.
HEALTH CARE SYSTEMS’ INTEGRATION OF MOBILE TECHNOLOGY
User-centered design means more than just the patient and clinician, said Ben-Zeev; it also includes the health care system and the deployment context. Thus, even if there are compelling clinical data to support the use of mobile health interventions, they may not make sense if providers, health care systems, and payers do not embrace their use.
According to Ben-Zeev, health organizations’ concerns include: How much will it cost to integrate these technologies and who will pay for it? Will it generate new revenue or savings? Does our organization have sufficient information technology capacity to use these tools, service and upgrade software, and provide adequate privacy and data security? Can our
culture absorb this new technology successfully? How will this technology integrate into our existing services and workflow without being disruptive?
The much faster pace of technology development in comparison with health care delivery presents a major challenge to the integration of these technologies into clinical care, said Mangravite. She suggested that change is likely to come from outside of the traditional system. As an example, she cited Propeller Health,2 a stand-alone company that provides services using Bluetooth-enabled inhalers to monitor patients with asthma. Propeller integrates data from the inhalers with patient reports on the context in which they are experiencing episodes as a means of helping patients to understand what is triggering these events and how they might avoid them. It even goes so far as to alerting people when particulate matter is above their threshold, suggesting they may want to exercise inside, said Mangravite.
Propeller provides universal benefits across stakeholder groups, said Mangravite. Clinicians can prescribe Propeller, but do not have to manage it. It reduces caregiver burden. For insurers, it reduces costs associated with emergency room visits. Mangravite suggested that these tools may also strengthen the relationships among clinicians, caregivers, and patients, allowing them to interact in a more holistic way. She added that although continuous monitoring may have benefits for people with chronic diseases, there is a different value proposition for healthy individuals, who first need to understand what they are getting into, what the value is, and what the potential consequences are.
The use of mobile technologies may also support a gradual shift among payers to outcome-based rather than fee-for-service–based reimbursement models, said Ben-Zeev. He suggested that payers may be willing to accept objective indicators of functional outcomes as pragmatic indicators of success, for example, if measures show that someone with a psychiatric disability is more socially engaged or is spending more time in the workplace. The promise is there, he said, although these technologies are in a very early stage of development and may not have been integrated into regular practice yet. This points to the necessity of engaging representatives of health care systems, providers, and caregivers and these early focus groups and usability testing projects, said Ben-Zeev. Lisa Holt of Intelligent Automation added that researchers should also be included in these focus groups to ensure that clinicians and patients are willing to give them the quality data they need in their studies to advance the science.
___________________
2 For more information about Propeller Health, see https://www.propellerhealth.com (accessed July 16, 2018).
The Health Information Technology for Economic and Clinical Health (HITECH) Act, signed into law in 2009, aims to promote the broad adoption and meaningful use of health information technology to improve health care.3 Ben-Zeev and colleagues have proposed inventing a new breed of health care professionals called “clinical technology specialists” as one mechanism to integrate these technologies in existing health care systems (Ben-Zeev et al., 2015). Not necessarily physicians or other licensed practitioners, these professionals would be embedded within a health care system, have both patient- and provider-facing functions, and be familiar with the range of digital health options available. They could assess and educate patients, provide technical troubleshooting, and support engagement over time, all while relieving busy clinicians of these responsibilities. They could also educate and train clinicians on the functions and resources relevant to their work, said Ben-Zeev.
___________________
3 For more information about the HITECH Act, see https://www.hhs.gov/hipaa/forprofessionals/special-topics/hitech-act-enforcement-interim-final-rule/index.html (accessed July 16, 2018).
This page intentionally left blank.








