2
THE NEED FOR CONTAINMENT LABORATORIES
NEEDS AND PURPOSES FOR BIOLOGICAL LABORATORIES IN LOW-RESOURCE SETTINGS
Throughout the workshop, several speakers addressed why biological containment laboratory capacity is needed. These include safe handling of potential pandemic pathogens (PPPs) and other infectious agents in research and medicine; making accurate and rapid diagnoses for PPPs and other infectious agents to ensure appropriate medical care; detecting antimicrobial resistance in infectious agents; facilitating epidemiological investigations of infectious disease outbreaks; and detecting biological attack agents. During his presentation on the need for containment laboratories, Tom Ksiazek, DMV, used the above list as a guide, reframing the needs as follows:
- Diagnostics—identifying etiology
- Supporting clinical care—identifying cases
- Supporting epidemiology
- identifying cases
- identifying chains of transmission using modern molecular tools
- Supporting and carrying out ecological investigations
- Applied research
- Testing and evaluating therapeutics—preclinical studies
- Testing and evaluating vaccines
- Basic research—developing pathways and targets for therapeutics and vaccines
Dr. Ksiazek, who has extensive experience working in settings ranging from rudimentary field laboratories to state-of-the-art research, focused on
alignment of the containment level with the laboratory purpose and the issues that funders must address when considering a laboratory project.
Basic and applied research are better and more safely performed in traditional high-containment labs in higher resource environments. But there remains a need to provide public health services and medical treatment, such as diagnostics and epidemiological work, in low-resource settings. This fact creates a dilemma for policy makers who must balance the need to improve public health with the need to prevent the use of hazardous pathogens as weapons of mass destruction. In the United States, only high-containment laboratories can conduct research with Biological Select Agents and Toxins (BSAT). These laboratories devote almost one-third of their budgets to meet the associated security requirements, which contributes to the extreme expense of doing such research. He noted, however, that the practicalities are that simpler labs in low-resource settings are capable of handling clinical and environmental samples for public health.
Diagnostic testing regimes can be carried out safely in “first tier” (non-BSL-3 and -4) facilities by trained and experienced personnel. Dr. Ksiazek stated that first tier labs usually do not cache or work with agents in quantities beyond what they receive in clinical or environmental samples and usually inactivate the materials they receive. This theme emerged throughout the workshop: that is, the importance of identifying the purpose of the laboratory when opening discussions about containment and safety and security. Public health–motivated testing in lower level laboratories does, however, require sustained adequate support for facilities, equipment, and training to protect the workers and others.
The field of etiology, that is, the branch of science concerned with the causes and origins of diseases, is a crucial component of safeguarding public health. Dr. Ksiazek stated that the failure to recognize the cause of serious disease in low-resource settings is partially due to a lack of diagnostic capabilities. This is problematic, because early direction of resources is very important in the fight against serious pathogens. The International Health Regulations (IHR) required rapid recognition and identification of PPPs after the epidemic of Severe Acute Respiratory Syndrome (SARS) in 2004. Delays in recognizing the serious nature of the early stages of the West African Ebola outbreak in 2014 are often cited as the reason for the disease’s uncontrolled spread by the time it was accurately identified. However, Dr. Ksiazek suggested that a lack of diagnostic facilities was not altogether to blame. Even if a laboratory exists, if it is not operated daily, then the personnel who know how to operate it may not be available in a time of crisis. In other words, the
prevailing local conditions may influence a laboratory’s ability to fulfill its purpose.
National or regional facilities can also support rapid recognition. In-depth comparisons of outbreak agents with reference strains, both by molecular and classical techniques, are highly desirable. Therefore, the early engagement of reference labs with appropriate containment capacity is desirable. However, moving samples to and from reference laboratories is becoming more and more problematic and restricted by regulations and security concerns. Ksiazek stated that as a result, it is now almost impossible for reference laboratories to obtain export/import licenses to share pathogens, which hinders not only research, but also the pursuit of potential regional approaches. In addition, although organism strain genome sequences are documented, the strains themselves are often not appropriately preserved. Consequently, the development of treatment options becomes more difficult, because the sequences alone are insufficient for the necessary research on countermeasures.
In addition, shippers are reluctant to handle pathogens. Federal Express, for example, ships 6 million packages per day through Memphis, Tennessee. However, because an Army laboratory inadvertently sent live samples rather than inactivated samples of Bacillus anthracis (anthrax) to scores of other laboratories, the company announced in July 2015 that it will no longer ship research samples of Select Agents through its regular shipping service. This leaves only very specialized, dedicated delivery services (e.g., FedEx Custom Critical) to deliver such samples, which costs orders of magnitude more than do ordinary shipments.
Dr. Ksiazek discussed the need for risk assessment. He separated risk into personal risk and environmental risk. Personal risk pertains to whether the pathogen is infectious to humans or a hazard for laboratory workers (through punctures or aerosol exposures) and whether vaccines and/or treatment options are available. Environmental risk pertains to whether a pathogen is contagious, indigenous, aerosol infectious, or agricultural. He presented a graph that plots personal protection against environmental containment, showing how the various biological safety levels are distributed along these two metrics (Figure 2.1). “Inside containment” requires physical barriers, animal caging systems, laminar flow hoods, centrifuge carriers, and air exchange and gradients, with the primary personal barrier being positive pressure suits. “Environmental containment” at a typical BSL-4 includes HEPA filtration and constant negative pressure for air handling, decontamination of lab equipment and of waste (autoclaving), personnel and biological samples, and lab effluents. He explained that high-containment (BSL-3 and -4) laboratories
are neither portable nor affordable for many low-resource settings. Most pathogens studied in high-containment labs in developed countries are not indigenous to the locations of these labs.
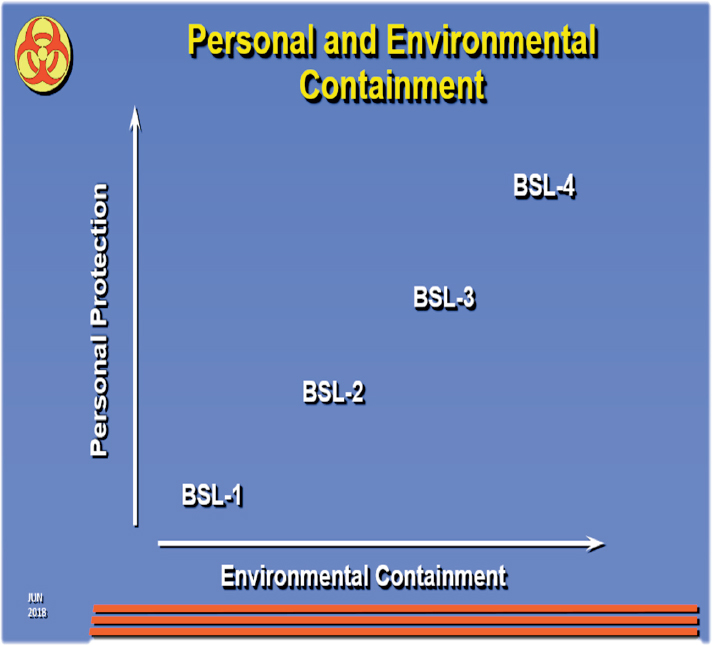
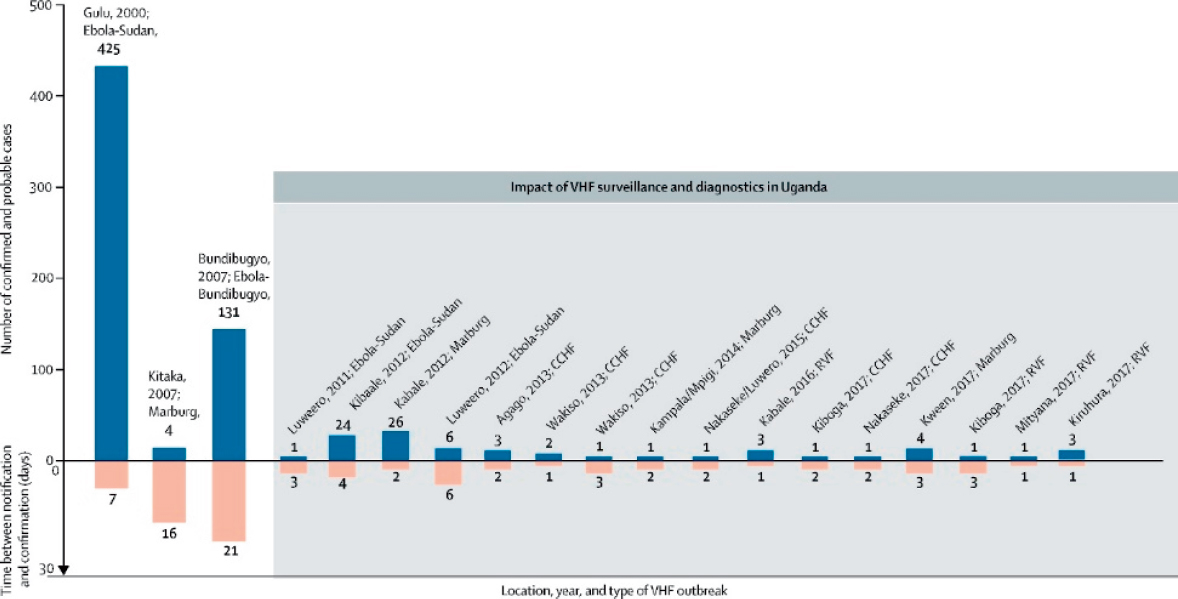
To conduct field studies or respond to epidemics, a researcher must travel to the environments where they exist. Dr. Ksiazek stated that there is no point in operating a BSL-4 facility for containment to prevent a pathogen from escaping to the environment if infectious patients or the organisms themselves are 20 paces away. As an example, he cited a paper by personnel from the Viral Special Pathogens Branch of the U.S. Centers for Disease Control and Prevention that described the field lab they established in Bo, Sierra Leone, during the 2014 Ebola outbreak (Flint et al., 2015). Although not a BSL-3 or BSL-4 facility, this laboratory was adequate to process more than 12,000 specimens from throughout Sierra Leone. He also cited a 2018 article (Shoemaker et al., 2018) on the impact
of enhanced viral hemorrhagic fever (VHF) surveillance on outbreak detection and response in Uganda (Figure 2.2). This paper showed that surveillance leading to early detection and outbreak responses in turn led to a significant decrease in intensity and duration of VHF outbreaks in Uganda. This successful project can serve as a role model for detecting and responding to international health threats.
Dr. Ksiazek also noted that the World Health Organization (WHO) Laboratory Biosafety Manual (LBM), now under revision, will follow a risk-based approach. Diagnostic laboratories can also be used to detect antibiotic resistance using molecular techniques if the resistance mechanisms and markers are known. These techniques would probably not require high-level containment. To detect biological attack agents and to distinguish engineered agents from naturally occurring outbreaks, researchers need epidemiological information to sort outbreak situations and early sequence data to assess what species or strain the attack agent is most closely related to. Depending on the nature of the agents, such situations may require either tier 1 or regional reference labs or both.
During the discussion following Dr. Ksiazek’s presentation, a participant asked whether compliance with the IHR requires BSL-3 capabilities to fulfill surveillance obligations. Dr. Ksiazek replied that a country would not need to operate a BSL-3 laboratory but would need to enter into some arrangement to access a BSL-3 laboratory when needed. He suggested that the possibility of obtaining BSL-3 lab capabilities to supplement the capabilities of existing lower-level labs might be an enticement for developing countries to comply with the IHR. Another participant stated that the risks associated with the work expected to be performed in a new or enhanced laboratory should drive the decision about what type of laboratory is really needed in that particular place. Because it may not be a BSL-3 lab, it might be preferable to move away from use of the term “BSL-3” as a descriptor. However, another participant noted that politicians in low-resource settings may ask for the highest tech facilities to bolster national pride without understanding the complexity and costs associated with such laboratories.
REVISION OF THE WHO LABORATORY BIOSAFETY MANUAL (LBM)
Dr. Kazunobu Kojima presented on the revision of the WHO LBM, which he is leading. The current version (third edition) was published in 2004 and is outdated, given the pace of the science and technology for infectious disease. WHO member states need an up-to-date manual to guide establishment, use, and dismantlement of needed laboratories. The IHR, which are legally binding, require WHO member states to develop minimum core national and international surveillance and to report capacities. Core Capacity 8 of the IHR pertains to laboratories and calls for policy and coordination, diagnostic capacity, laboratory biosafety and biosecurity, and laboratory-based surveillance. These regulations thus oblige member nations to have certain minimum public health-related capabilities in place, which translates to some form of biological laboratory. Dr. Kojima described how this requirement may be met without establishing laboratories with high containment levels where appropriate.
Adding to Dr. Ksiazek’s comments about risk assessment, Dr. Kojima noted that the LBM revision is meant to follow an “evidence-based” and “risk-based” approach, as the best way to inform the risk assessment process and policy instruments, allow logical prioritization to avoid overkill and overdesign of lab facilities, and learn from actual incidents to prevent recurrences. These considerations should facilitate resource optimization.1 He also discussed the findings from “Surveillance of laboratory exposures to human pathogens and toxins: Canada 2016.”2 In 2016, a total of 100 lab workers were accidentally exposed with no reports of secondary exposures. Most incidents (greater than 90 percent) occurred in risk group 2 or biocontainment safety level 2 (RG2 or BSL-2) facilities. The causes were failure to follow standard operating procedures (72 percent) and equipment failure (17 percent). These statistics illustrate the importance of having, and following, biosafety requirements. Regarding
___________________
1 Dr. Kojima recommended that the participants read “Evidence-Based Biosafety: A Review of the Principles and Effectiveness of Microbiological Containment Measures” by Kimman et al. (2008).
2 Bienek, A. M. Heisz, and M. Su. 2017. “Surveillance of laboratory exposures to human pathogens and toxins: Canada 2016.” Canada Communicable Disease Report 43-11:228-235.
risk tolerance, he asked “Who can decide what sort of capacity and authority is needed and on what grounds?”
Dr. Kojima defined “risk” as the sum of the severity of hazard plus the likelihood of exposure to the hazard. He noted that implementation of Good Microbiological Practices and Procedures (GMPP) depends on training rather than engineering controls. The Canadian data on laboratory accidents showed that the most well-designed and engineered laboratory is only as good as its least-trained worker, and that human factors are generally the cause of laboratory-acquired infections rather than malfunction of engineering controls. However, “risk (hazard) group” does not equate to biosafety level.
There are a variety of factors determining the consequences of an exposure to a pathogen, including:
- Low infectious dose
- High communicability
- Airborne route of transmission
- Availability of preventive or therapeutic treatment
- History of laboratory-acquired infection
- Exotic epidemiology (non-endemic)
- High susceptibility of population (e.g., immunocompromised, naïve)
These factors increase the severity and mortality resulting from pathogen exposure. Certain laboratory procedures increase the likelihood of exposure, as follows:
- Producing and using large volumes and high titers
- Following procedures that might generate aerosols (e.g., sonication, or deliberate generation of aerosols)
- Infecting animals
- Using sharps
- Necropsy where infection is suspected
- Increasing virulence
In contrast, some procedures present a low risk for exposures:
- Use of agar plates (e.g., streaking, spreading)
- Serial dilution
- Preparing/staining slides
- Nucleic acid extraction
- Inactivation
- Use of autoanalysers
- Enzyme-linked immunosorbent assay (ELISA)
- Polymerase chain reaction (PCR)
- Rapid diagnostic tests.
Dr. Kojima showed a graph that plots the consequences of infection against the likelihood of exposure. The revised WHO LBM will replace risk groups and biosafety levels with a thorough risk assessment and appropriate risk mitigation and control measures based on the consequence of infection from the pathogen and the risks associated with the procedures to be performed. Activities that have only “core requirements,” the minimum requirements for safely executing the majority of lab procedures, because there is a low process risk and low consequence of infection. The core requirements include codes of conduct, competent and appropriately trained staff, and GMPP, and are fundamental to safe work practices in any facility.
Most work can be performed using this set of minimum core requirements. Some work will require heightened control measures (e.g., a biological safety cabinet or extra personal protective equipment, segregated work area, task-specific equipment, or a combination). Heightened control measures are required with increased risk. Very few procedures will require high containment (i.e., BSL-4) control measures. Examples include working with eradicated diseases, such as smallpox, or using procedures that entail a high likelihood of worker exposure and/or release to the environment. This risk-based approach offers much needed flexibility to the way risk is controlled.
The revision of the WHO LBM seeks to produce a central core document with additional detailed monographs on risk assessment, biosafety program management, laboratory design and maintenance, biological safety cabinets, personal protective equipment, decontamination and waste management, and emergency outbreak response. Dr. Kojima circulated a recent news article from Science called “Risk-based reboot for global lab biosafety,”3 which discusses the revision of the manual that he is working on.
Finally, Dr. Kojima discussed the high heterogeneity of the regulatory situation among Member States. Some countries, such as the United
___________________
3 For more information, see Kojima, K., Makison Booth, C., Summermatter, K., Bennett, A., Heisz, M., Blacksell, S.D., and McKinney, M. 2018. “Risk-based reboot for global lab biosafety: New WHO guidance could expand access to lab facilities.” Science 360:260-262.
States, are highly regulated and have detailed biosafety and biosecurity legislation and regulations with well-defined responsibilities and processes. Other countries almost completely lack regulatory guidance in the form of legislation, standards, and regulations. WHO will undertake a new project to analyze the biosafety and biosecurity legislative framework of different WHO Member States and to develop a proposal for a harmonized international approach to ensure state-of-the art legislation for biosafety and biosecurity in biomedical laboratories.










