1
Introduction and Background1
Neurological and psychiatric disorders impose a tremendous human and economic burden on individuals, families, and societies, affecting more than 1 billion people globally and costing approximately $1.5 trillion per year in the United States alone (Feigin et al., 2017; Nager and Atkinson, 2016). Despite the significant unmet medical needs and large market potential, the development of new therapeutics in this area lags behind other diseases, and research and development are often characterized by costly, late-stage clinical trial failures (Hyman, 2012, 2016).
Although a number of factors contribute to this stalled therapeutic development, the challenge of translating scientific discoveries from rodent models to humans has been a major factor in slowing the development of new therapies for brain disorders, said Guoping Feng, the James and Patricia Poitras Professor of Neuroscience at the McGovern Institute for Brain Research, Massachusetts Institute of Technology. While research with rodent models has led to significant medical discoveries and increased fundamental understanding of brain function, pathology, and disease pathogenesis through basic science research, there are limitations to their use in studying human nervous system disorders, including the vast differences in brain structure and their inability to model many aspects of normal human cognition and behavior (IOM, 2013). About 93 percent of
___________________
1 The planning committee’s role was limited to planning the workshop, and the Proceedings of a Workshop was prepared by the workshop rapporteurs as a factual summary of what occurred at the workshop. Statements, recommendations, and opinions expressed are those of individual presenters and participants, and have not been endorsed or verified by the National Academies of Sciences, Engineering, and Medicine. They should not be construed as reflecting any group consensus.
drugs for nervous system disorders that show efficacy in rodent models fail in human clinical trials (Kola and Landis, 2004). Given the many recent failures of rodent-based treatments for neurological disease to translate to humans in clinical trials, neuroscientists are giving increasing thought to approaches to therapeutic development that do not rely on rodent models (see, e.g., IOM, 2014, and NASEM, 2017).
To address this challenge, the field needs to expand its bandwidth and think of new ways to conduct experiments and study disease, said William Newsome, the Vincent V. C. Woo Director of the Stanford Neurosciences Institute. Working with nonhuman primates and using powerful transgenic tools holds great promise in this regard, he added. One might ask whether the use of nonhuman primates for research is essential and necessary in order to better understand and develop therapies for neurological and psychiatric diseases. “We don’t know the answer to that fundamentally because we can’t predict the future,” said Newsome. “But what is essential is that we have a more diverse scientific ecosystem and a more diverse set of approaches to start understanding the biology underlying these diseases and how we might creatively take approaches to treat them.” Because nonhuman primates are much closer to humans from an evolutionary perspective—and therefore have more similar cognitive and behavioral functions, social cognition, and neuroanatomical organization (Belmonte et al., 2015; Jennings et al., 2016; Kaas, 2013)—Newsome suggested that increasing the use of animals such as marmosets and macaque monkeys for research is both justified and necessary.
The development of powerful transgenic tool applications—including transgenesis, viral gene delivery, genome editing, and cloning—in nonhuman primate models opens new potential opportunities to significantly advance neuroscience research and therapeutic development (see Okano and Kishi, 2018), and also to introduce scientific and bioethical questions that merit deep consideration.2 To examine the promise, concerns, and challenges related to neuroscience research using genetically modified nonhuman primates, the Forum on Neuroscience and Nervous System Disorders hosted a public workshop on October 4, 2018, bringing together an international
___________________
2 In September 2016, the National Institutes of Health hosted a congressionally requested workshop on Ensuring the Continued Responsible Oversight of Research with Non-Human Primates to review the appropriateness of research in nonhuman primates, including rationale and guidelines. For more information, go to https://osp.od.nih.gov/pastevent/nih-workshop-on-ensuring-the-continued-responsible-oversight-of-research-with-nonhuman-primates (accessed December 13, 2018).
group of experts and stakeholders representing academia, industry, laboratory animal management, disease-focused foundations, and federal agencies.
WORKSHOP OBJECTIVES
The workshop was designed to explore the current state and future promise of research using genetically modified nonhuman primate models of disease to understand the complex functions of the brain that control behavior, movement, and cognition in both health and disease states, said Frances Jensen, professor and chair of neurology at the Perelman School of Medicine, University of Pennsylvania (see Box 1-1). Many of these complex functions and systems cannot be replicated in a lower species such as a rodent, she said. Yet, the field must still grapple with the question of what merits taking the step of developing nonhuman primate models with genetic modifications, and if these models are deemed to be appropriate and essential, how the field will ensure the appropriate use of this extremely unique resource, said Jensen. For a comprehensive look at the issues, the workshop considered some of the distinct aspects of animal husbandry required to ensure optimal care of genetically modified nonhuman primates; the ethical considerations related to the use of these animals in research and the global regulatory and conduct codes that are in place or that need to be developed; and the infrastructure required worldwide to ensure that research achieves its greatest scientific impact through collaboration and partnerships. Finally, said Jensen, given the fact that the nonhuman primate research community is and will continue to be relatively small and specialized, attention must be given to training the next generation of scientists to continue working with these unique animals (see Figure 1-1). Workshop presentations primarily focused on models using the common marmoset, Cynomolgus macaque, or Rhesus macaque. Debating whether transgenic and chimeric nonhuman primate research should be undertaken was not within the scope of this workshop.
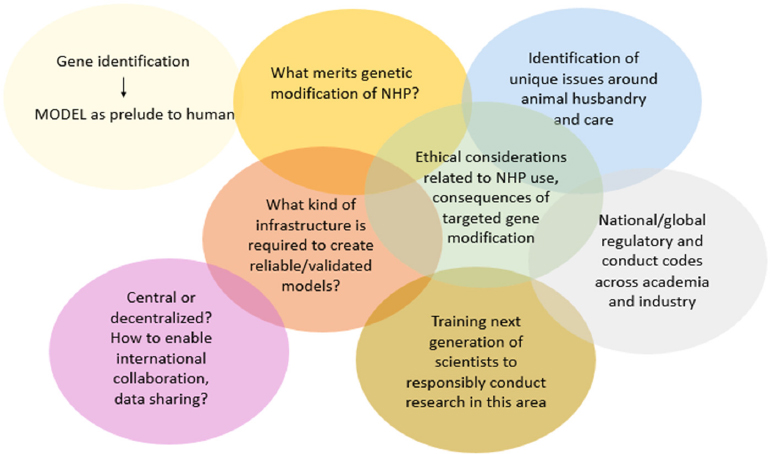
NOTE: NHP = nonhuman primate.
SOURCE: Presented by Frances Jensen, October 4, 2018.
ORGANIZATION OF THE PROCEEDINGS
Chapter 2 explores the rationale for developing genetically modified nonhuman primate models of nervous system disorders and provides an overview of challenges and opportunities associated with this area of research for academia, industry, and the public. Chapter 3 describes the development of several nonhuman primate models of human diseases using genetic modification and highlights a case study of how viral delivery of genes was used to develop the first successful gene therapy of a central nervous system disease. Chapter 4 discusses several approaches that have enabled the translation of discoveries from nonhuman primates to humans. Ethical issues related to nonhuman primate research are explored in Chapter 5. Chapter 6 focuses on the policy, infrastructure, and funding requirements to build and sustain a robust research enterprise, as well as other potential opportunities to move the field forward. Chapter 7 provides the final remarks.
This page intentionally left blank.






