2
Is the World Ready to Respond to the Next Influenza Pandemic?
The half-day pre-workshop event opened with welcome remarks from Victor Dzau, president of the National Academy of Medicine (NAM), who provided an overview of the progress, challenges, and opportunities for preparing and responding to the next influenza pandemic. Following his remarks, Laurie Garrett, science journalist and founder of the Anthropos Initiative, delivered a keynote presentation, setting the stage with an overview of historical influenza pandemics. In particular, Garrett explored the 1918 influenza pandemic and the 2009 H1N1 pandemic from both cultural and epidemiological perspectives and examined the potential impact of a severe influenza pandemic under today’s conditions.
OPENING REMARKS
In his opening remarks, Victor Dzau, president of the NAM, stated that 2018 marks the 100th anniversary of one of the deadliest disease outbreaks in human history: the 1918 influenza pandemic. Since then, scientific and medical advances in antibiotics, diagnostics, vaccines, and other areas have enabled the international community to better prepare for infectious disease outbreaks. However, outbreaks have continued to cause devastating outcomes over the years, and emerging infectious disease events are increasing significantly over time (Jones et al., 2008). According to Dzau, “The world is ill-prepared for another influenza pandemic.”
Dzau pointed out that the 2014–2016 Ebola outbreak in West Africa was a wakeup call with failures at all response levels: local, national, regional, and international (GHRF Commission, 2016). Ultimately, the
epidemic was contained, but the costs in human lives and in economic and social disruption were significant with more than 11,000 deaths. Most recently, the Ebola outbreak in the Democratic Republic of the Congo has resulted in more than 500 deaths so far.1 The ongoing conflict in the region has complicated this outbreak and has caused tremendous concern within the international community, he said. Although Ebola kills thousands of people and leads to devastating consequences, the health outcomes could be even worse if Ebola were airborne. With the potential for influenza to be transmitted by aerosols, Dzau underscored that the stakes are high and global health experts need to step up their efforts to prepare for the next influenza pandemic.
Dzau then discussed recent progress made that aims to strengthen pandemic preparedness. Since the Ebola outbreak in West Africa, the NAM, the Harvard Global Health Institute, the London School of Hygiene & Tropical Medicine, and the United Nations (UN) High-Level Panel on the Global Response to Health Crises have generated assessment reports to chronicle what went wrong during this outbreak and how infectious disease outbreaks can be managed better in the future (Moon et al., 2015; GHRF Commission, 2016; UN, 2016). These reports generated many common recommendations, and in fact, concrete changes have been made. In particular, the following programs and alliances have been created: the World Health Organization’s (WHO’s) Contingency Fund for Emergencies2 and Joint External Evaluation,3 the World Bank’s Pandemic Emergency Financing Facility,4 and the Coalition for Epidemic Preparedness Innovations.5
Furthermore, in response to the United Nations High-Level Panel’s recommendations to create a better monitoring system for disease outbreaks,
___________________
1 For the latest numbers, please see https://www.who.int/ebola/situation-reports/drc-2018/en (accessed March 25, 2019).
2 The Contingency Fund for Emergencies is a voluntary fund that allows WHO to disburse as much as $500,000 within 24 hours, filling the gap from when the need for emergency response is identified to the point when other financing mechanisms can activate. See http://www.who.int/emergencies/funding/contingency-fund/en (accessed February 26, 2019).
3 The Joint External Evaluation is a voluntary process to assess country capacities to prevent, detect, and respond to public health risks. The goal is to identify critical gaps in national health systems and prioritize actions toward better preparedness and response. See http://www.who.int/ihr/procedures/joint-external-evaluations/en (accessed February 26, 2019).
4 The Pandemic Emergency Financing Facility is another quick-disbursing financing mechanism that provides surge funds after a major infectious disease outbreak. It also offers the first insurance opportunity for pandemic risk, covering all low-income countries eligible for financing under the International Development Association. See http://www.worldbank.org/en/topic/pandemics/brief/pandemic-emergency-financing-facility (accessed February 26, 2019).
5 The Coalition for Epidemic Preparedness coordinates financing and research among public, private, philanthropic, and civil society organizations to prioritize development of vaccines against diseases with epidemic potential, and to enable equitable access to the resulting vaccines. See http://www.cepi.net (accessed February 26, 2019).
an independent board titled the Global Preparedness Monitoring Board (GPMB) was recently established for an initial 5 years. WHO’s Director-General and the president of the World Bank in 2018 co-led the creation of GPMB, which comprises 13 international members, including Dzau, at that time. He explained that the function of the board is to offer high-level monitoring and develop a framework for global health crisis preparedness; identify and then strategically prioritize gaps in preparedness; and advocate preparedness at the highest political forums (e.g., G7 and G20 summits). He noted that the framework would be organized around four domains: strengthening public health capacities, monitoring progress of research and development, monitoring public and private preparedness financing, and generating risk analyses that include economic and social vulnerabilities. Dzau said that to accomplish some of these goals, GPMB will recruit a secretariat and will commission and publish annual reports on the status of global preparedness for outbreak crises.
According to Dzau, continued progress in science and research, public health, and governance is critical for ensuring pandemic preparedness. He concluded, “Improving pandemic preparedness will require sustained attention to collective action across many sectors. This is why I am so pleased to see all of you come together for this workshop.”
HISTORICAL INFLUENZA PANDEMICS
Laurie Garrett, science journalist and founder of the Anthropos Initiative, explained that the historical record of influenza pandemics stretches back to the 1100s. Table 2-1 provides an overview of influenza pandemics known to have occurred from the 12th century through the 19th century. She noted that historical pandemics are often described as originating in Russia, but more likely, initial pandemics originated in China, spread across Siberia, entered Russia, and then moved into Europe. These pandemics probably spread from Europe to colonial outposts in the Americas, Africa, and Asia via trade routes, slavery, and general colonial mechanisms. Several pandemics in the 18th and 19th centuries had high mortality rates and the morbidity of the “Great Pandemic” in 1833 was as high as 80 percent (Patterson, 1986). Notably, the influenza pandemic of 1889, spread by Transatlantic shipping between the Americas and Africa, affected the entire world and had a high death toll for people between 21 and 40 years of age. Similar to subsequent influenza pandemics of the 20th century, peak mortality in several of these historical pandemics occurred among young adult populations (Patterson, 1986).
TABLE 2-1 Historical Influenza Pandemics, 1173–1889
| Year | Description and Context |
|---|---|
| 1173 | Europe |
| 1387 | Europe |
| 1510 | Unknown |
| 1557 | Unknown |
| 1580 | Spread from the Mediterranean to the Baltics in 4 months |
| 1708 | Started in Rome at Christmas and swept north |
| 1729 | Started in Moscow; swept west and reached North America in 1732 |
| 1732 | Started in Russia and spread west through Polanda |
| 1761 | Spread from Poland at the end of the 7 Years War |
| 1781 | Spread from China to Russia to Europeb |
| 1789 | Spread out of Russia through Europe to North and South Americas |
| 1799 | Spread from Russia across Europe during the Napoleonic War |
| 1830 | Spread from China, India, and Southeast Asia to Russia, Europe, and the Americas |
| 1830–1831 | Southeast Asia and Indonesia |
| 1833 | Spread out of Russiac |
| 1847 | Spread west out of Moscow and Constantinopled |
| 1889 | Spread from China to Londone |
a High rate of mortality in the United Kingdom; the outbreak in horses preceded the outbreak in humans.
b Highest mortality among young adults.
c Also known as the “Great Pandemic”; morbidity as high as 80 percent.
d Railroad links were completed across much of Europe by 1847.
e Spread by Transatlantic shipping to the Americas and to Africa multiple times; high mortality among adults 21 to 40 years of age.
SOURCES: Garrett presentation, November 26, 2018; Patterson, 1986.
AN IN-DEPTH EXAMINATION OF THE 1918 INFLUENZA PANDEMIC
According to Garrett, the 1918 influenza pandemic was exceptionally virulent because it behaved fundamentally differently from previous outbreaks in its ability to cause disease. The symptoms of the disease in many cases were very severe, including hemorrhage with profound bleeding, blood-laced vomit, high fever, delirium and hallucinations, miscarriage, heliotrope cyanosis, and acute respiratory distress. Garrett explained how the 1918 pandemic spread across the globe in 9 months through three waves: a mild first wave (May–August 1918), a severe second wave
(September–November 1918), and a moderate third wave (December 1918 to mid-1919). Although popularly dubbed “Spanish Influenza” (derived from the country’s neutrality during World War I), the pandemic was unrelated to Spain; it actually came out of U.S. military camps in Haskell County, Kansas, and spread through troop movements (Barry, 2005).
Historical and Cultural Context
Garrett then provided historical and cultural context leading to the turning point of the 1918 influenza pandemic (Crosby, 2003; Barry, 2005). She described her uncle’s experience as a young boy during the pandemic, noting that the generation who experienced that pandemic is now gone; only their recorded oral histories remain. World War I began in July 1914 and was well under way when the pandemic occurred. Despite the surge in influenza in 1918, the war continued. An estimated 9 million combatants and 7 million civilians died during the war, but it is unclear how many of those deaths may have been caused by the three waves of the influenza pandemic that spread worldwide between April 1918 and February 1919 (Chorba, 2018). The lowest estimate for influenza mortality during the pandemic is 21 million deaths, which represents millions of casualties more than the war claimed (Jordan, 1927; Mougel, 2011).
According to Garrett, warfare tactics likely contributed to influenza transmission. The first widespread use of gas warfare occurred during World War I, and thousands—if not millions—of civilians and soldiers were exposed to mustard gas and chlorine, which left many with permanent damage to their lungs, their eyes, and their general health. The effects of exposure likely contributed to people’s susceptibility to respiratory infections such as influenza. In addition, soldiers spent long periods in close contact in below-ground trenches to protect themselves from bombs, and this also may have contributed to increased influenza transmission. She explained that efforts to address the influenza pandemic in the United States were hampered by major deficits in the domestic health workforce during World War I; at the time of the initial outbreak, thousands of physicians were working on foreign battlefields and thousands of nurses had been recruited to serve on battlefields and in rehabilitation centers abroad (Crosby, 2003).
When influenza first emerged in the United States, the framework for public discourse about the pandemic was limited by congressional legislation that sought to suppress government criticism, said Garrett. The Espionage Act of 1917 prohibited many forms of speech, including “disloyal, profane, scurrilous, or abusive language about the form of government of the United States.”6 The Sedition Act, passed in May 1918, amended the
___________________
6 Espionage Act of 1917, Public Law 65-24, 99th Cong. (June 15, 1917).
Espionage Act and prohibited any form of speech or expressed opinion that cast the government or the war effort in a negative light or interfered with the sale of government bonds.7 She added that any attempts to criticize the government’s response to influenza—either among U.S. military personnel abroad or on the domestic front—were considered violations of the Sedition Act. Part of the reason the written history record of the pandemic is sparse, she noted, is because it was illegal to be critical of the influenza response in writing. In fact, media at the time published articles that misrepresented the severity of the pandemic to the public, stating that it was just a typical seasonal influenza outbreak (Bristow, 2010). Ultimately, however, the public in the United States and worldwide came to understand the severity of the pandemic.
Response to 1918 Influenza Pandemic
Garrett explained that because scientific knowledge about virology had just begun to burgeon in 1918, health professionals lacked clarity about what actually constituted the pandemic disease. At the time, the main theory proposed by the U.S. scientific establishment was that the pandemic was caused by Bacillus influenza, a bacterial disease. Conjecture and speculation were rampant about the nature and cause of the disease, as well as how people could protect themselves from it.8 A common presumption was that improved personal hygiene and public space cleanliness would somehow quash the pandemic; people also thought that face masks would provide protection against influenza, but no one understood what type of mask should be used, how it should be used, or how much protection a mask would offer. Nonetheless, many governments implored their populations to wear masks in public spaces, and in one case, a man in San Francisco was shot by law enforcement officers for refusing to wear a mask in public (University of Michigan Center for the History of Medicine, 2016). Because of confusion and misinformation about the disease, responses to the pandemic by state and local governments were disorganized and inconsistent. For example, some cities decided to close schools and cancel public events while others did not.
As the epidemic worsened around the world, quarantines were imposed and public and outdoor activities were restricted. Instances of undue optimism about the pandemic’s end were deflated by subsequent resurgences. Because
___________________
7 The Sedition Act was repealed in December 1920; the Espionage Act has been amended many times but still exists under Title 18 of the U.S. Constitution.
8 Garrett said that nakedness, fish contaminated by Germans, dirt or dust, unclean pajamas, open windows, closed windows, old books, and “cosmic influence” were all popularly posed as causes of influenza.
most of the country’s medical establishment was overseas at the time, the U.S. government heavily incentivized people who remained to volunteer to help the response. However, people were largely unwilling to volunteer because of their fears and concerns about becoming infected. Since hospitals and clinics in the United States were filled to capacity, giant warehouses were created worldwide and filled with patients. Garrett described these warehouses as crowded, makeshift wards, and she detailed how volunteers and trained personnel cared for patients and were also responsible for removing dead bodies for burial. She also noted that such warehouses may have contributed to influenza transmission among the uninfected people who entered. At the time, shortages in coffins and grave spaces were rampant across the United States, leading to mass burials and unmarked graves. Law enforcement officers became more aggressive in enforcing closures and prosecuting hygiene offenses such as spitting, coughing, or sneezing in public (Aimone, 2010). Churches and schools were often closed down or converted into orphanages or warehouses for people who were sick. Garrett suggested that Armistice celebrations for the end of World War I contributed to the third wave of the pandemic, because so many people were celebrating in congregate settings without masks (Byerly, 2010).
Mortality of 1918 Influenza Pandemic
The pandemic influenza strain had a distinct presentation in patients as early as July 1918; an excerpt from a coroner’s report in London noted, “The lung lesion, complex or variable, struck one as being quite different in character to anything one had met with at all commonly in the thousands of autopsies one has performed during the last 20 years. It was not the common broncho-pneumonia of ordinary years” (Barry, 2005). Garrett provided statistics to illustrate the pandemic’s devastating mortality. During the autumn of 1918, more than 20,000 people died of influenza in New York City in only 6 weeks, and during the same period, influenza killed 5 percent of Ghana’s population and 19 percent of Western Samoa’s population (Garrett, 2005; McLeod et al., 2008). Records from Kentucky indicate that 37 percent of all deaths in 1918 were classified as “respiratory.” The United States saw a huge increase in premature mortality among children under 1 year of age (Garrett, 1994).
Garrett explained that the 1918 event was a turning point in the recorded history of influenza pandemics. For the first time, an entirely bird-adapted virus had transformed into a human-to-human transmitting virus—without going through an intermediary species. The pandemic’s high mortality was bolstered as the virus spread via networks of expanding railroad and shipping lines across the world. Given the paucity of available records, 1918 death estimates range from 20 to 22 million, which
represented 1 percent of humanity at the time, to 100 million or more, representing about 5 percent of humanity (Jordan, 1927; Johnson and Mueller, 2002). To put this into perspective, Garrett explained that if a pandemic killed between 1 and 5 percent of humanity today, the number of deaths would likely range between 72 and 340 million. However, pandemic mortality rates varied widely across the world. For instance, all-cause mortality between 1918 and 1920 reached 40 percent in parts of Iran and was very high among some rural and indigenous communities in the United States (Brady et al., 2014; Hatami, 2016).
Garrett’s own analysis of mortality records suggests that in the United States, more than 1 million people died from any cause in 1917 and almost 1.5 million died in 1918. Death rates during the period increased across every U.S. state, and the number of deaths attributed to influenza and to “respiratory” causes skyrocketed nationwide between 1917 and 1918; according to Garrett, many deaths attributed to respiratory causes were likely caused by influenza.9 Overall, the generally cited estimate for influenza mortality in the United States is about 675,000 deaths, but she said that this is almost certainly an underestimate. The 1918 pandemic has the well-known “W effect” in the epidemiologic curve of median excess mortality by age. Excess mortality for seasonal influenza is typically U-shaped, affecting the very young and very old. For pandemic influenza, however, excess mortality tends to be greatest among young adults, which added a middle peak to the usual U-shape (see Chapter 4 for further discussions on the “W effect”). Garrett noted that the pandemic also affected life expectancy rates in the United States and Europe; however, the paucity of available accurate information makes it difficult to quantify that impact.
Garrett outlined a set of lessons from the 1918 outbreak:
- The 1918 H1N1 influenza virus developed from an avian influenza virus and spread worldwide;
- H1N1 can cause human outbreaks if the virus jumps from birds to people in a form that can spread through human-to-human transmission;
- All influenza A viruses in circulation among humans today are descendants of the 1918 H1N1 strain; and
- All deaths related to influenza A viruses over the past century are at least partially attributable to the 1918 virus.
___________________
9 Garrett said that among reported deaths in the United States, there were about 13,000 reported cases of influenza and about 124,000 deaths due to “respiratory” causes in 1917. In 1918, there were about 234,000 reported deaths due to influenza and about 245,000 deaths attributed to “respiratory” causes.
2009 H1N1 INFLUENZA PANDEMIC
Garrett shifted focus to the 2009 H1N1 pandemic and explored how the global response to pandemics has evolved since 1918. She explained that in 2009, the H1N1 influenza virus emerged in humans and was transmitted through swine. The first recorded human case was a young boy in Veracruz, Mexico, who survived and is sometimes called “Patient Zero” for the 2009 pandemic. However, she conjectured this pandemic probably originated in the Midwestern United States pork industry through isolated cases of pig-to-human transmission of the H1N1 strain when children were exposed directly to pigs at county fairs. In September 2008, for example, the Texas Department of Health Services reported that an individual had contracted a swine influenza A H1N1 triple reassortant virus from pigs, which appeared to be the same strain that passed from pigs to a teenage boy in Wisconsin in 2005 (Newman et al., 2008). In March 2009, health officials in Mexico noticed a spike in the number of reported influenza cases, and in the United States, two children in California contracted confirmed cases of H1N1 influenza (CDC, 2009). In April 2009, the virus infected several young people who returned to New York City from spring break vacation in Cancun, Mexico (CDC, 2009).
Response to the 2009 Influenza Pandemic
According to Garrett, the president of Mexico responded swiftly and contained the spread of the virus at the end of April 2009, announcing that many public services, government offices, private business, schools, churches, and mass transit modalities would be ordered to close. The military was deployed to distribute and enforce the use of face masks in public spaces and gatherings (Chowell et al., 2011). The Mexican government’s level of transparency and its willingness to draw international attention to the outbreak was laudable, said Garrett. However, the government’s response also gave a false impression that the virus was exclusively Mexico’s problem. This notion was dispelled as the virus quickly spread across the world. By April 29, 2009, 9 countries had officially reported a total of 148 cases, including 91 laboratory-confirmed cases with 1 death in the United States and 26 confirmed cases with 7 deaths in Mexico (WHO, 2009b). According to data from the Mexican Health Department, 71 percent of the cases in Mexico occurred among people 29 years of age or younger (Charu et al., 2011). The similarity of this highly atypical pattern to the epidemiology of the 1918 virus was cause for great concern, she said.
In response to the epidemic, many countries issued travel alerts that recommended against travel to Mexico, and many commercial flights were canceled. According to Garrett, many travelers on airplanes that originated
in or passed through Mexico were required to pass through thermal sensors at destination airports, and were quarantined if they had a fever. Many airports mandated the use of face masks, although such masks were likely ineffective in limiting transmission (Khan et al., 2013). Airline and leisure company shares decreased due to people’s fear that the outbreak would impact the global travel industry. On May 1, 2009, WHO issued a statement10 declaring that there was no reasonable rationale for travel restrictions related to the virus since imposing travel restrictions would do little to stop the spread of the virus but would be highly disruptive to the global community. Nevertheless, many countries’ travel sanctions and quarantine policies remained in place, and the Mexican tourist industry plummeted. Face mask production plants struggled to meet global demand for masks, and markets were flooded with fake antivirals because of the global medication shortage.
Mortality of the 2009 Influenza Pandemic
Garrett provided an overview of the mortality associated with the 2009 influenza pandemic. As of August 2010, WHO reported that more than 18,500 deaths were caused by laboratory-confirmed cases of H1N1 influenza (WHO, 2010). However, modeling studies have suggested that the actual mortality count could be as much as 10-fold greater. According to one study, the number of pandemic respiratory deaths worldwide may have exceeded 200,000 during the last 9 months of 2009 (Simonsen et al., 2013). Another study modeled global mortality during the first year of the 2009 pandemic, estimating more than 200,000 respiratory deaths and more than 80,000 cardiovascular deaths (Dawood et al., 2012).
As in the 1918 pandemic, younger adults were at greater risk of death during the 2009 pandemic; estimates have suggested that as many as 80 percent of respiratory and cardiovascular deaths occurred in people under 65 years of age (Dawood et al., 2012; Simonsen et al., 2013). Another similarity between the 1918 and 2009 pandemics is that mortality rates varied widely across the world, with more than half of the deaths occurring in Southeast Asia and Africa (Dawood et al., 2012). Figure 2-1 illustrates the global distribution of deaths associated with the 2009 pandemic during the first year. Garrett added that the global impact of the pandemic in terms of attack rates and mortality rates was relatively high, given that the 2009 H1N1 strain did not contain the known markers of pathogenicity (Mishra et al., 2010).
___________________
10WHO | No Rationale for Travel Restrictions. See https://www.who.int/csr/disease/swineflu/guidance/public_health/travel_advice/en (accessed December 18, 2018).
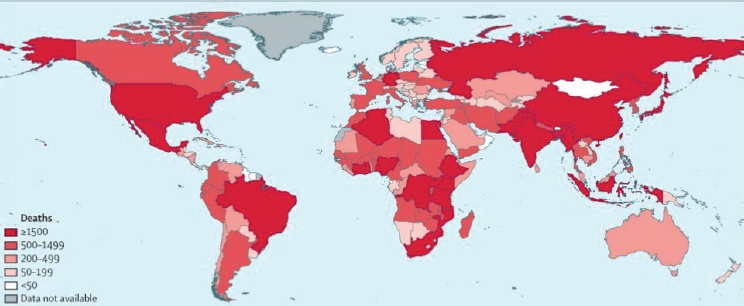
SOURCES: Garrett presentation, November 26, 2018; reprinted from The Lancet, Vol. 12, Fatimah S. Dawood et al., Estimated global mortality associated with the first 12 months of 2009 pandemic influenza A H1N1 virus circulation: A modelling study, 687–695, (2012), with permission from Elsevier.
POTENTIAL IMPACTS OF A MODERN PANDEMIC
Garrett considered the potential impacts a major influenza pandemic might have if it occurred today. According to an analysis that extrapolated the 1918–1920 mortality rates to the 2004 global population, between 50 and 80 million people could be killed by a modern influenza pandemic of similar severity to the one in 1918 (Murray et al., 2006).11 A more recent analysis estimated that more than 720,000 pandemic-related deaths each year could be expected despite of the use of currently available medical technology (Fan et al., 2018).
In contemporary contexts, the economic impact of pandemic influenza would be profound both nationally and internationally. Globally, the expected annual losses from pandemic risk has been estimated to be around $500 billion per year (0.6 percent of the global gross domestic product [GDP]) (Fan et al., 2018). An analysis of the potential economic consequences for the United States alone suggested that an influenza pandemic could cause national GDP losses of between $35 and $45 billion, depending on whether a vaccine is available (Prager et al., 2017). A macroeconomic analysis suggested that countries could face GDP losses of up to 2 percent in the event of an influenza pandemic, but that school closures
___________________
11 If this potential pandemic’s mortality were concentrated in a single year, it would increase global mortality by 114 percent (Murray et al., 2006).
and prophylactic absenteeism from workplaces could increase the economic losses by more than three-fold (Keogh-Brown et al., 2010).
Could a Modern Pandemic Be Mitigated?
In light of the projected devastating impacts on mortality and economic and societal functions, Garrett explored potential ways to mitigate an influenza pandemic if it occurred today. The current global landscape differs significantly from the landscape in 1918, when no effective treatments for influenza existed. In addition to the availability of vaccines, antibiotics, and antiviral drugs, the modern context benefits from improved respiratory care and support therapy, a larger and more skilled health labor force, the absence of intense combat and world war, WHO and other international governance mechanisms, and the Global Health Security Agenda (GHSA). She assessed each dimension in turn for its potential and pitfalls in mitigating the impact of future influenza pandemics.
Medical Countermeasures and Respiratory Care
Garrett challenged the assumption that the availability of vaccines will necessarily counteract outbreak mortality. Peak mortality during the 1918 pandemic occurred over a period of just 6 weeks, which is too short to mobilize mass vaccine production. However, she added, the current global vaccine supply is insufficient to vaccinate everyone who might need it, even if the supply were boosted with an adjuvant (McLean et al., 2016).12 She said that public misinformation about vaccination is another concern. A record number of children died from seasonal influenza during the 2017–2018 season, and 80 percent of those children had not been vaccinated (CDC, 2018a). According to a recent poll, one-third of all parents in the United States decline to vaccinate their children for influenza, and more than half believe that the vaccination itself can cause influenza (C.S. Mott Children’s Hospital National Poll on Children’s Health, 2018). Parents also rate the influenza vaccine as less important, less safe, and less effective than other childhood vaccines (C.S. Mott Children’s Hospital National Poll on Children’s Health, 2016).
Regarding the potential impact of antibiotics, Garrett noted that antibiotic resistance is increasing across all bacterial species relevant to respiratory infection treatments. However, only 12 new antibiotics have been licensed since 2000, and there are disparities in the global distribution of antibiotics (Kaufmann et al., 2017). Many pharmaceutical companies have
___________________
12 An adjuvant is a substance added to a vaccine that increases the body’s immune response to the vaccine.
now eliminated research and development for antibiotics, leaving only four major commercial companies in the antibiotic market—GlaxoSmithKline, Merck & Co., Inc., Pfizer Inc., and Roche. The efficacy of antivirals for treating influenza has also been compromised by rising drug resistance. Antivirals have never been widely available internationally, because of cost barriers and stockpiling of antivirals by wealthier countries. Domestically, in 2009, congressional funds only covered the cost for 31 million antiviral courses (Dimitrov, 2009).
The potential for improved respiratory care and support to mitigate a future pandemic is questionable, said Garrett. She noted that hospital bed capacity is lacking in many areas as a result of cost-cutting initiatives. Hospitals in the United States are largely operating at full capacity with no margin for surge capacity in personnel or facilities, she added, with limited capacity to use additional mechanical ventilators (Ajao et al., 2015).
Health Workforce
Garrett said with respect to the larger skilled health labor force, an adequately staffed health workforce cannot be depended on if an influenza pandemic occurs today. This is due to high absentee rates among the health workforce (driven by illness or fear), as well as the lack of day-to-day capacity—let alone surge capacity—to provide basic primary care. Furthermore, the number of health care personnel who reported receiving an influenza vaccine during the 2017–2018 season was suboptimal and varied among work settings, despite their regular exposure to infection (CDC, 2018b). Garrett also mentioned that while the absence of intense combat and world war is certainly a boon, attacks against health care workers are increasing around the world.13
International Governance and Global Health Security Agenda
Regarding the potential for international governance to mitigate the effects of a pandemic, Garrett commented that WHO is underfunded, and presumably, its existing funds continuously deplete with each new outbreak. Turning to the GHSA, Garrett said that only 39 countries have committed to contribute to the Action Packages.14 The United States invested $1 billion
___________________
13 For more on this, see WHO’s surveillance system for attacks on health care workers: https://publicspace.who.int/sites/ssa/SitePages/PublicDashboard.aspx (accessed February 26, 2019).
14 The Global Health Security Agenda was launched in 2014 to facilitate partnerships among countries, international organizations, and nongovernmental stakeholders to achieve core capacity targets outlined in 11 Action Packages. See https://www.ghsagenda.org (accessed February 25, 2019).
in health security under President Obama, but it is unclear whether support will continue under future administrations. No other country except Germany has so far matched this level of funding commitment, she said.
Garrett suggested that the brunt of a future pandemic will likely be borne by the poorest countries, which are more likely to have limited public health services, poor vaccine and antiviral availability, and inadequate disease management (Madhav et al., 2017). Without strong national- and global-level governance, she predicted, there will be vaccine hoarding, stockouts, travel restrictions, border closures, poor laboratory capacity, uncontrolled movement and spread of the virus, fear-mongering, and harsh military and police actions.
Impact of a Changing Planet
Garrett concluded by exploring how changes in the planet may be increasing the risk of a pandemic. Climate change is shifting the latitudes of the migration patterns, habitats, and movements of species that carry influenza. For example, migration patterns are shifting among marine species that are hunted by aquatic birds that carry influenza—as a species shifts, its predators also shift. Due to warming waters, more than 80 percent of global marine life is migrating north to escape the effects of climate change, with some marine species having migrated as many as 600 miles (Poloczanska et al., 2013). In response to climate change, ocean species are migrating 10 times faster than land species, and these shifts are expected to continue throughout the 21st century (Morley et al., 2018). The East Asian–Australasian Flyway is a key concern for influenza, said Garrett. Migratory aquatic birds on this flyway are the chief reservoirs of the influenza virus or family of viruses. The extent to which fish migratory patterns and other consequences of climate change are affecting the Asian flyway has not yet been quantified, she said, but it will likely affect influenza risk for both human and animal populations.
DISCUSSION
After the keynote presentation, the audience asked Garrett a couple of questions. Pia MacDonald, senior director of applied public health research at RTI International, asked whether the common belief that pandemic influenza strains originate in Asia is a misconception. Garrett explained that in every major pandemic H1N1 strain, some or all of its eight genes are descendants of the 1918 virus, which passed into Siberia from China. Although the second wave of the 1918 influenza pandemic appeared to emerge out of Haskell County, Kansas, it was based on the first-wave virus that had come from China. The descendant viruses do not disappear when
an outbreak or epidemic is quelled, Garrett added. The viruses continue to circulate before they are transmitted to humans from avian or swine species, particularly among concentrated livestock, as appears to have happened in the U.S. midwest pork industry in 2009.
Christopher Eddy, president of All1Health Systems, LLC, asked Garrett about the potential significance of future alternate transmission pathways for influenza, such as fecal-oral contaminated surfaces and fomites. Garrett replied that if a pandemic emerged with a highly virulent strain and efficient human-to-human transmission, the general population would immediately want to know how to protect themselves in practical ways. This would trigger evaluations of the virus and assessments of how to protect against it—for example, the effectiveness of handwashing, the distinction between risks of sneezing versus coughing, and clarifying the effectiveness of different types of masks. Much is still unknown about the relative impact of different interventions because very few large-scale studies have been conducted on this topic, said Garrett. She said that this area of research is underexplored and underfunded because it may not seem lucrative for product development, but the area warrants much more attention.
This page intentionally left blank.
















