4
Reflections on a Century of Infectious Disease Outbreaks and Pandemics
Building on discussions about pandemic influenza from the pre-workshop event, the 1.5-day workshop that followed delved into the world’s current state of readiness to prevent, detect, and respond to not only pandemic influenza but also other novel and emerging diseases. Rick Bright, director of the Biomedical Advanced Research and Development Authority (BARDA) in the Office of the Assistant Secretary for Preparedness and Response (ASPR) at the U.S. Department of Health and Human Services (HHS), delivered a keynote address on the impact of infectious disease outbreaks and pandemics on people, communities, and economies across the world. Arnold Monto, professor of public health in the Department of Epidemiology at the University of Michigan, followed with a plenary presentation in which he reflected on a century of evolving pandemic and emerging infectious disease challenges.
IMPACT OF OUTBREAKS AND PANDEMICS ON PEOPLE, COMMUNITIES, AND ECONOMIES
Rick Bright, director of BARDA, began by explaining the role of ASPR: Its mission is to save lives and to protect Americans from health security threats in the 21st century. ASPR tracks a wide range of microbial threats, which span emerging natural threats, scientific accidents, and threats imposed intentionally by individuals or state actors. In 2018, the U.S. Executive Office of the President released the first National Biodefense Strategy of the United States, which is aligned with the ethos of ASPR’s mission (USG, 2018) (see Box 4-1 for more details about the strategy).
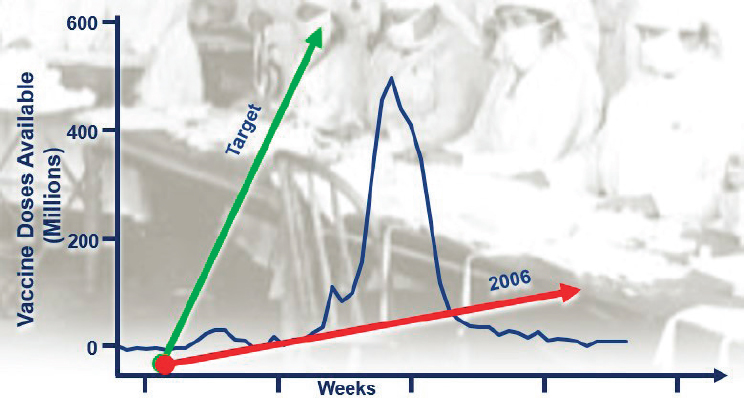
ASPR’s goal for pandemic influenza—as it is for any microbial threat—is to ensure that vaccine and countermeasure responses are in front of the epidemic curve. In 2006, ASPR conducted a national assessment that illustrated that the best national efforts to generate enough influenza vaccine to cover everyone in the country at two doses per person would be insufficient to curb the threat. Figure 4-1 illustrates ASPR’s pandemic vaccine response targets. By 2009, the country’s capacity to make more vaccines had improved but not quickly enough to get in front of the curve. In 2018, Bright reported that national capacity had improved to the extent that around 800 million doses of pandemic influenza vaccine could be produced if needed. The challenge now, he said, is to understand when to use the vaccine, where to use it, how to distribute it, and how to administer it as quickly as it can currently be produced.
Impact of Recent Outbreaks and Pandemics
Bright remarked that infectious disease outbreaks and pandemics continually emerge and evolve over time, but there are lessons that remain
SOURCE: Bright presentation, November 27, 2018.
to be gleaned from threats that have been encountered in the past—and from those that people face today. He advised that aggregating and applying those lessons in a cohesive way would strengthen preparedness for future threats. Since the 1918 influenza A H1N1 pandemic, Bright said, many impactful outbreaks and pandemic events have occurred over time:
- The U.S. Centers for Disease Control and Prevention (CDC) first reported HIV in 1981, and as of 2017, almost 37 million people were living with HIV worldwide and 35 million people have died of HIV/AIDS since the beginning of the epidemic (WHO, 2018d).
- Between 2002 and 2004, severe acute respiratory syndrome (SARS) emerged in China and caused almost 800 deaths worldwide (CDC, 2013b).
- Between 2009 and 2010, the influenza A H1N1 pandemic caused more than 200,000 deaths worldwide.1
- In 2012, Middle East respiratory syndrome (MERS) was first identified in Saudi Arabia and has since caused more than 800 deaths (Hajjar et al., 2018).
___________________
1 As noted in Chapter 2, the number of estimated deaths is unclear. CDC estimates that the pandemic caused 151,700 to 575,400 deaths, and this estimate is based on limited data and on untested cases that resulted in deaths from influenza-like illnesses (CDC, 2012).
- Between 2014 and 2016, the Ebola virus epidemic in West Africa caused more than 11,000 deaths (CDC, 2019a).
- In 2015, the Zika epidemic emerged and resulted in considerably fewer deaths but notably led to Congenital Zika Syndrome and Guillain-Barré syndrome (a rare neurological disorder) (WHO, 2016c).
Bright noted that those outbreaks and pandemics varied widely in mortality and morbidity, and in several cases, their overall economic impacts were more devastating in relative terms than their impacts on mortality and morbidity. For example, while the number of deaths from the 2003 SARS epidemic was relatively low, its global macroeconomic impact—estimated at between $30 billion and $100 billion—was severe (Smith, 2006). The Ebola epidemic was associated with $2.2 billion in lost gross domestic product (GDP) in 2015 across Guinea, Liberia, and Sierra Leone (CDC, 2018a); $3.6 billion was spent by Germany, the United Kingdom, the United States, and the World Bank to support the response to the epidemic (CDC, 2018a). Furthermore, the U.S. Congressional Budget Office has estimated that a modern moderate-to-severe influenza pandemic could have a macroeconomic impact in the United States of around 1 to 4.25 percent of the national GDP (CBO, 2005). Bright cautioned that even a severe outbreak of seasonal influenza—not necessarily a pandemic—could have a significant macroeconomic impact on the global economy. In 2016, the Commission on a Global Health Risk Framework projected the average expected economic losses from infectious disease crises in the future would be around $60 billion per year (GHRF Commission, 2016). A World Bank report on the emergence of antimicrobial resistance estimated that if resistance is unmitigated by 2050, the global economic losses could reach $100 trillion (World Bank, 2017).
Common Elements of an Outbreak Response
According to the World Health Organization (WHO), “Disease X represents the knowledge that a serious international epidemic could be caused by a pathogen currently unknown to cause human disease.”2 Bright explained that as many as 631,000 to 827,000 undiscovered viral species exist with zoonotic potential in mammal or bird hosts (Carroll et al., 2018). Furthermore, any one of the hundreds of thousands of additional organisms that are already known to circulate could emerge and cause total devastation in terms of morbidity, mortality, and economic loss. Bright
___________________
2 More information about Disease X is available at https://www.who.int/blueprint/prioritydiseases/en (accessed December 30, 2018).
added that this underscores the urgency of identifying common elements of effective outbreak response.
Bright offered a breakdown of the essential elements of an effective response: early detection, vaccination, behavioral countermeasures, and addressing gaps in host-based treatment. According to Bright, early detection of organisms that spread locally, regionally, nationally, or globally is a high priority because “we don’t know what’s coming our way until it’s too late . . . every day counts.” Knowledge about when and where to use vaccination and behavioral countermeasures is also critical, he added. Significant gaps remain in host-based treatment options, he said, and such gaps need to be addressed through a better understanding of immune modulation, cytokine storms,3 and common processes triggered in the body upon exposure to bacteria or viral organisms.
Potential Impact of Pandemic Influenza
To demonstrate the speed at which the death toll mounts during a pandemic similar to the one in 1918, Bright presented an animated simulation, the “Shattuck Flu Map,” developed by the Institute for Disease Modeling. Three months into the simulated pandemic, more than 10 million people had died around the world. After 4 months—the point at which factories would be able to start producing a pandemic influenza vaccine, given the world’s current best efforts—the death toll had increased to 22 million people.4 Six months into the simulated pandemic, when maximum vaccine production would be available, an estimated 33 million people had died. The simulation starkly illustrated the importance of early detection and timely warning systems, Bright said, and of the desperate need for new technologies to address this type of potential outbreak.
BARDA has developed an agent-based model to represent an unmitigated pandemic of comparable transmissibility to that of Shattuck Flu Map’s global simulation. The model covers a specific area in the United States—Washington, DC, and the states of Maryland and Virginia—and simulates the transmission of pandemic influenza among people in these areas. Every person is represented by a unique agent in the simulation, and each agent mixes among multiple interaction groups in which transmission could occur—at home, work, school, and general context settings. As a result, the specific risk of transmission across settings varies by age group, and transmission risks are calibrated to the results of influenza-transmission
___________________
3 A cytokine storm is an overproduction of immune cell cytokines, their activating compounds.
4 The animated simulation is available at http://www.idmod.org/news/node/296 (accessed December 27, 2018).
context studies. This agent-based model is specifically designed to simulate real-world pandemic conditions, Bright said. It simulates the interactions among individuals as they become aware of the pandemic or infection outbreak, which allows people to act in ways that impact transmission.
Layering Interventions to Change the Epidemic Curve
BARDA’s goal is to identify the most effective strategies for flattening the epidemic curve that underpins the vaccine-production targets modeled in Figure 4-1; such actions would push the curve outward and thus gain enough time to create vaccines. Bright outlined a set of factors that would contribute to changing the curve by 2030. He said that global collaboration is essential for early outbreak detection and for sharing data, knowledge, and virus samples—all of which are critical for getting ahead of the outbreak curve. He added that individuals also need to be empowered with actionable information so that people have adequate tools to intervene for themselves, take responsible action, and access treatment more quickly. He highlighted that investment in rapid, nimble production platforms for vaccines and for other medical countermeasures will also be critical for changing the curve by 2030.
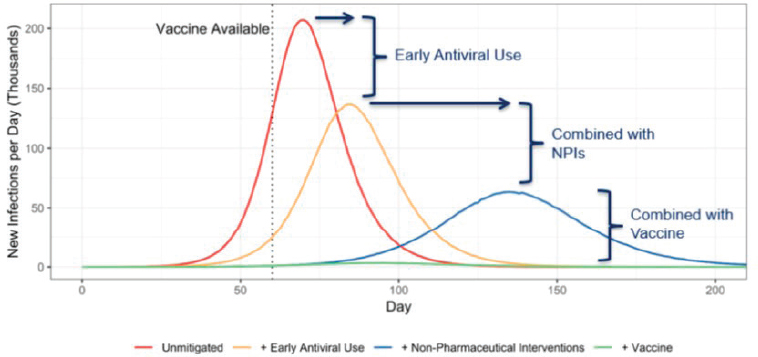
Bright explained that BARDA considers its agent-based model to be a layered mitigation that uses future technologies to explore the impact of various interventions on the epidemic curve. In particular, it has simulated the impact of three interventions: (1) early detection, which enables earlier treatment initiation, (2) targeted implementation of nonpharmaceutical intervention (NPI) measures, and (3) rapid vaccine and vaccination systems.
Specifically, the first step was to model the impact of early virus detection through at-home diagnostics and wearable devices, which enable earlier disease detection and treatment and may encourage people to take faster action and to seek faster treatment. He explained that at-home technology could send alerts to people who are still pre-symptomatic and notify them if they are exposed to influenza, for example; this alert could be augmented by messaging from local public health entities if there were an outbreak or pandemic in the community. Exposure information could then be shared with people’s health care providers through current telemedicine capabilities, and those providers could subsequently prescribe an influenza antiviral that would be delivered via curbside drop-off or drone delivery. He noted that these technologies are not futuristic; they are already in place, and they can enable people to receive faster treatment and to stay home, which may reduce further transmission of the virus. When BARDA modeled those interventions, Bright said, the impact of early antiviral use substantially flattened the epidemic curve by roughly 75,000 new infections per day and shifted the apex of the curve outward to about Day 80.
The next step was to model the targeted implementation of NPI measures. Various studies conducted in isolation have shown reduced transmission from hand hygiene, respiratory protection, confinement at home, school closures, and other interventions. However, because the relative impact levels of those interventions have not yet been established, BARDA combined those interventions in its model. Bright noted that the model assumed that individual agents will have a number of NPI options and that regardless of the specific option(s) they choose, individuals will act responsibly to try to reduce further transmission of the organism. Bright emphasized that the model suggested the synergistic impact of NPIs: When combined with early antiviral treatment, models of NPI implementation significantly flattened the epidemic curve to around 60,000 new infections per day and pushed out the apex of the curve by roughly an additional 50 days. Given the best current efforts at vaccine delivery, he said, this change in the epidemic curve effectively “buys time” for vaccines to become widely available and to begin affecting individuals’ exposures.
Vaccines and future vaccination technologies were then applied to the model. Bright noted that significant effort is currently being devoted to developing vaccine technologies, such as using recombinant-based technologies to increase the speed of vaccine production and exploring new ways to distribute those vaccines. He highlighted the importance of investing in technologies that make vaccines available and of investing in the creation of community-level distribution channels that allow vaccines to be administered easily and rapidly—which, in turn, elicits the necessary immune response during an outbreak. When new vaccines and vaccination technologies were added to the model, their synergistic interaction with early antiviral treatment based on early detection and notification and with NPIs dramatically flattened the epidemic curve (see Figure 4-2).
Bright reiterated that a vaccine’s impact depends on intervention synergy; in the context of current technologies, a vaccine intervention alone would only slightly—and insufficiently—shift the epidemic curve. The combined effects of multiple interventions are what effectively suppress the epidemic curve. Because of this, he explained, it is crucial to create a layered effect in a controlled, coordinated way in order to illustrate the true impact of early antiviral use, combined with NPI, then combined with an effective vaccine.
Potential Areas for Greatest Impact on Building Readiness for 21st-Century Threats
Bright concluded by delineating areas with the potential for the greatest impact on building readiness for threats in the 21st century (see Box 4-2). He noted that addressing end-to-end solutions in pandemic response is an
NOTE: NPI = nonpharmaceutical intervention.
SOURCE: Bright presentation, November 27, 2018.
important area of consideration, and researchers are conducting efforts on multiple fronts. These include cultivating better situational awareness and detection through new technologies; identifying and characterizing the most effective drugs and vaccines; and improving the design, validation, production, distribution, and administration of medical countermeasures. He noted that although each area is ripe for technological innovation, much of the current research is being carried out in isolation, which limits its potential benefit. For example, he explained that faster vaccine production needs to be tied to a faster early-notification system, incorporated into better
distribution channels, and streamlined through improved administration systems. He said, “We have to incorporate every single component of technology into an end-to-end solution, from early situational awareness all the way to the administration of the medical countermeasure.”
Bright emphasized that technology alone will not solve a pandemic crisis; response efforts need to be governed by strong leadership structures at the local, national, and global levels. He added that technology needs to be developed and made available in the context of local and national health care systems that are prepared for responses and have the support needed to implement them. Bright surmised that global collaboration and knowledge sharing is likely to be the most important factor in the success of any pandemic outbreak response.
Bright suggested that the production of medical countermeasures needs to be simplified and regionalized, shifting focus from scaling-up to scaling-out. He said it is critical for all countries around the world to have access to vaccines since the efforts of only one country cannot stop a pandemic. It is also important to routinely consider the impact of local-, community-, and hospital-level preparedness and of how such preparedness ties into global mechanisms, he added, because each country cannot have a unique national preparedness-response posture in an effective global effort. He added that this again underscores the importance of ensuring the functionality of global coordination, alignment, and collaboration mechanisms prior to a bioincident through implementing robust national planning and through testing to failure of countries’ pandemic response capabilities.
Finally, Bright noted that empowering individuals with tools and information is an important priority for both BARDA and ASPR. He said citizens should no longer need to wait passively for their governments to inform them about or rescue them from pandemics; rather, they should be given response tools that are already available, such as at-home diagnostics or indicators, technology that allows them to tether to a health care system from home, and actionable information in advance of an outbreak around how to reduce personal risk. According to Bright, this type of tool—which empowers the individual—may have the greatest impact on slowing, flattening, and pushing out the epidemic curve. He said:
As we invest in the large-scale infrastructure and invest in meetings and collaboration around the world at the highest levels of government and down to states and communities, we have to invest just as much in making sure each individual knows their role in a pandemic or emerging disease outbreak.
A CENTURY OF EVOLVING PANDEMICS AND EMERGING INFECTIOUS DISEASE CHALLENGES
Arnold Monto, professor of public health in the Department of Epidemiology at the University of Michigan, presented on a century of challenges created by evolving pandemics and emerging infectious diseases. In order to strengthen responses to future incidents, he highlighted some of the issues around and myths about influenza that have occurred over the years. He also noted that researchers have only recently re-identified important findings about pandemics (i.e., those that have emerged through analysis of past incidents) and remarked, “We keep reinventing influenza . . . if we had a better idea of what went on in the past, we might be better prepared for some of the things that should not be surprises as we go forward.”
Responses to Coronaviruses
Monto’s presentation focused primarily on pandemic influenza but began with a brief overview of global responses to SARS and MERS outbreaks. He noted that the SARS response in 2003 exemplified how response efforts should be—but often are not—successful. Partly because health teams that were focused on influenza were already in place and primed to respond to a pandemic, SARS coronavirus and its transmission sites were identified relatively quickly (Fouchier et al., 2003; Kuiken et al., 2003). He mentioned that at the initial stage, NPIs were the only available response approaches. SARS differs from influenza in a notable way: Its viral load is lower in the first 5 days of illness. This predicts that transmission is less likely early in the illness, which is supported by epidemiologic data (Lipsitch et al., 2003). Consequently, SARS transmission was mainly nosocomial because patients tended to be hospitalized and to receive invasive procedures during their peak viral load. Researchers determined that transmission could be interrupted if patients were detected early and then isolated.
Because patients flocked to hospitals at the time they posed the greatest transmission risk, community-level transmission of SARS was comparatively rare. This is reflected in the age distribution of SARS cases, which Monto noted was “peculiar” because it skewed so heavily toward young adults—many of whom worked at hospitals or in nursing homes where SARS patients arrived and then transmitted the disease—while sparing the elderly (Xu et al., 2014). People between 20 and 29 years of age were the most frequently affected population, followed by people between 30 and 50 years of age. People younger than 20 and older than 50 years of age were affected in much smaller numbers (Dawood et al., 2012; Simonsen et al., 2013). Monto added that it was difficult to show with serologic studies that young
children were being infected at all. He emphasized the importance of quickly determining the age distribution of an epidemic since that will inform how interventions will be distributed. Regarding the SARS transmission chain, he said it quickly became clear that airline travel was involved in the wide and rapid spread of SARS. The outbreak of SARS in 2003 differed from influenza outbreaks in another way: so-called “superspreaders” were clearly identified and they presented another opportunity for effective use of NPIs. Overall, Monto said, the response to SARS contained the epidemic within one calendar year.
Like SARS, MERS has been associated with nosocomial transmission and with superspreaders. In contrast, the majority of MERS cases occurred among patients who sought care in the same facilities as the index case, but SARS affected a larger proportion of health care workers (Chowell et al., 2015). Furthermore, Monto added that SARS was contained relatively quickly, but MERS is ongoing and that the 2015 MERS outbreak in Korea was particularly devastating. Monto attributed this to a lack of national preparedness capacity to recognize the threat quickly and to contain it early. He observed that the MERS outbreak exemplified how transmission is one of many factors that can complicate effective responses to pandemic and epidemic threats.
Influenza Pandemics of the Past Century
Monto discussed several influenza outbreaks and pandemics over the past century in order to highlight specific lessons that can productively inform future preparedness efforts. He drew lessons from the H1N1 pandemic in 1918, H2N2 pandemic in 1957, H1N1 nonpandemic in 1976, and the H1N1 pandemic in 2009.
1918 H1N1 Influenza Pandemic
Monto noted that the global mortality estimates for the 1918 H1N1 influenza pandemic have changed substantially over the years because the entire developing world was excluded from the initial estimate (around 20 million deaths). However, estimates about the pandemic’s devastating impact in developing countries were reported as early as 1927 (Jordan, 1927). For example, Monto said there were more than 12 million deaths in India—more than the bubonic plague—which illustrates how the devastating effect of any pandemic tends to be felt keenly in the world’s most highly populated areas and in places where people tend to reside in close proximity.
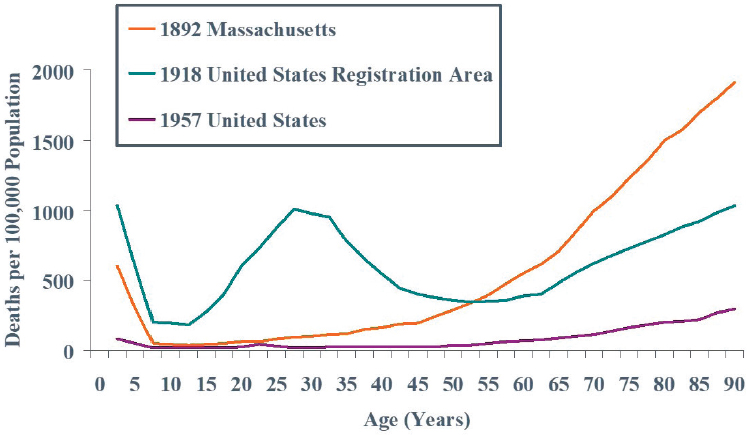
Monto presented Figure 4-3 to exemplify the famous W-shaped curve of pneumonia and influenza mortality in influenza pandemics. In this type of curve, mortality is high among three age groups: very young children,
SOURCES: Monto presentation, November 27, 2018; reprinted with permission from the American Thoracic Society. Copyright © 2019 American Thoracic Society. C. C. Dauer and R. E. Serfling/1961/American review of respiratory disease/83/15-28. The American Review of Respiratory Disease is an official journal of the American Thoracic Society.
young adults, and very old people (Dauer and Serfling, 1961). A more recent analysis of annualized 1918 data examined age-specific influenza death rates among females in England and Wales during a first-quarter wave of regular seasonal influenza and a fourth-quarter wave of pandemic influenza. This study showed that older individuals were spared, but young children were not (Nguyen-Van-Tam and Hampson, 2003). Monto said that a constant in every pandemic is that young children are at high risk, but this is often overlooked.5
Monto described how contemporary literature from the period also reflected that older individuals had been spared. A study among working-class persons covered by industrial policies at the Metropolitan Life Insurance Company (1917 versus 1918) revealed that age-specific excess total mortality rates were greatest among very young children and among people between 20 and 40 years of age (Craig and Dublin, 1919). Monto said the W-shaped curve, which represents what happens when older individuals are spared, was also evident in sex- and age-specific mortality rates for influenza and pneumonia during the Philadelphia outbreak in late
___________________
5 Monto noted that the impact of influenza on very young children is the reason why oseltamivir is now licensed for children as young as 2 weeks of age (Çiftçi et al., 2016).
1918—a time when the death rate was exceedingly high across a 2-week period after a Liberty Bond march.6 He remarked that these data are providing the foundation for new research. For example, virologists are investigating whether the population was sensitized during the 1889–1892 outbreak. If so, this may be linked to the higher mortality among young adults during the 1918 pandemic. He discussed the myth that the 1918 pandemic caused people to die within 1 or 2 days, and noted that in reality most people died between 9 and 11 days after the onset of illness as patients filled hospitals and strained surge capacity (Roberts, 1919). Pregnant women also experienced high risk during the 1918 pandemic (Jordan, 1927), but Monto added that this trend had to be “rediscovered” during the most recent influenza pandemic.
1957 H2N2 Influenza Pandemic
Monto used data from the 1957–1958 H2N2 influenza pandemic to illustrate how major outbreaks can occur at any time of the year when schools are open. For example, data from the U.S. pandemic that originated in Asia have shown that the incidence first peaked at 6,000 per 100,000 population in October and November 1957, dropped to 2,000 per 100,000 population during the winter school break, and then increased to a smaller peak of 3,000 per 100,000 population in February and March 1958 (Langmuir, 1961).
Analysis of data from the 1957 pandemic revealed the characteristic pattern of morbidity and mortality from influenza (Monto, 1987). A vaccine was eventually available, but it was delayed for 6 months because developers had to adjust its formulation (Murray, 1961). Monto added that this clearly illustrates the need for universal or next-generation vaccines in order to improve response efforts. He said that another observation made shortly after the 1957 pandemic was that ultraviolet lights can reduce the occurrence of an outbreak (Jordan, 1961), but this knowledge was only recently rediscovered by contemporary researchers. In addition, it has become increasingly evident, Monto noted, that some degree of airborne transmission occurs with influenza, which had not previously been supported by available data (McDevitt et al., 2012).
___________________
6 Source: U.S. Bureau of the Census, Special Tables for Mortality from Influenza and Pneumonia, September 1 to December 31, 1918. Available at https://www.cdc.gov/nchs/data/vsushistorical/morttable_1918.pdf (accessed March 22, 2019).
1976 H1N1 Influenza Nonpandemic
Monto also traced the course of the 1976 H1N1 influenza nonpandemic and sought to weigh the risks and benefits of inaction and to illustrate how an outbreak response may be viewed as an overreaction in retrospect. In January 1976, H1N1 influenza was first detected in the United States, and the national response was keen and swift; a program to vaccinate the entire population was announced in March, and vaccinations began in October. However, within weeks after initial vaccinations, it became clear that the influenza vaccine had caused Guillain-Barré syndrome in roughly 1 in every 100,000 people who received it (Escher, 2017). About 450 people developed the disorder before the vaccination program was suspended in December, and the repercussions of this mass vaccination campaign may still affect public sentiment about vaccines to this day (Escher, 2017).7 Monto stressed that this type of insight was only possible with the benefit of hindsight, and he asked, “If you didn’t react and something had happened, what then?”
Reflecting on the prepandemic use of pandemic vaccines, especially when adjuvants are available, Monto noted that vaccines are approved based on which induction of antibody titer researchers consider protective; antigen sparing is not an issue, and manufacturing capacity is generally available. He explained that vaccines can be given alone or combined with other antigens, which is similar to the strategy once used experimentally in the U.S. military. Monto noted that this strategy is currently under serious consideration by governments based on the premise that delivering vaccines to people directly is a better alternative to stockpiling. He said this holds particularly true when they are delivered prior to a pandemic scenario, in which it would be difficult to administer stockpiled vaccines to people in the midst of an incident.
2009 H1N1 Influenza Pandemic
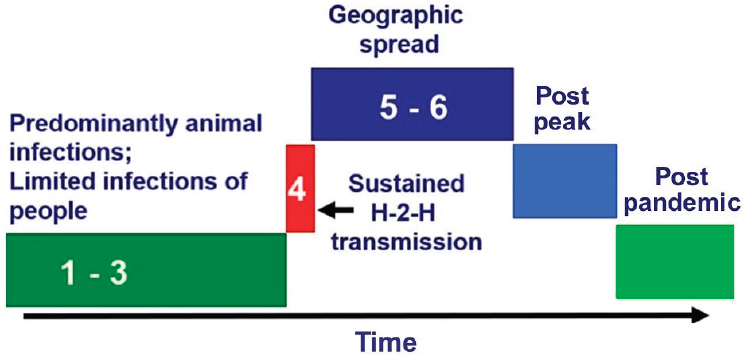
Before examining the 2009 H1N1 influenza pandemic, Monto explained that WHO proposed phases of epidemic and pandemic alert and response in 2008 (see Figure 4-4). Six phases were identified to illustrate the progression of an infectious disease through the peak of an outbreak. The
___________________
7 In a related aside, Monto discussed another example of resistant public sentiment about vaccines. He said some people view any appropriate pandemic planning as a marketing ploy, and often cite a particular publication as a peer-reviewed defense of this position (Doshi, 2013). In fact, the publication is an op-ed piece to argue for the existence of a plot to market antivirals and, to a lesser extent, vaccines. In response, a meta-analysis of oseltamivir data—which Monto deemed unnecessary work—was conducted to show that the already-licensed drug was as good as the clinical trials had shown (Dobson et al., 2015). This type of resistance needs to be considered, he cautioned, in order to prepare researchers to counter “bad science with good science.”
NOTE: H-2-H = human-to-human.
SOURCES: Monto presentation, November 27, 2018; adapted from WHO, 2009a. Reprinted from Current WHO phase of pandemic alert for pandemic (H1N1) 2009, Copyright (2009).
first three phases account for the period before sustained human-to-human transmission occurs in the fourth phase. Monto said the International Health Regulations were tested by a pandemic influenza epidemic in 2009, and this shifted the emphasis of response efforts toward the fourth phase. He noted that the fourth phase should be recognized as a key time; it is the point at which the virus becomes transmissible to humans, and health care professionals transition to pandemic preparedness as a result. Monto said current efforts have been situated in an interim scenario between the third and fourth phases for a number of years, and the primary threats of concern have been avian strains from Asia that have not yet exhibited sustained transmission in humans. He explained that sustained transmission is a bellwether: If a community-level outbreak were detected, and sustained human-to-human transmission were to occur, it would trigger urgent decisions about particular considerations (e.g., rapid containment and use of a pandemic vaccine). Monto explained that it remains impossible to predict when transmission is most likely, even when molecular work shows that resistance and transmission are possible. Consequently, he added, it will be necessary to prepare every time a potential threat arises, at great labor and expense, until the advent of a universal vaccine.
Monto explained that the 2009 H1N1 influenza pandemic occurred in two waves and that the second was a resurgence when schools opened in the autumn (Jhung et al., 2011). Similar to the 1918 pandemic, older people
were spared, and younger populations were hit hardest. Unlike the 1918 pandemic, he noted, some otherwise healthy young adults were at greater risk of a severe reaction or cytokine storm, but the mechanisms underlying that risk have not been identified. He added that pregnant women were discovered to be at higher risk during the 2009 pandemic—the same as in the 1918 pandemic—and that the entire program of antiviral distribution had to be adjusted (Louie et al., 2010). Indigenous populations in Canada were also found to have greater susceptibility for reasons that may include their living conditions, nutrition, genetics, or a combination of these (Kumar et al., 2009). Monto said that specific at-risk populations were identified in parallel all over the world and that this poses an important consideration for response elements (e.g., prioritizing the distribution of interventions).
Observations on a Century of Facing Evolving Pandemic and Emerging Infectious Disease Challenges
Monto concluded his presentation with a set of observations gleaned from a century of facing evolving pandemic and emerging infectious disease challenges. First, he said that although the clear virologic definition to describe what is a pandemic is no longer certain, the defining element should be that the virus is novel for much of the population. He noted that some people have questioned whether the 2009 H1N1 incident was a pandemic because it was not characterized by a new subtype of influenza. However, he noted that pandemics clearly occur through other mechanisms, such as when an avian segment enters the human population. The real matters of importance, he argued, are the antibody prevalence within a population and the likelihood that the virus will spread and cause a severe impact.
Second, he observed that the 1918 influenza pandemic remains an outlier in terms of morbidity and mortality. Although certain elements were repeated in the 2009 H1N1 incident, they were greatly reduced. Despite this, Monto cautioned that preparing for devastating epidemics remains a critical policy consideration. He also observed that each pandemic has unique characteristics, which makes complete generalization impossible and highlights the importance of early disease characterization. CDC has developed early assessment strategies to predict how a pandemic will progress and which populations it will affect. However, he added, these strategies are not infallible, and being nimble and being able to adjust to response efforts is critical. Finally, Monto observed that all modalities for intervention are imperative. If prior surveillance is sufficiently good, he said, some vaccines may even be made available in advance. NPIs are also crucial and warrant further evaluation, but Monto warned that high scientific standards for study designs have been set for this type of research, and they can be difficult to meet.
DISCUSSION
During the discussion, Peter Daszak, president of EcoHealth Alliance, asked Bright about progress toward high-tech solutions that promise to radically change the way diseases are diagnosed, such as new rapid and remote technologies. Bright responded that progress in this realm has been made in stages over the years and has included efforts to build centralized laboratories, large public health centers, and hospital laboratories; diagnostic technology has now filtered down to regional laboratories and to smaller units in “minute clinics” and mobile pharmacies. He said the next step is to push new diagnostics into people’s homes, but this raises concerns related to specific organisms, diseases, and outbreaks. With influenza, for example, there are concerns about how an individual might deal with a false-positive or a false-negative result. He said that risk–benefit analyses will be necessary to examine the consequences of starting unnecessary antiviral treatment based on false-positive tests. A concern, he explained, might be whether that outcome would promote antiviral resistance or squander limited supplies of a therapeutic resource in a pandemic context. Bright said that his agency’s broad aim is to encourage individuals to take responsible action to reduce transmission and to seek earlier antiviral treatment.
Furthermore, Bright noted that the capacity to miniaturize molecular technology to an affordable home-based system already exists, and the next step for at-home technologies is wearable devices. However, introducing such technology into homes creates another challenge: tethering the technology to a health care provider. He said tethering is important for individuals who receive a signal from an at-home device that instructs them to seek health care intervention. A notification pathway should be in place so that information from the indicator is coupled with a larger community database of health care. He said the current challenge is to identify appropriate biometrics within the regulatory pathway that can be coupled with an at-home test or with another type of intervention that occurs between an individual’s home and a doctor’s office. According to Bright, a plethora of wearables (e.g., tattoos, bandages, stamps, watches, rings, earrings, ear plugs) are already available and are being used to measure things that are “interesting but often useless.” The key is that these wearables motivate people to change their behaviors. He posited that similar technologies on the horizon will have huge potential during an outbreak, by indicating and motivating people to change their behaviors and to help create barriers to transmission.
Suerie Moon, director of research at the Global Health Centre of The Graduate Institute of International and Development Studies, remarked that layering mitigations—that is, implementing four or five different strategies at once—is feasible for a pathogen like influenza, which is well
understood, and in the context of a country such as the United States, which has relatively high technological development. However, she noted that pathogens and outbreaks also occur in countries where layering is not practical. Moon asked how response planners should prioritize interventions that are especially synergistic in settings where layering is not feasible.
Bright noted that although the United States appears to have the capacity and knowledge to do so, interventions in the country remain unlayered because layering requires extensive coordination and preparation from individual to federal levels. He explained that the concept of community-level mitigation, coupled with individual-level mitigation, is not commonly accepted, and efforts are generally not well coordinated—even in highly developed countries. He suggested prioritizing NPIs, especially in situations in which a novel organism emerges. Bright cautioned that the dataset for NPIs is not strong, but public messaging still needs to be supported by data; people must trust that the advice they receive will have an impact. The simple act of hand washing tremendously reduces virus transmission. He also noted that efforts are under way to reimagine respiratory protection technology (e.g., masks and respirators), which has remained largely unchanged for the past 50 or more years. He added that the best improvement in respiratory protection might actually be hand sanitation since people who wear masks take them on and off repeatedly, and this action can spread a virus or organism from a person’s hands to the person’s face and therefore transmit an infection. He advised that layering strategies for NPIs should be prioritized as well as better understanding the context of actions and behaviors related to NPIs.


















