Summary
One of the biggest challenges for the risk-assessment community is how to move from the traditional chemical-by-chemical approach to analyses that evaluate multiple chemicals together. The primary problems with the traditional approach are that chemicals on which data are insufficient are typically treated as not hazardous, that untested chemicals are often substituted for hazardous chemicals, and that cumulative exposure and risk are often ignored. One method of assessing multiple chemicals is a class approach in which chemicals that have similar chemical structures or physicochemical properties are evaluated together on the basis of an assumption that they have similar biologic activity. Over the last few decades, that approach has been used for a few chemical classes, including phthalates and cholinesterase-inhibiting pesticides. Although it is challenging to evaluate chemical groups, the number of chemicals in use today demands a new approach to risk assessment, and the class approach is a scientifically viable option.
In light of the momentum to regulate by chemical class, a coalition of organizations and individuals petitioned the Consumer Product Safety Commission (CPSC) to initiate regulatory action under the Federal Hazardous Substances Act (FHSA) to ban several products that contain nonpolymeric, additive organohalogen flame retardants (OFRs).1 To decide whether a ban should be enacted, CPSC must first conduct a hazard assessment to determine whether the chemical is toxic as defined in the FHSA. The term toxic is applied to “any substance that has the capacity to produce personal injury or illness through ingestion, inhalation, or absorption through any body surface”.2 If the chemical is found to be toxic, CPSC conducts a quantitative risk assessment in which dose–response relationships, bioavailability, and exposure are considered to determine whether the chemical is a “hazardous substance” under the FHSA.
ORGANOHALOGEN FLAME RETARDANTS AND THE PETITION
In the 1970s, flame retardants began to be added to synthetic materials to meet strict flammability standards. Over the years, diverse flame retardants have been manufactured and used in various products. Some flame retardants have migrated out of the products, and this has led to widespread human exposure and environmental contamination. There also is mounting evidence that many flame retardants are associated with adverse human health effects. As a result, some flame retardants have been banned, restricted, or voluntarily phased out of production and use.
The petition submitted to CPSC in 2015 specified four product categories that contain OFRs: infant, toddler, or children’s products; upholstered furniture; mattresses; and plastic electronic casings. The petitioners argued that the entire chemical class is toxic, that consumers are exposed to OFRs because they migrate from the products into the environment no matter how the products are used, and that their use therefore poses a risk to consumers. However, CPSC staff recommended that the commission deny the petition because OFRs constitute a broad chemical class that is defined primarily by function—to suppress combustion and increase the probability of escape from fire—rather than by any specific toxicity characteristic or chemical feature other than a halogen. Several other considerations also influenced the recommendation to deny the petition. The commission voted, however, to grant the petition, which required staff to proceed with the hazard assessment based on the chemical class. Because of the likely complexities of an assessment of this chemical class, CPSC asked the National Academies of Sciences, Engineering, and Medicine (the National Acad-
___________________
1 The abbreviation OFR in this report refers specifically to nonpolymeric, additive organohalogen flame retardants.
2 CPSC (Consumer Product Safety Commission). 2017. Staff briefing package in response to petition HP15-1, requesting rulemaking on certain products containing organohalogen flame retardants. May 24, 2017. Available: https://www.cpsc.gov/content/ballot-vote-petition-hp-15-1-requesting-rulemaking-on-certain-products-containing [accessed July 18, 2018].
emies) to develop a scoping plan to conduct the hazard assessment for OFRs as a chemical class.3 As a result of the request, the National Academies convened the Committee to Develop a Scoping Plan to Assess the Hazards of Organohalogen Flame Retardants, which prepared this report.
HAZARD-ASSESSMENT SCOPING PLAN
The committee’s recommended scoping plan is shown in Figure S-1 and described in detail in Chapter 2 of this report. The first step in the process is to determine whether a class approach to the chemicals of interest is viable for conducting a hazard assessment for CPSC. Answering that question might involve determining whether subclasses need to be formed if the chemicals in the class cannot all be assessed as a single class. Forming and evaluating broad subclasses is still a class approach. If a class approach is viable, the second step is to survey the literature to determine the availability of toxicity data (from human, animal, in vitro, and other relevant studies) and to identify relevant end points to investigate. If relevant data are available on any chemical for a given end point, the next steps are to extract, evaluate, and integrate the relevant data to reach a decision regarding potential hazard that can be applied to the entire class or subclass.
The committee conducted its own analysis to determine whether OFRs can be treated as a single class. It first created an inventory of 161 OFRs from several sources and then identified analogues on the basis of functional, structural, and predicted bioactivity information. To evaluate similarity, the committee compared the OFR inventory to the analogues and found that the OFRs cannot be treated as a single class for the purposes of a CPSC hazard assessment. The OFRs can, however, be divided into subclasses on the basis of chemical structure, physicochemical properties, and predicted biologic activity. The committee identified 14 subclasses that can be used to conduct a class-based hazard assessment and concluded that the best approach is to define subclasses as broadly as is feasible for the analysis; defining subclasses too narrowly could defeat the purpose of a class approach to hazard assessment.
The committee surveyed the literature and selected two subclasses—polyhalogenated organophosphates and polyhalogenated bisphenol aliphatics—to illustrate various aspects of its proposed scoping plan. In conducting its case studies, the committee identified four scenarios that would likely arise in a class-based hazard assessment of OFRs. Scenario 1 involves a subclass that has many data-rich members on which data are concordant. For that scenario, the hazard determination for the subclass should be relatively straightforward.
In Scenario 2, there are no relevant data on any subclass member that can be used to conduct the hazard assessment. The lack of data should not imply that an OFR subclass is not hazardous. The committee identified the following options to move the assessment forward:
- Option 2-1: Generate toxicity data on the subclass. The committee recommends a tiered approach that initially relies on new approach methodologies (NAMs) that encompass computational modeling, in vitro assays in animal and human cells and tissues, and toxicity testing that uses alternative animal species, such as zebrafish. The results of such studies can help to identify potential end points of interest and one or more chemicals in the subclass for targeted animal toxicity studies.
- Option 2-2: Expand the analysis beyond the set of chemicals that were identified as OFRs and use toxicity data on structurally related chemicals.
- Option 2-3: Reclassify the subclass so that data-poor members are distributed in other data-rich subclasses. Many OFRs have multiple functional groups and could be placed in multiple subclasses; reclassification might help to minimize the number of data-poor categories. Confidence in the reclassification can be increased when concordant biologic responses are seen among the members of the newly expanded subclasses, for example, if additional data show a common mechanism or effect.
In Scenario 3, there are sufficient coherent data on one or two chemicals for assessment, but there are few or no data on the other class members. The few available data, however, might suggest that the subclass members have similar biologic activity. In this case, the committee identified the following possible options to move the assessment forward:
- Option 3-1: Make a science-based policy decision, for example, to classify the subclass as potentially hazardous on the basis of the data-rich chemicals in the subclass.
- Option 3-2: Use the data-rich chemicals to serve as an anchor as suggested above and extrapolate or interpolate to other chemicals in the subclass.
- Option 3-3: Generate toxicity data on data-poor subclass members to the extent that satisfactory confidence is gained; testing could involve NAM studies, targeted animal testing, or a combination thereof.
Scenario 4 is the most difficult to address. There are data on some chemicals in the subclass but few or no data on others, and the available data are so heterogeneous or inconsistent with respect to biologic activity that a discor-
___________________
3 The verbatim statement of task is provided in Chapter 1 of this report.
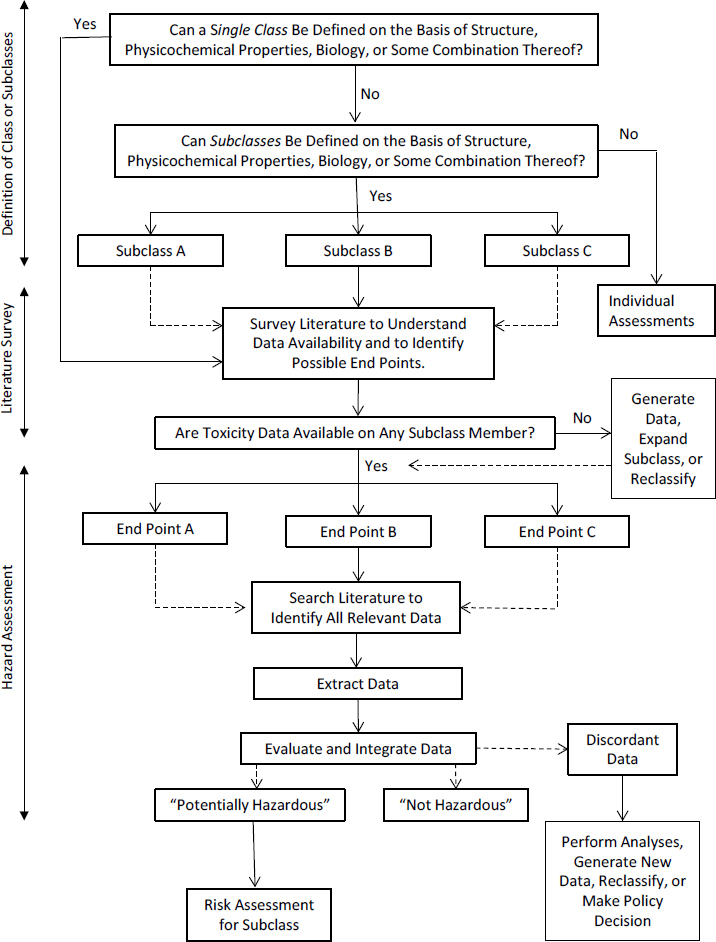
dant-data designation is reached. The committee identified the following possible options, which are discussed in further detail in Chapter 3:
- Option 4-1: Make a policy decision, for example, to extend the most conservative conclusion regarding hazard to the subclass.
- Option 4-2: Reclassify members in such a way that biologic similarity is improved; generate data to increase confidence that reclassification has resulted in biologically similar members.
- Option 4-3: Perform analyses that would help to explain the discordance and allow the assessment to move forward.
- Option 4-4: Generate new data that could increase clarity and the scientific basis of a decision.
CONCLUSIONS
In this report, the committee has provided a scoping plan for using a class approach to hazard assessment and illustrated aspects of the plan with case studies. Ultimately, the time and resources required to implement the plan will depend on several policy decisions that are beyond the committee’s charge. For example, CPSC will need to decide whether it will accept NAM data to set testing priorities for chemicals or to conduct its hazard and risk assessments. If not, the cost and time implications are
dramatic and a class-based hazard assessment for all relevant OFRs will be unlikely to be achieved. Specifically, relying solely on traditional (whole-animal) toxicology studies will require resources that are orders of magnitude greater than would be needed if NAM data or a combination of NAM data and targeted animal studies were used. CPSC will also have to determine the type and quantity of data necessary to achieve the confidence needed to draw conclusions about hazard and risk. If CPSC requires some data on each chemical in a subclass, the cost and time implications again are substantial. Moreover, as noted, the type of data required will have a strong bearing on the resources required. Ideally, the class approach provides a mechanism for extrapolating data on data-rich chemicals to data-poor chemicals and eliminates the need to collect data on all chemicals in a specific class.
The committee hopes that the scoping plan that it has described will give CPSC a means to use a class approach to assessing the hazards posed by OFRs. A class approach will likely result in increases in efficiency and decreases in cost compared with the traditional approach of evaluating individual chemicals. Although the challenges to a class approach might appear daunting, the alternative—individual assessments of hundreds of chemicals—is unrealistic. The only possible practical approach for a set of chemicals as large as the OFRs is a class approach.




