6
The Tropical Western Atlantic and Caribbean as a Case Study for Coral Interventions
The committee was tasked with assessing coral intervention strategies and their ability to meet objectives for sustaining coral reefs in the tropical western Atlantic (which includes the Caribbean and neighboring reefs in the Gulf of Mexico and outlying reefs in the northwest and southwest Atlantic, such as the Bahamas, Bermuda, and Brazil). These reefs vary widely in their condition and in the intensity of local stressors, and include areas that have experienced devastating coral reef losses in recent history. These losses have been due to the combined effects of a variety of factors, including disease outbreaks, bleaching events, and hurricane impacts, exacerbated by a variety of local stressors to which the reefs have had generally limited resilience.
Tailoring a decision strategy to a local area is a vital component of the decision process. In this chapter, the committee synthesizes the information about selecting and modeling interventions from the preceding chapters as it applies to the tropical western Atlantic and Caribbean. Specifically, which interventions may be particularly appropriate for consideration in this region given their context dependencies is explored (as discussed in Chapter 2). How the impacts of these interventions would be assessed in a model tailored to particular environments in this region is also considered (see Chapters 3 and 4). However, environmental context is only one component of a decision framework applied to a particular region; consultation with local stakeholders is also a necessary step in clarifying management objectives and identifying appropriate interventions (see Chapter 3). Because both stakeholder involvement and details
of the local reef ecosystem dynamics are needed to inform and refine model development, and because the region varies considerably in these respects, it is beyond the purview of the committee to conduct a formal assessment of objectives, alternatives, and intervention plans for the region as a whole. As an initial, framing contribution, this chapter lays out the unique nature of the reef ecosystems in this region that form the basis for undertaking this process, and outlines opportunities for pursuing some of the priority research described in Chapter 5.
TROPICAL WESTERN ATLANTIC AND CARIBBEAN REEF ECOSYSTEMS: GEOGRAPHY, DIVERSITY, ECOLOGY, AND RESILIENCE
The tropical western Atlantic and Caribbean region, including the Caribbean Sea, the Gulf of Mexico, Florida, the Bahamas, Turks and Caicos, Bermuda, and Brazil (hereafter referred to as “the wider Caribbean”) is an important reef area globally. Although these reefs represent only about 8% of the world’s total reef area (less than half that of Indonesia or Australia; Spalding et al., 2001), they are of considerable biological, ecological, cultural, and socioeconomic importance. There are 23 countries and 18 overseas colonies and territories in the region that have coral reef resources. These areas range from the poorest nation in the hemisphere (Haiti) to areas that remain politically affiliated with major western economies, such as Puerto Rico and the U.S. Virgin Islands (United States); Anguilla, Antigua and Barbuda, the Bahamas, Barbados, Bermuda, the British Virgin Islands, the Cayman Islands, Dominica, Grenada, Jamaica, Montserrat, St. Kitts and Nevis, and Turks and Caicos (United Kingdom); Guadeloupe, Martinique, St. Bart’s, and St. Martin (France); and Aruba, Bonaire, Curaçao, Saba, St. Eustatius, and St. Maarten (the Netherlands).
As a region, the wider Caribbean is oceanographically distinct. The Caribbean and the Gulf of Mexico represent a comparatively small and semi-enclosed region, influenced by nutrients from the outflows of the Amazon, Orinoco, and Mississippi River basins, as well as by iron from wind-blown dust (Pawlik et al., 2016; Roff and Mumby, 2012; Shinn et al., 2000). This means that, even on arid Caribbean islands, terrestrial influences tend to be higher than they are in the more oceanic parts of the Pacific and Indian oceans, with concomitantly higher nutrient loads and larger algal and microbial biomass. Hurricanes are common in the eastern and northern Caribbean, but are nearly absent from the southern Caribbean (Chollett et al., 2012a). Moreover, the southern Caribbean and Bermuda are potential refugia for climate change (Chollett et al., 2010), having experienced less frequent and less severe bleaching events and
tending to show longer times to annual severe bleaching (van Hooidonk et al., 2016).
The wider Caribbean has been completely isolated evolutionarily from the Pacific since the closure of the Isthmus of Panama about 2.8 million years ago (O’Dea et al., 2016), with periods of origination and extinction over the past 33 million years leading to its current distinctive and relatively low-diversity coral fauna (Budd, 2000). Today, the Caribbean and Pacific coral faunas are not closely related, with just a few shared genera (Siderastrea, Porites, and Acropora); notably, recent extinctions during the Plio-Pleistocene resulted in the loss of both Pocillopora and Stylophora from Caribbean reefs (Budd, 2000; Toth et al., 2015). Indeed, it is now recognized that, in a number of cases, Caribbean and Pacific scleractinian taxa that were originally thought to be congeners are actually in different families (Fukami et al., 2004), and some coral families are entirely or mostly restricted to the Caribbean (Fukami et al., 2008), with further endemism also characterizing Brazilian reefs. The characteristic algal symbionts associated with scleractinian corals also differ between the two reef regions (Baker and Rowan, 1997; LaJeunesse et al., 2003, 2018). Most corals in the Indo-Pacific host members of the genera Cladocopium and/or Durusdinium, whereas Caribbean corals commonly host Symbiodinium, Breviolum, Cladocopium, and/or Durusdinium. The latter symbiont genus may have been introduced from the Pacific (Pettay et al., 2015).
At the species level, reefs in the tropical western Atlantic are far less diverse than their analogs in the western Pacific; for example, there are only two species of Acropora (plus one hybrid) in the Caribbean, whereas there are hundreds in the western Pacific (Wallace, 2012). Similar diversity differences exist for reef fishes and other reef associates (Roberts et al., 2002). For this reason, it has been argued that Caribbean reefs lack the ecological/functional redundancy found on most Indo-Pacific reefs (Bellwood et al., 2004; McWilliam et al., 2018; Roff and Mumby, 2012). Nevertheless, until relatively recently (the 1970s), the percentage of coral cover was similar on reefs in the Caribbean and the Indo-Pacific, with reefs in both areas commonly reaching or exceeding 50% coral cover (Bruno and Selig, 2007; Gardner et al., 2003; Jackson et al., 2014). However, ecological decline of Caribbean reefs accelerated greatly beginning in the late 1970s, resulting in changes far greater than any observed during the previous 220,000 years (Jackson, 1997; Pandolfi and Jackson, 2006).
Basin-wide, major disease epizootics have been responsible for triggering some of the greatest changes. These began with the die-offs of structurally dominant elkhorn and staghorn corals (Acropora palmata and A. cervicornis) in the late 1970s (Aronson and Precht, 2001; Gladfelter, 1982) followed by the loss of a keystone herbivore, the black-spined sea urchin (Diadema antillarum) in the early 1980s (Lessios, 1988). Declines in
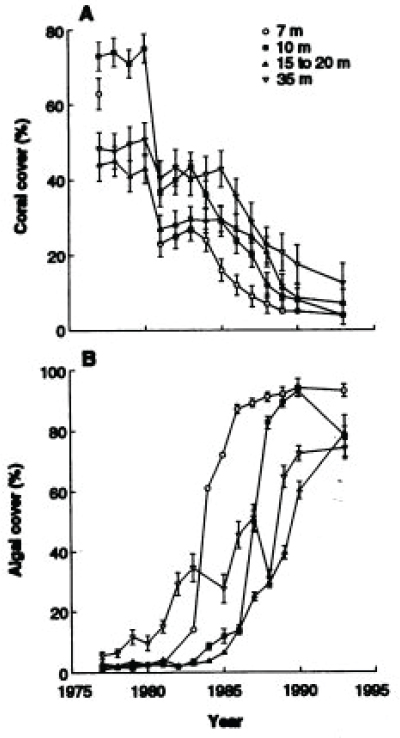
herbivory altered the competitive balance between fast-growing algae and slow-growing corals in a way that is now recognized as a fundamental property of coral reef ecosystems around the world (see Figure 6.1; Hughes, 1994). In some cases, corals have been replaced by other organisms such as octocorals or sponges (Norström et al., 2009; Ruzicka et al., 2013).
Other diseases have also directly affected coral populations (e.g., yellow-band disease, black-band disease, and various white syndromes), including the recent emergence of stony coral tissue loss disease (SCTLD), described below. It is possible that the wider Caribbean may be particularly vulnerable to disease due to its long isolation (Bak et al., 1984). Pathogenic agents are varied and remain generally unknown or uncertain (including some of the most important, such as the pathogen that
decimated D. antillarum). Nevertheless, it is likely that anthropogenic impacts (e.g., nutrient pollution, algal overgrowth, warming) have played a role in the rise of these diseases as ecological drivers (Lesser et al., 2007).
Diseases have not been the sole driver of coral decline in the region. Overfishing has compounded the impacts of the loss of D. antillarum (Hughes, 1994; Jackson et al., 2014), and the invasive lionfish, which has recently spread rapidly through the region, has aggravated the impacts of overfishing by preying on juvenile fish (Côté et al., 2013; Eckrich and Engel, 2013; Willette et al., 2014). Nutrient levels as well as differences in the diversity and abundance of fish herbivores, in particular, could help explain the apparent vulnerability of reefs in the wider Caribbean to algal overgrowth. Poor land-use practices (Bégin et al., 2013) and storm-driven wave surge (Knowlton, 2001) have also contributed to coral mortality by increasing sedimentation, nutrients, and turbidity. These sources of damage have in some cases led to the physical flattening of Caribbean reefs, that is, loss of three-dimensional structure, especially in shallow forereef and reef crest communities (Alvarez-Filip et al., 2009; Knowlton, 2001).
Taken together, these events and processes have had major impacts on Caribbean coral reef structure. Variation in these impacts, some local, some regional, some global, have led to marked heterogeneity in degradation among Caribbean reefs (Jackson et al., 2014; Knowlton and Jackson, 2008). At the same time, there has been widespread recruitment failure in some areas of the wider Caribbean, such as Florida, while recruitment has remained relatively high in other locations, such as Curaçao (Hartmann et al., 2018). Consequently, the trajectory of coral decline has not been uniform, and some areas have retained markedly higher coral cover than others (Jackson et al., 2014).
This suggests that the basic ecosystem dynamics of competition between fast-growing algae and slow-growing corals remain a powerful controlling force set by herbivory and growth rates, that local reef impacts due to fishing, land use, and tourism are common, and that local management still has an important role to play in potentially slowing or even reversing Caribbean degradation (Mcleod et al., 2019), even if these effects can be hard to detect (Bruno et al., 2019; Steneck et al., 2018).
Across this mosaic of local and regional effects, climate change impacts are increasing, and in the wider Caribbean, as elsewhere, there are limits to the effectiveness of local management. The western Atlantic has experienced background warming of about 0.015°C yr−1 on average (Glenn et al., 2015), but also shows substantial variability in rates of warming (Chollett et al., 2012b). This warming affects corals directly via heat-driven coral bleaching (Donner et al., 2007; Eakin et al., 2010), and indirectly by increasing intensities of tropical storms (Bhatia et al., 2019; Murakami et al., 2018) and more devastating coral disease epizootics
(Harvell et al., 1999). For example, the 2005 warming event that affected the northeastern Caribbean caused more than 10.2 degree heating weeks of thermal stress that resulted in more than 80% of corals bleaching and more than 40% of corals dying at many sites (Eakin et al., 2010). Bleaching was then followed by severe disease impacts (Miller et al., 2009; Smith et al., 2013). Severe bleaching was also observed in 2010 in some parts of the Caribbean (Alemu and Clement, 2014; Smith et al., 2013), also followed by disease outbreaks. Repetitive bleaching in Florida in 2014 and 2015 was also accompanied by disease (Gintert et al., 2018; Precht et al., 2016), as described below.
Recent Emergence of a Highly Lethal Coral Disease
Recently, a new disease syndrome, SCTLD, has emerged in Florida and parts of the central and northeastern Caribbean. The disease is an extremely virulent and persistent epizootic affecting a large proportion of coral species and consequently threatening coral diversity. This disease accompanied repetitive bleaching in Florida in 2014 and 2015 and is illustrative of the problem of compounding threats to coral persistence. It also serves as a reminder of how disease presence within coral populations might change how some interventions, such as those designed to increase thermal tolerance, might be implemented.
SCTLD was first documented in 2014 by contractors monitoring the environmental impacts of dredging at the Port of Miami at the northern end of the Florida Keys (Precht et al., 2016) and has since spread both north and south, reaching the northern limit for reef-building coral near Martin County, Florida, by 2017 and extending south to Looe Key (near Key West) by early 2018 (NOAA, 2019). As of May 2019, SCTLD had reached Sand Key, about 5 miles southwest of Key West (Florida DEP, 2019). SCTLD affects up to 22 species, but with significant variation among them in the severity and extent of the disease. Rates of mortality are high in susceptible species and can reach 100%, resulting in locally severe impacts on species diversity and potentially widespread reductions in genotypic diversity. The outbreak has prompted an interagency rescue effort—the Florida Reef Tract Rescue Project—to collect genotypes from ahead of the disease front and maintain them in ex situ land-based facilities until reintroduction and restoration can occur with a high chance of success. The causative agent (or agents) is not known, but waterborne transmission has been demonstrated. Disease lesions have been successfully treated with antibiotic pastes (Neely, 2018), suggesting a bacterial pathogen with potential co-relationships with viruses.
Unlike most diseases, which tend to flare up over the warmer summer season and then disappear over the cooler winter months, SCTLD
has persisted and continued to spread over multiple years. The initial appearance of the disease followed a bleaching event in late 2014, but then spread to affect corals that had already recovered from bleaching. The subsequent spread of the disease does not appear to have been linked to continued thermal stress. By spring 2018, corals with disease signs similar to SCTLD were reported in Jamaica (AGRRA, 2018), and by July 2018, it was reported in Puerto Morelos, Mexico (Alvarez-Filip, 2018). The disease had spread to St. Maarten by November 2018 (The Daily Herald, 2018), the U.S. Virgin Islands by February 2019 (Virgin Islands Consortium, 2019), and the Dominican Republic by March 2019 (Irazabal and Rodriguez, 2019). The relatively rapid and sudden appearance of the disease outside the Florida Reef Tract highlights the high interconnectivity of wider Caribbean reefs and the capacity for the rapid spread of diseases (which, with invasive species, is a risk factor for some interventions considered in this report), although the location of new outbreaks near ports may indicate transport by humans rather than via currents. There is concern that SCTLD, if it continues to spread regionally, has the ability to transform wider Caribbean reefs, as previously happened following the epizootic that decimated Diadema in 1983-1984, and the loss of Acropora from white-band disease in the 1970s and 1980s. In affected areas, such as Florida, it is not known if the disease will abate once susceptible individuals are removed from the population, or if the disease will remain a chronic problem once the initial outbreak has passed through.
IMPLICATIONS FOR SELECTING AND MODELING INTERVENTIONS
The environmental context of the wider Caribbean has direct implications for how to approach the decision to implement coral interventions. Here, the committee identifies the context dependencies that would influence analyses aimed at deciding which interventions to test and deploy (as discussed in Chapters 2, 3, and 4), as well as the environmental parameters that would be important to include in a dynamic coral model in which the risks and benefits of the intervention(s) may be evaluated.
Generally Poor Conditions and Intrinsic Vulnerability
In general, the Caribbean appears to have low resilience (a resilience “coolspot”; Roff and Mumby, 2012) with less functional redundancy in the wider Caribbean compared to the Indo-West Pacific (IWP) (McWilliam et al., 2018) and characteristics that promote algal overgrowth of corals. The wider Caribbean is generally further along the trajectory of decline than the Pacific (Gardner et al., 2003; Jackson et al., 2014; Roff and Mumby,
2012), although degradation of reefs in some areas of the Pacific is increasing (Bruno and Selig, 2007; De’ath et al., 2012; Hughes et al., 2018). As noted above, some wider Caribbean reefs have low coral cover and rampant disease, lack rapidly growing branching corals, and show very low coral recruitment. These attributes may imply potentially higher risk tolerance for interventions for some reef managers in these particularly degraded areas (e.g., Florida). On the other hand, lower resilience and poor condition may mean a higher risk of crossing a harmful tipping point or a lower likelihood of success when interventions are applied.
Generally poor conditions on such reefs demand models of interventions (e.g., as illustrated in Chapter 4) that include starting scenarios of low coral cover, poor recruitment, and vigorous algal growth. Additionally, processes representing local stressors, such as overfishing (which reduces herbivory) and poor water quality (which can accelerate disease and algal growth), will be important to evaluate to consider the impact of their management in comparison to the impact of, or need for, coral interventions. Reef models that evaluate interventions need to be parameterized with initial conditions that reflect current conditions of reefs in the wider Caribbean, including regional variability in reef ecosystem health, impacts of stressors, and community attitudes on risk acceptance.
Interconnectedness
The rapid spread of diseases and invasive species indicates that both the risks and benefits of biological interventions could spread quickly in the wider Caribbean. On the one hand, this suggests that there are relatively few isolated reefs that might be safe spots to test risky interventions in the field (see Chapter 2). On the other hand, local increase and spread of heat-tolerant genotypes, disease-resistant colonies, or heat-resistant symbionts might lead to regional benefits. It may be, for example, that the heat-resistant symbiont Durusdinium trenchii has already invaded the Caribbean from the Pacific (as opposed to being native; Pettay et al., 2015). For interventions that realize both benefits (through enhanced connectivity) and risks (through disease spread), such as assisted gene flow and assisted migration, incorporating this interconnectedness into a modeling framework can reveal whether interventions can outperform expectations from natural dispersal and assess the spatial scale of risks.
Differences in Diversity of Coral and Algal Symbionts
Algal symbiont diversity at the generic level is high in the Caribbean compared to the IWP, but total species diversity of coral hosts as well as algal symbionts is lower. The implications are uncertain: lower diversity
could mean either greater or lower responsiveness to interventions. Disentangling the role of diversity on multiple levels, including the relative efficacy of interventions to promote different types of diversity, would require a multispecies or genetic modeling framework of coupled host–symbiont dynamics.
Environmental Variability
The fact that Bermuda and the southern Caribbean may represent climate refugia, with long periods of time between bleaching events (van Hooidonk et al., 2016), has implications for the geography of interventions. These areas may have sites where protection of existing biodiversity is central to a suite of interventions, although these sites may also be less likely to contain genotypes of corals or symbionts that are highly heat resistant. Conversely, areas such as the Florida Keys, where reefs are already relatively highly degraded, may find riskier interventions to be acceptable in order to provide outcomes that enhance reef persistence. Making distinctions among different areas within the larger Caribbean region would require models with location-specific parameterizations including downscaled climate models and local stakeholder and management input.
Ongoing Acute Disease Outbreak
The presence of diseases, such as SCTLD, has the potential to change the way that thermal stress interventions are applied on Caribbean reefs, and provides a sobering lesson on the complexities of implementing interventions in the face of multiple stressors. Some interventions, such as pre-exposure, are likely to be much less effective during a disease outbreak because the application of an additional stressor to a diseased coral may be more likely to lead to mortality. In addition, bringing diseased corals into a laboratory setting and then redeploying them in the field may set the stage for laboratory-based transmission. Other interventions, such as managed relocation, may have extremely adverse effects if undertaken during a disease outbreak, for example, if corals are moved from an area where the disease is endemic to an area where the disease is not present. These interventions might well be rejected in such a case. Even moving corals in the opposite direction—from outside the diseased area into the diseased area—could exacerbate the disease by increasing the number of susceptible corals and fueling the spread of the disease in the endemic area.
Diseases such as SCTLD, by greatly reducing population size, also threaten to decrease the pool of potentially heat-resistant alleles available
for genetic interventions designed to promote the spread of those alleles. As a result of this concern, efforts are already under way to safeguard coral diversity in areas not yet exposed to SCTLD by removing representatives of susceptible species from reefs and holding them in ex situ facilities until such time as they can be used in a subsequent restoration program or applied to another conservation purpose. If the disease becomes chronic, continuously affecting susceptible species, these “rescued” corals may not be suitable for restoration inside the endemic area, and indeed corals sourced from inside these endemic areas may not be able to be used for restoration outside the same area. Such impacts underscore the need to prospect for coral genotypes that are both heat resistant and disease resistant (e.g., Muller et al., 2018) and to understand the genetic underpinnings of both as a part of a comprehensive intervention strategy, which is an ambitious goal. Interventions that focus on the treatment of disease (e.g., antibiotics, phage therapy, and microbiome manipulation) are clearly warranted in response to SCTLD, and testing such interventions is now a priority on affected reefs. A modeling framework that explicitly incorporates strong disease dynamics as they relate to both risks (disease spread for interventions that entail moving corals) and benefits (interventions that might enhance disease resistance) is particularly relevant to the wider Caribbean.
Finally, the rapid and extreme impacts of SCTLD in Florida—a relatively degraded coral reef system—may change managers’ risk tolerance and willingness to attempt interventions. The perceived risk of interventions may be considerably lower than the risks of disease-driven reef declines. The coral disease responses currently being executed in Florida include some activities that are starting points for coral interventions, such as the relocation of corals from outside the disease endemic zone to ex situ land-based facilities to preserve genetic diversity in advance of the disease spreading. Responses are now being considered that would have faced higher resistance until very recently, such as removals of infected colonies to reduce pathogen loading (currently being attempted in the U.S. Virgin Islands; Marilyn Brandt, personal communication, May 13, 2019).
Small But Politically Complex Region
Many countries effectively share coral reef resources and will also share risks and benefits of interventions. Some outliers, notably Bermuda and Brazil, are isolated from other reef systems (or at least represent the end points in a connectivity network), and thus may have the ability to pursue interventions more unilaterally. Several Caribbean possessions or territories are still governed by former colonial powers (France, the
Netherlands, the United Kingdom, and the United States), and these nations have jurisdictions that cover fairly broad and discontinuous areas. However, many other reef regions within the Caribbean (e.g., the Lesser Antilles) are managed by multiple countries.
Some interventions, such as cloud brightening to reduce surface water temperature or assisted gene flow over hundreds of miles, operate on a large enough scale to demand regional coordination. Other interventions, such as larval seeding or assisted gene flow over small distances, may be able to be accomplished within a very small jurisdiction. In the wider Caribbean, the degree of coordination among countries will affect the feasibility of using those interventions that necessarily cross borders (i.e., those that move corals from one area to another, or those that are likely to spread outside their direct area of implementation).
OPPORTUNITIES FOR INTERVENTIONS
The committee was tasked with assessing the interventions in expected near-future environmental conditions in the Caribbean region, relative to objectives and performance measures for conserving coral reefs. Selection of management objectives and associated metrics (e.g., Table 3.1) is a stakeholder-driven process that also includes an assessment of acceptable courses of action to meet these objectives (described in Chapter 3) that the committee did not undertake. The committee does not recommend specific objectives, and did not systematically review the applicability of all possible interventions for the Caribbean region, which are outlined in detail in the previous report (NASEM, 2019). Instead, the committee highlights interventions that stand out as being particularly well suited to the region, given the considerations of ecology, history, current state, and existing capacity outlined above. They therefore reflect the regional context and the context dependencies of the interventions, and reflect the assessments of Chapter 2. Depending on community objectives, these strategies, alone or in combination across different sites throughout the region, would be considered within a structured decision framework.
It should also be emphasized that these interventions are not intended as a substitute for conventional management actions, which continue to be essential across the Caribbean, with particular attention being paid to maintaining water quality and herbivory in local management jurisdictions. The importance of this point is supported by the fact that much of the decline of Caribbean reefs occurred prior to the onset of major bleaching events (Gardner et al., 2003), and also by the preliminary modeling results presented in Chapter 4. Similarly, these interventions cannot be expected to conserve coral reefs in the absence of actions to reduce climate change impacts such as through emissions reductions and carbon sequestration.
All interventions would benefit from additional research to improve their efficacy and understanding about their risks and benefits. Chapter 2 includes an assessment of which interventions are closest to being technically ready for implementation in the near term, were they to be identified as favorable options through a structured decision process. Ultimately, research is likely to be needed to understand and improve the efficacy of many other interventions in the Caribbean, and undertaking this decision process can help identify research priorities to improve promising interventions not ready for implementation. Some research needs are also mentioned here to highlight priority research relevant to the Caribbean more thoroughly explored in Chapter 5.
Identifying heat tolerant or disease resistant coral genotypes among the Caribbean standing stock to provide opportunities for assisted gene flow, managed breeding, and genetic interventions: Understanding the intraspecific range of thermotolerance would be helped by obtaining a better understanding of population structure of Caribbean species among habitats, localities, and regions. Moreover, quantitative data on the environmental differences among habitats would help with the interpretation of these data. Many species have habitat-specific subpopulations and, although not unique to the Caribbean, the restricted fauna offer an opportunity to fully understand key reef-building species and their potential range of thermal responses. In addition, a broad diversity of habitats and a historically species-poor fauna may have allowed significant habitat specialization within species. If so, this could be harnessed in interventions.
Some individuals of important reef-building Caribbean taxa (e.g., Orbicella and Acropora) have shown resistance to thermal stress, disease, and reduced water quality in the laboratory (Muller et al., 2018) and field (large colonies with low partial mortality after disturbance). Research into the basis of this resistance and co-resistance could identify alleles for use in propagation and managed breeding. More immediately, large-scale ongoing efforts to outplant corals could include monitoring of heat, disease, and water quality resistance among the varied suite of nursery colonies across these varied efforts. Because research has shown that colonies with different genotypes and symbionts can differ strongly in these traits (see Muller et al., 2018, for a Caribbean example), systems for tracking coral/symbiont genotypes in nursery settings are being developed (Kitchen et al., 2018). Similarly, identification of algal symbionts or microbiome components that confer resilience to stress would help restoration practitioners select resilient corals, or provide insight into how to manipulate holobionts in favor of resilient types (Cunning et al., 2018; Silverstein et al., 2015). A system of best practices for coral restoration that leverages these efforts
to provide information on resistant colonies would parallel similar longstanding efforts in terrestrial restoration and agriculture.
Leveraging existing coral restoration activities and infrastructure to create a comprehensive region-wide program to boost larval recruitment and survivorship: Given that the ultimate goal of asexual restoration is to produce sexually reproducing, self-sustaining populations that can maintain genetic diversity and therefore potentially be hardier in future conditions, more effort could be invested in determining the extent to which this is actually occurring in ongoing restoration programs. Restoration practitioners could be encouraged and incentivized to quantify the frequency, incidence, and output of sexual reproduction among outplanted corals, determine the biotic and abiotic parameters that are associated with sexual maturity and fecundity, and modify outplanting protocols to maximize eventual sexual reproduction. This leverages existing infrastructure and represents an opportunity to build supportive breeding activities into ongoing asexual restoration programs that are currently focused on outplanting, greatly increasing the long-term impact of these activities. Rather than representing an additional burden on restoration practitioners, incorporating breeding programs into nursery and outplanting activities could be encouraged and incentivized by funding agencies as part of scaling up. In addition to the value of producing coral recruits, sexual reproduction among colonies with diverse combinations of alleles can maximize opportunities to produce successful genotypes. This would be potentially advantageous for two objectives: (1) resisting impacts from climate change, but also (2) preserving diverse assemblages more capable of resisting the multiple stresses experienced in the most degraded Caribbean reefs.
A limiting factor in the success of these efforts is the relatively low natural levels of sexual reproduction and coral recruitment in some areas of the wider Caribbean region that might severely limit the success of managed breeding programs. Although progress is being made in developing interventions in some areas of the Caribbean that have high recruitment (e.g., in Curaçao; Hartmann et al., 2018), it is not known whether low success in other areas is a result of intrinsic factors that might in theory be resolved by interventions and restoration (e.g., low genetic diversity and inbreeding) or is driven by extrinsic environmental factors that cannot be directly improved by interventions considered here and is better managed through standard management actions (e.g., improving water quality or increasing herbivory).
Exploiting sexual restoration activities to test algal symbiont manipulations: Existing programs to collect, settle, and outplant coral larvae could also incorporate algal symbiont manipulations to test whether coral
recruits can be supplied with different algal symbiont types, in particular thermotolerant Durusdinium trenchii (Williamson et al., 2018). Such trials could also determine whether these symbionts are retained into adulthood and increase heat tolerance and could be used to assess potential tradeoffs, such as reduced growth rates (Little et al., 2004).
Expanding coral cryopreservation across the region to provide opportunities for managed breeding and assisted gene flow: Dissemination and training in protocols for gamete cryopreservation (e.g., Hagedorn et al., 2012, 2017) would allow researchers and restoration practitioners working on coral spawning to preserve gametes, both for long-term gene banking purposes to guard against continued loss of diversity and for immediate use in managed breeding and assisted gene flow (Hagedorn et al., 2018) programs, for which, by necessity, either sperm or eggs must be frozen and transported for later use in fertilization trials. Space could be acquired in existing national gene bank facilities to preserve these genetic materials long term.
Testing short-distance managed relocation (i.e., assisted gene flow) of corals across local thermal gradients, where disease incidence is not a limiting factor: Efforts could be made to prioritize projects with the potential to increase heat tolerance by sourcing resistant corals within management jurisdictions, across minimal relocation distances (Baker et al., 2018). Areas could be identified that are already under elevated thermal regimes, with adapted/acclimatized corals being used as source populations. Identifying locations where naturally occurring heat- or disease-resistant corals are more common might help efforts to provide raw material for nursery propagation and/or managed breeding, as above.
Extending relocations over larger distances would require balancing the potential gains in thermal tolerance (which exist due to large variation in thermal tolerance across the region), with the potential loss of fitness due to local selection (which may have already tuned adult corals to the particular physical environmental conditions at their respective natal sites), as well as the risks associated with spreading disease highlighted above. Testing whether heat-tolerant corals sourced from distant locations can survive equally well as their local counterparts under nonbleaching conditions remains an open question, not just for the Caribbean, but for translocations in other regions as well.
Leveraging restoration activities to test pre-exposure methods to increase stress tolerance of outplanted corals: Existing restoration activities in the region provide an opportunity to test pre-exposure interventions designed to increase thermal tolerance. For example, acute methods of pre-exposure, such as controlled bleaching and recovery of corals prior
to outplanting, may increase the abundance of heat-tolerant Durusdinium trenchii (Cunning et al., 2018), and pilot tests are determining the feasibility of this intervention, as well as assessing the longevity of its impacts and potential tradeoffs (Cabral et al., 2018; Winter et al., 2018). Similarly, testing whether chronic “stress-hardening” nurseries located at sites with more disturbed conditions can generate stocks of hardy corals for outplanting through pre-exposure might also be worthwhile. Such approaches can increase heat tolerance by promoting different algal symbionts (or microbiome communities) as well as other potential effects, such as front-loading of coral heat-tolerance genes (Barshis et al., 2013).
Assessing feasibility of environmental interventions to reduce heat stress at both local and subregional scales: Shading and cooling interventions may reduce the degree of thermal stress, and atmospheric shading may do so in a way that operates at large scales. Although early modeling suggests that the marine cloud layer is less suitable for cloud brightening in the Caribbean (Alterskjær et al., 2012), it would be important to determine if this applies to the warm season when bleaching conditions (doldrums) occur. Monitoring the growing risk from ocean acidification would also inform interest in implementing ocean acidification interventions.
Testing interventions, such as antibiotics, phage therapy, and microbiome manipulations, to halt the spread of emerging diseases, improve coral condition, and increase the success and/or feasibility of other interventions: The relatively high vulnerability of the Caribbean to disease, and the current spread of panzootics such as SCTLD, mean that this region is a high priority for testing and implementing disease interventions. There is a clear need to investigate techniques to slow, block, and halt the disease (e.g., antibiotics, phage therapy, and microbiome manipulations/probiotics) as well as approaches to preserve genetic resources prior to the arrival of a nearby disease front. There is an urgent need to develop quarantine and disinfection procedures to minimize the unintended spread of these interventions beyond the immediate site of interest. Active diseases in these areas also affect other interventions unrelated to disease that might introduce or spread pathogens (e.g., managed relocation). As discussed above for SCTLD, some interventions (e.g., pre-exposure or managed relocation) may be counterproductive if undertaken during an active disease outbreak.
Combining interventions where possible to increase resource efficiencies: As noted in Chapter 2, combined efforts can promote efficiency and could be profitably explored. For example, a sexual restoration program described above could incorporate both symbiont manipulations of larvae
and cryopreservation of gametes, be used to assess the heritability of resistant traits such as disease resistance, or cross disease-resistant genotypes with a native population in order to boost disease resistance.
Testing the efficacy of interventions under a range of different conditions by exploiting variability in the degree of degradation across the region: Areas of the wider Caribbean that are relatively intact (e.g., Bermuda, Bonaire, and Curaçao) could be compared with areas that are relatively more degraded (e.g., the Florida Keys) to test if the outcomes of interventions vary as a result of initial conditions, both for comparative modeling and for field testing. In addition, this same concept could also be applied within these areas where heterogeneity in reef degradation exists over local scales. It would be useful to know if certain areas of the Caribbean (e.g., the Florida Keys) are already unsuitable for interventions because underlying conditions from other stressors, apart from thermal stress, are already inimical to coral reefs.
More generally, resistance of Caribbean corals to thermal stress can be high in some cases (Smith et al., 2013), yet recovery, the other component of resilience, can often remain very low (Jackson et al., 2014; Roff and Mumby, 2012). This is fundamental to understanding the long-term trajectory of Caribbean corals and the benefit of interventions. Resilience science—understanding factors that promote or inhibit recovery from disturbance—is critical in the Caribbean and is central to understanding if interventions will have long-term success. For example, it is still an open question whether dust from the Sahara negatively impacts Caribbean resilience (Shinn et al., 2000), yet this could be fundamental to understanding the impact of interventions. In addition, interventions that promote recovery by breaking positive feedback cycles (e.g., restoration of genetically diverse stands of Acropora cervicornis; Roff and Mumby, 2012) could be identified.
Developing regional and multinational coordination and agreements to meet the scale of the challenge: The large number of jurisdictions in the Caribbean suggests that it may be beneficial if a program to manage and coordinate intervention efforts regionally were established and supported, perhaps as a working group of the Coral Restoration Consortium.1 Some of these efforts have already begun, for example, the multi-institution Florida Reef Tract Rescue Project currently ongoing for SCTLD within Florida (Florida DEP, 2019). These coordinated efforts could help share successes and avoid waste on duplicative failures, although not at the expense of innovation and risk taking. Internationally, large-scale cooperative research and funding agreements could be pursued, both through
___________________
1 See http://www.reefresilience.org/restoration/coral-restoration-consortium.
international agencies (e.g., World Bank, United Nations Environment Programme) and intergovernmental collaborations (e.g., a U.S.–Australia binational agreement).
Soliciting and incorporating stakeholder input on interventions to gauge and maximize acceptability/social license: Dramatic differences in reef health and economic resources across the region, as well as the multinational, multilingual, and multicultural nature of the Caribbean region, mean that assessments of intervention feasibility are likely to be variable across these gradients and will change with time.
Conclusion: Coral reef managers in the tropical western Atlantic/Caribbean region have a variety of interventions available to them depending on the localized management context and the specific objectives of stakeholders and decision makers. Available actions include leveraging existing restoration and propagation infrastructure, increasing sexual reproduction and genetic diversity of corals (managed breeding, gamete and larval capture and seeding, coral cryopreservation), capitalizing on thermally tolerant species and genotypes (managed selection, algal symbiont manipulation), accelerating reef connectivity to boost thermal tolerance when disease is not a factor (managed relocation), reducing disease spread (antibiotics, phage therapy, microbiome manipulation), and/or reducing exposure to stress (environmental interventions). The complex disease geography in the Caribbean requires particular care to ensure that interventions do not facilitate the spread or severity of ongoing disease outbreaks. These rapidly developing new interventions do not replace the need for direct management of local stressors.
Recommendation: The ongoing management and restoration efforts in the Caribbean provide a strong foundation on which to implement newly emerging interventions designed to increase the resilience of individual corals and coral populations. The modeling and decision-making tools outlined in this report should be used to inform more detailed assessments to evaluate which approaches might be appropriate for specific settings, including their interactions with more traditional management approaches. Maintaining genetic diversity in the face of multiple climate-driven stresses (e.g., bleaching and disease) is particularly important. Monitoring corals to maintain genetic diversity and identify resistant phenotypes should be simplified and standardized for research, ex situ propagation, and in situ restoration. Research programs to model and field test the risks, benefits, and efficacy of interventions in this multinational and highly interconnected region should be coordinated to maximize resources, co-learning opportunities, and the ability to achieve management objectives regionally.
This page intentionally left blank.


















