










Below is the uncorrected machine-read text of this chapter, intended to provide our own search engines and external engines with highly rich, chapter-representative searchable text of each book. Because it is UNCORRECTED material, please consider the following text as a useful but insufficient proxy for the authoritative book pages.
3 Key Issues for Salt Detection and Remediation This section provides guidance related to nine soluble salt detection and remediation questions. Following each question is a summary of the key points and a more detailed discussion of the issue. In some cases, the reader may also want to refer to the detailed technical discussion in the technical report that accompanies this project. When Is There a Significant Corrosion Risk Associated with Soluble Salt Contamination? Key Points ⢠Soluble salt contamination is most likely to be a significant concern in actively corroding (i.e., rusted) areas of bridges that are exposed to sources of soluble salt contamination (e.g., road salts, marine environments, industrial pollution). ⢠Soluble salt contamination is most likely to have an adverse impact when it is present on the structure to be coated, thorough cleaning is not performed, a less tolerant coating system is used, and/or the service environment is severe (e.g., high time-of-wetness, corrosive). Discussion The maximum level of salt that should be allowed on a surface is a function of the risk associated with coating failure, the feasibility of soluble salt remediation, and the desired performance. Table 1 includes specific risk factors that should be considered. In situations with one or more high-risk factors, increased testing and/or stricter accep- tance criteria should be specified. In low-risk situations, soluble salt remediation may not be a significant concern. The factors in Table 1 are grouped according to whether they predominately affect likelihood of contamination, feasibility of remediation, or consequence of residual salt contamination prior to painting. The first category of risk factors is the likelihood of soluble salt presence on an existing bridge before a bridge painting project begins. Salt contamination is most likely on bridges that are close to an obvious source of contamination such as road salting, seawater, or industrial contaminants. Absent those sources, contamination is less likely but certainly not impossible. Salt contamination on painted surfaces tends to be easily removed by pressure washing. In some cases, rain- water is able to remove salts from coated surfaces. As a result, surfaces with intact coatings prior to surface preparation are less likely to be contaminated. Surfaces with degraded coat- ings or heavy corrosion are likely to have high levels of salt contamination. It is prudent to consider heavily corroded areas (e.g., Condition C or D in SSPC VIS-1) a high risk for underlying soluble salt contamination. The second category of risk factors is the feasibility of removing soluble salts from a structure during surface preparation. Remediating salts from a well-coated area can be readily achieved using a pressure washer. This approach is recommended before mechanical surface preparation techniques to ensure that soluble salts are not transferred to a prepared surface prior to coating. As discussed under the heading âWhat Are the Best Practices to Remediate Soluble Salts?â in most cases abrasive blasting techniques can effectively remove soluble salts from steel bridges before painting. In heavily contaminated areas, supplemental wash- ing or multiple abrasive blasting iterations may be required to reduce salts below required limits. Note that it is more difficult to remove salts from complex structural details. In some cases, such as back-to-back members with heavy pack rust or very tight crevices, it is not possible to remove all soluble salts (and pack rust). It is also not possible to detect soluble salts in these areas using available field techniques. The third category of risk factors is the consequence of any residual soluble salt on a surface prior to coating appli- cation. Soluble salts impact coating performance on a steel C H A P T E R 2
4 bridge through two primary mechanisms. First, salts under a coating may osmotically draw moisture through the coat- ing to the steel-coating interface, forming osmotic blisters. Osmotic blistering depends on the availability of moisture in the environment (e.g., time of wetness, humidity) and the resistance of the coating to water vapor transmission (a function of the coating material and applied thickness). The second primary impact of salts is to depassivate the steel surface, facilitating increased corrosion rates where there is access to water and oxygen (e.g., coating defect and mois- ture). Coating systems that are higher build, have a sacrificial primer, and are free of defects are more tolerant of soluble salt contamination. Finally, the desired service life of the coating systems must be considered. Soluble salt contami- nation is also less consequential in arid environments or in situations where the desired service life is short. What Indicator of Soluble Surface Salts Should Be Measured? Key Points ⢠Both chloride ion density and total salt density calculated from conductivityi are preferred indicators of soluble salts for the purposes of evaluating cleanliness of a prepared surface. Conductivity, sulfate ion density, and nitrate ion density are also useful methods under certain circumstances. ⢠Since equivalence between chloride ion density and total salt density calculated from conductivity cannot be universally determined, specifications should either (a) only allow one method or (b) specify a limit associated with each criterion. If limits associated with each criterion are specified, they should be specified as acceptable alternatives (e.g., âX µg salt/cm2 or Y µg Clâ/cm2â). Since they are not equal metrics, potential conflicts may arise in the field, as one criterion may be accept- able while the other is not. Discussion Soluble salts can be measured using a variety of ion- specific or conductivity techniques. Figure 1 illustrates the hierarchy of soluble salt detection techniques. Both chloride ion density and total salt density calculated from conduc- tivity1 are highlighted as preferred indicators of soluble salts for the purposes of evaluating cleanliness of a prepared surface. It may be prudent to allow a contractor or inspector the flexibility to use either ion-specific or conductivity methods, however the specifier should note that the methods are not directly comparable as discussed later in this section. Issue Low Risk of Impact High Risk of Impact Predominately Impacting the Likelihood of Soluble Salt Contamination 1. Proximity to sources of soluble salt contamination No deicing salt use, location away from the coast and industrial pollutants Road salt use, coastal location, near significant pollution sources, or steel shipped to a job site in a manner that could become contaminated 2. Corrosion scale tends to retain more soluble salt than intact coating Primarily cleaning existing, adherent coating with minimal surface rust (e.g., Condition A or B in SSPC VIS-1) Primarily cleaning degraded coating with heavy rust scale (e.g., Condition C or D in SSPC VIS-1) Predominately Impacting the Feasibility of Soluble Salt Remediation 3. Effort to remediate soluble salts from a surface Minimal rust scale and negligible sources of soluble salt Heavy rust scale and significant source of soluble salt (e.g., heavy road salting or marine environment) 4. Surface preparation methods being used Abrasive blasting techniques requiring a high degree of cleanliness (e.g., near white metal) Power tool cleaning or other less thorough cleaning methods 5. Practicality of removing soluble salts from complex structures Simple structure with rolled beams and sheltered joints Structure with significant built-up members such as lacing bars and splice joints that are exposed to soluble salt sources Predominately Impacting the Consequence of Residual Soluble Salt Prior to Painting 6. Retained soluble salts interact with moisture to impact coating performance Arid environment, well drained structure High time of wetness caused by long periods of high humidity or collected water 7. Coating materials that are more tolerant of soluble salt contamination Three-coat systems with organic or inorganic zinc-rich primers Relatively thin, barrier coating systems with high water vapor permeability (e.g., 8 mils of alkyd) 8. Service life expectation before significant coating breakdown Maintenance painting for temporary protection (e.g., 3â7 years) Long life of a newly installed coatings system (15â35 years, depending on environment) Table 1. Impact of soluble salt during recoating of steel bridges.
5 Conductivity, sulfate ion density, and nitrate ion density are also useful methods under certain circumstances. When requiring or reporting soluble salt surface concen- trations, it should be clearly stated what is being measured. For example, calculated total salt density should be reported as âµg salt/cm2,â conductivity should be reported as âµS/cm,â chloride ion concentration should be reported as âµg Clâ/cm2,â and so forth as indicated in Figure 1. This will eliminate confusion when interpreting specifications, reviewing data, or performing any given test method. When using ion-specific techniques, chloride ions are most commonly measured. This is in part because much of the research into the effect of salts on coating perfor- mance was concerned with deicing salts and seawater, in both of which chloride ions are a significant constituent. Additionally, chloride-ion specific detection methods were the first to be used in the field. Test kits are also currently available to detect sulfate and nitrate ions. The effect of other salts on coating performance has not been as thoroughly studied. Conductivity is a relatively easy-to-measure indicator of the total soluble salts extracted from a surface. Conductivity instruments can detect lower levels of contamination than ion-specific techniques but are also more sensitive to small levels of interference. Contaminated test equipment, impuri- ties in extraction fluid, and other sources of contamination can influence the solution conductivity. Finally, conductivity is a volumetric property rather than a surface concentration. Conductivity of extraction fluid is a function of both the surface area tested and the volume of extraction fluid. ISO 8502-9 provides a formula to convert conductivity to surface density of salts. Each anion and cation contribute to the solution conductivity in proportion to its concentration and specific conductance. The formula contains a mathematical constant that is representative of the range of soluble salts that might be found in an industrial painting environment. The operator cannot determine the exact ions contributing to the conductivity without an ion-specific test. While it is not possible to quantitatively convert between chloride ion density and total salt density calculated from conductivity, pragmatically comparable values have been established by the U.S. Navy (see Table 2). However, recall that the conductivity measurement is a function of both the surface area tested and the volume of extraction fluid. There- fore, these comparable values are limited to measurements performed using the U.S. Navy procedure. Because conductivity is dependent on the measurement process, a specifier should indicate calculated total salt if a conductivity method is allowed. Since the total salt reading calculated from conductivity will almost always be higher than an ion-specific reading, a specifier can conservatively allow conductivity measurements to be used with the same allowable limit as the ion-specific technique. Alternatively, an acceptable total salt value would be higher than an acceptable ion-specific (e.g., chloride) value. When requiring or reporting soluble salt concentration as a conductivity value, the test method used should be clearly stated. Conductivity results from various methods may not be directly comparable unless converted to a total salt density. Other things being equal, methods that use more water or extract from a smaller surface area will detect a lower conductivity. Similarly, methods that use less water or extract from a larger surface area will detect a higher conductivity. Soluble Salt Detection Methods Measuring Conductivity Calculated "Total Salt" Density (µg salt/cm²) Measured Conductivity (µS/cm) Methods Measuring Specific Ions Measured Chloride Ion Density (µg Cl¯/cm²) Measured Sulfate Ion Density (µg SO4²-/cm²) Measured Nitrate Ion Density (µg NO3-/cm²) Figure 1. Soluble salt detection techniques (note the units of measurement).
6 How Much Salt Should Be Allowed on Surfaces Prior to Painting? Key Points ⢠When abrasive blasting and re-painting a bridge structure, maximum chloride ion level of 10 µg Clâ/cm2 is suitable for most environments. Where soluble salt is of greater con- cern (e.g., one or more of the high-risk factors identified Table 1, it is reasonable to lower the limit to 5 µg Clâ/cm2. Comparable values of total salt calculated from conductivity are 15 µg salt/cm2 and 7.5 µg salt/cm2, respectively. ⢠When power tool cleaning corroded areas during maintenance painting, soluble salt testing may not be cost-effective. In cases where corrosion is light, or a high degree of cleanliness is spec- ified (e.g., SSPC SP-11, Power Tool Cleaning to Bare Metal), or supplemental washing/cleaning methods are specified, a maximum chloride ion density of 10 µg Clâ/cm2 is achievable. Discussion The maximum level of salt that should be allowed on a surface is a function of the risk associated with coating failure, the likelihood of salt remediation, and the desired performance. See Table 1 for specific risk factors that should be considered. In situations with one or more high-risk factors, increased testing and/or stricter acceptance criteria should be specified. In low-risk situations, soluble salt reme- diation may not be a significant concern at all. However, it is recommended that some minimal amount of soluble salt testing be permitted or required in low-risk environments. As an example, a project that involves complete removal and replacement of a degraded coating with heavy rust scale in a coastal environment would be considered a high-risk project that should include a low soluble salt requirement (e.g., 5 µg Clâ/cm2) and relatively frequent testing (see âWhere and How Often Should an Inspector Measure Soluble Salt Concentrations?â). Alternatively, a project that involves maintenance of a coating in good condition with minor light rusting on a bridge in an arid environment without road salt would be considered a low-risk project that should include a higher salt requirement (e.g., 10 µg Clâ/cm2) and limit testing to that which the inspector deems necessary. Note that the risk of salt in these two examples is driven more by the degree of rust scale where coating has failed than the percentage of the coating surface area that is failing. Soluble salt ranges reported in various references are pre- sented in Table 2. In some cases, these are specification limits while in others they are guidance values. Regardless, they illustrate the magnitude of concern for industrial painting scenarios. Note that total salt calculated from conductivity is not commonly used, the total salt calculated from con- ductivity values in Table 2 have been calculated for this guide- book. SSPC SP-COM contains the following three commonly specified levels of soluble salts: ⢠Free of detectable levels of soluble contaminants; ⢠Less than 7 µg/cm2 (0.0007 grains/in2) of chloride contami- nants, less than 10 µg/cm2 (0.001 grains/in2) of soluble ferrous iron levels, or less than 17 µg/cm2 (0.0017 grains/in2) of sulfate contaminants; and ⢠Less than 50 µg/cm2 (0.005 grains/in2) of chloride or sulfate contaminants. As the number of risk factors increases, the specifier should consider setting lower soluble salt limits. Using the risk factors in Table 1 and the data in Table 2 as a point of reference, the matrix in Table 3 provides recommended soluble salt levels. Note that when power tool cleaning corroded areas during maintenance painting, soluble salt testing may not be cost-effective. How Should Soluble Salts Be Detected? Key Points ⢠There are several suitable field methods for detecting soluble salts on flat surfaces using ion-specific or conductometric methods. Source Low soluble salt limit High soluble salt limit Chloride ion Conductivity Chloride ion Conductivity FHWA-RD-91-011 recommendations 7 µg Clâ/cm² -- 50 µg Clâ/cm² -- MnDOT survey of DOT practices <5 µg Clâ/cm² undetectable 11-15 µg Clâ/cm² 26-50 µS/cm (3.1-6 µg salt/cm²) U.S. Navy Standard Item 009-32 requirements for âcriticalâ and ânon-criticalâ surfaces on Navy ships 3 µg Clâ/cm² 30 µS/cm (3.6 µg salt/cm²) 5 µg Clâ/cm² 70 µS/cm (8.4 µg salt/cm²) National Association of Corrosion Engineers (NACE)SP0213-2013 categories 1 µg NaCl/cm² 9 µS/cm* 40 µg NaCl/cm² 362 µS/cm* *The conductivity is calculated from NaCl concentration assuming a 12.5 cm² surface area and 3 mL of extraction fluid. Table 2. Soluble salt limits from various references.
7 ⢠The methods are sensitive to low levels of soluble salt conta- mination. Since typical industry requirements are near the detection limits for the technique, significant measurement variability may be observed in the field. ⢠None of the field methods can effectively detect salts in crevices and most are limited in their ability to extract salts from heavily pitted surfaces or other rough geometries. Discussion Figure 2 presents the hierarchy of detection methods as defined in SSPC Coatings Technology Guide 15, Field Methods for Extraction and Analysis of Soluble Salts on Steel and Other Nonporous Substrates. The guide is included as Appendix A. Appendix A and Appendix C contain details on use of the most common detection methods. Each method has advantages and limitations. The second volume of this report, titled Detection and Remediation of Soluble Salt Contamina- tion Prior to Coating Steel Highway Structures (available for download from the TRB website, trb.org by searching for âNCHRP Research Report 912) contains detailed data that support the following key issues of concern when using soluble salt detection techniques: ⢠All methods involve extraction of salts from surfaces by dissolving them in an aqueous solution at ambient tempera- tures (e.g., 40°F to 120°F). Field results are not comparable with laboratory evaluations where salts are extracted using boiling water. Application Criteria No high-risk factors One or more high- risk factors New coating system Chloride ion 10 µg Cl-/cm² 5 µg Cl-/cm² Salt from conductivity 15 µg salt/cm² 7.5 µg salt/cm² Spot/maintenance painting (if desired) Chloride ion 15 µg Cl-/cm² 10 µg Cl-/cm² Salt from conductivity 22.5 µg salt/cm² 15 µg salt/cm² Table 3. Recommended soluble salt levels. Field Methods for Retrieval and Analysis of Soluble Salts from Steel and Other Nonporous Surfaces Methods Measuring Conductivity Fully Automated Methods Soluble Salt Meter Surface Salinity Meter Continuous Flow Extraction Fiber Strip Multi-Step Methods ⢠Surface Swab or Wash ⢠Adhesively Bonded Latex Patch or Cell ⢠Saturated Special Filter Paper and Conductivity Meter Methods Measuring Specific Ions Fully Automated Methods None Available Multi-Step Methods Ten alternative approaches including combinations of swabbing, patches, sleeves, or blotting paper in conjunction with various analytical techniques Figure 2. Hierarchy of soluble salt detection methods from SSPC Guide 15 (Appendix A).
8 ⢠All methods are sensitive to very low levels of soluble salt contamination and should be performed with care. As a result, a significant relative standard deviation (on the order of 50%) of otherwise equivalent measurements may be observed. Multiple measurements should be performed to characterize a surface, and a significant scatter in the results should be expected. ⢠Laboratory testing shows that as salt contamination âagesâ the quantity of extracted salt decreases between 30% and 80% versus detection immediately after doping. As a practical matter, many surfaces in the field will signifi- cantly âageâ between the time salt is deposited on a surface and when the detection test is performed. This contributes to observed variability in field measurements. ⢠Research has shown that field techniques do not extract as much salt from a rusted surface as from a clean surface. ⢠None of the field methods can effectively detect salts in crevices. ⢠For methods measuring conductivity, ensure that the extraction fluid fully contacts the measuring electrodes. This may require that an instrument be oriented in a spe- cific direction, that a sufficient sample of extraction fluid is inserted in the device, and/or that a saturated filter paper is in full contact with instrument electrodes. ⢠Loss of extraction fluid due to leakage, evaporation, or spillage should be minimized. The impact of the extrac- tion fluid loss may vary depending on when it occurs (i.e., before or after salt dissolution) and whether all the fluid or only a sample is required for analysis. ⢠Electronic instruments commonly contain multiple measurement modes that electronically convert the data to account for various situations. For the most part, these modes perform a mathematical adjustment to the measured conductivity value. Some of the adjustments are common, while others are proprietary. For purposes of standardization, ANSI/NACE SP0508-2017 was developed to attempt to create consistency among the instruments. Most instruments call this a âBresle equivalentâ reading. The recommended best practice is to use a Bresle equivalent total salt density calculated from conductivity. Regardless of the method used, it is best practice to record instrument settings along with measured values. This helps determine what readings are comparable and may allow readings to be converted to values that would be obtained using other settings, or with other devices. ⢠Most electronic meters are able to perform temperature compensation; however, ISO 8502-9 is ambiguous on best practices (a meter with temperature compensation is required, but the standard does not suggest a reference temperature). Because of the wide range of possible test temperatures, it is good practice to record the measure- ment temperature and whether the value has been com- pensated to a standard temperature.2 For purposes of standardization, temperature correction to 25C (77°F) is the recommended best practice. ⢠The manufacturer recommends that ion detection tubes are measured immediately after the tubes are fully saturated. Readings have been observed to change within hours of the test being performed. ⢠Solution uptake into ion detection tubes can be slow or sometimes even appear to stall. Some operators mechani- cally enhance the capillary action by drawing a vacuum on the end of the tube open to the air. This draws solution into the tube faster and decreases measurement time. The practice is not explicitly allowed or forbidden by the manufacturer. While there is no compelling evidence that it impacts the test results, the capillary tube should be used as intended (i.e., allowing the solution to be drawn into the tube through capillary action) before additional vacuum force is applied to the tube. ⢠The electronic meter screens can be difficult to see and read in direct sunlight. Batching when available on electronic meters should be used to permanently record readings for later reference. Where and How Often Should an Inspector Measure Soluble Salt Concentrations? Key Points ⢠NACE SP0716-2016 is a good reference standard for soluble salt testing frequency and location. ⢠The inspectors should concentrate their measurements in areas where soluble salts are most likely to be found (e.g., corrosion, metal loss, coating failure, near joints, at drainage collection points). ⢠Rusting interferes with soluble salt measurements. Since high-performance coatings will require that this rust be removed, detection of salts on visibly rusted surfaces is often of no practical use. ⢠Typically, soluble salt contamination is only a concern on the prepared substrate. However, if there is a sufficient risk of soluble salt contamination (e.g., marine environment), soluble salt measurements may be made on primed steel and/or on intermediate coats of paint. Discussion When inspecting a prepared surface, an inspector should carefully observe the structure and perform measurements in areas that are most likely to have soluble salt contamination. When possible, the inspector should have a knowledge of the general history of the surface (e.g., the condition of the
9 steel prior to abrasive blasting or the environmental conditions since surface preparation). The inspector should also be aware of the following characteristics of soluble salts: ⢠Soluble salts tend to be non-uniformly distributed on a steel surface. ⢠Soluble salts are not usually visible except to the extent that they cause steel to rust or are present in sufficient concentrations to precipitate on the surface. ⢠During maintenance painting, soluble salts are less likely to be found on surfaces that previously had intact coating than on surfaces that were rusting or had a visibly deterio- rated coating ⢠The ability to dissolve salts in water (i.e., extraction effi- ciency) is reduced as a contaminated steel surface ages and on heavily pitted surfaces that cannot be fully wetted. ⢠In sufficiently high humidity, soluble salts will cause steel to discolor or rust back; in sufficiently low humidity, salts will not visibly affect the steel surface. ⢠Soluble salt sources tend to force them to collect in certain areas such as those that do not drain well, surfaces facing prevailing winds, or surfaces over which roadway runoff flows. ⢠Soluble salts are more likely to exist in crevices and pits. None of the field techniques can measure salts in crevices. The techniques can be challenging to use in heavily pitted areas that cannot be fully sealed and wetted with the extraction fluid. Inspectors should perform measurements as close as possible to crevices and on pitted areas where an effective seal can be established. SSPC Technology Guide No. 24, Soluble Salt Testing Frequency and Locations on New Steel Surfaces, provides useful guidance for sampling on new steel surfaces that may be applicable for shop coating of bridge steel. Note that the recommendations are not written as requirements that may be cited in a contract. NACE SP0716-2016, Soluble Salt Testing Frequency and Locations on Previously Coated Surfaces, designates where, how often, and the most likely places to test for the presence of surface soluble salts before application of a coating system. For a typical bridge recoating project, the standard would recommend five tests be performed in the first 1,000 square feet of each area, two tests in the second 1,000 square feet of each area, one test in each additional 1,000 square feet of each area. It further requires testing to be performed in locations where there is corrosion, metal loss, or coating failure. In the absence of these locations, one third of the tests are to be performed within 2 inches of a seam or joint. It is appropriate to cite this standard as a contractual requirement for bridge recoating projects. Table 4 contains recommended language for specifying the frequency and locations of soluble salt testing for locations of different risk. Note that when power tool cleaning corroded areas during maintenance painting, soluble salt testing may not be cost-effective. The inspector should develop a work plan that clearly states what level of contamination is allowed, how such contami- nation shall be measured, the specific equipment to be used to detect soluble salts, and the minimum required frequency of measurement. If these are contained in the contract doc- uments, they should be restated in the work plan. If these issues are ambiguous or not fully addressed, they should be clarified in the project kickoff meeting and in the inspection work plan. If more than one detection device is to be used, calculations or conversion factors to equate readings should also be addressed. In addition to testing the surface, inspectors should check conductivity of water and/or abrasive materials prior to and/ or during surface preparation. What Are the Best Practices to Remediate Soluble Salts? Key Points ⢠Washing of surfaces (with or without additives) is industry best practice that helps reduce soluble salt contamination prior to surface preparation. Application No high-risk factors One or more high risk factors Shop coating Develop a sampling procedure based on the guidance in SSPC Technology Guide 24. New coating system on an existing bridge Perform soluble salt testing on the prepared steel substrate prior to coating. Test three locations in the first 1,000 square feet and one location for each subsequent 1,000 square feet of each production area. Perform testing at the frequency and locations required in NACE SP0716-2016. Spot/maintenance painting (if desired) Perform testing at representative spots before painting each day. Perform testing at the frequency and locations required in NACE SP0716-2016. Table 4. Minimum recommended testing frequency and locations.
10 ⢠Dry abrasive blasting, wet abrasive blasting, and ultrahigh pressure water jetting to a high degree of cleanliness (e.g., SSPC-SP 10/NACE No. 2, Near-White Metal Blast Clean- ing; SSPC-SP 10 (WAB)/NACE WAB-2, Near White Metal Wet Abrasive Blast Cleaning, and Waterjet Cleaning of Metals; and SSPC-SP WJ-2/NACE WJ-2âVery Thorough Cleaning) are industry best practices for removing soluble salts during surface preparation. ⢠Cleaning to bare metal using power tools (SSPC SP-11, Power Tool Cleaning to Bare Metal) is the best practice to remediate soluble salt contamination using power tools. ⢠In instances where mechanical methods do not sufficiently remediate soluble salt contamination, water washing is effective at remediating the remaining soluble salt. However, supplemental mechanical preparation may be required to remove rusting which may occur because of the washing. Alternatively, repeated mechanical preparation may also reduce soluble salt contamination below the required levels. Discussion Remediating soluble salts prior to painting is generally accomplished using traditional surface preparation tech- niques. Table 5 presents advantages and disadvantages asso- ciated with using various surface preparation methods for soluble salt remediation. Remediation Method Examples Advantages Disadvantages Hand Tool Cleaning: Light Mechanical Remediation SSPC-SP 2: Hand Tool Clean SSPC-SP1: Solvent Cleaning Good practice as an initial method to remove loose salts, rust scale, and debris Not able to remediate soluble salts in tightly adhered rust Power Tool Cleaning: Medium Mechanical Remediation SSPC-SP3: Needle gun SSPC-SP11: Bristle impact Good methods for spot preparation, rework of small areas, or difficult to access areas Though surfaces may visibly appear âclean,â soluble salts may be present in pits or residual corrosion product Media Blasting: Heavy Mechanical Remediation SSPC-SP10: Near White Metal Blast WAB: Wet Abrasive Blast/ Slurry Blast Effectively remediates salt while removing rust and coating Can be difficult to remediate salts from tight crevices and pits too small for abrasive to enter Pressure Washing: Light Cleaning LPWC: 3,000-5,000psi LPWC with chloride reducing additives Removes loose salts, rust scale, and debris Can be used without damaging coating Readily available equipment Does not remediate tightly adherent coating or rust Rust remaining on the surface may still contain salts Will not produce a new surface profile Water Methods: Heavy Cleaning HPWC: 5,000-10,000psi HPWJ: 10,000-25,000psi More thorough surface and salt remediation than LPWC Higher pressures do a better job of getting into pits and larger crevices Higher pressures will remove most rust and many coatings Cannot fully remove tightly adherent rust and coatings Special equipment needed to reach these pressures Will not produce a surface profile Ultrahigh Pressure Waterjetting UHPWJ: over 25,000psi Can thoroughly remove coatings and rust Removes salts from pits and many crevices Surfaces tend to rust back due to high humidity produced during remediation Does not form a new profile, only exposes profile present on surface Special equipment needed to reach these pressures Combination Methods Power Tool Clean + LPWC Media Blast Clean + LPWC Able to gain a higher soluble salt remediation than physical or water remediation alone Combining methods can reduce dust from physical remediations Higher costs by completing two remediation steps Note: WAB = wet abrasive blast; LPWC = low-pressure water cleaning; HPWC = high-pressure water cleaning; HPWJ = high-pressure water jetting; UHPWJ = ultra-high-pressure water jetting. Table 5. Soluble salt remediation using surface preparation methods.
11 The contractor should develop surface preparation pro- cedures and processes that will reduce soluble salt levels in addition to removing the paint, rust, and mill scale. The plan should employ processes that recognize soluble salts are more likely to be present on corroding steel with pitting and/or pack rust than on surfaces from which an intact coating has been removed. The quality control plan should state what level of contamination is allowed, how such contamination shall be measured, and the minimum required frequency of measurement. It should also describe quality control tech- niques for monitoring conductivity of water and/or abrasive materials used during surface preparation. âPre-cleaningâ (generally by pressure washing) is an important part of the process whether it is driven by pre- job salt measurements or just as good practice. Pre-cleaning removes soluble salts, dirt, and debris before abrasive blast- ing, which can improve the overall effectiveness of abrasive blasting (especially with recycled abrasives). To varying degrees, all common coating surface prepa- ration methods remove soluble salts from surfaces. Soluble salts that are present on smooth surfaces in crystalline form are most easily removed by pressure washing. Soluble salts also migrate into rust scale where they are difficult to remove using pressure washing. In these cases, ultra-high-pressure water jetting (UHPWJ) or mechanical techniques (e.g., abrasive blasting or power tool cleaning) are required to remove the salt-contaminated rust. Dry mechanical cleaning techniques may be supplemented with pressure washing to effectively remove soluble salts in severely contaminated steel. Multiple blast operations are effective at reducing surface salt concentrations, though less effective than combining abrasive blasting with pressure washing. The data clearly show the benefit of performing a pressure wash after abra- sive blasting. Unfortunately, it is somewhat impractical to introduce and manage water after abrasive blasting has been performed. A pressure wash before blasting is less effective than after blasting, but more effective than not washing at all. As a practical matter, contractors typically pressure wash prior to abrasive blasting and blast areas that rust back multiple times. The rust back should be identified during quality assurance/quality control (QA/QC) inspections and are likely locations for excessive salt contamination. How Much Effort (Cost) Is Required to Remediate Soluble Salt Contamination? Key Points ⢠Experienced contractors can remediate soluble salts to low levels (e.g., less than 5 µg Clâ/cm2) with available technolo- gies when preparing a surface to SSPC SP-10/NACE No. 2 Near-White Metal Blast Cleaning. Extra work may be required to remediate high-risk areas such as expansion joints and heavily pitted steel; the cost premium of remediating salt contamination in these areas appears acceptable to most of the bridge industry. ⢠When power tool cleaning corroded areas during main- tenance painting, SSPC SP-11, Power Tool Cleaning to Bare Metal and/or supplemental washing/cleaning methods are specified. Pressure washing/cleaning between power tool cleaning steps will effectively achieve low soluble salt levels. Discussion SSPC SP-10/NACE No. 2 Near-White Metal Blast Clean- ing can remediate soluble salts to low levels (e.g., less than 5 µg Clâ/cm2) on most steel surfaces. Extra work may be required to remediate steel with substantial pitting, crevices and other geometries that abrasive blasting does not readily enter (e.g., less than 2 grit diameters wide). These conditions are often found near expansion joints, connection points, drainage areas, and complex structural detailing commonly found on older structures. If these geometries are present on a large surface area, the necessary rework, washing, and multiple abrasive blasting evolutions to reduce soluble salt to low levels may impact cost and schedule proportional to the affected surface area. While there may be a slight cost premium to getting the lowest levels of salt contamination in these areas, those who specify soluble salt levels in the bridge industry seem to find that the limits described in Table 4 provide an acceptable cost-benefit. It is more difficult to remove salts from rusted surfaces when using power tool cleaning methods to prepare a surface for painting. Additionally, it can be challenging to measure soluble salts on power tool cleaned surfaces as they are typi- cally rougher and may contain corrosion scale. However, if very high degrees of cleanliness (e.g., SSPC SP-11) and/or supplemental cleaning methods (e.g., hot water cleaning) are specified, low soluble salt levels may be achieved. As a practical matter, achieving low soluble salt levels when power tool cleaning localized corrosion does not seem to provide an acceptable cost-benefit. What Are Some Examples of Specification Language for Allowable Soluble Salt Concentration? Key Point ⢠Specification language should specify the acceptable soluble salt levels as well as detection methods and sampling frequency for verifying compliance.
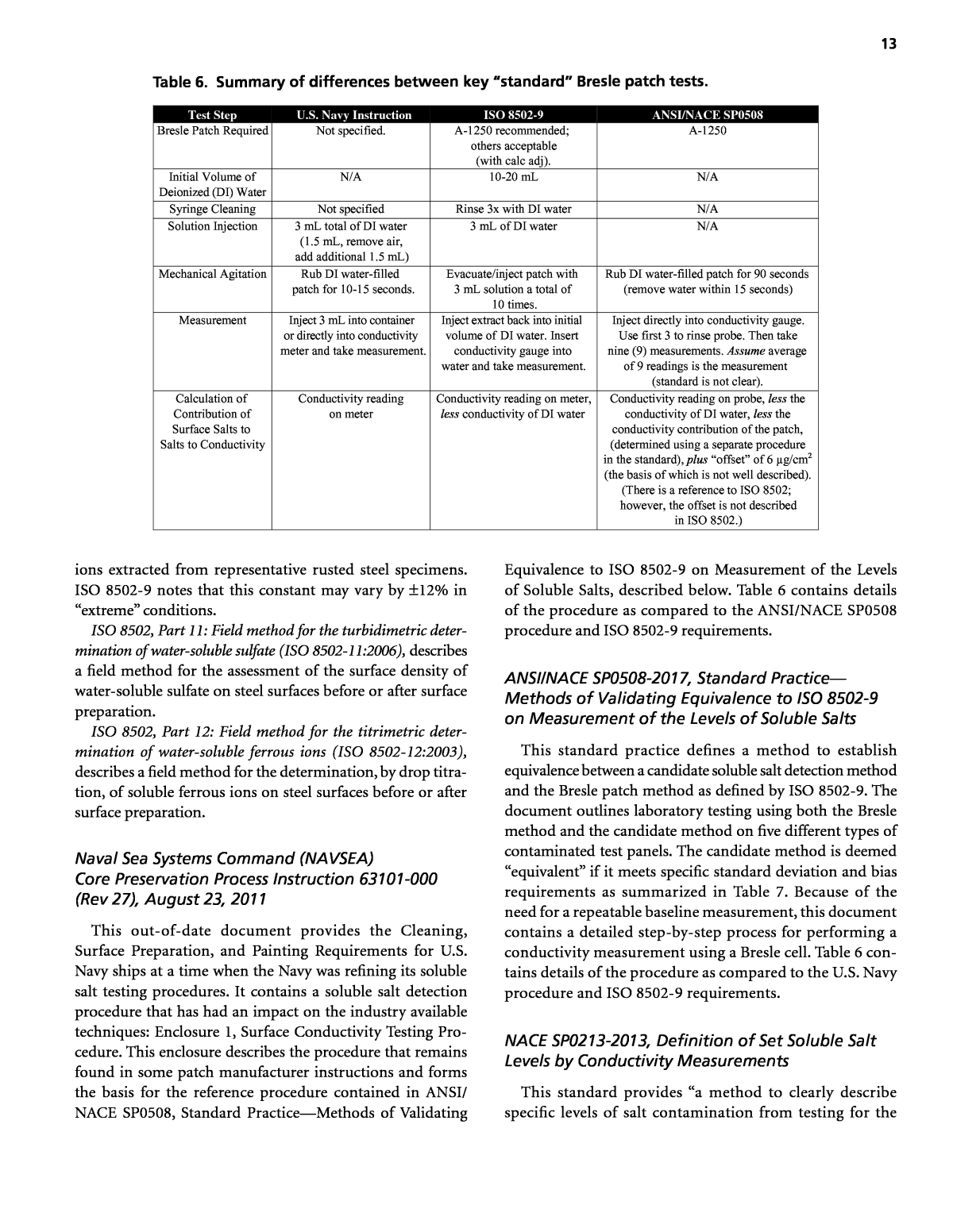
12 Discussion To ensure effective management of soluble salts prior to painting, the specifier should address them in the contract requirements. Specification statements should clearly state what level of contamination is allowable (see Table 3), how such contamination shall be measured (see Figure 1 and Figure 2), and the minimum required frequency of measure- ment (see Table 4). Following are two examples of specification statements: Immediately prior to the application of the coating, the surface extract shall not contain more than xx µg/cm2 of [insert specific contaminant, e.g., chloride, total soluble salt] when tested with [insert specified method(s), e.g., âa soluble salts test kit in accordance with SSPC-Guide 15â or âin accordance with ISO 9502-9â]. Perform tests at [insert frequency of testing, e.g., âthree locations in the first 1000 square feet and one location for each subsequent 1000 square feetâ]. Where possible, perform measurements in locations where soluble salt contamination is most likely to occur. Accomplish surface conductivity or chloride checks using available field or laboratory test equipment on the freshly pre- pared surface. Perform tests at [insert frequency of testing, e.g., âthe frequency and locations required in NACE SP0716-2016â]. Chloride measurements shall not exceed xx µg Clâ/cm2 (xx mg Clâ/m2); total salt determined from conductivity measure- ments shall not exceed xx µg salt/cm2 (xx mg salt/m2). Con- ductivity samples shall be collected using a product that meets the requirements of NACE SP0508-2010, Methods of Validating Equivalence to ISO 8502-9 on Measurement of the Levels of Soluble Salts, and converted to total salt as described in SSPC Guide 15. What Industry Standards Exist Relating to Soluble Salt Detection and Remediation? Key Point ⢠There are several available industry standards and guidelines that address various soluble salt detection and remediation issues. Following is a synopsis of several standards and guide- lines relevant to soluble salt detection. The Society for Protective Coatings (SSPC) Technology Guide 15, Field Methods for Extraction and Analysis of Soluble Salts on Steel and Other Nonporous Substrates The guide describes the most commonly used laboratory and field methods for the extraction and analysis of soluble salts on steel and other nonporous substrates. The guide con- tains a table summarizing 13 different field techniques and a discussion of each technique. It also includes a discussion of a laboratory reference technique and appendices with information on common conversions, relationships between conductivity and salt content, and references that discuss extraction efficiency. A complete copy of the guide is included as Appendix A. ISO Standards: ISO 8502, Preparation of Steel Substrates Before Application of Paints and Related ProductsâTests for the Assessment of Surface Cleanliness This standard was developed and is maintained by ISO Technical Committee ISO/TC 35, Paints and varnishes, Subcommittee SC 12, Preparation of steel substrates before application of paints and related products. ISO 8502 consists of 12 parts, six of which are relevant to this guideline. These six parts are summarized below. ISO 8502, Part 2: Laboratory determination of chloride on cleaned surfaces (ISO 8502-2:2005), describes a method for determining surface chloride contamination that incorpo- rates a swabbing technique for extracting the salts from a surface and a mercuric chloride titration for quantifying the chloride ion content of the wash water. ISO 8502, Part 5: Measurement of chloride on steel surfaces prepared for painting (ion detection tube method) (ISO 8502-5: 1998), describes a field test for the measurement of chloride ions using special detection tubes. The method is intended to be used in conjunction with a suitable surface sampling technique. ISO 8502, Part 6: Extraction of soluble contaminants for analysisâthe Bresle method (ISO 8502-6:2006), describes a method of extracting soluble contaminants from a surface using flexible cells in the form of adhesive patches. This part of ISO 8502 does not cover the subsequent analysis of the contaminants; other parts of ISO 8502 describe analysis methods. ISO 8502, Part 9: Field method for conductometric deter- mination of water-soluble salts (ISO 8502-9:1998), describes a field method for measuring the total surface density of water-soluble salts on steel surfaces. The method involves performing an extraction in accordance with ISO 8502-6, determining the solution conductivity, and converting that value to a theoretical surface density of soluble salts. This method does not determine individual surface densities of ionic species (chlorides, sulfates, etc.). Table 6 contains details of the procedure as compared to the U.S. Navy procedure and to the ANSI/NACE SP0508 procedure. Because the composition of the surface âsaltâ is rarely known, ISO 8502-9 provides a conversion formula that incorporates a constant to represent the specific conduc- tance of salt concentration under ânormal conditions.â This constant is reportedly based on calculation of the total con- ductivity from the specific conductivities of the individual
13 ions extracted from representative rusted steel specimens. ISO 8502-9 notes that this constant may vary by ±12% in âextremeâ conditions. ISO 8502, Part 11: Field method for the turbidimetric deter- mination of water-soluble sulfate (ISO 8502-11:2006), describes a field method for the assessment of the surface density of water-soluble sulfate on steel surfaces before or after surface preparation. ISO 8502, Part 12: Field method for the titrimetric deter- mination of water-soluble ferrous ions (ISO 8502-12:2003), describes a field method for the determination, by drop titra- tion, of soluble ferrous ions on steel surfaces before or after surface preparation. Naval Sea Systems Command (NAVSEA) Core Preservation Process Instruction 63101-000 (Rev 27), August 23, 2011 This out-of-date document provides the Cleaning, Surface Preparation, and Painting Requirements for U.S. Navy ships at a time when the Navy was refining its soluble salt testing procedures. It contains a soluble salt detection procedure that has had an impact on the industry available techniques: Enclosure 1, Surface Conductivity Testing Pro- cedure. This enclosure describes the procedure that remains found in some patch manufacturer instructions and forms the basis for the reference procedure contained in ANSI/ NACE SP0508, Standard PracticeâMethods of Validating Equivalence to ISO 8502-9 on Measurement of the Levels of Soluble Salts, described below. Table 6 contains details of the procedure as compared to the ANSI/NACE SP0508 procedure and ISO 8502-9 requirements. ANSI/NACE SP0508-2017, Standard Practiceâ Methods of Validating Equivalence to ISO 8502-9 on Measurement of the Levels of Soluble Salts This standard practice defines a method to establish equivalence between a candidate soluble salt detection method and the Bresle patch method as defined by ISO 8502-9. The document outlines laboratory testing using both the Bresle method and the candidate method on five different types of contaminated test panels. The candidate method is deemed âequivalentâ if it meets specific standard deviation and bias requirements as summarized in Table 7. Because of the need for a repeatable baseline measurement, this document contains a detailed step-by-step process for performing a conductivity measurement using a Bresle cell. Table 6 con- tains details of the procedure as compared to the U.S. Navy procedure and ISO 8502-9 requirements. NACE SP0213-2013, Definition of Set Soluble Salt Levels by Conductivity Measurements This standard provides âa method to clearly describe specific levels of salt contamination from testing for the Test Step U.S. Navy Instruction ISO 8502-9 ANSI/NACE SP0508 Bresle Patch Required Not specified. A-1250 recommended; others acceptable (with calc adj). A-1250 Initial Volume of Deionized (DI) Water N/A 10-20 mL N/A Syringe Cleaning Not specified Rinse 3x with DI water N/A Solution Injection 3 mL total of DI water (1.5 mL, remove air, add additional 1.5 mL) 3 mL of DI water N/A Mechanical Agitation Rub DI water-filled patch for 10-15 seconds. Evacuate/inject patch with 3 mL solution a total of 10 times. Rub DI water-filled patch for 90 seconds (remove water within 15 seconds) Measurement Inject 3 mL into container or directly into conductivity meter and take measurement. Inject extract back into initial volume of DI water. Insert conductivity gauge into water and take measurement. Inject directly into conductivity gauge. Use first 3 to rinse probe. Then take nine (9) measurements. Assume average of 9 readings is the measurement (standard is not clear). Calculation of Contribution of Surface Salts to Salts to Conductivity Conductivity reading on meter Conductivity reading on meter, less conductivity of DI water Conductivity reading on probe, less the conductivity of DI water, less the conductivity contribution of the patch, (determined using a separate procedure in the standard), plus âoffsetâ of 6 µg/cm2 (There is a reference to ISO 8502; however, the offset is not described in ISO 8502.) (the basis of which is not well described). Table 6. Summary of differences between key âstandardâ Bresle patch tests.
14 purposes of specifying surface cleanliness prior to coating.â The standard does not suggest what level of salt is acceptable in any given application. The standard provides an equation to convert a conductivity measurement to the equivalent NaCl density. Note that this equation is slightly different than the ISO 8502-9 equation for converting conductivity to âtotal salt.â Establishing NaCl as the standard salt equivalent requires a constant of 4.6 kg/m2S2 rather than 5.0 kg/m2S2 (8% lower than the ISO 8205-9 conversion). This standard identifies nine levels of âTotal NaCl Equiva- lent Salt Weightâ and names them SL-A (equal or less than 1 µg NaCl/cm2) through SL-J (equal or less than 40 µg NaCl/cm2). Note that based on the molecular weights of the elements, chlorides comprise 60.7% of the weight of sodium chloride. Therefore, 8.24 µg NaCl/cm2 is the same as 5.0 µg Clâ/cm2. As a practical matter, the standard establishes NaCl equiva- lent salt weight as an alternative to the conversion factor in ISO 8502-9. NACE SP0716-2016, Soluble Salt Testing Frequency and Locations on Previously Coated Surfaces The purpose of this standard is to establish industry best practices for soluble salt testing frequencies and locations. This standard applies to previously coated substrates and replacement substrate material. It does not contain any recom- mendations for allowable limits of soluble salts, procedures for measuring soluble salts, or discussion on the repeatability and reproducibility of such measurements. The standard designates where, how often, and the most likely places to test for the presence of surface soluble salts before application of a coating system. For a typical bridge recoating project, the standard would recommend five tests be performed in the first 1,000 square feet of each area, two tests in the second 1,000 square feet of each area, one test in each additional 1,000 square feet of each area. It further requires testing to be performed in locations where there is corrosion, metal loss, or coating failure. In the absence of these locations, one third of the tests are to be performed within 2 inches of a seam or joint. The testing is to be performed before application of the coating. A different sampling scheme is provided for spot painting. SSPC TECHNOLOGY GUIDE NO. 24, Soluble Salt Testing Frequency and Locations on New Steel Surfaces This guide is intended to assist owners and specifiers in determining the number and location of test sites and fre- quency of testing for measuring soluble salt on new steel surfaces prior to coating. It focuses on new construction, new equipment and components, and new fabrications of unpainted and shop-coated steel that will subsequently receive additional coats. The document provides specific guidance for the follow- ing type of components: ⢠Straight pipe lengths and other tubular components; ⢠Structural steel unit components (UC); ⢠Tanks, vessels, and equipment (TVE); and ⢠Assembled structures (AS). Tables are provided identifying the sampling popula- tion of components to be tested (e.g., 4 pieces from a lot of between 6 and 20 pieces) or the sampling per unit of surface area on assembled flat structures (e.g., 4 locations on an area between 1,001 and 4,000 square feet). The tables allow for a reduced inspection frequency if previous test results are below 75% of the acceptance criteria. The standard does not contain any recommendations for allowable limits of soluble salts, procedures for measuring soluble salts, or discussion on the repeatability and reproducibility of such measurements. Endnotes 1. The calculation of equivalent surface concentration of total salt from conductivity is described Section B1 of Appendix A, SSPC Guide 15. The formula is also described in ISO 8502-9. The calculated salt density value is referred to as âtotal saltâ in this report and by many instrument manufacturers. 2. Conductivity increases with increasing temperature. The magnitude of error is on the order of a few percent per degree Celsius for the ranges typically encountered in salt detection applications. This could be significant on industrial projects performed in natural outdoor environments. Criterion Requirement Standard deviation of test results on the panel condition contaminated to 85 mg/m² Less than 5.3 mg/m² Standard deviation of test results on three panel conditions contaminated to 50 mg/m² Less than 4.8 mg/m² Standard deviation of test results on the panel condition contaminated to 30 mg/m² Less than 5.3 mg/m² Table 7. Requirements for equivalence contained in ANSI/NACE SP0508.
