Summary
Opioid analgesics are commonly prescribed to treat chronic and acute pain. Marked increases in prescribing of opioids for acute and chronic pain occurred in the United States from the later 1990s to approximately 2012. Those increases, prompted by efforts to improve pain management, resulted in unanticipated morbidity and mortality in the United States, both in the civilian and the Department of Veterans Affairs/Department of Defense (VA/DoD) treatment environments. The population rate of opioid prescribing for pain has been declining since 2012, yet it remains at much higher levels than it was before 2000. Furthermore, multiple studies have demonstrated that a higher daily dosage of prescribed opioids is associated with a higher risk of death from overdose.
Whereas opioids are primarily prescribed for pain, benzodiazepines are prescription sedatives that are typically prescribed for anxiety or insomnia. Studies show that benzodiazepine prescribing also increased significantly from the later 1990s to approximately 2012 and has remained at high levels, despite evidence of severe adverse effects associated with chronic use. The concomitant use of opioids and benzodiazepines is also associated with increased rates of unintentional overdose and death and with higher rates of suicide.
Responding to the concern about opioid and benzodiazepine use in the veteran population, the VA requested that the National Academies of Sciences, Engineering, and Medicine “develop a protocol/study design to evaluate the relationship between concomitant opioid and benzodiazepine medication practices at the VA, for treating mental health and combat-related trauma, which potentially led to veteran’s deaths and suicides.”
BACKGROUND
Due to the effects of active duty and combat-related injuries, among other potential predisposing factors, the VA population has higher rates of chronic pain, traumatic brain injury, posttraumatic stress disorder, depression, substance use disorder, and accompanying mental health problems than civilians. Those co-occurring conditions increase the risk for suicidal ideation and suicide, and chronic opioid and benzodiazepine treatment is relatively common.1 Even so, current studies do not indicate that VA medical providers were prescribing opioids at higher doses or frequencies than civilian providers, and rates of opioid prescribing by VA providers have decreased in concert with the rates of civilian providers over the past few years. Of note, the VA and DoD published their Clinical Practice Guidelines for Opioid Therapy for Chronic Pain in 2010, updated in 2015, to disseminate recommendations for safer prescribing. The guideline highlights the most important risk factors associated with adverse events in chronic opioid treatment and provides guidance on appropriate dosing and duration of opioid treatment. Moreover, in response to increasing concerns about the risks of chronic opioid treatment, the VA implemented the Opioid Safety Initiative in 2013, which includes best-practice prescribing education for medical providers, overdose education and naloxone2 training for patients, and a clinical-decision-support tool for hospital leaders to track prescribing. That initiative was associated with a decreased prescribing of chronic opioid treatment during the ensuing 5-year period, 2013–2018.
Current studies do not clearly indicate to what extent opioid and benzodiazepine co-prescribing in the VA during that period contributed to severe adverse consequences, including opioid overdose deaths. Nevertheless, congressional leaders remained concerned about possible over-prescribing of opioids and benzodiazepines to veterans during the years 2010–2017. Thus, the Committee on Developing a Protocol to Evaluate the Concomitant Prescribing of Opioids and Benzodiazepine Medications and Veteran Deaths and Suicides was charged by the VA to “develop a protocol/study design to evaluate the relationship between concomitant opioid and benzodiazepine medication practices at the VA, for treating mental health and combat-related trauma, which potentially led to veteran’s deaths and suicides.” In responding to its task, the committee proposes observational studies using VA and Centers for Disease Control and Prevention (CDC)
___________________
1 Benzodiazepine users tend to report a longer history of opioid use and prior detoxifications; use higher doses of opioids; higher frequency of injection drug use, needle sharing, and co-occurring use of alcohol and cocaine; and greater criminal activity.
2 Naloxone is a medication approved by the Food and Drug Administration (FDA) to prevent overdose by opioids such as heroin, morphine, and oxycodone.
data to emulate hypothetical randomized trials, should the VA decide to conduct those studies.
DEFINING THE RESEARCH QUESTION
The committee interpreted its task to focus on the following overarching research question: What were the effects of opioid initiation and tapering (i.e., dosage reduction or discontinuation) strategies in the presence of benzodiazepines in veterans on all-cause mortality and suicide mortality from 2010 to 2017?3 The committee’s focus on that specific research question was a result of its interpretation of the intent of the Statement of Task and the committee’s review of the literature.
First, the committee determined that initiation and tapering represent two critical decision points in opioid treatment. A decision to initiate opioids is of necessity made for any person who eventually progresses to long-term opioid therapy. Tapering is the second decision point of focus identified by the committee. Given the focus of the Statement of Task on adverse consequences, tapering is highly relevant for two main reasons: (1) the avoidance of known adverse consequences of long-term opioid therapy, such as increased mortality, is often a motivation for the decision to taper opioid dosage rather than continuing treatment, and (2) some clinicians and patients have raised concerns that, rather than reducing the risk for harm, tapering a patient who is tolerant to opioids may contribute to adverse consequences, particularly suicide. The committee concluded that a study of the effect of opioid tapering on all-cause mortality and suicide would reduce clinical uncertainty and would be timely in the context of current opioid policy and practice decisions.
Second, the Statement of Task specifically focused on “concomitant opioid and benzodiazepine” prescribing. After reviewing the literature, the committee concluded that patients receiving both medications are at particularly high risk for adverse outcomes relative to patients on opioids alone, and there was very limited evidence regarding opioid prescribing strategies specifically for patients receiving benzodiazepines. Thus, although the effects of opioid initiation and tapering on patient outcomes are important areas of inquiry, the focus on patients prescribed benzodiazepines is more responsive to the Statement of Task and also addresses a particularly important sub-group of patients.
Finally, the Statement of Task specifically focused on the outcomes of “deaths and suicides.” The committee believed that a focus on all-cause
___________________
3 The Consolidated Appropriations Act, 2018, Number: 115-141, Session: 115th Congress (Second Session), from which the committee’s Statement of Task was written, specified an interest in the time period of “fiscal years 2010 to 2017.”
mortality appropriately reflected the fundamental importance to clinical decisions of evidence of increased or decreased risk of death from any cause. Furthermore, mortality from any cause was the outcome most likely to be complete within VA data resources, given the ability of VA patients to get care outside the VA. Additionally, the inclusion of suicide as an outcome separate from all-cause mortality was relevant to the concerns of patients, clinicians, and other stakeholders about suicide among veterans generally as well as among veterans with pain specifically.
The committee notes that the Statement of Task could be interpreted in other ways, such as the tapering of benzodiazepines for patients prescribed both opioids and benzodiazepines. Should the VA or others be interested in conducting related studies, those studies can be modeled on the committee’s approach.
THE TARGET TRIAL FRAMEWORK
Randomized trials are the preferred method for estimating the causal effects of treatment strategies on clinical outcomes. The primary advantage of a randomized trial is that the randomization assures a high likelihood that confounding variables are balanced across treatment groups. However, prospective randomized trials in which patients are enrolled and randomized into treatment groups are often infeasible for a variety of reasons, such as ethics and resource limitations, and the results from such trials may have limited generalizability to routine clinical settings. For that reason, studies using observational data are often appropriate for evaluating the connections between treatments and outcomes.
To develop the study strategy using observational data, the committee suggests the use of a “target trial” methodology, which involves describing a hypothetical randomized trial that is emulated (i.e., closely approximated) by an observational study. Specifying a target trial before emulating that trial using observational data can help mitigate some of the limitations of observational studies, for instance, by identifying and describing how to measure confounders. An observational study is most useful when any important confounding variables can be identified, measured, and therefore controlled for by using study design (matching or exclusion) or statistical analysis (stratification or mathematical modeling). In Chapter 2 the committee describes the rationale, advantages, and limitations of the target trial methodology and presents the target trials that the committee believes would best respond to the charge. Chapter 3 details considerations of how those target trials would be emulated using existing data from the VA and CDC. It should be noted that while the committee is familiar with those databases, it did not consider specifics such as which VA databases could be most useful.
For clarity, in this report the committee uses the term “target trial” to refer to the hypothetical randomized trial that would directly address the research question, “protocol” to include all components of the design (e.g., patient enrollment, treatment strategies, outcome) and analysis of the target trial, and “observational analysis” to refer to the data analysis proposed to emulate the target trial using existing observational data.
TARGET TRIAL PROTOCOL AND OBSERVATIONAL EMULATION
The committee developed protocols for two different hypothetical target trials, each of which would quantify effects on suicide and other causes of death. The first trial focuses on the initiation of opioids in the presence of benzodiazepines and the second on the tapering of opioids in the presence of benzodiazepines. Both studies were developed for patients receiving chronic benzodiazepine treatment. The committee then designed protocols and analytic strategies for those trials, recognizing that many other studies could also be of interest. The object of the trials is to determine preferred approaches for opioid initiation or reduction strategies for patients participating in the trials.
Figure S-1 illustrates the opioid initiation and tapering target trials proposed by the committee. The committee describes seven components in the protocol of the target trials: eligibility criteria, treatment strategies, treatment assignment, start and end of follow-up, outcomes, causal contrasts, and the statistical analysis plan.
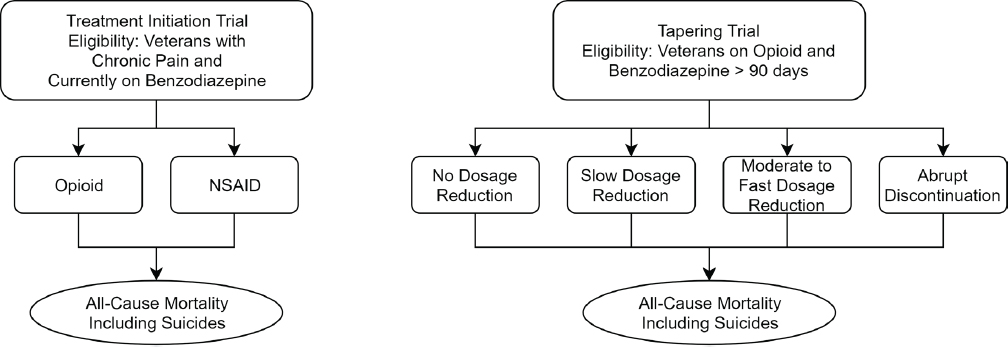
NOTE: NSAID = nonsteroidal anti-inflammatory drug.
In Table S-1, the committee illustrates a suggested set of initial choices for the required specification of the target trials. The choices listed in this table should be considered preliminary because the specification of the target trial components is an iterative process, with insights from pilot analyses of the available observational data resulting in changes to the definitions and choices that can be incorporated into a feasible and valid analysis plan.
TABLE S-1 Proposed Specifications for Initiation and Tapering Target Trials
| Protocol Component | Initiation Target Trial | Tapering Target Trial |
|---|---|---|
| Eligibility criteria | Chronic pain diagnosisa No prescriptions for opioids or non-aspirin NSAIDS in the last 90 days Long-term benzodiazepine therapy (defined based on pilot data) Exclude: Individuals with serious illnessb Individuals prescribed opioids used for treatment of opioid use disorder Individuals with surgery or acute painful injury within the past 90 daysc |
Long-term opioid therapy defined as 3+ opioid fills ≥21 days apart in a ≥84-day period for ≥84-day supply Average opioid MMEd/day is ≥30 over the prior 84 dayse Long-term benzodiazepine therapy (defined based on pilot data) Exclude: Individuals with serious illness Individuals prescribed opioids for the treatment of opioid use disorder Individuals with surgery or acute painful injury within the 90 days prior to baseline |
| Treatment strategies |
|
Participants who cannot tolerate their assigned dosage change will be excused from following their assigned strategy. Percentage of taper is relative to opioid dose at baseline and is calculated over the next 3 months. After that period, dosage is left to the physician’s discretion. |
| Protocol Component | Initiation Target Trial | Tapering Target Trial |
|---|---|---|
| Treatment assignment | Individual randomization, stratified on baseline dose | |
| Start and end of follow-up | Start of follow-up (baseline): start of treatment for chronic pain, defined as being dispensed one or more prescriptions of an opioid or NSAID for at least a 30-day supply over a 30-day period (this could be across multiple prescriptions), and also having a chronic pain diagnosis End of follow-up: the earliest of 18 months,g death, or administrative end of follow-up (end of the study) |
Start of follow-up (baseline): time of assignment to a treatment strategy End of follow-up: the earliest of 6 months,h death, or administrative end of follow-up (end of the study) |
| Outcomes |
|
|
| Causal contrast |
|
|
| Statistical analysis | Intention-to-treat analysis: check for balance on key variables, e.g., mental health diagnoses and substance use disorders. Per-protocol analysis: patients will be censored at the time they deviate from their assigned strategy. To adjust for the potential selection bias induced by censoring, inverse probability weighting will be used. The weights will be a function of the baseline and post-baseline (time-varying) confounders. Both analyses may require further adjustment for selection bias due to loss to follow-up. Pre-specified sub-groups to be examined for potential effect modification include, e.g., pain severity, history of overdose, history of suicide attempt, non-suicide death (for the suicide death analysis). |
|
a The intention of this definition is to exclude opioids and nonsteroidal anti-inflammatory drugs (NSAIDs) prescribed for acute pain. However, researchers should consider that there might be a large proportion of veterans prescribed opioids for whom there is not a chronic pain diagnosis.
b Serious illness is defined as a health condition that carries a high risk of mortality and negatively affects a person’s daily functioning. The committee recommends operationaliz-
ing this as any of the following conditions: cancer, chronic obstructive pulmonary disease, congestive heart failure, dementia, or severe neurologic disorder (e.g., amyotrophic lateral sclerosis, multiple sclerosis).
c 90 days was chosen to minimize likelihood of opioids being prescribed for acute rather than chronic pain conditions. However, the committee acknowledges that the choice of 90 as opposed to 30 or 60 is arbitrary.
d MME = morphine milligram equivalent.
e This threshold was used because labeling for OxyContin extended release defines “opioid tolerant” as consuming 30 MME/day. Researchers might consider a lower dose threshold if the purpose is to include anyone who could be considered for a taper.
f Speed of tapering: there is a lack of primary literature on the optimal rate of tapering speed (i.e., rate of dosage decrease per week/month). Within the context of concomitant opioid and benzodiazepine use and likely psychiatric comorbidity, a more conservative approach would be prudent.
g The committee believes that the 18-month timeline balanced the desire for a longer length of follow-up than prior initiation studies with the fact that there would be a greater degree of non-adherence from the assigned treatment group with the longer lengths of follow-up.
h The committee believes that the 6-month timeline balanced a desire for a longer length of follow-up with potential for non-adherence and a concern that suicide is a relatively short-term outcome in tapering studies.
Figure S-2 illustrates the proposed observational studies that would emulate those target trials using existing VA and CDC data.
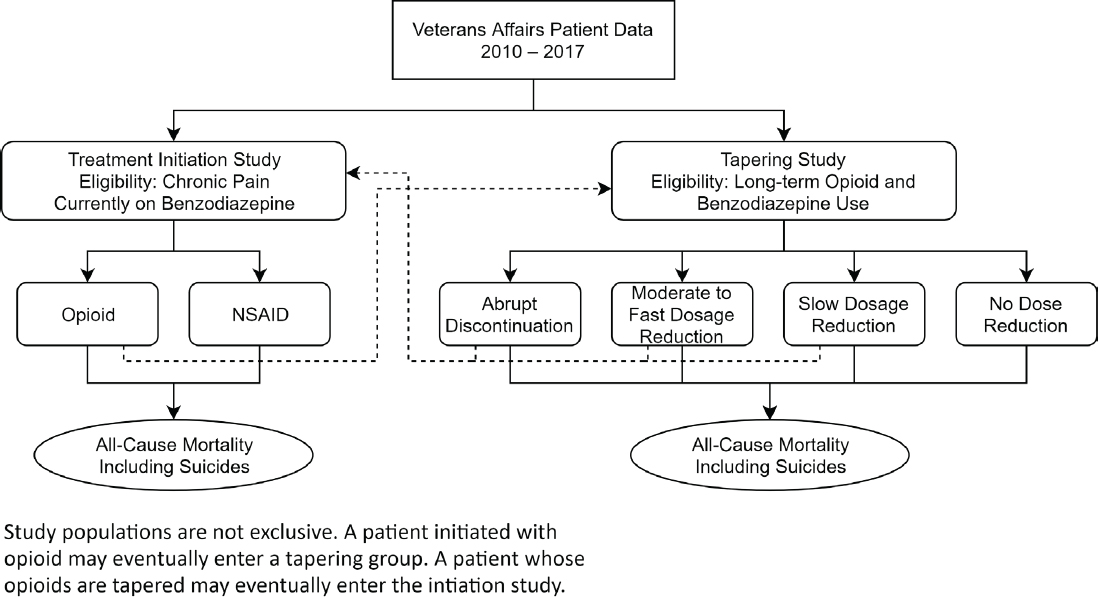
NOTE: NSAID = nonsteroidal anti-inflammatory drug.
Tables S-2 and S-3 reiterate the specifications of the target initiation and tapering target trials from Table S-1 in the “target trial” column and suggest emulation procedures using available observational data. Tables S-2 and S-3 describe an initial emulation strategy that has not been evaluated and would likely require modification after an initial review of the available data. Additional considerations for emulating the target trials using observational data and potential limitations are detailed in Chapter 3.
TABLE S-2 Opioid Initiation Target Trial Emulation
| Study Component | Target Trial | Emulation Using Observational Data |
|---|---|---|
| Eligibility criteria | Chronic pain diagnosis No prescriptions for opioids or non-aspirin NSAIDS in the last 90 days Long-term benzodiazepine therapy (defined based on pilot data) Exclude: Individuals with serious illnessa Individuals prescribed opioids used for treatment of opioid use disorder Individuals with surgery or acute painful injury within the past 90 daysb |
Data to determine the use of analgesics during the last 90 days and the use of benzodiazepines will come from pharmacy fills and will require information specifically on fill dates, dose, and supply duration. Data for the diagnoses of chronic pain, serious illnesses, surgeries, or acute painful injuries will come from the medical visit records. Opioid use for opioid use disorder treatment will be measured through a combination of pharmacy fills (for buprenorphine) and clinic codes for opioid treatment programs. |
| Treatment strategies |
|
Patients will be assigned to the strategies consistent with their pharmacy fill data, based on initiation at baseline with an opioid or NSAID. Adherence to the strategy is defined by continued fills during the year after baseline. An example of non-adherence would be if a patient could not tolerate over-sedation from opioid use and discontinued as a result. |
| Study Component | Target Trial | Emulation Using Observational Data |
|---|---|---|
| Treatment assignment | Individual randomization |
Assumed to be random conditional on baseline confounders, including, but not limited to
Diagnoses associated with clinical visits in VA medical records will be used to define these variables. |
| Start and end of follow-up | Start of follow-up (baseline): time of assignment to a treatment strategy End of follow-up: the earliest of 18 months, death, or the administrative end of follow-up (end of the study) |
Start of follow-up (baseline): time of assignment to a treatment strategy. End of follow-up: the earliest of 18 months, the date of death based on National Death Index records, or the administrative end of follow-up. |
| Outcomes |
|
Deaths ascertained from National Death Index data, with all-cause mortality measured as a death record with a date of death and suicide deaths as those records with underlying cause of death recorded as ICD-10 codes X60–X84, Y87.0, *U03.c |
| Causal contrast |
|
Observational analog of the intention-to-treat effect: this effect may be close to null and therefore relatively uninformative because adherence to the assigned treatment strategies is expected to be low in the observational data. Observational analog of the per-protocol effect. |
| Study Component | Target Trial | Emulation Using Observational Data |
|---|---|---|
| Statistical analysis | Intention-to-treat analysis: check for balance on key variables, e.g., mental health diagnoses and SUDs. Per-protocol analysis: patients will be censored at the time they deviate from their assigned strategy. To adjust for the potential selection bias induced by censoring, inverse probability weighting will be used. The weights will be a function of the baseline and post-baseline (time-varying) confounders. Both analyses may require further adjustment for selection bias due to loss to follow-up. Pre-specified sub-groups to be examined for potential effect modification include, e.g., patients with pain severity, history of overdose, or a history of suicide attempt. |
Intention-to-treat analysis: same as in target trial, except that an individual may have multiple eligibility points, and adjustment for baseline confounders is required. Per-protocol analysis: same, except that a single subject may have multiple eligibility points. All variables will be obtained from medical records, including clinic visit information, diagnoses, and pharmacy records. |
a Serious illness is defined as a health condition that carries a high risk of mortality and negatively affects a person’s daily functioning. The committee recommends operationalizing this as any of the following conditions: cancer, chronic obstructive pulmonary disease, congestive heart failure, dementia, or severe neurologic disorder (e.g., amyotrophic lateral sclerosis, multiple sclerosis).
b 90 days was chosen to minimize the likelihood of opioids being prescribed for acute rather than chronic pain conditions. However, the committee acknowledges that the choice of 90 as opposed to 30 or 60 is arbitrary.
c The researchers who perform the study should determine whether this definition is sufficiently accurate for their purposes.
TABLE S-3 Opioid Tapering Target Trial Emulation
| Study Component | Target Trial | Emulation Using Observational Data |
|---|---|---|
| Eligibility criteria | Long-term opioid therapy defined as 3+ opioid fills ≥21 days apart in a ≥84-day period for ≥84-day supply Average opioid MMEa/day is ≥30 over the prior 84 daysb Long-term benzodiazepine therapy (defined based on pilot data) Exclude: Individuals with serious illnessc Individuals prescribed opioids for the treatment of opioid use disorder Individuals with surgery or acute painful injury within the 90 days prior to baseline |
Data to determine opioid use will come from pharmacy fills and will require information specifically on fill dates, dose, and supply duration. Data for diagnoses qualifying as serious illnesses will come from the medical visit records. Opioid use for opioid use disorder treatment will be measured through a combination of pharmacy fills (for buprenorphine) and clinic codes for opioid treatment programs. |
| Treatment strategies |
Participants who cannot tolerate their assigned dosage change will be excused from following their assigned strategy. Percentage of taper is relative to opioid dose at baseline and is calculated over the next 3 months. After that period, dosage is left to the physician’s discretion. |
The treatment strategy to which a participant is assigned is determined by the average change in opioid dose during the 3-month period after baseline. This will minimize the impact of changes that are due to non-clinical reasons, as those changes should be followed by a correction (e.g., early prescription fill due to patient vacation, followed by a late fill). Tapering treatment strategies are defined the same as in the target trial. An example of non-adherence would be if a patient’s pain worsened and functioning declined and the patient returned to the original dosage after starting a taper. |
| Study Component | Target Trial | Emulation Using Observational Data |
|---|---|---|
| Treatment assignment | Individual randomization |
Assumed to be random conditional on the baseline confounders, including
Diagnoses associated with clinical visits in VA medical records will be used to define these variables. |
| Start and end of follow-up | Start of follow-up (baseline): time of assignment to a treatment strategy End of follow-up: the earliest of 6 months, death, or the administrative end of follow-up (end of the study) |
Start of follow-up (baseline): time of assignment to a treatment strategy. End of follow-up: the earliest of 6 months, the date of death based on National Death Index records, or the administrative end of follow-up. |
| Outcomes |
|
Deaths will be ascertained from National Death Index data, with all-cause mortality measured as a death record with a date of death and suicide deaths as those records with underlying cause of death recorded as ICD-10 codes X60–X84, Y87.0, *U03.e |
| Causal contrast |
|
Observational analog of the intention-to-treat effect: this effect may be close to null and therefore relatively uninformative because adherence to the assigned treatment strategies is expected to be low in the observational data. Observational analog of the per-protocol effect. |
| Study Component | Target Trial | Emulation Using Observational Data |
|---|---|---|
| Statistical analysis | Intention-to-treat analysis: check for balance on key variables, e.g., mental health diagnoses and SUDs. Per-protocol analysis: patients will be censored at the time they deviate from their assigned strategy. To adjust for the potential selection bias induced by censoring, inverse probability weighting will be used. The weights will be a function of the baseline and post-baseline (time-varying) confounders. Both analyses may require further adjustment for selection bias due to loss to follow-up. Pre-specified sub-groups to be examined for potential effect modification include, e.g., patients with pain severity, history of overdose, or a history of suicide attempt. |
Intention-to-treat analysis: N/A Per-protocol analysis: same as in target trial, except that a single subject may contribute two clones. All variables will be obtained from medical records, including clinic visit information, diagnoses, and pharmacy records. |
a MME = morphine milligram equivalent.
b This threshold was used because labeling for OxyContin extended release defines opioid tolerant as consuming 30 MME/day. Researchers might consider a lower dose threshold if the purpose is to include anyone who could be considered for a taper.
c Serious illness is defined as a health condition that carries a high risk of mortality and negatively affects a person’s daily functioning. The committee recommends operationalizing this as any of the following conditions: cancer, chronic obstructive pulmonary disease, congestive heart failure, dementia, or severe neurologic disorder (e.g., amyotrophic lateral sclerosis, multiple sclerosis).
d Speed of tapering: there is no generally accepted rate of tapering speed (i.e., rate of dosage decrease per week/month)—options would need to be explored using pilot data. The tapering speeds proposed for this trial should not be considered medical guidance.
e The researchers who perform the study should determine whether this definition is sufficiently accurate for their purposes.
The committee identified several challenges in their protocol development. The first was that prescribing practices and standards of care evolved in important ways from 2010 to 2017 and continue to evolve in response to increasing concerns about high-risk prescribing practices. A second challenge relates to the complexity of the patients who are prescribed both opioids and benzodiazepines, such as veterans with combat-related trauma or with co-occurring conditions. Thus, a consideration in developing a protocol for such an evaluation is that it will be important to identify prescribing practices that will lead to further reductions in inappropriate prescribing in a more challenging patient population for which existing approaches have not adequately addressed the problem. Furthermore, safe tapering protocols are necessary for reducing opioid and benzodiazepine use.
CLOSING COMMENTS
The committee emphasizes that the examples of study protocols in this report are only two possible target trials, chosen because they directly address the Statement of Task, are the minimum number of studies needed to address the Statement of Task, and address gaps in the literature. Adjustments to the protocols would likely be necessary after a preliminary examination of the observational data and a determination of how best to approach the studies and analyze the data. Many other studies would also be of interest beyond the outcomes of mortality and suicide in the population of veterans treated with opioids and benzodiazepines. For example, standardized self-report measures of pain, social and emotional functioning, depression, anxiety, and co-prescription of other central nervous system depressant medications could be examined to determine their effects on patient functioning over time. An examination of the clinical and functional outcomes of veterans who have been prescribed only opioids or only benzodiazepines would also be informative.
The VA medical record contains a wealth of clinical information that could be analyzed to determine the potential benefits, as well as risks, to patients with a wide variety of characteristics who were prescribed opioids and benzodiazepines. The committee views the proposed studies and any related investigations as an excellent opportunity to use the rich VA clinical databases to clarify the connections between important clinical conditions, changes in opioid and benzodiazepine prescribing practices over the years 2010–2017, and outcomes. Significant changes in prescribing practice occurred over that time period, so comparisons of outcomes of different treatment strategies could yield important insights into the best treatment practices. For example, because of concerns about high-dose opioid treatment, many practitioners in the United States have dramatically curtailed opioid prescribing in recent years in response to the increasing rates of
opioid use disorder, yet that leaves many patients struggling to cope with chronic pain problems for which they had previously relied on opioid medication. The proposed observational studies might reveal important insights into health care providers’ pain treatment practices, which could inform the use of opioid treatment as part of chronic pain management in the future.


















