Proceedings of a Workshop
| IN BRIEF | |
|
November 2019 |
Strategies for Identifying and Addressing Vulnerabilities Posed by Synthetic Biology
Proceedings of a Workshop—in Brief
BACKGROUND
In June 2018, the National Academies of Sciences, Engineering, and Medicine (the National Academies) released the report Biodefense in the Age of Synthetic Biology.1 The report was prepared for the Department of Defense (DOD), specifically for the office of the Deputy Assistant Secretary of Defense for Chemical and Biological Defense. The study process for the June 2018 report was conducted without access to any classified information and the report underwent security review prior to being publicly released as an unclassified document. Several months following the release of the report, a workshop with unclassified and closed discussions was held on October 11-12, 2018. The purpose of the workshop was to convene federal agency personnel and the report committee to explore the report and its implications for what steps DOD might explore to develop options to mitigate potential misuse of synthetic biology capabilities. Participants in the workshop included members of the committee that produced the report (see list at the end of this document); several invited scientists with knowledge of the topics addressed; and federal personnel representing the military, intelligence, and other communities. Committee members provided their individual views, not speaking on behalf of the entire committee or the original report except where noted. This proceedings document provides unclassified highlights from both the unclassified and closed workshop discussions and underwent security review prior to public release.
D. Christian Hassell of the DOD opened the meeting by describing why he requested the original National Academies report and the value provided by the report. The framework that the report provides to assess potential security vulnerabilities enabled by advancing synthetic biology and biotechnology capabilities is useful in informing DOD’s thinking on this topic and he noted that the framework could also be applied in different ways being explored by other agencies. He found the report’s recommendations very helpful and would like further discussion of implications of the concepts and recommendations in the report.
OVERVIEW OF THE REPORT
Michael Imperiale, chair of the study committee, provided a brief overview of the report. The task given to the study committee was first to develop a strategic framework to guide the identification of potential security vulnerabilities associated with advances in biotechnology, with a particular emphasis on synthetic biology. Specifically, DOD was asking:
- What are the possible security concerns on the horizon?
- What are the time frames of development of these concerns?
- What are the potential options for mitigating these concerns?
___________________
1 National Academies of Sciences, Engineering, and Medicine. 2018. Biodefense in the Age of Synthetic Biology. Washington DC: National Academies Press. The full report is available at: https://www.nap.edu/catalog/24890/biodefense-in-the-age-of-synthetic-biology.
The views expressed in this article are those of the author and do not reflect the official policy or position of the Department of Defense or the U.S. Government. Mention of any commercial product in this paper does not imply DoD endorsement or recommendation for or against the use of any such product. No infringement on the rights of the holders of the registered trademarks is intended.
![]()
The framework that the committee developed was presented to DOD as an interim report. The committee then used the framework to assess a set of synthetic biology-enabled capabilities and prepared a final report. The committee assessed 12 areas of capability related to:
- Synthesis and modification of pathogens;
- Production of chemicals, biochemicals, and toxins; and
- Modulation of human physiology.
The committee determined relative levels of concern for each area with regard to usability of a technology, usability as a weapon, requirements of actors, and potential for mitigation. They derived a list of higher, medium, and lower relative concerns for the capabilities assessed. For pathogens, synthetic biology is expected to expand the range of what can be produced for nefarious purposes, including making bacteria and viruses more harmful; decrease the amount of time required to engineer such organisms; and expand the range of actors who could undertake such efforts. This is because of the increasing accessibility of biotechnologies and starting materials. For chemicals, biochemicals, and toxins, synthetic biology blurs the line between chemical and biological weapons. High potency toxin molecules that can be produced through simple genetic pathways, of which there are many, are of greatest concern because they can be developed with modest resources and low organizational footprints. For modulation of human physiology, there may be novel ways to accomplish this, including inducing physiological changes in war fighters that differ from the typical effects of known pathogens and chemical agents. Synthetic biology expands the landscape of what may be possible in the future, by potentially allowing the in situ delivery of biochemicals by a biological agent or by allowing the engineering of the human microbiome or immune system.
Some malicious applications of synthetic biology may not seem plausible now but could be achievable if certain barriers and bottlenecks are overcome. A key message of the report is that it is important to monitor advances in biotechnology that may lower these barriers and bottlenecks. Developments in other areas, such as gene therapy, nanotechnology, automation, additive manufacturing, and health informatics, may also impact the ability to use synthetic biology-enabled weapons and make them more effective as weapons. Synthetic biology also poses mitigation challenges, such as the difficulty of monitoring and detecting synthetic biology-enabled weapons.
In the report, the committee concluded that a range of strategies is needed to prepare and respond to potential development and use of synthetic biology-enabled weapons. The committee thus recommended the continued exploration of strategies that are applicable to a wide range of biological and chemical threats. However, it also recommended evaluating the infrastructure for population-based surveillance, identification, and notification of both natural and purposeful health threats, as well as considering new strategies that manage emerging risk better than current agent-based lists and access control approaches. The report suggests several areas to explore for advancing mitigation opportunities: developing new capabilities to detect unusual ways in which synthetic biology-enabled weapons manifest; harnessing computational approaches for mitigation; and leveraging synthetic biology to advance detection, therapeutics, vaccines, and other countermeasures. More information and recommendations can be found in the full report.
DELIVERY AS A BARRIER TO MISUSE OF SYNTHETIC BIOLOGY CAPABILITIES
The next session of the workshop was devoted to a discussion of delivery mechanisms as a barrier to the misuse of synthetic biology to produce weapons. As Hassell noted, the topic of delivery mechanisms cuts across multiple threat agents and was of interest for the workshop.
Diane DiEuliis of the National Defense University and a member of the study committee briefly presented what the report states with regard to delivery. DiEuliis cited as factors that pose challenges to delivery for large-scale attacks the need for covertness, producing organisms that are transmissible among individuals and getting the pathogen to the desired cells in the victim’s body. For those agents intended to conduct or modify gene expression in the host organism, an additional factor is ensuring the expression of the right genes in the targeted cells. She noted that there are various “converging” technologies relevant to delivery (i.e., technologies being pursued outside of synthetic biology that may converge with synthetic biology advancements and ultimately reduce or eliminate barriers to the use of synthetic biology-enabled weapons), including both viral and non-viral vectors. Her presentation was followed by discussions among committee members and other participants, including Aravind Asokan of Duke University and Nick Fischer of the Lawrence Livermore National Laboratory, who specialize in two of the converging fields described in the report.
Asokan’s work deals with viral gene therapy vectors aimed at improved therapeutics. Viral vector options can include lentiviruses, adenoviruses, or adeno-associated viruses. These viruses differ in properties such as how immunogenic they are, the sizes of genes that could be packaged into them, and other factors that make certain types of viruses more suitable for varied uses. For example, Asokan noted that adeno-associated viruses are the front runners for gene therapy at the moment and are not especially immunogenic whereas adenoviruses provoke the most robust immunogenic host responses. Fischer’s work involves the development of non-viral delivery platforms for drugs and vaccines based on material science and nanotechnology. Such non-viral platforms can be made of a variety of materials including lipids, polymers, and metals, and can be produced in a wide range of sizes including as micrometer and nanometer-scale particles. Fischer’s group has developed a toolkit to adapt particles to synthetic biology components (like self-amplifying RNAs). He noted that particle size
and surface properties can be tuned, for example by conjugating targeting molecules, and optimized to affect where the particles will go in the body. However, rational design is not yet advanced enough to predict effectiveness. This is nevertheless a very active area of research. He noted that lipid-based nanoparticles are already commercially available, many other research groups are working on diverse non-viral delivery platforms, and these approaches are becoming as efficient as clinical grade viral-based vectors.
MODULATION OF THE HUMAN MICROBIOME FOR CHANGES TO FLORA AND FOR IN SITU SYNTHESIS OF BIOCHEMICALS
Committee member Kristala Jones Prather of the Massachusetts Institute of Technology gave a brief presentation on the report content related to the microbiome. The report indicates that it may be possible in the future to use synthetic biology to modulate human physiology in novel ways, such as modifying the human microbiome or other human systems. Prather said that, generally speaking, the human gut or skin microbiomes could represent opportunities to introduce harmful cargoes—modulatory molecules, toxic molecules, or molecules that cause dysbiosis (i.e., that perturb the normally healthy microbiome). For example, the manufacture of chemicals or biochemicals in situ could potentially be accomplished by engineering an organism, such as one that can survive in the human gut, to produce a biochemical and delivering this organism in such a way that it can release this biochemical in situ. An engineered organism capable of producing harmful biochemicals could be introduced with the goal of having it persist in the microbiome to release its cargo, or it could transfer its cargo via horizontal transfer to the native microbes, circumventing the challenges associated with establishing an engineered microbe in otherwise occupied niches. Prather stated that this type of technology would not be easy to use effectively at this time. But if it could be used, it might be difficult to recognize what was amiss, and attribution would be difficult. These difficulties are the reason in situ synthesis of biochemicals was of higher relative concern in the committee’s report. She noted that modulating the human microbiome is an active area of research, especially for therapeutics, so developments in this area need careful monitoring.
This session was joined by guest discussants Weston Whitaker of Novome Biotechnologies and Christina Smolke of Antheia. Whitaker’s company Novome has produced a bacterium engineered to act against a disease by colonizing the gut. Antheia is engineering yeast with pathways for making both opiates and non-opiates in situ. A wide variety of potential medical compounds might be made this way.
Engineered Microbes: Delivery, Colonization, and Persistence
A committee member suggested that key questions about microbiome manipulation remain: How can we effectively deliver an engineered microorganism and get it to persist, can we predict its effects, and what could attribution look like? The discussion did not delve into large-scale delivery challenges. But regarding persistence, Whitaker stated that if there is an open “niche,” such as a unique food source for an obligate feeder, it is not very difficult to get a bacterium established in the gut. For example, Novome Biotechnologies’ bacteria are fed an unusual carbohydrate and can reach levels of 10 percent of the microbiome, which is an enormous fraction. However, he agreed that, in general, colonization and persistence of microorganisms still remains a major barrier to a microbiome approach to delivery of harmful chemicals or biochemicals. When asked how important competition for resources in the gut is for metabolically-engineered organisms, Smolke described how there are multiple factors that impact the output of the desired chemical.
In some situations, persistence may not be the primary goal. A committee member asked whether there are organisms that could produce their product and then disappear so as to defeat attribution. Although another committee member noted that many of the harmful substances being discussed are easier to make by other means, one committee member suggested that if the release of a desired chemical could be timed at the exact moment that you want it, it would be something new. Another committee member noted that microbes introduced into the human gut may not only be pathogenic, but can also result in the transfer of antibiotic resistance and other undesirable traits from plasmids carried by the introduced organisms to the normal gut bacteria.
HARNESSING COMPUTATIONAL BIOLOGY APPROACHES
This session discussed the role of computation in synthetic biology, as well as potential opportunities computational tools might provide to mitigate concerns posed by misuse of synthetic biology capabilities through prevention, detection, attribution, or other opportunities to identify, monitor or constrain misuse. The session opened with a brief presentation by Douglas Densmore of Boston University, a member of the study committee. The discussion that followed was supplemented by four additional experts. James Diggans works on biosecurity issues at a DNA-synthesis company, Twist Bioscience. Jessica Green, of Phylagen works on the microbiome, including the microbiome of the built environment. Chris Myers of the University of Utah does work to enable the design-build-test paradigm of synthetic biology, including working on registries and hubs and developing tools for computational models. Eric Klavins of the University of Washington builds bio-products for customers, with bacteria, yeast, plants, and proteins.
Densmore described the report’s suggestion to explore computational biology as a means of studying opportunities to advance mitigation capabilities. He provided more ideas than were covered in the original report—concepts which are
captured here. Computational approaches in synthetic biology include: 1) approaches to understand and model biological phenomena, using tools like bioinformatics, big data, machine learning, etc.; and 2) bio-design automation, or how to transform high level functional requirements into a physical product automatically and manage the data along the way. Bio-design automation is more prevalent where “parts” are used in the design-build-test cycle, for example genetic circuit design, combinatorial library screening and optimization, and mutagenesis. It is not as prevalent in approaches that are more chemical engineering-based, such as making adjustments to a metabolic pathway.
A key message of Densmore’s presentation is that the computational stage cannot be bypassed in synthetic biology. Until recently the state of the art called for storing information about synthetic biology components (“parts”) in spreadsheets, but now more advanced software tools are enabling synthetic biology—so much so that new companies based on software are emerging. The user specifies what he/she wants, designs it with the help of biodesign tools, and does the physical assembly. Information can be recorded at each step to help control the outcome and stem the loss of data. This work flow is increasingly required for all product development, from the simple to the complex. Also, more and more information is being placed online. Densmore provided a list of potential opportunities for improving deterrence and prevention capabilities: Learn, Constrain, Register, Screen, and Track. Densmore elaborated on this list as noted here.
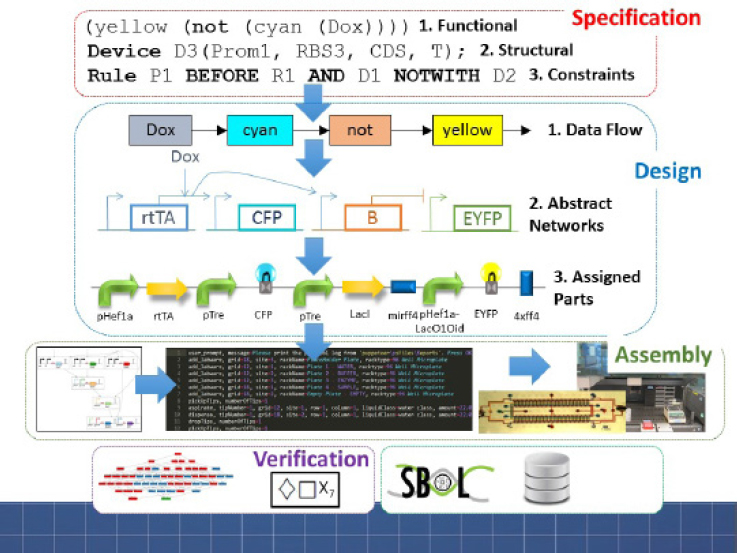
Learn – the screening of activities with machine learning to pick up patterns, is what Densmore believes is the most important opportunity computational approaches provide. We have never had as much data as we do now, and this learning can be stored as computational data structures (see Figure 1). A participant pointed out that this learning is a very difficult challenge if it is to be used to design mitigation measures, and another participant asked how we could get enough training data to develop the algorithms to recognize nefarious activity. Densmore agreed it will be difficult and a slow bottom-up process, but said that we need to begin the process of getting the biological community ready to think in terms of data sharing now. DNA orders provide glimpses into what people are trying to do, but another participant did not believe industry will want to share data with the government or third-party aggregators, making it difficult to assemble training sets. Others mentioned the proprietary nature of industrial synthetic biology work.
Constrain – systems to constrain design capabilities, and Screen – maintaining registries of known biological threats, are the next two areas described by Densmore. One participant noted that design constraints could aid researchers by preventing the combination of biological pieces that will not function well together, while such rules may also be useful in preventing the design of certain harmful products. However, he noted, the incorporation of rules into software systems that constrain certain synthetic biology designs would need to be transparent to users, but this transparency might enable engineering around the rules. A committee member asked if it was possible to build a system that can recognize and stop customers with nefarious intent. This was thought to be a continuing challenge, with another participant noting that biodesign software will not provide a magic bullet but rather can serve as one tool in combination with others to support security.
The opportunities created by the collection of customer information and the importance of looking at the supply chain were discussed. The synthetic biology field continues to advance, with a growing use of contract research organizations (CROs), “foundries,” that assist in synthetic biology production, and production facilities based outside of the United States.
More than just at the computational interfaces, business development units talk to each other to understand who is using various supply chain components such as foundries.
With regard to sequences of potential concern, the committee’s report described strategies based on agent-based lists such as the Federal Select Agents and Toxins list as insufficient for managing risks associated with synthetic biology, suggesting the U.S. government and scientific community consider strategies that manage emerging risk better. However, discussion of the select agent list was not a focus of the meeting described in this document because the list is being discussed in other venues.
Register – maintaining registries of known expertise and materials is an idea raised by Densmore and by synthetic biologists in other venues. The concept of registering synthetic biology practitioners was discussed at the meeting. A committee member asked if the practice of customer-screening for DNA orders could be made less burdensome to companies with a triage system. A committee member and another participant expressed concern that regulatory processes would foster anticompetitive approaches and would drive research underground or offshore, respectively. One committee member believes the government structure to enforce a registration system would be unwieldy and not provide enough benefit to justify it.
With regard to a different type of tracking, Green described how her company, Phylagen, can use metagenomic information to track materials goods, in terms of where they have been around the globe. This is accomplished by comparing the metagenomes of dust on the goods with the metagenomes of dust from various geographical areas. Artificial intelligence is used to analyze sample information and metadata and compare it to reference profiles to pinpoint location. For example, Green described using microbiome signatures on pills in determining whether those pills were made by non-authorized companies that are introducing counterfeit drugs into the market. Microbial signatures can be used for this geospatial location where there is sufficient baseline information characterizing the background.
MITIGATION
Several potential mitigation opportunities were discussed at the meeting, with mitigation defined broadly to include prevention, treatment, attribution, etc. Computational biology is described above; other concepts are discussed in this section.
Limitations of the Select Agent List Approach
A committee member said that the “low hanging fruit” in this area stems from the limitations of the list-based approach and that detection philosophies and methodologies need to be broadened. The list of specific biological threat organisms and toxins does not need to be abandoned, but for defense and public health it needs to be broadened. One participant pointed out there is no list that encompasses the widely distributed expertise of the scientific community on what is known about how agents can harm humans. Writing knowledge down in a structured way would assist those who need to judge whether a particular sequence is of concern. Another participant noted that each agency (e.g., the Commerce Department’s Bureau of Industry and Security) will have specific needs in terms of what substances need to be identified.
Diagnostics and Looking for Anomalies
When the committee was asked whether there are any “low hanging fruit” with mitigation potential, one member responded that genomic sequencing and proteomics for the military is one. As part of this discussion, it was noted that DOD may have unique opportunities to establish baseline measurements in populations of veterans and military personnel, and to monitor for changes in gene and protein expression over time. Such measurements could help to understand changes that occur normally compared to those associated with infections, and contribute significantly to developing monitoring capabilities. Using such information to look for anomalies in the microbiome is discussed in the microbiome section above.
Two participants discussed how the business case for commercial development of diagnostics, specifically point-of-care diagnostics, is frequently not sufficient for companies to pursue this area. The growing trend for recreational diagnostics was noted, as was the potential that some military personnel might welcome the opportunity to adopt new technologies for monitoring. Monitoring of individuals’ biology in general has become more ubiquitous, ostensibly for precision medicine. One participant noted that there are few measures other than glucose that need quick action, and this is why point-of-care diagnostics have failed. The commercial sector will be difficult to engage, he thinks, until reference laboratories do the necessary research to determine which measurements are needed. In his mind, that is the better goal for this space.
Engagement, Norms, Self-governance
In addition to other types of measures (regulations, sequence screening practices, etc.) the report includes a brief discussion on fostering norms of responsible conduct and promoting self-governance among members of the scientific community, and participants further touched on this topic. One committee member said that even though norms and self-governance will be messy, they are still worth having. He pointed out that when he was a student in another field, awareness of “dual use” issues was not part of the culture. But in the synthetic biology community, people are intentionally trying to engineer norms for their community in a transparent way. For example, another participant said that the Engineering Biology Research
Consortium community is working on how to encourage people to think about the implications of their work and to at least consider how an adversary would use their work. A committee member noted that engagement and the development of norms should include not only the synthetic biology community but also virologists and others who use synthetic biology tools. Another committee member noted that synthetic biology is a big and diverse enterprise; the norms in this space continue to evolve and develop, and what the norms will be in the future have not yet been fully envisaged.
A committee member noted that self-governance remains valuable because it is the people in the labs who need to make decisions about their research. Giving them the tools to do this is important. In addition, scientists need to continue to carve out time to discuss responsible conduct and biosecurity as part of training students. Another participant also noted that the existence and promulgation of norms of responsible behavior may aid in identifying individuals who are not complying with such norms and who may warrant additional scrutiny. Finally, another participant stated that the development of norms needs to happen at the international level to be effective, given the global nature of synthetic biology and biotechnology.
OTHER DISCUSSIONS
Beyond mitigation and the specific focus area of the meeting, participants also discussed several other points related to biotechnology in the age of synthetic biology.
Genetic Targeting
Participants asked about the targeting of specific genetic backgrounds and segregation of risk based on genetic predisposition. Several aspects of what one committee member referred to as the “dual use” potential of informatics topic are discussed in Chapter 7 of the report. The report states that while some fundamental barriers still likely limit the success of targeting, the availability of health and genomic data may increase the feasibility. The report states that it is crucial to continue to monitor developments that could facilitate targeting of particular populations.
DISCLAIMER: This Proceedings of a Workshop—in Brief was prepared by Marilee Shelton-Davenport, Katherine Bowman, and Frances Sharples as a factual summary of what occurred at the meeting. The statements made are those of the rapporteurs or individual meeting participants and do not necessarily represent the views of all meeting participants, the planning committee, the sponsor, or the National Academies of Sciences, Engineering, and Medicine.
The document has undergone security review by the Department of Defense.
Committee Members: Michael Imperiale (Chair), University of Michigan Medical School; Patrick Boyle, Ginkgo Bioworks; Peter A. Carr, Massachusetts Institute of Technology Lincoln Laboratory; Douglas Densmore, Boston University; Diane DiEuliis, National Defense University; Andrew Ellington, The University of Texas at Austin; Gigi Kwik Gronvall, Johns Hopkins Center for Health Security; Charles Haas, Drexel University; Joseph Kanabrocki, The University of Chicago; Kara Morgan, Quant Policy Strategies, LLC; Kristala Jones Prather, Massachusetts Institute of Technology; Thomas Slezak, Lawrence Livermore National Laboratory; Jill Taylor, New York State Department of Health; Marilee Shelton-Davenport (Study Director), Katherine Bowman (Senior Program Officer); Jenna Ogilvie (Research Associate); Jarrett Nguyen (Senior Program Assistant), National Academies of Sciences, Engineering and Medicine.
REVIEWERS: To ensure that it meets institutional standards for quality and objectivity, the Proceedings of a Workshop—in Brief was review by Peter Carr, Massachusetts Institute of Technology; Sarah Carter, Science Policy Consulting, LLC; Nancy Connell, Johns Hopkins Bloomberg School of Public Health; Gerald Epstein, National Defense University; and David Walt, Harvard University. Jennifer Gaudioso, Sandia National Laboratory, served as the review coordinator.
SPONSORS: This Proceedings of a Workshop—in Brief was sponsored by the Department of Defense.
For additional information regarding the activity, please contact the project director Marilee Shelton-Davenport at mshelton@nas.edu.
Suggested Citation: National Academies of Sciences, Engineering and Medicine. 2019. Strategies for Identifying and Addressing Vulnerabilities Posed by Synthetic Biology: Proceedings of a Workshop—in Brief. Washington, DC: the National Academies Press. https://doi.org/10.17226/25621.
Division on Earth and Life Studies

Copyright 2019 by the National Academy of Sciences. All rights reserved.






