Proceedings of a Workshop
| January 2020 | IN BRIEF |
Alzheimer’s Disease and Related Dementias: Experience and Caregiving, Epidemiology, and Models of Care
Proceedings of a Workshop—in Brief
SETTING THE STAGE
Mary Ellen O’Connell, executive director of the Division of Behavioral and Social Sciences and Education at the National Academies, provided a brief overview of the study—a decadal survey that will recommend AD/ADRD research priorities and investments for the next 10 years. She indicated that the study will focus on the profound effect Alzheimer’s Disease and other dementias have on individuals living with these diseases, as well as their families, caregivers, and communities. O’Connell noted that, like typical consensus studies at the National Academies, this study has a committee composed of researchers with expertise across a range of disciplines relevant to the study scope who will meet and deliberate, hold public sessions, and review relevant literature. However, as a decadal survey, it will also involve much more extensive outreach to the community than typical National Academies’ consensus studies. One major goal of the study, pursued via this extensive outreach, is to identify research ideas that are not well known but may have the potential to yield important advances.
Emphasizing the importance of outreach, Tia Powell, Albert Einstein College of Medicine and Montefiore Medical Center, and chair of the study committee, stated that the study will draw on extensive input from a wide range of individuals to assess the role of the social and behavioral sciences in supporting individuals living with dementia, their families, and caregivers. Individuals living with dementia and caregivers of those with AD/ADRD will provide input over the course of the study.
Karen Cook, Stanford University, and vice chair of the study committee, echoed Powell’s emphasis on the critical need to advance understanding about how best to support those affected by AD/ADRD. She stated that Alzheimer’s is the sixth leading cause of death in the United States and is the only disease in top 10 causes of death that cannot yet be treated or cured. Today, 5.8 million Americans are living with Alzheimer’s. By 2050, the number is projected to be around 14 million (Alzheimer’s Association, 2019). Additionally, Cook reported, more than 16 mil-
![]()
lion Americans provide unpaid care for people with Alzheimer’s disease or other dementias. In 2018, these individuals provided an estimated 18.5 billion hours of care, valued at nearly $234 billion (Alzheimer’s Association, 2019).
SPONSORS’ PERSPECTIVES
John Haaga, National Institute on Aging (NIA), explained that NIA is charged with being the lead agency in federal government for research on Alzheimer’s disease. He reported on research examining the increase in life expectancy at age 65 between 2000 and 2010, and whether increase in life added years in good cognitive health, or poor cognitive health (Crimmins, Saito, and Kim, 2016). Overall, he noted, there was an increase in the former, but disaggregating data by level of education shows that the increase is not evenly distributed (see Figure 1). The greatest decrease in years spent with dementia was for people with higher levels of education. People with lower levels of education had very little change in the number of years spent with dementia, and in life expectancy overall. Haaga commented that this relationship between early-life education and later cognitive health is not well understood, and examining the underlying causes of variation in the increase in later-life cognitive health is a research area worth pursuing.
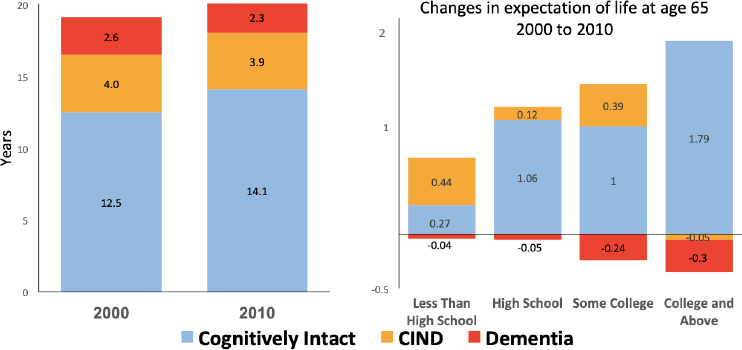
SOURCE: John Haaga, data from Crimmins et al. (2016) and Crimmins et al. (2018).
Haaga cautioned that the improvement in later-life cognitive health observed in the early part of the current century cannot simply be extrapolated forward. It is well known that the U.S. population is aging rapidly and approaching an inflection point in the population 85 and older; this population is going to grow very rapidly, and the oldest ages are at greatest risk for onset of dementia. In addition, there is a significant increase in some of the well-known risk factors for Alzheimer’s Disease among people who are now reaching older ages. Indeed, based on projections assembled by Hebert and colleagues (2013), the United States is facing a rapid increase in Alzheimer’s Disease in the coming years, Haaga reported.
Haaga briefly described the potential for the study to address the five broad goals of the National Plan to Address Alzheimer’s disease (see Figure 2). He added that all of the goals represent areas where behavioral and social sciences have a lot to contribute and this is the charge that NIA is putting forward to the committee. Elaborating on the goal of optimizing care quality and efficiency, Haaga noted that there is a lot of variation in services across the United States, even from county to county within states. What is available depends on who you are and precisely where you live. He commented that, “We can’t be doing it right everywhere if we’re doing it differently everywhere.”
Haaga outlined some of the areas where behavioral and social science research can contribute to understanding about aspects of AD/ADRD:
- Lessons from epidemiology about possible approaches to prevention
- How to anticipate future needs and plan services responsively and effectively
- How to study ways to improve services
- How to improve existing interventions
- Examining the causes and consequences of disparities and
- Identifying what data are needed to help future researchers.
Carole Sztalryd Woodle spoke on behalf of the U.S. Department of Veterans Affairs (VA). She stated that a major known risk factor for dementia is aging, and about 40 percent of veterans are over 65 years old. Sztalryd Woodle stated that the VA Office of Research and Development is particularly interested in supporting real-world impact
translational research that will guide clinical practices and policies and is looking to this study to inform that shift. She noted that one research area that is growing rapidly is the study of modifiable risk factors for dementia, which include traumatic brain injury (TBI) and posttraumatic stress disorder (PTSD)—conditions that affect many veterans as well as many in the general population. Sztalryd Woodle concluded by expressing hope that this study would inform the research agenda of the VA’s Strategic Plan for Research in Behavioral and Social Sciences to optimize health, independence, and well-being of veterans with AD/ADRD—an agenda that includes research to examine guidelines and performance measures; clinical policies; and effective services, patient outcomes, and safety.
Gavin Kennedy, Office of the Assistant Secretary for Planning and Evaulation (ASPE), U.S. Department of Health and Human Services (HHS) noted that one of the charges to ASPE is leading and managing the National Alzheimer’s Project Act (NAPA) Federal Advisory Committee. Kennedy stated that ASPE hopes future research can inform NAPA’s efforts to educate and expand the workforce, and to promote timely and accurate diagnosis, readiness for better diagnosis and improvements in treatment, identifying promising care models, and development of new, innovative care models. He noted that the current long-term care system is not meeting demand, and this is likely to worsen as care models advance. He concluded by stating that ASPE wants to ensure policy decisions are rooted in evidence and is also very interested in how to measure progress.
Sarah Tellock, Alzheimer’s Association and Alzheimer’s Impact Movement (AA/AIM), underscored the significant role social and behavioral sciences can play in reducing the burden of AD/ADRD, and stated that AA/AIM supports all of the proposed areas for review and subsequent research agenda should include the voices of individuals living with the disease, care partners, and caregivers.

SOURCE: John Haaga, data from the Office of the Assistant Secretary for Planning and Evaluation, U.S. Department of Education.
Tellock noted several areas or questions for further study, including examination of comprehensive outcomes like functionality, and resiliency; review of systems-level interventions, especially systems that include caregivers and integrate long-term and medical care; and consideration of data infrastructure needs, including looking to the field of biomedical research for parallels on measures of progress and consistency of those measures; here, Tellock highlighted biomarkers as an example.
Sarah Lock, representing AARP, indicated the organization’s interest is in three areas related to Alzheimer’s disease and other dementias—prevention, improving care, and innovations for a cure. She emphasized the need to better understand and disseminate information about how to reduce risks for cognitive decline. Lock also noted that many people are unaware of existing evidence, and asked that the study committee consider what can be done to drive the behavior change needed to accomplish what is possible. Education throughout the lifespan is important, Lock stated, not just early-life education. She underscored the need to eliminate health disparities and to determine how to help everyone reduce risk for cognitive decline as people age.
Lock noted the importance of listening carefully to the perspectives of individuals with dementia and their caregivers and note what they have to say about the outcomes that matter most. She also commented on the challenge of how best to point people to high-quality care. In response to the difficulty people face in finding information about resources and services, AARP and the Alzheimer’s Association have partnered to create a website where persons affected by dementia and their caregivers can search for locally available services by zip code (https://www.communityresourcefinder.org); Lock noted that AARP has not assessed the quality of the services.
Additionally, Lock expressed the hope that the study can quantify the benefits of improving cognitive health outcomes beyond just reducing health care costs. Likewise, she stated, a better understanding of many other elements that affect individuals with dementia and caregivers, including social isolation, transportation, housing, and en-
vironment, is critical. For example, what are the economic and other benefits to society if individuals retain good cognitive health later in life?
Speaking on behalf of The John A. Hartford Foundation, Rani Snyder expressed appreciation for the inclusion of individuals with dementia and caregivers. She commented that it is important to include the experiences and perspectives of those who are affected by Alzheimer’s and other dementias in research and in the development of programs. The John A. Hartford Foundation is particularly interested in detection and prevention of dementia, critical issues around caregiving, and the causes and consequences of health disparities.
PERSPECTIVES OF INDIVIDUALS LIVING WITH AD/ADRD AND AD/ADRD CAREGIVERS
During this panel discussion, three individuals living with Alzheimer’s disease, one individual living with Lewy Body dementia, and two individuals who have provided care for persons with Alzheimer’s disease shared information about their experiences. The panel was not intended to represent the full range of individuals with dementia or caregivers; rather, the aim for the session was to begin to learn about the experiences of individuals directly affected by Alzheimer’s and other dementias, and to develop a picture of how Alzheimer’s and other dementias can impact individuals and their families.
In her introduction to this panel, Margarita Alegria, Massachusetts General Hospital and Harvard Medical School, and a member of the study committee underscored the point made by several participants in the previous panel on the importance of considering the perspectives of individuals with dementia and caregivers in future work.
Cynthia Huling Hummel, who is living with Alzheimer’s disease, described the challenges she faced after beginning to experience symptoms at age 49. Her symptoms negatively affected her ability to conduct her work as a pastor at a busy Presbyterian Church. The process of pursuing a diagnosis was very challenging, with multiple referrals to specialists and tests, not all of which were covered by insurance. It was 8 years from the onset of symptoms to the time she received a diagnosis of amnestic mild cognitive impairment, most likely due to Alzheimer’s Disease. Huling Hummel commented that she did not receive any other information besides the diagnosis and follow-up appointment card that day, and that it would have been helpful to have received a referral to a support group or activities for individuals with an early-stage Alzheimer’s diagnosis. Postdiagnosis, she found that support services were almost exclusively aimed at caregivers. Though this was some time ago, Huling Hummel reported that she has heard of similar experiences from individuals diagnosed with Alzheimer’s disease much more recently.
Huling Hummel linked her own experiences to broader challenges. In some geographic areas there are few specialists and it is not unusual to have to wait months or travel hours away to see a dementia specialist. More health care providers who have the training and tools to make an early and accurate diagnosis are needed. Huling Hummel also commented that more awareness of Alzheimer’s clinical trials and reduced barriers to participation would be especially helpful. In addition to educating physicians and promoting research, Huling Hummel noted the need to continue building a robust dementia-capable workforce to support individuals living with AD/ADRD and caregivers. She called for the establishment of Alzheimer’s navigators—professionals who are trained to serve as liaisons who can guide persons living with dementia and care partners “through the maze and through the haze” of a dementia diagnosis, similar to those who work with cancer patients. Much more could be done to integrate clinical care with community-based support, Huling Hummel noted.
Following Huling Hummel’s remarks, J.R. Pagan, who is living with Lewy Body dementia, offered his perspectives. He urged removal of the term “demented” from the lexicon. He related a diagnosis experience with challenges similar to Huling Hummel’s, involving multiple years, appointments with several doctors, and little support at the time of diagnosis. When he did receive a diagnosis of a neurodegenerative disease via a telehealth consultation, Pagan reported, the medical provider ended the discussion without offering any further information about the condition or available supports.
Pagan, who was 53 at the time of the workshop, also commented on the issue of ageism, noting that often individuals younger than 65 are not eligible for services or resources that might be helpful in dealing with the effects of AD/ADRD. He expressed hope that the field would move to 50 as the lower-bound age for research and services, rather than 65. Pagan also raised a concern about the additional stigma faced by the LGBT community.
Ed Patterson, who is living with Alzheimer’s disease, described his experiences. Patterson noted that finding specialists such as neurologists and neuropsychologists who where accepting new patients and accepted his insurance proved to be challenging. Similar to Huling Hummel and Pagan, the time between the onset of symptoms
and diagnosis was several years—Patterson’s symptoms began when he was 63, with diagnosis at 71—and marked by a host of doctor’s visits and shaped by what insurance would and would not cover.
Patterson stated that his diagnosis of mild Alzheimer’s dementia came only after he connected with an organization that was conducting research studies focused on memory problems. Through this organization, Patterson was able to access amyloid PET scans, tau PET scans, and spinal taps that lead to his diagnosis. In addition to insurance coverage, Patterson identified the burden on caregivers as an area of concern.
Patterson experienced feelings of fear and stigma following the diagnosis,and waited for some time to share his diagnosis with family members. He closed by reminding participants that the disease does not define the person.
Brian Van Buren, who is living with Alzheimer’s disease, echoed his fellow panelists in noting that the diagnosis of younger onset Alzheimer’s is often accompanied by the experience of stigma. He also reiterated that younger onset Alzheimer’s often comes with a unique set of challenges affecting careers, families, and the individual’s financial future. Van Buren too experienced years of symptoms before receiving a diagnosis and struggled with insurance covering recommended diagnostic tests; he was unable to get a PET scan done until he received coverage through Medicare.
Van Buren stated that he represents three generations of Alzheimer’s disease. Recounting some of his experiences in caring for his mother and grandmother, Van Buren noted that Alzheimer’s disease is one of the costliest chronic diseases today, and, he continued, is projected to eventually cost more than $1 trillion annually (Alzheimer’s Association, 2015).
Van Buren briefly described the support he received through the Western Carolina Chapter of the Alzheimer’s Association, which began a pilot program for early-stage individuals shortly after he received his diagnosis. The program provided his first opportunity to disclose his diagnosis, connect in meaningful ways with others living with the disease, and relieve feelings of isolation.
Van Buren echoed Huling Hummel in underscoring the importance of participation in clinical studies. He suggested that barriers to participation should be minimized and stated that researchers should take steps to increase participation by minority populations. He also noted that results should be disclosed to participants, and that it is crucial to allow data to be used in more useful ways after a study concludes.
Marie Israelite was the first of two panelists providing the perspective of a caregiver. She began by stating that she is one of many sandwich-generation caregivers in the United States, providing care for her mother, who was diagnosed with Alzheimer’s disease in 2017, while also caring for her two young children. Echoing others’ remarks about the time lag between the onset of symptoms and diagnosis of Alzheimer’s, Israelite noted that her mother’s formal diagnosis came 5 to 7 years after noticeable changes began to occur.
Israelite observed reluctance among health care providers—in emergency medicine settings in particular—to talk about dementia and the changes her mother was experiencing in a frank manner. She wondered whether the discomfort and fear that she perceived in healthcare providers’ discussions of Alzheimer’s stems in part from a lack of understanding about what those with the disease and caregivers need in order to be supported. She noted that caregivers could also often benefit from guidance on how to communicate effectively with loved ones who are living with AD/ADRD.
Israelite identified a few areas of research that would be helpful to caregivers:
- Identifying promising practices for early interventions and treatments
- Increasing understanding about how social determinants of health, such as race, immigrant status, and socioeconomic status, impact patient access to early intervention and care
- Increasing understanding about how these social determinants affect caregiver access to information and support
- Examining how fields like social work can help caregivers learn to be better advocates for their loved ones in navigating systems and
- Identifying the types of assessments and supports that are effective for decreasing the stress of both the adults living with Alzheimer’s disease as well as the informal caregiver—helping caregivers understand how to reduce stress and how to reduce triggers for loved ones that they are caring for.
Seconding Huling Hummel’s call for the establishment of Alzheimer’s navigators, Israelite commented that even though she comes from a social services background, she found it challenging to navigate service and support systems. She also called for the development of practical tools to increase safety as the needs of older adults change so
that they can continue to live in their home or in the home of their caregivers and continue to thrive and maintain as much independence as possible.
Anxiety and depression are very much a part of the experience for many living with dementia, Israelite noted, commenting that when mental health issues are left undiagnosed and untreated, they can negatively impact willingness to comply with recommended treatment and programs. She added that ensuring treatment compliance is a concern, but so is maximizing overall wellbeing.
Another issue Israelite raised was culture as it relates to diagnosis, treatment, long-term care planning, and caregiver relationships. Speaking from her experience coming from an immigrant family, she noted that for many in the immigrant community, there is shame and stigma that comes with the loss of ability and independence on the part of older adults. There is often a cultural expectation that care will be provided and maintained strictly within that family unit; Israelite explained that because of this expectation, caregivers can also experience shame and stigma when they need assistance or outside support to navigate caregiving for a loved one. She noted that research on interventions and promising programs should really include considerations of what are culturallyappropriate modalities of communication about treatment and intervention options, including interventions and services that may be outside of cultural norms for the patient and his or her family.
Geraldine Woolfolk, who provided care to her husband Leonard Woolfolk from the time of his diagnosis in 2000 through his death in 2012, emphasized the importance of remembering that those with AD/ADRD are uniquely vibrant individuals, underscoring this point by describing the full life of her husband. She noted that people living with AD/ADRD have complicated lives, cultural variances, economic differences, and broad educational experiences, but they each must always be treated with respect and dignity.
Woolfolk indicated that her husband faced the same challenges in obtaining an accurate diagnosis that others shared, compounded by the fact that he was young at the onset of symptoms; doctors initially attributed his symptoms to stress. She also described some of the encounters she and her husband had that demonstrated the low value that society can place on the lives of people with Alzheimer’s disease and their care providers.
Communication is an important and powerful issue for researchers to consider, Woolfolk stated, adding that her husband was still able to communicate after he lost the ability to verbalize. However, his ability to communicate nonverbally was not always recognized or utilized by others outside of his family.
Woolfolk noted the importance of giving attention to what is working well currently, commenting that there may be valuable lessons to learn from data available from sources like the Alzheimer’s Association Help Line, adult day care centers, etc. She also highlighted the importance of improving resource availability in underserved urban settings and isolated rural areas; supporting families struggling with long-term care financing, and disseminating practical information that could be beneficial to individuals with AD/ADRD and their care partners.
EPIDEMIOLOGICAL PERSPECTIVES ON AD/ADRD
Deborah Blacker, Harvard Medical School and Harvard University, and a member of the study committee, offered her views on what epidemiology can contribute to understanding about disease by providing a sense of the size of the problem, both prevalence now and projected future incidence and prevalence over time. Epidemiology could also contribute to the identification of risk factors, some of which may be modifiable and thus might serve as a basis for prevention, she observed. Additionally, Blacker noted, epidemiology can contribute to the design of many types of other studies by providing an understanding of selection and sources of bias and modeling.
Kristine Yaffe, University of California, San Francisco, and a member of the study committee, noted that there are countries that are experiencing a much sharper increase in Alzheimer’s Disease and dementia than the United States, which has implications for workforce development. Overall, she reported, the number of cases of dementia is increasing, largely due to increased lifespan and demographic population shifts. However, there is some evidence that the incidence of dementia is not increasing for all segments of the population; data from studies in four high-income countries show stable or decreasing rates of dementia (see Figure 3). Yaffe commented that these findings raise the question of whether there is something happening in those populations that may be driving the observed trends. One possibility raised by researchers is the improvement in cardiovascular health experienced in populations that have seen stable or decreasing rates of dementia. However, Yaffe noted, it is difficult to disentangle what is happening because even as management of cardiovascular conditions has been improving, there have been increases in sedentary lifestyle, obesity, and screen time.
Yaffe also spoke about primary prevention for dementia. She reported that she and her colleague Deborah Barnes examined the literature and found the modifiable factors with the strongest evidence base were diabetes, mid-
life obesity, physical inactivity, depression, smoking, mid-life hypertension, and low education (Barnes and Yaffe, 2011). Yaffe explained that while one can predict a change in dementia at the population level when there is a population-level change in a relevant modifiable behavior, one cannot predict that a particular behavior change will prevent an individual from getting dementia. Therefore, she pointed out, the extent of the effect of modifying any of these behaviors has to be considered at the population level, not the individual level.
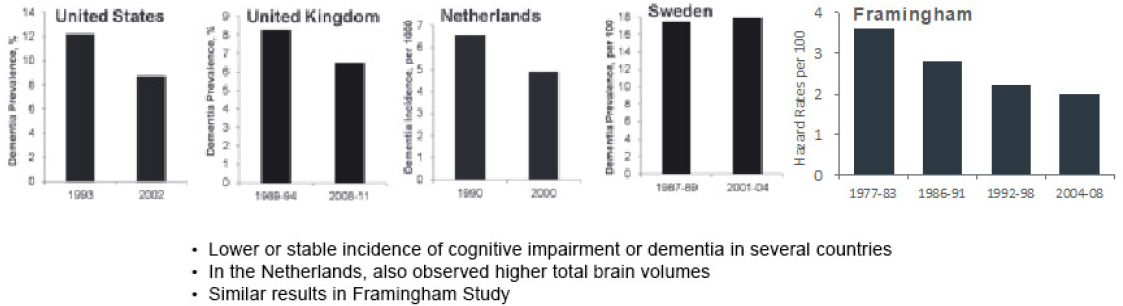
SOURCE: Kristine Yaffe, data from Langa et al. (2008); Schrijvers et al. (2012); Qiu et al. (2013); Matthews et al. (2013) and Satizabal et al. (2016).
Yaffe also discussed how standards of evidence can have a significant effect on the conclusions one can draw about a particular area of research, citing a report of the National Academies (National Academies of Sciences, Engineering, and Medicine, 2017) and an article in The Lancet (Livingston et al., 2017) about modifiable risk factors for dementia that were published in the same year that had very different conclusions, due in large part to the standards of evidence used. The National Academies reported that while there was evidence suggesting that there may be a relationship between dementia risk and three factors—cognitive training, blood pressure, and physical activity—the evidence was not strong enough to draw firm conclusions about the effectiveness of physical activity, managing hypertension, or cognitive training for reducing dementia risk. In contrast, Livingston and his colleagues concluded that taking an ambitious approach to prevent dementia was warranted, recommending interventions on nine factors: education, hypertension, obesity, hearing loss, smoking, depression, physical inactivity, social isolation, and diabetes. The reason for the different conclusions drawn in the two publications was the standard of evidence used, Yaffe reported. The National Academies’ study considered only randomized controlled trials (RCTs) involving at least 500 subjects followed for a minimum of 6 months; such studies are very expensive to conduct and there are not a large number of them. In contrast, Livingston and his colleagues based their conclusions on a much wider set of studies, including smaller RCTs and observational studies.
Maria Glymour, University of California, San Francisco, and a member of the study committee, discussed the importance of researchers being clear about the questions they want to answer. This includes both defining the outcomes of interest and distinguishing between causal and predictive questions. Glymour noted that it is not always obvious what the most important outcomes are. To illustrate this, she noted that one can measure cognitive reserve; Alzheimer’s-related pathology or other sources of pathology; resilience; functional outcomes; cognitive change or decline; or diagnostic criteria. These are all likely related to one another, she commented, but there is no particular reason to believe that the same determinants are relevant for pathology, reserve, or resilience.
Glymour suggested that from a public health perspective, research may get farther if it examines which risk factors are influencing which domain. One challenge is the gap between what researchers actually measure and what is really meaningful in people’s lives. Ideally, very rich aspects of people’s lives and their lived experiences would be measured, but instead researchers most often conduct brief psychometric tests that are not good proxies for the risk factors of interest; these tests miss a lot of what people’s lived experiences are, Glymour noted. Because of this limitation, measuring both impairment outcomes and level of change over time are important. When cognition is measured, the superimposition of multiple processes is seen—the developmental process that occurs throughout life and continues into old age, a disease process or perhaps multiple disease processes, and the resilience and plasticity that individuals experience as they encounter the world. She commented that there are multiple intervention foci for improving the level of cognitive health, including promoting brain health throughout life, helping
people experience cognitive growth and development longer and/or more quickly, delaying the onset of decline, slowing decline, and improving recovery.
Glymour commented that social differences in dementia are an indicator that social factors contribute to the likelihood of getting dementia; she noted that this can be viewed as good news because, as Yaffe had discussed, social factors are often modifiable. Glymour noted that education is an important social factor as it is modifiable via both individual behaviors and policy actions and presented several advantages of focusing on education: education appears to have few harmful side effects, has benefits beyond reduced incidence of dementia, and has broad exposure and therefore can have a large population benefit. In later life, other factors besides education are more relevant for change, such as income and possibly wealth (Marden et al., 2017; Cadar et al., 2018).
Glymour then noted that one can predict dementia risk at local or neighborhood levels, and also at macro levels like regions of the country. For example, in a study using data from Kaiser patients residing in Northern California, Gilsanz and colleagues (2017) found that those born in the Southeastern United States were at higher risk of dementia. There are a few studies that show neighborhood effects (e.g., Sheffield and Peek, 2009; Clarke et al., 2015), but Glymour noted that there may be publication bias in this line of research, with researchers less likely to publish results of neighborhood studies that do not demonstrate differences.
Another source of disparities in dementia rates that Glymour touched on is race and ethnicity. Using the data from Kaiser patients in Northern California, Mayeda and colleagues (2016) found that incidence rates for dementia are highest for African Americans and lowest for Asian Americans (see Figure 4). Glymour noted that there are variations, however. For example, in Mayeda’s study, Latinos and non-Latino whites had similar rates of dementia incidence, but other data show a different pattern for Latinos in Northern Manhattan and Latinos in California (Tang et al., 2001; Haan et al., 2003; Haan et al., 2007).
Glymour concluded by noting that in nearly every domain the evidence is inconsistent and somewhat noisy, and it can be difficult to determine whether findings are due to real differences or statistical challenges. She stated that her conclusion from the evidence is that childhood and early adulthood investments promote cognitive reserve and may have large effects on cognitive reserve or on dementia impairment. Later-life investments and resources may slow decline either by delaying disease or improving resilience and plasticity.
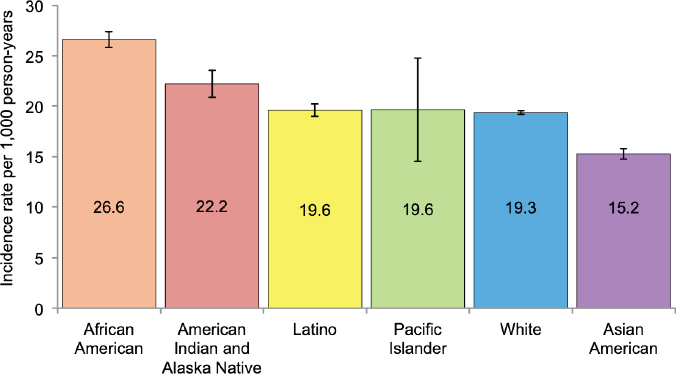
SOURCE: Maria Glymour, data from Mayeda et al. (2016).
Mika Kivimaki, University College London, picked up on Yaffe’s comments about standards of evidence, noting that limiting the evidentiary base to RCTs is problematic because there are many factors that could have bearing on risk for dementia but cannot be randomized for ethical reasons, such as smoking, alcohol use, and education. He added that to truly understand education effects, researchers would have to conduct 40 to 50 years of followup, which is not feasible.
Kivimaki stated that reverse causation is an underappreciated challenge in dementia research. He noted that there are very long periods of preclinical dementia where dementia-related physiological changes are taking place prior to a formal dementia diagnosis. Kivimaki added that when examining the relationship between dementia risk and factors such as BMI and blood pressure, it may be necessary to look at factors at least 10 years or even 20 years prior to diagnosis to discern the direction and strength of the relationship.
MODELS OF CARE INITIATIVES
Olivier Constant, Flanders Centre of Expertise on Dementia, described the “Forget Dementia, Remember the Person” campaign, which seeks to reduce stigma and make society more dementia-friendly in the Flanders region of Belgium. By creating materials such as animated videos that illustrate the experience of living with dementia (see https://youtu.be/ZofNJk9kCm0), the campaign aims to foster acceptance, understanding, adjusting to the pace of
a person in each stage of the condition, providing a safe environment for people with dementia, and talking about dementia without taboos.
Constant summarized the three central ideas of the “Forget Dementia, Remember the Person” campaign, suggesting they could also serve as a compass for future dementia research:
- Focus more on what people with dementia can still contribute to society. (Constant noted he set up the first-ever “Working group of people with dementia” in Flanders to meet this goal [see https://youtu.be/CqBKOJPxSLA].)
- Move forward on guidance toward tailored care for people with dementia and on acceptance of care and
- Develop strategies to maintain inclusion for people with dementia as long as possible.
Constant raised the importance of communication in medical and care settings, and the direct connection between stigma and diagnosis. Lag times between the onset of symptoms and formal diagnosis represent lost opportunities to retain quality of life, he commented. Noting the impact of stigma on care acceptance, Constant stated the campaign also works to help individuals with dementia move to acceptance. While a lot of information about dementia is available, people have to find their way to the that information and accept what is available.
Two other challenges Constant identified are advance care planning and social isolation of caregivers. Advance planning should start as soon as a formal diagnosis is made, he noted. Caregivers’ social networks decrease rapidly after they take on a caregiving role. Constant stated that this isolates caregivers, weakening their social ties, and also weakening of their own resilience.
Constant commented that there can be a one-sided focus on the later stages of dementia, one consequence of which is the creation of a somewhat self-fulfilling prophecy. When the media portray people with dementia as isolated, dependent on others, and losing capacities, the influence of this portrayal can lead people to mirror such behaviors, he noted. Constant called for research aimed at developing a more holistic approach to treating and supporting people with dementia that would include more focus on the person as a whole and what he or she can still do. In all stages of the condition, people with dementia retain a rich emotional life and a need for contact with others, and they still cherish dreams. Constant explained that this latter point is especially important to consider because when discussing care and policy for people with dementia; the tendency to look backward undermines attention to the fact that people with dementia still have the ability to learn and discover new things. Reducing people to their cognitive capacities gradually takes away the identity of a person with dementia, Constant cautioned.
Richard Fortinsky, UConn Health and UConn Center on Aging, underscored earlier speakers’ remarks about the challenge of obtaining a diagnosis, stating that numerous studies going back to the 1990s have found suboptimal practice patterns in the detection of cognitive impairment and diagnosis of dementia in the primary care setting (e.g., Fortinsky, Leighton, and Wasson, 1995; Borson et al., 2006). Early diagnosis of Alzheimer’s disease and other dementias is essential because earlier diagnosis then leads to more timely linkage of older adults and families with community-based educational and support services.
To address the problem of delayed diagnosis, the Gerontological Society of America assembled the Workgroup on Cognitive Impairment Detection and Earlier Diagnosis, Fortinsky reported. The Workgroup designed a four-step process (KAER) to spur an increase in cognitive impairment detection by primary care and other health care providers:
- Kickstart the cognition conversation
- Assess for cognitive impairment
- Evaluate for dementia
- Refer for community resources
These four steps together are intended to help older adults and doctors feel comfortable communicating with each other, talking about brain health, moving through a process of assessment and evaluation, and getting connected with resources—a critical part of postdiagnosis care.
Fortinsky also described the KAER toolkit, (see https://www.geron.org/images/gsa/kaer/gsa-kaer-toolkit.pdf) that includes a range of materials to facilitate the KAER process, such as suggestions for how doctors can communicate with patients about brain health and how patients and families can communicate with doctors. What often happens, he stated, is that neither party really wants to start the conversation. When doctors do not bring up the issue, patients may feel like there is nothing to talk about, and the reverse can happen, too. However, Fortinsky mused,
cognitive health is probably on everyone’s mind, and ideally such conversations would be included in the Medicare Annual Wellness Visit or other routine office visits.
Another component of the toolkit Fortinsky highlighted is the information that falls within the “Evaluate” portion of the KAER process. This information is meant to help patients and families understand the importance of following up the brief assessment with a full diagnostic workup; several studies have shown that many patients do not proceed to this step (e.g., Boustani et al., 2006).
To counter the argument that there is no financial incentive for doctors to engage in care planning and provide information on community resources, Fortinsky noted that there are billing and procedure codes that practitioners can use to get compensated by insurance providers for the time they devote to these activities.
Fortinsky shared a list of positive outcomes that might be fostered by following the KAER process (see Figure 5). He concluded by offering several avenues for future research, including usage of the KAER toolkit and other resources, how cognitive impairment and dementia affect the doctor-patient relationship, and pragmatic trials to test the effectiveness of the KAER pathway in real-world clinical settings.

SOURCE: Robert Fortinsky.
Jhamirah Howard, ASPE, focused her remarks on models that integrate acute care and long-term services and supports. To address challenges presented by AD/ADRD, Howard reported, HHS has identified the following priorities for improving care and addressing the complex care needs of people with AD/ADRD:
- Considering new person-centered models of care in Medicare and Medicaid that provide an integrated approach to addressing individuals’ medical, behavioral, long-term services and supports (LTSS), and other needs to maintain health, well-being, and independence.
- Promoting and implementing models connecting primary care, acute care, behavioral health care, and LTSS, that use health information technology effectively to facilitate transitions between care settings, especially for dually eligible Medicare-Medicaid enrollees.
- Advancing coordinated and integrated health and LTSS for individuals living with Alzheimer’s disease and related dementias.
Howard described one program for coordinating acute care and LTSS, the Program of All-inclusive Care for the Elderly (PACE). PACE involves two primary components: an adult day health center and an interdisciplinary care team. The care team includes a primary care provider; this role can be filled by either a physician or a nurse. Care teams can include many other types of care providers, such as drivers, home care coordinators, and physical therapists. Compared to other Medicaid and Medicare models, PACE is quite small, Howard noted, with a total of 47,000 participants. She added that 46 percent of PACE participants have Alzheimer’s disease or other dementias. PACE has been a Medicare option for several decades, and there is some data on outcomes for individuals who participate in PACE. Studies have found PACE performs well on a variety of outcomes, including inpatient hospitalization, nursing facility admissions, and mental health (Ghosh, Orfield, and Schmitz, 2014; Medicaid and CHIP Payment and Access Commission, 2019; Ghosh, Schmitz, and Brown, 2015), Howard reported.
Howard described another model aligning Medicare Advantage Special Needs plans with Medicaid Managed Long Term Services and Supports. Several states have begun to show interest in this model of aligning financing, and Minnesota has integrated these plans into a program called Minnesota Senior Health Options (MSHO). An evaluation of MSHO commissioned by ASPE (Anderson, Feng, and Long, 2016) found that enrollees had fewer hospital stays and emergency department visits than those in non-aligned plans, Howard reported.
Howard noted that several integrated health systems have adopted care coordination models wherein participants with dementia are identified and paired with care coordinators who help the patients and their caregivers connect
to social supports, as well as acute care, and help them manage their acute care. Many of these models also include a component that allows the individual and/or their caregivers access to their electronic health records.
Care coordination appears to have the strongest evidence of positive effects when it’s targeted, but after a certain point, further targeting yields diminishing returns, Howard commented. She added that it would be helpful to increase understanding about how to align care models that integrate long-term services and supports, acute care, and care coordination with models that are specific to dementia care. She concluded by stating that more evaluation of new and established models is needed. Models such as PACE have been around for a very long time, but Howard noted that they often have a woefully limited amount of evaluation both broadly and with a specific lens on populations that are already served by them, including the population with dementia.
Mary Mittelman, New York University School of Medicine, focused her remarks on the New York University Caregiver Intervention (NYUCI), a multicomponent intervention that provides comprehensive counseling, education, and support to caregivers for individuals with dementia. It is individualized to the needs of each family, Mittelman reported.
Mittelman described the intervention timeline, which begins with a comprehensive intake assessment, followed by a series of counseling sessions over the course of about 4 months. The first session is with the primary caregiver, followed by four family counseling sessions including people identified by the primary caregiver as important to him or her, and then a second individual session for just the primary caregiver.
After the initial series of six sessions, there are follow-up assessments every 4 to 6 months, and caregivers and participating family members have access to ad hoc (as needed) counseling, generally on the telephone. The clinicians also suggest caregivers join a support group that meets regularly for a source of ongoing support.
From 1987–2010, Mittelman and her colleagues conducted a randomized controlled trial of the intervention, following 406 spouse caregivers who were randomly assigned to NYUCI or usual care and followed for as long as 18 years. They found that family caregivers enrolled in NYUCI had reduced rates of depression (Mittelman et al., 2004a) and reduced stress (Mittelman et al., 2004b). The intervention also improved caregiver self-rated health (Mittelman et al., 2008). These effects lasted through nursing home placement (Gaugler et al., 2008) and death of the person with dementia and also through bereavement (Haley et al., 2008), Mittelman reported.
Mittelman identified social support as the active ingredient that resulted in better depression, stress, and health outcomes for the caregivers, noting that the most important aspects of social support were the number of people to whom the primary caregiver felt close and how satisfied he or she was with the support that he or she received from family (Roth et al., 2005).
One aspect of the study Mittelman found noteworthy was that during the time that the people with dementia were living at home, whether the caregivers were in the treatment group or the control group, less than five percent dropped out. She speculated that the reason for the low dropout rate was the availability of support through ad hoc counseling to all caregivers in the study, whether they were in the control group or the treatment group.
Mittelman emphasized that one important finding from the study was that by improving the well-being of caregivers, the time to a nursing home placement of people with dementia was delayed by a median of 557 days, or about a year and a half, which is about half the total average length of stay in a nursing home (Mittelman et al, 2006). She noted that this represents a very large cost savings, citing a model developed by Long and colleagues (2014) that showed that if every caregiver in the state of Minnesota received the NYUCI, the state would save $996 billion.
The program has been shown to work in other countries, including the United Kingdom and Australia (Mittelman et al., 2008), Mittelman noted, and they have developed an online training, which they have shown to be as effective as in-person training, so that clinicians in other areas can implement the intervention.
Mittelman stated that the social support paradigm demonstrated by NYUCI has informed the development of other interventions, including a couples counseling intervention, and Meet Me at MOMA, which is a program for people with dementia and their family caregivers centered around experiences at art museums; and a chorus for people with dementia and their family members (Mittelman and Papayannopoulou, 2018). She concluded by suggesting that the public and the medical professionals should reframe what they mean by care and dementia, as a more holistic approach has demonstrated benefits for people with dementia and their caregivers.
PUBLIC COMMENT SESSION
Following the formal panel sessions, the workshop included a public comment session during which thirteen individuals raised many topics worthy of future exploration, such as the tension between caregiving and employment,
stigma, communication, and research needs. Due to space constraints, the contributions during this session are not summarized here; the full content of the session is available at the link to the workshop materials above.
FINAL REMARKS
In her final remarks, Powell underscored the importance of the many topics discussed during the workshop. She noted that epidemiological studies reveal trends that can inform communication to the public about dementia and the design of interventions and educational models. The models of care session provided information about some existing efforts and what might be made more broadly available to people who could benefit from them. Most importantly, she stated, the perspectives of people living with dementia and those who care for them must be considered as the committee develops its guidance on the most useful and helpful directions for future research.
COMMITTEE ON THE DECADAL SURVEY OF BEHAVIORAL AND SOCIAL SCIENCE RESEARCH ON ALZHEIMER'S DISEASE AND ALZHEIMER'S DISEASE-RELATED DEMENTIAS
Patricia (Tia) Powell (Chair), Montefiore Einstein Center for Bioethics; Karen Cook (Vice Chair), Stanford University; Margarita Alegria, Massachusetts General Hospital and Harvard Medical School; Deborah Blacker, Harvard Medical School and Harvard T.H. Chan School of Public Health; Maria Glymour, University of California, San Francisco; Roee Gutman, Association of American Universities; Mark Hayward, Population Research Center and Population Health Initiative at the University of Texas at Austin; Ruth Katz, LeadingAge; Spero M. Manson, Centers for American Indian and Alaska Native Health in Colorado School of Public Health, University of Colorado, Denver; Terrie E. Moffitt, Duke University and King's College London; Vincent Mor, Brown University and Providence Veterans Administration Medical Center; David Reuben, UCLA Center for Health Sciences; Roland J. Thorpe, Jr., Johns Hopkins Alzheimer's Disease Research Center for Minority Aging and Hopkins Center for Health Disparities Solutions; Rachel Werner, University of Pennsylvania; Kristine Yaffe, University of California, San Francisco; and Julie Zissimopoulos, University of Southern California; Molly Checksfield, study director; Tina Winters, associate program officer.
DISCLAIMER: This Proceedings of a Workshop—in Brief was prepared by Tina Winters, rapporteur, as a factual summary of what occurred at the workshop. The statements made are those of the rapporteur or individual meeting participants and do not necessarily represent the views of all meeting participants; the committee; the Board on Behavioral, Cognitive, and Sensory Sciences; or the National Academies of Sciences, Engineering, and Medicine. The committee was responsible only for organizing the public session, identifying the topics, and choosing speakers.
REVIEWERS: To ensure that it meets institutional standards for quality and objectivity, this Proceedings of a Workshop—in Brief was reviewed by Mike Splain, Splain Consulting and Cognitive Solutions, LLC and David M. Bass, Benjamin Rose Institute on Aging. Kirsten Sampson Snyder, National Academies of Sciences, Engineering, and Medicine, served as review coordinator.
SPONSORS: The workshop was supported by the National Institutes of Health.
Suggested citation: National Academies of Sciences, Engineering, and Medicine. (2020). Alzheimer's Disease and Related Dementias: Experience, Caregiving, Epidemiology, and Models of Care: Proceedings of a Workshop—in Brief. Washington, DC: The National Academies Press. https://doi.org/10.17226/25694.
Division of Behavioral and Social Sciences and Education

Copyright 2020 by the National Academy of Sciences. All rights reserved.












