Proceedings of a Workshop
| January 2020 | IN BRIEF |
Challenging Questions about Epidemiology, Care, and Caregiving for People with Alzheimer's Disease and Related Dementias and Their Families
Proceedings of a Workshop—in Brief
SETTING THE STAGE
Tia Powell, Montefiore Einstein Center for Bioethics, opened the workshop with an introduction of members of the study committee and the advisory panel—people living with dementia and caregivers of people living with dementia—and highlighted the importance of the advisory panel in ensuring the voices of those living with the disease are reflected in the final report. Powell also introduced the sponsors for their support of this study and explained the format of the workshop.
INTERVENTIONS FOR CAREGIVERS OF INDIVIDUALS LIVING WITH AD/ADRD
Laura N. Gitlin, Drexel University, presented an overview of the in-progress paper coauthored with Joseph Gaugler, University of Minnesota, and Eric Jutkowitz, Brown University, about interventions for caregivers of individuals living with AD/ADRD. Gitlin noted that research on interventions supporting caregivers constitutes a huge body of literature; in her presentation, she outlined overarching key frameworks that are influencing the way the authors are framing the findings of that literature. She then touched on areas for further research, including new ways of thinking about how such interventions could be explored and how research might better include understudied populations.
Gitlin’s presentation focused on nonpharmacological interventions, though she pointed to the important role that pharmacological interventions also play. Labeling nonpharmacological interventions has been problematic for the paper’s coauthors because there is ambiguity and inconsistency in the literature as to how to describe interventions and their components, she noted. She explained that the authors have adopted a very broad set of parameters, thinking of nonpharmacological interventions in terms of helping caregivers manage cognitively (e.g., cognitive
![]()
reframing, positive coping strategies). These interventions also might address mood, enhance skills, address health issues, and provide resources and financial guidance.
The effectiveness of interventions for caregivers is based in part on identifying what that person needs and finding the right type of intervention for the individual. Those needs might change: research shows that they are affected by the needs of the person living with dementia who relies on the caregiver. Gitlin noted that caregiver needs are determined in part by culture, race, ethnicity, etc. The effectiveness of interventions for caregivers is also shaped in part by the needs of those they care for, she observed. Different kinds of etiologies at different stages of the disease, physical health, and cognitive functioning of the person living with dementia can all affect how caregivers participate in these interventions (see Figure 1). Furthermore, Gitlin noted, there is important research showing that the relationship between care partners and persons living with dementia affects not only the experience of caregiving, but also a caregiver’s approach to administering care, participation in and adherence to interventions, and the type of knowledge and skills that are needed and imparted.
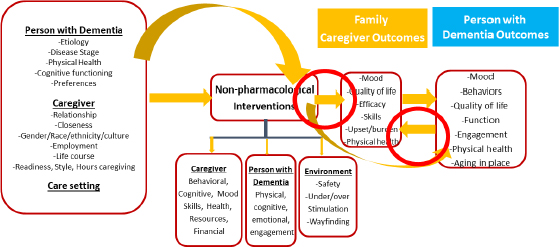
SOURCE: Laura Gitlin, adapted from Gitlin and Hodgson (2018).
Caregivers can also be affected indirectly by interventions that support the person living with dementia, Gitlin noted. Interventions that target the person living with dementia might improve the individual's quality of life and perhaps reduce excess functional disability and behavioral symptoms and mood, which in turn may enhance the well-being of family caregivers (Gitlin and Hodgson, 2018). Different kinds of interventions support the person living with dementia and/or caregiver through changes in care and enhanced job satisfaction or retention (for formal caregivers).
In addition to looking at interventions for family caregivers, Gitlin and her coauthors are also reviewing interventions that target formal caregivers in nursing homes or assisted-living facilities. These interventions primarily involve trainings to enhance knowledge and skills communicating. The authors are also reviewing domestic studies testing interventions for family caregivers as well as studies from Asia, Latin America, and Europe.
Gitlin explained that the paper will primarily review meta-analyses or other systematic reviews of efficacy studies and any reviews of studies evaluating translation of interventions into real-world settings, as well as protocol papers—publications of studies in progress. This includes more than 200 efficacy trials (Stage II or Stage III within the NIA Stage Model; Onkin, 2014) that have been summarized in more than 28 meta-analyses and systematic reviews of trials; the authors’ have found thus far that multicomponent interventions involving disease education, skill building, and support tend to be the most effective. The coauthors have also identified 16 published translational studies and recognize that there are many others in progress that will take time to be published (Stage IV within the NIA Stage Model), Gitlin stated.
Additionally, Gitlin and her team have identified more than 60 studies that target formal providers working primarily in nursing homes and also in assisted-living facilities, most of which also examine feasibility. The coauthors are looking at that literature both in order to determine what the characteristics of the intervention are and as a way of capturing outcomes that are relevant to stakeholders in the health care system.
Gitlin identified multiple suggested areas for future exploration based on their findings to date. First, she noted the need for improving how research is conducted such as improving the clinical relevance of trials through better measurement, adequately describing interventions to enable reproducibility, replication, adaptation, and widespread adoption. Second, she recommended examining outcomes in terms of relevance to different stakeholders and enhancing study designs, including deriving strategies for targeting at-risk caregivers and strategies for tailoring to caregiver needs, lived experiences, readiness, styles, dementia stage, person’s needs, and families from different
racial/ethnic groups. Gitlin’s final recommendation was to engage in implementation science in order to move existing proven interventions into real-world settings.
Gitlin ended her presentation asking the question of whether every caregiver needs an intervention. She concluded that not all caregivers need interventions, but all should be assessed for risk and provided basic information about the disease processes. Identifying who is at most risk is important, she noted, but accounting for the person’s readiness for support and his/her own needs is also an important step in the process of evaluating who should be targeted for such interventions.
INTERVENTIONS FOR CAREGIVERS OF INDIVIDUALS LIVING WITH AD/ADRD
Joeseph Gaugler, University of Minnesota, reviewed the status of his paper, coauthored with Gitlin and Jutkovitz, on interventions for individuals living with AD/ADRD. Gaugler noted that the paper will focus on reviews of efficacy and effectiveness of nonpharmacological interventions in dementia, and aim particularly to develop a better understanding of how to scale, evaluate, and eventually disseminate and implement interventions for people living with dementia.
Gaugler questioned whether current measures of the efficacy and effectiveness of interventions are right for promoting further dissemination and implementations so that more people could use them and benefit from them. He commented that further exploration in the paper will focus on diverse perspectives informed by culture, race, and socioeconomic status. There are efforts to harmonize measures utilized and evaluated across interventions, he stated, but more work needs to be done to ensure proper evaluation and dissemination to the community. The authors plan to incorporate additional recommendations from the Research Summit on Dementia Care Building Evidence for Services and Supports as well as the National Alzheimer’s Plan Act Council meetings and other key guidelines and reports.
Gaugler explained that researchers are putting multiple components in interventions and evaluating them, which has shown to be effective. He added that some key questions to be addressed in the paper include: How feasible is it to take those intervention components and implement them in real-world situations? Does one component work better than the other? Do different components interact? Is one component less effective? Can research identify these qualities more directly so as to expedite the pipeline from intervention, design, and evaluation, to implementation? Can this information be applied to dementia care science?
Gaugler stated that the field is stuck at efficacy testing, and pragmatic trials are one step toward improving the dissemination and implementation process. The authors are interested in what it will take to evaluate interventions in real-world settings more quickly and efficiently. In so doing, the authors believe researchers can get a better sense of whether these intervention models work in clinics, health care systems, and other types of health care provider situations.
Gaugler and his coauthors have observed that implementation science is rapidly emerging, and wonder how it might be applied to the study of non-pharmacological interventions for people with dementia. Implementation science takes a different mindset, with different methodological skills and repertoire to understand, Gaugler explained. He is interested in whether organizations are ready for the implementation phase, how interventions might be modified to fit within a health care system or community provider environment, and what should be measured and evaluated throughout that effort.
Gaugler noted that part of the paper will focus on dementia friendliness. In this, the authors will expand their scope beyond interventions solely targeting individual-level outcomes, health care systems, and community outcomes. He questioned how scientists can help these entities to become more dementia friendly and what that means for future evaluation of interventions.
Gaugler noted that the authors would like to explore the ramifications for the types of measures to be implemented and what kind of work needs to be done to develop them effectively. In particular, he highlighted the importance of reconsidering how interventions are evaluated so as to capture the lived experience of people with dementia and their family caregivers.
DATA ON AD/ADRD PREVALENCE
Julie Bynum, University of Michigan, presented about the status of her paper with coauthor Ken Langa, University of Michigan, on data sources for measuring population prevalence of AD/ADRD. Bynum explained that prevalence data are important for health policy, social policy, and many other issues that have broader impacts than just disease status. Prevalence data and epidemiological studies are important for understanding public health surveil-
lance, determining where populations live, and understanding demography of the population that supports those living with AD/ADRD. Prevalence data also helps researchers recognize what is happening at a national level and what is influencing those trends, she noted. Measuring at this broader level allows scientists to account for the people who do not participate in clinical trials; neglecting this broad view can result in a lack of understanding of what’s going on at a generalizable scale. Prevalence, special groups, and location play important roles in the study of how ADRD affects various populations.
Bynum then described the issue of evolving definitions of diseases variously known as dementia and AD/ADRD (see Figure 2). The first disease classification began in 1984. A research diagnosis followed in the 1980s, and a first drug was approved in 1987. Clinical diagnostic guidelines were revised in 2007, and 2011 saw major revisions that were partly driven by the emergence of the concept of mild cognitive impairment (MCI). In 2018, the divergence of a biologically defined disease and a clinical syndrome made that diagnosis much more explicit.
Bynum noted that because of these changes, not all definitions are stable yet, making it difficult to plan a 20-year trend population study. As definitions change, old data can lose relevance, collection of new data is uncertain, and there is disagreement in the scientific community about how to develop new definitions to properly account for population prevalence.
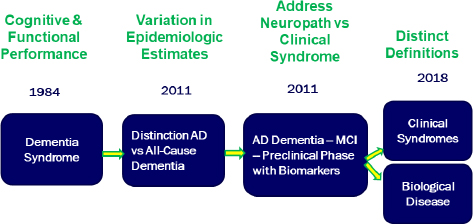
Source: Presentation by Julie Bynum.
Bynum described the variety of data sources available for population surveillance, including epidemiological data, which can be obtained through clinically adjudicated diagnosis or through survey instruments. Survey instruments are predominantly standardized and applied generally to larger populations, with the resulting scores used to develop data categories, Bynum explained. Some studies use standardized instruments, like the Health and Retirement Study (HRS) and National Health and Aging Trends Study (NHATS). She noted that various kinds of epidemiological studies use different data, in different regions, with different samples, and different goals; because of this, Bynum argued, it is not surprising that disagreement between epidemiological studies presents a major challenge.
Another challenge, Bynum explained, is that there are groups of people, distinguished by race, ethnicity, and possibly age and/or geography, who are not equally represented in these studies. Implementing a mechanism to gather nationally represented data will be a large, costly undertaking, and if the goal is to do in-person studies to get actual adjudicated diagnosis by type.
Bynum stated that data are also collected in the process of clinical care, including doctor’s notes and billing data; such data are increasingly being used and sometimes seen as reflecting disease prevalence. She noted that although these data are now much more available to researchers, she will explore whether this is the right set of data from which to derive prevalence. In her paper, she also plans to touch on electronic health record data, as this is an area with a tremendous amount of growth and an interesting tool to identify people who have this disease or may have it in the future. Bynum cautions that this clinical data’s use requires a nuanced understanding of the factors that influence whether a person obtains a disease label in the clinical setting. According to Bynum, there are changing diagnostic coding systems, changing clinical practice patterns, and differences in practices across regions, and clinicians’ summarizing data depend on individual expertise, which may be highly variable.
Bynum pointed to the Medicare payment system as one source of such data, functioning as a sort of a national repository. Every bill that a doctor or clinician submits must have a diagnosis on it to be paid, so data are rarely missing; and the system gives scientists access to information from every part of the United States. From a broader perspective, this can be seen as a good data source because data on a large portion of the population are available, which has been a problem for epidemiological studies, Bynum explained. She also noted that there are challenges, such as the exclusion of some immigrants. Bynum also touched upon the use of biomarkers and nontraditional data, noting particularly the challenge in incorporating that information into prevalence data. Some epidemiological studies include biomarkers, but these are regional and do not cover large groups. Bynum also noted that
because technology is still evolving, it is not yet clear what the best measure of biomarker data is going to be. This is exacerbated by the fact that all biomarker data come from clinical rather than population data groups; as such, there is potential for real biases since researchers do not also have general population biomarker data. Another point raised by Bynum was that most epidemiological data do not specify the type of dementia, which may have implications for disparities measurement.
Bynum also observed that there is a move to use artificial intelligence and machine learning to extract information from electronic health records to address these problems. While clinicians may not bill for this diagnosis, their comments in notes can identify people with symptoms. A number of algorithms have been developed to use electronic health record data to extract those missing patients.
HEALTH DISPARITIES AMONG ADRD
Ana Quiñones, Oregon Health & Science University, presented via Zoom about the status of her paper with Heather Allore, Yale University; Anda Botoseneanu, The University of Michigan; Jeffery Kaye, Oregon Health Services University; and Stephen Thielke, University of Washington, on health disparities. Quiñones stated that the paper will focus on accessing needed services, supports, and planning tools. Here, she noted, it is important to focus on not only the clinical approach to AD/ADRD, but also a population-based approach to improve dementia care—especially in thinking about dementia care that may or may not be reaching minority ethnic populations. Access to advanced care planning or palliative care—including eliciting patient values and preferences, initiating and continuing discussions of care, evaluating health care decisions, identifying health care surrogates, completing and registering advanced health care directives, and financial planning—are areas in need of improvement to effectively reach minority communities (Reuben et al., 2019).
Quiñones explained that exploring the drivers of health care-seeking behavior is another important theme that emerges in the paper. There are cultural norms, values, beliefs, and roles that shape definitions and differences in response to symptoms (Dilworth-Anderson and Gibson, 2002). There is also a distrust of formal clinical and medical research institutions and a history of systemic exclusion from care, as well as a history of discrimination, she noted. Quiñones will also examine domains of affordability, accessibility, availability, accommodation, and acceptability (McLaughlin et al., 2002). She explained that cumulative inequality theory places social structures as essential, such that early-life experiences can be translated to later-life outcomes; decisions to seek care do not necessarily derive from individual choices and actions but are more importantly structurally generated. Quiñones noted that the paper will devote time to thinking about a conceptual framework around inequality and health disparities.
According to Quiñones, there is a need to identify best practices and methods for outreach and communication in ethnic-minority populations—modes that are effective at conveying accurate information and debunking misconceptions. This includes both understanding and identifying the best mechanisms for transmission of information and integrating cultural and social information gained from a variety of intervention research about diverse groups. The paper will also focus on possible differences in dementia recognition and healthseeking behavior that are driven by self-assessment to the condition. She stated that the authors will evaluate the literature about whether or not work and workload may be more acutely felt in ethnic-minority populations, who, on average, have greater burdens and higher level and rates of multimorbidity (Spencer-Bonilla et.al., 2017).
Quiñones said that there will also be a focus in the paper on themes that emerge around the unfamiliarity with symptoms and the lack of general knowledge about AD/ADRD; welldesigned information campaigns, such as the Alzheimer’s Association may not be as effective in transmitting information across all populations. Another theme across the literature is normalization of symptoms as normal aging so that someone living with dementia or their caregiver might not perceive they have a problem, and will not seek help for it.
One important challenge Quiñones discussed is the need for more granularity and greater attentiveness to specific aspects of ethnic-minority populations rather than relying on race or ethnicity as markers or proxies for groupings that are heterogeneous. There are also calls for diversification of the workforce stemming all the way from training of existing workforce and staffing to recruiting and retaining diverse workforce to work in both the clinical and social services areas. The paper will address other challenges that have to do with accessibility to and availability of services. She will also explore whether specialized services are available when they are needed. There are also nuanced considerations when thinking about validation of dementia assessment tools specific to particular ethnic-minority populations. Quiñones views acculturation status and cultural orientation as important considerations in understanding the validity of these particular assessment tools, as questions arise about whether they are measuring what they are intended to measure.
ADRD: A HEALTH ECONOMICS AND PUBLIC POLICY PERSPECTIVE
Pei-Jung Lin, Tufts Medical Center, presented about the health economics and public policy perspective of AD/ADRD. Lin noted that Alzheimer’s disease imposes a substantial economic burden on patients, caregivers, and the health care system. The Alzheimer’s Association’s estimates health care costs totaling $290 billion a year, with Medicare and Medicaid as the primary payers of these costs; these include medical care, prescription drugs, long-term care, hospice (see Figure 3). She also mentioned that patient out-of-pocket costs are substantial for things like health insurance premiums, deductibles, co pays, and long-term care services that are not covered by other sources.
Lin added that it is well documented that AD/ADRD are also associated with substantial caregiver spillover costs and spillover health effects, such as lots of unpaid, informal care, and caregiver stress, which may increase the risk for anxiety, depression, and sleep disorders (Alzheimer’s Association, 2019).
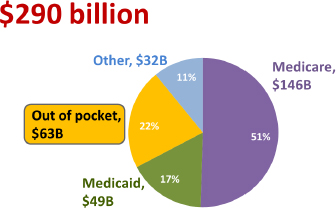
SOURCE: Pei-Jung Lin, from 2019 Alzheimer's Association Facts and Figures.
Population projections indicate that more than 9 million people will be living with AD/ADRD in year 2050, with an annual cost of $1.5 trillion; this assumes status quo, with no effective treatment to delay disease onset (Zissimopoulos et al., 2014), Lin noted. She explained that if the disease onset can be delayed, then there may be significant economic and longevity benefits.
A study led by Lin in 2016 found that people with AD/ADRD incur higher costs than those without the condition, even before they are diagnosed (Lin et al., 2016). In the year prior to diagnosis, Alzheimer’s patients spent, on average, $4,400 more than individuals not diagnosed with Alzheimer’s disease in Medicare expenditures, and that they continue to have higher costs postdiagnosis. Lin noted that annual costs of care are more than three times as high for individuals living with AD/ADRD; those costs typically rise around the time that a person is first diagnosed. AD/ADRD can complicate the management of comorbidities that are highly prevalent in people living with dementia, which can also substantially increase health care costs (Alzheimer’s Association, 2019).
Lin noted that total health care cost for a Medicare patient with AD/ADRD is nearly $49,000 a year, with in-patient nursing home and hospital care accounting for more than one-half (Alzheimer’s Association, 2019). Some admissions to nursing homes and hospitals may be preventable, such as those for unmanaged comorbidities like diabetes or diabetes complications, which could have been managed in an outpatient setting, she observed. Also preventable are unplanned admissions, which usually signal poor quality of care or poor care coordination.
Lin also highlighted data that showed that AD/ADRD cost drivers differed by care setting. In community-based settings, roughly 40 percent of the total patient expenditure covers direct health care costs, while 60 percent of costs are related to the informal care provided by family and friends (Schaller, 2015). This is in contrast to the institutionalized setting, where more than 85 percent of expenses are direct health care costs. It is well documented that AD/ADRD costs rise with increasing disease severity (Gustavsson et al., 2011).
Lin emphasized that, from a payer’s perspective, the real issue is budget impact. She explained that while there are more than 130 Alzheimer’s treatment agents in trial (Cummings, 2019), many are disease modifying, which likely means high-cost treatment that will likely result in controversies over drug pricing, coverage, and reimbursement issues. Lin noted that a drug might be cost effective but still not affordable. Costs of care and diagnostics also increase budget impact, she observed. All costs are anticipated to rise, Lin reported, and whether savings generated from early detection and treatment will offset costs associated with follow-up care depends on who is in the target population (Lin and Neumann, 2013).
Lin reported on a 2017 study by the RAND Corporation (Liu et al., 2017), which shows that based on historical and projected infrastructure estimates in 2017, roughly 7.5 million people needed to see a dementia specialist for evaluation for MCI or early AD in 2019 although the United States has about 27,000 dementia specialists, including neurologists, geriatricians, and geriatric psychiatrists, and this number seems unlikely to increase rapidly in the next 20 years, according to projections (Liu et al., 2017). Some may get amyloid testing, and about 3 million people who test positive for amyloid deposits will see a dementia specialist to learn more about treatment options. Lin reports that, given these numbers, the RAND study estimated that patients may have to wait 14 months to be seen by a dementia specialist (Liu et al., 2017).This problem may be even more prominent among some racial mi-
nority groups because they may have poorer access to specialist care to begin with.
Lin observed that while there are a lot of new, exciting developments in finding a cure for Alzheimer’s, it is going to be a while until a cure is discovered; and in the meantime, there is an ongoing care management issue. Most individuals living with the disease have other chronic conditions, so making sure that they are receiving appropriate dementia care and appropriate comorbidity management should be a priority, she argued.
In conclusion, according to Lin, the way we think about Alzheimer’s disease has changed over the years. The focus has shifted to recognizing earlier stages of the disease, whether by way of clinical symptoms or pathological and biomarker findings. If there is any hope of getting treatment for a good value, she added, it is going to depend on early diagnosis to identify who might benefit from such early treatment.
DEFINING TYPES OF DEMENTIA
David Bennett, Rush University Medical Center, spoke about the defining characteristics of different types of dementia and how these definitions have evolved over time. Bennett outlined the updated diagnostic criteria developed in 2011 for MCI and Alzheimer’s disease, especially noting the introduction of the preclinical stage of Alzheimer’s disease. He also explained the differences between MCI and Alzheimer’s disease: MCI is a change in cognition with evidence of cognitive impairment but preservation of independence and functional abilities (Albert et al., 2011)—that is, loss of cognition that does not impair function—as distinct from dementia, which is defined by loss of cognition that does impair function.
Bennett also described the differences among the types of dementia. The cerebral vascular disease field, for example, has had an advantage from a nomenclature perspective in that they have had for many years a separate term for the underlying biology—cerebral vascular disease—and a different term for the clinical syndrome, stroke. An individual could have cerebral vascular disease and not have a stroke, which is the equivalent of preclinical AD, where the pathology is present but symptoms are not. He also stated that diagnosing Lewy Body dementia and frontal temporal dementia are difficult to use in a population study because diagnoses of these diseases are targeted toward tertiary care clinicians who have to make a clinical diagnosis rather than researchers who need to implement the diagnoses in the field. Bennett noted that to properly capture population-level data for these diseases, one has to sample at a high rate to get precise prevalence data.
Bennett described the differences between prominent studies of dementia prevalence: the Chicago Health and Aging Project (CHAP) (Evans et al., 2003), the HRS, and the Aging, Demographics, and Memory Study (ADAMS) (Plassman et al., 2008). He reported that the studies found different levels of vascular dementia, some of which indicated Alzheimer’s disease plus vascular cognitive impairment. Bennett noted that these studies also raised the important point that clinicians are often forced to make a diagnosis even if they are not confident about the type of dementia prevalent, a situation that may exclude a number of people diagnosed. The level of cognition among people living with dementia as measured in HRS was much lower than in CHAP (Wilson et al., 2011).
Bennett also reviewed the longitudinal data from clinical patients who typically experience rapid rates of cognitive decline. For example, Wilson and colleagues (1999) cite data from community-based studies such as the East Boston study, which shows that older people are declining faster. By contrast, clinical data reveal that younger people living with AD/ADRD are declining faster (Wilson et al., 2000).
Bennett explained that there is limited longitudinal data from community-based studies of Blacks. In CHAP, Blacks and Whites decline at similar rates (Wilson et al., 2010). However, differences emerge with more nuanced examination. While Blacks living with AD/ADRD have lower abilities at baseline across multiple domains, they decline more slowly in semantic memory, perceptual speed, and visuospatial ability (Wilson et al., 2012). With additional waves of follow-up check-ins, a random change point model indicates that among persons who developed AD/ADRD or MCI, rate of change was faster for Whites once decline began (Kumar et al., 2017). Because of measurement invariance, it is not clear whether these subtle differences are biological or a result of psychometric differences in the cognitive tests. Bennett mentioned that the larger the study, the more likely those subtle effects will be significant, and the more likely it is that issues across ethnicity get more complicated.
Bennett then reviewed AD in light of the new NIA-AA AD Research Framework, which defines AD based on brain pathology (Jack et al., 2019). He noted that pathological AD only explained about one-third of Alzheimer’s dementia cases with another one-third explained by seven additional brain pathologies (Boyle, 2019). Further, in just over 1,000 people, they see almost 250 unique combinations of brain pathologies in people with or without dementia, with Alzheimer’s disease alone present in less than 6 percent (Boyle, 2018). Further, since many brain pathologies contribute to a loss of multiple cognitive domains, it is virtually impossible at this juncture to reliably distinguish different cases of dementia on a case-by-case basis in a community study (Wilson et al., 2019).
Bennett raised the point that the scientific community looks forward to utilizing blood-based biomarkers for AD at this level of the population. In an era that now distinguishes AD from its clinical consequence, the definition of risk factors for AD should be expanded. He suggests that large community studies should be banking serum and plasma so when those biomarkers are ready, they can be deployed in studies to help differentiate risk factor for dementia from risk factors for AD.
Bennett confirmed that he is in favor of having one term that refers to the biology and a different one for the clinical syndrome. He noted that he used the term “Alzheimer’s dementia” because he wants to make sure that future studies can be linked to past literature to ensure we do not lose secular trend data.
Bennett suggested that at a very high level, more data are needed on regional, sex, racial/ethnic differences in the prevalence of cognitive impairment across the United States. He suggested that gathering additional data on prevalence is a good start and pointed to incidence data as an important supplemental avenue of inquiry. Even though prevalence initiatives are being developed to present testing in Spanish, getting accurate prevalence estimates from community studies is difficult given the underlying heterogeneity of the Latinx population.
Bennett highlighted the need to develop simple, inexpensive approaches to diagnosis—a challenging goal made even more so by the importance of including populations that are traditionally underserved. Once inexpensive approaches are developed, trends need to be monitored over time, he noted. Generalizability and internal validity are competing for resources and studies, so beyond some social determinants, population-level data are not always the best way to accomplish a risk factor assessment, Bennett observed.
Bennett argued against current expansion of efforts toward a differential diagnosis of AD/ADRD. For political reasons, he noted, he would favor a general Alzheimer’s dementia diagnosis because there is currently more money available for AD/ADRD as defined by the 1984 criteria of progressive dementia with amnesia.
Bennett concluded that the accepted criteria for the clinical diagnoses of common dementia syndromes are made by and for clinical specialists, and it is imperative to adapt these for use in community-based studies. He also reiterated that Alzheimer’s dementia is on a continuum, where small differences in cut-points in how criteria are implemented can result in very large differences in estimated prevalence, especially when looking at studies like HRS and with CHAP, which focus on different communities and weight data differently.
MEASURING THE EFFECTS OF CAREGIVING
Rachel Werner, University of Pennsylvania, introduced the final panel, during which four individuals and two discussants reviewed measurement of the so-called “burden” of caregiving.
Julie Zissimopoulos, University of Southern California, presented her perspective on measuring the effects of caregiving. Zissimopoulos reviewed her team’s dynamic microsimulation model, called the Future Elderly Model for Understanding Alzheimer’s Disease (AD-FEM), which measures the economic costs over time of Alzheimer’s disease for persons with the disease, their caregivers, and society. Zissimopoulos explained that the model uses longitudinal data from the HRS to quantify and model the impact of social and economic factors and health behaviors on the onset of Alzheimer’s disease and how these factors affect other diseases and conditions that are risk factors for Alzheimer’s disease, such as hypertension and diabetes. The model can also be used in a number of other ways: to examine the impact of policy changes; to assess the long-term impact of new treatments and the outcomes for clinical trials measuring short-term or intermediate outcomes; and to measure a comprehensive set of caregiver outcomes including impact of interventions on the physical and mental health of caregivers or their well-being. Zissimopoulos noted that because of the flexibility, the underlying assumptions can be changed in the model, such as who participates in an intervention, who discontinues, and the heterogeneity effects across populations whether they are defined by race, ethnicity, or socioeconomic status.
Zissimopoulos reported that she and her team also use these data to measure how individuals use health care services and quantify how much informal and unpaid care they receive. They aggregate all these individual-level outcomes so they can also look at populations, health and spending outcomes, particularly health care costs and the value of unpaid caregivers for society. This model can be used as a mockup simulation tool to project future health and economic outcomes. FEM simulation results indicate that the annual value of unpaid caregiving in 2019 was $31,000, and 40 percent of combined health care and caregiving cost and that costs are higher for nonwhite caregivers compared to white caregivers, she reported.
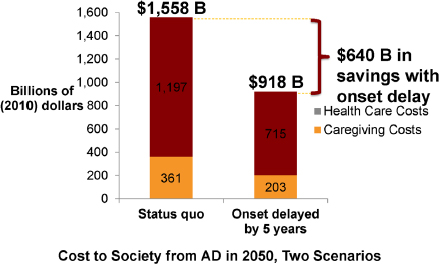
When describing the potential for costsavings, Zissimopoulos noted that in the future, Americans aged 70 and older with Alzheimer’s disease will increase by 125 percent (more than double the current rate) between now and 2050 (Zissimopoulos et al., 2014). Delaying onset by 5 years would lower those numbers by about 41 percent, she
observed. The aggregate population costs in 2050 are projected to be $1.5 trillion, with the value of unpaid caregiver time alone projected to be $361 billion. With the delay scenario (see Figure 4), there would be $640 billion in savings, over $150 billion of which represents caregiver cost savings.
Zissimopoulos noted that her team defines population burden as a function of the number of persons with disease, the duration of disease, and the cost of the disease. Even if all of these factors are calculated accurately, the total figure would still underestimate the full cost since there are other costs to the disease such as the impact on the physical and mental health of caregivers, impact on productivity on the workplace, lasting impacts of family wealth loss, etc.
Zissimopoulos suggested that policies can be implemented that might help reduce costs, but that it is important to consider whether the policies are reducing costs or simply cost shifting. Paying family caregivers, for example, could reduce the burden on them and their families, depending on how it is financed. Policies establishing reimbursement incentives meant to reduce postacute institutional care have been shown to reduce the cost to Medicare, but will likely shift costs to families. Better coordination of dementia care may reduce hospitalizations, delay nursing home entry, and may have benefits to caregivers through reduced stress.
Emily O’Brien, Duke University, began by discussing the importance of developing caregiver interventions that are comprehensive and intended to target and improve caregiver activities, particularly with respect to seeking support, and psychological activities that can then have a positive impact on caregiver outcomes and, indirectly, care recipient outcomes. She noted that a 2011 study found that the most commonly studied caregiver outcomes were burden and depression, and the most commonly studied care recipient outcomes were physical function and health care resource use (Van Houtven et al., 2011). O’Brien’s own presentation focused on two gaps of quantitative measures for caregiving: how caregivers perceive themselves to be recognized and valued by health care professionals, what the interactions look like with the health care team, and whether they feel their voices are heard; and the concept of home time.
O’Brien described the Caregiver Perceptions About Communication with Clinical Team members (CAPACITY) measure, an instrument that measures the extent to which caregivers feel that they are experiencing person- and family-centered care (Van Houtven et al., 2017). It is important for providers to understand the caregiver’s abilities and knowledge, as well as physical, cognitive, or emotional limitations can potentially affect their quality of life and the support they are able to provide. Because of this, the tool helps to optimize care of the patient in the home (Van Houtven et al., 2017).
Van Houtven and colleagues found that individuals with cognitive impairment, such as individuals living with AD/ADRD, may influence the caregiver’s perceived communication amongst care team members and also affect caregivers’ ability to provide support, O’Brien reported. In this population, O’Brien and her team found that the items in the survey fit the expected two-factor structure of communication and capacity and also found that higher cognitive functioning patients and higher literacy of health care partners was associated with low domain and overall scores for their measures, which O’Brien noted could warrant interesting future investigation (Van Houtven et al., 2017). Van Houtven and colleagues also found that poor care partner well-being was associated with lower CAPACITY scores.
According to O’Brien’s team’s data, associations between clinically salient outcomes such as medication adherence, rehospitalization mortality and capacity could be interesting to investigate in the future. O’Brien speculated that it would also be beneficial to look at what types of skills (clinical skills, support seeking skills, psychological skills) CAPACITY scores are associated with, and whether higher scores are associated with better reported emotional or physical well-being. O’Brien and her team are also interested in whether higher scores are associated with greater reports of caregiver satisfaction with the health care team
O’Brien also referenced another study of hers that looked at the correspondence between home time and patient-reported outcomes using data from the Medicare current beneficiary survey, and evaluated incidence of worsening patient-centered outcomes and home time (O’Brien et al, 2016). Another group of researchers found that less time spent at home over the course of a year was associated with greater rates of worsening patient-centered outcomes including self-rated health mobility impairment, depression, social activity, and difficulty in self-care (Lee et al., 2019). O’Brien emphasized that while a possible future area of focus around how home time may be patient centered, it is also important to investigate how increased days at home might translate to caregiver well-being. She urged the committee to think creatively about how to combine data sources to address that question.
Judy Kasper, Johns Hopkins University and principal investigator for the NHATS, explained that NHATS provides data that focuses on disability in later life from in-person interviews with more than 8,000 people and has been conducted annually since 2011. She also reviewed the work of the National Study of Caregiving (NSOC), which interviews caregivers to persons in the NHATS study about their caregiving experience. NSOC has been conducted periodically and will be conducted longitudinally starting in 2020.
Kasper explained that NHATS identifies activities for which an older person is receiving help and asks who is helping with activities of daily living (ADLs) and instrumental activities of daily living (IADLs) like self-care and mobility, as well as activities like transportation and medical activities. The extensive information from NHATS on the physical and cognitive functioning of study participants is also sufficient to characterize care recipients. These data help define the care recipient populations and also define who caregivers are.
Kasper reviewed a table of data from NHATS 2011 showing estimates of people receiving assistance with activities by dementia status of care recipient and types and levels of help. Activities included self-care and/or household activities by the dementia status of the care recipient. About 53 percent of recipients with dementia, were receiving help with self-care, compared to only about 11 percent of those without dementia, while assistance with three or more activities was observed in 40 percent of the dementia care recipients, and only 14 percent of those without (Kasper et al., 2015).
Also available via NSOC is an assessment of caregiving that includes positive and negative aspects and areas of difficulty that are directly tied to caregiving (see Figure 5), Kasper reported. This includes the overall well-being of persons who are caregivers, including depression, anxiety, and subjective well-being measured both in terms of current feelings and longer-term self-realization measures. Kasper emphasized that caregiver well-being is both an outcome and a contributing factor. She reviewed data from Jennifer Wolff, Johns Hopkins University, noting that caregivers who provide more assistance with health care activities such as medication management for the person with dementia they are caring for report much higher levels of reporting emotional difficulty, physical difficulty, financial difficulty, and inability to participate in other activities that they value (Wolff et al., 2016).

SOURCE: Judy Kasper, from NSOC in 2017.
Kasper explained that a lot can be learned from having perspectives from both care recipients and caregivers available in these data. Caregiver measurements include activities undertaken in the caregiving role; the level of effort involved in caregiving (which is often measured in terms of hours); the effect of caregiving on other activities such as work, family responsibilities; and other activities important to caregivers. The data also highlight the effect of caregiving on a person’s overall subjective well-being.
Kasper highlighted some elements of research that might be clarified. She noted that if scientists want to measure the effects of caregiving around areas of well-being, they need to be able to define caregiving activities and characteristics of the recipients. If they want to assess well-being, the perspective of the caregiver is of critical importance. Researchers also need to be able to clearly define and link who the caregivers are. An important aspect of these studies is also the characterization of the effects of caregiving across a range of outcomes. Kasper concluded that the upcoming availability of data for both NHATS and NSOC from 2020–2023 provides unique opportunities to understand changes over time in caregiving, as well as caregiving at the end of life.
Scott Beach, University of Pittsburgh, began his presentation on measures of objective burden by explaining that the caregiver burden does not end when a loved one is placed in a facility; caregivers are still monitoring and worrying about their loved one. Data show that once the care recipient passes away, there is sometimes a relief, and the depression felt in relation to the burden of caregiving while the person is alive lessens or evens out (Schulz et al., 2003).
Beach explained that in the literature, objective burden is measured through the number and types of tasks a caregiver is helping with, the hours per week spent caregiving, the level of heavy and physical care, problem behaviors of the patient, and the psychological symptoms of dementia. Subjective burden, on the other hand, measures personal strain and role strain through measurements like the Zarit Burden Interview.
Beach noted that of importance, and independent of objective burden indicators such as the burden of tasks, is the degree to which the caregiver believes that the care recipient is suffering psychologically, physically, and existentially. A grimacing care recipient, for example, has been found to be a significant independent predictor of things such as depression and increased antidepressive use (Schulz et al., 2010). Beach added that caregiver compassion is also an important lens through which to view this data. Perceived suffering of the care recipient could lead to intrusive thoughts, for example, where the caregiver cannot stop thinking about the care recipient. Beach and his team have extended their work into compassion and intrusive thoughts, and suggested a potential target for future interventions could be to measure the extent to which care recipient behavior is upsetting the caregiver and to help caregivers learn to deal with the suffering of the care recipient.
Of note, Beach stated, is the importance not only of recognizing the negative effects of caregiving, but exploring the positive. Recent review of literature about positive aspects of caregiving indicate that caregivers do report positive dimensions, such as a sense of personal accomplishment, an increase in cohesion, sense of personal growth. Beach described a concept of caregiver ambivalence, wherein caregivers experience both positive and negative effects, where their experience can be great one day and then very frustrating the next. Because of this, the quality of care can vary, Beach noted. He suggested that scholars might think of a broader range for interventions to enhance the positive and not just reduce the negative aspects of providing care.
Beach highlighted findings that care recipients who are reporting more unmet needs have caregivers that are stressed or strained (Beach et al., 2017). This study found that, despite receiving more than 100 hours a month of care by caregivers (skin care, tending wounds, etc.), nearly 30 percent of care recipients reported two or more unmet needs, which indicates a link between care burden and quality of care provided.
Beach concluded by noting that the current state of caregiving research is cross-sectional, and conducted with nonprobability clinic-based samples of caregivers with no care recipient; because of this, he recommends more longitudinal dyadic studies around mixed methods assessments of caregiver tasks and burdens. He explained that there should be a shift from efficacy to implementation research around interventions. He also noted the lack of research on caregiver burden as a risk factor for other outcomes and suggested further exploration of the caregiver stress theory that stressed caregivers cannot or will not provide adequate care.
John Richard Pagan, member of the advisory panel to the committee and living with Lewy Body dementia, provided his perspective on the effects of caregiving to round out the panel discussion. In his role as discussant, Pagan began by stating that he has encountered a paucity of discussion about that form of dementia or about early onset of the disease.
Pagan also underscored the importance of nomenclature. He noted that “care partner” is the suggested terminology instead of “caregiver” because providing care is a team effort that involves more than just one person and could include members of the community. He also recommended discontinuing use of the word “demented” and instead describing people with dementia as living with the disease. Pagan further recommended that the word “burden” be defined at the beginning of the report to reduce concern around the stigma attached to that word. He also encouraged the committee to recognize the importance of focusing on what the person living with dementia can do in his or her life and to help determine how to improve transitions after diagnosis.
Discussant Katie Jordan discussed her experience as a thirty-three-year-old whose father is living with dementia. Jordan noted that she is the only person in her friend group caring for a parent living with dementia. She commented that her father is not the burden, the disease is. She added that her dad never said he was forgetting things and did not want to talk about the fact that he was living with dementia, but she questioned if he may not have known he was losing himself. When he was living at home, he had a group of six men and women in various care-taking situations who made things easier on her, but she still had to step in to support him, which caused stress.
Jordan recollected that she has noticed changes in her father as the disease progresses. For example, she has gotten a number of phone calls from him in which he would not know who he was in relation to her but was calling because he knows she helps him. His opportunity to wander dissipated when he went to a nursing home, but Jordan
echoed Beach in observing that the type of stress she experienced changed as she swapped one set of stresses for another. Jordan explained that her father had to be admitted to a psychiatric unit in order to be medicated properly, and she relayed that she has seen neurologists and general practitioners hesitant to prescribe medicines to those living with dementia-related behavioral issues. She described how stressful that process was and said that medicating those with dementia is a frequent point of conversation in her dementia support group. She suggested that the committee explore the exorbitant cost of care and help support younger-generation caregivers navigate the complicated U.S. health care system.
FINAL REMARKS
Powell concluded the workshop by underscoring the importance of the topics discussed. She noted that there was an incredible amount of data put before the committee, and yet still more is needed. This workshop, she stated, was a step in the right direction in helping the committee to generate a final report to help the National Institute on Aging develop a 10-year research agenda in the behavioral and social sciences as it relates to AD/ADRD.
COMMITTEE ON THE DECADAL SURVEY OF BEHAVIORAL AND SOCIAL SCIENCE RESEARCH ON ALZHEIMER'S DISEASE AND ALZHEIMER'S DISEASE-RELATED DEMENTIAS
Patricia (Tia) Powell (Chair), Montefiore Einstein Center for Bioethics; Karen Cook (Vice Chair), Stanford University; Margarita Alegria, Massachusetts General Hospital and Harvard Medical School; Deborah Blacker, Harvard Medical School and Harvard T.H. Chan School of Public Health; Maria Glymour, University of California, San Francisco; Roee Gutman, Association of American Universities; Mark Hayward, Population Research Center and Population Health Initiative at the University of Texas at Austin; Ruth Katz, LeadingAge; Spero M. Manson, Centers for American Indian and Alaska Native Health in Colorado School of Public Health, University of Colorado, Denver; Terrie E. Moffitt, Duke University and King's College London; Vincent Mor, Brown University and Providence Veterans Administration Medical Center; David Reuben, UCLA Center for Health Sciences; Roland J. Thorpe, Jr., Johns Hopkins Alzheimer's Disease Research Center for Minority Aging and Hopkins Center for Health Disparities Solutions; Rachel Werner, University of Pennsylvania; Kristine Yaffe, University of California, San Francisco; and Julie Zissimopoulos, University of Southern California; Molly Checksfield, study director; Tina Winters, associate program officer.
DISCLAIMER: This Proceeding of a Workshop—in Brief was prepared by Molly Checksfield, rapporteur, as a factual summary of what occurred at the workshop. The statements made are those of the rapporteur or individual meeting participants and do not necessarily represent the views of all meeting participants; the committee; the Board on Behavioral, Cognitive, and Sensory Sciences; or the National Academies of Sciences, Engineering, and Medicine. The committee was responsible only for organizing the public session, identifying the topics, and choosing speakers.
REVIEWERS: To ensure that it meets institutional standards for quality and objectivity, this Proceedings of a Workshop—in Brief was reviewed by Karen Love, Dementia Action Alliance and Katie Maslow, the Gerontological Society of America. Kirsten Sampson Snyder, National Academies of Sciences, Engineering, and Medicine, served as review coordinator.
SPONSORS: The workshop was supported by the National Institutes of Health.
Suggested citation: National Academies of Sciences, Engineering, and Medicine. (2020). Challenging Questions about Epidemiology, Care, and Caregiving for People with Alzheimer's Disease and Related Dementias and Their Families: Proceedings of a Workshop—in Brief. Washington, DC: The National Academies Press. https://doi.org/10.17226/25706.
Division of Behavioral and Social Sciences and Education

Copyright 2020 by the National Academy of Sciences. All rights reserved.












