2
Pivotal Role of Innovations in Tackling Microbial Threats: Lessons from Past Outbreaks
The workshop opened with two keynote addresses from speakers who explored how innovations can be pivotal in changing the course of an outbreak. Ananda Bandyopadhyay, program officer, Bill & Melinda Gates Foundation, described lessons learned from innovations in polio eradication over the past several decades. Jonathan Towner, team lead, disease ecology group, viral special pathogens branch, U.S. Centers for Disease Control and Prevention (CDC), examined the role of innovation on the ground in the fight against the Ebola virus, with a focus on the outbreak in West Africa (2014–2016) and the outbreak in eastern Democratic Republic of the Congo (DRC) that began in 2018.
LESSONS LEARNED FROM INNOVATION IN POLIO ERADICATION
Ananda Bandyopadhyay explored lessons learned from the global effort to achieve polio eradication over the past several decades, with a focus on how innovation can be leveraged in practical, impactful ways. He explained that disease eradication can be defined in different ways, but a simple approach is to define it as a permanent reduction of the worldwide incidence of a given infection to zero (Cochi and Dowdle, 2013). Until and unless the three criteria of zero incidence, permanency, and global scale are satisfied, a disease cannot be categorized as eradicated, he continued. Bandyopadhyay noted that the only human disease that has been eradicated is smallpox (Cochi and Dowdle, 2013). In contrast, polio is an acute viral illness that is passed from person to person, primarily through contact with feces; is highly
subclinical; and although very infectious, it presents with the characteristic signs and symptoms of paralysis in only a small subset of those infected—about 1 in 200 infections (WHO, 2019a). He warned that as long as polio exists somewhere in the world, it is just a plane ride away from the countries or regions that have eliminated polio. As Bandyopadhyay explained, immunization is the only way to prevent the disease, and there is no cure for the disease, while rehabilitative therapy is the only treatment option for people who have already been paralyzed with polio (WHO, 2019a).
Progress in the Eradication of Polio
Bandyopadhyay provided an overview of the substantial progress to date toward the eradication of polio worldwide. A cadre of 20 million volunteers deliver vaccinations that reach around 400 million children each year, resulting in the prevention of paralysis on a large scale of about 18 million people who would have been paralyzed had they not been vaccinated (DFID, 2019). Wild polio cases have declined by 99.9 percent over the past 30 years, from 350,000 cases across 125 countries in 1988 to 33 cases reported in 2018 (WHO, 2019a). When the eradication program started in 1988 with the formation of the Global Polio Eradication Initiative, roughly 1,000 cases of paralysis related to polio were being reported every day, he noted. He attributed the remarkable scale of decrease in the number of paralysis cases to the eradication program, but noted that the effort has yet to achieve eradication, and there is still work to be done.
A challenge in addressing polio is that it is a combination of many diseases from the surveillance, diagnostic, and prevention perspectives, said Bandyopadhyay. Polio has been broadly categorized into the wild or naturally occurring disease type, which is further subcategorized into wild poliovirus types 1, 2, and 3. Wild types 2 and 3 were certified as eradicated in 2015 and 2019, respectively (WHO, 2019a,b). However, wild polio type 1 is still actively circulating, has the highest case-infection ratio, and spreads rapidly (WHO, 2019b). He explained that an additional burden of polio disease is related to the oral polio vaccine (OPV), referred to as OPV-related polio, which is subclassified into two categories. Vaccine-associated paralytic polio myelitis (VAPP) is an aberrant neuroparalytic reaction to the oral live polio vaccine virus in an individual who received the vaccine or was in close contact with the vaccine (WHO, 2019c). The overall risk of VAPP in low- and middle-income countries is about 1 case per 4–5 million OPV doses (Platt et al., 2014). The other subcategory, vaccine-derived polio viruses (VDPVs), are revertant strains of the live OPV virus that are transmitted from one person to another, particularly in settings of very low population immunity (WHO, 2019c). Through serial transmission, the viruses become revertant and neurovirulent. Most VDPVs are circulating (cVDPVs), with type 2 cVDPVs
accounting for about 90 percent of all cVDPVs (WHO, 2019c). He explained that cVDPVs are essential public health threats because they revert and become transmissible from person to person and can also cause paralysis.
The endgame phase of polio eradication requires taking into account these different subcategories of polio, Bandyopadhyay said. Although VAPP and VDPVs are public health threats of major concern, they are rare, so the OPV is still the mainstay of interrupting person-to-person transmission in high-risk settings. However, in populations with sustained low levels of population immunity, OPV strains can lead to paralysis or transmission. A balance needs to be struck between these two considerations, he said.1
Innovations in Polio Surveillance, Diagnostics, and Strategic Approaches
Bandyopadhyay explained that because polio is a highly subclinical disease, surveillance is critical. He explained that the overall purpose of polio surveillance is to detect in a timely manner any circulation of polio viruses in any part of the world. The Bill & Melinda Gates Foundation polio program closely tracks the polio virus in around 70 countries by testing paralyzed children and collecting environmental samples to detect virus transmission, he explained. Two forms of polio surveillance are typically used. Acute flaccid paralysis (AFP) surveillance is a clinical syndromic surveillance system whereby reports of sudden-onset paralysis in a specified age group are investigated. Environmental surveillance involves collecting sewage samples from strategically selected areas in different countries to rule out or confirm the existence of polio viruses (Bill & Melinda Gates Foundation, n.d.). He added that multiple countries contribute to polio surveillance formally or informally, with support from an extensive global network of around 150 accredited laboratories. Another objective of polio surveillance is to generate evidence to support the certification process of eradication. Maintaining an adequate quality of surveillance through appropriate methods can help identify prolonged periods without detection and contribute to the documentation around certification of polio-free status.
Many innovations are ongoing or under development in the spectrum of polio surveillance, said Bandyopadhyay. Methods to improve electronic and mobile phone–based reporting systems include the SMS-based Auto-Visual AFP Detection and Reporting project, Integrated Supportive Supervision, and the eSURV electronic surveillance tool. New tools have also been developed for sewage collection and filtration. Innovations in data and analytics include site characterization and sensitivity assessment as well as digital tools, such as digital elevation model databases, geospatial applications, and
___________________
1 This was updated after prepublication release to remove a figure.
facial recognition technology. New direct detection and molecular methods include next-generation sequencing, MinION platforms, and methodologies that would enhance the environmental surveillance technologies. He said that these innovations are facilitating earlier and more sensitive detection, as well as creating integrated methods of detection in which polio surveillance can be combined with surveillance systems like antimicrobial resistance or typhoid surveillance.
Bandyopadhyay emphasized that despite these exciting innovations, real-world polio surveillance on the ground is largely done using traditional methods that require workers to seek out and manually transport patients to care, often across difficult terrain. He explained that to reach and protect more children, the polio program is implementing innovative strategies to achieve the aim of vaccinating every child. For instance, teams are deployed house to house as well as to transit points and health centers to vaccinate children. In areas that are high risk, the programs engage community mobilizers and religious leaders to help ensure that communities will accept the vaccine.
Innovations in Vaccines for Polio Prevention
Bandyopadhyay provided an overview of the spectrum of current innovations in vaccines, prevention, and immunologic interventions (Bandyopadhyay et al., 2015). Research on inactivated polio vaccines (IPVs) is looking to reduce costs, to increase supply, to make the vaccines more immunogenic, and to improve the delivery technologies. Clinical studies are being planned or are under way to evaluate aluminum salts and other adjuvants for IPV, to find novel routes of IPV administration that can be used concomitantly with other vaccines (e.g., disposable jet injectors and microneedle patches), and to create IPV from less infectious or noninfectious sources, such as Sabin and virus-like particles. Antiviral therapies are being developed for people who are immunodeficient and for chronic excretors who shed polio virus for long periods. He explained that antiviral therapies hold promise in interrupting the shedding and reducing the risk of community spread.
A novel genetically stabilized OPV is currently in development to strengthen outbreak control by reducing the risk of VDPVs and VAPP, said Bandyopadhyay. These vaccines are developed with inherent qualities of genetic stability, so they are more stable than the Sabin OPVs and have a lower risk of reverting into neurovirulence. He noted that this is the first new OPV development effort in about 60 years since the licensure of the Sabin vaccine. Human clinical trials began in 2017 and target-population data were generated in 2019. This effort is a large-scale partnership across many organizations and has enabled accelerated development of this vaccine to
counter the threat of VDPVs. A first-in-human study was recently conducted in Belgium, where 30 individuals stayed for approximately 30 days in contained settings with extensive monitoring to understand the characteristics of the new vaccine virus strains (Van Damme et al., 2019a). Preliminary results are all promising, he said (Van Damme et al., 2019b). Researchers found clear evidence of replication in the gut and of immunogenicity. The vaccine is more genetically stable and less neurovirulent compared with the Sabin OPV, which were primary aims of developing the new vaccine.
Looking Forward to Eradicating Polio
Bandyopadhyay remarked that although multiple vaccine options are now a reality—OPV, IPV, and potentially the novel oral polio vaccine—this will not necessarily solve the issue of eradication because the core issue is vaccination, not vaccines. Regardless of a vaccine’s quality, it is not effective while contained in a vial. The difference is made by people who hand carry the vaccines to children, often through seemingly impassible conditions. He lauded these people working on the ground as the champions and sources of real-world, practical innovations that have contributed to the dramatic reduction in polio cases worldwide. Despite the historic progress in reducing polio transmission, Bandyopadhyay remarked that overcoming the remaining challenges to achieve and sustain eradication will depend on concerted global efforts as well as innovation around new surveillance methodologies, vaccine formulations, and delivery technologies. The risk of reintroduction and resurgence of eradicated types of polio will also need to be managed carefully. Finally, he emphasized that the development and deployment of new tools and technologies will need to be tailored based on local need and feasibility in underserved areas—innovative strategies have to be rooted in the ground if they are to reach the last child.
ADVANCING INNOVATION ON THE GROUND IN THE FIGHT AGAINST EBOLA
In his keynote address, Jonathan Towner focused on the role of innovation on the ground in the fight against the Ebola virus. He explained that Ebola virus disease is a severe, often fatal disease with initial symptoms that are nonspecific: fever, severe headache, fatigue, muscle pain, vomiting, diarrhea, and abdominal pain. Unexplained hemorrhage is one of the disease’s main features, but it only occurs in less than half of cases, he clarified. The incubation period of the disease ranges between 2 and 21 days with an average of 8 to 10 days (CDC, 2019a). The Ebola virus is transmitted primarily via direct contact through broken skin or unprotected mucous membranes with blood or body fluids, including but not limited to urine, saliva, sweat,
feces, vomit, semen, and breast milk (CDC, 2019a). It can also be spread through contaminated objects, such as needles and syringes, as well as through infected animals, including apes, monkeys, and possibly bats (CDC, 2019a). Follow-up studies of survivors of the 2014 outbreak in West Africa show that the virus can persist in semen for long periods after recovery (Crozier, 2016). Historically, case fatality rates from Ebola range from to 25 to 90 percent (WHO, 2020).
Origin and Ecology of Ebola Virus
Towner explained that there are six different species of Ebola virus in the genus Ebolavirus within the family of filoviruses. The Zaire ebolavirus was the cause of the outbreak in West Africa, but three other species are known to have caused human disease: Sudan ebolavirus, Tai Forest ebolavirus, and Bundibugyo ebolavirus. The Reston ebolavirus and the Bombali ebolavirus are not known to cause disease in humans at this point, he said. The first filovirus, discovered in 1967, was the Marburgvirus species, which causes high-fatality disease like the Zaire ebolavirus and has been responsible for several relatively large outbreaks. The origin of the Ebola virus remains poorly understood, he added. Various spillover events have been linked to contact with infected nonhuman primates, but the original source is thought to be the bat, which is supported by the discovery of the Bombali ebolavirus in Sierra Leone, Guinea, and Kenya. Further support comes from findings that the Egyptian fruit bat (Egyptian rousette) is the natural reservoir for at least 25 different Marburgvirus isolates (Amman et al., 2012; Swanepoel et al., 2007; Towner et al., 2009).
Epidemiology of Ebola Virus Outbreaks
Towner said that the majority of filovirus outbreaks have occurred in the equatorial region of Central Africa, including the 2014 outbreak in West Africa at the nexus of Guinea, Liberia, and Sierra Leone. The first documented outbreak of Ebola virus occurred in 1976 and regular outbreaks have occurred since, including an ongoing outbreak in the eastern part of DRC that is the second largest ever recorded (CDC, 2019b). He explained that the current outbreak in DRC began in July 2018 and was ongoing as of December 2019, meaning that it has persisted much longer than typical outbreaks, which tended to last no longer than 3 months. The epidemic curve of the DRC outbreak also differs from the 2014 outbreak in West Africa; although the latter was also lengthy in comparison to previous outbreaks, it was also more explosive than the DRC outbreak in terms of the number of cases (see Figure 2-1). As a result, the West Africa outbreak tested the public
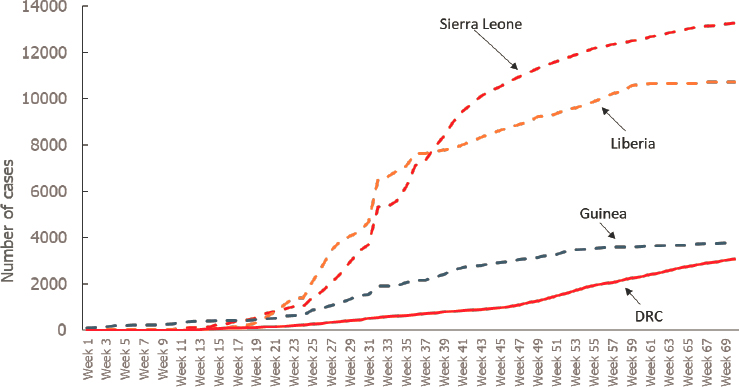
NOTE: DRC = Democratic Republic of the Congo.
SOURCES: Towner presentation, December 4, 2019; data from the U.S. Centers for Disease Control and Prevention Viral Special Pathogens Branch Emergency Operation Center for Ebola Epidemic Team.
health infrastructure of the affected countries and led the United States and Europe’s exportation of multiple clinicians and health care workers for the first time. He noted that 900 of the total 28,000 cases during that outbreak were health care workers.
Progress in Diagnostics, Genomics, and Case Investigations for Ebola Virus
Towner outlined progress in diagnostics, genomics, and case investigations for Ebola virus disease. The classic response to Ebola focuses on stopping human-to-human transmission and isolating infected patients, as well as contact tracing and patient management. Aggressive infection control practices in homes and health care settings, safe burial practices, community engagement, and good data management are also major components of Ebola outbreak control. He explained that the mobile laboratory emerged as the hub for polymerase chain reaction (PCR) diagnostics during the outbreak in Gulu, Uganda, in 2000. Until the West Africa outbreak, the Gulu outbreak of 425 cases had been the largest on record. CDC fielded a team to St. Mary’s Lacor Hospital in Gulu and established a laboratory that con-
sisted of the antigen capture and ELISA2 methods (immunoglobulin G and immunoglobulin M), he continued. These methods had been used before, but they had never been done in PCR for acute-case diagnostics that were focused on finding infected people as quickly as possible. This was done singly in phenol extractions, nested PCR gel boxes, and some marine gels, which are now antiquated technologies.
Not long after that outbreak ended in 2001, there was a major push to develop higher-throughput platforms for detecting potential bioterrorism agents, with Ebola, Marburg, and filoviruses as high priorities. This drove the development of high-throughput RNA extraction and Ebola detection through the titration of Ebola Zaire in whole blood over a 6-log range, which were later used during the Marburg virus outbreak in Angola in 2005—the first time these high-throughput platforms were used in the field. He noted that this response also included the first widespread use of oral swabs for detecting Ebola or filovirus patients, albeit with some controversy.
Towner explained that the 2014 West Africa Ebola virus outbreak spurred an all-hands-on-deck approach to field diagnostics, with laboratories from different countries fielding different PCR methodologies. CDC helped provide some of the high-throughput testing capability to a South African laboratory at Lakka in Freetown, Sierra Leone. CDC also fielded a “hot” laboratory in Bo, Sierra Leone, that was very spartan but highly effective. The Bo laboratory converted a small house into a laboratory with 96-well MagMAX traction platforms and a Biorad 96 real-time PCR machine, which was resistant to the brownouts that occurred frequently.
Ultimately, the Bo laboratory remained operational for more than 400 days, supported by 28 teams of personnel from 17 different branches throughout CDC. They processed more than 27,000 specimens over that period, sometimes processing 150–200 samples per day. He noted that a limiting step was assessing the data from case investigation forms and trying to read illegible handwriting, which monopolized time that could have been spent on diagnostic testing. The diagnostic testing included more than 500 semen specimens to assess the viral persistence in male survivors as well as samples from the Ebola vaccine trials that were ongoing at the time.
Field Diagnostic Laboratory Challenges in the West Africa Outbreak
Towner described some of the challenges they encountered while running the field diagnostic laboratory. Some issues related to the types of specimens and sample transport, which had to be managed in impromptu and ad hoc ways—from motorbikes to helicopters—in the face of the large
___________________
2 The enzyme-linked immunosorbent assay (ELISA) is a commonly used immunological assay used to measure antigens, proteins, and antibodies in biological samples.
and dynamic West Africa outbreak. There were additional challenges related to the assays. Many different real-time PCR assays were being used across multiple laboratory networks, which necessitated attempts to standardize not only assays but also quality-control proficiency panels to identify poor performers. CDC distributed panels in Sierra Leone and Guinea and found that two of the six laboratories in Sierra Leone had 10 percent incorrect results, which enabled them to implement improvements.
Different assays also had implications for different cutoffs and interpretations in terms of criteria for releasing patients from overcrowded Ebola treatment units (ETUs). He said that health care workers were forced to make difficult decisions in certain circumstances: for example, setting criteria for release that a patient had to be clinically well and PCR negative or have a cycle threshold value of less than 35 if the ETU was at capacity and beds were at a premium. To create some correlation and decrease the risk of false positives and false negatives, ETUs developed a two-target Ebola assay and used cell RNA (β 2M or RNaseP) PCR controls. He noted that this turned out to be useful in the deployment of Ebola glycoprotein expressing vaccines by providing the capacity to differentiate between vaccinated and truly infected individuals.
Use of GeneXpert in the Democratic Republic of the Congo Outbreak
GeneXpert (Cepheid) is a cartridge-based platform that has now been implemented widely in the current outbreak in eastern DRC and seems to be working well, said Towner (Pettitt et al., 2017; Raftery et al., 2018; Semper et al., 2016; van Vuren et al., 2016). It is a very sensitive real-time assay with a turnaround time of 98 minutes; it accepts whole-blood or buccal swab specimens. The cartridge component makes disposal and management of the sample and remnants more manageable than other platforms. More than 120,000 field tests had been performed in DRC as of November 2019, said Towner. A caveat with GeneXpert, Towner continued, is that the only commercially available filovirus assay is for Ebola Zaire, but suggested that assays for other filoviruses will be developed.
Using Next-Generation Sequencing to Fight Ebola
Towner remarked that virus whole-genome sequencing is now possible in the field with rapid turnaround time. This next-generation sequencing (NGS) was a valuable innovation in fighting Ebola in West Africa in the later stages of the outbreak when researchers were dealing with unexplained clusters that emerged separately from any known ongoing transmission (Diallo et al., 2016; Goldstein et al., 2018; Towner et al., 2008). For example, molecular field epidemiology conducted using MinION, a handheld NGS platform, enabled researchers to have full-length genomes within 24 hours
to investigate an unexplained cluster in Guinea. This genetic fingerprint of the virus was used to determine that it was a case of sexual transmission originating from a male survivor who was still shedding infectious virus 500 days after convalescence. Towner explained that this technology saves resources by narrowing down epidemiological investigations and establishing whether a virus is newly introduced, a spillover, or part of a previously unknown lengthy chain of transmission.
NGS has also been used effectively in pathogen discovery, for both the Bundibugyo ebolavirus in 2007 in Uganda and the Bombali ebolavirus in 2017 in Sierra Leone. NGS enables rapid whole-genome genetic characterization that can assess how well current diagnostic assays will be able to perform against a newly emergent virus. Knowledge of the full-length genome can also be used to rescue and test viruses when clinical samples are not available, or if samples are available but people are reluctant to handle or transport them, he said. To test truant antivirals or evaluate how well the current diagnostic assays work, the virus can be rescued from an infectious clone. A recent study tested diagnostic assays with a known mutation in a circulating virus as well as testing potential therapeutic monoclonal antibodies (McMullan et al., 2019).
Innovation in Case Detection
Simple innovations can also be highly effective, said Towner. For instance, a mobile case investigation app—the Ebola Exposure Window Calculator—has been developed to support epidemiologists by estimating the period of time during which a person could have been exposed to the Ebola virus. The information provided by this tool can be used in collaboration with other known information about a person to identify potential cases of infection.
Innovation in Capacity Building and Infection Control
Towner emphasized that investing long term in foreign diagnostic laboratory infrastructure can dramatically reduce the size, duration, and cost of outbreaks of emerging infectious diseases. Since 2010, CDC has invested in the Uganda Virus Research Institute to establish and maintain an enhanced comprehensive surveillance and diagnostics program for viral hemorrhagic fevers (CDC, 2019c). The investment has had a substantial effect by diminishing the scope of outbreaks, decreasing the number of cases, and reducing the time it takes to identify the agent of an outbreak. Towner noted that more than 1,000 health care workers have been infected with Ebola virus during the West Africa and DRC outbreaks. To improve infection control and protect health care workers, CDC and other agencies developed a train-
ing course for workers going into the field (Narra et al., 2017). The course consists of a didactic component as well as practical, hands-on training conducted in full protective equipment to simulate activities such as patient care, waste disposal, and disinfection.
Innovation in Vaccines and Therapeutics
Innovations in Ebola vaccines and therapeutics are under way and appear to be effective, said Towner. It was first demonstrated in 2000 that Ebola virus disease could be prevented by a vaccine (Sullivan et al., 2000). A new vaccine—a live attenuated recombinant vesicular stomatitis virus—is now being implemented in eastern DRC, with more than 250,000 doses already administered. Initial reports suggest that its efficacy is greater than 97 percent. Towner surmised that because the vaccine was rolled out early on in the DRC outbreak, it may have had a dramatic effect in attenuating the trajectory of the virus compared to the West Africa outbreak.
Another simple but effective innovation was developed in response to the Gates Millennium Challenge—a portable cooler that can keep the vaccine at the necessary temperature of –60 degrees Celsius for 7 days in tropical temperatures, even with repeated entries into the unit (Jusu et al., 2018). This was valuable during vaccine efforts, he said. Towner concluded with optimism that Zaire Ebola may soon be treatable. Two antibody-based cocktails hold promise as therapeutics for Zaire Ebola virus disease, said Towner. Ridgeback Biotherapeutics’s mAb114 and Regeneron Pharmaceuticals’s REGN-EB3 appear to decrease mortality rates by half compared to rates without those treatments (Kupferschmidt, 2019).
DISCUSSION
Peter Daszak, president of EcoHealth Alliance, noted that the current Ebola outbreak in DRC is larger and longer lasting than any previous outbreak in the region. He asked about the likelihood of future outbreaks being long-term, chronic threats or explosive outbreaks that are more similar to the 2014 outbreak in West Africa. Towner replied that one of those issues, the likelihood of spillover, has more to do with interaction with the reservoir. In the West Africa outbreak, the explosion occurred when the virus entered into the major population centers and overwhelmed the system’s ability to respond in a timely way to control the outbreak through contact tracing, isolation, and other activities. It also required the international community to deal with the challenges involved in engaging with three different governments simultaneously.
He suggested that the current outbreak in eastern DRC could have been quelled in 2018 if contact tracing had been conducted effectively, but it was not possible owing to the insecure environment. Dozens of militia groups
operate in the area and at least two ETUs were burned down and attacked, leading many of the international teams to pull out of the area. Cases who are not found cannot be isolated, protracting the outbreak even though the new vaccine is available. He added that fortunately, the population centers in DRC are not as large as Conakry, Freetown, and Monrovia.
Jyoti Joshi, head of the South Asia Center for Disease Dynamics, Economics & Policy, commented on the differences between the two diseases: polio being a slow and persistent threat, while Ebola was a sudden and huge threat. The innovation in the response to Ebola tended to involve laboratories reaching out to people rather than vice versa. Both cases demonstrate that innovations need to synergize with local context or reach will be limited. Joshi asked about how to implement innovations in a context-appropriate way. Bandyopadhyay remarked that each context is different, and each approach should be evaluated and adapted as it is implemented. This involves understanding the geopolitical context, engaging local communities, and ensuring local ownership of programs.
He said that the current concerted, integrated drive toward immunization has benefited the polio program, particularly in endemic areas. In terms of diagnostics, he suggested relying on culture-based methods because they are highly specific and sensitive for diseases such as polio, in which a single case can trigger an outbreak. However, as the number of polio cases continues to decrease, interest is growing in direct detection. Pilots are already under way to explore this molecular method of detection and to bring the laboratory closer to the field, but the advantages of these innovations need to be balanced with the need for specificity and sensitivity in planning for outbreak response.
James Lawler, director, International Programs and Innovation, Global Center for Health Security, University of Nebraska, highlighted the crosscutting issue of misinformation campaigns that contributed to the outbreak of polio in northern Nigeria, hindered Ebola outbreak control efforts in eastern DRC, and is currently driving measles outbreaks in Europe and the United States. Towner commented that community engagement to develop “street cred” and to build trust is critical for the success of outbreak control, particularly in settings such as eastern DRC.
Greg Armstrong, director, Office of Advanced Molecular Detection Program, CDC, asked if the innovation of emergency operations centers has been helpful in improving timeliness and completeness of response, bringing partners together for better coordination, and giving governments a degree of engagement and control that they would not otherwise have. Bandyopadhyay replied that emergency operations centers have strengthened polio response efforts, especially in high-risk settings and in situations where response time is critical. They have also been used as platforms to
build infrastructure and capacity to respond to other pathogens and threats, such as Ebola.
Cristina Cassetti, deputy division director, Division of Microbiology and Infectious Diseases, National Institute of Allergy and Infectious Diseases, asked if surveillance for acute flaccid paralysis during the polio eradication campaign also tests for related viruses that can cause paralysis, such as the enterovirus EV-D68. Bandyopadhyay replied that surveillance for acute flaccid paralysis partly involves clinical surveillance, with reporting based on symptoms of sudden-onset flaccidity in people aged less than 15 years, but it does include laboratory testing for nonpolio enteroviruses. Testing is categorized by whether a sample has polio, and if so, intratypic differentiation is conducted to classify whether it is wild or vaccine derived, which serotype it is, and if it contains nonpolio enteroviruses. They also do an ongoing assessment of nonpolio enterovirus prevalence in some of the settings, he added.
Keiji Fukuda, director and clinical professor, The University of Hong Kong School of Public Health, asked about the place of vaccine-associated polio phenomena within the overall goal of eradication. He suggested that political will and financing may dissipate for these polio campaigns. Bandyopadhyay emphasized the success of vaccination campaigns in reducing the number of wild cases outside of Afghanistan and Pakistan, although there are concerns that transmission is ongoing in those two countries. He reiterated that there is a conundrum around using the OPV. It is essential to interrupt transmission in the settings where a live vaccine is needed to provide intestinal immunity, but it is associated with risk in settings with consistently low immunization coverage.
He also expressed concern about the expanding nature of VDPVs, 90 percent of which are from the type 2 component of the virus, which is the component of the OPV that has been withdrawn from routine immunization use. He highlighted the global vulnerability around type 2 vaccine transmission. Outbreak response will need to be intensified using the available tools that can disrupt this transmission; but, at the same time, innovations are needed to develop improved vaccines with greater genetic stability. He added that a wild type 3 vaccine-derived outbreak would be concerning, but there are tools available to interrupt it if used effectively. Alternative tools are also under development that could solve the issue of seeding more vaccine-derived outbreaks, such as the novel OPV that is more genetically stable.
This page intentionally left blank.














