8
Maternal Nutrient Intake and Early-Life Programming
A mother’s intake from the prenatal through postnatal periods influences programming of the growth and development of her child. The mechanisms by which this takes place involve a complex interplay of systems, including the microbiome, metabolome, placenta, and epigenetic changes. The seventh session of the workshop, moderated by Emily Oken of the Harvard Medical School and the Harvard Pilgrim Health Care Institute, explored the evidence on these systems and the implications of maternal nutrition on early-life programming. Highlights from the session presentation and panel discussion are presented in Box 8-1.
THE ROLE OF THE PLACENTA IN DELIVERING NUTRIENTS AND IN DEVELOPMENTAL PROGRAMMING
“I firmly believe the placenta is the director of pregnancy. It’s in charge,” said Leslie Myatt, professor of obstetrics and gynecology, director of perinatal research, and Endowed Professor in the Bob and Charlee Moore Institute for Nutrition & Wellness at the Oregon Health & Science University, Portland, to set the stage for his remarks. The placenta not only transfers nutrients to the infant and removes waste products, it also communicates with the mother and fetus, he explained. Over the course of his presentation, Myatt reviewed advances in evidence on developmental programming, roles of the placenta, sexual dimorphism in fetal outcomes, placental growth and development, and factors that affect the placenta and developmental programming.
Advances in Evidence on Developmental Programming
The rise in obesity and diabetes in the population over the past 30 years affects pregnancy outcomes. The bulk of human existence has faced nutritional deficits. Accordingly, the placenta and fetus adapted to develop mechanisms to handle these deficits. In modern times, however, there is an excess of nutrients, explained Myatt.
Originally called the Barker Hypothesis, the Developmental Origins of Health and Disease (DOHaD) suggests that risk for chronic diseases—particularly cardiovascular disease, diabetes, metabolic syndrome, and neurobehavioral outcomes—are determined in utero. Myatt remarked that there can be differential effects to the same insult, and that male fetuses tend to be more severely affected than female fetuses. Differences in the type and level of nutrition, physical factors, environmental contaminants, and other factors affect fetal growth, development, and programming. These factors tend to operate through epigenetic mechanisms. The resulting DNA methylation and histone modification affects gene expression and transcription.
Roles of the Placenta
Myatt described a variety of roles the placenta plays, in addition to nutrient, oxygen, and waste exchange. The placenta serves as an immune barrier between mother and fetus and helps prevent the mother from rejecting the semiallogenic fetus. Notably, the placenta makes hormones and other metabolites that enter maternal circulation. These chemical signals can alter the amount of nutrients in maternal circulation thus changing the availability of glucose, fatty acids, and amino acids for transport to the fetus. The placenta metabolizes a substantial portion of the substrates to generate adenosine triphosphate for its own processes. A current line of investigation is exploring the role of placental metabolites (e.g., serotonin) in fetal development.
Characterizing the placenta as “selfish,” Myatt explained that the placenta regulates the supply and composition of nutrients coming from the mother, and provides the fetus what it does not consume itself. The placenta is four to six times more metabolically active per unit weight than the fetus. At term, the placenta is about one-sixth the size of the fetus and yet consumes approximately half of the fuel and oxygen delivered to the uterus. One-third of the oxygen consumed by the placenta is used for the de novo generation of peptides, while another third is used to maintain the cation gradient needed for transmembrane transport. The placenta uses different mechanisms for transport, depending on the substrate, including flow-mediated diffusion, active transport, facilitated transport, and endocytosis and exocytosis.
The placenta controls the transfer of nutrients and has adaptive responses to changes in supply. It plays a role in regulating maternal metabolism so that substrates are released for transport (i.e., the supply of nutrients), and it appears to be involved in fetal growth and development (i.e., the demand for nutrients). The placenta can also change the flow of nutrients to the fetus by altering the expression of various transporters. With the ability to store certain nutrients, the placenta may also serve as a buffer. From an evolutionary standpoint, Myatt suggested that the placenta likely developed adaptive mechanisms in response to altered nutrient supplies. These mechanisms appear to include altering regulatory signals and promoting epigenetic changes.
Sexual Dimorphism in Fetal Outcomes
“It’s good to be a girl in utero … conversely it’s not so good to be a boy in utero,” said Myatt to preface his discussion of sexual dimorphism in fetal outcomes. Animal studies have shown that changing maternal nutrition can alter the ratio of male-to-female fetuses. Feeding mice omega-3
polyunsaturated fatty acids can increase the ratio of male pups born, added Myatt. In humans, female fetuses appear to grow at a rate to optimize survival, whereas male fetuses tend to continue growing throughout gestation. Rates of adverse outcomes—including preterm births, placenta previa, preeclampsia, lagging lung development, macrosomia, and late stillbirths—are higher among pregnancies with male fetuses. Male fetuses also appear to affect maternal beta-cell function, increasing risk of gestational diabetes. The sexual dimorphism also occurs in the placenta (see Box 8-2).
Placental Growth and Development
Placental growth, structure, and function is dynamic and increases in substrate transport efficiency over the course of gestation. There are several critical periods of placental development, and the placenta appears to adapt to insults in an effort to keep the fetus alive. The same insult occurring at different times of gestation can exert different effects. Although difficult to study in humans, this concept has been demonstrated with hyperglycemia. Women with insulin-dependent diabetes who entered pregnancy with elevated blood glucose concentrations tended to have large-for-gestational-age infants and had alterations to placental glucose transport. In contrast, women who developed gestational diabetes and experienced hyperglycemia later in pregnancy did not experience the placental changes.
The epigenome is responsive to changes in the timing and availability of nutrients. For instance, DNA methylation was found to be different depending on whether a woman was in her first or third trimester dur-
ing the Dutch Hunger Winter.1 The tricarboxylic acid cycle is affected by various nutritional factors, and the resulting epigenetic changes can have implications for placental gene expression, placental function, and developmental programming (see Figure 8-1). As the placenta is fetal tissue, it is likely that epigenetic changes are also taking place in the fetus, but the extent of this is currently unknown, added Myatt.
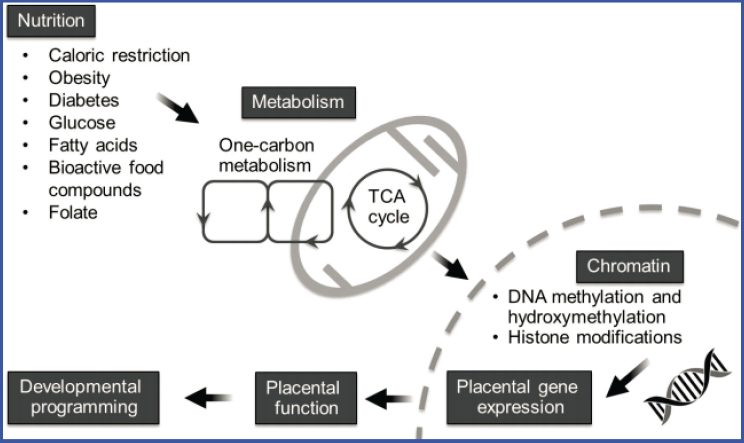
NOTE: TCA = tricarboxylic acid.
SOURCES: Presented by Leslie Myatt. Reprinted from Myatt, L. and Mitsuya, K. 2015. Epigenetics and the placenta: Impact of maternal nutrition. In Human placental trophoblasts, edited by Asim K. Duttaroy and Sanjay Basak: CRC Press, pp. 37–52.
Factors That Affect the Placenta and Early-Life Programming
Nutrients such as betaine, choline, folate, vitamin B6, and vitamin B12, which are involved in one-carbon metabolism, affect epigenetic modifications. A host of other nutrients and dietary factors also appear to play a role. Butyrate and curcumin, for instance, affect histone conformation. More broadly, various states and conditions can also have implications for
___________________
1 The Dutch Hunger Winter was a famine that took place in the Netherlands during the winter of 1944–1945.
the placenta and early-life programing, including obesity and gestational diabetes, oxidative stress, and substrate use, said Myatt.
Obesity and Gestational Diabetes
Obesity and gestational diabetes both have implications for early-life programming. For instance, obesity alters placental DNA methylation. A recent analysis found a 15 percent increase in DNA methylation and a corresponding decrease in hydroxymethylation in the placentas of women with obesity, as compared to lean women (Mitsuya et al., 2017). These changes often translated into differences in gene expression. Many of the 262 genes affected were found on chromosomes 17 and 19. Increasing maternal adiposity can also decrease mitochondrial respiration, which is potentially related to increased risk of stillbirth, noted Myatt. Mitochondrial respiration is also decreased in women who develop gestational diabetes. In an effort to generate energy, the placenta adapts by increasing in size and increasing glycolysis.
Oxidative Stress
Pregnancy is a state of elevated oxidative stress, with the placenta being a major source of reactive oxygen species. Pathological pregnancies, such as those with obesity and gestational diabetes, further increase placental oxidative stress and can deplete antioxidant defenses, often in a sexually dimorphic manner. The highest antioxidant defenses are found in placentas of male fetuses carried by lean women, whereas the lowest antioxidant defenses are found in the placentas of male fetuses carried by women with obesity (Evans and Myatt, 2017). There are different types of antioxidant defenses, including extracellular antioxidants and intracellular reducing elements, such as superoxide dismutase and glutathione peroxidase. Mechanisms to handle reactive oxygen species within a cell are likely context and location specific. “Perhaps it is not surprising, with our failure of things like vitamin C and vitamin E, to actually be able to effectively reduce oxidative stress if they are actually not reaching the site of reactive oxygen species production,” said Myatt.
Substrate Use
Glucose, fatty acids, and glutamine are substrates used in placental mitochondrial respiration. Different cell types prefer different substrates, but they have metabolic flexibility to switch between substrates, should one become limited. In lean women, the three substrates are used in approximately equal proportions and there are differences between male and female
fetuses. Women who have hyperglycemia and hyperlipidemia stemming from obesity and gestational diabetes have a greater reliance on glucose and fatty acids and decreased metabolic flexibility but only among women carrying male fetuses (Wang et al., 2019).
Docosahexaenoic acid (DHA) and arachidonic acid are two key fatty acids that are essential for brain development, particularly as the brain undergoes dramatic growth in the third trimester of pregnancy and in early childhood. Male infants of women supplemented with these brain-specific fatty acids during pregnancy had increased brain size (Ogundipe et al., 2018). Expression of different fatty acid oxidation enzymes in the placenta shows sexually dimorphic differences. In women with obesity, there is an increase in expression of enzymes specific to short-chain fatty acids among male placentas, whereas female placentas show a preference for long-chain fatty acids. The male placentas of women who developed gestational diabetes also showed a decrease in triglycerides containing DHA. These differences result in more medium- and short-chain fatty acids enzymes in the placentas for male fetuses. Aligned with these findings, O’Neill et al. (2018) reported the women with diabetes had higher concentrations of medium chain fatty acids in their amniotic fluid, and more long-chain fatty acids if the fetus was female.
Summarizing Key Points
Myatt ended his remarks by summarizing select key points from his presentation. He emphasized that the placenta influences maternal metabolism to support fetal growth and development. The placenta is responsive to changes in nutrient supply, and these adaptations may lead to fetal programming via epigenetic mechanisms. Given the responsiveness of the placenta, Myatt suggested that, “at delivery, the placenta can be used as a diary of fetal exposure.” Finally, he underscored that there is a sexual dimorphism in placental function and effect that should be considered and further studied. It is currently unknown if interfering with the adaptive effects is potentially harmful or if interventions are addressing a primary dysfunction of the placenta.
PANEL DISCUSSION
After Myatt’s presentation, Kjersti Aagaard of the Baylor College of Medicine and Michael Goran of Children’s Hospital Los Angeles and the University of Southern California joined him for a panel discussion. Moderator Oken had the panelists consider upcoming, applied, and future innovations and considerations related to each of their work.
Oken asked the panelists to first share their ideas on what they thought were clear next-step, highest-yield interventions. From his perspective,
Goran thought sugar is a dietary culprit that should be addressed, given its caloric, metabolic, and developmental effects. He admitted that investigating different sugar exposures would be complicated, time-consuming, and costly, but he emphasized that sugar is a dietary factor frequently found to be related to maternal and infant outcomes. Reducing sugar intake would shift dietary intakes toward a more wholesome diet, suggested Goran, who cautioned the focus should be reducing the craving for sweetness rather than displacing intake with artificial sweeteners.
Aagaard stated that there is some data to suggest the periconception period is a key time for intervention, but acknowledged that this approach would be difficult to use at a population level, as approximately half of pregnancies are unplanned. Taking a more global view, she thought that current dietary patterns are not sustainable and locally and regionally grown nutritious food should be made more available. She also believed a clear next step would be to improve maternal diets, thereby improving breast milk composition. With approximately half of the maternal population affected by obesity and/or gestational diabetes, Myatt underscored that these two conditions are critically important to address, especially given the implications for the placenta. He noted that placental research is difficult, as samples only become available at term, and thought that being able to get tissue samples earlier could help identify important time points for intervention. Findings from the few studies that have been able to assess placentas earlier in gestation have aligned with animal studies, suggesting changes in the preconception period and early gestation could have implications for the entire pregnancy, he added. Given this, Myatt suggested that there should be greater focus on diet and care in the preconception period and that gestational diabetes should be screened for sooner than 26 weeks gestation.
In her second question, Oken asked if any of the research the panelists presented is ready for clinical application. From Myatt’s perspective, he thought the amount of testing a woman undergoes during pregnancy is fairly minimal and suggested the time is right for more testing of placental health. He mentioned that, as a result of efforts like the Human Placenta Project, two biomarkers of placental health (PlGF and sFlt-1) have recently entered clinical practice in the United Kingdom. Used as a screening in the third trimester, the biomarkers identify women at increased risk for adverse outcomes in the last portion of their pregnancies. Myatt indicated additional biomarkers are emerging but need improvements in their sensitivity and specificity.
Aagaard thought that, in general, women undergo a number of screenings during pregnancy. She was concerned about the undue stress positive screening tests can cause, especially when there is no readily identifiable therapeutic but a known adverse outcome in the end. She suggested any
screening that that is implemented should be tied to clinical management that would lead to a change or improvement in an outcome. Aagaard also raised the issues of cost and patient burden related to additional screenings. Shifting her remarks to the microbiome and epigenomics, Aagaard did not think either was ready for clinical applications at this time. She stated that she supported the American College of Obstetricians and Gynecologists statement that inoculation with vaginal swabs is reserved for the setting of a clinical trial and research purposes only, and cautioned that the risks of such transfers are not fully understood nor has benefit been demonstrated.
From his perspective, Goran thought oligosaccharides were not ready for clinical applications yet. The existing clinical trials on oligosaccharides have limitations, including study design and short durations, he indicated. Given the state of the science, Goran suggested high-quality trials supplementing infant formulas and supplementing lactating women were warranted. “We’re talking about turning breast milk into a more individual, precision-type of nutrition that can be individualized for the infant to offset any problems such as environmental toxins,” he said. Goran emphasized the importance of diet quality, to shift intakes away from sugar, environmental toxins, and contaminants. He remarked, “The question is how to get people there and how to modify the food system to make that more broadly available to more people.”
To conclude the panel discussion, Oken asked for ideas on where the research on these topics should go. Myatt indicated there should be a movement toward determining individualized, optimal nutrition. From a practical perspective, Aagaard suggested that synchronizing and linking well-child and postnatal visits in the same time and space could be incredibly informative. She also thought that there is a need for locally sourced diets specific to each region of the world across seasons, in an effort to reduce the need for supplementation. Until the information is digestible, however, personalized nutrition will not be fully embraced, she suggested. Myatt added, “We need to get the message across that pregnancy and lactation is an important time of life in development and [give] it the attention and funding it needs.” To make strides toward personalized nutrition, research needs to tease apart differences, he remarked, adding that a common set of data across different populations could facilitate much more rapid comparisons.
QUESTIONS FROM THE AUDIENCE
After the panel discussion, Oken open the discussion to members of the audience. Questions were raised about collaboration and futures studies, evidence related to the placenta, and dietary quality and components.
Collaboration and Future Studies
A member of the webinar viewing audience wondered to what extent toxicologists and nutritionists collaborate, given shared interests in the role of environmental contaminants on fetal programming. Aagaard acknowledged the interplay among nutrition, environmental contaminants, and fetal programming. Even nutritious food can be exposed to chemical toxicants depending on how and where it is grown. Proper nutrition helps to ensure the right microbes are present so they can detoxify these chemical toxicants, she explained. Agreeing that there is a connection, Goran said he personally has had such cross-disciplinary collaborations, but thought more interdisciplinary work is needed.
Referring to remarks made on the continuum of maternal health and nutrition, Anna Maria Siega-Riz of the University of Massachusetts Amherst asked for ideas of how best to effectively study long-term outcomes. Oken said the Environmental influences on Child Health Outcomes (ECHO) Program is one example in the United States. Building on this, Aagaard added that the ECHO Program brings together data from different studies to help fill in gaps. She suggested that artificial intelligence and the use of electronic health records could also be valuable. Aagaard viewed the 5-year timeline for National Institutes of Health funding as a limitation to long-term endeavors, and she thought there was a need to develop funding mechanisms to support lengthy longitudinal cohorts.
Evidence Related to the Placenta
Siega-Riz wanted to know at what point the placenta starts to play a more prominent role in delivering nutrients to the fetus. Myatt explained that blood flow in the placental intervillious space begins at about 12 weeks gestation and increases over the course of pregnancy. He noted both the fetus and the surface area of the placenta undergo exponential growth during the third trimester. On the topic of fetal and placental growth, Aagaard offered that there are interesting examples in the animal literature. For instance, marmosets are New World nonhuman primates that do not have singleton births, but rather birth twins. Approximately 70–80 of the 135 days of gestation are spent investing in placentation, which is the opposite of what humans do, she explained. Marsupials can make a choice between gestations and lactation, depending on nutrient availability. “Decisions around investments in placentation and investments in fetal growth are ones that different species have come up with different solutions for,” said Aagaard.
A webcast audience member asked Myatt about the underpinning for the sexual dimorphic growth in utero. There is an argument that, from an evolutionary standpoint, males want to grow larger so they can compete for
a mate even though it places them in a stressful situation, suggested Myatt. “Boys live dangerously in the womb,” he said, in a quote he attributed to David Barker.
Dietary Quality and Components
With respect to comments made earlier in the workshop, a webcast audience member asked about the relationship between fiber and the microbiome. Indicating fiber serves a beneficial role, Aagaard stated that to her knowledge there are no pregnancy-specific recommendations for fiber intake related to supporting the microbiome.
A member of the webcast audience questioned whether quality of fat should be prioritized over quantity of total fat. Noting that certain fats are critical for brain growth and development, Myatt indicated that there are mechanisms to preferentially transport and use different types of fat. More research is needed to determine the ideal mix of different types of fat, he said. Goran added that quality should also be considered for the other macronutrients, as well.
__________________
This page intentionally left blank.












