Approaches for Improving Army Medical Infrastructure Planning
DARPA’S APPROACH TO MILITARY MEDICAL INNOVATION
Dr. Geoff Ling (Colonel, U.S. Army, retired), currently CEO for NED Biosystems, outlined his experiences as DARPA’s Biological Technologies Office founder. He described DARPA’s approach to military medical innovation. DoD expects the unexpected, and DARPA is there to solve problems by saying “Yes, not No” and by “doing science to build capability.” He identified the following key risk reduction elements: pursue an idea if it makes sense; sufficiently resource it to succeed, and manage it closely with metrics, milestones, and timelines; if necessary, fail early and fast; and identify, early on, transition and commercialization partners.
Ling described a range of projects. A simple project was an automated ventilator that enabled first responders to accomplish other critical tasks and was procured by the thousands for COVID-19. A complicated project is the Predicting Health and Disease program, which has the goal of keeping the fighting force 100 percent available by identifying sickness early so that the military billet can be filled with mission-ready persons. Humans can exhibit subclinical signs of illness, which can be detected with near certainty days before becoming symptomatically ill from a virus—information that can be used to employ appropriate therapeutics or take administrative actions. The technology is promising because in studies, exposure to disease resulted in changes in hundreds of biomarkers. A small subset (2 to 5 per disease) was used in nonlinear, predictive classifiers for emergence of symptoms of specific diseases. A secondary subset of markers was used to create a classifier indicative for
all three viruses studied. Ling discussed immediately available solutions, with a portable platform and usability with standard laboratory equipment, for detecting the presence of SARS-CoV-2 (severe acute respiratory syndrome) virus even 2 to 4 days prior to typical symptoms and response signatures in presymptomatic patients.
Ling described another innovative project, Pharmacy on Demand (Figure 4). Research began when it was observed that medicines needed to treat service members in Afghanistan were not available but could have been made with a chemistry set. A partnership between Massachusetts Institute of Technology scientists and DARPA resulted in the development of a miniaturized manufacturing unit the size of a household refrigerator. Pharmacy on Demand features proprietary microreactors and continuous-flow synthetic chemical processes to make active pharmaceutical ingredients and final formulated medicines. This capability has the potential of transforming current ways to make drugs, and in the future, it could lead to a stand-alone machine producing multiple drugs on demand, thus reducing rising drug prices and problems associated with U.S. drugs and their ingredients not being made in America. He concluded that medical innovation is about expecting the unexpected and preparing for the unthinkable. Success always favors the prepared.
PERFORMANCE IMPROVEMENT
Russ S. Kotwal (Colonel, U.S. Army, retired), Strategic Projects, DoD Joint Trauma System (JTS), defined performance improvement as “systematic data-guided activity designed to effect delivery of a service (e.g., health care) in near real-time,” and he emphasized that a well-designed system should use performance improvement to help guide and prioritize research. Referring to the cyclical pattern of wartime improvement in medical support followed by postwar performance dips, which represent potential lives lost, he identified the three recurring themes: (1) postwar military reductions falling disproportionately on medical services, (2) failure of medical services to be represented in senior operational planning, and (3) failure to adequately capture lessons learned during a conflict and incorporating them into doctrine and training and into the civilian sector.
Kotwal illustrated how organizations can improve their performance by recounting experiences of the 75th Ranger Regiment. Combat death data showed approximately 8 to 9 out of 10 combat deaths occurred prehospital. TCCC became focused on prehospital guidelines, such as MARCH (Massive hemorrhage control, Airway, Respiratory, Circulation, and Hypothermia) prevention, infection and pain control, documentation, and evacuation. Importantly, tactical leadership then took

ownership of TCCC and included medical training as part of “the big 4 training priorities.” Cultural changes were achieved by having both leaders and subordinates accept ownership and personal accountability and by reinforcing unit cohesion, all aimed at the strategy of eliminating preventable deaths. The importance of documentation was emphasized to provide data for after-action reviews, analyses, and studies to inform future performance-improvement decisions. The end result was a casualty response system integrated with a performance-improvement cycle to continuously validate, refine, and solidify TCCC standards.
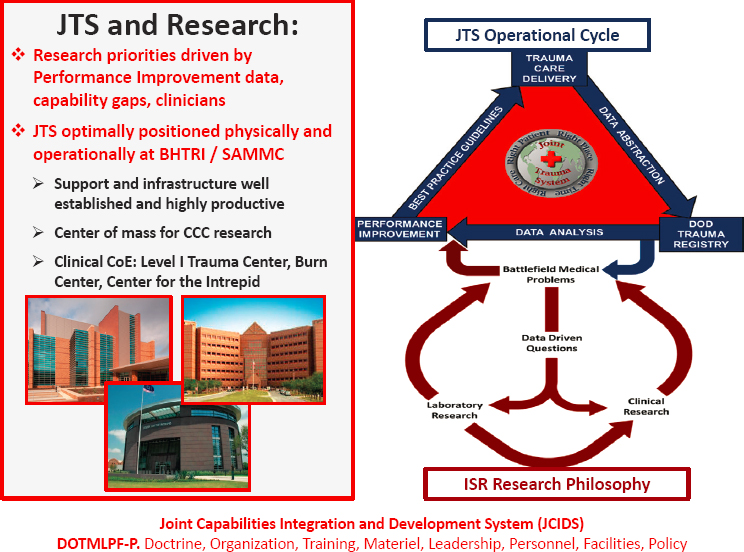
Kotwal outlined the JTS. The JTS concept of collecting and analyzing data began in 1996 and was formally aligned under the DHA in 2018. Its development was funded by R&D monies but maintained by operations and maintenance (O&M) funds. The JTS mission is to improve trauma readiness and outcomes through evidence-driven performance improvement. The JTS and medical research are connected because research priorities are driven by performance-improvement data and capability gaps, as shown in Figure 5. From this point, Kotwal displayed numerous DoD
studies of trauma mortality, injuries, and time, along with military trauma mortality reviews, emphasizing that “wisdom is found at the intersection of knowledge, experience, and judgment.” Among the many references he cited was the 2016 National Academies report A National Trauma Care System.6 Kotwal concluded performance improvement should be used to preserve and advance lessons learned and inform and prioritize R&D. This will provide military solutions; for JTS, 18 years of data have produced approximately 90,000 separate casualty events for analysis.
LESSONS FROM FOUNDING CENTERS AND STAKEHOLDER COLLABORATION
S. Shankar Sastry, Professor, Electrical Engineering and Computer Science Department, University of California, Berkeley, began with an overview of TRUST (Team for Research in Ubiquitous Secure Technology), which conducts core research with integrated education and knowledge transfer. He made a point that TRUST’s success is not measured just by numbers of technical papers produced; rather, it is by making an impact on society. Figure 6 provides a visual overview of TRUST’s tightly integrated areas, (1) education and diversity, aimed at the next generation of computer and social scientists and engineers; (2) research, leading to a science base and delivering breakthrough advances in trust for systems under attack; and (3) knowledge transfer, disseminating and transitioning results along with much external collaboration.
Sastry described grand challenges for financial, health, and physical system infrastructures under attack, such as infrastructures for supervisory control and data acquisition, distributed control systems, smart-grid security, and privacy. He outlined TRUST’s research influence in terms of leadership and engagement with a range of institutions, including the Air Force Office of Scientific Research and HHS-related strategic health care information technology. He noted the value added in education and diversity—for example, undergraduate research experiences that address security, privacy, and policy issues. He noted that TRUST has established a science base with multidisciplinary teams for building trustworthy systems and is a true academic, government, and industry undertaking and public-private partnership model, with integrative research areas and projects at the forefront of many emerging threats.
___________________
6 National Academies of Sciences, Engineering, and Medicine, 2016, A National Trauma Care System: Integrating Military and Civilian Trauma Systems to Achieve Zero Preventable Deaths After Injury, Washington, DC: The National Academies Press, https://doi.org/10.17226/23511.

OTHER COLLABORATIVE MODELS AND PROJECT OUTCOMES
Sastry went on to describe several programs that he believes illustrate successful collaborative models, including the following:
- The Blum Center for Developing Economies illustrated a new model of development innovation with a strong technology component and university-generated solutions for poverty reduction, sustainable management of innovation, and other global challenges. Laboratory personnel explore solutions in areas like food production, energy and water systems, humanitarian and disaster relief, and technologies to detect, diagnose, and treat disease. These activities are congruent with what military expeditionary forces also experience.
- Big Ideas @ Berkeley is a campus-wide annual competition that provides funding and encouragement to graduate and undergraduate interdisciplinary teams with “Big Ideas.” A spin-off includes a company that designs portable solar suitcases that power critical lighting and medical devices in areas without reliable electricity.
- The Development for Impact Lab brings together world-class science, engineering, and economics to change the way new technologies are designed, evaluated, and scaled for the developing world. An illustrative project is a mobile application to monitor electricity quality and reliability.
- The Health Technologies Innovation laboratory has a diagnostic capability to rapidly detect SARS-CoV-2 ribonucleic acid and a continuous intravascular blood monitor for remote patient monitoring.
Sastry discussed other activities involving interdisciplinary learning and problem solving for the Department of State and DoD. He also described a Digital Transformation Institute that has the mission to attract the world’s leading scientists to join in a coordinated and innovative effort to advance digital transformation of business, government, and society.
He noted that technology is changing society in unexpected ways, as with intelligent machines, artificial intelligence, augmented and virtual reality, the Internet of Things, and next-generation infrastructures. For the future, he believed that changes like these will bring about deep technology innovation, resulting in societal changes like better health and improved quality of life along with an entrepreneurial ecosystem capable of devising solutions and strategies to address society’s greatest challenges. He believes these types of organizations assist in translating R&D into practical applications.
EXPERIENCES WITH EXPERIMENTAL MEDICINE—ARMY AND ACADEMIA
Chad Roy, a professor at Tulane University and director of its Infectious Disease Aerobiology scientific core, spoke from the viewpoints of both the Army and academia. He received a Ph.D. in preventive medicine, worked with USAMRIID for several years, and then went to Tulane’s National Primate Research Center. During his time with the Army, Roy concentrated on aerosol threats—investigating toxicity and infectivity of aerosolized biological threat agents. He developed appropriate animal models involving pathogenesis and mechanisms of toxicity and worked to determine the efficacy of vaccines and therapeutics in animal models, considering surrogates of protection and mechanisms of immunity. Roy also participated in mission-driven research with directed funding for performance and a focus on developing medical countermeasures.
Turning to research at the Primate Research Center, he discussed a range of disease models, such as AIDS, Lyme disease, and West Nile virus, and then focused on biodefense and emerging infectious diseases. He described development of appropriate animal models of disease and attention to nonhuman primate models with emphasis on aerosol routes of infection. He also described evaluation of experimental medical products (vaccine efficacy and therapeutics). Illustrating performance metrics for infectious disease aerobiology over the past 5 years, he summarized extensive work on hundreds of discrete aerosol challenges, bioaerosol characterizations, and reagent delivery activities; a range of infectious agents or toxins; numerous challenges and experiments on animal exposures; dozens of bioaerosol characterization studies; and dozens of aerosol reagent drug delivery studies with animals.
He discussed different categories of biodefense and emerging infectious disease–related projects and pathogens related to studies that have been funded, are under way, or have submitted research applications, as well as completed studies with no active work. Directly related to Army and DoD interests, he described ricin-related research and offered the following two observations: (1) prophylactic immunization to protect large populations against a terrorist attack is not realistic and (2) the most likely use of a ricin vaccine will be in military personnel or first responders. A relevant focus would be on developing ricin-specific postexposure treatments and efforts to develop field tests to rapidly detect ricin use. Roy’s discussion of his experimental medicine experiences in the Army, coupled with experiences after moving into academia, such as the challenging switch from direct funding into competitive funding and the “insider” knowledge that a transitioning Army person has compared to a new graduate, were helpful in showing the value of having experts, new students, faculty, and personnel in both academia and the Army understand
the goals, processes, and limitations of both organizations with respect to medical R&D. This kind of understanding contributes to achieving successful collaborations between the Army and outside organizations.
OPPORTUNITIES IN THE R&D LABORATORIES
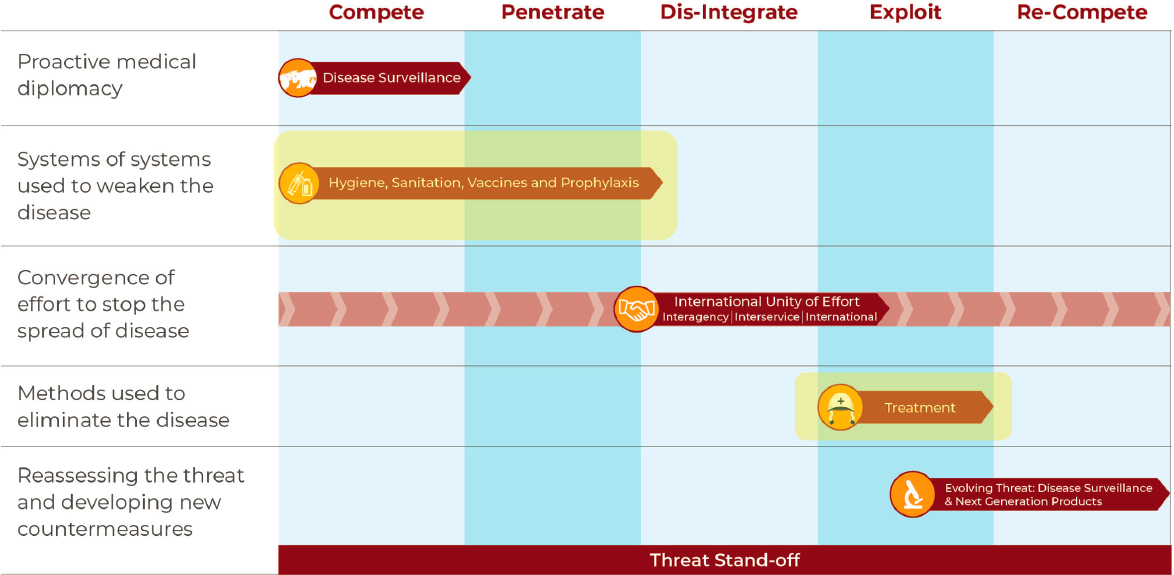
Robert Kaminski, Chief, Department of Diarrheal Disease Research, Bacterial Diseases Branch, Center for Infectious Disease Research at WRAIR, explained that WRAIR offers unique opportunities for scientists, such as science-focused product development, unique customers with unique needs, problems needing solutions outside the normal scope of U.S. public interests, unmatched global capabilities, and a diverse research portfolio. He discussed combating the enteric disease threat across the MDO phases of compete, penetrate, dis-integrate, exploit, and re-compete (Figure 7). The first step, proactive medical diplomacy via disease surveillance, would be followed by weakening the disease through systems of hygiene, sanitation, vaccines, etc. The handshake symbol in the third step down in Figure 7 (convergence of effort to stop the spread of disease) symbolizes the broad unity of effort needed to achieve this goal. The final step in the process, “reassessing the threat and developing new countermeasures,” highlights the necessity of continuing efforts to combat enteric diseases in order to achieve a standoff.
Kaminski described a pipeline of enteric-countermeasure activities—consisting of fermentation, biochemical analyses, animal models, manufacturing (vaccines, therapeutics), and clinical trials for safety, efficacy, and FDA approval. Collaborators included strong worldwide involvement in understanding and treating or preventing these diseases, which are high priorities for the DoD medical community, and leveraged nonDoD assets, because of high rates of infection and decreased-performance or out-of-action days for military personnel.
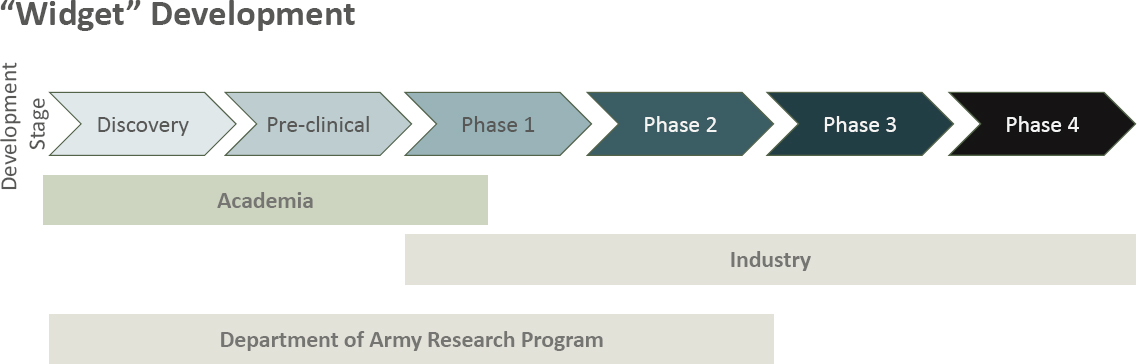
Kaminski viewed the medical research enterprise as “vibrant and opportunity filled,” and outlined impediments to funding and support. He noted that funding allocations did not align with DoD priorities, as diarrheal diseases ranked as the leading infectious disease threat to U.S. troops, yet the scope has been reduced to nonvaccine countermeasures and funding reductions over the past 4 years, with stagnant funding for the previous 10 years. Also, development of medical products and devices for human use is inherently expensive in terms of time and funding, and necessary R&D budgets are difficult to link to fiscal-year spending plans. Kaminski suggested that the potential funding life of projects should be extended from 12 months to 18 months, with influx funding provided during key product-development stages. Kaminski highlighted the differences between academic-to-industry translation and DoD-to-industry
translation (Figure 8), showing that development often extends through several industry phases after early academic discovery and preclinical work, whereas Army research programs normally stop before the end of the industry phase. Thus, Army researchers are in a unique situation, partly academic in nature, and partly industry.
Regarding research funding, Kaminski suggested comprehensive 5-year proposals, funding extensions, and linking funding to priorities. On research support, he suggested building teams linked with scientists and unique procurement needs. Comparing the Army to academia and industry, he highlighted the need to understand the niche, set expectation boundaries appropriately and work within those boundaries, but also thrive in that very special place.
NOVEL ACADEMIC MODELS FOR TRANSLATIONAL RESEARCH
Stephen Quake, professor of bio-engineering and applied physics at Stanford University and co-president of the Chan Zuckerberg Biohub, began with the Biohub organization initiation and noted receiving early inspiration from Don Ingber (the next speaker) regarding the research-institute concept. He was asked by Mark Zuckerberg and Priscilla Chan to start a new biomedical research institute in the Bay area with co-president Joseph DeRisi. The Biohub began operating in 2016 with a budget of approximately $60 million per year. The Biohub vision is to focus on understanding underlying mechanisms of disease and develop new technologies leading to actual diagnostics and effective therapies—an important, bold, and long-term (100-year) venture aimed at curing human diseases. Because of the long timescale, Biohub invests in basic science and technology aimed at revolutionary change over decades. Collaborations were developed with three nearby universities and an industry-experienced Biohub team to enable moving with urgency and flexibility. The Biohub budget is divided roughly into thirds, as follows: funds for exciting university research too risky for federal grants; funds for cutting-edge technology platforms that can advance science frontiers; and funds for an internal research program, which is currently focused on infectious disease and cell biology. As of 2020, the Biohub operates with 111 employees—85 percent are scientists and engineers, half with Ph.D. degrees, with a lean administrative staff. Biohub-related activities have resulted in publication of more than 1,000 research articles and 30 patent applications. Quake ensures that the researchers have great flexibility to use the money any way they want, following their best ideas and not being tied down to a proposal.
Quake then discussed recent achievements. The Tabula Muris Senis, or “Mouse Aging Cell Atlas,” provides molecular information about how
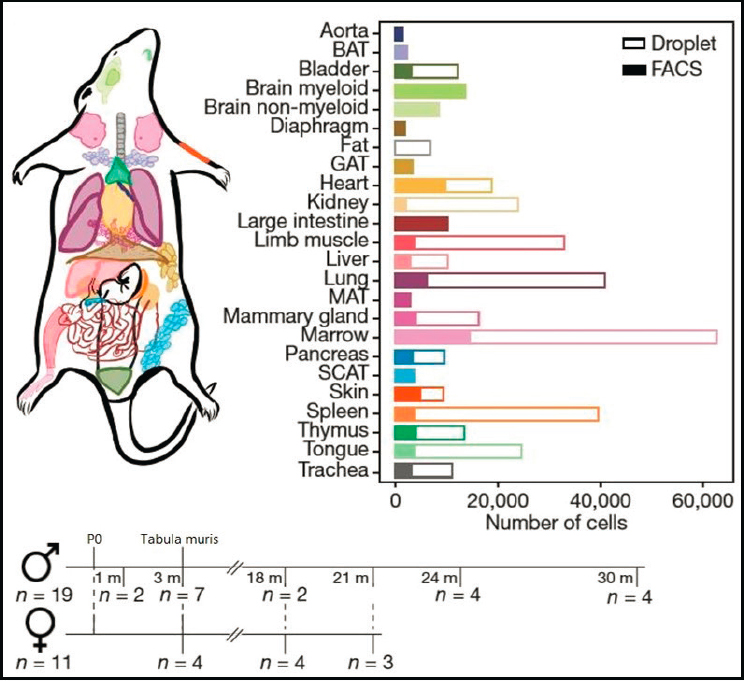
the most important hallmarks of aging are reflected in a broad range of tissues and cell types. The work of the Tabula Muris Consortium was a recent Biohub achievement and was published in Nature.7 This work depicts the aging process and diseases and disorders of the cell (Figure 9). He also discussed an “open cell,” which is an attempt to map out all the
___________________
7 Almanzar, N., Antony, J., Baghel, A.S. et al. A single-cell transcriptomic atlas characterizes ageing tissues in the mouse. Nature 583, 590-595 (2020). https://doi.org/10.1038/s41586-0202496-1.
interactions of proteins within cells, such as images of where proteins are localized, and measuring to provide a catalog. Quake outlined other advancements such as novel forms of microscopy and imaging software. On the infectious disease side, he said they were focused mostly on biology, with the notion of working on ways to detect, respond, treat, and prevent diseases. That overview led to a description of doing hypothesis-free infectious disease epidemiology, IDSeq,8 a software package that is now distributed for use around the world. In that connection, he mentioned work with the Gates Foundation to help people in the developing world implement the system.
Quake outlined the Biohub’s intellectual property and patent policies, where the inventors get to decide. Recognizing that project information is widely shared, if collaborators want to protect their inventions with patents, anything invented with Biohub funds receives Biohub help with the licensing (universities let Biohub do the licensing). Biohub had signed on to the guidelines used by major universities for patent licensing, and Biohub seeks patent protection only in high-income countries—leaving the rest of the world free to practice Biohub inventions without restrictions. He closed with information relating to the COVID-19 pandemic and how his small and nimble organization could pivot and make contributions, such as in testing, developing a simple low-cost ventilator, understanding the cells in human lungs, and gaining insights into immune-system responses.
NEW INNOVATION MODEL THAT SPANS THE ACADEMIC-INDUSTRIAL-MILITARY INTERFACE
Don Ingber, founding director, Wyss Institute for Biologically Inspired Engineering, Harvard University, began by indicating that engineering has transformed medicine over the past 30 years, and looking 30 years ahead, as boundaries between disciplines break down, biological principles can be leveraged to inspire future engineering opportunities. Ingber explained the mission of the Wyss Institute is to transform health care and the environment by emulating the way nature builds. Recognizing that breakthrough discoveries cannot change the world if they do not leave the laboratory, the Wyss vision is to translate disruptive ideas into “commercializable” products. Measures of success are international recognition for scientific leadership and recruitment of world-class faculty, students, and
___________________
8 Chan Zuckerberg Biohub. 2018. Chan Zuckerberg Biohub and Chan Zuckerberg Initiative Announce First-of-its-kind “IDSeq” Platform and Service to Enable Real-Time Global Disease Surveillance and Prevention. October 16. Available at https://www.czbiohub.org/infectious-disease-idseq.
fellows along with a rich patent portfolio, corporate alliances, licensing agreements and startups, and bio-inspired technologies being sold or “productized.” The thrust of these measures is a positive near-term impact on our world. Wyss was founded in 2009 with a focus on high-risk research and technology development that spans academia and industry and is structured to harness the entire Boston–Cambridge region. Co-located activities spawn collaboration among 40 expert technical staff recruited from industry who are able to perform multidisciplinary research at a new level. The institute’s Advanced Technology Team staff add broad industrial experience. Large donated gifts (in millions of dollars) provide funding. Wyss is a product pipeline for biomedical, industrial, and environmental applications; an array of its platforms and initiatives is shown in Figure 10.
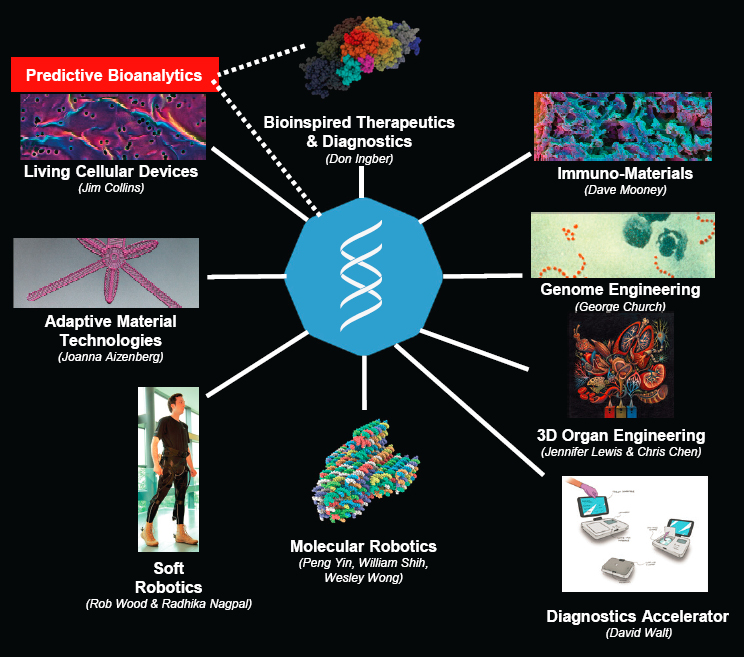
Ingber described Wyss as the “Skunk Works9 of academia,” a translation model that resembles a funnel for innovation, starting with idea generation, narrowing to concept refinement, narrowing again to technology validation and optimization, and finally resulting in the licensing of a few created products. He discussed a range of Wyss technologies that cross the academic-industrial-military interface—for example, engineering human blood elements to enable new sepsis therapeutics and diagnostics, a nonstick slippery liquid-infused porous surface that could enable coatings for medical devices, soft exo-suits for the military, human organs on chips, participation in the DARPA COVID-19 program, injectable therapeutics, hand-held electro-chemical sensors for robust field-able diagnostics, and ways to preserve limbs and organ transplants with drugs instead of cold transport. Ingber noted many academic and translation successes, including 2,300 publications and 66 total licensing deals. His recommendations for Army research include the following:
- Reducing formal requirements for technology development,
- Increasing speed and flexibility in contracting,
- Providing teams and leaders with more autonomy,
- Creating funding sources independent of managed 5-year budgets,
- Developing interdisciplinary collaborations within the research enterprise,
- Integrating multidisciplinary approaches into the research enterprise, and
- Focusing more on creativity and entrepreneurship rather than administration.
Finally, he recommended creating a Skunk Works within each Army research site with an appropriate organizational structure, budget flexibility, and agility so any solution can be brought to the table. He also recommended the development of mechanisms to quickly apply follow-on funding to successful DARPA programs that focus on military-relevant technologies, regardless of technology readiness level.
___________________
9 The popular name for Lockheed Martin’s Advanced Development Programs effort, which has produced a number of innovations in military aircraft design.
















