Early life exposure to neurotoxicants can impact a child’s developmental trajectory in multiple and sometimes unexpected domains, said David Bellinger. It can affect how well an individual is able to respond to neurological insults even later in life, possibly by reducing resilience or cognitive reserve, which may provide a link between neurodevelopment and neurodegeneration, he added. Bellinger noted, however, that effects at the individual level may be small and affected by many other factors, including the extent and time course of the exposure. Thus, in order to estimate the societal impact of neurotoxicant exposure, studies must look at the population rather than individual level, he said.
Bellinger also noted that the effects of neurotoxicants on brain development have been recapitulated in animal models. For example, a study in rats showed that animals that were exposed to lead recovered more poorly to an induced brain injury (Schneider, 2007).
NEURODEVELOPMENT, AUTISM, AND ATTENTION-DEFICIT HYPERACTIVITY DISORDER
As discussed in Chapters 2 and 3, exposure to neurotoxicants such as lead and pesticides has been associated with increased rates of neurodevelopmental disorders such as autism and attention-deficit hyperactivity disorder (ADHD). With about 1 in 68 individuals affected by autism, according to Mark Zylka, this has become a major health problem. The linkages between environmental exposures and autism have already been discussed extensively; here we explore in more detail the association with ADHD.
In addition to effects of lead exposure on IQ, which was discussed in Chapter 2, lead exposure has also been reliably linked to other domains of neurodevelopment, such as attention, executive function, and impulse control, said Bellinger (Braun et al., 2006). Children with higher levels
of lead exposure in early life do less well in school, are more likely to be classified as learning or behaviorally exceptional, and less likely to be considered advanced or intellectually gifted (Miranda et al., 2007, 2010). Bellinger added that the impact of the same level of lead exposure on school achievement and behavior was considerably greater for children already at risk of having these problems due to low socioeconomic status and low parental education levels. As adults, children with higher blood lead levels achieve lower socioeconomic status (Reuben et al., 2017). Putting this all together, said Bellinger, suggests that childhood lead exposure has cascading downstream effects with real consequences on quality of life, including, for some children, greater criminal activity later in life (Boutwell et al., 2017; Coulton et al., 2020; Emer et al., 2020; Nkomo et al., 2017).
Bellinger acknowledged that the effect sizes are small at an individual level, but noted that on a population level, lead has a huge impact on cumulative IQ loss and thus is responsible for a substantial loss of societal intellectual resources (Bellinger, 2012).
Difficulties with attention and impulse control manifest in many children as a diagnosis of ADHD. Indeed, according to Jason Richardson, ADHD affects about 8 to 12 percent of children in the United States, with boys diagnosed about three to four times as often as girls. Like other neurodevelopmental and neurodegenerative disorders, ADHD has a strong genetic basis, but is also associated with multiple other factors, including environmental exposures, he said. In a recent meta-analysis of twin studies, the heritability of ADHD was estimated to be more than 70 percent (Faraone and Larsson, 2019), said Richardson, but factors such as low birthweight, perinatal hypoxia, and lead exposure are also associated with increased risk of ADHD. Environmental toxicants such as polychlorinated biphenyls (PCBs) may also be linked to ADHD, but have been less well studied, he added.
Richardson and colleagues used a gestational lactational exposure paradigm to study the behavioral and neurobiological effects of the pyrethroid insecticide deltamethrin in mice (Richardson et al., 2015). Using doses from 4- to 40-fold lower than the “no observable adverse effect level” established by the Environmental Protection Agency, pregnant mice were fed deltamethrin in peanut butter starting at gestation day 6 through weaning on postnatal day 22. At 6 weeks of age (equivalent to early adolescence in humans), a dose-related increase in locomotor activity was observed in males, which was attenuated by treatment with methylphenidate. Male mice also scored significantly higher on a task assessing impulsivity, said Richardson. The behavioral findings in mice were recapitulated in a separate human study, where pyrethroid pesticide exposure in children (assessed by measuring urinary levels of a pyrethroid metabolite) was shown to be
associated with ADHD, particularly in boys with hyperactive-impulsive symptoms (Wagner-Schuman et al., 2015).
In an effort to understand the mechanism underlying the effect of pyrethroids on locomotor activity, Richardson and colleagues showed significantly increased locomotor activity in male mice exposed to deltamethrin and given a dopamine-1 (D1) receptor agonist. This increase in activity was reduced to control levels by administration of a D1 receptor antagonist. Using receptor autoradiography, they also demonstrated increases in D1 receptors in the nucleus accumbens of these mice, a brain region associated with impulse control and substance abuse (Richardson et al., 2015). They also observed increases in D1 and dopamine transporter mRNA, which persisted through 1 year of age and suggested a possible epigenetic effect, said Richardson. His lab has proceeded to show in cell culture that knocking down DNA methyltransferase increases D1 receptor mRNA, which further supports an epigenetic mechanism.
NEURODEGENERATIVE DISORDERS: ALZHEIMER’S DISEASE, PARKINSON’S DISEASE, AND AMYOTROPHIC LATERAL SCLEROSIS
A complex combination of genetic and environmental factors is thought to contribute to the pathogenesis of neurodegenerative disorders such Alzheimer’s disease (AD), Parkinson’s disease (PD), and amyotrophic lateral sclerosis (ALS), although what initiates the process of neurodegeneration may be different from what drives it subsequently, said J. Timothy Greenamyre. Indeed, several of the mechanisms associated with neurotoxicant exposure that were discussed in Chapter 3, including effects on synaptic function and endolysosomal pathways, oxidative stress, epigenetic changes, and cellular mosaicism, have been linked to neurodegenerative disorders. Greenamyre and Jason Cannon have suggested that common mechanisms may underlie most neurodegenerative disorders (Cannon and Greenamyre, 2011).
Alzheimer’s Disease
Accumulating data indicate that exposure to high levels of air pollution increase the risk for all-cause dementia, said Andrew Petkus (Peters et al., 2019). Indeed, according to Caleb Finch, ARCO Professor and William F. Kieschnick Chair in the Neurobiology of Aging at the University of Southern California, half of individual AD risk may be environmental (Gatz et al., 2006). Finch and Alexander Kulminski of the Duke Population Research Institute and Duke’s Biodemography of Aging Research Unit, recently proposed an AD exposome as a framework for understanding
endogenous and exogenous environmental factors that may contribute to AD as well as gene–environment interactions (Finch and Kulminski, 2019).
Finch’s research focuses on the role of air pollution and cigarette smoke as risk factors for accelerated aging and AD. He calls both of these “sterile gerogens”—sterile because they are toxicants rather than pathogens, and gerogens because they accelerate aspects of aging processes. Both air pollution and cigarettes shorten life span by about 5 to 10 years and accelerate disease of aging, said Finch. He noted that both contain fine and ultrafine particles that are deposited in the lungs as well as incompletely burned carbon particles, carcinogens, neurotoxicants, polyaromatic hydrocarbons, and the toxic metals iron and lead. In the Vietnam Era Twin Study of Aging, cigarette smoking was associated in a dose-dependent manner with brain volume loss (Prom-Wormley et al., 2015); and in the Women’s Health Initiative Memory Study (WHIMS), exposure to particulate matter from air pollution was associated with accelerated loss of both gray and white matter as well as with a decline in episodic memory (Casanova et al., 2016; Younan et al., 2020).
Gene–environment interactions have been demonstrated as well, said Finch. In the Washington Heights-Inwood Columbia Aging Project (WHICAP) cohort, higher concentrations of air pollution were associated with more rapid cognitive decline, particularly in carriers of the APOE4 allele, which is the strongest genetic risk factor for AD (Kulick et al., 2020). Finch’s lab is exploring in mouse models the mechanisms underlying this association. In APOE4-carrying mice, they have shown that oxidative stress from exposure to nano-sized, traffic-related air pollution particulate matter (nPM) results in accelerated production of the amyloid-β protein by reorganizing key enzymes involved in processing the amyloid precursor protein (Cacciottolo et al., 2020). They have also demonstrated that the effects of nPM on gene transcription differ depending on sex as well as APOE allele (Haghani et al., 2020).
Finch also noted “remarkable overlap” in the developmental impact of air pollution, cigarette smoke, and lead exposure (Finch and Morgan, 2020). Synergistic effects from exposure to cigarette smoke and air pollution also have been demonstrated for multiple morbidities, including cognitive aging, he said (Forman and Finch, 2018).
The oxidative stress and inflammation triggered by air pollution contribute to the accumulation of hallmark neuropathologies associated with AD, including amyloid β and tau tangles, brain atrophy, cognitive decline, and eventually dementia, said Petkus. A decline in episodic memory is typically the first cognitive sign of AD, he said (Petkus et al., 2020). Petkus added that air pollution induces variable effects on different aspects of episodic memory; for example, fine particulate matter (referred to as PM2.5) appears to be more strongly associated with the encoding aspect of episodic
memory compared to retrieval and long-term recall aspects, suggesting that it may be impacting brain regions associated with learning new material versus long-term recall (Petkus et al., 2020). However, imaging studies using structural magnetic resonance imaging to assess brain atrophy have produced mixed findings, he said.
Parkinson’s Disease
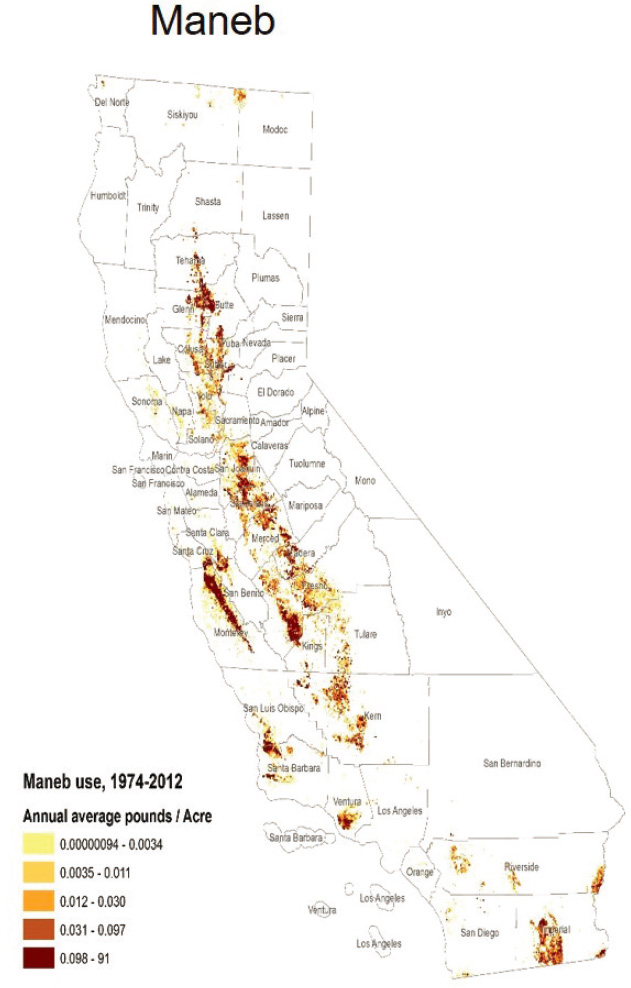
Pesticides have long been linked to onset of PD, although until recently it has been difficult to determine which specific pesticide exposures may be associated with PD, said Beate Ritz. For the past two decades, Ritz and Jeff Bronstein, director of the Movement Disorders Program at the University of California, Los Angeles, have collected biosamples and data on pesticide use in California in the Parkinson’s Environment and Gene Study. By combining data on the timing and geo-location of pesticide application with lifelong address data, they have been able to determine long-term pesticide exposure across a large population. These modeled exposures have been validated against biomarkers of exposure (Paul et al., 2018b; Ritz and Costello, 2006). For example, they estimated levels of two pesticides—paraquat and maneb (see Figure 4-1). They also determined that when these two agents come together, there is an approximately 75 percent increase in risk of developing PD (Costello et al., 2009). The results of their study supported earlier work indicating that exposure to paraquat and maneb increase the risk of PD amongst those who carry risk alleles in the dopamine transporter by as much as five-fold, said Ritz (Kelada et al., 2006; Ritz et al., 2009).
Ritz described another way that gene–environment interactions contribute to the development of PD. Organophosphates (OPs) are another widely used class of pesticides that have been linked to an increased risk of PD. Ritz and colleagues have shown that this risk is affected by variants in the gene for paraoxonase (PON1), an enzyme that detoxifies OP pesticides. These gene variants determine how fast a person metabolizes OPs: those who are slow metabolizers have a much greater increased risk of developing PD, said Ritz (Lee et al., 2013). For example, people who frequently use OPs in their households and are slow metabolizers have about 2.5-fold increased risk of developing PD compared to people who are slow metabolizers, but do not use OPs frequently (Narayan et al., 2013). In other words, said Ritz, genetic susceptibility alone does not increase the risk of PD in the absence of exposure. She added that the combination of PON1 slow metabolizers and OP exposure also contributes to a decline in cognitive function over time, both in populations with ambient residential and occupational exposures from agricultural applications in a population of older Mexican Americans (Paul et al., 2017, 2018a).
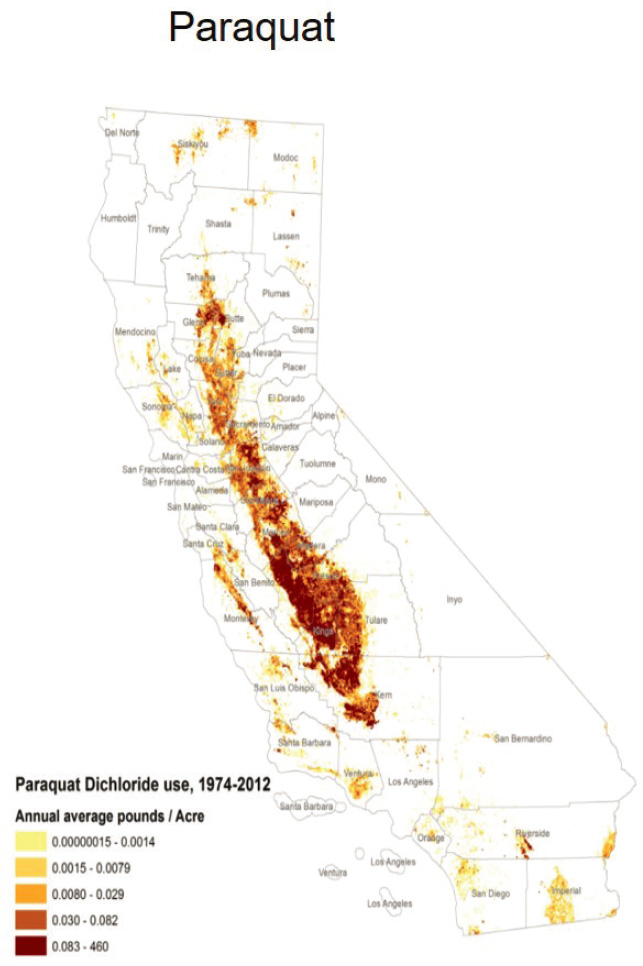
SOURCES: Presented by Beate Ritz, June 25, 2020; California Department of Pesticide Regulation.
Ritz and colleagues have also been investigating the impact of air pollution on the development of PD. A study in Denmark combined sophisticated traffic-related exposure data over a 40-year period with incidence of PD and showed that people highly exposed to traffic had an increased risk of PD (Ritz et al., 2016). They also demonstrated a gene–environment interaction by showing that those exposed to traffic-related air pollution who also carried a polymorphism in the interleukin-1β gene, which is known to increase inflammatory responses in the brain, was associated with a three-fold increased risk of PD in those with high exposure to traffic (Lee et al., 2016).
Amyotrophic Lateral Sclerosis
ALS is a progressive, incurable degenerative disease of the motor neurons in the brain, brainstem, and spinal cord, which has a prevalence in North America of approximately 3.5 per 100,000. Eva Feldman, the Russell N. DeJong Professor of Neurology and director of the ALS Center of Excellence Pranger ALS Clinic at the University of Michigan, said that while heritability clearly plays a role, multiple studies show there is an environmental component as well. Scientists believe that in ALS, as in other neurodegenerative diseases, a clear genetic load or genetic predisposition combines with aging and cell damage and with environmental exposures to initiate a self-perpetuating decline to death, she said (Al-Chalabi and Hardiman, 2013).
Michigan is a hotspot with a high prevalence of ALS cases, said Feldman, which she suggested may result from the large number of uncleaned Superfund sites in the state (see Figure 4-2).
Against this backdrop, Feldman has been collaborating with Stephen Goutman, director of the Pranger ALS Clinic at Michigan Medicine, on a project designed to identify environmental and occupational toxic exposures, the sources of these exposures, and the effects of these exposures on the metabolome of 156 individuals with ALS compared to 128 controls. Using whole blood gas chromatography and mass spectrometry, they measured levels of three groups of chemicals: chlorinated pesticides, brominated flame retardants, and PCBs. In addition to being highly toxic, Feldman noted that these chemicals are also highly persistent in the environment, sometimes lasting for decades before dissipating. Their results, published in 2016, reported an association between persistent environmental pollutants and ALS. Chlorinated pesticides were associated with the greatest risk of developing ALS, said Feldman (Su et al., 2016).
Because they also saw that organic pollutants were highly correlated with each other in terms of case exposure, Goutman, Feldman, and colleagues collaborated with Stuart Batterman and Bhramar Mukherjee from the University of Michigan School of Public Health to assess exposures to
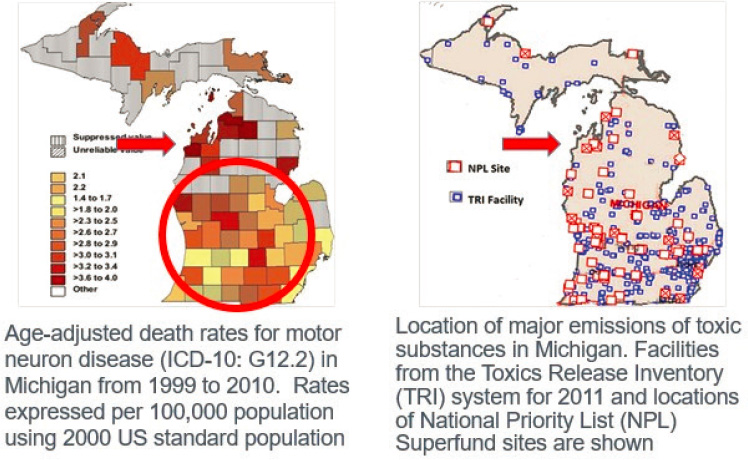
SOURCE: Presented by Eva Feldman, June 25, 2020.
multiple pollutants. Using a mathematical model, they showed that the cumulative environmental risk score for mixtures of pollutants in ALS patients was more than seven times that of controls. They also showed that exposure to persistent organic pollutants influence survival. Individuals with the lowest environmental risk scores survived twice as long as those with the highest scores, said Feldman (Goutman et al., 2019).
Feldman and colleagues, led by Manish Aurora at Mount Sinai in New York City, have also explored how exposures during childhood and adolescence affect ALS risk. Using laser analysis of metals in teeth, this joint collaboration showed that 11 metal toxicants present in teeth—including zinc, chromium, and manganese, but not lead—were clearly associated with increased ALS risk (Curtin et al., 2020; Figueroa-Romero et al., 2020). Feldman added that metabolomics studies conducted by her lab have also demonstrated a significant difference in the metabolome of ALS patients associated with exposure to the chlorinated pesticide pentachlorobenzene. She noted that more environmental studies are needed to advance this research, especially in regard to recruiting and maintaining cohorts to later support more hypothesis-based studies.










