Throughout the workshop, several participants highlighted potential opportunities to address policy concerns in the field, foster multidisciplinary collaborations to advance research, and motivate action among stakeholders.
POLICY IMPLICATIONS FOR ENVIRONMENTAL NEUROSCIENCE
Improving human brain health by reducing exposures to environmental toxicants will require both an expanded research enterprise, as discussed in Chapter 5, as well as public policy changes that prioritize prevention of neurodevelopmental and neurodegenerative disorders, said David Bellinger. To accomplish this, new and existing data will need to be packaged and presented to stakeholders in a way that demonstrates the devastating impact of environmental exposures on brain development and function, and strengthens the case for increases in attention and resources. Experts from multiple fields must also advocate for these changes publicly with a unified voice, said Eva Feldman. Deborah Cory-Slechta added that these experts’ voices must also be present on review committees and committees that are doing risk assessment at the Environmental Protection Agency (EPA).
The framework Bellinger proposed included
- Developing a broader and more integrated story about how early life exposure to neurotoxicants can impact a child’s developmental trajectory in multiple and sometimes unexpected domains.
- Demonstrating how early life exposure to neurotoxicants can affect how effectively an individual is able to respond to neurological insults by reducing resilience and cognitive reserve; and thus, how
- these exposures may be linked to neurodevelopmental and neurodegenerative disorders.
- Estimating the societal impact of neurotoxicant exposures by examining data at a population rather than an individual level; for example, by highlighting the association between lead exposure and criminal behavior in adolescence and young adulthood.
To make these messages resonate with policy makers, population health research—which focuses on the multiple factors that affect health outcomes in and across groups of people—may play an important role, said Allison Willis. She and her colleagues in the Division of Economic Research at the Leonard Davis Institute of Health Economics at the University of Pennsylvania have developed a public health framework for neurotoxicant exposure, disease risk, and public health outcomes. Most of the data presented at this workshop fit into this framework, she said, allowing policy holders to understand the data in their language and see the long-term and population health outcomes that are at risk from chemical exposures.
Carl Hill questioned what kind of framework would create momentum to move the field forward. He suggested bringing thought leaders from different fields together to develop a taxonomy that crosses these disciplines. Devon Payne-Sturges agreed, adding that more interdisciplinary work is needed to understand how the cumulative impact of environmental exposures and social stressors may help to explain the differences in prevalence of neurodevelopmental and neurodegenerative disorders by socioeconomic status, race, or ethnicity.
Convincing regulators of the relevance of biological science to exposure risk is an additional challenge, which will require cross-disciplinary dialogue among clinicians, epidemiologists, basic researchers, and toxicologists, added Stanley Barone. The screening and testing of chemicals is not a trivial pursuit, and cannot be done on all chemicals in the marketplace with current testing approaches, he said. Barone reinforced comments made by Helena Hogberg on the need to prioritize the worst chemicals to be tested first and to develop higher throughput approaches using molecular and cellular approaches.
Patrick Breysse, director of the National Center for Environmental Health/Agency for the Toxic Substances and Disease Registry (NCEH/ATSDR) within the Centers for Disease Control and Prevention, said one of the challenges is how to evaluate the multiplicity of exposures and incorporate that information into policies that are protective and that can be implemented at the state or health department level. NCEH/ATSDR works to translate scientific knowledge into protective measures that improve public health, according to Breysse. However, he noted that many public and environmental health practitioners fail to recognize the wide variety of
neurotoxicants in the environment and the wide range of biological effects. Thus, there is no sense of urgency to address these issues going forward, said Breysse.
New approaches are also needed within academic institutions and government agencies, including the National Institutes of Health (NIH), to encourage and reward scientists to get out of their silos and work collaboratively, said Richard Woychik. He advocated creating mechanisms that reward people for doing complementary and synergistic science across disciplines, including environmental science, neuroscience, biochemistry, and genetics, among others. Bringing the powerful tools from each of these scientific disciplines to bear on the questions raised in this workshop will also be essential, particularly in regard to the exposome, he said.
Payne-Sturges added that undergraduate and graduate programs in neuroscience also need to incorporate into their programs of study courses that raise awareness about the policy implications of neuroscience research and that emphasize the importance of interdisciplinary collaborations between neuroscientists and environmental health scientists, with a focus on research translation.
OPPORTUNITIES FOR MULTIDISCIPLINARY COLLABORATION
A broad approach that brings together expertise from multiple disciplines is needed to craft solutions to the problems imposed by environmental exposure to neurotoxicants, said Feldman. For example, she said multidisciplinary collaborations are needed to integrate what is known about persistent organic pollutants with longitudinal exposure data, and then coupling that with genome-wide association studies to determine polygenic risk scores. No one laboratory can do all of this, she said.
Andrew Petkus agreed that team science is essential to address the complex approaches used to estimate environmental exposures and understand their associations with multifactorial and heterogeneous disorders such as Alzheimer’s disease (AD). His team, for example, includes neurologists and psychologists, as well as experts in exposure science with particular technical expertise in the complicated methodologies that enable linking EPA monitoring data with geographic covariates such as population density and urban versus agricultural land use.
Several collaborative programs have already been established by NIH and the National Institute of Environmental Health Sciences (NIEHS). For example, Jason Richardson mentioned that his work with the Collaborative Centers for Parkinson’s Disease Environmental Research, funded by NIEHS, brought together cutting-edge neuroscience and environmental science, and set the standard for the types of collaborations that can occur.
Richardson said he has also received funding through NIEHS’s Virtual Consortium for Translational/Transdisciplinary Environmental Research program. In one of these projects, he is collaborating with Brenda Plassman at Duke University to develop a comprehensive dataset, including cognitive measurements from her Agricultural Health Study of Memory, a panel of serum biomarkers associated with AD, and measurements of serum pesticides.
In 2015, NIH established the Environmental Influences on Child Health Outcomes (ECHO) Program,1 a transinstitute research initiative to examine the effects of environmental influences on the health of children. ECHO compiled data from existing longitudinal cohorts comprising about 50,000 children across the United States. As part of this work, Tracey Woodruff and colleagues developed an approach for prioritizing chemicals to be studied for their contributions to child health including neurodevelopmental conditions in children as a pathway toward prevention. Criteria include (1) the presence of the chemical in consumer products; (2) biomonitoring of the chemical demonstrates levels greater than 10 percent in biospecimens or greater than 20 percent in environmental media such as air, house dust, food, or drinking water; and (3) potential for toxicity including neurotoxicity using data from EPA ToxCast, a high-throughput assay program that has screened thousands of compounds from many different endpoints including neurotoxicity. The panels selected chemicals that fall into five classes: organophosphorus and alternative flame retardants, alternative plasticizers, aromatic amines, environmental phenols, and pesticides, said Woodruff (Pellizzari et al., 2019). A pilot project has been launched to measure 100 of these novel chemicals prenatally, she said.
Meanwhile, NIEHS has funded the Toxicant Exposures and Responses by Genomic and Epigenomic Regulators of Transcription (TaRGET)-II Consortium2 to explore exposure-induced epigenomic signatures and to determine whether target tissues can be used to assess the epigenetic consequences of various exposures (Wang et al., 2018), said Marisa Bartolomei. Consortium members will use the same exposure model that Bartolomei described in her studies of bisphenol A (see Chapter 3, in which pregnant mice are exposed to agents through food, water, or air; offspring continue to be exposed through weaning after which the exposure ends; and then tissues are sampled from progeny at various time points for epigenomic and transcriptomic analysis) (see Figure 6-1).
Among the questions to be addressed by the consortium are whether epigenetic perturbations in mice are relevant to human phenotypes and
__________________
1 For more information, see https://www.nih.gov/research-training/environmental-influences-child-health-outcomes-echo-program (accessed September 8, 2020).
2 For more information, see https://targetepigenomics.org (accessed September 8, 2020).
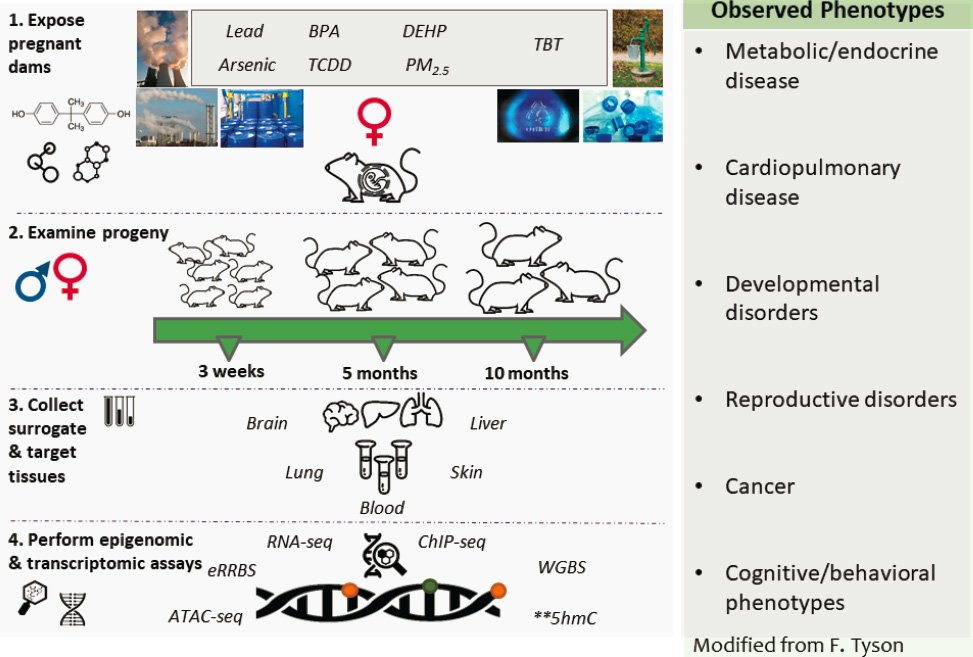
SOURCE: Presented by Marisa Bartolomei, June 25, 2020.
whether there is an exposure signature that can be assessed in certain surrogate tissues, said Bartolomei. She added that while the consortium is addressing only single exposures, more complex studies with multiple exposures will also need to be addressed in the future.
To increase understanding of gene–environment interactions, Richard Woychik from NIEHS advocated more intersection between environmental health scientists and neuroscientists, especially neuroscientists studying genetics. Beate Ritz noted that her research has only been possible because of funding for a multidisciplinary center for neurodegeneration at the University of California, Los Angeles. This enabled crosstalk among neuroscientists, animal experimenters, population scientists, and epidemiologists to determine which pesticides to select as well as which genes and pathways to interrogate.
MOTIVATING ACTION
Neurodegenerative disorders—that is, AD, Parkinson’s disease, and amyotrophic lateral sclerosis—together account for more disability than cardiovascular disease, infectious disease, and cancer, said Ray Dorsey, neurologist at the University of Rochester. “And to a great extent, we’ve brought these diseases upon ourselves,” he said, through the use of agricultural and industrial chemicals and air pollution.
Although more research is clearly needed, motivation to take action has been largely absent, said Dorsey. Economic considerations have kept chemicals such as paraquat and trichloroethylene on the market in the United States, although they have been banned by many other countries, despite strong linkages to Parkinson’s disease and the availability of safer alternatives, he said.
Woychik said he believes there are high-profile geneticists who agree that environmental exposures have a significant impact on genes and genetic disorders and who are willing to work with environmental scientists to advance this research. The key, he said, is to bring together people with complementary points of view and different skillsets to discuss neurodevelopmental and neurodegenerative disorders from environmental, genetic, and neuroscience standpoints.
This page intentionally left blank.








