INTRODUCTION
The committee calls for a new and broader focus on sexual health and new and expanded ownership and accountability for confronting sexually transmitted infections (STIs) across society. At the same time, the committee recognizes and supports the health system as having primacy in improving STI outcomes. Nonetheless, within the health system (including public health, public and private health insurance programs, and other health care safety-net programs), with its decentralized regulation and complex organization and financing, it is unclear which entities or professions are accountable for improving STI outcomes. It is the committee’s view that the answer is not to centralize responsibility into one or more types of specialists or institutions. Rather, as with the recommendations for community stakeholders (see Chapter 12), the committee finds that it is necessary to broaden responsibility for STI screening, prevention, and treatment within the health system and increase accountability across payers and programs.
PAYING FOR STI PREVENTION AND TREATMENT SERVICES
Health care and clinical preventive care in the United States are paid for by a patchwork of private insurance plans and government-supported programs, which finance STI services for the vast majority of, but not all,
U.S. residents. About half the U.S. population relies on employer-sponsored health insurance to pay for STI screening, testing, and treatment services (see Figure 10-1). One in five individuals, nearly all of whom are low income, are covered by Medicaid, a program that is operated by states under broad federal guidelines but jointly funded by state and federal governments. Both Medicare and Medicaid offer their beneficiaries a defined set of benefits and guarantee coverage of medically necessary services to those who qualify, which differs from other discretionary programmatic funding discussed in Chapter 4 that is constrained by the amount of funding appropriated by Congress. Despite the broad reach of employment-based coverage, the availability of subsidized individual plans to purchase coverage through Patient Protection and Affordable Care Act (ACA) marketplaces, and the important role of Medicare and Medicaid, a sizable fraction (about 9 percent) of U.S. residents are uninsured and do not have a direct payment source for their care (see Figure 10-1). Those who lack coverage rely on public health services and other discretionary programs (see Chapter 4). The following sections discuss the role of publicly and privately funded coverage programs in providing access to and coverage of STI services for U.S. residents.
While sexually active individuals of all ages are at risk for acquiring STIs, teens and reproductive-aged adults are disproportionately affected (see Box 10-1 for information about confidentiality and its importance in adolescent care).
Furthermore, as Figure 10-2 shows, 6 in 10 individuals aged 15–49 receive coverage through the workplace, either through their own employer or as a dependent (typically as a dependent child up to age 26
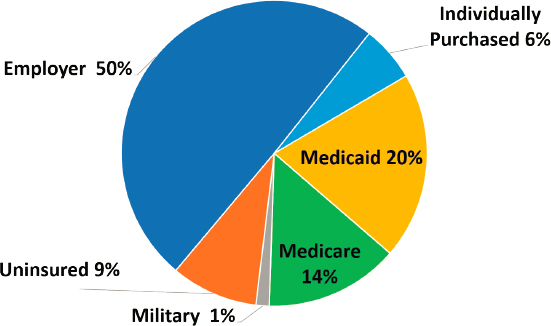
NOTE: Total excludes Puerto Rico.
SOURCE: Data from KFF, 2019a.
or a spouse). Nearly 8 percent obtain individually purchased insurance, many securing subsidies based on their household income to purchase coverage through federal or state marketplaces established by the ACA. Under the ACA, nearly all of these plans cover a wide range of preventive services, including for STIs, such as chlamydia screening for sexually active women younger than age 25, and are not permitted to charge the policyholder any cost sharing for those services (see later in this chapter for a discussion of these programs).
Medicaid plays a particularly important role in providing coverage for low-income individuals and Black and Hispanic populations, who disproportionately have low incomes. In addition, a higher share of women than men relies on the program, which stems from eligibility policy originating in categorical programs, including cash assistance and pregnancy-related care. State-established Medicaid eligibility levels must fall within
broad federal guidelines, meaning that states must cover parents but can set their income standards and must extend eligibility to all pregnant people with incomes below 135 percent of the federal poverty level (FPL).
The ACA included a Medicaid expansion that allows states to enroll all eligible individuals with income up to 138 percent of the FPL; before that, only individuals who fell into federally established categories, including pregnant women, parents of dependent children, individuals 65 and older, and those with disabilities, could qualify. The ACA originally required all states to make this expansion, but a legal challenge that reached the Supreme Court in 2012 (National Federation of Independent Business v. Sebelius) resulted in a ruling that allowed states to be able to choose whether to expand Medicaid to other populations and higher income categories. This state choice has resulted in inconsistent coverage policies across the country. As of September 2020, 38 states and the District of Columbia have adopted a Medicaid expansion.
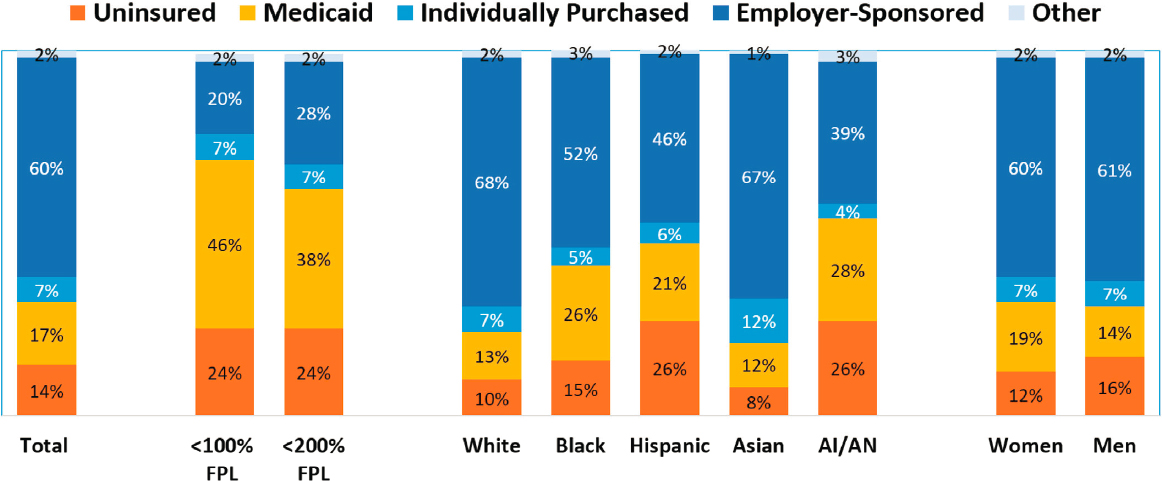
NOTES: The Census Bureau federal poverty level (FPL) was $13,300 for a nonelderly individual. “Other” includes those covered under the military or Veterans Health Administration and nonelderly Medicare enrollees. AI/AN = American Indian/Alaska Native.
SOURCE: Kaiser Family Foundation, unpublished analysis of 2019 Census Bureau’s American Community Survey; data file available upon request from PARO@nas.edu.
Twelve states1 have not expanded Medicaid and still limit eligibility to low-income individuals in the categorical groups (pregnant women, parents, individuals 65 and older, and those with disabilities) and often those with incomes far below the FPL (KFF, 2020e). In all of these states, adults aged 19–49 are not eligible, no matter their income level, if they are not pregnant, parents, and/or living with a disability. Eligibility for parents in these states ranges from a low of 17 percent of the FPL in Texas to 100 percent of the FPL in Wisconsin. This policy results in many in these states losing pregnancy-related Medicaid eligibility (which ends 60 days postpartum). In non-expansion states, those who have Medicaid coverage for pregnancy do not have a pathway to affordable coverage 2 months after delivery, and half become uninsured (Daw et al., 2019). This also has been documented to a lesser degree in expansion states, where coverage churn affects one-third of those who were covered by Medicaid during their pregnancy (Daw et al., 2019). This state inaction has prompted advocates and policy leaders to call for federal and state responses to address this coverage gap (Eckert, 2020; Ranji et al., 2020).
State refusal to adopt the ACA Medicaid expansion, in particular, has contributed to the considerable variation in uninsured rates (see Figure 10-3), from 4 percent to 26 percent of adults under age 65. A county-level analysis of STI rates across the United States found that 64.3 percent of counties without current Medicaid expansion were in the highest two STI quartiles compared to 42.2 percent of counties with Medicaid expansion (p < 0.0001) (Rietmeijer et al., 2021).2 Thus, across quartiles, higher combined STI rates were significantly associated with a lack of Medicaid expansion.
Medicaid also plays an outsize role in financing family planning services, including STI screening, testing, and treatment for individuals. Family planning services are mandatory under Medicaid, although the phrase is not specifically defined in regulation or statute. Many states have used a waiver mechanism to establish a Medicaid financed limited-scope family planning program that typically includes STI services along with contraceptive services and supplies (Ranji et al., 2019). The ACA has enabled states to establish these programs using a state plan amendment
___________________
1 Alabama, Florida, Georgia, Kansas, Mississippi, North Carolina, South Carolina, Tennessee, Texas, Wisconsin, and Wyoming.
2 Rates of reportable STI (chlamydia, gonorrhea, and primary and secondary syphilis) were examined at the county level (3,218 counties) for counties located in states with (2,177) or without (1,041) Medicaid expansion using data from the Census Bureau’s American Community Survey. For simplicity, data for the three STIs were combined. County data were grouped by quartile of combined STI rates, and a simple chi-square analysis was used to test for statistical significance across quartiles. Odds ratios were calculated comparing the highest quartile with a combination of the lowest three.
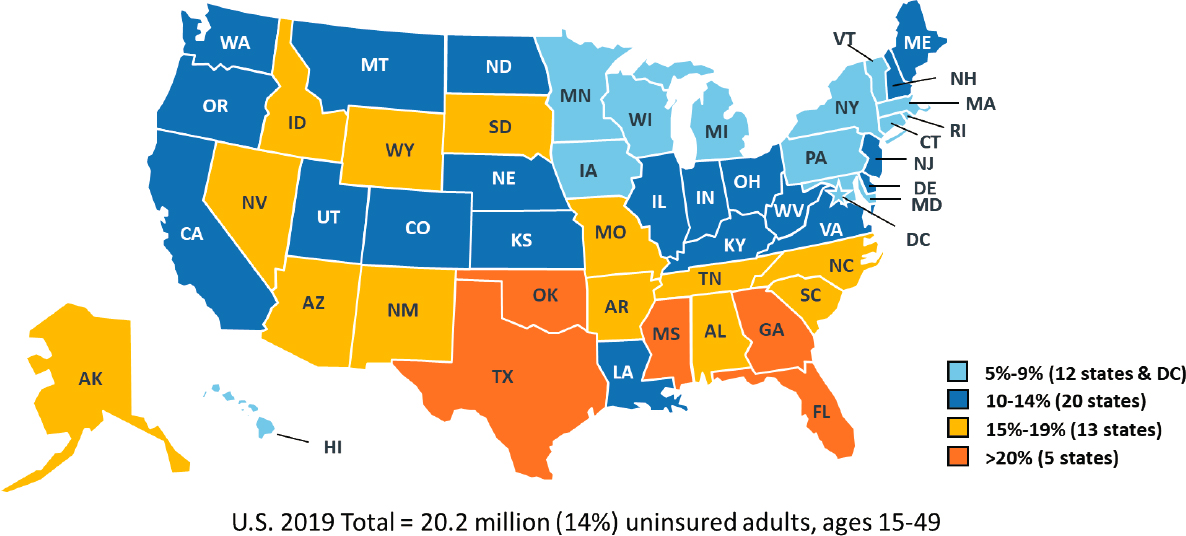
SOURCE: Kaiser Family Foundation, unpublished analysis of 2019 Census Bureau’s American Community Survey; data file available upon request from PARO@nas.edu.
(Ranji et al., 2016). There are currently 26 states operating these limited-scope programs, and many, but not all, also include men (KFF, 2020d). Eligibility for these programs is based on income, loss of Medicaid due to the end of pregnancy-based eligibility (ends 60 days postpartum in non-expansion states), or loss of Medicaid eligibility for other reasons.
A notable gap in Medicaid has been a ban on coverage of immigrants lawfully present in the United States, who are generally ineligible for Medicaid (unless they are asylees or refugees) for the first 5 years of legal residency unless their state of residency has chosen to expand coverage to those who are children or pregnant, which half of the states have done (KFF, 2020a). Undocumented individuals, however, lack a pathway to any type of publicly supported insurance coverage, including Medicaid, Medicare, and subsidized ACA marketplace coverage, and are disproportionately uninsured unless they obtain coverage through an employer.
Uninsured individuals, particularly those who are low income, have fewer STI screening and treatment options and typically depend on a network of safety net providers, including STI clinics and health departments, federally qualified health centers (FQHCs), and family planning providers, for free or low-cost screening and treatment services. These providers typically rely on a patchwork of federal, state, and local funding to sustain these services (see Chapter 4). The decrease in funding to support STI services and federal family planning funding has been well documented (Gift et al., 2018). Clinic closures and reduced staffing and hours of care have directly impacted the ability of uninsured individuals, particularly those that are lowest income and more likely to be medically underserved, to obtain care.
HEALTH INSURANCE COVERAGE REQUIREMENTS
Federal Requirements for Coverage of STI and Related Services
Coverage programs also have a large role in establishing payment, performance, and service delivery standards. While the ACA was primarily focused on providing more coverage to the uninsured, it also contains several provisions that shape the scope of coverage for STI services for individuals with commercial or employer plans, Medicare, or Medicaid.
Section 2713 of the ACA (KFF, 2015) includes a requirement that health insurance plans cover preventive services, without any patient cost sharing, that receive an A or a B rating from the United States Preventive Services Task Force (USPSTF), are endorsed by the Advisory Committee on Immunization Practices (ACIP), or are recommended for women by the Health Resources and Services Administration (HRSA) (KFF, 2020b).
The law stipulates that all private plans cover these services—self-funded and fully insured employer plans, as well as individual policies. Only plans that have “grandfathered” status (i.e., have been in effect without substantive changes since before the ACA was enacted) are exempt.
Medicaid expansion coverage must offer the same preventive services that are required of private health plans. For those who would have qualified under the pre-ACA income eligibility levels (i.e., traditional Medicaid), however, preventive services are considered “optional” (states can decide not to cover them). Nevertheless, many states have aligned this coverage and do not differ between the services they cover for expansion populations and those who qualify under traditional pathways. Importantly, children have extra protections under Medicaid’s Early and Periodic Screening, Diagnostic and Treatment (EPSDT) benefit, which requires states to periodically screen people under age 21 for certain conditions; in addition, any coverable service under Medicaid must be provided when medically necessary to children, even if it is optional for adults.
Table 10-1 shows a breakdown of which recommended preventive STI services must be covered by most private insurance plans, Medicare, ACA Medicaid expansion programs, and traditional Medicaid plans. Only preventive services recommended by USPSTF, ACIP, HRSA’s Bright Futures Project, and HRSA’s Women’s Preventive Services Guidelines are covered without cost sharing (KFF, 2020b). While the ACA’s provisions set a floor for coverage, they do not cover all aspects of STI preventive care and treatment. For example, STI treatment is typically covered by most public and private plans but may be subject to cost sharing for follow-up visits and prescriptions. In addition, private plans or Medicaid programs are not required to pay for partner evaluation and management, including expedited partner therapy (EPT), which provides the patient with medication or a prescription for an affected sexual partner (see below and Chapter 7 for more on EPT). That risk assessment alone, however, is insufficient for prevention and control, as persons may deny risk due to stigma or be unaware of the risk that their partners create for them, which is a shortcoming in general for STI screening guidelines.
The ACA preventive services coverage provision sets a standard for coverage, and the recommendations in its requirements are all issued by independent committees (USPSTF, ACIP, Bright Futures, and Women’s Preventive Services Guidelines) that make their recommendations based on evidence-based reviews of findings from peer-reviewed studies. Screening and testing recommendations for young adult men, however, are generally lacking. While Bright Futures recommends screening for all adolescents, many men under 25 are diagnosed with STIs, and no recommendation suggests routine screening for them. Indeed, a shortcoming of current recommendations is that they are focused on large average risk
TABLE 10-1 Preventive Services Required to Be Covered by Different Payers Without Cost Sharing
| Service | Recommending Authority: Eligible Populations | Medicare | Private Insurers* (federally required) | Medicaid Expansion (federally required) | Traditional Medicaid |
|---|---|---|---|---|---|
| Chlamydia screening | USPSTF: Screen all sexually active women age 24 and younger, all sexually active women (aged 25+) who are at increased risk | Women ≤24 annually; women >24 if at increased risk; up to 2 screenings in pregnancy | √ | √ | Required for children under EPSDT Covered at state option for adults |
| Bright Futures: Screen all sexually active adolescents for STIs (11–21 years old) | Χ | √ | √ | Required for children under EPSDT Covered at state option for adults |
|
| Gonorrhea screening | USPSTF: Screen all sexually active women age 24 and younger, all sexually active women (aged 25+) who are at increased risk | Women ≤24 annually; women >24 if at increased risk; up to 2 screenings in pregnancy | √ | √ | Covered at state option for adults |
| Bright Futures: Screen all sexually active adolescents for STIs (11–21 years old) | Χ | √ | √ | Required for children under EPSDT Covered at state option for adults |
| Service | Recommending Authority: Eligible Populations | Medicare | Private Insurers* (federally required) | Medicaid Expansion (federally required) | Traditional Medicaid |
|---|---|---|---|---|---|
| Syphilis screening | USPSTF: Screen all pregnant women and persons at risk | Pregnant women at start of pregnancy and third trimester, at delivery if high risk; all men and other women annually if at risk | √ | √ | Covered at state option for adults |
| Bright Futures: Screen all sexually active adolescents for STIs (11–21 years old) | Χ | √ | √ | Required for children under EPSDT | |
| PPX for ocular gonorrhea | USPSTF: Provide prophylactic ocular topical medication to all newborns to prevent gonococcal ophthalmia neonatorum | Χ | √ | √ | Covered at state option for adults |
| Service | Recommending Authority: Eligible Populations | Medicare | Private Insurers* (federally required) | Medicaid Expansion (federally required) | Traditional Medicaid |
|---|---|---|---|---|---|
| HPV testing | Women’s Preventive Services Guidelines: For women aged 21–29, screen with cytology every 3 years; for women aged 30–65, screen with cytology and HPV testing every 5 years or cytology alone every 3 years | Covers screening once every 5 years if asymptomatic for ages 30–65 | √ | √ | Covered at state option for adults |
| USPSTF: Screen women aged 21–65 with cytology every 3 years or, for women aged 30–65 who want to lengthen screening interval, screen with cytology and HPV testing every 5 years | √ | √ | Covered at state option for adults | ||
| HPV vaccine | ACIP: Routine vaccination for men and women aged 11–12; also vaccinate the following groups if they have not been vaccinated previously or have not completed the three-dose series: women aged 13–26, men aged 13–21, and men who have sex with men and immunocompromised persons through age 26 | Χ | √ | √ | Required for children under EPSDT |
| Covered at state option for adults |
| Service | Recommending Authority: Eligible Populations | Medicare | Private Insurers* (federally required) | Medicaid Expansion (federally required) | Traditional Medicaid |
|---|---|---|---|---|---|
| STI and HIV prevention counseling | USPSTF: Provide intensive behavioral counseling to prevent STIs to all sexually active adolescents and for adults at increased risk for STIs | Up to two 20–30-minute behavioral counseling sessions annually for those at high risk | √ | √ | Required for children under EPSDT Covered at state option for adults |
| Women’s Preventive Services Guidelines: Annual counseling on STIs, including HIV, for all sexually active women | Χ | √ | √ | Covered at state option for adults |
NOTE: ACIP = Advisory Committee on Immunization Practices; EPSDT = Early and Periodic Screening, Diagnostic and Treatment; HPV = human papillomavirus; PPX = prophylaxis; USPSTF = United States Preventive Services Task Force.
* Applies to non-grandfathered plans.
SOURCES: CDC, 2020d; CMS, n.d.; KFF, 2020b; Medicare.gov, n.d.-c.
populations by design and so neglect subpopulations such as adolescent males, racial and ethnic minorities, men who have sex with men (MSM), and others that are at greater risk for acquiring STIs. Without guidance from USPSTF or clinical professional organizations, pathways to provide these services to men and avenues for reimbursement and out-of-pocket payment protections are both limited.
STI Testing and Treatment Guidelines Issued by Health Professional Organizations
The Centers for Disease Control and Prevention (CDC) and USPSTF provide national standards and guidance for the prevention, screening/testing, and treatment of STIs. Health professional organizations, however, sometimes adapt these recommendations for their own members or have their own recommendations. Appendix B presents the different recommendations issued by leading health professional organizations and highlights how they compare to the CDC/USPSTF recommendations. These organizations include the American Academy of Family
Physicians, the American Academy of Pediatrics, the American College of Obstetricians and Gynecologists, the American College of Physicians, the American Academy of Physician Assistants, the American Public Health Association, and various nursing organizations (e.g., American Nurses Association).
For the most part, recommendations published by professional organizations are consistent or mostly consistent with CDC and USPSTF guidelines. These organizations are more likely to publish guidance when there is a paucity of information, however, than to publish inconsistent information. Many organizations direct readers to CDC or USPSTF for more detailed guidelines and focus more on the guidelines that pertain only to their population of specialization (e.g., women for the American College of Obstetricians and Gynecologists, pediatrics for the American Academy of Pediatrics). Medical societies are also more likely to publish specific screening/treatment recommendations than organizations representing nurses, physician assistants, and public health practitioners are, as the latter tend to rely on existing policy recommendations.
ASSESSING SYSTEMS OF CARE AND ACCOUNTABILITY
Performance Measures on STIs
Despite broad recognition that the United States falls short in obtaining health care value and quality for its outsized investment, the nation still lacks a uniformly accepted and widely adopted method for measuring quality. This is an especially notable gap for sexual and reproductive health services, including STI prevention and treatment for sexual minority groups, such as LGBTQ populations, particularly MSM, bisexual, and transgender individuals who are at higher risk of exposure to STIs.
The measures most commonly used by providers and plans are those promulgated by the Healthcare Effectiveness Data and Information Set (HEDIS), the National Quality Forum (NQF), and the Medicaid Core Set. The measures that have been approved and adopted largely focus on services for women, teens, and infants, including chlamydia screening for women, human papillomavirus (HPV) vaccination, and cervical cancer screening. NQF also collects data on the share of persons with HIV being screened for STIs and vaccination rates for hepatitis A and B, although these are not typically the focus of STI prevention efforts. Screening and treatment completion indicators for gonorrhea are absent in the HEDIS and Medicaid Core Set, as are targeted screening and treatment completion services for men. Because these performance measures are designed to capture recommended services that pertain to a large population rather than targeted at a population at risk, plans and payors are not able to
provide information about the effectiveness of these systems in serving these often marginalized and high-risk populations.
In contrast, the CDC performance measures for state health departments track the performance of STI clinics, family planning sites, and adult jails along with measures to track partner services and data reporting. These STI measures are considerably more robust than those that are used by payors and plans with HEDIS and Medicaid. In addition, CDC measures include timelines of treatment and counseling, which are critical in terms of tracking the recommended responses to identified cases yet are not addressed by the HEDIS, NQF, or Medicaid measures. These measures, however, have not been updated since 2011 (CDC, 2011). Table 10-2 lists the performance measures on STIs for health plans, Medicaid, and CDC.
Overall, completion rates for the few STI-relevant measures tracked by HEDIS are quite low. For example, according to 2018 HEDIS data on teens who had a health care visit in the past year, only 30 percent enrolled in a health maintenance organization (HMO), 25 percent enrolled in a preferred provider organization (PPO), and 38 percent enrolled in a Medicaid HMO had completed the HPV vaccine series by their 13th birthday. Similarly, HEDIS reports that only half of sexually active women aged 16–24 who were enrolled in an HMO or PPO and had a health care encounter had a chlamydia screening during the measurement year; the rates were slightly higher for those in a Medicaid HMO (58 percent). STI screening under NQF is limited to persons with HIV. No STI-related measures for men are represented in HEDIS, NQF, or the Medicaid core measures promulgated by the Centers for Medicare & Medicaid Services. It also should be noted that HEDIS measures are calculated based on health plan enrollees who received a service within a given year. Enrollees who did not have a claim or health care visit are not included in the denominator, so HEDIS measures likely overestimate the true screening rates among enrollees.
Given the number of enrollees who are now eligible for Medicaid or individual policies through the ACA exchanges, the absence of measures for men represents a major gap for a population that is served by these programs. With the growth and interest in developing value-based payment strategies to incentivize the provision of recommended care, the absence of measures that track a broader range of STI screening, testing, and treatment for men and other populations that are at high risk of exposure and also experience poor access to care represents a significant gap. In addition to improving the completion rates for services that affect women and teens, there is an urgent need to develop tools to track whether providers and plans are meeting the standards of care for often marginalized populations that are at higher risk for contracting STIs, including MSM, transgender men and women, and other gender nonconforming individuals discussed in Chapter 3. Finally, the lack of STI performance measures for services needed by these populations will
TABLE 10-2 Health Plan, Medicaid, and CDC Performance Measures for Sexually Transmitted Infections
| HEDIS Measures | |
| HPV immunizations (NCQA, n.d.-c) |
|
| Chlamydia screening in women (NCQA, n.d.-b) |
|
| Cervical cancer screening (NCQA, n.d.-a) |
|
| NQF-Endorsed Measures | |
| Chlamydia screening in women (NQF, n.d.) |
|
| STI screening for persons with HIV (NQF, n.d.) |
|
| Hepatitis B/hepatitis A vaccines (NQF, n.d.) |
|
| Medicaid Performance Measures (data collected by state) | |
| Cervical cancer screening (adult core) (Medicaid.gov, n.d.-a) |
|
| Chlamydia screening in women (adult core and child core) (Medicaid.gov, n.d.-a, n.d.-b) |
|
| Immunizations for adolescents (child core) (Medicaid.gov, n.d.-b) |
|
| CDC Performance Measures for Public Health Departments (CDC, 2011) MLS | |
| Chlamydia testing in juvenile detention facilities |
|
| Chlamydia positivity in juvenile detention facilities |
|
| Timely treatment of women with chlamydia at family planning sites |
|
| Timely treatment of women with gonorrhea at family planning sites |
|
| Timely treatment of women with chlamydia at STD clinics |
|
| Timely treatment of women with gonorrhea at STD clinics |
|
| Timely treatment of P&S syphilis cases |
|
| Syphilis testing of women at select adult jails |
|
| New syphilis cases diagnosed in select adult jails |
|
| Timely syphilis treatment in select adult jails |
|
| CDC Partner Services | |
| Timeliness of P&S syphilis interviews |
|
| Timeliness of prophylactic treatment for contacts to P&S syphilis cases (CSPS PS2a) |
|
| Timeliness of treating contacts of P&S syphilis cases who are themselves infected (CSPS PS2b) |
|
| Timeliness of gonorrhea interviews (CSPS PS3) (required for non-high-morbidity areas only) |
|
| CDC Statistics and Data Management for Health Departments | |
| Completeness of data |
|
| Timeliness of data |
|
| Completeness of data |
|
NOTE: CDC = Centers for Disease Control and Prevention; CT = Chlamydia trachomatis; GC = Neisseria gonorrhoeae; HEDIS = Healthcare Effectiveness Data and Information Set; HMO = health maintenance organization; HPV = human papillomavirus; IPP = Infertility Prevention Program; MLS = Medical and Laboratory Services; NETSS = National Electronic Telecommunications System for Surveillance; NQF = National Quality Forum; P&S = primary and secondary; PPO = preferred provider organization; STD = sexually transmitted disease; STI = sexually transmitted infection.
also pose a barrier to the future development of a sexual and reproductive health reimbursement bundle that could potentially include payment for the recommended screening and treatment services for STIs, counseling, and other services.
CLINICAL STI SERVICES
In 2020, CDC released recommendations on improving quality of STI clinical services, including sexual history and physical examination,
prevention, screening, partner services, evaluation of STI-related conditions, laboratory, treatment, and referral to a specialist for complex STI or STI-related conditions. The recommendations distinguish between basic services that should be available in primary care settings and more extensive services in STI specialty settings (Barrow et al., 2020). These guidelines represent an aspirational standard of STI care, but there are many settings where STI care is delivered differently depending on the population served, with the priority being that STI care and prevention account for and address the medical needs of these populations and the logistical and resource constraints in each setting. From a pragmatic and strategic point of view, it is important to examine each of these settings in detail to determine their optimal role in STI care and prevention.
The next sections discuss different components of STI clinical care, followed by a detailed review of STI services in the spectrum of care settings that identifies the relative strengths and weaknesses and how they may fit together in a continuum of services rather than a fragmented system.
Sexual Health Assessment
The more we focus on teaching doctors how to take a really good sexual history, the better. A lot of doctors don’t feel comfortable taking a sexual history and asking questions about sex—and maybe when they do, they may make assumptions and moral judgments. Patients sense this, and that adds to the discomfort they already feel.
—Participant, lived experience panel
Assessing sexual health is the force driving subsequent interventions for STI prevention and control and can normalize sexual health as a component of overall health. A basic sexual health assessment needs to be an integral part of a “review of systems” as a standard in most health care settings—both offered routinely by the provider and expected by the patient/client. At a minimum, this assessment includes questions on sexual orientation and gender identification (SOGI) and a review of the “Five Ps”: partners (gender, number), (sexual) practices (receptive and insertive vaginal, anal, oral sex), prevention (condom use, contraception), previous history of STIs, and pregnancy (CDC, 2015a). Besides being good clinical practice, the consistent collection and review of SOGI data at the health systems level allows for sexual health policy development and funding allocation.
While the fundamentals of sexual health assessments are routinely taught as part of health care professional training, their use in the context of routine care typically atrophies over time, particularly when training
is carried out in acute care and inpatient settings. CDC recommends routine sexual health assessments in U.S. care settings, but a review showed generally low adherence to these guidelines (Brookmeyer et al., 2020). Thus, these sexual health assessments need to be further routinized and automated and protocols developed that will reduce provider reliance to determine who is at risk for STIs. Technological advances may provide opportunities, such as clinical prompts in an electronic health record (EHR; see Chapter 6 for more on EHRs). Information gathering, such as tablet-based self-reporting of recent sexual activity, would allow data collection in clinicians’ waiting rooms, with little impact on visit time. Such assessments could be age and gender specific.
Following the principles laid out in Chapter 1, a sexual health assessment needs to be free of prejudice and stigmatizing language. When conducted appropriately, it provides a window into the patient’s sexual well-being and suggests potential intervention steps. Conducting such an assessment needs to be a basic competency addressed in medical and nursing school curricula, and health systems could consider accommodations, such as EHRs, to facilitate recording the details. This information could lead to action prompts, such as recommended chlamydia screening for sexually active women under the age of 25 (see Chapter 6 for more on this topic).
While a regular sexual health assessment is critical in sexual health promotion and STI prevention, it also has limitations. For example, given the continued stigma surrounding sexuality, sexual activity may be under-reported, especially among adolescents and young adults and people with multiple partners. Furthermore, STI acquisition depends on partners’ sexual behavior, which may include exposures unknown to the individual seeking care, and STIs may be missed when testing is limited to those who verbalize potential exposure. Thus, STI screening programs, such as opt-out chlamydia screening for high-prevalence groups (e.g., those under 25) regardless of sexual health assessment and even bypassing the provider, could be developed. Evidence from cost-effectiveness modeling studies suggests that such approaches may be cost saving (Owusu-Edusei et al., 2016).
Behavioral Interventions
Health promotion and risk reduction counseling is the most commonly used behavioral intervention in the health care setting and serves a variety of health objectives, including quitting smoking, promoting healthy eating and drinking, addressing substance use, and enhancing physical activity. When performed correctly, counseling can have positive impacts on health behavior. Effective counseling models have
been developed and implemented in care settings, including the widely adopted motivational interviewing (Miller and Rollnick, 1991). A similar technique called “client-centered counseling” has been proven to be effective for STI prevention (Kamb et al., 1998), but a follow-up study conducted 15 years later could not confirm its effectiveness (Metsch et al., 2013), possibly related to overall changes in attitudes following the introduction of highly active antiretroviral therapy that dramatically changed the perception of HIV as an invariably deadly infection. Nonetheless, the principles of motivational interviewing, including those applied in client-centered counseling, and shared medical decision making have been widely adopted and influenced the provider–client interaction (Elwyn et al., 2014).
The following steps can be achieved in a single brief counseling session (Dreisbach et al., 2014):
- Discuss how various sexual behaviors can expose a person to STIs.
- Assess the patient’s understanding and beliefs about STI transmission.
- Assess the circumstances that affect the patient’s sexual behavior.
- Assess the patient’s readiness to change.
- Negotiate a behavioral goal.
- Identify a concrete and realistic first step toward the goal.
One common critique of counseling is that it is time intensive; even when providers can bill for it, it is often not implemented. The Safe in the City waiting room video intervention was shown to be effective in reducing incident STIs in a large, controlled study involving more than 40,000 patients in three STI clinics (Warner et al., 2008) and was highly cost effective and even cost saving in many settings (Williams et al., 2020). Follow-up studies demonstrated widespread adoption in STI clinics (Harshbarger et al., 2012). Health care settings, including clinics and doctors’ offices, could incorporate more of these brief behavioral interventions and other types of behavioral interventions (e.g., family interventions; see Chapter 8) to increase behavioral assessment, improve patient education, and establish behavior change goals to increase positive sexual health practices and decrease STI risk. To stay relevant and fresh, however, these interventions need regular updates. Unfortunately, despite its cost effectiveness, no additional funding has been available to develop follow-up videos for Safe in the City.
Immunizations
An inventory of recommended immunizations is obtained routinely in many practices and needs to include approved and recommended immunizations for sexually transmitted pathogens, including hepatitis B virus and HPV. As discussed in Chapter 7, hepatitis B vaccination is now widely recommended as part of the routine childhood series, so coverage is increasing. In 2017, 73.6 percent of infants received it within 3 days of birth (Hill et al., 2018). As the cohort that received standard childhood immunization ages, overall coverage will also increase. HPV immunization coverage has lagged behind; only 51.1 percent of adolescents aged 13–17 were fully immunized in 2018 and 48.6 percent in 2017 (Walker et al., 2019), although the vaccine has had large positive effect on HPV infection (McClung et al., 2019) (see Chapter 7 for a discussion of barriers). Uptake has been hampered in part by the cost of the three-dose schedule. Thus, the recent shift in recommendations toward a two-dose schedule (at baseline and 6–12 months later) for 9–14-year-olds will likely improve coverage (CDC, 2019a). Unlike other routine childhood immunizations, however, HPV vaccination is often presented as optional. This linkage to (future) sexual activity creates a barrier for providers to offer and for parents to accept it. Emphasizing its importance as a cancer prevention tool and de-emphasizing the link with sexual activity has been suggested as a more effective way in routinizing this intervention (Offit, 2014).
Testing and Screening
The reality is that we are all sexual creatures, so we are all at risk, and we should all be screened.
—Participant, lived experience panel
Chapter 7 extensively discusses the technical details of diagnostics. From an implementation perspective, STI testing can be considered to fall into four not entirely mutually exclusive groups: screening, opportunistic testing, on-demand testing, and diagnostic testing. Screening is universal or age-based testing for populations considered to be at specific risk for a certain pathogen and may include opportunistic testing (i.e., offering a test, typically in a health care setting, to a person who makes a visit unrelated to the test, such as chlamydia testing for a sexually active woman visiting her primary care provider for an upper respiratory tract infection and who has no STI symptoms or concerns). On-demand testing typically occurs for a specific concern (i.e., exposure to an STI but otherwise asymptomatic). Finally, diagnostic testing is conducted for symptomatic STIs to determine etiology and select appropriate treatment.
While many providers will offer testing in different categories, some types are more appropriate for certain venues than others. For example, online testing services may be very appropriate for on-demand testing but not for diagnostic testing, as symptomatic patients should be encouraged to seek out a health care provider. Making these distinctions will prove useful for recommendations to enhance interventions for STI prevention, as detailed below.
As discussed in Chapter 7, the widespread implementation of nucleic acid amplification tests (NAATs) for gonorrhea and chlamydia since the mid-1990s has revolutionized the armamentarium of STI diagnostics. These tests are highly sensitive and specific and, most importantly, can be performed on specimens, including urine specimens and self-obtained vaginal, throat, or anal swabs, that no longer require invasive procedures. As a result, STI testing is now conducted in a variety of settings, which has effectively broadened the available venues beyond the traditional STI specialty clinic, and extragenital (i.e., pharyngeal and anal) testing should now be the standard of care for all providers of basic STI services per CDC guidelines (Barrow et al., 2020).
Availability of point-of-care (POC) testing promises to further enhance diagnostic capability. Some POC testing (Gram-stained smear to differentiate gonococcal from nongonococcal urethritis and wet preps of vaginal discharge to differentiate trichomoniasis, bacterial vaginosis, and yeast infection) and rapid treponemal (Syphilis Health Check) and nontreponemal (such as rapid plasma reagin test) testing for syphilis, however, have long been available but are not widely used outside the STI specialty clinic setting due to regulatory and logistical issues. Studies, including demonstration projects, need to be conducted to elucidate barriers and facilitators to implementing POC testing in all venues that provide STI care, as it is likely that tests on the horizon may face the same hurdles as those now available.
Prenatal Screening for Syphilis and Other STIs
All cases of congenital syphilis are failures of the health care system. To eliminate the disease, prenatal services need to be universally available without cost or access barriers (as many cases are missed due to lack of prenatal care), and screening needs to be universal to treat infection in the pregnant person and prevent neonatal infection (see Chapter 3 for more information). Universal antepartum screening for syphilis (as well as chlamydia and gonorrhea) is recommended because treatment provided appropriately during pregnancy will prevent adverse outcomes for the pregnant person and the infant in nearly all cases (Cheng et al., 2007; Newman et al., 2013). The U.S. Public Health Service, CDC, the American
College of Obstetricians and Gynecologists, and other medical, nursing, and public health organizations recommend STI screening during the first prenatal visit. Many states require syphilis testing during prenatal care, as of 2018: 42 states have laws requiring syphilis screening in the first visit, only 15 states require a second screening in the third trimester, and 5 require screening only if high risk. Only six states require testing at delivery (and six others do for those at high risk), in addition to prenatal care screening. State policies regarding prenatal syphilis screening are diverse but may be an alternative to address screening policies (CDC, 2020e; Warren et al., 2018). Ideally, HIV screening would always be accompanied by other STI screening and vice versa (Rac et al., 2017; Taylor et al., 2017). Guidelines also typically recommend that women at higher risk be rescreened at the beginning of their third trimester and again at delivery or after exposure to a partner who may be infected. Universal screening for syphilis and other STIs can reduce stigma because testing is offered to all pregnant people irrespective of symptoms or risk behavior. Compared to single testing, repeat testing for HIV and syphilis during pregnancy is cost effective and improves outcomes for the pregnant person and the infant for both congenital syphilis and HIV during pregnancy and at birth in higher-prevalence priority populations (Albright et al., 2015; Hersh et al., 2018). Therefore, offering unrestricted access to prenatal care and effectively deploying screening services to key marginalized persons are essential in decreasing rates of congenital syphilis. This will require improving access to prenatal care and more widespread testing (including easier access points for care) in populations of those most impacted and their sex partners.
STI Testing and Pre-/Post-Exposure Prophylaxis for HIV Prevention
As discussed in other parts of this report, the use of antiretrovirals in pre- and post-exposure prophylaxis (PrEP and PEP, respectively) is a highly recommended intervention for HIV prevention yet may lead to increases in condomless sex and associated incidence of non-HIV STIs (Jenness et al., 2017; Volk et al., 2015). As this intervention is being implemented in a variety of settings, guidelines for PrEP and PEP stress regularly screening for STIs, including serological syphilis testing and testing for chlamydia and gonorrhea at all exposed anatomical sites (CDC, 2018). (See Chapter 5 for additional discussion on the intersection of STIs and HIV.)
“Express” Visits for STI Testing
NAATs have obviated the need for invasive anogenital sampling, so some STI clinics have explored the possibility of testing-only visits (i.e., not including physical examination of asymptomatic patients) with the
main objective of increasing the efficiency of clinic operations in times of dwindling resources. These are known as “express” or “fast-track” visits, and early proof-of-concept studies have shown their feasibility, with up to 25 percent of visits qualifying for the express option, resulting in decreased clinic time compared to traditional, full-exam visits (Heijman et al., 2007; NACCHO, 2019; NAPA, 2018; Shamos et al., 2008). Subsequent studies have confirmed that this approach is safe, efficient, and acceptable to patients (Chambers et al., 2017, 2018), and many clinics have since adopted some form of it. Taking this approach a step further, the Dean Street EXPRESS clinic3 in London is the first and best-known example of a clinic that only offers the express option, but it is organizationally linked to a full-service HIV/STI clinic across the street that allows for easy referral for treatment or clinical follow-up (Whitlock et al., 2018). In the United States, CDC supports a project overseen by the National Association of County and City Health Officials to develop demonstration projects exploring feasibility and acceptability of express visit options in different clinical environments. Challenges include insurance reimbursement for services not involving medical providers. In addition, challenges for standalone STI express clinics include lack of funding and the need for expeditious referral to full-service clinics for additional follow-up and care. Nonetheless, the evolving landscape of express STI clinical services is an excellent example of how technological advancements, such as POC tests, can leverage innovative approaches and new options in clinical care and prevention (Gaydos et al., 2020). Further developments include offering testing-only options in nonclinical settings, including mobile outreach units, pharmacies, community organizations, and other venues where people disproportionally affected by STIs may congregate. Future availability and implementation of POC testing will be particularly important in the evolution of the express visit option to decrease the time to treatment for clients who otherwise do not present with an immediate reason to treat.
Treatment
At the provider level, STI treatment either follows a positive STI test (etiologic treatment) or is implemented presumptively based on patient symptoms (syndromic treatment) or partner infection (epidemiologic treatment). Syndromic treatment has been used for patients with urethral discharge, vaginal discharge, genital ulcer disease, and pelvic inflammatory disease. It has some obvious advantages (no need for onsite laboratory tests) but also many disadvantages, including both overtreatment with unnecessary antibiotics and under-treatment for those without
___________________
3 See https://dean.st (accessed January 26, 2021).
symptoms. The CDC Treatment Guidelines (CDC, 2015b) (discussed in Chapter 7) cover both etiologic and syndromic STI management. As a result of the COVID-19 pandemic and its restrictions on in-clinic patient visits, CDC has issued interim guidelines that include expanding syndromic management in certain cases (Bachmann et al., 2020).
Delays between diagnosis and treatment mean a patient may transmit the infection; treatment needs to follow diagnosis as quickly as possible (hence the need for POC testing), would ideally be delivered at the point of care, and would preferably be a single dose to maximize adherence. For example, studies show that 15–20 percent of persons seen in emergency departments (EDs) and STI clinics with positive screening tests for chlamydia do not receive treatment within 30 days of testing, and of those who return for treatment, approximately 4 percent develop complications, such as pelvic inflammatory disease or epididymitis (Hook et al., 1994; Schwebke et al., 1997). Unfortunately, neither POC testing nor treatment are available in many clinic settings, and implementation studies are necessary to better understand how and with what means the quality of STI care in these settings may be improved (Gaydos et al., 2019).
Partner Services and Expedited Partner Therapy (EPT)
A challenge that I personally face in my practice is contradictory guidance on EPT. In our Oklahoma laws, it’s very hazy. If I have a patient who tests positive for chlamydia, and I want to send partner treatment home with my patient, it’s a bit hazy whether or not I am covered by Oklahoma law according to the CDC references. Now, I do it, but it is hazy. For some of my colleagues who are maybe not as public health oriented, I think it keeps them from treating partners, and I think that should be changed across the board in some way.
—Participant, lived experience panel
After diagnosis and treatment, prompt evaluation and treatment of identifiable recent sex partners is the second pillar of effective STI control and prevention. Partner notification (PN) and referral for STI care, however, often has been carried out irregularly. Historically, PN has been conducted in differing ways: initiated by the patient/client after encouragement from the provider (client referral), sometimes aided by issuing referral cards, or performed directly by the provider if clients feel uncomfortable communicating with their partners (provider referral). A third type of notification is a hybrid of the first two, where the client is given a certain period to notify and the provider will take over if the client has not done so, also known as “contract referral.” In the real world, partner services exist along a continuum that includes these three options. For example, one can think about assisted and unassisted client referral
based on the level of counseling and follow-up and whether the patient receives a referral card. State and local health departments often provide PN and referral through disease intervention specialists (DIS). DIS can be effective in case finding, but they are time consuming and costly. As a result, DIS-initiated partner services are prioritized for STIs with the highest morbidity, including HIV and early syphilis, while some PN may be initiated for chlamydia or gonorrhea in the context of co-occurring HIV infection and/or pregnancy. Thus, partner services for most chlamydial and gonococcal infections rely almost exclusively on patient/client referral (Hogben et al., 2016).
EPT (partner treatment without an intervening professional evaluation) has been promoted and is becoming more common. CDC has concluded that EPT is a “useful option” for partner treatment, especially for male partners of women with chlamydia or gonorrhea (CDC, 2020b). EPT can be implemented by providing patients with additional medication or a prescription for their partner(s). EPT has been demonstrated to be effective in preventing reinfection in the index patient in a number of randomized clinical trials (Golden et al., 2005, 2015), and CDC has recommended it since 2005 for heterosexual men and women with chlamydial or gonococcal infections (Douglas, 2015; Kissinger et al., 2005; Schillinger et al., 2003). EPT has been demonstrated to be particularly effective for gonococcal infections (Golden et al., 2005), even though it is given as oral medications, which are not recommended as a first-line treatment for gonorrhea, given concerns of emerging resistance. In certain jurisdictions this has hampered widespread adoption. Thus, developing new effective oral antibiotics for the treatment of gonorrhea (see Chapter 7) may positively affect more widespread EPT implementation. EPT is not recommended by CDC for use among MSM, in large part because of missed opportunities to diagnose HIV infection and syphilis when MSM present as contacts in the clinical setting (Stekler et al., 2005). This issue requires further study.
EPT implementation varies by jurisdiction. According to CDC, it was legally available in 45 states and the District of Columbia (as of May 2020) and is potentially available in 4 states4 as well as Guam and Puerto Rico. Only South Carolina explicitly prohibits it (CDC, 2020c). EPT has become more common among publicly funded family planning clinics; a study found that 79 percent provided EPT at the same visit (Zola and Frost, 2016).
Not all states, even those where there are no legal barriers, permit the patient’s insurance plan to be billed for EPT (KFF, 2020c). This practice limits access to EPT and can serve as an obstacle to it. A community-level trial demonstrated that it is feasible to implement EPT at the population
___________________
4 Alabama, Kansas, Oklahoma, and South Dakota.
level if the medication is made widely available through public funding (Golden et al., 2015), suggesting that EPT effectiveness may increase when it is considered as a public health rather than individual health intervention. This also implies that 340B funding should be available for EPT drugs, as has been successfully implemented in a number of jurisdictions (FPNTC, 2017). Finally, safety issues (such as allergic reactions), perceived legal obstacles, potential for intimate partner violence, and possible contributions to progressive antimicrobial resistance are important consequences that impede EPT. Extensive experience in the past decade, however, has allayed these concerns (Hogben et al., 2016; Jamison et al., 2018, 2019), and the committee supports continued evaluation of the best use of EPT for controlling STIs.
Telehealth
Telehealth services use electronic information and telecommunication technologies to support primary, behavioral, and specialty health care. Telemedicine is a subcategory that involves two-way, real-time communication for clinical care. Telemedicine coverage policies are complex, dynamic, and often geographically specific. Telemedicine is playing an increasingly important role due to the growth of new technologies and the effects of the COVID-19 pandemic on the need for technologies to facilitate physical distancing and replace in-person visits to health settings (Hartnett et al., 2020; Koonin et al., 2020).
Many states only permit physicians licensed in those particular states to provide telemedicine services. Some pre-COVID-19 policies have limited the extent to which patients can have an STI visit remotely through strict requirements as to the type of provider and sometimes patient location and communication medium. The COVID-19 pandemic, however, has transformed the role of telemedicine in the future of health care, including STI care (Nagendra et al., 2020). Illinois serves as a case example (Young and Schneider, 2020). Both academic medical centers, such as the University of Chicago, and FQHCs, such as Howard Brown Health, transitioned scheduled patients to telemedicine beginning in March 2020, developing telemedicine protocols, billing algorithms, and Health Insurance Portability and Accountability Act–compliant communication videoconferencing platforms. This occurred rapidly (within a few days), with patient acceptability examined and clinical flow modified with experience. STI and HIV care was provided remotely, and patient engagement was substantial (Young and Schneider, 2020). Furthermore, studies have shown that individuals can successfully collect their own samples for HIV and STI screening (Carnevale et al., 2020; Melendez et al., 2020). As telemedicine increases as a result of COVID-19, using home-collected samples for mail-in STI testing to a laboratory has also been shown to be
efficient (Carnevale et al., 2020; Melendez et al., 2020; Ogale et al., 2019; Patel et al., 2019).
From an implementation science perspective, several important questions remain regarding telemedicine’s effectiveness, feasibility, acceptability, and contextual variation (Tuckson et al., 2017). For example:
- To what degree should sustainability of telehealth be pursued for sexual health/wellness, and what hybrid models (telehealth/in person) models are useful at the clinic and client levels?
- How can community-based testing approaches (i.e., mobile unit care, home visits) become next generation through telehealth that allows for busy providers to join remotely and potentially manage multiple programs simultaneously?
- How does acceptability vary by population subtype, and what models can be optimized for care of those without or with limited technology access?
- What strategies and interventions are needed to bridge the digital divide experienced by individuals and clinics and community-based organizations in underserved communities?
While COVID-19 has positioned telehealth as an important solution for physical distancing, how it will evolve once better therapies and vaccines for COVID-19 exist and how STI client visits will be affected for counseling, diagnosis, treatment, and PN remain unclear.
Furthermore, telemedicine for STI care has its downsides, especially for symptomatic patients, which has led to temporary expansion of syndromic management guidelines (Bachmann et al., 2020). It remains to be seen how telemedicine will change the STI care landscape in the post-COVID-19 era. Studies are necessary to evaluate the extent of telehealth provision for STI care during the epidemic and to elucidate lessons learned for future applications.
Given the dearth of U.S. clinical specialists who are experts in sexual health and STI care and treatment, telemedicine will likely hold appeal beyond the COVID-19 era. Rural areas may benefit the most, as these have many fewer specialized providers available (AIDS United and MAO of Alabama, 2017). Even urban clients may prefer telemedicine after their COVID-19-related experiences because of the potential convenience and privacy. Telemedicine will also benefit from new dermatological probes and pathological diagnostic aids, often as simple as using a cell phone camera with a special attachment. The future of telemedicine can bring convenient and affordable services for positive sexual health and stigmatized conditions among clients reluctant to seek such services otherwise.
STI SYSTEMS OF CARE
STI diagnostic and treatment services are available from many clinical providers, each with specific client/patient populations, opportunities, and challenges. The following sections map national STI care and highlight ways for a fragmented system to become better integrated.
Primary Care
The majority of reportable STIs (gonorrhea, chlamydia, and syphilis) are diagnosed outside STI specialty clinics; a large proportion originate from primary care providers and others, including those associated with HMOs and other practice systems (CDC, 2019b). With more than 200,000 primary care physicians in the United States, however, and many more nurses, nurse practitioners, physician assistants, and others in primary care, the average primary care provider diagnoses only a few STIs annually. Thus, STIs do not constitute a high priority in a setting where more prevalent conditions, including cardiovascular disease, pulmonary disease, and diabetes, require the most attention. On the other hand, the “secret weapon” of primary care providers is continuity of care, as care in these settings is built on a continuous patient–provider relationship that allows issues that are not immediately top priorities to be addressed in future visits, effectively breaking up the ongoing conversation, interventions, and follow-through over time. These are advantages not easily replicated in other settings, such as STI clinics.
Given time and resource constraints, the greatest contribution that primary care can provide in STI control lies in opportunistic testing, including offering annual chlamydia screening to all sexually active women under the age of 25, as recommended by CDC, USPSTF, and other organizations (USPSTF, 2014) (see Table 10-1). According to data from HEDIS, however, recommendation adherence is less than 50 percent in most settings (NCQA, n.d.-b), leaving substantial room for improvement that may be accomplished by educating the general public to expect their primary care provider to offer a basic set of sexual health services.
As discussed in Chapter 6, the widespread implementation of EHRs in primary care settings is a promising tool in completing sexual health assessments and expanding STI screening, including opt-out testing for selected persons. For example, research at Phoenix Indian Medical Center (the largest urban health care provider for American Indians in Arizona, which is also an Indian Health Service [IHS] facility) demonstrated increases in gonorrhea case finding and positivity after increasing provider training, EHR reminder prompts, and bundled laboratory orders (Patton et al., 2016). The latter two are scalable interventions, particularly in IHS facilities that use a common information technology platform, but
need to be combined with other interventions in order to be sustainable and combat alert fatigue (Patton et al., 2016). Research at Phoenix Indian Medical Center also indicates the potential effectiveness of other possible interventions, including incorporating STI screening into routine clinical testing (Taylor et al., 2011) and conducting EPT (Taylor et al., 2013), to increase STI screening rates. Systems-level interventions, including computerized test reminders to enhance chlamydia screening, however, have not been successful across the board (Hocking et al., 2018; Scholes et al., 2006). Therefore, while some projects have demonstrated proof of concept (Taylor et al., 2016), well-designed demonstration projects are needed to develop best practices in the appropriate use of EHRs for STI prevention and examine the challenges with lack of interoperability of data systems. In this context, opt-out testing (e.g., routine chlamydia screening for certain age categories, regardless of risk) should also be considered, as discussed previously.
Primary care providers can also provide on-demand testing for patients who have a specific concern about STIs even though they may not be symptomatic. A request for on-demand testing is an important cue for a detailed sexual health assessment. Some primary care providers may offer diagnostic POC testing for those with symptoms (e.g., microscopy of vaginal discharge samples that may indicate etiologic treatment for yeast infections, trichomoniasis, and bacterial vaginosis). Where such diagnostics are not available, women with vaginal discharge would be treated syndromically. Unfortunately, Gram-stained smears of urethral discharge specimens in men with urethritis are typically unavailable in primary care settings. The future availability of easy-to-use and affordable POC tests (as discussed in Chapter 7) holds great promise for the improvement of etiologic treatment in primary care settings.
Finally, primary care providers need to have easy access to resources that offer technical assistance in implementing appropriate STI services, clinical consultation, and referral. Ideally, such resources would be available locally or regionally (see Chapter 11). Meanwhile, the online STD Clinical Consultation Network, developed by the National Network of Clinical STD Prevention Training Centers, is available to all U.S. clinical providers (Caragol et al., 2017).
STI Specialty Care
As outlined in this chapter, a wide and growing array of clinics, venues, and other resources exist for STI testing. However, STI specialty clinics will continue to play a critical and central role in the STI prevention and control infrastructure (ASTDA Board of Directors, 2020). First, while access to care has increased in certain jurisdictions, many people continue
to seek care at STI clinics for reasons that include clinical expertise, confidentiality, easy walk-in access, and cost. Second, due to increasing patient volume and greater focus on STIs, many STI clinics have developed into specialty clinics that offer consultation to and referral from providers in the region. Third, STI specialty clinics are important to sentinel surveillance (Rietmeijer et al., 2009), STI research, and workforce development (Stoner et al., 2019). Finally, in contrast to other STI service providers, publicly funded STI specialty clinics have a primary STI control mission and could thus play a central role in regional control and prevention efforts. A number of examples in various U.S. geographic regions are doing so and could serve as models to expand such clinics across the nation (e.g., see Barrow et al., 2020). Unfortunately, existing levels of financial support will not be sufficient to address these needs, and alternative funding streams must be developed to expand the provision of STI specialty care to all cities and high-morbidity nonurban regions of the United States.
Family Planning
Women accessing family planning services are typically young and sexually active and are therefore an important target for opportunistic STI screening, especially for chlamydia, gonorrhea, and syphilis. Nearly all publicly supported family planning clinics offered testing for chlamydia and gonorrhea (98 percent) and STI treatment (97 percent) in 2015 (Guttmacher Institute, 2019a), but only 52 percent of users were actually tested for chlamydia in 2018, including 61 percent of women under the age of 25 (OPA, 2019). In addition, more than 90 percent of publicly funded family planning clinics offer HPV vaccination, but data on actual coverage are lacking. Family planning clinics are encouraged to expand their services to male clients; however, men only accounted for about 13 percent of visitors in 2018 (OPA, 2019). An important consideration, however, is how to implement such an expansion in light of new Title X restrictions (discussed in Chapter 4), as facilities receiving these funds can no longer provide counseling on pregnancy termination, which has led a number of grantees and clinics, including Planned Parenthood clinics, to decline Title X funding altogether, limiting the services that they can provide (KFF, 2019b).
The committee finds that family planning clinics play an important role in diagnosing, treating, and preventing STIs among their clients. As is discussed in Chapter 4, funding for these programs has not kept up with inflation, and, despite their critical role serving their communities, the number of clients they have served has fallen in the past decade (OPA, 2020). In other parts of this report, the paucity of STI services for men has been described as an important deficit in current U.S. STI control
programs. While their focus on women is vital and not to be undermined, family planning clinics need to be encouraged to further expand their services to male clients and train their staff accordingly. The large network of clinics employs a sizable cadre of providers who are well trained in providing confidential and sensitive services, including STI care, which could be an important foundation to expand the availability of STI services to not only traditional family planning populations but also broader communities who are disproportionately affected by STIs. Many of these clinics are already invested in serving socially and financially marginalized communities and could be used to address the current dearth of services available to individuals who are at high risk for exposure to STIs but lack access and coverage.
Emergency Departments
ED patients are disproportionally affected by STIs. Consequently, people are increasingly visiting EDs for STI care and treatment, especially if other STI services, such as STI clinics, are not easily accessible (Batteiger et al., 2019). Ease of access and perceptions of clinical expertise may be additional reasons to visit EDs for nonurgent purposes (Moskop, 2010). Interventions have sought to capitalize on these developments. For example, building on an existing opt-out HIV testing program, researchers at the University of Chicago developed an ED-based routine syphilis testing intervention in a region of the city with high syphilis incidence. With automatic order sets and close collaborations with local sexual health/STI clinics and the health department, large numbers of tests are performed with high rates of newly diagnosed cases unrelated to the reason for the visit (Stanford et al., 2020). Eckman et al. (2021) recently found that targeted screening and universally offered screening are both cost-effective strategies for identifying chlamydial and gonococcal infections in adolescents and young adults who access acute care at EDs. Thus, some see EDs as an important and promising access point for STI care and advocate for expanding such services and improving STI skills among ED staff. On the other hand, providing nonurgent care in the ED is controversial, as such services may be unnecessarily costly and undermine the primary mission of the ED (Moskop, 2010). Further studies are needed to investigate the risks and benefits of the expansion of ED-based STI services and associated costs.
HIV Care and Treatment
Providers of HIV care and treatment have seen a dramatic increase in STIs, specifically gonorrhea and syphilis, among their clientele. While a
large proportion of these infections are detected due to screening guidelines, a rise in symptomatic infections means that HIV care providers are becoming increasingly knowledgeable about non-HIV STIs. As a result, historically HIV-focused care providers have become STI clinical experts, effectively forming the rationale for further development of STI specialty clinics in traditional HIV clinics, as discussed in a previous section of this chapter. In addition, these clinics are increasingly branded as “sexual health” clinics that are fully integrated to address not only STIs and HIV treatment but also family planning, sexual dysfunction, and other related services, such as gender-affirming care for transgender and non-binary community members. Such clinics tend to be early adopters of newer approaches to testing, results notification, and wrap-around services that are key to enabling them to be as client centered as possible. This development needs to be encouraged, and clinics moving in this direction need to enhance their STI services, as stated in the Recommendations for Providing Quality Clinical STD Services (Barrow et al., 2020).
Resources for On-Demand Testing
Asymptomatic persons who feel at risk of an STI can access traditional, clinic-based services to request testing. In addition, the availability of noninvasive NAATs and the proliferation of Internet resources have spawned numerous for-profit and not-for-profit resources for on-demand testing. Some of these services are quite costly, are of dubious quality, and may test for pathogens otherwise not recommended (e.g., Ureaplasma urealyticum or Mycoplasma hominis). An additional challenge is that home collection of STI samples has not been cleared by the Food and Drug Administration for a commercial assay and often may not be accurate, as there is no regulatory oversight or guidance for such venues (Owens et al., 2010).
On the other end of the spectrum, online services, including home testing, have been developed and quality controlled by academic institutions and offered for free as part of public health campaigns (Gaydos et al., 2006). Some STI specialty clinics are developing online portals for home collection and mailing specimens to reduce the clinic burden from asymptomatic patients (Hogenson et al., 2019; Jordan et al., 2020). Restricted clinical access due to COVID-19 has further encouraged the use of mail-in testing programs (Melendez et al., 2020). To the extent that these services provide additional options for persons who would otherwise not receive testing, these developments need to be encouraged, especially if they are integrated into a full spectrum of STI prevention and care. Persons with STI symptoms, however, need to be discouraged from using these services and instead encouraged to seek clinical care.
School-/College-/University-Based Health
Gonorrhea and chlamydia prevalence are highest among adolescents and young adults aged 15–25 (CDC, 2019b). According to the 2017 Youth Risk Behavioral Survey, 41.4 percent of male and 37.7 percent of female high school students report ever having had sexual intercourse (Kann et al., 2018). These data provide a rationale for opportunistic testing among sexually active high school and college/university students. A 2016 literature review on STI screening guidance concludes that school-based programs are a feasible and cost-effective way to test large numbers of adolescents for chlamydia and gonorrhea and provide counseling and treatment to almost everyone with an infection (Lewis et al., 2016). STI testing coverage in educational venues varies and depends on the policies of individual school districts and other educational organizations. As detailed in Chapters 8 and 12, parents’ and educators’ involvement is critical to develop a comprehensive sexual health discourse in educational settings and create and implement school-based STI prevention programs, including regular STI screening and HPV vaccination.
Correctional Facilities
Based on the evidence that chlamydia and gonorrhea rates are generally higher among persons in correctional facilities, especially juvenile detention centers, than among demographically similar individuals in the general community, CDC recommends that women and men up to ages 35 or 30, respectively, in correctional facilities be screened for both diseases at entry, if symptomatic/following exposure, and at discharge (CDC, 2015b). The National Commission on Correctional Health Care issued a similar position statement, recommending chlamydia/gonorrhea testing for women up to age 25 and, when possible, age 35, pregnant women regardless of age, and men up to age 30 (NCCHC, 2014). Studies indicate that correctional screening programs may have a positive impact on the community (Barry et al., 2009). Actual coverage (i.e., the number of persons tested across detention centers), however, is not well known. A study by CDC published in 2014 covering 126 geographically diverse juvenile detention centers indicated an overall screening rate of 55.2 percent and an overall chlamydia positivity rate of 14.7 percent among 149,923 women tested (Satterwhite et al., 2014).
An important challenge of STI testing and treatment in correctional settings, especially jails and, to some extent, juvenile detention centers, is the rapid turnover of detained persons, as many will have left the facility when their test results become available. The future availability and implementation of POC testing will thus be particularly meaningful in
these settings. (See Chapters 3 and 9 for more information on STIs within criminal justice–involved populations.)
Military Personnel Health Care Facilities
The demographic composition of military personnel, including age, race and ethnicity, and social background, as well as intensive social interaction and housing, disproportionally favors STI transmission (Deiss et al., 2016; Gaydos and Gaydos, 2008) (see Chapter 3 for more information on STI rates and drivers). Sexuality in the military is a taboo subject, as it is seen as undermining morale and cohesion in the ranks even though the military is becoming increasingly diverse with regard to gender identity and sexual orientation. The high prevalence of STIs among military personnel, and the many reports of sexual coercion and violence, has prompted an ongoing discourse on sexual health in the military (Deiss et al., 2016; Schuyler et al., 2017). The military does offer universal health services, including STI care and prevention, through military health care facilities and the Veterans Health Administration system for active and discharged personnel. These care systems, as discussed in greater detail in Chapter 3, present an important opportunity for screening and periodic sexual health assessment, including of sexual safety. Thus, while continuing sexual health discourse is a priority for the entire military community, military health care providers need to be a driving force in initiating and supporting this discourse and, through proactive STI screening, can significantly impact personnel’s STI epidemiology.
Integration of STI Services: The Role of Public Health
A positive perspective on the wide array of STI care services outlined above would be described as a mosaic composed of complementary pieces. A more critical eye might see a fragmented system that, without an integrating framework, produces duplication of efforts, inefficiencies, and, most importantly, gaps in coverage and services for those populations with the greatest needs. The overarching challenge is that most STI care providers do not have STI prevention as their primary mission; for many, it is overtaken by more pressing health issues and behaviors, including nutrition, substance abuse, physical activity, and now COVID-19. Ideally, public health would serve as the indispensable glue that holds all the pieces together. Unfortunately, public health is fragmented as well; federal public funding for STI prevention through block grants to states and some local jurisdictions is primarily devoted to surveillance, epidemiology, and disease intervention, while STI diagnosis and treatment has traditionally been seen as a responsibility of local health jurisdictions.
A holistic approach that incorporates structural, societal, and community perspectives, as espoused throughout this report, needs to be applied to organize STI care and prevention. Decision-making collaboratives could be developed on the basis of a number of principles. First, they need to have a local or regional focus to address local epidemiological patterns and priority populations. Second, they need to be led by organizations that have a primary STI-focused mission and can be held accountable for the collaborative’s implementation and outcomes. Such leading partners include state and local public health departments, specialty clinics, and STI prevention training centers. Third, these collaboratives need to engage a wide array of stakeholders, including care providers, public health representatives, community leaders, and activists. Their center of gravity will vary depending on local resources and STI leadership. Collaboratives could form around an STI specialty clinic with strong ties to the community or evolve from HIV care centers, family planning clinics, or community clinics that have expanded into STI specialty care. The collaborative’s functions could include assessment (epidemiological and availability of and gaps in services); policy development and advocacy; and assurance of services, including developing new service arrangements if deemed necessary.
This idea of STI collaboratives is not new. A number of them have formed in recent years (e.g., St. Louis STI Regional Response Team), and CDC has experience with a similar concept through the Community Approaches to Reduce STDs projects. Since 2011, 3 grant cycles have funded 12 sites, including Los Angeles, Baltimore, and Chicago (CDC, 2020a). Published experiences from the first two funding cycles suggest a number of key elements in developing community engagement for STI prevention: commitment to engagement, partner flexibility, talented and trusted leadership, participation from diverse sectors, establishing a clear vision and mission, open communication, minimizing power differentials, working through conflict, identifying and leveraging resources, and building a shared history (Rhodes et al., 2020). Experiences from these projects, whether grassroots or CDC funded, need to be further evaluated and leveraged as a guideline for further rollout. Specific community- and structural-level interventions discussed in Chapter 9 can be deployed to build and support critical community engagement in the development of such collaboratives.
In this context, it is important to point out that community planning has been a long-standing feature of national HIV care and prevention efforts, through the Ryan White HIV/AIDS Program, HIV Prevention Community Planning, and, more recently, the Ending the HIV Epidemic Initiative. These efforts are much more heavily resourced than community-based STI initiatives, and integrating both STI and HIV prevention
in a combined community planning initiative may make good sense from a fiscal perspective and avoid duplicating efforts.
As described in Chapter 11, the committee envisions creating STI Resource Centers to provide technical assistance to such community collaboratives and easily accessible clinical consultation to STI care providers. These centers would operate on the level of states or large jurisdictions and include an array of STI experts, including epidemiologists, DIS, and clinical providers, with support from the regional STI Clinical Prevention Training Centers.
CONCLUSIONS AND RECOMMENDATION
Conclusion 10-1: Effective public health efforts to control the transmission of infectious diseases should benefit the whole population, yet many people are not reached by the current fragmented health care system. Many individuals experience financial barriers to STI screening and treatment services because of their insurance status, immigration status, or ability to afford copayments and/or coinsurance for treatment and services. The committee supports ongoing efforts to broaden no-cost care options for STI prevention and treatment to all individuals.
Conclusion 10-2: As recommended in the 2015 STD Treatment Guidelines, expedited EPT is an important evidence-based tool in the treatment of contacts of individuals with chlamydia, trichomonas, and, to a lesser extent, gonorrhea. The barriers to implementation of EPT, such as financial obstacles (lack of insurance coverage) and ambiguity about the legal status of state policies regarding EPT, however, have limited the use of this tool. In addition, further research is needed to develop approaches to better use EPT in certain populations, such as men who have sex with men.
Conclusion 10-3: There are notable gaps in measures that assess program and plan performance in addressing STIs in populations and subpopulations at risk. In particular, measures that track performance in screening, testing, and treatment services for men are needed, as well as for MSM and other gender expansive populations. The addition of these new measures could provide incentives for the development of payment bundles that could improve the scope and quality of sexual health services offered by plans and programs.
Conclusion 10-4: The ongoing transition of local STI clinics to comprehensive sexual health clinics taking place in some jurisdictions in the United States is an important trend that needs to be supported and accelerated.
Conclusion 10-5: The Title X family planning program provides an important foundation for STI services to low-income women. Its current structure could be built on to expand access to sexual health more broadly and specifically to improve the availability of STI services to men, including heterosexual as well as LGBTQ and other gender nonconforming individuals. The current program, however, is not adequately resourced or structured to serve this function. While the focus on women and contraception is critical, a broader lens on sexual health in Title X and among the providers who participate could open up the pathway for care to populations who are disproportionately affected by STIs and have limited care options.
Conclusion 10-6: Targeted initiatives for creating new, improved, and easier care access points for sexual health care are needed. Promising initiatives include readily accessed STI screening and treatment services venues, such as urgent care centers and pharmacies. Access points could also include rapid and on-demand testing sites, sexual and gender diverse clinics, and emergency departments. Title X sites, family planning clinics, and FQHCs could provide the foundation for these expanded and specialized services. Long-term sustainability plans and new financing models to support these services are also needed.
Recommendation 10-1: The Department of Health and Human Services and state governments should identify and support innovative programs to ensure that sexually transmitted infection (STI) prevention and treatment services are available through multiple venues and ensure that federal and state governments maximize access opportunities for individuals who face health care access barriers. Priority populations for these efforts should include persons ineligible for coverage, persons who face affordability barriers (including high out-of-pocket costs), and persons who will not access STI services without confidentiality guarantees (such as adolescents and young adults with insurance coverage through parents or guardians).
While some changes may require greater consideration, prompt exploration of new initiatives could include the following:
- Incentives to expand full scope Medicaid or other well-subsidized options to fill gaps in coverage, particularly in states that have not opted to expand Medicaid or not fully enrolled eligible populations.
- Special Medicaid-funded programs, such as developing guidance to permit the expansion of Section 1115 Medicaid family planning waivers or state plan amendments to include those who do not
- otherwise qualify for Medicaid because of their state of residence, immigration status, and/or other factors.
- Establishment of a federally supported supplemental grant program to provide funds to FQHCs, family planning providers, state health departments, Title X providers, and STI providers to offer STI testing and treatment services (including EPT) at no cost for those who are uninsured or underinsured; to support outreach, for the services that they offer; and to facilitate the expansion of a broader range of sexual health services at those sites.
- Promulgation of new 340B drug pricing program guidance that deems the provision of STI prevention and treatment medications (including population-based EPT services and HIV PrEP services) to be allowable uses of program income.
- New funding initiatives to support and expand access to comprehensive sexual health clinics and create new access points for sexual health services in a variety of settings and venues.
CONCLUDING OBSERVATIONS
STI clinical services are available from a large array of clinical specialties, with the level of services varying by specialty, from basic testing and treatment in primary care settings to in-depth evaluation, diagnosis, and management of complicated cases in STI specialty clinics. The committee recognizes the unique contribution that each of these specialties brings to STI diagnosis, treatment, and prevention. Emerging opportunities, including online testing and increased involvement of nontraditional providers, hold promise in further strengthening the STI care system. There are many missed opportunities for sexual health assessment and STI evaluation in many settings, however, and better coordination of private and public STI services at the local level would move the field from fragmentation to integration, with better overall outcomes for STI prevention and control. Public and private insurance coverage options are important sources of payments for STI prevention, screening, and treatment services, but gaps in access to coverage and coverage of STI services persist. Many people in the United States are uninsured or underinsured, have no pathways to affordable coverage, and therefore rely on a patchwork of providers to obtain care. The committee finds that the scope of coverage for preventive STI services established under the ACA is extensive, but there are few recommendations for screening, testing, and treatment services for men. In light of these coverage gaps, the committee recognizes that policy changes addressing both who is eligible for coverage and the scope of that coverage could reduce barriers to STI services that are accessible and affordable.
REFERENCES
AAP (American Academy of Pediatrics). 2016. Confidentiality protections for adolescents and young adults in the health care billing and insurance claims process (statement of endorsement). Pediatrics 137(5):e20160593.
AIDS United and (Medical Advocacy and Outreach) of Alabama. 2017. Making connections: A guide to starting a rural HIV care telemedicine program. Washington, DC: AIDS United and Medical Advocacy and Outreach.
Albright, C. M., J. B. Emerson, E. F. Werner, and B. L. Hughes. 2015. Third-trimester prenatal syphilis screening: A cost-effectiveness analysis. Obstetrics & Gynecology 126(3).
ASTDA (American Sexually Transmitted Diseases Association) Board of Directors. 2020. ASTDA position statement: In support of the publicly-funded sexually transmitted infections specialty clinic. Sexually Transmitted Diseases 47(8):503-505.
Bachmann, L., P. Thorpe, G. Bolan, and J. Mermin. 2020. Dear colleague letter STD treatment options. https://www.cdc.gov/std/dstdp/DCL-STDTreatment-COVID19-04062020.pdf (accessed November 4, 2020).
Barrow, R. Y., F. Ahmed, G. A. Bolan, and K. A. Workowski. 2020. Recommendations for providing quality sexually transmitted diseases clinical services, 2020. MMWR Recommendations and Reports 68(5):1-20.
Barry, P. M., C. K. Kent, K. C. Scott, J. Goldenson, and J. D. Klausner. 2009. Is jail screening associated with a decrease in chlamydia positivity among females seeking health services at community clinics?—San Francisco, 1997-2004. Sexually Transmitted Diseases 36(Suppl 2):S22-S28.
Batteiger, T. A., B. E. Dixon, J. Wang, Z. Zhang, G. Tao, Y. Tong, W. Tu, S. A. Hoover, and J. N. Arno. 2019. Where do people go for gonorrhea and chlamydia tests: A cross-sectional view of the Central Indiana population, 2003-2014. Sexually Transmitted Diseases 46(2):132-136.
Brookmeyer, K. A., A. Coor, R. E. Kachur, O. Beltran, H. E. Reno, and P. J. Dittus. 2020. Sexual history taking in clinical settings: A narrative review. Sexually Transmitted Diseases. doi: 10.1097/OLQ.0000000000001319.
Caragol, L. A., K. A. Wendel, T. S. Anderson, H. C. Burnside, A. Finkenbinder, J. D. Fitch, D. H. Kelley, T. W. Stewart, M. Thrun, and C. A. Rietmeijer. 2017. A new resource for STD clinical providers: The Sexually Transmitted Diseases Clinical Consultation Network. Sexually Transmitted Diseases 44(8):510-512.
Carnevale, C., P. Richards, R. Cohall, J. Choe, J. Zitaner, N. Hall, A. Cohall, S. Whittier, D. A. Green, M. E. Sobieszczyk, P. Gordon, and J. Zucker. 2020. At home testing for sexually transmitted infections during the COVID-19 pandemic. Sexually Transmitted Diseases 48(1):e11-e14.
CDC (Centers for Disease Control and Prevention). 2011. 2011 performance measures: Quick reference guide. https://www.cdc.gov/std/program/2011PerformanceMeasuresQuickGuidel.pdf (accessed January 25, 2021).
CDC. 2015a. 2015 sexually transmitted diseases treatment guidelines. https://www.cdc.gov/std/tg2015/default.htm (accessed October 22, 2019).
CDC. 2015b. Sexually transmitted diseases treatment guidelines, 2015. MMWR Recommendations and Reports 64(3).
CDC. 2018. U.S. public health service: Preexposure prophylaxis for the prevention of HIV infection in the United States—2017 update: A clinical practice guideline. Atlanta, GA: Centers for Disease Control and Prevention.
CDC. 2019a. HPV vaccine schedule and dosing. https://www.cdc.gov/hpv/hcp/schedules-recommendations.html (accessed June 15, 2020).
CDC. 2019b. Sexually transmitted disease surveillance 2018. Atlanta, GA: Department of Health and Human Services.
CDC. 2020a. Community approaches to reducing sexually transmitted diseases. https://www.cdc.gov/std/health-disparities/cars.htm (accessed February 21, 2021).
CDC. 2020b. Expedited partner therapy. https://www.cdc.gov/std/ept/default.htm#:~:text=The%20usual%20alternative%20is%20to,refer%20their%20partners%20for%20treatment.&text=CDC%20has%20concluded%20that%20EPT,with%20chlamydial%20infection%20or%20gonorrhea (accessed December 8, 2020).
CDC. 2020c. Legal status of expedited partner therapy (EPT). https://www.cdc.gov/std/ept/legal/default.htm (accessed February 10, 2021).
CDC. 2020d. Sexually transmitted disease preventive services coverage. https://www.cdc.gov/nchhstp/highqualitycare/preventiveservices/std.html (accessed May 5, 2020).
CDC. 2020e. State statutory and regulatory language regarding prenatal syphilis screenings in the United States, 2018. https://www.cdc.gov/std/treatment/prenatal-syphilis-screening-laws-web-document-cleared-01-24-2020.pdf (accessed February 1, 2021).
Chambers, L. C., L. E. Manhart, D. A. Katz, M. R. Golden, L. A. Barbee, and J. C. Dombrowski. 2017. Evaluation of an automated express care triage model to identify clinically relevant cases in a sexually transmitted disease clinic. Sexually Transmitted Diseases 44(9).
Chambers, L. C., L. E. Manhart, D. A. Katz, M. R. Golden, L. A. Barbee, and J. C. Dombrowski. 2018. Comparison of algorithms to triage patients to express care in a sexually transmitted disease clinic. Sexually Transmitted Diseases 45(10):696-702.
Cheng, J. Q., H. Zhou, F. C. Hong, D. Zhang, Y. J. Zhang, P. Pan, and Y. M. Cai. 2007. Syphilis screening and intervention in 500,000 pregnant women in Shenzhen, the People’s Republic of China. Sexually Transmitted Infections 83(5):347-350.
CMS (Centers for Medicare & Medicaid Services). n.d. Screening for sexually transmitted infections (STIs) & high intensity behavioral counseling (HIBC) to prevent STIs (NCD 210.10). https://www.cms.gov/Medicare/Prevention/PrevntionGenInfo/medicarepreventive-services/MPS-QuickReferenceChart-1.html#STI (accessed December 8, 2020).
Daw, J. R., K. B. Kozhimannil, and L. K. Admon. 2019. High rates of perinatal insurance churn persist after the ACA. Health Affairs Blog (September 16). https://www.healthaffairs.org/do/10.1377/hblog20190913.387157/full (accessed February 22, 2021).
Deiss, R., R. J. Bower, E. Co, O. Mesner, J. L. Sanchez, J. Masel, A. Ganesan, G. E. Macalino, and B. K. Agan. 2016. The association between sexually transmitted infections, length of service and other demographic factors in the U.S. military. PLoS One 11(12):e0167892.
Douglas, J. 2015. Dear colleague letter—regarding the use of expedited partner therapy. https://www.cdc.gov/std/dearcolleagueept5-10-05.pdf (accessed February 10, 2021).
Dreisbach, S., H. Burnside, K. Hsu, L. Smock, P. Coury-Doniger, C. Hall, J. Marrazzo, G. Nagendra, C. Rietmeijer, A. Rompalo, and M. Thrun. 2014. Improving HIV/STD prevention in the care of persons living with HIV through a national training program. AIDS Patient Care and STDS 28(1):15-21.
Eckert, E. 2020. It’s past time to provide continuous Medicaid coverage for one year postpartum. Health Affairs Blog (February 6). https://www.healthaffairs.org/do/10.1377/hblog20200203.639479/full (accessed February 22, 2021).
Eckman, M. H., J. L. Reed, M. Trent, and M. K. Goyal. 2021. Cost-effectiveness of sexually transmitted infection screening for adolescents and young adults in the pediatric emergency department. JAMA Pediatrics 175(1):81-89.
Elwyn, G., C. Dehlendorf, R. Epstein, K. Marrin, J. White, and D. Frosch. 2014. Shared decision making and motivational interviewing: Achieving patient-centered care across the spectrum of health care problems. Annals of Family Medicine 12:270-275.
FPNTC (Family Planning National Training Center). 2017. 340B drug pricing program frequently asked questions for Title X family planning agencies. https://www.fpntc.org/sites/default/files/resources/340b_faq_2017-12-08.pdf (accessed June 10, 2020).
Gaydos, C. A., and J. C. Gaydos. 2008. Chlamydia in the United States military: Can we win this war? Sexually Transmitted Diseases 35(3):260-262.
Gaydos, C. A., K. Dwyer, M. Barnes, P. A. Rizzo-Price, B. J. Wood, T. Flemming, and M. T. Hogan. 2006. Internet-based screening for Chlamydia trachomatis to reach non-clinic populations with mailed self-administered vaginal swabs. Sexually Transmitted Diseases 33(7):451-457.
Gaydos, C. A., M. C. Ako, M. Lewis, Y. H. Hsieh, R. E. Rothman, and A. F. Dugas. 2019. Use of a rapid diagnostic for Chlamydia trachomatis and Neisseria gonorrhoeae for women in the emergency department can improve clinical management: Report of a randomized clinical trial. Annals of Emergency Medicine 74(1):36-44.
Gaydos, C. A., M. Barnes, J. Holden, B. Silver, R. Smith, J. Hardick, and T. C. Quinn. 2020. Acceptability and feasibility of recruiting women to collect a self-administered vaginal swab at a pharmacy clinic for sexually transmissible infection screening. Sexual Health 17(4):392-394.
Gift, T. L., K. M. Cuffe, and J. S. Leichliter. 2018. The impact of budget cuts on sexually transmitted disease programmatic activities in state and local health departments with staffing reductions in fiscal year 2012. Sexually Transmitted Diseases 45(11):e87-e89.
Gleeson, C., M. Robinson, and R. Neal. 2002. A review of teenagers’ perceived needs and access to primary health care: Implications for health services. Primary Health Care Research & Development 3:184-193.
Golden, M. R., W. L. Whittington, H. H. Handsfield, J. P. Hughes, W. E. Stamm, M. Hogben, A. Clark, C. Malinski, J. R. Helmers, K. K. Thomas, and K. K. Holmes. 2005. Effect of expedited treatment of sex partners on recurrent or persistent gonorrhea or chlamydial infection. New England Journal of Medicine 352(7):676-685.
Golden, M. R., R. P. Kerani, M. Stenger, J. P. Hughes, M. Aubin, C. Malinski, and K. K. Holmes. 2015. Uptake and population-level impact of expedited partner therapy (EPT) on Chlamydia trachomatis and Neisseria gonorrhoeae: The Washington State community-level randomized trial of EPT. PLoS Medicine 12(1):e1001777.
Guttmacher Institute. 2019a. Fact sheet: Publicly supported family planning services in the United States. https://www.guttmacher.org/fact-sheet/publicly-supported-FP-services-US (accessed November 17, 2019).
Guttmacher Institute. 2019b. Minors’ access to STI services. https://www.guttmacher.org/state-policy/explore/minors-access-sti-services (accessed December 23, 2020).
Harshbarger, C. L., L. N. O’Donnell, L. Warner, A. D. Margolis, D. B. Richardson, S. R. Novey, L. C. Glover, J. D. Klausner, C. K. Malotte, and C. A. Rietmeijer. 2012. Safe in the city: Effective prevention interventions for human immunodeficiency virus and sexually transmitted infections. American Journal of Preventative Medicine 42(5):468-472.
Hartnett, K. P., A. Kite-Powell, J. DeVies, M. A. Coletta, T. K. Boehmer, J. Adjemian, A. V. Gundlapalli, and National Syndromic Surveillance Program Community of Practice. 2020. Impact of the COVID-19 pandemic on emergency department visits—United States, January 1, 2019-May 30, 2020. Morbidity and Mortality Weekly Report 69:699-704.
Heijman, T. L., A. K. Van der Bij, H. J. De Vries, E. J. Van Leent, H. F. Thiesbrummel, and H. S. Fennema. 2007. Effectiveness of a risk-based visitor-prioritizing system at a sexually transmitted infection outpatient clinic. Sexually Transmitted Diseases 34(7):508-512.
Hersh, A. R., C. J. Megli, and A. B. Caughey. 2018. Repeat screening for syphilis in the third trimester of pregnancy: A cost-effectiveness analysis. Obstetrics and Gynocology 132(3):699-707.
Hill, H. A., L. D. Elam-Evans, D. Yankey, J. A. Singleton, and Y. Kang. 2018. Vaccination coverage among children aged 19-35 months—United States, 2017. Morbidity and Mortality Weekly Report (67):1123-1128.
Hocking, J. S., M. Temple-Smith, R. Guy, B. Donovan, S. Braat, M. Law, J. Gunn, D. Regan, A. Vaisey, L. Bulfone, J. Kaldor, C. K. Fairley, and N. Low. 2018. Population effectiveness of opportunistic chlamydia testing in primary care in Australia: A cluster-randomised controlled trial. Lancet 392(10156):1413-1422.
Hogben, M., D. Collins, B. Hoots, and K. O’Connor. 2016. Partner services in sexually transmitted disease prevention programs: A review. Sexually Transmitted Diseases 43(2 Suppl 1):S53-S62.
Hogenson, E., M. Jett-Goheen, and C. A. Gaydos. 2019. An analysis of user survey data for an Internet program for testing for sexually transmitted infections, I Want the Kit, in Maryland and Washington, DC. Sexually Transmitted Diseases 46(12):768-770.
Hook, E. W., 3rd, C. Spitters, C. A. Reichart, T. M. Neumann, and T. C. Quinn. 1994. Use of cell culture and a rapid diagnostic assay for Chlamydia trachomatis screening. JAMA 272(11):867-870.
Jamison, C. D., T. Chang, and O. Mmeje. 2018. Expedited partner therapy: Combating record high sexually transmitted infection rates. American Journal of Public Health 108(10):1325-1327.
Jamison, C. D., J. S. Coleman, and O. Mmeje. 2019. Improving women’s health and combat-ting sexually transmitted infections through expedited partner therapy. Obstetrics & Gynecology 133(3):416-422.
Jenness, S. M., K. M. Weiss, S. M. Goodreau, T. Gift, H. Chesson, K. W. Hoover, D. K. Smith, A. Y. Liu, P. S. Sullivan, and E. S. Rosenberg. 2017. Incidence of gonorrhea and chlamydia following human immunodeficiency virus preexposure prophylaxis among men who have sex with men: A modeling study. Clinical Infectious Diseases 65(5):712-718.
Jordan, N. N., M. Jett-Goheen, Y. H. Hsieh, J. C. Gaydos, and C. A. Gaydos. 2020. Detection of three sexually transmitted infections by anatomic site: Evidence from an Internet-based screening program. Sexually Transmitted Diseases 47(4):243-245.
Kamb, M. L., M. Fishbein, J. M. Douglas, F. Rhodes, J. Rogers, G. Bolan, J. Zenilman, T. Hoxworth, C. K. Malotte, M. Iatesta, C. Kent, A. Lentz, S. Graziano, R. H. Byers, and T. A. Peterman. 1998. Efficacy of risk-reduction counseling to prevent human immunodeficiency virus and sexually transmitted diseases: A randomized controlled trial. Project respect study group. JAMA 280(13):1161-1167.
Kann, L., T. McManus, W. A. Harris, S. L. Shanklin, K. H. Flint, B. Queen, R. Lowry, D. Chyen, L. Whittle, J. Thornton, C. Lim, D. Bradford, Y. Yamakawa, M. Leon, N. Brener, and K. A. Ethier. 2018. Youth risk behavior surveillance—United States, 2017. Morbidity and Mortality Weekly Report: Surveillance Summaries 67(8):1-114.
KFF (Kaiser Family Foundation). 2015. Preventive services covered by private health plans under the Affordable Care Act. https://www.kff.org/health-reform/fact-sheet/preventive-services-covered-by-private-health-plans (accessed December 23, 2020).
KFF. 2019a. Health insurance coverage of the total population. https://www.kff.org/other/state-indicator/total-population/?activeTab=map¤tTimeframe=0&selectedDistributions=uninsured&sortModel=%7B%22colId%22:%22Location%22,%22sort%22:%22asc%22%7D (accessed February 22, 2021).
KFF. 2019b. The status of participation in the Title X federal family planning program. https://www.kff.org/interactive/the-status-of-participation-in-the-title-x-federal-family-planning-program (accessed July 9, 2020).
KFF. 2020a. Health coverage of immigrants. https://www.kff.org/racial-equity-and-health-policy/fact-sheet/health-coverage-of-immigrants (accessed December 23, 2020).
KFF. 2020b. Preventive services tracker. https://www.kff.org/report-section/preventive-service-tracker-sexual-health (accessed February 22, 2021).
KFF. 2020c. Sexually transmitted infections (STIs): An overview, payment, and coverage. https://www.kff.org/womens-health-policy/fact-sheet/sexually-transmitted-infections-stisan-overview-payment-and-coverage (accessed December 23, 2020).
KFF. 2020d. States that have expanded eligibility for coverage of family planning services under Medicaid. https://www.kff.org/medicaid/state-indicator/family-planning-services-waivers/?currentTimeframe=0&sortModel=%7B%22colId%22:%22State%20Has%20Secured%20a%20Waiver%20or%20State%20Plan%20Amendment%20(SPA)%20from%20CMS%20to%20Cover%20Services%22,%22sort%22:%22asc%22%7D (accessed January 25, 2021).
KFF. 2020e. Status of state Medicaid expansion decisions: Interactive map. https://www.kff.org/medicaid/issue-brief/status-of-state-medicaid-expansion-decisions-interactive-map/#:~:text=To%20date%2C%2039%20states%20(including,available%20in%20a%20table%20format (accessed November 18, 2020).
Kissinger, P., H. Mohammed, G. Richardson-Alston, J. S. Leichliter, S. N. Taylor, D. H. Martin, and T. A. Farley. 2005. Patient-delivered partner treatment for male urethritis: A randomized, controlled trial. Clinical Infectious Diseases 41(5):623-629.
Koonin, L. M., B. Hoots, C. A. Tsang, Z. Leroy, K. Farris, T. Jolly, P. Antall, B. McCabe, C. B. R. Zelis, I. Tong, and A. M. Harris. 2020. Trends in the use of telehealth during the emergence of the COVID-19 pandemic—United States, January-March 2020. Morbidity and Mortality Weekly Report (69):1595-1599.
Leichliter, J. S., C. Copen, and P. J. Dittus. 2017. Confidentiality issues and use of sexually transmitted disease services among sexually experienced persons aged 15-25 years—United States, 2013-2015. Morbidity and Mortality Weekly Report 66(9):237-241.
Lewis, F. M., P. Dittus, M. E. Salmon, and M. J. Nsuami. 2016. School-based sexually transmitted disease screening: Review and programmatic guidance. Sexually Transmitted Diseases 43(2 Suppl 1):S18-S27.
McClung, N. M., R. M. Lewis, J. W. Gargano, T. Querec, E. R. Unger, and L. E. Markowitz. 2019. Declines in vaccine-type human papillomavirus prevalence in females across racial/ethnic groups: Data from a national survey. Journal of Adolescent Health 65(6):715-722.
McKee, M. D., S. E. Rubin, G. Campos, and L. F. O’Sullivan. 2011. Challenges of providing confidential care to adolescents in urban primary care: Clinician perspectives. Annals of Family Medicine 9(1):37.
Medicaid.gov. n.d.-a. Adult health care quality measures. https://www.medicaid.gov/medicaid/quality-of-care/performance-measurement/adult-and-child-health-care-quality-measures/adult-health-care-quality-measures/index.html (accessed November 19, 2020).
Medicaid.gov. n.d.-b. Children’s health care quality measures. https://www.medicaid.gov/medicaid/quality-of-care/performance-measurement/adult-and-child-health-care-quality-measures/childrens-health-care-quality-measures/index.html (accessed November 19, 2020).
Medicare.gov. n.d.-c. Preventive & screening services. https://www.medicare.gov/coverage/preventive-screening-services (accessed December 8, 2020).
Melendez, J. H., M. M. Hamill, G. Armington, C. A. Gaydos, and Y. C. Manabe. 2020. Home-based testing for sexually transmitted infections: Leveraging online resources during the COVID-19 pandemic. Sexually Transmitted Diseases 48(1):e11-e14.
Metsch, L. R., D. J. Feaster, L. Gooden, B. R. Schackman, T. Matheson, M. Das, M. R. Golden, S. Huffaker, L. F. Haynes, S. Tross, C. K. Malotte, A. Douaihy, P. T. Korthuis, W. A. Duffus, S. Henn, R. Bolan, S. S. Philip, J. G. Castro, P. C. Castellon, G. McLaughlin, R. N. Mandler, B. Branson, and G. N. Colfax. 2013. Effect of risk-reduction counseling with rapid HIV testing on risk of acquiring sexually transmitted infections: The AWARE randomized clinical trial. JAMA 310(16):1701-1710.
Miller, W., and S. Rollnick. 1991. Motivational interviewing: Preparing people to change addictive behavior. New York: Guilford Press.
Moskop, J. C. 2010. Nonurgent care in the emergency department—bane or boon? Virtual Mentor 12(6):476-482.
NACCHO (National Association of County and City Health Officials). 2019. STI express services: Increasing access and testing while maximizing resources. https://www.naccho.org/uploads/downloadable-resources/issue-brief_STI-Express-Services.pdf (accessed January 25, 2021).
Nagendra, G., C. Carnevale, N. Neu, A. Cohall, and J. Zucker. 2020. The potential impact and availability of sexual health services during the COVID-19 pandemic. Sexually Transmitted Diseases 47(7):434-436.
NAPA (National Academy of Public Administration). 2018. The impact of sexually transmitted diseases on the United States: Still hidden, getting worse, can be controlled. Washington, DC: National Academy of Public Administration.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2019. Promoting positive adolescent health behavior and outcomes: Thriving in the 21st century. Washington, DC: The National Academies Press.
NCCHC (National Commission on Correctional Health Care). 2014. STD testing for adolescents and adults upon admission to correctional facilities. https://www.ncchc.org/std-testing-upon-admission (accessed November 17, 2019).
NCQA (National Committee for Quality Assurance). n.d.-a. Cervical cancer screening (CCS). https://www.ncqa.org/hedis/measures/cervical-cancer-screening (accessed November 19, 2020).
NCQA. n.d.-b. Chlamydia screening in women. https://www.ncqa.org/hedis/measures/chlamydia-screening-in-women (accessed January 2, 2019).
NCQA. n.d.-c. Immunizations for adolescents (IMA). https://www.ncqa.org/hedis/measures/immunizations-for-adolescents (accessed November 19, 2020).
Newman, L., M. Kamb, S. Hawkes, G. Gomez, L. Say, A. Seuc, and N. Broutet. 2013. Global estimates of syphilis in pregnancy and associated adverse outcomes: Analysis of multinational antenatal surveillance data. PLoS Medicine 10(2):e1001396.
NQF (National Quality Forum). n.d. Measures, Reports & Tools. http://www.qualityforum.org/measures_reports_tools.aspx (accessed November 19, 2020).
Offit, P. A. 2014. Let’s not talk about sex. The New York Times. https://www.nytimes.com/2014/08/20/opinion/lets-not-talk-about-sex-HPV-vaccine.html (accessed November 13, 2020).
Ogale, Y., P. T. Yeh, C. E. Kennedy, I. Toskin, and M. Narasimhan. 2019. Self-collection of samples as an additional approach to deliver testing services for sexually transmitted infections: A systematic review and meta-analysis. BMJ Global Health 4(2):e001349.
OPA (Office of Population Affairs). 2019. Title X family planning annual report—2018 national summary. https://www.hhs.gov/opa/sites/default/files/title-x-fpar-2018-national-summary.pdf (accessed November 17, 2019).
OPA. 2020. Title X family planning annual report: 2019 national summary. Rockville, MD: Office of Population Affairs.
Owens, S. L., N. Arora, N. Quinn, R. W. Peeling, K. K. Holmes, and C. A. Gaydos. 2010. Utilising the Internet to test for sexually transmitted infections: Results of a survey and accuracy testing. Sexually Transmitted Infections 86(2):112-116.
Owusu-Edusei, K., Jr., K. W. Hoover, and T. L. Gift. 2016. Cost-effectiveness of opt-out chlamydia testing for high-risk young women in the U.S. American Journal of Preventive Medicine 51(2):216-224.
Patel, A. V., S. M. Abrams, C. A. Gaydos, M. Jett-Goheen, C. A. Latkin, R. E. Rothman, and Y.-H. Hsieh. 2019. Increasing HIV testing engagement through provision of home HIV self-testing kits for patients who decline testing in the emergency department: A pilot randomisation study. Sexually Transmitted Infections 95(5):358-360.
Patton, M. E., R. D. Kirkcaldy, D. C. Chang, S. Markman, M. Yellowman, E. Petrosky, L. Adams, C. Robinson, A. Gupta, and M. M. Taylor. 2016. Increased gonorrhea screening and case finding after implementation of expanded screening criteria—urban Indian Health Service facility in Phoenix, Arizona, 2011-2013. Sexually Transmitted Diseases 43(6):396-401.
Rac, M. W., P. A. Revell, and C. S. Eppes. 2017. Syphilis during pregnancy: A preventable threat to maternal-fetal health. American Journal of Obstetrics & Gynecology 216(4):352-363.
Ranji, U., Y. Bair, and A. Salganicoff. 2016. Medicaid and family planning: Background and implications of the ACA. https://www.kff.org/report-section/medicaid-and-family-planning-medicaid-family-planning-policy (accessed January 25, 2021).
Ranji, U., A. Salganicoff, L. Sobel, and I. Gomez. 2019. Financing family planning services for low-income women: The role of public programs. https://www.kff.org/womens-health-policy/issue-brief/financing-family-planning-services-for-low-income-women-therole-of-public-programs (accessed January 25, 2021).
Ranji, U., I. Gomez, and A. Salganicoff. 2020. Expanding postpartum Medicaid coverage. https://www.kff.org/womens-health-policy/issue-brief/expanding-postpartum-medicaid-coverage (accessed January 25, 2021).
Rhodes, S. D., J. Daniel-Ulloa, S. S. Wright, L. Mann-Jackson, D. Johnson, N. Hayes, and J. Valentine. 2020. Critical elements of community engagement to address disparities and related social determinants of health: The CDC community approaches to reducing STDs (CARS) initiative. Sexually Transmitted Diseases 48(1):49-55.
Rietmeijer, C. A., J. Donnelly, K. T. Bernstein, J. M. Bissette, S. Martins, P. Pathela, J. A. Schillinger, M. R. Stenger, H. Weinstock, and L. M. Newman. 2009. Here comes the SSuN: Early experiences with the STD Surveillance Network. Public Health Reports 124(Suppl 2):72-77.
Rietmeijer, C. A., B. C. Hauschild, H. C. Burnside, and K. A. Wendel. 2021. Medicaid expansion and rates of reportable sexually transmitted infections in the United States—A county-level analysis. Sexually Transmitted Diseases. E-pub ahead of print.
SAHM (Society for Adolescent Health and Medicine). 2016. Confidentiality protections for adolescents and young adults in the health care billing and insurance claims process. Journal of Adolescent Health 58(3):374-377.
SAHM. 2020. The 21st Century Cures Act & adolescent confidentiality. https://www.adolescenthealth.org/Advocacy/Advocacy-Activities/2019-(1)/NASPAG-SAHM-Statement.aspx (accessed January 26, 2021).
Satterwhite, C. L., D. Newman, D. Collins, and E. Torrone. 2014. Chlamydia screening and positivity in juvenile detention centers, United States, 2009-2011. Women Health 54(8):712-725.
Schillinger, J. A., P. Kissinger, H. Calvet, W. L. Whittington, R. L. Ransom, M. R. Sternberg, S. M. Berman, C. K. Kent, D. H. Martin, M. K. Oh, H. H. Handsfield, G. Bolan, L. E. Markowitz, and J. D. Fortenberry. 2003. Patient-delivered partner treatment with azithromycin to prevent repeated Chlamydia trachomatis infection among women: A randomized, controlled trial. Sexually Transmitted Diseases 30(1):49-56.
Scholes, D., L. Grothaus, J. McClure, R. Reid, P. Fishman, C. Sisk, J. E. Lindenbaum, B. Green, J. Grafton, and R. S. Thompson. 2006. A randomized trial of strategies to increase chlamydia screening in young women. Preventative Medicine 43(4):343-350.
Schuyler, A., S. Kintzle, C. L. Lucas, H. Moore, and C. Castro. 2017. Military sexual assault (MSA) among veterans in Southern California: Associations with physical health, psychological health, and risk behaviors. Traumatology 23(3):223.
Schwebke, J. R., R. Sadler, J. M. Sutton, and E. W. Hook, 3rd. 1997. Positive screening tests for gonorrhea and chlamydial infection fail to lead consistently to treatment of patients attending a sexually transmitted disease clinic. Sexually Transmitted Diseases 24(4):181-184.
Sedlander, E., C. D. Brindis, S. H. Bausch, and K. P. Tebb. 2015. Options for assuring access to confidential care for adolescents and young adults in an explanation of benefits environment. Journal of Adolescent Health 56(1):7-9.
Shamos, S. J., C. J. Mettenbrink, J. A. Subiadur, B. L. Mitchell, and C. A. Rietmeijer. 2008. Evaluation of a testing-only “express” visit option to enhance efficiency in a busy STI clinic. Sexually Transmitted Diseases 35(4):336-340.
Stanford, K. A., A. Hazra, and J. Schneider. 2020. Routine opt-out syphilis screening in the emergency department: A public health imperative. Academic Emergency Medicine 27(5):437-438.
Stekler, J., L. Bachmann, R. M. Brotman, E. J. Erbelding, L. V. Lloyd, C. A. Rietmeijer, H. H. Handsfield, K. K. Holmes, and M. R. Golden. 2005. Concurrent sexually transmitted infections (STIs) in sex partners of patients with selected STIs: Implications for patient-delivered partner therapy. Clinical Infectious Diseases 40(6):787-793.
Stoner, B. P., J. Fraze, C. A. Rietmeijer, J. Dyer, A. Gandelman, E. W. Hook, 3rd, C. Johnston, N. M. Neu, A. M. Rompalo, G. Bolan, and National Network of STD Clinical Prevention Training Centers. 2019. The national network of sexually transmitted disease clinical prevention training centers turns 40—a look back, a look ahead. Sexually Transmitted Diseases 46(8):487-492.
Taylor, M. M., B. Reilley, S. Tulloch, M. Winscott, A. Dunnigan, M. Russell, and J. T. Redd. 2011. Identifying opportunities for chlamydia screening among American Indian women. Sexually Transmitted Diseases 38(10):947-948.
Taylor, M. M., B. Reilley, M. Yellowman, L. Anderson, L. de Ravello, and S. Tulloch. 2013. Use of expedited partner therapy among chlamydia cases diagnosed at an urban Indian Health Centre, Arizona. International Journal of STD & AIDS 24(5):371-374.
Taylor, M. M., J. Frasure-Williams, P. Burnett, and I. U. Park. 2016. Interventions to improve sexually transmitted disease screening in clinic-based settings. Sexually Transmitted Diseases 43(2 Suppl):S28-S41.
Taylor, M. M., M. Kamb, D. Wu, and S. Hawkes. 2017. Syphilis screening and treatment: Integration with HIV services. Bulletin of the World Health Organization 95(9):610.
Tebb, K. P., E. Sedlander, S. Bausch, and C. D. Brindis. 2015. Opportunities and challenges for adolescent health under the Affordable Care Act. Maternal and Child Health Journal 19(10):2089-2093.
Tuckson, R. V., M. Edmunds, and M. L. Hodgkins. 2017. Telehealth. New England Journal of Medicine 377(16):1585-1592.
Tylee, A., D. M. Haller, T. Graham, R. Churchill, and L. A. Sanci. 2007. Youth-friendly primary-care services: How are we doing and what more needs to be done? Lancet 369(9572):1565-1573.
USPSTF (United States Preventive Services Task Force). 2014. Final recommendation statement chlamydia and gonorrhea: Screening. https://www.uspreventiveservicestaskforce.org/uspstf/document/RecommendationStatementFinal/chlamydia-and-gonorrheascreening (accessed June 2, 2020).
Volk, J. E., J. L. Marcus, T. Phengrasamy, D. Blechinger, D. P. Nguyen, S. Follansbee, and C. B. Hare. 2015. No new HIV infections with increasing use of HIV preexposure prophylaxis in a clinical practice setting. Clinical Infectious Diseases 61(10):1601-1603.
Walker, T. Y., L. D. Elam-Evans, D. Yankey, L. E. Markowitz, C. L. Williams, B. Fredua, J. A. Singleton, and S. Stokley. 2019. National, regional, state, and selected local area vaccination coverage among adolescents aged 13-17 years—United States, 2018. Morbidity and Mortality Weekly Report 68(33):718-723.
Warner, L., J. D. Klausner, C. A. Rietmeijer, C. K. Malotte, L. O’Donnell, A. D. Margolis, G. L. Greenwood, D. Richardson, S. Vrungos, C. R. O’Donnell, C. B. Borkowf, for the Safe in the City Study Group. 2008. Effect of a brief video intervention on incident infection among patients attending sexually transmitted disease clinics. PLoS Medicine 5(6):e135.
Warren, H. P., R. Cramer, S. Kidd, and J. S. Leichliter. 2018. State requirements for prenatal syphilis screening in the United States, 2016. Maternal and Child Health Journal 22(9):1227-1232.
Whitlock, G. G., D. C. Gibbons, N. Longford, M. J. Harvey, A. McOwan, and E. J. Adams. 2018. Rapid testing and treatment for sexually transmitted infections improve patient care and yield public health benefits. International Journal of STD & AIDS 29(5):474-482.
Williams, A. M., T. L. Gift, L. N. O Donnell, C. A. Rietmeijer, C. K. Malotte, A. D. Margolis, and L. Warner. 2020. Assessment of the cost-effectiveness of a brief video intervention for sexually transmitted disease prevention. Sexually Transmitted Diseases 47(2):130-135.
Young, S. D., and J. Schneider. 2020. Clinical care, research, and telehealth services in the era of social distancing to mitigate COVID-19. AIDS and Behavior 24(7):2000-2002.
Zola, M., and J. Frost. 2016. Publicly funded family planning clinics in 2015: Patterns and trends in service delivery practices and protocol. New York: Guttmacher Institute.


















































