5
Breast Cancer
Breast cancer is the leading cause of cancer death among women globally and the second leading cause of cancer death (after lung cancer) among women in the United States (DeSantis et al., 2019; Torre et al., 2017). It is also the second most frequently diagnosed cancer in women after skin cancer (DeSantis et al., 2019). Although the U.S. incidence of breast cancer for women older than 50 years has remained about same over the past 10 years, its incidence has been rising slightly in women under 40 years (Scott et al., 2020; Ward et al., 2019). However, breast cancer treatment has improved outcomes for many women, especially those with more aggressive subtypes and those with metastatic disease (that is, breast cancer that has spread to a distant site in the body). Fortunately, only about 6% of patients have metastatic breast cancer at the time of their initial diagnosis (Howlader et al., 2020). Although breast cancer is also diagnosed in men, it is rare, with an incidence rate of 1.32 per 100,000 people in 2017 (Howlader et al., 2020). As of 2020, it is estimated that there are about 3.5 million adults alive who have or have had breast cancer (SEER, n.d.-a). The epidemiology of breast cancer is discussed in Chapter 3.
Risk factors for breast cancer include family history (with or without an identified genetic predisposition), reproductive factors (early menarche, late menopause, late or no pregnancy), the use of exogenous hormones at menopause, radiation exposure to the chest wall in adolescence or young adult years, and regular use of alcohol. Obesity and lifetime weight gain are primarily risk factors for postmenopausal breast cancer. The identification of women who have a hereditary risk for breast cancer (e.g., BRCA1 and BRCA2 mutation carriers) but who are unaffected (do not have cancer)
has potential to reduce the risk of disease through chemoprevention or prophylactic mastectomy.
In this chapter the committee describes the care trajectory (see Figure 4-1) for breast cancer from initial screening and diagnosis through breast cancer staging (including its major subtypes) to the treatment options for both local and metastatic disease. These treatment options include surgery, radiation, and systemic therapies. The focus of the treatment section is on approaches that are currently standard of care for breast cancer specifically. Finally, the committee considers the long-term and late-onset impairments that may be experienced by women (and men) following the diagnosis and treatment of their breast cancer. To help manage these impairments and functional limitations as well as the adverse effects from both the cancer and its treatment, palliative and supportive care may be recommended for patients at the time of their diagnosis.
As noted in Chapter 3, only about 1% of breast cancer cases are diagnosed in men. Although this chapter focuses on breast cancer in women, men with breast cancer face similar decisions and complexity regarding the diagnosis and treatment of their disease and must take into account many of the same factors when determining appropriate treatments. The vast majority of research on breast cancer is conducted in women and this is reflected in the studies cited in this chapter.
SCREENING
Breast cancer is usually detected in one of two ways: as a result of a screening test (such as mammogram) in an asymptomatic individual or by a clinical evaluation of an individual who presents with symptoms such as a breast mass, bloody nipple discharge, or change in the skin of the breast. The majority of women with breast cancer are currently diagnosed while asymptomatic at the time of screening. However, younger women (i.e., those younger than 40–45 years) are not typically candidates for breast cancer screening and thus are more likely to be diagnosed when symptomatic (usually with a mass).
Several organizations (e.g., the U.S. Preventive Services Task Force and the American Cancer Society) have developed guidelines for breast cancer screening (see Table 4-1). Mammograms are typically used to screen for breast cancer. Although the timing of initiation and the intervals of screening vary among the clinical guidelines, mammographic screening has been shown to reduce mortality from breast cancer compared with no screening, particularly in women aged 50–69 years (Nelson et al., 2009). Studies have demonstrated that screening mammograms lead to an earlier stage of disease at diagnosis and to reduced mortality.
A newer form of mammogram called digital breast tomosynthesis (also called a 3-D mammogram) is becoming more widely used. In addition, magnetic resonance imaging (MRI) is increasingly being used to supplement mammograms as a screening tool, particularly in women with high-risk factors such as a pathogenic variant in a cancer susceptibility gene, such as BRCA1 or BRCA2, or who have a family history of the disease (NCCN, 2020a; Saadatmand et al., 2019).
As described in Chapter 4, decisions about when to start and how often to undergo breast cancer screening should take into account a woman’s risk of breast cancer as well as how she values the specific benefits (e.g., small reductions in the likelihood of dying of breast cancer) and harms (false positive findings, unnecessary biopsies, and the potential for overdiagnosis—detecting a cancer that would not have become clinically evident in a woman’s lifetime without screening—and related overtreatment) (Nelson et al., 2016). Some evidence suggests that screening mammography has only marginally reduced the rate at which women present with advanced cancer (Bleyer and Welch, 2012). Ongoing studies are assessing whether improvements in screening technologies and the ability to identify overdiagnosed cancers that do not require treatment can improve the benefit-to-harm ratio of breast cancer screening (Kanbayashi et al., 2019).
DIAGNOSIS AND STAGING
When a patient presents with changes to his or her breast, no matter whether the changes have been detected with a mammogram or MRI, or by presentation of symptoms such as detection of a lump in the breast tissue, a diagnosis of breast cancer requires a tissue biopsy. Image-guided biopsy—directed by mammogram, ultrasound, or MRI—is usually the first approach and is performed by radiologists or surgeons. In particular, core needle biopsy is the standard way to diagnose breast cancer and does not involve going to the operating room or undergoing general anesthesia, although sometimes a surgical biopsy is necessary. Metastasis to lymph nodes can be diagnosed either by a biopsy, such as a core needle biopsy or a fine-needle aspiration, which uses a smaller needle than a core biopsy, or by surgery.
Once the biopsy tissue has been extracted, it is examined by pathologists who determine whether cancer is present and, if so, the type of breast cancer and the tumor grade and margins. Tumor grade is the description of how abnormal the tumor cells and the tumor tissue appear under a microscope. It is an indicator of how aggressive a tumor is.
Pathologists also test the biopsy tissue for specific biomarkers, particularly hormone receptors (HRs) (i.e., estrogen receptors [ERs] and progesterone receptors [PRs]) and human epidermal growth factor receptor
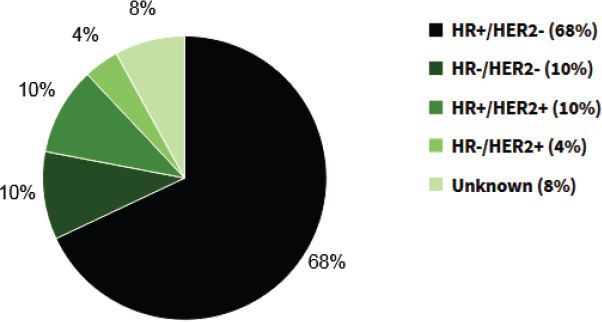
2 (HER2). These biomarkers, along with grading and staging the tumor, help direct the treatment strategy. In data from the Surveillance, Epidemiology, and End Results (SEER) program from 2013–2017 (SEER, n.d.-b), 68% of breast cancers were HR-positive and HER2-negative, 10% were HR-positive and HER2-positive, 10% were triple-negative (ER-, PR-, and HER2-negative), 4% were HR-negative and HER-positive, and the biomarker status was unknown for 8% of breast cancers (see Figure 5-1).
Breast cancer staging describes the size of the tumor in the breast (primary tumor) and whether or how much it has spread in the body. Breast cancer is either invasive or noninvasive. Invasive breast cancers are those that have spread into surrounding breast tissue, the most common of which is invasive ductal carcinoma. In situ breast cancer (ductal carcinoma in situ, discussed below) is a noninvasive cancer that starts in a milk duct and is confined there. A cancer’s stage has implications not only for outcomes, but also for treatment decisions, such as the need for and extent of surgery (e.g., sentinel node dissection versus axillary lymph node dissection) and the use of systemic therapy. An earlier stage at diagnosis is associated with improved outcomes.
As noted in Chapter 4, the most commonly used staging system for breast cancer is that of the American Joint Committee on Cancer (AJCC). The AJCC anatomical staging system incorporates the size of the breast tumor (T), the spread to lymph nodes (N), and metastasis to distant organs (M) for a TNM stage based on information from imaging studies, physical examination, and biopsy results.
The clinical stage describes the initial stage at the time of diagnosis based on imaging studies such as mammogram or ultrasound (e.g.,
SOURCE: SEER, n.d.-b.
cT1N0M0). After the patient has had surgery, the pathologic stage is based on a microscopic analysis of the tumor and lymph nodes that were removed with surgery (e.g., pT2N1). The TNM combinations can be summarized by anatomic stages from stage 0 (noninvasive cancer) to stage IV (metastatic cancer beyond the breast and regional lymph nodes) (see Table 5-1). In addition to TNM factors and the expression of the ER, PR, and HER2 biomarkers in breast cancer, genomic tests such as OncotypeDX® and Mammaprint® are also incorporated into staging since they are known to be associated with a patient’s prognosis and responsiveness to therapy. In 2017 this change was incorporated into the AJCC staging manual eighth edition (Amin et al., 2017). The inclusion of ER, PR, HER2 biomarkers increases the complexity of the AJCC staging system and while it cannot be fully illustrated in this report, the details can be found in AJCC staging manual, which is updated periodically.
TABLE 5-1 Anatomic Staging of Invasive, Nonmetastatic Breast Cancer
| Stage | Detailed Stage | TMN Stage |
|---|---|---|
| I | Stage IA |
|
| Stage IB |
|
|
| II | Stage IIA |
|
| Stage IIB |
|
|
| III | Stage IIIA |
|
| Stage IIIB |
|
|
| Stage IIIC |
|
|
NOTE: M = presence or absence of distant metastasis; N = extent of regional lymph node spread; T = size or extent of the primary tumor (see Chapter 4 for more information on cancer staging).
SOURCES: Used with the permission of the American College of Surgeons, Chicago, Illinois. The original source for this information is the AJCC Cancer Staging Manual, Eighth Edition (2017) published by Springer International Publishing: the American College of Surgeons. Amin, M.B., Edge, S.B., Greene, F.L., et al. (Eds.). AJCC Cancer Staging Manual, 8th Ed. Springer New York, 2017. See https://cancerstaging.org/references-tools/deskreferences/Documents/AJCC%208th%20Edition%20Breast%20Cancer%20Staging%20System.pdf (accessed January 15, 2021) for the AJCC cancer staging manual 8th edition breast cancer chapter.
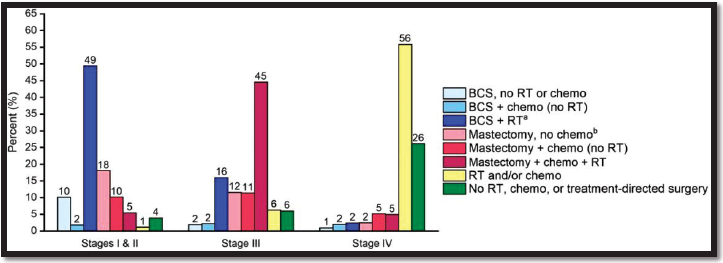
NOTES: BCS = breast‐conserving surgery; chemo = chemotherapy, including targeted therapy and immunotherapy; RT = radiation therapy.
a A small number of these patients received chemotherapy.
b A small number of these patients received RT.
SOURCE: Reprinted from DeSantis, C.E., J. Ma, M.M. Gaudet, L.A. Newman, K.D. Miller, A. Goding Sauer, A. Jemal, and R.L. Siegel. 2019. Breast cancer statistics, 2019. CA: A Cancer Journal for Clinicians 69(6):438–451; used with permission from John Wiley and Sons.
The staging and the treatment of breast cancer have both evolved rapidly over the past decade. Consequently, determining the most appropriate treatment for an individual patient can be complicated and may include multiple options. In general, earlier-stage breast cancers tend to be treated with breast-conserving surgery (BCS) that is less extensive and with less chemotherapy than more advanced cancers. However, as seen in Figure 5-2, the breast cancer stage does not correlate directly with the type of treatment, which can be heterogeneous across stages. For example, some stage I and stage II patients may be treated with more extensive surgery (total mastectomy) and chemotherapy, whereas some stage III patients may be treated with BCS without chemotherapy based on tumor biological factors, surgical margins, and response to therapy. Treatment decisions are ideally made in a multidisciplinary, patient-centered manner that involves the surgeon, medical oncologist, radiation oncologist, and other clinicians (such as the plastic reconstructive surgeon) as well as the patient herself (or himself).
TREATMENT OF NONINVASIVE BREAST CANCER
Ductal carcinoma in situ (DCIS) is the earliest form of breast cancer and refers to cancer cells that arise in the breast milk ducts but have not spread outside the duct. DCIS is considered to be noninvasive. It is also
called intraductal carcinoma, pre-invasive cancer, or stage 0 breast cancer. Between 2000 and 2017 the incidence rate of the disease ranged from about 12 to 14.5 cases per 100,00 for women younger than 50 years and from about 81.5 to 96.5 cases per 100,000 for women older than 50 years (Howlader et al., 2020).
DCIS is usually detected with screening mammography. DCIS cells are not capable of metastasizing or spreading outside the breast to lymph nodes or other distant organs, so lymph node dissection is generally not needed. There is a low risk of DCIS becoming invasive, and therefore the current standard of care is to treat DCIS with BCS with or without radiation therapy or tamoxifen or else to treat it with a mastectomy in a manner similar to what is typical for invasive breast cancer. With complete resection and radiation treatment as indicated, the prognosis is excellent for patients with DCIS, with a 20-year mortality rate of 3.3% (Narod et al., 2015; Subhedar et al., 2015). The 20-year risk of recurrence of invasive disease in the same breast (ipsilateral) that had the initial DCIS diagnosis is 5.9% and 6.2% in the contralateral breast (Narod et al., 2015). Endocrine therapy with tamoxifen or an aromatase inhibitor is offered to patients with ER-positive, stage 0 breast cancer because although there is no survival benefit, endocrine therapy may reduce the risk of ipsilateral and contralateral recurrence (Staley et al., 2014).
TREATMENT OF INVASIVE, LOCALIZED BREAST CANCER
Invasive breast cancer can be either localized, that is, it has not spread outside the breast and regional lymph nodes, or metastatic, that is, it has spread to other, often distant, parts of the body. More than 90% of breast cancer patients present with localized breast cancer. The treatment for localized breast cancer, while complex, is intended to cure and aims to minimize recurrences both locally (in the breast, chest wall, and lymph nodes) and systemically (e.g., liver, lungs, bone, and brain). The particular treatment given to an individual patient depends on a number of factors, particularly the cancer stage (including the tumor size and presence of involved lymph nodes), tumor grade, and HR and HER2 receptor status. Other factors that are considered when determining the treatment plan include the patient’s comorbidities and menopausal status, the response of the tumor to prior treatments, and the patient’s personal preferences.
Treatment for invasive, localized breast cancer may be local (i.e., surgery with or without radiation), systemic (e.g., chemotherapy, endocrine therapy), or, more frequently, a combination of both (see Figure 5-3). This section begins with a discussion of the types and extent of surgery that may be used to remove the tumor and, when necessary, the affected lymph nodes. The two general surgical approaches are breast conservation with
resection and mastectomy. Following this discussion, the several types of radiation therapy that may be used to treat localized, invasive breast cancer are considered. Next is a synopsis of the various kinds of systemic therapies that are currently used based on the hormone and biomarker status of the
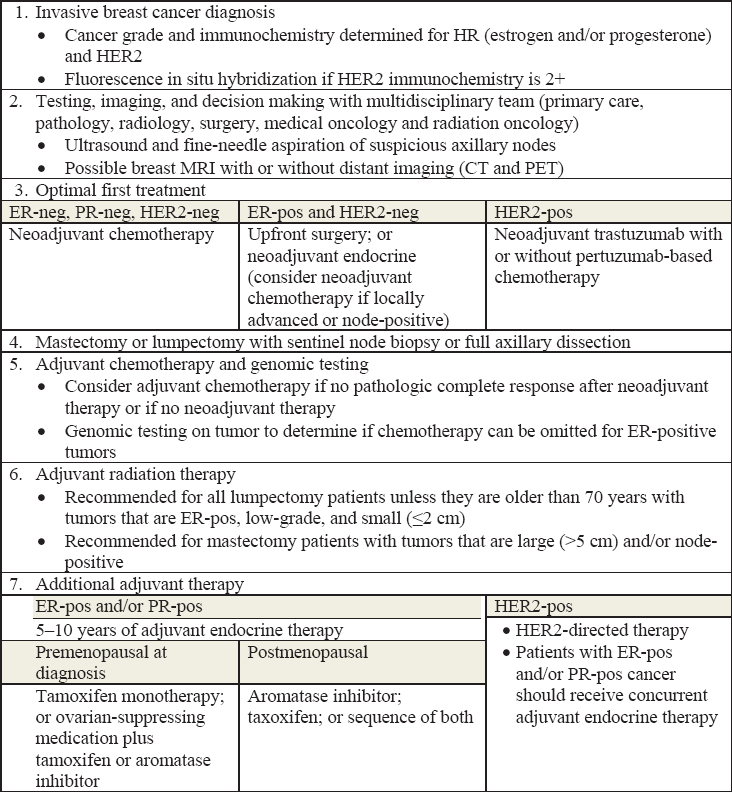
NOTE: CT = computed tomography; ER = estrogen receptor; HER2 = human epidermal growth factor receptor 2; HR = hormone receptor; MRI = magnetic resonance imaging; PET = positron emission tomography; PR = progesterone receptor.
SOURCE: Ruddy and Ganz, 2019, reproduced with permission from JAMA; copyright © (2021), American Medical Association, all rights reserved.
cancer. Systemic and radiation therapies may be used before (neoadjuvant) or after (adjuvant) surgery. Some forms of systemic therapy, such as endocrine therapy, may be used for HR-positive breast cancer for many years after surgery and radiation are completed. Other systemic therapies include chemotherapy and therapies that target the HER2 protein. The treatment of metastatic breast cancer, discussed after the treatment of localized breast cancer, focuses primarily on systemic therapy, although surgery and radiation are used in certain situations.
Surgical Treatments
Decisions about the surgical management of the primary breast tumor and the lymph nodes are made almost separately, depending on anatomic stage, biomarkers, and patient preferences. These decisions are often influenced by other treatment recommendations from the medical oncologist, radiation oncologist, breast surgeon, and plastic surgeon as well as by the patient’s preferences, reflecting the truly multidisciplinary nature of breast cancer treatment. This section discusses BSC, mastectomy, and the surgical management of axillary nodes in the breast area. The committee then considers breast reconstruction during or after surgery for cancer treatment.
Breast-Conserving Surgery
The options for resecting a breast tumor are (1) BCS (partial mastectomy, segmental mastectomy, or lumpectomy), which removes only the tumor and leaves the remaining breast tissue; or (2) total mastectomy, which removes the tumor along with the entire breast, possibly including the skin and nipple-areolar complex. Breast-conserving therapy often consists of BCS plus radiation therapy. Multiple randomized trials have shown that breast-conserving therapy and mastectomy are equally effective, with no difference in disease-free survival and overall survival (Fisher et al., 2002; Litiere et al., 2012; Veronesi et al., 2002).
There are several relative contraindications to breast-conserving therapy, including a tumor that is large relative to breast size, multicentric disease (i.e., multiple tumors in different sections of the breast), and contraindications to radiation therapy (e.g., prior chest wall irradiation). For invasive breast cancers, BCS requires a complete resection of the tumor such that the excised tissue has no cancer at the margins (i.e., negative surgical margins) (Moran et al., 2014). For in situ breast cancer, which tends toward more diffuse distribution compared with invasive breast cancer, the consensus guidelines recommend a 2 mm margin for optimal local control for BCS with whole-breast irradiation (Morrow et al., 2016). The effective use
of BCS depends on the relative size of the tumor compared with the native breast size. Even for fairly “small” tumors, the location of the tumor may be unfavorable (e.g., upper inner chest quadrant or superior quadrant) and can be associated with a sub-optimal cosmetic outcome after lumpectomy and radiation therapy. Finally, for multiple tumors in one breast, it may not be feasible to get an acceptable cosmetic result with BCS unless the patient has a large breast or is receiving partial-breast reconstruction.
Patient preference is also a factor when deciding whether to perform BCS (combined with radiation) or mastectomy. Despite equivalent survival rates for the two procedures, BSC-eligible patients are increasingly choosing to have mastectomies for several reasons, including a reluctance to undergo radiation therapy, fear of recurrence, and a desire for symmetry (DeSantis et al., 2019). In a review of 1.2 million U.S. breast cancer patients, 35.5% of patients with early-stage disease (i.e., those who are most likely to be eligible for BCS) underwent mastectomy. The rate of mastectomy increased from 2003 to 2011, when the study ended (Kummerow et al., 2015; Lee et al., 2009).
Mastectomy
Mastectomy is performed for patients who are not eligible for BCS or for those who choose it. A modified radical mastectomy refers to a mastectomy with axillary lymph node dissection. A mastectomy can be performed with preservation of the skin or nipple or both if some form of immediate breast reconstruction is planned. A contralateral prophylactic mastectomy refers to a mastectomy performed on a breast without a diagnosis of cancer and is often offered to women with a genetic mutation that confers an increased risk of breast cancer (e.g., BRCA1, BRCA2, TP53, PTEN) in order to reduce that risk. Women without a known deleterious mutation or other risk factors have also elected to undergo contralateral prophylactic mastectomy at increasing rates (Chagpar, 2014), although there is no evidence of improved survival. The decision to undergo this surgery has been shown to be based primarily on a fear of breast cancer as well as cosmetic reasons, such as a desire for symmetry (Ager et al., 2016; Sando et al., 2018). Low physician knowledge about contralateral breast cancer and the risk of local recurrence also was found to be associated with favorable recommendations about contralateral prophylactic mastectomy for patients at average risk (Kantor et al., 2019).
Surgical Management of Axillary Nodes
A metastasis of breast cancer to the axillary lymph nodes is determined by physical exam and imaging, commonly by ultrasound. Lymph nodes
that appear suspicious in ultrasound images can be biopsied to confirm metastasis (clinically node-positive disease). Less commonly, breast cancer can metastasize to other nodal basins such as the internal mammary, infraclavicular, or supraclavicular lymph nodes (see Figure 5-4).
For patients with breast cancer, lymph nodes may be removed for the purpose of staging and prognosis to help guide treatment recommendations as well as for potentially reducing regional recurrence for patients with locally advanced disease. In order to minimize the adverse effects from lymph node removal such as lymphedema, neuropathy, and seromas, surgeons may perform a sentinel lymph node dissection (SLND, or sentinel lymph node biopsy) to remove only the first few axillary lymph nodes draining a breast tumor; this is performed for early-stage clinically node-negative breast cancer. Sentinel lymph nodes are the first lymph nodes to which cancer cells are most likely to spread from a primary tumor. A positive sentinel lymph node biopsy indicates that the cancer has spread to the lymph nodes, in which case an axillary lymph node dissection (ALND) may be performed. ALND entails a more complete removal of axillary lymph nodes (levels I/II, and occasionally level III) and results in higher rates of adverse effects. ALND is typically performed for patients with clinically node-positive disease.
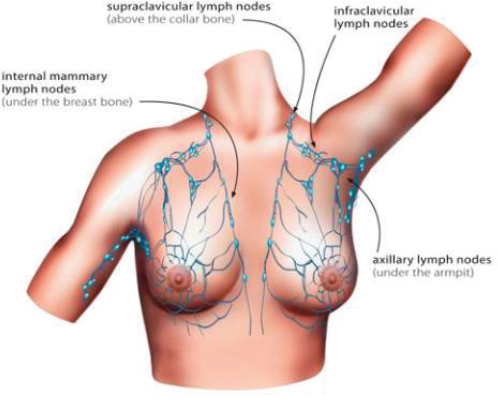
SOURCE: Illustration reproduced with permission from Cancer Australia, 2021.
Breast Reconstruction
Patients who undergo total mastectomy may elect to have breast reconstruction to improve self-esteem and psychosocial well-being, including their emotional health, sexuality, and body image (Cordova et al., 2019). There are many options for reconstruction, including the use of a breast implant or a patient’s own tissue (autologous flap), and reconstruction may be performed at the same time as the mastectomy or delayed. Immediate breast reconstruction is often appropriate for patients who do not have significant medical comorbidities, have healthy mastectomy skin flaps, and are not expected to require radiation after mastectomy. Although the rates of breast reconstruction after mastectomy have been increasing, only about 24% of U.S. patients with invasive breast cancer undergo reconstruction at the same time as their mastectomy (Kamali et al., 2019), suggesting that breast reconstruction may be underutilized. Delayed reconstruction may be recommended if patients have risk factors for delayed wound healing (e.g., poorly controlled diabetes, smoking, or obesity) or if they are likely to be treated with postmastectomy radiation. A third approach is staged reconstruction, also referred to as “delayed-immediate” reconstruction, for patients who may need postmastectomy radiation (Kronowitz, 2010). With this approach, a temporary tissue expander is placed under the skin of the breast at the time of the mastectomy to preserve the three-dimensional shape of the breast skin envelope, and after the completion of radiation, definitive reconstruction surgery is performed. The potential benefits of the staged approach for selected patients include avoiding the complications associated with radiation to the breast implant or autologous flap and an improved cosmetic result (Kronowitz, 2010). In the first 2 years after breast reconstruction with either implant or autologous flap, the most common adverse effects are wound complications (4.4–9.5%), infection (20.5–20.7%), fat necrosis (15.7%), and implant removal (24.7%). The addition of radiation increases the risks of infection and implant removal (Jagsi et al., 2016; Olsen et al., 2017).
In addition to reconstruction following mastectomy, an increasing number of breast cancer patients are undergoing reconstructive procedures with BCS (oncoplastic reconstruction). These patients may have tumors that are large relative to their native breast size or multiple breast lesions (multicentric), or the tumor is located in an unfavorable quadrant in the breast. These patients are not good candidates for BCS alone because a poor cosmetic outcome is anticipated following BCS and radiation (Clough et al., 2015). Oncoplastic reconstruction techniques involve rearranging the patient’s remaining native breast tissue and filling the defect resulting from the lumpectomy, a breast reduction and lift (mastopexy), or using adjacent local breast tissue to restore breast volume. This surgery is usually done on
an outpatient basis, and it has a significantly lower rate of postoperative complications than mastectomy with reconstruction (Carter et al., 2016) and rates of locoregional recurrence and survival that are similar to those following BCS without oncoplastic reconstruction or total mastectomy (Carter et al., 2016; Down et al., 2013).
Adverse Effects of Breast Surgery
Both BCS and mastectomy are relatively low-risk surgeries that by themselves do not commonly result in long-term adverse effects after healing is complete. However, particularly when combined with radiation, surgery for the breast and axillary lymph nodes can result in adverse sequelae, such as recurrent seromas (fluid collections) that may require repeated drainage; wound complications such as infection, necrosis, or delayed healing; breast and nipple deformities; pain or neuropathy (e.g., numbness and burning sensations); and weakness and limited movement in the affected shoulder and arm. Furthermore, lymph node resection has various potential adverse effects, including lymphedema, neuropathy, and seromas that can impair quality of life. Several of these potential long-term adverse effects are discussed later in this chapter and in Chapter 9. To reduce the risk of these adverse effects, a number of innovative approaches to axillary node surgery have been developed; these are discussed in Chapter 8.
Radiation Therapy
Radiation therapy is an integral component of optimal local-regional therapy for the management of stage I–III breast cancer. Decisions regarding whether a patient is likely to benefit from adjuvant radiation therapy take into account a number of factors, including the surgical approach (lumpectomy or mastectomy), tumor biology based on receptor status, lymph node status, and patient age. In this section the indications for radiation therapy and fractionation schedules for patients treated with lumpectomy or mastectomy are presented. A general overview of radiation therapy for many cancers is given in Chapter 4.
Radiation Therapy After Lumpectomy
The main goals of radiation therapy after lumpectomy for women with invasive breast cancer are to decrease recurrences, decrease the risk of death from breast cancer, conserve the breast with an acceptable cosmetic outcome, and avoid mastectomy in women who desire to preserve the breast. On the basis of numerous randomized clinical trials and meta-analyses, lumpectomy followed by radiation therapy to the whole breast
is considered to be equivalent to mastectomy in terms of cancer control and survival rates (Fisher et al., 2002; Poggi et al., 2003; van Dongen et al., 2000; Veronesi et al., 2002). While whole-breast irradiation has been the standard of care, other approaches include partial-breast irradiation, regional nodal irradiation, and a determination to omit radiation treatment (see Table 5-2).
TABLE 5-2 Radiation Approaches for Breast Cancer
| Radiation Approach | Indications | Comments |
|---|---|---|
| Whole breast |
|
|
| Partial breast |
|
|
| Regional nodal |
|
|
| Radiation omission |
|
|
NOTE: EBRT = external beam radiation therapy; ER = estrogen receptor; Gy = gray, a unit denoting the joules of radiative energy deposited per kilogram of body mass; HER2 = human epidermal growth factor receptor 2; IORT = intraoperative radiation therapy; PR = progesterone receptor.
Whole-Breast Irradiation
For patients who have undergone a lumpectomy, the standard of care typically involves conventionally fractionated radiation therapy (45–50.4 Gy in 25–28 fractions) over 5–5.5 weeks followed by an additional booster dose to the lumpectomy cavity with an additional 4–8 treatments (NCCN, 2020b). However, hypofractionated whole-breast radiation (see Table 5-2), has the same tumor control rate as longer courses of radiation with fewer side effects (Haviland et al., 2013; Whelan et al., 2010). Some women will receive a boost dose to the lumpectomy cavity, which can add an additional week to the treatment duration. There was some initial concern that certain types of breast cancer (triple-negative breast cancer, grade 3 tumors) were not well suited for hypofractionated whole-breast radiation therapy, but consensus guidelines now recommend it for nearly all patients receiving whole-breast radiation (ASTRO, 2018; Smith et al., 2018). Most recently, a hypofractionated schedule of 26 Gy delivered in 5 treatments given on consecutive days was shown to produce results similar to 40 Gy delivered in 15 treatments (Murray Brunt et al., 2020). As a result, this 1-week schedule of whole-breast irradiation may become more common in the near future.
Partial-Breast Irradiation
When patients who do not receive radiation therapy after a lumpectomy develop an in-breast tumor recurrence, the recurrence tends to occur near the site of the original tumor. This led to randomized clinical trials comparing whole-breast irradiation with partial-breast irradiation, which is defined as radiation therapy targeting the tumor bed (lumpectomy cavity) and approximately 2 cm of tissue surrounding the tumor bed. On the basis of these large, randomized trials comparing partial-breast irradiation to whole-breast irradiation, partial-breast irradiation results in equivalent cancer control rates to whole-breast irradiation in appropriately selected patients (Hepel and Wazer, 2020). Tumor biology and patient factors (age or a familial history) are important selection factors for partial-breast irradiation; specifically, the factors indicating partial-breast irradiation use include an age of greater than 50 years and lymph-node-negative and ER-positive tumors that are smaller than 2 cm. For patients who do not meet these criteria, the recurrence rates are slightly but significantly higher with partial-breast irradiation than with whole-breast irradiation.
There are various partial-breast irradiation delivery methods, including external beam, brachytherapy, and intraoperative radiation therapy (see Chapter 4 for more details). The most common approach in North America is external beam radiation therapy, which is typically delivered two times per day (3.85 Gy per fraction), with each treatment separated by at least 6
hours, for 5 consecutive days (total dose, 38.5 Gy). Alternatively, a once-per-day (6 Gy per fraction) partial-breast irradiation regimen given on nonconsecutive days for a total of 5 days (30 Gy) is a good option for patients who are inconvenienced by the twice-per-day schedule (Livi et al., 2015).
Regional Lymph Node Irradiation
Although most patients who undergo lumpectomy will receive whole-breast irradiation or partial-breast irradiation, there are a growing number of patients who may benefit from radiation therapy that treats at-risk lymph node regions, including the axillary, supraclavicular, infraclavicular, and internal mammary lymph nodes. The classic indication for radiation to these regions was for a woman to have four or more involved lymph nodes, large tumors (>5 cm), and at least one involved lymph node (i.e., a woman with anatomic stage III breast cancer). However, recent studies have demonstrated that women with one to three involved lymph nodes or those with tumors that are in the inner portion of the breast (near the breast bone) may also benefit from radiation therapy that includes the lymph node regions and they may have a significantly lower risk of incurable distant metastases (Poortmans et al., 2015; Whelan et al., 2015). In women who receive neoadjuvant chemotherapy, regional lymph node irradiation is indicated if there is lymph node involvement prior to the start of chemotherapy or if there are any pathologically involved axillary lymph nodes at the time of surgery.
Omission of Radiation Therapy
Omission of radiation therapy is considered mainly for women aged 70 years or older. For these women who have tumors that are ER-positive, <2 cm in size, and lymph-node-negative, data suggest that radiation provides a modest decrease in local recurrences in the breast but has no benefit in reducing mortality, provided that these women take endocrine therapy (Hughes et al., 2013). Similar results have been demonstrated in women 65 or older (with ER-positive, pathologically lymph-node-negative tumors up to 3 cm in size) in a separate randomized study, though mature results have not yet been reported (Kunkler et al., 2015). Therefore, provided that a patient accepts a slightly higher risk of an in-breast tumor recurrence in her lifetime with the understanding that it will not affect how long she may live, not having radiation therapy may be appropriate for these low-risk women.
Radiation Therapy After Mastectomy
Postmastectomy radiation is generally used for patients with stage III breast cancer (i.e., 4–9 involved lymph nodes; tumors >5 cm with at
least one involved lymph node; 10 or more nodes with any tumor size; inflammatory breast cancer). However, new studies have demonstrated that patients with one to three involved nodes may also receive a significant benefit from radiation therapy in terms of reduced recurrences and mortality. Furthermore, these studies have also shown that women who have no involved lymph nodes received no benefit from radiation therapy in terms of reduced recurrences or mortality. In women who receive neoadjuvant chemotherapy, data suggest that it is safe to omit radiation therapy only for those women who present with node-negative disease; it remains unknown whether women who receive neoadjuvant chemotherapy that renders negative nodes that were initially positive may omit postmastectomy radiation. Otherwise, postmastectomy radiation therapy is indicated in women with persistent nodal disease after neoadjuvant chemotherapy.
Radiation therapy after mastectomy is generally delivered over 25 treatments (5 weeks) (NCCN, 2020b). A booster dose of radiation (additional 5 treatments) is sometimes given to the mastectomy scar, although this has not been studied well in clinical trials. Hypofractionated radiation courses (15–16 treatments) are sometimes used for postmastectomy radiation. This approach is being investigated in a large clinical trial specifically of patients who have undergone or are planning to undergo breast reconstructive surgery.
Adverse Effects of Radiation Therapy
As discussed in Chapter 4, skin irritation and fatigue are among the most common acute adverse effects of radiation therapy to any part of the body. The acute and long-term adverse effects of radiation for breast cancer depend on the extent of treatment (e.g., whole-breast versus regional nodal irradiation) because the larger the volume that is irradiated, the greater the risk of adverse effects. The acute and long-term effects for radiation therapy for breast cancer are summarized in Table 5-3.
Systemic Treatments
Systemic treatment refers to medications given to prevent the development of recurrent breast cancer whether locally (in the same breast, opposite breast, or lymph nodes) or distantly (e.g., in the bones, liver, lungs, or brain). The decision whether to use adjuvant or neoadjuvant systemic therapy for localized breast cancer depends primarily on the tumor stage, HR status, and HER2 status, along with other factors.
Each of the major types of systemic therapies—chemotherapy, endocrine therapy, and targeted therapies—may be used for localized breast cancer. In the section below the committee considers the use of these therapies
TABLE 5-3 Acute and Long-Term Effects Following Radiation Treatment for Breast Cancer
| Radiation Treatment | Acute Effects | Long-Term Effects |
|---|---|---|
| Whole-breast irradiation |
|
|
| Partial-breast irradiation |
|
|
| Regional lymph node irradiation or postmastectomy radiation therapy |
|
|
b Cheng et al., 2017; Wennstig et al., 2020.
for HR-positive and HR-negative and HER2-positive and HER2-negative breast cancers. Table 5-4 presents common scenarios for the use of these therapies, but whether a particular patient will receive any or all of them depends on his or her specific cancer staging, grade, biomarker status, comorbidities, and personal preferences. In addition, the committee considers the use of systemic therapy for inflammatory breast cancer, a rare and aggressive invasive cancer.
TABLE 5-4 Systemic Therapies Used for Stage I–III (Localized) Breast Cancer
| Breast Cancer Subtype | Endocrine Therapy | Chemotherapy | HER2-Directed Therapy | Use of Neoadjuvant Therapy |
|---|---|---|---|---|
| HR+, HER2– | Yes | If node-negative, use OncotypeDx® or Mammaprint® assay to determine chemotherapy use; if node-positive, often use chemotherapy | No | Sometimes |
| HR–, HER2– | No | Yes | No | Often |
| HR+, HER2+ | Yes | Yes | Yes | Often |
| HR–, HER2+ | No | Yes | Yes | Often |
Endocrine Therapy
Endocrine therapy is used to treat HR-positive cancers (see Table 5-4) by blocking the production of hormones or preventing them from working. The selective estrogen receptor modulator (SERM) tamoxifen acts to block the effects of estrogen in breast tissue. Luteinizing hormone-releasing hormone (LHRH) agonists, such as goserelin and leuprolide, decrease the body’s estrogen and progesterone by suppressing the production of these hormones in the ovaries. Aromatase inhibitors, such as anastrozole, letrozole, and exemestane, decrease the body’s estrogen by blocking an enzyme called aromatase from turning androgen into estrogen.
Chemotherapy
Chemotherapy regimens for breast cancer are numerous and complex (see Table 5-5) and usually involve a mixture of agents. The choice of a chemotherapy regimen for breast cancer depends on a number of factors, including the clinical status of the patient and patient preference. Unlike endocrine therapy, which typically is used after local therapies (i.e., surgery and radiation) are complete, chemotherapy may be used before or after local therapies. Chemotherapy agents used in the adjuvant setting are intravenous and often given as combinations; for example, doxorubicin (adriamycin) is used with cyclophosphamide.
HER2-Directed Therapy
There are several types of HER2-directed therapies for localized breast cancer, such as the monoclonal antibodies trastuzumab and pertuzumab,
TABLE 5-5 Examples of Systemic Therapies for Localized or Metastatic Breast Cancer
| Drug Class | Examples |
|---|---|
| Endocrine Therapy | SERM: tamoxifen Aromatase inhibitors: anastrazole, letrozole, exemestane SERD: fulvestrant LHRH: goserelin, leuprolide, triptorelin |
| Chemotherapy | doxorubicin, cyclophosphamide, paclitaxel, docetaxel, capecitabine, eribulin, vinorelbine, gemcitabine, and others |
| Targeted Therapies | |
| HER2-targeted | trastuzumab, pertuzumab, ado-trastuzumab-emtansine, trastuzumab deruxtecan, lapatinib, neratinib, tucatinib |
| CDK4/6 inhibitors | palbociclib, ribociclib, abemaciclib |
| mTOR inhibitor | everolimus |
| PARP inhibitors | olaparib, talazoparib |
| PI3 kinase inhibitor | alpelisib |
| Immunotherapy (immune checkpoint inhibitors) | atezolizumab |
NOTE: CDK = cyclin-dependent kinase; HER2 = human epidermal growth factor receptor 2; LHRH = luteinizing hormone-releasing hormone; mTOR = mammalian target of rapamycin; PARP = poly (ADP-ribose) polymerase; PI3 = phosphoinositide 3-kinases; SERD = selective estrogen receptor degrader; SERM = selective estrogen receptor modulator.
the antibody conjugate ado-trastuzumab-emtansine, and the small molecule neratinib (see Table 5-5). Adjuvant treatment of HER2-positive breast cancer includes chemotherapy in addition to HER2-directed therapies.
Hormone-Receptor-Positive, HER2-Negative Breast Cancer
Most breast cancer, in both women and men, is estrogen- or progesterone-positive or both (ER+/PR+) as well as HER2-negative. Endocrine therapy is the key component of systemic treatment. Chemotherapy is considered in certain situations (discussed below) but always in the context of what additional benefit it may add to endocrine therapy.
In invasive breast cancer, adjuvant endocrine therapy is recommended after surgery and radiation (if used) to reduce the risk of local and systemic recurrence. The type of endocrine therapy used depends on the patient’s menopausal status (see Figure 5-3). Women who are premenopausal are treated with tamoxifen or with a combination of ovarian suppression (usually with an injection of an LHRH agonist) and tamoxifen or with an aromatase inhibitor, depending on clinical factors and patient preference.
Postmenopausal women usually receive an aromatase inhibitor, although tamoxifen is also an option for those women who are unable to tolerate the adverse effect of aromatase inhibitors. Men with breast cancer are generally treated with adjuvant tamoxifen. Previously the optimal length of endocrine therapy was considered to be 5 years; however, several studies have demonstrated some benefit from a longer duration of use. Decisions about the length of endocrine therapy (5 versus 10 years) consider the risk of recurrence as well as the patient’s tolerance of and preference for these endocrine therapies.
When considering the additional benefit that using chemotherapy with endocrine therapy is likely to confer, clinicians take into account tumor stage, tumor assays, and patient age. For many years individuals with a node-negative, ER-positive tumor larger than 1 cm in size were offered adjuvant chemotherapy to prevent distant recurrence. However, molecular assays performed on primary breast tumors, such as OncotypeDx® and Mammaprint®, can identify patients who will not benefit from chemotherapy and therefore should not receive it (Beumer et al., 2016; Sparano et al., 2018, 2019). Such assays are both prognostic (tumors with lower scores for recurrence have a better prognosis at baseline) and predictive (they predict the benefit of chemotherapy) and are critical components of treatment planning for node-negative, ER-positive breast cancer. At present, most women with node-negative, ER-positive breast cancer do not receive chemotherapy, which is a significant change from 10 years ago. This change reflects a growing understanding of tumor biology, which in turn drives decisions on optimal treatments.
In patients with node-positive, ER-positive breast cancer, the decision whether to add chemotherapy to endocrine therapy is based on the number of involved nodes and the patient’s menopausal status. In certain situations, particularly for postmenopausal women with limited nodal involvement, there appears to be little benefit from adding chemotherapy to endocrine therapy. Studies are investigating the use of the molecular assays, such as OncotypeDx®, in patients with limited lymph node involvement. Early data in postmenopausal women suggest that these assays may be helpful in determining which women may safely avoid chemotherapy, and studies are under way to try to answer this important question.
The most common chemotherapy regimens used in ER-positive/HER2negative breast cancer include “TC” (taxotere [docetaxel] and cytoxan [cyclophosphamide]) chemotherapy given every 3 weeks for four cycles and “dd (dose-dense) AC-T” (adriamycin and cyclophosphamide given every 2 weeks for four cycles followed by taxol [paclitaxel] every 2 weeks for four cycles). Chemotherapy is often administered with white blood cell growth factors (e.g., pegfilgrastim or filgrastim) to prevent the low white blood cell counts and infection that may result from the chemotherapy alone.
Hormone-Receptor-Negative, HER2-Negative Breast Cancer (Triple-Negative Breast Cancer)
Triple-negative breast cancer (TNBC) is more common in African-American women and premenopausal women. TNBC has the worst prognosis of the three main subtypes of breast cancer (HR-positive/HER2-negative, HR-negative/HER2-negative, or HER2-positive), although, women with stage 1 (localized) TNBC still have a good prognosis, with a 5-year survival of 91%, and those with regional TNBC have a 5-year survival rate of 65% (SEER, n.d.-b).
At the present time, chemotherapy is the sole systemic treatment used for early-stage disease, although clinical trials are testing other agents. Increasingly, neoadjuvant chemotherapy is being used for TNBC, as this approach allows for (1) clinical trials investigating new therapies in addition to standard therapies to improve the response rate and (2) risk-adapted treatment decisions that determine the need for additional therapy based on the response of the tumor. When chemotherapy is given prior to surgery, the pathological complete response (pCR, that is, no tumor detected after the chemotherapy) can be determined when the breast tissue is examined during the postsurgical pathology assessment. A pCR is associated with improved disease-free survival in TNBC. Studies indicate that adding carboplatin to taxol in the dd AC-T chemotherapy regimen described above for HR-positive/HER2-negative breast cancer can improve pCR for TNBC (Loibl et al., 2018). Clinical trials have suggested that neoadjuvant immune therapy (including pembrolizumab) may also improve pCR, but these drugs are not approved by the U.S. Food and Drug Administration (FDA) for this cancer at this time.
If there is residual tumor following chemotherapy, administration of capecitabine for 6 months can reduce the risk of recurrent disease. The combination of chemotherapy, surgery, possible radiation, and then 6 months of capecitabine means that TNBC treatment may extend for more than 1 year. Numerous clinical trials are under way to assess whether there are benefits of treating women with residual TNBC, including, for example, studies of women who have received capecitabine (or chemotherapy) therapy with either pembrolizumab (Lewis, 2019) or with atezolizumab (Keenan and Tolaney, 2020).
HER2-Positive Breast Cancer
The central component of HER2-positive breast cancer treatment, whether it is HR-positive or HR-negative, is HER2-directed therapy, specifically trastuzumab in conjunction with chemotherapy. Depending on the underlying biological risk of recurrence of the tumor, there are various chemotherapy regimens can be used with trastuzumab, including paclitaxel,
TC (taxotere [docetaxel] and carboplatin), and dd AC-T (see above). An additional HER2-targeted therapy, pertuzumab, is added in cases in which tumors are at higher risk of recurrence.
Neoadjuvant chemotherapy is increasingly being used in HER2-positive cancers. The antibody drug conjugate trastuzumab–emtansine significantly reduces the risk of distant disease in patients for whom neoadjuvant chemotherapy did not result in a pCR of the tumor. If HER2-positive tumors are also ER-positive, then the hormone targeted therapies described earlier are also used.
Bisphosphonates and Denosumab
Bone-acting agents such as bisphosphonates and denosumab have been studied in women with breast cancer to determine their effect on cancer recurrence. Adjuvant bisphosphonates have been recommended for postmenopausal women who have had breast cancer to reduce the risk of recurrence (Curigliano et al., 2017). Postmenopausal women with early-stage invasive breast cancer treated with bisphosphonates for 2 to 5 years had highly significant reductions in recurrence, distant recurrence, bone recurrence, and breast cancer mortality; however, in premenopausal women, bisphosphonates had no effect on any of these outcomes (EBCTCG, 2015). Another study of women with early-stage breast cancer showed that bisphosphonates not only significantly decreased the risks of locoregional or distant recurrence, new primary breast cancers, and breast cancer mortality, but that these risk reductions were seen in both pre/peri and postmenopausal women, in both ER-negative and ER-positive breast cancers, and for both earlier and later recurrences (Korde et al., 2018).
Inflammatory Breast Cancer
Inflammatory breast cancer is a rare (1–5% of all breast cancers) and very aggressive disease, which is characterized by the skin of the breast having a discolored and thickened appearance (ACS, 2019). It is diagnosed as a locally advanced disease (stage III). It is more common in African-American women, younger women, and women who are overweight or obese.
The treatment of inflammatory breast cancer includes chemotherapy (with the exact chemotherapy dependent on HER2 status), mastectomy, and postmastectomy chest wall radiation. It may also include endocrine therapy if the cancer is HR-positive. Despite aggressive multimodality treatment, inflammatory breast cancer has a poor prognosis, with a 5-year survival rate of 52% for women with regional disease and 18% for those with distant disease (ACS, 2019).
Adverse Effects of Systemic Treatments
Although some adverse effects may be more common with specific systemic treatments, in general the following effects are seen with adjuvant chemotherapy: hair loss, mouth sores, diarrhea, constipation, fatigue, difficulty concentrating, induction of menopause, infertility, low blood counts, fever, infection, bone pain, and neuropathy, along with others. Long-term adverse effects, described in the section on Life After Breast Cancer Diagnosis and in Chapter 9, include cardiotoxicity, chronic fatigue, and chemotherapy-induced peripheral neuropathy. The toxicity of HER2-directed therapy includes infusion reactions, cardiotoxicity, pneumonitis, and diarrhea.
The adverse effects of tamoxifen include hot flashes and night sweats; vaginal dryness, discharge, or bleeding; uterine cancer; blood clots; and cataracts. The adverse effects of aromatase inhibitors include muscle aches and pains (arthralgias and myalgias), bone loss and bone fractures, vaginal dryness, and hot flashes.
Treatment-related new primary cancers are relatively rare. Lung cancer from radiation, uterine cancer from tamoxifen, and leukemia from anthracyclines and other chemotherapy agents such as cytoxan have been reported (Bhatia et al., 1996; Dracham et al., 2018; Travis et al., 1999, 2000). See Chapter 4 for more discussion of treatment-related cancers.
TREATMENT OF METASTATIC BREAST CANCER
Metastatic breast cancer at diagnosis (stage IV) is infrequent; most metastatic disease occurs several years after initial diagnosis of earlier stage disease and represents a recurrence of the original breast cancer that has spread to another part of the body, most commonly the liver, brain, bones, or lungs. Although the length of time that individuals live with metastatic breast cancer has been increasing with improved therapies, metastatic disease is generally felt to be incurable. About 6% of all women and about 9% of Black women are diagnosed with metastatic breast cancer at the time of their initial diagnosis (Howlader et al., 2020). Most women and men who develop metastatic disease do so at some point after the diagnosis of a potentially curable breast cancer which then recurs outside the breast (see section on recurrence). For example, among women with ER-positive breast cancer who received 5 years of adjuvant endocrine therapy, the risk of distant metastases ranged from 10% to 41% in the 15 years after cessation of endocrine therapy, with the risk being strongly correlated with the original TN status and tumor grade (Pan et al., 2017). While firm estimates of the number of women living with recurrent or metastatic disease breast cancer are not available, some reports suggest it may be more than 150,000 individuals (Mariotto et al., 2017) (see Figure 5-5). This is an important
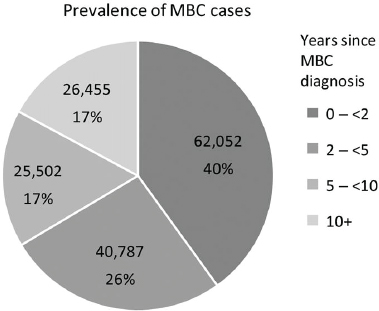
SOURCE: Mariotto et al., 2017, with permission from the American Association for Cancer Research.
population of patients, as these individuals are now living for extended periods of time on chronic cancer therapies, including many new investigational agents that are effectively targeting HR-positive and HER2-positive disease. See Chapter 10 for a discussion of the palliative and supportive care needs of cancer patients and survivors.
Younger women are more likely to be diagnosed with de novo metastatic disease, likely due to the absence of screening and delays in early diagnosis. This diagnosis has significant implications for the prognosis and treatment of these women. However, most women who die from breast cancer are initially diagnosed with localized disease and subsequently have a systemic recurrence (i.e., metastatic disease).
The treatment goals for metastatic disease are to improve longevity and quality of life. The National Comprehensive Cancer Network and other organizations encourage enrollment in clinical trials whenever possible as the best choice for treatment. Treatment for metastatic breast cancer, as with localized disease, is determined by HR and HER2 status, the timing and type of prior therapies, the presence of visceral crisis (i.e., the need to achieve a therapeutic response quickly because of impending or actual organ damage), and patient preference. TNBC is more likely to recur than the other subtypes, with a 5-year survival rate of approximately 77% for triple-negative tumors versus 90–94% for HR-positive and HER2-positive cancers (SEER, n.d.-b). Once breast cancer is metastatic, the median 5-year survival is estimated to be 27% (SEER, n.d.-a). However, due to the increasing number of effective medications for metastatic breast cancer, the
median survival of stage IV breast cancer is increasing, particularly for HER2-positive and ER-positive disease.
In this section, the systemic treatments and radiation therapy used for metastatic breast cancer are discussed.
Systemic Treatments
Systemic treatments for metastatic breast cancer include the use of endocrine therapies and chemotherapy as well as targeted therapies. In general, treatments are used until there is too much toxicity to continue or until the cancer grows. Breaks from therapy are often considered in the context of shared decision making with the patient.
Endocrine Therapy
Individuals with metastatic HR-positive tumors will generally receive a sequence of endocrine therapies until the time of toxicity or disease progression (Caswell-Jin et al., 2018; Cristofanilli et al., 2016). These therapies include tamoxifen, aromatase inhibitors, and fulvestrant (see Table 5-5). Often these endocrine therapies are used in combination with targeted therapies (see below) such as CDK4/6 inhibitors, everolimus, or alpelisib (see Table 5-5). When all endocrine options have been tried or if the clinical situation warrants, chemotherapy may then be used. Some metastatic tumors respond extremely well to endocrine therapies, and individuals can be on these medications for years or, rarely, even decades.
Chemotherapy
The choice of chemotherapy for metastatic breast cancer depends on a number of factors, including the clinical status of the patient and patient preference. For example, capecitabine is an oral chemotherapy medication that does not generally cause hair loss. Although sequential single chemotherapy agents are generally used for metastatic disease, combination chemotherapy with two of more agents is sometimes used, particularly if the patient has significant visceral disease and a rapid response to therapy is felt to be necessary. FDA has approved an extensive list of chemotherapy agents for metastatic breast cancer (NCI, 2020).
Targeted Therapies
Targeted therapies are frequently used in metastatic breast cancer. It is standard of care to combine endocrine therapy with other forms of targeted therapy. HER2-directed therapy is, by its nature, targeted therapy. Thus,
in both HER2-positive and HR-positive metastatic breast cancer, targeted therapies are frequently used either alone or in combination with other treatments.
HER2-Directed Therapy
For HER2-positive metastatic disease, many treatment options are available (see Table 5-5). There has been an explosion in available HER2targeted treatments in the past 10 years, which have led to an improvement in the overall survival for metastatic breast cancer. Some of these therapies are administered on their own (e.g., ado-trastuzumab-emtansine or trastuzumab deruxtecan), and some are administered in combination with chemotherapy agents such as lapatinib and tucatinib. Patients can be on these therapies for extended periods of time.
PARP Inhibitors
Multiple poly (ADP-ribose) polymerase (PARP) inhibitors have been approved by FDA as cancer therapeutics. Tumors in which there are defects in homologous DNA repair, such as those associated with mutations in BRCA1 and BRCA2, appear particularly susceptible to PARP inhibitors (Domchek et al., 2020). About 60% of women who inherit either a BRCA1 or BRCA2 mutation develop breast cancer by age 66 years (Kuchenbaecker et al., 2017). Approximately 5–10% of all breast cancers are associated with hereditary factors such as BRCA1/2 mutations (PDQ® Adult Treatment Editorial Board, 2020). FDA approved the PARP inhibitors talazoparib and olaparib for BRCA1/2-associated metastatic breast cancer (Litton et al., 2018; Robson et al., 2017). The side effects of PARP inhibitors include nausea, fatigue, and low blood counts.
CDK4/6 Inhibitors
FDA has approved cyclin-dependent kinase (CDK) 4/6 inhibitors in combination with endocrine therapy for both first-line and second-line treatment of HR-positive, HER2-negative, advanced or metastatic breast cancer. Cyclin-dependent kinase is an enzyme important in cell proliferation, and therefore these drugs work by targeting the cell cycle machinery. These oral medications include palbociclib, ribociclib, and abemaciclib and are given in combination with endocrine therapy such as aromatase inhibitors, tamoxifen, or fulvestrant (Cristofanilli et al., 2016; Goetz et al., 2017; Johnston et al., 2019; Turner et al., 2015, 2018). Multiple studies have demonstrated that CDK4/6 inhibitors plus endocrine therapy have significantly higher progression-free survival than endocrine therapy alone.
Patients can be on these for a prolonged period of time, with median time of progression-free survival in the range of 2 years.
mTOR Inhibitor
Everolimus is a mammalian target of rapamycin (mTOR) inhibitor approved for use in metastatic breast cancer in combination with endocrine therapy. mTOR is a protein that helps control cell division.
PI3K Kinase Inhibitors
Approximately 40% of ER-positive, HER2-negative metastatic breast cancers have a detectable PI3K (phosphoinositide 3-kinase) mutation. Alpelisib is a cyclin-dependent kinase inhibitor which, in combination with endocrine therapy, improves progression-free survival for these metastatic breast cancer patients. The side effects of alpelisib include elevated blood sugars, diarrhea, and rash (André et al., 2019).
Immunotherapy
Atezolizumab is a PD-1 (programmed cell death-1) inhibitor used to treat metastatic breast cancer. Atezolizumab in combination with the chemotherapeutic agent nab-paclitaxel (abraxne) has been shown to improve disease progression-free survival in patients with TNBC and PD-L1 (programmed death ligand-1) expression when administered as a first-line treatment. Immune side effects (such as thyroiditis, pneumonitis, colitis, and myocarditis) are common and can be serious (Schmid et al., 2018). In November 2020, FDA approved pembrolizumab, another PD-L1 inhibitor to treat locally recurrent unresectable or metastatic TNBC (FDA, 2020).
Bisphosphonates and Denosumab
A review by the Cochrane Collaborative found that in women with metastatic breast cancer with bone metastases, bisphosphonates reduced the risk of skeletal-related events by 14%, but had no effect on overall survival. Denosumab was slightly more effective in reducing the risk of skeletal-related events (22%), but also had no effect on survival (O’Carrigan et al., 2017).
Surgical Treatment
Occasionally surgery is used for the treatment of metastatic breast cancer. Situations in which surgery may be considered include the
removal of a single focus of metastatic breast cancer, the prevention of severe complications of metastatic breast cancer (such as malignant cord compression or pathological fracture of the leg), or the management of an existing complication of metastatic breast cancer (such as a pathological fracture of the leg). However, a recent clinical study has shown that compared with optimal systemic therapy, early local therapy with BCS followed by radiation therapy or mastectomy (with or without radiation therapy) does not improve survival in patients with de novo metastatic breast cancer and an intact primary tumor. Although there was an increased risk of local disease progression without local therapy, surgical removal of the intact primary tumor did not lead to improved quality of life (Khan et al., 2020).
Radiation Treatment
Palliative Radiation Therapy
Although the first-line treatment of metastatic breast cancer is systemic therapy, local treatments such as radiation therapy can be used to help improve or palliate a symptom caused by the disease, particularly pain from bone metastases. Radiation therapy reduces pain from bone metastases in more than 60% of patients and has been shown to eliminate pain completely in 21–25% of patients (Chow et al., 2007; Spencer et al., 2018).
Most palliative radiation therapy courses involve 1–10 treatments (Hartsell et al., 2005; Howell et al., 2013; Lutz et al., 2011), and are delivered using standard 3-D-conformal radiation therapy. Stereotactic body radiation therapy (SBRT), which delivers an ablative dose of radiation in 1–5 fractions, is sometimes used to treat painful bone metastases in the spine, particularly if the metastasis has already received prior palliative radiation therapy. Compared with standard palliative radiation therapy doses and techniques, the data on using SBRT to treat a de novo bone metastasis are mixed (Loi et al., 2019; Spencer et al., 2019) and clinical studies are under way. The biology of the breast cancer and the disease course are important for determining how many treatments a patient will receive. For instance, a patient with ER-positive breast cancer receiving endocrine therapy and a CDK4/6 inhibitor who is experiencing pain in a rib bone would most likely benefit from 10 radiation treatments instead of one treatment because her life expectancy is on the order of years and the likelihood of re-treatment for pain in that rib is high. By contrast, the optimum treatment for a patient with TNBC with the same degree of rib pain but whose cancer has progressed after three prior types of therapy may be only one radiation treatment because her life expectancy is much shorter.
Stereotactic Body Radiation Therapy for Oligometastatic Breast Cancer
In addition to its role in palliating painful bone metastases, SBRT is increasingly being used in patients with very limited (“oligometastatic”) cancers with the goal of completely eradicating the known sites of disease and potentially curing patients with limited metastatic disease (see Chapter 4). The approach of ablating oligometastases is commonly used in lung cancer on the basis of small, randomized studies, some of which included patients with breast cancer (see Chapter 6). In some cases, surgery can also be used to ablate sites of metastatic disease. SBRT has an advantage over surgery because there is less disruption in the delivery of systemic therapy and less acute morbidity. While the treatment of up to four metastases with SBRT has been deemed safe in a clinical trial (Chmura et al., 2019), SBRT can be associated with significant toxicities depending on the proximity of the metastasis to normal tissues, and some of these toxicities can be fatal (Palma et al., 2019). The results of a randomized clinical trial (NCT02364557) comparing SBRT to standard-of-care therapy (systemic therapy and palliative radiation, if indicated) are pending, and if SBRT proves to be beneficial by improving disease outcomes, it may become the standard of care for patients with limited metastatic breast disease (Chmura et al., 2019). Selecting patients with oligometastatic breast cancer for SBRT should carefully weigh the risks of treating the metastases against the potential benefits.
Recurrence
For women who have HR-positive cancers—the vast majority of breast cancer—treatment may result in a cure, but there is a continuing risk of cancer recurrence in the decades following the initial diagnosis (see Figure
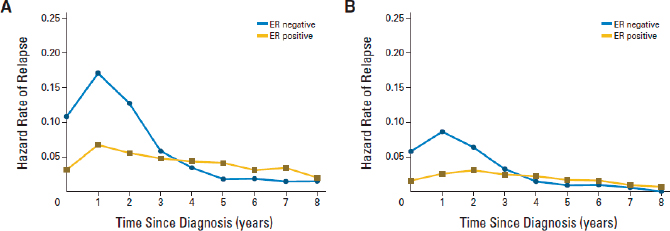
NOTE: ER = estrogen.
SOURCE: Cosetti et al., 2015, reprinted with permission © (2015) American Society of Clinical Oncology, all rights reserved.
5-6). These women may have recurrent metastatic cancer 10 to 25 years after their initial diagnosis, and these recurrent cancers can lead to morbidity, impairments, and functional limitations. In such cases the treatment for recurrence adds to any toxicity and morbidity that may have resulted from the initial cancer treatments.
LIFE AFTER BREAST CANCER DIAGNOSIS
Physical and mental impairment following a diagnosis of breast cancer can be due to the toxicity of the treatments used to cure a person or caused by the development of incurable metastatic disease. Thus, looking solely at the prognosis and expected mortality from breast cancer is an incomplete measure of the impairments and subsequent functional limitations that can result from a diagnosis of the disease.
To a large extent the post-treatment experience of early-stage breast cancer survivors is influenced by the specific local treatments they receive, such as the extent of breast surgery or radiation treatment. Many of the long-term and late-onset effects of cancer treatment, such as fatigue or cognitive complaints, may result from a variety of treatments used for local disease. For women with de novo metastatic or recurrent breast cancer, the physical effects often dominate and are influenced by the specific sites of metastatic disease (e.g., bone, lung, liver, or brain). Thus, the experience for survivors after a diagnosis of breast cancer is heterogeneous and cannot be easily categorized.
The impacts of cancer and its treatment on survivor functioning can be classified into two types: (1) long-term effects, which are symptoms or difficulties that begin during treatment and persist for some time after treatment ends (e.g., pain, fatigue, insomnia, neuropathy) (see Chapter 9); and (2) late-onset effects, which are problems that are not evident during active treatment but that emerge months and years after treatment ends (e.g., new-onset lymphedema, congestive heart failure, osteoporosis, and fracture). While these two categories of problems seem distinct, they often overlap, and their time of origin may not be easy to delineate. Long-term effects are more common and sometimes reflect the “new normal” for cancer survivors, while the late-onset effects are often rarer and more serious and can take the survivor by surprise, as they occur at a time that may be distant from the initial treatment. Furthermore, long-term and late-onset effects may occur individually, as clusters, or be synergistic; all of these effects may increase the likelihood of impairments and functional limitations.
In the sections below the committee briefly summarizes some of the long-term and late-onset effects that may result from breast cancer and its treatments. More information on many of these effects that result not only from breast cancer but other cancers considered in this report can also be found in Chapter 9.
Common Long-Term Effects of Breast Cancer Treatment
The most direct effects of treatment result from breast cancer surgery and radiation treatment, if given, and include pain, lymphedema, fatigue, cognitive impairments, and depression. Other late-onset and long-term effects include chemotherapy-induced peripheral neuropathy and reproductive system dysfunction.
Pain
Pain in the residual breast tissue and chest wall is common after breast surgery. Although this pain is most severe in the early months after surgery, for 25% of women it can persist indefinitely after surgery (Miaskowski et al., 2012). Pain often extends into the ipsilateral arm (i.e., the arm adjacent to the breast undergoing surgery) and axilla. Breast surgery may also limit the range of motion and result in a frozen shoulder if physical therapy is not performed early in the course of postoperative pain (Stubblefield and Custodio, 2006). Lumpectomy with radiation was expected to reduce the likelihood of these post-treatment physical effects, but that has not been observed (Shimozuma et al., 1999). Women may also have difficulty sleeping and resuming some physical activities, such as lifting, as a result of their chronic chest and breast pain.
Lymphedema
Lymphedema (swelling of the arm) on the side associated with breast surgery is more likely to occur in women who had more extensive axillary node surgery (axillary lymph node dissection versus sentinel lymph node dissection) in combination with radiation to the axillary area (Erickson et al., 2001; Naoum et al., 2020). Swelling of the breast can also be a problem if radiation is carried out after BCS. Other risk factors for lymphedema include weight gain, cellulitis, skin injury, and trauma. Edema may occur early in the postoperative period and can be helped by physical therapy interventions, such as manual lymphatic drainage, but it can become chronic, causing pain and arm dysfunction. Edema of the arm can also occur as a late-onset effect, appearing months to years after the end of treatment. Patients who have SLND are at much lower risk for arm pain and lymphedema in the year after surgery than those undergoing ALND. Nevertheless, a small number of survivors undergoing either surgery may have persistent pain and lymphedema out to 36 months (Land et al., 2010).
Fatigue
Fatigue is one of the most common acute side effects of cancer treatment and is generally most severe in those who receive multimodal therapy
(i.e., surgery, radiation, and chemotherapy). It often takes months to 1 year or more after treatment ends for the fatigue to resolve. In about 20% of breast cancer survivors, treatment-associated fatigue does not resolve and may persist for years (Bower et al., 2000, 2006). Fatigue after breast cancer treatment appears to be associated with persistent levels of inflammation, and some individuals appear to be genetically predisposed to have more difficulty recovering from that inflammation (Bower, 2014; Bower and Ganz, 2015; Bower et al., 2002, 2005). Because patients often appear well and have no laboratory evidence that explains the fatigue (i.e., normal chemistries, no anemia, normal thyroid function), the diagnosis of fatigue must be based on patient reports; given the lack of laboratory tests for fatigue, this complaint may be underreported and underappreciated (Bower, 2019). Fatigue patients are often unable to resume normal activities, must carefully manage their energy expenditure during the day, and have less time for leisure activities. Fatigue often co-occurs with cognitive difficulties (Bower and Ganz, 2015). A recent study that examined the trajectory for post-treatment fatigue in breast cancer survivors found that women with depressive symptoms and childhood adversity were at a higher risk of fatigue out to 6 years of follow-up (Bower et al., 2018).
Because of the prevalence of fatigue in breast cancer survivors, a number of interventions have been tested to address this ongoing problem. For example, integrative medicine interventions, such as yoga, have been found to be beneficial in addressing cancer-related fatigue in breast cancer survivors and may be effective in reducing inflammation (Bower et al., 2012, 2014). Other treatments for cancer-related fatigue are discussed in Chapter 9.
Cognitive Impairment
Cognitive impairment after breast cancer treatments can be associated with fatigue or may be an independent complaint. While only 15–20% of women may experience this problem, many are unable to do their usual home and professional work due to having difficulty remembering, planning, and multitasking. The issue was often referred to as “chemobrain” or “chemofog” because when early reports of this problem first appeared in the 1990s it was at a time when many women were receiving adjuvant chemotherapy for breast cancer, and high-dose chemotherapy with stem cell transplants was being used in an attempt to cure women with metastatic or locally advanced breast cancer (Ganz, 1998; van Dam et al., 1998). Since then it has been found that various treatments other than chemotherapy, including the endocrine therapies that are the mainstay of treatment for HR-positive breast cancer, may be causing the cognitive complaints reported by breast cancer patients (Ganz and Van Dyk, 2020; Ganz et al., 2014; Wagner et al., 2020). Premature menopause, a common result of adjuvant
chemotherapy in younger women, and also ovarian suppression therapy, now widely used in premenopausal breast cancer patients, may exacerbate the cognitive changes associated with adjuvant chemotherapy.
Individuals who are having persistent cognitive difficulties in association with breast cancer treatments should be evaluated by a neuropsychologist who is familiar with this complication of cancer treatment. While standardized neurocognitive test batteries can be used, there are some specific areas of potential deficit that should be investigated. In addition, uncontrolled depressive symptoms and insomnia may be important co-factors that need to be excluded or managed. Interventions that have been found to be helpful include cognitive rehabilitation and cognitive behavioral therapy (CBT). More information on the treatment of cognitive impairments from cancer treatments can be found in Chapter 9.
Depression
In addition to the disruptive physical symptoms faced by younger and mid-life women, the psychosocial impact of breast cancer can be substantial and can affect all aspects of life. Many women may have persistent anxiety, depression, and associated insomnia after a diagnosis of breast cancer or its treatment (IOM, 2004). These symptoms are often not identified by the cancer care team during clinical visits, and psychosocial counseling services for cancer patients may be unavailable. Although cancer survivors may appear to have recovered (e.g., their hair has grown back), they may still have mental health issues; this is particularly true for younger breast cancer survivors (Howard-Anderson et al., 2012). Depression occurs at a high rate in breast cancer survivors, and it can be lead to functional limitations. One study of newly diagnosed women found that 38% had elevated symptoms of depression at a 16-month follow-up (Stanton et al., 2015). While most women with breast cancer will recover from the psychosocial impact of their diagnosis and treatment, clinical interventions and medications may be needed for those with persistent symptoms (Stanton and Bower, 2015). Concomitant problems often include anxiety and insomnia, both of which may benefit from specific medications and behavioral interventions, such as CBT, or from supportive therapies such as Tai Chi Chih (Irwin et al., 2017). More information on depression can be found in Chapter 9.
Chemotherapy-Induced Peripheral Neuropathy
For the past two decades, taxane chemotherapy has been increasingly used as a component of adjuvant chemotherapy. While highly effective in the treatment of breast cancer, this class of drugs may result in serious chemotherapy-induced peripheral neuropathy (CIPN), which can then
become a dose-limiting toxicity. CIPN is common after breast cancer treatments end and does not resolve in a substantial number of breast cancer survivors (Rivera et al., 2018). Persistent CPIN can lead to impaired quality of life and functioning (Rivera et al., 2018) as well as an increased risk of falls (Winters-Stone et al., 2017). More information on CIPN can be found in Chapter 9.
Other Long-Term and Late-Onset Effects
Other important physical effects of cancer treatments relate to disruptions in the endocrine or menopausal status of women. Specifically, younger premenopausal women who are treated with cytotoxic chemotherapy may have an abrupt cessation of menses and the subsequent onset of vasomotor (hot flashes, night sweats, palpitations) and vaginal (dryness, pain with intercourse) symptoms, along with the potential for infertility (Barton and Ganz, 2015; Knobf, 2006). The cessation of menses may be temporary or permanent, with the latter more common in women aged 40 years or older (Ganz et al., 2011). If endocrine therapy with tamoxifen is used following chemotherapy-induced amenorrhea, then the amenorrhea is likely to persist much longer, although menses may return (Ganz et al., 2011; Knobf, 2006). However, endocrine therapy may cause the same vasomotor symptoms and lead to substantial impacts on quality of life. This therapy is given for 5–10 years and is difficult for many women to tolerate.
There may be other complications from breast cancer treatment, such as blood clots from tamoxifen or osteoporosis and fracture risk from premature menopause or endocrine therapy. New primary or subsequent cancers, such as endometrial cancer, can result from tamoxifen, especially in postmenopausal women. Some of the menopausal symptoms can be managed with medications. Rarely, women may experience serious depression in association with these hormonal changes. Impaired fertility is another loss these women face, and although there are opportunities to preserve fertility by the pretreatment collection of oocytes from the ovaries, which can be preserved with or without fertilization to create an embryo, the costs may be prohibitive for many women (Howard-Anderson et al., 2012; Pinto and de Azambuja, 2011).
For women with de novo or recurrent metastatic breast cancer, treatments may be chronic as long as the woman is alive, perhaps for as many as 15–20 years. This may include multiple episodes of recurrent/progressive disease, with use of radiation at various times to control pain or other symptoms, along with systemic treatments. Late-onset effects of cancer treatments include cardiac toxicity from chemotherapy and radiation as well as osteoporosis and fractures that may lead to additional functional limitations beyond the presence of metastatic disease itself.
FINDINGS AND CONCLUSIONS
Findings
- As of 2020 there are an estimated 3.5 million women and men with a past or current diagnosis of breast cancer alive in the United States.
- More than 90% of breast cancer patients present with localized disease that has not spread outside of the breast and regional lymph nodes. Most breast cancer is hormone (estrogen and/or progesterone)-receptor-positive and HER2-negative (68%); about 10% of breast cancer is hormone-receptor-negative and HER2negative (triple-negative breast cancer); 10% is hormone-receptor-positive and HER2-positive; and about 4% of breast cancer is hormone-receptor-negative and HER2-positive.
- Triple-negative breast cancer is more common in African American women as well as premenopausal women and has the worst prognosis of the subtypes of breast cancer; it is also the most likely to recur.
- Treatment is determined by guidelines that are based on clinical and biological factors, stage of cancer, family history, patient age, and patient preferences, and include a combination of local treatments (i.e., surgery and radiation therapy) or local treatments with systemic therapy.
- For localized breast cancer, lumpectomy followed by radiation therapy to the whole breast is considered equivalent to mastectomy in terms of cancer control and survival rates.
- Lymph node dissection has potential adverse effects including lymphedema, neuropathy, and seromas that can impair a survivor’s quality of life.
- For breast cancer that is hormone-receptor-positive and HER2negative, endocrine therapy (tamoxifen or an aromatase inhibitor) is a key component of systemic treatment. Adverse effects of aromatase inhibitors that may cause impairments include muscle aches and pains (arthralgias and myalgias) and bone loss.
- The long-term and late-onset effects of intensive chemotherapy include leukemia, cardiac dysfunction, chronic fatigue, and neuropathy, which can be life threatening and lead to functional limitations.
- The post-treatment experience of early-stage breast cancer, which may relate to impairments and functional limitations, is influenced by specific local treatments, such as the extent of breast surgery or radiation therapy.
- The impairments resulting from breast cancer and from its treatment fall into two main groups: long-term effects, which are symptoms or problems that start during treatment and persist long after treatment ends (e.g., pain, fatigue, insomnia, neuropathy, and cognitive complaints); and late-onset effects (i.e., impairments that emerge months to years after treatment ends—lymphedema, congestive heart failure, osteoporosis, and fracture).
- Most women who die from breast cancer are initially diagnosed with localized disease but subsequently have a systemic recurrence, resulting in metastatic disease.
- Women who present with de novo metastatic breast cancer at the time of their initial diagnosis are more likely to be younger. Metastatic breast cancer has significant implications for their prognosis and treatment. For women with de novo metastatic or recurrent metastatic breast cancer, the physical effects often dominate and are influenced by the sites of the metastatic disease (e.g., bone, lung, liver, or brain); however, these women may also have substantial ongoing psychosocial concerns.
Conclusions
- The 5-year survival for people with early-stage breast cancer (specifically, stage I, hormone-receptor-positive, and HER2-negative), the most commonly diagnosed breast cancer, is excellent, at 94–99%.
- Impairments associated with breast cancer and its treatment, even early-stage breast cancer, are highly variable and depend on many individual characteristics and the specific treatments the patient receives.
- There are many impairments that may result from breast cancer and its treatment, especially pain, fatigue, cognitive complaints, neuropathy, psychological issues, and lymphedema, which are often untreated and may lead to persistent functional limitations.
REFERENCES
ACS (American Cancer Society). 2019. Inflammatory breast cancer. https://www.cancer.org/cancer/breast-cancer/understanding-a-breast-cancer-diagnosis/types-of-breast-cancer/inflammatory-breast-cancer.html (accessed August 11, 2020).
Ager, B., P. Butow, J. Jansen, K.-A. Phillips, D. Porter, and CPM DA Advisory Group. 2016. Contralateral prophylactic mastectomy (CPM): A systematic review of patient reported factors and psychological predictors influencing choice and satisfaction. The Breast 28:107–120.
AJCC (American Joint Committee on Cancer). 2017. AJCC cancer staging manual, eighth edition. New York: Springer International Publishing.
Amin, M.B., F.L. Greene, S.B. Edge, C.C. Compton, J.E. Gershenwald, R.K. Brookland, L. Meyer, D.M. Gress, D.R. Byrd, and D.P. Winchester. 2017. The Eighth Edition AJCC Cancer Staging Manual: Continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA: A Cancer Journal for Clinicians 67(2):93–99.
André, F., E. Ciruelos, G. Rubovszky, M. Campone, S. Loibl, H.S. Rugo, H. Iwata, P. Conte, I.A. Mayer, B. Kaufman, T. Yamashita, Y.-S. Lu, K. Inoue, M. Takahashi, Z. Pápai, A.-S. Longin, D. Mills, C. Wilke, S. Hirawat, and D. Juric. 2019. Alpelisib for PIK3CAmutated, hormone receptor–positive advanced breast cancer. New England Journal of Medicine 380(20):1929–1940.
ASTRO (American Society for Radiation Oncology). 2018. ASTRO guideline on radiation therapy for the whole breast. https://www.astro.org/Patient-Care-and-Research/Clinical-Practice-Statements/ASTRO-39;s-guideline-on-whole-breast-irradiation (accessed August 21, 2020).
Barton, D.L., and P.A. Ganz. 2015. Symptoms: Menopause, infertility, and sexual health. Advances in Experimental Medicine and Biology 862:115–141.
Beumer, I., A. Witteveen, L. Delahaye, D. Wehkamp, M. Snel, C. Dreezen, J. Zheng, A. Floore, G. Brink, B. Chan, S. Linn, R. Bernards, L. van ‘t Veer, and A. Glas. 2016. Equivalence of Mammaprint array types in clinical trials and diagnostics. Breast Cancer Research and Treatment 156(2):279–287.
Bhatia, S., L.L. Robison, O. Oberlin, M. Greenberg, G. Bunin, F. Fossati-Bellani, and A.T. Meadows. 1996. Breast cancer and other second neoplasms after childhood Hodgkin’s disease. New England Journal of Medicine 334(12):745–751.
Bleyer, A., and H.G. Welch. 2012. Effect of three decades of screening mammography on breast-cancer incidence. New England Journal of Medicine 367(21):1998–2005.
Bower, J.E. 2014. Cancer-related fatigue—Mechanisms, risk factors, and treatments. Nature Reviews Clinical Oncology 11(10):597–609.
Bower, J.E. 2019. The role of neuro-immune interactions in cancer-related fatigue: Biobehavioral risk factors and mechanisms. Cancer 125(3):353–364.
Bower, J.E., and P.A. Ganz. 2015. Symptoms: Fatigue and cognitive dysfunction. Advances in Experimental Medicine and Biology 862:53–75.
Bower, J.E., P.A. Ganz, K.A. Desmond, J.H. Rowland, B.E. Meyerowitz, and T.R. Belin. 2000. Fatigue in breast cancer survivors: Occurrence, correlates, and impact on quality of life. Journal of Clinical Oncology 18(4):743–753.
Bower, J.E., P.A. Ganz, N. Aziz, and J.L. Fahey. 2002. Fatigue and proinflammatory cytokine activity in breast cancer survivors. Psychosomatic Medicine 64(4):604–611.
Bower, J.E., P.A. Ganz, and N. Aziz. 2005. Altered cortisol response to psychologic stress in breast cancer survivors with persistent fatigue. Psychosomatic Medicine 67(2):277–280.
Bower, J.E., P.A. Ganz, K.A. Desmond, C. Bernaards, J.H. Rowland, B.E. Meyerowitz, and T.R. Belin. 2006. Fatigue in long-term breast carcinoma survivors. Cancer 106(4):751–758.
Bower, J.E., D. Garet, B. Sternlieb, P.A. Ganz, M.R. Irwin, R. Olmstead, and G. Greendale. 2012. Yoga for persistent fatigue in breast cancer survivors: A randomized controlled trial. Cancer 118(15):3766–3775.
Bower, J.E., G. Greendale, A.D. Crosswell, D. Garet, B. Sternlieb, P.A. Ganz, M.R. Irwin, R. Olmstead, J. Arevalo, and S.W. Cole. 2014. Yoga reduces inflammatory signaling in fatigued breast cancer survivors: A randomized controlled trial. Psychoneuroendocrinology 43:20–29.
Bower, J.E., J. Wiley, L. Petersen, M.R. Irwin, S.W. Cole, and P.A. Ganz. 2018. Fatigue after breast cancer treatment: Biobehavioral predictors of fatigue trajectories. Health Psychology 37(11):1025–1034.
Cancer Australia. 2021. Illustration of lymph nodes near the breast. https://www.canceraustralia.gov.au/affected-cancer/cancer-types/breast-cancer/about-breast-cancer/what-breast-cancer/lymphatic-system (accessed January 4, 2021).
Carter, S.A., G.R. Lyons, H.M. Kuerer, R.L. Bassett, Jr., S. Oates, A. Thompson, A.S. Caudle, E.A. Mittendorf, I. Bedrosian, A. Lucci, S.M. DeSnyder, G. Babiera, M. Yi, D.P. Baumann, M.W. Clemens, P.B. Garvey, K.K. Hunt, and R.F. Hwang. 2016. Operative and oncologic outcomes in 9861 patients with operable breast cancer: Single-institution analysis of breast conservation with oncoplastic reconstruction. Annals of Surgical Oncology 23(10):3190–3198.
Caswell-Jin, J.L., S.K. Plevritis, L. Tian, C.J. Cadham, C. Xu, N.K. Stout, G.W. Sledge, J.S. Mandelblatt, and A.W. Kurian. 2018. Change in survival in metastatic breast cancer with treatment advances: Meta-analysis and systematic review. JNCI Cancer Spectrum 2(4):pky062.
Chagpar, A.B. 2014. Prophylactic bilateral mastectomy and contralateral prophylactic mastectomy. Surgical Oncology Clinics of North America 23(3):423–430.
Cheng, Y.J., X.Y. Nie, C.C. Ji, X.X. Lin, L.J. Liu, X.M. Chen, H. Yao, and S.H. Wu. 2017. Long-term cardiovascular risk after radiotherapy in women with breast cancer. Journal of the American Heart Association 6(5):e005633.
Chmura, S.J., K.A. Winter, H.A. Al-Hallaq, V.F. Borges, N.T. Jaskowiak, M. Matuszak, M.T. Milano, J.K. Salama, W.A. Woodward, and J.R. White. 2019. NRG-BR002: A phase IIR/III trial of standard of care therapy with or without stereotactic body radiotherapy (SBRT) and/or surgical ablation for newly oligometastatic breast cancer (NCT02364557). Journal of Clinical Oncology 37(15_suppl):TPS1117.
Chow, E., K. Harris, G. Fan, M. Tsao, and W.M. Sze. 2007. Palliative radiotherapy trials for bone metastases: A systematic review. Journal of Clinical Oncology 25(11):1423–1436.
Clough, K.B., D. Benyahi, C. Nos, C. Charles, and I. Sarfati. 2015. Oncoplastic surgery: Pushing the limits of breast-conserving surgery. The Breast Journal 21(2):140–146.
Cordova, L.Z., D.J. Hunter-Smith, and W.M. Rozen. 2019. Patient reported outcome measures (PROMS) following mastectomy with breast reconstruction or without reconstruction: A systematic review. Gland Surgery 8(4):441–451.
Cossetti, R.J., S.K. Tyldesley, C.H. Speers, Y. Zheng, and K.A. Gelmon. 2015. Comparison of breast cancer recurrence and outcome patterns between patients treated from 1986 to 1992 and from 2004 to 2008. Journal of Clinical Oncology 33(1):65–73.
Cristofanilli, M., N.C. Turner, I. Bondarenko, J. Ro, S.-A. Im, N. Masuda, M. Colleoni, A. DeMichele, S. Loi, S. Verma, H. Iwata, N. Harbeck, K. Zhang, K.P. Theall, Y. Jiang, C.H. Bartlett, M. Koehler, and D. Slamon. 2016. Fulvestrant plus palbociclib versus fulvestrant plus placebo for treatment of hormone-receptor-positive, HER2-negative metastatic breast cancer that progressed on previous endocrine therapy (PALOMA-3): Final analysis of the multicentre, double-blind, phase 3 randomised controlled trial. The Lancet Oncology 17(4):425–439.
Curigliano, G., H.J. Burstein, E.P. Winer, M. Gnant, P. Dubsky, S. Loibl, M. Colleoni, M.M. Regan, M. Piccart-Gebhart, H.J. Senn, B. Thürlimann, F. André, J. Baselga, J. Bergh, H. Bonnefoi, S.Y. Brucker, F. Cardoso, L. Carey, E. Ciruelos, J. Cuzick, C. Denkert, A. Di Leo, B. Ejlertsen, P. Francis, V. Galimberti, J. Garber, B. Gulluoglu, P. Goodwin, N. Harbeck, D.F. Hayes, C.S. Huang, J. Huober, K. Hussein, J. Jassem, Z. Jiang, P. Karlsson, M. Morrow, R. Orecchia, K.C. Osborne, O. Pagani, A. H. Partridge, K. Pritchard, J. Ro, E.J.T. Rutgers, R. Sedlmayer, V. Semiglazov, Z. Shao, I. Smith, M. Toi, A. Tutt, G. Viale, T. Watanabe, T.J. Whelan, and B. Xu. 2017. De-escalating and escalating treatments for early-stage breast cancer: The St. Gallen International Expert Consensus Conference on the Primary Therapy of Early Breast Cancer 2017. Annals of Oncology 28(8):1700–1712.
DeSantis, C.E., J. Ma, M.M. Gaudet, L.A. Newman, K.D. Miller, A. Goding Sauer, A. Jemal, and R.L. Siegel. 2019. Breast cancer statistics, 2019. CA: A Cancer Journal for Clinicians 69(6):438–451.
Domchek, S.M., E. Mardis, J.W. Carlisle, and T.K. Owonikoko. 2020. Integrating genetic and genomic testing into oncology practice. American Society of Clinical Oncology Educational Book 40:e259–e263.
Down, S.K., P.K. Jha, A. Burger, and M.I. Hussien. 2013. Oncological advantages of oncoplastic breast-conserving surgery in treatment of early breast cancer. Breast Journal 19(1):56–63.
Dracham, C.B., A. Shankar, and R. Madan. 2018. Radiation induced secondary malignancies: A review article. Radiation Oncology Journal 36(2):85–94.
EBCTCG (Early Breast Cancer Trialists’ Collaborative Group). 2015. Adjuvant bisphosphonate treatment in early breast cancer: Meta-analyses of individual patient data from randomised trials. The Lancet 386(10001):1353–1361.
Erickson, V.S., M.L. Pearson, P.A. Ganz, J. Adams, and K.L. Kahn. 2001. Arm edema in breast cancer patients. Journal of the National Cancer Institute 93(2):96–111.
FDA (U.S. Food and Drug Administration). 2020. FDA grants accelerated approval to pembrolizumab for locally recurrent unresectable or metastatic triple negative breast cancer. https://www.fda.gov/drugs/drug-approvals-and-databases/fda-grants-accelerated-approval-pembrolizumab-locally-recurrent-unresectable-or-metastatic-triple (accessed December 7, 2020).
Fisher, B., S. Anderson, J. Bryant, R.G. Margolese, M. Deutsch, E.R. Fisher, J.H. Jeong, and N. Wolmark. 2002. Twenty-year follow-up of a randomized trial comparing total mastectomy, lumpectomy, and lumpectomy plus irradiation for the treatment of invasive breast cancer. New England Journal of Medicine 347(16):1233–1241.
Ganz, P.A. 1998. Cognitive dysfunction following adjuvant treatment of breast cancer: A new dose-limiting toxic effect? Journal of the National Cancer Institute 90(3):182–183.
Ganz, P.A., and K. Van Dyk. 2020. Cognitive impairment in patients with breast cancer: Understanding the impact of chemotherapy and endocrine therapy. Journal of Clinical Oncology 38(17):1871–1874.
Ganz, P.A., S.R. Land, C.E. Geyer, R.S. Cecchini, J.P. Costantino, E.R. Pajon, L. Fehrenbacher, J.N. Atkins, J.A. Polikoff, V.G. Vogel, J.K. Erban, R.B. Livingston, E.A. Perez, E.P. Mamounas, N. Wolmark, and S.M. Swain. 2011. Menstrual history and quality-of-life outcomes in women with node-positive breast cancer treated with adjuvant therapy on the NSABP B-30 trial. Journal of Clinical Oncology 29(9):1110–1116.
Ganz, P.A., L. Petersen, S.A. Castellon, J.E. Bower, D.H. Silverman, S.W. Cole, M.R. Irwin, and T.R. Belin. 2014. Cognitive function after the initiation of adjuvant endocrine therapy in early-stage breast cancer: An observational cohort study. Journal of Clinical Oncology 32(31):3559–3567.
Goetz, M.P., M. Toi, M. Campone, J. Sohn, S. Paluch-Shimon, J. Huober, I.H. Park, O. Tredan, S.C. Chen, L. Manso, O.C. Freedman, G. Garnica Jaliffe, T. Forrester, M. Frenzel, S. Barriga, I.C. Smith, N. Bourayou, and A. Di Leo. 2017. MONARCH 3: Abemaciclib as initial therapy for advanced breast cancer. Journal of Clinical Oncology 35(32):3638–3646.
Hartsell, W.F., C.B. Scott, D.W. Bruner, C.W. Scarantino, R.A. Ivker, M. Roach, 3rd, J.H. Suh, W.F. Demas, B. Movsas, I.A. Petersen, A.A. Konski, C.S. Cleeland, N.A. Janjan, and M. DeSilvio. 2005. Randomized trial of short- versus long-course radiotherapy for palliation of painful bone metastases. Journal of the National Cancer Institute 97(11):798–804.
Haviland, J.S., J.R. Owen, J.A. Dewar, R.K. Agrawal, J. Barrett, P.J. Barrett-Lee, H.J. Dobbs, P. Hopwood, P.A. Lawton, B.J. Magee, J. Mills, S. Simmons, M.A. Sydenham, K. Venables, J.M. Bliss, and J.R. Yarnold. 2013. The UK standardisation of breast radiotherapy (START) trials of radiotherapy hypofractionation for treatment of early breast cancer: 10-year follow-up results of two randomised controlled trials. The Lancet Oncology 14(11):1086–1094.
Hepel, J.T., and D.E. Wazer. 2020. Update on partial breast irradiation. Clinical Breast Cancer S1526-8209(20):30081.
Howard-Anderson, J., P.A. Ganz, J.E. Bower, and A.L. Stanton. 2012. Quality of life, fertility concerns, and behavioral health outcomes in younger breast cancer survivors: A systematic review. Journal of the National Cancer Institute 104(5):386–405.
Howell, D.D., J.L. James, W.F. Hartsell, M. Suntharalingam, M. Machtay, J.H. Suh, W.F. Demas, H.M. Sandler, L.A. Kachnic, and L.B. Berk. 2013. Single-fraction radiotherapy versus multifraction radiotherapy for palliation of painful vertebral bone metastases-equivalent efficacy, less toxicity, more convenient: A subset analysis of radiation therapy oncology group trial 97-14. Cancer 119(4):888–896.
Howlader, N., A.M. Noone, M. Krapcho, D. Miller, A. Brest, M. Yu, J. Ruhl, Z. Tatalovich, A. Mariotto, D.R. Lewis, H.S. Chen, E.J. Feuer, and K.A. Cronin. 2020. SEER cancer statistics review,1975–2017. https://seer.cancer.gov/csr/1975_2017 (accessed September 1, 2020).
Hughes, K.S., L.A. Schnaper, J.R. Bellon, C.T. Cirrincione, D.A. Berry, B. McCormick, H.B. Muss, B.L. Smith, C.A. Hudis, E.P. Winer, and W.C. Wood. 2013. Lumpectomy plus tamoxifen with or without irradiation in women age 70 years or older with early breast cancer: Long-term follow-up of CALGB 9343. Journal of Clinical Oncology 31(19):2382–2387.
IOM (Institute of Medicine). 2004. Meeting psychosocial needs of women with breast cancer. Washington, DC: The National Academies Press.
Irwin, M.R., R. Olmstead, C. Carrillo, N. Sadeghi, P. Nicassio, P.A. Ganz, and J.E. Bower. 2017. Tai Chi Chih compared with cognitive behavioral therapy for the treatment of insomnia in survivors of breast cancer: A randomized, partially blinded, noninferiority trial. Journal of Clinical Oncology 35(23):2656–2665.
Jagsi, R., J. Jiang, A.O. Momoh, A. Alderman, S.H. Giordano, T.A. Buchholz, L.J. Pierce, S.J. Kronowitz, and B.D. Smith. 2016. Complications after mastectomy and immediate breast reconstruction for breast cancer: A claims-based analysis. Annals of Surgery 263(2):219–227.
Johnston, S., M. Martin, A. Di Leo, S.A. Im, A. Awada, T. Forrester, M. Frenzel, M.C. Hardebeck, J. Cox, S. Barriga, M. Toi, H. Iwata, and M.P. Goetz. 2019. MONARCH 3 final PFS: A randomized study of abemaciclib as initial therapy for advanced breast cancer. NPJ Breast Cancer 5:5.
Kamali, P., A. van Bommel, B. Becherer, R. Cooter, M.A.M. Mureau, A. Pusic, S. Siesling, R. van der Hulst, S.J. Lin, and H. Rakhorst. 2019. Immediate breast reconstruction in The Netherlands and the United States: A proof-of-concept to internationally compare quality of care using cancer registry data. Plastic and Reconstructive Surgery 144(4):565e–574e.
Kanbayashi, C., A.M. Thompson, E.-S.S. Hwang, A.H. Partridge, D.W. Rea, J. Wesseling, T. Shien, T. Mizutani, T. Shibata, and H. Iwata. 2019. The international collaboration of active surveillance trials for low-risk DCIS (LORIS, LORD, COMET, LORETTA). Journal of Clinical Oncology 37(15 Suppl):TPS603.
Kantor, O., C. Chang, R.J. Bleicher, M. Moran, J.L. Connolly, S.H. Kurtzman, K. Yao, and the National Accreditation Program for Breast Centers Data Working Group. 2019. Physician knowledge of breast cancer recurrence and contralateral breast cancer risk is associated with increased recommendations for contralateral prophylactic mastectomy: A survey of physicians at NAPBC-Accredited centers. Annals of Surgical Oncology 26(10):3080–3088.
Keenan, T.E., and S.M. Tolaney. 2020. Role of immunotherapy in triple-negative breast cancer. Journal of the National Comprehensive Cancer Network 18(4):479–489.
Khan, S.A., F. Zhao, L.J. Solin, L.J. Goldstein, D. Cella, M. Basik, M. Golshan, T.B. Julian, B.A. Pockaj, C.A. Lee, W. Razaq, J.A. Sparano, G.V. Babiera, I.A. Dy, S. Jain, P. Silverman, C. Fisher, A.J. Tevaarwerk, L.I. Wagner, and G.W. Sledge. 2020. A randomized phase III trial of systemic therapy plus early local therapy versus systemic therapy alone in women with de novo stage IV breast cancer: A trial of the ECOG-ACRIN Research Group (E2108). Journal of Clinical Oncology 38(18_suppl):LBA2.
Knobf, M.T. 2006. The influence of endocrine effects of adjuvant therapy on quality of life outcomes in younger breast cancer survivors. Oncologist 11(2):96–110.
Korde, L.A., D.R. Doody, L. Hsu, P.L. Porter, and K.E. Malone. 2018. Bisphosphonate use and risk of recurrence, second primary breast cancer, and breast cancer mortality in a population-based cohort of breast cancer patients. Cancer Epidemiology, Biomarkers & Prevention 27(2):165–173.
Kronowitz, S.J. 2010. Delayed-immediate breast reconstruction: Technical and timing considerations. Plastic and Reconstructive Surgery 125(2):463–474.
Kuchenbaecker, K.B., J.L. Hopper, D.R., Barnes, K.-A. Phillips, T.M. Mooij, M.-J. Roos-Blom, S. Jervis, F.E. van Leeuwen, R.L. Milne, N. Andrieu, D.E. Goldgar, M.B. Terry, M.A. Rookus, D.F. Easton, A.C. Antoniou, and the BRCA1 and BRCA2 Cohort Consortium. 2017. Risks of breast, ovarian, and contralateral breast cancer for BRCA1 and BRCA2 mutation carriers. JAMA 317(23):2402–2416.
Kummerow, K.L., L. Du, D.F. Penson, Y. Shyr, and M.A. Hooks. 2015. Nationwide trends in mastectomy for early-stage breast cancer. JAMA Surgery 150(1):9–16.
Kunkler, I.H., L.J. Williams, W.J. Jack, D.A. Cameron, and J.M. Dixon. 2015. Breast-conserving surgery with or without irradiation in women aged 65 years or older with early breast cancer (Prime II): A randomised controlled trial. Lancet Oncology 16(3):266–273.
Land, S.R., J.A. Kopec, T.B. Julian, A.M. Brown, S.J. Anderson, D.N. Krag, N.J. Christian, J.P. Costantino, N. Wolmark, and P.A. Ganz. 2010. Patient-reported outcomes in sentinel node-negative adjuvant breast cancer patients receiving sentinel-node biopsy or axillary dissection: National surgical adjuvant breast and bowel project phase III protocol B-32. Journal of Clinical Oncology 28(25):3929–3936.
Lee, M.C., K. Rogers, K. Griffith, K.A. Diehl, T.M. Breslin, V.M. Cimmino, A.E. Chang, L.A. Newman, and M.S. Sabel. 2009. Determinants of breast conservation rates: Reasons for mastectomy at a comprehensive cancer center. Breast Journal 15(1):34–40.
Lewis, D. 2019. Strategies emerge for treating residual TNBC post chemo. Targeted Therapies in Oncology, 8(15). https://www.targetedonc.com/view/strategies-emerge-for-treating-residual-tnbc-post-chemo (accessed December 10, 2020).
Litiere, S., G. Werutsky, I.S. Fentiman, E. Rutgers, M.R. Christiaens, E. Van Limbergen, M.H. Baaijens, J. Bogaerts, and H. Bartelink. 2012. Breast conserving therapy versus mastectomy for stage I–II breast cancer: 20 year follow-up of the EORTC 10801 phase 3 randomised trial. Lancet Oncology 13(4):412–419.
Litton, J.K., H.S. Rugo, J. Ettl, S.A. Hurvitz, A. Gonçalves, K.-H. Lee, L. Fehrenbacher, R. Yerushalmi, L.A. Mina, M. Martin, H. Roché, Y.-H. Im, R.G.W. Quek, D. Markova, I.C. Tudor, A.L. Hannah, W. Eiermann, and J.L. Blum. 2018. Talazoparib in patients with advanced breast cancer and a germline BRCA mutation. New England Journal of Medicine 379(8):753–763.
Livi, L., I. Meattini, L. Marrazzo, G. Simontacchi, S. Pallotta, C. Saieva, F. Paiar, V. Scotti, C. De Luca Cardillo, P. Bastiani, L. Orzalesi, D. Casella, L. Sanchez, J. Nori, M. Fambrini, and S. Bianchi. 2015. Accelerated partial breast irradiation using intensity-modulated radiotherapy versus whole breast irradiation: 5-year survival analysis of a phase 3 randomised controlled trial. European Jounal Cancer 51(4):451–463.
Loi, M., J.J. Nuyttens, I. Desideri, D. Greto, L. Livi. 2019. Single-fraction radiotherapy (SFRT) for bone metastases: Patient selection and perspectives. Cancer Management and Research 11:9397–9408.
Loibl, S., J. O’Shaughnessy, M. Untch, W.M. Sikov, H.S. Rugo, M.D. McKee, J. Huober, M. Golshan, G. von Minckwitz, D. Maag, D. Sullivan, N. Wolmark, K. McIntyre, J.J. Ponce Lorenzo, O. Metzger Filho, P. Rastogi, W.F. Symmans, X. Liu, and C.E. Geyer. 2018. Addition of the PARP inhibitor veliparib plus carboplatin or carboplatin alone to standard neoadjuvant chemotherapy in triple-negative breast cancer (BrighTNess): A randomised, phase 3 trial. The Lancet Oncology 19(4):497–509.
Lutz, S., L. Berk, E. Chang, E. Chow, C. Hahn, P. Hoskin, D. Howell, A. Konski, L. Kachnic, S. Lo, A. Sahgal, L. Silverman, C. von Gunten, E. Mendel, A. Vassil, D.W. Bruner, and W. Hartsell. 2011. Palliative radiotherapy for bone metastases: An ASTRO evidence-based guideline. International Journal of Radiation Oncology, Biology, Physics 79(4):965–976.
Mariotto, A.B., R. Etzioni, M. Hurlbert, L. Penberthy, and M. Mayer. 2017. Estimation of the number of women living with metastatic breast cancer in the United States. Cancer Epidemiol Biomarkers & Prevention 26(6):809–815.
Miaskowski, C., B. Cooper, S.M. Paul, C. West, D. Langford, J.D. Levine, G. Abrams, D. Hamolsky, L. Dunn, M. Dodd, J. Neuhaus, C. Baggott, A. Dhruva, B. Schmidt, J. Cataldo, J. Merriman, and B.E. Aouizerat. 2012. Identification of patient subgroups and risk factors for persistent breast pain following breast cancer surgery. The Journal of Pain 13(12):1172–1187.
Moran, M.S., S.J. Schnitt, A.E. Giuliano, J.R. Harris, S.A. Khan, J. Horton, S. Klimberg, M. Chavez-MacGregor, G. Freedman, N. Houssami, P.L. Johnson, and M. Morrow. 2014. Society of Surgical Oncology–American Society for Radiation Oncology consensus guideline on margins for breast-conserving surgery with whole-breast irradiation in stages I and II invasive breast cancer. Annals of Surgical Oncology 21(3):704–716.
Morrow, M., K.J. Van Zee, L.J. Solin, N. Houssami, M. Chavez-MacGregor, J.R. Harris, J. Horton, S. Hwang, P.L. Johnson, M.L. Marinovich, S.J. Schnitt, I. Wapnir, and M.S. Moran. 2016. Society of Surgical Oncology–American Society for Radiation Oncology–American Society of Clinical Oncology consensus guideline on margins for breast-conserving surgery with whole-breast irradiation in ductal carcinoma in situ. Journal of Clinical Oncology 34(33):4040–4046.
Murray Brunt, A., J.S. Haviland, D.A. Wheatley, M.A. Sydenham, A. Alhasso, D.J. Bloomfield, C. Chan, M. Churn, S. Cleator, C.E. Coles, A. Goodman, A. Harnett, P. Hopwood, A.M. Kirby, C.C. Kirwan, C. Morris, Z. Nabi, E. Sawyer, N. Somaiah, L. Stones, I. Syndikus, J.M. Bliss, and J.R. Yarnold. 2020. Hypofractionated breast radiotherapy for 1 week versus 3 weeks (fast-forward): 5-year efficacy and late normal tissue effects results from a multicentre, non-inferiority, randomised, phase 3 trial. The Lancet 395(10237):1613–1626.
Naoum, G.E., S. Roberts, C.L. Brunelle, A.M. Shui, L. Salama, K. Daniell, T. Gillespie, L. Bucci, B.L. Smith, A.Y. Ho, and A.G. Taghian. 2020. Quantifying the impact of axillary surgery and nodal irradiation on breast cancer–related lymphedema and local tumor control: Long-term results from a prospective screening trial. Journal of Clinical Oncology 38(29):3430–3438.
Narod, S.A., J. Iqbal, V. Giannakeas, V. Sopik, and P. Sun. 2015. Breast cancer mortality after a diagnosis of ductal carcinoma in situ. JAMA Oncology 1(7):888–896.
NCCN (National Comprehensive Cancer Network). 2020a. NCCN clinical practice guidelines in oncology: Breast cancer. Version 6.2020. https://www.nccn.org/professionals/physician_gls/pdf/breast.pdf (accessed October 13, 2020).
NCCN. 2020b. NCCN guidelines for patients: Invasive breast cancer. https://www.nccn.org/patients/guidelines/content/PDF/breast-invasive-patient.pdf (accessed October 13, 2020).
NCI (National Cancer Institute). 2020. Drugs approved for breast cancer. https://www.cancer.gov/about-cancer/treatment/drugs/breast (accessed August 11, 2020).
Nelson, H.D., K. Tyne, A. Naik, C. Bougatsos, B.K. Chan, and L. Humphrey. 2009. Screening for breast cancer: An update for the U.S. Preventive Services Task Force. Annals of Internal Medicine 151(10):727–737.
Nelson, H.D., M. Pappas, A. Cantor, J. Griffin, M. Daeges, and L. Humphrey. 2016. Harms of breast cancer screening: Systematic review to update the 2009 U.S. Preventive Services Task Force recommendation. Annals of Internal Medicine 164(4):256–267.
O’Carrigan, B., M.H. Wong, M.L. Willson, M.R. Stockler, N. Pavlakis, and A. Goodwin. 2017. Bisphosphonates and other bone agents for breast cancer. Cochrane Database of Systematic Reviews 10(10):CD003474.
Olsen, M.A., K.B. Nickel, I.K. Fox, J.A. Margenthaler, A.E. Wallace, and V.J. Fraser. 2017. Comparison of wound complications after immediate, delayed, and secondary breast reconstruction procedures. JAMA Surgery 152(9):e172338.
Palma, D.A., R. Olson, S. Harrow, S. Gaede, A.V. Louie, C. Haasbeek, L. Mulroy, M. Lock, G.B. Rodrigues, B.P. Yaremko, D. Schellenberg, B. Ahmad, G. Griffioen, S. Senthi, A. Swaminath, N. Kopek, M. Liu, K. Moore, S. Currie, G.S. Bauman, A. Warner, and S. Senan. 2019. Stereotactic ablative radiotherapy versus standard of care palliative treatment in patients with oligometastatic cancers (SABR-COMET): A randomised, phase 2, open-label trial. Lancet 393(10185):2051–2058.
Pan, H., R. Gray, J. Braybrooke, C. Davies, C. Taylor, P. McGale, R. Peto, K.I. Pritchard, J. Bergh, M. Dowsett, and D.F. Hayes for the Early Breast Cancer Trialists’ Collaborative Group. 2017. 20-year risks of breast-cancer recurrence after stopping endocrine therapy at 5 years. New England Journal of Medicine 377(19):1836–1846.
PDQ® Adult Treatment Editorial Board. 2020. PDQ breast cancer treatment (adult). Bethesda, MD: National Cancer Institute. https://www.cancer.gov/types/breast/patient/breast-treatment-pdq (accessed November 2, 2020).
Pinto, A.C., and E. de Azambuja. 2011. Improving quality of life after breast cancer: Dealing with symptoms. Maturitas 70(4):343–348.
Poggi, M.M., D.N. Danforth, L.C. Sciuto, S.L. Smith, S.M. Steinberg, D.J. Liewehr, C. Menard, M.E. Lippman, A.S. Lichter, and R.M. Altemus. 2003. Eighteen-year results in the treatment of early breast carcinoma with mastectomy versus breast conservation therapy: The National Cancer Institute randomized trial. Cancer 98(4):697–702.
Poortmans, P.M., S. Collette, C. Kirkove, E. Van Limbergen, V. Budach, H. Struikmans, L. Collette, A. Fourquet, P. Maingon, M. Valli, K. De Winter, S. Marnitz, I. Barillot, L. Scandolaro, E. Vonk, C. Rodenhuis, H. Marsiglia, N. Weidner, G. van Tienhoven, C. Glanzmann, A. Kuten, R. Arriagada, H. Bartelink, and W. Van den Bogaert. 2015. Internal mammary and medial supraclavicular irradiation in breast cancer. New England Journal of Medicine 373(4):317–327.
Rivera, D.R., P.A. Ganz, M.S. Weyrich, H. Bandos, and J. Melnikow. 2018. Chemotherapy-associated peripheral neuropathy in patients with early-stage breast cancer: A systematic review. Journal of the National Cancer Institute 110(2):djx140.
Robson, M., S.-A. Im, E. Senkus, B. Xu, S.M. Domchek, N. Masuda, S. Delaloge, W. Li, N. Tung, A. Armstrong, W. Wu, C. Goessl, S. Runswick, and P. Conte. 2017. Olaparib for metastatic breast cancer in patients with a germline BRCA mutation. New England Journal of Medicine 377(6):523–533.
Ruddy, K.J., and P.A. Ganz. 2019. Treatment of nonmetastatic breast cancer. JAMA 321(17):1716–1717.
Saadatmand, S., H.A. Geuzinge, E.J.T. Rutgers, R.M. Mann, D.B.W. de Roy van Zuidewijn, H.M. Zonderland, R.A.E.M. Tollenaar, M.B.I. Lobbes, M.G.E.M. Ausems, M. van ‘t Riet, M.J. Hooning, I. Mares-Engelberts, E.J.T. Luiten, E.A.M. Heijnsdijk, C. Verhoef, N. Karssemeijer, J.C. Oosterwijk, I.-M. Obdeijn, H.J. de Koning, M.M.A. Tilanus-Linthorst, C.H.M. van Deurzen, C.E. Loo, J. Wesseling, M. Schlooz-Vries, S. van der Meij, W. Mesker, K. Keymeulen, C. Contant, E. Madsen, L.B. Koppert, J. Rothbarth, W.B. Veldhuis, A.J. Witkamp, E. Tetteroo, C. de Monye, M.M. van Rosmalen, J. Remmelzwaal, H.B.W. Gort, R. Roi-Antonides, M.N.J.M. Wasser, and E. van Druten. 2019. MRI versus mammography for breast cancer screening in women with familial risk (FaMRIsc): A multicentre, randomised, controlled trial. The Lancet Oncology 20(8):1136–1147.
Sando, I.C., J.I. Billig, S.W. Ambani, C.T. Kraft, K.M. Kidwell, L. Zhong, K.C. Chung, and A.O. Momoh. 2018. An evaluation of the choice for contralateral prophylactic mastectomy and patient concerns about recurrence in a reconstructed cohort. Annals of Plastic Surgery 80(4):333–338.
Schmid, P., S. Adams, H.S. Rugo, A. Schneeweiss, C.H. Barrios, H. Iwata, V. Diéras, R. Hegg, S.A. Im, G. Shaw Wright, V. Henschel, L. Molinero, S.Y. Chui, R. Funke, A. Husain, E.P. Winer, S. Loi, and L.A. Emens. 2018. Atezolizumab and Nab-paclitaxel in advanced triple-negative breast cancer. New England Journal of Medicine 379(22):2108–2121.
Scott, A.R., K.C. Stoltzfus, L.T. Tchelebi, D.M. Trifiletti, E.J. Lehrer, P. Rao, A. Bleyer, and N.G. Zaorsky. 2020. Trends in cancer incidence in US adolescents and young adults, 1973–2015. JAMA Network Open 3(12):e2027738.
SEER (Surveillance, Epidemiology, and End Results). n.d.-a. Cancer stat facts: Female breast cancer. https://seer.cancer.gov/statfacts/html/breast.html (accessed September 25, 2020).
SEER. n.d.-b. Cancer stat facts: Female breast cancer subtypes. https://seer.cancer.gov/statfacts/html/breast-subtypes.html (accessed October 29, 2020).
Shaitelman, S.F., Y.J. Chiang, K.D. Griffin, S.M. DeSnyder, B.D. Smith, M.V. Schaverien, W.A. Woodward, and J.N. Cormier. 2017. Radiation therapy targets and the risk of breast cancer-related lymphedema: A systematic review and network meta-analysis. Breast Cancer Research and Treatment 162(2):201–215.
Shimozuma, K., P.A. Ganz, L. Petersen, and K. Hirji. 1999. Quality of life in the first year after breast cancer surgery: Rehabilitation needs and patterns of recovery. Breast Cancer Research and Treatment 56(1):45–57.
Smith, B.D., J.R. Bellon, R. Blitzblau, G. Freedman, B. Haffty, C. Hahn, F. Halberg, K. Hoffman, K. Horst, J. Moran, C. Patton, J. Perlmutter, L. Warren, T. Whelan, J.L. Wright, and R. Jagsi. 2018. Radiation therapy for the whole breast: Executive summary of an American Society for Radiation Oncology (ASTRO) evidence-based guideline. Practical Radiation Oncology 8(3):145–152.
Sparano, J.A., R.J. Gray, D.F. Makower, K.I. Pritchard, K.S. Albain, D.F. Hayes, C.E. Geyer, E.C. Dees, M.P. Goetz, J.A. Olson, T. Lively, S.S. Badve, T.J. Saphner, L.I. Wagner, T.J. Whelan, M.J. Ellis, S. Paik, W.C. Wood, P.M. Ravdin, M.M. Keane, H.L. Gomez Moreno, P.S. Reddy, T.F. Goggins, I.A. Mayer, A.M. Brufsky, D.L. Toppmeyer, V.G. Kaklamani, J.L. Berenberg, J. Abrams, and G.W. Sledge. 2018. Adjuvant chemotherapy guided by a 21-gene expression assay in breast cancer. New England Journal of Medicine 379(2):111–121.
Sparano, J.A., R.J. Gray, P.M. Ravdin, D.F. Makower, K.I. Pritchard, K.S. Albain, D.F. Hayes, C.E. Geyer, E.C. Dees, M.P. Goetz, J.A. Olson, T. Lively, S.S. Badve, T.J. Saphner, L.I. Wagner, T.J. Whelan, M.J. Ellis, S. Paik, W.C. Wood, M.M. Keane, H.L. Gomez Moreno, P.S. Reddy, T.F. Goggins, I.A. Mayer, A.M. Brufsky, D.L. Toppmeyer, V.G. Kaklamani, J.L. Berenberg, J. Abrams, and G.W. Sledge. 2019. Clinical and genomic risk to guide the use of adjuvant therapy for breast cancer. New England Journal of Medicine 380(25):2395–2405.
Spencer, K., R. Parrish, R. Barton, and A. Henry. 2018. Palliative radiotherapy. BMJ 360:k821.
Spencer, K.L., J.M. van der Velden, E. Wong, E. Seravalli, A. Sahgal, E. Chow, J.J. Verlaan, H.M. Verkooijen, and Y.M. van der Linden. 2019. Systematic review of the role of stereotactic radiotherapy for bone metastases. Journal of the National Cancer Institute 111(10):1023–1032.
Staley, H., I. McCallum, and J. Bruce. 2014. Postoperative tamoxifen for ductal carcinoma in situ: Cochrane systematic review and meta-analysis. Breast 23(5):546–551.
Stanton, A.L., and J.E. Bower. 2015. Psychological adjustment in breast cancer survivors. Advances in Experimental Medicine and Biology 862:231–242.
Stanton, A.L., J.F. Wiley, J.L. Krull, C.M. Crespi, C. Hammen, J.J.B. Allen, M.L. Barrón, A. Jorge, and K.L. Weihs. 2015. Depressive episodes, symptoms, and trajectories in women recently diagnosed with breast cancer. Breast Cancer Research and Treatment 154(1):105–115.
Stubblefield, M.D., and C.M. Custodio. 2006. Upper-extremity pain disorders in breast cancer. Archives of Physical Medicine and Rehabilitation 87(3 Suppl 1):S96–S99.
Subhedar, P., C. Olcese, S. Patil, M. Morrow, and K.J. Van Zee. 2015. Decreasing recurrence rates for ductal carcinoma in situ: Analysis of 2996 women treated with breast-conserving surgery over 30 years. Annals of Surgical Oncology 22(10):3273–3281.
Torre, L.A., F. Islami, R.L. Siegel, E.M. Ward, and A. Jemal. 2017. Global cancer in women: Burden and trends. Cancer Epidemiology, Biomarkers & Prevention 26(4):444–457.
Travis, L.B., E.J. Holowaty, K. Bergfeldt, C.F. Lynch, B.A. Kohler, T. Wiklund, R.E. Curtis, P. Hall, M. Andersson, E. Pukkala, J. Sturgeon, H. Storm, E.A. Clarke, J.D. Boice, M. Gospodarowicz, and M. Stovall. 1999. Risk of leukemia after platinum-based chemotherapy for ovarian cancer. New England Journal of Medicine 340(5):351–357.
Travis, L.B., M. Andersson, M. Gospodarowicz, F.E. van Leeuwen, K. Bergfeldt, C.F. Lynch, R.E. Curtis, B.A. Kohler, T. Wiklund, H. Storm, E. Holowaty, P. Hall, E. Pukkala, D.T. Sleijfer, E.A. Clarke, J.D. Boice, Jr., M. Stovall, and E. Gilbert. 2000. Treatment-associated leukemia following testicular cancer. Journal of the National Cancer Institute 92(14):1165–1171.
Turner, N.C., J. Ro, F. André, S. Loi, S. Verma, H. Iwata, N. Harbeck, S. Loibl, C. Huang Bartlett, K. Zhang, C. Giorgetti, S. Randolph, M. Koehler, and M. Cristofanilli. 2015. Palbociclib in hormone-receptor–positive advanced breast cancer. New England Journal of Medicine 373(3):209–219.
Turner, N.C., D.J. Slamon, J. Ro, I. Bondarenko, S.-A. Im, N. Masuda, M. Colleoni, A. DeMichele, S. Loi, S. Verma, H. Iwata, N. Harbeck, S. Loibl, F. André, K. Puyana Theall, X. Huang, C. Giorgetti, C. Huang Bartlett, and M. Cristofanilli. 2018. Overall survival with palbociclib and fulvestrant in advanced breast cancer. New England Journal of Medicine 379(20):1926–1936.
van Dam, F.S., S.B. Schagen, M.J. Muller, W. Boogerd, E. vd Wall, M.E. Droogleever Fortuyn, and S. Rodenhuis. 1998. Impairment of cognitive function in women receiving adjuvant treatment for high-risk breast cancer: High-dose versus standard-dose chemotherapy. Journal of the National Cancer Institute 90(3):210–218.
van Dongen, J.A., A.C. Voogd, I.S. Fentiman, C. Legrand, R.J. Sylvester, D. Tong, E. van der Schueren, P.A. Helle, K. van Zijl, and H. Bartelink. 2000. Long-term results of a randomized trial comparing breast-conserving therapy with mastectomy: European Organization for Research and Treatment of Cancer 10801 trial. Journal of the National Cancer Institute 92(14):1143–1150.
Veronesi, U., N. Cascinelli, L. Mariani, M. Greco, R. Saccozzi, A. Luini, M. Aguilar, and E. Marubini. 2002. Twenty-year follow-up of a randomized study comparing breast-conserving surgery with radical mastectomy for early breast cancer. New England Journal of Medicine 347(16):1227–1232.
Wagner, L.I., R.J. Gray, J.A. Sparano, T.J. Whelan, S.F. Garcia, B. Yanez, A.J. Tevaarwerk, R.C. Carlos, K.S. Albain, J.A. Olson, Jr., M.P. Goetz, K.I. Pritchard, D.F. Hayes, C.E. Geyer, E.C. Dees, W.J. McCaskill-Stevens, L.M. Minasian, G.W. Sledge, Jr., and D. Cella. 2020. Patient-reported cognitive impairment among women with early breast cancer randomly assigned to endocrine therapy alone versus chemoendocrine therapy: Results from TAILORx. Journal of Clinical Oncology 38(17):1875–1886.
Ward, E.M., R.L. Sherman, S.J. Henley, A. Jemal, D.A. Siegel, E.J. Feuer, A.U. Firth, B.A. Kohler, S. Scott, J. Ma, R.N. Anderson, V. Benard, and K.A. Cronin. 2019. Annual report to the nation on the status of cancer, featuring cancer in men and women age 20–49 years. Journal of the National Cancer Institute 111(12):1279–1297.
Wennstig, A.K., C. Wadsten, H. Garmo, I. Fredriksson, C. Blomqvist, L. Holmberg, G. Nilsson, and M. Sund. 2020. Long-term risk of ischemic heart disease after adjuvant radiotherapy in breast cancer: Results from a large population-based cohort. Breast Cancer Research 22:10.
Werner, E.M., M.C. Eggert, S. Bohnet, and D. Rades. 2019. Prevalence and characteristics of pneumonitis following irradiation of beast cancer. Anticancer Research 39(11):6355–6358.
Whelan, T.J., J.-P. Pignol, M.N. Levine, J.A. Julian, R. MacKenzie, S. Parpia, W. Shelley, L. Grimard, J. Bowen, H. Lukka, F. Perera, A. Fyles, K. Schneider, S. Gulavita, and C. Freeman. 2010. Long-term results of hypofractionated radiation therapy for breast cancer. New England Journal of Medicine 362(6):513–520.
Whelan, T.J., I.A. Olivotto, W.R. Parulekar, I. Ackerman, B.H. Chua, A. Nabid, K.A. Vallis, J.R. White, P. Rousseau, A. Fortin, L.J. Pierce, L. Manchul, S. Chafe, M.C. Nolan, P. Craighead, J. Bowen, D.R. McCready, K.I. Pritchard, K. Gelmon, Y. Murray, J.-A. W. Chapman, B.E. Chen, M.N. Levine, for the MA.20 Study investigators. 2015. Regional nodal irradiation in early-stage breast cancer. New England Journal of Medicine 373(4):307–316.
Winters-Stone, K.M., F. Horak, P.G. Jacobs, P. Trubowitz, N.F. Dieckmann, S. Stoyles, and S. Faithfull. 2017. Falls, functioning, and disability among women with persistent symptoms of chemotherapy-induced peripheral neuropathy. Journal of Clinical Oncology 35(23):2604–2612.
















































