9
Cardiometabolic Diseases
Deaths due to cardiometabolic diseases include the following cause-of-death categories: endocrine, nutritional, and metabolic (ENM) diseases (e.g., thyroid conditions, diabetes, hyperlipidemia, obesity); hypertensive heart disease (e.g., heart disease caused by prolonged exposure to high blood pressure); and ischemic heart disease and other diseases of the circulatory system (e.g., reduced blood supply to the heart, including atherosclerosis and coronary heart disease (CHD), stroke, and other cardiovascular conditions). Collectively, cardiometabolic diseases were responsible for more than 4.8 million deaths among the U.S. working-age (ages 25–64) population between 1990 and 2017 (Centers for Disease Control and Prevention [CDC], 2020b). ENM diseases accounted for 703,247 deaths; hypertensive heart disease for 360,309 deaths; and ischemic heart disease and other diseases of the circulatory system for the largest share, 3,782,186 deaths (CDC, 2020b).
The contribution of cardiometabolic mortality to the recent rise in working-age mortality is complex and involves several countervailing trends. Death rates due to ENM diseases and hypertensive heart disease generally increased from 1990 to 2017, especially since 2010. While significant long-term reductions in mortality from ischemic heart disease and other diseases of the circulatory system had occurred since 1990, much of that progress appears to have stalled after 2010. The combination of these trends operated to increase all-cause working-age mortality after 2010 because the slowdown in declines in mortality from ischemic heart disease and other circulatory diseases no longer offset the rise in mortality from ENM diseases and hypertensive heart disease.
Within the working-age population, certain subgroups experienced greater relative increases in mortality from ENM diseases and hypertensive heart disease over the past three decades and slower declines in mortality from ischemic heart disease and other circulatory diseases in the recent decade—notably younger adults (ages 25–44) of all racial/ethnic groups, non-Hispanic (NH) White (White) males and females, NH Black (Black) males (in the recent decade), and those living in rural areas (exceptions are detailed in the next section). These troubling changes in cardiometabolic mortality have been most pronounced in the South and in nonmetropolitan areas.
Among the potential explanations for these patterns are three relevant trends: the obesity epidemic; diminishing returns of medical advances; and social, economic, and cultural changes. While all three sets of factors played some role in cardiometabolic mortality trends, the evidence suggests that rising obesity—an epidemic that has now spanned four decades—has exerted the greatest influence. The evidence also shows that younger adults, particularly those born in the late 1970s and early 1980s, who have been exposed to obesogenic environments for their entire lives, have experienced more adverse endocrine, metabolic, and cardiovascular consequences relative to older adults who averted this exposure as children and young adults. Moreover, vulnerable populations (racial/ethnic minorities, less-educated Whites, and rural populations) experienced higher rates of obesity and chronic stress due to social and economic conditions, together with less access to effective medical interventions for cardiometabolic disease, contributing to persistent disparities in cardiometabolic mortality across population subgroups and geographic areas. This chapter first summarizes the trends in cardiometabolic mortality by age, sex, race and ethnicity, and geography, and then reviews the evidence salient to explaining changes and disparities in these trends. The chapter ends with several recommendations for future research and policy evaluations.
TRENDS IN CARDIOMETABOLIC MORTALITY
Endocrine, Nutritional, and Metabolic Diseases
During the study period (1990–2017), trends in mortality from ENM diseases were largely similar among working-age males and females within each age group (Figure 9-1). Among younger working-age adults (25–44) mortality from these diseases increased slowly throughout the 2000s and 2010s, with the largest increases occurring among Black males. Among older working-age adults (45–64), Whites experienced gradual increases in mortality from ENM diseases throughout the period, while trends among Blacks and Hispanics were marked by decreasing mortality in the 2000s
and a subsequent leveling off and/or increase during the 2010s. In every age group, the largest increase in ENM mortality in the 2010s occurred among Black males.
Differences between White and Hispanic adults in ENM mortality were minimal among those ages 25–54. Among older adults (ages 55–64), Hispanics experienced higher mortality than their White counterparts in the 1990s, but this gap began to narrow in the 2000s and had disappeared by the end of the study period primarily because of declining ENM mortality among Hispanics. Although working-age Blacks experienced larger changes in mortality from ENM diseases over time, these changes preserved the sizable racial inequalities in mortality from these diseases as the rates for Blacks remained roughly twice those for Whites and Hispanics throughout the period (Figure 9-1).
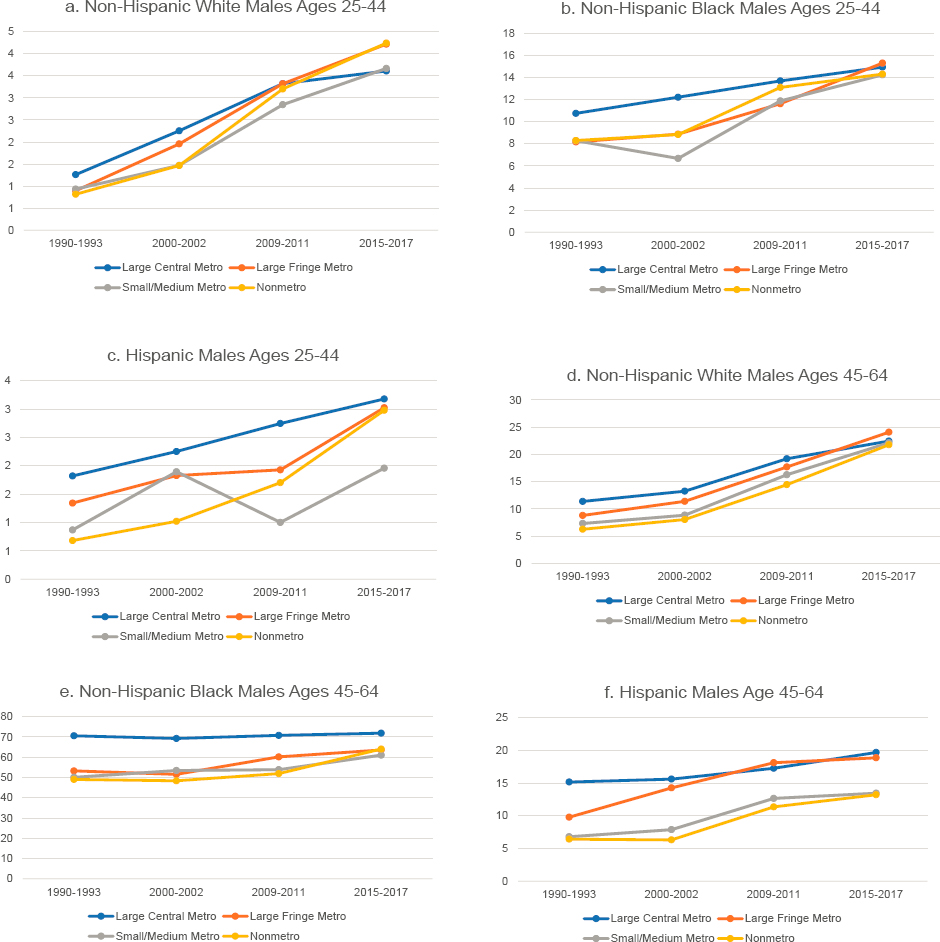
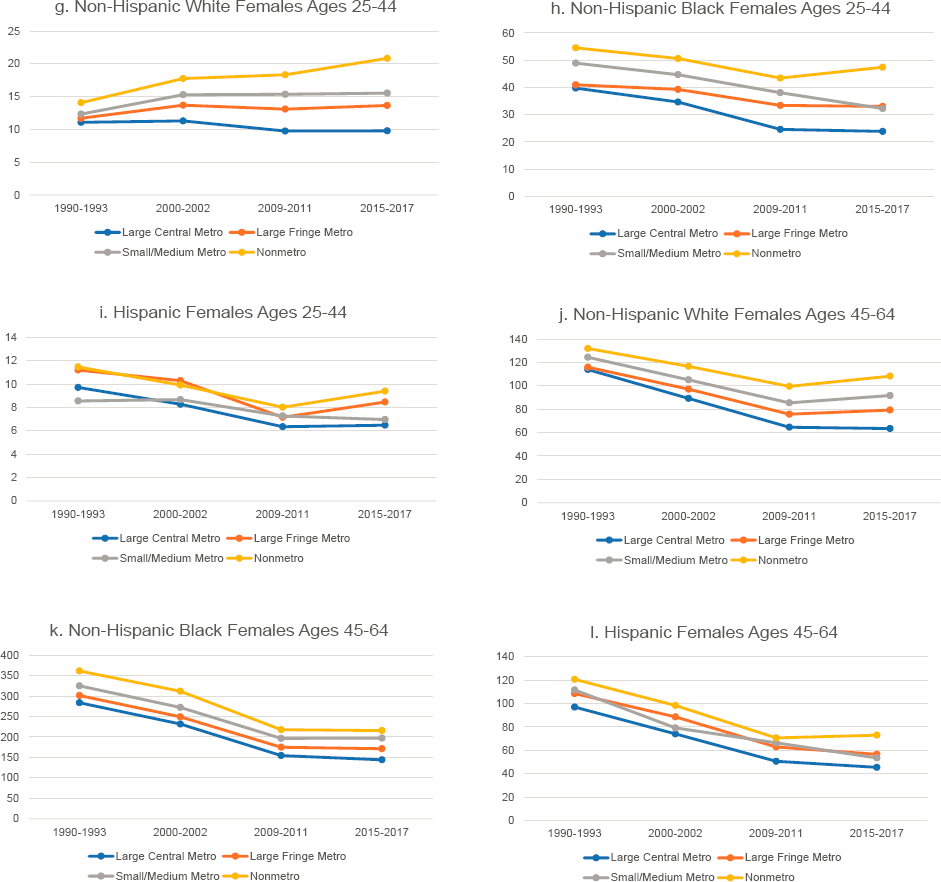
Working-age adults living in large central metropolitan areas (hereafter referred to as “large central metros”) generally experienced lower mortality from ENM diseases relative to those in less-populated areas throughout the study period (Figure 9-2). Mortality in nonmetropolitan areas (hereafter referred to as “nonmetros”) was not always the highest, but it increased more rapidly (or decreased more slowly) than in other areas throughout the 1990s. By 2000–2002, mortality from ENM diseases was highest in nonmetros, and this continued to be the case throughout the remainder of the study period among all working-age adults except Black males ages 25–44. The largest increases among these younger Black males occurred in small and medium metropolitan areas (hereafter referred to as “small/medium metros”). The net result was that disparities across metropolitan areas widened over time, particularly among Whites and Blacks.
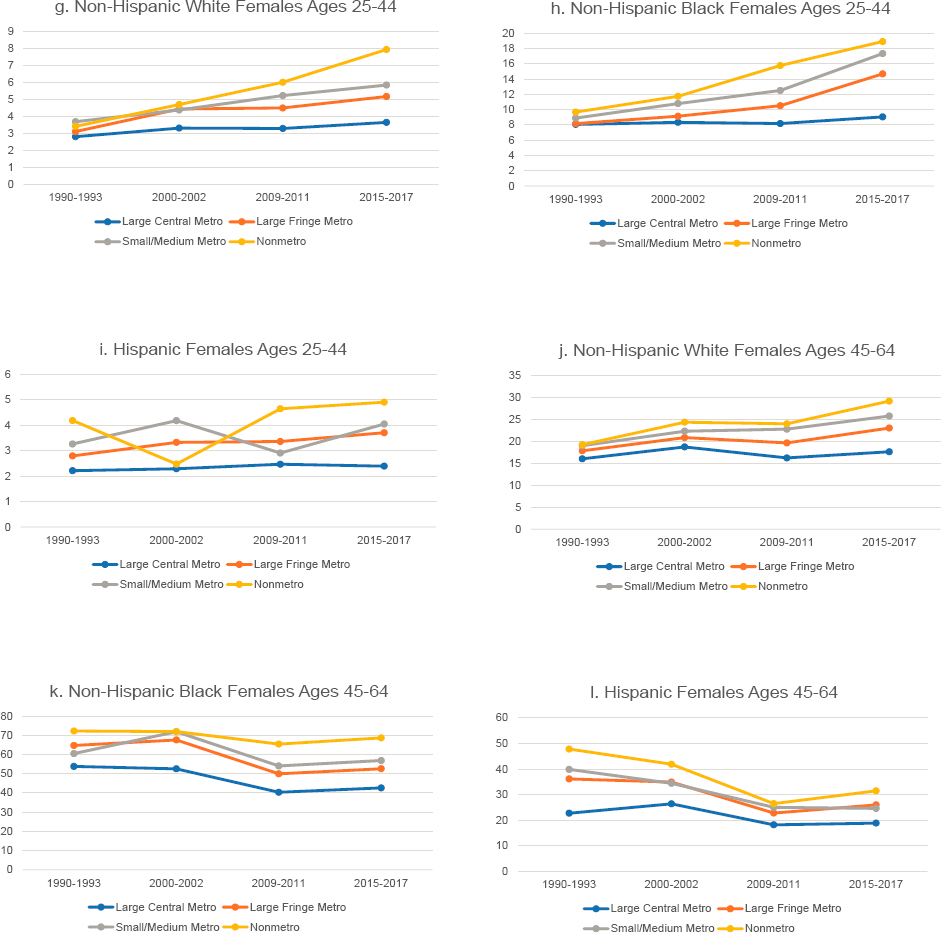
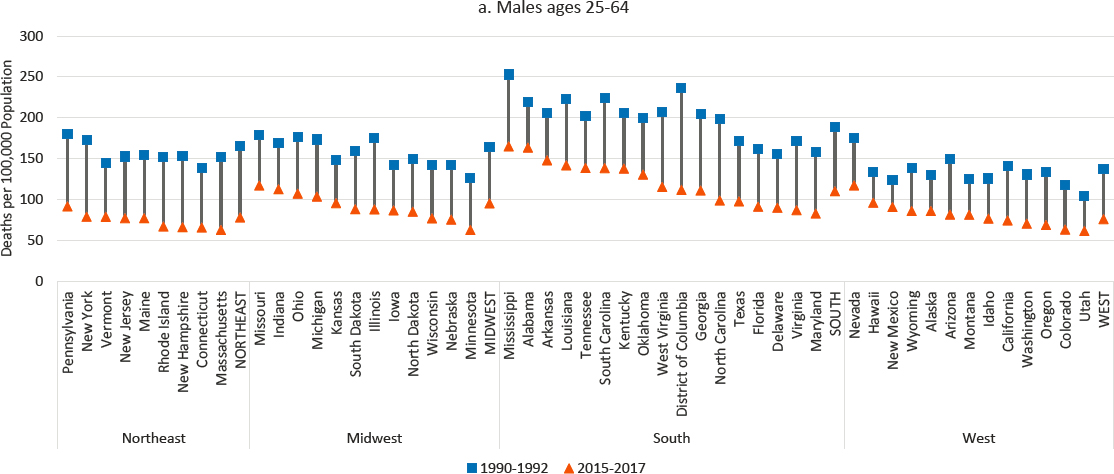
At the state level, mortality from ENM diseases increased among males in all but two states (New Jersey and Vermont) and the District of Columbia, with particularly large increases in several Southern and Western states and in South Dakota (Figure 9-3). Among females, the rates increased in 38 states. There was very little change in the rates across the Northeastern states, but small increases occurred across most of the Midwest and West, and comparatively large increases occurred in several Southern states, most notably West Virginia, Mississippi, Arkansas, Kentucky, and Oklahoma. Among both males and females, there were relatively large declines in the District of Columbia.
Hypertensive Heart Disease
White adults across all working ages experienced increased mortality from hypertensive heart disease during the study period, while the trends among Hispanic and Black adults differed by age and sex (Figure 9-4). Although rates of mortality due to hypertensive heart disease were lower
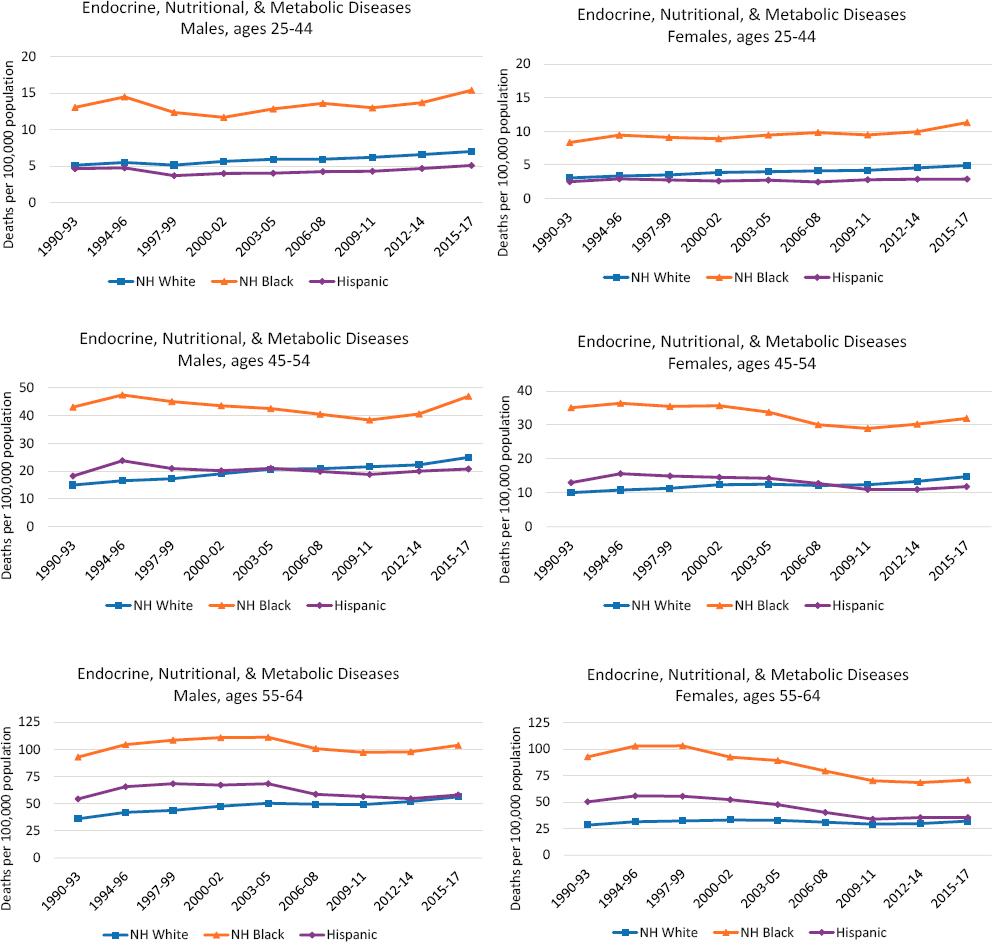
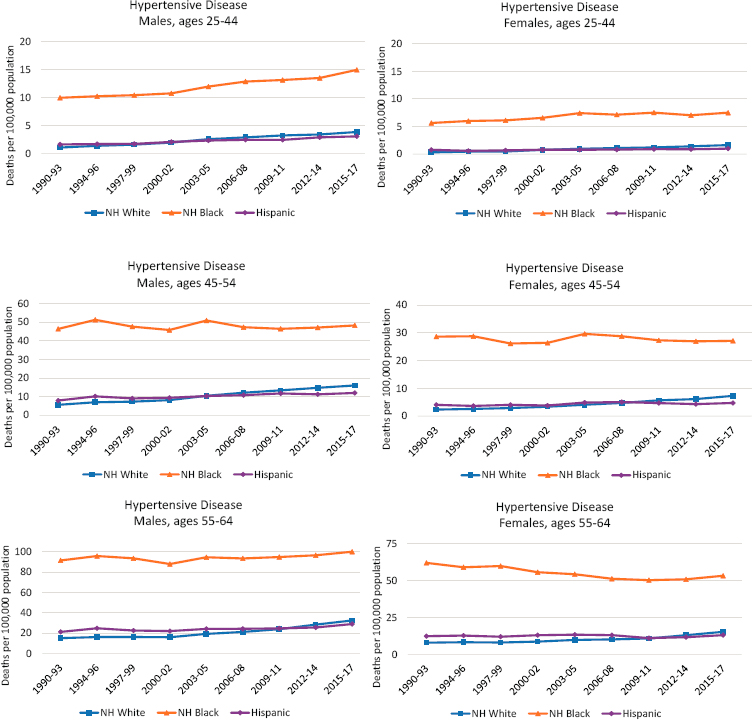
NOTE: Each panel shows mortality rates for non-Hispanic (NH) Whites (blue line), NH Blacks (orange line), and Hispanics (purple line). Mortality rates for males are shown in the lefthand panels, while those for females are shown in the righthand panels. Mortality rates are shown for three age groups: 25–44 (top panels), 45–54 (middle panels), and 55–64 years (bottom panels). Rates are age-adjusted to reflect a standard population age distribution.
SOURCE: Data from National Vital Statistics System Detailed Mortality Files, https://www.cdc.gov/nchs/nvss/deaths.htm.
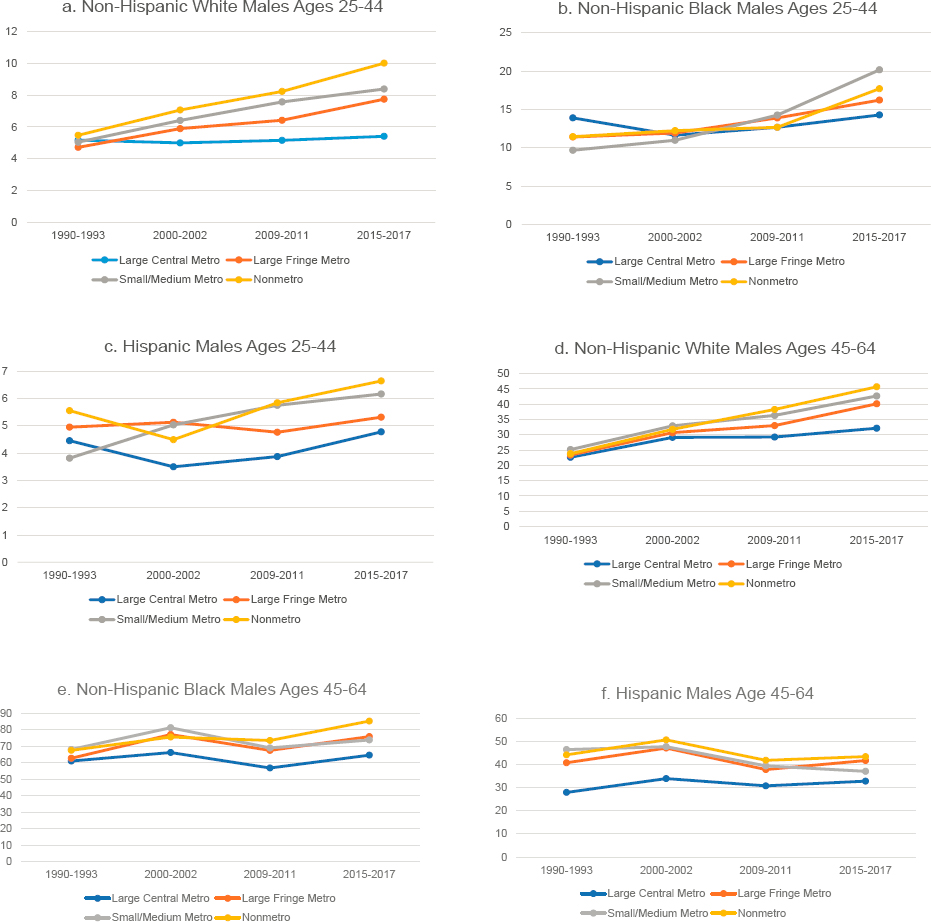
NOTE: Mortality rates are shown for those ages 25–44 (panels a-c and g-i) and 45–64 (panels d-f and j-l) across four levels of metropolitan status: (1) large central metropolitan areas (blue lines), (2) large fringe metropolitan areas (orange lines), (3) small or medium metropolitan areas (gray lines), and (4) nonmetropolitan areas (yellow lines). Trends in these four groups are presented separately by sex (males in panels a-f, females in panels g-l) and for non-Hispanic (NH) Whites (panels a, d, g, and j), NH Blacks (panels b, e, h, and k), and Hispanics (panels c, f, i, and l). Rates are age-adjusted by 10-year age group.
SOURCE: Data from https://www.cdc.gov/nchs/nvss/nvss-restricted-data.htm.
among younger (ages 25–44) than older (ages 45–64) adults, they increased slowly in this younger age group throughout the period among White and Black males and females and Hispanic males, with the largest increases occurring in the early 2000s and the most recent period, from 2012–2014 to 2015–2017; the increases were greater among males than females and among Blacks in this age group. Mortality rates among Hispanic females in all age groups remained stable over the study period. Older Black females ages 55–64 were the only group of working-age adults to experience declining mortality from hypertensive heart disease over the period, although these declines slowed in the 2010s and trended slightly upward after 2012. Among Black males ages 45–64 and Black females ages 45–54, mortality due to hypertensive heart disease showed small fluctuations during the 1990s and 2000s before stagnating (Black females) or increasing slightly (Black males) in the 2010s.
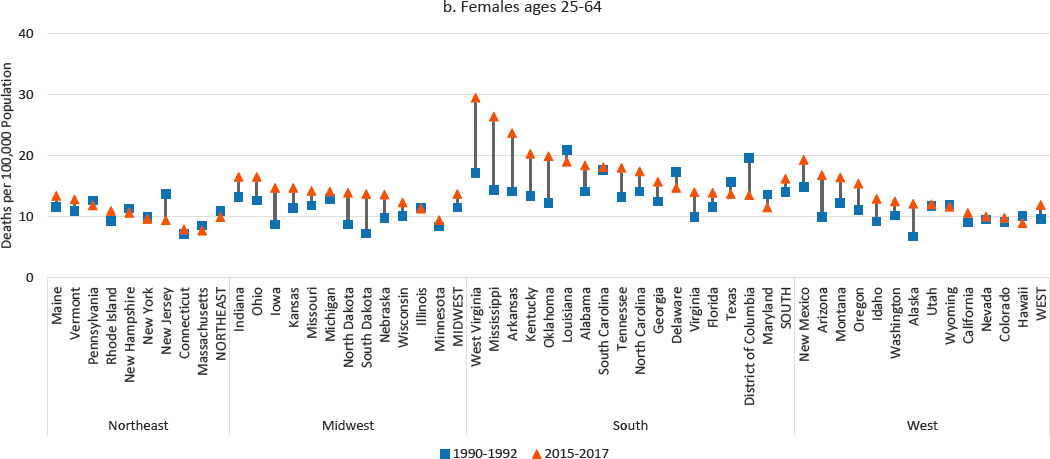
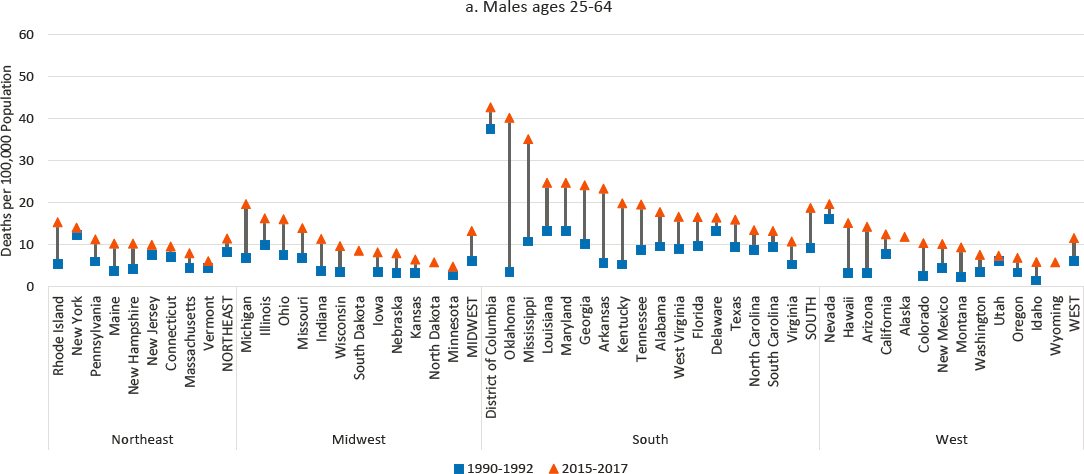
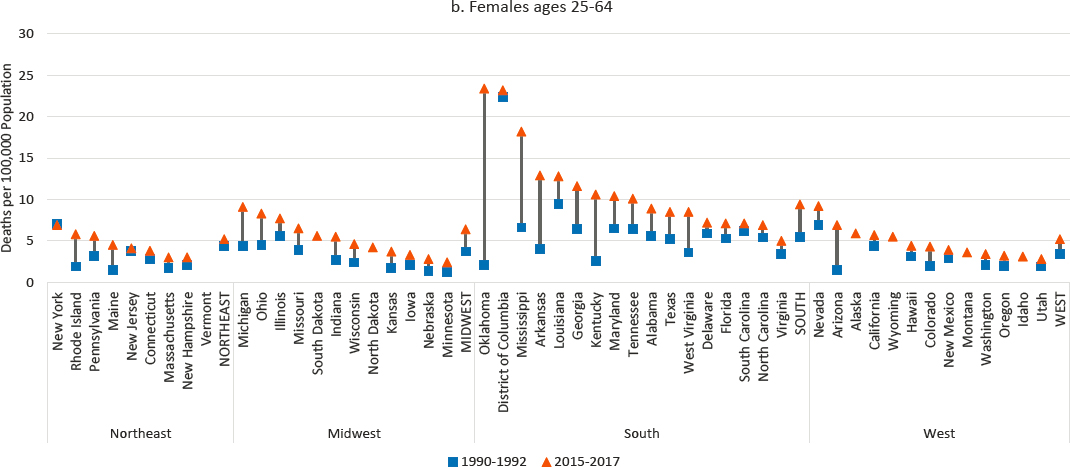
NOTE: Mortality rates are shown for 1990–1992 (blue squares) and 2015–2017 (orange triangles), along with the changes over time (black connecting lines). Mortality rates for males are shown in panel a, while those for females are shown in panel b. Rates are age-adjusted by 10-year age band. For males, the 1990–1992 rate for Alaska represents 1991 and 1992 only; the rate was suppressed for 1990. The District of Columbia is excluded for females for the same reason. States are ordered from highest to lowest mortality rate in 2015–2017 within region.
SOURCE: Data from CDC WONDER Online Database, https://wonder.cdc.gov.
NOTE: Each panel shows mortality rates for non-Hispanic (NH) Whites (blue line), NH Blacks (orange line), and Hispanics (purple line). Mortality rates for males are shown in the lefthand panels, while those for females are shown in the righthand panels. Mortality rates are shown for three age groups: 25–44 (top panels), 45–54 (middle panels), and 55–64 years (bottom panels). Rates are age-adjusted to reflect a standard population age distribution.
SOURCE: Data from National Vital Statistics System Detailed Mortality Files, https://www.cdc.gov/nchs/nvss/deaths.htm.
Throughout the study period, working-age mortality from hypertensive heart disease was more than three times higher among Blacks than among Whites and Hispanics. Because mortality increased more quickly among younger (ages 25–44) Black adults than among their White and Hispanic peers, the mortality gap widened in this age group. However, the Black–White mortality gap narrowed slightly among older working-age adults (ages 45–64), as mortality increased among Whites but declined or stagnated among Blacks. While working-age White and Hispanic adults experienced similar mortality rates throughout the period, the mortality rates in 1990–1993 were slightly higher among Hispanic adults in every age group, but by 2015–2017, the rates were slightly higher among White adults.
While the substantive differences are not large, interesting patterns emerge when these trends are examined separately by metropolitan status (Figure 9-5). Mortality from hypertensive heart disease in large central metros was highest in 1990–1993 but increased less than in other areas, while mortality in nonmetros increased more rapidly. In 1990–1993, large central metros had the highest mortality from hypertensive heart disease among White adults and nonmetros the lowest, the exception being White females ages 25–44, among whom mortality did not differ by metropolitan status. During the 2010s, however, mortality from hypertensive heart disease increased sharply in nonmetro areas, while large central metros experienced much smaller increases. By 2015–2017, mortality from hypertensive heart disease among working-age Whites was highest in nonmetros and lowest in large central metros. The exception was mortality among older White males (ages 45–64), which ended the period with little difference across metro status because mortality in nonmetros did not increase as sharply in this population.
The same trends occurred among working-age Black males (higher mortality in large central metros in 1990–1993 and smaller increases, or larger decreases, over time than in other areas), reducing the gap in mortality by metro size. Among older Black females, the overall declines in mortality from hypertensive heart disease that occurred in the 1990s and 2000s were driven primarily by decreasing mortality in large central metros; mortality stagnated or increased elsewhere. Despite the favorable trends in large central metros; mortality remained highest in these areas for most working-age Blacks. The exception was younger working-age Black females, among whom mortality from hypertensive heart disease increased within small/medium metros and large fringe metropolitan areas (hereafter referred to as “large fringe metros”), but decreased within both nonmetros and large central metros in the 2010s.
Mortality from hypertensive heart disease among older working-age Hispanics (ages 45–64) deviated from the overall trend among older males and females. Among older Hispanic males, mortality increased most rapidly
NOTE: Mortality rates are shown for those ages 25–44 (panels a-c and g-i) and 45–64 (d-f and j-l) across four levels of metropolitan status: (1) large central metropolitan areas (blue lines), (2) large fringe metropolitan areas (orange lines), (3) small or medium metropolitan areas (gray lines), and (4) nonmetropolitan areas (yellow lines). Trends in these four groups are presented separately by sex (males in panels a-f, females in panels g-l) and for non-Hispanic (NH) Whites (panels a, d, g, and j), NH Blacks (panels b, e, h, k), and Hispanics (panels c, f, i, and l). Rates are age-adjusted by 10-year age group.
SOURCE: Data from https://www.cdc.gov/nchs/nvss/nvss-restricted-data.htm.
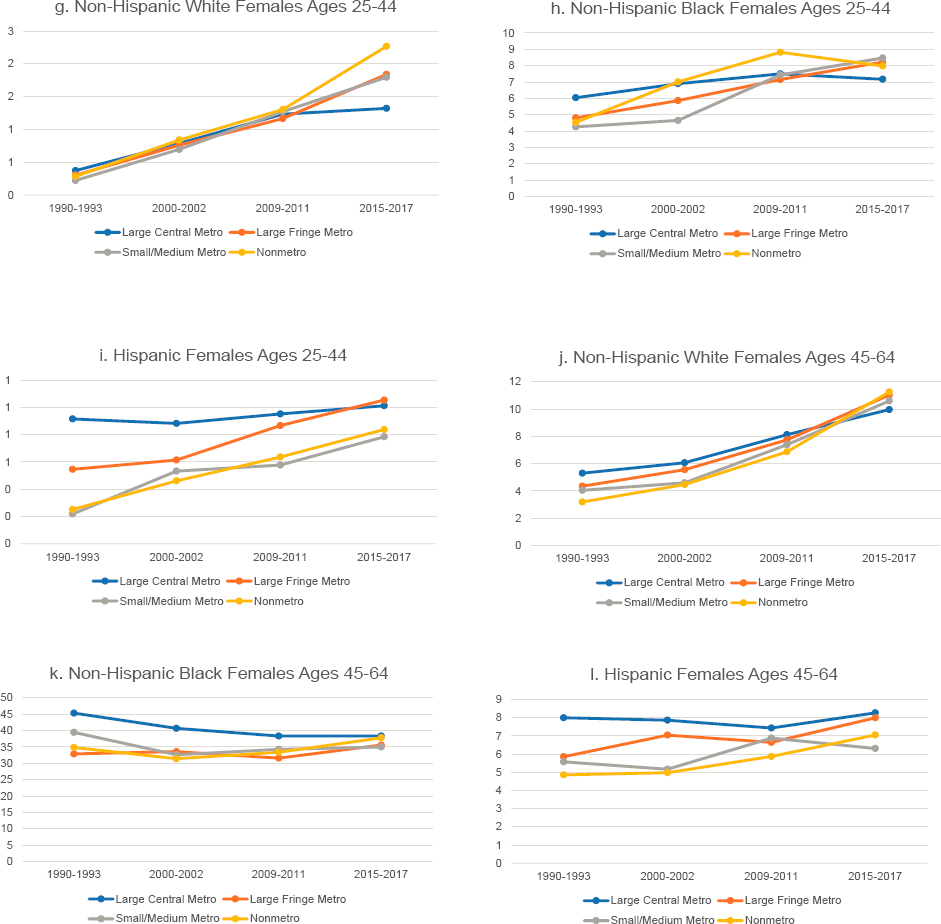
in large fringe metros; by 2015–2017, the mortality rates were similar in large central metros and large fringe metros and lower in nonmetros and small/medium metros. Among older Hispanic women, mortality from hypertensive heart disease remained steady throughout the study period, a pattern driven by mortality trends in large central metros. Outside of large central metros, mortality in this population increased between 1990 and 2017. Even with these increases, however, mortality from hypertensive heart disease remained relatively low among Hispanic men and women.
At the state level, mortality from hypertensive heart disease increased among males in every state (Figure 9-6), a pattern of state-level consistency found for only two other causes of death (drug poisoning and liver cancer). Mortality among females increased in every state but New York (where the rate of mortality from hypertensive heart disease, the highest in the
NOTE: Mortality rates are shown for 1990–1992 (blue squares) and 2015–2017 (orange triangles), along with changes over time (black connecting lines). Mortality rates for males are shown in panel a, while those for females are shown in panel b. Rates are age-adjusted by 10-year age band. For males, the 1990–1992 rates were suppressed for North Dakota, South Dakota, Alaska, and Wyoming. For females, the 1990–1992 rates were suppressed for North Dakota, South Dakota, Alaska, and Wyoming, Montana, and Idaho. Rates for females in both 1990–1992 and 2015–2017 were suppressed for Vermont. States are ordered from highest to lowest mortality rate in 2015–2017 within region.
SOURCE: Data from CDC WONDER Online Database, https://www.cdc.gov/nchs/nvss/deaths.htm.
Northeast region, remained virtually unchanged). The largest increases and highest mortality rates occurred in several Southern states (notably Oklahoma, Mississippi, Arkansas, and Kentucky). In contrast, mortality from hypertensive heart disease changed comparatively little in the Northeast. Although increases among both males and females were small in the District of Columbia, it maintained the highest rate of mortality from hypertensive disease for males and the second highest for females (after Oklahoma) in 2015–2017.
Ischemic Heart Disease and Other Circulatory System Diseases
By far the largest contributor to the recent plateau in all-cause mortality among working-age adults was the slowing pace of improvements in mortality from ischemic heart disease and other circulatory diseases. The remarkable declines in mortality from these diseases seen throughout the 1990s and 2000s stalled in the 2010s (Figure 9-7).1 Working-age Blacks had much higher rates of mortality from ischemic heart disease and other circulatory diseases relative to their White and Hispanic counterparts, although they also experienced the largest reductions between 1990 and 2017. The more rapidly declining death rates among Blacks reduced the mortality gap between them and both Hispanics and Whites over time. Although Hispanic adults had lower mortality from these diseases relative to White or Black adults, the trends among Whites and Hispanics were similar over time. The flattening of the mortality trend in ischemic heart disease and other circulatory diseases after 2010 was similar across all working-age adults.
Perhaps because ischemic heart disease and other circulatory diseases are a leading cause of death, differences in mortality from this category of causes by metropolitan status (Figure 9-8) largely mirror the patterns noted earlier for all-cause mortality. That is, mortality from ischemic heart disease and other circulatory diseases was highest in nonmetros and lowest in large central metros throughout the study period. Among working-age
___________________
1 The plateau in mortality from ischemic heart disease and other circulatory system diseases obscures continued progress in lowering the death rate for ischemic heart disease—progress that was offset by increasing death rates from other circulatory diseases. As detailed in Chapter 4, this latter category of miscellaneous causes of death from circulatory diseases spans International Classification of Diseases (ICD)-10 codes I00–I09, I26–I28, I30–I51, I60–I69, I70–I78, I80–I89, and I95–I99 and includes deaths from arrhythmias, cardiomyopathy, heart failure, cardiac arrest, myocarditis, and valvular and pericardial disease. Whereas age-adjusted working-age mortality from these causes increased between 2011 and 2018 (from 29.9 to 32.2 per 100,000 population), mortality from ischemic heart disease decreased (from 38.6 to 35.2 per 100,000 population) (CDC, 2018). The combination of these trends produced what appears to be a plateau and belies a mixed picture of progress and setbacks for this category of cardiometabolic mortality (Shah et al., 2020).
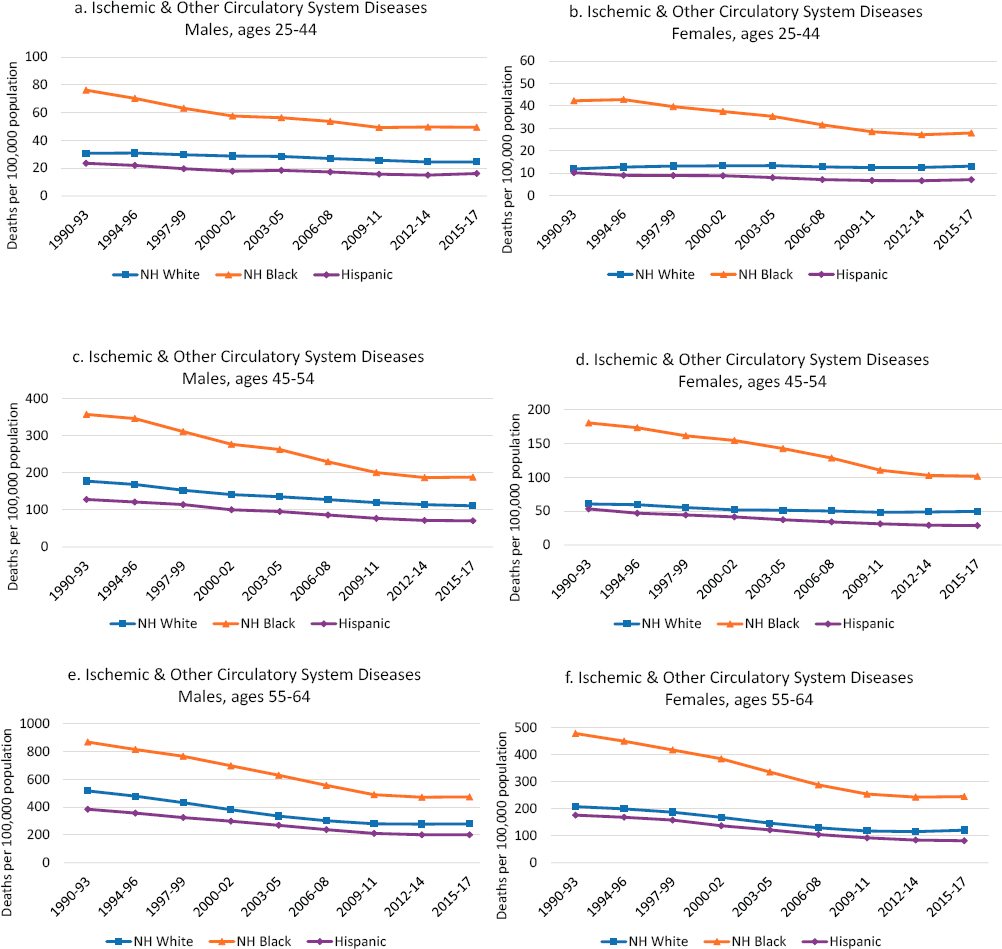
NOTE: Each panel shows mortality rates for non-Hispanic (NH) Whites (blue line), NH Blacks (orange line), and Hispanics (purple line). Mortality rates for males are shown in the lefthand panels, while those for females are shown in the righthand panels. Mortality rates are shown for three age groups: 25–44 (top panels), 45–54 (middle panels), and 55–64 years (bottom panels). Rates are age-adjusted to reflect a standard population age distribution.
SOURCE: Data from National Vital Statistics System Detailed Mortality Files, https://www.cdc.gov/nchs/nvss/deaths.htm.
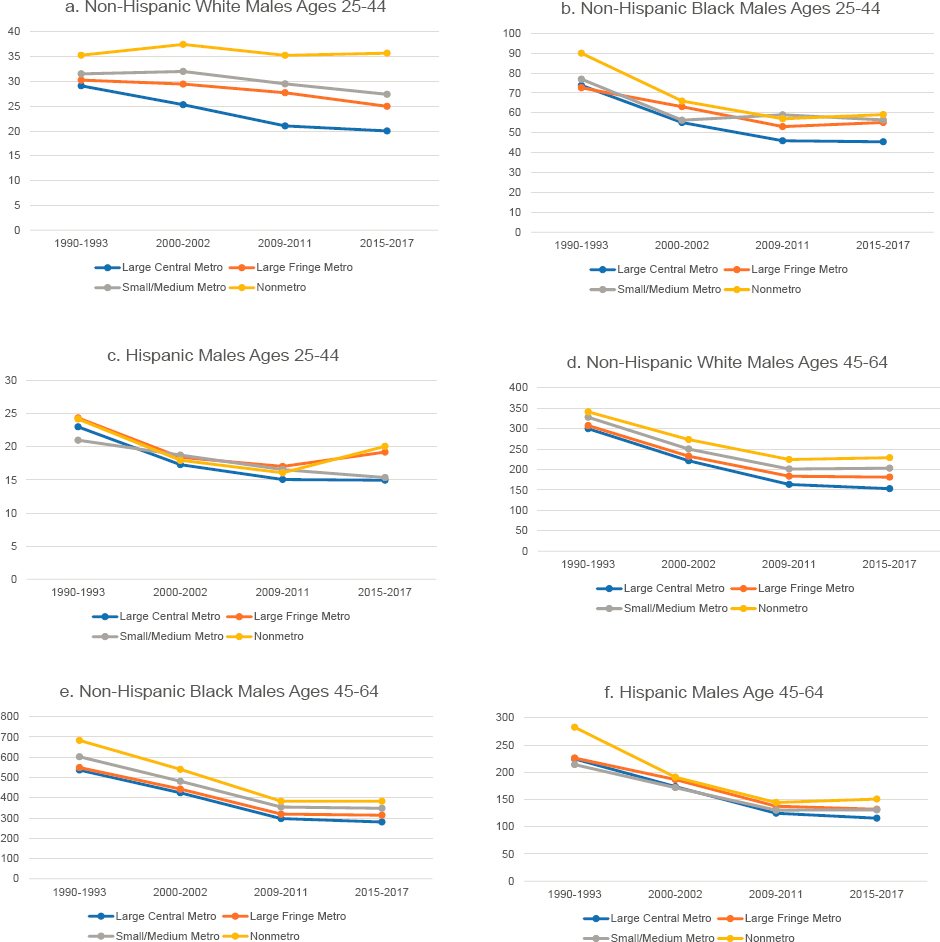
NOTE: Mortality rates are shown for those ages 25–44 (panels a-c and g-i) and 45–64 (panels d-f and j-l) across four levels of metropolitan status: (1) large central metropolitan areas (blue lines), (2) large fringe metropolitan areas (orange lines), (3) small or medium metropolitan areas (gray lines), and (4) nonmetropolitan areas (yellow lines). Trends in these four groups are presented separately by sex (males in panels a-f, females in panels g-l) and for non-Hispanic (NH) Whites (panels a, d, g, and j), NH Blacks (panels b, e, h, and k), and Hispanics (panels c f, i, and l). Rates are age-adjusted by 10-year age group.
SOURCE: Data from https://www.cdc.gov/nchs/nvss/nvss-restricted-data.htm.
White adults, disparities between large central metros and nonmetros widened, just as they did for all-cause mortality. They widened among younger working-age White adults (ages 25–44) because mortality decreased in large central metros but increased elsewhere, particularly in nonmetros. Mortality also declined among older working-age Whites (ages 45–64), but these declines were largest within large central metros and smallest within nonmetros. Among younger working-age Blacks and Hispanics, the mortality gap by metropolitan status was steady throughout the period until 2010, when it widened among Hispanics.
Between 1990 and 2017, working-age mortality from ischemic heart disease and other circulatory diseases declined in every state (Figure 9-9). The District of Columbia experienced the largest absolute and relative declines. In general, the largest declines occurred in Northeastern states and the smallest in several Western and Southern states. As of 2015–2017, the South accounted for 8 of the 10 states with the highest rates of male
NOTE: Mortality rates are shown for 1990–1992 (blue squares) and 2015–2017 (orange triangles), along with the changes over time (black connecting lines). Mortality rates for males are shown in panel a, while those for females are shown in panel b. Rates are age-adjusted by 10-year age band. States are ordered from highest to lowest mortality rate in 2015–2017 within region.
SOURCE: Data from CDC WONDER Online Database, https://wonder.cdc.gov.
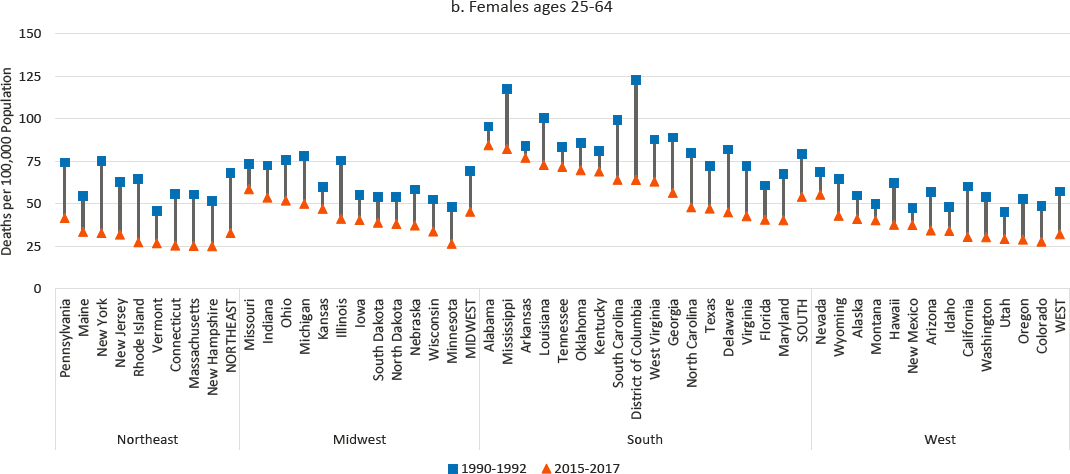
mortality from these diseases (the two other states were Nevada and Missouri); all 10 states with the highest female rates were in the South. This was the case despite the comparatively large declines in mortality that occurred in the South over the study period.
Summary of Trends
Cardiovascular disease (CVD) was the leading cause of death among working-age males and Black females throughout the study period, and the second leading cause of death (after cancer) among White and Hispanic females. Mortality from CVDs declined substantially throughout the 1990s and 2000s among working-age adults, particularly Black males and females. These improvements were due primarily to steady reductions in death rates from ischemic heart disease and other circulatory diseases during the 1990s and 2000s. However, these mortality reductions in ischemic heart disease and other circulatory diseases masked concerning trends in mortality due to hypertensive heart disease and ENM diseases that were rising among young adults, White adults, and Hispanic males during the 1990s and 2000s. These countervailing mortality forces ended when reductions in cardiovascular mortality among all population groups stalled in the 2010s. From 2009–2011 to 2017, mortality from ischemic heart disease and other circulatory system diseases remained largely unchanged among U.S. working-age adults (though see footnote 1 above). At the same time (after 2010), mortality from hypertensive heart disease increased among younger (ages 25–44) working-age adults (except Hispanic females), White working-age adults, and older (ages 55–64) working-age adults, and remained steady among other working-age adults, while mortality due to ENM diseases increased among all working-age adults, particularly Blacks.
These troubling changes in cardiometabolic mortality were particularly pronounced in the South and in areas outside of large central metros, which, along with the Northeast, generally experienced the most favorable trends in cardiometabolic mortality. As a result of these trends, the gap in mortality by metropolitan size grew over time, particularly among White working-age adults.
The contribution of cardiometabolic mortality to the recent rise in all-cause working-age mortality was therefore due to the net increases in mortality from cardiometabolic diseases after 2010, when mortality declines in ischemic heart disease and other circulatory system diseases stalled and no longer offset the rising rates of mortality from ENM diseases and hypertensive heart disease. These trends in cardiometabolic mortality are likely related; that is, the factors causing the rise in mortality from ENM diseases and hypertensive heart disease may also be related to the recent slowdown in reductions in mortality from ischemic heart disease and other
circulatory diseases. Moreover, the magnitude of the long-term decline in mortality from ischemic heart disease and other circulatory diseases has masked the significant implications of the recent stagnation in this trend since 2010. For example, recent research by Mehta, Abrams, and Myrskylä (2020) demonstrates that the slowing pace of decline in cardiovascular mortality was an important factor in the longer-term stagnation in U.S. life expectancy, playing a much larger role since 2010 relative to other causes of death, including drug-related causes. These authors show that drug-related mortality contributed to the shorter-term decline in life expectancy over 2014–2017, whereas stagnation in mortality due to CVD contributed to the longer-term flattening trend in life expectancy. In simulations of life expectancy at age 25, the authors found that the gains in life expectancy would have been greater by a factor of 4 for males and 8 for females if mortality from CVD had continued its rate of decline after 2010, compared with no increase in drug-related deaths since 2010. Their findings indicate that the slowdown in progress in CVD has been more consequential for U.S. life expectancy relative to increasing drug-related mortality. Similarly, Masters, Tilstra, and Simon (2018) found that without “cohort-based increases in metabolic disease mortality risk, the recent increases in middle-age U.S. White women’s overall mortality rates likely would not have occurred” (p. 87). Indeed, the stalling reductions in mortality from CVD among U.S. working-age adults will likely continue to shape U.S. mortality trends in the future (more on this below).
Turning next to explanations for the related trends in mortality from ENM diseases, hypertensive heart disease, and ischemic heart disease and other circulatory diseases between 1990 and 2017, three important themes are relevant. First, the obesity epidemic, fueled by expanding obesogenic environments, has been a major cause of increasing disease, affecting multiple body systems and leading to deaths from hypertension, diabetes, stroke, and CHD (Mensah et al., 2017). Second, while declining smoking rates and medical advances over the past 50 years have greatly lowered mortality due to CVD, declines in smoking have slowed, and the contributions of further medical innovation have had less impact in recent years. Third, cardiometabolic diseases may be rising as a result of other social, economic, and cultural changes that have undermined economic security, intergenerational mobility, and social support networks, which in turn have led to increased chronic stress among the working-age population. The next section presents the committee’s assessment of the evidence related to each of these explanations.
EXPLANATIONS FOR THE TRENDS IN CARDIOMETABOLIC MORTALITY
Explanations for the above trends in cardiometabolic mortality point to the three themes noted above: rising rates of obesity; diminishing returns of medical advances; and social, economic, and cultural change.
Rising Rates of Obesity
Prevalence and Subgroup Differences
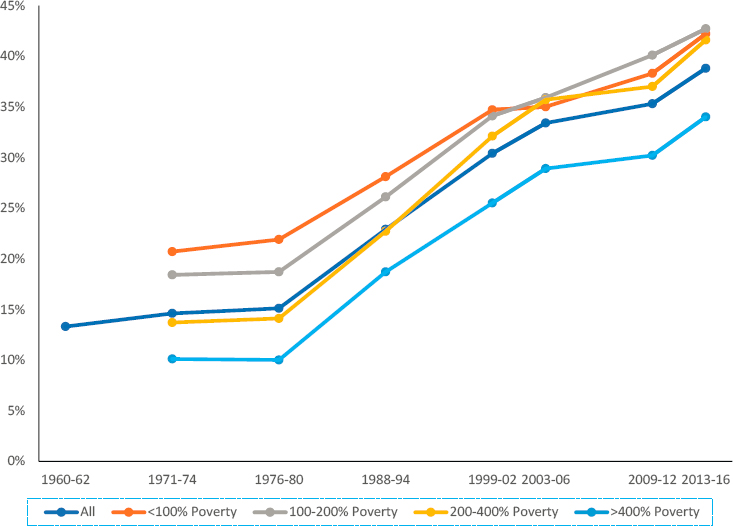
The most important trend potentially explaining the rise in working-age mortality due to ENM diseases and hypertensive heart disease and the stagnation in improvements in mortality due to ischemic heart disease and other diseases of the circulatory system is the increased prevalence of obesity in the U.S. population (Lloyd-Jones et al., 2016; Mensah et al., 2017; Sidney et al., 2016). This report uses the Centers for Disease Control and Prevention’s (CDC’s) definition of obesity as weight that is higher than what is considered a healthy weight for a given height based on body mass index (BMI). BMI is calculated as weight in kilograms divided by height in meters squared, rounded to one decimal place. Obesity in adults is indicated when BMI is greater than or equal to 30 as a measure of abnormal or excessive fat accumulation that presents a risk to health (Hales et al., 2020). Rates of obesity began to rise in the early 1980s, a trend that has continued as a period-based phenomenon affecting children and adults of all ages (Reither, Hauser, and Yang, 2009). The prevalence of obesity among U.S. adults nearly tripled, increasing rapidly from the late 1970s through the early 2000s, with sustained but less rapid increases since then. The age-adjusted prevalence of adult obesity, defined as the percentage of adults ages 20–74 with a BMI of 30 kg/m2 or above, was 42.4 percent in 2017–2018 compared with 15.0 percent in 1976–1980 (Fryar, Carroll, and Ogden, 2018; Hales et al., 2020).
Obesity rates vary by age, sex, race, ethnicity, and socioeconomic status. Older adults tend to have higher rates of obesity relative to younger adults (ages 20–39), although age differences in obesity have diminished as its prevalence has increased over time (Fryar, Carroll, and Ogden, 2018; Hales et al., 2020). Black women have higher obesity rates than Black men, but there are no significant differences in prevalence between men and women among White, Asian, or Hispanic adults (Hales et al., 2020). In 2017–2018, 56.9 percent of Black women were obese, compared with 41.1 percent of Black men. Overall, obesity rates are highest among Blacks (49.6%), followed by Hispanics (44.8%), Whites (42.2%), and Asians (17.4%).
NOTE: Data are from the National Health and Nutrition Examination Survey (NHANES). The sample is ages 20–74 for 1960–1962, 1971–1974, and 1976–1980 and ages 20+ thereafter.
SOURCE: NCHS (2019a).
Rates of obesity have increased in a similar fashion among adults in all income categories, as shown in Figure 9-10, and higher-income populations consistently have lower rates of obesity relative to those with lower income. Working-age adults with a college degree have a lower prevalence of obesity compared with their less-educated peers (Figure 9-11), although this pattern is not consistent by race and ethnicity and sex. For example, Black men show no difference in rates of obesity by educational attainment (Ogden et al., 2017).
Given differentials in obesity by socioeconomic status, some research has examined whether obesity explains differentials by socioeconomic status in the recent rise in working-age mortality. The evidence to date suggests that changes in obesity are not exerting differential impacts on working-age mortality by socioeconomic subgroups of the population. Cutler, Meara, and Richards-Shubik (2011) show that changing rates of obesity were not
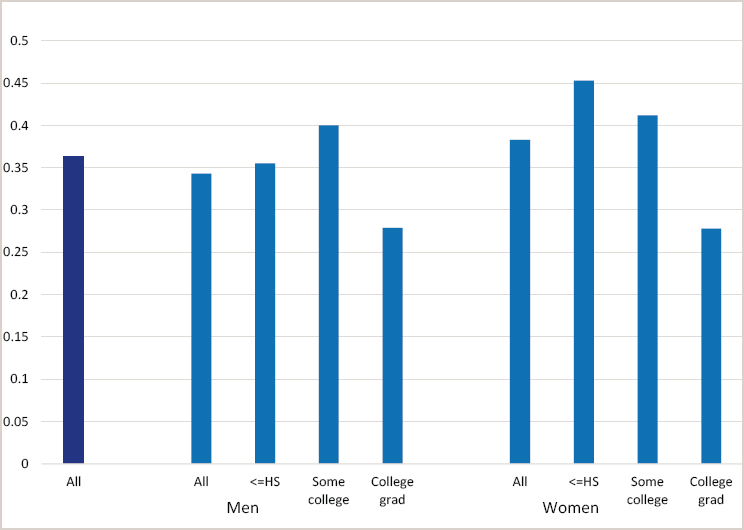
SOURCE: Prevalence of Obesity Among Adults, by Household Income and Education—United States, 2011–2014, Morbidity and Mortality Weekly Report, https://www.cdc.gov/mmwr/volumes/66/wr/mm6650a1.htm.
responsible for widening educational differences in mortality among U.S. adults (ages 25–74) between the 1970s and early 2000s. And Geronimus and colleagues (2019) demonstrate that changes in mortality at working ages (25–64) due to CVD and diabetes between 1990 and 2015 were highly similar for low- and high-educated Black and White men and women. Together, these studies suggest that changes in obesity, while clearly contributing to working-age mortality for key causes of death over the past 25 years, has had little impact on socioeconomic disparities in those trends.
Obesity-Related Mortality
In the face of such dramatic increases in obesity in recent decades, a body of work has examined obesity’s role in shaping mortality patterns and trends. The evidence that obese adults are at higher risk of mortality from cardiometabolic diseases relative to nonobese adults is extensive. Obesity increases the risks of hypertension, stroke, CHD, type 2 diabetes,
and several site-specific cancers and related mortality (Adair et al., 2014; Basu et al., 2014; Di Angelantonio et al., 2016; Eisenmann, 2003; Manson et al., 1990; Prospective Studies Collaboration, 2009; Yu et al., 2017; Zhang et al., 2008). The Prospective Studies Collaboration (2009) carried out a meta-analysis of the association between BMI and mortality among 900,000 individuals in 57 prospective studies designed primarily to evaluate risk factors for CVD. That meta-analysis showed that both overweight (BMI = 25.0–29.9 kg/m2) and obesity (BMI >30 kg/m2) status were associated with increased all-cause mortality; on average, each additional 5 BMI units over normal (e.g., BMI = 20–25 kg/m2) was associated with a 30 percent increase in all-cause mortality. The proportionate increase in mortality risk was greatest for overweight and obese younger adults ages 35–59 and decreased slightly in linear fashion in each subsequent 10-year age group. Increasing BMI greatly increased cardiovascular mortality, including deaths from ischemic heart disease, stroke, heart failure, and hypertensive heart disease, as well as from diabetes and kidney disease. The meta-analysis shows that the effects of BMI on heart disease and stroke are mediated in part by increases in blood pressure and diabetes (Prospective Studies Collaboration, 2009). However, the analysis includes smokers and those with preexisting cancer, who tend to have lower BMIs as a result of illness, likely leading to an underestimate of the association between BMI and all-cause mortality.
A follow-up study by Berrington de Gonzales and colleagues (2010) examined the relationship between BMI and all-cause mortality in a pooled analysis of 19 prospective studies that included 1.46 million White adults. The analysis was limited to healthy participants ages 19–84 who had never smoked, had had their BMI measured, and had been followed for a median of 10 years to ascertain mortality events. The authors found that hazard ratios of death from any cause were higher among White men and women with overweight and obesity relative to those with normal weight and that the risks were greater for those with overweight or obesity at younger ages. Their findings suggest that overweight or obesity in young adulthood versus later in life may have a greater effect on heightening the risks of disease and death (although older survey participants introduce a health bias; see below). Importantly, the hazard ratios associated with overweight and obesity were higher for cardiovascular mortality than for mortality from cancer and other causes.
Subsequent studies improved upon prior study designs to better understand how obesity is related to mortality. For example, studies with multiple measures of BMI across the life course or with retrospective reports of BMI at an earlier life stage have attempted to estimate when in the life course weight gain or high BMI is most related to subsequent morbidity and mortality risks, and whether the life-course patterns of obesity exposure explain
differential mortality risks (Abdullah et al., 2011; Cao, 2015; Engeland et al., 2004; Ferraro, Thorpe, and Wilkinson, 2003; Gray et al., 2011; Owen et al., 2009; Preston, Mehta, and Stokes, 2013; Stokes and Preston, 2016; Tirosh et al., 2011; Zheng et al., 2017). The evidence indicates that obesity experienced in early life, especially young adulthood, is associated with higher subsequent mortality because obesity tracks across the life course and operates through increased risks of cardiometabolic diseases (e.g., diabetes, hypertension, and CVD) (Engeland et al., 2004; Gray et al., 2011; Tirosh et al., 2011; Zheng et al., 2017). Moreover, any exposure to obesity, even among individuals who subsequently lose weight and achieve a normal BMI, is associated with higher subsequent mortality (Stokes and Preston, 2016). It is clear that longer durations of obesity across the life course are associated with greater cardiometabolic disease risks and mortality (Abdullah et al., 2011; Everhart et al., 1992; Harris et al., 2020; Reis et al., 2013).
Finally, several studies show the need to consider the cohort-based prevalence of obesity when estimating the percentage of mortality attributable to obesity (Masters, Powers, and Link, 2013; Masters et al., 2013; Olshansky et al., 2005; Reither, Olshansky, and Yang, 2011; Reither, Hauser, and Yang, 2009). Masters, Powers, and Link (2013) and Masters and colleagues (2013) demonstrate how cohort variation confounds age-specific hazard estimates of the obesity–mortality relationship. When cohort membership is not considered, the age-specific mortality hazards of BMI/obesity appear to weaken with age (Berrington de Gonzales et al., 2010; Kuk and Ardern, 2009; Prospective Studies Collaboration, 2009; Stevens et al., 1998). Masters and colleagues (2013) found that age-related sampling biases affect the obesity–mortality relationship because of attrition: older obese individuals are disproportionately missing in survey samples because of illness and higher death rates. After accounting for survey selection bias and cohort differences in the prevalence of obesity, these authors found that the obesity–mortality relationship remained strong at all ages. Because obesity prevalence has increased with successive cohorts (Lee et al., 2010, 2011; Robinson et al., 2013), mortality risks are expected to increase as younger cohorts age. Indeed, Masters and colleagues (2013) show that obesity has accounted for an increasing share of U.S. deaths in recent decades—about 18 percent of all deaths between the ages of 40 and 85 during the period 1986–2006. These figures are consistent with estimates by Stokes and Preston (2016) that attribute about 16 percent of U.S. deaths among adults ages 50–84 to obesity.
While the obesity epidemic swept through the entire age distribution of the U.S. population in a specific time period, its health consequences have followed a cohort-specific pattern (Masters et al., 2013). Thus as noted earlier, those who were younger when the epidemic occurred (i.e., those born in the 1980s) and are now in their 30s have been exposed to obesogenic
environments for a longer period of their lives relative to previous cohorts of young adults. Indeed, younger adults in more recent birth cohorts have been exposed to obesogenic environments for most of their lives and are therefore most at risk of its cardiometabolic health consequences compared with when older birth cohorts were the same age (Robinson et al., 2013). Data from the National Longitudinal Study of Adolescent to Adult Health (Add Health) study on a cohort born in the late 1970s and early 1980s who have been followed to midlife confirm this sobering fact. When members of the Add Health cohort were in their late 20s in 2008, 37 percent were obese; 10 years later, in 2016–2018, when they were in their late 30s, 47 percent were obese. The cardiometabolic consequences were already emerging for this cohort in 2008 when 26 percent had hypertension, 6 percent had diabetes, and 27 percent had prediabetes (Harris et al., 2019). The cardiometabolic profile of the Add Health cohort represents the new generation of working-age adults in the United States, and is reflected in the trend data showing that among Black and Hispanic younger adults (ages 25–44), increases in mortality from ENM diseases and hypertensive heart disease in 1990–2017 make up a larger share of the rise in all-cause mortality at these ages relative to older Black and Hispanic adults (see Tables 4-2 and 4-3, respectively, in Chapter 4).
Explaining the Increase in Obesity
Identifying the determinants of the rise in obesity can point to modifiable factors for potential interventions to reduce cardiometabolic mortality. This section reviews the scientific evidence on the causes of the U.S. obesity epidemic following the conceptual framework in Chapter 6, which differentiates downstream (i.e., proximate) from upstream (meso-level and macrostructural) drivers. Note that the focus is on how upstream and downstream factors changed to bring about the increase in obesity and by extension, a change in cardiometabolic mortality. For obesity and mortality risks to change, the determinants of obesity and mortality must have changed as well.
Proximate determinants
The most proximate causes of obesity involve genetics and behavior. Cardiometabolic conditions and diseases are at least partly heritable, so genes always play a role (Ehret et al., 2011; Nikpay et al., 2015; Ravussin and Ryan, 2018). Given that inherited genes are fixed throughout one’s lifespan, however, genetic factors can matter in the rise in obesity and related changes in cardiometabolic mortality only if they combine with changes in behavior. Health behaviors such as diet, smoking, alcohol use, and physical exercise affect cardiometabolic health risks. Sedentary lifestyles and workplace environments, including long commutes
and car-based work and prolonged screen time, reduce physical activity and increase the risks of obesity (Church and Martin, 2018; Ekelund et al., 2011). Diets high in fat, sugar, and carbohydrates and low in fiber, associated with regular consumption of processed foods, increase the risks of overweight and obesity (Hall, 2018; Malik et al., 2010; Walker et al., 2020). How have these health behaviors changed since 1990 to affect the risks of obesity and cardiometabolic mortality?
Obesity results from an imbalance between calories consumed and calories expended. The evidence on the relative contribution of each of these factors is not extensive, but the literature offers some insights. Since the 1980s, caloric intake has increased substantially. Cutler, Glaeser, and Shapiro (2003) report that calories consumed per adult increased by about 200 per day between the late-1970s and mid-1990s. In steady state, 200 calories per day amounts to about 20 pounds of weight annually. At the same time, caloric expenditure appears to have changed little. Between 1975 and 1995, time spent in housework fell, but time spent at work rose (Cutler, Glaeser, and Shapiro, 2003, Table 3). TV hours increased, but so did active recreation time. By this analysis, the major issue to explain is why caloric intake has increased so greatly.
However, that is not the only interpretation of the evidence. Over the first half of the 20th century, caloric expenditure fell as people moved off farms and into cities. At the same time that the need for calories fell, caloric intake fell apace. Thus, weights did not change because people made adjustments in what they consumed to match their falling needs. One might ask why the same cannot be said about the more recent period. Indeed, by 2010, the most recent year for which caloric consumption data are available, average daily calories continued to climb by another 100 additional calories over the mid-1990s level (Desilver, 2016). As caloric intake has increased, why have people not increased their caloric expenditure to match their greater intake? Trends in physical activity provide part of the answer. Research suggests that regular recreational exercise has not changed over the past few decades, but occupational activity has (Church and Martin, 2018). By 2006, only 20 percent of American jobs required high levels of physical activity, compared with 1960 when more than 50 percent of jobs required levels of physical activity that met the current daily physical activity goals. The increased low levels of occupational activity mean that many individuals have such low energy expenditure that caloric intake has become uncoupled from expenditure (Church and Martin, 2018).
Environmental determinants
Much attention has been devoted to the role of the obesogenic environment, defined as an environment that promotes weight gain and is not conducive to weight loss within the home or workplace (Swinburn, Egger, and Raza, 1999; Townshend and Lake, 2017).
Such features of the built environment as automobile-dependent design, decreased walkability, and limited access to green spaces and recreational opportunities combine with food advertising and availability to help shape patterns of physical activity and diet that influence the risks of obesity and cardiometabolic disease (Hill et al., 2003). To the extent that these environmental factors have changed over time, they may be influencing mortality trends, although there would be long lag times in their effects given the lengthy causal chains linking these more distal factors to cardiometabolic mortality. Thus, changes to the built environment in the mid-20th century, from the construction of interstate highways in the 1950s and 1960s to the emergence of fast food restaurants, could explain increases in obesity in subsequent decades and an increase in current rates of obesity-related mortality. Moreover, as the obesity epidemic spread, exposure to these period-based changes in the obesogenic environment has been greater for more recent cohorts, increasing their susceptibility to becoming obese.
A large body of research has examined how aspects of the obesogenic environment have contributed to obesity levels by influencing physical activity and dietary behaviors at the individual and community levels (Cobb et al., 2015; Townshend and Lake, 2017). Yet the wide range of study designs, methods, metrics, and environmental variables employed in various studies makes cross-comparison and strong evidence elusive (Church and Martin, 2018; Davis, Plaisance, and Allison, 2018; Hall, 2018; Mackenbach et al., 2014; Sturm and An, 2014). While the basic drivers of obesity are straightforward (more energy consumed than expended), its etiology is multifactorial and complex. The combination of multiple factors (including built environment and social changes) may have created the “perfect storm” for obesity and its consequences for cardiometabolic mortality risk (Ravussin and Ryan, 2018). Rigorous studies have identified the singular role of these factors: (1) inactivity in daily life resulting from automobile dependence (Frank et al., 2007; Lopez-Zetina, Lee, and Friis, 2006), (2) longer commute times and reduced time to prepare foods (Christian, 2012; Zhang et al., 2014), (3) greater reliance on ultraprocessed and high-calorie foods (Juul et al., 2018; Steele et al., 2016), (4) extensive availability and marketing of relatively low-cost processed and high-calorie products (Drewnowski, 2004; Hruby and Hu, 2015), and (5) more sedentary lifestyles linked to work and leisure activities that rely increasingly on passive behaviors (changes in the nature of work and use of electronic devices for entertainment) (Lakdawalla and Philipson, 2009; Sturm and An, 2014). The sum of this research provides substantial evidence for how physical environments have changed to become increasingly obesogenic and how obesogenic environments are associated with obesity rates. Remaining to be understood, however, are the causal pathways by which the changing environment fueled the level and spread of obesity, and, by extension, the
rise in mortality from ENM diseases and hypertensive heart disease or the slowing declines in mortality from ischemic heart disease and other circulatory diseases.
Research on the impact of the food environment on diet focused initially on proximity to healthy and unhealthy food sources (often using types of food stores as a crude proxy for the availability of healthy food). A number of earlier studies suggested that proximity to sources of healthy food (e.g., stores selling fruits and vegetables) was related to healthier diets (Larson, Story, and Nelson, 2009; Morland, Wing, and Roux, 2002) and, conversely, that proximity to fast food outlets was related to greater consumption of calorie-rich diets (Davis and Carpenter, 2009; Lopez, 2007). However, longitudinal evidence documenting the relationship between changes in food environments and healthy versus unhealthy diets is less common and not conclusive (Caspi et al., 2012; Sturm and An, 2014).
It may be that the food environment operates at a much larger spatial scale than has been studied in existing empirical work. For example, food environments may operate primarily through the ubiquitous availability (and promotion) of low-cost, high-calorie foods (and drinks) in multiple contexts encountered by individuals in the course of their daily lives. Consumption of processed and ultraprocessed foods is also ubiquitous in home, work, school, and leisure environments (Poti et al., 2015; Steele et al., 2016), representing 25–60 percent of total daily energy intake (Steele et al., 2016). Ultraprocessed foods undergo multiple physical, biological, and/or chemical processes and generally contain food additives, almost none of which are nutritious or healthy (Monteiro et al., 2019). Not surprisingly, the evidence indicates that consumption of these foods contributes to the risks of obesity (Mendonça et al., 2016), hypertension (Mendonça et al., 2017), diabetes (Srour et al., 2020), and CVDs (Srour et al., 2019).
Macrostructural determinants
Macro-level technological changes have greatly altered the way people produce, obtain, and consume food, especially highly processed, palatable, and cheap food with high amounts of sugar, fat, salt, and flavor additives (Cutler, Glaeser, and Shapiro, 2003; Hall, 2018). Such innovations as vacuum packing, improved preservatives, deep freezing, artificial flavors, and microwaves have enabled food manufacturers to cook food centrally and ship it to consumers for rapid consumption. Agricultural innovations and the mechanization of work once performed by humans have increased the efficiency of food production as well as the quantity of food produced, and thereby provided Americans with the cheapest food in the world (Lakdawalla and Philipson, 2009). Access to processed food has greatly increased through vending machines, takeaways, cafes, convenience stores, and fast food restaurants (Morland and Evenson, 2009). As a result, traditional normative eating behaviors have shifted over time from
time-consuming home cooking to more ubiquitous snacking and consumption of large-portion-size meals, often at restaurants.
Cutler, Glaeser, and Shapiro (2003) examined how technological change affected rates of obesity in the United States. They argue that obesity resulted from technological changes that reduced the price—in terms of both money and time—of obtaining food, especially less-healthy food. Whereas the technology for producing and distributing fresh food (e.g., fruits and vegetables) has not changed much over time, the technology for producing, preserving, and distributing less-healthy food has increased markedly. For example, soda can be mass produced and distributed more readily than in the past, advances in preservatives have made such foods as potato chips and cookies available on virtually every street corner, and fresh pizza can now be made easily and sold in more locations. Not surprisingly, people consume more food as its monetary and time costs decline. Internationally, countries, especially English-speaking countries, that allow more mass production of food have experienced greater increases in obesity (Bleich et al., 2008).
Given the evidence of direct links between technological changes in food production and distribution and the rise in obesity, one role for government is to provide incentives for the production and distribution of healthy foods and disincentives for the mass production and promotion of unhealthy foods. Recent innovations have led to the use of many traditional technologies, such as fermentation, extraction, encapsulation, fat replacement, and enzyme technology, to produce new healthy food ingredients, reduce or remove undesirable food components, add specific nutrient or functional ingredients, modify the composition of foods, mask undesirable flavors, and stabilize ingredients (Hsieh and Ofori, 2007). New techniques currently in development include the use of high-pressure processing to substantially extend the shelf life of healthy food products. This technique preserves the sensorial and nutritional properties of food by not involving heat treatment and maintains the food’s original freshness throughout its shelf life without the addition of chemical preservatives (McFadden, 2018).
Unfortunately, some innovations in food science, such as the introduction of synthetic sweeteners, have also contributed to the obesity epidemic. Beginning in the 1980s, farm subsidies authorized by Congress encouraged an oversupply of corn, thereby lowering its cost and encouraging the production of high-fructose corn syrup, a sweetener that has made a major contribution to obesity. U.S. per capita consumption of high-fructose corn syrup increased from 0.8 g per day in 1970 to 91.6 g in 2000 (Bray, Nielsen, and Popkin, 2004), and added sugar intake from corn sweetener rose by 359 percent from 1970 to 2007 (Sturm and An, 2014).
Finally, the food industry—aided by legislation and budget decisions promoted by lobbyists and politicians from agricultural states—has
successfully leveraged advertising and marketing techniques to boost consumption of calorie-dense foods (Sadeghirad et al., 2016). Likewise, restaurants promoting inexpensive, unhealthy, and “all you can eat” menus have proliferated, especially in the socioeconomically disadvantaged neighborhoods that are at greatest risk for obesity.
The need for solutions is widely recognized in the public and private sectors, driven not only by public health concerns but also by the threat obesity poses for employers, the business community, and the armed services. Further work is needed to build on recent efforts—some led by the food industry itself and others by public health authorities—to discourage the production and purchase of unhealthy foods or at least give consumers better information with which to make healthier food choices. Examples include self-regulation of or restrictions on misleading advertising (Graff, Kunkel, and Mermin, 2012), pricing and tax strategies (Blecher, 2015; Novak and Brownell, 2011; Powell et al., 2013), “Nutrition Facts” product labels mandated by the Food and Drug Administration (FDA) (Food and Drug Administration [FDA], 2020), menu labeling by restaurants (VanEpps et al., 2016), and zoning restrictions limiting the proliferation of fast food restaurants. The fact that U.S. caloric intake per capita outpaces that of other high-income countries (Institute of Medicine and National Research Council [IOM and NRC], 2013) suggests the need to identify structural causes, from lax restrictions on the food industry and advertisers to cultural differences in lifestyle, such as levels of physical activity.
Governments, for example, could seek to address the issues that prevent people from offsetting the consequences of ever-cheaper food. The increase in food availability has occurred in parallel with people’s having a good deal of additional time; this is the case particularly for women, who no longer spend as many hours cooking and cleaning as they once did (Sturm and An, 2014). At the societal level, people could use this additional time to work off the added calories from prepared food and still have time left over (Cutler, Glaeser, and Shapiro, 2003). Many people talk about wanting to make this trade, but society at large has not chosen to do so, perhaps because it is easy to eat today and delay exercise until tomorrow. Helping people engage in the behavior that is necessary to reduce obesity is a key public priority.
The promotion of healthy foods and diets among children and youth is especially critical given that healthy habits and lifestyles are formed early in life, when health trajectories tend to become set into young adulthood (Harris et al., 2006; Kane et al., 2018). School breakfast and lunch offerings, access to sugary drinks, and the contents and locations of vending machines are increasingly being monitored to promote access to healthy snacks and daytime meals for children. Some evidence indicates that increasing the costs of nutritionally less-desirable foods is most likely to
have an effect on body weight among youth, people of low socioeconomic status, and those at risk of obesity (Powell et al., 2013). Taxes on sugary drinks and educational programs on the health consequences of poor food choices might help steer the public toward more healthy diets, and more research is needed to evaluate the effects of such policies and programs on the consumption of healthier food.
Prevention and Treatment of Obesity
Extensive reviews of treatment programs to reduce obesity and prevention programs to avoid obesity and maintain healthy weights have provided limited evidence of the long-term success of these programs (Jeffery et al., 2000; Johnston et al., 2014; Stice, Shaw, and Marti, 2006), although many local interventions and programs have shown short-term success with weight loss and increased physical activity (Community Guide, 2020a, 2020b; Johnston et al., 2014; Wang et al., 2013). Research clearly points to the need to alter the food environment to reduce obesity (Mattes and Foster, 2014); the challenge appears to be how to maintain healthy lifestyle changes for more permanent reductions in weight and body mass (Sobol-Goldberg, Rabinowitz, and Gross, 2013). In terms of prevention, the evidence points overwhelmingly to avoiding obesity at all costs. As noted earlier, a growing body of research shows that any experience of obesity over one’s life course, longer life-course durations of obesity, and earlier life-course exposure to obesity result in heightened morbidity and mortality risks (Abdullah et al., 2011; Harris, Duncan, and Boisjoly, 2002; Owen et al., 2009; Reis et al., 2013; Stokes, Ni, and Preston, 2017; Zheng et al., 2017). Thus, prevention programs need to target children and adolescents most at risk of obesity (e.g., racial/ethnic minorities, females, people living in poverty and with low socioeconomic status) and those who are overweight or experiencing weight gain to intervene before obesity trajectories become set throughout adulthood. Overall, the lack of long-term success for weight loss and exercise programs in maintaining healthy weights speaks to the need for program development; experimental designs; and quantitative and qualitative research to uncover the social, biological, and behavioral impediments to permanent weight loss.
Diminishing Returns of Medical Advances
Starting in the late 1960s, there was a remarkable turnaround in the century-long trend of increasing cardiovascular mortality in the United States. By the 1970s, death rates due to CVD, and in particular CHD and stroke, were in sharp decline, registering a 70 percent decline by 2010 (Mensah et al., 2017). Medical innovations in drug development
and prevention, treatment, and control of chronic diseases, together with increasing knowledge about the health effects of cigarette smoking, fueled this decline. Studies suggest that medical advances and surgical treatments explain about 47 percent of the decline, while reductions in major risk factors (e.g., smoking, poor diet, lack of physical activity) explain approximately 44 percent (Ford et al., 2007; Mensah et al., 2017).
The 1964 Surgeon General’s Report on Smoking and Health publicized the dangers of cigarette smoking, triggering a series of tobacco control efforts that successfully reduced tobacco use (see Chapter 11). The benefits of treating moderate hypertension were demonstrated in the early 1970s, and the benefits of lowering cholesterol emerged in the early 1980s, leading to the establishment of the first standards and targets for blood pressure and cholesterol management in 1987 (Mensah et al., 2017). By the late 1980s, powerful new drugs for managing blood pressure (e.g., angiotensin-converting enzyme inhibitors and beta-adrenergic blocking agents) and cholesterol (e.g., statins) also contributed to lowering CVD mortality. Furthermore, the late 1980s introduced thrombolysis2 and angioplasty as standard treatments for acute myocardial infarction, stroke, and pulmonary embolisms. Coronary stents were approved by the FDA in the 1990s and widely used thereafter.
As discussed previously, the 21st century brought a slowdown in the impact of medical advances in treating CVD (Mensah et al., 2017). The new century saw some additional clinical trials of statin drugs, a focus on treating isolated systolic hypertension3 in the elderly, and a lowering of blood pressure targets to increase blood pressure control, but innovations after 2000 had a smaller impact relative to those in prior decades. Stagnation in improvements in CVD mortality became evident after 2010, garnering public attention (e.g., Bever, 2019; McKay and Winslow, 2016; Painter, 2019) and stimulating research to find an explanation (Ma et al., 2015; Mensah et al., 2017; Shah et al., 2019; Sidney et al., 2016). Several explanations for the diminishing returns of medical advances in lowering CVD mortality after 2010, detailed in the next section, have been proposed: more incremental development of medical innovations, rising obesity that blunted the impact of medical advances, and differential delivery of or access to the medical advances that enabled the reductions in CVD mortality.
International data do not support the explanation that a lack of medical innovations after 2010 is responsible for the stalling improvements in CVD. The flattening of the CVD mortality trend in the United States
___________________
2 Thrombolytic agents dissolve dangerous blood clots in blood vessels to improve blood flow and prevent organ damage.
3 Isolated systolic hypertension is the most common form of high blood pressure in adults over age 65. It occurs when diastolic blood pressure is less than 80 mm Hg and systolic blood pressure is 130 mm Hg or higher.
after 2010 has occurred in other industrialized countries as well (Case and Deaton, 2017; Lopez and Adair, 2019; Ma et al., 2015; Preston, Vierboom, and Stokes, 2018; Shah et al., 2019; Sidney et al., 2016). International comparisons are shown in Figure 9-12 (adapted from Mehta, Abrams, and Myrskylä, 2020). Although all of the 10 countries with high life expectancy in this figure experienced flattening trends in CVD mortality after 2010, the United States has not kept pace with the reductions in CVD mortality seen in these other countries, suggesting that other factors specific to the United States have been stalling continued improvements. Prime among those factors is obesity and its cardiometabolic consequences, which, as noted above, typically lag obesity exposure (Mensah et al., 2017; Olshansky et
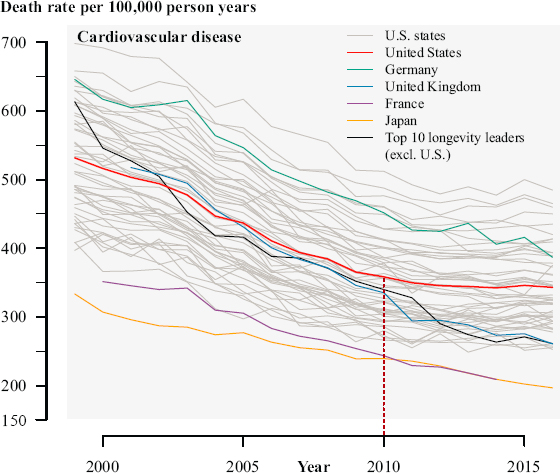
NOTES: Age-standardized death rates (per 100,000 person-years) for CVD from 1999 to 2017. Gray shaded lines are trends for individual U.S. states and DC. “Longevity leaders” denotes the average of countries that had the highest life expectancy at birth in 2010 (Japan, Switzerland, Singapore, Australia, Spain, Iceland, Italy, Israel, Sweden, and France). Sexes are combined. CVD deaths include deaths from heart attacks and strokes (International Classification of Diseases [ICD]-10 codes I00 to I78).
SOURCE: Adapted from Mehta, Abrams, and Myrskylä (2020). This figure is an excerpt of the original, licensed under Creative Commons Attribution-NonCommercial-NoDerivatives License 4.0 (CC BY-NC-ND).
al., 2005). Preston, Vierboom, and Stokes (2018) estimate that rising levels of BMI during 1988–2011 reduced life expectancy at age 40 by .9 years in 2011 and have reduced the impact of many beneficial factors working to improve mortality, such as declining smoking rates and medical advances that include pharmacological treatments for several obesity-related chronic conditions, such as hypertension, hyperglycemia, and dyslipidemia. Indeed, the cardiometabolic consequences of the obesity epidemic were beginning to emerge in data in 2009 showing an increase in the prevalence of high blood pressure and cholesterol, a decrease in physical activity, and an increase in BMI and high blood glucose (Mannsverk et al., 2016; McEwen et al., 2011; Pilkerton et al., 2015). Thus, the slowdown in improvements in mortality, particularly CVD mortality, in the past decade may be due to the lagged cardiometabolic consequences of the obesity epidemic, now under way for more than 30 years.
Another factor unique to the U.S. context that may explain the diminishing returns of medical advances in reducing CVD mortality is the persistent racial, ethnic, and socioeconomic disparities in chronic health conditions and control of chronic conditions related to cardiometabolic mortality that slow continued gains once those most at risk or those with greater health care access are treated. When new pharmaceuticals and other therapeutic treatments to control hypertension and heart disease were developed, delivery of these treatments was naturally targeted to those with the highest levels of disease (e.g., the highest levels of cholesterol or blood pressure), sharply reducing CVD mortality in the 1970s and 1980s (Weisfeldt and Zieman, 2007). Treating those with more moderate or mild cardiometabolic conditions, by contrast, results in marginal gains in improvement of CVD mortality, so that progress slowed after 2000.
In addition, many individuals may not be receiving or correctly using medications that are effective in controlling hypertension, diabetes, and heart disease, and many may be struggling to discontinue tobacco use. Research has shown that poor adherence to treatment results from a variety of barriers, including those experienced by patients (Ho, Bryson, and Rumsfeld, 2009; Rashidi et al., 2020) and those introduced by the health care system (Banerjee et al., 2016). Baroletti and Dell’Orfano (2010) divide the barriers faced by patients into three categories: socioeconomic (e.g., lack of adequate health care coverage, unemployment, poverty, concerns about cost), communication related (e.g., inadequate instructions, language barriers, mental illness), and motivational (e,g., not appreciating the gravity of these conditions or benefits of treatment, fears and concerns, and cultural beliefs) (Baroletti and Dell’Orfano, 2010). Understanding the barriers faced by patients and designing solutions to overcome those barriers is thus a priority for the control of cardiometabolic mortality and chronic diseases more generally.
When cardiovascular treatments were first introduced and when the dangers of smoking were first recognized decades ago, individuals with greater access to care, higher education, and other socioeconomic advantages were more likely to receive care and to quit smoking (Hiscock et al., 2012; Kanjilal et al., 2006; Ma et al., 2005; Sterling et al., 2019; U.S. Department of Health and Human Services [HHS], 2011). Much of the progress in lowering CVD mortality in prior decades may reflect the widespread uptake of these interventions among these more advantaged population groups, but adults who faced greater barriers to care may have been left behind. People of color and those of lower socioeconomic status face greater barriers to adherence to treatment targets and have higher rates of tobacco use (Cokkinides et al., 2008; Kiefe et al., 2001). The field of implementation science (also known as translational and health services research) studies interventions to close this gap between recommended care and the care actually received by individuals, especially members of vulnerable groups (Bauer et al., 2015; University of Washington, n.d.). The magnitude of this gap first became apparent in 2003, when a classic study documented that U.S. adults received only 54.9 percent of recommended care (McGlynn et al., 2003).
That gap has persisted, especially among marginalized populations that have limited access to care and live in settings that make adherence difficult. For example, fewer than half of U.S. adults (44%) with hypertension have achieved optimal blood pressure control (Dorans et al., 2018), and slightly more than half (55%) who could benefit from lipid-lowering agents are currently taking that medicine (Mercado et al., 2015). Disparities in the prevalence of chronic diseases mirror disparities in control of those diseases. In 2015–2016, for example, Blacks had higher rates of hypertension (57.3%) but lower rates of blood pressure control (37.2%) relative to Whites (43.8% and 45.7%, respectively) (Dorans et al., 2018). Moreover, individuals with hypertension, hyperlipidemia (high cholesterol levels), and insulin resistance may be asymptomatic and escape detection. This is the case especially among younger adults, who get fewer regular medical checkups and have less access to screenings for disease at work or in other community settings (Zhang and Moran, 2017). National estimates show that three-quarters of young adults ages 24–32 with measured hypertension were unaware of their condition (e.g., had not received a medical diagnosis), and two-thirds with measured diabetes were similarly unaware (Nguyen et al., 2011, 2014).
It is well documented that both research to identify strategies for reducing these disparities in care and the evidence-based programs and policies that emerge from translational research have been less heavily funded relative to investments in new drugs and technologies (Dzau and Balatbat, 2019; Woolf, 2008). This imbalance is counterproductive with respect to
reducing mortality. For example, improving adherence to older-generation treatments can save more lives than developing newer agents with incrementally greater efficacy (Woolf and Johnson, 2005). Thus, after years of declining CVD mortality due to the broad uptake of pharmacotherapy and smoking cessation among advantaged populations, the recent decline in the pace of progress may reflect the failure to invest adequately in closing the prevention and treatment gap among high-risk populations. If so, a resurgence in progress in lowering CVD mortality in the United States could be stimulated by more robust efforts to help these populations overcome socioeconomic and environmental barriers to receiving effective cardiovascular treatments and successfully discontinue tobacco use.
A closer look at tobacco use is illuminating. The reduction in U.S. smoking first occurred among men in the early 1950s and later among women beginning in the late 1970s, producing a substantial decrease in mortality for U.S. adults (Cutler, 2008; Wang and Preston, 2009). Following the U.S. Surgeon General’s Report on Smoking and Health (1964), cigarette smoking rates declined 67 percent, from 42.6 percent in 1965 to 13.7 percent in 2018 (American Lung Association, n.d.; Creamer et al., 2019). However, an array of new tobacco products, including e-cigarettes, has entered the market in the past decade, with 19.7 percent of U.S. adults reporting use of any tobacco product in 2018 (Creamer et al., 2019). That prevalence is even higher among males, adults less than 65 years old, non-Hispanic American Indians/Alaska Natives, those with a high school education or less, those with an annual household income of less than $35,000, and those with a disability or serious psychological distress (Creamer et al., 2019).
The educational disparities in the reduction of smoking are especially noteworthy. While smoking declined for all groups, greater progress occurred among adults with more education (Figure 9-13). Studies by Cutler, Meara, and Richards-Shubik (2011) and Montez and Zajacova (2013) show that reductions in smoking behavior have been greater among the highly educated than among the less educated. Over the period 1974–2016, the smoking rate declined by 76 percent among people with a college degree but by just 35 percent among those with only a high school degree. Yet despite this widening educational disparity in smoking, only a modest portion of the widening educational difference in U.S. adult mortality in recent decades is attributable to smoking because widening educational disparities in other trends (e.g., use of drugs) were more important contributors. Further research is needed to address the barriers to smoking cessation faced by populations that continue to smoke at higher rates, but whether interventions to overcome these barriers can refuel the decline in smoking and reductions in CVD mortality remains to be seen.
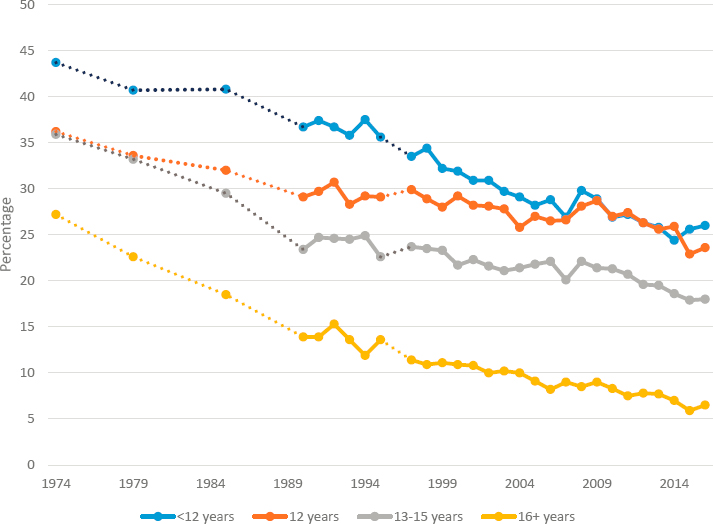
NOTES: The years shown in this figure are those for which data have been published. Data for 1992 and later are not strictly comparable with data for earlier years because of a change in the National Health Interview Survey (NHIS). Because of the redesign of the NHIS in 1997, comparisons with data from prior years must be conducted with caution.
SOURCES: Data for 1974–2007 are from Centers for Disease Control and Prevention, National Center for Health Statistics, Health, United States, 2009, Table 61. Data for subsequent years are as follows: 2008–2009: Health, United States, 2010, Table 59; 2010–2012: Health, United States, 2013, Table 57; 2013–2014: Health, United States, 2015, Table 48; 2015–2016: Health, United States, 2017, Table 48.
Social, Economic, and Cultural Change
Social, economic, and cultural shifts over the past 50 years have profoundly changed the family, work, and community environments of daily life in America, and the possibility that these shifts may have contributed to cardiometabolic morbidity and mortality cannot be dismissed. Changes in family structure and rising family instability over the past 50 years have been well documented (Cavanagh and Fomby, 2019; Cherlin, 2009). Since the 1970s, increases in divorce, cohabitation, and nonmarital childbearing have greatly altered the family contexts in which children are raised and the financial and social capital to which they have access (Bianchi, 2011; Casper and Bianchi, 2001; Cherlin, 2009; McLanahan and Sandefur, 1994;
Sayer, Bianchi, and Robinson, 2004). During this period, families were affected by macroeconomic changes that included the loss of manufacturing jobs, which provided employment opportunities for many low-educated and unskilled males, and the growth of the service sector, which employed largely females and more highly educated individuals. Single mothers were now able to support a family on their own—albeit at much-reduced wages and earnings relative to a two-worker family—and the economic status of men, especially those with less education and fewer job skills, suffered (Oppenheimer, 2000, 2003). The rise in family instability, combined with additional economic distress caused by the Great Recession, permeated entire family systems and disrupted the family-based safety net on which many working-age adults once relied. Communities hardest hit by the decline in manufacturing—especially in the textile, mining, and steel industries—experienced widespread unemployment, economic insecurity, diminished access to health care, and increasing individual- and familial-level stress. Recent cohorts of middle- and working-class young adults now face the realization that they will not live a better life than their parents, as the American Dream of intergenerational mobility has drastically declined (Cherlin, 2014; Chetty et al., 2017).
These shifts coincided with other social and cultural changes in American life. Jobs now required increasing educational credentials, resulting in rapidly increasing college enrollments but also burdening many young people, especially those lacking a 4-year degree, with college debt that caused both their finances and their careers to suffer (Velez and Woo, 2017; Wei and Horn, 2013). The digital era ushered in new lifestyles featuring ubiquitous, multiplatform, and uninterrupted access to online, electronic, and digitally streamed information and introduced new forms of social interaction and network formation (IOM and NRC, 2015). Individuals without the skills to access these forms of communication remained isolated from job opportunities and social interaction. Almost all forms of daily activity at work, at home, and in social groups were being accomplished at a faster pace than in prior generations (Tapscott, 2008; Vogels, 2019). The speed and volume of information in daily life created new stresses for those managing work, home, and family responsibilities and new forms of trauma related to body image, bullying, and other adverse experiences (McEwen and McEwen, 2017).
These daily, gradual, and long-term influences from the social and physical environments have contributed to cardiometabolic morbidity and mortality risks through multiple pathways, including the influence of financial hardships on health care access and affordability and on adverse living and work conditions that affect endocrine and cardiovascular diseases (Lian, 2018; Steptoe and Kivimäki, 2013). For example, job loss means loss of health insurance, and financial strains put out-of-pocket costs for health
care (and other household needs, such as housing, food, and transportation) out of reach. Individuals with chronic conditions and comorbidities cannot afford prescription medications, blood glucose testing, or doctor’s appointments, and economic pressures force many families to resort to consuming inexpensive, calorie-dense foods; to live in unhealthy housing and neighborhoods; and to work in jobs that discourage physical activity. Job loss, family breakdowns, and economically depressed neighborhoods increase social isolation and erode social supports that are vital to health and longevity (House, Landis, and Umberson, 1988; Yang et al., 2016).
These cascading and multiplicative social, economic, and cultural changes have led to increasing levels of chronic stress with known adverse effects on cardiometabolic health. For example, adults who experience social isolation and workplace stress have a 1.5-fold and 1.3-fold increased risk, respectively, of CHD (Steptoe and Kivimäki, 2013). Numerous studies with rigorous repeated cross-sectional or longitudinal designs have found that chronic exposure to low socioeconomic status is associated with increased risk of the metabolic syndrome (Elovainio et al., 2011; Loucks et al., 2007; Manuck et al., 2010; Ramsay et al., 2008). Analyses of other stressors, including loneliness (Whisman, 2010), marital stress (in women but not men) (Whisman, Uebelacker, and Settles, 2010), and workplace stress (Chandola, Brunner, and Marmot, 2006), have found a longitudinal association with the onset of the metabolic syndrome, even after controlling for socioeconomic status. Chronic stress and psychological distress are also associated with the components of the metabolic syndrome, such as higher waist circumference (central obesity); high BMI (general obesity); and dyslipidemia, indicated by higher triglycerides and lower HDL cholesterol (Chandola et al., 2008; Kivimäki et al., 2009). Several meta-analyses likewise have found an association between stress and obesity (Nyberg et al., 2012; Wardle et al., 2011).
The increasing daily stresses that characterize contemporary society are felt more intensely among certain population groups. Racial/ethnic discrimination, lack of educational opportunities and economic advancement, and incarceration are the norm rather than the exception for some of the nation’s most vulnerable groups. Additional stresses associated with social and economic inequality and exposure to structural racism and microaggression have been widely documented and are associated with higher rates of cardiometabolic disease and other adverse health outcomes (Chetty et al., 2020; Gee and Ford, 2011; Link and Phelan, 1995; Phelan and Link, 2015; Williams, 1999; Williams, Lawrence, and Davis, 2019). The stresses associated with the increased pace of daily life may be especially acute for younger adults who came of age in the digital era but lack financial or career stability (IOM and NRC, 2015). As discussed earlier, two decades of research has documented that exposure to adverse childhood experiences,
including poverty, leaves a lifelong imprint on the brain and body and undermines long-term health, increasing the incidence of substance abuse, heart disease, diabetes, and poor health behaviors (Flaherty et al., 2013; McEwen and McEwen, 2017; Su et al., 2015).
Chronic stress can cause both biological and behavioral responses that affect health. Repeated activation of the stress response system keeps levels of stress hormones elevated, which in turn increases levels of blood glucose, inhibits insulin production to prevent glucose from being stored, increases glucose intolerance, contributes to the buildup of fat tissue and weight gain, increases pulse rate and blood pressure, and damages blood vessels and arteries (Lupien et al., 2006; McEwen and Lasley, 2002; Seeman et al., 1997). Stress may also induce unhealthy coping behaviors, such as smoking, alcohol and drug abuse, and unhealthy eating habits that may affect cardiometabolic risks and help explain observed mortality trends (Geronimus et al., 2010; Jackson, Knight, and Rafferty, 2010). For example, the stress hormone cortisol triggers cravings for high-fat, sugary, and salty foods (Chao et al., 2017; Tryon et al., 2013), and the extra calories consumed as a result are converted to fat deposits that increase the risk of heart disease, diabetes, high blood pressure, and stroke.
Chronic stress can bring on feelings of despair and hopelessness. When individuals lack hope for their future and feel that they have nothing to lose, they may engage in unhealthy and risky behaviors because they are apathetic about their health or the risk of premature death (Gaydosh et al., 2019; Goldman, Glei, and Weinstein, 2018; Harris, Duncan, and Boisjoly, 2002). To the extent that despair and hopelessness are associated with chronic stress, an increase in those feelings may also play a role in increasing rates of obesity, hypertension, stroke, and diabetes, although as detailed in Chapter 7, the evidence that rates of despair have increased or are responsible for the increase in working-age mortality is inconclusive.
Substantial research has documented these profound shifts in U.S. social and economic conditions since the 1970s (Cherlin, 2014; McLanahan and Sandefur, 1994; Wilson, 1987) and accompanying cultural changes in American work and home lives in the digital era, but the ways in which such macro-level long-term changes affect cardiometabolic mortality are difficult to ascertain. Research is only beginning to illuminate how daily adverse environmental exposures that chronically activate the brain’s stress management system can lead to dysregulation of individual and multiple body systems, including the immune, metabolic, and cardiovascular systems, involved in predisease pathways (Hertzman and Boyce, 2010; McEwen and Lasley, 2002; NRC, 2001). Whether such long-term shifts that have occurred over decades and are codependent and synergistic can lead to population-level changes in cardiometabolic mortality is unknown, and the research tools in the form of data, study design, and methodology with
which to address this broad and important question are not yet available. There is more evidence, however, for how certain population groups are more vulnerable to adverse social and economic changes and the associated biological consequences of chronic stressors, and this avenue of research may be more fruitful in policy and programmatic efforts to reduce such disparities and thereby achieve health equity within American society.
SUMMARY
The contribution of cardiometabolic mortality to the recent rise in working-age mortality is complex and involves several countervailing trends. Death rates due to ENM diseases and hypertensive heart disease generally increased during 1990–2017, and especially since 2010. While there have been significant long-term reductions in mortality from ischemic heart disease and other diseases of the circulatory system since 1990, much of that progress stalled after 2010. The combination of these trends acted to increase all-cause mortality after 2010 because the slowdown in mortality declines from ischemic heart disease and other circulatory diseases no longer offset the rise in mortality from ENM diseases and hypertensive heart disease.
Certain populations were especially vulnerable to rising cardiometabolic mortality. Mortality due to hypertensive heart disease increased among young adults, White adults, and Hispanic males during the 1990s and 2000s. Mortality due to ENM diseases increased during the 1990s among all working-age adults except younger (ages 25–44) Hispanics. Most younger working-age adults saw additional increases in mortality from ENM diseases through the 2000s.
The stalling of improvements in CVD mortality in the 2010s has exposed these troubling trends in cardiometabolic mortality. Since the 2009–2011 period, mortality from ischemic heart disease and other circulatory system diseases has remained largely unchanged among working-age adults, while mortality from hypertensive heart disease and ENM diseases has increased among all younger (ages 25–44) working-age adults except Hispanic females; all White working-age adults; and all working-age males, particularly Blacks. These worrisome changes in cardiometabolic mortality have been particularly pronounced in the South and in nonmetros and other areas outside of large central metros. Large central metros and the Northeast region of the country have generally experienced the most favorable trends in cardiometabolic disease mortality over time. Because of these trends, the gap in mortality by metropolitan size has grown over time, particularly among White working-age adults.
There are three potential explanations for these trends: the obesity epidemic, which has been identified as a major cause of increasing mortality
risks affecting multiple cardiovascular and metabolic systems; the diminishing returns of medical advances in lowering CVD mortality as the lagged cardiometabolic consequences of the obesity epidemic blunted the impact of medical innovations, and disparities in awareness of, access to, and use of drug therapies and smoking cessation persisted; and other social, economic, and cultural changes that increased multiple stressors among the working-age population, especially vulnerable subgroups. Although all three explanations have some support, the increased prevalence of obesity in the U.S. population is the most significant.
Obese adults are at higher risk of mortality from cardiometabolic diseases relative to nonobese adults; obesity increases the risks of hypertension, stroke, CHD, and type 2 diabetes. Rates of obesity have increased in a similar fashion among adults in all income and educational categories, such that over time, income and educational differentials have remained more or less the same: higher-income versus lower-income populations have a lower prevalence of obesity, and working-age adults with a college degree have a lower prevalence of obesity compared with their less-educated peers. As a result, changes in obesity are having little impact on socioeconomic disparities in working-age mortality trends.
The most proximate causes of obesity involve health behaviors that produce an imbalance between calories consumed and calories expended. The question remains as to what factors have led people to consume more and not expend as many calories as they are consuming. Much attention has been devoted to the role of the obesogenic environment, defined as an environment that promotes weight gain and is not conducive to weight loss within the home and environment. While substantial evidence indicates how Americans’ physical environments have changed to become increasingly obesogenic, the multifactorial and complex pathways by which these changing environments have increased the level and spread of obesity and, by extension, the rise in mortality from ENM diseases and hypertensive heart disease or the slowing declines in mortality from ischemic heart and other circulatory diseases represent major research challenges.
Medical advances in drug development and prevention, treatment, and control of chronic diseases, together with increasing knowledge about the health effects of cigarette smoking, played a large role in the long-term decline in mortality from ischemic heart disease and other circulatory diseases that began in 1970, but their impact lessened after 2010, when progress in reducing CVD mortality stalled. The diminishing returns of medical advances after 2010 appear to be due to the lagged cardiometabolic consequences of the rise in obesity to which adults of working age had been exposed for 30 years. In particular, young adults approaching midlife have been exposed to obesogenic environments their entire lives and are experiencing severe cardiometabolic consequences (e.g., rising rates
of diabetes, hypertension, and CVD) compared with their peers of prior generations. Indeed, the rising trends in mortality from ENM diseases and hypertensive heart disease among young adults reflect this cohort effect of the obesity epidemic. Therapeutic approaches can address the cardiometabolic consequences of obesity, but disparities in access to and uptake of these treatments may also have slowed progress in reducing CVD mortality. Increasing investments in closing the prevention and treatment gap in high-risk populations could help reduce these disparities to achieve greater equity in therapeutic care and spur continued reductions in CVD mortality.
Finally, social, economic, and cultural changes over the past 50 years represent a natural progression among all advanced societies around the world. These changes have increased the pace and efficiency of work and social interactions but have also necessitated more education, training, and technological skills to keep up with the faster pace of life and dwindling opportunities for social mobility. These shifts have left those without the necessary education and training behind, as more vulnerable populations have lost the ability to find decent-paying jobs to support a family or live in a good neighborhood. Falling behind exacts a biological and emotional toll on those with less education; racial/ethnic minorities; those living in rural areas; and young adults in particular, who lack the resources to avoid the multiple daily stressors that can slowly and permanently damage their endocrine, metabolic, and cardiovascular systems and thereby increase their mortality risks. While research has established these links between chronic stress and CVD risks, it has not provided evidence that long-term social, economic, and cultural changes have brought about the recent changes in cardiometabolic mortality.
IMPLICATIONS FOR RESEARCH AND POLICY
The evidence suggests that increasing obesity is the most important explanation for rising rates of cardiometabolic diseases, including diabetes, hypertension, and CVD, in the United States, driving up rates of mortality from ENM diseases and hypertensive heart disease and stalling progress in mortality due to ischemic heart disease and other circulatory diseases. Moreover, research indicates that the earlier in life one becomes obese, the earlier and more severe are the adverse health consequences because most people who become obese do not become nonobese in their lifetime (Harris et al., 2020). Thus the promotion of healthy weights and the prevention of obesity are a critical research and policy priority. This priority is not new; what is new is mounting evidence that obesity is a key driver of cardiometabolic morbidity and mortality trends, especially among young working-age
adults, and will continue to be so for some time in the future unless progress is made in reducing obesity in the population and disparities therein.
The 2012 National Academies’ report Accelerating Progress in Obesity Prevention: Solving the Weight of the Nation (IOM, 2012a) advances a number of policy recommendations regarding obesity prevention that are highlighted here as starting points for the committee’s research recommendations. That report uses a systems perspective to advocate for change in the environments related to five areas for obesity prevention: environments for physical activity, food and beverage environments, messaging environments, health care and work environments, and school environments. Similar to the committee’s conceptual framework in Chapter 6 (Figure 6-1), the 2012 report outlines the ways in which upstream environmental change in societal messaging about (un)healthy food and diets filters down to and influences the meso-level environments of the physical neighborhood, schools, health care, work, and food and beverage consumption in independent and overlapping ways to affect obesity among individuals nested within families and communities. The present report views obesity prevention through the same lens and calls for the use of a life-course, multilevel, multifactorial approach to identify the multiple interactive causal pathways and drivers most responsible for the rise in obesity. In 2021, the research community is in a better position to undertake this work than it was in 2012, with new and additional data sources needed to apply this life-course, multilevel approach.
Research applying such an approach is needed to enable the development of a multipronged public policy strategy for mitigating this major public health problem. Almost all obesity scholars point to the important role of obesogenic factors in the physical and food environments, including individual health behaviors involving diet and physical exercise and societal-level changes in food production, transportation systems, access to green spaces, and sedentary work environments. For example, there is evidence that technological changes in the way food is produced, distributed, and consumed have contributed to the increase in obesity, and that public health policy can play a role in improving the production of healthy foods and reducing the distribution and consumption of unhealthy foods, especially among children and adolescents. The research reviewed in this chapter also documents the success of healthy diets in achieving short-term weight loss, and regular exercise is almost always beneficial in reducing overweight and obesity. However, efforts to maintain lifestyle changes that support healthy diets and programs to promote regular exercise (e.g., worksite health promotion programs, free gym memberships) may not always work or be sustainable. Thus, research is needed to explore the factors that erode short-term successes in diet and exercise changes and, conversely, the factors that promote long-term lifestyle changes that can reduce obesity, with a focus
on environmental drivers (e.g., occupational activity, access to recreational space and activities; exposure to chemicals, food deserts, economic inequality, residential segregation, duration of use of electronic screens, increased consumption of medications). In sum, the committee recommends research to evaluate the effectiveness of programs and policies designed to improve cardiometabolic health using a multilevel framework.
Many programs and policies implemented at the societal or community level are upstream and vary in their impact on individuals’ behavior and health because the mechanisms through which they operate vary by local context and individual characteristics. Since the 2012 National Academies’ report was issued (IOM, 2012a), more data sources enabling examination of how macro-level policies and programs are linked to individuals and the environments in which they live and work have become available with which to conduct the multilevel research recommended in the present report. For example, several national longitudinal studies, such as the Health and Retirement Study, the National Longitudinal Study of Adolescent to Adult Health (Add Health), the Fragile Families and Child Well-Being Study, the Panel Study of Income Dynamics, and the National Longitudinal Study of Youth, provide data at multiple levels (state, county, census tract, family, individual) and measure behavioral and health outcomes of individuals throughout their life course. With such multilevel longitudinal data, researchers will be able to identify the intervening multilevel mechanisms (e.g., family poverty, consumption of sugary drinks, physical activity) through which macro-level policies and programs (e.g., soda taxes, urban development) operate to affect cardiometabolic morbidity and mortality.
RECOMMENDATION 9-1: Federal agencies, in partnership with private foundations and other funding entities, should support research that evaluates the effectiveness of programs and policies designed to improve U.S. cardiometabolic health and that considers the impact of changes at multiple levels of analysis:
- At the individual level, research should continue to evaluate the effectiveness of programs and policies that promote consumption of healthy foods (e.g., mandatory labeling of food ingredients or components, fruit and vegetable subsidies) and the adoption of healthy lifestyles (e.g., subsidies for sports activities; urban development that prioritizes walking, biking, and transit). Likewise, research should continue to evaluate the effectiveness of programs and policies that discourage the consumption of poor-quality foods (e.g., sugar and soda taxes, nutritional standards and dietary guidelines from the U.S. Department of Agriculture and the U.S. Department of Health and Human Services) and unhealthy lifestyles (e.g.,
- insurance rating based on poor health habits such as smoking, zoning laws for fast food restaurants and alcohol outlets).
- At the societal level, research should consider systemic changes in food production, workplace systems, and transportation and other societal-level changes in the United States that foster and sustain obesogenic environments and sedentary lifestyles to determine the pathways through which such environments have deleterious consequences for population health.
Identifying the upstream to downstream factors that can explain how the physical environment leads to rising obesity and thus to rising cardiometabolic morbidity and mortality is a research priority, but it is a challenging one posing substantial data demands and requiring longitudinal studies or well-designed experiments and interventions. Although experimental designs tend to lack generalizability, they provide some of the strongest evidence of causal impacts—a major gap in the current literature on the effects of obesogenic factors in the environment. The committee therefore endorses experimental research designs that examine the impact of changes in such obesogenic factors as access to green space, walkability, dependence on automobiles, density of fast food restaurants and alcohol outlets, and availability and promotion of high-calorie and processed foods on changes in obesity prevalence and BMI levels. There may be opportunities to take advantage of existing neighborhood experimental and evaluation projects, such as Moving to Opportunity or the New York City Housing and Neighborhood Study, by linking built environment data elements to the pre- and post-neighborhood geographic locations of families in these studies.
RECOMMENDATION 9-2: Federal agencies, in partnership with private foundations and other funding entities, should support research that uses experimental designs and takes advantage of existing neighborhood experimental projects to examine the causal role of factors in the obesogenic environment and determine which have the greatest role in the rise in obesity prevalence and body mass index levels.
The food industry and policy makers alike have contributed to the obesity epidemic in both intentional and unintentional ways. The food industry has successfully leveraged advertising and marketing techniques to boost consumption of calorie-dense foods. The U.S. Department of Agriculture, which for decades has promulgated dietary guidelines to promote healthy eating and combat obesity, issued farm subsidies in the early 1980s that generated an oversupply of corn, thereby lowering its cost and encouraging the production of high-fructose corn syrup, the major sweetener added to the food supply. The use of high-fructose corn syrup and the wide distribution
of low-cost, calorie-dense foods have combined to raise U.S. caloric intake per capita far above that of other high-income countries. Some efforts are under way to regulate food advertising, tax unhealthy foods and beverages, mandate product labels, list calorie content on restaurant menus, and implement zoning of fast food restaurants and alcohol outlets. But further action on the part of both the food industry and public health authorities is needed to discourage the production and purchase of unhealthy foods, or at least provide consumers with better information with which to make healthier food choices. Tighter regulations to improve the nutritional content of foods and beverages, prohibit misleading advertising, alter pricing to encourage the consumption of healthy foods, or use zoning and land use policies to influence the location of food stores and restaurants could make it easier for the public to make healthier dietary choices but also encroach on the free market rights of the food and restaurant industries.
POLICY CONCLUSION 9-1: To reduce the per capita calorie consumption and body mass index levels of the U.S. population, policy makers will need to implement laws and regulations that preserve a healthy balance between the rights of the food industry, advertisers, grocers, and restaurants to enjoy free market competition and the public health imperative to limit the promotion and consumption of foods and beverages that contribute to obesity.
Mounting evidence shows that the current generation of young adults nearing midlife (i.e., those born in the late 1970s and early 1980s), who have spent their entire lives amid an obesity epidemic, is at high risk of morbidity and mortality from obesogenic conditions. Those born in the 1990s, 2000s, and 2010s have also experienced an obesogenic environment their entire lives, with foreboding health consequences for future cohorts of young and middle-age adults. Obesity needs to be avoided in early life because any exposure, and especially prolonged exposure, can greatly increase risks for cardiometabolic diseases and mortality. In addition, the early stages of chronic disease associated with obesity often present as asymptomatic risk factors (e.g., hypertension, hyperlipidemia, glucose intolerance), especially among young adults for whom permanent biological damage has not yet manifested in disease. Young adults often fall through the cracks of regular medical care if they do not have stable employment or health insurance that provides for regular health care screening. U.S. cohort studies make clear that the obesity epidemic has made the cardiometabolic health of young adults today much worse than that of their peers in previous cohorts. The alarming health status of today’s young adults portends a shorter work life, with societal consequences for productivity and growth, and escalating medical care costs for individuals, families, and society
as a whole. The committee therefore believes that prevention of obesity should focus on children and young adults, especially those in vulnerable populations.
RECOMMENDATION 9-3: Designers of obesity prevention programs should focus on developing programs that start early in life and target children and adolescents most at risk of obesity (e.g., racial/ethnic minorities, females, people living in poverty and in neighborhoods of low socioeconomic status) and those who are overweight or gaining weight, thus intervening before obesity trajectories become set throughout adulthood.
Persistent disparities in access to evidence-based preventive and therapeutic care for the control of chronic diseases hinder continued progress in lowering CVD mortality. Appropriate use of medications that are effective in controlling hypertension, diabetes, and heart disease and thus in lowering CVD mortality is not as widespread as would be ideal, and persistent disparities in successful management of these chronic diseases continue to slow progress in mortality reduction. Because much of this shortfall may result from individuals not fully understanding how to care for their conditions (because of inadequate instructions, language barriers, or poor communication) or being unable to implement or maintain care (because of challenges with accessing and affording medications, providers, and other resources), improved systems for solving these challenges is an important priority for continued improvement in cardiometabolic mortality. Efforts to improve systems for delivering existing treatments might yield more benefit in lowering mortality relative to incremental gains from innovative new treatments. For cardiometabolic conditions in particular, more implementation research is needed to better understand barriers to accessing and maintaining effective control and use of highly effective medications and other treatments. A priority for such research is to design solutions that address the needs of high-risk populations, including communities of color and less-educated and lower-income populations who face higher rates of hypertension and other cardiometabolic risk factors, whose conditions often escape early detection, and who face added challenges in accessing information and clinical services to treat these conditions. Further research likewise is needed to address the barriers to smoking cessation faced by populations that continue to smoke at high rates, especially those of lower socioeconomic status.
RECOMMENDATION 9-4: To improve systems for delivering preventive care (e.g., smoking cessation) and existing treatments for hypertension, diabetes, and heart disease, federal agencies, in partnership
with private foundations and other funding entities, should support research focused on better understanding the barriers to prevention and control of cardiometabolic disease faced by individuals—especially less-educated and lower-income populations—and evaluating potential solutions for removing those barriers.
Finally, social, economic, and cultural shifts over the past 50 years have profoundly changed the family, work, and community environments of daily life in the United States, and many of these changes have increased stress in the lives of Americans. This increasing and chronic stress may be contributing to the rising trends in mortality documented in this report, including deaths due to substance use, suicide, and cardiometabolic diseases. The daily sources of stress are not likely to abate, and they are felt more intensely among certain subgroups of the population. Exposure and responses to stress are strongly graded by socioeconomic status such that those with less education, lower income, greater job instability, and long-term unemployment experience much more stress in their lives and are more likely than those of higher socioeconomic status to engage in unhealthy behaviors (e.g., smoking, alcohol and drug use, overeating of ultraprocessed foods, self-harm) as a way of coping with that stress. Policy approaches in this realm include upstream macro-level approaches to reduce downstream socioeconomic disparities and promote healthy behaviors that lessen stress (e.g., exercise, social interaction, sleep, meditation). The committee therefore developed a cross-cutting recommendation for more research to explore the sources of increasing stress in the lives of Americans and to identify which population subgroups are most affected by that stress (see Chapter 11, Recommendation 11-5).




















































