10
Enhancing Research in Primary Care
Primary care is the only function within the health care system responsible for all people in the population. It is the platform where more than one-third of all health care visits are made (Johansen et al., 2016), and, for many, the only place they seek care. Nonetheless, with few exceptions, primary care largely depends on evidence derived from research on subspecialty care, hospital settings, or single-disease cohorts (Petterson et al., 2014). Primary care practice–based research networks (Hickner and Green, 2015; Phillips et al., 2007b) have famously turned our understanding of disease and treatment on its head regarding brown recluse bites (Mold and Thompson, 2004), depression, upper respiratory infections, heart failure, unstable angina, and radiologic examinations of headaches, among many other topics (Westfall et al., 2007), by studying the epidemiology of symptoms, conditions, and treatments in the settings where illness often first presents (Westfall et al., 2007). Research has also demonstrated that how primary care clinical data are collected and organized can enable translating electronic clinical data into probability engines for clinical decision making and research into how symptoms become disease and how disease treatment affects outcomes (Eberl et al., 2008; Soler et al., 2008).
The neglect of basic primary care research (PCR), and lack of research that draws on primary care–specific databases, such as clinical registries, not only adversely affects primary care outcomes but also leads to the lack of a population-based understanding of illness and disease along the health care spectrum. Better PCR support could lead to answers to questions that are critically important for improving population health. For example, adequate support could propel pioneering work regarding how best to
incorporate data on the social determinants of health (SDOH) into clinical decision making. Such studies could also help health care systems and society at large better address patients’ important social needs as a means of improving health outcomes (Cottrell et al., 2018; DeVoe et al., 2016; Gold et al., 2017).
Many leading experts in biomedical and scientific inquiry predict that future scientific breakthroughs and advancements will require transdisciplinary collaboration (Méndez, 2015; NRC, 2015). PCR teams are naturally transdisciplinary and span the boundaries of the biological, physical, and social sciences. Primary care and health care have “wicked” problems that have not been addressed with traditional methods and siloed researchers (Camillus, 2008); tackling them will require alliance building and collaboration with a range of expertise in both qualitative and quantitative research designs. Moreover, once discoveries are made in a few settings, important questions about how to most effectively disseminate, implement, and scale evidence-based interventions will be key, demonstrating the critical role of implementation scientists on PCR teams.
Returning to the fundamental question of why PCR is needed, Richard Hobbs of the Nuffield Department of Primary Care Health Sciences at Oxford University summarized it well:
Given that most patient contacts originate and end in primary care in most developed health systems, the necessity to research more within primary care is obvious. The full spectrum of disease is represented, the long trajectory of disease is discoverable, and the patient subjects are representative of the total population and demonstrate the full range of behaviours … care in the community should be based on evidence from community populations, whether for diagnostic test performance and thresholds or for therapeutic interventions. (Hobbs, 2019, p. 424)
Taking this one step further, care in the community should be based on evidence from community populations that is gathered in partnership with community-based research teams that understand the context and players and bring both a primary care and an equity lens to their work.
THE 1996 INSTITUTE OF MEDICINE REPORT
The report Primary Care: America’s Health in a New Era (IOM, 1996) declared that the science base for primary care is modest and the infrastructure underlying the knowledge base is skeletal at best. It added that current clinical research may have little to offer to primary care clinicians, lessons from well-done PCR are not available to inform the larger picture of health care organization and delivery, and the paucity of PCR and development leaves primary care insufficiently prepared to confront the challenges and
opportunities inherent in the committee’s definition of primary care. The current committee believes that these three findings remain unchanged.
These findings led to four recommendations in the 1996 Institute of Medicine (IOM) report that the current committee still finds relevant:
Recommendation 8.1 Federal Support for Primary Care Research called for a declaration of a lead PCR agency and adequate research infrastructure funding.
Recommendation 8.2 National Database and Primary Care Data Set called for a new national health care needs database beyond existing national health surveys.
Recommendation 8.3 Research in Practice-Based Primary Care Research Networks called for providing “adequate and stable financial support to practice-based primary care research networks” (p. 12).
Recommendation 8.4 Data Standards called for new data standards for primary care clinical data collection akin to the International Classification for Primary Care, particularly for capturing episodes of care.
The first recommendation remains relevant because there is still no federal agency charged with developing and advancing a robust program of PCR and funded to support that mission. (See later in this chapter for more on Recommendation 8.1.) For 8.2, the 1996 report’s findings about relying on important but insufficient national health surveys to assess the health of the general public, their care-seeking behaviors, their care use, and the quality of the care they receive still hold true. In fact, the difficulties with response rates and sampling challenges have only grown since 1996. Re-evaluating these surveys and considering alternative ways to sample population and primary care data remain a priority.
Practice-based research networks (PBRNs) (Recommendation 8.3) are increasingly important for frontline health and health equity research, and yet they continue to struggle to find infrastructure and sustain funding (Gaglioti et al., 2016; Goldstein et al., 2018; Hall-Lipsy et al., 2018; Westfall et al., 2019).
Finally, for Recommendation 8.4, the electronic health record (EHR) evolved to optimize payment for delivering health care, but it has not fulfilled its potential to support research and improve health care or population health. Criticism of EHRs is abundant, but their lack of functionality to capture and organize data in a way that could inform primary care is notably problematic (Krist et al., 2014, 2015; Phillips et al., 2015). Currently, researchers rely on the National Ambulatory Medical Care Survey (NAMCS) from the National Center for Health Statistics, which samples some 3,000 physicians, to capture what is happening in outpatient care. In 2016, the criteria were sufficiently outdated that nearly half the initial
sample needed to be excluded. As a result, 1 week in the lives of 209 primary care physicians (263 if obstetrics and gynecology is included) has been used to determine most of what is known about more than 400 million visits (Rui and Okeyode, 2016). In an era when nearly 90 percent of office-based primary care physicians use EHRs (ONC, 2019) and an increasing number of EHRs collect patient-reported outcomes, the nation has opportunities to assimilate large regional and national databases, standardize and normalize these data, and use this information to enhance the understanding of population health and health disparities and the relationship of primary care to both. Tapping EHR and claims data fully for research would meet the 1996 IOM report’s data recommendation partway, but doing so fully also requires having better data sources on people who do not seek care. NAMCS remains incomplete for capturing the contribution of other primary care team members, and EHR and/or claims data could be assembled to better characterize their contributions and care effects.
THE STATUS OF RESEARCH SUPPORT IN PRIMARY CARE
As noted in the previous section, the 1996 IOM report recommended that the U.S. Department of Health and Human Services (HHS) should “identify a lead agency for primary care research and … the Congress of the United States [should] appropriate funds for this agency in an amount adequate to both build the infrastructure required to conduct primary care research and fund high-priority research projects” (p. 11). Today, the Agency for Healthcare Research and Quality (AHRQ) is the only federal agency with a mandate for PCR, but its National Center for Excellence in Primary Care has consistently had no specific research funding and a nebulous structure, at best, limiting its ability to meaningfully contribute to the field (CAFM, 2019). In 2018, the National Academies held a workshop on the Future of Health Services Research (NASEM, 2018b). As in the 1996 report, primary care was presented as highly overlapping with health services research (HSR), as both share struggles for support, though with some distinctly different needs. This workshop also discussed strategies to develop quality measures and embedding research skills in care delivery, along with models of engaging care-seekers and communities throughout the research process. Central to the discussion was the lack of federal investment in studying the process of health care delivery and factors that influence effective delivery and positive outcomes. This workshop also highlighted that current support for HSR focuses mainly on hospital settings, with a minority of resources going to primary care.
Between 2002 and 2014, family medicine, the specialty that provides more than one-quarter of all outpatient visits across the health care system, consistently received around 0.2 percent of total research funding dollars
and 0.3 percent of all awards by the National Institutes of Health (NIH) (Cameron et al., 2016). A related qualitative study found that NIH officials valued the clinical relationships in family medicine but saw no research home for it at NIH (Lucan et al., 2009). A later study of the Patient-Centered Outcome Research Institute (PCORI) found that primary care fared better there but that less than one-third of the studies it supported involved or were relevant to primary care (Mazur et al., 2016).
This long-standing lack of funding for PCR led to a provision in H.R. 1625, the Consolidated Appropriations Act of 2018,1 which directed and authorized AHRQ to contract with an independent entity for a study on HSR and PCR supported by federal agencies.
The goal of the study was to provide an independent assessment of the current breadth, scope, and impact of HSR and PCR supported by HHS’ 11 operating divisions and the Department of Veterans Affairs (VA) since fiscal year 2012. In support of this goal, the study was to identify research gaps and propose recommendations to AHRQ for maximizing the outcomes, value, and impact of HSR and PCR investments during the next five to 20 years. (Mendel et al., 2020a, p. 1)
The study, completed by the RAND Corporation in 2020, found that between 2012 and 2018, federal agencies had funded 1,090 primary care projects and another 8,845 that could be classified as HSR and/or PCR. Of the studies that were purely about PCR, NIH funded 750, and the VA was second at 150; however, across all research agencies, PCR represented only 1 percent of all funded projects (Mendel et al., 2020a).
RAND noted that AHRQ’s distinct focus on health care systems, synthesis of evidence, and dissemination of innovations across settings and populations is unique among government agencies, most of which (including NIH) tend to focus on more specific topics, such as diseases, populations, or settings. The RAND study offered recommendations for HSR and PCR (Mendel et al., 2020a) (see Box 10-1).
The RAND report also noted that PCR has emerged as a distinct field in its own right, addressing a central component of the health care system. Study participants mentioned challenges to coordination of HSR and PCR portfolios, including the breadth and volume of research activities across the federal HSR and PCR enterprise, differing research time frames among agencies, and the lack of targeted funding for a lead agency to coordinate PCR in particular.
The committee agrees that a separate interagency prioritization process for PCR would ensure that the country’s vital needs for primary care
___________________
1 Consolidated Appropriations Act of 2018, Public Law 115-141 (March 23, 2018).
knowledge and evidence base are attended to, incorporating the stakeholders needed to inform prioritization and spanning the full breadth of the primary care basic research questions. Federal PCR efforts are not yet aligned but could be by coordinating both PCR across federal agency research portfolios and the funding to achieve it (Mendel et al., 2020b). As discussed in Chapter 8, this committee notes that one role of a Secretary’s Council, supported by an advisory committee formed under the Federal Advisory Committee Authority, could be to coordinate PCR activities. One of the tasks of the Council and the advisory committee would be to assess the adequacy of PCR support across HHS agencies and to direct interagency efforts to support PCR and research infrastructure (see Chapter 12 for the committee’s recommendation related to this).
The current structure and priority setting of institutes within NIH and programs within AHRQ often overlook the need for knowledge about integrating, personalizing, and prioritizing whole-person care (Thomas et al., 2018). These essential primary care functions require a science base able to support the work of primary care as a force for integration in a fragmented health care system (Cebul et al., 2008; Hughes et al., 1977; Stange, 2009b). In addition, there is a prevalent but unfounded notion that primary care is simple (Martin and Sturmberg, 2009; Muche-Borowski et al., 2017), and can be supported merely by adding up knowledge gained from outside settings and disease- or organ-specific research studies (Creighton, 2013). These factors have led to a diminished appreciation for the generalist function in health care (Alston et al., 2019; Gerard et al., 2008; Mazzone et al., 2015; Tuggy et al., 2015) and left it insufficiently supported by a relevant knowledge base (IOM, 1996; Stange et al., 2001). The lack of support for the mechanisms by which primary care functions has resulted in a devaluing of relationship-centered whole-person care (Beach and Inui, 2006; Rudebeck, 2019) and an increasing commodification (Heath, 2006; Lown, 2007; NASEM, 2018a) and depersonalization of the health care enterprise (Rotenstein et al., 2018; Shippee et al., 2018).
Disease-specific knowledge can be very useful in informing many aspects of primary care. However, most clinical trials exclude people with the comorbid conditions (Fortin et al., 2006), social and medical complexity (Peek et al., 2009; Ronis et al., 2019), and undifferentiated illness (Epstein et al., 2006; Heath, 1995) that are the norm in primary care. Evidence-based guidelines ignore the complexity of primary care (Casalino, 1999), and the unintended consequences of focusing quality improvement only on narrowly defined evidence are profound (Galvin, 2006; Lipsitz, 2012; McDonald and Roland, 2009; Sutton et al., 2009). No current funding home exists to support research on the integrating, personalizing, and prioritizing functions that provide much of the added value of primary care in functional health care systems.
Where the RAND report judged that the risks associated with moving AHRQ into NIH outweigh the potential advantages, a both/and strategy may be warranted as their individual foci are distinct. The 2007 series of IOM reports on emergency medicine (IOM, 2007a,b,c) are credited on the NIH website (NIH, 2019) for the formation of the Office of Emergency Care Research, which has the important task of working across NIH institutes to identify, coordinate, and support research relevant to emergency medicine. The formation of an Office of Primary Care Research at NIH operating with similar goals and functions could be tremendously helpful to making PCR a more robust part of the NIH portfolio. This would not obviate the need for the National Center for Excellence in PCR at AHRQ; it could still fill this important role, but only if Congress and HHS dedicate resources to it. This would allow AHRQ to continue as a focused funder of PCR but build an internal advocacy and coordination unit within NIH to frame categorical (e.g., disease-specific) PCR relevant to particular Institutes and propose crosscutting primary care/generalist research questions.
There are precedents for new investment in big challenges that create distributed innovation networks and that the committee believes could serve as models for future NIH commitments to support PCR. For example, the National Cancer Act of 19712 created 71 National Cancer Institute (NCI)-designated Cancer Centers, 58 of which have research as a focus (NCI, 2019). In 2006, NIH launched the Clinical and Translational Science Awards (CTSA) program with the goals or increasing the quality of clinical and translational research through scientific breakthroughs and enhancing collaborations among institutions, disciplines, and researchers (Frechtling et al., 2012). In 2011, NIH swept these into a new NIH center, the National Center for Advancing Translational Sciences, building a network of more than 50 institutional hubs.
NEEDED RESEARCH IN PRIMARY CARE
The preceding chapters of this report have identified specific PCR needs. All of these foci of functionality suffer from a lack of research infrastructure and sustained support within primary care. This section focuses on these fundamental needs and the inflexibility of national research infrastructure, including funding agencies, to address them.
Primary Care Basic Research
PCR methods, laboratories, and theories exist but are largely unsupported. PCR has been a pioneer in the integration of quantitative and
___________________
2 National Cancer Act of 1971, Public Law 218 (December 23, 1971).
qualitative methods (Crabtree and Miller, 1999; Stange and Zyzanski, 1989; Stange et al., 1994) and in participatory methods (Borkan, 2004; Creswell and Hirose, 2019; Macaulay and Nutting, 2006; Westfall et al., 2009) that bring together the numbers and narratives and the diverse perspectives needed to understand how care can be integrated for whole people, families, and communities (Aungst et al., 2019; Homa et al., 2015; Macaulay et al., 1999; Stange et al., 2017).
Primary care also has a robust theoretical basis (Brown et al., 1986; Checkland, 2007; Donner-Banzhoff, 2018; Greenhalgh, 2007; McWhinney, 1972, 1977; Miller et al., 2010; Starfield, 1992, 1996, 1998, 2001) but has suffered from having to force a fit between interest in relationship-centered, family-centered, whole-person care and disease- and organ-based, reductionist worldviews and methods in order to be funded. Relevant theories grounded in the wisdom of practice (Green and Lutz, 1990; Stange, 2009a) and in complexity science (Grant et al., 2011; Greenhalgh and Papoutsi, 2018; Litaker et al., 2006; Peek et al., 2009) are worthy of testing but typically seen as out of scope by categorical funders (Stange et al., 2001). The growing recognition of the inadequacy of the science base (Fortin et al., 2005) to understand and support care for the large number of Americans living with multiple chronic conditions (Parekh et al., 2011; Tinetti et al., 2019; Ward and Schiller, 2013) shows the need for research on this vital use case for primary care.
Primary Care Basic Science Research Questions
Many PCR questions can be pursued through categorical funding mechanisms, such as those available through NIH and the HSR tracks at AHRQ. Research questions that use primary care in service of a narrowly focused disease or medical condition question tend to be advantaged over those needed to build primary care delivery knowledge (Slawson et al., 2001), advantaged in the review processes that value a narrow focus, and advantaged by the tacit assumption that rigor must be equated with rigid adherence to prespecified protocols over identifying processes and outcomes that emerge from participatory and complex system processes (deGruy et al., 2015; Peek et al., 2014). The current research funding environment has prevented addressing meaningful questions critical to the advancement of primary care. No meaningful area of medicine or science can survive and thrive without sufficient support for discovery and innovation.
Here are a few examples of research questions that go unasked in the current limited funding and peer-review environment:
- What are the mechanisms by which primary care works to advance the health of people and populations, while controlling health care costs and increasing equity and system-level quality of care?
- How do we explain the paradox of primary care—that despite the apparently poorer quality of care for disease-specific measures, health care systems based on primary care have greater population health, equity, and quality of care, at lower cost?
- What are the outcomes of investing in health care as a relationship rather than as a series of transactions?
- How can care be integrated for people living with multiple chronic conditions?
- How do continuity, comprehensiveness, and coordination of care work together to affect person and system outcomes?
- How do primary care clinicians approach prioritizing care, particularly for those who have multiple conditions with potentially competing evidence-based guidelines?
- How are trust and trustworthiness developed over time in relationships between care-seekers, families, and communities and their primary care clinicians? What are the trade-offs and outcomes?
- How do we measure what matters in/from care of the whole person, not just their individual diseases?
- How does primary care prioritize care based on a whole-person focus, and how does that affect person and system outcomes?
- How can we network the primary care platform to share best evidence and best practices for prioritizing and managing care during a public health crisis or pandemic?
- What kind of variability in primary care is useful? How do we distinguish between variability that involves personalizing care and variability that represents missing opportunities for evidence-based care that affects outcomes?
- What are the new information management and science roles required in a high-performing primary care setting?
- How can primary care serve as a force for integration in a fragmented health care system and society?
- How do we prioritize individual patient-directed goals with guideline recommendations and quality metrics?
- What does it mean to provide care in the context of family and community? How do different approaches affect outcomes?
- How should training of primary care clinicians change in order to stress the benefits of a whole-person approach to care, rather than approaching the care of people as a sum of what we learn in pieces, studying their organs and systems?
- By what mechanisms does primary care affect health care and health equity?
- How can care be optimized for people presenting with early, undifferentiated illness?
- How does primary care best provide care for the many people whose illnesses don’t fit into current disease categories?
- Are intermediate clinical outcomes the best approach for assessing quality and value in primary care?
- How can primary care of whole people be best supported—at the level of the community, practice, local health care system, and state and national policy?
- How do we make it easier for clinicians to consistently deliver the right care at the right time?
In thinking about the need for the types of studies listed above, it is clear that the questions cannot be answered by one discipline alone. The complexity of primary care requires a transdisciplinary approach that brings together scientists from medicine and other health professions but also public health, psychology, sociology, economics, social justice, and equity, to name a few.
Epidemiologic Research
Basic research about the prevalence and presentation of symptoms and illness is largely driven by sample surveys when it no longer need be. The basic understanding of the content and complexity of primary care is woefully lacking. For example, SDOH inform whether someone stays healthy, seeks care, carries out treatment or prevention strategies, and needs acute care and receives it in a timely manner. Epidemiological methods to capture the complex layers of factors that influence health and illness trajectories are unfortunately lagging behind the recognition of their importance. Understanding how SDOH, or drivers, affect health and testing upstream interventions to protect at-risk people has a natural fit in primary care. Moving toward interventions needs information on prevalence and effects that remains largely unexplored in primary care.
Clinical Research
Studies of diagnostic and therapeutic strategies should be relevant to primary care, incorporate the individual’s perspective with respect to acceptability and feasibility, and incorporate, rather than exclude, multimorbidity (De Maeseneer et al., 2003). The Primary Care and Interventions Unit of the United Kingdom’s National Health Service occasionally asks primary care physicians about gaps in research that would help them care for people (Lecky et al., 2020). The United States has no such systematic inquiry. Given that many of the guidelines and therapeutic options for specific diseases are created based on studies of populations without multiple
conditions, studies are critically needed for patients in primary care who typically have more than one condition to understand the real-world risks and benefits of care pathways.
Practice-Based Research Networks
A 2007 JAMA article, “Practice-Based Research—‘Blue Highways’ on the NIH Roadmap,” effectively made the case for the need for connections between the “interstates” of academic science and ambulatory practices (Westfall et al., 2007, p. 404):
A potential solution to these problems is the expansion of practice-based research, which is grounded in, informed by, and intended to improve practice. Practice-based research occurs in the office, where most patients receive most of their care most of the time and may be the essential link between bench discoveries, bedside efficacy, and everyday clinical effectiveness. Practice-based research and practice-based research networks (PBRNs) may help because they can (1) identify the problems that arise in daily practice that create the gap between recommended care and actual care; (2) demonstrate whether treatments with proven efficacy are truly effective and sustainable when provided in the real-world setting of ambulatory care; and (3) provide the “laboratory” for testing system improvements in primary care to maximize the number of patients who benefit from medical discovery.
The authors offered a model (see Figure 10-1) that has led to greater engagement with NIH by using their language and imagery, but the impact has been limited. A few related NIH programs have been launched to formalize laboratories for implementation science, including NCI’s Implementation Science Centers in Cancer Control, but these are not limited to primary care, and many have been established in academic health center systems.
The current NIH Roadmap for Medical Research includes two major research laboratories (bench and bedside) and two translational steps (T1 and T2). Historically, moving new medical discoveries into clinical practice (T2) has been haphazard, occurring largely through continuing medical education programs, pharmaceutical detailing, and guideline development. Proposed expansion of the NIH Roadmap (blue) includes an additional research laboratory (practice-based research) and translational step (T3) to improve incorporation of research discoveries into day-to-day clinical care. The research roadmap is a continuum, with overlap between sites of research and translational steps. The figure [10-1] includes examples of the types of research common in each research laboratory and translational
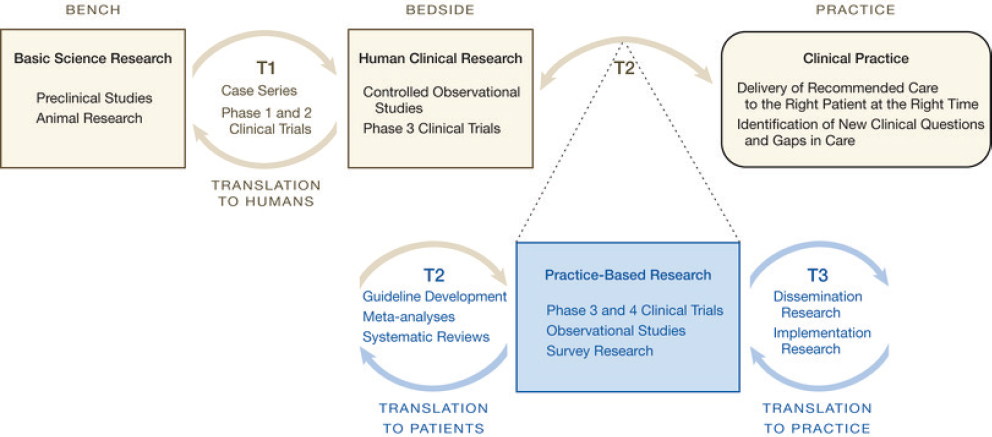
SOURCE: Westfall et al., 2007.
step. This map is not exhaustive; other important types of research that might be included are community-based participatory research, public health research, and health policy analysis. (Westfall et al., 2007, p. 405)
PBRNs are vital participatory community laboratories for “reuniting practice and research around the problems most of the people have most of the time” (Nutting and Green, 1994, p. 335). PBRNs overcome the problem of translating research into practice (Brownson et al., 2012; Cohen et al., 2008; Davis and Taylor-Vaisey, 1997) by making the research questions, settings, and populations served immediately relevant to the real problems faced by patients and primary care practices (DeVoe and Sears, 2013; Westfall et al., 2011). These reasons suggest the nation should establish and maintain an infrastructure to support PBRNs and use them to conduct research to generate the real-world evidence that primary care clinicians need to practice effectively.
AHRQ provided early support for PBRNs (AHRQ, 2012), as did the CTSA program in its early years (Fagnan et al., 2010), but AHRQ support for PBRNs has dwindled, and the CTSA program currently sees clinical practice networks primarily as vehicles for enrolling participants in clinical trials (Riley-Behringer et al., 2017). Despite its importance to the health and health care of the nation, no ongoing primary care cohort or data source exists outside of high-level health service use data. NIH’s investments in CTSA flow through the National Center for Advancing Translational Sciences, dedicated solely to this distributed research mechanism. This model could work nicely for PCR centers or PBRNs.
Secondary Data
As noted above, EHRs lack functionality to capture and organize data in a way that could inform primary care. In addition, data are currently privatized and monetized by EHR and digital health vendors. Primary care practices and even primary care researchers struggle to access existing clinical data, and they have to ask vendors for them. This unacceptable situation is stifling PCR. Clinicians and patients should be able to access and share their data with primary care researchers. These data hold great promise with adequate investment and an organized PCR infrastructure to harness them.
Health information exchanges hold a large volume of ambulatory data, and a few clinical registries do too, but these were rarely systematically used for research before the COVID-19 pandemic, and even most of these studies are based on hospital data (Lavery et al., 2020). AHRQ supports secondary research on the Medical Expenditure Panel Survey, and the National Library of Medicine has recently made notice of supporting EHR data research, but there has not been systematic support of basic studies of data collected in primary care that would harness data from more than 450 million visits per year. The capacity to apply sophisticated analyses, including machine learning, to these data could replace reliance on national surveys for understanding patterns of symptoms, illness, and treatment and offer far greater reliability.
Even so, researchers struggle with the utility of the data because of the way they are captured and classified. Clinical classification codes used worldwide have become increasingly specific because they are critical to health care business transactions, but this drastically reduces their utility for understanding patterns of care and outcomes in primary care. As noted earlier, the International Classification for Primary Care captures reason for visit and episodes of conditions, key elements for studying how symptoms relate to disease, which medications are effective or dangerous, which tests are useful, and likely outcomes for patients (Okkes et al., 2002b). This classification is in use in more than 30 countries (Basílio et al., 2016; Okkes et al., 2002a; PH3C, 2011; USYD, 2020; van Boven et al., 2017), but it has no foothold in the United States despite being in the Unified Medical Language System of the National Library of Medicine (NLM, 2020), recognized by the World Health Organization, integrated into the Systematized Nomenclature of Medicine, and able to produce International Classification of Diseases codes in the course of care.
A shared primary care data model would facilitate the accurate capture of clinical processes and enable accurate assessment for individuals, families, and communities within the larger health care enterprise (Green and Klinkman, 2015). The United States currently relies on an outdated Framingham Study, clinical trials in other settings, or meta-analysis to
create decision support tools. With the right methods and research, existing primary care data could radically improve the ability to use probabilistic, predictive models at point of care, manage panels of patients, and inform population health work (Phillips et al., 2007a).
Research Capacity
It is difficult to find primary care researchers who have not pivoted to disease-specific research to fit the NIH or PCORI models or HSR to fit AHRQ’s paradigm (Robinson and Westfall, 2011). Successful PCR departments exist around the country, and many share common characteristics that could be replicated with support (Liaw et al., 2019). Fellowship funding supports successful research training centers, often with Institutional Career Development Awards or T32 National Research Service Award mechanisms (NIH, 2020b). National Research Service Award Fellowships are also important but do not support faculty salaries (NIH, 2020a). Stimulating Access to Research in Residency Transition Scholar awards are also being implemented in some primary care residency programs. The mechanisms are in place, but without a particular preference for primary care and the limited hubs for PCR, the pipeline for primary care researchers will remain small and lacking in significant and sustained investment.
Other countries have successfully organized research around primary care, and the United States often relies on their research. Leading examples include the Netherlands Institute for Health Services Research, which grew out of general practice but has migrated increasingly toward HSR and a broader, European focus. Nevertheless, it has produced some of the most important research about the strength of primary care across developed countries and relationship to outcomes (NIVEL, 2020). The United Kingdom has a specific focus on PCR within the National Institute for Health Research, which funded 562 primary care studies through its Clinical Research Network in 2018 and 2019; these represented 9.2 percent of the 6,106 studies conducted by the network (NIHR, 2020). Similarly, the Canadian Institutes of Health Research has initiatives focused on community-based primary health care and primary and integrated health care innovations (CIHR, 2020).
FINDINGS AND CONCLUSIONS
Given the data showing that primary care accounts for one-third of all health care visits and the paucity of published PCR, a substantial need is clear for primary care–oriented research that could identify the practices that improve the delivery of high-quality primary care. With few exceptions, the committee has determined that primary care largely depends on
evidence derived from research done in subspecialty care, hospital settings, or among single-disease cohorts, even though primary care PBRNs have famously yielded important insights regarding many health conditions and treatments. While disease-specific knowledge can be useful in informing many aspects of primary care, most clinical trials exclude people with the comorbid conditions, social and medical complexity, and undifferentiated illness that are the norm in primary care. Evidence-based guidelines ignore the complexity of primary care, and the unintended consequences of focusing quality improvement only on narrowly defined evidence are profound.
The neglect of basic PCR, and lack of research that draws on primary care–specific databases, such as clinical registries, not only adversely affects primary care outcomes but also leads to the lack of a population-based understanding of illness and disease along the health care spectrum. However, better support of PCR could lead to answers to questions that are critically important for improving population health, such as how to incorporate data on SDOH into clinical decision making.
This situation is not new, as it was noted in the 1996 IOM report. Nonetheless, 25 years later, current clinical research has little to offer to primary care clinicians. Moreover, lessons from well-done PCR are not available to inform the larger picture of health care organization and delivery. As a result, the paucity of PCR and development leaves primary care insufficiently prepared to confront the challenges and opportunities inherent in this committee’s definition.
An important reason for the neglect PCR suffers is that no federal agency is charged with developing and advancing a robust PCR program and funded to support that mission. In fact, PBRNs continue to struggle to find infrastructure and sustained funding, even though they are increasingly important for frontline health and health equity research. While AHRQ has a federal mandate to conduct PCR, its National Center for Excellence in Primary Care has consistently had no specific research funding and is under constant budgetary threat, limiting its ability to meaningfully contribute to the field. Family medicine, the primary care specialty that accounts for more than 25 percent of all outpatient visits, has consistently received some 0.2 percent of total research and 0.3 percent of all NIH awards. A dedicated office of PCR at NIH could ensure that PCR becomes a more robust part of the NIH portfolio.
In summary, while primary care is the most widely used service in health care, research that could improve its delivery is in need of a significant boost in emphasis and funding. The lack of an agency or office within the federal government whose primary mission is to emphasize, coordinate, and fund research on primary care is a critical impediment to generating the type of knowledge that would benefit both those who provide care in
the primary setting and all Americans who receive most of their care in that same setting.
REFERENCES
AHRQ (Agency for Healthcare Research and Quality). 2012. Primary care practice-based research networks: An AHRQ initiative. https://www.ahrq.gov/research/findings/factsheets/primary/pbrn/index.html (accessed December 23, 2020).
Alston, M., J. Cawse-Lucas, L. S. Hughes, T. Wheeler, and A. Kost. 2019. The persistence of specialty disrespect: Student perspectives. PRiMER 3:1.
Aungst, H., M. Baker, C. Bouyer, B. Catalano, M. Cintron, N. B. Cohen, P. A. Gannon, J. Gilliam, H. Gullett, K. Hassmiller-Lich, S. C. Horner, R. N. Karmali, Ó. B. Kristjánsdóttir, R. Martukovich, J. Mirkovic, J. E. Misak, S. M. Moore, N. Ponyicky, A. Reichsman, M. C. Ruhe, C. Ruland, D. S. Schaadt, K. C. Stange, U. Stenberg, S. A. Sweeney, A. van der Meulen, R. F. Weinberger, J. Williams, and J. Yokie. 2019. Identifying personal strengths to help patients manage chronic illness. Washington, DC: Patient-Centered Outcomes Research Institute (PCORI).
Basílio, N., C. Ramos, S. Figueira, and D. Pinto. 2016. Worldwide usage of international classification of primary care use. Revista Brasileira de Medicina de Família e Comunidade 11:1.
Beach, M. C., and T. Inui. 2006. Relationship-centered care. A constructive reframing. Journal of General Internal Medicine 21(Suppl 1):S3–S8.
Borkan, J. M. 2004. Mixed methods studies: A foundation for primary care research. Annals of Family Medicine 2(1):4–6.
Brown, J., M. Stewart, E. McCracken, I. R. McWhinney, and J. Levenstein. 1986. The patient-centered clinical method. 2. Definition and application. Family Practice 3(2):75–79.
Brownson, R. C., G. A. Colditz, and E. K. Proctor, eds. 2012. Dissemination and implementation research in health: Translating science to practice. New York: Oxford University Press.
CAFM (Council of Academic Family Medicine). 2019. Fund AHRQ’s primary care research center. Washington, DC: Council of Academic Family Medicine.
Cameron, B. J., A. W. Bazemore, and C. P. Morley. 2016. Lost in translation: NIH funding for family medicine research remains limited. Journal of the American Board of Family Practice 29(5):528–530.
Camillus, J. C. 2008. Strategy as a wicked problem. https://hbr.org/2008/05/strategy-as-a-wicked-problem (accessed November 18, 2020).
Casalino, L. P. 1999. The unintended consequences of measuring quality on the quality of medical care. New England Journal of Medicine 341(15):1147–1150.
Cebul, R. D., J. B. Rebitzer, L. J. Taylor, and M. E. Votruba. 2008. Organizational fragmentation and care quality in the U.S. healthcare system. Journal of Economic Perspectives 22(4):93–113.
Checkland, K. 2007. Understanding general practice: A conceptual framework developed from case studies in the UK NHS. British Journal of General Practice 57(534):56–63.
CIHR (Canadian Institutes of Health Research). 2020. About us. https://cihr-irsc.gc.ca/e/37792.html (accessed November 11, 2020).
Cohen, D. J., B. F. Crabtree, R. S. Etz, B. A. Balasubramanian, K. Donahue, L. C. Leviton, E. C. Clark, N. F. Isaacson, K. C. Stange, and L. W. Green. 2008. Fidelity versus flexibility: Translating evidence-based research into practice. American Journal of Preventive Medicine 35:S381–S389.
Cottrell, E. K., R. Gold, S. Likumahuwa, H. Angier, N. Huguet, D. J. Cohen, K. D. Clark, L. M. Gottlieb, and J. E. DeVoe. 2018. Using health information technology to bring social determinants of health into primary care: A conceptual framework to guide research. Journal of Health Care for the Poor and Underserved 29(3):949–963.
Crabtree, B. F., and W. L. Miller, eds. 1999. Doing qualitative research. 2nd ed. Thousand Oaks, CA: Sage Publications.
Creighton, S. 2013. “Why waste a medical education on primary care?” Op-Med. https://opmed.doximity.com/articles/why-waste-a-medical-education-on-primary-care-edc497ed-fc19-4ddf-be32-a86533975a21 (accessed December 22, 2020).
Creswell, J. W., and M. Hirose. 2019. Mixed methods and survey research in family medicine and community health. Family Medicine and Community Health 7(2):e000086.
Davis, D. A., and A. Taylor-Vaisey. 1997. Translating guidelines into practice. A systematic review of theoretic concepts, practical experience and research evidence in the adoption of clinical practice guidelines. Canadian Medical Association Journal 157(4):408–416.
De Maeseneer, J. M., M. L. van Driel, L. A. Green, and C. van Weel. 2003. The need for research in primary care. The Lancet 362(9392):1314–1319.
deGruy, F. V., B. Ewigman, J. E. DeVoe, L. Hughes, P. James, F. D. Schneider, J. Hickner, K. Stange, T. Van Fossen, A. J. Kuzel, and R. Mullen. 2015. A plan for useful and timely family medicine and primary care research. Family Medicine 47(8):636–642.
DeVoe, J. E., and A. Sears. 2013. The Ochin Community Information Network: Bringing together community health centers, information technology, and data to support a patient-centered medical village. Journal of the American Board of Family Practice 26(3):271–278.
DeVoe, J. E., A. W. Bazemore, E. K. Cottrell, S. Likumahuwa-Ackman, J. Grandmont, N. Spach, and R. Gold. 2016. Perspectives in primary care: A conceptual framework and path for integrating social determinants of health into primary care practice. Annals of Family Medicine 14(2):104–108.
Donner-Banzhoff, N. 2018. Solving the diagnostic challenge: A patient-centered approach. Annals of Family Medicine 16(4):353–358.
Eberl, M. M., R. L. Phillips, Jr., H. Lamberts, I. Okkes, and M. C. Mahoney. 2008. Characterizing breast symptoms in family practice. Annals of Family Medicine 6(6):528–533.
Epstein, R. M., C. G. Shields, S. C. Meldrum, K. Fiscella, J. Carroll, P. A. Carney, and P. R. Duberstein. 2006. Physicians’ responses to patients’ medically unexplained symptoms. Psychosomatic Medicine 68(2):269–276.
Fagnan, L. J., M. Davis, R. A. Deyo, J. J. Werner, and K. C. Stange. 2010. Linking practice-based research networks and clinical and translational science awards: New opportunities for community engagement by academic health centers. Academic Medicine 85(3):476–483.
Fortin, M., L. Lapointe, C. Hudon, and A. Vanasse. 2005. Multimorbidity is common to family practice: Is it commonly researched? Canadian Family Physician 51:244–245.
Fortin, M., J. Dionne, G. Pinho, J. Gignac, J. Almirall, and L. Lapointe. 2006. Randomized controlled trials: Do they have external validity for patients with multiple comorbidities? Annals of Family Medicine 4(2):104–108.
Frechtling, J., K. Raue, J. Michie, A. Miyaoka, and M. Spiegelman. 2012. The CTSA national evaluation final report. Rockville, MD: Westat.
Gaglioti, A. H., J. J. Werner, G. Rust, L. J. Fagnan, and A. V. Neale. 2016. Practice-based research networks (PBRNs) bridging the gaps between communities, funders, and policymakers. Journal of the American Board of Family Medicine 29(5):630–635.
Galvin, R. 2006. Pay-for-performance: Too much of a good thing? A conversation with Martin Roland. Interview by Robert Galvin. Health Affairs 25(5):w412–w419.
Gerard, K., C. Salisbury, D. Street, C. Pope, and H. Baxter. 2008. Is fast access to general practice all that should matter? A discrete choice experiment of patients’ preferences. Journal of Health Services Research and Policy 13(2):3–10.
Gold, R., E. Cottrell, A. Bunce, M. Middendorf, C. Hollombe, S. Cowburn, P. Mahr, and G. Melgar. 2017. Developing electronic health record (EHR) strategies related to health center patients’ social determinants of health. Journal of the American Board of Family Practice 30(4):428–447.
Goldstein, K. M., D. Vogt, A. Hamilton, S. M. Frayne, J. Gierisch, J. Blakeney, A. Sadler, B. M. Bean-Mayberry, D. Carney, B. DiLeone, A. B. Fox, R. Klap, E. Yee, Y. Romodan, H. Strehlow, J. Yosef, and E. M. Yano. 2018. Practice-based research networks add value to evidence-based quality improvement. Healthcare 6(2):128–134.
Grant, R. W., J. M. Ashburner, C. C. Hong, Y. Chang, M. J. Barry, and S. J. Atlas. 2011. Defining patient complexity from the primary care physician’s perspective. Annals of Internal Medicine 155(12):797–804.
Green, L. A., and M. Klinkman. 2015. Perspectives in primary care: The foundational urgent importance of a shared primary care data model. Annals of Family Medicine 13(4):303–311.
Green, L. A., and L. J. Lutz. 1990. Notions about networks: Primary care practices in pursuit of improved primary care. In Primary care research: An agenda for the 90s, edited by J. Mayfield and M. L. Grady. Washington, DC: Agency for Health Care Policy and Research. Pp. 125–132.
Greenhalgh, T. 2007. Primary health care: Theory and practice. Malden, MA: Blackwell/ BMJ Books.
Greenhalgh, T., and C. Papoutsi. 2018. Studying complexity in health services research: Desperately seeking an overdue paradigm shift. BMC Medicine 16(1):1–6.
Hall-Lipsy, E., L. Barraza, and C. Robertson. 2018. Practice-based research networks and the mandate for real-world evidence. American Journal of Law & Medicine 44(2-3):219–236.
Heath, I. 1995. The mystery of general practice. London, UK: Nuffield Provincial Hospitals Trust.
Heath, I. 2006. Patients are not commodities. BMJ 332(7545):846–847.
Hickner, J., and L. A. Green. 2015. Practice-based research networks (PBRNs) in the United States: Growing and still going after all these years. Journal of the American Board of Family Practice 28(5):541–545.
Hobbs, R. 2019. Is primary care research important and relevant to GPs? British Journal of General Practice 69(686):424–425.
Homa, L., J. Rose, P. S. Hovmand, S. T. Cherng, R. L. Riolo, A. Kraus, A. Biswas, K. Burgess, H. Aungst, K. C. Stange, K. Brown, M. Brooks-Terry, E. Dec, B. Jackson, J. Gilliam, G. E. Kikano, A. Reichsman, D. Schaadt, J. Hilfer, C. Ticknor, C. V. Tyler, A. Van der Meulen, H. Ways, R. F. Weinberger, and C. Williams. 2015. A participatory model of the paradox of primary care. Annals of Family Medicine 13(5):456–465.
Hughes, J. R., R. Grayson, and F. C. Stiles. 1977. Fragmentation of care and the medical home. Pediatrics 60(4):559.
IOM (Institute of Medicine). 1996. Primary care: America’s health in a new era. Washington, DC: National Academy Press.
IOM. 2007a. Emergency care for children: Growing pains. Washington, DC: The National Academies Press.
IOM. 2007b. Emergency medical services: At the crossroads. Washington, DC: The National Academies Press.
IOM. 2007c. Hospital-based emergency care: At the breaking point. Washington, DC: The National Academies Press.
Johansen, M. E., S. M. Kircher, and T. R. Huerta. 2016. Reexamining the ecology of medical care. New England Journal of Medicine 374(5):495–496.
Krist, A. H., J. W. Beasley, J. C. Crosson, D. C. Kibbe, M. S. Klinkman, C. U. Lehmann, C. H. Fox, J. M. Mitchell, J. W. Mold, W. D. Pace, K. A. Peterson, R. L. Phillips, R. Post, J. Puro, M. Raddock, R. Simkus, and S. E. Waldren. 2014. Electronic health record functionality needed to better support primary care. Journal of the American Medical Informatics Association 21(5):764–771.
Krist, A. H., L. A. Green, R. L. Phillips, J. W. Beasley, J. E. DeVoe, M. S. Klinkman, J. Hughes, J. Puro, C. H. Fox, T. Burdick, and the NAPCRG Health Information Technology Working Group. 2015. Health information technology needs help from primary care researchers. Journal of the American Board of Family Medicine 28(3):306–310.
Lavery, A. M., L. E. Preston, J. Y. Ko, J. R. Chevinsky, C. L. DeSisto, A. F. Pennington, L. Kompaniyets, S. D. Datta, E. S. Click, T. Golden, A. B. Goodman, W. R. Mac Kenzie, T. K. Boehmer, and A. V. Gundlapalli. 2020. Characteristics of hospitalized COVID-19 patients discharged and experiencing same-hospital readmission—United States, March–August 2020. Morbidity and Mortality Weekly Report 69(45).
Lecky, D. M., S. Granier, R. Allison, N. Q. Verlander, S. M. Collin, and C. A. M. McNulty. 2020. Infectious disease and primary care research—what English general practitioners say they need. Antibiotics (Basel) 9(5).
Liaw, W., A. Eden, M. Coffman, M. Nagaraj, and A. Bazemore. 2019. Factors associated with successful research departments: A qualitative analysis of family medicine research bright spots. Family Medicine 51(2):87–102.
Lipsitz, L. A. 2012. Understanding health care as a complex system: The foundation for unintended consequences. JAMA 308(3):243–244.
Litaker, D., A. Tomolo, V. Liberatore, K. C. Stange, and D. C. Aron. 2006. Using complexity theory to build interventions that improve health care delivery in primary care. Journal of General Internal Medicine 21(Suppl 2):S30–S34.
Lown, B. 2007. The commodification of health care. PNHP Newsletter (Spring):40–44.
Lucan, S. C., F. K. Barg, A. W. Bazemore, and R. L. Phillips, Jr. 2009. Family medicine, the NIH, and the medical-research roadmap: Perspectives from inside the NIH. Family Medicine 41(3):188–196.
Macaulay, A. C., and P. A. Nutting. 2006. Moving the frontiers forward: Incorporating community-based participatory research into practice-based research networks. Annals of Family Medicine 4(1):4–7.
Macaulay, A. C., L. E. Commanda, W. L. Freeman, N. Gibson, M. L. McCabe, C. M. Robbins, and P. L. Twohig. 1999. Participatory research maximises community and lay involvement. North American primary care research group. BMJ 319(7212):774–778.
Martin, C., and J. Sturmberg. 2009. Complex adaptive chronic care. Journal of Evaluation in Clinical Practice 15(3):571–577.
Mazur, S., A. Bazemore, and D. Merenstein. 2016. Characteristics of early recipients of Patient-Centered Outcomes Research Institute funding. Academic Medicine 91(4):491–496.
Mazzone, M., N. Bhuyan, G. M. Dickson, J. W. Jarvis, L. Maxwell, W. F. Miser, K. Mitchell, S. Schultz, T. Shaffer, and M. Tuggy. 2015. A prescription to advocate for graduate medical education reform. Annals of Family Medicine 13(2):184–185.
McDonald, R., and M. Roland. 2009. Pay for performance in primary care in England and California: Comparison of unintended consequences. Annals of Family Medicine 7(2):121–127.
McWhinney, I. R. 1972. Beyond diagnosis: An approach to the integration of behavioral science and clinical medicine. New England Journal of Medicine 287(8):384–387.
McWhinney, I. R. 1977. The naturalist tradition in general practice. Journal of Family Practice 5(3):375–378.
Mendel, P., C. A. Gidengil, A. Tomoaia-Cotisel, S. Mann, A. J. Rose, K. J. Leuschner, N. S. Qureshi, V. Kareddy, J. L. Sousa, and D. Kim. 2020a. Health services and primary care research study: Comprehensive report. Santa Monica, CA: RAND Corporation.
Mendel, P., C. A. Gidengil, A. Tomoaia-Cotisel, S. Mann, A. J. Rose, K. J. Leuschner, N. S. Qureshi, V. Kareddy, J. L. Sousa, and D. Kim. 2020b. Investing in the future of health care: A strategic assessment of federally funded health services research and primary care research. Santa Monica, CA: RAND Corporation.
Méndez, F. 2015. Transdiscipline and research in health: Science, society and decision making. Colombia Médica 46(3):128–134.
Miller, W. L., B. F. Crabtree, P. A. Nutting, K. C. Stange, and C. R. Jaén. 2010. Primary care practice development: A relationship-centered approach. Annals of Family Medicine 8(Suppl 1):S68–S79, S92.
Mold, J. W., and D. M. Thompson. 2004. Management of brown recluse spider bites in primary care. Journal of the American Board of Family Practice 17(5):347–352.
Muche-Borowski, C., D. Lühmann, I. Schäfer, R. Mundt, H. O. Wagner, M. Scherer, and the Guideline Group of the German College of General Practice and Family Medicine (DEGAM). 2017. Development of a meta-algorithm for guiding primary care encounters for patients with multimorbidity using evidence-based and case-based guideline development methodology. BMJ Open 7(6):e015478.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2018a. Crossing the global quality chasm: Improving health care worldwide. Washington, DC: The National Academies Press.
NASEM. 2018b. The future of health services research: Advancing health systems research and practice in the United States. Washington, DC: The National Academies Press.
NCI (National Cancer Institute). 2019. NCI-designated cancer centers. https://www.cancer.gov/research/infrastructure/cancer-centers (accessed December 14, 2020).
NIH (National Institutes of Health). 2019. OECR history. https://www.ninds.nih.gov/CurrentResearch/Trans-Agency-Activities/Office-Emergency-Care-Research/OECR-History (accessed November 3, 2020).
NIH. 2020a. NIH National Research Service Award (NRSA) fellowships. https://www.nlm.nih.gov/ep/NRSAFellowshipGrants.html (accessed November 11, 2020).
NIH. 2020b. Ruth L. Kirschstein Institutional National Research Service Award. https://researchtraining.nih.gov/programs/training-grants/T32 (accessed November 11, 2020).
NIHR (National Institute for Health Research). 2020. Primary care. https://www.nihr.ac.uk/explore-nihr/specialties/primary-care.htm (accessed November 11, 2020).
NIVEL (Netherlands Institute for Health Services Research). 2020. QUALICOPC. https://www.nivel.nl/en/international-projects/qualicopc (accessed November 11, 2020).
NLM (National Library of Medicine). 2020. ICPC (International Classification of Primary Care)—synopsis. https://www.nlm.nih.gov/research/umls/sourcereleasedocs/current/ICPC/index.html (accessed June 2, 2020).
NRC (National Research Council). 2015. Enhancing the effectiveness of team science. Washington, DC: The National Academies Press.
Nutting, P. A., and L. A. Green. 1994. Practice-based research networks: Reuniting practice and research around the problems most of the people have most of the time. Journal of Family Practice 38(4):335–336.
Okkes, I. M., S. K. Oskam, and H. Lamberts. 2002a. The probability of specific diagnoses for patients presenting with common symptoms to Dutch family physicians. Journal of Family Practice 51(1):31–36.
Okkes, I. M., G. O. Polderman, G. E. Fryer, T. Yamada, M. Bujak, S. K. Oskam, L. A. Green, and H. Lamberts. 2002b. The role of family practice in different health care systems: A comparison of reasons for encounter, diagnoses, and interventions in primary care populations in the Netherlands, Japan, Poland, and the United States. Journal of Family Practice 51(1):72–73.
ONC (Office of the National Coordinator for Health Information Technology). 2019. Office-based physician electronic health record adoption. dashboard.healthit.gov/quickstats/pages/physician-ehr-adoption-trends.php (accessed December 8, 2020).
Parekh, A. K., R. A. Goodman, C. Gordon, and H. K. Koh. 2011. Managing multiple chronic conditions: A strategic framework for improving health outcomes and quality of life. Public Health Reports 126(4):460–471.
Peek, C. J., M. A. Baird, and E. Coleman. 2009. Primary care for patient complexity, not only disease. Families, Systems, & Health 27(4):287–302.
Peek, C. J., R. E. Glasgow, K. C. Stange, L. M. Klesges, E. P. Purcell, and R. S. Kessler. 2014. The 5 R’s: An emerging bold standard for conducting relevant research in a changing world. Annals of Family Medicine 12(5):447–455.
Petterson, S., B. F. Miller, J. C. Payne-Murphy, and R. L. Phillips Jr. 2014. Mental health treatment in the primary care setting: Patterns and pathways. Families, Systems, & Health 32(2):157–166.
PH3C (Primary Health Care Classification Consortium). 2011. Members. http://www.ph3c.org/4daction/w3_CatVisu/?wCatFonc=membres&chMembreNom=&chMembrePrenom =&chMembreAnnee=&chMembreActif=&aff=liste&wNbEnrPage=20&wNumPage=1& wCritTri=3 (accessed June 2, 2020).
Phillips, R. L., Jr., M. Klinkman, and L. A. Green. 2007a. Conference report: Harmonizing primary care clinical classification and data standards. Washington, DC: Robert Graham Center and Agency for Healthcare Research and Quality.
Phillips, R. L., Jr., J. Mold, and K. Peterson. 2007b. Research-based research networks. In The learning healthcare system: Workshop summary, edited by L. Olsen, D. Aisner, and J. M. McGinnis. Washington, DC: The National Academies Press.
Phillips, R. L., Jr., A. W. Bazemore, J. E. DeVoe, T. J. Weida, A. H. Krist, M. F. Dulin, and F. E. Biagioli. 2015. A family medicine health technology strategy for achieving the triple aim for US health care. Family Medicine 47(8):628–635.
Riley-Behringer, M., M. M. Davis, J. J. Werner, L. J. Fagnan, and K. C. Stange. 2017. The evolving collaborative relationship between practice-based research networks (PBRNs) and clinical and translational science awardees (CTSAs). Journal of Clinical and Translational Science 1(5):301–309.
Robinson, K., and J. Westfall. 2011. NAPCRG puts the increase of primary care research funding at the top of the priority list. Annals of Family Medicine 9(5):468–469.
Ronis, S. D., R. Grossberg, R. Allen, A. Hertz, and L. C. Kleinman. 2019. Estimated non-reimbursed costs for care coordination for children with medical complexity. Pediatrics 143(1).
Rotenstein, L. S., M. Torre, M. A. Ramos, R. C. Rosales, C. Guille, S. Sen, and D. A. Mata. 2018. Prevalence of burnout among physicians: A systematic review. JAMA 320(11):1131–1150.
Rudebeck, C. E. 2019. Relationship based care—how general practice developed and why it is undermined within contemporary healthcare systems. Scandinavian Journal of Primary Health Care 37(3):335–344.
Rui, P., and T. Okeyode. 2016. National Ambulatory Medical Care Survey: 2016 national summary tables. Atlanta, GA: Centers for Disease Control and Prevention.
Shippee, N. D., T. P. Shippee, P. D. Mobley, K. M. Fernstrom, and H. R. Britt. 2018. Effect of a whole-person model of care on patient experience in patients with complex chronic illness in late life. American Journal of Hospice & Palliative Care 35(1):104–109.
Slawson, D. C., A. F. Shaughnessy, and H. Barry. 2001. Which should come first: Rigor or relevance? Journal of Family Practice 50(3):209–210.
Soler, J. K., I. Okkes, M. Wood, and H. Lamberts. 2008. The coming of age of ICPC: Celebrating the 21st birthday of the international classification of primary care. Family Practice 25(4):312–317.
Stange, K. C. 2009a. The generalist approach. Annals of Family Medicine 7(3):198–203.
Stange, K. C. 2009b. The problem of fragmentation and the need for integrative solutions. Annals of Family Medicine 7(2):100–103.
Stange, K. C., and S. J. Zyzanski. 1989. Integrating qualitative and quantitative research methods. Family Medicine 21(6):448–451.
Stange, K. C., W. L. Miller, B. F. Crabtree, P. J. O’Connor, and S. J. Zyzanski. 1994. Multimethod research: Approaches for integrating qualitative and quantitative methods. Journal of General Internal Medicine 9(5):278–282.
Stange, K. C., W. L. Miller, and I. McWhinney. 2001. Developing the knowledge base of family practice. Family Medicine 33(4):286–297.
Stange, K. C., S. T. Cherng, R. L. Riolo, L. Homa, J. Rose, P. S. Hovmand, and A. Kraus. 2017. No longer looking just under the lamp post: Modeling the complexity of primary health care. In Growing inequality: Bridging complex systems, population health, and health disparities, edited by G. A. Kaplan, A. V. Diez Roux, C. P. Simon, and S. Galea. Washington, DC: Westphalia Press. Pp. 81–107.
Starfield, B. 1992. Primary care: Concept, evaluation, and policy. New York: Oxford University Press.
Starfield, B. 1996. A framework for primary care research. Journal of Family Practice 42(2):181–185.
Starfield, B. 1998. Primary care: Balancing health needs, services, and technology. Revised edition. New York: Oxford University Press.
Starfield, B. 2001. New paradigms for quality in primary care. British Journal of General Practice 51(465):303–309.
Sutton, M., R. Elder, B. Guthrie, and G. Watt. 2009. Record rewards: The effects of targeted quality incentives on the recording of risk factors by primary care providers. Health Economics 19(1):1–13.
Thomas, H., G. Mitchell, J. Rich, and M. Best. 2018. Definition of whole person care in general practice in the English language literature: A systematic review. BMJ Open 8(12):e023758.
Tinetti, M. E., A. R. Green, J. Ouellet, M. W. Rich, and C. Boyd. 2019. Caring for patients with multiple chronic conditions. Annals of Internal Medicine 170(3):199–202.
Tuggy, M., N. Bhuyan, G. M. Dickson, J. W. Jarvis, L. Maxwell, M. Mazzone, W. F. Miser, K. Mitchell, S. Schultz, and T. Shaffer. 2015. Training implications of family medicine for America’s health: A preview. Annals of Family Medicine 13(1):91–92.
USYD (University of Sydney). 2020. Bettering the evaluation and care of health (BEACH). https://www.sydney.edu.au/medicine-health/our-research/research-centres/bettering-theevaluation-and-care-of-health.html (accessed June 2, 2020).
van Boven, K., A. A. Uijen, N. van de Wiel, S. K. Oskam, H. J. Schers, and W. J. J. Assendelft. 2017. The diagnostic value of the patient’s reason for encounter for diagnosing cancer in primary care. Journal of the American Board of Family Medicine 30(6):806–812.
Ward, B. W., and J. S. Schiller. 2013. Prevalence of multiple chronic conditions among us adults: Estimates from the National Health Interview Survey, 2010. Preventing Chronic Disease 10:E65.
Westfall, J. M., J. Mold, and L. Fagnan. 2007. Practice-based research—“blue highways” on the NIH roadmap. JAMA 297(4):403–406.
Westfall, J. M., L. J. Fagnan, M. Handley, J. Salsberg, P. McGinnis, L. K. Zittleman, and A. C. Macaulay. 2009. Practice-based research is community engagement. Journal of the American Board of Family Practice 22(4):423–427.
Westfall, J. M., L. Zittleman, E. W. Staton, B. Parnes, P. C. Smith, L. J. Niebauer, D. H. Fernald, J. Quintela, R. F. Van Vorst, L. M. Dickinson, and W. D. Pace. 2011. Card studies for observational research in practice. Annals of Family Medicine 9(1):63–68.
Westfall, J. M., R. Roper, A. Gaglioti, and D. E. Nease, Jr. 2019. Practice-based research networks: Strategic opportunities to advance implementation research for health equity. Ethnicity & Disease 29(Suppl 1):113–118.
























