2
The Science of Human Neural Organoids, Transplants, and Chimeras
Neurological and psychiatric disorders take a tremendous toll (Gooch et al., 2017). Together they represent the leading cause of morbidity worldwide and account for substantial mortality. Their lifetime prevalence in the aggregate exceeds 30 percent of the population. The suffering they cause is enormous; indeed, two of the five disabilities Americans most fear are Alzheimer’s disease and irreversible blindness (Scott et al., 2016), both of which result from loss of neurons. As discussed in Chapter 1, their economic impact, in the United States and globally, is massive as well in terms of both direct health care costs and indirect costs, such as lost productivity (Trautmann et al., 2016). And their incidence profile is devastating: Psychiatric diseases, which lead to lifelong disability, typically manifest by the early 20s, while lethal neurodegenerative diseases are age-related and are therefore likely to increase two-fold or more in the next few decades as the population ages.
Given the frequency and burden of brain disorders, a large community of researchers is working to understand and treat them. Progress has been slow, however, for at least three reasons. First, the brain is by far the most complex of human organs, with nearly 100 billion neurons (plus an even larger number of glial cells) comprising thousands of distinct types, interconnected in complex circuits, with some neurons making or receiving thousands of synaptic connections (see Box 2-1). Second, tools needed to probe these circuits—such as methods with which to measure activity from hundreds to thousands of neurons at the same time, to map connectivity in comprehensive ways, or to characterize the molecular differences among neuronal types or between normal and dysfunctional neurons—are still being developed. Third, while small animal and cell culture models of human disease have been extremely valuable, their limitations have been widely recognized and are particularly acute for brain disorders (Sierksma et al., 2020). Brains and brain cells of mice and rats, the most com-
monly used model species, differ from those of humans with respect to their size, structure, molecular composition, and pharmacological responses (Hodge et al., 2019). Furthermore, mice are most useful in modeling diseases that are caused by mutations in single genes, which can be manipulated in the mouse genome. In contrast, most prevalent human brain diseases are polygenic, meaning that their genetic underpinnings result from the combined effects of many genomic variants; at present, these complex genotypes cannot be replicated in model organisms (Hyman, 2018; Quadrato et al., 2016). Another limitation is that some neural disorders are likely due to defects in brain regions that are difficult to study in mice. For example, the prefrontal cortex, which plays a key role in executive function, is extremely underdeveloped in mice relative to humans (Wise, 2008); and the leading cause of irreversible blindness in the United States—age-related
macular degeneration—results from loss of light-sensitive neurons in a structure called the macula that is completely absent in mice (Bringmann et al., 2018). There are also many cell types that are unique to primates or differ in gene expression between rodents and primates (see, e.g., Oberheim et al., 2009; Krienen et al., 2020). These and other differences limit the value of model organisms for research aimed at understanding the human brain, and are likely to account at least in part for the frequent failure of potential therapies developed in current animal neurological disease models to translate effectively to humans (King, 2018; Sierksma et al., 2020). The problem is more dire still for psychiatric diseases. Such disorders as autism and schizophrenia are characterized and defined by disruptions of behaviors that may not be present and certainly cannot be adequately measured in mice (Pankevich et al., 2014).
Responding to the limitations of animal models, scientists have developed several ways to analyze the human brain directly. Several methods, including magnetic resonance imaging (MRI), functional MRI (fMRI), and positron emission tomography (PET), are noninvasive, allowing studies of the structure, function, and in some cases molecular composition of the human brain in awake, living individuals (Filippi, 2015). However, the spatial resolution of these methods does not allow analysis at the cellular level, and the temporal resolution is 100-fold less than that needed to capture key neural signals. Moreover, even when these modalities can detect markers of disease or disease progression, protections afforded to human subjects limit the ability to test interventions or potential therapies in human beings. Higher resolution is provided using living brain tissue removed during surgery (often for intractable epilepsy) or obtained postmortem. Slices of such ex vivo brain tissue have been used for analyses of neural activity and molecular composition of defined regions and cell types but are severely limited in quality and quantity. Moreover, when they come from patients with neurological diseases, conclusions drawn from them may not be applicable to normal subjects or to patients with other diseases. Researchers have
also generated cultures from human neurons or stem cells induced to form neurons (discussed further below), but these two-dimensional cultures fail to form stereotyped, complex circuits.
Experimental models of the human brain are therefore needed. As discussed in Chapter 1, three sets of model systems have been developed that allow scientists to analyze human neural cells in powerful new ways: neural organoids, neural cell transplants, and neural chimeras. In this report we refer to these three systems according to the following definitions:
- Neural organoids are three-dimensional cultures derived from pluripotent stem cells that have been treated in ways that lead them to generate neurons and glia (see Figure 1-1 in Chapter 1). Organoids can contain multiple neuronal and glial cell types and, unlike classical neuronal cultures, exhibit complex synaptic interactions among types. They represent an important complement to conventional “monolayer” cell cultures and animal models.
- Neural cell transplants. Neural cells or, in some cases, neural organoids, can be transplanted directly into the brain of a nonhuman animal, either
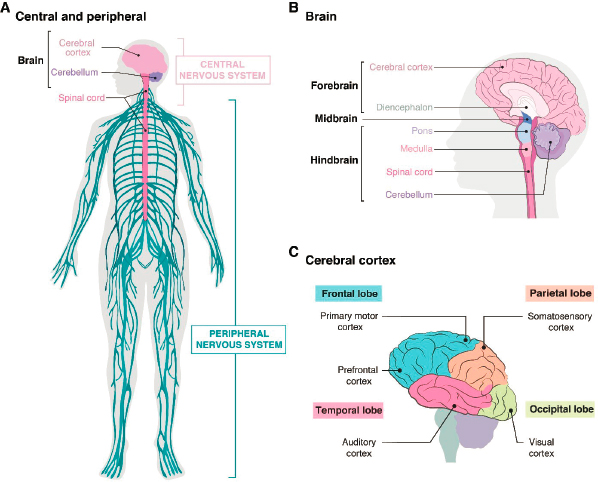
IMAGE SOURCE: Maria Diaz de la Loza, Ph.D.
- during development or in adulthood. They are examples of “xenotrans-plants,” a term that refers to insertion of cells from one species into a host of another species. Transplanted neuronal precursors can mature into functional neurons and integrate into the host nervous system, receiving synaptic inputs from and providing synaptic input to host neurons. Transplanted glia can also interact with host cells. Cells in neural cell transplants vary in where and how they interact with the recipient’s brain, but they seldom if ever contribute to other host tissues or organs.
- Neural chimeras are a form of transplant in which donor cells are introduced into the nonhuman animal at an embryonic stage prior to formation of the nervous system. The donor cells can therefore develop in parallel with host cells, enabling high levels of integration. In some cases, host cells of particular types are ablated by genetic methods, so donor cells injected at a very early stage (the blastocyst) can provide the major contribution to a tissue or organ. This method is called blastocyst complementation.
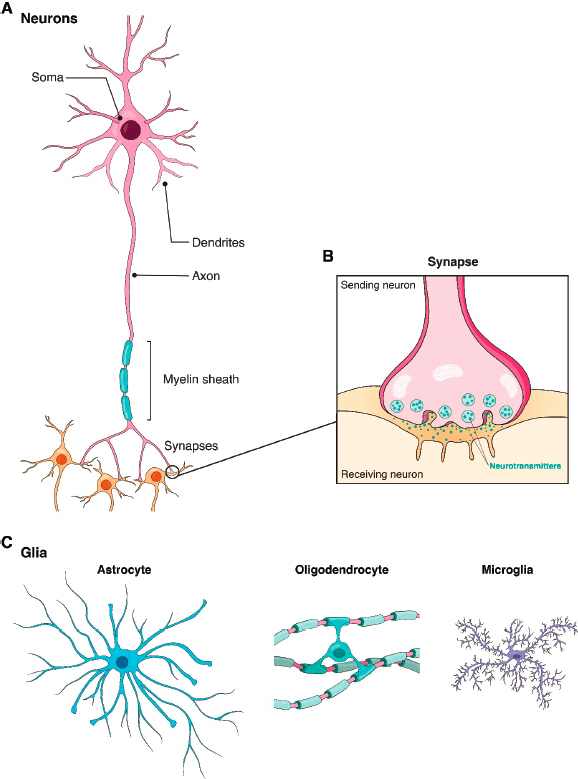
IMAGE SOURCE: Maria Diaz de la Loza, Ph.D.
The distinction between a neural cell transplant and a neural chimera rests largely on whether the introduced cells remain limited to the nervous system or contribute substantially to other organs. This distinction is not always clear-cut in practice. A useful dividing line at present is the gastrula stage of embryogenesis, at which cells are fated to generate specific organs. In general, scientists in the field view introduction of exogenous cells to an organism prior to gastrulation as generating chimeras and after gastrulation as generating transplants.
Organoids and in vivo animal models that are derived from or incorporate human neural cells have raised ethical and philosophical concerns, based in part on the use of human material and in part on the capabilities they might acquire. As the methods become more powerful, these concerns will intensify. The remainder of this chapter surveys the current state of these technologies and considers likely improvements over the next several years. Because many concerns about these models arise from issues related to consciousness, awareness, or sentience, the discussion includes a review of current understanding of those capacities and methods for assessing or monitoring them. Ethical and moral issues are discussed in Chapter 3, and regulatory and oversight mechanisms in Chapter 4.
HUMAN NEURAL ORGANOIDS
Organoids are three-dimensional cell cultures in which multiple cell types are arranged in patterns that recapitulate some features of the corresponding organ in vivo. They are usually derived from stem cells that have the potential to mature into many types of cells, such as embryonic stem cells (ESCs) or induced pluripotent stem cells (iPSCs) (see Figure 2-3). ESCs are obtained from early embryos, at a time when cells are still totipotent—that is, capable of differentiating into any cell type. They are maintained in culture using methods that allow them to retain this capacity. When rodent ESCs are injected into the blastocyst of the same species, they can give rise to a complete embryo. iPSCs are obtained from postnatal specimens, usually skin or blood. Cells are treated with a cocktail of factors that lead them to differentiate into a pluripotent state. They can then be used as an alternative to ESCs (see Takahashi and Yamanaka, 2006). To generate organoids, ESCs or iPSCs are cultured under conditions that promote their aggregation, growth, and differentiation into multiple cell types and the “self-assembly” of these types into structures that display features of an organ. Expanding on early attempts to generate more complex cultures (see, e.g., Zhang et al., 2001; Watanabe et al., 2007), Eiraku and colleagues (2008) used the method to generate aggregates with features characteristic of forebrain structures. This strategy was soon applied to generate organoids resembling many other tissues (Clevers, 2016; Eiraku et al., 2011; Sato et al., 2009).
Neural organoids, in which cells are predominantly if not entirely neural (i.e., neuronal or glial), are typically classified into two main groups (see Figure 1-1). In one, called self-patterning or whole-brain organoids, cells take on identities typical of multiple brain regions. They have the advantage that interactions normally occurring among regions can in principle be analyzed in a single structure. On the other hand, their organization differs vastly from that of any particular part of an actual brain, and they display high levels of organoid-to-organoid variability. In the other group, called prepatterned organoids, cells are directed to generate cells typical of specific, restricted brain regions. This class of organoid was pioneered by Sasai and colleagues, who showed that supplementing the
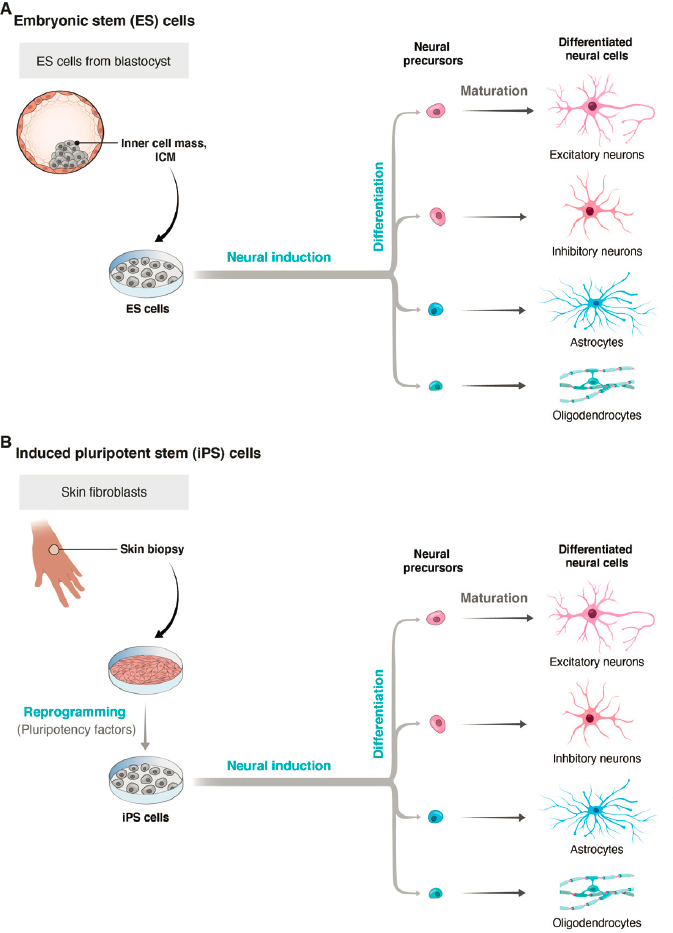
IMAGE SOURCE: Maria Diaz de la Loza, Ph.D.
media with agents known to regulate neural development in vivo and including extracellular matrix components (for better scaffolding) (Ueno, et al., 2006) could influence the identity of the structures that formed—for example, causing them to resemble distinct parts of the forebrain. Later, protocols were developed to generate organoids with some characteristics of other regions, including striatum, retina, thalamus, and spinal cord (Andersen et al., 2020; Cowan et al., 2020; Kadoshima et al., 2013; Miura et al., 2020; Velasco et al., 2019).
For self-organizing cortical organoids, pioneering work was reported by Lancaster and colleagues (2013), who devised standardized methods for generating organoids from iPSCs and demonstrated the self-organizing capacity of whole-brain organoids. Building on this work, many groups have improved the method to extend culture times (to more than 9 months) and to recapitulate key features of brain development, such as neurogenesis from progenitor zones, patterns of gene activity, migration of specific cell types, aspects of neural circuitry, and generation of spontaneous and induced electrical activity (Le Bail et al., 2020; Pasca, 2018; Quadrato et al., 2017). Using patient-derived iPSCs and stem cells engineered to carry mutations associated with human diseases, organoids have provided insight into several human diseases (see Figure 2-4).
For example, Lancaster and colleagues (2013) generated iPSCs from a patient with microcephaly caused by a mutation in a specific gene (CDK5RAP2) and showed that neural organoids derived from that cell line exhibited features characteristic of the disease. Subsequently, researchers used neural organoids to investigate neurodevelopmental changes in individuals with severe idiopathic autism spectrum disorder, elucidating molecular mechanisms that underlie overproduction of inhibitory neurons (Mariani et al., 2015). Cells from individuals with 22q11.2 deletion syndrome (DiGeorge syndrome) were differentiated into neural organoids to identify defects in spontaneous neuronal activity and calcium signaling commonly associated with this syndrome and other genetic forms of the neuropsychiatric disease (Khan et al., 2018). Ye and colleagues (2017) produced organoids from cells donated by a schizophrenia patient with a mutation in a gene called DISC1 (disrupted-in-schizophrenia 1) and documented significant disruptions in cellular processes caused by the mutation. Birey and colleagues (2017) analyzed Timothy syndrome, a devastating neurodevelopmental disorder, in a similar way. Neural organoids have also been valuable in investigating the mechanisms underlying Zika virus (ZIKV)–associated microcephaly in infants, with ZIKV-infected human iPSC-derived neural organoids showing a range of neurodevelopmental abnormalities (Birey et al., 2017; Qian et al., 2017). More recently, these models were used to elucidate the effects of SARS-CoV-2 on the choroid plexus and the blood–brain barrier (Pellegrini et al., 2020).
Organoids differ from the human brain in several significant respects. First, they are small, generally less than 4 mm in diameter, and contain fewer than 2–3 million cells. In contrast, an adult human brain measures approximately 1,350 cubic centimeters and contains some 100 billion cells (neurons and glia)—an
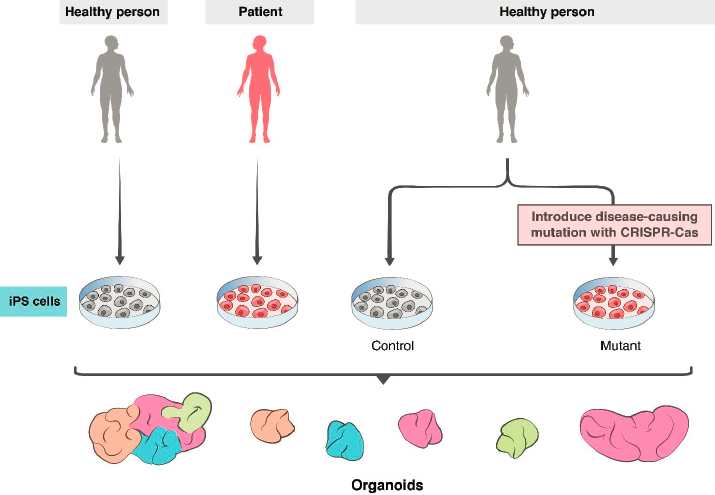
IMAGE SOURCE: Maria Diaz de la Loza, Ph.D.
approximately 40,000-fold difference. One major constraint on growth is that neural organoids do not have blood vessels to supply oxygen and nutrients or to remove metabolic waste, so their size is limited in part by diffusion. Moreover, the hypoxic (oxygen-poor) core can become necrotic. Second, organoids do not recapitulate characteristics of many brain regions, and therefore cannot form the complex networks of connections among regions that typify the brain. Third, at present, circuits that do form lack many features that underlie information processing in the brain. For example, while major cell classes are represented, neural organoids do not display the full diversity of individual cell types found in the brain, nor do they exhibit patterns of organization, lamination, and precise connectivity observed in vivo (Bhaduri et al., 2020; Velasco et al., 2020). Fourth, most neural organoids lack cells of nonneural origin, such as microglia, endothelial cells, and vascular cells, that are critical for brain health and function. Finally,
the neurons, glia, and synapses that form in organoids currently fail to mature beyond those stages typically found in neonatal brains, limiting their utility for analyzing the mature human brain.
Researchers are working to overcome these limitations (Del Dosso et al., 2020). Recent and ongoing efforts include the following:
- Vascularization. Providing organoids with a blood supply would, in principle, give the interior of the organoid access to oxygen and nutrients and enable the elimination of waste products. It is unclear, however, whether vascularization alone would enable growth to a larger size. Vessels are generated from nonneural cells, so they are not present in organoids formed from neural stem cells. Researchers are working to integrate synthetic vasculature into the three-dimensional matrix used for organoid formation (Karzbrun and Reiner, 2019). Alternatively, when neural organoids are implanted into the brains of adult mice, host vessels invade the implant and supply it with nutrients (Mansour et al., 2018). In this case, the organoid and host brain can also become synaptically interconnected.
- Long-range connectivity among brain regions. Connections among multiple brain regions are required for most neural computations in the nervous system and all motor behaviors and perceptions. Currently, many organoids contain a mixture of cells that resemble those found in multiple regions, but without the cellular organization or regional segregation found in the brain. In other cases, organoids are generated under conditions that favor differentiation into structures resembling a single region, such as the cerebral cortex or the retina. In neither case can orderly long-range connections form. As a way to promote orderly and predictable long-range connectivity, an “assembloid” method has been devised in which organoids are grown separately under conditions that promote their acquisition of features characteristic of distinct regions (see Figure 1-1). They are then placed in close proximity to each other, whereupon they form interregional connections (Birey et al., 2017). The most recent studies include combinations of organoids directed to differentiate into aggregates resembling cortex, striatum, spinal cord, and skeletal muscle (Andersen et al., 2020; Miura et al., 2020).
- Individual variation among organoids. Variation among organoids was a vexing problem in early studies, even for organoids generated from the same iPSC line and cultured under seemingly identical conditions. This variability impeded the ability to identify, for example, disease- or patient-specific features. More recently, standardization of conditions has led to a decrease in this variability (Cowan et al., 2020; Velasco et al., 2019; Yoon et al., 2019).
- Use of microfluidic devices (Rifes et al., 2020; Uzel et al., 2016) and specialized materials (Sood et al., 2019). Use of these methods to sup
- port organoid formation enables greater control over the size, uniformity, and patterning of the organoids. As these methods improve, they may also help address issues of reproducibility and generation of higher-order structure.
The major unaddressed limitation is immaturity. Neither neurons nor glia mature to typical adult states in organoids generated to date (Bhaduri et al., 2020). Several groups are using innovative methods to enhance neuronal survival and axon outgrowth and to maintain organoids for longer periods (Giandomenico et al., 2019, 2020), but the inability to model adult patterns of gene expression or connectivity in organoids is a persistent problem. Nonetheless, recent and foreseeable advances hold promise that the use of organoids can advance from modeling disease to providing a platform for testing potential treatments. Already, Esk and colleagues (2020) have been able to screen organoids to identify genes responsible for microcephaly; based on the results of the screen, they elucidated a dysregulated intracellular signaling pathway that led to the disease phenotype. Nonneural organoids are already being used for drug screens (Driehuis et al. 2019; Schuster et al., 2020), with parallel efforts using neural organoids in progress. The future will surely see increased use of iPSCs obtained from adults for whom extensive phenotypic and genotypic data are available; from people with complex polygenic diseases; from children with diseases that are fatal before adulthood; and from patients with diseases in which nonneural symptoms complicate analysis of neurological features. These patient-specific organoids present a unique opportunity for modeling diseases and testing potential treatments (see Figure 2-4).
HUMAN NEURAL TRANSPLANTS
There is a long history of transplanting neural progenitors from one animal into the brain of another. Among the first such efforts were studies aimed at treating Parkinson’s disease. Parkinson’s results from the death of dopaminergic neurons, so several groups proposed implanting dopaminergic precursors or progenitors derived from fetal tissue into patients with the disease to replace those that had been lost (see Figure 1-2 A). Studies in rodent and then nonhuman primate models in the 1970s led to initial clinical trials in patients in the 1980s (Sladek and Gash, 1988). Although efficacy was insufficient for expanded use, results were sufficiently encouraging that efforts in this line of research continue to this day (Kim et al., 2020). This pioneering work also paved the way for many groups to transplant neuronal progenitors derived from rodent embryos or rodent neuronal cell lines into multiple sites within the neonatal rodent brain, with the dual aims of studying neuronal development and testing therapeutic potential (see Figure 1-2 B) (Björklund and Lindvall, 2000).
A major advance came with the ability to generate neurons from human stem cells, particularly iPSCs. As was the case for organoids, discussed above, these
reagents freed the field from reliance on scarce and ethically challenging fetal tissue. In appropriate mouse models, for example, transplantation of midbrain neuronal progenitors generated from human embryonic stem cells attenuates Parkinsonian symptoms (Kim et al., 2020; Xiong et al., 2020); transplantation of interneurons generated from iPSCs attenuates epilepsy (Cunningham et al., 2014; Harward and Southwell, 2020; Southwell et al., 2014; Upadhya et al., 2019); and transplantation of similar inhibitory interneurons into spinal cord attenuates neuropathic pain (Braz et al., 2017; Hunt and Baraban, 2015). Other studies have transplanted small numbers of neurons derived from human iPSCs into mouse cortex and studied them with histological and physiological methods to analyze human-specific developmental features that would not be accessible using human fetal material (Linaro et al., 2019). In addition, to investigate disease mechanisms, stem cells to be transplanted can be genetically modified or derived from patients with specific diseases (see Figure 2-4).
Finally, although the majority of transplantation studies have focused on engraftment of neuronal progenitors, exciting results have also been reported for glial transplants, including all of the three major glial types: oligodendrocytes, which form myelin; astrocytes, which support neuronal metabolism and signaling; and microglia, which are critical for responses to injury and inflammation (see Box 2-1). For example, human glial progenitors can restore oligodendrocytes that form myelin in a mutant mouse that lacks oligodendrocytes (Windrem et al., 2020). Given that several human diseases, including multiple sclerosis, result from myelin loss, this preclinical study suggests a promising therapeutic approach. In another experiment, Hasselmann and colleagues (2019) populated the mouse brain with microglia derived from human iPSCs and showed that these microglia respond to brain injury and inflammation in ways similar to endogenous microglia. Han and colleagues (2013) transplanted human astrocyte precursors into neonatal mouse cortex and found, remarkably, that as adults, the hosts exhibited improved performance in learning and memory tasks compared with controls. One possible explanation is that the human astrocytes were better able than their mouse counterparts to support the neuronal functions responsible for the behaviors.
HUMAN NEURAL CHIMERAS
Transplants vary markedly in the number and type of human neural cells introduced and the stage at which they are introduced into the nonhuman host. In general, the extent of integration is likely to be greater the earlier the cells are engrafted because they can then develop and interact in parallel with host cells. In this respect, introduction into the embryonic blastocyst of a nonhuman mammal allows maximal engraftment.
The blastocyst is a hollow ball of cells within which a small group, called the inner cell mass, is destined to give rise to the entire embryo. (The cells that form
the ball itself are the precursors of the placenta.) In this method, ESCs or iPSCs are microinjected into the blastocyst cavity, where they mix with cells of the host inner cell mass (see Figure 1-2 C). The embryo is then implanted into the uterus of a female of that animal species, where it can develop to term and give rise to live, healthy, chimeric offspring. The method was initially developed for generation of genetically engineered mouse “knock-out” lines, in which genetically engineered embryonic stem cells contribute to all tissues, including the gonads, generating what are called germline chimeras. In blastocyst complementation, the host blastocyst is engineered so that cells of a particular organ either fail to form or are eliminated at an early stage. The introduced cells can still populate all tissues, but they make their greatest contribution to the eliminated organ because they do not need to compete there with host cells. Specificity can be enhanced by engineering the donor embryonic stem cells or iPSCs to eliminate their ability to generate particular cell types, most importantly germ cells. This method has been used to generate chimeric mice, rats, and pigs with donor-derived organs including pancreas, but the only chimeras generated from human cells to date have not survived past extremely early embryonic stages (Masaki and Nakauchi, 2017; Wu et al., 2017).
Recently, Chang and colleagues (2018) used blastocyst complementation to generate mice in which most forebrain neurons were derived from another mouse. They showed extensive replacement of principal (excitatory) neurons of the host by those from the mouse donor, and demonstrated that the offspring were healthy and, to the extent tested, structurally and behaviorally intact. They also generated chimeras using embryonic stem cells from a mouse in which they had inactivated a gene implicated in human intellectual disability. The offspring recapitulated structural and behavioral phenotypes characteristic of the mouse mutant, demonstrating the power of this method to model diseases.
In most chimeras described to date, the donor and host are from the same species, but in a few cases, they differ—for example, rat embryonic stem cells can contribute extensively to nonneural mouse organs (Kobayashi et al., 2010; Wu et al., 2017). At present, formation of neural chimeras from injection of human ESCs or iPSCs into mouse blastocysts has not been reported, and there are good reasons to believe it would be infeasible for brains at this time. One major impediment is that maturation times of human and mouse neurons are roughly proportional to the gestation times of the species, which differ by more than 10-fold, and the temporal mismatch persists when human neural cells are transplanted into mouse brain (Linaro et al., 2019; Masaki and Nakauchi, 2017; Rayon et al., 2020). Thus, even though the introduced human cells might begin to develop in parallel with the host mouse cells, the temporal mismatch would likely prevent formation of a functional chimeric brain. Attempts to improve integration of human cells into rodents in chimerism are under way, but the contributions to the nervous system have to date been minimal (Hu et al., 2020). It is possible that transplantation of human stem cells into a nonhuman primate with greater genetic
similarity to humans and a more similar gestation time (gestation time for macaques is around 5 months) might reduce this mismatch and allow some degree of chimerism to occur. Moreover, chimeras generated in nonhuman primates may well be better models of human disorders, particularly psychiatric disorders, relative to chimeras generated in rodents. Results from macaque models of autism, generated by gene editing, support this idea (Qiu et al., 2019; Zhou et al., 2019).
CAPACITIES OF HUMAN NEURAL ORGANOIDS, TRANSPLANTS, AND CHIMERAS
Human neural organoids, transplants, and chimeras all contain neurons that can generate and propagate electrical signals (action potentials) and form synapses with other neurons. For organoids, synaptic partners are within the organoid, whereas donor neurons in transplants and chimeras can form synapses with host neurons. Neurons in organoids have already been shown to generate electrical signals in response to a sensory stimulus (light) and to evoke contractions in cocultured muscle cells (Andersen et al., 2020; Quadrato et al., 2017). Even simple neural circuits, such as those that form in conventional monolayer (two-dimensional) cultures, are capable of rudimentary information processing (Ju et al., 2015), and the same is true of neural organoids. For neural transplants and chimeras, synaptic connections with the host imply that the introduced neurons could respond to sensory inputs, influence motor outputs, and participate in sophisticated computations.
As discussed in Chapter 3, some ethical concerns about human neural organoids, transplants, and chimeras require further scientific knowledge to address, and some do not. Of those ethical concerns that require more research, the most prominent regarding neural organoids revolve around the possibility that they might develop certain capacities, such as perception, consciousness, or the ability to experience pain. These capacities are not thought to be present in current neural organoids or in monolayer culture systems. For neural transplants and chimeras, a key concern is that as a consequence of introducing human neurons, existing capacities of the host might be altered in ways that would make their experience of the world much more similar to that of humans. This concern would be heightened if the host were a nonhuman primate, because more deference is accorded to primates used in research than to other species, such as mice and rats (see below). Capacities of greatest concern include the capacity to experience pain and the quality termed consciousness.
Pain
The subjective experience of feeling pain is certainly not a uniquely human quality, and behavioral and physiological metrics for assessing pain in nonhuman animals are well established. Such measurements can be applied to animals
with neural cell transplants and to chimeras. The idea that organoids now or in the future might feel pain is a more challenging and perhaps more concerning question to some.
But what is pain? At the simplest level it involves activation of a set of sensory neurons called nociceptors, which respond to such potentially damaging stimuli as corrosive chemicals, intense pressure, or extreme heat. However, pain is an experience, not simply a sensation, and nociceptors are neither necessary nor sufficient to evoke it.1 For example, patients who have had limbs amputated sometimes report pain in those limbs even in the absence of nociceptors. Conversely, stimulation of nociceptors fails to evoke pain in the presence of analgesics that act on the central nervous system. Instead, pain may be more closely associated with activity in regions of the brain to which nociceptors project, directly or indirectly—for example, the anterior cingulate and insular regions of the cerebral cortex. Indeed, pain can be elicited by direct activation of these and other brain areas, without nociceptive stimulation (Harriott et al., 2021; Sun et al., 2020). However, these circuits remain incompletely defined, so it is difficult to provide guidance on what circuits would need to be present in an organoid for it to generate a percept akin to pain. Whatever the relevant circuits are, they are vastly more sophisticated than any circuitry present in current organoids or assembloids, and the prospects for generating such complex circuits in the foreseeable future are remote.
Even less well defined is the potential to experience pain. Some experts point to this capacity as being as important as pain itself because it does not rely on any specific stimulation paradigm. At present, however, the capacity to experience pain can be assessed only in the context of a painful stimulus. This situation may change as the circuitry required to experience pain becomes better understood, but at present neuroscientists would not be able to recognize neural circuits that confer the potential for pain in an organoid even if they existed.
Thus, two related but logically distinct concerns about pain in human neural transplants, chimeras, and organoids might be raised: that the entity actually experiences pain, and that the entity has the capacity to experience pain (or the potential to develop that capacity). Chapter 3 considers the issue of the experience of pain in research animals from the perspective of animal welfare regulations, which form the current basis for oversight of animal research, as well as from the perspective of animal rights.
Consciousness
A key capacity of concern is consciousness, sometimes termed sentience or awareness. These terms are notoriously difficult to define and are even more
___________________
1 Allan Basbaum, University of California-San Francisco, presentation to the committee, August 11, 2020, virtual meeting.
difficult to measure. Because these terms are often used interchangeably, we use “consciousness” hereafter, even though its equivalence with other terms is not universally accepted.
Consciousness, in the sense that elicits substantial ethical concern, denotes a subjective experience, people’s introspective awareness of the external world and the internal states of their own bodies, generally tied to a cognitive and/or emotional impact. In the absence of subjective experience, moral or ethical concerns are substantially diminished. For example, before surgery is performed on human patients (and most nonhuman animals), subjective experience is abolished by means of anesthetic drugs to prevent the experience of suffering. Entities that lack the capacity for subjective experience, such as plants, are accorded lower moral status relative to animals known or believed to have the capacity for consciousness.
A reasonable intuition for consciousness thus defined is provided by the experience of visual perception. A person’s typical perception of a visual scene consists of recognizable objects and living beings that move (or do not) in familiar ways. This conscious experience is merely the tip of an iceberg of neural processing; it lies atop a vast complex of unconscious visual processing. People are not consciously aware, for example, that their brain actually receives two visual images—one from each eye—and combines them to form a conscious perception of a single world. Nor are people aware of the processing that causes them to perceive a single, stable visual world even though their eyes move a few times per second (saccades), sending changing views to the brain that must be stitched into a single perceived world. Analogous conscious experiences (and underlying icebergs of unconscious processing) exist in all sensory domains, as well as in the internal cognitive realms of learning, memory, affect, and attention.
The adoption of a working definition of consciousness is necessary for specifying ethical issues involved in research with human neural organoids, transplants, and chimeric animals. Once specified, ethical judgments could be critically informed by knowledge of underlying neural mechanisms. Many theories and definitions of consciousness are framed in ways that cannot readily be applied to brain function. In some cases, however, philosophers and neuroscientists working together have endeavored to provide explanations of consciousness that are amenable to scientific inquiry (Doerig et al., 2020; Linkenhoker, 2019). Two of the most prominent are the integrated information theory and the global workspace theory. According to the integrated information theory, consciousness is correlated with the ability of a system to integrate information (Tononi et al., 2016). Therefore, the level of consciousness in a brain could theoretically be measured and compared between species, individuals, and circumstances. The perturbational complexity index (PCI), described below, provides one potential metric. The global workspace theory holds that consciousness relates to an ongoing, changing broadcast of a person’s mental contents (the tips of the unconscious
processing icebergs) to a diverse network of brain circuits by means of long-range anatomical connections (Dehaene, 2014; Mashour et al., 2020).
Whatever the definition of consciousness, a key question is the extent to which it is a particularly human attribute. Higher levels of subjective experience are almost always studied in human subjects, who are able to report their experiences verbally. That does not mean, however, that consciousness is confined to humans. Indeed, the view that consciousness, particularly self-consciousness, is a defining attribute of humans has been challenged by philosophers, ethicists, animal behaviorists, and ethologists. The current consensus is that all vertebrates and some cephalopods possess consciousness to some extent. It has been suggested that mammals have a higher level of consciousness compared with nonmammalian vertebrates, primates compared with other mammals, and humans compared with other primates (DeGrazia, 2009). Few people who have lived or worked closely with vertebrate animals—for example, pet owners and research scientists—harbor any doubt that these animals have subjective experiences of the sensory world and the inner world of memory, affect, and self-movement toward goals (i.e., purpose).
Whether any of the higher levels of consciousness, such as self-awareness—that is, the ability to reflect on one’s own subjective experiences—are restricted to humans remains an open question. One framework for thinking about self-awareness was proposed by DeGrazia (2009). Although his model is not explicitly framed in evolutionary terms, it maps onto phylogenetic distinctions. DeGrazia posits four levels of self-consciousness. The first, agential, is awareness that one’s body is distinct from the rest of the environment and subject to one’s direct control. DeGrazia argues that many vertebrates have this capacity. The second level, social, involves awareness of one’s position within social relationships, such as dominance hierarchies and kinship groups. It appears to be characteristic of many mammals. The third, introspective, denotes awareness of one’s own mental status, beliefs, or feelings (metacognition). Current evidence suggests that this level is well developed in primates and may be present to some degree in other mammals as well. The fourth level, autobiographical identity, involves awareness of oneself as an individual, having a rich remembered past; entertaining multiple possibilities for the future; and possessing a narrative of some sort connecting past, present, and future. This level may be specific to humans, although it may be present to some extent in their closest relatives, the great apes (e.g., chimpanzees and gorillas). This evolutionary conception of consciousness views it as a graded quality rather than one divided into distinct groups of “have” and “have not” species based on specific abilities, such as the ability of an animal to recognize itself in a mirror (the so-called “mirror self-recognition” test (see Figure 2-5) (Anderson and Gallup, 2015).
A second set of critical questions about consciousness is neurobiological: What neural circuits are required for consciousness, and where in the brain are they housed? Answers to these questions can ground judgments about the pos-
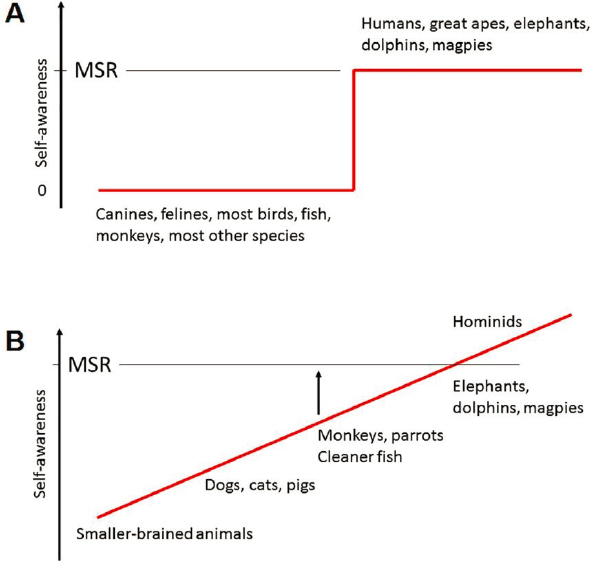
SOURCE: de Waal, 2019. Copyright: © 2019 Frans B. M. de Waal. This figure is reproduced from an open access article distributed under the terms of the Creative Commons Attribution License (see https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. The figure caption has been adapted from the original figure text.
sibilities of conscious experience in human neural organoids, transplants, and chimeras.
Research in this area has proceeded on two fronts. The first involves an attempt to define the neural circuits responsible for consciousness, often called “neural correlates of consciousness” (Koch et al., 2016). Several promising can-
didates have been proposed, but there is no consensus on which are most relevant. Detailed analysis of circuitry is generally carried out in model systems but has been extended to human subjects using fMRI (Dehaene, 2014; Huang et al., 2020). For example, Dehaene (2014) presents subliminal stimuli to subjects and asks which brain regions are activated by those that make the subject consciously aware of a stimulus that would otherwise not reach consciousness.
The other front for neurobiological research involves determining which brain regions house the highest levels of consciousness. Since these are, as far as is known, strictly human qualities, the best evidence comes from studies of human subjects who have suffered injuries to these regions. One example is blindsight, in which people with lesions of their visual cortex are nonetheless able to respond to visual stimuli without being aware they have seen anything (Fox et al., 2020). Their behavioral responses are likely to be mediated through midbrain structures, as is the case with lower vertebrates, whereas their denial of sight suggests a predominantly cortical locus for visual awareness. Another example comes from lesions to the prefrontal cortex, which can rob individuals of the ability to generate long-term plans and express individual personalities (Miller et al., 2002). In this respect, it is noteworthy that the prefrontal cortex is so poorly developed in rodents that some believe it is absent; it is substantial in primates and largest in humans. The result that executive functions, which require entertaining multiple possibilities about the future and connecting them to a remembered past, are housed in a structure that is (nearly) primate-specific and largest in humans supports DeGrazia’s view described above that autobiographical consciousness may be restricted to humans and perhaps their closest relatives.
A final question of great relevance to judging the potential of organoids for consciousness is the extent to which it depends on experience. The vertebrate central nervous system is initially assembled through a genetically determined plan, but this “rough draft” is then refined by experience as transduced into electrical neural activity (Sanes, 2021). Experience-dependent refinement occurs to some extent in all vertebrates, but appears to play a larger role in mammals than in other vertebrate classes, a larger role in primates than in other mammalian orders, and a larger role in the cortex than in subcortical structures. These relationships map well to what is known about the evolution and localization of consciousness. Thus, to the extent that higher levels of consciousness are housed in the human cortex, it is reasonable to imagine that they develop in an experience-dependent fashion. Moreover, to the extent that consciousness is defined in terms of subjective experience, it can occur only in the context of objective experience.
However, the capacity for consciousness is also a concern, as noted above, for pain, and here the need for experience is less clear. Complex neural circuitry can develop in the absence of external experience and even in the absence of electrical activity. In a classic experiment, a newt (axolotl) embryo was allowed to develop under conditions of paralysis and anesthesia, so that external stimulation and motor responses were eliminated. When the anesthetic was washed
out, the tadpole swam away (Twitty, 1937). More recently, mice were studied in which synaptic transmission is prevented by mutation of key components of the neurotransmitter release apparatus. Again, the mice developed to term with brains relatively normal in structure, although they did not survive postnatally. In view of these results, it remains possible that the substrate for consciousness could form in the absence of experience even if experience were required for manifestation of consciousness.
It is generally agreed, however, that even the capacity for consciousness relies on complex patterns of circuitry that include multiple brain regions and connections between them. Given the current properties of organoids, the prospects for organoids achieving this level of complexity in the near future are remote. Likewise, transplants of human neural cells into nonhuman animals currently involve far too few cells to generate any capacity approaching consciousness. Chimeras, in contrast, raise distinct issues, which are considered in Chapter 3.
Emotion
Suffering can arise from sources other than physical pain—for example, sadness, fear, and anxiety. The neural bases of these emotions have been studied in at least three ways (Adolphs and Anderson, 2018; Anderson and Adolphs, 2014). First, a rich literature of human neuroimaging studies has pinpointed areas in the brain in which activity is correlated with negative emotions generally and with particular negative emotions specifically. These include such areas as prefrontal cortex, amygdala, and hypothalamus, together forming what is called the limbic system. Second, systems neuroscientists have used rodents, particularly genetically engineered mice, to trace neural circuits underlying such negative emotions as anxiety and fear. Third, an increasing number of studies have demonstrated responses in invertebrates (flies) that resemble, in several respects, states called aggression, fear, and anxiety in vertebrates.
Interpretation of work on emotions in rodents and especially invertebrates remains controversial because emotions are traditionally defined in terms of human behaviors accessible by introspection and self-report. Indeed, some experts argue against assuming that emotions of the sort recognized in humans are even present in other animals (LeDoux, 2012). Others, however, argue that “emotional behaviors are a class of behaviors that express internal emotion states. These emotion states exhibit certain general functional and adaptive properties that apply across all specific human emotions, such as fear or anger, as well as across phylogeny” (Anderson and Adolphs, 2014). Indeed, recent studies have even provided evidence that mice exhibit empathy: If one mouse is exposed to another mouse experiencing pain or fear, the observer will become more sensitive to painful or frightening stimuli (Klein and Gogolla, 2021). This transfer is related to a “theory of mind,” once thought to be restricted to primates, in which an animal has some awareness of the experience of another animal. Based on this
accumulation of evidence, it appears likely that rodents do have emotions. Therefore, assessment of emotional capacity in transplants or chimeras would have to be based not on acquisition of emotional responses, but rather on assessment of alterations in the emotional capability of the transplant or chimera compared with an unmanipulated host.
MEASURING CHARACTERISTICS OF HUMAN NEURAL ORGANOIDS, TRANSPLANTS, AND CHIMERAS
As research on human neural organoids, transplants, and chimeras proceeds, it will be increasingly important to devise methods for assessing their characteristics and capacities. These include consciousness, which, as discussed above, has resisted clear definition and is therefore difficult to assess. For other measures of complexity, however, some metrics exist.
Human Neural Organoids
At present, neural organoids lack complex and precise circuitry, are missing critical diversity among cell types, and do not include more than very limited representations of the multiple brain regions and long-range circuitry thought to underlie consciousness (Alves et al., 2019; Zirui et al., 2020). Thus, there is currently no objective basis for ascribing consciousness to organoids. Moreover, to the extent that consciousness requires experience and/or goal-directed behavior, it may be impossible for organoids to acquire consciousness in the foreseeable future. As organoid technology advances, however, assessment of their capacities may become more relevant, not only for purely scientific purposes but also to guide decisions about whether and under what circumstances they should or should not be used in research. The most reliable ways of assessing pain, consciousness, and related capacities are behavioral. It is possible that behavioral outputs could be inferred in cases in which human neural organoids are transplanted into the brains of host animals. For organoids maintained in culture, however, it is extremely unlikely that behavioral metrics will be feasible in the foreseeable future: Even when these organoids are connected to an output device, such as by innervating muscle cells, any outputs will be reflexive responses, which are inadequate for assessing consciousness or pain.
A feasible alternative will be to track how closely neural organoids and assembloids resemble actual brains. Anatomical, molecular, and physiological metrics can be assessed. Anatomical metrics include organoid size, number of cells, dendritic complexity of neurons, and synaptic connectivity. Immunohistochemical markers of specific cell classes or types can be applied to assess the degree to which neurons have differentiated (Albanese et al., 2020), the extent to which they are arranged into laminar or nuclear structures reminiscent of the brain, and the extent to which they display circuit motifs and activity dynamics
characteristic of specific brain regions. Assessment can also include analyses of key glial types and properties, such as myelination, that are critical for normal neural function.
Molecular metrics, such as transcriptomic and epigenomic profiling using high-throughput single-cell or single-nucleus RNA-seq (scRNA-seq and snRNAseq, respectively) (Quadrato et al., 2017), can be used to assess gene expression of many thousands of single cells per organoid. These methods are increasingly being applied to postmortem samples of developing and adult human brains, making it possible to compare gene expression patterns in organoids with those in vivo. From the comparison, one can assess the levels of differentiation and maturation of cells in the organoids, their diversification into distinct cell types, and the extent to which they resemble their in vivo counterparts. Proteomic and metabolomic measures can currently be made from whole organoids and are rapidly progressing to the single-cell level.
Physiological methods can be applied to assess patterns of neuronal activity and synaptic connectivity in neural organoids, comparing results with those obtained, for example, from human samples collected ex vivo. EEG (electroencephalogram) techniques have already been used in neural organoids (Trujillo et al., 2019). Physiological methods can be used to assess synaptic plasticity, which is thought to be the physical basis of learning and memory, and to determine whether synapses are regulated by such neuromodulatory substances as dopamine, norepinephrine, serotonin, and acetylcholine, which regulate plasticity and brain state. All of these methods are in common use and have already been applied to neural organoids to some extent, so technical challenges to their use are minimal.
At a higher level, the complexity of a neural system can be evaluated and even quantified by proposed physiological measures, such as the aforementioned PCI (Casali et al., 2013). This index is calculated from measurements of the complexity of an EEG response in both time and space following delivery of a pulse of transcranial magnetic stimulation to the brain. PCI levels have been shown to correlate with the level of consciousness in healthy humans (i.e., awake vs. sleep) and in patients who are anesthetized, minimally conscious, or comatose, indicating the PCI’s potential utility in measuring consciousness. Thus far, the PCI has not been used across species to assess potentially informative differences, nor has it been applied to neural organoids. It has, however, been adapted for in vitro use to study connectivity and complexity in slices of mouse brain (D’Andola et al., 2018).
It should be noted, however, that these approaches cannot by themselves demonstrate the presence of consciousness. It is more useful to think of these characteristics as being necessary but not sufficient for consciousness. As the cell types, circuits, signaling mechanisms, and regional specializations of neural organoids become increasingly brain-like, concerns about conscious states will need to be reconsidered.
Human Neural Cell Transplants and Chimeras
Most of the tests described above for neural organoids can also be applied to transplants and chimeras. A critical difference, however, is that behavioral tests can be used to determine whether any capacities of animals with human neural cell transplants or chimeric animals differ from those of their unmanipulated counterparts. Cognitive capabilities can be measured with assays for learning and memory that are well established in animal models (such as those used by Han and colleagues [2013]). A promising recent advance is the introduction of methods for capturing and quantifying behavior at high spatial and temporal resolution (Mathis and Mathis, 2020; Mathis et al., 2018; Oikarinen et al., 2019; Wiltschko et al., 2020). These new methods can identify discrete, elementary behavioral motifs that are stitched together in different ways to generate more sophisticated, goal-directed behaviors. They come closer than traditional methods to harmonizing the scales of behavioral and physiological measures, and therefore improve the ability to assess possible consequences of augmenting nonhuman brains with human neurons.
Researchers have also developed behavioral metrics for other types of awareness or emotional capabilities that range from whether nonhuman animals feel pain (e.g., learned avoidance of painful stimuli) to tests of self-awareness (e.g., a mirror test to determine whether an animal recognizes itself in a mirror [Anderson and Gallup, 2015]; see Figure 2-5), metacognition (Smith, 2010), and capacity for empathy (de Waal and Preston, 2017; Klein and Gogolla, 2021). However, many of these assays can be difficult to design and interpret because nonhuman animals may not express these traits in a way that is obvious to humans (de Waal, 2019). In view of the difficulty of measuring consciousness directly, Pennartz and colleagues (2019) have proposed an alternative approach, based on assessment of multiple qualities they call “indicators” of consciousness, none individually decisive but highly suggestive in the aggregate (see, e.g., Edelman et al., 2005; Seth et al., 2005). They include goal-directed behavior, the presence of circuits that mediate multimodal integration, episodic memory, susceptibility to illusions, and specific visuospatial behaviors. Going forward, it will be essential to reach agreement on an operational definition of consciousness and on acceptable methods for assessing its presence. However difficult to attain, this community consensus will provide a basis for oversight as transplant and chimera research progresses.
This page intentionally left blank.


























