Proceedings of a Workshop
| IN BRIEF | |
April 2021 |
Improving the Evidence Base for Treatment Decision Making for Older Adults with Cancer
Proceedings of a Workshop—in Brief
The National Academies of Sciences, Engineering, and Medicine hosted the virtual workshop Improving the Evidence Base for Treatment Decision Making for Older Adults with Cancer on January 22, 25, and 27, 2021. This workshop—sponsored by the U.S. Food and Drug Administration (FDA)—was convened by three forums of the National Academies: the National Cancer Policy Forum; the Forum on Drug Discovery, Development, and Translation; and the Forum on Aging, Disability, and Independence. Workshop speakers examined the persistent underrepresentation of older adults in cancer clinical trials across the drug development continuum and identified potential solutions to strengthen the evidence base to inform treatment decision making for older adults with cancer, discussed throughout this Proceedings of a Workshop—in Brief and highlighted in Box 1. Participants examined the importance of adequate representation of older adults in cancer drug development and discussed why some traditional approaches to clinical trial design and recruitment hinder enrollment of older adults in cancer clinical trials. Many speakers described examples of study design and real-world evidence development approaches that may provide more meaningful insights for treating older adults with cancer, and addressed policy changes and ethical considerations to improve inclusion of older adults in cancer drug development. Participants underscored the urgency of the problem and pointed to myriad opportunities for clinicians, researchers, drug companies, regulators, journals, patients, and patient advocacy groups to work together to proactively build the evidence base to improve cancer care for older adults.
![]()
BACKGROUND AND CONTEXT
Monica Bertagnolli from Brigham and Women’s Hospital and Harpreet Singh from FDA discussed the impetus for the workshop and its key goals. As the U.S. population ages, the overall number of individuals diagnosed with cancer is increasing and older adults also account for a growing proportion of both cancer diagnoses and cancer deaths.1 Clinicians, along with patients with cancer, and their families rely on clinical research for information on the potential benefits and harms of different treatment options, so that they can make informed treatment decisions that are consistent with patients’ needs, values, and preferences (IOM, 2013). However, the evidence base for older adults with cancer is limited by a pervasive underrepresentation of this population in cancer clinical trials. Singh pointed to an FDA analysis that found older adults, especially those over age 75, are underrepresented in the registration trials of new cancer therapies (Singh et al., 2017) (see Figure 1).
Laura Levit from the American Society of Clinical Oncology (ASCO) and Heidi Klepin from the Wake Forest School of Medicine reviewed previous efforts to understand and address the issue. These include the 2013 report Delivering High-Quality Cancer Care from the Institute of Medicine (IOM, 2013), recommendations issued by ASCO and Friends of Cancer Research (Hurria et al., 2015; Kim et al., 2017, 2021; Lichtman et al., 2017), a symposium organized by ASCO and FDA (Levit et al., 2018), and draft guidance from FDA (2020). While these steps have been important, Levit stressed that the issue is far from resolved; in a recent analysis of more than 300 cancer trials, trial participants were on average 6.5 years younger than the median age of the patient population for each disease—a gap that has increased over time (Ludmir et al., 2019). Levit outlined four main strategies that emerged from previous efforts to improve the evidence base for older adults with cancer. The first strategy is to increase enrollment of older adults in cancer clinical trials through approaches such as working with FDA to devise plans to enroll representative numbers of older adults in clinical trials, broadening eligibility criteria, opening more community-based clinical trial sites, and adhering to the policy on Inclusion Across the Lifespan from the National Institutes of Health (NIH, 2017). A second strategy is to design trials that collect information relevant to older adult populations; for example, including geriatric assessments and patient-reported outcomes (PROs) as co-primary or composite endpoints, and the characterization of patients enrolled
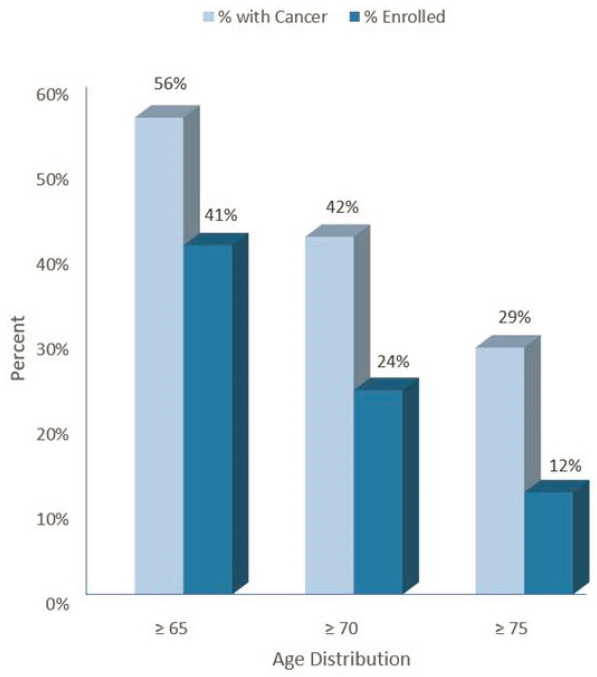
SOURCES: Presented by Singh on January 22, 2021; adapted from Singh et al., 2017.
___________________
1 See https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2019/cancer-facts-and-figures-special-section-cancer-in-the-oldest-old-2019.pdf (accessed March 19, 2021).
(e.g., comorbidity and functional status). A third strategy is to expand the collection of real-world data (RWD), such as from electronic health records (EHRs), for use in cancer research. Finally, Levit said, the fourth strategy is to advance all of these efforts through collaboration, advocacy, and policy action.
The underrepresentation of older adults in cancer clinical trials contributes to a lack of understanding of both the efficacy and tolerability of cancer drugs in this population and how they affect patient quality of life. Supriya Mohile from the University of Rochester noted that approximately two-thirds of older adults are considered vulnerable or frail, which puts them at greater risk for adverse outcomes. Rebecca Pentz from the Winship Cancer Institute and the Emory University School of Medicine said there is an ethical imperative to improve the evidence base for older adults with cancer. Without data regarding how well treatments work in older patients with cancer, clinicians, the older patients they care for, and family caregivers are making life-altering decisions without adequate evidence-based guidance (Fentiman et al., 1990; Kearney and Miller, 2000).
OVERARCHING ISSUES: DESIGNING MORE INCLUSIVE STUDIES
A number of speakers discussed reasons why older adults may be left out of trials and suggested opportunities for designing more inclusive studies. Overly stringent eligibility criteria commonly limit participation, said Mary Whitehead, Patient Advocate with the Cancer and Aging Research Group. Because older adults are more likely to take several medications, have a previous cancer diagnosis, or have organ dysfunction, Lilli Petruzzelli from InCyte said loosening medication or comorbidity restrictions, or adjusting dosages for these patients, could enable more older adults to participate. Petruzzelli, Hyman Muss from the University of North Carolina at Chapel Hill, and Stuart Lichtman from the Memorial Sloan Kettering Cancer Center suggested minimizing exclusionary criteria, especially those criteria that are unrelated to drug performance or are more common in older adults, such as renal function, concomitant medication usage, or the presence of comorbidities. Rather than using age as a cutoff, Michael Maitland from the Inova Health System, and other participants suggested using more nuanced measures of patients’ health status, such as a comprehensive risk score (Hurria et al., 2016). Several speakers encouraged increased use of geriatric assessments, and Muss urged a greater emphasis on endpoints reflecting functional status and quality of life, in addition to traditional endpoints. For example, Muss said PROs can be used to track symptoms such as nausea, neuropathy, and fatigue. Lichtman agreed and suggested using simple, established strategies to track factors such as patient gait, weight loss, and depression.
Mohile and many other speakers posited that different trial design strategies could be utilized to support the enrollment of older adult populations. Geriatric oncology trials conducted by the National Cancer Institute (NCI) Community Oncology Research Program have demonstrated that the evidence base can be strengthened by having an age-relevant research question, a multidisciplinary team, stakeholder input in trial design, and relevant assessment and tolerability endpoints (Mohile et al., 2020a,b). Mina Sedrak from the City of Hope outlined opportunities to improve involvement of older adults throughout the drug development process. One suggestion was to create trials or cohorts specifically for older, high-risk, and frail adults, with more robust data collection on how cancer treatment affects patient outcomes among this population. Another is to embed aging considerations throughout the drug development life cycle by expanding safety considerations to include impacts on underlying biological aging processes, such as cognition and function, Sedrak said. In addition, Martine Extermann from the Moffitt Cancer Center suggested that investigators could seek to establish the minimum effective dose of a drug rather than a maximum tolerable dose, which could also improve the safety profile, particularly in older, frail populations. Finally, a number of speakers suggested that post-marketing registries could inform clinicians about more representative patient outcomes in community-based clinical practice.
OVERARCHING ISSUES: ADDRESSING PERVASIVE BARRIERS
Many speakers discussed a variety of factors influencing older adults’ willingness and ability to enroll in cancer clinical trials. Practical factors, such as the location of a trial, lack of transportation or family support, and financial constraints, can pose significant barriers for older adults, Whitehead and Petruzzelli said. Participating in a clinical trial can be disruptive to patients’ lives, and traveling to distant trial sites can be particularly challenging. Several speakers, including Mihaela Popa-McKiver from Bristol Myers Squibb, Mary Lou Smith from the Research Advocacy Network, and Extermann suggested that clinical trial protocols should condense study appointments and associated tests as much as possible and enable individuals enrolled in a trial to participate from a community oncology practice and use local lab or imaging facilities. Larissa Nekhlyudov from Brigham and Women’s Hospital also encouraged involvement of primary care clinicians in discussions of enrolling in a clinical trial, because patients—especially older adults—often have developed long-term relationships with their clinicians and trust their input on whether a clinical trial may be right for them. Home-based clinical trial participation strategies—including telemedicine, home visits, wearables, or self-administered
sampling protocols—could also improve access to clinical trials for older adults with cancer. Several speakers—including Gwen Darien from the National Patient Advocate Foundation—noted that recent moves toward trial decentralization in response to the COVID-19 pandemic could facilitate greater adoption of these strategies, where feasible (Pennell et al., 2021).
Smith and Sedrak cautioned that many clinicians do not discuss the option of enrolling in a clinical trial with older patients. This can be due to a variety of factors, including implicit biases, ageism, or concerns about whether a trial is well suited to an older patient (e.g., whether all arms of the study may be tolerable), particularly if that patient is frail, has comorbidities, or may have inadequate caregiver support (Daher, 2013; Kearney et al., 2000; Surbone, 2008). Smith added that patient–clinician communication about cancer care and the option to participate in a clinical trial can be hampered by undiagnosed hearing loss among older patients. Many speakers stressed that older adults should not be denied the opportunity to enroll in cancer clinical trials. Pentz argued that the ethical principle of justice is thwarted if older adults are denied access to appropriate trials.
Whitehead and Muss suggested engaging patients and patient advocates in the trial design process to help inform more patient-friendly protocols. Smith suggested investigators take a more patient-centered approach to the informed consent process, consider patient financial burdens associated with clinical trial participation, and improve communication to help participants feel more invested and empowered when participating in clinical trials. Whitehead also stressed the importance of communicating effectively about a study’s purpose and results.
CHALLENGES AND OPPORTUNITIES IN EARLY PHASE CANCER DRUG DEVELOPMENT
Early phase clinical trials, designed for initial assessment of dosing and safety, are a critical component of the cancer drug development process. R. Donald Harvey from the Emory University School of Medicine noted that the clinical benefit and the impact of a phase I trial in a drug’s overall pathway to approval has increased tremendously in recent years. Maitland agreed and noted that breakthroughs in biomarkers, molecular pathology, and technologies are enabling researchers to fast-track drug development; while this can have benefits, a drive toward smaller, faster studies can also limit the ability to generate knowledge on drug performance or patient metabolic response (Lyauk et al., 2019). Researchers may assume older adults are too frail to help determine a maximum tolerated dose, said FDA’s Nam Atiqur Rahman; as a result, drug interaction and pharmacokinetic studies often include older adults only when testing the safe starting dose or during dose tailoring. Maitland, Harvey, and Rahman suggested employing clinical pharmacology methods and principles to develop optimal dosing strategies for novel cancer drugs in older adult populations. Klepin added that researchers could harness geroscience principles to target specific biological mechanisms of aging and improve therapeutic development, but this requires the inclusion of older adults in every phase of drug development.
J. Jack Lee from the University of Texas MD Anderson Cancer Center described how researchers can leverage statistical methods to extract more information from research, increase representation of older adults, and reduce sources of bias and variability. Extermann suggested that early phase clinical trials could be improved by incorporating assessment of patient comorbidities with the Cumulative Illness Rating Scale-Geriatric, measuring functional status with the instrumental activities of daily living score, using performance tests to obtain objective physical data, and collecting PROs relevant to older adults. Several speakers discussed opportunities to use adaptive trial designs to assess different age subgroups and dosing strategies (Hobbs et al., 2018; Zhu et al., 2020). The Bayesian Hierarchical Model for Synthesizing Information can help analyze clinical trial data, especially across subgroups, and can synthesize information from the trial with real-world evidence, Lee noted (Chen and Lee, 2019, 2020). Extermann described opportunities to use adaptive design to progressively modify inclusion criteria as early results become available, and how stratification of phase II trials can yield insights into safety for different patient groups. Petruzzelli and Lee noted that a potential downside of grouping patients into cohorts in early phase trials is that it could reduce trial efficiency; Lee suggested that “borrowing” data across groups, with continuous covariate adjustments, could help address this. Smith and Lee added that personalized dosing approaches, once validated, could also enhance the ability of older adults to participate in early phase trials.
CHALLENGES AND OPPORTUNITIES IN CLINICAL TRIALS WITH REGISTRATIONAL INTENT
Singh and Rajeshwari Sridhara, a contractor with FDA, discussed recommendations from FDA’s 2020 draft guidance on increasing trial participation among older adults (FDA, 2020), as well as Project Silver, FDA’s initiative to improve the evidence base via regulatory policy, advocacy, outreach, and global engagement. They noted the importance of these efforts in all phases of drug development. Early phase trials can provide more information for older adults on safety, tolerability, dose modifications, drug interactions, and assessment feasibility. Confirmatory trials conducted with registrational intent can be designed to group patients by age or risk, or can use hierarchical or adaptive design to address safety concerns. Post-marketing studies, including the use of RWD from clinical practice, can provide information on
the safety and effectiveness of cancer drugs in broader populations. Sridhara and Singh said key considerations when designing trials to increase older adult enrollment include the comorbidities, toxicity risks, or drug interactions that are acceptable; the proportion of older adults who should be included; whether hierarchical testing is feasible; how safety will be assessed in high-risk populations; whether serious adverse events can be correctly interpreted and attributed; how patients will access care facilities; and what can be learned during post-marketing studies.
Efrat Dotan from the Fox Chase Cancer Center, Kevin Tang from Odonate Therapeutics, Richard M. Stone from the Dana-Farber Cancer Institute, and Monica Giovannini from Novartis described strategies to involve older adults in clinical trials for pancreatic cancer,2 breast cancer (O’Shaughnessy et al., 2019), acute myeloid leukemia (DiNardo et al., 2020; Lancet et al., 2016), and lung cancer (Wolf et al., 2020), respectively. Hans Wildiers from the University Hospital Leuven, Belgium, shared examples of trials conducted in Europe (Wildiers et al., 2013). To make trials feasible and meaningful for older adults, several speakers emphasized keeping eligibility criteria broad; employing geriatric assessments; administering medications orally when possible; offering telemedicine or home visits; assessing endpoints such as quality of life, functioning and independence, peripheral neuropathy, and cause of death, because older adults may die from causes other than their cancers; modifying dose based on patient fitness; and fostering collaboration among community oncology practices and drug developers. Smith noted that it is critical to elicit the input of older adults and their caregivers—through surveys, focus groups, or other mechanisms—to better understand what aspects of clinical trial design are of greatest importance to the participants involved in cancer drug development.
CHALLENGES AND OPPORTUNITIES WITH POST-MARKETING STUDIES
In the absence of adequate data from clinical trials, several speakers said that various types of post-marketing studies and data sources (including RWD such as administrative claims data, EHRs, or cancer registries) can help inform treatment decision making for older adults with cancer (FDA, 2013, 2018). Bindu Kanapuru from FDA suggested that drug sponsors should discuss post-marketing data-collection strategies with FDA early in the drug development process, and that they should take advantage of multiple data sources to improve the evidence base.
Robert Miller from CancerLinQ; Deborah Schrag from the Dana-Farber Cancer Institute; and Neal Meropol from Flatiron Health described the potential of RWD to fill evidence gaps, but also noted certain limitations of using RWD. These include the prevalence of missing data; the possibility of poor data quality; the potential for biases, some that may be challenging to measure; and particularly for registry data, long lag times for data availability. Despite these challenges, they noted that analyses using RWD can provide valuable insights, especially with regard to studying disease burden, adherence to protocols for cancer treatment, and access to care. Meropol added that it is important to consider the specific characteristics of each RWD source, including generalizability, to understand its appropriate use in a particular clinical context.
The sheer size of some observational data sets can be advantageous, and several participants said that linking different data sources together also can provide valuable insights. Lisa Hess from Eli Lilly and Company suggested greater collaboration among industry, academia, and community oncology practice settings. Dawn Hershman from Columbia University described how combining observational and randomized controlled trial data can help inform both treatment decisions and efforts to increase enrollment of older adults in clinical trials (Hershman et al., 2018; Unger et al., 2006). Schrag said that combining NCI’s Surveillance, Epidemiology, and End Results (SEER) Program registry data with Medicare claims data can create large, representative cohorts to answer specific questions about care or outcomes (Enewold et al., 2020). Jennifer Malin from UnitedHealthcare added that claims data can provide insights on patterns of care, comorbidities, adverse events, and outcomes. Miller stressed the need to further improve the quality of RWD by implementing data standards such as mCODE.3
POLICY OPPORTUNITIES
Many participants expressed frustration that the evidence gaps for treating older adults diagnosed with cancer have persisted despite decades of attention and guidance aimed at increasing the representation of older adults in cancer drug development. A number of participants suggested policy actions that could help to close evidence gaps. Ishwaria Subbiah from the University of Texas MD Anderson Cancer Center said that Congress should act to expand FDA’s authority to improve inclusion of older adults across the cancer drug development continuum by requiring that clinical trial participants adequately reflect the patient population of a given disease. Sue Peschin from the Alliance for Aging Research agreed and added that pharmaceutical companies are unlikely to change their practices unless they are required to do so. She pointed to the NIH policy on Inclusion Across the Lifespan and the European Medicines Agency’s
___________________
2 See https://clinicaltrials.gov/ct2/show/NCT04233866 (accessed March 18, 2021).
3 Minimal Common Oncology Data Elements. See https://mcodeinitiative.org (accessed March 19, 2021).
Geriatrics Medicines Strategy4 as possible models on which to build. Eric Rubin from Merck Research Laboratories said that industry is a willing partner in discussions of these issues, and he was optimistic about the use of extension cohorts or subsequent clinical trials with relaxed eligibility criteria as an approach to enable the evaluation of safety and efficacy in older patients without slowing drug development. Aaron Kesselheim from Brigham and Women’s Hospital/Harvard Medical School reviewed current rules that extend the period of market exclusivity as an incentive to increase clinical trials of approved drugs in children. He argued that such an approach would not be an appropriate strategy to improve the evidence base for older adults with cancer, because this incentive in the context of pediatric trials has proven to be wasteful and has not consistently incentivized timely accumulation of useful information (Hwang et al., 2019; Sinha et al., 2018). Instead, Kesselheim suggested focusing on measures such as mandates for new drug applications to include clinical trials with a representative sample of older adults; post-marketing commitments with greater enforcement mechanisms such as requiring FDA to re-review drugs after a certain period of time or placing restrictions on the pricing of drugs for which essential post-approval data are not collected; or targeted governmental funding for clinical trials involving older adults with cancer. Several speakers suggested that regulators should require clinical trial protocols to include safety, efficacy, and dosing recommendations for older adults; engage community oncologists; make trial enrollment easier; broaden eligibility criteria; encourage the use of adaptive trial designs and geriatric assessment tools; and include geriatric experts on advisory boards. Richard Schilsky from ASCO and Randall Oyer from Penn Medicine Lancaster General also suggested that FDA update its drug labeling practices to incentivize inclusion of older adults in trials and post-marketing research. Although passing new legislation is difficult, it may be feasible to add provisions to upcoming legislation, such as the 2022 Prescription Drug User Fee Act reauthorization, Kesselheim noted.
Many participants also pointed to opportunities for collaborations among professional societies, journals, and health care organizations to advance policies that facilitate more inclusive practices. Bertagnolli and Schilsky urged updates to the policies of NCI’s National Clinical Trials Network and ASCO’s quality certifications aimed at increasing representation of older adults. Several speakers suggested that journals could require investigators to include improved data on patient demographics—including, at a minimum, patient age, performance status, a validated comorbidity measurement, and a validated self-report measure—and refuse to publish clinical trials that unnecessarily exclude older adults. Several participants also discussed how EHRs could be improved by simplifying data entry, capturing outcomes that are meaningful to older adults, and incentivizing good data practices, but noted that regulation might be required to facilitate adoption. Lichtman and Schilsky suggested adequately reimbursing clinicians for the time it takes to discuss potential clinical trial options with patients, and to help them through the process.
CLOSING REMARKS
“We have an ethical obligation to improve evidence development for older adults with cancer,” stressed Bertagnolli. “Our society must deliver the best possible care for all patients, and a key requirement for this is to eliminate health disparities across all groups, including those defined by age, gender, race, and socioeconomic status. One of the most glaring disparities in the field of oncology is the lack of data to guide care for older adults.”
Singh agreed, adding, “Oncology is geriatric oncology and vice versa. While we must count on our experts in geriatric oncology to guide us, advocacy for research to address the needs of older adults cannot rest with them alone. We need cancer centers, clinical trial groups, community oncology programs, and industry all involved, and clinical oncology evidence generation [is needed] to correct this disparity so that our older citizens do not miss out on the opportunities provided by our advances in health care.” ◆◆◆
REFERENCES
Chen, N., and J. J. Lee. 2019. Bayesian hierarchical classification and information sharing for clinical trials with subgroups and binary outcomes. Biometric Journal 61(5):1219–1231. doi: 10.1002/bimj.201700275.
Chen, N., and J. J. Lee. 2020. Bayesian cluster hierarchical model for subgroup borrowing in the design and analysis of basket trials with binary endpoints. Statistical Methods in Medical Research 29(9):2717–2732. doi: 10.1177/0962280220910186.
Daher, M. 2013. Ethical issues in the geriatric patient with advanced cancer “living to the end.” Annals of Oncology 24(Suppl 7):vii55–vii58. doi: 10.1093/annonc/mdt262.
DiNardo, C. D., B. A. Jonas, V. Pullarkat, M. J. Thirman, J. S. Garcia, A. H. Wei, M. Konopleva, H. Dohner, A. Letai, P. Fenaux, E. Koller, V. Havelange, B. Leber, J. Esteve, J. Wang, V. Pejsa, R. Hajek, K. Porkka, A. Illes, D. Lavie, R. M. Lemoli, K. Yamamoto, S. S. Yoon, J. H. Jang, S. P. Yeh, M. Turgut, W. J. Hong, Y. Zhou, J. Potluri, and K. W. Pratz. 2020. Azacitidine and venetoclax in previously untreated acute myeloid leukemia. The New England Journal of Medicine 383(7):617–629. doi: 10.1056/NEJMoa2012971.
__________________
4 See https://www.ema.europa.eu/en/human-regulatory/research-development/medicines-older-people#:~:text=Geriatric%20medicines%20strategy,-EMA's%20geriatric%20medicines&text=The%20strategy%20aims%20to%3A,on%20the%20use%20of%20medicines (accessed March 19, 2021).
Enewold, L., H. Parsons, L. Zhao, D. Bott, D. R. Rivera, M. J. Barrett, B. A. Virnig, and J. L. Warren. 2020. Updated overview of the SEER-Medicare data: Enhanced content and applications. Journal of the National Cancer Institute Monographs 2020(55):3–13. doi: 10.1093/jncimonographs/lgz029.
FDA (U.S. Food and Drug Administration). 2013. Best practices for conducting and reporting pharmacoepidemiologic safety studies using electronic healthcare data sets. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/best-practices-conducting-and-reporting-pharmacoepidemiologic-safety-studies-using-electronic (accessed April 11, 2021).
FDA. 2018. Meta-analyses of randomized controlled clinical trials to evaluate the safety of human drugs or biological products. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/meta-analyses-randomized-controlled-clinical-trials-evaluate-safety-human-drugs-or-biological (accessed April 11, 2021).
FDA. 2020. Inclusion of older adults in cancer clinical trials: Draft guidance for industry. FDA-2019-D-5572. https://www.fda.gov/media/135804/download (accessed March 24, 2021).
Fentiman, I. S., U. Tirelli, S. Monfardini, M. Schneider, J. Festen, F. Cognetti, and M. S. Aapro. 1990. Cancer in the elderly: Why so badly treated? The Lancet 335(8696):1020–1022. doi: 10.1016/0140-6736(90)91075-L.
Hershman, D. L., C. Till, S. Shen, J. D. Wright, S. D. Ramsey, W. E. Barlow, and J. M. Unger. 2018. Association of cardiovascular risk factors with cardiac events and survival outcomes among patients with breast cancer enrolled in SWOG clinical trials. Journal of Clinical Oncology 36(26):2710–2717. doi: 10.1200/JCO.2017.77.4414.
Hobbs, B. P., N. Chen, and J. J. Lee. 2018. Controlled multi-arm platform design using predictive probability. Statistical Methods in Medical Research 27(1):65–78. doi: 10.1177/0962280215620696.
Hurria, A., L. A. Levit, W. Dale, S. G. Mohile, H. B. Muss, L. Fehrenbacher, A. Magnuson, S. M. Lichtman, S. S. Bruinooge, E. Soto-Perez-de-Celis, W. P. Tew, M. A. Postow, H. J. Cohen, and the American Society of Clinical Oncology. 2015. Improving the evidence base for treating older adults with cancer: American Society of Clinical Oncology statement. Journal of Clinical Oncology 33(32):3826–3833. doi: 10.1200/jco.2015.63.0319.
Hurria, A., S. Mohile, A. Gajra, H. Klepin, H. Muss, A. Chapman, T. Feng, D. Smith, C. L. Sun, N. De Glas, H. J. Cohen, V. Katheria, C. Doan, L. Zavala, A. Levi, C. Akiba, and W. P. Tew. 2016. Validation of a prediction tool for chemotherapy toxicity in older adults with cancer. Journal of Clinical Oncology 34(20):2366–2371. doi: 10.1200/jco.2015.65.4327.
Hwang, T. J., L. Orenstein, A. S. Kesselheim, and F. T. Bourgeois. 2019. Completion rate and reporting of mandatory pediatric postmarketing studies under the U.S. Pediatric Research Equity Act. JAMA Pediatrics 173(1):68–74. doi: 10.1001/jamapediatrics.2018.3416.
IOM (Institute of Medicine). 2013. Delivering high-quality cancer care: Charting a new course for a system in crisis. Washington, DC: The National Academies Press.
Kearney, N., and M. Miller. 2000. Elderly patients with cancer: An ethical dilemma. Critical Reviews in Oncology/Hematology 33(2):149–154. doi: 10.1016/S1040-8428(99)00065-7.
Kearney, N., M. Miller, J. Paul, and K. Smith. 2000. Oncology healthcare professionals’ attitudes toward elderly people. Annals of Oncology 11(5):599–601. doi: 10.1023/a:1008327129699.
Kim, E. S., S. S. Bruinooge, S. Roberts, G. Ison, N. U. Lin, L. Gore, T. S. Uldrick, S. M. Lichtman, N. Roach, J. A. Beaver, R. Sridhara, P. J. Hesketh, A. M. Denicoff, E. Garrett-Mayer, E. Rubin, P. Multani, T. M. Prowell, C. Schenkel, M. Kozak, J. Allen, E. Sigal, and R. L. Schilsky. 2017. Broadening eligibility criteria to make clinical trials more representative: American Society of Clinical Oncology and Friends of Cancer Research joint research statement. Journal of Clinical Oncology 35(33):3737–3744. doi: 10.1200/jco.2017.73.7916.
Kim, E. S., T. S. Uldrick, C. Schenkel, S. S. Bruinooge, R. D. Harvey, A. Magnuson, A. Spira, J. L. Wade, M. D. Stewart, D. M. Vega, J. A. Beaver, A. M. Denicoff, G. Ison, S. P. Ivy, S. George, R. P. Perez, P. A. Spears, W. D. Tap, and R. L. Schilsky. 2021. Continuing to broaden eligibility criteria to make clinical trials more representative and inclusive: ASCO–Friends of Cancer Research joint research statement. Clinical Cancer Research. doi: 10.1158/1078-0432.CCR-20-3852.
Lancet, J. E., A. Hoering, G. L. Uy, J. E. Cortes, L. F. Newell, T. L. Lin, E. K. Ritchie, R. K. Stuart, S. A. Strickland, D. Hogge, S. R. Solomon, R. M. Stone, D. L. Bixby, J. E. Kolitz, G. J. Schiller, M. J. Wieduwilt, D. H. Ryan, M. T. Chiarella, A. C. Louie, and B. C. Medeiros. 2016. Survival following allogeneic hematopoietic cell transplantation in older high-risk acute myeloid leukemia patients initially treated with CPX-351 liposome injection versus standard cytarabine and daunorubicin: Subgroup analysis of a large phase III trial. Blood 128(22):906. doi: 10.1182/blood.V128.22.906.906.
Levit, L. A., H. Singh, H. D. Klepin, and A. Hurria. 2018. Expanding the evidence base in geriatric oncology: Action items from an FDA–ASCO workshop. Journal of the National Cancer Institute 110(11):1163–1170. https://doi.org/10.1093/jnci/djy169.
Lichtman, S. M., R. D. Harvey, M. A. Damiette Smit, A. Rahman, M. A. Thompson, N. Roach, C. Schenkel, S. S. Bruinooge, P. Cortazar, D. Walker, and L. Fehrenbacher. 2017. Modernizing clinical trial eligibility criteria: Recommendations of the American Society of Clinical Oncology–Friends of Cancer Research Organ Dysfunction, Prior or Concurrent Malignancy, and Comorbidities Working Group. Journal of Clinical Oncology 35(33):3753–3759. doi: 10.1200/JCO.2017.74.4102.
Ludmir, E. B., W. Mainwaring, T. A. Lin, A. B. Miller, A. Jethanandani, A. F. Espinoza, J. J. Mandel, S. H. Lin, B. D. Smith, G. L. Smith, N. A. VanderWalde, B. D. Minsky, A. C. Koong, T. E. Stinchcombe, R. Jagsi, D. R. Gomez, C. R. Thomas, Jr., and C. D. Fuller. 2019. Factors associated with age disparities among cancer clinical trial participants. JAMA Oncology 5(12):1769–1773. doi: 10.1001/jamaoncol.2019.2055.
Lyauk, Y. K., D. M. Jonker, and T. M. Lund. 2019. Dose finding in the clinical development of 60 U.S. Food and Drug Administration-approved drugs compared with learning vs. confirming recommendations. Clinical and Translational Science 12(5):481–489. doi: 10.1111/cts.12641.
Mohile, S. G., R. M. Epstein, A. Hurria, C. E. Heckler, B. Canin, E. Culakova, P. Duberstein, N. Gilmore, H. Xu, S. Plumb, M. Wells, L. M. Lowenstein, M. A. Flannery, M. Janelsins, A. Magnuson, K. P. Loh, A. S. Kleckner, K. M. Mustian, J. O. Hopkins, J. J. Liu, J. Geer, R. Gorawara-Bhat, G. R. Morrow, and W. Dale. 2020a. Communication with older patients with cancer using geriatric assessment: A cluster-randomized clinical trial from the National Cancer Institute Community Oncology Research Program. JAMA Oncology 6(2):196–204. doi: 10.1001/jamaoncol.2019.4728.
Mohile, S. G., M. R. Mohamed, E. Culakova, H. Xu, K. P. Loh, A. Magnuson, M. A. Flannery, E. E. Ramsdale, R. F. Dunne, N. Gilmore, S. Obrecht, A. Patil, S. Plumb, L. M. Lowenstein, M. C. Janelsins, K. M. Mustian, J. O. Hopkins, J. L. Berenberg, R. Gaur, and W. Dale. 2020b. A geriatric assessment (GA) intervention to reduce treatment toxicity in older patients with advanced cancer: A University of Rochester Cancer Center NCI community oncology research program cluster randomized clinical trial (CRCT). Journal of Clinical Oncology 38(15 Suppl):12009. doi: 10.1200/JCO.2020.38.15_suppl.12009.
NIH (National Institutes of Health). 2017. Inclusion across the lifespan. https://grants.nih.gov/policy/inclusion/lifespan.htm (accessed February 21, 2021).
O’Shaughnessy, J., M. Piccart, L. S. Schwartzberg, J. Cortes, N. Harbeck, S.-A. Im, H. S. Rugo, M. Untch, D. A. Yardley, I. Bondarenko, S. Chan, V. Dieras, M. D. Pegram, S. Kroll, J. P. O’Connell, J. Vacirca, T. Wei, K. Tang, and A. D. Seidman. 2019. CONTESSA: A multinational, multicenter, randomized, phase III registration study of tesetaxel plus a reduced dose of capecitabine in patients (pts) with HER2−, hormone receptor + (HR+) locally advanced or metastatic breast cancer (LA/MBC) who have previously received a taxane. Journal of Clinical Oncology 37(15 Suppl):TPS1107. doi: 10.1200/JCO.2019.37.15_suppl.TPS1107.
Pennell, N. A., M. Dillmon, L. A. Levit, E. A. Moushey, A. S. Alva, S. Blau, T. L. Cannon, N. R. Dickson, M. Diehn, M. Gonen, M. M. Gonzalez, J. O. Hensold, L. J. Hinyard, T. King, S. C. Lindsey, A. Magnuson, J. Marron, B. L. McAneny, T. M. McDonnell, K. F. Mileham, S. F. Nasso, G. S. Nowakowski, K. R. Oettel, M. I. Patel, D. A. Patt, J. Perlmutter, T. A. Pickard, G. Rodriguez, A. R. Rosenberg, B. Russo, C. Szczepanek, C. B. Smith, P. Srivastava, E. Teplinsky, R. Thota, T. A. Traina, R. Zon, B. Bourbeau, S. S. Bruinooge, S. Foster, S. Grubbs, K. Hagerty, P. Hurley, D. Kamin, J. Phillips, C. Schenkel, R. L. Schilsky, and H. A. Burris, 3rd. 2021. American Society of Clinical Oncology Road to Recovery report: Learning from the COVID-19 experience to improve clinical research and cancer care. Journal of Clinical Oncology 39(2):155–169. doi: 10.1200/jco.20.02953.
Singh, H., B. Kanapuru, C. Smith, L. A. Fashoyin-Aje, A. Myers, G. Kim, and R. Pazdur. 2017. FDA analysis of enrollment of older adults in clinical trials for cancer drug registration: A 10-year experience by the U.S. Food and Drug Administration. Journal of Clinical Oncology 35(15 Suppl):10009. doi: 10.1200/JCO.2017.35.15_suppl.10009.
Sinha, M. S., M. Najafzadeh, E. K. Rajasingh, J. Love, and A. S. Kesselheim. 2018. Labeling changes and costs for clinical trials performed under the U.S. Food and Drug Administration pediatric exclusivity extension, 2007 to 2012. JAMA Internal Medicine 178(11):1458–1466. doi: 10.1001/jamainternmed.2018.3933.
Surbone, A. 2008. Ethical considerations in conducting clinical trials for elderly cancer patients. Aging Health 4(3):253–260. https://doi.org/10.2217/1745509X.4.3.253.
Unger, J. M., C. A. Coltman, Jr., J. J. Crowley, L. F. Hutchins, S. Martino, R. B. Livingston, J. S. Macdonald, C. D. Blanke, D. R. Gandara, E. D. Crawford, and K. S. Albain. 2006. Impact of the year 2000 Medicare policy change on older patient enrollment to cancer clinical trials. Journal of Clinical Oncology 24(1):141–144. doi: 10.1200/JCO.2005.02.8928.
Wildiers, H., M. Mauer, A. Pallis, A. Hurria, S. G. Mohile, A. Luciani, G. Curigliano, M. Extermann, S. M. Lichtman, K. Ballman, H. J. Cohen, H. Muss, and U. Wedding. 2013. End points and trial design in geriatric oncology research: A joint European Organisation for Research and Treatment of Cancer–Alliance for Clinical Trials in Oncology–International Society of Geriatric Oncology position article. Journal of Clinical Oncology 31(29):3711–3718. doi: 10.1200/JCO.2013.49.6125.
Wolf, J., T. Seto, J. Y. Han, N. Reguart, E. B. Garon, H. J. M. Groen, D. S. W. Tan, T. Hida, M. de Jonge, S. V. Orlov, E. F. Smit, P. J. Souquet, J. Vansteenkiste, M. Hochmair, E. Felip, M. Nishio, M. Thomas, K. Ohashi, R. Toyozawa, T. R. Overbeck, F. de Marinis, T. M. Kim, E. Laack, A. Robeva, S. Le Mouhaer, M. Waldron-Lynch, B. Sankaran, O. A. Balbin, X. Cui, M. Giovannini, M. Akimov, and R. S. Heist for the GEOMETRY mono-1 Investigators. 2020. Capmatinib in MET exon 14-mutated or MET-amplified non-small-cell lung cancer. The New England Journal of Medicine 383(10):944–957. doi: 10.1056/NEJMoa2002787.
Zhu, H., J. Piao, J. J. Lee, F. Hu, and L. Zhang. 2020. Response adaptive randomization procedures in seamless phase II/III clinical trials. Journal of Biopharmaceutical Statistics 30(1):3–17. doi: 10.1080/10543406.2019.1657439.
DISCLAIMER: This Proceedings of a Workshop—in Brief was prepared by Erin Balogh, Anne Johnson, and Sharyl Nass as a factual summary of what occurred at the workshop. The statements made are those of the rapporteurs or individual workshop participants and do not necessarily represent the views of all workshop participants; the planning committee; or the National Academies of Sciences, Engineering, and Medicine.
*The National Academies of Sciences, Engineering, and Medicine’s planning committees are solely responsible for organizing the workshop, identifying topics, and choosing speakers. The responsibility for the published Proceedings of a Workshop—in Brief rests with the rapporteurs and the institution. The planning committee includes Monica Bertagnolli (Chair), Brigham and Women’s Hospital and Dana-Farber Cancer Institute; Gwen Darien, National Patient Advocate Foundation; R. Donald Harvey, Emory University School of Medicine; Samir N. Khleif, Georgetown University Medical Center; Heidi Klepin, Wake Forest School of Medicine; Larissa Nekhlyudov, Brigham and Women’s Hospital; Randall Oyer, Penn Medicine Lancaster General; Rebecca D. Pentz, Winship Cancer Institute and Emory University School of Medicine; Barbara Radziszewska, National Institute on Aging; Eric H. Rubin, Merck Research Laboratories; and Ishwaria Subbiah, University of Texas MD Anderson Cancer Center.
REVIEWERS: To ensure that it meets institutional standards for quality and objectivity, this Proceedings of a Workshop—in Brief was reviewed by Mary Lou Smith, Research Advocacy Network, and Martine Extermann, Moffitt Cancer Center. Leslie Sim, National Academies of Sciences, Engineering, and Medicine, served as the review coordinator.
STAFF: Rachel Austin, Erin Balogh, Lori Benjamin Brenig, Annalee Gonzales, Tracy Lustig, Sharyl Nass, Carolyn Shore, Micah Winograd, Health and Medicine Division, National Academies of Sciences, Engineering, and Medicine.
SPONSORS: This workshop was supported by the U.S. Food and Drug Administration.
For additional information regarding the workshop, please visit https://www.nationalacademies.org/our-work/improving-the-evidence-base-for-treatment-decision-making-for-older-adults-with-cancer-a-workshop.
Suggested citation: National Academies of Sciences, Engineering, and Medicine. 2021. Improving the evidence base for treatment decision making for older adults with cancer: Proceedings of a workshop—in brief. Washington, DC: The National Academies Press. https://doi.org/10.17226/26157.
Health and Medicine Division

Copyright 2021 by the National Academy of Sciences. All rights reserved.










