2
Prevention and Protective Factors
Multiple lines of evidence suggest that a large proportion of all dementia could be prevented, delayed, or slowed by social or behavioral changes. Researchers have not yet established, however, which specific risk factors are most important or how interventions to modify behaviors and conditions could have the greatest impact on dementia. Many aspects of an individual’s life—socioeconomic resources; education level; health-relevant behaviors, including diet and exercise patterns; trauma; medical and psychiatric conditions; and characteristics of the physical and social environment—play a role in dementia and intersect with genetic risks. Yet while social and behavioral factors may influence the risk of developing dementia or the progression of disease, their impacts are not inevitable. Modifications, either at the individual level (through changes in behaviors or exposures) or at the population level (through changes to local, state, or federal policies or reorganization of institutional regulations, programs, and practices) may affect outcomes.
Although clear causal relationships are challenging to establish definitively, some researchers have suggested that as much as 40 percent of dementia may be attributable at least in part to modifiable risk factors (Barnes and Yaffe, 2011; Norton et al., 2014; Livingston et al., 2020).1 Identifying a firm percentage would be challenging, however, because dementia risk is associated with many factors beginning very early in life,
___________________
1 Modifiable risk factors account for an additional portion of the overall impact of dementia that is not explained by either identified genetic or identified environmental factors (Livingston et al., 2020).
which vary significantly across population groups. Although some genetic factors are important, healthier social and behavioral patterns predict lower dementia risk, virtually regardless of genetic background (Lourida et al., 2019).2 Overall, these estimates are approximate at best and fail to account for the joint impact of multiple correlated risk factors or changes in the pattern of risk factors over time, as discussed below. What is important to note is that prevention efforts can alter the risk of dementia in any population, at any time of life.
Several public health initiatives to modify harmful influences on health—including reductions in the prevalence of smoking (leading to extraordinary declines in cardiovascular disease), reductions in motor vehicle crash fatalities per mile traveled, and reductions in exposure to lead—have been quite successful (Ruiz-Hernandez et al., 2017). Although these initiatives are by no means completed and often were slow to initiate, they have yielded tremendous public health gains and offer insight into how to approach complex public health problems (Gielen and Green, 2015). These successful campaigns highlight the importance of targeting multiple levels of influence, as was done, for example, in the campaign to reduce smoking, which targeted both individual behavior (e.g., smoking cessation classes) and factors at the population or systems level (e.g., cigarette taxes and bans on smoking in public spaces).3
Successes in combating cardiometabolic conditions are especially relevant because many types of dementia are influenced by mechanisms or processes that contribute to other diseases (e.g., vascular changes, metabolic dysregulation, inflammation). A substantial proportion of cases of late-onset dementia (defined as dementia symptoms starting at age 65 and older [McMurtray et al., 2006]) reflect the combined effects of mixed pathologies, such as amyloid and tau deposition and vascular changes. Improved understanding of the relationships among these factors and how to exploit them to slow disease progression, reduce the severity of disease, or prevent or delay disease in some people could be an important means of reducing the incidence and severity of dementia (Schneider et al., 2009; Barnes et al., 2015; Kawas et al., 2015). Likewise, such population trends as the obesity epidemic and associated increasing prevalence of diabetes are relevant for anticipating future increases in the risk of dementia.
This chapter examines the state of the research on prevention and risk factors for dementia, beginning with an overview of the nature of the
___________________
2 This statement does not apply to rare autosomal-dominant dementias that occur early in life.
3 See Frieden (2010) for discussion of the tension between interventions that require increasing individual effort (e.g., counseling, education, or clinical interventions) and interventions that are likely to have large population impact (e.g., socioeconomic factors or systems changes to make healthy choices the default).
available evidence. The chapter then looks in detail at two primary lenses for thinking about prevention. First, it explores evidence about how various factors affect cognitive health and disease in individuals, including evidence about preventive strategies and policies that have been pursued in targeting chronic diseases that are medically linked to dementia, such as cardiovascular disease and diabetes. The chapter then turns to the evidence about broader social and environmental factors and policies that play a role in increasing or diminishing risk over the life span.
INTERPRETING THE EVIDENCE
In assessing the evidence about risk and protective factors for dementia, the committee was able to rely on systematic reviews, meta-analyses, and the work of previous National Academies of Sciences, Engineering, and Medicine committees that have synthesized relevant research. In general, these syntheses make clear that the body of evidence provided by randomized controlled trials (RCTs) is limited. RCTs are regarded as providing the highest-quality evidence for establishing a causal link between an exposure and an outcome in at least some people. But very few RCTs targeting behavior change and including follow-up for dementia or related outcomes have been carried out. Moreover, existing RCTs in this area are often underpowered, involving modest sample sizes, relatively brief interventions, and short follow-up periods. The findings they yield are sometimes inconsistent across trials and between outcomes within the same trial. And even when RCTs are available, they often do not reflect the demographic characteristics or health status of the broader population, and so must be augmented with evidence on heterogeneity of effects and generalizability to other populations. There are also many situations in which conducting RCTs can be considered unethical, such as when the intervention is known to be beneficial for another outcome and it is not offered to one group. And in other settings, RCTs are infeasible because the time periods over which risk factors are thought to operate are so long.
A significantly larger body of observational (non-RCT) data implicates several factors associated with subsequent dementia risk. However, many of these observational studies are vulnerable to at least one of two sources of bias. One is “confounding,” the term used when an association is ascribed to one factor, the putative cause, but is driven by another factor that is associated with both the putative cause and the outcome. For example, cognitive activity appears to reduce the risk of dementia, but it is possible that some or all of the apparent effect may result from the association of cognitive activity with other protective factors, such as education. Researchers address this problem by using analytic adjustment for such confounding factors, but residual or unmeasured confounders are always
a concern in observational studies. A second potential source of bias is reverse causation—when early symptoms of the disease outcome lead to changes in the apparent risk factor rather than the other way around. For example, mild cognitive changes could make cognitive activities less enjoyable and thus decrease participation, leading to a spurious impression that lower cognitive activity increases the risk of dementia (Floud et al., 2021; Sajeev et al., 2016). Similarly, while social isolation may be a risk factor for dementia, incipient dementia may also lead to social isolation.
In addition to these two forms of bias, other challenges need to be considered when evaluating observational studies, including
- difficulties measuring cognition, especially subtle cognitive changes or change in those with very high or low levels of education, and while this is true in all dementia research, in observational studies it can interact with the hypothesis or a key covariate and lead to bias;
- selective survival, or differences in the characteristics of populations that survive to older ages that can lead to spurious statistical associations;
- a lack of diversity in the samples or nonrepresentative samples of the population;
- short follow-up periods in many studies, particularly given that many risk factors are thought to contribute to risk over long periods of time;
- selective recruitment into and retention in research studies, leading to both uncertainty (because of small sample sizes) and spurious associations (caused by selection bias); and
- testing of multiple hypotheses without correction for the multiplicity of tests.
These concerns are well known, but biases can be minimized, quantified, or possibly avoided with appropriate study designs and analytical tools, and ancillary evidence may aid in the interpretation of results from observational studies. Nevertheless, biases and the other challenges outlined above remain a significant concern if observational findings are to be translated into preventive interventions. These issues are discussed further in Chapter 8.
INFLUENCES ON COGNITIVE HEALTH IN INDIVIDUALS
Researchers have explored factors that may affect the risk of dementia directly, influences on cardiovascular health that in turn have implications for cognitive health, and other possible culprits. This section explores the evidence and reviews the implications for individuals and public health experts.
Evidence About Risk and Protection for Cognitive Health
Interest in preventing or minimizing the impact of dementia has stimulated a wealth of research, as well as the development of many hypotheses and recommendations to the public. Expert advice is neither unanimous nor conclusive, however. Different experts who have assessed the available work have reached somewhat different conclusions as a result of decisions about which type of evidence to focus on or how to weigh the evidence. Two National Academies’ committees and a group writing for the medical journal The Lancet have reviewed the available research and summarized their conclusions about preventive factors. These summaries offer a clear picture of the current state of the research.
As a backdrop, a 2015 National Academies’ report titled Cognitive Aging: Progress in Understanding and Opportunities for Action focuses not on preventing such diseases as dementia but on optimal cognitive aging overall. The report summarizes ways to support cognitive health and functioning as people experience the “process of gradual, ongoing, yet highly variable changes in cognitive functions that occur as people get older” (Institute of Medicine, 2015, p. 2). The authors offer broad recommendations about steps individuals can take to support their cognitive health (p. 7):
- Be physically active.
- Reduce and manage cardiovascular disease risk factors (including hypertension, diabetes, and smoking).
- Regularly discuss and review health conditions and medications that might influence cognitive health with a health care professional.
The report also endorses some actions more cautiously, indicating that they “may” promote cognitive health:
- Be socially and intellectually engaged and engaged in lifelong learning.
- Get adequate sleep and receive treatment for sleep disorders if needed.
- Take steps to avoid the risk of cognitive changes due to delirium if hospitalized.
The authors suggest careful evaluation of products advertised to consumers for improving cognitive health, such as medications, nutritional supplements, and cognitive training.
The willingness of these authors to recommend these actions likely was based on the observation that they have other potential benefits (e.g., prevention of cardiovascular disease) and are certainly unlikely to harm anyone, rather than on the strength of evidence that they ameliorate cognitive aging per se. The authors also reviewed evidence about other factors, such
as exposure to pollution, tobacco smoke, and stress; diet; and such conditions as hearing loss and depression, but found it difficult to draw conclusions about this evidence because of variation in the available studies and the paucity of studies for some topics. In general, the quality of evidence was such that it was difficult to support recommendations for anything one might not already recommend for other reasons.
2017 National Academies’ Report
The 2017 National Academies of Sciences, Engineering, and Medicine report Preventing Cognitive Decline and Dementia: A Way Forward summarizes the evidence about interventions that may be effective in preventing dementia and other types of cognitive decline (National Academies of Sciences, Engineering, and Medicine [NASEM], 2017). The report examines evidence that changes in the brain associated with dementia and other forms of cognitive impairment may begin many years before they are expressed as symptoms. This evidence provides reason to think that interventions implemented long before a person is impaired could have significant impact and that making changes decades before the typical age of onset may even be essential. This possibility is supported by the data showing declines in both the incidence and prevalence of dementia in high-income countries (see Chapter 1), suggesting that shifts in risk factors have influenced dementia risk.
The authors of the 2017 report coordinated their work with a systematic review commissioned by the Agency for Healthcare Research and Quality (AHRQ), which focused on evidence from RCTs. However, they note the difficulty of conducting this type of investigation on interventions targeting conditions that develop later in life and often in conjunction with other medical conditions. For example, they observe that the National Institute on Aging, the primary funder of research related to dementia, generally funds only research on older adults, which would not encompass risk factors that occur in earlier phases of the life course. Because of these challenges, the report’s authors found very few randomized studies that could support public health recommendations. They supplemented their work with a review of observational data, studies of risk factors, and assessments of the possible effects of interventions on the body, but also note the limitations of these studies.
The authors found that three types of interventions are “supported by encouraging although inconclusive evidence” (NASEM, 2017, p. 7):
- cognitive training—a broad set of interventions, such as those aimed at enhancing reasoning, memory, and speed of processing—to delay or slow age-related cognitive decline;
- blood pressure management for people with hypertension to prevent, delay, or slow clinical Alzheimer’s-type dementia; and
- increased physical activity to delay or slow age-related cognitive decline.
The report notes that methods of cognitive training—including structured exercises designed to improve reasoning or problem solving, boost memory, and increase processing speed, as well as cognitively stimulating activities, such as learning a new language or playing challenging games—can improve performance on the task involved, at least in the short term. But the authors found only limited or mixed evidence about whether such benefits would translate to improved capacity on other cognitive tasks, to general cognitive functioning over time, or to decreased risk of dementia.
With respect to hypertension, the authors cite research that has established multiple connections between dementia and the health of the brain’s vascular system. Atherosclerosis in blood vessels in the brain, microbleeds, and silent strokes, for example, have been identified as contributors to dementia. It is possible that vascular risk factors increase dementia risk by other mechanisms as well (e.g., decreased blood flow in the brain may lessen the body’s capacity to clear Alzheimer’s disease proteins or increase their production). In any event, reducing the risk of stroke or other cerebrovascular disease could plausibly reduce the risk of dementia. Since the publication of this 2017 National Academies’ report, results reported from the SPRINT-MIND RCT have demonstrated that more aggressive systolic blood pressure management led to a lower risk of dementia and mild cognitive impairment (SPRINT-MIND Investigators et al., 2019).
Physical activity, including aerobic activities, resistance or weight training, and stretching, is generally recognized as important for healthy aging, sustaining physical functioning and reducing the risk of cardiovascular disease. While there is substantial observational evidence to support the hypothesis that physical activity may reduce the risk of cognitive decline or dementia, the 2017 report’s authors (drawing on a previous systematic review from AHRQ) found that the data from intervention studies designed to confirm this effect remained sparse. Most studies the authors identified were not of sufficient duration or size to detect plausible effect sizes. Other study designs (e.g., isolating different types of physical activity) might have identified truly heterogeneous effects.
The caution that characterizes the conclusions of the 2017 report reflects the authors’ strict filters for the types of evidence on which they would rely, particularly the decision to accord the greatest weight to evidence from RCTs. The authors also highlight the need for further research and methodological improvements to build understanding of the differences among populations and other issues.
Lancet Commission Report
A 2020 Lancet Commission report also summarizes the evidence on dementia prevention and possible interventions, offering a more expansive view of the possibilities for prevention (Livingston et al., 2020). The authors interpreted a wide array of expert opinion in building on findings from their own 2017 report on the subject, which identified nine potentially modifiable risk factors for dementia that could account for approximately 35 percent of dementia cases (Livingston et al., 2017). By 2020, Livingston and colleagues had found the evidence to be stronger and identified three additional factors for which they found recent evidence to be compelling. The resulting set of 12 factors is
- lower education levels,
- hypertension,
- hearing impairment,
- smoking,
- obesity,
- depression,
- physical inactivity,
- diabetes,
- low social contact,
- excessive alcohol consumption,
- traumatic brain injury, and
- air pollution.
These lifestyle behaviors are linked to the development of other diseases, particularly cardiovascular disease, and may also be linked to dementia risk. For example, cigarette smokers have been found to be at higher risk for developing dementia relative to those who do not smoke. Thus, targeting these lifestyle risk behaviors has the dual benefit of reducing the risk of common chronic diseases while likely reducing the risk of dementia as well.
The authors found that these 12 factors collectively “account for around 40 percent of worldwide dementias,” meaning that that proportion of dementias could be “prevented or delayed” with intervention (Livingston et al., 2020, p. 413). This estimate should be interpreted with caution, however, because it depends on the current distribution of risk factors in a population and overall population risk, which differ across current demographic and other population features and are expected to change over time. In addition, these calculations do not take into account the simultaneous impact of multiple risk factors, many of which are highly correlated. In any event, the authors suggest that the potential benefits are likely to be highest in low-income countries.
The report advocates for broad prevention efforts, such as providing all children with primary and secondary education and reducing exposure to air pollution. It also makes very specific recommendations, such as maintaining a systolic blood pressure of 130 mm Hg or less from age 40. The recommendation regarding blood pressure likely reflects the influence of the above-mentioned clinical trial data from the SPRINT-MIND study, in which tighter blood pressure control was associated with better outcomes. However, it should be noted that data specific to older individuals, for whom lower blood pressures may not be as well tolerated, were limited (Yaffe, 2019; SPRINT-MIND Investigators et al., 2019).
The authors of this 2020 Lancet Commission report based their conclusions on observational studies and expert opinion, with limited evidence from experimental or even quasi-experimental studies. As noted above, these observational studies are vulnerable to two important sources of bias—confounding and reverse causation—as well as measurement errors and selection bias. Because some recommendations based on observational evidence appear to be innocuous and may have ancillary benefits, the assumptions under which that evidence would support causal inferences may be evaluated less rigorously.
Potentially Important Risk Factors That Have Received Less Attention
The role of medications and polypharmacy receives little attention in the reports discussed above but may be important. An estimated 85 percent of adults ages 65 and older live with at least one chronic condition, and nearly 60 percent have two or more such conditions. The conditions associated with dementia risk (e.g., diabetes, hyperlipidemia, depression) are among the most prevalent, and most older Americans use drug therapies to treat them. There are questions about whether some drug therapies themselves may be associated with that risk, even though they offer the benefits of treating a condition that increases dementia risk. Some drugs may also interact with dementia-related pathophysiological pathways by way of mechanisms unrelated to their original therapeutic indication. For example, such drugs as benzodiazepines (used for anxiety), antispasmodics (for overactive bladder), and anticholinergics (used for a variety of conditions and also present in over-the-counter sleep medications) have been associated with increased risk of dementia (Barthold et al., 2020; Chatterjee et al., 2020; see Thunell et al., 2021, for an overview of research on the relationship between pharmaceuticals and dementia risk).4 However, the caveats
___________________
4 In the case of benzodiazepines for anxiety, the association may be driven by reverse causality, as anxiety can be a prodromal symptom of dementia.
noted above regarding confounding, reverse causation, measurement error, and selection bias should be noted regarding some of these findings.
The physical environment also likely influences dementia risk. While the effects of specific environmental factors on dementia risk are generally not well understood, there is compelling evidence that a wide variety of toxic exposures are influential (Finch and Kulminski, 2019; Mortamais et al., 2021). For example, female participants in the Women’s Health Initiative Memory Study who were exposed to air pollution above the risk standard identified by the Environmental Protection Agency in 2012 had nearly twice the risk of dementia compared with their counterparts who did not have that exposure (Cacciottolo et al., 2017). Similarly, exposure to lead is associated with dementia risk. As climate change increases temperature extremes and volatility and such adverse events as wildfires, these effects are likely to influence the risk of incident dementia and the well-being of people living with dementia (Wei et al., 2019; Milton and White, 2020). Because environmental risk factors are unequally distributed in terms of geographic location, socioeconomic conditions, and other social factors, research on this topic needs to be linked with the research on disparities discussed below.
Significant research and funding have also been devoted to identifying modifiable risk factors that may help prevent cancer, hypertension, and other diseases. As noted above, cardiovascular diseases themselves may increase the risk of dementia; moreover, many of the risk factors that have been studied appear to affect the risk of multiple diseases, including dementia. Thus, the reduction of risk factors for these other diseases can contribute to reducing the risk for dementia. Successful efforts to reduce risk for these diseases can also offer insights for public health activities targeting dementia directly. Cardiovascular disease in particular may contribute to dementia risk, and relationships among major chronic conditions, including hypertension, heart disease, and stroke, and dementia heighten the importance of attention to modifiable factors that affect risk.5
Use of Emerging Evidence to Promote Public Health
The major challenge related to findings such as those reported above has been identifying ways to act on the evidence and change long-term behaviors, which may involve addictive substances or strong social norms and are constrained by built and social environments, as well as socioeconomic resources. There is little evidence to suggest that telling individuals to change their behavior will bring about enduring behavior change in most
___________________
5https://www.ninds.nih.gov/Disorders/Patient-Caregiver-Education/Preventing-Strokeresearch
of the population. However, a look at the campaign to reduce tobacco use illustrates the possibilities for altering harmful individual behaviors. Smoking declined by 58 percent among adults between the 1960s and the early 2000s, and this campaign has been identified as one of the most successful public health efforts of the 20th century (Institute of Medicine, 2007) and a major driver of declines in lung cancer and cardiovascular disease (Lu et al., 2019). As noted earlier, the campaign to reduce smoking, which is still ongoing, demonstrates the importance of targeting multiple levels of influence for prevention, including individual behavior change and population- or systems-level factors. Other major public health achievements, such as reductions in motor vehicle crashes and lead exposure, were also achieved through multiple levels of intervention. Another successful example is a clinical trial testing the effectiveness of lifestyle interventions for preventing diabetes—the Diabetes Prevention Program6—and the program’s successful dissemination in a wide variety of settings and populations (Jiang et al., 2013; Ackermann et al., 2015).
The campaign to reduce tobacco use was a massive effort based on strong evidence for the causal relationship between smoking and serious adverse health outcomes. In general, sound decisions about devoting resources to such interventions rest on solid evidence that
- the change being promoted has the capacity to reduce risk,
- changing the behavior or environment to a degree likely to have a significant effect is feasible and sustainable, and
- there is a tested means of effecting the change that could work in the intended setting or circumstances.
There is evidence that interventions can improve or maintain cognitive function in older individuals (see, e.g., Ngandu et al., 2015). But in the context of health-related behaviors with possible implications for dementia risk, it is important to weigh a variety of competing considerations. The causal role of some risk factors that have been linked with dementia remains uncertain, and the estimated impact of any one individual risk factor may be small. In other words, the fraction of dementia cases that could potentially be prevented if it were possible to convince everyone to adopt a particular behavior might be small. Researchers also have not yet been able to establish whether combined risk factors have multiplicative effects. Thus, it would be reasonable to prioritize efforts to modify behaviors by considering the feasibility of changing those risk factors, the opportunity cost of such changes (given that most would have impact if achieved in
___________________
middle age or earlier), and the potential population impact of such changes on the overall incidence of or disparities in dementia.
Conveying the above uncertainties clearly so that individuals can make informed decisions about behavioral changes will be key to the usefulness of behavioral interventions. The greatest success may be achieved with interventions that are collaborative rather than prescriptive and help people decide what they believe is worth doing and how to do it if they are interested. A holistic perspective that considers the potential impact of a behavior change on overall health and quality of life may be key to helping individuals navigate these types of decisions.7 Interventions that target systems or structures to make healthy behaviors the default (e.g., active transportation options, policies that subsidize or increase access to healthy dietary patterns or tax or decrease access to unhealthy ones) are likely to be especially valuable.
The impact of such behavior changes on common risk factors could be substantial for a population, even if individual-level benefits were small. However, the population-level effects of behavior changes that reduce the risk of cardiovascular and other health conditions associated with dementia could also bring corresponding changes in population mortality risk. If reduced risk in associated health conditions resulted in longer lives, there could be a corresponding increased cumulative lifetime risk of dementia as more people reached older and older ages (Zissimopoulos et al., 2018). Thus, from a population perspective, it would make sense to base action on improved understanding of the point in the life course at which interventions would both improve population health and longevity and reduce the cumulative lifetime risk of dementia.
SOCIOECONOMIC RISK
The advances in understanding of how individual behaviors may influence the development of dementia and related diseases discussed in the first part of this chapter offer valuable benefits, but researchers have also looked more broadly at influences on risk. Work that has emerged in the past few decades from economics, epidemiology, and neuropsychology, among other disciplines, has substantially broadened understanding of how social, environmental, and economic factors contribute to risk, as well as how racism and racial discrimination have interacted with those factors to amplify risk for certain groups.
A full understanding of these influences starts with a look at the entire life course, as noted in Chapter 1. Beginning in infancy and early childhood,
___________________
7 For an example of this type of communication, see https://siteman.wustl.edu/prevention/ydr.
such factors as health and nutrition, material well-being, social ties, stressful experiences, and education affect how an individual develops physically, cognitively, and emotionally in ways that, decades later, can have substantial impacts on cognitive health (see, e.g., Zhang et al., 2010; Lövdén et al., 2020; Sharp and Gatz, 2011; Jirout et al., 2019). As the individual ages, health behaviors, leisure activities, and factors associated with an array of social advantages and disadvantages continue to shape later cognitive health outcomes (see, e.g., Bowling et al., 2016; Nelson et al., 2020; Arpino et al., 2018). And, as discussed above, the development of such adult disease conditions as diabetes and stroke, many of which are themselves associated with these social factors, is associated with risk of dementia.
It is increasingly clear that factors operating at multiple geographic levels (neighborhood, city, state) and through such social institutions as school, workplaces, and houses of worship have the potential to modify the associations between risk factors and both the incidence and progression of dementia. It is also important to examine who lives in a particular environment and why (e.g., racial segregation), and the characteristics of an area (e.g., levels of crime, availability of such resources as health care providers and support networks, exposures to such toxins as air pollution) that may also be influential.
Taking a life-course approach sensitizes researchers to the importance of historical factors (e.g., technological developments, policies, and such cultural forces as racism). For example, the so-called Jim Crow laws that enforced segregation in the South were not ended until the Civil Rights Act passed in 1965. Thus, many Black Americans alive today were born into a racialized caste system that substantially affected their lives: they attended primary schools legally segregated by race, which were systematically underresourced, and experienced higher rates of childhood adversity compared to White populations (Zhang et al., 2016). Indeed, de facto segregation persists in many American localities, and despite minor advances toward racial equity, the United States can still be described as a racialized caste system (Wilkerson, 2020). The effects of these experiences will likely continue to contribute to racial disparities in dementia risk for years to come (see, e.g., Zuelsdorff et al., 2020; Coogan et al., 2020; Caunca et al., 2020). For instance, analyses of the results of the Health and Retirement Study between 1998 and 2010 found that higher rates of childhood adversity among Black populations put Black adults at significantly greater risk of cognitive impairment in later life (Zhang et al., 2016).
Figure 2-1 illustrates the life-course approach to the development of dementia. The figure highlights two major phases of the human life course—the developmental phase and the aging phase. The developmental phase corresponds roughly to childhood, when a substantial amount of cognitive and brain development occurs. During the aging phase, general
declines in functioning occur in healthy individuals; neurodegeneration and pathology can lead to dementia when a threshold is crossed in terms of the individual’s comparative cognitive functioning.
The figure roughly illustrates how the effects of experience and environmental exposures compound over a lifetime, pushing in both positive and negative directions. The blue curve represents possible trajectories in the course of a lifetime and shows that the individuals who are most affected by risks across the life span may have significantly poorer cognitive health after decades of such exposures and experiences, relative to their counterparts who experience fewer risks and much greater protections. In other words, outcomes diverge significantly as people age because their cumulative experiences may combine either to support cognitive health or to make dementia more likely.
The figure significantly oversimplifies these processes. An individual’s trajectory is not likely to follow a smooth arc, and it is difficult to portray the interplay of influences and effects that occur across the life course, such as exposures or learning occurring later in life. What is too complex to represent in a figure is what lies behind the differences in exposures: the social, environmental, and economic factors that multiply the risks for certain groups and afford cushions of protection for others. Nevertheless, it highlights the critical influence of environment and experience on cognitive health.
The precise biological, behavioral, and psychosocial mechanisms through which such life-course risk factors influence dementia and the timing of greatest influence are not clearly established. Researchers have posited numerous possible pathways, including cellular (e.g., neuroplasticity), behavioral (e.g., physical activity), material (e.g., lead exposure), and medical (e.g., hypertension control) mechanisms. For example, children who grow up in poverty may experience nutritional deprivation, which may harm brain development. Growing up in poverty may also limit an
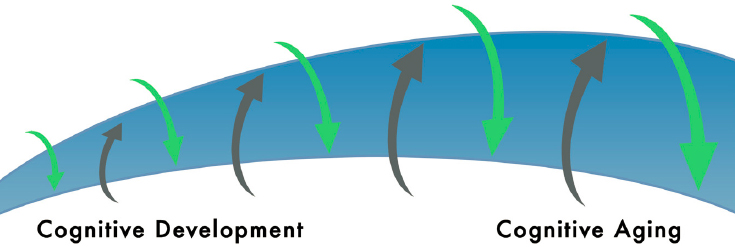
NOTE: The upward arrows represent experiences and exposures that protect cognitive health; the downward arrows represent risk factors that may impair it.
individual’s employment opportunities in adulthood and therefore also limit access to high-quality medical care that would manage hypertension—increasing the risk of dementia. And both pathways contribute to risk. Such hypotheses are plausible, but establishing their relative impact is not easy.
Pinpointing such connections is challenging in part because ways of measuring mediators (factors that explain the links among variables) in this context are not well developed (see Chapter 8). For example: What mediates the association between years of education in early life and dementia in late life? Does education have a direct effect on brain development, or does its impact come through its effects on income, access to health care, health behaviors, or other factors, or some combination of these and other factors? Many questions about how factors combine over the life course to influence the risk of dementia have not been answered. For example, few of the existing studies of associations among these factors were designed to address questions about the accumulation of risk from childhood through adulthood. Emerging data sources along with improved statistical methods will support progress in understanding these mechanisms in the coming decade, as discussed in Chapter 8.
Although much more work is needed to understand these issues, there is a growing body of work on three socioeconomic factors that influence dementia risk: education, occupation, and financial resources. There is also a growing literature on the role of race and ethnicity in disparities in dementia prevalence and incidence. These factors are discussed in turn below.
Socioeconomic Factors
Policies to alter socioeconomic conditions, including employment, education, and financial security, hold the potential to bring substantial benefits to the cognitive health of future cohorts of people as they age. Because policy remedies are expensive, however, clear evidence that they could have meaningful impact on dementia would be important. Improving understanding of the effects of socioeconomic factors on dementia will also contribute to understanding of the effects of other modifiable risk factors because nearly every behavior (physical activity, diet, alcohol use, smoking) is influenced by socioeconomic conditions. Yet few studies of these issues have included measures of socioeconomic conditions that would make it possible to disentangle such related factors, and there is limited systematic information on the magnitude of bias potentially introduced by this omission. A detailed exploration of these areas was beyond the scope of this study, but the issues related to education, occupation, and financial resources are briefly discussed below.
Education
The contribution of educational inequities in early life to disparities in the incidence of dementia in later life is perhaps the best studied of the socioeconomic factors (Walsemann and Ailshire, 2020). The causal evidence linking education and dementia risk includes both observational and quasi-experimental findings in many settings. For example, a systematic review of studies of the effects of lower education on risk for dementia showed a significant relationship (Sharp and Gatz, 2011). A study that explored the effects of changes in rules about compulsory schooling showed that individuals who completed more years of school had better cognitive outcomes and lower dementia risk decades later (Nguyen et al., 2016). And a meta-analysis of dose-response and dementia suggests that higher education significantly reduces dementia risk (Xu et al., 2016). Similar results from another longitudinal study indicated that these associations were robust when models controlled for indicators of childhood IQ (Wolters et al., 2020).
This finding points to a promising area for further study. Cohorts now reaching old age benefited from dramatic increases in educational opportunity and attainment since the mid-20th century—increases in K–12 enrollment, high school graduation, and college enrollment and graduation, for example (National Center for Education Statistics, 1993)—and recently documented declines in age-specific dementia incidence have been linked to educational improvements (Hayward et al., 2021).
Education may offer protection from dementia in several ways. It may strengthen cognitive reserve, the brain’s ability to optimize or maximize performance by recruiting different brain networks and tapping alternative cognitive strategies (Stern, 2009). It may also prevent the progressive pathophysiologic processes that lead to dementia (e.g., cerebrovascular disease or amyloid deposition) or offer protection in other ways. Research on the protection that may be afforded by education has explored links between cognitive decline and educational attainment in varied settings and at varied stages of the life cycle (see, e.g., Crimmins et al., 2018; Seblova et al., 2019; Lövdén et al., 2020; Weden et al., 2018).
However, there is some inconsistency across studies, which may be the result of chance or sampling or measurement issues, or may reflect limits on the power of education to protect an individual from other negative forces. For example, despite significant improvements in the quality of and access to education in the United States in the 1960s and 1970s, race-based discrimination often prevented Black men from obtaining jobs appropriate to their education levels, thus denying them many of the social benefits of education (Hayward et al., 2021). Some evidence suggests that although education may strengthen an individual’s cognitive level, later-life
influences, such as level of income or wealth, have greater impacts on the rate of cognitive change (Glymour and Manly, 2008; Marden et al., 2017). Finding a factor that is theoretically modifiable throughout life, such as income level, affects cognitive health would be useful, as it would point to opportunities for intervention.
Although education may seem a simple concept to measure, it is a multifaceted construct, and each of those facets is challenging to measure. Nearly all evidence on the effects of education is based on easily quantifiable measures, such as test scores, number of years completed, or major credentials (see, e.g., Zahodne et al., 2015). However, more difficult-to-measure aspects of educational quality are likely as important for cognitive development and health. At the same time, there is strong evidence that some educational assets—such as high-quality preschool—have multiple benefits, such as for social and emotional development, academic attainment, and earnings, that are observable even decades later (see, e.g., Child Trends, 2018). On the other hand, there is surprisingly little evidence on the benefits of educational experiences people have later in life, although such experiences are quite common. These same ambiguities apply to nearly every other social determinant of health, such as work and retirement, financial resources, and social networks and support.
Occupation
Employment and occupation merit special consideration because of the multiple mechanisms through which they may influence dementia risk. Researchers have examined the effects of types of occupation, occupational autonomy, stress, and unemployment. For example, a study of work during midlife and later dementia risk found that mentally stimulating or complex work is associated with lower risk of dementia and may even compensate for the risks that come with lower educational attainment (Karp et al., 2009).
There is growing evidence that retirement is associated with cognitive deterioration, increasing the risk of dementia (Karp et al., 2009; Rohwedder and Willis, 2010; Celidoni et al., 2017). However, individuals’ responses to retirement vary significantly, by number of years in retirement, occupation, and postretirement environment (Denier et al., 2017). Assessing the role of retirement is also challenging because of the possibility of reverse causality (early cognitive decline may cause someone to retire early), as well as selection bias (those with physically demanding jobs may be more likely to retire early). Because retirement age reflects both personal preferences and policy interventions, evidence on how retirement influences dementia risk and what aspects of postretirement life may reduce the risk would be valuable. The link between education and cognition is sometimes interpreted as
support for the “use it or lose it” hypothesis of cognitive aging, that is, the idea that cognitive demands are important for maintaining cognitive health (Denier et al., 2017). If so, enhancing retirement from the paid labor force with alternative cognitively engaging activities may provide opportunities for reducing cognitive decline.
Financial Resources
A factor related to many others that have possible effects on cognitive health is inequality in wealth and income. Establishing direct links between cognitive health and poverty, economic hardship, and financial security would open intriguing possibilities for population-level interventions. The pernicious effects of poverty on human health and well-being have been documented, but policy responses are still emerging (see, e.g., NASEM, 2019a, 2019b).
Racial/Ethnic Disparities in Dementia Risk
As noted in Chapter 1, significant disparities across racial/ethnic groups in dementia prevalence and incidence persist in the United States, although the magnitude of these disparities has shifted over time (Matthews et al., 2019; Mayeda et al., 2016). Box 2-1 summarizes some key recent evidence documenting these disparities in dementia outcomes.
The magnitude of racial/ethnic disparities in dementia risk and the heterogeneity of these disparities across subpopulations point to the potential impact of social and interpersonal experiences on people’s risk for dementia. Multiple explanations for the disparities in dementia risk have been posited, many of which emphasize structural racism (Zhang et al., 2016; Plassman et al., 2007). For example, researchers studying racial/ethnic differences in dementia risk found that environmental factors and social determinants of health could be responsible for inequities in dementia risk (Plassman et al., 2007; Yaffe et al., 2013). Structural racism that has been embedded in policies and laws as well in the delivery of medical care has contributed to disparities across health outcomes (Bailey et al., 2021; Park and Chen, 2020). Disparities in access to care, quality of care, and health outcomes of care for communities of color in the United States all likely contribute to disparities in dementia risk and outcomes (Chen and Zissimopoulos, 2018; Lines et al., 2014; Werner, 2019). In a 2021 survey, 36 percent of Black Americans, 18 percent of Hispanic Americans, and 19 percent of Asian Americans reported that discrimination is a barrier to receiving dementia care and that they expected to be treated differently because of their race, color, or ethnicity (Alzheimer’s Association, 2021).
Respondents also reported that it is more difficult for them to get excellent dementia care, and caregivers also reported that they had witnessed racial discrimination in their recipient’s health care settings.
Researchers have focused on effects of the interlocking processes that maintain race-based power inequalities in the United States, which
- expose people of color to interpersonal racism;
- reduce their socioeconomic opportunities, such as educational attainment;
- impose intense psychosocial stressors, such as threats of violence and incarceration; and
- create barriers to quality medical care (Glymour and Manly, 2008).
These processes operate across the life course and across generations. They tend to be cumulative, and it is therefore difficult to isolate single
factors contributing to the observed disparities. Nevertheless, as researchers have sought to identify modifiable targets for which change is likely to have large impacts, their attention has increasingly turned to the interplay among the effects of race and ethnicity, risk factors for dementia, and the role of public policy. A detailed analysis of how domestic policies in the United States have contributed to current disparities is beyond the scope of this study, but several examples can illustrate the importance of these issues.
An example is education. As discussed above, there is reason to believe that having limited educational experiences (including both attainment and quality of schooling) plays a role in the development of dementia. At the same time, minority and immigrant populations that are disproportionately affected by dementia are also more likely than other groups to have experienced limited educational opportunities (Garcia et al., 2018). An extensive body of work in education has documented inequities by race and ethnicity as well as income in educational opportunity (see, e.g., Duncan and Murnane, 2011). Looking specifically at dementia risk, researchers have explored differences across groups. For example, one study showed that although foreign-born Latina women and Black males are more likely to experience cognitive impairment and dementia relative to their White counterparts, these differences are attenuated after adjusting for years of education (Garcia et al., 2018).8 Yet the benefits of education in terms of healthy cognitive life expectancy appear greatest for Black men and women and U.S.-born Latina women with some college education (Garcia et al., 2021). As evidence of the importance of education in explaining disparities in healthy cognitive life expectancy grows, it will underscore the urgency of addressing educational disparities and the role of local, state, and federal policy in ameliorating them (Montez et al., 2019; see especially Farina et al., 2020).
Policy makers also influence economic well-being, which, as discussed above, has an influence on dementia risk. A few examples illustrate the connections between economic policies and health. Increasing state minimum wages and expanding earned income tax credits has been associated with decreasing the risk of disability, such as being deaf or blind or being unable to perform activities of daily living (Montez et al., 2017). Similarly, expanding Medicaid to cover working-age adults with incomes up to 138 percent of the federal poverty level can significantly narrow the insurance gap between Black and Latinx households and White households (Buchmueller et al., 2016; Griffith et al., 2017). Increasing opportunities for health care coverage would contribute to opportunities for early diagnosis of many health conditions, including hypertension and diabetes, both
___________________
8 The disparities persist for U.S.-born Latinx and Black older adults.
of which increase the risk for dementia. Insurance policies that reduce financial obstacles in health care can contribute to improved access to and quality of care and health outcomes (Sommers et al., 2015; Simon et al., 2017; Guth et al., 2020). Also of note, an estimated 65 percent of Medicaid enrollees lose their Medicaid coverage when they transition to traditional Medicare at age 65, which means they begin to bear substantially higher costs for services just at the stage of life when they may have increased need for care, such as provision of hearing aids and blood pressure control, that may contribute to dementia prevention (Kaiser Family Foundation, 2020).
Limited access to high-quality health care may also have significant effects on cognitive health, as evidence on American Indian populations illustrates. Both limitations of the Indian Health Service (a division of the U.S. Department of Health and Human Services) and very low rates of health insurance among many Native American groups have been documented (Artiga and Orgera, 2019). American Indian and Alaska Native adults are more likely than their White counterparts to have been unable to see a doctor in the past year because of cost (19% vs. 13%) and to have delayed care for other reasons (36% vs. 19%). Lost opportunities for preventive care (e.g., having blood pressure under control or obtaining hearing aids) and for connections with community resources (i.e., avoiding isolation) likely have significant effects on health, including cognitive outcomes, although researchers have not effectively disentangled the effects of race from the effects of disadvantaged socioeconomic status (Zahodne et al., 2017). Furthermore, access to health care, including such services as cognitive impairment screening (discussed further in Chapter 3), is critical to the early detection of dementia, which in turn can positively influence decision making and promote better health outcomes (Patnode et al., 2020).
Evidence for other population groups supports the importance of insurance and health care access. For example, a study of social correlates of likely dementia for the oldest old Mexican origin populations in Mexico and the United States showed that most of these individuals depend on their extended family for care (Mejia-Arango et al., 2020). Lack of medical, rehabilitative, and preventive services for this group, and others, can translate to significant economic challenges for families (e.g., paying for costly hearing aids that are important to many outcomes for the elderly [Brewster et al., 2020]). Inadequate nutrition, financial hardship, environmental stressors, and reduced opportunities for physical and social activity in their neighborhoods also play a key role in cognitive outcomes, and the groups they hit hardest are both members of minority populations and those who are economically disadvantaged.
The strong relationship between the race and ethnicity of a population and places where its members reside and the benefits they can access is discussed in greater detail in Chapter 5, but here we note that the United States spends significantly less on social services for the elderly compared with 11 other high-income countries (Osborn et al., 2017). The resulting financial strains affect the racial/ethnic subgroups that are also disproportionally poor. The conditions in homes and neighborhoods experiencing these strains are associated with an increased risk of premature mortality that is comparable to the risks posed by obesity, smoking, and a sedentary lifestyle (Fulmer et al., 2021). In other words, it is the intersection of many risk factors at the policy, community, and individual levels that either increases risk for or offers protection against dementia for racial/ethnic minorities.
Many researchers who study these issues use the concept of structural racism to capture the intersection of the multiple processes that drive differential outcomes in dementia risk (as well as many other health outcomes).9 Much more research is needed to identify and document the effects of inequitable policies and systems in health care, education, housing, and social services; to identify specific discriminatory practices; and to document unequal distribution of resources that has adverse impacts on the health of populations of color. Also needed is evaluation of initiatives that have begun to address such sources of inequality—such as Seattle’s Race and Social Justice Initiative, an effort to eliminate racial disparities and foster racial equity.10 Needed as well is study of the interactive, embedded, and reciprocal dynamics that operate in the relationships among patient, provider, community, service systems, and policy to identify additional pathways for mitigating the impact of institutional racism on cognitive health.
RESEARCH DIRECTIONS
A look across the landscape of preventive and protective factors suggests that a large proportion of dementia could be prevented or delayed, but there is limited rigorous causal evidence with enough precision to guide evidence translation and the development of interventions. The uncertainties relate to nearly every domain of prevention, including behavioral changes, socioeconomic conditions, and structural and interpersonal racism and
___________________
9 Structural racism has been defined as the “totality of ways in which societies foster racial discrimination through mutually reinforcing systems of housing, education, employment, earnings, benefits, credit, media, health care, and criminal justice” (Zinzi et al., 2017, p. 1453).
discrimination. This chapter has noted as well important potential impacts of risk factors that have received less attention, including polypharmacy and environmental conditions. For all these factors, there is an urgent need for better evidence on how to translate their well-documented correlations with the prevalence, incidence, and course of dementia into effective policies, system changes, or interventions.
For example, although there is robust evidence that people who take such common-sense measures as eating a healthy diet, exercising regularly, maintaining a healthy weight, and reducing cardiovascular risk have a lower risk of dementia, it remains unclear whether interventions on these factors would reduce dementia risk. The specifics of how to design such interventions are even less clear: Must healthy behavior be sustained throughout life, or can older adults who have recently modified their behavior derive benefits? What duration and intensity of exercise are necessary, and does it matter whether the exercise is primarily aerobic or strength training? Are there some groups of people who would benefit more or less from particular behavioral interventions? For individuals seeking to reduce their risk of dementia, the research provides little firm evidence but rather suggests a set of behaviors that might be helpful for brain health, are almost certainly not harmful, and probably have ancillary benefits for other health domains.
There are similar gaps regarding the causal effects of structural racism, socioeconomic disadvantage, and negative social interactions (e.g., perceived overt racism and discrimination), with the exception of relatively strong evidence that education improves cognitive reserve. To understand how to design interventions for any of these factors may require better understanding of mechanisms and mediators. To the extent that socioeconomic resources, such as education, do reduce dementia risk, these factors may well operate through behavior changes. Likewise, socioeconomic disadvantage constrains the ability of all adults to engage in health-promoting behaviors. Inequities and their consequences also point to questions about the possible benefits of public health interventions designed to reduce dementia risk at the population level, such as by promoting changes in access to resources (e.g., education, housing) or health behaviors (e.g., encouraging physical activity) or by altering the environment (e.g., improving access to exercise venues).
Scientific answers to these questions are urgently needed to support evidence-based, easily transferable, and timely interventions to ameliorate the starkly disparate effects of dementia. Because early interventions are key, rigorous yet rapid research methods that produce findings that can be translated to meet the needs of varied groups and regions will be extremely valuable. Methodological issues are discussed on Chapter 8, but we note
here that resources available to support researchers in engaging diverse communities in basic research and intervention development and evaluation include guidebooks developed by the Centers for Disease Control and Prevention and others (see, e.g., Alzheimer’s Association and Centers for Disease Control and Prevention, 2019; Portacolone et al., 2020; Ejiogu et al., 2011; Quiñones et al., 2020; Gershon et al., 2020; Streitz et al., 2020).
Having surveyed the landscape of research related to reducing the risk of dementia, the committee identified high-priority research needs in this domain in six broad areas. These areas are summarized in Conclusion 2-1; Table 2-1 provides detailed directions for research in each area.
CONCLUSION 2-1: For health care and public health professionals to take advantage of modifiable factors to prevent Alzheimer’s disease and related dementias or reduce or delay their symptoms, research is needed in six broad areas:
- The causal effects of social factors on the incidence and rate of progression of dementia, including factors from multiple domains (socioeconomic resources, social network, structural drivers of exposure); at multiple levels (individual, family, and community); and at multiple life-course periods (e.g., childhood, early to mid-adulthood, old age).
- The effects of health-related behaviors and their management over the life course.
- Modifiable drivers of racial/ethnic inequality in dementia incidence, as well as other dimensions of inequality (e.g., geography).
- The mechanisms through which socioeconomic factors influence brain health, including physiologic changes, behavioral mechanisms, and medical care pathways.
- Detailed understanding of identified risk factors to support more precise recommendations to individuals about decision making and inform population-level policies for altering social contexts, modifying the environment, or changing social policies/systems to promote brain health.
- Effective means of communicating the magnitude and degree of potential risk and protective factors to support informed decision making.
TABLE 2-1 Detailed Research Needs
| 1 and 2: Causal Effects of Social Factors and Health-Related Behaviors Over the Life Course |
|
| 3: Inequality in Dementia |
|
| 4: Mechanisms Through Which Socioeconomic Factors Operate |
|
| 5: Interventions Involving Changes in Policies, Systems, or Individual Behaviors |
|
| 6: Effective Means of Communicating About Risk and Protective Factors |
|
REFERENCES
Ackermann, R.T., Liss, D.T., Finch, E.A., Schmidt, K.K., Hays, L.M., Marrero, D.G., and Saha, C.A. (2015). Randomized comparative effectiveness trial for preventing type 2 diabetes. American Journal of Public Health, 105(11), 2328–2334. https://doi.org/10.2105/AJPH.2015.302641
Alzheimer’s Association. (2021). Special Report: Race, Ethnicity and Alzheimer’s in America. https://www.alz.org/media/Documents/alzheimers-facts-and-figures-special-report.pdf
Alzheimer’s Association and Centers for Disease Control and Prevention. (2019). Healthy Brain Initiative: Road Map for Indian Country. Chicago, IL: Alzheimer’s Association. https://www.cdc.gov/aging/healthybrain/pdf/HBI-Road-Map-for-Indian-Country-508.pdf
Arpino, B., Gumà, J., and Julià, A. (2018). Early-life conditions and health at older ages: The mediating role of educational attainment, family and employment trajectories. PloS One, 13(4), e0195320. https://doi.org/10.1371/journal.pone.0195320
Artiga, S., and Orgera, A. (2019, November 12). Key Facts on Health and Health Care by Race and Ethnicity. Kaiser Family Foundation. https://www.kff.org/racial-equity-and-health-policy/report/key-facts-on-health-and-health-care-by-race-and-ethnicity
Bailey, Z.D., Feldman, J.M., and Bassett, M.T. (2021). How structural racism works—Racist policies as a root cause of U.S. racial health inequities. New England Journal of Medicine, 384(8), 768–773. https://doi.org/10.1056/NEJMms2025396
Barnes, D.E., and Yaffe, K. (2011). The projected effect of risk factor reduction on Alzheimer’s disease prevalence. Lancet Neurology, 10(9), 819–828. https://doi.org/10.1016/S1474-4422(11)70072-2
Barnes, L.L., Leurgans, S., Aggarwal, N.T., Shah, R.C., Arvanitakis, Z., James, B.D., Buchman, A.S., Bennett, D.A., and Schneider, J.A. (2015). Mixed pathology is more likely in black than white decedents with Alzheimer dementia. Neurology, 85(6), 528–534.
Barthold, D., Marcum, Z.A., Gray, S.L., and Zissimopoulos, J. (2020). Alzheimer’s disease and related dementias risk: Comparing users of non-selective and M3-selective bladder antimuscarinic drugs. Pharmacoepidemiology and Drug Safety, 129(12), 1650–1658. https://doi.org/10.1002/pds.5098
Bowling, A., Pikhartova, J., and Dodgeon, B. (2016). Is mid-life social participation associated with cognitive function at age 50? Results from the British National Child Development Study (NCDS). BMC Psychology, 4(1), 58. https://doi.org/10.1186/s40359-016-0164-x
Brewster, K.K., Pavlicova, M., Stein, A., Chen, M., Chen, C., Brown, P.J., Roose, S.P., Kim, A.H., Golub, J.S., Brickman, A., and Galatioto, J. (2020). A pilot randomized controlled trial of hearing aids to improve mood and cognition in older adults. International Journal of Geriatric Psychiatry, 35(8), 842–850.
Buchmueller, T.C., Levinson, Z.M., Levy, H.G., and Wolfe, B.L. (2016). Effect of the Affordable Care Act on racial and ethnic disparities in health insurance coverage. American Journal of Public Health, 106(8), 1416–1421.
Cacciottolo, M., Wang, X., Driscoll, I., Woodward, N., Saffari, A. Reyes, J., Serre, M.L., Vizuete, W., Sioutas, C., Morgan, T.E., Gatz, M., Chui, H.C., Shumaker, S.A., Resnick, S.M., Espeland, M.A., Finch, C.E., and Chen, J.C. (2017). Particulate air pollutants, APOE alleles and their contributions to cognitive impairment in older women and to amyloidogenesis in experimental models. Translational Psychiatry, 7(1), e1022. https://doi.org/10.1038/tp.2016.280
Caunca, M.R., Odden, M.C., Glymour, M.M., Elfassy, T., Kershaw, K.N., Sidney, S., Yaffe, K., Launer, L., and Zeki Al Hazzouri, A. (2020). Association of racial residential segregation throughout young adulthood and cognitive performance in middle-aged participants in the CARDIA study. JAMA Neurology, 77(8), 1000–1007. https://doi.org/10.1001/jamaneurol.2020.0860
Celidoni, M., Dal Bianco, C., and Weber, G. (2017). Retirement and cognitive decline: A longitudinal analysis using SHARE data. Journal of Health Economics, 56, 113–125. https://doi.org/10.1016/j.jhealeco.2017.09.003
Chatterjee, S., Talwar, A., and Aparasu, R.R. (2020). Anticholinergic medications and risk of dementia in older adults: Where are we now?. Expert Opinion on Drug Safety, 19(10), 1251–1267. https://doi.org/10.1080/14740338.2020.1811227
Chen, C., and Zissimopoulos, J. (2018). Racial and ethnic disparities in dementia prevalence and risk factors from 2000 to 2012 in the United States. Alzheimer’s and Dementia Translational Research, 4(1), 510–520. https://doi.org/10.1016/j.trci.2018.08.009
Child Trends. (2018). High-Quality Preschool Can Support Healthy Development and Learning. https://www.childtrends.org/wp-content/uploads/2018/05/PreschoolFadeOutFactSheet_ChildTrends_April2018.pdf
Coogan, P., Schon, K., Li, S., Cozier, Y., Bethea, T., and Rosenberg, L. (2020). Experiences of racism and subjective cognitive function in African American women. Alzheimer’s & Dementia (Amsterdam, Netherlands), 12(1), e12067. https://doi.org/10.1002/dad2.12067
Crimmins, E.M., Saito, Y., Kim, J.K., Zhang, Y.S., Sasson, I., and Hayward, M.D. (2018). Educational differences in the prevalence of dementia and life expectancy with dementia: Changes from 2000 to 2010. Journals of Gerontology, Series B, 73(suppl_1), S20–S28. https://doi.org/10.1093/geronb/gbx135
Denier, N., Clouston, S., Richards, M., and Hofer, S. (2017). Retirement and cognition: A life course view. Advances in Life Course Research, 31, 11–21. https://doi.org/10.1016/j.alcr.2016.10.004
Denny, A., Streitz, M., Stock, K., Joyce, E., Balls-Berry, L.L., Barnes, G., Byrd, G.S., Croff, R., Gao, S., Glover, C.M., Hendrie, H.C., Hu, W.T., Manly, J.J., Moulder, K.L., Stark, S., Thomas, S.B., Whitmer, R., Wong, R., Morris, J.C., and Lingler, J.H. (2020). Perspective on the “African American participation in Alzheimer disease research: Effective strategies” workshop, 2018. Alzheimer’s and Dementia, 16(12), 1734–1744. https://doi.org/10.1002/alz.12160
Duncan, G.J., and Murnane, R.J., Eds. (2011). Whither Opportunity? Rising Inequality, Schools, and Children’s Life Chances. New York: Russell Sage Foundation.
Ejiogu, N., Norbeck, J.H., Mason, M.A., Cromwell, B.C., Zonderman, A.B., and Evans, M.K. (2011). Recruitment and retention strategies for minority or poor clinical research participants: Lessons from the Healthy Aging in Neighborhoods of Diversity across the Life Span study. Gerontologist, 51(Suppl 1), S33–S45. https://doi.org/10.1093/geront/gnr027
Farina, M.P., Hayward, M.D., Kim, J.K., and Crimmins, E.M. (2020). Racial and educational disparities in dementia and dementia-free life expectancy. Journals of Gerontology. Series B, 75(7), e105–e112. https://doi.org/10.1093/geronb/gbz046
Finch, C., and Kulminski, A. (2019). The Alzheimer’s disease exposome. Alzheimer’s & Dementia, 15(9), 1123–1132. https://doi.org/10.1016/j.jalz.2019.06.3914
Floud, S., Balkwill, A., Sweetland, S., Brown, A., Mauricio Reus, E., Hofman, A., Blacker, D., Kivimaki, M., Green, J., Peto, R., Reeves, G., and Beral, V. (2021). Cognitive and social activities and long-term dementia risk: The prospective UK Million Women Study. Lancet Public Health, 6(2), e116–e123. https://doi.org/10.1016/S2468-2667(20)30284-X
Frieden, T.R. (2010). A framework for public health action: The health impact pyramid. American Journal of Public Health, 100(4), 590–595. https://doi.org/10.2105/AJPH.2009.185652
Fulmer, T., Reuben, D.B., Auerbach, J., and Fick, D.M. (2021). Actualizing better health and health care for older adults. Health Affairs (Project Hope), 40(2), 219–225. https://doi.org/10.1377/hlthaff.2020.01470
Garcia, M., Saenz, J., Downer, B., and Wong, R. (2018). The role of education in the association between race/ethnicity/nativity, cognitive impairment, and dementia among older adults in the United States. Demographic Research, 38, 155–168. https://doi.org/10.4054/DemRes.2018.38.6
Garcia, M., Downer, B., Chiu, C-T., Saenz, J., Ortiz, K., and Wong, R. (2021). Educational benefits and cognitive health life expectancies: Racial/ethnic, nativity, and gender disparities. Gerontologist, 61(3), 330–340. https://doi.org/10.1093/geront/gnaa112
Gershon, R., Nowinski, C., Peipert, J.D., Bedieti, K., Ustinovich, V., Hook, J., Fox, R., and Weintraub, S. (2020). Use of the NIH Toolbox for assessment of mild cognitive impairment and Alzheimer’s disease in general population, African-American and Spanish-speaking samples of older adults. Alzheimer’s & Dementia, 16(S6). https://doi.org/10.1002/alz.043372
Gielen, A., and Green, L. (2015). The impact of policy, environmental, and educational interventions: A synthesis of the evidence from two public health success stories. Health Education & Behavior, 42(1 Suppl), 20S–34S. https://doi.org/10.1177/1090198115570049
Glymour, M.M., and Manly, J.J. (2008). Lifecourse social conditions and racial and ethnic patterns of cognitive aging. Neuropsychology Review, 18(3), 223–254.
Griffith, K., Evans, L., and Bor, J. (2017). The Affordable Care Act reduced socioeconomic disparities in health care access. Health Affairs, 36(8), 1503–1510.
Guth, M., Artiga, S., and Pham, O. (2020). Effects of the ACA Medicaid Expansion on Racial Disparities in Health and Health Care. Kaiser Family Foundation. https://www.kff.org/report-section/effects-of-the-aca-medicaid-expansion-on-racial-disparities-in-health-and-health-care-issue-brief
Hayward, M.D., Farina, M.P., Zhang, Y.S., Kim, J.K., and Crimmins, E.M. (2021). The importance of improving educational attainment for dementia prevalence trends from 2000-2014, among older non-Hispanic Black and White Americans. Journals of Gerontology. Series B, gbab015. Advance online publication. https://doi.org/10.1093/geronb/gbab015
Institute of Medicine. (2007). Ending the Tobacco Problem: A Blueprint for the Nation. Washington, DC: The National Academies Press. https://doi.org/10.17226/11795
———. (2015). Cognitive Aging: Progress in Understanding and Opportunities for Action. Washington, DC: The National Academies Press. https://doi.org/10.17226/21693
Jiang, L., Manson, S.M., Beals, J., Henderson, W., Huang, H., Acton, K., Roubideaux, Y., and the Special Diabetes Program for Indians Diabetes Prevention Demonstration Project. (2013). Translating the Diabetes Prevention Program into American Indian and Alaska Native communities: Results from the Special Diabetes Program for Indians Diabetes Prevention demonstration project. Diabetes Care, 36(7), 2027–2034. https://doi.org/10.2337/dc12-1250
Jirout, J., LoCasale-Crouch, J., Turnbull, K., Gu, Y., Cubides, M., Garzione, S., Evans, T.M., Weltman, A.L., and Kranz, S. (2019). How lifestyle factors affect cognitive and executive function and the ability to learn in children. Nutrients, 11(8), 1953. https://doi.org/10.3390/nu11081953
Kaiser Family Foundation. (2020). Premium and Cost-sharing Requirements for Selected Services for Medicaid Adults. https://www.kff.org/0a320d8
Karp, A., Andel, R., Parker, M., Wang, H-X., Winblad, B., and Fratiglioni, L. (2009). Mentally stimulating activities at work during midlife and dementia risk after age 75: Follow-up study from the Kungsholmen project. American Journal of Geriatric Psychiatry, 17(3), 227–236. https://doi.org/10.1097/JGP.0b013e318190b691
Kawas, C.H., Kim, R.C., Sonnen, J.A., Bullain, S.S., Trieu, T., and Corrada, M.M. (2015). Multiple pathologies are common and related to dementia in the oldest-old: The 90+ study. Neurology, 85(6), 535–542.
Lines, L., Sherif, N., and Wiener, J. (2014). Racial and Ethnic Disparities among Individuals with Alzheimer’s Disease in the United States. RTI Press. https://doi.org/10.3768/rti-press.2014.RR.0024.1412
Livingston, G., Sommerland, A., Ortega, V., Costafreda, S., Huntley, J., Ames, D., Ballard, C., Banerjee, S., Burns, A., Cohen-Mansfield, J., Cooper, C., Fox, N., Gitlin, L., Howard, R., Kales, H., Larson, E., Ritchie, K., Rockwood, K., Sampson, E., Samus, Q., Schneider, L., Selbæk, G., Teri, K., and Mukadam, N. (2017). Dementia prevention, intervention, and care. Lancet, 390(10113), 2673–2734. https://doi.org/10.1016/S0140-6736(17)31363-6
Livingston, G., Huntley, J., Sommerland, A., Ames, D., Ballard, C., Banerjee, S., Brayne, C., Burns, A., Cohen-Mansfield, J., Cooper, C., Costafreda, S., Dias, A., Fox, N., Gitlin, L., Howard, R., Kales, H., Kivimäki, M., Larson, E., Ogunniyi, A., Orgeta, V., Ritchie, K., Rockwood, K., Sampson, E., Samus, Q., Schneider, L., Selbæk, G., Teri, L., and Mukadam, N. (2020). Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. Lancet, 396(10248), 413–446. https://doi.org/10.1016/S0140-6736(20)30367-6
Lourida, I., Hannon, E., Littlejohns, T., Langa, K., Hyppönenm E., Ku´zma, E., and Llewellyn, D. (2019). Association of lifestyle and genetic risk with incidence of dementia. Journal of the American Medical Association, 322(5), 430–437. https://doi.org/10.1001/jama.2019.9879
Lövdén, M., Fratiglioni, L., Glymour, M.M., Lindenberger, U., and Tucker-Drob, E.M. (2020). Education and cognitive functioning across the life span. Psychological Science in the Public Interest, 21(1), 6–41. https://doi.org/10.1177/1529100620920576
Lu, T., Yan, X., Huang, Y., Zhao, M., Li, M., Ma, K., Yin, J., Zhan, C., and Wang, Q. (2019). Trends in the incidence, treatment, and survival of patients with lung cancer in the last four decades. Cancer Management and Research, 11, 943–953. https://doi.org/10.2147/CMAR/S187317
Marden, J.R., T chetgen T chetgen, E.J., Kawachi, I., and Glymour, M.M. (2017). Contribution of socioeconomic status at 3 life-course periods to late-life memory function and decline: Early and late predictors of dementia risk. American Journal of Epidemiology, 186(7), 805–814. https://doi.org/10.1093/aje/kwx155
Matthews, K.A., Xu, W., Gaglioti, A.H., Holt, J.B., Croft, J.B., Mack, D., and McGuire, L.C. (2019). Racial and ethnic estimates of Alzheimer’s disease and related dementias in the United States (2015-2060) in adults aged ≥65 years. Alzheimer’s & Dementia, 15(1), 17–24. https://doi.org/10.1016/j.jalz.2018.06.3063
Mayeda, E.R., Glymour, M.M., Quesenberry, C.P., and Whitmer, R.A. (2016). Inequalities in dementia incidence between six racial and ethnic groups over 14 years. Alzheimer’s & Dementia, 12(3), 216–224.
Mayeda, E.R., Glymour, M.M., Quesenberry, C.P., Johnson, J.K., Pérez-Stable, E.J., and Whitmer, R.A. (2017a). Heterogeneity in 14-year dementia incidence between Asian American subgroups. Alzheimer Disease and Associated Disorders, 31(3), 181.
———. (2017b). Survival after dementia diagnosis in five racial/ethnic groups. Alzheimer’s & Dementia, 13(7), 761–769. https://doi.org/10.1016/j.jalz.2016.12.008
McMurtray, A., Clark, D.G., Christine, D., and Mendez, M.F. (2006). Early-onset dementia: Frequency and causes compared to late-onset dementia. Dementia and Geriatric Cognitive Disorders, 21(2), 59–64. https://doi.org/10.1159/000089546
Mehta, K., and Yeo, G. (2017). Systematic review of dementia prevalence and incidence in United States race/ethnic populations. Alzheimer’s & Dementia, 13(1), 72–83. https://doi.org/10.1016/j.jalz.2016.06.2360
Mejia‐Arango, S., Aguila, E., López‐Ortega, M., Gutiérrez‐Robledo, L.M., Vega, M., Drumond Andrade, F., Rote, S., Grasso, S., Markides, K., and Angel, J. (2020). Health and social correlates of dementia in oldest‐old Mexican‐origin populations. Alzheimer’s & Dementia, 6(1), e12105. https://doi.org/10.1002/trc2.12105
Milton, L.A., and White, A.R. (2020). The potential impact of bushfire smoke on brain health. Neurochemistry International, 139, 104796. https://doi.org/10.1016/j.neuint.2020.104796
Montez, J.K., Hayward, M.D., and Wolf, D.A. (2017). Do U.S. states’ socioeconomic and policy contexts shape adult disability? Social Science & Medicine (1982), 178, 115–126. https://doi.org/10.1016/j.socscimed.2017.02.012
Montez, H., Hayward, M., and Zajacova, A. (2019). Educational disparities in adult health: U.S. states as institutional actors on the association. Socius, 5. https://doi.org/10.1177/2378023119835345
Mortamais, M., Gutierrez, L.A., de Hoogh, K., Chen, J., Vienneau, D., Carrière, I., Letellier, N., Helmer, C., Gabelle, A., Mura, T., and Sunyer, J. (2021). Long-term exposure to ambient air pollution and risk of dementia: Results of the prospective Three-City Study. Environment International, 148, 106376.
NASEM (National Academies of Sciences, Engineering, and Medicine). (2017). Preventing Cognitive Decline and Dementia: A Way Forward. Washington, DC: The National Academies Press. https://doi.org/10.17226/24782
———. (2019a). Fostering Healthy Mental, Emotional, and Behavioral Development in Children and Youth. Washington, DC: The National Academies Press.
———. (2019b). Roadmap to Reducing Child Poverty. Washington, DC: The National Academies Press.
National Center for Education Statistics. (1993). 120 Years of American Education: A Statistical Portrait. https://nces.ed.gov/pubs93/93442.pdf
Nelson, C.A., Scott, R.D., Bhutta, Z.A., Harris, N.B., Danese, A., and Samara, M. (2020). Adversity in childhood is linked to mental and physical health throughout life. BMJ (Clinical Research Ed.), 371, m3048. https://doi.org/10.1136/bmj.m3048
Ngandu, T., Lehtisalo, J., Solomon, A., Levälahti, E., Ahtiluoto, S., Antikainen, R., Bäckman, L., Hänninen, T., Jula, A., Laatikainen, T., Lindström, J., Mangialasche, F., Paajanen, T., Pajala, S., Peltonen, M., Rauramaa, R., Stigsdotter-Neely, A., Strandberg, T., Tuomilehto, J., Soininen, H., and Kivipelto, M. (2015). A 2 year multidomain intervention of diet, exercise, cognitive training, and vascular risk monitoring versus control to prevent cognitive decline in at-risk elderly people (FINGER): A randomised controlled trial. Lancet (London, England), 385(9984), 2255–2263. https://doi.org/10.1016/S0140-6736(15)60461-5
Nguyen, T.T., T chetgen T chetgen, E.J., Kawachi, I., Gilman, S.E., Walter, S., Liu, S.Y., Manly, J.J., and Glymour, M.M. (2016). Instrumental variable approaches to identifying the causal effect of educational attainment on dementia risk. Annals of Epidemiology, 26(1), 76.e3. https://doi.org/10.1016/j.annepidem.2015.10.006
Norton, S., Matthews, F.E., Barnes, D.E., Yaffe, K., and Brayne, C. (2014). Potential for primary prevention of Alzheimer’s disease: An analysis of population-based data. Lancet Neurology, 13(8), 788–794. https://doi.org/10.1016/S1474-4422(14)70136-X
Osborn, R., Doty, M.M., Moulds, D., Sarnak, D.O., and Shah, A. (2017). Older Americans were sicker and face more financial barriers to health care than counterparts in other countries. Health Affairs (Project Hope), 36(12), 2123–2132. https://doi.org/10.1377/hlthaff.2017.1048
Park, S., and Chen, J. (2020). Racial and ethnic patterns and differences in health care expenditures among Medicare beneficiaries with and without cognitive deficits or Alzheimer’s disease and related dementias. BMC Geriatrics, 20(1), 482. https://doi.org/10.1186/s12877-020-01888-y
Patnode, C.D., Perdue, L.A., Rossom, R.C., Rushkin, M.C., Redmond, N., Thomas, R.G., and Lin, J.S. (2020). Screening for cognitive impairment in older adults: Updated evidence report and systematic review for the U.S. Preventive Services Task Force. Journal of the American Medical Association, 323(8), 764–785. https://doi.org/10.1001/jama.2019.22258
Plassman, B.L., Langa, K.M., Fisher, G.G., Heeringa, S.G., Weir, D.R., Ofstedal, M.B., Burke, J.R., Hurd, M.D., Potter, G.G., Rodgers, W.L., Steffens, D.C., Willis, RJ., and Wallace, R.B. (2007). Prevalence of dementia in the United States: The Aging, Demographics, and Memory study. Neuroepidemiology, 29(1-2), 125–132. https://doi.org/10.1159/000109998
Portacolone, E., Palmer, N.R., Lichtenberg, P., Waters, C.M., Hill, C.V., Keiser, S., Vest, L., Maloof, M., Tran, T., Martinez, P., Guerrero, J., and Johnson, J.K. (2020). Earning the trust of African American communities to increase representation in dementia research. Ethnicity & Disease, 30(Suppl 2), 719–734. https://doi.org/10.18865/ed.30.S2.719
Quiñones, A.R., Kaye, J., Allore, H.G., Botoseneanu, A., and Thielke, S.M. (2020). An agenda for addressing multimorbidity and racial and ethnic disparities in Alzheimer’s disease and related dementia. American Journal of Alzheimer’s Disease and Other Dementias, 35, 1533317520960874. https://doi.org/10.1177/1533317520960874
Rohwedder, S., and Willis, R.J. (2010). Mental retirement. Journal of Economic Perspectives, 24(1), 119–138. https://doi.org/10.1257/jep.24.1.119
Ruiz-Hernandez, A., Navas-Acien, A., Pastor-Barriuso, R., Crainiceanu, C.M., Redon, J., Guallar, E., and Tellez-Plaza, M. (2017). Declining exposures to lead and cadmium contribute to explaining the reduction of cardiovascular mortality in the U.S. population, 1988–2004. International Journal of Epidemiology, 46(6), 1903–1912.
Sajeev, G., Weuve, J., Jackson, J., VanderWeele, T., Bennett, D., Grodstein, F., and Blacker, D. (2016). Late-life cognitive activity and dementia: A systematic review and bias analysis. Epidemiology, 27(5), 732–742. https://doi.org/10.1097/EDE.0000000000000513
Schneider, J.A., Arvanitakis, Z., Leurgans, S.E., and Bennett, D.A. (2009). The neuropathology of probable Alzheimer disease and mild cognitive impairment. Annals of Neurology, 66(2), 200–208.
Seblova, D., Brayne, C., Machu, V., Kuklová, M., Kopecek, M., and Cermakova, P. (2019). Changes in cognitive impairment in the Czech Republic. Journal of Alzheimer’s Disease, 72(3), 693–701. https://doi.org/10.3233/JAD-190688
Sharp, E.S., and Gatz, M. (2011). Relationship between education and dementia: An updated systematic review. Alzheimer Disease and Associated Disorders, 25(4), 289–304. https://doi.org/10.1097/WAD.0b013e318211c83c
Simon, K., Soni, A., and Cawley, J. (2017). The impact of health insurance on preventive care and health behaviors: Evidence from the first two years of the ACA Medicaid expansions. Journal of Policy Analysis and Management, 36(2), 390–417.
Sommers, B.D., Gunja, M.Z., Finegold, K., and Musco, T. (2015). Changes in self-reported insurance coverage, access to care, and health under the Affordable Care Act. Journal of the American Medical Association, 314(4), 366–374.
SPRINT-MIND Investigators for the SPRINT Research Group, Williamson, J., Pajewski, N., Auchus, A., Bryan, R.N., Chelune, G., Cheung, A., Cleveland, M., Coker, L., Crowe, M., Cushman, W., Cutler, J., Davatzikos, C., Desiderio, L., Erus, G., Fine, L., Gaussoin, S., Harris, D., Hsieh, M-K., Johnson, K., Kimmel, P., Tamura, M., Launer, L., Lerner, A., Lewis, C., Martindale-Adams, J., Moy, C., Nasrallah, I., Nichols, L., Oparil, S., Ogrocki, P., Rahman, M., Rapp, S., Reboussin, D., Rocco, M., Sachs, B., Sink, K., Still, C., Supiano, M., Snyder, J., Wadley, V., Walker, J., Weiner, D., Whelton, P., Wilson, V., Woolard, N., Wright, J., and Wright, C. (2019). Effect of intensive vs standard blood pressure control on probable dementia: A randomized clinical trial. Journal of the American Medical Association, 321(6), 553–561. https://doi.org/10.1001/jama.2018.21442
Stern, Y. (2009). Cognitive reserve. Neuropsychologia, 47(10), 2015–2028. https://doi.org/10.1016/j.neuropsychologia.2009.03.004
Thunell, J., Joyce, G., Chen, Y., Barthold, D., Brinton, R., and Zissimopoulos, J. (2021). Drug therapies for chronic conditions and risk of Alzheimer’s disease and related dementias: A scoping review. Alzheimer’s & Dementia, 17, 41–48. https://doi.org/10.1002/alz.12175
Walsemann, K., and Ailshire, J. (2020). Early educational experiences and trajectories of cognitive functioning among U.S. adults in midlife and later. American Journal of Epidemiology, 189(5), 403–411. https://doi.org/10.1093/aje/kwz276
Weden, M.M., Shih, R.A., Kabeto, M.U., and Langa, K.M. (2018). Secular trends in dementia and cognitive impairment of U.S. rural and urban older adults. American Journal of Preventive Medicine, 54(2), 164–172. https://doi.org/10.1016/j.amepre.2017.10.021
Wei, Y., Wang, Y., Lin, C.K., Yin, K., Yang, J., Shi, L., Li, L., Zanobetti, A., and Schwartz, J.D. (2019). Associations between seasonal temperature and dementia-associated hospitalizations in New England. Environment International, 126, 228–233.
Werner, P. (2019). Reflections on quality of care for persons with dementia: Moving toward an integrated, comprehensive approach. International Psychogeriatrics, 31(3), 307–308. https://doi.org/10.1017/S1041610219000346
Wilkerson, I. (2020). Caste: The Origins of Our Discontent. New York: Random House.
Wolters, F.J., Chibnik, L.B., Waziry, R., Anderson, R., Berr, C., Beiser, A., Bis, J.C., Blacker, D., Bos, D., Brayne, C., Dartigues, J.-F., Darweesh, S.K.L., Davis-Plourde, K.L., De Wolf, F., Debette, S., Dufouil, C., Fornage, M., Goudsmit, J., Grasset, L., Gudnason, V., Hadjichrysanthou, C., Helmer, C., Ikram, M.A., Ikram, M.K., Joas, E., Kern, S., Kuller, L.H., Launer, L., Lopez, O.L., Matthews, F.E., McRae-McKee, K., Meirelles, O., Mosley, T.H., Pase, M.P., Psaty, B.M., Satizabal, C.L., Seshadri, S., Skoog, I., Stephan, B.C.M., Wetterberg, H., Wong, M.M., Zettergren, A., and Hofman, A. (2020). Twenty-seven-year time trends in dementia incidence in Europe and the United States. Neurology, 95(5), e519–e531. https://doi.org/10.1212/WNL.0000000000010022
Xu, W., Tan, L., Wang, H.F., Tan, M.S., Tan, L., Li, J.Q., Zhao, Q.F., and Yu, J.T. (2016). Education and risk of dementia: Dose-response meta-analysis of prospective cohort studies. Molecular Neurobiology, 53(5), 3113–3123. https://doi.org/10.1007/s12035-015-9211-5
Yaffe, K. (2019). Prevention of cognitive impairment with intensive systolic blood pressure control. Journal of the American Medical Association, 321(6), 548–549. https://doi.org/10.1001/jama.2019.0008
Yaffe, K., Falvey, C., Harris, T.B., Newman, A., Satterfield, S., Koster, A., Ayonayon, H., Simonsick, E., and Health ABC Study. (2013). Effect of socioeconomic disparities on incidence of dementia among biracial older adults: Prospective study. BMJ (Clinical Research Ed.), 347, f7051. https://doi.org/10.1136/bmj.f7051
Zahodne, L.B., Stern, Y., and Manly, J.J. (2015). Differing effects of education on cognitive decline in diverse elders with low versus high educational attainment. Neuropsychology, 29(4), 649–657. https://doi.org/10.1037/neu0000141
Zahodne, L.B., Manly, J.J., Smith, J., Seeman, T., and Lachman, M.E. (2017). Socioeconomic, health, and psychosocial mediators of racial disparities in cognition in early, middle, and late adulthood. Psychology and Aging, 32(2), 118–130. https://doi.org/10.1037/pag0000154
Zhang, Z., Gu, D., and Hayward, M.D. (2010). Childhood nutritional deprivation and cognitive impairment among older Chinese people. Social Science & Medicine (1982), 71(5), 941–949. https://doi.org/10.1016/j.socscimed.2010.05.013
Zhang, Z., Hayward, M.D., and Yu, Y.L. (2016). Life course pathways to racial disparities in cognitive impairment among older Americans. Journal of Health and Social Behavior, 57(2), 184–199. https://doi.org/10.1177/0022146516645925
Zinzi, D.B., Krieger, N., Agénor, M., Graves, J., Linos, N., and Bassett, M.T. (2017). Structural racism and health inequities in the USA: Evidence and interventions. Lancet, 389(10077), 1453–1463. https://doi.org/10.1016/S0140-6736(17)30569-X
Zissimopoulos, J., Tysinger, B., St. Clair, P., and Crimmins, E. (2018). The impact of changes in population health and mortality on future prevalence of Alzheimer’s Disease and other dementias in the United States. Journals of Gerontology: Series B, 73(suppl_1), S38–S47. https://doi.org/10.1093/geronb/gbx147
Zuelsdorff, M., Okonkwo, O.C., Norton, D., Barnes, L.L., Graham, K.L., Clark, L.R., Wyman, M.F., Benton, S.F., Gee, A., Lambrou, N., Johnson, S.C., and Gleason, C.E. (2020). Stressful life events and racial disparities in cognition among middle-aged and older adults. Journal of Alzheimer’s Disease, 73(2), 671–682. https://doi.org/10.3233/JAD-190439


































