3
Patient Perspectives on Data Needs
This chapter summarizes data needs conveyed by patient organizations. Speakers in this session were asked to focus on the questions below. The brief overview of the input received from the presenters is followed by the committee’s conclusions.
- Looking ahead, what are the main data needs?
- What are the implications of the (recently broadened) statutory scope for patient-centered outcomes research (PCOR)?
- What questions cannot be answered and who is not served by the current PCOR data infrastructure?
- What new data sources could be incorporated into the PCOR data infrastructure?
- What data capacity challenges is the U.S. Department of Health and Human Services (HHS) best positioned to address in the context of its public mission, authorities, programs, and data resources?
Rebekah Angove shared her perspective based on her role as vice president for patient experience and program evaluation at the Patient Advocate Foundation (PAF), a nonprofit organization that provides case management services and financial aid to those with chronic, life-threatening, and debilitating illnesses. Angove also previously served as engagement director of REACHnet, a clinical research network that is part of the National Patient-Centered Clinical Research Network.
As part of its work focused on assisting patients, PAF also collects data from patients with the goal of translating evidence into research and
policy work focused on improving health care and the patient experience. Angove said that because PAF works with patients who experience access and affordability challenges, its patient and caregiver network represents populations that are often underrepresented in research initiatives because they are underinsured or uninsured and, consequently, are less likely to be represented in large health care systems that have robust data collections and clinical trials.
Angove discussed challenges associated with obtaining patient agreements for participating in data collection as well as broader engagement in research. PAF’s experience and the research it has done on this topic indicate that most patients have a very limited understanding of research, and especially of terminology such as “comparative effectiveness” research and “patient-centered outcomes” research. Patients are also often confused or uninformed about how research data are used and the implications of giving their consent for the use of their data. She argued that confusion in these areas leads to mistrust or distrust.
Angove highlighted several characteristics of meaningful patient engagement. First, she noted that engagement requires careful thinking about the range of experiences that are included in order to achieve representativeness. Beyond the dimensions of diversity discussed in Chapter 1, diversity along additional dimensions such as treatment experiences, life experiences, urban vs. rural, and ability to pay for and access health care also need to be represented. If the patients engaged in and contributing to research are not representative of a broad range of experiences, the findings could exacerbate disparities. Angove also underscored the role transparency plays in meaningful patient engagement. This means being clear about who owns the data and how the data are being used, as well as better communication about how patients are involved in the process.
Angove argued that meaningful patient involvement means involvement in all activities that are part of PCOR, and not just involvement in an activity that is specifically carved out for patient involvement (for example, recruitment and patient committees). For example, patients could be more involved in conversations about methodology, about how the results are interpreted, or about how information about the research is communicated. Angove emphasized that the fact that the data are patient reported does not necessarily mean they are patient centered. Validated measures for patient-reported outcomes (PROs) have historically been developed without patient involvement. For example, when patients are involved in projects, they often point out issues related to the PRO measurement scales that are used, but ultimately their input is not incorporated because researchers are reluctant to deviate from PRO measures that have been validated.
Another aspect of meaningful patient engagement discussed by Angove is training in how research is done, not only for patients but also for the
researchers themselves, as well as policy makers and health care workers. This is particularly important given the sensitive nature of the health information that patients are being asked to share. Angove also emphasized the importance of communicating to patients the value of their contributions.
Gary Epstein-Lubow, Brown University, discussed his experiences as team leader for the stakeholder engagement team for the National Institute on Aging’s Imbedded Pragmatic Alzheimer’s Disease and AD-related Dementias Clinical Trials Collaboratory (IMPACT Collaboratory). The IMPACT Collaboratory’s goal is to build the nation’s capacity to conduct pragmatic clinical trials of interventions imbedded within health care systems for people at risk of dementia, people living with dementia, and their family members and care partners.
While there are no disease-modifying treatments available for dementia, nonpharmacologic interventions show promise, although they have had limited adoption. Epstein-Lubow said that to address this, embedded pragmatic clinical trials (ePCTs) need improved patient-centered outcomes data that are systematically available. Patients and caregivers are important stakeholders who provide input to the trial implementation, and the interventions need to be integrated into the routine clinical flow and not add a reporting burden. To do this, it is essential for electronic health records to capture patient-centered outcomes in a systematic way, with special attention paid to confirming that data are collected from underrepresented groups and groups disproportionately harmed by dementia. Finally, outcomes of ePCTs must be relevant and usable by decision makers, including health care systems.
Epstein-Lubow said that there are special considerations for ePCTs when it comes to dementia research. From the perspective of patients, applicable ethical considerations and regulations deserve particular attention, because people living with dementia are a vulnerable population and there are questions about their capacity to provide informed consent and to self-report their preferences. Another area that deserves attention is the role of care partners and family members, including the potential for linking caregiver data with patient data.
Concerning data needs in the area of dementia, Epstein-Lubow highlighted five needs, namely the need for:
- information that can lead to improvements in person-centered care;
- improved reporting on functional status, including physical, social, occupational, and emotional functioning, in addition to cognitive functioning;
- data linkages between information provided by people living with dementia and their caregivers;
- strategies for capturing information about lived experiences; and
- methods for standardizing proxy reporting for people who have partial capacity or who lack capacity to report directly.
Epstein-Lubow argued that the broadened statutory scope for PCOR increases opportunities for learning about patient-centered outcomes but also involves some risks. For example, if inadequate attention is paid to underrepresented groups, including members of groups at higher risks of negative health effects of dementia, this could lead to underrepresented patient-centered outcomes data in these areas. There is also the risk of potential added burden for family members and caregivers in their roles as proxy respondents and missed opportunities for data linkage in the case of missing data from caregivers.
Additional data challenges exist for research on dementia, according to Epstein-Lubow, including the following:
- There is no standard measure set for people living with dementia.
- There is no standard measure set for family caregivers.
- Quality measures for dementia care are optional in most reporting systems.
- There are challenges regarding the collection of patient-centered outcomes from people living with moderate or severe dementia.
- Accommodations for data collection may be required for people with limited health literacy.
- There are methodological challenges associated with linking data from people living with dementia and their caregivers.
Epstein-Lubow highlighted several dementia measures that are part of the Merit-Based Incentive Payment System (MIPS), which is a Centers for Medicare & Medicaid Services (CMS) program that eligible health care clinicians can participate in to report data to better connect care quality with Medicare payments. These dementia measures are optional, but Epstein-Lubow argued that they could be required. The measures highlighted include
- dementia-associated behavioral and psychiatric symptom screening and management;
- dementia: cognitive assessment;
- dementia: education and support of caregivers for patients with dementia;
- dementia: functional status assessment; and
- dementia: safety concern screening and follow-up for patients with dementia.
In addition, Epstein-Lubow suggested that the Consumer Assessment of Healthcare Providers and Systems questions on patient and caregiver experience of care could be expanded to include proxy reports of dementia.
To strengthen the PCOR data infrastructure in ways that could benefit people living with dementia, Epstein-Lubow said that HHS would be particularly well positioned to address one of the recommendations made by the public members of the HHS Advisory Council on Alzheimer’s Research, Care and Services, which urged HHS to “Encourage further development, evaluation, and use of health care models for AD/ADRD that align performance measures, the experience of care by persons living with AD/ADRD and their caregivers, and payment.”1
Epstein-Lubow further suggested using the definitions of care “value” used by CMS and studying models that enhance value. One way to do this would be to rely on the CMS “Meaningful Measures” initiative, including use of new care planning codes, the annual wellness visit, and the MIPS dementia measures discussed earlier.
Elisabeth Oehrlein discussed insights from her work at the National Health Council (NHC), a nonprofit association of more than 140 health-related organizations, including leading patient advocacy groups. She identified a list of key data needs based on what the NHC’s patient groups are hearing from the patients these organizations serve, and based on NHC’s work in the areas of regulations, real-world evidence, and value assessment:
- patient-centered outcomes and impacts that really matter to patients, collected consistently;
- burden, including costs incurred by patients and their families;
- social determinants of health (for example, transportation, housing);
- evidence based on representative populations;
- quality and satisfaction with care, defined from the perspective of patients;
- accessibility of the research results to patients; and
- interoperability.
Oehrlein said that in addition to measuring the outcomes and impacts that patients truly care about, it is also important to use language that patients will use. For example, in the case of alopecia areata (an autoimmune disease that causes hair loss), the patient-reported outcome measure has traditionally been the percentage of the skin that is covered in hair, but talking to patients made it clear that what they care about is not
___________________
1https://aspe.hhs.gov/public-members-advisory-council-alzheimers-research-care-and-services-2020-recommendations#clinical, see recommendation 5.
necessarily the difference between a 20 percent or 40 percent improvement in skin covered, but rather whether or not they need to wear a wig.
In terms of increasing patient accessibility of the research results and evidence, Oehrlein mentioned the expansion of dashboards that patients can use to enter certain criteria and find out what treatments might work better for them or what the outcomes experienced by others with similar conditions have been. These types of communication vehicles could be a model going forward.
To further illustrate the types of outcomes that matter to patients, Oehrlein discussed findings from a Food and Drug Administration report focused on chronic fatigue syndrome and myalgic encephalomyelitis (Table 3-1).2 The table shows that many aspects of a patient’s experience that are traditionally considered important to measure are indeed important to patients. These aspects include disease-related impacts, feelings, and functions. However, patients also care about and want more information on treatment-related impacts, financial impacts, and caregiver impacts. This type of patient input can help narrow the scope of the data that are collected in order to better focus resources and reduce the burden on patients.
Oehrlein said that the broadened statutory scope for PCOR presents opportunities to more effectively assess treatment alternatives and value from the patients’ perspective. In particular, she finds that patient advocates and patients are often surprised to discover that data on out-of-pocket costs and other costs important to patients have not been systematically collected. Having these types of data available would lead to more holistic evidence relevant to patient decision-making and more informed decisions as patients navigate their options and understand what the treatment impacts might be, beyond clinical outcomes. These types of decisions increasingly have an impact on patients’ access to care.
As an illustration of a model for moving forward, Oehrlein discussed the work of the EveryLife Foundation for Rare Diseases. Its report assessing the total economic burden of rare diseases was born out of the realization that the data that have been collected to date on direct medical costs really do not reflect the full patient experience, especially when it comes to rare diseases.3 EveryLife Foundation researchers have thought carefully about which costs are important to patients and how those data might be collected.
In terms of data needs that HHS would be best positioned to address, Oehrlein highlighted the need for better data for underserved populations and communities. Supporting dashboard-type solutions to make more of the data available to inform individual patient decisions would also be
___________________
2https://www.fda.gov/media/86879/download.
3https://everylifefoundation.org/wp-content/uploads/2021/02/The_National_Economic_Bur-den_of_Rare_Disease_Study_Summary_Report_February_2021.pdf.
| Disease-Related Impact on Feelings and Function | Treatment-Related Impacts | Financial-Related Impacts | Caregiver-Related Impacts |
|---|---|---|---|
|
|
|
|
SOURCE: U.S. Food and Drug Administration (2013). https://www.fda.gov/media/86879/download. Workshop presentation by Elisabeth Oehrlein, May 3, 2021.
useful. The COVID-19 dashboard operated by the National Center for Advancing Translational Sciences is an example of a government initiative along those lines.
Bray Patrick-Lake, Evidation Health, shared her insights in part based on her career working in patient advocacy, leading a patient foundation, and working on national research programs, such as All of Us and PCORnet. Her current work is focused on measuring what matters most to patients in everyday life. Evidation Health is a digital research and health engagement company whose members participate in research studies. Members provide person-generated health data, which enable continuous monitoring of health outcomes at the individual level. The platform collects 750 million data points daily. The data include individually generated, individually permissioned data, such as data from wearables and environment data, as well as system-generated individually permissioned data, such as data from electronic health records. Figure 3-1 illustrates the range of data sources considered by Evidation.
Patrick-Lake argued that the traditional sources of data (shown on the left side of Figure 3-1) provide episodic snapshots of the experiences of patients living with disease over time. Most of what constitutes people’s lives, and therefore the richest data about patients’ experiences with disease,
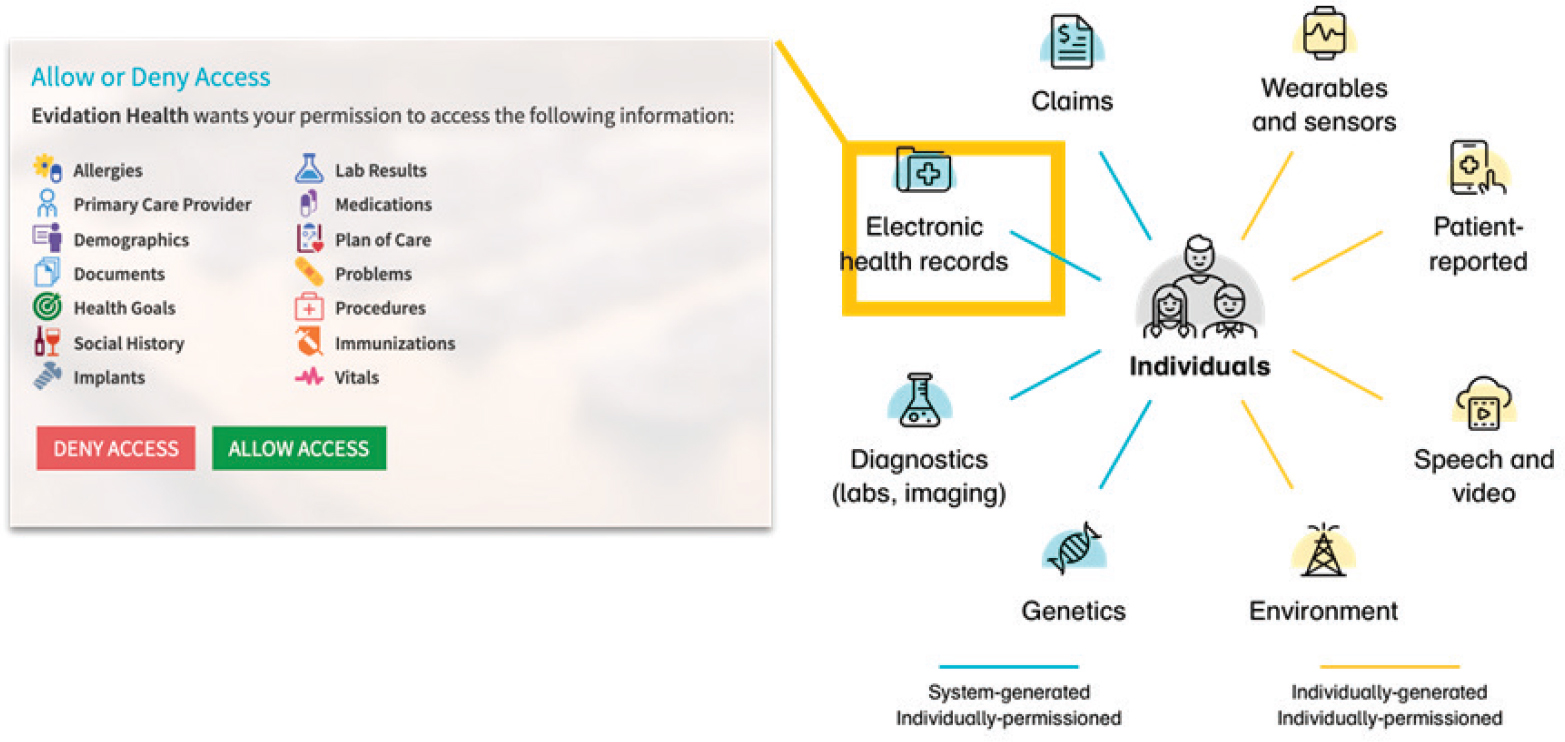
SOURCE: Workshop presentation by Bray Patrick-Lake, May 3, 2021. Evidation Health (2021).
is currently invisible to the health care system. Figure 3-2 illustrates how real-world evidence data points (i.e., evidence obtained from real-world data, such as data generated during routine clinical practice) contrast with data from digital technologies that have the potential to continuously and passively collect data, without substantial burden to patients.
Patrick-Lake argued that digital technologies that continuously collect data have the potential to result in:
- better characterization and understanding of living with the disease;
- better understanding of disease progression;
- earlier identification of at-risk individuals;
- real-world, objective Quality of Life and Activities of Daily Living measures; and
- pattern detection for public health.
Patrick-Lake echoed a point made by other speakers, namely that traditional sources of real-world data might not capture what is truly important to patients. For example, a measure used in the context of cardiac care is mortality, but a patient living with cardiac disease would more likely want information on how to improve his or her quality of life, what the progression of the disease might be like, and how the disease might impact activities of daily living over time.
As an illustration of how data from wearable devices can greatly enhance real-world data, Patrick-Lake suggested considering two asthma patients who seem to be nearly identical based on traditional sources of real-world data: they both have moderate or severe asthma; they are both nonsmokers; they are on the same inhaled medications; and they are both adhering to their physician-prescribed treatments. In terms of symptom control, one person might report waking up often (every night or almost every night) due to asthma symptoms, while the other reports waking up 2 or fewer days a month due to asthma symptoms. When data from (the same brand of) wearable device are added, it becomes clear that the first person is asleep 49 percent of the time, while the second person is asleep 90 percent of the time, while in bed.
Patrick-Lake argued that digital technologies can result in data that accelerate and enhance clinical care, accelerate clinical research, and improve public health. Examples discussed included predicting flare events in an autoimmune condition using wearable and survey data; enhancing recovery modeling for limb surgeries with personalized predictions of outcome tailored to individual characteristics; and early detection, monitoring, and management of COVID-19 in everyday life.
For a potential roadmap of how to achieve the full potential of person-generated health data and digital clinical measures, Patrick-Lake referenced
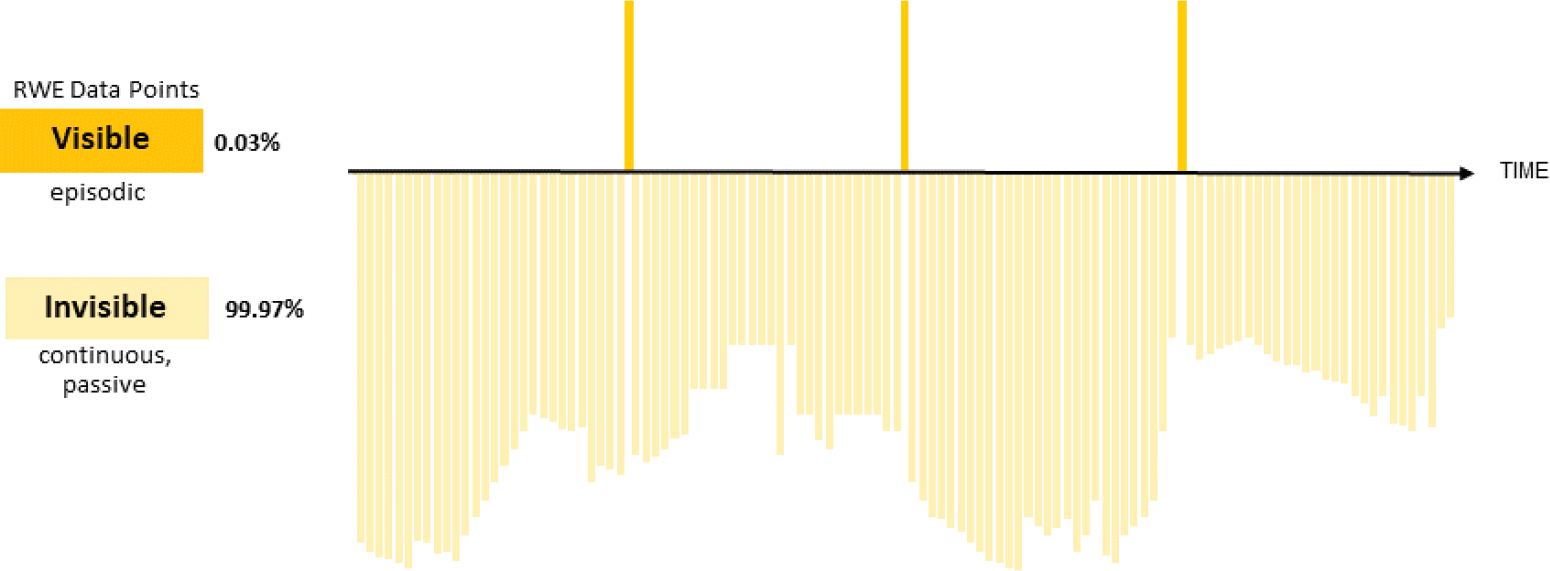
SOURCE: Workshop presentation by Bray Patrick-Lake, 2021. Evidation Health (2021).
The Playbook, a document released by the Digital Medicine Society.4 She highlighted the need for standards for organizing and analyzing digital data and to serve as a foundation for further developing methodologies for their use. There is also a need to develop an evidentiary framework, which would make this type of data more broadly accepted in health care, research, and public health settings.
Patrick-Lake argued that HHS could have a role in developing incentives and rewards that could reduce barriers to collaboration and promote the use and reuse of innovations. She said that there is also need for policy leadership to advance data rights and the work of the National Human Genome Research Institute’s Ethical, Legal and Social Implications Research Program, which supports the appropriate balance between individual protections and public benefit, ensuring that individuals are well informed and protected against discrimination based on their digital specimens.
CONCLUSIONS
Representatives of patient organizations argued that PCOR data are often not focused on the types of issues that are truly important to people and that would enable them to find answers to the questions they tend to have about their treatment options and potential outcomes. Information on costs was highlighted as particularly important, which is in line with the goals of the recently broadened scope of PCOR to take into consideration “the potential burdens and economic impacts of the utilization of medical treatments, items, and services.”
CONCLUSION 3-1: The patient-centered outcomes research data infrastructure has not reached its full potential to provide data that can answer questions that matter to patients and enable them to make informed decisions. Information about the cost of care was highlighted among the types of data that would be particularly useful.
While data on patient-centered outcomes are available in many areas that are important to patients, the information is rarely available in ways that would make it truly accessible to them for decision-making purposes. More widespread dissemination of information that is easy to use could also increase engagement.
CONCLUSION 3-2: Dissemination and translation of the research findings could be greatly enhanced by using forms of communication that are relevant to those outside of the research community.
___________________
This page intentionally left blank.












