Proceedings of a Workshop
| IN BRIEF | |
|
August 2021 |
Achieving Excellence in the Diagnosis of Acute Cardiovascular Events
Proceedings of a Workshop—in Brief
On April 23, 2021, the Board on Health Care Services of the National Academies of Sciences, Engineering, and Medicine hosted a virtual workshop titled Achieving Excellence in the Diagnosis of Acute Cardiovascular Events.1 The workshop, which focused on the diagnosis of acute coronary syndrome (ACS), stroke, and pulmonary embolism (PE), featured invited speakers and discussions to examine the current scientific landscape and research opportunities for improving diagnosis within the context of the U.S. health care system. The workshop highlighted system improvement strategies for diagnosis, approaches to promote equity in diagnosis, and novel diagnostic tools. This workshop was the second in a series on diagnostic excellence funded by the Gordon and Betty Moore Foundation.2 This Proceedings of a Workshop—in Brief highlights the presentations and discussions that occurred at the workshop.3
BACKGROUND
Daniel Yang from the Gordon and Betty Moore Foundation described the concept of diagnostic excellence, noting that it goes beyond the avoidance of diagnostic error and encompasses “getting to the right diagnosis, in the shortest amount of time, with the least amount of resources, in a way that is patient-centered.” He added that the diagnostic process often requires trade-offs among these objectives, but innovations in diagnostic systems and technologies have the potential to improve quality across all domains of diagnosis and increase clarity for front-line clinicians tasked with critical decision making. Yang noted that cardiovascular events are one of three clinical areas4 that the Gordon and Betty Moore Foundation has selected as priorities because errors, delays, and inefficiencies in diagnosis in these areas can be especially harmful for patients and are responsible for a disproportionate share of serious patient harm and preventable death (Newman-Toker et al., 2019).
THE BURDEN OF DIAGNOSTIC ERROR
Laura Zwaan, cognitive psychologist at the Erasmus Medical Center, described diagnostic error as a serious threat to patient safety. She noted that 10–15 percent of physician diagnoses are not entirely correct, which contributes to avoid
![]()
__________________
1 The workshop agenda and presentations are available at https://www.nationalacademies.org/event/04-23-2021/achievingexcellence-in-cardiac-diagnosis-a-workshop (accessed August 12, 2021).
2 More information about the workshop series is available at https://www.nationalacademies.org/our-work/advancing-diagnosticexcellence-a-workshop-series (accessed June 14, 2021).
3 This Proceedings of a Workshop—in Brief is not intended to provide a comprehensive summary of information shared during the workshop. The information summarized here reflects the knowledge and opinions of individual workshop participants and should not be seen as a consensus of the workshop participants, the planning committee, or the National Academies of Sciences, Engineering, and Medicine.
4 The other clinical areas include sepsis and cancers.
able patient morbidity and mortality and leads to malpractice claims (Berner and Graber, 2008; Bishop et al., 2011; Zwaan et al., 2010). Clinicians face three primary challenges when diagnosing patients: disease evolution, balancing over- versus under-diagnosis, and dealing with uncertainty. Zwaan noted that as a disease evolves, patients often move from a state of health through biochemical changes and presymptomatic disease before presenting with symptomatic illness. Clinicians often encounter patients after the onset of symptomatic disease, although symptoms may change over time, complicating the process of diagnosis. Zwaan also described the tension between the avoidance of over- and under-diagnosis, explaining that over-diagnosis arises when there is no possible benefit of diagnosing a condition and under-diagnosis arises from missed, delayed, or incorrect diagnoses. She said both over- and under-diagnosis are associated with substantial risks for patient harm.
Zwaan also addressed the difficulty of managing and communicating uncertainty throughout the diagnostic process. She noted that it is nearly impossible and potentially harmful for clinicians to pursue every possible diagnosis; as a result, the most likely diagnoses are identified using heuristics—shortcuts in problem solving. These heuristics may point clinicians to the appropriate diagnosis, but can also lead to errors in judgment and cognitive biases. Zwaan said that the most effective way to counteract these biases and avoid diagnostic errors is to enhance clinician knowledge of features that distinguish between similar diseases.
OVERVIEW OF DIAGNOSIS IN ACUTE CARDIOVASCULAR EVENTS
Acute Coronary Syndrome
Basmah Safdar, associate professor of emergency medicine at the Yale School of Medicine, provided an overview of the diagnosis of ACS,5 noting that the high volume of patients with ACS presents a significant burden on the U.S. health care system. She highlighted the time sensitivity of ACS diagnosis, noting, “Time is muscle. The earlier you diagnose, the earlier you intervene, the better the prognosis.” However, Safdar explained that ACS is one of many potential diagnoses clinicians consider when evaluating chest pain; clinicians need to differentiate between cardiac chest pain and non-cardiac chest pain, differentiate life threatening and non–life threatening symptoms, and also need to consider alternate diagnoses.
Safdar provided the example of myocardial infarction (MI) to illustrate the complexity of diagnosis: many patients who present with MI do not have classic chest pain symptoms and instead experience shortness of breath, nausea, or syncope (fainting). Symptoms of MI and other forms of ACS can differ depending on the sex of the individual, and although overall mortality rates have decreased over the past decade, women experiencing MI have higher mortality rates than men (Virani et al., 2020). She noted that diagnosis is also complicated by cognitive biases resulting from reliance on “classic” symptom presentation of ACS (e.g., chest pain), which may result in either over- or under-testing.
Safdar added that recent advances in biomarkers and non-invasive imaging have improved diagnosis, but there are still limitations, including missed diagnoses for non-classic phenotypes and presentations of ACS and over-testing. Safdar suggested that future diagnostic strategies for ACS should incorporate the evolving understanding of ACS pathophysiology and the differences in disease and symptom presentation among different patient populations. She also suggested that future work should ensure that existing protocols for diagnosis are adapted to incorporate new data from technological advancements and that future research should investigate all phenotypes of ACS and their influence on patient outcomes, diagnostic precision and risks of diagnostic errors, and health care costs.
Deborah Diercks, chair of emergency medicine at the University of Texas Southwestern Medical Center, noted that when a patient presents with chest pain, the two potential diagnoses associated with the greatest morbidity and mortality occur with the lowest frequency (ST segment elevation MI and non-ST segment elevation MI6). The majority of patients with chest pain are at intermediate or low risk for ACS. Diercks also emphasized the time sensitivity of an ACS diagnosis and described the regionalization of care through which high-risk patients in low-resource settings are quickly identified and transferred to previously designated hospitals capable of providing specialized care. Diercks described several complexities in the diagnosis of ACS, including the risk of false positives when using high-sensitive troponin assays7 and the difficulty in accurately assessing the risk for MIs. She suggested that future research should investigate the optimization of risk scores and their utility for reducing over-testing.
__________________
5 ACS includes medical conditions where blood flow to the heart is blocked, such as myocardial infarction and unstable angina.
6 ST segment elevation MI is a type of heart attack that may not cause changes on an electrocardiogram (ECG). However, chemical markers in the blood indicate that damage has occurred to the heart muscle. Non-ST segment elevation MI is a type of heart attack that is caused by an abrupt and prolonged blocked blood supply. It affects a large area of the heart muscle and causes changes on the ECG as well as in the blood levels of key chemical markers. See https://my.clevelandclinic.org/health/diseases/16713-cad-acute-coronary-syndrome (accessed July 6, 2021).
7 High-sensitivity troponin assays detect troponin, a biomarker that can signal heart muscle injury, such as a myocardial infarction (Brush et al., 2016). See https://www.uptodate.com/contents/troponin-testing-clinical-use (accessed July 19, 2021).
Stroke
Ava Liberman, assistant professor of neurology at the Montefiore Medical Center/Albert Einstein College of Medicine, explained that a stroke can be defined as a “neurological impairment caused by a disruption in blood supply to a region of the brain.” She described three types of stroke: ischemic stroke, intracerebral hemorrhage, and subarachnoid hemorrhage. Ischemic stroke (blockage in a vessel supplying blood to the brain) is the most common type of stroke in the United States. Liberman stressed that ischemic stroke is a time-sensitive diagnosis, noting that “time is brain”: the longer the duration between symptom onset and treatment, the less likely the patient is to have a favorable outcome (Saver and Levine, 2010). Liberman also suggested that the diagnosis of stroke must be made clinically; the diagnosis cannot be derived solely through radiology or other diagnostic testing.
Liberman said that the first step in stroke diagnosis is the detection of symptoms in the community setting, adding that a delay commonly occurs in this first step. She noted that patients with atypical and transient symptoms often do not seek medical care, and current community education strategies focus only on typical symptom presentation (e.g., facial asymmetry and arm weakness), which may make patients reluctant to report less severe or atypical symptoms (e.g., leg weakness or vision disturbances) (Wolters et al., 2018). Liberman added that delays may also arise through inefficiencies in the emergency medical services’ (EMS’s) detection and dispatch and the hesitance of patients to use EMS for hospital transport. In addition to the risk presented by delayed diagnosis and care, there is also a risk to patients from false positive diagnosis of stroke, particularly when a false positive stroke diagnosis precludes delivery of effective therapies for the underlying condition. Liberman suggested multiple strategies to improve stroke diagnosis, such as community education on stroke detection, better understanding of the heterogeneity of stroke symptoms and presentation, and providing clinicians with the tools to recognize atypical symptoms that may reduce the time between stroke diagnosis and treatment.
Opeolu Adeoye, professor and chair of emergency medicine at Washington University, said that individuals experiencing a stroke in minority communities are often less likely to recognize symptoms of stroke and are less likely to use EMS transport. Adeoye described the ideal prehospital triage as one in which patients with a severe stroke are taken directly to a large hospital with the resources for complex care while patients with a less severe stroke are taken to the closest hospital to reduce delays in treatment. However, limitations in EMS recognition of stroke symptoms, as well as heterogeneity in symptom presentation, create challenges for fast and effective triage. While there are several clinical scales to assist in stroke risk assessment, they often result in over-triage and the bypass of proximal hospitals for patients with less severe stroke, which unnecessarily delays treatment (Duvekot et al., 2021). Adeoye agreed that approaches to improve stroke diagnosis should focus on the prehospital setting and the development of tools to improve EMS recognition of stroke and facilitate triage. He also stressed the need to develop additional devices, biomarkers, and imaging technologies to improve diagnosis in the emergency department (ED). Finally, Adeoye called for more effective education campaigns to improve public awareness of stroke symptoms and suggested that interventions to improve diagnosis should be implemented in ways that will reduce disparities in access to care and outcomes.
Pulmonary Embolism
John Sather, assistant professor of emergency medicine at the Yale School of Medicine, noted that 10 million Americans visit the ED every year for symptoms of PE, such as chest pain, difficulty breathing, or hemoptysis (coughing up blood) (Feng et al., 2013). He described PE as a “can’t miss diagnosis” because of its high morbidity and mortality. However, there are also risks associated with over-testing and over-treatment, such as unnecessary exposure to radiation from imaging and unnecessary use of anticoagulants. To balance these risks, guidelines have been developed and implemented to optimize and standardize diagnosis and treatment.
Sather noted that diagnosis of PE is complicated by heterogeneity in disease presentation and the wide range of vulnerable patients. He added that clinicians should consider the probability that a patient has a PE in evaluating the need for diagnostic testing and treatment. Sather also emphasized the importance of characterizing the risks of morbidity and mortality associated with each case of PE to appropriately select treatment. Sather suggested that future efforts to improve the diagnosis of PE should refine diagnostic pathways, develop novel technologies to support clinical decision making, and implement measurement and feedback approaches to improve performance. He also recommended further research to understand PE risk across demographic groups and vulnerable populations, noting that identifying populations that are likely to be misdiagnosed, to receive delayed diagnoses, or be subjected to over-testing will help to close the gap in diagnostic accuracy.
Jeffrey Kline, professor of emergency medicine at the Indiana University School of Medicine, commented that PE is “simultaneously the most over-tested yet under-diagnosed condition in medicine.” He stated that low-value testing is common, noting that many computed tomography (CT) scans are performed without first administering blood tests (e.g., D-dimer testing8) that could potentially rule out the possibility of a blood clot without exposing patients to radiation. This radiation exposure is associated with increased cancer risk, particularly in young women. Additionally, Kline noted that as many as 5 percent of PE diagnoses based on scans are false positives, which results in unnecessary exposure to anticoagulant medications (Schissler et al., 2013). Despite these risks, over-testing is common because failure to diagnose PE is associated with significant morbidity and mortality. He suggested that age-adjusted D-dimer testing prior to the use of CT, together with the implementation of effective clinical decision support tools, have the potential to improve the diagnosis of PE. Kline also suggested that public information campaigns should be used to increase knowledge about the signs and symptoms of PE to facilitate prompt diagnosis and treatment.
SYSTEM IMPROVEMENT EFFORTS FOR DIAGNOSING ACUTE CARDIOVASCULAR EVENTS
Pascale Carayon, professor of industrial and systems engineering at the University of Wisconsin–Madison, said that diagnosis is a complex, iterative process embedded within multiple work systems, not a single event9 (see Figure 1). It begins when a patient identifies a health problem and seeks medical care, and entails an iterative process of gathering, interpreting, and integrating clinical information to develop a working diagnosis. This diagnosis is then communicated to the patient and treatment is initiated. Carayon noted that while steps of the diagnostic process may be subject to errors, there are also opportunities to build in mechanisms for error recovery within the diagnostic process and to capture data that can be used to learn from these errors and improve the process for future patients.
Christine Goeschel, system leader at the MedStar Health Institute for Quality and Safety, reviewed health system–level initiatives to improve diagnosis. She noted that cardiovascular disease is a devastating and costly chronic illness, and is “fraught with errors in diagnosis and treatment.” She added that meaningful progress toward diagnostic excellence requires engagement and accountability from the entire health care team, including clinical and administrative leaders: “Accountability in any area of health care involves all of us.” Goeschel emphasized the critical importance of involving patient and family voices in the diagnostic process. She suggested that efforts targeted at improving diagnosis should be integrated with existing quality and safety improvement programs, and also called for the deliberate and intentional measurement of diagnostic quality to support quality improvement. She said that achieving diagnostic excellence will require providing feedback on diagnostic performance, building workforce capacity, and ensuring appropriate time and resources are dedicated. “Diagnostic excellence for cardiovascular disease is … worthy of our effort,
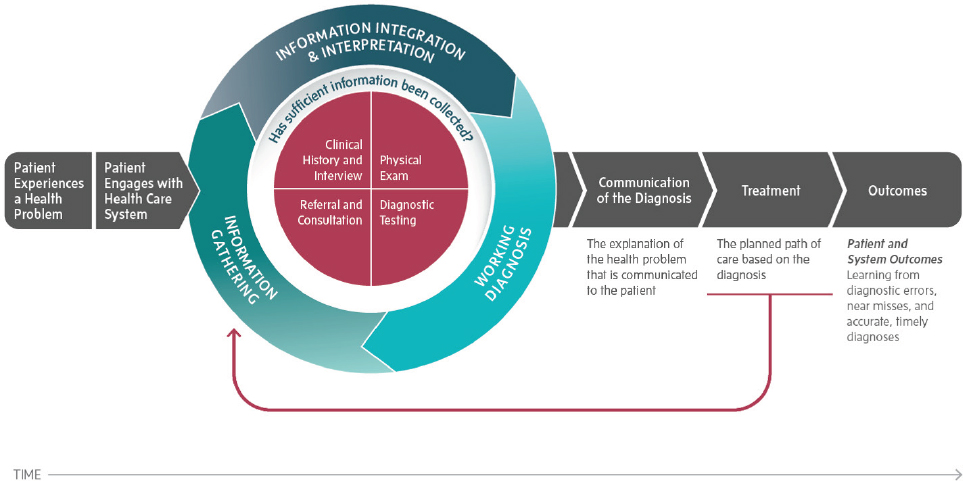
SOURCES: Pascale Carayon presentation, April 23, 2021; NASEM, 2015, p. 33.
__________________
8 Blood levels of D-dimer—a protein fragment—can be used to help clinicians assess whether a patient may or may not be experiencing a PE (Adam et al., 2009; Gao et al., 2018). See https://medlineplus.gov/lab-tests/d-dimer-test (accessed July 19, 2021).
9 For more information on this model, see Improving Diagnosis in Health Care (NASEM, 2015).
as the number one killer and highest cost chronic disease in the country,” she concluded, “and health systems need to urgently strive toward achieving diagnostic excellence today.”
Don Casey, founder of Improving Patient Outcomes for Health (IPO4Health), described the biologic and health system delivery complexities of ACS that require multiple sequential diagnostic steps to inform specific therapeutic interventions. He emphasized the importance of utilizing current ACS clinical practice guidelines from the American College of Cardiology and the American Heart Association (which he co-authored) that provide explicit scientific evaluation and grading of evidence and recommendations for diagnostic and treatment interventions (Amsterdam et al., 2014; O’Gara et al., 2013).
Casey also described the complex system for ACS care, which is time dependent and involves an intricate composite of many “Structure/Process/Outcome” variables (in accordance with the Donabedian conceptual model for health care quality). The complexities and interconnected nature of these domains often contribute to diagnostic uncertainty. Because delays in seeking care for suspected ACS symptoms can contribute to worse patient outcomes, he said that there is a need to better understand patient decision making prior to engaging in the health care system, including activation of EMS. “Just getting a person to the right place at the right time with the right resources is the big challenge,” noted Casey.
Elliott Haut, vice chair for quality, safety, and service in surgery at the Johns Hopkins University School of Medicine, discussed the use of guidelines in the diagnosis of acute cardiovascular events. He noted that the implementation of guidelines can improve the safety and quality of care, and can also reduce inappropriate practice variations and mitigate disparities. Haut discussed the application of care guidelines to the prevention of venous thromboembolism (VTE), emphasizing the importance of active dissemination and implementation. He described a pair of studies conducted by Haut et al. (2012) and Streiff et al. (2012) in which a computerized clinical decision support tool for VTE risk stratification and prophylaxis was incorporated into clinicians’ normal workflows. This system-level intervention improved care by increasing the use of risk-appropriate prophylaxis and reducing preventable harm. Haut also discussed the application of care guidelines to diagnosis, noting that the process is complicated by the need to identify which diagnostic options could be explored prior to referencing a particular guideline. He suggested that it may be more effective to develop guidelines for diagnostic evaluation and management strategies that cover multiple conditions.
ACHIEVING EQUITY IN DIAGNOSIS AND MITIGATING DISPARITIES IN ACUTE CARDIOVASCULAR EVENTS
Clyde Yancy, vice dean of diversity and inclusion at the Northwestern University Feinberg School of Medicine, discussed racial and ethnic disparities in acute decompensated heart failure, noting that the condition disproportionately affects middle-aged Black men (Virani et al., 2021). He said that despite precise clinical guidelines for diagnosis and treatment of the condition, Black patients are less likely than White patients to be referred to high-level care. Yancy emphasized that there is no physiological basis for these disparities in care and stressed that standards of care should be applied uniformly, regardless of an individual’s sex, gender, race, ethnicity, or age. He stated that failure to apply established standards of care in acute decompensated heart failure is driven by stereotyping, bias, and institutional racism, but noted that large-scale quality improvement efforts may be able to partially counteract these effects. Yancy also said that in some cases, this bias has been incorporated into algorithms used for hospital triage. He added that such algorithms should be examined to ensure that they do not systematically disadvantage Black patients, and that professional diversity in discovery science10 is extremely important to help mitigate disparities.
Urmimala Sarkar, professor of medicine at the University of California, San Francisco (UCSF), discussed the importance of patient–clinician communication for equity in the diagnosis of acute cardiovascular events. She noted that implicit bias may drive clinical decision making in ways that lead to lower referral rates to appropriate cardiac care for women and Black patients (Schulman et al., 1999). Sarkar noted that disparities in care and outcomes occur at multiple stages of the diagnostic process. To achieve a timely diagnosis, patients need to know when to seek care and how to recognize disease symptoms. After seeking medical care, disparities may arise from failures in patient–clinician communication. Sarkar noted that communication within clinical visits is “a complex and multi-level activity”; clinicians need to understand a patient’s disease state, barriers to care, and social determinants of health, while also providing them with information about how to manage their disease. Language skills and health literacy are crucial to this complex process, Sarkar said. She noted that non-White and non-English speaking patients are less likely to return for appropriate follow-up care, often because of perceived disrespect or lack of information about screening and diagnosis (Allen et al., 2008; Karliner et al., 2012). Sarkar suggested that to improve patient–clinician communication and reduce dispari-
__________________
10 Discovery science encompasses basic, translational, and computational science with the aim to discover new therapies (Juffermans et al., 2020).
ties in the diagnosis of acute cardiovascular events, it is important to support evidence-based communication practices, such as teach-back11 and literacy-appropriate communication. She also suggested that clinicians and health systems should work to combat implicit bias in clinical encounters, and that patient safety research should include diverse populations and systematically collect data that can be used to assess and address disparities. She added that among low-income, non-White, limited English proficiency populations, there is high interest in using telehealth and other digital modalities. Additionally, Sarkar noted that increased workforce diversity will go a long way in helping patients, especially immigrants, feel comfortable in health care settings.
Herman Taylor, professor of cardiology at the Morehouse School of Medicine, discussed disparities in the earliest stages of identifying and diagnosing acute cardiovascular events, noting that it is crucial to better understand the “epidemiology of delay.” He said that Black patients often experience delays in diagnosis relative to White patients. These delays are particularly salient for health outcomes after acute cardiovascular events; every 30 minutes of delayed treatment during an acute MI increases 1-year mortality by 7.5 percent (De Luca et al., 2004). Taylor noted that patient denial and misattribution of symptoms are common reasons for delays in seeking care. Among Black patients, misattribution of symptoms to other conditions, such as asthma or digestive problems is particularly common (Raczynski et al., 1994). Black patients may also face additional social barriers to seeking care, such as job insecurity and lack of trust in the medical system.
Taylor noted that another contributing factor for delayed diagnosis is disparities in prehospital triage and EMS response. He described a study analyzing data from more than 63,000 cardiac arrests, which found that EMS response and transport times were longer for low-income patients compared to high-income patients. He explained that a key driver of extended EMS transport times is hospital diversion,12 which is more common in hospitals that serve minority communities (Shen and Hsia, 2016). Diversion and subsequent delays in care are associated with a nearly 10 percent increase in mortality from MI (Shen and Hsia, 2016). To address disparities in the diagnosis of acute cardiovascular events, Taylor recommended community-engaged research to better understand patient perspectives and potential strategies to address barriers to seeking timely care. He also emphasized the importance of diversity at every level of the health care team, from EMS dispatchers to surgeons, describing the results of a study demonstrating that Black–White cardiovascular disparities could be reduced by improving the racial and ethnic diversity of clinicians (Alsan et al., 2018).
NOVEL DIAGNOSTIC TOOLS AND STRATEGIES FOR ACUTE CARDIOVASCULAR EVENTS
Rima Arnaout, associate professor of medicine at UCSF, discussed the potential of machine learning (ML) tools to reduce diagnostic gaps in cardiovascular care. She said that the goal of these tools is to ensure an accurate, timely, reproducible diagnosis that is robust across diverse patient populations and care settings. “When diagnostic gaps exist,” she said, “they can be due to insufficient scientific knowledge or to variability in applying available knowledge because of conscious or unconscious biases, or simply because … to quote a colleague, ‘the complexity of medicine now exceeds the capacity of the human mind’” (Obermeyer and Lee, 2017).
Arnaout noted that ML algorithms differ from traditional rules-based algorithms because ML algorithms learn patterns from data. Well-designed, appropriately validated ML algorithms should perform reliably across settings and patient populations. This presents the opportunity to reduce diagnostic gaps due to clinician bias or lack of expertise. She noted that in the future, ML algorithms may also be able to surpass human judgment by identifying data characteristics not easily apparent to human perception (Quer et al., 2021).
Arnaout described her use of ML algorithms to screen for prenatal congenital heart disease: by applying ML tools to second trimester ultrasound images, the project was able to double the sensitivity and specificity of the test (Arnaout et al., 2021). She noted that while ML offers exciting potential for improving diagnosis, its future depends on scientific rigor and adoption by the medical community. Arnaout called for ML researchers to develop algorithms to address specific clinical problems and to clearly describe model performance to enable clinicians to assess clinical value. She also called for leaders in cardiovascular diagnostics to be critical consumers of ML advancements, to participate in responsible data sharing, and to support the implementation and integration of validated ML tools. Finally, Arnaout emphasized the importance of supporting data infrastructure and ensuring that data sets used to develop algorithms are representative of diverse patients and populations to mitigate the potential for ML tools to perpetuate biases.
__________________
11 Teach-back is a technique clinicians can employ to ensure that they have explained medical information clearly so that patients and their families understand what is communicated to them. See https://www.ahrq.gov/patient-safety/reports/engage/interventions/teachback.html (accessed July 19, 2021).
12 Diversion occurs when an ED’s patient load exceeds its capacity and incoming patients are sent to alternate, often more distant, hospitals.
Derick En’Wezoh, vice president of growth at Viz.ai,13 echoed Liberman’s earlier comments about the time-critical nature of timely stroke diagnosis and treatment, stating that for every minute delay in intervention, patients lose nearly 2 million neurons (Saver, 2006). Stroke care and workflow is complex and challenging, explained En’Wezoh, and involves numerous clinicians, imaging modalities, and the coordination of services. He noted that there are a growing number of technology solutions to improve stroke diagnosis in the face of these challenges, including mobile units with CT technology and helmets that can diagnose a stroke outside of medical settings.
En’Wezoh emphasized the potential of artificial intelligence (AI)-powered systems to improve stroke diagnosis and care. He presented the example of Viz.ai’s tool for stroke care, which uses AI to improve care coordination and facilitate the early identification and treatment of stroke for patients in hospital settings. The system comprises an AI detection tool that analyzes CT data to identify patients with stroke, as well as real-time automated alerts that are sent to members of the patient’s care team. This enables the synchronization of stroke care across clinical specialties, facilitating early treatment. En’Wezoh noted that Viz.ai’s tool has demonstrated substantial clinical improvements over traditional care, including reduced treatment delays, shorter lengths of stay in the intensive care unit and hospital, and reduced stroke-related disability after 90 days (Hassan et al., 2020; Morey et al., 2020).
En’Wezoh noted that, despite broad academic interest, there are relatively few AI diagnostic tools currently in clinical use. He said that this is partially due to the challenges associated with bringing technologies to market, including the complexity of the regulatory environment, the difficulty of commercializing and selling an AI tool to health systems, and the difficulty of achieving clinician adoption and integration in clinical care. En’Wezoh emphasized the importance of demonstrating that new technologies for diagnosis make meaningful differences in patient outcomes and experiences. He also noted that as AI-powered technologies become more common, payer organizations (e.g., the Centers for Medicare & Medicaid Services) are increasingly opening pathways for reimbursement for their use.
Eric Topol, director of the Scripps Research Translational Institute, reiterated that ML and AI technologies have the potential to improve the quality of diagnosis for acute cardiovascular events and to improve patients’ experiences during the diagnostic process. He added that research on ML and AI applications for diagnosis is in its infancy, with many additional tools in development. “It’s an exciting time—I don’t know anything more transformative than machine learning applications to improve health care,” he said. However, he also emphasized the importance of ensuring that tool development, validation, and implementation result in meaningful improvements to patient outcomes. Topol also stated that the creation and maintenance of large, high-quality clinical data sets is a key requirement for the development of ML and AI diagnostic tools. He suggested that data sharing among health systems and investments in data engineering and infrastructure will help to achieve this goal.
A VISION FOR THE FUTURE OF DIAGNOSIS
In a final session, panelists reflected on their visions for diagnostic excellence in acute cardiovascular events. Carolyn Thomas of Heart Sisters recounted the misdiagnosis of her own heart attack and emphasized the importance of collecting data on diagnostic errors. She added, “we don’t have mandatory reporting of diagnostic error, so it’s hard to address something or fix something when we don’t measure it.”
Building on Thomas’s comments about measurement, Marina Del Rios, associate professor of emergency medicine at the University of Illinois at Chicago, stated that achieving diagnostic excellence should include equity as an outcome and offered three primary suggestions: diversify the health workforce to mitigate inequities in diagnosis and outcomes; diversify discovery science, especially in the design and use of new technology; and include equity as an outcomes measure to achieve diagnostic excellence. Gari Clifford, professor of biomedical informatics and biomedical engineering at Emory University and the Georgia Institute of Technology, agreed and reflected on the importance of diversity in discovery science, adding “the coding teams and the data wranglers need to be diverse and see themselves in the data.” Del Rios discussed other strategies to mitigate inequalities in diagnosis, including the increased use of evidence-based guidelines, though she cautioned that these tools are used primarily for treatment after diagnosis and much of the evidence is based on clinical trials that lack diversity. She added that implementation science research is needed to evaluate how new technologies, new diagnostic algorithms, and established evidence-based guidelines are performing in diverse patient populations. She noted that payment models can also affect care quality, though punitive actions may have unintended consequences for hospitals that serve marginalized populations.
Bernadette Boden-Albala, professor at the University of California, Irvine, School of Medicine, added that the accurate and timely diagnosis of cardiovascular events spans both the community and the health system and will require thinking about each step and participant along the entire care continuum. She added that educating and engaging with at-risk communities, especially prior to the occurrence of a cardiovascular event (i.e., “preparedness actions”),
__________________
13 As of June 14, 2021, Derek En’Wezoh is affiliated with Susa Ventures.
are extremely important. Del Rios agreed, adding that “one of the biggest mistakes that we as health care providers make is confusing outreach with engagement … what you really need to do is have conversations.”
Building on these comments, Goeschel emphasized that “everybody in the health care domain is part of the diagnostic team,” and that it is important that “patients are engaged in every aspect of this work, from research to policy formation and implementation, to ongoing operational issues.” Thomas agreed with the importance of patient engagement in the diagnostic process, adding that patients are the best source of information on their own body. Goeschel said that in order to affect change at the health system level, leadership buy-in is pivotal, and that means showing organizations concrete data, such as patient satisfaction and outcomes, that will open their eyes to opportunities for diagnostic improvement.
Reflecting on new technologies for diagnostic excellence in cardiovascular events, Clifford said that he was most concerned about trust in commercialized AI algorithms, and he stressed that aftermarket vigilance (following U.S. Food and Drug Administration approval) is crucial to assess whether an algorithm is working well in real-world clinical settings. He added that algorithms are sequestered and constantly updated by independent entities, so a set of standards and databases to benchmark AI algorithms with comparable metrics of success is crucial to avoid unreliable or biased algorithms. John Halamka, president of the Mayo Clinic Platform, predicted that “the future belongs to continuous non-episodic monitoring of devices that you wear, devices in your home, and devices that you carry that give you the benefit of AI algorithms tuned to your disease and your demographics and phenotype, with reasonable event notification when there is a material change” in your health status. He added that when it comes to new technologies, it is important to meet patients at their level of technological comfort and address disparities in access. Clifford agreed and emphasized the importance of communication with patients and communities when developing new technologies. Individual speakers made further suggestions for achieving excellence in the diagnosis of acute cardiovascular events (see Box 1).
In his summary remarks, Andrew Bindman of Kaiser Permanente emphasized a theme seen throughout the workshop: “diagnosis is ultimately a team sport.” He cautioned that although new diagnostic tools—in the form of biomarkers, AI, and ML—offer tremendous opportunities to gain new insights, researchers and clinicians should be on alert for unintended effects, including increased over-diagnosis. Bindman closed the workshop with a cross-cutting observation from the discussions: “it is no longer acceptable to pursue improvement in diagnosis for some, we must pursue diagnostic excellence for all.”
BOX 1
Suggestions from Individual Workshop Participants to Achieve Excellence in the Diagnosis of Acute Cardiovascular Events
Improving Patient and Clinician Education
- Enhance disease-specific knowledge and features that distinguish between similar presenting diseases to counteract cognitive biases (Zwaan).
- Improve clinician recognition of the heterogeneous nature of acute cardiovascular events, including evolving phenotypes and atypical or minor symptoms, to avoid delays in diagnosis and treatment (Liberman, Safdar, Taylor, Thomas).
- Increase community engagement and patient awareness of stroke symptoms, including atypical and transient symptoms, to improve timely diagnosis and treatment (Adeoye, Boden-Albala, Kline, Liberman).
- Encourage and support effective communication between patients and their clinicians in the diagnostic process (Goeschel, Sarkar, Thomas).
- Prioritize interventions focused on prehospital settings and those that address patient-related factors that contribute to delays in seeking care for acute cardiovascular events. (Adeoye, Casey, Liberman, Sather, Taylor).
Developing Innovations in Technology and Care Delivery
- Provide guidance for data sharing and transparency to achieve the full potential of artificial intelligence and machine learning (Arnaout, En’Wezoh, Halmaka, Topol).
- Increase the use of D-dimer testing prior to computed tomography scans to reduce over-testing for pulmonary embolism (Kline).
- Utilize telemedicine to make quicker diagnoses and provide health care access to resource-limited and rural communities (Liberman, Sather).
- Employ community participatory research methods and more rigorous evaluation of the implementation of digital tools in diverse communities (Sarkar).
Strategies to Improve Diagnostic Equity
- Increase diversity in the clinical and discovery science workforce and ensure patient diversity in clinical trials (Clifford, Del Rios, Sarkar, Taylor, Yancy).
- Improve education on how cardiovascular conditions affect different patient populations and how implicit bias among clinicians can influence patient outcomes (Sarkar, Yancy).
- Utilize practice guidelines to avoid inappropriate variations in care and to reduce disparities in care (Haut).
- Implement evidence-based communication practices, such as the teach-back method, to ensure all patients understand their diagnosis and care plan (Sarkar).
Opportunities for Future Research Initiatives
- Identify how symptom presentation varies among different patient populations (Safdar, Sarkar, Sather, Yancy).
- Standardize data collection on the social determinants of health for cardiovascular disease (Arnaout, Goeschel).
- Investigate how risk scores can be used to optimize diagnostic test use and reduce over-testing (Diercks).
- Develop practices and tools to improve prehospital care, such as devices to detect hemorrhage and portable advanced imaging (Adeoye).
- Investigate the epidemiology of care delays, especially in minority communities (Taylor).
NOTE: This list is the rapporteurs’ summary of points made by the individual speakers identified and the statements have not been endorsed or verified by the National Academies of Sciences, Engineering, and Medicine. They are not intended to reflect a consensus among workshop participants.
REFERENCES
Adam, S. S., N. S. Key, and C. S. Greenberg. 2009. D-dimer antigen: Current concepts and future prospects. Blood 113(13):2878–2887.
Allen, J. D., R. C. Shelton, E. Harden, and R. E. Goldman. 2008. Follow-up of abnormal screening mammograms among low-income ethnically diverse women: Findings from a qualitative study. Patient Education and Counseling 72(2):283–292.
Alsan, M., G. Owen, and G. C. Graziani. 2018. Does diversity matter for health? Experimental evidence from Oakland. American Economic Review 109(12):4071–4111.
Amsterdam, E. A., N. K. Wenger, R. G. Brindis, D. E. Casey, T. G. Ganiats, D. R. Holmes, A. S. Jaffe, H. Jneid, R. F. Kelly, M. C. Kontos, G. N. Levine, P. R. Liebson, D. Mukherjee, E. D. Peterson, M. S. Sabatine, R. W. Smalling, and S. J. Zieman. 2014. 2014 AHA/ACC guideline for the management of patients with non-ST-elevation acute coronary syndromes. Circulation 130(25):e344–e426.
Arnaout, R., L. Curran, Y. Zhao, J. C. Levine, E. Chinn, and A. J. Moon-Grady. 2021. An ensemble of neural networks provides expert-level prenatal detection of complex congenital heart disease. Nature Medicine 27(5):882–891.
Berner, E. S., and M. L. Graber. 2008. Overconfidence as a cause of diagnostic error in medicine. The American Journal of Medicine 121(5 Suppl):S2–S23.
Bishop, T. F., A. M. Ryan, and L. P. Casalino. 2011. Paid malpractice claims for adverse events in inpatient and outpatient settings. JAMA 305(23):2427–2431.
Brush, J. E., S. Kaul, and H. M. Krumholz. 2016. Troponin testing for clinicians. Journal of the American College of Cardiology 68(21):2365–2375.
De Luca, G., H. Suryapranata, J. P. Ottervanger, and E. M. Antman. 2004. Time delay to treatment and mortality in primary angioplasty for acute myocardial infarction: Every minute of delay counts. Circulation 109(10):1223–1225.
Duvekot, M. H. C., E. Venema, A. D. Rozeman, W. Moudrous, F. H. Vermeij, M. Biekart, H. F. Lingsma, L. Maasland, A. D. Wijnhoud, L. Mulder, K. C. L. Alblas, R. P. J. van Eijkelenburg, B. I. Buijck, J. Bakker, A. S. Plaisier, J. H. Hensen, G. J. Lycklama, À. Nijeholt, P. J. van Doormaal, A. van Es, A. van der Lugt, H. Kerkhoff, D. W. J. Dippel, and B. Roozenbeek. 2021. Comparison of eight prehospital stroke scales to detect intracranial large-vessel occlusion in suspected stroke (presto): A prospective observational study. The Lancet Neurology 20(3):213–221.
Feng, L. B., J. M. Pines, H. R. Yusuf, and S. D. Grosse. 2013. U.S. trends in computed tomography use and diagnoses in emergency department visits by patients with symptoms suggestive of pulmonary embolism, 2001–2009. Academic Emergency Medicine 20(10):1033–1040.
Gao, H., H. Liu, and Y. Li. 2018. Value of D-dimer levels for the diagnosis of pulmonary embolism: An analysis of 32 cases with computed tomography pulmonary angiography. Experimental and Therapeutic Medicine 16(2):1554–1560.
Hassan, A. E., V. M. Ringheanu, R. R. Rabah, L. Preston, W. G. Tekle, and A. I. Qureshi. 2020. Early experience utilizing artificial intelligence shows significant reduction in transfer times and length of stay in a hub and spoke model. Interventional Neuroradiology 26(5):615–622.
Haut, E. R., B. D. Lau, F. S. Kraenzlin, D. B. Hobson, P. S. Kraus, H. T. Carolan, A. H. Haider, C. G. Holzmueller, D. T. Efron, P. J. Pronovost, and M. B. Streiff. 2012. Improved prophylaxis and decreased rates of preventable harm with the use of a mandatory computerized clinical decision support tool for prophylaxis for venous thromboembolism in trauma. Archives of Surgery 147(10):901–907.
Juffermans, N. P., P. Radermacher, J. G. Laffey, on behalf of the Translational Biology Group. 2020. The importance of discovery science in the development of therapies for the critically ill. Intensive Care Medicine Experimental 8(1):17.
Karliner, L. S., A. Auerbach, A. Nápoles, D. Schillinger, D. Nickleach, and E. J. Pérez-Stable. 2012. Language barriers and understanding of hospital discharge instructions. Medical Care 50(4):283–289.
Morey, J. R., E. Fiano, K. A. Yaeger, X. Zhang, and J. T. Fifi. 2020. Impact of Viz LVO on time-to-treatment and clinical outcomes in large vessel occlusion stroke patients presenting to primary stroke centers. medRxiv 2020.2007.2002.20143834.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2015. Improving diagnosis in health care. Washington, DC: The National Academies Press.
Newman-Toker, D. E., A. C. Schaffer, C. W. Yu-Moe, N. Nassery, A. S. S. Tehrani, G. D. Clemens, Z. Wang, Y. Zhu, M. Fanai, and D. Siegal. 2019. Serious misdiagnosis-related harms in malpractice claims: The “big three”—vascular events, infections, and cancers. Diagnosis 6(3):227–240.
Obermeyer, Z., and T. H. Lee. 2017. Lost in thought—the limits of the human mind and the future of medicine. New England Journal of Medicine 377(13):1209–1211.
O’Gara, P. T., F. G. Kushner, D. D. Ascheim, D. E. Casey, M. K. Chung, J. A. d. Lemos, S. M. Ettinger, J. C. Fang, F. M. Fesmire, B. A. Franklin, C. B. Granger, H. M. Krumholz, J. A. Linderbaum, D. A. Morrow, L. K. Newby, J. P. Ornato, N. Ou, M. J. Radford, J. E. Tamis-Holland, C. L. Tommaso, C. M. Tracy, Y. J. Woo, and D. X. Zhao. 2013. 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction. Circulation 127(4):e362–e425.
Quer, G., R. Arnaout, M. Henne, and R. Arnaout. 2021. Machine learning and the future of cardiovascular care: JACC state-of-the-art review. Journal of the American College of Cardiology 77(3):300–313.
Raczynski, J. M., H. Taylor, G. Cutter, M. Hardin, N. Rappaport, and A. Oberman. 1994. Diagnoses, symptoms, and attribution of symptoms among black and white inpatients admitted for coronary heart disease. American Journal of Public Health 84(6):951–956.
Saver, J. L. 2006. Time is brain—quantified. Stroke 37(1):263–266.
Saver, J. L., and S. R. Levine. 2010. Alteplase for ischaemic stroke—much sooner is much better. The Lancet 375(9727):1667–1668.
Schissler, A. J., A. Rozenshtein, M. E. Kulon, G. D. N. Pearson, R. A. Green, P. D. Stetson, D. J. Brenner, B. D’Souza, W.-Y. Tsai, N. W. Schluger, and A. J. Einstein. 2013. CT pulmonary angiography: Increasingly diagnosing less severe pulmonary emboli. PLOS ONE 8(6):e65669.
Schulman, K. A., J. A. Berlin, W. Harless, J. F. Kerner, S. Sistrunk, B. J. Gersh, R. Dubé, C. K. Taleghani, J. E. Burke, S. Williams, J. M. Eisenberg, W. Ayers, and J. J. Escarce. 1999. The effect of race and sex on physicians’ recommendations for cardiac catheterization. New England Journal of Medicine 340(8):618–626.
Shen, Y.-C., and R. Y. Hsia. 2016. Do patients hospitalized in high-minority hospitals experience more diversion and poorer outcomes? A retrospective multivariate analysis of Medicare patients in California. BMJ Open 6(3):e010263.
Streiff, M. B., H. T. Carolan, D. B. Hobson, P. S. Kraus, C. G. Holzmueller, R. Demski, B. D. Lau, P. Biscup-Horn, P. J. Pronovost, and E. R. Haut. 2012. Lessons from the Johns Hopkins multi-disciplinary venous thromboembolism (VTE) prevention collaborative. BMJ 344:e3935.
Virani, S. S., A. Alonso, E. J. Benjamin, M. S. Bittencourt, C. W. Callaway, A. P. Carson, A. M. Chamberlain, A. R. Chang, S. Cheng, F. N. Del-ling, L. Djousse, M. S. V. Elkind, J. F. Ferguson, M. Fornage, S. S. Khan, B. M. Kissela, K. L. Knutson, T. W. Kwan, D. T. Lackland, T. T. Lewis, J. H. Lichtman, C. T. Longenecker, M. S. Loop, P. L. Lutsey, S. S. Martin, K. Matsushita, A. E. Moran, M. E. Mussolino, A. M. Perak, W. D. Rosamond, G. A. Roth, U. K. A. Sampson, G. M. Satou, E. B. Schroeder, S. H. Shah, C. M. Shay, N. L. Spartano, A. Stokes, D. L. Tirschwell, L. B. VanWagner, and C. W. Tsao. 2020. Heart disease and stroke statistics—2020 update: A report from the American Heart Association. Circulation 141(9):e139–e596.
Virani, S. S., A. Alonso, H. J. Aparicio, E. J. Benjamin, M. S. Bittencourt, C. W. Callaway, A. P. Carson, A. M. Chamberlain, S. Cheng, F. N. Del-ling, M. S. V. Elkind, K. R. Evenson, J. F. Ferguson, D. K. Gupta, S. S. Khan, B. M. Kissela, K. L. Knutson, C. D. Lee, T. T. Lewis, J. Liu, M. S. Loop, P. L. Lutsey, J. Ma, J. Mackey, S. S. Martin, D. B. Matchar, M. E. Mussolino, S. D. Navaneethan, A. M. Perak, G. A. Roth, Z. Samad, G. M. Satou, E. B. Schroeder, S. H. Shah, C. M. Shay, A. Stokes, L. B. VanWagner, N.-Y. Wang, and C. W. Tsao. 2021. Heart disease and stroke statistics—2021 update. Circulation 143(8):e254–e743.
Wolters, F. J., L. Li, S. A. Gutnikov, Z. Mehta, and P. M. Rothwell. 2018. Medical attention seeking after transient ischemic attack and minor stroke before and after the UK face, arm, speech, time (FAST) public education campaign: Results from the Oxford vascular study. JAMA Neurology 75(10):1225–1233.
Zwaan, L., and H. Singh. 2015. The challenges in defining and measuring diagnostic error. Diagnosis (Berlin, Germany) 2(2):97–103.
Zwaan, L., M. de Bruijne, C. Wagner, A. Thijs, M. Smits, G. van der Wal, and D. R. Timmermans. 2010. Patient record review of the incidence, consequences, and causes of diagnostic adverse events. Archives of Internal Medicine 170(12):1015–1021.
DISCLAIMER: This Proceedings of a Workshop—in Brief was prepared by Erin Balogh, Emily Zevon, Megan Snair, Ruth Cooper, and Jennifer Lalitha Flaubert as a factual summary of what occurred at the workshop. The statements made are those of the rapporteurs or individual workshop participants and do not necessarily represent the views of all workshop participants; the planning committee; or the National Academies of Sciences, Engineering, and Medicine.
*The National Academies of Sciences, Engineering, and Medicine’s planning committees are solely responsible for organizing the workshop, identifying topics, and choosing speakers. The responsibility for the published Proceedings of a Workshop—in Brief rests with the rapporteurs and the institution.
The planning committee comprises Andrew Bindman (Chair), Kaiser Permanente; Kirstin Bibbins-Domingo, University of California, San Francisco; Pascale Carayon, University of Wisconsin–Madison; Gari Clifford, Emory University; Marina Del Rios, University of Illinois at Chicago; Kathryn McDonald, Johns Hopkins University; Robert Neumar, University of Michigan; and Saul Weingart, Tufts University.
REVIEWERS: To ensure that it meets institutional standards for quality and objectivity, this Proceedings of a Workshop—in Brief was reviewed by Basmah Safdar, Yale University. Leslie J. Sim, National Academies of Sciences, Engineering, and Medicine, served as the review coordinator.
STAFF: Jennifer Lalitha Flaubert, Emily Zevon, Erin Balogh, Ruth Cooper, Cyndi Trang, Anesia Wilks, and Sharyl Nass, Board on Health Care Services, Health and Medicine Division, National Academies of Sciences, Engineering, and Medicine.
SPONSOR: This workshop was supported by the Gordon and Betty Moore Foundation.
For additional information regarding the workshop, visit https://www.nationalacademies.org/event/04-23-2021/achieving-excellence-in-cardiac-diagnosis-a-workshop.
Suggested citation: National Academies of Sciences, Engineering, and Medicine. 2021. Achieving excellence in the diagnosis of acute cardiovascular events: Proceedings of a workshop—in brief. Washington, DC: The National Academies Press. https://doi.org/10.17226/26309.
Health and Medicine Division

Copyright 2021 by the National Academy of Sciences. All rights reserved.











