Below is the uncorrected machine-read text of this chapter, intended to provide our own search engines and external engines with highly rich, chapter-representative searchable text of each book. Because it is UNCORRECTED material, please consider the following text as a useful but insufficient proxy for the authoritative book pages.
Chemical Composition of Petroleum Hydrocarbon Sources INTRODUCTION Sources of hydrocarbons enter ing the mar ine environment are numerous, and the number of individual hydrocarbon components are quite large. Thus, the analytical chemist faces a challenge in der iving detailed compositional data on a given environmental sample as does the bio- geochemist in associating a given complex hydrocarbon a~s~mblaa" with one or more sources. The interpretive dilemma becomes further aggra- vated by chemical and microbial alterations that occur after intro- duction of a particular set of hydrocarbon compounds to the mar ine environment, a set originally attributable to a source but subsequently modif led. This section bight ights the main compositional character- istics of the hydrocarbon sources to the mar ine environment and distinguishes features of each source. ~7 CRUDE OILS Petroleum formation and composition have been discussed in detail recently by Tissot and Welte (1979) and Hunt (1980), and unless otherwise noted, those texts are the sources for the following information. The chemical composition of crude oils from different producing regions, and even from within a particular formation, can vary tremendously. Crude oils contain thousands of different chemical compounds owing to processes dur ing petroleum formation resulting in "molecular scrambling. " Hydrocarbons are the most abundant compounds in crude oils, accounting for 50-989 of the total composition (R.C . Clark and Brown , 1977 I, although the major ity of crude oils contain the higher relative amounts of hydrocarbons. While carbon (80-879~) and hydrogen (10-15~) are the main elements in petroleum, sulfur (0-10%) nitrogen (0-1%), and oxygen (0-5%) are important minor constituents present as elemental sulfur or as heterocyclic constituents and functional groups. Compounds containing N. S. O as constituents are often collectively referred to as NSO compounds. Crude oils contain widely varying concentrations of trace metals such as V, Ni, Fe, Al, Na, Ca, Cu. and U (Posthuma, 1977~. 17
18 Table 1-1 presents examples of the composition of crude oils and fuel oils. Petroleum hydrocarbons (Figure 1-1) consist of alkanes, cycloalkanes, and aromatic compounds containing at least one benzene ring. The alkanes, or aliphatic hydrocarbons, consist of the fully saturated normal alkanes (also called paraffins) and branched alkanes of the gener al molecular formula (CnH2n+2 ), with n r ang ing f rom 1 to usually around 40, although compounds with 60 carbons have been reported. Above C13, the most important group of branched compounds is the isoprenoid hydrocarbons consisting of isoprene building blocks. Pristane (Cl") and phytane (C20) are usually the most abundant isoprenoids, and while the C10-C20 isoprenoids are often ma jor petroleum constituents, extended ser. ies of isoprenoids (C20-C40) have been repor ted (Albaiges, 1980 ~ . Many of the cycloalkanes or saturated r ing structures, also called cycloparaff ins or naphthenes, consist of important minor constituents that, 1 ike the isoprenoids, have specif ic animal or plant precursors ~ e . g ., ster anes , d iterpanes , tr iterpanes ~ and that serve as impor ten t molecular markers in of' spill and geochemical studies (Albaiges and Albrecht, 1979; Dastillung and Albrecht, 1976 ~ . Aromatic hydrocarbons, usually less abundant than the saturated hydrocarbons, contain one or more aromatic (benzene) r ings connected as fused rings (e.g., naphthalene) or lined rings (e.g., biphenyl). Petroleum contains many homologous ser ies of aromatic hydrocarbons consisting of unsubstituted or parent aromatic structures (e.g., phenanthrene) and like structures with alkyl side chains that replace hydrogen atoms. Alkyl substitution is most prevalent in 1-, 2-, and 3-r inged aromatics, although the higher polynuclear aromatic compounds (>3 r ings) do contain alkylated (1-3 carbons) side groups. The polycyclic aromatics with more than 3 r ings consist mainly of pyrene, chrysene, benzanthracene, benzopyrene, benzofluorene, benzof l uor anthene, and perylene structures. The naphthenoaromatic compounds consist of mixed structures of aromatic and saturated cyclic rings. This series increases in importance in the higher boiling fractions along with the saturated naphthenic series. The naphthenoaromatics appear related to resins, kerogen, and sterols. Petroleum generation usually involves the formation of some naphthenoaromatic structures. The nonhydrocarbon petroleum constituents, such as examples in Figure 1-lc, can be grouped into s ix classes according to Posthuma (1977~: sulfur compounds, nitrogen compounds, porphyrins, oxygen compounds, asphaltenes, and trace metals. Sulfur compounds compr ise the most important group of nonhydrocarbon constituents. Most sulfur present is organically bound, e.g., heterocyclic. The organosulfur compounds consist of thiols, disulfides, sulfides, cyclic sulfides ~ e . g ., th iacyclohexanes ), and th iophenes . The benzoth iophenes and d ibenzoth iophenes are important constituents of the h igher-molecular- weight aromatic fractions of env~.ronmental samples, with the tetramethyl dibenzothiophenes apparently having the highest molecular weight of the sulfur heterocyclics (Jewel!, 1980) . Nitrogen is present in all crude oils in compounds such as pyr i- dines, quinolines, benzoquinolines, acr idines, pyrroles, indoles, carbazoles, and benzcarbazoles (R.C. Clark and Brown, 1977; Posthuma,
19 TABLE 1-la Crude Oils Physical Characteristics and Chemical Properties of Several Characteristic or Component Crude Oil Prudhoe Baya South . . b Lou~s~ana Kuwait: API gravity (20°C) (°API)* Sulfur (wt %) N itrogen (wt 9 N ickel (ppm) Vanadium (ppm) Naphtha fractior~ (wt %) Par af f ins Naphthenes Aromatics Benzenes Toluene C~ aromatics C' aromas ics C1O aromas ics Cll aromatics Indans H igh-boil ing f ractione (wt 9e Satur ates e-paraffins Cl1 C12 C13 C14 C15 C16 C17 C18 C19 c2o C C22 C23 C24 C25 C26 C27 C28 C29 C30 C31 C32 plUS Isoparaffins 27.8 0.94 0.23 10 20 23.2 12.5 7.4 3.2 0.3d 0.6 0.5 0.06 __ 76.8 14.4f 5.89 0.12 0.25 0.42 0.50 0.44 0.50 0.S1 0.47 0.43 0.37 0.32 0.24 0.21 0.20 0.17 0.15 0.10 0.09 0.08 0.08 0.08 0.07 1-ring cycloparaffins 9.9 2-ring cycloparaffins 7.7 34.5 0.25 0.69 2.2 1.9 18.6 8.8 7.7 2.1 0.2 0.4 0.7 O .5 0.2 O .1 __ 81.4 56.3 5.2 0.06 0.24 0.41 0.56 0.54 0.58 0.59 0.40 0.38 0.28 0.20 0.15 0.16 0.13 0.12 O .09 0.06 O .05 O .05 0.04 0.04 o 14.0 12.4 9.4 31.4 2.44 0.14 7.7 28. 22.7 16.2 4.1 2.4 0.1 0.4 0.8 0.6 0.3 O .1 O .1 77.3 34.0 4.7 0.12 0.28 0.38 0.44 0.43 0.45 0.41 0.35 0.33 0.25 0.20 0.17 0.15 0.12 0.10 0.09 0.06 0.06 0.05 O.07 0.06 0.06 13.2 6.2 4.5
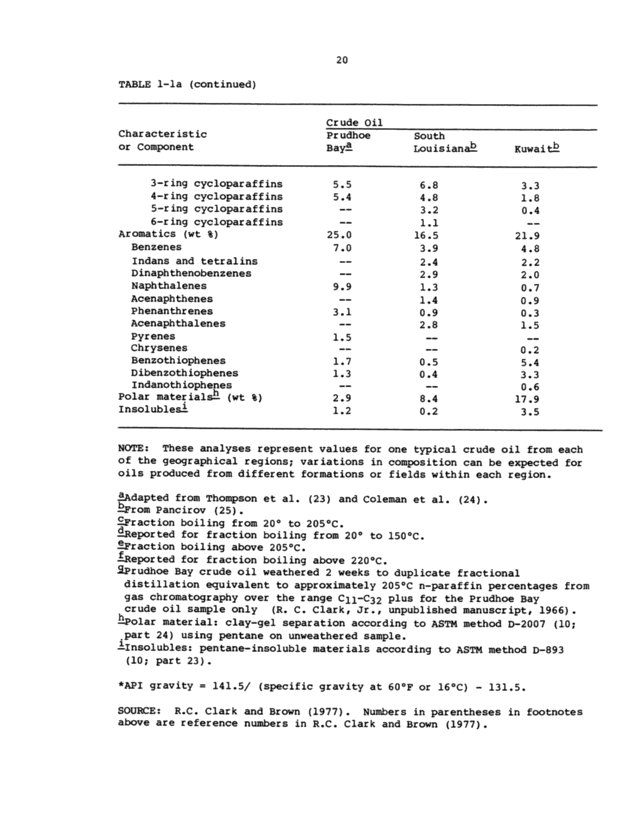
20 TABLE 1-la (continued) Characteristic or Component Crude Oil . Prudhoe South Baya Louisian ~Kuwaitb 3-ring cycloparaffins5.5 6.83.3 4-ring cycloparaffins5.4 4.81.8 5-ring cycloparaffins-- 3.20.4 6-ring cycloparaffins-- 1.1- Aromatics (wt %)25.O 16.521.9 Benzenes7.0 3.94.8 Indans and tetralins-- 2.42.2 Dinaphthenobenzenes-- 2.92.0 Naphthalenes9.9 1.30.7 Acenaphthenes-- 1.40.9 Phenanthrenes3.1 0.90.3 Acenaphthalenes-- 2.81.5 Pyrenes1.5 --- Chrysenes-- --0.2 Benzothiophenes1.7 0.55.4 Dibenzothiophenes1.3 0.43.3 Indanothiophenes-- --0.6 Polar materialsh (wt %)2.9 8.417.9 Insoluble=)1.2 0.23.5 - NOTE: These analyses represent values for one typical crude oil from each of the geographical regions; variations in composition can be expected for oils produced from different formations or fields within each region. Adapted from Thompson et al. (23) and Coleman et al. (24) ~ rom Pancirov (25~. Fraction boiling from 20° to 205°C. epor ted for fraction boiling from 20° to 150 °C. ~Fraction boiling above 205°C. epor ted for fraction boiling above 220°C. ~Prudhoe Bay crude oil weathered 2 weeks to duplicate fractional distillation equivalent to approximately 205°C e-paraffin percentages from gas chromatography over the range Cll-C32 plus for the Prudhoe Bay crude oil sample only (R. C. Clark, Jr., unpublished manuscript, 1966~. hPolar material: clay-gel separation according to ASTM method D-2007 (10; .part 24) using pentane on unweathered sample. ~Insolubles: pentane-insoluble materials according to ASTM method D-893 (10; part 23~. . *API gravity = 141.5/ (specific gravity at 60°F or 16°C) - 131.5. SOURCE: R.C. Clark and Brown (1977~. Numbers in parentheses in footnotes above are reference numbers in R.C. Clark and Brown (19771.
21 TABLE 1-lb Physical Character istics and Chemical Properties of Two Ref ined Products Character istic No. 2 Bunker C or Component Fuel Oila Fuel Oil API gravity (20°C) (°API) * 31.6 7.3 Sulfur (wt 9~) 0.32 1.46 Nitrogen (wt %) 0 . 024 0 . 94 Nickel (ppm) 0.5 89 Vanadium (ppm) 1.5 73 Saturates (wt %) 61.8 21.1 e-par af f ins 8 . 0 7 1 . 73 Clo ~ C11 1.26 0 C12 0.84 0 C13 0.96 0.07 C14 1.03 0.11 C15 1.13 0.12 C16 1.05 0.14 C17 0.65 0.15 C18 0.55 0.12 C19 0.33 0.14 C20 0.18 0.12 C21 0 .09 0 .11 C22 0 0.10 C23 0 o . os C24- 0 0.08 C25 0 0 .07 C26 0 0.05 C27 0 0.04 C28 0 0.05 C29 0 O .04 C30 0 O .04 C31 0 0.04 C32 plus O O .05 Isoparaffins 22.3 S.O 1-ring cycloparaffins 17.5 3.9 2-r ing cycloparaff ~ns 9 .4 3 . 4 3-r ing cyclopar af f ins 4 .5 2 . 9 4 -r ing cyclopar af f ins 0 2 . 7 5-r ing cyclopar af f ins 0 1.9 6-r ing cyclopar af f ins 0 0.4 Aromatics (wt % ~38 .2 34 . 2 Benzenes 10 .3 1. 9 Indans and tetralins 7.3 2.1 Dinaphthenobenzenes 4.6 2.0 Naphthalenes 0.2b Methyloaphthalenes 2. ~2.6 Dimethylnaphthalenes 3.2b
22 Table 1-lb (continued) Characteristic No. 2 Bunker C or Component Fuel Oila Fuel Oil Other naphthalenes 0.4 Acenaphthenes 3.8 3.1 Acenaphthalenes 5.4 7.0 Phenanthrenes 0 11.6 Pyrenes 0 1.7 Chrysenes 0 0 Benzothiophenes 0.9 1.5 Dibenzothiophenes 0 0.7 Polar materially (wt %) O 30.3 Insolubles (pentane)C (wt %) 0 14.4 NOTE: These analyses represent typical values for two different refined products; variations in composition can be expected for similar materials from different crude oil stocks and different refineries. From Pancirov (257. This is a high aromatic mater ial; a typical No. 2 fuel oil would have an aromatic content closer to 20-259~. From Vaughan (26~. tFrom Vaughan (26~. ESee footnotes h and ~ for Table 1-2. *API gravity = 141.5/(specific gravity at 60°F or 16°C) - 131.5. SOURCE: R.C. Clark and Brown (19771. Numbers in parentheses in footnotes above are reference numbers in R.C. Clark and Brown (1977~. 1977; Hunt, 1979; TisSot and Welte, 1978~. The porphyrins are nitrogen- containing compounds derived from chlorophyll and consisting of four 1 inked pyrrole r ings . Porphyr ins occur as organometall ic complexes of vanadium and nickel. Oxygen compounds in crude oils (0-2%) are found pr imar fly in distillation fractions above 400°C and consist of phenols, carboxylic acids, ketones, esters, lactones, and ethers. Petroleum contains a significant fraction (0-20%) of material of h igher molecular weight ~ 1 , 000-10 , 000 ), cons isting of both hydrocarbon and NSO compounds called asphaltenes. These compounds, consisting of 10-20 fused rings with aliphatic and naphthenic side chains, contribute signif icantly to the properties of petroleum in geochemical formations and in spill situations in relation to emulsification behavior.
23 TABLE 1-lc Examples of Individual Polynuclear Aromatic Hydrocarbon Concentrations in Petroleum (10-6 gig Petroleum) South Louisiana Kuwait Crude Crude No. 2 Fuel Oil Bunker C Pyrene 4.3 4.5 4123 Fluoranthene 6.2 2.9 37240 Benzanthracene 3.1 2.3 1.290 Chrysene 23 6.9 2.2196 Triphenylene 13 2.8 1.431 Benzo~aipyrene 1.2 2.8 0.644 Benzo~eipyrene 3.3 0.5 <0.110 SOURCE: Pancirov et al . ( 1980 ~ . Vanadium and nickel are the most abundant metallic constituents of crude petroleum, sometimes reaching thousands of parts per million. They are pr imar fly present in porphyr in complexes and other organic compounds (R.C. Clark and Brown, 1977; Yen, 1975~. The stable isotope ratio of 13C to 12C in whole crude oils, in oil fractions, and in the total and lipid fractions of sediments and organisms is being used to identify sources of carbon and to charac- terize or n fingerprint" various types of petroleum. Values in the literature are generally expressed in terms of 613C, where 613C (in o/oo) = ~ (l3C/l2C)sample/(l3C/l3C)standard - 131,000 and the standard is the Chicago PDB material (Craig, 1953~. The 613C compositions of natural crude oils range from about -18 to -35 o/oo (Silverman and Epstein, 1958~. Relative to the composition of a given whole crude, C, CH4 values are as negative as -40 o/oo and C values as positive as +2 o/oo for up to C15 hydrocarbons (Silverman, can be 1963). REFINED PRODUCTS Ref ined petroleum products introduced to the mar ine environment include gasoline, kerosene, jet fuels, fuel oils (No. 2, No. 4, No. S. No. 6) or Bunker fuel oils, and 1ubricatino oils. Figure 1-2 illustrates the t~Pes of common uroducts obtained from crude oil distillation and cracking. As refining processes and terminologies differ worldwide, For _ _ example, distillation, catalytic and thermal cracking, polymerization, and reforming yield products that are blended together to achieve desired chemical properties. They contain all of the hydrocarbon classes previously mentioned, but with narrower boiling ranges than comparisons of comDositions of refined products vary widely.
24 n-ALKANES CH4 CH3- CH3 METHAN E ETHANE I SOALKAN ES CH 3 - ( C H 2 ) - CH 3 (n = 1 - 58 ) C1 13 - C H - C H2 - C H3 cH3 CH3 CH - C - CH - CH - CH 3 1 2 1 3 ISOOCTANE ISOBUTANE CH3 CH3 CH3 CH3 CH3 CH3 CH3 - C - ( CH2 )n ~ C - ( CH2 )n ~ C ~ (CH2)n C CH3 ( n = 3 ) PR I STA N E (an isoprenoid hydrocarbon) CYCLOALKANES Carbon ond Hydrogen atoms Onot shown for example ~I I represented A CYCLOHEXANE DECALI N CH2 CH2 by \ / CH2 CYCLOPENTAN E W^' >~ HOPANE (general class of similar structures are friterpanes. ,~ CH OLESTA NE genera I class of simi lar structures are steranes.) FIGURE 1-la Chemical structure of petroleum hydrocarbons. Isooctane was formed in cracking process for gasoline production.
25 Benzene I ndane tOlOJ 2-Methyl Phenanthrene 3,4-Benzopyrene and 3,4-Benzo [a] pyrene Hi, Naphthalene 1-Methyl Naphthalene 2,5-Dimethyl Naphthalene F luorene Fl uoranth rene Phenanthrene Benzanth racene Pyrene FIGURE 1-lb Chemical structure of petroleum hydrocarbons. corresponding crude oils. In addition, cracking operations generate olefins (alkenes and cycloalkenes), which occur in concentrations as high as 30% in gasoline and about 1% in jet fuel. Olefins are not present in crude petroleum and are present only in minor amounts in other refined products. Alkylation processes yield many branched compounds such as isooctane (Figure 1-la). An excellent discussion of the chemical properties of refined products pertinent to fate and effects in the environment is found in R.C. Clark and Brown (19771. OIL SEEPS AND ANCIENT SEDIMENTS The composition of seep oil is similar in many respects to crude oil pumped from wells, but can be influenced by a variety of physical, chemical, and biological processes to be discussed in a later section.
26 SULFUR COMPOUNDS CH3- CH2 - SH Ethanethio I CH3- CH2 - S - S - C H - CH 3~4 Dithia hexane Thiacyclohexane Thiophene NITROGEN COMPOUNDS Dibenzothiophene H H Pyridine Quinoline Indoline Carbazole OXYGEN COMPOU N D S [ - ~OH o Fluorenone Phenol Dibenzofuran FIGURE 1-lc Nonhydrocarbon petroleum constituents: NSO compounds. Hydrocarbons and other compounds associated with ancient sediments range in composition from that of many crude oils to that of biogenic and early diagenetic compounds found in recent nonpolluted sediments. 8IOGENIC HYDROCARBONS Hydrocarbons are synthesized by most mar ine plants and animals, includ- ing microbiota (Han and Calvin, 1969; J.B. Davis, 1968), phytoplankton (Blumer et al., 1971; Clark and Blumer, 1967), zooplankton (Blumer et al. , 1969 ; Blumer and Thomas, 1965a,b; Avignan and Blumer , 1968), benthic algae (Youngblood et al., 1971; Youngblood and Blumer, ~ 973; Clark and Blumer, 1967), and fishes (Blumer et al., 1969; Blumer and Thomas, 1965b) . Organisms can both produce their own hydrocarbons and acquire them from food sources. Species of marine organisms synthesize limited numbers of hydro- carbon constituents over relatively narrow boiling ranges. For example, odd-numbered carbon chains predominate in mar ine biotic systems (C15-C2 1 normal alkanes in phytoplankton) , although the biogenic production of even-numbered carbon chains has been observed (R.C . Clark, unpubl ished manuscript, 19661. Pristane (C19) is a major component of calanoid copepods and, consequently, of some f ishes (Blumer et al . , 1963 ; Blumer ,
27 FRACTIONAL D ISTI LLATION DISTR I BUTION STRAIGHT-RUN MIDDLE DISTILLATES WIDE-CUT RESIDUAL GASO L I N E GAS Ol LS Ol LS O1 1 200 r, 400 By CC z o m 600 800 1000 GASOLINE FR ACTI ONS my_ KEROSINE HEATI NG | / DIESEL FU EL JET FU EL LIGHT LUBES I HEAVY ' LUBES FIGURE 1-2 Boil ing point range of fractions of crude petroleum. o -1 00 -200 o UJ CO Z a: a: -300 ~ By _ - o 400 m 500 600 NOTE: Bunker oil (not shown) is an oil of high viscosity used as a fuel oil. A given Bunker oil may be a mixture of two or more of the distillate cuts shown in the f igure or it may be a residual oil from a distillation run. SOUPBONE: Adapted from Bureau of Naval Personnel by R.C. Clark and Brown (1977 ~ . 1967~. Although normal and branched alkanes are biosynthesized, alkenes are the most abundant biosynthetic compounds in all trophic levels. Terrestrial plants (and sargassum) produce C21-C33 odd-chain n-alkanes, with the C21-C29 compounds dominating in marsh grasses and the C27-C33 alkanes associated with the waxy coatings of grasses and leaves. These are major hydrocarbon components of most "cleans coastal sediments (Wakeham and Farrington, 1980; Simoneit, 19781. Other compounds that have been detected in mar ine organisms include certain of the tr iterprenoid (hopane) hydrocarbons in mar ine bacter ia (Our isson et al ., 1979 ~ and naphthenes containing 1-3 r ings in land herbs and plants (Blumer, 1969 ~ . Although there have been reports of the synthes is of polycyclic aromatic hydrocarbons by algae and higher plants (e.g., Borneff et al., 1968a,b), these contentions are disputed (Gr immer and Duval , 1970 ; Hase and H ites , 1976 I, and at present the issue remains unresolved.
28 DIAGENETIC SOURCE S Biogenic precursor molecules (e.g., terpenes , sterols, carotenoid pigments) may be altered after deposition in sediment by microbially mediated and chemical processes to yield a variety of chemical compounds. Diagenetic hydrocarbon constituents include aliphatic hydrocarbons, cycloalkanes, sterenes, polyouclear aromatic hydrocarbons, and penta- cycl ic tr iterpanes . Among the most s ignif icant sets of diagenetic products are the PAH compounds, including some compounds that are also found in petroleum and other hydrocarbon sources (Wakeham et al., 19811. These diagenetic compounds may constitute important components of recent sediment hydrocarbon assemblages. Perylene and retene are among those compounds formed in reducing sediment (Hites et al., 1980; A izenshtat, 1973 ~ . COMBUSTION SOURCES AND COMPARISON TO PETROLEUM Particulate matter in urban air contains saturated (Heuser and Pattison, 1972) and aromatic hydrocarbons formed during the high-temperature incomplete combustion or pyrolysis of fossil fuels (coal, oil, and wood) (M.L. Lee et al ., 1977 ~ . Polynuclear aromatic hydrocarbons (PAM) formed dur ing combustion processes are transported seaward via direct deposition c; n the sea sur face or r ainout over land followed by storm- water runoff. PAH compounds are, therefore, ubiquitous chemical com- ponents of mar ine systems throughout the wor ld (Laf lamme and H ites, 1978; Pancirov and Brown, 1977; Youngblood and Blumer, 1975; Windsor and Hites, 1979; R.A. Brown and Weiss, 1978~. Aromatic hydrocarbons from combustion sources are characterized by a lesser degree of alkylation than aromatics from petroleum. The degree of alkylation within a homologous series of aromatics (e.g., phenanthrenes) in a given PAH assemblage is dependent on the temperature of formation of the PAH; high temperature processes (incomplete combus- tion or pyrolysis) favor less alkylation, while relatively low tempera- ture geological processes (petroleum maturation) favor higher degrees of alkylation. Figure 1-3 illustrates the principle that allows for the differentiation of combustion-related inputs from fresh and ,7eathered petroleum by considerations of alkyl homolog distr ibutions (AHD) (Blumer, 1976; M.L. Lee et al ., 1977; Hites and Bieman, 1975; Youngblood and Blumer, 1975) . Combustion sources contain relatively low quantities of 2-ringed aromatic families (e.g., naphthalenes). Therefore, in some cases the relative inputs of petroleum and com- bustion sources can be discerned from AHD plots of 2- to 5-ringed aromatics. Figure 1-4a illustrates a case where substantial quantities of petroleum- (2-3 rings) and combustion-related (3-5 rings) hydrocar- bons are present, while Figure 1-4b gives results for a sample comprised mainly of 3- to 5-ringed aromatics of pyrolytic or igin . Saturated hydrocarbons derived from combustion and weathered petroleum may have similar composition and thus may not be diagnostic for interpreting source mater ial . However, there has been less research on saturated hydrocarbons from combustion sources than on PAH.
29 EXAMPLE: TWO ALKYL CARBONS ON PHENANTHRENE ~ CH3 (2,3-DI METHYLPHENANTH RENE) ~ ~ ·_ ~ 0 24 60 2 4 60 2 4 60 2 4 6 NUMBER OF ALKYL CaRBONS ON aROMaT/C RINGS HIGH TEMPERaTURE MEDIUM TEMPERATURE LOW TEMPERATURE ( 2,000°C ) ( 800 °- 400°C ) (1 50° - 1 00°C ) FIGURE 1-3 Relative abundance of parent aromatic compound and alkyl substituents as influenced by temperature of formation. OTHER SOURCES Anthropogenic hydrocarbons may be introduced through a variety of sources (dredge spoil, sewage sludge, fly ash, industrial wastes) containing mixed inputs of hydrocarbon material (petroleum plus combus- tion materials. In addition, the direct introduction of coal may be significant in certain areas. Saturated and aromatic hydrocarbons in coal (Tripp et al., 1981) are very similar to those in petroleum, both being formed through low temperature processes, although careful evalua- tion of AHD plots may differentiate oil and coal (Hites et al., 1980~. DIAGNOSING SOURCES OF HYDROCARBONS The task of determining the sources of hydrocarbons in environmental samples is often difficult due to the multiplicity of sources that might be present and due to the postintroduction or postdeposition environ- mental modification of source materials. However, the proper composi- tional evaluation is often as important as determining the absolute values of hydrocarbon components.
30 25 0 ~ll ~CIay I r I ~<~oSilt [ INote Scale Change) l l l ~ ~ Sand N = Naphthalenes F = Fluorenes l l l | PA = Phenanthrenes + Anthracenes ;lgig 10- | | | | FPY = Fiuoranthenes+ Pyrenes BACH = Benzanthracenes+ Chrysenes I ~ I ~I | BP = Benzopyrenes + Benzofluoranthrene | ~ l f /1 1 1 0BT= Dibenzothiophenes m/e 0128142 1 563 70 1 84 1 98 166 180194 208 1 84 198212 226 240178192 206 220 234 202 216230244 228242 256 252 266 ~N - '. f - ' I OBT '~ PA ' ~ FPY ' iBACHJ ~ BP~ . ~-! ~ ~ ~ Number of Rings 2 2 3 1 1 1 3 4 4 5 ~1 1 1 B 240 220 200 180 160 140 ngig 1 20 100 80 60 40 n \ \ . 281i21561iO184 1661801942081841982;2i26240 1i8192206220234 202216230 228242256 262266 1- N - l I --. FJOBT- I PA ~ ~ FPY~ LBACH ~ L BpJ . ,,,,_,, PAH Alkyl Homologue Distributions showing Sewage Sludge-Mainly Petroleum Der~ved (A) and Dredge Spoil-Mainly Combustion Derived (B); within each Homologous Series FIGURE 1-4 PAH alkyl homolog distr ibutions for sewage sludge and dredge spoils. SOURCE: Boehm ( 1983 )
31 Many analytical techniques can aid in compositional evaluation as well as determination of absolute amounts of ind ividual compounds or groups of compounds. However, some of the methodology has been devel- oped in only one or two laboratories for certain specific purposes and has yet to be r igorously tested in a variety of situations in several laboratories. Thus, while working guidelines can be presented for differentiating between various sources of input, many of these guide- l~nes need further testing and may not provide sufficient sensitivity when applied to samples containing low concentrations of compounds (l-ppm level or less) from several different sources. Biogenic Sources Character istics of biogenic hydrocarbons are controlled by biosynthetic/ metabol ic pathways that usually result in mixtures of compounds of limited complexity due to structural specificity relations to specific biological functions. Thus: 1. Biogenic hydrocarbons of recent origin show a high predominance of odd-carbon n-alkanes. Possible exceptions are the n-C22 to n-C30 alkanes present at low concentrations in phytoplankton and bacteria. 2. Terrigenous plant detrital inputs are characterized by odd- numbered n-alkanes in the C23-C33 region; marine biogenic inputs are often marked by the presence of odd-chain n-alkanes n-C15, n-C17, and n-Clg . 3. Biogenic inputs are often noted by the predominance of a single isoprenoid, usually pr istane . The ratio of pr istane to phytane is usually much greater than one in Biogenic sources. Phytane is rarely found as a biolipid except in some bacteria. 4. One or several aliphatic olefins or cyclic alkenes may occur in narrow ranges of molecular weight. 5. Biogenic compounds often include polyolefins such as heneicosa- hexaene in algae and squalene in higher animals . Other polyolef ins ar e abundant in many organisms and compr ise most of the mater ial in "aromatic/saturate" silica gel adsorption column eluates. 6. Biogenic aromatics do not occur often or in high concentrat~ons. 7 . Stable carbon isotope ratios of biogenic mater ial are often much different than isotopically heavier petroleum (Degens, 1969~. Characteristics of Undegraded Petroleum 1. Petroleum contains a much more complex mixture of hydrocarbons over wider boiling ranges than biogenic inputs. 2. Crude oil contains no olefins. 3. The ratio of odd to even carbon number n-alkanes in various molecular weight ranges expressed as either the odd-even preference {OEP; Scalan and Smith , 1970 ~ or carbon preference index (CPI ; Farrington and Tripp, 1977) is near unity. The alkane n-C16 is rarely found in biolipids (Tbompson and Eglinton, 1978~.
32 4. Petroleum contains several homologous series of compounds (e.g., normal alkanes; branched alkanes; cycloalkanes; isoprenoid alkanes, including branched cyclohexanes, steranes, and triterpanes). 5. Petroleum contains homologous series of alkylated aromatics (e.g., mono-, di-, trim, and tetra-methyl benzenes; naphthalenes; fluorenes; dibenzothiophenes; phenanthrenes). 6. Petroleum contains numerous naphthenic and naphthenoaromatic compounds. 7. Petroleum contains numerous heterocyclic compounds containing S. N. and 0. 8. Petroleum contains trace metals, with Ni and V often present in ng/g quant it ies . 9. Hydrocarbons of a petroleum or igin should have little 14C activity. 10. Stable carbon isotope ratios are isotopicall-y heavier than b iogen ic inputs . - All character istics are attr ibutable to petroleum and ref ined products, although the composition of distillate cuts is narrower in boiling range than the corresponding crude oil. Light distillate cuts may contain olef inic mater ial and little, if any, trace metal content unless added. However, one caveat pertains to carbon-number ratios. Smooth distributions of alkanes (CPI or 0EP = 1) within the crude oil nonvolatile molecular weight range have been reported for marine bacteria (Han and Calvin, 1969) and have been detected in marine fish (Whittle et al., 1977a,b; Boehm, 1980~. ThUS, paraffinic tar and biogenic alkanes may be very similar in the C20-C30 range. Furthermore, smooth n-alkane distributions have been noted in urban air (Heuser and Pattison, 1972) and in laboratory dust (Gelpi et al., 1970) samples. Thus, n-alkane distributions alone, in environmental samples and especially in marine fish, cannot be attributable to oil pollution without corroboration by other petroleum compositional features. Characteristics of Petroleum Altered by Physical, Chemical, and Biological Processes Most often, except for recent spill studies, environmental samples contain altered, rather than "fresh, n petroleum. Thus, some diagnostic features associated with petroleum may be reduced in importance, and other or new diagnostic parameters become more important. The compos ition can be altered on time scales varying from days to years past the point where of] can be easily attributed to a particular source. Certain chemical marker compounds survive longer than others, and the time course and degree of change in composition vary with each spill or source of input, sample type, and environmental conditions. An example of a time series for fresh petroleum and samples subjected to alteration by natural processes is shown in Figure 1-5. The following changes in composition occur:
33 1. Loss of low boiling (<C20) aromatic and saturated hydrocarbons through evaporation. 2. Loss of low boiling (<C15) aromatic hydrocarbons through dissolution. 3. An increased relative importance of unresolved naphthenic and naphthenoaromatic compounds (i.e., the unresolved complex mixture or UCM). 4. An increased importance of highly branched aliphatic hydro- carbons (i.e., isoprenoids) relative to straight chain and s ingly methyl-branched molecules due to selective depletion of n-alkanes by biodegradation. 5. An increased importance of alkylated (dimethyl to tetramethyl) phenanthrene and dibenzothiophene compounds relative to other aromatics through combined weathering processes. 6. An increased importance of polycyclic aliphatic (e.g., penta- cyclic triterpanes) compounds relative to all saturated components. Weathered petroleum and the marine environment adjacent to weathering petroleum residues are affected by the oxidation products of both photochemical and microbial oxidation. Photooxidation, photodecomposition, and polymerization of spilled oil components (Overtop et al., 1979, 1980; Larson et al., 1977; Burwood and Speers, 1974; Freegarde et al., 1971; Parker et al., 1971) may result from sunlight-induced reactions. A variety of reaction products may result from free radical reactions, including aliphatic and aromatic ketones, aldehydes, carboxylic acids, fatty acids, esters, epoxides, sulfoxides, sulfones, phenols, anhydrides, quinones, and aliphatic and aromatic alcohols. Tn addition, polymer ization reactions occurr ing during photolysis affect the chemical and physical properties of the oil, perhaps increasing its emulsif ication rate. Burwood and Speers (1974) observed an increase in UCM content of samples as photo- oxidation proceeded, the mater ial presumably resulting from the formation of a var iety of high-molecular-weight sulfoxides. As photolysis produces a var iety of chemical reaction products, it also decomposes petroleum, thus affecting the residual hydrocarbon assemblage. The relative extent of photooxidation of spilled oil and the production of reaction products versus other weathering-induced changes in the oil's chemistry are largely unknown, although photolysis reactions are believed to play major roles in altering the oil's chemical composition after evaporation ceases to be the major process acting on oil. The relative "competition" of photolysis and microbial oxidation/ degradation remains poorly understood. Microbial oxidation can result in the formation of the same class of reaction products as does photolysis (Overtop et al., 19801. However, microbial processes are more selective due to enzymatic preferences for specific isomers, while photooxidative processes tend to be less selective. AS a demonstration of this, Overton et al. (1980) compared the composition of samples of benzoic acid and its C1-C3 alkylbenzoic acid homologs from photo- chemical and microbial oxidation experiments and determined that the C3+ homologs differed significantly between the experiments, the photochemical reaction products being more structurally diverse.
34 A R EF ERENCE MOUSSE 1 C ~ CD ~ . - Cat ~ a, O - ~ ~ ~ - - - .L - - -A,- ~- ~ Cad ~ ~ Cad UCM ~ C "ONE YEAR EXPOSURE l c |> UCM NAAI ~1 _~ FIGURE 1-5 Glass capillary gas chromatograms of time series for fresh petroleum and petroleum subjected to alteration by natural processes in sediments. To date, the various transformation products have not been used as markers for particular sources of inputs, although useful indicator compounds may emerge with further research. Characteristics that have proven useful are the following:
35 D STAG E 3 WEATH E R I NG (Saturated Hydrocarbons) In PCA=Polycycl ic Al iphatics UCM PCA it_ 1 _` E STAGE 4 WEATHERING (Saturated Hydrocarbons) O , At' 1.~ In _ ~ ~' UCM PCA l i.. .) my_ Aim_ _ .. . /. . . F BACKGROUND (Saturated Hydrocarbons) i t~ ~ ~1~ L4, . . ~ . - C~ ~ g Cal ~1~i FIGURE 1-5 (continued). 1. An unresolved complex mixture is characteristic of weathered oils (see Chapter 3, Gas Chromatography section). 2. The normal alkane-to-isoprenoid ratio, which in fresh oils is much greater than 1, decreases as biodegradation proceeds (Boehm et al., 1981; Atlas et al., 1981~. That is, pristane, phytane, and farnesane become dominant saturated hydrocarbon components of weathered oils until they, too, are degraded.
36 3. Extended series of isoprenoids (C20-C40) become useful indicators of petroleum in weathered samples (Albaiges, 1980~. 4. C27+ pentacyclic triterpanes (e.g., hopanes; Figure 1-6), being relatively resistant to degradation, become prominent marker compounds (Dastillung and Albrecht, 1976; Albaiges and Albrecht, 1979; Boehm et al., 1981) as the presence of paired peaks (22R and S diastereomers) of the Cal, C32, and C33 17H,21H-hopanes is unique to petroleum (see Figure 1-61. 5. Alkylated phenanthrenes and alkylated dibenzothiophenes sometimes are the prominent aromatic components of weathered petroleum (Teal et al., 1978; Ber thou et al., 1981; Boehm et al., 1981; Overton et al., 1981~. 6. The relative amount of polar (N. S. O) material increases as degradation proceeds due to oxidation reactions (J.R. Payne et al., 1980a,b). 7. Stable isotope ratios of carbon, hydrogen, and sulfur do not vary greatly with weathering, and thus may be useful to identify weathered oils (Sweeney et al., 1980; Sweeney and Kaplan, 1978~. Character istics of Combustion-Related Hydrocarbons Little is known about the saturated hydrocarbon composition of the combustion products of fossil fuels. Most compositional information is based on polynuclear aromatic hydrocarbons. 1. PAH compounds generally occur in the 2- to 6-ring range. 2. Fluoranthrene and pyrene are often the most abundant PAH in pyrolysis-related samples together with phenanthrene, benzanthracene, chrysene, and the benzopyrenes. 3. Unsubstituted (nonalkylated parent) compounds are much more abundant than alkylated members of any homologous series. ThiS important difference from weathered petroleum is often most striking for the phenanthrene series. Alkylated phenanthrene members are often the most abundant aromatic constituents of weathered oils. 4. Dibenzothiophene is relatively far less abundant than in oils.
37 REFERENCE MOUSSE 91 .0 T] 91 .a T] In" ~ Hopane Norhopane _ Low Homohopanes Trisnorhopanes ~ 1 |4 61 yes ~s"'.~"'~s"'L'i'lllli'l~ll~l6'llll~l `~'6i'i~`'lLl~`r BAY OF MORLAIX SEDIMENT November t978 ~__ _ ~=~__ 1)~ t.~1 111. 11~4 I.. t _ 101111 2. 2 ~He ~ - ca CA en '^ ~ - ~__ FIGURE 1-6 Gas chromatographic mass spectrometry selected ion searches for pentacyclic tr iterpanes (hopanes) in Amoco Cadiz reference oil and November 1978 weathered oil in sediments.
38 REFERENCES Aizenshtat, A. 1973 . Perylene and its geochemical signif icance. Geochim. Cosmochim. Acta 37: 559-567 . Albaiges, J . 1980 . Fingerpr inking petroleum pollutants in the Mediterranean Sea, pp. 69-81. In J. Albarges, ed. Analytical Techniques in Env ironmental Chemistry. Pergamon, New York. Albaiges, J., and P. Albrecht. 1979. Fingerprinting marine pollutant hydrocarbons by computer ized gas chromatography-mass spectrometry. Intern. J. Environ. Anal. Chem. 6: ~ 71-190 . Atlas, R.M., P.D. Boehm, and J.A. Calder, 1981. Chemical and biological weathering of oil from the Amoco Cadiz oil spillage, within the 1 ittoral zone. Estuarine Coastal Mar. Sci. 12:589-608. Avignan, J., and M. Blumer. 1968. On the origin of pristane in marine organisms. J. Lipid Res. 9:350-352. Ber thou, F., Y. Gourmelun, Y. Dreano, and M.P. Friocourt. 1981. Application of gas chromatography on gas capillary columns to the analysis of hydrocarbon pollutants from the Amoco Cadiz oil spill. J. Chromatogr. 203:279-292. Blumer, M. 1967. Hydrocarbons in digestive tract and liver of a basking shark. Science 156:390-391. Blumer, M. 1969. Oil pollution of the ocean, pp. 5-13. In D.P. Hoult ed. Plenum, New York. Blumer, M. 1976. Polycyclic aromatic compounds in nature. Sci. Am. 234:34-45. Blumer, M., and D.W. Thomas. 1965a. Phytodienes in zooplankton. Science 147:1148-1149. Blumer, M., and D.W. Thomas. 1965b. Zamene, isomeric Cl9 monoolefins from marine zooplankton, fishes, and mammals. Science 148:370-371. Blumer, M., M.M. Mullin, and D.W. Thomas. 1963. Pristane in zooplankton. Science 140:974. Blumer, M., J. Gordon, J.C. Robertson, and J. Sass. 1969. Phytol-derived Clg and di- and tri-olefinic hydrocarbons in mar ine zooplankton and f ishes . Biochemistry 8: 4067-4074 . Blumer, M., R.R.L. Guillard, and T. Chase. 1971. Hydrocarbons of mar ine phytoplankton. Mar . Biol . 8 :183-189 . Boehm, P.D. 1980. Gulf end Atlantic survey--Cape Hatteras to Gulf of aine survey for selected organic pollutants in f inf ish and benthic animals. Final Report. NOAA Contract NA-80-FA-C-00046. NOAA/NMFS Northeast Fisheries Center, Sandy Hook, N.J. Boehm, P.D. 1983 . Coupl ing of organic pollutants between the estuary and continental shelf and the sediments and water column in the New York Bight region. Can. J. FiSh. Aquat. Sci. 40(Suppl. 21:262-276, Fig. 6. Boehm, P.D., D.L. Fiest, and A. ElSkUS. 1981. Comparat~ve weathering patterns of hydrocarbons from the Amoco Cadiz oil spill observed at a variety of coastal environments, pp. 159-173. In Proceedings, Amoco Cadiz: Fate and Effects of the Oil Spill, November 19-22, 1979. Centre Nationale pour 1'Exploitation des Oceans, COB, Brest, Fr ance .
39 Borneff, J., F. Selenka, H. Kunte, and A. Maximos. 1968a. Die synthese van 3,4-benzpyren and anderen plyzyklischen, aromatischem kohnlenwasserstoffen in pflanzen. Arch. Hyg. Bakt. 152:279-282. Borneff, J., F. Selenka, H. Kunte, and A. Maximos. 1968b. Experimental studies on the formaton of polycyclic aromatic hydrocarbons in plants. Environ. Res. 2:22-29. Brown, R.A., and F.T. Weiss. 1978. Fate and effects of polynuclear aromatic hydrocarbons in the aquatic environment. Publication 4297. American Petroleum Institute, Environmental Affairs Department, Washington, D.C. Bur wood, R., and G.C. Speers. 1974. Photooxidation as a factor in the environmental dispersal of crude oil. Estuarine Coastal Mar. Sci. 2:117-135. Clark, R.C., Jr., and M. Blumer. 1967. Distribution of e-paraffins in marine organisms and sediment. Limnol. Oceanogr. 12:79-87. Clark, R.C., Jr., and D.W. Brown. 1977. Petroleum: properties and analyses in biotic and abiotic systems, pp. 1-89. In D.C. Malins, ed. Effects of Petroleum on Arctic and Subarctic Marine Environments and Organisms. Vol. 1. Nature and Fate of Petroleum. Academic Press, New York. Craig, H. 1953. The geochemistry of stable carbon isotopes. Geochim. Cosmochim. Acta 3:53-92. Dastillung, M., and P. Albrecht. 1976. Molecular test for oil pollution in surface sediments. Mar . Pollut. Bull . 7 :13-15. Davis, J .B. 1968 . Par aff inic hydrocarbons in the sulfate-reducing bacterium Desulfovibrio desulfuricans. Chem. Geol. 3:1SS-160. Degens, E.T. 1969. Biogeochemistry of stable carbon isotopes, pp. 304-329 . Tn G . Egl inton and M.T.J . Murphy, eds . Organic Geochemistry. Springer-Verlag, New York. Far r ington, J .W., and B.W. Tr ipp. 1977. Hydrocarbons in western North Atlantic surface sediments. Geochim. Cosmochim. Acta 41:1627-1641. Freegarde, M., C.G. Hatchard, and CoA~ Parker. 1971. Oil spilt at sea its identification, determinatzon and ultimate fate. Lab. Practice 20~1~:35-40. Gelpi, E., D.W. Nooner, and J. Oro. 1970. The ubiquity of hydrocarbons in nature: aliphatic hydrocarbons in dust samples. Geochim. Cosmochim. Acta 34:421-425. Grimmer, G., and D. Duval. 1970. Investigations of biosynthetic formation of polycyclic hydrocarbons in higher plants. Naturforsch. 25b:1171-1175. Han, J., and M. Calvin. 1969. Hydrocarbon distribution of algae and bacteria and microbiological activity in sediments, pp. 436-443. In Proceedings, National Academy of Sciences. Vol. 64. Washington, D.C. Hase, A., and R.A. Hites . 1976. On the or igin of polycyclic aromatic hydrocarbons in recent sediments: biosynthesis by anaerobic bacter ia. Geochim. Cosmochim. Acta 40: 549-555 . Hauser, T.R., and J.N. Pattison. 1972. Analysis of aliphatic fraction of air particulate matter . Environ. Sci . Technol . 6: 549-555 . Hites, R.A., and W.G. Bieman. 1975. Identification of specific organic compounds in a highly anoxic sediment by gas chromatographic-mass spectrometry and high resolution mass spectrometry, pp. 188-201. In
40 R.P. Gibbs, ed. Analytical Methods in Oceanography. Advances in Chemistry Series 147. American Chemical Society, Washington, D.C. Hites, R.A., R.E. Laflamme, and J.G. Windsor, Jr. 1980. Polycyclic aromatic hydrocarbons in marine/aquatic sediments, pp. 289-311 . In L. Petrakis and F e T . Wiess , eds . Petroleum in the Mar ine Environment. Advances in Chemistry Ser. ies 185 . Amer ican Chemical Society, Washington, D.C. Hunt, J.M. 1979. Petroleum Geochemistry and Geology. W.H. Freeman and Co ., San Fr anc isco . 6 1 7 pp . Jewell, D.M. 1980. The role of nonhydrocarbons in the analysis of virgin and biodegraded petroleum, pp. 219-235. In L. Petrakis and F.T. Weiss, eds . Petroleum in the alar ine Environment. Advances in Chemistry Series 185. American Chemical Society, Washington, D.C. Laflamme, R.E., and R.A. Hites. 1978. The global distribution of polycycl ic aromas i c hydrocarbons in recent sediments . Geochim. Cosmochim. Acta 42:289-304. Larson, R.A., L.L. Hunt, and D.W. Blankenship. 1977. Formation of toxic products from a No. 2 fuel oil by photooxidation. Environ. Sci. Technol. 11~5~:492-496. Lee, M.L., G.P. Prado, J.B. Howard, and R.A. Hites. 1917. Source identif ication of urban airborne polycyclic aromatic hydrocarbons by gas chromatographics mass spectrometry and high resolution mass spectrometry. Biomed. Mass. Spec. 4:182-186. Our isson , G ., P . Albrecht, and M. Rohmer. 1979. The hopanoids : paleo-chemistry and biochemistry of a group of natural products. Pure Appl. Chem. 51:709-729. Overton, E.B., J.R. Patel, and J.L. Laseter . 1979. Chemical character ization of mousse and selected environmental samples from the Amoco Cadiz oil spill, pp. 169-214. In Proceedings, 1979 Oil Spill Conference (Prevention, Behavior, Control, Cleanup) . Amer ican Petroleum Institute, Wash ington, D.C . Over ton, E.B., J.L. Laseter, W. Mascarella, C. Raschke, I . Nuiry, and J.W. Far r ington. 1980 . Photochemical oxidation of Ixtoc I oil, pp. 41-386. In Proceedings of the Conference on the Preliminary Scientif ic Results from the Researcher/Pierce Cruise to the Ixtoc I Blowout. NOAA, Office of ~arine Pollution Assessment, Rockville, ~Id. Over ton , E.B., J . McFall , S.W. Mascarella, C.F. Steele, S.A. Antoine , I.R. Politzer, and J.L. Laseter. 1981. Petroleum residue sources identification after a f ire and oil spill , pp. 541-546 . In Proceedings, 1981 Oil Spill Conference (Prevention, Behavior, Control, Cleanupl. American Petroleum Institute, Washington, D.C. Pancirov, R.J., and R.A. Brown. 1977. Polynuclear aromatic hydrocarbons in mar ine tissues . Environ. Sci. Tech . 11: 989-992 . Pancirov, R.J., T.D. Seal, and R.A. Brown. 1980. Methods of analysis for polynuclear aromatic hydrocarbons in environmental samples, pp. 123-142 . In L . Petrakis and F.T. Weiss, eds. Petroleum in the Mar ine Environment . Advances in Chemistry Ser. ies 1 85 . Amer ican Chemical Society, Washington, D.C. Parker, C.A., ~q. Freegarde, and C.G. Hatchard. 1971. The effect of some chemical and biological factors on the degradation of crude oil at sea, pp. 237-244. ~n P. Hepple, ed. Water Pollution by Oil. Institute of Petroleum, London.
Payne, J.R., G. Smith, P.J. Mankiewicz, R.F. Shokes, N.W. Flynn, V. Moreno, and J . Altamirano. 1980a. Hor izontal and vertical transport of dissolved hydrocarbons from the Ixtoc I blowout, pp. 239-266. In Proceedings of Symposium on the Preliminary Results from the Researcher/Pierce Cruise to the Ixtoc I Blowout. NOAA, Office of Marine Pollution Assessment, Rockville, Md. Payne, J.R., N.W. Flynn, P.J. Mankiewicz, and G.S. Smith. 1980b. Surface evaporation/dissolution partitioning of lower-molecular-weight aromatic hydrocarbons in a down-plume transect from the Ixtoc I wellhead. In Proceedings of the Conference on the Preliminary Scientific Results from the Researcher/Pierce Cruise to the Txtoc I Blowout. NOAA, Office of Marine Pollution Assessment, Rockville, Md. Posthuma, J. 1977. The composition of petroleum. Rapp. P.-v. Reun. Cons. Int. Explor. Mer 171:7-16. Scalan, R.S., and J.E. Smith. 1970. An improved measure of the odd-even predominance in the normal alkanes of sediment extracts and petroleum. Geochim. Cosmochim. Acta 34: 611-620 . Silverman, S.R. 1963 . Investigations of petroleum or igin and evolution mechanisms by carbon isotope studies, pp. 92-102 . In H. Craig, S.L. Miller, and G.J. Wasserburg, eds. Isotopic and Cosmic Chemistry. Nor th-Holland, Amsterdam. S ilverman, S.R., and S . Epstein. 1958 . Carbon isotopic compositions of petroleum and other sedimentary organic mater ials. Bull . Am. Assoc . Petrol. Geol. 42: 998-1012 . Simoneit, B.R.T. 1978. Organic chemistry of marine sediments, pp. 233-311 . In R. Chester and J.P . Riley, eds . Chemical Oceanography . Vol. 7. Academic Press, New York. Sweeney, R.E., and I . R. Kaplan . 1978 . Character ization of oils and seeps by stable isotope ratio. Proc. Energy/Environ. Calif. SPIB: 281-293 . Sweeney, R.E., R.I. Haddad, and I.R. Kaplan. 1980. Tracing the dispersal of the Ixtoc I oil using C, H. S and N stable isotope ratios, pp. 89-118. In Proceedings, Symposium on the Preliminary Results from the Researcher/Pierce Cruise to the Ixtoc I Blowout. NOAA, Office of Mar ine Pollution Assessment, Rockville, Md. Teal, J.M., K. Burns, and J . Farrington. 1978. Analyses of aromatic hydrocarbons in intertidal sediments resulting from two spills of No. 2 fuel oil in Buzzards Bay, Mass. J. FiSh. Res. Board Can. 35:510-520. Thompson, S., and G. Eglinton. 1978. Composition and sources of pollutant hydrocarbons in the Sever n Estuary. Mar. Pollut. BU11. 9:133-136. Tissot, B.P., and D.H. Welte. 1978. Petroleum Formation and Occurrence. Spr inger-Verlag, New York. 538 pp. Tripp, B.W., J .W. Far r ington, and J.M. Teal. 1981. Unburned coal as a source of hydrocarbons in sur face sediments . ~lar . Pollut . Bull . 12: 122-126. Wakeham, S.G., and J.W. Farrington. 1980. Hydrocarbons in contemporary aquatic sediments, pp. 3-32. In R.A. Baker, ed. Contaminants and Sediments. Vol. 1. Ann Arbor Science Publishers, Ann Arbor, Mich.
42 Wakeham, S.G., C. Schaffner, and W. Giger. 1981. Diagenic polycyclic aromatic hydrocarbons in recent sediments: structural information obtained by high per for mance 1 iquid chromatography, pp. 353-363. In J. Maxwell and A. Douglas, eds . Advances in Organic Geochemistry. Macmillan, New York. Whittle, K.J., J. Murray, P.R. Mackie, R. Hardy, and J. Farmer. 1977a. Fate of hydrocarbons in f ish. Rapp. P.-v. Reun. Cons . Int. Explor . Mer 171: 139-142. Whittle, K.J., P.R. Mackie, R. Hardy, A.D. McIntyre, and R.A.A. Blackman. 1977b. The alkanes of mar ine organisms from the united Kingdom and surrounding waters. Rapp. P.-v. Reun. Cons. Int. Explor . Mer 171: 72-78 . Windsor, J.G., Jr., and R.A. Hites. 1979. Polycyclic aromatic hydrocarbons in Gulf of Maine sediments and Nova Scotia soils. Geochim. Cosmochim. Acta 43: 27-33 . Yen, T.F . 1975 . The Role of Trace Metals in Petroleum. Ann Arbor Scientific Publishers, Ann Arbor, Mich. Youngblood , W.W., and M. Blumer. 1973. Alkanes and alkenes in marine benthic algae. Mar. Biol. 21:163-172. Youngblood, W.W., and M. Blumer. 1975. Polycyclic aromatic hydrocarbons in the environment: homologous series in soils and recent marine sediments . Geochim. Cosmochim. Acta 39 :1303-1314 . Youngblood, W.W., M. Blumer, R.L. Guillard, and F. Fiore. 1971. Saturated and unsaturated hydrocarbons in marine benthic algae. Mar. Biol. 8:190-201.