15
Tyrosine and Stress: Human and Animal Studies
Harris R.Lieberman1
INTRODUCTION
In a variety of combat scenarios, enhancement or prevention of stress-induced decrements in mental performance would be desirable. The use of nutrients to enhance performance may be one strategy for increasing the combat effectiveness of soldiers. Providing even a small edge in effectiveness by using a comparatively benign nutritional intervention could, in theory, significantly improve performance on the battlefield, reduce casualties, and conserve critical material resources. In the classic combat scenario, fear, loss of sleep, and exposure to harsh environmental conditions can be expected to produce substantial decrements in performance. In other situations, such as sentry duty or vehicle operation, boredom and sleepiness can reduce vigilance and cause critical errors. A nutrient that is useful in all circumstances would be of the greatest value as a performance-enhancing ration component. However, it is more likely that a substance that is beneficial in one scenario will have no effect in another scenario or will even have adverse effects if used inappropriately. Therefore, it is likely that the choice of a food constituent
or constituents will depend on both the environment and the operational demands of the mission in which it will be used.
One nutrient that may prevent performance decrements caused by exposure to highly stressful environmental or operational conditions is the amino acid tyrosine. This substance is the precursor of several key brain neurotransmitters and may protect against the severe mental fatigue associated with exposure to life-threatening stressors that can occur in combat and certain other critical military operations. Tyrosine does not appear to have the stimulant-like activity of caffeine, so it is not likely to be useful in situations in which performance is degraded by boredom or sleepiness.
BACKGROUND
Tyrosine, a large neutral amino acid (LNAA) normally present in protein-containing foods, is the precursor of the catecholamine (CA) neurotransmitters dopamine (DA), norepinephrine (NE), and epinephrine. When it is systemically administered in pharmacologic quantities, it can, under highly stressful conditions, increase brain CA concentration and turnover (Gibson and Wurtman, 1978; Wurtman et al., 1974, 1981). There are no known adverse effects from tyrosine administration. Also, because tyrosine is normally present in substantial quantities in ordinary foods and is rapidly metabolized, its administration is unlikely to have long-term toxicity or unwanted side effects. In fact, since tyrosine exerts its effects only when a localized deficiency state exists, its effects appear to be system specific and present only when needed, such as when local CA stores are expended. Therefore, tyrosine’s actions are likely to be more specific than those of most drugs, but perhaps less potent.
Precursor Coupling
The availability of tyrosine is rate-limiting for the synthesis of its neurotransmitter products only when a higher than normal level of transmitter release by catecholaminergic neurons is occurring. When CA-containing neurons are firing frequently and therefore releasing more transmitter (DA or NE), they may require more of the precursor (tyrosine) that is the substrate for transmitter synthesis. Frequent neuronal firing may enhance the kinetic properties of tyrosine hydroxylase, causing this rate-limiting, CA-synthesizing enzyme to be more susceptible to control by this amino acid or may deplete the tyrosine pools within nerve terminals (Lovenberg et al., 1975; Weiner et al., 1978).
The ability of tyrosine supplementation to enhance the synthesis of CAs in and their release from rapidly firing neurons but not from relatively quiescent cells has been demonstrated by using a variety of experimental manipulations (for a review, see Milner and Wurtman [1986]). Tyrosine administration increases brain levels of the norepinephrine metabolite methoxyhydroxyphenylethylgylcol sulfate (MHPG-SO4) in cold-stressed rats (Gibson and Wurtman, 1978) and in the brains of spontaneously hypertensive rats (Sved et al., 1979) but not in those of control, normotensive animals. Increases in MHPG-SO4 levels after tyrosine treatment have also been observed in rats stressed by tailshock (Lehnert et al., 1984a,b; Reinstein et al., 1984). Dopaminergic metabolism also appears to be accelerated by tyrosine administration (Brady et al., 1980).
Catecholamines and Stress
The observation that the function of catecholaminergic neurons can be precursor dependent is the basis for the hypothesis that tyrosine mitigates the adverse effects of acute stress, because such neurons regulate, in part, the behavioral, cardiovascular, and neuroendocrine consequences of stress. It is well established that the noradrenergic neurons of the locus ceruleus and hypothalamus participate in the regulation of a variety of functions that are altered during acute stress. Alertness, attention, activity, anxiety levels, blood pressure, and the secretion of certain hormones are all controlled, at least in part, by these neurons (Antelman and Caggiula, 1977; Gray, 1982; Murphy and Redmond, 1975; Stone, 1975). Certain dopaminergic neurons also appear to participate in the regulation of some of these functions.
There is a great deal of evidence demonstrating that CA-containing neurons play a key role in the regulation of arousal level and anxiety. Data from animal neurochemical and behavioral studies, from animal and human psychopharmacology, and also from clinical observations all contribute to the complex literature that exists on this topic. Because of the diversity of this literature and the extraordinary difficulties associated with any attempt to establish the neurochemical substrate of complex behavioral-cognitive states, such as anxiety, helplessness, and depression, there are many controversial issues in this area. However, in attempts to integrate the diverse data, investigators are in agreement on many key issues (Gray, 1982; Stone, 1975). For example, it is agreed that when animals are subjected to acute stress, especially when the stress cannot be avoided and is highly aversive, an array of behavioral, cardiovascular, neuroendocrine, and neurochemical changes are present (for a review, see Stone [1975]). It appears that the neurochemical substrate for this syndrome is, at least in part, the depletion of readily available
central NE stores. Stressful conditions that can deplete central NE stores include cold, heat, restraint, exercise, and footshock (Stone, 1975). Acutely stressed animals become less responsive to their environment, explore less, are less aggressive and more submissive, cannot learn as readily, and generally seem debilitated (Maier and Seligman, 1976; Rapaport and Maier, 1978. The term learned helplessness has been applied to certain aspects of this syndrome because animals fail to respond appropriately (escape or avoid) to aversive stimuli when they are given the opportunity to do so (Maier and Seligman, 1976; Maier and Jackson, 1979; Minor et al., 1984; Weiss et al., 1976). This phenomenon has been used to model human clinical depression and posttraumatic stress disorder (Gray, 1982; van der Kolk et al., 1985).
The effects of acute stress are not limited to learning and similar behaviors, but rather, impairments in these behaviors are representative of a broad range of behavioral deficits. The underlying factor that characterizes most of the changes induced by acute stress appears to be inhibition of both spontaneous and stimulus-dependent behaviors. Acutely stressed animals, for the most part, regardless of the specific nature of the stress, generally appear to be unable to function. Not only are complex stimulus-dependent behaviors like aggression, learning, and exploration depressed, but even eating and sleep are disturbed. With the appearance of such behavioral deficits (Gray, 1982; Stone, 1975), brain NE turnover increases substantially and NE stores are depleted. (When the stress is chronic, more complex changes in NE function take place.) It is widely believed that catecholaminergic neurons participate directly in the onset of certain aspects of this acute stress-induced syndrome, which has, in fact, been termed noradrenergic helplessness (for a comprehensive review, see Gray [1982]).
The critical role that noradrenergic systems play in this behavioral syndrome is supported by several lines of evidence, including the effects that various pharmacologic interventions known to modify CA transmission have on behavior. Catecholaminergic agonists, for example, tricyclic antidepressants, monoamine oxidase inhibitors (MAO-I), and levodopa (a precursor of dopamine and NE and an intermediate metabolite of tyrosine), acutely reverse some of the negative consequences of stress in animals (Anisman and Sklar 1979; Glazer et al., 1975; Sherman et al., 1979). Also, CA antagonists, for example, inhibitors of tyrosine hydroxylase and dopamine-ß-hydroxylase, impair performance in avoidance or escape tasks. Drugs that primarily block the synthesis of NE, like FLA-63, also produce escape deficits, as do dopamine receptor blockers like haloperidol and pimozide. However, brain dopamine levels are more resistant to stress than brain NE levels (Gray, 1982; Stone, 1975).
Acute Behavioral Consequences of Combat
Little is known about the acute effects of combat and similar life-threatening stressors on human brain function. There is a large, mostly anecdotal literature on the effects of combat on mental processes. Many popular books and articles, as well as military histories, refer to the adverse consequences of combat stress on the performance of soldiers on the battlefield. In one of the few research studies conducted in a combat situation, the affect of Special Forces soldiers in an isolated outpost, deep in enemy territory during the earlier stages of the war in Vietnam, was assessed (Bourne et al., 1968). The primary symptom among these seasoned soldiers was hostility toward higher authorities at headquarters. Among these elite troops, anxiety and depression were not especially elevated. In one of the few laboratory studies that addressed the issue, Villoldo and Tarno (1984) modeled combat using battlefield noise (including simulated ordnance detonations), temperature extremes, chemical protective clothing, and a variety of other physical and psychological stressors. In that study, the subjects’ performance on a variety of standardized tests of mental performance were significantly degraded.
Efforts have also been made to formally describe the acute behavioral syndrome that occurs among some soldiers as a consequence of exposure to combat. It has been termed combat stress reaction (CSR), and its principal symptoms are anxiety, fear of death, helplessness, crying, and tiredness. Sleep is also disturbed (Solomon et al., 1989). The behavioral manifestations of the CSR syndrome appear to resemble the helplessness syndrome described in animals. It is likely that the underlying alterations in brain CA function documented in animals subjected to acute stress are also present in humans exposed to the life-threatening stress that characterizes combat.
In many battles, casualties from the CSR syndrome have been extremely high. For example, in Okinawa during World War II, CSR casualties among U.S. soldiers made up 48 percent of all wounded-in-action U.S. battlefield casualties. For the 60 days following D-Day (June 6, 1944) in France, 40 percent of battlefield casualties were due to CSR. On some battlefields, such as the early stages of the 1973 Arab-Israeli war, casualties that resulted from CSR actually exceeded those that resulted from enemy fire (Flora, 1985). These severe losses of personnel that occur during intense combat and that are attributable to CSR indicate that an intervention that would reduce such casualties would be of great benefit. Several recent review articles have discussed the use of tyrosine as a potential countermeasure to improve performance during highly stressful military operations (Owasoyo et al, 1992; Salter, 1989).
TYROSINE AND ACUTE STRESS: STUDIES IN ANIMALS
Many drugs, like the tricyclic antidepressants or MAO-I’s, that enhance central catecholaminergic transmission and that are highly effective for the treatment of human depression (after they have been administered for several weeks) have numerous acute and chronic side effects. Their acute side effects, such as drowsiness and impaired cognitive performance (Baldessarini, 1985; Curran, 1992), largely preclude their use under any circumstance in which performance enhancement is the desired outcome. Other drugs that are used to treat human anxiety disorders and acutely reduce anxiety, such as the benzodiazepines, produce acute side effects, such as drowsiness, reduced ability to maintain vigilance, and impaired memory (Koelega, 1989; McNair, 1973). Therefore, one strategy for developing performance-enhancing ration components should emphasize utilization of a nutrient that can increase catecholaminergic activity with minimal side effects.
One nutritional strategy to reduce the consequences of acute stress could be use of the dietary precursor of the CAs, tyrosine. In a number of studies, administration of tyrosine, either systemically just prior to initiation of the stress or as a dietary supplement, has been shown to partially protect animals from both the neurochemical and the behavioral consequences of the stress (Brady et al., 1980; Lehnert et al., 1984a,b; Lieberman et al., 1992; Luo et al, 1992; Rauch and Lieberman, 1990). After acute stress, animals pretreated with tyrosine more actively engage in a variety of normal behaviors in their environment compared with untreated but stressed control animals. Also, unlike stressed control animals not receiving tyrosine, the brain NE levels in treated animals were not depleted (Lehnert et al., 1984a,b).
In several of these studies (Lehnert et al., 1984a,b), tailshock was used to produce acute stress in rats. Following 60 min of such shocks, animals were permitted to recover for 15 min and were then placed in an open-field, hole poke apparatus. After observing their spontaneous behaviors for the next 10 min, the rats were euthanized and their brains were removed. Exposure to this experimental stress paradigm significantly decreased (by approximately 80 percent) open-field locomotor activity and several other spontaneous behaviors, such as rearing and hole poking, in stressed rats compared with the behaviors of unstressed control animals. However, in another group of animals that were given a diet supplemented with tyrosine and that were stressed, the frequency of these spontaneous behaviors did not differ significantly from those in the unstressed control animals. Increased dietary tyrosine apparently protected these animals from the behavioral inhibition produced by the stressor, presumably by augmenting noradrenergic, but also perhaps dopaminergic, neurotransmission (Lehnert et al., 1984a). When a similar study was conducted with rats given single intraperitoneal doses of tyrosine (200 mg/kg of body
weight) or placebo immediately prior to tailshock, tyrosine also protected the animals from the acute behavioral depression induced by the stress (Lehnert et al., 1984b). In both of those studies, among the stressed but untreated animals, declines in NE levels of 30 to 40 percent were noted in specific brain regions, such as the hypothalamus, locus coeruleus, and hippocampus. Both studies of Lehnert and colleagues reported that tyrosine administration blocked this depletion. Tyrosine also increased NE turnover in specific regions of the brain of stressed animals, as measured by regional differences in brain MHPG-SO4 levels.
Tyrosine has also been shown to facilitate several other types of animal behavior under stressful conditions. For example, it has been found to restore normal levels of aggressive behavior in animals that are subjected to cold-water stress (Brady et al., 1980). Additionally, in a stressful behavioral procedure sometimes considered to be a learned helplessness paradigm and used to screen drugs for their antidepressant activities (the Porsolt swim test [Porsolt et al., 1978]), significant dose-related potentiation of escape behavior following tyrosine administration has been observed (Gibson et al., 1982). The Porsolt test is conducted by placing the animal in a cylinder containing cold water for 3 min and assessing the duration of time that they spend actively swimming versus the amount of time that they maintain a characteristic immobile posture. Animals pretreated with tyrosine and phenylalanine (which is metabolized to tyrosine) continued to swim significantly longer than placebo-treated controls.
Studies conducted at the U.S. Army Research Institute of Environmental Medicine (USARIEM) have replicated and extended these initial studies in animals. In one series of studies (Luo et al., 1992, Rauch and Lieberman, 1990), cold-induced stress was used to produce decrements in performance. In another study, hypobaric hypoxia was the primary stressor (Lieberman et al., 1992). In the initial study of cold-induced stress (Rauch and Lieberman, 1990), rats were pretreated with 400 mg of tyrosine per kg, and their core body temperature was lowered to 30°C by immersing the animals in a cold-water bath for approximately 30 min. The Porsolt swim test was then used to assess the effects of tyrosine and cold-induced stress. As shown in Figure 15–1, the mean duration of immobility in this task increased as the core body temperature was reduced. This demonstrates that as the intensity of cold-induced stress increases, animals are less responsive to the environment. Figure 15–2 shows that when rats were pretreated with tyrosine, the duration of immobility declined significantly. In fact, tyrosine restored performance to the normal level typically observed in animals not exposed to cold-induced stress. Luo et al. (1992) replicated the findings of Rauch and Lieberman (1990) and demon-
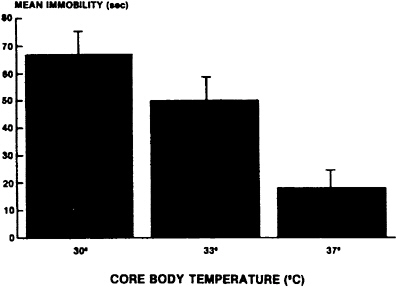
FIGURE 15–1 Changes in mean duration of immobility (in seconds) assessed in the Porsolt swim test (Porsolt et al., 1978) as a function of core body temperature. As core body temperature was reduced, the swim time decreased. Results are expressed as mean±standard error of the mean. Source: Rauch and Lieberman (1990), used with permission.
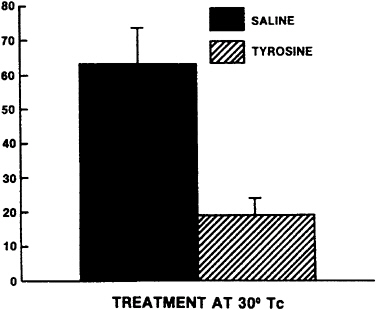
FIGURE 15–2 Effect of tyrosine (400 mg/kg given intraperitoneally) pretreatment on duration of immobility in the Porsolt swim test. Just prior to testing, the core body temperature (Tc) was lowered to 30°C. When rats were pretreated with tyrosine, the duration of immobility significantly decreased compared with that of placebo-treated rats. Results are expressed as mean ±standard error of the mean. Source: Rauch and Lieberman (1990), used with permission.
strated that the effects of tyrosine on cold-stressed animals are dose dependent (Figure 15–3).
Studies examining the effects of tyrosine on rats and humans subjected to cold-induced stress have also been conducted by the Naval Medical Research Institute (Shurtleff et al., 1992; see also Chapter 16). These studies support the hypothesis that tyrosine protects against the adverse effects of acute cold-induced stress on memory consolidation (see Chapter 16).
In another animal study conducted at USARIEM, tyrosine reversed the adverse effects of acute exposure to hypobaric hypoxia (Lieberman et al., 1992). In that study, rats were exposed to a simulated altitude of 5,950 m (19,500 ft) for 8 h. Performance was assessed by the Morris water maze, a test of spatial learning and memory (Brandeis et al., 1989) at 2 and 6 h into the exposure. A total of 400 mg of tyrosine per kg was administered intraperitoneally on two occasions: at 1.5 and 5.5 h after the start of exposure to hypoxia. This task requires animals to learn to find and remember the location of a hidden platform in a tank of water. Since the platform is not visible to the
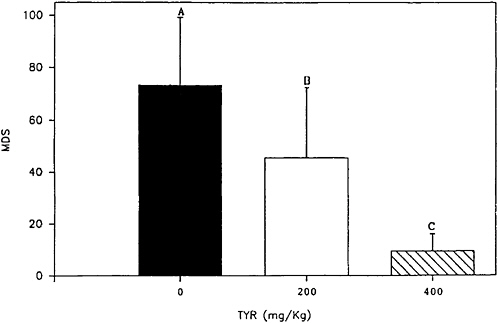
FIGURE 15–3 Effect of two doses of tyrosine (TYR; 200 and 400 mg/kg given intraperitoneally) on the duration of immobility in the Porsolt swim test (Porsolt et al., 1978). Prior to testing, the core body temperature was lowered to 30°C. The data shown here demonstrate that the effects of tyrosine on cold-stressed animals are dose dependent. Values are expressed as mean difference scores (MDS), which equals hypothermia immobility time minus normothermia immobility time. Results are expressed as mean±standard error of the mean. Letters (A-C) identify conditions that are significantly different one from another (P=0.5).
animal, spatial cues must be employed. Although hypoxia itself clearly impairs learning and memory, the water maze task probably increases the stressfulness of the situation. When this task is used to assess performance, animals are exposed to an unusual environment in which they are required to swim to escape. In addition, the task may produce thermal stress that would contribute to the overall stressfulness of the situation. The combination of hypoxia and other stressors clearly impaired performance on this task; tyrosine reduced the decrements in performance related to working (short-term) memory (Figure 15–4).
Tyrosine has also been shown to have other potentially beneficial effects in stressful environments. For example, its acute administration can lower blood pressure in spontaneously hypertensive rats that are subjected to stressful testing conditions (Sved et al., 1979) and raise blood pressure in animals made hypotensive by blood loss, which is a model of hemorrhagic shock (Conlay et al., 1981, 1985). Tyrosine also decreases the vulnerability of the canine heart to ventricular fibrillation in a dose-dependent manner and may therefore pre-
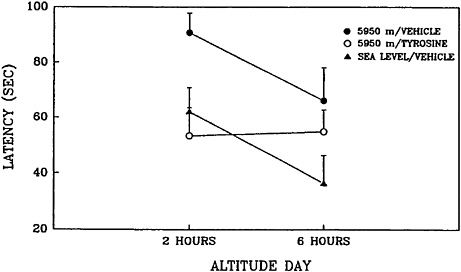
FIGURE 15–4 Effects of tyrosine and vehicle on working (short-term) memory as assessed by the Morris water maze. Rats were exposed to hypobaric hypoxia (5,950 m) or sea level conditions. A total of 400 mg of tyrosine per kg was administered intraperitoneally on two occasions: at 1.5 and 5.5 h after the start of exposure to hypoxia. Hypoxia clearly impaired performance on this task and tyrosine reduced the decrements in performance, as assessed by the latency to locate the hidden platform. Results are expressed as mean±standard error of the mean.
vent sudden, stress-induced cardiac arrest (Scott et al., 1981). It may also have beneficial effects on the neuroendocrine response to stress, since tyrosine blocks the rise in plasma corticosterone levels that occurs after unavoidable stress (Reinstein et al., 1985). It has been reported that animals who successfully cope with avoidable stress by escaping have lower levels of plasma corticosterone than animals that fail to cope (Swenson and Vogel, 1983). Some of these effects may be of particular value on the battlefield, especially tyrosine’s ability to increase blood pressure in models of hemorrhagic shock.
These observations taken together suggest that tyrosine, or any treatment that potentiates CA neurotransmission without significant side effects, may be useful for treating many of the adverse consequences of severe acute stress in humans. Presumably, combat and other operations in which soldiers are exposed to life-threatening danger are situations in which tyrosine or other catecholaminergic agonists may have some beneficial effects.
TYROSINE AND ACUTE STRESS: STUDIES IN HUMANS
Tyrosine has been administered to normal human subjects in only a few studies, although it may have beneficial effects on the mood states of certain subgroups of depressed patients (Gelenberg et al., 1983). In studies with normal males, no significant behavioral effects of tyrosine administered orally at a dose of 100 mg/kg of body weight were noted (Lieberman et al., 1983). However, subjects in that study did not experience experimental stressors, and it is under stressful conditions that tyrosine would be expected to have its positive effects on behavior.
Hypobaric Hypoxia
The first study in which the behavioral effects of tyrosine were examined in humans subjected to acute stress was conducted at USARIEM (Banderet and Lieberman, 1989). The experimental stressor was acute exposure (4 h) to a combination of hypobaric hypoxia (4,200 and 4,700 m [13,800 and 15,500 ft]) and cold (15°C [60°F]). A double-blind, placebo-controlled crossover design was used, and mood, symptoms, and mental performance were assessed by using a battery of standardized behavioral tests. Exposure of humans to such simulated altitudes rapidly produces numerous adverse changes. Cognitive performance is significantly impaired on a wide variety of simple and complex aspects of behavior. Simultaneously, mood state is adversely affected, and a series of typical symptoms appear, such as headache, lightheadedness, nausea, and general malaise. There are considerable individual differences in the
patterns and severities of these symptoms (for a review, see Banderet and Burse [1991]). Exposure to the secondary stressor, an ambient air temperature of 15°C, would not be expected to produce similar impairments in performance but certainly contributed to the overall stressfulness of the environment. In this study, tyrosine was administered as a total oral dose of 100 mg/kg of body weight over a 40-min period in two equal portions. There were substantial decrements in many of the parameters assessed among the volunteers in that study due to exposure to the stressors, and there were significant individual differences in the severities and patterns of the deficits. When these differences were accounted for, tyrosine appeared to have robust effects on those individuals whose performances on a given behavioral task were most severely affected. Tyrosine significantly mitigated many of the decrements in symptoms, mood, and performance induced by these stressors (Figures 15–5 to 15–7, respectively), including functions believed to be regulated by catecholaminergic neurons such as vigilance, alertness, and anxiety (Banderet and Lieberman, 1989).
To replicate and extend these findings, a second study was conducted at USARIEM (Lieberman et al., 1990). In that follow-up study, the duration of exposure to hypobaric hypoxia was extended to 7 h, a simulated altitude of 4,700 m was used, and the ambient air temperature was held at 17°C. Two oral doses of tyrosine were tested: 85 and 170 mg/kg. As in the previous study (Banderet and Lieberman, 1989), each treatment was given as a divided dose. The results of the second study were similar to those of the first study. Tyrosine significantly reduced impairments on many of the same performance-, mood-, and symptom-dependent variables among those individuals whose performances of the particular measure were most affected. However, the effects did not appear to be dose dependent.
Acute Cardiovascular Stress
The effects of tyrosine on humans exposed to acute stress have also been examined at the U.S. Air Force Armstrong Laboratory. In a double-blind, placebo-controlled crossover study, the effects of tyrosine on individuals subjected to an acute cardiovascular stressor, lower body negative pressure (LBNP), were assessed (Dollins et al., 1990; Lieberman et al., 1991). LBNP is a technique that is used to simulate gravitational stress (orthostasis) by exposing the lower body to subatmospheric pressures. This causes blood and interstitial fluids to pool in the lower extremities because of decreased venous return and increased sympathetic drive (Bonde-Petersen et al., 1984). Subjects exposed to LBNP initially respond with decreased blood pressure and increased heart rate. These changes continue until the cardiovascular system
cannot maintain homeostasis, and blood pressure and heart rate fall precipitously. At this stage consciousness is lost if the LBNP exposure is not terminated. LBNP was generated in the Armstrong Laboratory study by placing subjects in a specially constructed airtight chamber from the waist down. A vacuum was then applied to the chamber and subjects were exposed to two consecutive 40-min LBNP sessions separated by a brief break. The maximum negative pressure was -50 mm Hg. When tyrosine, at a dose of 100 mg/kg, was administered orally over a 1 h period (in two 50-mg/kg oral boluses), subjects were able to maintain higher pulse pressures (Figure 15–8). In addition, changes in auditory-evoked potentials suggestive of increased central nervous system activity were also observed as a consequence of tyrosine administration (Figure 15–9).
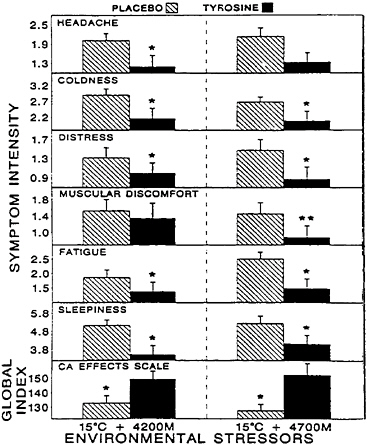
FIGURE 15–5 Effects of tyrosine treatment on a variety of parameters assessed with self-reported symptom questionnaires. The experimental stressor was a 4 h exposure to a combination of hypobaric hypoxia (4,200 m and 4,700 m) and cold (15°C). Asterisks indicate the level of statistical significance (*, P<0.05; **, P<0.01). Results are expressed as mean±standard error of the mean. CA, catecholamine. Source: Banderet and Lieberman (1989), used with permission.
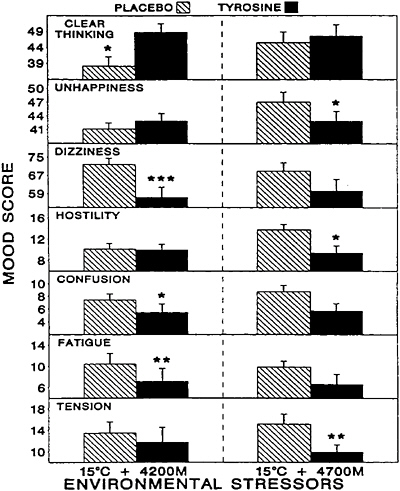
FIGURE 16–6 Effects of tyrosine treatment on mood. The experimental stressor was a 4-h exposure to a combination of hypobaric hypoxia (4,200 and 4,700 m) and cold (15°C). Asterisks indicate the level of statistical significance (*, P<0.05; **, P<0.01; ***, P<0.001). Results are expressed as mean±standard error of the mean. Source: Banderet and Lieberman (1989), used with permission.
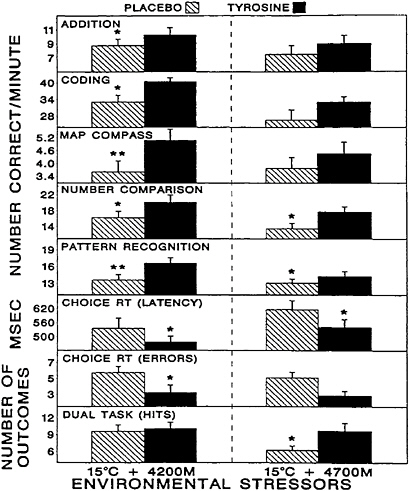
FIGURE 15–7 Effects of tyrosine treatment on cognitive, reaction time (RT), and vigilance performance. The experimental stressor was a 4-h exposure to a combination of hypobaric hypoxia (4,200 and 4,700 m) and cold (15°C). Asterisks indicate the level of statistical significance (*, P<0.05; **, P<0.01). Results are expressed as mean±standard error of the mean. Source: Banderet and Lieberman (1989), used with permission.
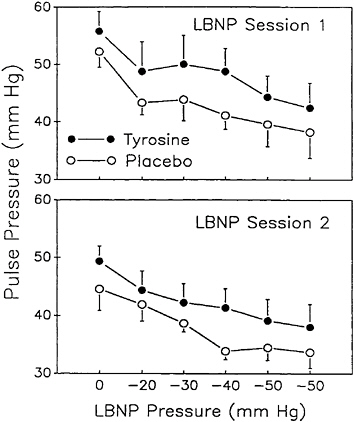
FIGURE 15–8 Change in mean±standard error of the mean, pulse pressure following treatment with 100 mg of tyrosine per kg of body weight. The experimental stressor was exposure to lower body negative pressure (LBNP).
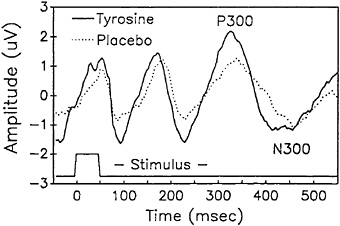
FIGURE 15–9 Change in mean amplitude of middle latency auditory evoked potential (P 300 and N 300 waves during exposure to lower body negative pressure (LBNP) stress. Subjects were treated with a total of 100 mg of tyrosine or placebo per kg of body weight.
CONCLUSIONS
Results of the studies discussed above and research presented elsewhere in this volume (see Chapter 16) suggest that the amino acid tyrosine may have beneficial effects on humans and other animals that are subjected to acute stressors. The adverse effects of hypoxia, cold, lower body negative pressure, and psychological stresses have all been reduced by treatment with tyrosine. This amino acid may have protective effects on behavioral and cardiovascular parameters because it prevents the depletion of central and peripheral catecholamines caused by acute stress. Since a variety of environmental and psychological stressors appear to deplete catecholamines, tyrosine may have wide application as a performance-enhancing ration component (PERC). However, the currently available data cannot be considered to be definitive in establishing that tyrosine should be added to a combat ration. Key issues that must be addressed by additional research include the utility of tyrosine in combat or similar situations, the generalizability of tyrosine’s effects across different stressors, the appropriate dose, and in particular, whether tyrosine should be used acutely or as a routine part of the diet.
Tyrosine can be expected to be beneficial only when the stress is severe. Intense combat and similar highly stressful military operations are therefore the situations in which tyrosine could have the greatest benefit to the armed forces. A variety of considerations, however, including ethical concerns, make it difficult to test tyrosine in a realistic manner. Previous attempts to use psychological manipulations to generate stress have not shown tyrosine to be beneficial, presumably because the stress was not sufficiently intense or prolonged (H.R.Lieberman and G.Garfield, unpublished observations). It is therefore essential that studies with this nutrient continue in animals in conjunction with studies in humans. Such studies should use environmental stressors, combinations of environmental and operational stressors, or sustained exposures to stressors. Attempts to develop a suitable human model of combat stress that can be used to evaluate tyrosine and other PERCs are also necessary.
In addition to tyrosine, a variety of other nutrients have been discussed as potential PERCs. Currently, the food constituent with the most clearly demonstrated ability to enhance behavioral performance is caffeine (for recent reviews, see Lieberman [1992] and Penetar [Chapter 20, this volume]). The circumstances in which caffeine has its clearest effects on performance are, in many respects, quite different from those in which tyrosine would be expected to be useful. Caffeine appears to improve performance when individuals are engaged in long-duration, boring activities such as driving or sentry duty. Tyrosine appears to enhance performance when acute stress degrades function. If development of both compounds as PERCs continues, then different types
of rations, or specially labeled supplements within rations, may be needed, depending on the operational situation. Caffeine would not be expected to have beneficial effects in high-stress scenarios, and since it can have adverse effects on sleep, it would not be advisable to add it to rations as a generic supplement. Although no adverse effects of tyrosine have been demonstrated, the apparent requirement for it to be used in high doses may preclude its use except in a specially identified form. The consumption of reduced levels of rations during combat, especially during its most stressful periods such as initial exposure to enemy action (Popper et al., 1984), also suggests that tyrosine could best be provided in a combat ration as a special stress-reduction item, perhaps in combination with an easily digested carbohydrate. Carbohydrate may facilitate tyrosine’s uptake into the brain and could also provide the needed energy under such circumstances (Wurtman et al., 1981).
In summary, the critical issues that should be addressed in studies in animals and/or humans include the following:
-
Demonstrate the generalizability of tyrosine’s effects across a wider range of stressors.
-
Establish the dose-response function for tyrosine’s beneficial effects.
-
Assess the risks and benefits of acute versus chronic administration of tyrosine.
-
Determine whether tyrosine has efficacy in chronic stress paradigms.
-
Determine the safety of tyrosine administration.
-
Determine the most appropriate method for using combinations of PERC-type rations.
ACKNOWLEDGMENTS
The author thanks MAJ Mary Mays for her helpful comments on earlier versions of this chapter.
REFERENCES
Anisman, H., and L.Sklar 1979 Catecholamine depletion in mice upon reexposure to stress: Mediation of the escape deficits produced by inescapable shock. Comp. Physiol. Psychol. 93:610.
Antelman, S.M., and A.R.Caggiula 1977 Norepinephrine-dopamine interactions and behavior. A new hypothesis of stress-related interactions between brain norepinephrine and dopamine is proposed. Science 195:646.
Baldessarini, R.J. 1985 Drugs and the treatment of psychiatric disorders. Pp. 387–445 in The Pharmacological Basics of Therapeutics, 7th ed., A.G.Gilman, L.S.Goodman, T.W.Rall, and F.Murad, eds. New York: Macmillan Publishing Co.
Banderet, L.E., and R.L.Burse 1991 Effects of high terrestrial altitude on military performance. Pp. 233–254 in The Handbook of Military Psychology, R.Gal and D.Mangelsdorff, eds. New York: Wiley.
Banderet, L.E., and H.R.Lieberman 1989 Treatment with tyrosine, a neurotransmitter precursor, reduces environmental stress in humans. Brain Res. Bull. 22:759–762.
Bonde-Petersen, F., M.Suzuki, and N.J.Christensen 1984 Cardiovascular and hormonal responses to bicycle exercise during lower body negative pressure. Adv. Space Res. 4(12):31–33.
Bourne, P.G., W.M.Coli, and W.E.Datel 1968 Affect levels of ten special forces soldiers under threat of attack. Psych. Rep. 22:363–366.
Brady, K., J.W.Brown, and J.B.Thurmond 1980 Behavioral and neurochemical effects of dietary tyrosine in young and aged mice following cold-swim stress. Pharmacol. Biochem. Behav. 12:667–674.
Brandeis, R., Y.Brandys, and S.Yehuda 1989 The use of the Morris water maze in the study of memory and learning. Int. J. Neurosci. 48:29–69.
Conlay, L.A., T.J.Maher, and R.J.Wurtman 1981 Tyrosine increases blood pressure in hypotensive rats. Science 212:559–560.
Conlay, L.A., T.J.Maher, and R.J.Wurtman 1985 Tyrosine accelerates catecholamine synthesis in hemorrhaged hypotensive rats. Brain Res. 333:81–84.
Curran, H.V. 1992 Antidepressant drugs, cognitive function and human performance. Pp. 319–336 in Handbook of Human Performance, Vol. 2, Health and Performance, A.P.Smith and D.M.Jones, eds. Boston: Academic Press.
Dollins, A B., L.P.Krock, W.F.Storm, and H.R.Lieberman 1990 Tyrosine decreases physiological stress caused by lower body negative pressure (LBNP). Aviat. Space Environ. Med. 61:491.
Flora, D. 1985 Battlefield Stress: Cause, Cures and Counter Measures. Report No. ACSC-85–3232. Fort Leavenworth, Kans.: Army Command and General Staff College.
Gelenberg, A.J., J.D.Wojcik, C.J.Gibson, and R.J.Wurtman 1983 Tyrosine for depression. J. Psychiatr. Res. 17(2):175.
Gibson, C.J., and R.J.Wurtman 1978 Physiological control of brain norepinephrine synthesis by brain tyrosine concentration. Life Sci. 22:1399–1406.
Gibson, C.J., S.M.Deikel, S.N.Young, and Y.M.Binik 1982 Behavioral and biochemical effects of tryptophan, tyrosine and phenylalanine in mice. Psychopharmacology 76:118–121.
Glazer, H.I., J.M.Weiss, L.A.Pohorecky, and N.Miller 1975 Monoamines as mediators of avoidance-escape behavior. Psychosom. Med. 37:535.
Gray, J.A. 1982 The Neuropsychology of Anxiety. Oxford: Clarendon Press.
Koelega, H.S. 1989 Benzodiazepines and vigilance performance: A review. Psychopharmacology 98:145–156.
Lehnert, H., D.K.Reinstein, B.W.Strowbridge, and R.J.Wurtman 1984a Neurochemical and behavioral consequences of acute, uncontrollable stress: Effects of dietary tyrosine. Brain Res. 303:215–223.
Lehnert, H., D.K.Reinstein, and R.J.Wurtman 1984b Tyrosine reverses the depletion of brain norepinephrine and the behavioral deficits caused by tail-shock stress in rats. Pp. 81–91 in Stress: The Role of the Catecholamines and Other Neurotransmitters, E.Usdin, R.Kvetnansky, and J.Axelrod, eds. New York: Gordon and Beach.
Lieberman, H.R. 1992 Caffeine. Pp. 49–72 in Handbook of Human Performance, Vol. 2, Health and Performance, A.P.Smith and D.M.Jones, eds. Boston: Academic Press.
Lieberman, H.R., S.Corkin, B.J.Spring, J.H.Growdon, and R.J.Wurtman 1983 Mood, performance, and pain sensitivity changes induced by food constituents. J. Psychiatr. Res. 17(2):135–145.
Lieberman, H.R., L.E.Banderet, and B.Shukitt-Hale 1990 Tyrosine protects humans from the adverse effects of acute exposure to hypoxia and cold. Soc. Neurosci. Abstr. 16:272.
Lieberman, H.R., A.B.Dollins, and R.J.Wurtman 1991 Strategies to sustain and enhance performance in stressful environments: The effects of tyrosine pre-treatment on lower body negative pressure stress. Air Force Office of Scientific Research Report TR-90–0403, AD A221224 Bolling Air Force Base, Washington, D.C.: Air Force Office of Scientific Research.
Lieberman, H.R., B.Shukitt-Hale, S.Luo, J.A.Devine, and J.F.Glenn 1992 Tyrosine reduces the adverse effect of hypobaric hypoxia on spatial working memory of the rat. Soc. Neurosci. Abstr. 18:715.
Lovenberg, W., E.S.Bruck, and I.Hanbauer 1975 ATP, cyclic AMP, and magnesium increase the affinity of rat striatal tyrosine hydroxylase for its cofactor. Proc. Natl. Acad. Sci. USA 72:2955–2958.
Luo, S., L.Villamil, C.J.Watkins, and H.R.Lieberman 1992 Further evidence tyrosine reverses behavioral deficits caused by cold exposure. Soc. Neurosci. Abstr. 18:716.
Maier, S.F., and R.L.Jackson 1979 Learned helplessness: All of us were right (and wrong): Inescapable shock has multiple effects. In Psychology of Learning and Motivation 13, G.H.Bower, ed. New York: Academic Press.
Maier, S.F., and M.E.P.Seligman 1976 Learned helplessness: Theory and evidence. J. Exp. Psychol. 105:3.
McNair, D.M. 1973 Antianxiety drugs and human performance. Arch. Gen. Psychiatry 29:611.
Milner, J.P., and R.J.Wurtman 1986 Catecholamine synthesis: Physiological coupling to precursor supply. Biochem. Pharmacol. 35:875–881.
Minor, T.R., R.L.Jackson, and S.F.Maier 1984 Effects of task-irrelevant cues and reinforcement delay on choice-escape learning following inescapable shock: Evidence for a deficit in selective attention. J. Exp. Psych. 10:543.
Murphy, D.L., and D.E.Redmond 1975 The catecholamines: Possible role in affect, mood and emotional behavior in man and animals. Pp. 73–117 in Catecholamines and Behavior, A.J.Freidhoff, ed. New York: Plenum Press.
Owasoyo, J.O., D.F.Neri, and J.G.Lamberth 1992 Tyrosine and its potential use as a countermeasure to performance decrement in military sustained operations . Aviat. Space Environ. Med. 63:364–369.
Popper, R.D., G.J.Smits, H.L.Meiselman, J.Moy, and H.B.Masor 1984 Eating in combat: A survey of how much and what foods U.S. Marines eat in action. U.S. Army Natick Research, Development and Engineering Center Technical Report TR-85–010. Natick, Mass.: U.S. Army Research Institute of Environmental Medicine.
Porsolt, R.D., G.Anton, N.Blavet, and M.Jalfre 1978 Forced swimming in rats: Hypothermia, immobility and the effects of imipramine. Eur. J. Pharmacol. 47:379–391.
Rapaport, P.M., and S.F.Maier 1978 Inescapable shock and food-competition dominance in rats. Animal Learning Behav. 6:160.
Rauch, T.M., and H.R.Lieberman 1990 Tyrosine pretreatment reverses hypothermia induced behavioral depression. Brain Res. Bull. 24:147–150.
Reinstein, D.K., H.Lehnert, and R.J.Wurtman 1984 Tyrosine prevents behavioral and neurochemical correlates of an acute stress in rats. Life Sci. 34:2225–2231.
1985 Dietary tyrosine suppresses the rise in plasma corticosterone following acute stress in rats. Life Sci. 37:2157–2163.
Salter, C. 1989 Dietary tyrosine as an aid to stress resistance among troops. Milit. Med. 154:144–146.
Scott, N.A., R.A.DeSilva, B.Lown, and R.J.Wurtman 1981 Tyrosine administration decreases vulnerability to ventricular fibrillation in the normal canine heart. Science 211:727–729.
Sherman, A.D., G.L.Allers, F.Petty, and F.A.Henn 1979 A neuropharmacologically-relevant animal model of depression. Neuropharmacology 18:891.
Shurtleff, D., J.R.Thomas, J.Schrot, K.Kowalski, R.Harford, M.O.Thornton, P.A.Shea, and M.Malik 1992 Tyrosine reverses a cold-stress-induced memory deficit in humans. Soc. Neurosci. Abstr. 18:1210.
Solomon, Z., M.Mikulincer, and R.Benbenishty 1989 Combat stress reaction—clinical manifestations and correlates. Milit. Psychol. 1:35–47.
Stone, E.A. 1975 Stress and catecholamines. Pp. 31–71 in Catecholamines and Behavior, A.J.Freidhoff, ed. New York: Plenum Press.
Sved, A.F., J.D.Fernstrom, and R.J.Wurtman 1979 Tyrosine administration reduces blood pressure and enhances brain norepinephrine release in spontaneously hypertensive rats. Proc. Natl. Acad. Sci. USA 76:3511–3514.
Swenson, R., and W.Vogel 1983 Plasma catecholamine and corticosterone as well as brain catecholamine changes during coping in rats exposed to stressful footshock. Pharmacol. Biochem. Behav. 18:689–693.
Van der Kolk, B., M.Greenberg, H.Boyd, and J.Krystal 1985 Inescapable shock, neurotransmitters and addiction to trauma: Toward a psychobiology of post traumatic stress. Biol. Psychiatry 20:314–325.
Villoldo, A., and R.L.Tarno 1984 Measuring the performance of EOD equipment and operators under stress. Naval Explosive Ordnance Disposal Technology Center, NAVEODTECHCEN Technical Report T-270, AD-B083850L, San Francisco, Calif: Frederick Burk Foundation Research Center.
Weiner, N., F.L.Lee, E.Dreyer, and E.Barnes 1978 The activation of tyrosine hydroxylase in noradrenergic neurons during acute nerve stimulation. Life Sci. 22:1197–1216.
Weiss, J., H.Glazer, and L.Pohorecky 1976 Coping behavior and neurochemical changes: An alternative explanation for the original ‘learned helplessness’ experiments. Pp. 141–173 in Animal Models in Human Psychobiology, G.Serban and A.King, eds. New York: Plenum Press.
Wurtman, R.J., F.Larin, S.Mostafapour, and J.D.Fernstrom 1974 Brain catechol synthesis: Control by brain tyrosine concentration. Science 185:183–184.
Wurtman, R.J., F.Hefti, and E.Melamed 1981 Precursor control of neurotransmitter synthesis. Pharmacol. Rev. 32:315–335.
DISCUSSION
EDWARD HORTON: I am fascinated by the hypoxia studies, having spent a couple of summers myself up on Mount Logan studying and experiencing acute mountain sickness. I was wondering whether you have looked at the possible peripheral effects of tyrosine? For example, have you monitored arterial blood gas and looked at pCO2 [partial carbon dioxide pressure] and pH because they have big effects on cerebral blood flow as well.
HARRIS LIEBERMAN: Our studies were designed to study the behavioral effects of tyrosine. In our first study, we did look at blood pressure and heart rate and did not see any changes associated with the administration of tyrosine, but we did not focus on those indicators in particular because they are such crude measures of the effect of hypoxia.
JOEL GRINKER: I have two questions. Have you done any other studies, and would you expect to see a threshold effect with dose-response work?
HARRIS LIEBERMAN: As the answer to the first question, have you done studies on other neurotransmitters, yes, we focused on acetylcholine initially
and norepinephrine as well. The data for the other neurotransmitters are not complete at this point, and I cannot present any information on that. The other question had to do with dose-response studies. Unless I have other information, I would always assume that a dose-response function rather than a step function will be found.
JOEL GRINKER: I just wanted some clarification as to what the feeding paradigm that you referred to in the tyrosine study was, and when you said there was a significant factor, what you meant. You mentioned the feeding paradigm.
HARRIS LIEBERMAN: We always gave tyrosine by injection. Richard Wurtman and Henrik Lehnert did studies in which they fed tyrosine in the diet and found effects. I believe they gave it for a week in advance of the study, or at least several days. Dick Wurtman could probably give you more detail on that. Does this answer your question?
JOEL GRINKER: No, I think it was your dependent measures that you were talking about.
HARRIS LIEBERMAN: Oh, that was Tim Maher, and I would rather he would comment on that.
TIMOTHY MAHER: We made rats hyperphagic by removing food for 4 h and then tested the effects of a number of compounds alone or in combination with tyrosine. Although tyrosine had no effect on food intake, it significantly potentiated the effects of these factors.
























