16
Tyrosine and Glucose Modulation of Cognitive Deficits Resulting from Cold Stress
Stephen T.Ahlers,1 John R.Thomas, John Schrot, and David Shurtleff
INTRODUCTION
COLD STRESS AND WORKING MEMORY
Research at the Naval Medical Research Institute on the effects of nutritional components on performance has centered primarily on alleviation of cold-induced impairment of short-term or working memory by the catecholamine precursor tyrosine and the simple sugar glucose. Efforts have focused on measures of working memory specifically, since research has shown that working memory is uniquely susceptible to disruption by cold stress. This does not imply that other cognitive abilities are not deleteriously affected by exposure to cold stress. Indeed, it is well established that impaired
cognitive function results from exposure to cold-induced stress that induces core hypothermia (Coleshaw et al., 1983a,b; Webb, 1984). Rather, research at the Naval Medical Research Institute has focused on the effects of exposure to cold air stress that does not result in an obvious drop in core temperature, as determined by standard thermometry methods. Under these conditions, exposure to cold ambient air (2–5°C) for periods of even as short as 1 h produces reliable and robust impairment of working memory, as determined by a delayed matching-to-sample (DMTS) test (Thomas et al., 1989, 1991a).
Figure 16–1 displays a diagram of a typical trial on the DMTS procedure used to test human subjects. At the beginning of the trial, a subject is presented with a sample stimulus matrix on a computer screen for 2 s. The screen is then blanked for a randomly determined delay of 2, 8, or 16 s. Following the delay the subject is presented with the original sample matrix and another matrix that differs slightly in that the color of one square is reversed. For a correct response, the subject must match the original sample stimulus. A typical session consists of 90 trials and takes approximately 30 min to complete. Subjects perform the DMTS task in the last 30 min of a 60-to 90-min exposure session in which the ambient temperature of the environmental chamber is either 4° or 23°C. The effect of exposure to cold stress on
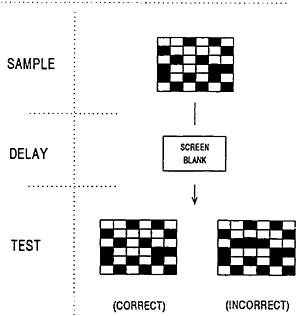
FIGURE 16–1 A typical trial with the computerized version of the human delayed matching-to-sample test. Stimuli are presented on a computer screen, and subjects respond appropriately on a keyboard.
DMTS task performance in humans is depicted in Figure 16–2. When subjects perform the task under normothermic conditions there is a gradual decrease in matching accuracy as the delay between the termination of the sample stimulus and the presentation of the comparison stimulus is increased. During exposure to cold-induced stress, this descending slope function is increased substantially, indicating that exposure to cold air causes the information in an individual’s working memory to be forgotten more quickly.
It is important to note that matching accuracy at the 2-s delay was unimpaired by the cold stress. The fact that cold stress did not decrease matching accuracy at the 2-s delay might be interpreted to indicate that moderate levels of exposure to cold stress does not impair the ability of an individual to encode the stimulus into memory, but specifically affects memory retention over time. In cases in which performance at the shorter delay times is also decreased, i.e., the y intercept is reduced, it is generally agreed that the initial encoding of information into short-term memory is impaired (Bushnell, 1990). Ahlers et al. (1993) recently observed that a longer exposure to cold
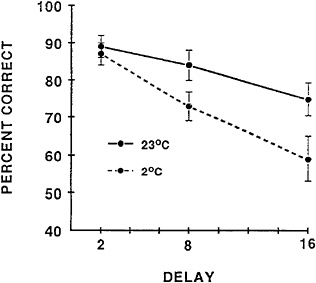
FIGURE 16–2 Matching accuracy of human subjects performing the delayed matching-to-sample task during exposure to ambient temperatures of 23° and 4°C. Delay is measured in seconds.
stress often produces impaired performance after both short and long delays. Changes in the y intercept (encoding) and in the slope function (retention) may reflect distinct neurobiological processes which, as described below, appear to influence the conditions in which nutritional factors ameliorate cold-induced memory decrements.
To elucidate the basic mechanisms that underlie the cold-induced memory deficit and to test prospective agents that could alleviate these effects, an animal model was developed in which rats performed a DMTS task. Although there are obvious procedural differences in the human and rat versions of the DMTS task, it is interesting to note that the decay of information over time function in short-term memory is similar in both species over a relatively brief retention interval at a normal temperature (23°C) (Figure 16–3). More striking perhaps is the similarity in the performance of humans and rats during exposure to cold-induced stress. Since rats show cold-induced impairment of working memory similar to what is observed in human subjects exposed to cold stress, initial studies of pharmacological treatments to improve performance during cold stress used this animal model.
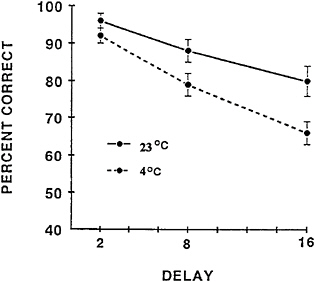
FIGURE 16–3 Matching accuracy of rats performing the delayed matching-to-sample task during exposure to ambient temperatures of 23° and 2°C. Delay is measured in seconds. Source: Thomas et al. (1991a), used with permission.
TYROSINE EFFECTS ON COLD-INDUCED MEMORY IMPAIRMENT
As shown by the extensive work by Wurtman, Lieberman, and others, administration of the catecholamine precursor tyrosine can alleviate many of the deleterious effects of acute stress by increasing the amount of catecholamine neurotransmitter depleted by a stressful event (Banderet and Lieberman, 1989; Lehnert et al., 1984; Rauch and Lieberman, 1990; Reinstein et al., 1984). Although previous studies have demonstrated that exposure to ambient cold air stress increases the turnover rate of catecholamines, which could possibly deplete neuronal stores (Gibson and Wurtman, 1978), there was still uncertainty whether the deficit in working memory that they observed was due to a stress effect that could be reversed with tyrosine or whether the deficit was derived from a direct temperature effect on neural function. The reason for this was that although the cold-induced impairment of working memory appeared to occur in the absence of core hypothermia, as determined with standard rectal thermistor probes. A study by Ahlers et al. (1991) indicated that the cold-induced memory deficit may in fact be due to a direct effect on brain temperature and, thus, may have very little to do with activation of stress pathways. They found that during exposure to cold air, the temperature in the hippocampus, a key brain structure involved in the modulation of working memory, was decreased by 1°C compared with the temperature when the DMTS task was performed during normothermic conditions. This finding added some weight to the suggestion that subtle temperature gradients in the brain may underlie many cold-induced deficits in cognition (Pozos, 1986). Because of the uncertainty regarding the nature of the cold-induced deficit in working memory, investigators examined the effects of the stress- activating hormone corticotropin-releasing factor (CRF) in the rodent model system. There were at least two compelling reasons for using CRF to examine the effects of stress on working memory. First, CRF produces the release of brain catecholamines (Dunn and Berridge, 1987; Lenz et al., 1987). Second, administration of CRF has been shown to produce behavioral deficits that are attenuated by pretreatment with tyrosine (Ahlers et al., 1992).
Cold-Induced Amnesia as a Stress Effect
Administration of CRF produced dose- and delay-dependent impairments (Figure 16–4). Low doses of CRF (0.1 and 0.3 µg) decreased DMTS task accuracy at the long delays, whereas the 1.0-µg dose of CRF decreased DMTS task accuracy across all of the delays. The pattern of impaired DMTS performance with observed increasing doses of CRF is similar to the impair-
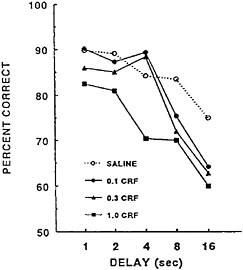
FIGURE 16–4 Effects of corticotropin-releasing factor (CRF; in micrograms) on the delayed matching-to-sample task matching accuracy in rats. Rats were administered CRF or saline intracerebroventiculary 30 min prior to the test session.
ment observed with increasing exposure to cold air stress. This suggested that the impairment of working memory may in fact be due to activation of stress pathways that would produce a sustained release and an eventual depletion of endogenous brain catecholamines.
Initial Studies with Tyrosine
Because the impairment of working memory caused by cold-induced stress may in fact result from activation of the stress response system that could deplete catecholamine stores, Shurtleff et al. (1993) initially investigated whether administration of tyrosine would alleviate the cold deficit in rats exposed to cold ambient air-induced stress. The doses of tyrosine used ranged from 50 to 200 mg/kg of body weight and were administered approximately 30 min prior to the cold exposure. The results of the study with a rodent model (Shurtleff et al, 1993) indicated that tyrosine partially alleviated the deficit in matching accuracy.
Given the success in reducing the cold-induced memory impairment in rats, the emphasis was shifted to examine the effects of tyrosine in human subjects exposed to cold stress in the environmental chamber at the Naval Medical Research Institute, Bethesda, Maryland (Shurtleff, et al., 1994). In that study, administration of 150 mg/kg of tyrosine completely reversed the cold-induced memory impairment. What was particularly relevant in this finding
was that tyrosine was most effective under conditions in which there was a substantial cold-induced decrement only at the longest delay time (16 s). These data clearly indicate that tyrosine effectively blocks an impairment in working memory when cold-induced stress specifically affects memory retention, i.e., in a situation in which accuracy was impaired only at the long delay.
Tyrosine Studies During Field Operations in the Cold
Once the tyrosine-induced alleviation of a cold-induced impairment of working memory in the laboratory chamber with humans was demonstrated, the next logical step was to determine whether tyrosine pretreatment would be beneficial in field situations with military personnel operating in a cold environment. Figure 16–5 shows the effects of tyrosine pretreatment on military personnel under field conditions. In this situation, military personnel were performing maneuvers in a cold weather environment in which the ambient temperature was -20°C. After spending nearly all day performing operations in the cold, half of the personnel were given 75 mg/kg of tyrosine and the other half were given a placebo. The subjects performed the DMTS task 1 h after ingestion of the placebo or tyrosine. In personnel given tyrosine
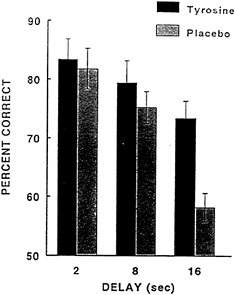
FIGURE 16–5 Effect of tyrosine pretreatment on military personnel. Tyrosine or placebo was administered to military personnel during cold weather field exercises prior to performing the delayed matching-to-sample task.
there was a substantial improvement in task accuracy at the 16-s interval compared with that in the subjects given the placebo. These results were very similar to the effects of tyrosine observed in the environmental chamber study.
The finding that tyrosine was efficacious during field studies is significant in two ways. First, it demonstrates the effectiveness of tyrosine under operational conditions most similar to conditions for which it might ultimately be recommended and used. Second, but no less significant, it validates the observation that working memory is impaired when military personnel perform duties during exposure to cold air stress. When taken together with previous research demonstrating tyrosine’s beneficial effects in military personnel under a variety of stressful conditions with several different test parameters (Banderet and Lieberman, 1989), it is clear that tyrosine administration is capable of improving cognitive performance in military personnel exposed to cold stress.
The Effects of Tyrosine on Cold-Induced Impairment in Timing
In the original observation demonstrating the effects of moderate cold stress on working memory in rats, Thomas et al. (1991a) observed that in addition to a deficit in matching accuracy, rats’ latencies to respond to the test stimulus decreased, whereas their latencies to respond to the sample stimulus were unchanged. In humans performing the DMTS task during cold exposure in an environment of 4°C, sample response times were slower, and test response latencies were shorter (Thomas et al., 1989). Collectively, these findings indicate that a concomitant effect of the cold-induced impairment of short-term memory may be an alteration in the speed with which cognitive operations are performed. Recent data are consistent with the notion that cold-induced stress might modulate the internal clock speed. During exposure to cold air stress, the rate of response in rats performing a differential reinforcement of low rate (DRL) procedure, which requires subjects to time their responses to a fixed time interval, increased substantially (Thomas et al., 1991b).
Although the degree to which a decrement in timing and impairment of working memory produced by exposure to cold stress may reflect some common physiological dysfunction has yet to be elucidated, investigators have examined whether tyrosine would also ameliorate the effects of cold stress exposure on timing behavior. For this experiment, rats trained on the DRL procedure were administered tyrosine during exposure to cold and normothermic environments. These data are shown in Figure 16–6. Increasing doses of the catecholamine precursor attenuated the increase in the rate of DRL responding during cold stress. The two highest doses of tyrosine essentially
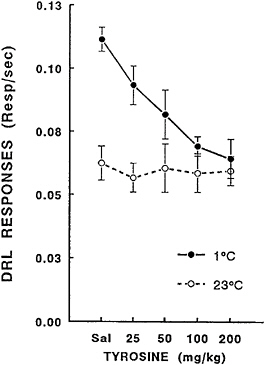
FIGURE 16–6 Effects of tyrosine administration on cold-induced increases in differential reinforcement of low rate (DRL) procedure in rats exposured to ambient temperatures of 23° and 1°C. Sal=saline.
normalized the rats’ ability to meet the requirements for the DRL procedure. These results indicate that administration of tyrosine completely blocked cold-induced changes in response timing.
Conclusions and Recommendations Regarding the Use of Tyrosine
The observation that tyrosine alleviates cold-induced impairment of working memory and timing behavior is consistent with the demonstrated efficacy of tyrosine in a variety of test situations with both human and animal subjects. Of particular relevance to the recommendation that tyrosine might be given to deployed military personnel are the findings of Banderet and Lieberman (1989), who have also demonstrated the efficacy of tyrosine in human subjects. Their findings demonstrate that tyrosine can reverse a number of the neuropsychological and emotional effects of the combined stresses of
exposure to cold and high altitudes. Tyrosine has been found to block effectively the cold-induced deficits in working memory observed in humans both in the laboratory and in military field operations in cold weather. Taken together, these studies indicate that tyrosine certainly has potential as a nutritional component for improving the compromised performance of military personnel operating under stressful conditions (Owasoyo et al.,1992; Salter, 1989).
Situations in Which Tyrosine is Less Effective
As encouraging as these results with tyrosine may be, some caution is warranted since investigators have found that tyrosine is not universally effective in ameliorating performance decrements resulting from cold-induced stress. For example, administration of tyrosine is less effective in individuals with cold-induced learning deficits measured by a repeated acquisition (RA) task. In the RA procedure, subjects are required to learn a new sequence of appropriate responses on a daily basis. In contrast to the DMTS procedure, the RA task places a much greater demand on an individual’s ability to learn new information. For this reason, it takes many trials for an individual to learn the new sequence and makes the RA task particularly sensitive in revealing learning (encoding) deficits. In laboratory and field studies, exposure to cold-induced stress consistently impairs an individual’s ability to learn new information using the RA task, such that subjects make more errors over the course of a session. In studies in both humans and rats, tyrosine has not demonstrated consistent effects in modifying cold-induced decrements in the ability to learn a new response sequence as part of the RA task.
Tyrosine and Chronic Stress
The potential restrictions on circumstances in which tyrosine is effective in modifying a stress effect highlight the more general concern of defining the conditions in which tyrosine most effectively blocks stress-induced impairments. Clearly, more focused research is needed to determine these situations before it is recommended as a nutritional supplement for military personnel. Another important consideration for agents such as tyrosine is whether the efficacy may change as a function of the duration that subjects are exposed to cold temperatures. Quite apart from a concern for potential shifts in a tyrosine dose-response curve is the question of whether there might be conditions in which tyrosine administration would be less beneficial or even harmful to the health and safety of the personnel who would use it.
Several studies in individuals with chronic stress have shown that there are significant changes in the synthesis and disposition of endogenous catecholamines in the central nervous system (CNS) that could affect the pharmacodynamics of tyrosine. For example Nisenbaum et al. (1991) have shown that animals chronically exposed to the cold show a greater increase in NE release in the rodent hippocampus during acute restraint stress, as measured by in vivo microdialysis. In subsequent studies, Nisenbaum and Abercrombie (1992) demonstrated augmented release of NE in rats exposed to chronic cold-induced stress is due to a significant increase in the amount of tyrosine hydroxylase (TH), the rate-limiting enzyme for the synthesis of the catecholamines. These findings suggest that the effects of tyrosine in the brains of individuals who have been exposed to cold-induced stress for a long duration may be considerably altered.
There is still another concern about what effects tyrosine might have in chronically stressed organisms. In a recent study, Ahlers and Salander (1993) demonstrated that the acute disruptive effects of CRF are lessened considerably when CRF is administered chronically. What is particularly relevant to the use of tyrosine is the fact that a recent study by Melia and Duman (1991) demonstrated that chronic administration of CRF produces a dramatic increase in TH in the locus ceruleus. These studies indicate that chronic stress produces enduring alterations in the noradrenergic and CRF stress response systems. These changes, while adaptive, may also be the physiological antecedent conditions that lead to stress-induced pathological disease states (Chrousos and Gold, 1992; Stone, 1987). Significant alterations in the noradrenergic and CRF systems in individuals with chronic stress may not only modify the effects of tyrosine, but they may also be modified by the presence of tyrosine. These observations underscore the need for additional information on how tyrosine may modulate, or be modulated by, chronic stress.
Conditions in Which Tyrosine Would Be Contraindicated
Although tyrosine may have positive or negative effects in the CNS, depending upon situational factors, there is also the possibility that supplemental tyrosine might augment untoward peripheral actions of catecholamine release under conditions of cold stress. This has yet to be examined empirically. It is well established that peripheral NE is released in significant amounts during cold stress (Thomas et al., 1990). This drives the vasoconstriction response and helps to shunt blood to the core and maintain body temperature. There is some evidence that hypertension brought about by stress experienced in a cold environment may potentiate peripheral vasoconstrictive mechanisms and thus predispose military personnel to cold-induced vasoneuropathy such
as nonfreezing cold injury or frostbite injury (Sampson, 1984). Before tyrosine is given to troops in the field, it will be important to determine whether it has any augmented effect on peripheral vasoconstriction during exposure to cold stress.
GLUCOSE EFFECTS ON COLD-INDUCED MEMORY IMPAIRMENT
The use of glucose as a nutritional factor for enhancing working memory during cold-induced stress was a result of studies demonstrating its effectiveness in improving learning and memory rather than from the development of treatments to specifically alleviate stress. Glucose has been shown to enhance memory and to alleviate memory deficits resulting from a variety of conditions (see Gold [1991] for a review). Although a majority of studies demonstrating the enhancing effects of glucose on memory have focused on the modulation of long-term memory, several reports have shown that glucose can also enhance working memory as well (Means and Fernandez, 1992; Ragozzino and Gold, 1991; Stone et al., 1991). Because of its reported effectiveness, the DMTS task (Thomas et al., 1991a) was used to examine the effects of glucose administration on the cold-induced impairment of working memory in rats. In that study, the exposure to the cold air stress produced an impairment of matching accuracy at all of the delays (Figure 16–7).
Recall from the previous discussion that when cold induces a decrease in matching accuracy at the short delays, the effect of cold on working memory is considered to be at least partially derived from an inability to initially encode information. This is of particular importance since the effects of glucose on cold-induced impairment of matching accuracy in rats are specific to only the short delays. Doses of glucose of between 10 and 100 mg/kg of body weight completely blocked the cold-induced impairment in accuracy at the 1- and 2-s delays. At the 4-s delay, all but the lowest glucose dose alleviated the decrease in matching accuracy resulting from exposure to cold stress. Although there appeared to be some improvement in performance at the 8-s delay with glucose, this did not reach statistical significance. Glucose did not alleviate the cold-induced decrement at the 16-s delay. There was no detectable improvement in performance with glucose administration in that study when rats performed in an ambient temperature of 23°C (data not shown). This was likely due to the fact that rats were at an asymptotic level of performance, especially at the short delay intervals when glucose appeared to be most effective in rats exposed to cold stress.
If the glucose preferentially modulates working memory processes by enhancing encoding, then one would expect there to be a greater effect of glu-
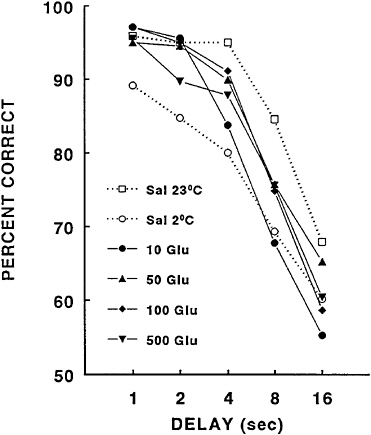
FIGURE 16–7 Glucose effects on performance impairment when rats are exposed to cold, stress (2°C). Sal=saline; Glu=glucose. Source: Ahlers et al. (1993), used with permission.
cose in a procedure that is more sensitive to encoding deficits. For this reason, the effects of glucose were examined by using the RA procedure, in which rats are required to learn a novel response sequence each day. The data showing the effects of a 50-mg/kg dose of glucose on the number of errors within a test session as a function of cold stress are depicted in Figure 16–8. In a procedure in which the acquisition of the response sequence is protracted over multiple trials, glucose not only decreased the number of errors across the session during cold stress, but it also produced fewer errors during exposure to 23°C.
Several interesting aspects of these data are worth considering. First, when utilizing the RA procedure, glucose was effective in alleviating a cold-induced decrement in performance, whereas tyrosine had marginal efficacy, at least in rats. In the case of impairment of working memory resulting from exposure to cold stress, glucose would appear to modulate the encoding of information,
whereas tyrosine would appear to affect the retention of already encoded information over time. Second, administration of glucose improved performance under nonstressed conditions. This situation is quite different from the effects of tyrosine, which is effective only during stress.
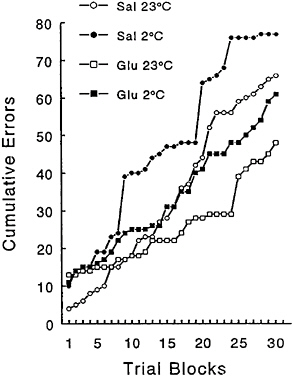
FIGURE 16–8 Effects of administration of 50 mg/kg of glucose on the number of errors committed in the repeated acquisition procedure during exposure to 23° and 2°C. Sal=saline; Glu= glucose.
Mechanisms of Action of Glucose on Working Memory
The cold-induced encoding observed when rats perform the DMTS task is similar to the effects of scopolamine, an acetylcholine (ACh) antagonist, on similar tests of working memory in rats (Bushnell, 1990). This is important since one of the proposed mechanisms for the effects that glucose has on memory is an increase in ACh levels when glucose acts as precursor to acetylcoenzyme A (Gold, 1991; Messier et al., 1991). Support for the fact that glucose modulates working memory by this type of mechanism is found in several studies demonstrating that glucose alleviates the impairment of performance produced by a cholinergic blockade (Ragozzino and Gold, 1991; Stone et al., 1991).
Another possible mechanism of action of glucose for the enhancement of performance during cold-induced stress may stem from a direct increase in glucose-mediated metabolism in specific regions of the brain. Reduced glucose utilization in those regions of the brain associated with working memory is correlated with impaired performance (Gage et al., 1984). If brain temperature were an indication of metabolic activity and glucose uptake, then the observation that the cold-induced impairment of working memory is associated with a 1°C decrease in hippocampal temperature (Ahlers et al., 1991) may have relevance to the effects of glucose on the cold-induced deficit. It is possible that administration of glucose offsets the reduction in metabolic activity and brain temperature and thus attenuates the cold-induced decrement.
Although the mechanism of action of glucose on working memory during cold stress is not known, it is fair to say that there is an abundance of data to support the notion that glucose plays an important modulatory role in cognitive processes. In addition, there has been recent speculation, supported by experimental data, that many of the putative cognition enhancers, i.e., nootropic drugs, which might be recommended for use in improving cognitive performance in military personnel, produce their beneficial effects by increasing the availability and uptake of glucose in the CNS (Wenk, 1989).
Conclusions and Recommendations Regarding Glucose as a Cognition Enhancer
The results of studies with glucose indicate that, as a nutritional component, it has the potential to enhance the performance of military personnel in operational environments. Like tyrosine, however, the circumstances in which glucose is effective in blocking a stress-induced impairment need further study.
CONCLUSIONS
-
Studies have demonstrated that as nutritional supplements, tyrosine and glucose improve particular components of working memory during cold stress. A particularly important aspect of the research with tyrosine is the demonstration that it is effective in alleviating performance decrements in military personnel working under operational cold stress conditions.
-
Under most circumstances, the demonstration of tyrosine efficacy in these situations would be the prelude to recommending that tyrosine be added as a staple in the diet when personnel are likely to operate under stressful conditions.
-
As promising as tyrosine is, however, more research is needed to examine the effects of tyrosine in terms of how the whole organism is affected by tyrosine administration, especially under conditions of chronic stress.
-
Progress with glucose as a performance enhancer during cold-induced stress is not as advanced as progress with tyrosine. The logical next step for glucose is to examine its effects in military personnel in studies in controlled environmental chambers and in field exercises and to consider its effects under acute and chronic stress conditions as well.
-
In addition, given that treatments with such compounds as tyrosine and glucose may have distinct neurological effects, studies should examine the interaction of these agents when administered in combination, since, in all probability, no one treatment is likely to be universally effective in all situations.
ACKNOWLEDGMENTS
The authors gratefully acknowledge the input of Patricia M.Thornton. This research was supported by Naval Medical Research and Development Command Research and Technology Work Units 61152N.MR00001.001.1383, 62233N.MM33C30.004–1002, and 63706NM0095.004.1008.
The experiments with rats were conducted according to the principles set forth by the National Research Council (1986).
REFERENCES
Ahlers, S.T., and M.K.Salander 1993 Chronic administration of corticotropin releasing factor (CRF) results in tolerance to the suppression of performance observed with acute administration. Pharm. Biochem. Behav. 44:375–380.
Ahlers S.T., J.R.Thomas, and D.L.Berkey 1991 Hippocampal and body temperature changes in rats during delayed matching-to-sample performance in a cold environment. Physiol. Behav. 50:1013–1018.
Ahlers, S.T., M.K.Salander, D.Shurtleff, and J.R.Thomas 1992 Tyrosine pretreatment alleviates suppression of schedule-controlled responding produced by corticotropin releasing factor (CRF) in rats. Brain Res. Bull. 29:567–571.
Ahlers, S.T., D.Shurtleff, J.Schrot, J.R.Thomas, and F.Paul-Emile 1993 Glucose attenuates cold-induced impairment of delayed matching-to-sample performance in rats. Psychobiology 21:87–92.
Banderet, L.E., and H.K.Lieberman 1989 Treatment with tyrosine, a neurotransmitter precursor, reduces environmental stress in humans. Brain Res. Bull. 22:759–762.
Bushnell, P.J. 1990 Modelling working and reference memory in rats: Effects of scopolamine on delayed matching-to-position. Behav. Pharm. 1:419–427.
Chrousos, G.P., and P.W.Gold 1992 The concepts of stress and stress system disorders: Overview of physical and behavioral homeostasis. J. Am. Med. Assoc. 267:1244–1252.
Coleshaw, S.R.K., H.M.Davis, W.R.Keatinge, R.N.M.VanSomeren, and A.H.Wolff 1983a Memory registration at mildly depressed deep body temperatures. J. Physiol. 325:58.
Coleshaw, S.R.K., R.N.M.VanSomeren, A.H.Wolff, H.M.Davis, and W.R.Keatinge 1983b Impaired memory registration and speed of reasoning caused by low body temperature. J. Appl. Physiol. 55:27–31.
Dunn, A.J., and C.W.Berridge 1987 Corticotropin-releasing factor administration elicits a stress-like activation of cerebral catecholaminergic systems. Pharmacol. Biochem. Behav. 27:685–691.
Gage, F.H., P.A.T.Kelly, and A.Bjorklund 1984 Regional changes in brain glucose metabolism reflect cognitive impairments in aged rats. J. Neuroscience 4:2856–2865.
Gibson, C.J., and R.J.Wurtman 1978 Physiological control of brain norepinephrine synthesis by brain tyrosine concentration. Life Sci. 22:1399–1406.
Gold, P.E. 1991 An integrated memory system: From blood to brain. Pp. 391–418 in Peripheral Signaling of the Brain: Role in Neural-Immune Interactions and Learning and Memory, R.C.A. Frederickson, J.L.McGaugh, and D.L.Felten, eds. Lewiston, N.Y.: Hogrefe & Huber Publishers.
Lehnert, H., D.K.Reinstein, B.W.Strowbridge, and R.J.Wurtman 1984 Neurochemical and behavioral consequences of acute, uncontrollable stress: Effects of dietary tyrosine. Brain Res. 303:215–223.
Lenz, H.J., A.Raedler, H.Greten, and M.R.Brown 1987 CRF initiates biological actions within the brain that are observed in response to stress. Am. J. Physiol. 252(Reg. Integr. Comp. Physiol.):R34–R39.
Means, L.W., and T.J.Fernandez 1992 Daily glucose injections facilitate performance of a win-stay water-escape working memory task in mice. Behav. Neurosci. 106:345–350.
Melia, K.R., and R.S.Duman 1991 Involvement of corticotropin-releasing factor in chronic stress regulation of the brain noradrenergic system. Proc. Nat. Acad. Sci. USA 88:8382–8386.
Messier, C., T.Durkin, O.Mrabet, and C.Destrade 1991 Contribution of hippocampal acetylcholine synthesis to the memory-improving action of glucose. Pp. 473–477 in Peripheral Signaling of the Brain: Role in Neural-Immune Interactions and Learning and Memory, R.C.A.Frederickson, J.L.McGaugh, and D.L. Felten, eds. Lewiston, N.Y.: Hogrefe & Huber Publishers.
National Research Council 1986 Guide for the Care and Use of Laboratory Animals. Institute of Laboratory Animal Resources. Washington, D.C.: National Academy Press.
Nisenbaum, L.K., and E.D.Abercrombie 1992 Enhanced tyrosine hydroxylation in hippocampus of chronically stressed rats upon exposure to a novel stressor. J. Neurochem. 58:276–281.
Nisenbaum, L.K., M.J.Zigmond, A.F.Sved, and E.D.Abercrombie 1991 Prior exposure to chronic stress results in enhanced synthesis and release of hippocampal norepinephrine in response to a novel stressor. J. Neurosci. 11(5):1478–1484.
Owasoyo, J.O., D.F.Neri, and J.G.Lamberth 1992 Tyrosine and its potential use as a countermeasure to performance decrement in military sustained operations. Aviat. Space Environ. Med. 63:364–369.
Pozos, R.S. 1986 Cold stress and its effects on neural function. Pp. 25–35 in Human Performance in the Cold, G.A.Laursen, R.S.Pozos, and F.Hempel, eds. Bethesda, Md.: Undersea Medical Society.
Ragozzino, M.E., and P.E.Gold 1991 Glucose effects on mecamylamine-induced memory deficits and decreases in locomotor activity in mice. Behav. Neural Biol. 56:271–282.
Rauch, M.T., and H.R.Lieberman 1990 Tyrosine pretreatment reverses hypothermia-induced behavioral depression. Brain Res. Bull. 24:147–150.
Reinstein, D.K., H.Lehnert, N.A.Scott, and R.J.Wurtman 1984 Tyrosine prevents behavioral and neurochemical correlates of an acute stress in rats. Life Sci. 34:2225–2231.
Salter, C.A. 1989 Dietary tyrosine as an aid to stress resistance among troops. Milit. Med. 154:144–146.
Sampson, J.B. 1984 Anxiety as a factor in the incidence of combat cold injury: A review. Milit. Med. 149:89–91.
Shurtleff, D., J.R.Thomas, S.T.Ahlers, and J.Schrot 1993 Tyrosine ameliorates a cold-induced delayed matching-to-sample performance decrement in rats. Psychopharmacology. (Berl.) 112:228–232
Stone, E.A. 1987 Brain noradrenergic mechanisms in models of depression. Pp. 263–277 in Hormones and Depression, U.Halbreich, ed. New York: Raven Press.
Stone, W.S., B.Walser, S.D.Gold, and P.E.Gold 1991 Scopolamine- and morphine-induced impairments of spontaneous alternation performance in mice: Reversal with glucose and with cholinergic and adrenergic agonists. Behav. Neurosci. 105:264–271.
Thomas, J.R., S.T.Ahlers, J.F.House, and J.Schrot 1989 Repeated exposure to moderate cold impairs matching-to-sample performance. Aviat. Space Environ. Med. 60:1063–1067.
Thomas, J.R., S.T.Ahlers, J.Schrot, J.F.House, K.F.Van Orden, M.M.Winsborough, R.L. Hesslink, and S.B.Lewis 1990 Adrenergic response to cognitive activity in a cold environment. J. Appl. Physiol. 68:962–966.
Thomas, J.R., S.T.Ahlers, and J.Schrot 1991a Cold-induced impairment of delayed matching in rats. Behav. Neural Biol. 55:19–30.
Thomas, J.R., S.T.Ahlers, and D.Shurtleff 1991b Thermal stress modulates temporal patterns of responding on a multiple DRL-FR schedule. Physiol. Behav. 50:437–442.
Webb, P. 1984 Impaired performance from prolonged mild body cooling. Pp. 391–400 in Underwater Physiology, A.J.Bachrach and M.M.Matzen, eds. Bethesda, Md.: Undersea Medical Society.
Wenk, G.L. 1989 An hypothesis on the role of glucose in the mechanism of action of cognition enhancers. Psychopharmacology 99:431–438.
DISCUSSION
JOHN MILNER: I have a question dealing with multiple exposures to tyrosine. I am concerned that on the basis of Harris Lieberman’s information about the interaction of tyrosine with some drugs, amphetamines in particular, one might, in fact, be looking at some inducible types of enzyme problems, that the response might occur once but it might not occur a second time. Do you have any information on that?
STEPHEN AHLERS: Our studies with tyrosine are replicated several times for each subject, so we do not use one dose of tyrosine; we use several doses two or three times and then use the aggregate data for analysis.
JOHN FERNSTROM: So there is no diminished response?
STEPHEN AHLERS: There is no diminishing of tyrosine’s efficacy when it, in fact, works. I assume Harris Lieberman has found the same thing.
HARRIS LIEBERMAN: We have administered tyrosine repeatedly in the course of studies and have not seen any evidence that there is a diminution in effect. If you are talking about the chronic administration that Steve Ahlers was talking about at the end of his discussion [Chapter 16]—and I believe Carol Greenwood also referred to the possibility of potentiating this type of enzyme—then there may be an issue with chronic administration. Actually, your point had to do more with chronic stress than with chronic administration.
STEPHEN AHLERS: Exactly. A chronically stressed organism is probably a different organism. By the way, those changes were not transient; the tolerance to CRF persisted for 3 to 6 months.
GILBERT LEVEILLE: I have a concern that is somewhat analogous to John Fernstrom’s, and it is really one of safety as we think about using materials such as tyrosine. Most of the paradigms that we have seen are the use of single bolus administrations to look for an effect. But Harris Lieberman mentioned eventually putting tyrosine in rations, and we know how these substances can be readily abused. What do we know about the toxicology of something like tyrosine administered at these dose levels?
STEPHEN AHLERS: I do not think there is much in terms of toxicology. Very rarely do you see an effect of tyrosine by itself, although we saw some effects, but I am not sure what caused those effects.
HARRIS LIEBERMAN: My response is the same as Steve Ahlers. I am not aware of anything like that with tyrosine.
STEPHEN AHLERS: Yes.
CHANDON PRASAD: And also, the cold ambient temperature is a problem. Therefore, the implication would be the release of corticotropin-releasing factor. Have you tried to give the corticotropin-releasing factor antagonist and really prove it?
STEPHEN AHLERS: That is one of our next experiments. That is actually the reason we used CRF to induce the stress response, to get around the potentially confounding differences involved with the administration of physical stressors. You are quite right, the potential of the corticotropin-releasing factor antagonist to block the effects of cold stress is very important. It is our next study.




















