5
Biochemical Strategies for Ration Design: Concerns of Bioavailability
C.Patrick Dunne1
INTRODUCTION
This chapter presents a strategic overview that shows how some modern technologies and biochemistry interact with nutrition and physiology research efforts in the design and development of future ration systems. These ration systems will enhance the performance of troops under stressful battlefield conditions.
Investigators are seeking detailed information to fill the knowledge gaps in the optimum nutrition concept postulated by Walter Mertz, among others (Mertz, 1981). The curve shown in Figure 5–1 is a simplification of a multidimensional response surface. The Committee on Military Nutrition Research has been seeking some definitions of the optimum nutrition plot on the y axis that will measure a response in terms of performance on a military-related task. Most current nutritional standards are historically based on the avoidance of deficiency. Animal nutrition standards may be based on the experimental measurement of growth. Also, one must take with caution the
oversimplification of a curve such as the one shown in Figure 5–1 for a single nutrient, in that it overlooks several types of nutrient-nutrient interactions; the presence or absence of any other nutrient may shift the optimum curve for a single nutrient either to the left or to the right in a three-dimensional surface. An additional complexity and concern is the issue of the fourth dimension of time. Time scales on classic nutrient deficiency studies are, by nature, much longer than the shorter time responses that might be the focus of research on nutritional supplements for performance enhancement. A related issue that should be addressed is the importance of time intervals both for the duration of feeding of any supplements and paired control or deficient diets and the intervals between consumption and testing the performance-related responses (Willcutts et al, 1988).
Military nutrition standards are shown in Table 5–1. These standards provided by the Office of the Surgeon General of the Army, published in AR 40–25 (U.S. Department of the Army, 1985), provide a prescription that must be met by ration developers. The military ration is based on a 3,600-kcal daily
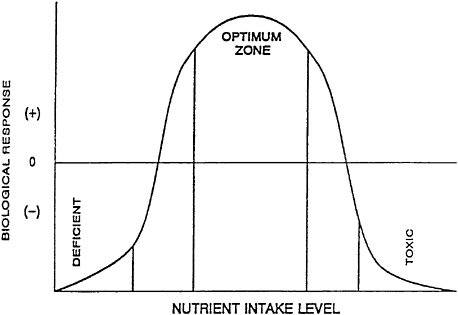
FIGURE 5–1 Optimal nutrition concept. Source: Adapted from Mertz (1981).
TABLE 5–1 Nutritional Standards for Operational Rations
|
Nutrient |
Units |
AR 40–25* |
RDA M-19–22† |
|
Energy |
kcal |
3,600 |
2,900±400 |
|
Protein |
g |
100 |
56 |
|
Carbohydrate |
g |
440 |
- |
|
Fat |
g |
160 |
- |
|
Vitamin A |
µg RE‡ |
1,000 |
1,000 |
|
Vitamin D |
IU |
400 |
300 |
|
Vitamin E |
mg TE§ |
10 |
10 |
|
Ascorbic acid |
mg |
60 |
60 |
|
Thiamin |
mg |
1.8 |
1.5 |
|
Riboflavin |
mg |
2.2 |
1.7 |
|
Niacin |
mg |
24 |
19 |
|
Vitamin B6 |
mg |
2.2 |
2.2 |
|
Folacin |
µg |
400 |
400 |
|
Vitamin B12 |
µg |
3 |
3 |
|
Calcium |
mg |
800 |
800 |
|
Phosphorus |
mg |
800 |
800 |
|
Magnesium |
mg |
400 |
350 |
|
Iron |
mg |
18 |
10 |
|
Zinc |
mg |
15 |
15 |
|
Sodium¶ |
g |
5–7 |
1.1–3.3 |
|
Potassium¶ |
g |
1.88–5.62 |
1.88–5.62 |
|
*U.S. Department of the Army (1985). †RDA, Recommended Dietary Allowances for males, ages 19–22 (NRC 1980). ‡RE, retinol equivalents. §TE, a-tocopherol equivalents. ¶Estimated safe and adequate dietary intakes. |
|||
energy requirement, so many of the nutrient requirements have been increased by a calorie multiplier when compared with Recommended Dietary Allowances of the National Research Council (National Research Council, 1980). The Military Recommended Daily Allowances (MRDA) contain a specified minimum value for carbohydrates (440 g or 48.9 percent of calories) because of the established link between carbohydrate intake and optimal performance in high-demand exercise (Costill, 1988). Investigators are now addressing the issue of which specific types of carbohydrates in what mixtures would be best for selected scenarios (Guezennec et al., 1993).
One other difference in recommended macronutrient composition is the high protein requirement of 100 g for the MRDA. It must be recognized that protein is not only used for growth, replacement of injured tissue, and tissue turnover but also as a key source of the precursors of biogenic amines and many of the other secondary metabolites of the protein-derived amino acids.
Additional nutrients that are of concern to ration developers are listed in Table 5–2. One concern is that, since packaged rations may be used for extended periods of time, the nutrients found at trace levels in a normal mixed diet may not be available at appropriate levels. Another concern, which has also been expressed by Irwin Taub (see Chapter 4), is the possible use of some of the nutrients as metabolic enhancers or to meet stress-induced extra metabolic demands. In particular, some combat rations are high in fat, and individuals may be metabolizing fat at high levels. Heinonen et al. (1992) among others have conducted research on carnitine, which is needed to transport the fatty acids into the mitochondria for oxidation. Zeisel et al. (1991) have summarized the recent studies of choline, which point to the possibility that choline is a required nutrient for humans, especially under conditions of stress. Of the trace elements, Anderson (1989) was especially interested in chromium because of its reported links to carbohydrate metabolism.
A key goal is to identify any special nutritional needs that may be created by stressful situations in combat, that include extremes of heat, cold, or altitude, in addition to physical and mental demands. Investigators would then work at engineering foods or special-purpose food supplements to be used in
TABLE 5–2 Other Important Micronutrients
|
Nutrient |
Safe and Adequate Intake* |
||
|
Biotin |
100–200 µg |
||
|
Choline |
400–900 mg |
||
|
Inositol |
_ |
||
|
Carnitine |
_ |
||
|
Vitamin K |
70–140 µg |
||
|
Pantothenic acid |
4–7 mg |
||
|
Iodine |
150 µg (RDA)† |
||
|
Chloride |
1.7–5.1 |
||
|
Sulfur |
?‡ |
||
|
Chromium |
50–200 µg |
||
|
Manganese |
2.5–5 mg |
||
|
Copper |
2–3 mg |
||
|
Molybdenum |
150–500 µg |
||
|
Selenium |
50–200 µg |
||
|
Fluoride |
1.5–4.0 mg |
||
|
Dietary fiber |
? |
||
|
*National Research Council (1980). †RDA, Recommended Dietary Allowance. ‡A safe and adequate intake of sulfur is assumed if the RDA/MRDA for protein is met. |
|||
testing in conjunction with the Military Nutrition Division of the U.S. Army Research Institute of Environmental Medicine (USARIEM).
The current objectives for the ongoing effort to develop performance-enhancing ration supplements are listed below:
-
To develop fortified liquid or solid ration components that satisfy the special nutritional needs of troops under stress.
-
To provide optimal nutritional support for physiological defense mechanisms against environmental hazards.
-
To seek a definition of the special vitamin and mineral requirements of troops.
-
To test the net available fortifying micronutrients in formulations after processing, storage, and reconstitution for use.
-
To develop a selection procedure for specific protein, peptide, or amino acid supplementation by consideration of precursors of stress-related biogenic amines.
PRACTICAL CONSIDERATIONS FOR RATION DESIGN
In Chapter 4, Irwin Taub provided examples of some of the kinds of structured foods that can be developed and made stable to serve the requirements of military personnel. In any prescription for an optimal nutrient content of rations, it must be recognized that the nutrients must be available after processing, after storage for up to 3 years at 27°C (80°F), to meet military shelf-life requirements, and after any form of reconstitution of the preparation that may be required before use. Some of the technical concerns for ration development are listed in Table 5–3. Certain of those may be of special interest to the Committee on Military Nutrition Research. One key issue is the choice of a carrier for supplemental nutrients. An ideal carrier should maintain the stability of the nutrient, and it should have sensory characteristics that will encourage consumption.
Investigators also need to follow guidelines for fortification that not only match the physical and chemical characteristics of the nutrient with the carrier but also the expectations that a certain food item carries about its nutritional content. For example, it might be best to add caffeine to a cocoa- or coffee-flavored beverage or bar, but not to a fruit bar or drink, where caffeine would not be expected.
The military shelf-life requirements demand that some ration formulations contain food additives as stabilizers or antioxidants. Whenever possible, investigators try to select functional ingredients that would also have a nutritional value. Dunne (1987) developed stable high-fat dairy bars that use a synergistic combination of antioxidants centered on vitamin E (a-tocopherol)
and ascorbyl palmitate. Investigators also select food-grade lecithin, which is commonly used as an emulsifier, for the provision of choline. Purified phosphatidylcholine in liposome vesicles is also being explored as a carrier for labile nutrients.
The bottom line regarding the technical concerns listed in Table 5–3 is taste and acceptability; this is also the bottom line for any food item selected for its special nutritional value. If soldiers do not eat the foods, the foods are not nutritious. As a major design consideration, investigators give attention, in addition to nutrient content, to the nutrient-nutrient interactions that may affect sensory properties (color, flavor, and odor). At least three levels of interactions should be considered: (1) chemical interactions that may occur during processing or storage of rations, (2) interactions during digestion and absorption, and (3) metabolic interactions.
Chemical interactions may be either positive or negative. Examples of negative interactions include the interaction of iron, copper or vitamin B12 with ascorbate. All three of these reactions require air, and they represent the catalysis of oxidation of ascorbate by redox-active metal ions. In the case of vitamin B12 and ascorbate, a mutual destruction has been observed. This destruction is postulated to occur through the formation of active oxygen species [superoxide or hydroxyl radicals] involving the participation of both the CO+2 and CO+3 states of vitamin B12 (Gossamer et al., 1977). Although this destructive interaction caused some concern about negative effects of megadoses of ascorbate supplements, the physiological organometallic coenzyme forms of vitamin B12 are quite unlikely to participate in such destructive redox cycles
TABLE 5–3 Technical Concerns for Ration Development
|
Liquid versus Solids—stability differences, sterilization required |
|
Long-term storage requirements |
|
Minerals and electrolytes—choice of salts |
|
Vitamins—choice of form |
|
Requirements for additives |
|
Stabilizers-antioxidants |
|
pH control buffers and acidulents for liquids |
|
Packaging the product to maximize stability |
|
Taste and acceptability—as a function of temperature |
(Dunne, 1971). Another possible destructive interaction is the riboflavin-catalyzed photooxidation of unsaturated lipids. Chemical interactions may also be positive, as in the antioxidant couple, whereby ascorbyl palmitate may serve to regenerate the active form of vitamin E (a-tocopherol) (Cort, 1974).
There are examples of both positive and negative interactions during digestion and absorption that can be used to guide the formulation of nutrient-rich products (Roe, 1991). Negative interactions that retard absorption include the complexing of certain minerals (iron or zinc) with inositol phosphates (phytate), producing insoluble, less available forms or the complexing of calcium or magnesium with the fatty acids produced by digestion of fats, forming insoluble soaps. Examples of positive interactions that may enhance absorption include the interaction of ascorbate with iron and sugars with calcium.
Food technologists may avoid some of the negative chemical interactions by use of encapsulation techniques to shield one or more of the potential reactants. For example, either metal salts of iron or zinc can be purchased as encapsulated forms, and so can ascorbate. The encapsulation agents can be chosen to be released or removed during digestion, so that the positive effects of ascorbate on iron intake can be derived without the risk of loss of nutrient potential during storage. Some of the types of encapsulation are shown in Figure 5–2.
After digestion and absorption there may be continued metabolic interactions involving dietary nutrients that should be considered in the design of supplements (Bodwell and Erdman, 1988). Again, both positive and negative metabolic interactions have been observed. For example, the branched-chain amino acids, being large neutral amino acids, compete with the aromatic amino acids tyrosine, tryptophan, or phenylalanine for the transport system involved in the uptake of these amino acids across the capillaries into the brain. The negative folate-vitamin B12 interaction is a subtle and pernicious one, whereby folate masks some of the symptoms of vitamin B12 deficiency, but the neurological damage caused by the hidden vitamin B12 deficiency is the reason why folate is not allowed as a single nutrient supplement.
This case of vitamin B12 and folate may be just one example of the trouble that can be caused by single-nutrient supplements. A single nutrient, especially when given at high doses, does have the potential to perturb metabolic pathways such that the metabolism of other nutrients may be negatively affected. For a brief summary of single-nutrient problems, see the paper by Bendich (1992).
There are positive higher metabolic interactions that should also be considered. Many of these involve product-precursor relationships or the sharing of a common pool of chemical functional groups, such as choline and methionine, which are methyl group donors and part of the key one-carbon
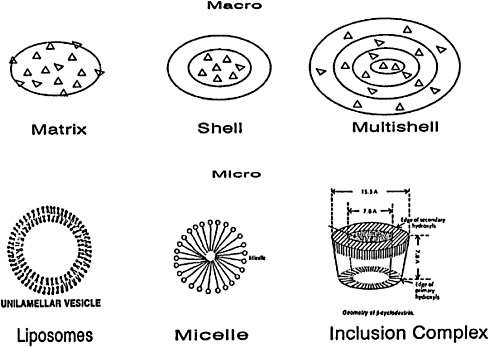
FIGURE 5–2 Encapsulation types. Source: Information on the sizes and structures of macro forms is adapted is from Risch and Reineccius (1988). Information on micro forms is from Brooks and McManus (1990) for liposomes, used with permission; and adapted from Yesair (1989) for micelles; and information for cyclodextrins is from Beesley (1985), used with permission.
metabolism scheme that animal nutritionists, especially those in the poultry industry, have known for some time. These two nutrients are listed among the lipotropic factors in older textbooks. Vitamin E and vitamin A have been shown to interact positively both chemically and metabolically (Machlin and Langseth, 1988). Certain product-precursor relationships are of interest because they involve nutrients that are topics of other chapters in this volume. These nutrients with positive metabolic interactions include phenylalanine and tyrosine, tryptophan and niacin, and lysine and carnitine. A much more complete discussion of nutrient interaction is found in the monograph edited by Bodwell and Erdman (1988). One interesting question is whether all of the aspartame consumed in both commercial and military products can lead to increased phenylalanine- or tyrosine-derived neuroendocrine factors.
BIOCHEMICAL STRATEGIES FOR CONTROLLED DELIVERY OF NUTRIENTS
Investigators at the U.S. Army Natick Research, Development and Engineering Center are exploring some strategies for what can be termed targeted nutrient delivery. It is akin to approaches taken by a pharmacokineticist in the design of an oral delivery system for a new drug. Techniques of sustained or controlled release may be explored, but first, one must consider a whole host of barriers or reactants that nutrients and their carriers must traverse on their way through the digestive tract. Figure 5–3 is provided as a guide to the stresses and membrane barriers that exist during enteral nutrition. During normal oral delivery of nutrients, few nutrients may pass directly into the bloodstream through oral cavity membranes. However, the buccal route is exploited for direct delivery of some active water-soluble pharmaceutical agents, including enzymes, which can be absorbed by the body by holding tablets under the tongue, thereby allowing the agents to gain entry to the bloodstream without the delay resulting from the passage of the agent through the digestive tract and thus bypassing a potential first-pass metabolism by the liver through the portal circulation. Some digestion may be initiated in the oral cavity by the action of amylase and a lingual lipase that may continue its action downstream in the digestive tract. In the stomach there is a pH shift plus the addition of the protease pepsin, which is active at acid pH. Again, membrane barriers are significant, so not much leaks out into the bloodstream; one exception, however, is alcohol. As nutrients and carriers enter the small intestine, the medium changes drastically, whereby, along with carbonate to neutralize acid, the pancreatic enzymes and the bile salts enter the system. The bile salts prove to be important at changing the structures of native dietary fats
and also of fats or phosphatidylcholine vesicles (liposomes) that might be used as encapsulates for nutrients. In the intestinal mucosal cell there is more or less a division point where the majority of water-soluble nutrients, either by diffusion or facilitated transport, enter the portal circulation system to go to the liver, which is metabolically active in the transformation of many nutrients into final circulating forms. The portal route may take from 1 to 4 h to show evidence of nutrient intake by an increase in the general blood concentration of those nutrients; a commonly studied indicator is the blood glucose level, which peaks about 2 h after the intake of common carbohydrates, including simple sugars. An alternative route is taken by lipids, including long-chain fatty acids. They enter the lymphatic system and are later deposited in the general circulation via the thoracic duct; this pathway is slower, taking 2 to 7 h, but the nutrients are able to avoid first-pass liver metabolism.
One key area of concern is the uptake and subsequent metabolism of amino acids and protein. When considering oral delivery routes for amino acids that
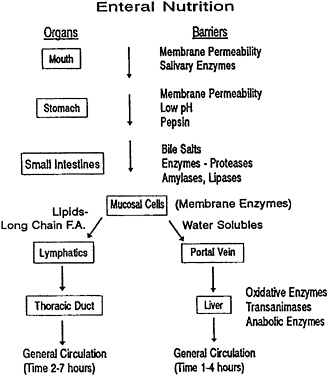
FIGURE 5–3 Diagram indicating the barriers to nutritional delivery where F.A.=fatty acids. SOURCE: Adapted from Briggs and Calloway (1984).
are to serve as biotransmitter precursors, one should consider two sometimes overlooked facts: (1) that certain amino acids, especially the aromatic ones, are classic inducers of catabolic liver enzymes (Ip and Harper, 1973), and (2) that, in some cases, uptake of peptides may be facilitated compared with uptake of single amino acids (Siemensma et al., 1993). A hypothesis can be constructed that, with certain amino acids such as tyrosine and tryptophan, repeated or chronic supplementation of the free amino acids may induce the catabolic liver enzymes, such as tryptophan pyrrolase to such an extent that more precursor amino acid will be oxidized or otherwise changed and less will get to the target tissues, such as the adrenal glands or the brain, to be used for biogenic amine synthetic pathways.
One might then consider alternative strategies that could be used to minimize the effects of diversionary metabolic pathways on those amino acids that are intended to serve as neurotransmitter precursors. One strategy is to restrict the doses to large single bolus forms, such that liver enzyme induction is not a factor, or a candy drop (i.e., pill) that is held under the tongue can be used to enhance buccal uptake. In either of these cases, one may face the issue of deciding whether the delivery is that of a drug or a food; John Vanderveen addresses this issue in Chapter 23. Both of these types of delivery seem limited both by mass and by permeability factors, so they might be best for potential performance-enhancing nutrients that are effective in relatively small quantities (milligrams rather than grams) and that are fairly water soluble.
To provide protection to nutrients either during processing and storage or during the passage through the digestive system, a variety of techniques are available for encapsulating nutrients. The top of Figure 5–2 shows examples of larger microencapsulation products that are found in commercial food, fragrance, or pharmaceutical products (Risch and Reineccius, 1988); these range in size from 2 µm to 1 mm. Only some are true shell-type encapsulated products, such as in the dry powder forms of fat-soluble vitamins. The shell might be made of gelatin or an acacia gum, which can protect fat-soluble vitamin A or D from damage during processing or storage. These shells would resist organic solvents, but they would release their contents in aqueous environments. Mulitshell encapsulants may offer added protection or offer the possibility of a controlled or an extended duration of release of the contents. Many of the commercial encapsulated micronutrients use a spray-drying process to produce matrix “encapsulated” products in which the active nutrient is embedded in a matrix made of starch, for example, but is not truly isolated from the environment.
The bottom of Figure 5–2 shows smaller encapsulated forms, sometimes called nanocapsules, which may range in size from 20 nm to 200 mm. Inclusion complexes of the cyclodextrins are included in Figure 5–2 because of their potential to trap and carry small, volatile molecules (Bender and
Komiyama, 1978), but they are not yet approved for use as a food in the United States. Micelles and derivatives of micelles are starting to receive consideration as carriers to exploit the alternative lymphatic route to the bloodstream (Yesair, 1989); these may be as small as 5 to 10 nm in diameter. The liposome shown in Figure 5–2 is a simple unilamellar vesicle, perhaps 50 to 200 nm in diameter, that can be made from phosphatidylcholine or related polar lipids (Deamer and Uster, 1983). Larger multilamellar liposomes are also commonly used as potential microencapsulants or carriers; these onion-like structures may form spontaneously (Brooks and McManus, 1990). Investigators at the U.S. Army Natick Research, Development and Engineering Center are finding that they can get a stable vesicle by sonicating the mulitilamellar vesicles and waiting for subsequent thermal fusion or curing of the resulting smaller unilamellar vesicles. Whitburn and Dunne (1991) have been able to stabilize the vesicles to some degree against freeze-drying, providing the possibility of creating stable dry dosage nanocapsules.
To date investigators have not found that liposomes are true magic bullets for the oral delivery of high-impact nutrients. The location of the carried cargo nutrient, the amount of cargo that can be carried, and the stability of vesicles depend on the nature of the cargo, the nature of the polar lipids forming the vesicle, and the preparation procedures. Investigators have not had much success encapsulating caffeine, for instance. Investigators have been assessing the effects on loading efficiencies and cargo leakage as a function of (1) the presence of membrane additives (e.g., cholesterol), (2) bilayer phospholipid chain length, and (3) medium additives. There is still much to learn about matching the carried nutrient to the encapsulating agent to achieve a true targeted and controlled nutrient delivery system. The information presented in this volume should help investigators focus their efforts on the most promising nutrients that could be enhanced by a controlled or sustained delivery system.
CONCLUSIONS AND RECOMMENDATIONS
Not all of the strategy described here is a nutritional “Star Wars” concept, in that many of the nutrients or food ingredients considered to have potential performance-enhancing capabilities are available as food-grade materials. Investigators have been using a strategy of the dual-purpose use of such ingredients as shown in Table 5–4, which lists examples of substances with both nutritional function and special functionalities in the food technology sense. The strategy is to emphasize the proper form of the ingredients that will best fill their functional and nutritional roles. A simple example is a-tocopherol acetate, which is present in many multivitamin preparations. When used in this form, the product is not protected by the chemical antioxidant function of free a-tocopherol.
TABLE 5–4 Dual-Function Ingredients for Ration Bars
|
Ingredient |
Function |
Nutritional Role |
|
a-tocopherol |
Antioxidant |
Vitamin E |
|
Ascorbyl palmitate |
Antioxidant |
Vitamin C |
|
Lecithin |
Surfactant and emulsifier |
Choline source |
|
Pectin |
Binder and texturizer |
Soluble dietary fiber |
|
Whey protein |
Binder |
Essential amino acids |
|
Bran |
Matrix Structure |
Insoluble dietary fiber |
|
SOURCE: Dunne (1987), used with permission. |
||
Investigators have developed several criteria for the optimal fortification of supplementation that may well be applied to any performance-enhancing supplements. These criteria are listed below:
-
The supplementation regimen should be based on knowledge of the nutrient content of an individual’s basic diet. This knowledge may be simpler for combat rations than for normal diets, but many advances in analytic techniques may be required.
-
Supplements should be in a form that is acceptable and consumable by troops under battlefield conditions. Carriers must be highly preferred items.
-
Fortified items should be designed to maximize nutrient retention in the form that they are consumed by users. Both storage stability and nutrient stability when reconstituted for use need to be optimized.
-
The interactive effects among different components must be considered and controlled. Reactive micronutrients will be separated by encapsulation or by use of separate carriers.
-
The levels and specific forms of individual fortifying nutrients should be chosen to give maximum bioavailability, with little or no chance of negative effects from either under- or oversupplementation.
-
Supplementation should be based on knowledge of the metabolic roles of the supplement’s components and the basic dietary components.
Finally, four major concerns related to the issues listed above set the stage for later issues. These are:
-
Where do certain potential performance-enhancing supplements lie in the gray zone between food and drugs?
-
What will be the acceptance by users, and how can it be maximized?
-
What real tests of efficacy are available to establish the positive effects of any supplement on mental and/or physical performance?
-
How do investigators establish and use proper time frames for both usage and testing of supplements?
A related issue linking the first and last questions is to consider the differences between a bolus dose of a single amino acid, which might well be considered a drug, and graduated regular doses over a longer term of that amino acid in peptide forms, which is more akin to a food.
A bit of history outlined in Table 5–5 may put the task in perspective. In the prime period of Army human nutrition research Consolazio and colleagues established a time course for the onset of nutrient deficiency syndromes (Consolazio, 1983). From the data there seems to be a hierarchy of effects, with fatigue being a major and leading effect. However, one must note that with the exception of water, the time course for the first signs of deficiency is in days.
The present task is to come back 20 years later to determine whether it is possible to observe the effects in hours, not days, by modulating intakes of specific nutrients under stressful conditions in which subtle differences in performance can be detected. There may be improved technology for the delivery of nutrients, but investigators must still deal with the major issues of evaluating the effects of nutrients on performance.
TABLE 5–5 Time Course for Onset of Nutrient Deficiency Effects
|
Nutrient |
Time for Deprivation Effect |
Primary Symptom of Deprivation |
|
Water |
A few hours |
Fatigue, mental confusion |
|
Total energy |
2–3 days |
Fatigue |
|
Electrolytes (Na+CL-) |
=3 days |
Fatigue, muscle cramps |
|
Carbohydrate |
=3 days |
Fatigue |
|
B Vitamins |
1–2 weeks |
Fatigue, neurological effects |
|
Vitamin C |
Several weeks |
Fatigue |
|
Protein |
Several weeks |
Varies |
|
Vitamin A |
Several months |
Uncertain |
|
Fats |
Many months |
Varies; essential fatty acid deficiency |
|
Trace Minerals |
Several months |
Anemias or blood cell functions |
|
SOURCE: Consolazio (1983), used with permission. |
||
REFERENCES
Anderson, R.A. 1989 Essentiality of chromium in humans. Pp. 75–81 in The Science of the Total Environment. Amsterdam: Elsevier Sciences Publishers.
Army Regulation 40–25 1985 See U.S. Departments of the Army, the Navy, and the Air Force.
Beesley, T.E. 1985 Inclusion Complexing: A new Basis for HPLC Selectivity. American Laboratory 17(5):78–87.
Bender, M.L., and M.Komiyama 1978 Cyclodextrin Chemistry. Berlin: Springer-Verlag.
Bendich, A. 1992 Safety Issues Regarding the Use of Vitamin Supplements. Ann. N.Y. Acad. Sci. 669:300–310.
Bodwell, C.E., and J.W.Erdman, Jr. Eds. 1988 P. 389 in Nutrient Interactions. New York: Marcel Dekker, Inc.
Brooks, G.J., and R.C.McManus 1990 Finding new uses for liposomes in cosmetics. INFORM (Am. Oil Chem. Soc.) 1:891–895.
Consolazio, C.F. 1983 Recommended dietary allowances. Prog. Food Nutr. Sci. Nutr. Performance 7:13–27.
Cort, W.M. 1974 Antioxidant activity of tocopherols, ascorbyl palmitate, and ascorbic acid and their mode of action. J. Am. Oil Chem. Soc. 51:321–325.
Costill, D.L. 1988 Carbohydrates for exercise: Dietary demands for optimal performance. Int. J. Sports Med. 9:1–18.
Deamer, D.W., and P.S.Uster 1983 Pp. 27–51 in Liposome Preparation: Methods and Mechanisms, in Liposomes, M.J.Ostro, Ed. New York: Marcel Dekker.
Dunne, C.P. 1971 Reactions of the Carbon-Cobalt Bond of Alkyl Vitamin B12 Derivatives. Ph.D. Thesis. Brandeis University, Waltham, Mass.
1987 Fundamental research and engineered caloric dense rations-introduction: Some nutritional perspectives. Activities Report of the R&D Associates 39:145–150.
Gossamer, A., B.Grüning, L.Ernst, W.Becker, and W.S.Sheldrick 1977 Reaction of dicyanocobyrinic heptamethyl ester with ascorbic acid. Angew. Chem. Int. Ed. Engl. 16:481–482.
Guezennec, C.Y., P.Satabin, F.Duforez, J.Koziet, and J.M.Antoine 1993 The role of type and structure of complex carbohydrates response to physical exercise. Int. J. Sports Med. 14:224–231.
Heinonen, O.J., J.Takala, and M.H.Kvist 1992 Effect of carnitine loading on long-chain fatty acid oxidation, maximal exercise capacity and nitrogen balance. Eur. J. Appl. Physiol. 65:13–17.
Ip, C.C.Y., and A.E.Harper 1973 Effects of dietary protein content and glucagon administration on tyrosine metabolism and tyrosine toxicity in the rat. J. Nutr. 103:1594–1607.
Machlin, L.J., and L.Langseth 1988 Pp. 287–311 in Vitamin-Vitamin Interactions in Nutrient Interactions, C.E.Bodwell and J.W.Erdman, Jr., eds. New York: Marcel Dekker.
Mertz, W. 1981 The essential trace elements. Science 213:1332–1338.
National Research Council 1980 Recommended Dietary Allowances, 9th Ed. Washington, D.C.: National Academy of Sciences Press.
Risch, S.J., and G.A.Reineccius 1988 Flavor Encapsulation. ASC Symposium Series 370. Washington, D.C.:American Chemical Society.
Roe, D.A. 1991 Pp. 459–471 in Bioavailability of Nutrients Added to Human Foods in Nutrient Additions to Food—Nutritional Technological and Regulatory Aspects, J.C.Bayernfeind and P.A.Lachance, eds. Trumbell, Conn.: Food and Nutrition Press, Inc.
Siemensma, A.D., W.J.Weijer, and H.J.Bak 1993 The importance of peptide lengths in hypoallergenic infant formulae. Trends Food Sci. Technol. 4:16–21.
U.S. Departments of the Army, the Navy, and the Air Force 1985 Army Regulation 40–25/Naval Command Medical Instruction 10110.1/Air Force Regulation 160–95. Nutrition Allowances, Standards, and Education. May 15. Washington, D.C.
Willcutts, K.F., A.R.Wilcox, and K.K.Grunewald 1988 Energy metabolism during exercise at different time intervals following a meal. Int. J. Sports Med. 9:240–243.
Whitburn, K.D., and C.P.Dunne 1991 Pp. 161–181 in Evaluation of Stabilizers for Synthetic Vesicles and Milk Fat Globules Under Drying Stress in Microemulsions and Emulsions in Foods. M.El-Nokaly and D. Cornell, eds. ACS Symposium Series 448. Washington, D.C.: American Chemical Society.
Yesair, D.W. 1989 Composition for Delivery of Orally Administered Drugs and Other Substances. U.S. Patent No. 4,874,795. October 17, 1989;
Zeisel, S.H., K.A.DaCosta, P.D.Franklin, E.A.Alexander, J.T.Lament, N.F.Sheard, and A.Beiser 1991 Choline, an essential nutrient for humans. FASEB. 5:2093–2098.
DISCUSSION
AUDIENCE MEMBER: Dr. Dunne, you made the distinction between taking a bolus as opposed to taking an encapsulated form and putting it into the food, making the encapsulated form a food. Those of us who work in this field need guidelines as to what the U.S. Food and Drug Administration will accept and how we should proceed in trying to formulate this complex.
PATRICK DUNNE: Our strategy is that if we are going to put any nutrient in rations on a regular basis, it must be an additive approved for use in foods. So levels of use would be set by certain parameters, whether it is generally recognized as safe (GRAS) or as an approved food additive. There is an upper value of how much you would be able to take in on a daily basis if it is a
food. So we bring in that regulatory response and we would do it on a regular basis. With the bolus, however, I would say that first, your level of use might be different for a drug dose, and the timing factors would be different.
RICHARD WURTMAN: The trouble is, amino acids are not GRAS. In addition to that, compounds that are GRAS are GRAS for specific functionalities. The effects of amino acids on performance and behavior do not qualify.
PATRICK DUNNE: My own feeling is we would get somewhere into a middle ground if we focused on peptides or protein hydrolysates that happen to have the right enrichment in the amino acids that we are more interested in.
RICHARD WURTMAN: Then, of course, you get into the problem that the efficacy of a compound like tyrosine will be specifically impaired if it is presented along with proteins unless you make a peptide that has tyrosine plus nonessential amino acids, and that gets very expensive.
PATRICK DUNNE: The question of making versus selecting a natural peptide is where we might also have a distinction. We would prefer not to have synthetic peptides, but there are ways with specific receptor technology to enrich for tyrosine, for instance, in a casein hydrolysate.


















