2
Understanding HIV Transmission
This chapter presents an overview of knowledge about the transmission of HIV, focusing on the sexual and substance-using behaviors that have been most implicated in the spread of the disease. Although the general categories of risk behavior are well known, the specific acts responsible for HIV transmission—use of injection drugs and sexual practices, for example—have not been adequately researched or discussed publicly because of their sensitive nature. Yet, in order to develop effective prevention and treatment strategies, it is crucial to understand the specific behaviors involved. A review of knowledge gained to date from biological, epidemiological, psychological, and social research on HIV transmission—much of it supported by NIAAA, NIDA, and NIMH—has helped to elucidate relevant behaviors. However, much remains to be learned. In particular, the prevalence and spread of those behaviors within certain communities is unknown.
SEXUAL TRANSMISSION
In 1992, the World Health Organization estimated that as many as 10 million people around the globe were infected with HIV, and that 40 million would be infected by the end of the century (Eckholm, 1992). Although this estimate is indefinite (for reasons described below), it reflects the general consensus that in the absence
of effective intervention, the epidemic will spread vastly and rapidly, primarily through sexual transmission.
As with any sexually transmitted disease (STD), transmission of HIV is highly dependent on the number of sex partners one has and the specific types of sexual practices in which one engages. Sexual intercourse has been and continues to be a very important mechanism of HIV transmission. In particular, anal intercourse carries the highest risk for HIV acquisition, for both receptive and insertive partners. Penile-vaginal intercourse places individuals at next highest risk for HIV transmission, and is followed by oralgenital intercourse. These sexual behaviors can place anyone at risk for HIV, regardless of his or her sexual orientation or gender.
In major studies of prevalent HIV infection among gay men, receptive anal intercourse has been the primary mode of transmission when researchers controlled for other risk factors, including number of sex partners (Winkelstein et al., 1987). Similarly, receptive anal intercourse has been shown to be the primary mode of transmission of HIV among gay men in cohort studies in which seroconversion has been analyzed during follow-up (Kingsley et al., 1987; Winkelstein et al., 1989).
Among people who engage in anal intercourse, a series of cofactors for HIV infection affect transmission. Cofactors include any physical conditions, behavioral practices, or microbiological agents that facilitate the transmission of HIV. Two major categories of cofactors have been investigated: those that might affect acquisition of HIV before or during sexual contact (such as foreplay and other ancillary sexual practices) and those, of a more general nature, that might enhance susceptibility. Cofactors include two major types of ancillary sexual practices: those that are likely to disrupt sensitive tissues in the rectum, thereby facilitating infection, and those that interfere with judgment, thereby rendering sex partners less likely to take precautions against acquiring infection. Under the first group of sexual practices that place individuals at risk are anal intercourse itself, rectal douching, and ''fisting." The tissues inside the rectum are highly delicate, and minor tearing of these sensitive membranes is possible during any form of anal intercourse. Even when lubricants are used, vigorous intercourse can cause rectal trauma. Moreover, it is not uncommon for people to insert fingers and dildos into the rectum during sexual activity, which contributes to the potential for trauma. Data from various longitudinal studies have documented that enemas and douching before sexual contact increase the risk of HIV infection during receptive anal intercourse (Kingsley et al., 1987;
Polk et al., 1987). The findings of a large-scale study of risk factors for STD infection in gay men attending five public health clinics for STD treatment were consistent with observations of an association between douching and hepatitis B virus (HBV) infection (Schreeder et al., 1980). Specifically, men who practiced rectal douching in association with receptive anal-genital intercourse were found to be at greater risk for HBV infection. One can logically speculate that if this practice provides both a source of HBV-infectious blood and a point of introduction for HBV infection, the same would be the case for HIV infection. Finally, likelihood for rectal injury exists with "fisting," anal penetration by the fingers or hand into the rectum and possibly into the sigmoid colon (Browning, 1993). As with rectal douching, receptive fisting has been found to be significantly related to HBV infection (Schreeder et al., 1980).
Under the second group of ancillary sexual practices, those that interfere with judgment, are the use of psychoactive drugs such as nitrite inhalants. The nitrites—principally amyl and butyl nitrite—are liquid compounds that were first introduced into medicine more than a century ago for the treatment of angina. They have had other therapeutic uses, as muscle relaxants and vasodilators. The primary effect of these drugs is the relaxation of all smooth muscles in the body, including those in the blood vessels, thus allowing a greater flow of oxygenated blood to the brain (Nickerson, 1975). Amyl and butyl nitrite are quick-acting drugs, taking effect in 15 to 30 seconds, with a duration of two to three minutes. On inhalation, there is a distinct "rush." The recreational popularity of the nitrites results from their reputation as aphrodisiacs (Louria, 1970; Seymour and Smith, 1987). Nitrites—and their association with the sexual activities of homosexual men—have received a great deal of attention, especially with respect to their potential role as a cofactor in Kaposi's sarcoma (Archibald et al., 1992; Haverkos, 1990; Ross and Drew, 1991). However, alcohol and other mood-altering drugs popular among a broader range of people may be just as likely to influence high-risk sexual behaviors, although the evidence of this is mixed (Bolton et al., 1992; Leigh and Stall, 1993).
The biological variables that determine HIV infectivity (the tendency to spread from host to host) and susceptibility (the tendency for a host to become infected) are incompletely understood. HIV has been isolated from the semen of infected men, and it appears that it may be harbored in the cells of pre-ejaculate fluids or sequestered in inflammatory lesions (Fischl et al., 1987). Furthermore,
there is evidence that women can harbor HIV in vaginal and cervical secretions in varying concentrations at different phases of the menstrual cycle (Vogt et al., 1986, 1987; Wofsy et al., 1986). Also, there are a number of concurrent or prior infections that represent potential cofactors, particularly those infections associated with sexually transmitted diseases, such as syphilis, gonorrhea, anal and genital warts, and herpes. These cofactors may operate to enhance HIV susceptibility during either anal, vaginal, or oral sex.
A number of studies have suggested that cofactors affect the likelihood of vaginal (heterosexual) transmission of HIV. For example, in one study, despite repeated sexual exposure, an average of only 15 percent of women who were steady partners of HIV-infected men acquired the infection (European Study Group, 1989; Johnson, 1988). These and other studies suggest that the risk of contracting HIV through a single sexual exposure is not particularly high in the great majority of instances (Hearst and Stephen, 1988; Holmberg et al., 1989). However, there is evidence that, although some people remain uninfected after hundreds of episodes of unprotected sex, others have become infected after only one or just a few sexual encounters (Padian, Wiley, and Glass, 1988).
Three groups of factors influencing the probability of acquiring HIV through vaginal contact have been suggested: (1) sexual behavior and risk duration; (2) infectiousness of the HIV-positive partner; and (3) host susceptibility (Johnson and Laga, 1988). Surprisingly, a majority of early studies of heterosexual transmission failed to show a relationship between risk of infection and either the frequency of sexual intercourse or the duration of a relationship with an infected partner (Goedert et al., 1988; Johnson, Petherick, Davidson, et al., 1989; Laga et al., 1988; Peterman et al., 1988). These results were counterintuitive, and may have been the result of measurement error, improper statistical analysis, and the failure to account for other factors. More recent investigations have found that the number of exposures to an infected partner is indeed associated with transmission. Important findings in this regard came from the California Partners' Study, an investigation that surveyed the opposite-sex partners of individuals infected with HIV or diagnosed with AIDS (Padian et al., 1990). Of 150 female partners of infected men recruited for the study during the second half of the 1980s, 48 percent were partners of bisexual men, 15 percent were partners of injection drug users, 23 percent were partners of hemophiliacs, 5 percent were partners of men infected
through contaminated blood transfusions, 5 percent were partners of men in multiple risk groups, and 4 percent were partners of men in unidentified risk groups. Of the 85 women who had 0 to 200 sexual contacts with their infected partners, 13 percent were HIV positive; of the 65 women who had 201 to 2,170 sexual contacts with their infected partners, 37 percent were HIV positive.
With regard to the infectiousness of the seropositive partner, a higher rate of infection among the female partners of men in advanced clinical stages of HIV disease has been well documented (Laga et al., 1989). This is probably due to the fact that declining immune function enhances infectivity in an HIV-positive individual, and as such, bodily fluids such as blood and semen contain higher concentrations of HIV. One factor in this regard is a history of sexually transmitted diseases (CDC, 1987).
One of the more comprehensive studies of heterosexual transmission to examine the full range of risk behaviors was conducted by the Italian Study Group on HIV Heterosexual Transmission (Lazzarin et al., 1991). The sample included 368 women whose only potential exposure to HIV was having a male sex partner who was HIV positive, and 27.7 percent of these women were seropositive. The findings of the study documented risk duration, type and frequency of sexual intercourse, the man's infectiousness, and the woman's susceptibility as the key factors. With respect to risk duration, for example, women having a relationship with an infected male for 1 to 5 years had the highest prevalence of seropositivity, and a frequency of sexual intercourse more than twice a week was associated with a twofold increased risk of infection. Seropositive women reported anal intercourse twice as frequently as seronegative women, but condom use had a clear association with reduced rates of heterosexual transmission. In addition, infected women were more frequently those whose partners' disease had progressed to AIDS. Finally, seropositive women also reported histories of syphilis, genital warts, or genital herpes more so than seronegative women.
All of these studies document that, although the transmission of HIV from an infected male to a female partner is not always certain with any amount of exposure, considerable risk is always present. Such risk can increase dramatically depending on the type and frequency of the sexual contacts and the immunological state of the partners.
Female-to-male sexual transmission of HIV is supported by biological plausibility, equal numbers of male and female AIDS cases in African and Asian countries, case reports of males with no risk
factors other than heterosexual intercourse, and seroconversion of male sex partners of infected women that occurred while the couples were being studied prospectively. With respect to the biological plausibility of female-to-male transmission, it has been argued that since other sexually transmitted diseases are bidirectional in nature, it is not unreasonable to assume that HIV can spread in the same manner. A number of studies have documented that African and Indian men who have multiple female sex partners or sexual contact with commercial sex workers are at highrisk for becoming infected with HIV (Cameron, Plummer, and Simonsen, 1987; Carswell, Lloyd, and Howells, 1989; Clumeck et al., 1985; Kreiss, Koech, and Plummer, 1986). The most persuasive case reports of female-to-male transmission have been those in which (1) the female acquired the infection from a transfusion or organ transplant and her male partner (without other known risk factors) subsequently seroconverted (L'Age-Stehr, Schwarz, and Offermann, 1985) and (2) a sequential chain of male-to-female-to-male transmission was observed (Calabrese and Gopalakrishna, 1986).
Although significant numbers of female-to-male infections have been documented in Africa (Barnett and Blaikie, 1992; Panos Institute, 1988; Shannon, Pyle, and Bashshur, 1991), such a mode of transmission has been reported only infrequently in the United States, and the majority of the more recent case reports have come from investigators in Europe (Beck et al., 1989; European Study Group, 1992; Johnson, Petherick, Davidson, et al., 1989; Lefrere et al., 1988). Several explanations have been offered for the differences in female-to-male transmission rates between the United States and certain countries in Africa. A number of researchers have suggested that the documentation of infrequent heterosexual transmission from women to men in this country may be a function of the history of the epidemic, since the initial phase was largely confined to men who have sex with men and injection drug users, so that during that time the number of infected women was low, and the possibility of female-to-male transmission was small. Because the majority of AIDS cases occurring today reflect infections that were acquired during the early years of the epidemic, most heterosexually acquired infections among men may still be in the asymptomatic or latent stage (Friedland and Klein, 1987; Osmond, 1990). Also, the fact that the infectivity of an HIV carrier increases over time may magnify the effects alluded to above. Following this line of reasoning, it has been argued that virus concentration in genital secretions may also increase over
the course of the infection (Burke and Redfield, 1988). Researchers from NIDA recently have argued that the relative frequency of female-to-male transmission has been underestimated (primarily as a result of the way that cases are classified), suggesting that it represents a more significant public health concern than is generally believed (Haverkos and Battjes, 1992).
In sum, although many highrisk practices and cofactors have been observed most predominantly among men who have sex with men, they are not limited to that population. Nor are they the only behaviors linked to sexual risk. As the AIDS epidemic spreads more widely among women and heterosexual men, a broader range of sexual practices must be considered for their potential in transmitting HIV, including the possibility of female-to-female sexual transmission, which has been notably understudied. Although lesbians and bisexual women have been considered to be a low-risk group for contracting and transmitting HIV, the fact that women can transmit STDs such as chlamydia, herpes, and genital warts to one another makes it likely that they could similarly transmit HIV. There is some evidence that among injection drug users, women who have sex with women are at particular risk for HIV (Friedman, Des Jarlais, Deren, et al., 1992; Jose et al., 1993; Reardon et al., 1992).
HIV RISK AND INJECTION DRUG USE
The sharing of hypodermic needles, syringes, and other injection paraphernalia is the most likely route of HIV transmission among intravenous and other injection drug users. When the blood of the previous user is lodged in the needle, the syringe, or some other part of the works (drug paraphernalia), that blood serves as the vector for transmission. Levels of risk may vary depending on the particular injection practice, but this has not been well studied. One practice is skin-popping—the subcutaneous or intramuscular injection of cocaine, narcotics, and other drugs. Skin-popping (or simply popping ) is a common method of heroin use by experimenters and novice and casual users who mistakenly believe that addiction cannot occur through this route (Baden, 1975; Kaplan, 1983). Skin-popping also is done by long-term injectors who can no longer find veins into which they can inject.
Another injection method is booting, also known as kicking, a process that uses a syringe to draw blood from the user's arm, mixes the drawn blood with the drug already taken into the syringe, and injects the blood-drug mixture into the vein. Booting
leaves traces of blood in the needle and syringe, thus placing subsequent users of the injection equipment at risk (Inciardi, 1990a). There are three reasons for booting. First, most injectors draw blood into the syringe for the sake of vein registration, that is, to ensure that the needle is properly placed in the vein. Second, many injectors draw large amounts of blood into the syringe, pumping it in and out several times to mix the blood with the drug solution, believing that this practice potentiates a drug's effects. Third, many users wish to test the strength or effect of the drug before injecting the entire amount (Greenfield, Bigelow, and Brooner, 1992).
Jacking is another technique used by drug injectors. It is a practice more common to cocaine injectors than users of other injection drugs. Jacking is staged shooting, in which the user injects a portion of the dissolved cocaine, pulls blood back into the syringe, and waits for the rush to subside. The user then repeats this process with a larger amount of the drug until all of the drug is mixed with blood and injected and no cocaine is left in the syringe (Ouellet et al., 1991).
Although most injection drug users are generally aware of the risks associated with booting, jacking, and needle sharing, they may be less aware of risks associated with other aspects of the injection process. Shared water used in the injection process represents a potential reservoir for disease. Virological studies have indicated that HIV can survive in ordinary tap water for extended periods of time. In a series of experiments conducted at the Laboratory of Tumor Cell Biology at NIH, infectious cell-free virus was recovered from dried material after up to three days at room temperature, and in an aqueous environment virus survived longer than 15 days at room temperature (Resnick et al., 1986). Similar results were found in complementary studies conducted at the Institute Pasteur in Paris (Barre-Sinoussi, Nugeyre, and Chermann, 1985; Martin, McDougal, and Lsokoski, 1985).
Injection drug users require water both to rinse their syringes and to mix with their drugs to liquify them for injection. Rinsing is not done for hygienic purposes, but to make sure a syringe does not become clogged with blood and drug residue, so that it can be used again. Rinse water is often shared. As such, water contaminated through the rinsing of a syringe is used for rinsing other syringes and for mixing the drug. Similarly, spoons, cookers, and cottons—components of the injection kit—also represent potential reservoirs of disease. Spoons and cookers are the bottle caps, spoons, baby food jars, and other small containers used for mixing
the drug, and cottons are any materials placed in the spoon to filter out undissolved drug particles. Filtering is considered necessary since undissolved particles tend to clog injection equipment. Spoons and cottons are frequently shared, even by drug users who carry their own syringes, thus increasing the risk of HIV transmission.
Viral contamination may also result from frontloading and backloading , techniques for distributing a drug solution among a drug-injecting group (Grund et al., 1989, 1990). In frontloading, the drug is transferred from the syringe used for measuring by removing the needle from the receiving syringe and squirting the solution directly into its hub. Common in many shooting galleries (places where injection equipment is rented and shared and drugs are injected, discussed below) is the intercontamination of drug doses through the mixing and frontloading of speedball (a heroin and cocaine combination). Since heroin is cooked (heated in an aqueous solution) during its preparation for injecting, whereas cocaine is not, separate containers are used for the mixing process. Those who share speedball draw the heroin into one syringe and the cocaine into another, remove the needle from the cocaine syringe and discharge the heroin into it through its hub, and return half the speedball mixture back into the syringe that originally contained the heroin. If either syringe contains virus at the start of such an operation, both are likely to contain it afterward (Inciardi and Page, 1991).
The backloading of speedball has also been observed in shooting galleries. Backloading involves essentially the same process, but the plunger, rather than the needle, is removed from the receiving syringe. Backloading has been found to be a risk factor for HIV transmission among injection drug users in New York City (Jose et al., 1993). Frontloading seems to be the preferred mixing/sharing method; backloading is substituted when syringes with detachable needles are unavailable. An alternative method of drug sharing is referred to by some drug injectors as shooting back and drawing up. This practice has been observed in instances when every member of the drug-sharing group has a syringe. After the heroin, cocaine, or speedball is thoroughly mixed, it is discharged from the mixing syringe into a common spoon, cap, or container. Each member of the sharing group then draws a specific amount.
All forms of needle sharing tend to occur among running partners—drug users who are lovers, good friends, crime partners, or who live together. They serve as lookouts for one another—one watching for police and other intruders while the other purchases,
prepares, or injects the drug. Running partners also provide other elements of safety, such as monitoring each other's responses to the drugs in order to prevent overdoses or other acute reactions (Des Jarlais, Friedman, and Strug, 1986).
HIV risk is associated not only with certain drug-using practices, but also with certain locations where drug use occurs. In most urban locales where rates of injection drug use are high, common sites for injecting (and sometimes purchasing) drugs are the neighborhood shooting galleries, typically referred to in some settings as safe houses or get-off houses. After purchasing heroin, cocaine, amphetamines, or other injectable substances in a local drug-selling area, users are faced with three logistical problems: how to get off the street quickly to avoid arrest for possession of drugs, where to obtain a set of works with which to administer the drugs, and where to find a safe place to get off (inject and then experience the effects of the drugs). Shooting galleries occupy a functional niche in the world of injection drug use, where for a fee of two or three dollars users can rent an injection kit and relax while getting off. After using a syringe and needle, the user generally returns them to a central storage place in the gallery where they are held until someone else rents them. On many occasions, however, these works are simply passed to another user in the gallery.
Shooting galleries have not been systematically studied. However, based on the observations of a number of investigators combined with reports from a variety of ethnographic and other research studies in drug communities in several parts of the United States (Agar, 1973; Fiddle, 1967; Gould et al., 1974; Hanson et al., 1985; Johnson et al., 1985; Murphy and Waldorf, 1991; Rettig, Torres, and Garrett, 1977), their more obvious roles and characteristics can be described. Reports suggest many similarities from city to city. Most shooting galleries are situated in basements and back rooms, apartments and hotel rooms, and even house trailers in the rundown sections of cities where drug use rates are high. Other galleries are in abandoned buildings, darkened hallways, alleys, and under railroad bridges and highway ramps. Characterized by the stench of urine and littered with trash, human feces, garbage, and discarded injection paraphernalia, shooting galleries are typically unfurnished and extremely unsanitary. Rarely is there heat, running water, or functional plumbing.
Most galleries are run by drug users, drug dealers, or people who are both. Neighborhood heroin and/or cocaine sellers may operate galleries as a service to customers—providing them with a
convenient location to inject for a slight charge. More often, however, gallery operators are drug users themselves who provide a service for a small fee or a sample of someone else's drugs.
For the majority of injection drug users, shooting galleries are considered to be the least desirable places to patronize. Most prefer to use their own homes or apartments or those of drug-using friends, since these are considered safer than galleries. Few users truly relish having to pay a fee to use someone else's drug paraphernalia. Some hard-core injectors do consider personal hygiene a priority. For many drug injectors, however, the use of shooting galleries is routine and commonplace. Moreover, there are repeated occasions in the lives of all injection drug users, including the most hygienically fastidious, when galleries become necessary. If they have no works of their own, or if friends or other running partners have no works, then a neighborhood gallery is the only recourse. Similarly, users who purchase drugs far from home also gravitate toward galleries. This tendency is based on the heightened risk of arrest when carrying drugs and drug paraphernalia over long stretches. In addition, for the heroin or cocaine user undergoing withdrawal, going somewhere close by to inject after purchasing drugs is imperative. Moreover, the gallery operator often serves as a middleman between drug user and drug dealer, thus making the get-off house the locus of exchange.
Because of the way that injection equipment is cleaned and distributed in galleries, the potential for coming in contact with an HIV-infected needle and syringe is high (Marmor et al., 1987; Schoenbaum et al., 1989). As the user has paid his or her fee to use the gallery, a needle/syringe is taken from those available on a table or in a container. In some galleries, the user has no choice in the selection of a syringe, but is merely given one by the gallery operator. Regardless of who selects the syringe, the equipment is not usually scrutinized for traces of blood, but rather for dull or clogged needles. Should such an impairment be evident, only then will the user return it for a substitute. Moreover, needles are generally not cleaned prior to use. Shooting up as quickly as possible is of primary importance. After injecting and before returning the needle and syringe to the common container, the user is expected to rinse them—again, not necessarily for the sake of decontamination, but to prevent any drug or blood residue from hardening and causing an obstruction. Sometimes the paraphernalia are indeed rinsed, but with water, which does not deactivate HIV. And sometimes they are rinsed with infected water taken from a container used to rinse other needles.
While systematic research and clinical observation suggest that the use of shooting galleries, the sharing of needles and other drug paraphernalia, and the practices of booting and jacking combine to explain the increasing proportion of injection drug users infected with HIV, little is known about the prevalence of HIV antibodies in needle and syringe combinations utilized by drug injectors. To address this question, samples of needle and syringe combinations from major shooting galleries in Dade County (Miami) Florida were collected and their contents analyzed for the presence of HIV antibodies (Chitwood, McCoy, Inciardi, et al., 1990). All needle and syringe combinations were labeled and visually graded by their condition—''clean" if they contained no visible dirt, stains, or blood; "dirty" if they contained dirt or stains but no visible blood; and "visible blood" if they appeared to contain any liquid or dried blood. Of a total of 212 needle and syringe combinations collected, 62 could not be analyzed for the presence of HIV antibodies because of clogging, broken plungers, or other physical damage, leaving 150 available for laboratory analysis. Of the 150 needles tested, 15 were found to be seropositive—133 were seronegative, and in two cases serostatus was indeterminate. The overall seropositivity rate was 10.1 percent with no significant differences between sites. In addition, although the number of customers frequenting these galleries tended to vary from one day to the next, further analysis demonstrated that there were no apparent changes in seropositivity rates by day of the week. However, a strong relationship was found to exist between the graded condition of needle and syringe combinations and the presence of HIV antibodies. Of the 55 combinations graded as "clean" through visual inspection, only 5.5 percent were found to be seropositive, with a similar rate (4.7 percent) for "dirty" needles and syringes. By contrast, 20 percent of the needle and syringe combinations containing visible blood were found to be HIV positive. This study indicated a clearly significant relationship between the appearance of a needle and syringe and the presence of HIV antibodies. A "clean" needle/syringe had a significantly lesser chance of containing HIV antibodies. Conversely, 1 out of 5 needle/syringe combinations containing visible blood also contained HIV antibodies. Moreover, the data were indicative of the high rate of seropositivity among users of the three shooting galleries selected for this study and the highrisk a user has in choosing a needle/syringe containing blood. Thus, it would appear that shooting galleries represent a significant health problem as far as the spread of the HIV infection is concerned.
CRACK COCAINE AND HIV RISK: THE INTERSECTION OF DRUG USE AND SEXUAL BEHAVIOR
Although the potential for viral transmission is considerable in shooting galleries as a result of the multiple use of injection equipment, exchanges of sex for drugs are not particularly common. They occur, but not with great frequency. By contrast, however, sex-for-drugs exchanges are commonplace in many crack houses—places where crack cocaine is sold and smoked.
Crack is a variety of cocaine base, produced by cooking cocaine hydrochloride in boiling water and baking soda. It has been called the "fast-food" variety of cocaine, and is popular in the United States because it is cheap, it is easy to conceal, it vaporizes with practically no odor, and the gratification is swift: a short-lived (up to five minutes) but nevertheless intense, almost sexual euphoria. Smoking cocaine as opposed to snorting it results in more immediate and direct absorption of the drug, producing a quicker and more compelling high, which greatly increases the potential for dependence (Inciardi, 1987, 1992; Wallace, 1991). For many users, once crack is tried it is not long before it becomes a daily habit.
Users typically smoke for as long as they have crack or the means to purchase it—money, personal belongings, sexual services, stolen goods, or other drugs. It is rare that smokers have only a single "hit" of crack. More likely they spend $50 to $500 during a three-or four-day binge, smoking almost constantly. During these cyclical binges, crack users neglect food, sleep, and basic hygiene, severely compromising their physical health. In addition, mouth ulcerations and burned lips and tongues from the hot stems of the pipes are not uncommon, and many smokers have reported and have been observed to have untreated STDs (Inciardi, Tims, and Fletcher, 1993; McCoy and Miles, 1992; Ratner, 1993).
The use of crack first became popular in many inner-city communities in the United States during the mid-1980s, and shortly after the drug was noticed by the media, press and television reports began describing crack use as an "epidemic" and a "plague" that was devastating entire communities (Inciardi, 1987). Considerable focus was placed on how the high addiction liability of the drug instigated users to commit crimes to support their habits, how crack engendered a so-called hypersexuality among users, and how the drug was contributing to the further spread of HIV and AIDS (Gross, 1985; Hackett and Lerner, 1987; Kerr, 1986a,b, 1988; Lawlor, 1986; Lee, 1988; Minebrook, 1989; Morganthau and McKillop, 1986; Raab and Selwyn, 1988; Seligman, 1986; Time, 1988).
Since the beginnings of the crack epidemic, the drug has been viewed as a sexual stimulant and enhancer, as well as the cause of excessive sexual behavior in many users. Reports from the field and in the media have indicated that crack is the "ultimate turn-on"; that crack users readily engage in a variety of sexual activities, at any time and under any circumstances, and with an abundance of partners; that crack use has initiated a "new prostitution" and the crack house has become the "new brothel"; and that the numerous rates of sex-for-crack exchanges in some locales are increasing the spread of HIV infection (Chiasson et al., 1991; Fullilove and Fullilove, 1989; Grant, 1988; Greve, 1989; Inciardi, 1989). The association between crack use and apparent excessive sexual behavior has been evident in numerous ethnographic analyses of the crack scene (Ratner, 1993). Indeed, the tendency of crack users to engage in high-frequency sex with numerous, anonymous partners is a feature of crack dependence and crack house life in many locales (Bourgois, 1989; Hamid, 1990; Inciardi, 1992, 1993; Treaster, 1991; Williams, 1992). Much of the sexual activity associated with crack houses occurs in a separate bedroom specifically provided for sexual activities. However, sex acts also occur in the more public smoking rooms as well. Although many street prostitutes who barter sex for money to purchase drugs often insist that their customers use condoms, this is not usually the case with crack house sex. In fact, condoms are rarely seen in crack houses. Given the health status of crack users (including a high likelihood of compromised immune systems), the incidence of STDs (many of which go untreated), and general lack of condom use, many of the conditions that have contributed to the heterosexual transmission of HIV in Africa exist in crack houses in Miami, New York, Philadelphia, and other urban areas across the United States. In addition, given the frequency of sex and the large number of partners associated with crack house sex, the potential for coming into contact with HIV through heterosexual sex is even greater.
To understand this situation, one must consider a few things about cocaine and crack. First, cocaine's (and hence crack's) potent psychic dependence has been well documented (Washton and Gold, 1987). Compulsive users seek the extreme mood elevation, elation, and grandiose feelings of heightened mental and physical prowess induced by the drug. When these sensations begin to wane, a corresponding deep depression is felt, which strongly motivates users to repeat the dose and restore their euphoria. Thus, when chronic users try to stop using crack, they often are plunged
into a severe depression from which only the drug can arouse them.
Second, cocaine is problematic as an aphrodisiac, in either its powdered or its base form. Researchers have found considerable differences in sexual responses to the same dosage level of cocaine, depending primarily on the setting of the use and the background experiences of the user. Among male recreational users, cocaine not only helps to prevent premature ejaculation, but at the same time permits prolonged intercourse before orgasm. Among female recreational users, achieving a climax under the influence of cocaine is often quite difficult. For both, however, when an orgasm finally occurs, it is quite intense. Medical accounts generally conclude that because of the disinhibiting effects of cocaine, its use among new users does indeed enhance sexual enjoyment and improve sexual functioning, including more intense orgasms (Grinspoon and Bakalar, 1985; Weiss and Mirin, 1987). These same reports maintain, however, that among long-term addicts, cocaine decreases both sexual desire and sexual performance (MacDonald et al., 1988).
The association between crack and sex appears to be both pharmacological and sociocultural in nature. The pharmacological explanation begins with psychopharmacology: one effect of all forms of cocaine, including crack, is the release of normal inhibitions on behavior, including sexual behavior. The disinhibiting effect of cocaine is markedly stronger than that of depressants such as alcohol, Valium, or heroin. While the latter drugs typically cause a release from worry and an accompanying increase in self-confidence, cocaine typically causes elation and an accompanying gross overestimation of one's capabilities. Moreover, because the effects of cocaine have a rapid onset, so too does the related release of inhibitions.
Often, the association between crack and sex results from the need of female crack addicts to pay for their drugs. This connection has a pharmacological component—crack's rapid onset, extremely short duration of effects, release of inhibitions, and high addiction liability combine to result in compulsive use and in a willingness to obtain the drug through any means. In addition, although overdose is a constant threat, crack use does not pose the kind of physiological limit on the maximum needed (or possible) daily dosage that other drugs do. Whereas the heroin addict typically needs four doses per day, and an alcoholic commonly passes out after reaching a certain stage of intoxication, the heavy crack user typically uses until the supply is gone—be that minutes,
hours, or days. The consequent financial burden can be staggering. Other parts of the economic relationship between crack and sex, however, are strictly sociocultural. As in the legal job market, women's access to income in the illegal, street subcultures is typically more limited than men's. Prostitution has been the easiest, most lucrative, and most reliable means for women to finance drug use (Goldstein, 1979).
Because crack makes its users ecstatic and yet is so short-acting, it has an extremely high addiction potential. Use rapidly becomes compulsive use. Crack acquisition thus becomes enormously more important than family, work, social responsibility, health, or personal values. This makes sex-for-crack exchanges psychologically tolerable as an economic necessity. Furthermore, the disinhibiting effects of crack enable users to engage in sexual acts they might not otherwise even consider.
Although the bartering of sex for crack was mentioned in the popular media at the very beginnings of the crack epidemic (Gross, 1985; Lamar, 1986; Lawlor, 1986), the first empirical study of the phenomenon did not appear in the scientific literature until 1989. In that analysis, drawn from a larger study of drug use and street crime among serious delinquents in Miami, the potential for HIV acquisition and transmission through sex-for-crack exchanges was addressed (Inciardi, 1989). Of 100 girls ranging in age from 14 to 17 years, 27 had bartered sex for crack during the one-year period prior to interview. Of these, 11 had traded sex for drugs on fewer than six occasions, but had nevertheless exchanged sex for money on an aggregate of 6,850 occasions.
At about the same time that this research was being reported, several observers began to notice rising rates of syphilis and other STDs among crack users (Bowser, 1989; Fullilove and Fullilove, 1989; Fullilove, Fullilove, Bowser, et al., 1990; Kerr, 1989; Knopf, 1989a,b). Shortly thereafter, sex-for-crack exchanges were targeted for systematic study by CDC and NIDA. However, one of the difficulties in assessing the nature of HIV risks associated with crack use remained the fact that most crack users engage in multiple risk behaviors.
A study of risk factors for HIV infection was conducted at an STD clinic in an area of New York City where the cumulative incidence of AIDS among adults through mid-1990 was 9.1 per 1,000 population and where the use of illicit drugs, including crack smoking, was common (Chiasson et al., 1991). Overall seroprevalence among the 3,084 volunteer subjects was 12 percent, with 80 percent of those reporting risk behaviors associated with HIV infections
including male-to-male sexual contact, intravenous drug use, and heterosexual contact with an injection drug user. The seroprevalence rate in individuals denying these risks was 3.6 percent in men (50 of 1,389) and 4.2 percent in women (22 of 522). Among women, the behaviors associated with infection were prostitution and the use of crack, and among men, a history of syphilis, crack use, and sexual contact with a crack-using sex worker were associated with HIV infection.
The potential for a male in sex-for-crack exchanges to come into contact with an HIV-positive female partner was demonstrated in a study of 87 New York City women who had been admitted to a municipal hospital with a diagnosis of pelvic inflammatory disease (Des Jarlais, Abdul-Quader, and Minkoff, 1991). Crack use was reported by 56 percent of the subjects (n = 49), and of these, 20 percent were HIV positive. Crack use was significantly related to both traditional AIDS risk behaviors (injecting drugs and having sex with an injection drug user) and other unsafe sexual behaviors (exchanging sex for money or drugs and having casual sex partners).
Given the potential for sex-for-crack exchanges to spread HIV to new populations, in 1989 NIDA began supporting ethnographic studies of the phenomenon in eight cities—Chicago, Denver, Los Angeles, Miami, Newark, New York, Philadelphia, and San Francisco (Ratner, 1993). A total of 340 crack users (69 percent of whom were women) were interviewed in depth. Of the 233 women, 108 had participated in sex-for-crack exchanges, as had 69 of the men. HIV testing was done with 168 of the subjects, and a total of 14 percent were found to be positive for HIV antibodies. Of the 24 males who were non-injectors and who had engaged in heterosexual sex-for-crack exchanges, 3 were HIV positive.
ALCOHOL USE AND SEXUAL TRANSMISSION
Only very recently has the possible link between alcohol consumption and risky sexual behavior been explored in the context of HIV transmission. This is somewhat surprising, given that it is a fairly common belief that alcohol use and risky sexual behavior are causally linked. Alcohol is perceived to reduce inhibitions, causing impulsive or out-of-control behavior and thereby increasing the likelihood of risky sexual activity. Nevertheless, despite this widespread—and intuitively appealing—belief, direct causality of alcohol use and risky sexual behavior has not been established.
Recent research on the relationship between alcohol and risky sexual behavior has produced contradictory findings. For example, a study of a group of 461 gay and bisexual men in England and Wales found no statistically significant difference in rates of risky sexual behaviors between sexual encounters that involved alcohol use and those that did not. Furthermore, for those men who engaged in sexual behavior while under the influence of alcohol, the quantity of alcohol consumed had no effect on sexual behavior (Weatherburn et al., 1993). Studies of gay men in two cities in Australia found that in Melbourne drinking was not significantly associated with unsafe sex, but in Sydney it was (Leigh and Stall, 1993). Additionally, a study of gay and bisexual men in San Francisco discovered higher rates of risky sexual behavior among men in an outpatient alcohol and drug abuse treatment program than among gay and bisexual men who were not in the program and who were not identified as having a substance abuse problem (Paul, Stall, and Davies, 1993). At the same time, however, a study of 2,174 students in England found that those students who drank more also had higher rates of unsafe sex. The respondents themselves associated alcohol with sexual risk taking, reporting, for example, that when drinking they were more likely to have sex without contraceptives or with someone they know has had many previous sexual partners (McEwan et al., 1992).
In a recent study of HIV seroprevalence rates among heterosexual alcoholics in treatment, researchers in San Francisco found that, after controlling for injection drug use, the rate of HIV infection was 3 percent in men and 4 percent in women (it was 5 percent overall, including injection drug users). People in this study also had a high prevalence of unsafe sexual behaviors (Avins et al., 1994).
The reasons for the mixed results from the studies cited above are not yet understood, but taken together, the studies suggest that it is premature to assume a causal relationship between alcohol use and risky sexual behaviors. Indeed, much of the research to date could be consistent with a number of explanations, including causality, correlation, and coincidence (Leigh and Stall, 1993). The lack of consistent findings may be due in part to the variety of methodologies and measures used in studying the relationship between alcohol and risky behaviors. A great variety of research foci, methods, study populations, variables and measures, sampling and weighting issues, and definitions of "risky sex" and "alcohol use" has negated the possibility of reliable synthesis of research results. Some studies, for example, only compare behavioral
extremes, which distorts their generalizability. Other studies are conducted with populations for which the overall patterns of alcohol use or sexual behavior have changed over time. This variability of study design and purpose has made comprehensive synthesis difficult. But it is also complicated by a difference among researchers in the conceptualization of the issue. Some focus on the individual, proposing that the links among alcohol, risky sexual behavior, and HIV may be a result of a certain personality type—that is, one who may be predisposed both to drink and to engage in risky sexual behavior (and thereby put himself or herself at risk of HIV). Others view the link between alcohol and sexual risk as a contextual phenomenon, that is, alcohol may be related to unsafe sex because sexual encounters often begin in drinking establishments or contexts (e.g., parties) where people drink to subconsciously rationalize or facilitate their subsequent risky behavior.
It has been argued elsewhere (Leigh and Stall, 1993) that, given the lack of certainty resulting from research to date on the links between alcohol, risky sexual behavior, and HIV/AIDS, causality need not be established for additional research and public policy efforts to be undertaken. Future research to assess the alcohol-HIV link should include alternative approaches such as correlational studies designed to identify other variables that are relevant (e.g., determining the usefulness of the concept of a risk-taking personality, examining other psychosocial variables). This approach would provide more information on causality by characterizing the nature and relevant weight of the variables involved in risky sexual behaviors. Observational studies of the association of alcohol use and risky sex might determine if there is indeed an association between the two, in which case reducing the levels of substance abuse might lower the levels of risky sex. Finally, using excessive drinking as a marker for risky sexual behavior, even in the absence of any casual relationship between the two behaviors, would provide a useful shorthand for a complex set of social and psychological variables that result in higher risk. For all markers, there is no claim that such characteristics cause risky sexual behaviors, only that the correlation is useful in designing public health and information efforts (Leigh and Stall, 1993; Stall and Leigh, 1994).
Because HIV infection is transmitted by intimate acts, the epidemic spread of the virus through social and sexual networks has illuminated the personal connections that link the disparate parts of the world together. HIV infection occurs because individuals
are socially and sexually engaged with each other. These intimate acts take place between specific people, acting in specific places. The central problem in AIDS epidemiology, from the perspective of the social and behavioral sciences, is to describe the connections between individual acts and social settings that lead to the spread of infection. On the basis of the careful delineation of the social structure of viral transmission, scientists can develop targeted strategies for prevention campaigns.
MONITORING THE EPIDEMIC
Epidemic surveillance—numbers of AIDS cases and HIV infection—is an essential first element for delineating the social and geographic structure of the epidemic. Those basic data provide the framework for more detailed analysis of the specific social settings and interpersonal interactions that will promote or contain further spread of infection, and have been useful for estimating the magnitude of the AIDS epidemic. As with all estimates, however, the number of AIDS cases is an imperfect reflection of the magnitude of the epidemic. AIDS cases reflect only the numbers of people with advanced illness. AIDS is characteristic of the end stage of HIV disease, and may follow a period of latency or milder illness lasting, on average, about 10 years. AIDS cases thus reflect the patterns of transmission and infection that occurred at some time in the past. The criteria for AIDS, originally established in 1981, have been altered in 1985, 1987, and 1993 to reflect the information accumulated about the natural history of HIV disease. Finally, AIDS case reporting is incomplete. Although in the United States a vast majority of cases are reported, this is not true in many parts of the world. Despite these limitations, AIDS case reporting has been a central tool for examining the epidemic, and has revealed much about its spread over the past 13 years.
In the United States, the epidemic has disproportionately affected men, who constituted more than 85 percent of the adults diagnosed with AIDS as of December, 1993. Early in the epidemic, the predominance of cases among men who had sex with men gave rise to the stereotype that AIDS was a "gay" disease. However, this stereotype has been challenged as the list of groups at risk for contracting HIV has expanded to include injection drug users, their sexual partners, infants born to mothers infected with HIV and, in very rare cases, those exposed to blood or blood products by occupational activities or transfusion.
AIDS cases have also been disproportionately reported among African Americans and Hispanics/Latinos (Selik, Castro, and Pappaioanou, 1988), and this excess risk for AIDS has increased as the epidemic has progressed. African Americans and Hispanics/Latinos compose approximately 20 percent of the U.S. population. Early in the epidemic, 42 percent of those with AIDS were African American or Hispanic/Latino; by 1993, that proportion had reached 48 percent. The analysis of this excess risk for AIDS has required that researchers develop specific hypotheses regarding the role of race and ethnicity as explanatory factors in HIV infection (Osborne and Feit, 1992). Little support has emerged for biological explanations of excess risk for infection. More support has emerged for social hypotheses that examine race/ethnicity and socioeconomic status as factors in the organization of interpersonal networks within which people have sex or use drugs (Neaigus, Friedman, Curtis et al., 1994; Potterat et al., 1985; Wallace, 1988).
The number of AIDS cases reported in the United States has grown rapidly, surpassing 100,000 in 1988, 200,000 in 1990, and 300,000 in 1992. A comparison of the second 100,000 cases with the first demonstrated some of the trends of the epidemic. For example, while the first set of cases were reported between 1981 and 1988, the second set of cases were reported in the two-year period from 1988 to 1990. There was a larger proportion of cases attributable to heterosexual transmission in the second 100,000 than there was in the first 100,000. The proportion of cases among African American and Hispanics/Latinos increased from 42 percent to 48 percent of cases. Among the first 100,000 people with AIDS, 9 percent were women, while women represented 12 percent of the second 100,000 (CDC, 1992a).
Sixty percent of adults and 53 percent of children diagnosed with AIDS have died. In November 1993, the Centers for Disease Control and Prevention reported that by 1992 HIV infection had become the leading cause of death for men aged 25 to 44 and the fourth leading cause of death for women in this age group. AIDS is the cause of 19.9 percent of deaths among men and 7.3 percent of deaths among women in this age group (Figure 2.1). Factors such as socioeconomic status and access to health care are thought to affect length of survival and quality of life after diagnosis (Curtis and Patrick, 1993).
As AIDS has spread in numbers and through risk groups, it has also spread geographically. From the major urban centers where it was first identified, AIDS was disseminated through hierarchical
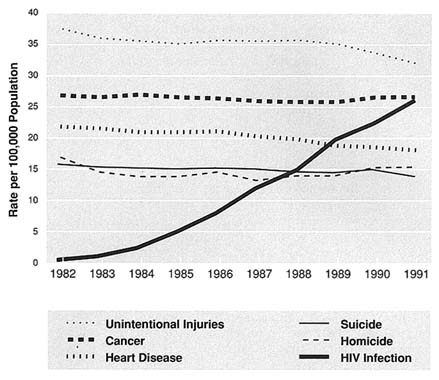
FIGURE 2.1 Death Rates for Leading Causes and HIV Infection for Persons Aged 25–44, 1982–1991. Note: 1991 rates are provisional. Data are from NVSS, NCHS, DVS. Source: National Vital Statistics System, CDC, NCHS.
diffusion, spatial contagion, and social diffusion. AIDS spread from major cities to smaller cities connected to them by transportation routes (hierarchical diffusion). AIDS also spread within the commuting field of each of the affected cities, extending outward from local epicenters (spatial contagion). Within a particular area, AIDS has been transmitted within the social and sexual networks of infected people (social diffusion). The characteristics of these networks, including the frequency and density of highrisk intimate interactions among individuals within them, appear to determine the rate and extent of spread both within and between local networks. Networks are placed at risk because individual members of one network may have connections to other networks. This spread of infection into new populations has characterized the AIDS pandemic worldwide.
PREVALENCE AND INCIDENCE OF HIV INFECTION
Measuring HIV prevalence (the number of infections at a point in time) and incidence (the number of new infections over time) is critical for monitoring the course of the epidemic. Descriptions of prevalence and incidence of HIV infection provide a more complete picture of the magnitude of the epidemic than do AIDS case reports, because they do not rely on presentation of end-stage disease. Efforts to determine HIV prevalence have been based on a range of seroprevalence studies—small cross-sectional studies and large-scale surveys. The most difficult challenge in conducting seroprevalence studies is the selection of the sample. Samples drawn from the general population, such as household surveys, will include many at low risk for infection, but few at highrisk for infection. Conversely, samples chosen to select those at higher risk cannot be generalized to the population at large. As a partial solution to this problem, surveys have been conducted among highrisk subgroups as well as in the general population. The combined insights provided by this ''family of surveys" have guided estimates of seroprevalence.
CDC has conducted seroprevalence surveys of sex workers, drug users (in and out of treatment), patients in STD clinics and sentinel hospitals, childbearing women, and newborn infants. The surveys have been of two types: blinded surveys, in which the serum cannot be connected to the donor, and unblinded surveys, in which people provide consent for their blood to be tested and the results can be linked to the donor. All branches of the military have conducted HIV testing on new recruits as well as enlisted personnel. The Jobs Corps has conducted HIV testing of its applicants. The National Center for Health Statistics has conducted a national household study of seroprevalence. Local studies of the general population have been conducted in Dallas, San Francisco, and Chicago. Studies of special populations, such as psychiatric patients, homeless psychiatric patients, runaway adolescents, and prisoners, have been conducted in localities throughout the United States.
These seroprevalence studies demonstrate that HIV infection spread rapidly through the large, tightly knit, geographically concentrated networks of gay men and injection drug users that occurred in certain cities. The San Francisco Cohort, a study that began in the 1970s as a study of the hepatitis vaccine, was able to document retrospectively that substantial spread of HIV had transpired before the first cases were officially recognized in 1981.
Aside from hemophiliacs, the highest seroprevalence rates have been documented among drug users in the New York metropolitan area: as early as 1984, rates of infection as high as 50 percent were reported in some studies (Marmor et al., 1987). However, alarming HIV seroprevalence rates also have recently been observed among other groups, including female Job Corps students, young gay men, alcoholics in treatment, and psychiatric patients.
While there is some evidence that HIV rates among gay males in some cities have plateaued (San Francisco Department of Public Health, 1994), other trends give cause for alarm. For example, the overall prevalence of HIV among Job Corps students, at 3 per 1,000 is high given their youth (Conway et al., 1993). Among female Job Corps students, the HIV rate rose from 2.1 per 1,000 in 1988 to 4.2 per 1,000 in 1992. The rate among male students decreased during this time from 3.6 to 2.2 per 1,000.
One indicator of trouble is the relapse to unsafe sex by men who had adopted safer practices (Stall et al., 1990; Stall et al., 1992). A second indicator of possible negative future trends is the high rate of unsafe behavior reported by the low-risk population (Catania et al., 1992; Tanfer et al., 1993). In fact, rates of risk behavior may provide a more accurate window to the future than that offered by AIDS case rates or HIV seroprevalence.
With respect to injection drug users, numerous HIV seroprevalence studies have been conducted in many cities around the world, and have generated prevalence estimates ranging from 0 percent to greater than 50 percent of the relevant populations. A number of these cities have seroprevalence rates of 30 percent or higher among drug injectors, and several have seen the rapid spread of HIV among drug injectors soon after the introduction of HIV into the drug-using population (Brown and Beschner, 1993; CDC, 1990, 1993b; Des Jarlais et al., 1989; Des Jarlais, Friedman, Choopanya, et al., 1992; Hahn et al., 1989).
Although most seroprevalence studies have been performed among convenience samples, and most have involved prevalent rather than incident HIV infections, they have generated useful information regarding the levels of HIV infection within these defined population groups. HIV incidence can be an important predictor for the future course of the epidemic. Although some studies have measured incidence among various highrisk groups (including gay men and injection drug users), there is a need for additional data on HIV incidence (Metzger et al., 1993; Moss et al., 1994; Rezza et al., 1990; van Ameijden et al., 1992; Vlahov et al., 1990; Vlahov, 1994). Such data provide insight into the spread of
HIV infection among various populations, and may ultimately provide the basis for intervention programs aimed at curtailing further transmission.
Among injection drug users, in particular, it is also critical to develop effective epidemiologic surveillance mechanisms for identifying new drug injectors within populations, to be able to track trends in drug injection over time. Due in part to difficulties in identifying and gaining access to the population groups initiating or relapsing into drug injection, most of the HIV epidemiologic studies among drug users to date have focused on long-term, chronic injectors. While older injectors have generally had higher documented levels of prevalent HIV infection, the group of younger, new injectors may be at particularly highrisk of acquiring and transmitting HIV infection, and thus may be an important group for preventive interventions in the future.
Another population at potentially highrisk for HIV infection, but one that has been neglected in the research until very recently, is the seriously mentally ill (Box 2.1). Recent seroprevalence studies among the seriously mentally ill reveal rates of HIV infection higher than the general population. Because risk among this group relates to comorbidity of mental illness, substance abuse, and unprotected sex, the seriously mentally ill are an important population for monitoring concurrent epidemics.
FACTORS INFLUENCING THE EPIDEMIC'S COURSE: RISKY ACTS, SOCIAL NETWORKS, AND UNSAFE PLACES
As has been noted, HIV infection is spread through specific behaviors, particularly sexual intercourse without barrier protection and the sharing of contaminated drug injection paraphernalia. In order to understand the potential course of the epidemic, social and behavioral research has focused on determining the likelihood that infected individuals in one group will have contact with uninfected individuals in the other; identifying the contexts that influence risk taking; investigating whether people in high- and low-risk groups are adopting recommended preventive behaviors; and examining how the spread of HIV infection will be altered by changing patterns in concurrent epidemics of drug use and infectious diseases.
The risk posed by unprotected sexual intercourse is linked to the probability that the sexual partner is HIV infected. Several studies have examined the risk status of sex partners of people who are neither homosexually active men nor intravenous drug
|
Box 2.1 HIV Risk Among the Seriously Mentally Ill It is estimated that 2.8 million Americans suffer from a serious mental illness, yet little is known about their sexual and drug-using behavior, or about the incidence and prevalence of HIV/AIDS among them. A series of recent studies conducted in facilities in New York City show HIV seroprevalence rates among the seriously mentally ill ranging from 4.0 percent to 19.4 percent (Cournos et al., 1991; Sacks et al., 1992; Susser, Valencia, and Conover, 1993; Volavka et al., 1991; Zamperetti et al., 1990). In all cases, these rates are higher than for the general population. These data suggest that people with severe mental illness may constitute a significant highrisk group for HIV infection. Only very recently has the seriously mentally ill population been considered a group of people at risk of contracting or transmitting HIV, even though it is generally assumed that cognitive impairments, problems in judgment, affective instability, and impulsivity increase the likelihood that people will engage in unsafe sexual behavior and drug use (Brady and Carmen, 1990). Indeed, comorbidity of substance abuse and psychiatric illness has been recognized for quite some time (Cournos et al., 1991), and recently the "triple diagnosis" of HIV, substance abuse, and psychiatric disorder has been identified (Batki, 1990a). With respect to sexual risk, contrary to the longstanding stereotype of the mentally ill population as asexual, recent studies suggest that one-half to two-thirds of seriously mentally ill patients have been sexually active within the previous year, and among those approximately one-half to two-thirds have had more than one sex partner during that time (Cournos et al., 1994; Kalichman et al., 1994). Chronically mentally ill women are more sexually active than are their male counterparts (Lyketsos et al., 1993; Test and Berlin, 1981). Moreover, many mentally ill persons engage in homosexual or bisexual activities, even though they may not identify themselves as homosexual or bisexual (Brady and Carmen, 1990; Cournos et al., 1994; Kalichman et al., 1994). Psychiatric symptoms may impede the ability of these individuals to perceive their own sexual risk for HIV or to employ necessary prevention measures, such as using condoms (Cournos et al., 1991; Cournos et al., 1994; Kalichman et al., 1994). |
users. CDC's study of HIV infection in Belle Glade, Florida, interviewed residents of the community at home and in clinic settings. Those with HIV infection were significantly more likely than those who were HIV negative to report a highrisk sex partner (Castro et al., 1988). Castro and colleagues suggested that the lessons of Belle Glade might be relevant to other cities.
|
The seriously mentally ill also are at risk for HIV because of their aggregate high rates of comorbidity with substance abuse. Data from the Epidemiological Catchment Area Survey sponsored by NIMH indicate that the rate of alcohol and drug abuse is 47 percent among schizophrenic patients and 61 percent among patients with bipolar disorder (Grinspoon, 1994). Moreover, substance abuse often is a cofactor in risky sexual behavior among the mentally ill (as well as other populations). Two recent studies, in New York and Milwaukee, found that a majority of sexually active psychiatric patients used alcohol or drugs in conjunction with sex—more commonly alcohol than other substances (Cournos et al., 1994; Kalichman et al., 1994). Approximately 50 percent of these patients also exchanged sex for money, drugs, or other goods. Women were more likely to sell sex and men were more likely to buy it (Cournos et al., 1994; Kalichman et al., 1994). While these studies provide important information about HIV risk and prevalence among the seriously mentally ill, they are limited in a number of ways. First, and most obviously, nearly all research has been conducted in one place—New York City—and may not, therefore, be generalizable. The particular demographic characteristics of that city's seriously mentally ill population—for example, the overrepresentation of racial/ethnic minorities—may not be common to the rest of America. Second, except for one study of mentally ill homeless men in a shelter (Susser, Valencia, and Conover, 1993), all research has taken place among psychiatric patients in hospitals or clinics. The population of seriously mentally ill who have not undergone treatment has not been adequately examined. Third, a number of researchers report that relating serostatus to specific risk behaviors among the mentally ill has been made difficult by the fact that hospitals and clinics often do not take sexual and drug-using histories from patients upon arrival (Cournos et al., 1991). Moreover, many inpatients are released without a record of their serostatus, suggesting a lack of testing and reporting of HIV status on their charts (Mahler et al., 1994). HIV testing and reporting among the mentally ill raise significant ethical issues, which undoubtedly contribute to this situation. (For a discussion of some of the ethical issues of testing in this population, see Satriano and Karp, 1993.) |
The AIDS in Multiethnic Neighborhoods (AMEN) Study was designed to examine the possibility that similar risky sexual networks existed in an urban setting. The AMEN Study interviewed single adults residing in highrisk neighborhoods adjacent to the Castro district, the AIDS epicenter of San Francisco. Among the 1,573 heterosexually active people who did not inject drugs interviewed
in that study, the prevalence of risk behaviors (sex with an HIV-infected person or homosexually active man or injection drug user, unprotected sexual intercourse with more than 4 partners, and having a sexually transmitted disease) was 12 percent overall, with specific race-gender estimates ranging from 5 percent among Hispanic/Latina women to 21 percent among white women (Fullilove et al., 1992). Similar findings were reported from a study of two highrisk neighborhoods in Chicago (Wiebel et al., 1993). The behavioral bridge between low-risk and highrisk populations is the potential pathway for further spread of HIV infection.
That spread may be addressed by examining the interaction between social and individual risk taking. Unstable communities created by war, social upheaval, or social marginalization have been called "risky situations," contexts in which people are predisposed to engage in risky behavior and thus are at greater risk of HIV infection and other diseases (Obbo, 1993; Wallace, 1988; Zwi and Cabral, 1991). For example, sharing of drug injection equipment is concentrated among those who use illicit drugs, particularly heroin, cocaine, and amphetamines. Because of the illegal nature of drug use, drug users are forced to live in hidden subcultures, mostly located in poor communities in urban centers (Drucker, 1991). One such setting is the South Bronx section of New York City. This section of New York was partially destroyed by a program of "planned shrinkage," which curtailed city fire services in the 1970s. A substantial portion of the housing in the area—up to 80 percent of some blocks—was burned down. The social networks of poor people, including those of intravenous drug users, were greatly disrupted by this process. It has been argued that the social disruption of the 1970s, which occurred just as HIV infection was established in that neighborhood, contributed to subsequent high rates of HIV seroprevalence (Wallace, 1988).
A 1990 study of the social setting and kinship networks of women heroin addicts in the Bronx demonstrated the social character of drug use that has evolved in that community (Pivnick et al., 1994). Drug use was rare among the parents of the 126 women studied. However, of the 589 siblings of the subjects, 31 percent were reported to have a history of drug abuse, and 6 percent had a history of alcohol abuse. Drug use patterns were influenced by the drug use of the women's sex partners. In addition, half of the women lived with one or more of their children, supporting a perception of drug use as normative. The drug-using social networks, located in a distressed social setting, create a risky situation
in which the likelihood of HIV infection is greatly increased for anyone having unprotected sexual intercourse or using needles for drug injection.
Social settings and networks are also implicated in efforts to reduce HIV risk. In a recent study of injection drug users in Brooklyn, consistent condom use was shown to be a characteristic of social relationships rather than an individual attribute (Friedman et al., 1994). Condom use by an individual varied from relationship to relationship. Relationship characteristics, as well as peer norms, influenced the extent of consistent condom use. Overall, condoms were consistently used in about two-thirds of the relationships between HIV positive injection drug users and their non-drug using partners.
The HIV epidemic is tightly linked to a complex set of diseases whose prevalence in the community will change over time. The progression of the other, concurrent epidemics—infectious disease epidemics such as STDs and behavioral epidemics such as drug use—can be expected to influence either the incidence and prevalence of HIV infection or the course of HIV disease. Several examples may serve to highlight this process.
New drug use epidemics can alter the patterns of needle use or sexual behavior, increasing the prevalence of risk behavior in the community. An increase in the number of people practicing risky behaviors implies an increase in the risk for spread of infection. The crack epidemic has demonstrated the increased spread of HIV infection through the confluence of new epidemics. As discussed earlier, the risk for HIV infection among crack users is thought to be attributable to the widespread practice of risky sex-for-drugs exchanges.
Some diseases will influence the course of HIV disease, rather than the spread of new infections. HIV-infected people are more likely to move rapidly from tuberculosis infection to active tuberculosis disease. The prognosis for tuberculosis in the HIV-infected person appears to be poor and requires early diagnosis and aggressive treatment. Thus, infection with tuberculosis may shorten the course of HIV disease for those dually infected.
CONCLUSION AND RECOMMENDATIONS
CONCLUSION
The AIDS epidemic in the United States, while still predominantly affecting the gay male community, has spread through sexual contact and injection drug use to other communities and populations.
Epidemiological and social research have contributed much to an understanding of the primary modes and conditions of HIV transmission, which is important both for charting and for predicting the course of HIV/AIDS and concurrent epidemics of other infectious diseases and drug use. Qualitative research has played a particularly important role in explicating the specific activities, conditions, and meanings of sexual and substance-using behaviors among the people engaged in them. Knowledge of this sort is essential for designing effective prevention and treatment interventions, especially among hard-to-reach populations.
At the same time, serious gaps remain in the information available on the prevalence of behaviors that put people at risk for HIV. Nationally representative data on sexual and substance-using behaviors are still unavailable, so it is impossible to estimate with any accuracy how many people are indeed at risk for HIV infection from any particular behaviors. Nor is it known to what extent individuals engage in multiple risk behaviors. Until these kinds of data exist, it will be difficult to monitor the epidemic and to design and target truly effective HIV interventions.
RECOMMENDATIONS FOR UNDERSTANDING HIV TRANSMISSION
2.1 The committee recommends that a national survey be undertaken to determine the prevalence and correlates of HIV risk-taking behavior. NIAAA, NIDA, and NIMH should collaborate to sponsor such a survey.
2.2 The committee recommends that NIAAA, NIDA, and NIMH support studies of the social, psychological, and biological determinants of HIV risk-taking behavior using a variety of methods complementing the national survey. These studies would allow an understanding of the detailed mechanisms of such behavior, the social dynamics within which they occur, the differing conceptualization and terminology in specific communities, and the role of concurrent health events.
2.3 The committee recommends that NIAAA, NIDA, and NIMH develop studies of the highrisk settings, such as shooting galleries and crack houses, that may contribute to epidemic spread and implement prevention efforts in those settings.
2.4 The committee recommends that NIAAA, NIDA, and NIMH support research that integrates basic biological, epidemiological,
psychological, and social research to better understand transmission of HIV through sex and drug use.
2.5 The committee recommends that NIAAA, NIDA, and NIMH support research on AIDS risk and behavior change among seriously mentally ill men and women and people with other cognitive impairments, including those not in psychiatric treatment. Such research should be conducted in a range of geographic locations.
2.6 The committee recommends that the Public Health Service coordinate interagency efforts to monitor and respond to concurrent epidemics (such as drug use, violence, and infectious diseases) that will alter the course of the HIV epidemic.































