
7 Dioxin Toxicity
|
Environmental ALERT…
|
This monograph is one in a series of self-instructional publications designed to increase the primary care provider’s knowledge of hazardous substances in the environment and to aid in the evaluation of potentially exposed patients. The Agency for Toxic Substances and Disease Registry (ATSDR) and the Centers for Disease Control (CDC) designate this continuing medical education activity for 1 credit hour in Category 1 of the Physician’s Recognition Award of the American Medical Association and 0.1 continuing education units for other health professionals. See pages 17 to 19 for further information.
|
Guest Contributor: |
Raymond Demers, MD, MPH |
|
Guest Editor: |
Eugene Perrin, MD |
|
Peer Reviewers: |
Charles Becker, MD; Jonathan Borak, MD; Joseph Cannella, MD; Bernard Goldstein, MD; Alan Hall, MD; Richard J.Jackson, MD, MPH; Jonathan Rodnick, MD; Robert Wheater, MS; Brian Wummer, MD |

U.S. DEPARTMENT OF HEALTH & HUMAN SERVICES
Public Health Service
Agency for Toxic Substances and Disease Registry
Case Study
A 5-year-old boy with a vesicular facial rash one day after contact with an herbicide
A 5-year-old boy is brought to your office after acute onset of a rash on his face and arms. The rash consists of small blisters with surrounding erythema, which the patient states itch and burn. The boy’s mother says she noticed the rash on her son’s face last evening, and by morning it had spread to both arms. The child also complains of a headache and stomachache that began early this morning. His temperature is normal.
Further history reveals that the patient and his family moved to this Midwest rural area 2 years ago; their farm is adjacent to a wooded area. Two days ago, workmen from a utility company sprayed beneath the high-voltage power lines that traverse the back edge of the property where the children frequently play during the summer. On questioning, the workmen told the mother they were using an herbicide, but assured her the area would be safe for the children in a few hours. The mother asks you if the defoliant could have contained dioxins, and, if so, whether her child has been affected. Her concern stems from what she has heard about alleged effects of Agent Orange on Vietnam veterans.
Medical history and chart review reveal the child has had no major illnesses. He has had a normal pattern of growth and development, both physical and psychosocial. Immunizations are up to date.
![]()
(a) What further questions would you ask while taking the medical history?
_________________________________________________________________
(b) On what aspect of the boy’s physical examination should you focus to address the mother’s concern?
_________________________________________________________________
(c) What laboratory tests are appropriate to aid in the diagnosis?
_________________________________________________________________
(d) Are dioxins likely to be the cause of the boy’s rash? Explain.
_________________________________________________________________
Answers to the Pretest are incorporated in Challenge answers (6) through (10) on page 16.
Exposure Pathways
❑ The general population’s principal route of dioxin exposure is through the food chain.
❑ Other minor dioxin sources are diesel exhaust, chlorine-bleached paper products, and incineration gases.
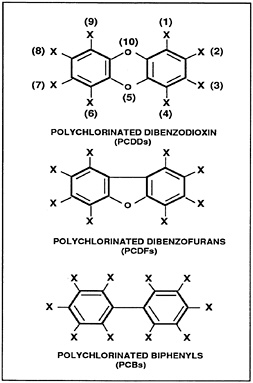
Dioxins and furans are terms applied to polychlorinated dibenzo-p-dioxins and polychlorinated dibenzofurans (Figure 1). They are believed to produce similar health effects and are known to coexist as unwanted contaminants in various materials. Because 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) is the most thoroughly studied and most toxic of the 75 dioxin isomers, the term TCDD is used interchangeably with dioxins throughout this monograph. As a pure solid, TCDD is colorless, odorless, lipid-soluble, and only sparingly soluble in water.
Figure 1. Chemical structure of dioxins and related compounds
Dioxins, as well as furans, have received much media attention in the past few decades and hence have become a public concern. Dioxins’ reputation as extremely toxic is based primarily on tests performed on guinea pigs, the most dioxin-sensitive mammalian species. By comparison, the hamster is 5000 to 10,000 times more resistant in similar toxicity tests. Extrapolation to humans from data based on animal studies is difficult because species vary widely in their sensitivity to dioxins and human risk assessment models are uncertain.
Several human populations exposed to dioxin-contaminated compounds have been studied extensively for health effects. Some Vietnam veterans were potentially exposed to dioxins through the military use of the defoliant Agent Orange (a mixture of 2,4,5-trichlorophenoxyacetic acid [2,4,5-T] and 2,4-dichlorophenoxyacetic acid [2,4-D] contaminated with TCDD). Several areas of the United States have been contaminated as a result of industrial discharges or spraying of dirt roads with dioxin-contaminated waste oils. Three areas that have received notoriety are Love Canal in Niagara Falls, New York; Times Beach, Missouri; and Newark, New Jersey. The most highly publicized accident affecting a residential population was an explosion at a chemical plant that resulted in an airborne discharge of dioxins over Seveso, Italy, in 1976. More than 37,000 people lived in an area that may have been contaminated by dioxins. In all cases, there were no deaths from acute poisoning, and concomitant exposure to other toxic substances may have contributed to the effects that were observed.
Dioxins are formed during the production of many chlorinated organic solvents, hexachlorophene, and the herbicide 2,4,5-T. Emissions from coal-burning power plants, exhaust from diesel engines, and the incomplete burning of wastes containing chlorine, such as PVC plastic form both dioxins and furans; they are also naturally produced in small amounts by volcanoes and forest fires. In nature, dioxins are found adsorbed on air and soil particles. TCDD concentrations in water and on vegetation are generally below present detection limits.
Extremely small quantities of dioxins are found nearly everywhere in the developed world. Minute amounts of TCDD have been detected in paper pulp, formed when chlorine is added during the bleaching process. TCDD reportedly has been leached from milk cartons and from coffee filters (less than 50 parts per trillion [ppt]). The amount of dioxins detected in paper-based personal care products such as disposable diapers, facial or toilet tissue, and paper towels is considered insignificant.
Because dioxins bioaccumulate in the food chain, the major route of human exposure is through food, especially fish, meat, and dairy
products. It has been estimated that food accounts for 98% of total adult exposures. TCDD has been detected in fish from Saginaw Bay, contaminated sections of the Great Lakes, and some Michigan rivers, although levels in both the water and fish have decreased over time.
Use of products known or thought to be contaminated by dioxins and furans has been significantly restricted in the last few decades. The U.S. Environmental Protection Agency (EPA) has removed the herbicide 2,4,5-T from the commercial market, and heat-transfer agents using polychlorinated biphenyls (PCBs), which form dioxins during combustion, are being phased out of production and use.
|
(1) Whom would you contact to obtain further information about the herbicide used? _________________________________________________________________ _________________________________________________________________ |
Who’s at Risk
❑ Workers in the chemical industry may have increased likelihood of exposure.
❑ Overt clinical effects from dioxin exposure have been seen primarily after major industrial accidents involving these compounds.
❑ Fetuses and nursing infants may be at increased risk of dioxin exposure if the mothers have been exposed.
Dioxins are no longer manufactured in the United States, except for small amounts used for scientific research. Nevertheless, some workers may encounter dioxins as contaminants in certain industrial processes such as the manufacture of chlorinated herbicides, germicides, and organic solvents. Firefighters and cleanup crews involved with capacitor or transformer fires and hazardous waste accidents may also be exposed to dioxins through dermal absorption or inhalation. Municipal and waste incinerator workers may encounter dioxins in smoke, gases, or fly ash formed during combustion processes. Major industrial accidents have been the source of most overt clinical effects from dioxins.
Because TCDD crosses the placenta and accumulates in breast milk, fetuses and nursing infants of contaminated mothers are potentially at increased risk of exposure. Ingestion of contaminated soil due to pica or normal hand-to-mouth activities may contribute to a child’s dioxin body burden. No cases of dioxin toxicity have been reported by either of these routes.
|
(2) Would you consider the patient described in the case study at increased risk from dioxin exposure? Explain. _________________________________________________________________ _________________________________________________________________ _________________________________________________________________ |
Biologic Fate
❑ Dioxins and furans accumulate in adipose tissue.
❑ In animals, the major routes of absorbed TCDD elimination are through lactation and excretion of urine and bile.
Dioxins enter the body by ingestion, inhalation, and dermal absorption. In humans, the following percentages of total absorption have been reported for various exposure routes: inhalation, 25% to 29%; ingestion of contaminated soil, 20% to 26%; and ingestion of contaminated fish, 50% to 80%. Skin was found to absorb up to 3% of the dioxin in contaminated soil.
The metabolic pathway of TCDD for humans has not been established. TCDD orally administered to animals is metabolized relatively slowly, but once metabolites are formed, they are rapidly excreted in the urine and bile. Metabolism is primarily by hepatic detoxification, with the major metabolites consisting of hydroxylated or methoxylated TCDD derivatives, which are then excreted as glucuronide and sulfate conjugates. Unabsorbed TCDD is excreted through direct elimination in the feces. Because of the lipophilic nature of milk, nursing females decrease their body burden of TCDD through lactation. Dioxins distribute to organs according to lipid content and readily accumulate in body fat. In the general population, the background level of TCDD in adipose tissue may be as high as 20 ppt.
The half-life of TCDD ranges from several hours (on the surface of plants) to 7.1 years (in human serum and adipose tissue). On the soil’s surface, where TCDD undergoes photodegradation when exposed to ultraviolet light, the half-life is 1 to 3 years. Beneath the soil’s surface, the half-life of TCDD can be over 10 years.
|
(3) Assuming the utility company mentioned in the case study sprayed the area with a dioxin-containing herbicide, when will it be safe for the children to return to the area to play? _________________________________________________________________ _________________________________________________________________ _________________________________________________________________ |
Physiologic Effects
❑ Only two effects of dioxin exposure have been confirmed in humans: chloracne and transient mild hepatotoxicity.
Dioxins are very toxic to some animal species, but the evidence for corresponding toxicity in humans has not been established. No deaths due to systemic dioxin toxicity in humans have been reported. Only two clinical effects have been repeatedly observed in exposed populations: chloracne and transient hepatic effects. Soft-tissue sarcomas, lymphomas, peripheral neuropathy, birth defects, and reproductive effects have been studied but remain unconfirmed. Because human dioxin exposure always involves mixed exposures, the specific effects of dioxin are difficult to ascertain.
In some animals, TCDD is a potent carcinogen and causes reproductive effects and birth defects. TCDD’s toxicity also appears in animals as pathologic effects in the liver, peripheral nerves, hematopoietic and reticuloendothelial systems, and skin. Acute toxicity develops after a latent period of 7 to 10 days, after which the animals experience a rapid wasting syndrome not seen in humans.
Dermatologic Effects
❑ Chloracne is a hallmark of dioxin toxicity in humans and can persist for years after exposure ceases.
Acneform lesions may appear as early as 1 to 3 weeks after dioxin exposure. The lesions are small, pale yellow cysts, which arise from altered differentiation of acinar sebaceous basal cells into keratinocytes. The lesions primarily involve the face, especially the periorbital, temporal, and malar areas, as well as the upper body. Most cases of chloracne resolve in 1 to 3 years; one case has been reported to persist for as long as 32 years. Although TCDD is one of the most potent acne-producing agents, chloracne can also be
caused by chloro- and bromonaphthalenes, polychlorinated and polybrominated biphenyls (PCBs, PBBs), pentachlorophenol, and tetrachlorobenzene.
Chloracne is the only overt effect of dioxin exposure in human populations; however, the absence of this effect does not rule out dioxin exposure. There is no acceptable dose-response model for chloracne in exposed human populations. It may develop weeks or months after exposure and may be dependent upon individual predisposition. It can result from inhalation, ingestion, or dermal contact and may indicate systemic toxicity.
Six workers who developed chloracne and other illnesses after an industrial accident in Germany had an estimated mean TCDD body burden shortly after the accident of 44 micrograms (µg) with a range of 9.7 to 124 µg. Based on this data, researchers believe that a body burden of 9.7 µg as measured in adipose tissue may be the lowest observable adverse effect level for TCDD-related chloracne in humans. Thirty-two years after exposure, these workers still had detectable levels of TCDD in adipose tissue, and one still had chloracne.
In addition to chloracne, other reported dermal effects include hyperpigmentation, hirsutism, increased skin fragility, and vesicular eruptions on exposed areas of the skin. Concomitant exposure to other agents, however, may have contributed to these effects.
Neurologic Effects
❑ Human epidemiologic data are inconsistent regarding TCDD’s neurotoxic effects.
Although dioxins have produced neurotoxic results in laboratory animals, data from human studies have been inconsistent and inconclusive. Peripheral nervous system involvement was studied 6 years after the Seveso accident in 152 victims with chloracne. No case of peripheral neuropathy was found; however, subclinical peripheral nerve impairment was reported in 16 victims. A study of railroad workers exposed to TCDD during cleanup of a tank car spill reported that 43 of 45 workers had findings suggestive of peripheral neuropathy 6 years after exposure, but the validity of the study has been questioned and numerous other epidemiologic studies have found no evidence of neurologic impairment due to dioxin exposure.
Hepatic Effects
❑ Persons exposed to dioxin-contaminated agents had transiently elevated liver enzyme levels.
Although hepatotoxicity has been observed in a variety of animal species, there is no evidence that TCDD causes long-term hepatotoxicity in humans. Some studies have shown a transient increase in liver enzymes without clinical disease. A study of Seveso children potentially exposed to TCDD revealed hepatomegaly and slightly
elevated serum levels of GGTP (GGT) and SGPT (ALT); however, no apparent liver function impairment was found, and the enzyme levels subsequently returned to normal. Studies of liver function in people in the Times Beach, Missouri, incident also suggested sub-clinical hepatic effects. Epidemiologic studies of TCDD-exposed workers, however, have shown no difference in serum levels of hepatic enzymes compared with those of matched controls.
Reproductive and Developmental Effects
❑ Evidence is lacking that TCDD is a reproductive toxicant in humans.
Studies of Vietnam servicemen possibly exposed to Agent Orange revealed no overall increase of debilitating birth defects in progeny. A study of the Missouri incident encompassing 410 births reported no statistically significant increase in risk ratios for infant, fetal, and perinatal death, low birth weight, or for several subcategories of birth defects. Studies of the Seveso area have also failed to demonstrate increased risk of birth defects due to dioxin exposure.
Numerous reproductive and developmental effects have been noted in animals exposed to dioxins. These effects include spontaneous abortions in the Rhesus monkey and birth defects in a number of other mammalian species.
Carcinogenic Effects
❑ TCDD carcinogenicity in animals is well established; however, human epidemiologic data are inconclusive.
❑ EPA and NIOSH consider TCDD alone to be a probable human carcinogen, and a “cancer promoter” in conjunction with certain other chemicals.
Although evidence for human carcinogenicity is inconclusive, TCDD is irrefutably an animal carcinogen. The exact mechanisms of action are unknown; therefore extrapolation from animal studies to humans is impossible. Before TCDD’s potential human carcinogenic capabilities can be determined, long-term epidemiologic studies in humans are necessary and concomitant exposures must be further defined.
Much of the research in humans has focused on TCDD’s ability to induce soft-tissue sarcomas. Epidemiologic studies have produced conflicting results. A study of Swedish workers potentially exposed to TCDD-containing phenoxyacetic acid herbicides indicates an association between herbicide exposure soft-tissue sarcomas at various sites, stomach cancers, and lymphomas. This study was criticized for bias recall, and results of similar research in Finland and New Zealand refute the Swedish study. Of the more than 200 workers exposed to dioxin in a plant explosion in Nitro, West Virginia in 1949, 122 developed chloracne. However, no excess deaths due to cancer were found among the workers 30 years after the incident.
EPA and the National Institute of Occupational Safety and Health (NIOSH) consider TCDD to be a “cancer promoter” in conjunction with certain other chemicals. NIOSH classifies TCDD as a “potential
occupational carcinogen.” EPA considers TCDD to be a probable human carcinogen. The category of “probable human carcinogen” indicates that EPA considers animal evidence of carcinogenicity “sufficient” and human evidence “inadequate.”
Immunotoxic Effects
❑ Assessment of immunologic effects in TCDD-exposed patients has no clinical value.
Abundant animal data indicate that immunotoxicity may be one of the most sensitive toxicologic outcomes for TCDD. Investigations of this endpoint in humans are limited; thus, the importance of TCDD-induced immunotoxicity in humans cannot be evaluated and assessing this endpoint in patients has no clinical value.
|
(4) If the child described in the case study had been exposed to dioxin, what health effects might he experience? _________________________________________________________________ _________________________________________________________________ (5) What is the child’s risk of developing cancer from the exposure described in the case study? _________________________________________________________________ _________________________________________________________________ |
Clinical Evaluation
History and Physical Examination
❑ A detailed history and physical examination are needed to discover all possible sources of exposure.
Because dioxins are extremely persistent and bioaccumulate in the food chain, a patient’s entire exposure history must be taken into account. If exposure to dioxins is suspected, the history should include the following:
past and present occupational duties
military service in Vietnam involving handling or spraying of herbicides
proximity of residence or work to waste sites or incinerators
contact with herbicides, fungicides, and germicides
dietary composition and habits
history of breast-feeding
In the physical examination, the clinician should review all systems, with particular attention to the skin, liver, and peripheral nervous system. Discovery of a person with dioxin toxicity should suggest that others, such as coworkers, friends, or family members, may have been exposed similarly.
Signs and Symptoms
❑ Chloracne and elevated liver enzymes may be noted in persons with dioxin toxicity.
The onset of symptoms after acute exposure to TCDD-containing substances can take days to weeks. Symptoms reported by hazardous waste cleanup crews who later developed chloracne were skin, eye, and respiratory tract irritation; headache; dizziness; and nausea. Other reported symptoms of exposure to TCDD-contaminated compounds include loss of appetite, weight loss, loss of libido, sensory changes, severe fatigue, pain in the abdomen and extremities, memory impairment, uncharacteristic bouts of anger, and insomnia. Many of these signs and symptoms are nonspecific and commonly observed with exposure to other chemicals. Since exposure to TCDD always involves mixed exposures, it is impossible to state with certainty that dioxins caused these effects.
Laboratory Tests
Direct Biologic Indicators
❑ Blood and adipose tissue tests for TCDD levels are not recommended unless the exposure is massive.
Adipose tissue and blood serum analyzed for the presence of TCDD by gas chromatography-mass spectrometry (GC-MS), can be quantified to 100 parts per quadrillion. Some researchers believe that serum levels correlate with adipose tissue levels in persons with long-term exposure. However, analyses of serum or fat TCDD levels
by GC-MS are expensive and time-consuming; therefore, unless exposure has been massive, they are not recommended. Levels of 20 ppt in adipose tissue have been measured in persons with no known exposure to TCDD. In 50 persons exposed in the Missouri incident, the mean ratio of the TCDD level in adipose tissue to that in serum was 1.09 after adjusting both samples for lipid content. Some of the exposed people in the Seveso, Italy, accident had TCDD levels a thousand times greater than the average found in the general unexposed population and did not experience illness.
Indirect Biologic Indicators
❑ Cases of chloracne should be reported to public health authorities.
Chloracne may be an indicator of dioxin toxicity, although its absence does not necessarily rule out exposure. New cases should be reported to local or state public health authorities. In the absence of chloracne, liver function tests may be the most sensitive indicator of dioxin exposure. This measure, however, is nonspecific, and normal results do not rule out significant exposure or elevated body burden.
|
(6) What issues should you address in obtaining the medical history of the child described in the case study? _________________________________________________________________ _________________________________________________________________ (7) What would be the focus of the physical examination if dioxin exposure were a possibility? _________________________________________________________________ (8) What laboratory tests, if any, would you use to evaluate this child? _________________________________________________________________ (9) What symptoms might develop after exposure to a dioxin-contaminated compound? _________________________________________________________________ |
Treatment and Management
❑ No antidote for dioxin toxicity is known; symptomatic and supportive care is the only therapy.
Treatment of chronic exposure to dioxin-containing agents is primarily supportive, and depends on the agent involved and the presenting signs and symptoms. It is most important to remove the patient from the source of exposure. Chloracne is often refractory and unresponsive to common acne treatments. Long-term topical treatment with dilute retinoic acid and administration of tetracycline to treat secondary pustular follicles have been used. In severe cases, acne surgery or dermabrasion may be effective.
|
(10) What is likely to be the cause of the child’s rash and other symptoms mentioned in the case study? What treatment will you recommend? _________________________________________________________________ _________________________________________________________________ _________________________________________________________________ |
Standards and Regulations
Before 1965, it was not uncommon for TCDD to be present as a product contaminant in herbicides at concentrations exceeding 30 mg/kg (30ppm). Today, however, the concentration of TCDD as a product contaminant is limited to 0.01 to 0.05 ppm.
Workplace
Air
❑ OSHA has not set a workplace standard for dioxins.
No permissible exposure limit (PEL) has been set for dioxin by the Occupational Safety and Health Administration (OSHA), which is responsible for workplace standards. NIOSH considers TCDD a “probable” human carcinogen and recommends that workplace exposure be reduced to the lowest feasible level.
Environment
Air
❑ Dioxins in ambient air are not currently regulated.
EPA currently has no standard for TCDD or other dioxins in ambient air.
Water
❑ The legal level for TCDD in drinking water is 0.05 ppt.
The EPA has set a maximum contaminant level for TCDD in drinking water of 5×10−8 mg/L, or 0.05 ppt. A maximum contaminant level goal of zero will be proposed by EPA in June 1990.
Food
❑ Fish is the only food regulated for dioxin content.
Fish is the greatest potential food source of dioxin contamination. Dioxins readily adsorb to aquatic sediment, and can be ingested by bottom-feeding organisms that are subsequently ingested by preda tory fish, thereby bioconcentrating these substances. The U.S. Food and Drug Administration’s (FDA) limit for dioxins in fish is 25 ppt TCDD. Since livestock are herbivores, the dioxin bioconcentration potential in these mammals is minimal unless they are grazing on contaminated pasture. There are no standards for meats or other foodstuffs.
Suggested Reading List
General
Barinaga M. Agent Orange: Congress impatient for answers. Science 1989;245:249–50.
Fishbein L. Health-risk estimates for 2,3,7,8-terachlorodibenzo-p-dioxin: an overview. Toxicol Ind Health 1987;3:91–134.
Gochfeld M, Gallo M, Kahn P. Dioxin exposure and health. N J Med 1988;85:907–12.
Skene SA, Dewhurst IC, Greenberg M. Polychlorinated dibenzo-p-dioxins and polychlorinated dibenzofurans: the risks to human health. A review. Hum Toxicol 1989;8:173–204.
Exposure Assessment and Epidemiologic Studies
Bertazzi PA, Zocchetti C, Pesatori AC, Guercilena S, Senarico M, Rodice L. Ten-year mortality study of the population involved in the Seveso incident in 1976. Am J Epidemiol 1989;129:1187–1200.
Bond GG, McLaren EA, Lipps TE, Cook RR. Update of mortality among chemical workers with potential exposure to the higher chlorinated dioxins. J Occup Med 1989;31:121–3.
Centers for Disease Control. Preliminary Report: 2,3,7,8-tetrachlorodibenzo-p-dioxin exposure to humans— Seveso, Italy. JAMA 1989;261:831–2.
Kimbrough RD, Falk H, Stehr P, Fries G. Health implications of 2,3,7,8-tetratchlorodibenzodioxin (TCDD) contamination of residential soil. J Toxicol Environ Health 1984;14:47–93.
Ott MG, Olson RA, Cook RR, Bond GG. Cohort mortality study of chemical workers with potential exposure to the higher chlorinated dioxins. J Occup Med 1987:29:422–9.
Stehr-Green PA, Andrews JS, Webb KB, Hoffmann RE, Schramm WF. An overview of the Missouri USA dioxin studies. Arch Environ Health 1988;43:174–7.
Stern AH, Munshi AA, Goodman AK. Potential exposure levels and health effects of neighborhood exposure to a municipal incinerator bottom ash landfill. Arch Envir Health 1989;44:40–8.
Thiess AM, Frentzel-Beyme R. Mortality study of persons exposed to dioxin following an accident which occurred in the BASF on 13 November, 1953. Am J Ind Med 1982;3:179–89.
Webb K, Evans RG, Stehr P, Ayres SM. Pilot study on health effects of environmental 2,3,7,8-TCDD in Missouri. Am J Ind Med 1987;11:685–92.
Dermal Effects
Caputo R, Moti M, Ermacora E, et al. Cutaneous manifestations of tetrachlorobenzo-p-dioxin in children and adolescents. Follow-up 10 years after the Seveso, Italy, accident. J Am Acad Dermatol 1988;19:812–9.
May G. Chloracne from the accidental production of tetrachlorobenzodioxin. Br J Ind Med 1982;39:128–35.
Moses M, Prioleau PG. Cutaneous histologic findings in chemical workers with and without chloracne with past exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin. J Am Acad Dermatol 1985;12:497–506.
Reproductive and Developmental Effects
Mastroiacovo P, Spagnolo A, Marni E, Meazza L, Bertollini R, Segni G. Birth defects in the Seveso area (Italy) after TCDD contamination. JAMA 1988:259:1668–72.
Silbergeld EK, Mattison DR. Experimental and clinical studies on the reproductive toxicology of 2,3,7,8-tetrachlorodibenzo-p-dioxin. Am J Ind Med 1987;11:131–44.
Neurologic Effects
Barbieri S, Pirovano C, Scarlato G, Tarchini P, Zappa A, Maranzana M. Long-term effects of 2,3,7,8-tetrachlorodibenzo-p-dioxin on the peripheral nervous system. Clinical and neurophysiological controlled study on subjects with chloracne from the Seveso area. Neuroepidemiology 1988;7:29–37.
Levy CJ. Agent Orange exposure and posttraumatic stress disorder. J Nerv Ment Dis 1988;176:242–5.
Carcinogenic Effects
Fingerhut MA, Halperin WE, Honchar PA, Smith AB, Groth DH, Russel WO. An evaluation of reports of dioxin exposure and soft tissue sarcoma pathology among chemical workers in the United States. Scand J Work Environ Health 1984;10:299–303.
Pratt CB, George SL, O’Connor D, Hoffman RE. Adolescent colorectal cancer and dioxin exposure (letter). Lancet 1987;2:803.
Wiklund K, Dich J, Holme L-E. Soft tissue sarcoma risk in Swedish licensed pesticide applicators. J Occup Med 1988;30:801–4.
Woods JS, Polissar L, Severson RK, Heuser LS, Kulander BG. Soft tissue sarcoma and non-Hodgkin’s lymphoma in relation to phenoxyherbicide and chlorinated phenol exposure in western Washington. J Natl Cancer Inst 1987;78:899–910.
Related Government Publications
Agency for Toxic Substances and Disease Registry. Toxicological profile for 2,3,7,8-tetrachlorodibenzo-p-dioxin. Atlanta: U.S. Department of Health and Human Services, Public Health Service, 1990. NTIS report no. PB/ 89/214522/AS.
Centers for Disease Control. Preliminary report: 2,3,7,8-tetrachlorodibenzo-p-dioxin exposure to humans— Seveso, Italy. MMWR 1988;37(48):733–6.
Centers for Disease Control. Serum 2,3,7,8-Tetrachlorodibenzo-p-dioxin levels in Air Force health study participants—preliminary report. MMWR 1988;37(20):309–11.
Centers for Disease Control. Serum dioxin in Vietnam-era veterans—preliminary report. MMWR 1987;36 (28):470–5.
Centers for Disease Control. Results of a pilot study of health effects due to 2,3,7,8-tetrachlorodibenzodioxin contamination—Missouri. MMWR 1987;36(28):470–5.
Environmental Protection Agency. Estimating exposures to 2,3,7,8-TCDD. Washington,DC: Exposure Assessment Group, Office of Environmental Assessment, 1987. Report no. EPA/600/6–88/005A.
National Cancer Institute. Dioxins and dibenzofurans in carcinogenesis, 1980–1986. In: Greenlee, WF, ed. Oncology overview. Washington, DC: International Cancer Data Bank, National Cancer Institute, 1987.
Answers to Pretest and Challenge Questions
Pretest is found on page 1. Challenge questions begin on page 4.
-
A call to the medical director or the health and safety department of the utility company should provide the answer to the type of herbicide used and its contents. Dioxin-containing herbicides are not likely to have been used in this case since they are no longer being manufactured in the United States.
-
The patient could be at increased risk of dioxin exposure because he is living in an area of possible soil contamination. Normal hand-to-mouth activity of children can result in ingestion of contaminated soil. Because of the child’s age, it is unlikely that he has pica (the abnormal ingestion of nonfood items, commonly found in children aged 2 to 6 years), which could significantly increase the boy’s soil intake. Children consume large quantities of milk, which can be a source of dioxins if it comes from cows grazing on contaminated vegetation. The small amount of dioxin leached from paper milk cartons is negligible.
If the family raises its own foodstuffs and if the previous owner of the farm used contaminated herbicides that still may be present in the soil, the current root crops could contain small amounts of dioxins. (Evidence for translocation of dioxins is sparse and inconclusive.) Even though production of herbicides such as 2,4,5-T were discontinued in the United States in 1976, the half-life of dioxin in soil may be 10 years or more, depending on the type of soil. However, this source is likely to be insignificant in terms of health risk.
-
Even if the herbicide did contain dioxins, these compounds photodegrade rapidly, resulting in a half-life on vegetation of several hours and several days in air. The half-life of dioxins in surface soil is 1 to 3 years, while dioxins beneath the soil surface could have a half-life of 10 years or more. However, dermal absorption from TCDD-contaminated soil is less than 5%. If the children do not ingest the soil, the danger is minimal.
-
The primary human health effects of dioxin exposure are chloracne, and secondarily, hepatomegaly, elevated liver enzyme levels, and possibly peripheral neuropathy (subclinical changes in nerve conduction velocity).
-
Although dioxins are proven carcinogens in some animals, their carcinogenic effect in humans requires further study. Even if the herbicide contained TCDD, the risk of cancer for this patient is likely to be insignificant from a one-time exposure that caused no acute effects.
-
Some of the issues you might address in obtaining the medical history are the following: the type and extent of farming carried on by the family; their lifestyle before coming to this farm; dietary habits, including present or past pica in the child.
-
During the physical examination, the skin should be carefully examined for evidence of rash, particularly chloracne. Chloracne is a papular, sometimes pustular, lesion located principally on the upper facial areas. The onset of chloracne is not acute, as was the rash described in the case study. In addition, an examination of the abdomen should be conducted, looking for hepatomegaly or hepatic tenderness. A neurologic examination might also be undertaken, with a mental status examination to assess more subtle CNS effects.
-
Analytical tests for TCDD (adipose tissue, serum) are very specialized and expensive, and generally are not recommended in clinical practice, especially since interpretation in individual cases is difficult. Dioxins may be associated with hepatotoxicity, and liver function tests would be appropriate if there has been known exposure to dioxin.
-
Symptoms associated with acute exposure to dioxin-containing substances include skin and mucous membrane irritation, headache, fatigue, abdominal pain, memory and personality changes, and insomnia. However, such symptoms are nonspecific and may have other etiologies.
-
The cause of the child’s rash is more likely to be poison ivy, which is common in the wooded areas of the Midwest, an allergy, or exposure to some chemical other than a dioxin. This conclusion is suggested by the acute onset of the rash, its appearance, and its burning nature. Referral to a dermatologist may be warranted if standard measures of treating the rash are not efficacious. The child’s symptoms of headache and stomachache may be a result of such factors as stress, food intolerance, or viral infection. If symptoms do not resolve within a day or two, further investigation may be warranted.

















