
22 Cholinesterase-Inhibiting Pesticide Toxicity
|
Environmental ALERT…
|
This monograph is one in a series of self-instructional publications designed to increase the primary care provider’s knowledge of hazardous substances in the environment and to aid in the evaluation of potentially exposed patients. See page 23 for more information about continuing medical education credits and continuing education units.
|
Guest Contributor: |
Laurence J.Fuortes, MD; Amadu Ayebo, PhD; Burton Kross, PhD |
|
Guest Editor: |
Sheldon Wagner, MD |
|
Peer Reviewers: |
John Ambre, MD, PhD; Charles Becker, MD; Jonathan Borak, MD; Joseph Cannella, MD; Richard J.Jackson, MD, MPH; Howard Kipen, MD; Jonathan Rodnick, MD; Brian Wummer, MD |

U.S. DEPARTMENT OF HEALTH & HUMAN SERVICES
Public Health Service
Agency for Toxic Substances and Disease Registry
Case Study
A 30-year-old, comatose male with miosis, diaphoresis, and incontinence
You are alerted that a 30-year-old, unconscious male is being brought by ambulance to the Emergency Department where you are on duty. While at home, the patient suddenly developed headache, dizziness, weakness, nausea, vomiting, and diarrhea. En route to the Emergency Department, he lost consciousness and experienced urinary and fecal incontinence.
When the patient arrives at the Emergency Department, you note that he has fixed pinpoint pupils, generalized paralysis, fasciculations, and is unresponsive to deep pain. Corneal and gag reflexes are absent. He has profuse salivation, diaphoresis, and excess lacrimation. Vital signs include the following: blood pressure 140/90 mm Hg, temperature 99.2°F (37.2°C), pulse 58 beats/minute and regular. Rales are noted during chest auscultation. Heart examination is unremarkable except for an S4 gallop. Abdominal examination reveals no detectable masses, organomegaly, or hyperactive bowel sounds. Mucoid secretions are suctioned from the trachea at the time of intubation, and mechanical respiratory support is instituted. You initiate treatment with Narcan* for possible opiate ingestion, with no effect.
From the patient’s brother, you learn that the patient returned yesterday from a 5-day vacation in Arizona. This morning he changed into work clothes and began mixing pesticides for subsequent tree spraying in their family orchard. About noon, the patient became nauseated and started sweating profusely. The brother cannot recall any unusual events before his brother’s illness and assures you that they had performed the tree-spraying operations many times in the past with no ill effects.
After treatment and antidotal therapy, the patient improves remarkably—he has spontaneous respirations and regains consciousness within 4 hours. Mechanical ventilation is discontinued, and the patient is well enough to be discharged 3 days after admission.
|
* |
Use of trade names is for identification only and does not imply endorsement by the Public Health Service or the U.S. Department of Health and Human Services. |
![]()
(a) What should be included in this patient’s problem list? What is the differential diagnosis?
_________________________________________________________________
(b) What important information did the patient’s brother provide? What further information will you seek?
_________________________________________________________________
_________________________________________________________________
(c) What laboratory tests could you order to confirm your diagnosis?
_________________________________________________________________
(d) What was the treatment and antidotal therapy that resulted in the patient’s remarkable recovery?
_________________________________________________________________
_________________________________________________________________
Answers to the Pretest questions are on page 21.
Exposure Pathways
❑ Most occupational pesticide poisonings result from dermal contact during handling or contact with residues on sprayed plants and soils.
❑ As insects develop greater resistance to insecticides, new toxic compounds are necessary.
The use of organophosphate and carbamate compounds (Figure 1) as insecticides began in the 1930s and has increased markedly since many organochlorine insecticides were banned in the 1970s. In contrast to organochlorine insecticides, organophosphate and carbamate insecticides degrade rapidly in the environment and do not accumulate or concentrate in the food chain. Thus, organophosphate and carbamate insecticides have less potential for chronic health effects or environmental contamination than do organochlorine insecticides and pose less risk to consumers of food products. However, organophosphate and carbamate compounds have a greater potential for acute toxicity in humans than do chlorinated compounds. Even among the organophosphate or carbamate pesticides, however, a wide spectrum of potency exists. As insects develop greater resistance, the trend is to use more potent, and consequently, more lipid-soluble and longer-lasting insecticides.
Figure 1. General chemical structures of organophosphate and carbamate pesticides
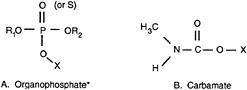
*R1 and R2 are usually alkyl groups (typically ethyl or methyl); X is also commonly an alkyl group that is replaced by a hydrogen atom during the “aging” of the organophosphate-enzyme complex. Pesticides that have a sulfur in place of the oxygen double-bonded to the phosphorus are called thiophosphates.
❑ Improper storage of pesticides often leads to accidental ingestion and contact by children and adults.
Most organophosphate and carbamate insecticides are used for crop spraying in commercial agriculture. Approximately 75% of all insecticides are used on three crops: cotton, corn, and soybeans. Ethyl parathion (Parathion), which will soon be phased out of production, and malathion are the most widely used organophosphate insecticides. Aldicarb (Temik) and carbaryl (Sevin) are well-known carbamate insecticides. Organophosphate and carbamate compounds degrade in the environment at varying rates; half-lives range from days to months, although generally they are longer in dry climates and at low temperatures.
Most occupational exposures to organophosphates and carbamates occur from skin absorption, although inhalation may be an important route of exposure during pesticide manufacture and application. Skin absorption can occur when dermal contact is made during handling and application of the insecticide, and when contact is made with chemical residues on plants, fruits, and foliage; in soil; and on dust particles after spraying. Some organophosphate insecticides (thiophosphates) are applied in the sulfur-containing (-thion) form but readily undergo desulfuration (removal of a sulfur atom from the molecule) to form a more toxic oxygen-containing (-oxon) form. In the field, this conversion occurs slowly under the influence of oxygen and light, often producing residues that are not only more toxic but more readily absorbed through the skin. The conversion can also occur during storage, making it important to promptly dispose of unused sulfur-containing pesticides.
Federal law prohibits field workers and structural pest control workers from entering a sprayed area without protective equipment until a specified interval of time has elapsed. Premature entry into sprayed fields or fumigated homes has caused acute organophosphate toxicity and death. Poisonings have occurred even when persons entered treated areas after the prescribed interval.
Although a few cases of chronic and mildly acute dietary poisonings have been reported, hazards to consumers from surface contamination of food appear to be minimal. If fruits and vegetables are thoroughly washed and the time elapsed between spraying and consumption is sufficient for insecticide degradation, pesticide poisoning is unlikely. However, some organophosphate and carbamate insecticides act systemically and can be translocated into foliage and fruit when applied at an improper time before harvest. Outbreaks of aldicarb intoxication, which affected hundreds of watermelon and cucumber consumers, occurred during the 1980s from misapplication of this systemic-acting insecticide.
Organophosphate and carbamate compounds, either alone or in combination with other insecticides, account for most pesticide-related poisonings. Poisonings have resulted from suicidal, homicidal, and accidental exposures. With improper storage, children, as well as adults, can mistake these chemicals for liquor, medicine, or other consumables. Pesticides usually contain solvent carriers such as toluene or xylene that are labeled as inert ingredients but may produce toxic effects in a pesticide ingestion. (The inert ingredients included on the labels of most pesticide containers refer to their application as a pesticide and not to their toxicologic activity.) Hence, the treatments suggested on pesticide container labels often are incomplete. Consumers should consult a physician or the regional poison control center if poisoning is suspected.
Who’s at Risk
❑ Farm workers, pesticide formulators and applicators, and their families are at greatest risk of exposure to pesticides.
Seasonal or migrant farm workers (estimated 2.5 million), agricultural workers involved in the formulation and application of pesticides, and structural pest-control workers have the highest incidence of pesticide exposure. The improper use of pesticide containers as drinking or cooking utensils by field workers and their families and contamination of open water containers during aerial pesticide spraying have resulted in exposure. Other persons who may be exposed to organophosphates include workers in chemical manufacturing plants, truckers, longshoremen, chemical warfare (nerve gas) producers, and military personnel.
❑ Proper precautions should be taken while mixing or applying insecticides because dermal absorption can be substantial and even lead to death.
Despite extensive regulation, labeling, and educational efforts, the public remains unaware that fatal amounts of organophosphates can be absorbed through the skin. Because dermal absorption is so great, workers have been poisoned by wearing work clothes contaminated with these chemicals. Laundering may not be effective in removing some organophosphates; contaminated clothing may have to be burned, especially leather articles. Although the concentrations and toxicities of insecticides for home use are sometimes less than the concentrations and toxicities of the insecticides used in commercial agriculture, home gardeners should avoid all dermal contact and follow the label instructions carefully.
❑ Children are at increased risk of exposure to pesticides and are more susceptible to their adverse health effects.
Children can be exposed to pesticides by playing in areas where the chemicals have been sprayed, spilled, or improperly stored, and by playing with packages or instruments used in spraying. The mouthing and exploratory behaviors of young children and their tendency to play on the floor or ground increases their risk of exposure to pesticides or pesticide residues. Infants under 6 months of age have incompletely developed acetylcholinesterase systems and immature livers, and this may increase the susceptibility of young infants to cholinesterase-inhibiting pesticides.
Emergency personnel responding to pesticide spills, accidents, or poisonings are at risk not only of primary contamination but also of secondary contamination from contaminated persons and equipment. Appropriate protective clothing should be worn and care should be exercised during the decontamination of victims. In one case, the hospital emergency department staff were affected by xylene, which vaporized from the vomitus of a patient who had ingested a pesticide; the vomitus had been left in an open basin for an extended period of time.
Other persons who may be susceptible to the adverse effects of organophosphate pesticides include a small percentage of the population who have an atypical variant of plasma cholinesterase. This genetic abnormality decreases the amount of cholinesterase available and renders persons unusually sensitive to succinylcholine, a paralytic
agent used in emergency intubations and abdominal surgeries. They may also be vulnerable to poisoning when cholinesterase activity is further depressed by insecticide exposure (although this has not been proven). Low plasma cholinesterase levels are also exhibited by long-distance runners; women in early pregnancy or using birth control pills; and persons who have advanced liver disease, chronic alcoholism, malnutrition, or dermatomyositis. Persons who have asthma and are exposed to organophosphate pesticides may be at increased risk because many of these compounds cause narrowing of the airways, which can exacerbate breathing difficulties.
|
Additional information for the case study: During your conversation with the patient’s brother, you learn that a spill occurred in the pesticide formulation area 2 days ago. The patient’s brother had been mixing several chemicals, including an organophosphate insecticide, and spilled the mixture on the floor and on his clothes. After quickly removing his clothing, which consisted of rubber gloves, a cartridge respirator, a pair of coveralls kept in the formulation area, and his street clothes, he immediately showered. He cleaned the floor in the formulation area using a mixture of sand and other inert materials kept on hand for that purpose. He then buried the spilled contents. He took precautions to avoid contact with the chemicals during clean-up, and his wife laundered the coveralls separately from their other clothing. He saw no reason to mention the incident to his brother, who was on vacation at the time. (1) What are the most likely routes of exposure for the patient in the case study? _________________________________________________________________ _________________________________________________________________ (2) Who else could have been, or yet may be, at risk from this accident? _________________________________________________________________ _________________________________________________________________ |
Biologic Fate
❑ Organophosphate and carbamate insecticides can cause systemic poisoning when absorbed by any route.
❑ Thiophosphate insecticides may be converted in vivo and in the environment to an organophosphate that has greater skin penetration and clinical effects than the parent compound.
Organophosphate and carbamate pesticides are absorbed readily by inhalation, ingestion, and skin absorption. The water solubility of some organophosphate and carbamate pesticides allows them to be absorbed by plants and also to act as systemic poisons in both insects and mammals. Because these compounds are distributed rapidly throughout the body, they typically are associated with rapid onset of symptoms, rarely longer than a few hours after a toxic exposure. However, the more lipid-soluble organophosphates, such as chlorfenthion and fenthion, can undergo initial lipid storage with subsequent redistribution and may not produce medical crisis for several days. Symptoms can persist for several weeks and periodic relapses can occur, requiring additional therapy.
In the body, as well as in the environment, some organophosphates can be converted from the -thion form to the more toxic -oxon form; rates of conversion vary widely but are more rapid in vivo than in the environment. In the body, conversion is brought about chiefly by hepatic microsomal esterases. Ultimately, both the -thion and -oxon forms are usually metabolized to alkyl phosphates and other products that are of relatively low toxicity and are excreted rapidly. Carbamates are also metabolized in the liver, and the products are excreted in urine without evidence of significant accumulation.
Physiologic Effects
Neurobehavioral Effects
❑ Acetylcholinesterase, which is critical to control of nerve impulse transmission, is inhibited by organophosphate and carbamate pesticides.
Organophosphate and carbamate compounds share a common pathophysiology—they combine with and thereby inhibit cholinesterase enzymes, of which acetylcholinesterase (AChE) in nerve tissue is the most important. Inactivation of AChE results in accumulation of acetylcholine at the neuroreceptor transmission site, resulting in massive overstimulation of the cholinergic system.
Initially, a weak reversible bond is formed between AChE and the organophosphate or carbamate pesticide. With time, however, a more permanent AChE-phosphate bond forms that inactivates the
❑ Because organophosphates may “age” in the body, antidote(pralidoxime) must be administered within 12 to 48 hours after exposure to be effective.
enzyme and requires an antidote to break. This process is known as “aging.” If an antidote (pralidoxime [2-PAM]) is not given within 24 to 48 hours after most organophosphate exposures, the AChE-phosphate bond becomes so strong that physiologic recovery will depend on de-novo synthesis of AChE. At the nerve junction, AChE is restored in an average of 2 weeks; in the body as a whole, it may require 1 to 3 months to restore enzyme activity to near normal levels.
Compared with the AChE-phosphate bond, the AChE-carbamate bond is relatively weak, and because the AChE-carbamate complex is inherently transient, aging does not occur. Therefore, carbamate pesticides produce an acute toxicity of much shorter duration than organophosphate pesticides and usually without persistent sequelae. Antidotes in the form of enzyme reactivators (pralidoxime) are rarely required treatment for carbamate poisonings because the formation of the AChE-carbamate complex is spontaneously reversible. However, the use of the atropine antidote is valuable even in carbamate poisonings, especially in cases where maintenance of airway and respiration is important.
❑ Several organophosphate pesticides can cause delayed peripheral neuropathy and chronic central neurotoxicity.
Inhibition of AChE causes acetylcholine to accumulate at synaptic sites. The accumulation results initially in overstimulation and then in paralysis of neurochemical transmission. The character, degree, and duration of the resulting physiologic effects are directly related to the amount and rate of AChE enzyme inhibition at certain receptor sites in the central and peripheral nervous systems. Some critical mass of AChE must be inactivated before the signs and symptoms of poisoning manifest.
Accumulation of acetylcholine in the brain causes sensory and behavioral disturbances, incoordination, depressed cognition, and respiratory failure (the usual cause of death). Other clinical manifestations are variable and depend on the balance of enzyme inhibition at cholinergic neuroeffector junctions (muscarinic effects) and at voluntary skeletal nerve-muscle junctions and autonomic ganglia (nicotinic effects) (Table 1). Organophosphate pesticides cause muscarinic, nicotinic, and central-nervous-system (CNS) effects, whereas carbamate pesticides cause predominantly muscarinic effects.
Table 1. Common manifestations of acetylcholinesterase inhibition by site
Delayed Neuropathy
Symmetric distal axonal degeneration is a systemic health effect caused by some organophosphate pesticides that is not due to AChE inhibition. The degeneration is a “dying back” of large diameter axons and their myelinic sheaths in distal parts of the peripheral nerves and in long spinal-cord tracts. Studies indicate the degeneration is caused by inhibition of a neuronal, nonspecific carboxylesterase known as neuropathic target esterase, which appears to have a role in lipid metabolism in neurons.
The resulting clinical syndrome, organophosphate-induced distal neuropathy (OPIDN), typically begins about 7 to 14 days after treatment, after other signs of organophosphate poisoning have improved or resolved. These late neuropathies are primarily distal and sensorimotor in nature; predominant overall presentation is that of a motor disorder. Usual manifestations are flaccidity or paralysis and paresthesia of the extremities, predominantly the legs. (Foot drop, spasticity, and hyperactive reflexes are not uncommon.) In severe cases, paralysis can extend to involve the hands and muscles of the forearms and may mimic Guillain-Barré syndrome.
Only some organophosphate compounds (e.g., Trichlorphon, Merphos, triorthocresyl phosphate [TOCP], and triorthotolyl phosphate [TOTP]) have-been associated with delayed neuropathy in humans, and no carbamate compounds have been reported to cause this condition. During the Depression of the 1930s, thousands of people in the United States fell victim to “ginger jake paralysis,” a condition caused by drinking an alcoholic extract of Jamaican ginger that had been adulterated with TOTP, a compound used as a plasticizer and as an additive in aviation fuels and lubricants. In 1959, several thousand people in Morocco were poisoned when jet fuel containing TOCP was mixed with olive oil and sold as cooking oil. Effects of these ingestions, which commonly included calf pain and tingling in the hands and feet initially, persisted for weeks to years.
Another syndrome of delayed onset neuropathy has recently been described by clinicians in Sri Lanka in conjunction with suicide attempts involving organophosphate insecticides. This paralytic condition, known as “intermediate syndrome,” consists of a sequence of neurologic signs that appear 24 to 96 hours after acute cholinergic crisis but before the expected onset of OPIDN. The major effects are weakness of proximal limb muscles, neck flexors, and respiratory muscles. Cranial nerve palsies are common. Knee and ankle reflexes may be absent. Unlike OPIDN, this sequence does not cause sensory impairment. Risk of death during this time interval is considerable, however, because of respiratory depression and distress.
Other Effects
❑ Organophosphate and carbamate pesticides are primary skin irritants.
Dermal lesions are more common than systemic poisonings among persons who encounter pesticides in the course of their occupational activities. About one-third of all reported pesticide-related diseases are dermatologic. Many organophosphate and carbamate pesticides cause primary irritant dermatitis; only a few are known to cause allergic contact dermatitis (e.g., parathion, malathion, and dichlorvos), and some can trigger phototoxic dermatitis (see Case Studies in Environmental Medicine: Skin Lesions and Environmental Exposures). Because pesticides are often used in combination, it can be difficult to identify the pesticide causing the problem.
❑ Some cholinesterase-inhibiting pesticides are suspected animal or human carcinogens; some have been found to cause developmental effects in experimental animals.
Results of experimental animal studies and human epidemiologic studies suggest an association between certain organophosphate compounds and cancer. A proposed mechanism of action is altered cellular immunity. However, the cholinesterase-inhibiting compounds commonly used as insecticides have not been found to be carcinogenic.
Some organophosphate compounds caused reduced rates of conception, reduced fertility, and adverse developmental effects in rats. However, other species of experimental animals showed no reproductive or teratogenic effects from parathion. No evidence exists that cholinesterase-inhibiting insecticides are significant teratogenic risks to humans.
|
(3) Are the signs and symptoms exhibited by the patient in the case study consistent with poisoning by an organophosphate pesticide? Explain. _________________________________________________________________ _________________________________________________________________ _________________________________________________________________ (4) Would you treat the patient differently if he had been exposed to a carbamate rather than an organophosphate pesticide? Explain. _________________________________________________________________ _________________________________________________________________ _________________________________________________________________ |
Clinical Evaluation
The relative intensity of clinical manifestations in patients poisoned by organophosphate or carbamate pesticides is strongly influenced by (1) the toxicity and route of absorption of the pesticide, and (2) the degree and rate of AChE inhibition with subsequent accumulation of acetylcholine.
History and Physical Examination
Diagnosis of organophosphate or carbamate toxicity is based on the history of exposure and the presence of characteristic muscarinic, nicotinic, and CNS manifestations. In suspected organophosphate poisoning, the physical examination should include careful evaluation of the cardiovascular system and the nervous system. The medical history should emphasize the following elements:
-
Occupational history (particularly involving exposures to insecticides)
-
Residence: age, pest control programs (both structural and grounds), and location in relation to certain industrial facilities (sites where pesticides and related products are produced or stored)
-
Twenty-four hour recall of all foods and liquids ingested
-
Hobbies: management of playing fields, gardening
-
Medications, such as pilocarpine for glaucoma
(See Case Studies in Environmental Medicine: Taking an Exposure History.)
Signs and Symptoms
Acute Exposure
❑ The onset of symptoms of acute organophosphate or carbamate poisoning usually occurs shortly after exposure but can be delayed up to 12 hours.
Because clinical manifestations are typical, patients who are severely ill from acute pesticide exposure seldom present a diagnostic challenge if the history of exposure is also available. Miosis, fasciculations, excess secretions, and hyperactive bowels constitute a classic presentation of organophosphate poisoning. Often a petro-chemical-like odor emanates from the patient’s breath or clothing.
Depending on the route of exposure, symptoms of acute organophosphate and carbamate poisoning usually develop within 4 hours of contact, but may be delayed for up to 12 hours. Initial symptoms may be headache, dizziness, nausea, and abdominal pain. Signs of
❑ Respiratory failure is the usual cause of death after acute poisoning by cholinesterase-inhibiting pesticides.
severe poisoning may include coma, pulmonary edema, ataxia, toxic psychosis (manifested as confusion or bizarre behavior), dyspnea, fasciculations, bradycardia, cardiac dysrhythmias, and weakness or paralysis. The actual cholinergic symptomatology may depend somewhat on the route of exposure and the balance between nicotinic (organophosphate pesticides) and muscarinic receptors (organophosphate and carbamate pesticides). (See Table 1, page 8.) The symptoms of carbamate poisoning typically are less severe and of shorter duration than those of organophosphate poisoning.
Chronic Exposure
Poisoning due to chronic exposure to cholinesterase-inhibiting insecticides may not be readily apparent because the symptoms are sometimes nonspecific and resemble other illnesses such as influenza, heat exhaustion, alcohol toxicity, or simple fatigue. Also the history of exposure may not be particularly remarkable in such cases. Repeated absorption of organophosphates at subacute concentrations may cause persistent anorexia, weakness, and malaise (“orange-picker’s flu”), a condition seen in farm workers engaged in crop spraying. Certain neurobehavioral effects may persist after chronic exposure, as well as after acute toxicoses.
Despite the lack of distinct symptomatology, it is important to aggressively investigate all suspected cases of chronic pesticide poisoning because such a case may represent a sentinel event, indicating a workplace hazard or other populations at risk. In some states, failure to report a pesticide-related illness can result in a fine. (Check with your state health department.)
Laboratory Tests
A combination of history, physical examination, and laboratory tests will provide the most appropriate approach to diagnosing pesticide-related illnesses.
Direct Biologic Indicators
❑ Analysis of blood and urine for direct evidence of organophosphate or carbamate exposure is valuable, but usually is available only from reference laboratories.
Detection of intact organophosphate or carbamate compounds in blood is usually not possible except during or soon after exposure. In general, cholinesterase-inhibiting pesticides do not remain unhydrolyzed in the blood more than a few minutes or hours, unless the quantity absorbed is large or the hydrolyzing liver enzymes are inhibited.
The metabolites of organophosphates (i.e., the corresponding alkyl phosphates) and the unique metabolites of N-methyl carbamates can often be detected in the urine up to 48 hours after exposure. The appearance of these urinary metabolites can demonstrate
pesticide absorption at lower dosages than those required to depress cholinesterase activity and at much lower dosages than those required to produce signs and symptoms. However, testing for these excretion products generally is available only from reference laboratories, and the results may be delayed because of the complexity of the analyses.
Indirect Biologic Indicators
❑ Measurement of red blood cell or plasma cholinesterase activity can help to confirm exposure to an organophosphate or carbamate pesticide.
Determination of cholinesterase activity is useful in routine biologic monitoring of workers chronically exposed to cholinesterase-inhibiting pesticides, as well as in diagnosing acute poisoning. Cholinesterase depression is usually apparent within a few minutes or hours after significant absorption.
Acetylcholinesterase enzymes measured in red blood cells (RBC) (referred to as true cholinesterase) and in plasma or serum (referred to as pseudocholinesterase) are actually surrogate indicators for activity occurring at neuroreceptor sites. Both plasma and RBC cholinesterase should be determined on each sample because the two tests have different meanings, and the results must be considered in combination for proper interpretation.
Plasma cholinesterase is more labile than RBC cholinesterase and is therefore less reliable in reflecting actual depression of enzyme activity at neuroeffector sites. Plasma cholinesterase is generally more rapidly inactivated by exposure to organophosphates, and it may also be slightly depressed by factors such as infection, alcohol, hepatic disease, birth control pills, and pregnancy. Because plasma cholinesterase is produced in the liver, it can be regenerated quickly.
RBC cholinesterase is the same enzyme as that found in the nervous system. It is depressed more slowly than plasma cholinesterase, but because the depression is more specifically attributable to organophosphates, its level more accurately reflects the degree of actual enzyme inactivation at neuroeffector sites. RBC cholinesterase activity is generally restored only as new red blood cells are formed. (Regeneration of red blood cells takes place at a rate of about 1% per day.) RBC cholinesterase activity is slightly affected by certain rare conditions that damage the cell membrane, such as hemolytic anemias or other causes of reticulocytosis. (Hemolytic anemias will cause a relatively elevated level of plasma cholinesterase but a lowered RBC cholinesterase as the cholinesterase of the lysed cells is liberated into the plasma.)
It is difficult to interpret cholinesterase inhibition without baseline values because normal human cholinesterase levels vary widely. The laboratory normal range is not useful because upper and lower limits of normal range may differ by a factor of 4 with some common laboratory methods. Inhibitions of from 25% to 50% of the individual’s
baseline enzyme activity are generally regarded as evidence of toxicity. However, in both acute and chronic exposures, clinical symptoms have been absent even when cholinesterase levels were inhibited by 50% of baseline. The development of signs and symptoms is related to both the rate of decline in enzyme activity and the absolute level of enzyme activity.
Because of marked variation among different methods of analysis and among laboratories using the same method, cholinesterase tests, including baseline determinations and serial follow-up testing, should be performed by the same laboratory, using the same method. It is misleading to extrapolate from one method to another or from one laboratory’s results to another’s. A laboratory performing cholinesterase tests should have a rigorous quality-control program and an accurate and reproducible method of analysis.
Other laboratory evaluations for patients seriously exposed to organophosphate or carbamate pesticides may include CBC and determinations of electrolytes, glucose, BUN, creatinine, and liver enzymes. Arterial blood gases and chest radiography are useful in cases of inhalation exposure or respiratory compromise.
|
(5) What is the differential diagnosis for the patient in the case study? _________________________________________________________________ (6) What laboratory tests will you request? _________________________________________________________________ (7) The patient has an initial RBC cholinesterase that is 25% of the laboratory’s normal value. What factors, other than organophosphate poisoning, could account for this dramatic depression of RBC cholinesterase? _________________________________________________________________ _________________________________________________________________ |
Treatment and Management
❑ Nicotinic and muscarinic effects of acute pesticide poisoning require immediate, aggressive emergency management.
Organophosphate poisoning is one of the few cases of acute toxicity for which specific antidotes are available. The severity of the poisoning, among other considerations, should dictate the institution of the management procedures. If a patient has been poisoned by organophosphate exposure, decontamination should proceed concurrently with resuscitative and antidotal measures.
Protective clothing (including rubber gloves) should be worn when decontaminating patients exposed to organophosphate compounds. If patients are able and cooperative, they may assist with their own decontamination. Remove clothing and personal belongings and double-bag them. Clothing and leather items are difficult to decontaminate, and in most cases, should be incinerated.
❑ Atropine is the primary antidote for adverse muscarinic effects.
Flush exposed skin and hair with plain water for 2 to 3 minutes (preferably under a shower), then wash twice with mild soap. Be certain to clean under fingernails and in all skin folds. Rinse thoroughly with water. If the patient’s eyes were exposed, irrigate them with plain water or saline for 5 minutes; remove contact lenses if present.
❑ Pralidoxime (2-PAM) is the antidote of choice to control nicotinic manifestations in organophosphate poisoning.
In cases of ingestion, gastric lavage is preferred over emesis because CNS depression may develop rapidly. The effectiveness of lavage, however, diminishes rapidly with time. The airway must be vigorously protected from aspiration of gastric contents. A slurry of activated charcoal with a cathartic (sorbitol, magnesium sulfate or citrate) should be administered even though most organophosphate compounds are not efficiently adsorbed to charcoal.
The recommended treatment for serious poisoning is as follows:
(a) Establish a clear airway by aspiration of secretions. If necessary, intubate the patient and consider mechanical means to assist respiration. (Atropine, which is recommended in the next step, can cause ventricular fibrillation when administered to an hypoxic patient.)
(b) Intravenously administer atropine sulfate (adult: 2–4 milligrams [mg]; <12 years: 0.05 mg/kilogram [kg] body weight) and repeat at 3- to 10-minute intervals until signs of atropinization appear (e.g., reversal of excess salivation, bronchial secretions, and sweating). Maintain atropinization by repeated dosage for at least 24 hours; total doses up to 50 mg/day have been given. (In one reported case, 1000 mg/day was given.)
Atropine controls muscarinic manifestations but has no effect on nicotinic or CNS manifestations. Severely poisoned patients may
exhibit remarkable tolerance to atropine and massive amounts may be required. Reversal of muscarinic signs and symptoms, not an arbitrary dose limit, is the endpoint. If signs of atropine toxicity (fever, muscle fibrillations, and delirium) appear, atropine administration should be discontinued temporarily while the severity of poisoning is reevaluated. The clinician inexperienced in handling patients poisoned by cholinesterase-inhibiting pesticides should contact a regional poison control center or a medical toxicologist for specific guidelines before giving further treatment.
(c) Obtain a blood sample (heparinized) for cholinesterase determination. (The blood sample should be taken before administering the antidote pralidoxime chloride [2-PAM, Protopam] recommended in the next step because this antidote quickly reactivates the enzyme.)
(d) If the patient exhibits muscle weakness, twitching, or respiratory depression, administer 2-PAM intravenously, preferably as an infusion in 100 mL of saline over a 15- to 30-minute period (adult: 1–2 grams [gm]; <12 years: 20–50 mg/kg body weight). Rapid injection of 2-PAM can cause tachycardia, laryngeal spasm, muscle rigidity, and transient neuromuscular blockage.
Pralidoxime is an oxime that breaks the bond in the AChE-phosphate complex and should be used for most clinically significant organophosphate poisonings. However, in certain carbamate poisonings (e.g., carbaryl) 2-PAM is generally of limited value and may be hazardous. If a combination of carbamate and organophosphate poisoning has occurred, or if the pesticide cannot be identified, 2-PAM should be administered.
Administer 2-PAM as soon as possible but within 48 hours after organophosphate exposure. Beyond this time, AChE can become firmly bound to the phosphate in an “aging” process, which results in the enzyme becoming refractory to reactivation.
(e) Repeat the dosage of 2-PAM in 1 to 2 hours, then at 10- to 12-hour intervals, if needed. If testing is readily available, the RBC and plasma cholinesterase activity of the patient can be monitored to determine the effect of 2-PAM.
(f) Observe the patient closely for at least 72 hours for recurrence of toxicity. Monitor pulmonary ventilation and cardiac status, and administer symptomatic treatment as required. If convulsions occur despite atropine and 2-PAM therapy, diazepam or other anticonvulsants may be administered. Slowly administer the anticonvulsant intravenously, while watching for hypotension and hypoventilation.
Medications that may be contraindicated in organophosphate or carbamate pesticide poisoning include morphine, aminophylline,
and phenothiazines because they have weak anti-cholinesterase activity. However, they are not contraindicated unequivocally and should be administered if there are other extenuating medical reasons for their use.
(g) Notify the health department of all pesticide exposures resulting in poisoning. Patients should not return to an environment where further exposure is possible until their blood cholinesterase activity has reached a recommended minimum (typically 80% of the baseline level). Neither atropine nor 2-PAM should be administered prophylactically to workers or others exposed to organophosphates or carbamates. Oral atropine should not be used in treating pesticide poisonings.
|
(8) What agents would be contraindicated for the patient in the case study? _________________________________________________________________ _________________________________________________________________ (9) Why should atropine or 2-PAM not be used prophylactically? _________________________________________________________________ _________________________________________________________________ (10) Describe the probable course and outcome for this patient. _________________________________________________________________ _________________________________________________________________ |
Standards and Regulations
The U.S. Environmental Protection Agency (EPA) was given wide-ranging authority over domestic pesticide usage by regulations contained in the Federal Insecticide, Fungicide, and Rodenticide Act (FIFRA), passed in 1947 and amended in 1972 and 1988. EPA is responsible for registering all pesticides for use in the United States, establishing guidelines for registration procedures, certifying individual state programs for licensing pesticide applicators, establishing reentry intervals, specifying label information and use instruction, and setting acceptable tolerance levels in food and water.
Workplace
The Occupational Safety and Health Administration (OSHA) mandates permissible exposure limits (PELs) for pesticides in the workplace. PELs for organophosphate pesticides are typically time-weighted averages for an 8-hour workshift and range from 0.05 mg/cubic meters (m3) of air to 15.0 mg/m3. PELs depend on the pesticide’s toxicity, which is determined by the LD50 (lethal dose to 50% of the test animals exposed to a chemical administered in a single dose by any route other than inhalation) value and other hazard indicators such as corrosiveness, dermal rating, and carcinogenicity.
Environment
Water
The enforceable maximum contaminant level (MCL) set by EPA for drinking water varies for each pesticide. The EPA Drinking Water Hotline ([800] 426–4791) and Office of Pesticide Programs ([703] 557–2440) can provide information on specific pesticide regulations.
Food
EPA, in conjunction with the Food and Drug Administration (FDA) and the U.S. Department of Agriculture (USDA), monitors and regulates pesticide residues and their breakdown products in food. Established tolerance limits for most cholinesterase-inhibiting pesticides and their metabolites in various raw agricultural commodities range from 0.05 parts per million (ppm) to 10 ppm.
Suggested Reading List
General
Ames RG, Brown SK, Mengle DC, Kahn E, Stratton JW, Jackson RJ. Cholinesterase activity depression among California agricultural pesticide applicators. Am J Ind Med 1989;15:143–50.
Baker SR, Wilkinson CF, eds. The effects of pesticides on human health: proceedings of a workshop, May 9–11, 1988, Keystone, Colorado. Advances in modern environmental toxicology; vol 18. Princeton, New Jersey: Princeton Scientific Publishing Co., Inc., 1990.
Costa LG, Schwab BW, Murphy SD. Tolerance to anticholinesterase compounds in mammals. Toxicol 1982;25:79–97.
Clifford NJ, Niew AS. Organophosphate poisoning from wearing a laundered uniform previously contaminated with parathion. JAMA 1989;262(21):3035–6.
Gallo MA, Lawryk NJ. Classes of pesticides. In: Hayes WJ, Laws ER, eds. Handbook of pesticide toxicology; vol 2. New York: Academic Press, Inc., 1991:917–1123.
Goldman LR, Smith DF, Neutra RR, et al. Pesticide food poisoning from contaminated watermelons in California. Arch Environ Health 1990;45(4):229–36.
Griffith J, Duncan RC. Grower reported pesticide poisonings among Florida citrus fieldworkers. J Environ Sci Health; B20
Meister R, ed. Farm chemicals handbook. Willoughby, Ohio: Meister Publishing Co., 1991 (published yearly).
Moses M. Pesticides. In: Rom WN, ed. Environmental and occupational medicine. New York: Little, Brown and Company, 1983.
Rosenberg J. Pesticides. In: LaDou J, ed. Occupational medicine. Norwalk, Connecticut: Appleton & Lange, 1990.
Tafuri J, Roberts J. Organophosphate poisoning. Ann Emerg Med 1987;16:193–202.
World Health Organization. Organophosphorus insecticides: a general introduction. Environmental Health Criteria 63. Geneva: World Health Organization, 1986.
Neurotoxicity
Abou-Donia MB, Lapadula DM. Mechanisms of organophosphorus ester-induced delayed neurotoxicity: type I and type II. Ann Rev Pharmacol Toxicol 1990;30:405–40.
Hansen LG. Biotransformation of organophosphorus compounds relative to delayed neurotoxicity. Neurotoxicology 1983;4:97– 111.
Lotti M, Becker CE, Aminoff MJ. Organophosphate polyneuropathy: pathogenesis and prevention. Neurology 1984;34:658–62.
Savage EP, Keefe TJ, Mounce LM, Heaton RK, Lewis JA, Burcar PJ. Chronic neurological sequelae of acute Organophosphate pesticide poisoning. Arch Environ Health 1988:43(1):38–45.
Maroni M, Bleecker ML. Neuropathy target esterase in human lymphocytes and platelets. J App Toxicol 1986;6 (1):1–7.
Treatment and Management
California Department of Health Services. Medical supervision of pesticide workers: guidelines for physicians. Berkeley, California: California Department of Health Services, 1988.
Lotti M, Becker CE. Treatment of acute organophosphate poisoning: Evidence of a direct effect on central nervous system by 2-PAM. J Toxicol Clin Toxicol 1982;19:121–7.
Midtling JE, Barnett PG, Coye MJ, et al. Clinical management of field worker organophosphate poisoning. West J Med 1985;142:514–8.
Laboratory Evaluation
Coye MJ, Lowe JA. Biological monitoring of agricultural workers exposed to pesticides: I. Cholinesterase activity determinations. II. Monitoring of intact pesticides and their metabolites. J Occup Med 1986;28:619–28.
Silk E, King J, Whittaker M. Assay of cholinesterase in clinical chemistry. Ann Clin Biochem 1979;16:57–75.
Related Government Documents
Agency for Toxic Substances and Disease Registry. Toxicological profile for methyl parathion. Atlanta: US Department of Health and Human Services, Public Health Service, 1991.
Morgan DP. Recognition and management of pesticide poisonings. Washington, DC: US Environmental Protection Agency, Office of Pesticide Programs, 1989. EPA report no. EPA-540/9–88–001.
Environmental Protection Agency. Pesticide fact handbook. Washington, DC: US Environmental Protection Agency, Office of Pesticide Programs , 1985.
Sources of Information
More information on treating and managing patients exposed to cholinesterase-inhibiting pesticides can be obtained from ATSDR, the National Pesticide Telecommunications Network (24-hour hotline, [800] 858–7378), your state and local health departments, the plant division of your state’s department of agriculture, county agricultural commissioner and cooperative extension agents, and university medical centers. Case Studies in Environmental Medicine: Cholinesterase-Inhibiting Pesticide Toxicity is one of a series. To obtain other publications in this series, please use the order form on the inside back cover. For clinical inquiries, contact ATSDR, Division of Health Education, Office of the Director, at (404) 639–6204.
Answers to Pretest and Challenge Questions
Pretest questions are on page 1. Challenge questions begin on page 5.
Pretest
-
The patient’s problem list includes headache, dizziness, nausea, vomiting, hypersalivation, diaphoresis, miosis, altered mental status, paralysis, fasciculations, and autonomic dysfunction.
The differential diagnosis might include the following:
organophosphate or carbamate poisoning
heroin/opiate overdose
phenothiazine overdose
pontine tumor or subarachnoid hemorrhage
CNS infection, such as encephalitis, meningitis, or brain abscess
botulism, gastroenteritis
congestive heart failure
heat exhaustion
alcohol toxicosis
-
The history provided by the patient’s brother and the signs and symptoms are consistent with poisoning due to a cholinesterase-inhibiting pesticide (see Challenge answer 3). It is important to discover the route of exposure so that future exposures can be prevented. (See Challenge question and answer 1.) Further questioning may reveal whether the pesticide was an organophosphate or carbamate. You should also query the brother and other family members about the patient’s liquid and food consumption for the past 24 hours and medications that he may be taking.
-
Laboratory confirmation of organophosphate poisoning can be sought by measuring the activities of RBC and plasma cholinesterase—both enzymes are affected by organophosphates. Although individual normal values vary markedly, poisoning is considered to have occurred when cholinesterase activity is 25% to 50% of normal. However, not only is the degree of cholinesterase depression important, but the rate of depression is also pertinent. A repeat acetylcholinesterase test should be performed in 2 to 3 weeks to determine whether the value has increased significantly.
-
The treatment that resulted in the patient’s remarkable recovery included the administration of atropine and 2-PAM.
Challenge
-
The most likely routes of exposure for the patient are inhalation and dermal contact. The brothers apparently wore adequate protective equipment, and they have performed the pesticide mixing operations many times without ill effects. It is significant that only one of the men became ill. A possibility exists that the illness is related to the pesticide spill in the formulating area.
Using investigative results by the State Department of Agriculture and by the process of eliminating other conditions, such as a loose cartridge respirator, you conclude that contamination most likely occurred via the coveralls that had been worn during the spill.
-
Anyone wearing the coveralls may be similarly exposed. Laundering, even multiple washings, may not completely remove some pesticides. In the process of laundering, the coveralls could also contaminate the clothing of other family members. The coveralls should be burned.
-
Yes. The patient manifests the classic signs and symptoms of organophosphate poisoning. The effects can be classified into three categories: muscarinic or hollow-organ parasympathetic manifestations, nicotinic or autonomic ganglion and somatic motor effects, and CNS effects. Carbamate poisoning can be distinguished from organophosphate poisoning by the absence of nicotinic effects. Carbamate poisoning is also immediately reversible by a small dose of atropine, compared with the large doses of atropine needed in organophosphate intoxications.
The muscarinic effects involve the bronchial tree, sweat and lacrimal glands, heart, pupils, and ciliary body. Muscarinic effects are easily remembered by the acronym SLUDGE—salivation, lacrimation, urination, defecation, gastrointestinal distress, and emesis.
Nicotinic effects typically include muscle fasciculations, cramping, and weakness that can progress to paralysis, areflexia, hypertension, tachycardia, pupillary dilation, and pallor. Respiratory failure may occur secondary to weakness of the pulmonary muscles or paralysis of the diaphragm. Hypertension and pupillary dilation have also been noted.
CNS effects may include restlessness, emotional lability, headache, tremor, drowsiness, delirium, psychosis, coma, and cardiorespiratory depression.
-
Yes, if the patient had been exposed to a carbamate, administration of 2-PAM to reactivate the AChE from the AChE-carbamate complex would usually be unnecessary because the carbamate complex is spontaneously reversible. Recovery from carbamate poisoning is typically more rapid than from organophosphate poisoning and without persistent sequelae.
-
See Pretest answer (a) above.
-
See Pretest answer (c) above.
-
Diet, medications, and reticulocytosis may lower the RBC acetylcholinesterase activity. Reticulocytosis may be the result of recovery from hemorrhage, hemolysis, liver disease, jaundice, hepatitis, pregnancy, and pernicious anemia or other anemias. However, these conditions cannot account for the severe lowering of the RBC cholinesterase activity seen in this patient. (The RBC cholinesterase activity is 25% of normal.)
-
Drugs that are contraindicated for nearly all organophosphate-poisoned patients include opiates and phenothiazines; they may increase the risk of cardiac dysrhythmias. A small portion of the population possess a genetic variant of plasma cholinesterase that can cause death if succinylcholine is administered to the patient.
-
Atropine and 2-PAM should not be administered prophylactically because they cause blurred vision and lack of sweating. The loss of sweating may cause hyperthermia under certain conditions. Administration of antidote can mask signs and symptoms of pesticide poisoning, thus allowing dangerously prolonged exposure.
-
After 4 weeks, the patient shows no sign of delayed neuropathy or other adverse effects. The prognosis is, therefore, excellent. Chronic effects similar to cerebral dysfunction have been noted in some patients acutely poisoned.























