SESSION II
Preembryo Research
Overview: Modern Ideas in Embryo Research
ROBERT EDWARDS1
Bourn Hall Clinic, Cambridge, United Kingdom
Much of the preembryo research conducted over the past 20 years has been related to the development of in vitro fertilization (IVF), now an accepted procedure worldwide. In thinking about IVF, we must always think first of the gametes: the sperm and egg. Some recent findings about gametes may have major bearing on IVF both scientifically and ethically. Two involve new mechanisms for sperm selection.
In the process of meiosis, through which gametes form, precursor cells called spermatocytes give rise to spermatozoa. Spermatocytes are diploid: they contain two sets of chromosomes. They divide into haploid cells, each containing one set of chromosomes. In experiments with a strain of mice, it was observed that the two classes of spermatozoa arising from meiosis were not equally successful in fertilizing eggs; one class fertilized 80 percent of the eggs. It was subsequently determined that this unexpected disparity results from differences in the way in which genetic information encoded in the DNA of one specific locus is transcribed in the two classes of spermatids. The controlling gene sequence—the string of ''letters'' that spell our the genetic message—has been identified in mouse spermatozoa. Similar sequences have been found in human spermatozoa, but their function in humans is as yet unknown.
This discovery provided a long-sought answer to the question of whether sperm are governed genetically by the haploid or diploid genome, at least with
regard to their ability to fuse with eggs. This research avenue may lead to the possibility of sperm selection to avoid genetic transmission of disease genes as an alternative to the current methods of prenatal testing (e.g., amniocentesis), which is done after embryo implantation in the uterus.
In other experiments, researchers have been developing refined methods for separating X and Y spermatozoa. X and Y are the chromosomes that determine an offspring's sex. Because may inherited diseases are X-linked, this new methodology may present another opportunity for preselecting spermatozoa that contain the Y chromosome. It could also intensify ethical quandaries concerning parental choice of a child's sex for other than medical reasons.
Research on female gametes (oocytes) is limited by lack of supply. Even superovulation, induced by fertility drugs in IVF, yields, at best, a small number of oocytes. The problem is now compounded by the availability of cryopreservation of embryos. Many researchers feel strongly that all eggs obtained from IVF patients should be fertilized, and the healthy embryos that result kept solely for the patient, in case of initial implantation failure or for future pregnancies. Aside from other considerations, IVF is a costly procedure. Consequently, the research that can be done with normal IVF embryos has been almost completely confined to what is directly relevant to the patient.
A potentially abundant source of oocytes exists in aborted female fetuses. Experiments with egg follicles extracted from infantile mice show that the follicles can be grown in culture to form artificial ovaries that ovulate, and the mature eggs can be fertilized to produce offspring. Governmental permission to conduct experiments with human fetal egg follicles has recently been granted in the United Kingdom, and experiments have been initiated to study ovarian growth and oocyte development. Permission has not yet been sought for fertilization of human fetal oocytes.
Fetal oocytes could conceivably be used for IVF, but such use raises serious ethical concerns. One concern is the sheer number of embryos that could be generated from oocytes from a single fetus. Another problem is compliance with consent requirements from an egg donor when that donor is an aborted fetus.
There has been considerable progress in preembryo science in animals and in techniques for making IVF safer and easier. For example, key information found regarding the mechanism by which the sperm attaches to and penetrates the zona pellucida (the egg's outer coat) led to the more successful practice of implanting those oocytes that exhibited zona-bound sperm. Understanding the role of calcium in activation of sperm has been important to micromanipulation of gametes. In addition, fascinating reproductive biological principles have been elucidated, such as the role of sperm-produced proteins in egg activation. Yet, the rate of successful implantations in patients remains low. The reasons are still not understood, and many studies are being directed toward solving the problem.
Science and medical practice need to know more about the maturation of male gametes and the way in which cells are allocated within embryos. Better
culture methods need to be developed and improved standards need to be defined by consensus of experts in the field. Much remains to be known about the fundamental biology of embryos, especially preimplantation embryos, with a view toward identifying vulnerable periods and states, both in vitro and in vivo.
Unexpected events may help to shed some light. It has lately been discovered that acyclic women, including women with no ovaries and postmenopausal women, have a much higher rate of IVF success than normally cyclic IVF patients. In 50-year-old women, the pregnancy rate was almost as high as in normal women of 25, and there have been successful pregnancies in women in their 60s. Why this is so, whether it has to do with the condition of the donated eggs or the uterus or some other factors, is unclear. What is clear is that the possibility of babies from fetal eggs and postmenopausal pregnancies ushers in a new set of ethical issue. Examples of these include the potential births of scores of genetic half-siblings and births of children to mothers who may not live long enough to raise the children to adulthood.
MICROMANIPULATION RESEARCH IN CLINICAL EMBRYOLOGY
JACQUES COHEN
Cornell University Medical Center, New York, New York
Research on important areas of human embryology, such as gamete interaction, embryo implantation, and abnormal development, has focused largely on efforts aimed at improving success rates of assisted reproduction. These studies have been advanced by the adaptation of powerful new laboratory tools. Micromanipulation is a technology that employs very fine glass microtools attached to robot arms hooked to a microscope and moved by remote control (Figure 2).
In embryo studies in experimental animals, micromanipulation is used to extract cells or cell components for genetic analysis and diagnosis, to inject sperm into eggs, to transplant cell nuclei for genetic study, or to remove damaged tissue. In human embryology it is used mainly to facilitate fertilization in cases where sperm cannot penetrate the zona pellucida and to help embryos hatch out of the zona, although its use for genetic analysis of preimplantation embryos is close at hand.
Male infertility is a widespread problem for which conventional IVF works in only a small percentage of cases. Assisted fertilization is a new and developing technology in which a number of approaches are being tried. In the most dramatic approach, a single sperm is placed directly into an oocyte. Although success rates are still relatively low and unpredictable, what is significant is that a man with virtually no sperm, or none that are motile, has a chance of becoming a father.
At present, IVF culture systems provide less than optimal environments, especially for older women, for the embryo to hatch out of the zona pellucida
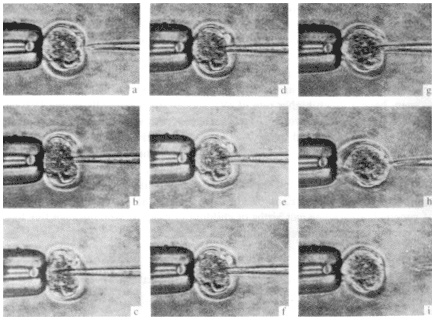
Figure 2
Aspiration of the blastocoelic cavity of a mouse blastocyst. (a–c) The blastocyst is secured to the holding pipette close to the inner cell mass and held by gentle suction. The sharply pointed biopsy pipette is introduced to the cavity via the mural trophectoderm. The outline of the cavity is not visible clearly in this figure, which is always focused on the point of the biopsy pipette. (d) The pipette is withdrawn slightly away from the inner cell mass. (e–f) Suction is applied to the biopsy pipette. The cavity collapses. Collapse has occurred in the vertical plane in this specimen, and no major change in volume is evident in the figure. (g–h) The biopsy pipette containing aspirated cells is withdrawn. The biopsy can then be expelled into a drop of medium for observation at high magnification. (i) The collapsed blastocyst seen in this figure reexpanded within three hours of the biopsy taken. SOURCE: Edwards, R.G., Preconception and Preimplantation Diagnosis of Human Genetic Disease, 1993. Reprinted, with permission, from Cambridge University Press.
and implant itself in the uterus. In human embryos the zona comes in different shapes and sizes, some thick, some thin. Thicker zonas yield low pregnancy rates. This problem can often be corrected by micromanipulating an opening in the zona shortly before the embryo is returned to the woman. In a series of assisted hatchings involving 790 patients, 339 became pregnant—much more than 50 percent of the younger patients, and more than 20 percent of patients over 39.
Research examining the chromosome abnormalities in human blastomeres suggests that such abnormalities account for a high percentage of IVF embryos that fail.2 This represents one of the most pressing research questions in the field. Thus, methods of molecular biology, adapted for studying genetic anomalies in single embryonic cells removed by micromanipulation, offer great promise. One of these methods is called fluorescence in situ hybridization (FISH) in which complementary pieces of single-strand DNA with identified sequences are used to bind to single-strand DNA in the blastomere nucleus to identify specific sequences. The bound pieces are visualized with fluorescence microscopy. In this context, FISH can be used to identify specific chromosomes, including sex chromosomes in interphase nuclei and, thus, to determine the frequency of aneuploid blastomeres. Implantation would be limited to those embryos that are chromosomally normal.
Progress in Reproductive Biology and Implications for the New Reproductive Technologies in Human IVF
ALAN TROUNSON
Monash University Medical Center, Victoria, Australia
As with micromanipulation, a number of other areas of reproductive research are progressing toward innovations that should prove beneficial in IVF. A procedure now used with animals, which looks promising for adaptation to human patients, would lessen or eliminate the need for fertility drugs.
In human IVF as currently practiced, fertility drugs are used to induce superovulation. In a method that has proved very productive in artificial cattle breeding, developing follicles are directly removed from the ovaries of slaughterhouse animals and cultured and matured in vitro. Then, oocytes are fertilized and implanted. As modified for human patients, the technique would involve recovering immature eggs at any time during the natural ovulatory cycle, without any drug treatment or after very brief treatment with follicle-stimulating hormone.
There are several advantages to this technique. Oocyte recovery is markedly simpler than superovulation, which requires that the patient be closely monitored. It is significantly cheaper, which would make IVF available to more people. It garners many more eggs from a donor. It avoids or reduces adverse side effects of fertility drugs, such as premature births, deleterious effects of superovulation on uterine receptivity to implantation, and the potential for hormone-stimulated pathologies.
In another area, progress in cryopreservation techniques has simplified and improved the success of gamete as well as embryo preservation. Freezing gametes rather than embryos avoids the technical difficulties associated with keeping embryos and the ethical dilemma created by increased numbers of unwanted embryos.
It is important to consider that not all techniques successful in animal work are likely to be transferred to human IVF. Embryo bisection is one of these, as are nuclear transfer and insertion of DNA.
The overriding concern in IVF is achieving healthy embryos. Methods are being developed for identifying and reducing the 30 to 40 percent of chromosomally abnormal embryos that occur in human IVF. (The rate of abnormalities is also high in natural conception.) Culturing methods for embryos need to be optimized by studying their nutritional requirements in detail. Results of studies in mice and sheep have yielded very different results, so that experiments on the growth conditions for human embryos are essential. As a consequence of improved embryo viability, the number of embryos implanted in IVF could be lowered, thus decreasing the frequent multiple births that arise from implantations and the attendant physical and familial problems.
Much of what has been learned about the antigens present on the zona pellucida or on sperm could be used to design reversible contraceptive agents, which would be free of the side effects of conventional drug therapies. From the perspective of a researcher working outside the United States, the present state of embryo research in the United States certainly reduces the keen competition from talented American scientists, but also limits the discoveries possible. This occurs at a time when, with techniques now available or being developed, answers to the major questions in reproductive biology are at our fingertips.
OPEN DISCUSSION PERIOD
All three presentations stimulated discussion concerning chromosomal abnormalities in fetuses and the dilemma of how research is to be pursued in this area if IVF embryos are kept solely for patients and oocyte fertilization is held in abeyance because of potential controversy. Numerous contrasting views were expressed on these topics and, most striking, the research questions and ethical dilemmas were intimately interwoven throughout the discussion.
Prior to the ban on federal funding, an Ethics Advisory Board (EAB) existed in the United States that considered the ethical issues related to IVF research and served as the forum for national debate. It is important to note that although this body discussed the use of extra embryos from IVF in research, no explicit approval was given. The Ethics Advisory Board, in fact, did not even address the issue of harvesting male and female gametes for the purpose of producing IVF embryos—for research or any other purpose. Research advances in other countries have now made such questions more immediate. Thus, the conference
discussion highlighted the new ethical questions that have emerged since the EAB and other national advisory bodies were dissolved.
Many of the conferees thought that couples undergoing IVF should be permitted to donate extra embryos for research purposes, and it was pointed out by some that the National Institutes of Health regulations on the use of embryos for research were being reconsidered.
Nowhere was the need for national debate through the reestablishment of some kind of ethics advisory body more apparent than in the discussion concerning the harvesting and fertilization of immature fetal oocytes. The many discordant views expressed could provide only a hint of the complexity of issues raised by this technical advance. For example, some thought it reasonable to fertilize fetal oocytes for embryo research, but not for implantation. Others considered implantation of such embryos as fundamentally similar to the presently accepted use of donated male and female gametes for IVF. A cautionary note was raised that fertilized fetal oocytes should first be studied to determine if they are normal. This concept was challenged by the suggestion that determining the normalcy of such embryos would be severely complicated by the lack of phenotypic information on the fetal donor. Assessment, then, could include only genetic analysis, which is not complete and may not be sufficient to ensure the health of the resulting child. As discussion deepened, other broad issues emerged such as the potential for wide dissemination of a single genotype, because many thousands of oocytes can be harvested from a single fetal donor.
Despite the compelling nature of the discussion outlined above, other technical and medical questions were raised concerning embryo implantation in postmenopausal women, the continued implantation of multiple embryos in IVF, and the selection of subjects for micromanipulation techniques. Although postmenopausal women up to 60 years of age have carried IVF pregnancies to term, concern was expressed regarding the greater medical risks and the social implications for the children of mothers of advanced age.
Regarding implantations of multiple embryos, a participant asked why, with the availability of cryopreservation for IVF embryos, this practice was still routine when it is well known that multiple births and their ensuing dangers (e.g., premature births, growth retardation, infant mortality, and personal burdens of triplets and quadruplets) are the common result. The reason is largely a matter of economics—multiple implantations increase the chance that one procedure will result in a viable pregnancy. Each implantation procedure is expensive, and whether or not this cost is covered by insurance is an important factor. In Australia, where IVF costs are covered, the number of embryos implanted in a single procedure has been significantly reduced, resulting in many fewer multiple births.
Experiences with microsurgical fertilization techniques for treatment of male infertility were shared in an attempt to arrive at selection criteria. There is growing evidence that severity of sperm deficiency does not correlate with poor
results. Rather, sperm with the most severe profiles appear to do best when injected into eggs through these techniques. A participant involved in data collection in this area reported that given the differences in technical competence at IVF centers, microsurgical fertilization does not result in fetal malformation any more significantly than normal IVF.
Preimplantation Genetic Analysis of Single Human Blastomeres
MARK HUGHES
Baylor College of Medicine, Houston, Texas
Until recently, a couple who carries the gene for a genetic disease had three recourses: elect not to have children, adopt or use artificial insemination for recessive disorders, or terminate pregnancy if prenatal testing revealed the presence of disease in the unborn child. Preimplantation genetic analysis now provides a fourth option for some diseases.
Cystic fibrosis (CF), characterized by obstruction of the lungs leading to early death, is among the most devastating inherited disorders. One out of 25 Americans of northern European ancestry is a carrier of the CF gene. The disease is caused by a variety of mutations in a single gene that encodes a protein involved in chloride flux across cell membranes. Preimplantation analysis for CF and other genetic diseases has become possible through newly emerging techniques of molecular biology.
Developing a test that would definitively diagnose the presence of CF mutations requires being able to take the DNA in a single blastomere (one of the cells resulting from division of the fertilized egg) and generate huge quantities of it. A technique of molecular biology called polymerase chain reaction (PCR) can multiply a piece of DNA millions of times. This permits repeated comparative analyses of genes. The CF test that was developed, which yields results in a few hours, appears to be virtually 100 percent accurate for those cases in which the mutations in both parents have been identified, and it has been used successfully in a number of pregnancies.
Preimplantation analysis has also been developed for Lesch-Nyhan Disease, a severe neuromuscular disorder whose sufferers display bizarre self-mutilation behavior. To create this test it was necessary to amplify whole chromosomes, a feat achieved by a variation of PCR called primer extension preamplification, or PEP. PEP has also been used to develop a test for Duchenne's muscular dystrophy, which can arise from deletions in different parts of a single, enormous gene.
Other tests can now detect mutations of two forms of Tay-Sachs disease and sickle cell anemia. Blastomere diagnosis will be applicable to an inherited disorder for which the gene mutation has been identified. This technology can even be employed in certain diseases for which the defective genes are not yet identi-
fied. For example, in X-linked recessive disease, only male children will be affected. Thus, if one can identify the sex of the IVF embryos, one could implant only those that were female. Although identifying sex from multiple cells is relatively easy, the chances of misdiagnosis in the analysis of single cells from embryos with conventional methods are still high. In an elegant test that amplifies regions from two related genes, one on the X and the other on the Y chromosome, unequivocal sex diagnoses can be made.
It is unclear how many couples who can conceive normally will choose preimplantation analysis, which involves going through IVF, rather than conventional prenatal testing. Certainly many couples have serious difficulties with the idea of terminating a pregnancy. The decision for other couples may involve more than religious or moral positions on termination of their pregnancies. Many of the couples already have a child affected with the same disease, and the cycle of pregnancy, prenatal testing, and possible pregnancy termination carries with it an undesirable message that although the parents love their child, they do not want another child like that one. The spur to developing the test for Lesch-Nyhan disease was a woman who already had an afflicted son, four subsequent pregnancies that were aborted because of the presence of the disease, and five spontaneous abortions. To date, hundreds of patients have inquired about preimplantation analysis at IVF centers all over the world.
The molecular biological techniques involved in preimplantation diagnosis require those doing the testing to have extensive training and experience. As the demand for this approach widens, a serious concern is that IVF centers may attempt preimplantation analysis without adequate competence in medical genetics and molecular biology. What is needed is guidance by the National Institutes of Health and the establishment of a preimplantation working group to provide a mechanism for interaction among molecular biologists, medical geneticists, and obstetricians.
Studies on DNA Sequences in Single Human Gametes
NORMAN ARNHEIM
University of Southern California, Los Angeles, California
As with preimplantation embryo studies, PCR and PEP are being applied to genetic analysis of human gametes. Because of the difficulty in obtaining oocytes, most of this research has concentrated on sperm. Among the diseases being studied are the muscle-wasting diseases spinobulbar muscular atrophy and myotonic dystrophy. Huntington's disease, a severe and ultimately fatal neurological disorder, is also being studied with this technique.
A common characteristic of all these diseases is abnormal repetition of DNA sequences within specific genes. The number of repeats marks the difference between being an unaffected carrier for the disease and being afflicted with it.
Current research, looking at genes in sperm from men who start out with different numbers of repeats, is aimed at learning about mutation rates for the gene expansion, and possible contraction, events in these conditions.
In the double set of chromosomes within diploid cells, one inherited from the mother and the other from the father, paired genes may be identical or different in their DNA sequences. The different forms of the gene are called alleles. Studies of sperm by using PCR and PEP are being applied to the investigation of recombination or crossing-over events during meiosis, in which chromosomes intertwine and exchange sections that are detected as new allele combinations. Aberrant types of crossing-over events can sometimes lead to altered genes that can cause disease. A critical problem concerns the fact that crossing-over events occur at different frequencies in male and female gametes. Because of limited access to human female gametes, studies are conducted mostly with laboratory animals, but human oocyte research will ultimately be needed to resolve these questions.
Access to early human embryos is also critical for understanding fragile X syndrome, and possibly other diseases involving DNA repeats. Recent evidence suggests that the repeat expansion that leads to full-blown disease may occur not during meiosis, but shortly after fertilization. Therefore, it is questionable whether a full expansion could be detected by blastomere diagnosis.
In strict Mendelian genetics there is no functional difference between a gene inherited from the male or female parent. In mammals, however, some genes are expressed differently in an offspring—depending on which parent contributed that gene—through a process called genetic imprinting. A number of human diseases have been found to be associated with genetic imprinting, particularly certain types of juvenile-onset cancer. An experimental system is needed to investigate the mechanisms that control the differential marking of genes in sperm and oocyte meiosis that affects their later expression in the embryo.
OPEN DISCUSSION PERIOD
Discussion opened with a debate regarding the percentage of the genome that is actually amplified by PEP and means by which to optimize the technique. The usefulness of early embryos for studies of imprinting was also questioned because imprinting may occur only postfertilization.
Technical questions were focused on the relative merits of PCR techniques versus FISH. Whereas FISH can clearly identify certain chromosomal abnormalities, PCR techniques are more suited for identification of mutations and abnormalities in discrete regions of chromosomes. Further investigation of both these, and other, methods to analyze genes and chromosomes in single cells from embryos is needed.
A participant asked the cost of preimplantation genetic analysis. Because it is still a non-billed experimental protocol, its ultimate cost is difficult to esti-
mate, but should not be much higher than prenatal testing on chorionic villi sampling or amniotic material with the same DNA technology. The cost of IVF was a recurrent topic of concern. In the United States it can range from $1,500 to $12,000 a cycle. On average, three or four cycles are performed in the course of treatment. An interesting comparison may emerge between IVF pregnancy rates of parents utilizing preimplantation genetic analysis, who have normal reproductive systems, and rates among the more common IVF parents who have reproductive difficulties.
A persistent question concerning genetic analysis is confidentiality, for example, when an embryo is implanted that is known to be an unaffected carrier of genetic disease. Such concerns exist against a broader background involving a growing number of genes being sequenced and the way in which decisions will, or should, be made—and by whom—regarding access by government or private insurance companies to a patient's genetic profile.3 As one participant pointed out, everyone carries genetic traits potentially detrimental to one's self or one's offspring. As information accrues, the way in which insurance risks are calculated may have to be reconsidered.











