SESSION III
Fetal Research
Overview: The Fetus as a Patient: The Origin of the Specialty
JOHN QUEENAN
Georgetown University Medical Center, Washington, D.C.
Before the last half-century, the fetus in the womb was essentially inaccessible to medical diagnosis or treatment. The beginnings of fetal medicine came in the late 1940s with the use of penicillin to treat syphilis in pregnant women, which prevented the congenital effects of the disease in their offspring. Over the last 40 years, revolutionary new technologies have opened the way to direct intervention to correct fetal disorders, initiated in large part through efforts to combat Rh disease.
The Rh factor is a substance on the surface of red blood cells. In a small percentage of the population the factor is missing. Rh incompatibility between a mother and fetus can incite the mother's immune system to attack the fetal blood cells. Normally, an Rh-negative woman's first such pregnancy is not adversely affected, but with the immune reaction established, subsequent pregnancies become jeopardized. Before effective therapy was developed, the mortality rate was 50 percent.
The first leaps forward in the treatment of Rh disease came in the 1950s with exchange blood transfusion of the baby immediately after birth, or preterm delivery, when the mother's Rh history was known. These measures cut the death rate in half, but there remained the problem of undiagnosed disease. The application of amniocentesis, in which a sample of amniotic fluid is drawn by needle from the uterus and analyzed for various genetic and chemical characteristics, provided the means for detecting and evaluating a fetus at risk. In the
1960s, methods were developed to perform intrauterine transfusions. As a result of these advances, the Rh disease death rate dropped to 5 percent.
With intrauterine transfusion a fetus could, for the first time, be treated directly to correct the anemia and heart failure that can occur in Rh disease. However, because the blood was transfused into the peritoneal cavity (the fluid-filled space between the abdominal wall and abdominal organs), it entered the fetal bloodstream too slowly to be very effective against hydrops, a severe, potentially fatal edema, or fluid accumulation. In the past few years, it has become possible to transfuse the fetus intravascularly, directly through the umbilical vein, and reverse hydrops.
When amniocentesis and intrauterine transfusion began, there were virtually no methods for accurately guiding a needle into the uterus. Ultrasound, in which sound waves bouncing off the fetus create a moving picture in real time, has made access to the womb substantially safer. Ultrasound and intrauterine intravascular transfusion have now lowered fetal mortality from Rh disease to 2 percent.
Ultrasound and intravascular transfusion have revolutionized fetal medicine. In addition to Rh disease, ultrasound detects life-threatening obstructions in the urinary tract, chest, and skull; diaphragmatic hernias; and cardiac dysrhythmias. Intrauterine intravascular intervention can reverse nonimmune hydrops, a fetal edema unrelated to Rh hydrops. The causative agents of this condition, including viral infections, number 100 or more. Untreated, the mortality rate can range from 50 to 90 percent.
Many fetal disorders cannot yet be treated, or treated adequately. Surgical interventions to repair severe developmental abnormalities have yielded uneven results and require further exploration. Innovative approaches are needed, which may solve such problems as inborn errors of metabolism, blood diseases, and structural abnormalities. Among technologies on the horizon, fetal gene therapy and new applications of intravenous intervention hold promise for eliminating a variety of fetal ills.
Percutaneous Umbilical Blood Sampling
KENNETH MOISE, JR.
Baylor College of Medicine, Houston, Texas
The procedure for intravenous entry into the fetal bloodstream is called by different names. The term PUBS, for percutaneous umbilical blood sampling, is commonly used in the United States; equivalent terms include cordocentesis and funipuncture. In PUBS, a needle guided by ultrasound is introduced into a blood vessel (usually the vein) of the umbilical cord to assess fetal status and treat fetal disease, particularly diseases of the blood.
PUBS was developed primarily in France, in the 1980s, to diagnose fetal toxoplasmosis, an infectious disease that can be transmitted by a pregnant wom-
an to her fetus and can damage the fetal nervous system. Although relatively rare in the United States, toxoplasmosis is a common problem in France and elsewhere.
Between 1987 and 1991, an international registry monitored the applications of PUBS. Data collected from 7,000 procedures conducted in 11 centers showed PUBS being used most frequently to obtain chromosome diagnoses faster (within 48 hours) than is possible with amniocentesis (which requires weeks for results) in situations where delay might have serious clinical implications. In addition to toxoplasmosis, Rh disease, and nonimmune hydrops, PUBS has been used to diagnose fetal thrombocytopenia, a hemorrhaging condition caused by maternal autoimmune antibodies to platelets that cross the placenta, and to monitor drug levels and hyperthyroidism.
Therapeutically, PUBS has been indispensable in reversing hydrops in Rh disease. Data on 400 Rh fetuses transfused by PUBS, as reported at a 1989 PUBS conference, showed an 82 percent survival rate for hydropic fetuses. Survival rates vary from center to center, probably reflecting the severity of the cases handled. As PUBS is tried for reversing other conditions, varying degrees of success are being reported. Eight fetuses have been treated by means of PUBS for nonimmune hydrops caused by one of the recently identified agents, B19 parvovirus. All eight procedures resulted in the birth of healthy babies. In five attempts to correct chronic loss of blood between the fetus and its mother, only one transfusion was successful. Efforts to transfuse blood platelets into the fetus in cases of maternal-fetal platelet incompatibility proved impractical because platelets have a short half-life and multiple transfusions are required. Platelet transfusions performed near term, however, have averted the necessity for cesarean delivery.
Stem-cell transfusion is a new promising application of PUBS in which the cells that give rise to different classes of blood cells are transfused into fetuses with some type of blood cell disease. Of five stem-cell transfusions reported, three of the fetuses survived. One survivor had bare lymphocyte syndrome, a condition in which white blood cells fail to produce cell-surface molecules essential to immune response. Another had severe combined immunodeficiency (SCID). The third had beta thalassemia, an anemia widespread among people of Mediterranean origin, in which hemoglobin production is defective.
PUBS is not without risk. The PUBS registry's overall fetal fatality rate was around 1 percent. Other surveys have shown somewhat higher or lower rates. Survival is critically related to the particular disease and how far it has advanced; in fetal infection, mortality can be 11 percent, and in nonimmune hydrops as high as 31 percent. There is a risk of slowed heart rate, usually transient but more common if the umbilical artery is used, and of hemorrhage. In PUBS transfusions, there is the danger of the fetus moving and bumping into the needle, causing lacerations of the blood vessels. To prevent this, the fetus is often given drugs that induce temporary paralysis. When such risks are clearly
explained to the mother, PUBS can be a very effective and lifesaving procedure. The risks, however, mitigate against the use of PUBS for prenatal screening when there are no indications of fetal illness.
Embryoscopy and New Advances in Fetal Diagnosis and Treatment
E. ALBERT REECE
Temple University, Philadelphia, Pennsylvania
Although great strides have been made in fetal medicine, current diagnostic and therapeutic techniques have, for the most part, been limited to the second and third trimesters of pregnancy. In the early stages of pregnancy, ultrasound produces only a generalized, blurry picture of the embryo. Conventional fetoscopy, in which a viewing scope inside a needle is inserted into the womb, requires a needle too large to be used safely for early embryonic examination. Embryoscopy, a newly developed refinement of fetoscopy using a much finer needle, provides both direct visualization of an early embryo in exquisite detail and access into the embryonic circulation.
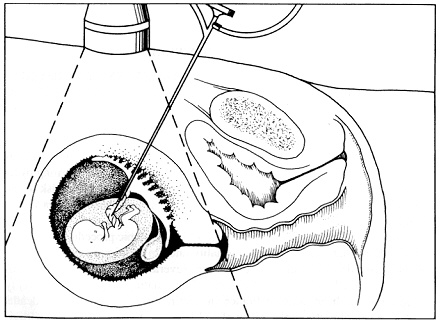
Figure 3
Schematic of embryoscopy method using transabdominal approach. SOURCE: Reece, E.A. ''Embryoscopy: New Developments in Prenatal Medicine,'' Current Opinion in Obstetrics and Gynecology, 4:450, 1992. Reprinted with permission.
Embryoscopy can be performed through the vagina or the abdomen. In the transvaginal approach the scope is guided through the cervical canal, the chorion (the outer embryonic membrane), and into the exocoelomic space. The limitation is around 12 weeks, when the chorion and amnion fuse and there is risk of rupturing the amnionic membrane. To reduce complications, a new transabdominal approach has been developed, similar to amniocentesis, in which the needle, guided by ultrasound, goes directly through the uterine wall and into the amniotic cavity (see Figure 3 on p. 29). Recent trials of transabdominal embryoscopies were performed on more than 12 patients undergoing pregnancy terminations. They yielded complete anatomical surveys of the fetus. Blood sampling and infusion of dye were conducted and did not harm the fetus or the mother.
Transabdominal embryoscopy should significantly enhance capabilities for early diagnosis and therapy. It could pave the way for treatment of genetic and stem-cell diseases during a period when the fetus is immunologically naive, thus avoiding the problem of immune rejection of transplanted or injected donor cells. Many disease genes have been identified, and model systems for human gene delivery have been applied successfully in experiments in both culture and laboratory animals. Although the optimal times for intervention would have to be worked out, it is altogether conceivable that stem-cell and gene therapy could be initiated early enough in gestation to reduce or prevent irreversible damage to a developing embryo.
OPEN DISCUSSION PERIOD
The advisability of using paralyzing drugs during PUBS was challenged by a participant who also employs the procedure. Their use was argued to be necessary for PUBS transfusion, which takes longer than blood sampling and so runs a greater risk of fetal movement. In an early case in which the fetus was not immobilized, it moved and dislodged the needle, which caused fatal lacerations to the vessels. Another discussant brought up the possibility of a woman contemplating PUBS being misled by the PUBS registry statistics concerning overall safety. In all cases, counseling should be based on the particular risk to the individual fetus, and the higher risks associated with specific fetal diseases should be outlined clearly.
A questioner wanted to know how many embryoscopic procedures had been performed, in what context, and the mortality rate. In addition to the 12 transabdominal cases described in the presentation, several hundred transvaginal embryoscopies have been conducted in cases of terminating pregnancies. Seventy procedures have been done in France in continuing pregnancies; two deaths resulted. Regarding the applicability of embryoscopy to gene therapy, a participant wondered whether sufficient blood could be drawn to retrieve enough cells into which altered genes could be inserted. Blood sampling in early embryos is difficult, and the maximum amount that can be taken has not been determined.
Maternal Nutrition and Metabolism: Role in Intrauterine and Long-Term Development of the Offspring
BOYD METZGER
Northwestern University, Chicago, Illinois
That good maternal and infant nutrition is essential for optimal pre-and postnatal development is universally accepted. Exactly what constitutes good nutrition, and how malnutrition impedes development, have not been clearly defined. Recent advances in epidemiology and clinical medicine have uncovered important clues linking nutrition and metabolism during prenatal development to long-term health. Pregnancy complicated by maternal diabetes is a situation in which these issues have been explored in depth.
The environment in which a fetus grows is conditioned by the maternal metabolism and the function of the placenta, through which all nutrients must pass. The placenta has the capacity to modify nutrients and contribute its own metabolic and hormonal input. Insulin plays an essential role in determining the composition of the maternal metabolic environment and the fetus's nutrient intake.
In the traditional model that has been used to explain the effects of maternal diabetes on fetal development, insulin deficiency in the mother disturbs insulin-sensitive nutrients, including glucose and many amino acids, the subunits of proteins. These nutrients, modified by the placenta, produce an altered nutrient mix that impinges on the beta cells of the fetal pancreas and leads to fetal hyperinsulinism. (Insulin is made by clusters of beta cells, called the islets of Langerhans, dispersed through the pancreas.) Excessive insulin production alters fetal development and the maturation of insulin-sensitive cells, including lung, heart, liver, and especially, fat cells.
A study was set up 15 years ago to test the model in a group of diabetic mothers and their fetuses, and to follow long-term effects on the offspring. As is typical of babies of diabetic mothers, the babies in the study were fat at birth and returned to a normal weight level shortly after. The situation later reversed, so that by about age six the incidence of obesity was very high, and by early adolescence so was the incidence of impaired glucose metabolism.
The most important insight that has come out of these studies to date is that the tendency toward obesity and metabolic abnormality in the children correlated directly with their state of hyperinsulinism during gestation. Furthermore, the effects were the same whether the mother was diabetic before pregnancy or developed gestational diabetes. In addition, impaired fetal islet-cell development has been linked to pre- or postnatal growth deficiency, in particular, secondary to protein deficiency in infancy.
The propensity to obesity and impaired glucose tolerance in the offspring of diabetic mothers establishes a setting in which these conditions may be perpetuated congenitally from generation to generation. Confirming data from animal
studies show that altered fetal islet function is sustained through subsequent generations. Among the Pima Indians of Arizona, who tend to marry within their own group, a large percentage of the population suffers from severe obesity and diabetes.
The next step is to learn how to intercede in this process. If intrauterine malnutrition and growth retardation in the first year of life could be prevented, and maternal diabetes more effectively treated so as to prevent fetal hyperinsulinism, the cycle of congenital diabetes could be broken and the ever-increasing incidence of the disease arrested.
Hormonal Regulation of Human Fetal Growth
STUART HANDWERGER
Children's Hospital Medical Center, Cincinnati, Ohio
Although there is considerable information about the hormonal factors that regulate postnatal growth, these factors appear to have scant effect, if any, on the fetus. Relatively little is known about hormonal regulation of fetal growth. Gaining such knowledge is of critical importance clinically since aberrations in fetal growth are frequently involved in diabetes and in many of the other common pathological conditions of pregnancy. Intrauterine growth retardation may have a lifelong effect on the incidence of illness and early death.
A number of findings have demonstrated that the pituitary growth hormone so important to growth after birth (deficiency leading to short stature and excess to gigantism) is not at work prenatally—infants born without a pituitary gland are normal in size. Although pituitary growth hormones are present in a normal fetus, fetal tissues lack receptors for them. (Receptors are the molecules on cells that initiate cellular response to hormones.) Insulin-like growth factor (IGF-1), whose production is increased postnatally by the action of pituitary growth hormone, does appear to be at work in fetal growth. Recent experiments have shown that the absence of IGF-1 leads to significant fetal growth failure. This suggests that in the fetus, something other than pituitary growth hormone is driving IGF production.
Fetal growth seems to be regulated, to a large extent, by a hormone in the placenta called human placental lactogen (hPL). Its role was first suggested by the striking similarities in its structure and function to pituitary growth hormone and to prolactin, another pituitary hormone, and the genes that encode the three hormones are closely related. Placental lactogen is made in the placenta and secreted into the maternal blood beginning at about six weeks into gestation. Production continues to increase, reaching a concentration at week 34 greater than that of any other hormone in the mother. Decreased concentrations in the mother have also been correlated with fetal growth failure.
Placental lactogen is active in both maternal metabolism and fetal tissue. In
the mother, hPL contributes, along with growth hormone and prolactin, to preparing the breast for lactation, and it stimulates maternal IGF production, which may be involved in increasing uterine size. More importantly, it increases the availability of glucose and amino acids for transport across the placenta to the fetus. In fetal tissues, placental lactogen directly stimulates IGF production, uptake of amino acids, and the activity of an enzyme important in protein and carbohydrate synthesis.
Contrary to expectation, the release of hPL is not regulated by the factors that regulate growth hormone or prolactin, but by high-density lipoproteins (HDLs). Lipoproteins are complex compounds of lipids and protein. Almost all of the effects of HDL that have been observed postnatally (in cholesterol metabolism) involve the lipid part of the compound. In contrast, the release of hPL in the fetus is effected by the protein part.
Aberrations in hPL secretion have been detected in many complications of pregnancy, including diabetes and preeclampsia, a condition associated with intrauterine growth retardation. It may be possible to prevent these conditions by administering placental lactogen or drugs that increase hPL concentrations. Before that can be ascertained, however, considerably more human research is required to understand the mechanisms involved.
Clinical Evaluations of Fetal Loss: Relevance and New Directions
CYNTHIA CURRY
Valley Children's Hospital, Fresno, California
Approximately 15 percent of all clinically recognized pregnancies end in spontaneous abortion. The increasing tendency of women to postpone childbearing has made such loss all the more distressing, because in some cases a spontaneous abortion is the first clue to problems that may compromise every succeeding pregnancy. In these and most other cases of spontaneous abortion, knowing the underlying causes is important to the parents and critical to a physician's ability to counsel patients concerning future pregnancies. Yet, with some notable exceptions, clinical evaluation of fetal loss, despite its diagnostic and psychological benefit, has been largely neglected. Numerous factors are responsible for this neglect. For example, complete evaluations require the expertise of geneticists, pathologists, and radiologists, among others. Thus, the evaluations require careful planning and coordination, and they must be financially supported in some way. In addition, it is often unclear which physician should be in charge.
To be complete, evaluation of fetal loss should include a history of the pregnancy, examination of the fetus, chromosomal studies, photographs, x rays, autopsy, and examination of the placenta and cord. The mother's history should be taken, including results of fetal monitoring tests. It is important to know
about the mother's glucose control as well as any maternal illnesses. Congenital myotonic dystrophy is an inherited condition that can be difficult to diagnose in a mother who is asymptomatic; yet a significant percentage of future pregnancies carry a 50 percent risk of being affected by it.
Causes of death differ in fetuses that have become macerated (e.g., not delivered for sometime after fetal death has occurred) and those that are freshly expelled. Macerated fetuses have a significantly higher chance of having been aborted because of chromosomal abnormality, a placental problem, or multiple malformations. Fresh fetuses are more likely to have died of nonrecurrent causes, such as cord accidents or infection. Chromosome studies need to be performed in fetuses when the mother has had recurrent losses. Such studies should also be done in any fetus with ultrasound-detected or obvious external abnormalities and in fetuses with hydrops, growth retardation, or maceration.
In a study of 500 cases of pregnancy loss, a cause could be established in 90 percent of intact and 30 percent of nonintact fetuses. Chromosomal abnormalities accounted for 19 percent; placental abnormalities, 11 percent; infections, 7 percent; cord abnormalities, 6 percent; neural tube defects, 8 percent; renal, central nervous system, or cardiac abnormalities, 6 percent; twins, 5 percent; recognizable syndromes, 4 percent; skeletal deformities, 2 percent; and other causes, 11 percent. Of particular significance was the finding of maternal immune rejection (possibly preventable) in about 10 percent of the losses. Certain placental pathologies were typical of this condition and underscore the importance of examining the placenta in cases of fetal loss. The fetal findings in maternal "immune rejection" included growth retardation and hydrops. Most of the women also had a history of recurrent miscarriages.
Many fetal conditions are yielding to advanced diagnostic and therapeutic modalities. The role of the placenta in fetal loss offers a major new direction for exploration. Families want and deserve answers as to why a miscarriage happened and what can be done to prevent its happening again. The best way to provide such answers is to know more about the precise mechanisms of fetal loss.
OPEN DISCUSSION PERIOD
A participant asked whether altered islet-cell function in the children in the diabetes study involved permanent down-regulation of insulin receptors. The question cannot be answered at present. Insulin sensitivity and beta-cell function continue to be measured in the children who show impaired glucose tolerance, and comparative information is being sought through animal studies.
Another participant asked what is known about the receptor for placental lactogen and where it is distributed in the fetus. Studies of sheep fetuses show the presence of hPL as early as six weeks into gestation, binding to cells in the liver, ovary, testis, and adrenal gland. In mice there is also a high degree of
binding in the brain, which raises the question of whether hPL could affect maternal behavior. Isolating and purifying the fetal hPL receptor, which has been pursued for a number of years, has proved extraordinarily difficult. Most of the studies of human placental lactogen have been done on tissues at term, well past the period of most rapid fetal growth. Second trimester studies have not been done because of the difficulties of obtaining tissue.
As to the evaluation of aborted fetuses, a medical school dean wondered who pays for the tests. Patient insurance covers examination of a fetus less than 20 weeks as a surgical procedure. After 20 weeks, placental examination is covered. The study reported in the presentation was supported by grants. Nevertheless, such tests are often supported by academic medical institutions as part of overall research and training and, as such, are threatened by increasing financial constraints on these institutions.
Therapeutic Interventions in Utero
RICHARD BERKOWITZ
Mount Sinai School of Medicine, New York, New York
Current methods for intrauterine fetal therapy encompass noninvasive measures, principally bed rest and transplacental drug delivery, and invasive procedures, such as preterm delivery, blood sampling and transfusion, percutaneous shunts, and open surgery.
Bed rest, the most common form of noninvasive therapy, is generally thought of as cheap and without significant side effects. Bed rest in the hospital, however, is not cheap, and being confined to bed for weeks can have major side effects. More important, it rarely works. Transplacental therapy, in which drugs given to the mother pass through the placenta to the fetus, effectively treats a variety of conditions. Drugs or vitamins that cross the placenta have been used to accelerate fetal lung maturation, reduce the risk of neural tube defects, and manage cardiac arrhythmias.
Among invasive therapies, percutaneous umbilical blood sampling, which provides direct entry into the fetal blood circulation, enables diagnosis, intravenous transfusions, and drug delivery. PUBS has been used recently to diagnose and track the effectiveness of therapy for alloimmune thrombocytopenia.
Like Rh disease, alloimmune thrombocytopenia is caused by maternal-fetal incompatibility. In this case, maternal antibodies attack the platelets in fetal blood, which causes a marked reduction in their number (i.e., thrombocytopenia) and may lead to devastating intracranial hemorrhaging. The treatment that has been used is gamma globulin administered intravenously to the mother, sometimes with steroid hormones.
To evaluate and refine this therapy, a trial was conducted with women who
had previously delivered babies with the disease and had been shown, by PUBS testing, to be carrying severely thrombocytopenic fetuses. Half of the group was given gamma globulin alone, and half, gamma globulin with low doses of a steroid (dexamethasone). Fetal platelet counts were reassessed with PUBS three weeks after the beginning of the regimen and again six weeks later. If at three weeks the fetal platelet count was less than 20,000/ml3, the mothers were additionally given high doses of a steroid, prednisone, for the remainder of the pregnancy. Among the women who did not require the additional therapy and have since delivered, there was no essential difference in outcome with or without hormones.
A therapeutic success was defined as a birth platelet count greater than 30,000/ml3 since there have been no reports of intracranial hemorrhage with higher platelet concentrations. This was achieved in 82 percent of cases. Fifty percent of those fetuses whose initial platelet count was less that 20,000/ml3 showed a therapeutic response at the time of the second PUBS. Of greatest importance, however, is the fact that regardless of the platelet count at birth, no infant in this study had an intracranial hemorrhage. These data strongly suggest that medical therapy for this disorder will be effective in the vast majority of cases.
An additional finding from this multicenter study was the critical role of fetal platelet concentration as a risk factor for PUBS-induced fetal loss. Eight untreated fetuses, all with a platelet concentration of less than 20,000/ml3 (six actually had counts of less than 10,000/ml3), died following the initial diagnostic PUBS procedure. As a result, the investigators recommended obtaining measurement of the platelet concentration before removing the needle from the umbilical blood vessel and, if the concentration was less than 50,000/ml3, transfusing previously prepared antigen-negative packed platelets into the fetus prior to removal of the needle. No fetal losses have occurred after adoption of this regimen.
Percutaneous shunting is a form of intervention designed to drain abnormal accumulations of fluid within the fetus that can severely compromise the growth and development of adjacent organs. The shunts are put into the obstructed area by a needle guided by ultrasound. Shunts have been used to alleviate obstructions of the bladder outlet, pleural space, and ventricles of the brain. A registry of bladder shunt procedures showed an overall survival of about 40 percent, but with a high risk of renal disease later in life. Long-term results have been more encouraging for hydrothorax, which hampers development of the heart and lungs. Shunting in utero has been much less successful for hydrocephalus, however, where fluid builds up within the ventricular system of the brain. Although 83 percent of the shunted hydrocephalic fetuses survived, more than half had severe brain impairment, and as a result, there is now a moratorium on the procedure for this indication.
The most radical form of invasive intervention is now being attempted out
side the uterus. In a small number of selected cases where closed procedures cannot be performed, the uterus has been opened and surgery performed directly on the fetus. The results of this approach, have thus far been mixed, and its ultimate role remains to be determined.
Fetal Therapy
MITCHELL GOLBUS
University of California Medical Center, San Francisco, California
A number of advances, derived through animal research, made it possible to think of attempting open surgical interventions (outside the uterus) on fetuses whose prospects for survival in the absence of intervention were dismal, but for whom interventions inside the uterus were not possible. The first fetal surgeries were performed on fetal bladders to correct urinary tract obstructions in instances in which shunting was not advisable. A shunt tends to clog after a number of weeks and, so, will only buy enough time for a fetus close to the point at which it can be safely delivered. In earlier stages, it becomes necessary to replace the shunt repeatedly, thereby putting the fetus in extreme danger.
Despite technical advances, though, such surgeries present major challenges. Specific anesthetics must be used that provide uterine relaxation as well as anesthesia. Opening the uterus causes preterm labor, which must be prevented by continuous intravenous administration of relaxing (tocolytic) agents through indwelling catheters for the remainder of the pregnancy. Fetal status during the surgery requires monitoring.
Of the eight original bladder surgeries, only three were successful. Two of the failures resulted from gastrointestinal problems that could not be recognized prenatally. Two involved kidney abnormalities at a time when it was not known how to evaluate fetal renal status. The fifth was caused by the failure of the mother to stay on tocolytic medications, which resulted in premature labor and delivery. Of the surviving children one required a kidney transplant and the other two are doing well.
The failures from inadequate diagnosis of other compromising conditions can now be alleviated by improved evaluation and testing. Although such tests can predict which fetuses are likely to survive the surgery and be delivered, they cannot predict future renal function. Nevertheless, many couples appear willing to accept the idea of a child who may need a kidney transplant, because they give consent to the surgery after being fully informed about the risk of later renal failure. Even so, the investigators wonder whether such couples can really appreciate the enormity of the burdens and concomitant risks of kidney transplants in the absence of experiential knowledge and in the face of trying to save their fetus.
Diaphragmatic hernia surgery was attempted next. These hernias cause the
stomach and other organs to lie in the thorax, crowding the heart and lungs and preventing their normal development. Although postnatal surgery can be done, mortality is high. In situations where the bowel is in the chest, there is not enough room for the lungs to develop. The bowel can be surgically moved back into the abdomen in the prenatal surgical procedure, but because the mass is too large for the abdomen that has developed, the gap must be covered with a patch and closed postnatally. This prenatal surgery could not be tolerated before 22 weeks. Unlike bladder surgery, which takes 10 minutes from the time the uterus is opened until it is closed, hernia repair requires an hour. In addition, surgery to bring the liver down from the chest proved impossible. Of the first 30 prenatal diaphragmatic hernia surgeries, only four fetuses survived. Among 100 fetuses with diaphragmatic hernia who did not have in utero surgery, the survival rate was 40 percent. Thus, despite the enormous knowledge gained, the majority of investigators have instituted a moratorium on prenatal procedures for diaphragmatic hernia.
Two other types of fetal surgery have shown more promise. Fetuses with cystic adenomatoid malformation of the chest, which impedes development of the lungs, can survive without intervention, but not if the condition is compounded by nonimmune hydrops. Surgery on six such fetuses resulted in four that did well, although intervention early in the hydrops was critical to their survival. Of five surgeries performed to remove an anomalous twin that was threatening the life of the living twin, four survived—a rate considerably better than nature's 50 percent.
Although a good deal has been learned about fetal conditions that are more or less amenable to open surgery and there have been some successful outcomes, it does not seem likely that the existing technology offers an approach that will save large numbers of endangered fetuses.
Transplantation of Fetal Liver Hematopoietic Stem Cells in Utero
ESMAIL ZANJANI
Ioannis A. Lougaris Medical Center, Reno, Nevada
Hematopoietic stem cells (HSCs) give rise to all the elements of the blood. In addition, they have the ability to self-replicate, so that their numbers do not decrease as mature cells form. In the adult, HSCs are found primarily in bone marrow. It is possible to transplant these stem cells to replace defective marrow cells, but in an immunologically competent adult, all of the patient's own marrow cells must be destroyed and the marrow repopulated with donor cells. Because the number of donors with compatible cell types is limited, there is the danger of graft rejection and the resulting need for sustained immunosuppressant therapy. In addition, there is a danger of graft-versus-host disease (GVHD), which results from the immunologically mature donor cells attacking the host's
cells. As is well known, fetal cells are not mature immunologically and, thus, make excellent donor cells for transplant.
Many of the congenital abnormalities that can be treated with HSC transplantation after birth can be diagnosed early in gestation. The question arises then of whether one can transplant stem cells into a compromised fetus to take advantage of an immature immune system and therapy avoid the problems of rejection and immunosuppressant therapy. The additional use of fetal donor cells, produced abundantly by the fetal liver, could circumvent the possibility of GVHD.
In early experiments in sheep, stem cells from adult animals were transplanted into fetuses. The sheep that were born showed acceptance (engraftment) of the transplanted cells, but they all developed graft-versus-host disease and died. In subsequent experiments, stem cells from fetal donors were used and none of the recipients developed GVHD. In fact, the donor cells were still active in the sheep several years after birth.
The next question was whether fetal HSC transplantation could actually correct a disease. Batten's disease causes blindness in sheep. Animals diagnosed with the disease in utero and treated with donor fetal HSCs were born normal. They were sacrificed at 15 months of age for pathological examination, and the data, although not yet complete, indicate that at the time of death the sheep were still free of disease.
Human victims of Hurler's disease, which is due to an enzymatic deficiency that causes accumulation of mucopolysaccharides in many organs, usually die before the age of 10 from respiratory infection and heart failure. In 1990 a fetus diagnosed with Hurler's was given fetal HSC transplants, injected into the peritoneal cavity. The child was born prematurely and showed signs of pulmonary disorder typical of early birth, but exhibited no indication of GVHD. However, the child was enzyme deficient at birth and, by six months, clearly exhibited the disease. Starting at about one year, tests began to show low levels of the missing enzyme, a trend that continued over the next year. The child is now two years old and still shows evidence of disease. Whether failure to cure him resulted from problems inherent in the particular condition or an insufficient level of cell transplantation is not known.
Research to intervene early in fetal life to alleviate specific diseases is hampered in humans by the difficulty of obtaining human fetal tissue for experimentation, the condition of the tissue that can be obtained (most of which comes from spontaneous abortions and is infected or genetically abnormal), and the social and political issues surrounding fetal tissue use.
Studies are now under way to improve methods of purification and preservation of stem cells and to determine persistence of engraftment. For example, incubation of stem cells in medium with certain growth factors seems to improve the engraftment of the donor cells in sheep. Fetal sheep stem cells have been grown in culture and their numbers expanded, with the idea that transplanting
more cells will improve success. Human fetal stem cells have also been successfully transplanted in sheep. Among the questions being explored is whether the pool of human HSCs could be expanded by using animals as a kind of ''living'' culture system. Will a small population of human fetal cells transplanted in fetal sheep or pigs expand in the adult animal's bone marrow? Will the cells be accepted by and beneficial to human recipients? These and other questions await further research.
OPEN DISCUSSION PERIOD
The data reported from the alloimmune thrombocytopenia trial led one conferee to suggest that the time had come for gamma globulin treatment to be considered cost-effective for coverage by third-party payers. It costs more than $1,000 a week per patient. Even with extended treatment, however, it is less than the cost of caring for a hemorrhaging infant or the cost of repeated transfusions.
In a discussion of success rates for treating obstructions that affect renal function, the point was made that the condition is often not diagnosed until the kidney damage is too extensive to save the fetus. Another participant wondered whether the collective incidence of prenatal problems that could be diagnosed and treated if caught early warranted the use of ultrasound in every pregnancy. Ultrasound scanning at 18 weeks would allow appropriate dating important for the evaluation of intrauterine growth retardation, including AIDS pregnancy, and is done for every pregnancy in Britain, Scandinavia, Germany, and France. Although not contesting its medical value, another participant replied that a recent literature search did not support the statistical cost-effectiveness of routine ultrasonography in pregnancy.
The feasibility of fetal cardiac surgery was raised. Some cardiac conditions might be candidates for prenatal surgery in the future, and animal studies have begun to explore this.
The discussions kept returning to the issue of the ultimate value and usefulness of fetal surgery. One point made was that given the difficulties encountered in surgical trials for diaphragmatic hernia, it was best that the trials were limited to one institution rather than being conducted by different centers using different protocols. In the view of one of the speakers, as more is learned about molecular and cellular factors controlling fetal growth and development, new approaches, such as stem-cell transplantation and gene therapy, stand to help many more patients than fetal surgery.
A participant questioned the use of intraperitoneal rather than intravenous transfusion for stem-cell transplantation. Technical difficulties have made intravenous cell transplantation less successful than intraperitoneal transplantation in sheep, but there are likely species differences. The resulting questions about how many cells were transplanted by the two routes led to an interesting scheme for "preparing" a fetus to receive a stem-cell transplant after birth, which has
been tried in mice. This approach would be to transfuse or intraperitoneally inject stem cells into a fetus in low numbers, with the assumption that the fetal immune system as it matured would recognize the transplanted cells as "self." After birth, a full stem-cell transplantation with cells from the original donor pool could take place with a much reduced chance of graft rejection.
Applications of Molecular Biology and Genetics to Developmental Toxicology
DANIEL NEBERT
University of Cincinnati Medical Center, Cincinnati, Ohio
The role of environmental substances in causing infertility, in utero toxicity, and birth defects is an important public health issue. Studies of the genetic mechanisms that control metabolic pathways have begun to clarify the ways in which toxic substances induce abnormalities in the developing fetus.
Enzymes of a group collectively called cytochrome P450 (P450) are involved in metabolizing the foods, drugs, and environmental chemicals that enter the body. Cytochrome P450 enzymes belong to a group called Phase I enzymes that add oxygen molecules to incoming substances. The oxygen functions as a sort of handle that Phase II enzymes, another group, use to hook or conjugate the substance to other chemical compounds in the body as a preparation for excretion. Many substances, such as foods or certain chemicals in cigarette smoke, enter the body already oxygenated. It has long been known that such oxygenated "intermediate" compounds cause cancer and birth defects in mice—for example, benzopyrene metabolites cause epithelial tumors, cleft palate, and kidney lesions, among other problems. In some cases, Phase II enzymes may actually potentiate the toxicity of an oxygenated compound by blocking its breakdown, thereby allowing the concentration of the toxic compound to increase.
In the 1960s, research was initiated to describe the functional relationships between various genes that encode drug-metabolizing enzymes (DMEs), including P450, that process a specific class of incoming compounds known as aromatic hydrocarbons. An early study that examined the P450 concentrations in human placenta found greater concentrations of the enzyme in placentas from women who had smoked more cigarettes during pregnancy. Other research focused on the receptors, proteins that "recognize" and bind certain molecules, for classes of incoming compounds. These and other lines of investigation provided a rich understanding of the mechanisms of chemical toxicity and laid a useful foundation for more intensive work. For example, it became possible in the 1970s to identify the genes for two types of receptors that bind aromatic hydrocarbons, one of which binds such compounds with vigor (high affinity) and one of which binds only weakly (low affinity).
Knowledge of aromatic hydrocarbon (Ah) receptor differences allowed investigators to give benzopyrene to pregnant mice and demonstrate that the offspring of mice with the low-affinity receptor had more birth defects and in utero toxicity. This finding is supported by work in humans showing that pregnant women with the low-affinity Ah receptor had babies with more birth defects than mothers with the high-affinity receptor, despite smoking the same number of cigarettes.
Another series of experiments was set up to examine the interrelationship of two Phase I and four Phase II DME genes in mice. In normal function, Phase II genes turn on and off to maintain a steady-state balance of metabolic activity. In the study, all six of the DME genes were activated by benzopyrene and by dioxin. It was assumed in the early 1970s that these DMEs existed only after birth and were not present in the fetus. Thus, the role of DMEs in birth defects could be attributed to the genetic susceptibility of the mother.
Doubt was cast on that assumption by findings from studies of the mechanisms of birth defects in mice, which suggested that the response in the embryo to certain environmental chemicals given to the mother occurred as early as day 5 and varied with the genetic makeup of the fetus. With the advent of more sensitive molecular techniques, it has been possible to confirm and expand this finding. By using methods of in situ hybridization in which genetic probes search out and combine with complementary genetic material, P450 expression was localized in mouse embryos. It occurred first in endothelial cells lining the uterus, then in the developing yolk sac, and by about day 12 or 14, in the developing liver and lung.
Recent experiments have been aimed at pinpointing the specific function of P1450, an extensively studied specific P450, in normal fetal development. By using a variant of the very sensitive technique of polymerase chain reaction, which provides a large amount of a particular gene for study, a striking increase in the expression of a gene encoding P1450 was discovered in the fertilized egg before the two-cell stage—expression that decreases in later stages of embryonic development. This finding supports the idea that the gene and its product are important at critical times in the preimplantation embryo.
Extrapolation from animal models provides an incomplete picture of human biology. There are drug-metabolizing enzymes in mice that are not present in humans and vice versa. One way to attack this problem is to make transgenic mice and insert human genes into these mice, thus inducing the production of proteins and enzymes that react as they would in humans to incoming chemicals. The use of transgenic mice can help to further our understanding of the complex interplay among enzymes that process drugs and toxic substances from food and the environment. In addition to the P450 genes, other DME genes are now implicated in developmental toxicity. Armed with sensitive molecular techniques and assays, we can now begin to manipulate this complex system experimentally in developing embryos. A complete understanding of the mechanisms
through which toxic substances cause birth defects clinically, however, can come only from application of the knowledge gained thus far to the study of human fetal tissue.
OPEN DISCUSSION PERIOD
A participant asked how many human genes would have to be expressed in transgenic mice to provide an adequate model of drug and environmental responses in humans. The number of genes involved in the important responses is estimated to be between 10 and 20. With so-called knockout technology, mouse genes can be made nonfunctional and replaced by comparable human genes. Transgenic mice can be bred to be simultaneously deficient for more than one gene. As the pharmacogenetic variations at P450 genes in humans become well-characterized, test tube assays can be set up to determine which women should or should not receive certain prescribed drugs on the basis of individual Phase I and Phase II cytochrome P450 alleles.


















