5
BIOLOGIC MARKERS IN EXTRAPOLATION
Extrapolation is common in many scientific disciplines—so common that it can easily go unrecognized. Extrapolation is concerned with the translation or transfer of relationships (e.g., clinical measures and mathematical variables) observed in one setting to another setting. When we extrapolate from experimental systems to humans, we assume that some cause-effect relationships are the same in humans as in the experimental systems. The more we understand about the variables in an experimental situation, the more we will understand about the validity of such extrapolation to human situations. In the context of biologic markers and urinary toxicity, our goal is to gain a better understanding of the overall relationship between exposure and disease by examining those markers. The relationships among markers that can be observed and tested in experimental systems might be extrapolatable from those systems to situations of concern with respect to human health risks, such as occupational or environmental exposures to urinary toxicants.
The main purpose of extrapolation in any context is to make it possible to predict. In the clinical setting, the observation of particular symptoms in a patient leads clinicians to conclude that the patient has a particular disease or will soon manifest other symptoms. In that case, the clinicians are extrapolating from their experience with some patients to a new patient. Extrapolation is required to support prediction and the design of a suitable treatment.
Extrapolation from epidemiologic studies is commonly used to predict risks to other cohorts. Every epidemiologic study is restricted to some population. To extrapolate from a study population to other potentially affected populations, one must consider the differences between the study population and the potential target populations. Variables related to and affecting the development of the health effect under investigation must be considered; differences between the study and target populations with respect to those variables will influence how the extrapolation is completed by influencing or modifying
the relationships between variables and the health effect.
Consider the situation of a new chemical that is proposed for use or that will be a byproduct of some new operation. Direct evidence that the chemical causes adverse effects in humans is lacking. Some important considerations include the potential for adverse health effects in humans exposed to the chemical and, if adverse effects do result from exposure, the magnitude of the effects after exposures of different severities. It is clear that predictions are required. However, the basis for the predictions cannot be the previous human experience; there is no previous experience. It might be possible to extrapolate, but the model from which the extrapolation is made will of necessity be a nonhuman model.
In the scenario just described, the need for extrapolations from experimental systems to humans is apparent. Many other scenarios, both clinical and "population-based," will require the prediction of human responses from data obtained in nonhuman test systems, especially in light of the thousands of chemicals that are produced, used, and released into the environment as byproducts of our way of life. Indeed, it is the desire to be predictive that drives the need to develop and apply good experimental systems. Such systems have at least four advantages: they allow predictions of human health effects and the magnitude of those effects before human exposure occurs or before adverse effects are manifested in exposed populations; they can be altered to clarify aspects of the process leading from exposure to adverse health effects when similar experimentation in humans would be unethical; they can be designed to eliminate many factors that confound the determination of cause-effect relationships in epidemiologic studies; and they can suggest directions for epidemiologic investigation by providing the hypotheses that epidemiologic studies might be able to test.
Previous chapters have focused on markers of susceptibility, exposure, and effect (particularly early effect) and their value in clinical situations; the sooner a disease state or precursor of a disease state can be identified, the greater the chance of successful therapy or treatment. This chapter focuses on the prediction of effects, not in an individual patient but rather in a (hypothetical) population of humans potentially exposed to a supposed toxicant. In this context, one is concerned about maintaining the health of the population by predicting whether an activity or an exposure is likely to produce harmful consequences in that population—often without previous observations of humans exposed at the magnitudes of interest. The objective is to learn how to tie chemical exposure under various scenarios to the dose or amount of the chemical that reaches the body, to the amount that is absorbed and distributed to target tissues, and ultimately to the effect.
How does one make such predictions? The discipline of risk assessment addresses that question. Human-health risk assessment is a complex,
multifaceted process that relies on data and scientific principles from many disciplines to determine whether a chemical is toxic and the likelihood of manifestation of detrimental effects under specific conditions. Risk assessment draws on a variety of methods and models to examine and evaluate information about the toxicity of a chemical. A useful and well-described system for looking at toxicity information is the risk-assessment paradigm originally depicted by the National Research Council in the 1983 publication Risk Assessment in the Federal Government: Managing the Process. That system for organizing and analyzing risk information follows an understandable series of steps portraying qualitative and quantitative aspects and typically includes some or all of the following: hazard identification, dose-response assessment, exposure assessment, and risk characterization (NRC, 1983). The result is a characterization of the potential adverse health effects of human exposures to a chemical.
The characterization of potential adverse human health effects requires extrapolation. That typical of many risk assessments includes extrapolation from animal test species to humans, from large exposure to small exposure, and from one route of exposure to another. Risk assessments are not infallible, and the relative accuracy of a risk assessment depends not only on the scope and quality of the scientific data but also on the reliability of the methods and the validity of the models used. The degree of confidence in a risk assessment depends on how well data and model quality are validated and on the extent to which uncertainty is quantified.
As described in Chapter 2, various aromatic amines are recognized human bladder carcinogens (NRC, 1981). Consider, for example, an assessment of human bladder-cancer risk associated with dermal exposure to one of those aromatic amines—4,4'-methylene-bis(2-chloroaniline), or MOCA. For such an assessment, we might be required to extrapolate relationships that were observed in dogs exposed to MOCA in their diet (Stula et al., 1977) at doses far greater than the human exposures of interest. The conclusion that the occurrence of bladder cancers in dogs exposed to MOCA implies a bladder-cancer risk for humans assumes that the qualitative relationship between MOCA exposure and bladder cancer can be extrapolated from dogs to humans. Such cross-species extrapolation is typical of the hazard-identification component of risk assessment, that is, the determination of the existence of a cause-effect relationship between chemical exposure and adverse health effect.
Mathematical extrapolation is particularly relevant to risk assessment of a most useful kind, i.e., quantitative risk assessment. Quantitative risk assessment is a means of providing a measure of the risk of some harm as a result of a specific exposure to some substance or activity (Almeder and Humber, 1987), and mathematical extrapolation can be conceived of as the transfer of the quantitative relationships estimated in one scenario to another scenario, whether
those scenarios differ in species (animal-to-human extrapolation), in magnitude of exposure (dose extrapolation), or in route of exposure (route-to-route extrapolation). Again, it is assumed, lacking data to the contrary, that a relationship observed in one scenario is valid in the other. Risk can be stated as the magnitude of exposure that is estimated to be without substantial likelihood of harmful effect, or stated as the probability of occurrence of harmful effect.
The remainder of this chapter concerns answers to two questions: What is the basis for concluding that extrapolation is reasonable? Can it be said that one approach to extrapolation is better than another approach? It is in relation to the second question that the relevance and utility of biologic markers in risk assessment become apparent.
BASIS OF EXTRAPOLATION
Animal Studies
A fundamental principle of toxicology is that results of animal studies can be applied to humans. The scientific basis for assuming that animals are good surrogates for humans and therefore a suitable basis for extrapolation to humans is overwhelming. If one considers that the genetic makeup of a mouse or a rat is more than 95% and of a monkey is more than 99% identical with that of a human, it is reasonable to assume that these animals in particular and mammals, in general, will react to infectious agents and chemical stressors much as humans will. Among mammals, most of the host defense mechanisms (barrier and immune) and metabolic (anabolic and catabolic) systems are similar. In particular, the urinary systems of most mammals are very similar. Although specific, often subtle, differences between humans and other animals with respect to renal function have been demonstrated, the vast majority of human renal responses to xenobiotics mimic what has been observed in other species. Biologic markers of renal transport, concentrating, and metabolic functions of the kidney are reproduced in many species, including humans, although quantitative differences have been demonstrated. Therefore, it is reasonable to use animal models for extrapolation to humans unless specific information on specific chemicals dictates otherwise.
Many epidemiologic investigations have, in fact, been suggested as a result of animal studies, and the epidemiologic findings have tended to support the results of the animal studies. For example, several of the current epidemiologic studies of heavy-metal toxicity, including small exposures, were initiated in response to urinary toxicity observed in a large number of animal studies and were undertaken specifically because of the likelihood that the human response would mimic that seen in animal test species. Mechanistic studies of chemically induced nephrotoxicity in animals influenced or stimulated epidemiologic studies of a variety of substances, including many of the halogenated hydro-carbons, such as chloroform, hexa-
chlorobutadiene, and bromobenzene. Much of the epidemiologic investigation and mechanistic understanding of anesthetic, analgesic, and antibiotic nephropathy has been driven by observations of nephrotoxicity in animals. Classic examples of the direct application of animal studies to epidemiology of known nephrotoxicants are the use of antineoplastic and immunosuppressant drugs (e.g., cisplatin and cyclosporin, respectively).
Studies of carcinogenic responses in laboratory animals and in humans have revealed substantial correlations (Crump et al., 1989). The results suggest that there are reasonable approaches to extrapolating cancer responses observed in test species—approaches that appear to predict fairly well the responses in humans.
Identification of chemical hazards should include assimilation and evaluation of all relevant information. Appraisal of physical and chemical properties and structure-activity relationships can sometimes provide important indications of potential toxic characteristics. Markers of urinary function and chemical toxicity in experimental animals can be studied at various levels of tissue structure and organization (Table 5-1). This is in contrast with human studies, in which only noninvasive studies of renal function are possible. Markers identified through in vitro studies of systems that use animal and human cells or tissues in culture can often give insight into potential toxicity. However, because of the intricacy of the body, only whole-animal studies or observations in humans provide information on the operation of multiple cells, tissues, and organs under the influence of complicated feedback mechanisms. Animals are necessary in the study of chemical-induced toxicity and for the development and validation of markers because studies that involve modulation of cellular responses and tissue sampling cannot be performed in humans.
Traditionally, experimental animal Studies have been of most value for identifying markers that can be used to predict target-organ effects, understand dose-response relationships, and study mechanisms of action. The studies include measurements of function, blood chemistry, urinalysis (including cytology), histopathology, and electron microscopy. Metabolism and transport peculiar to the kidney are often routine parts of such studies.
Animal models developed as surrogates for humans in the study of renal and urinary function should conform to some general principles, which are applicable to any organ system, although they are discussed here in the context of renal and urinary toxicology. They include the following:
-
The animal model should be reproducible within and among laboratories. It should not be so complex that only a few laboratories could do the study.
-
The model should be peculiar to the part of the urinary tract under consideration. This characteristic can be realized only with sophisticated procedures that permit study of discrete nephron segments.
-
The model should be sensitive enough to differentiate normal from abnormal changes in structure or function.
-
The model should be able to measure alterations in renal structure or function caused by exogenous agents.
Whole-animal studies are usually the first step in evaluating the potential toxicity of a given agent, and these studies involve assessment of urine and plasma for markers indicative of organ function and toxicity. Noninvasive or nondestructive studies can be followed by application of histopathologic techniques to determine markers of target-organ or tissue-site injury. In most cases, rodents suffice, but there might be instances when only a primate can properly represent the human situation. For example, the route of administration might be important if direct extrapolation to humans is likely. With nephrotoxicants as with other toxicants, acute, subchronic, and chronic exposures are used to determine potential toxicity. The determination of markers of urinary toxicity is generally easier with acute protocols than with subchronic or chronic exposures. Studies often involve single exposures of both sexes of at least two species, usually rodents. Depending on the results of the rodent studies and the questions being asked, one might decide to study the agent in higher mammals, such as dogs or monkeys. The use of subchronic or chronic exposure regimens is usually driven by the nature of the potential human exposure, the agent being studied, and the possibility of chronic toxicity, including carcinogenicity.
Renal Parenchymal Injury
The difficulties in diagnosing renal injury and predicting its health consequences are considerable. That is primarily because the kidney can undergo substantial chemically induced injury without any clinical manifestation; subtle injury can be negligible, given the considerable functional reserve of the kidney. For example, the single cross-sectional measurement of glomerular filtration rate (GFR) might show only severe acute or chronic renal damage, as discussed in Chapter 2. Most studies indicate that quantitative urinary-enzyme secretion patterns cannot reveal either the type or the severity of renal injury, and often they do not correlate with structural or functional changes, as discussed in greater detail in Chapter 4. The need, therefore, is for standard diagnostic criteria that are sensitive enough to serve as markers of renal damage in the presence of renal functional reserve.
Much of the nephrotoxicity that follows the administration of inert, relatively nontoxic chemicals is related to the formation of reactive electrophiles during their metabolism (Ford and Hook, 1984). It is thought that the electrophilic products can react covalently with various nucleophilic sites on renal macromolecules and, by some mechanism yet to be defined, lead to renal damage. Measurement of the re-
active electrophiles or the covalently bonded compound with sensitive techniques might yield markers of renal damage (Harris et al., 1987; Omichinski et al., 1987; Reddy et al., 1984; Tyson and Mirsalis, 1985). However, those procedures require renal tissue; although they might yield useful markers of renal damage in experimental studies, they are not suitable for use in humans exposed to potentially electrophilic products of nontoxic chemicals.
Glomerular Filtration Rate
Evaluation of the blood-urea nitrogen concentration (BUN) is a common procedure for the indirect assessment of the GFR in experimental animals. As discussed in Chapter 4, it is unsuitable for quantitative purposes but might have utility in establishing the course of chronic renal failure in an experimental setting if renal damage is severe enough. Measurement of the serum creatinine concentration and urinary creatinine excretion, with calculation of the creatinine clearance, is generally preferred as an indicator of GFR. However, in some animal models, variable amounts of creatinine can be excreted via tubular secretion, and that reduces its utility as a marker of GFR. More subtle changes in GFR can be assessed by evaluating the clearance of various exogenous substances, such as inulin, EDTA, and iodothalamate.
Sensitive analytic procedures are available for measurement of those markers and GFR. Again, however, the extent of reduction of GFR in the face of a nephrotoxic insult might be hidden by the inherent renal reserve, and even measurements of GFR often are not sensitive enough to detect modest renal damage.
GFR can be assessed in either conscious or anesthetized animals. In both cases, the same markers can be used and their clearance determined with standard renal physiologic techniques. Both creatinine and inulin are used commonly to determine GFR. The use of anesthetized animals permits a more accurate determination of urinary flow than the use of conscious animals housed in metabolism cages. The use of anesthetized animals also permits the collection of precisely timed blood samples for determination of the marker under study. However, if conscious animals are used, GFR can be determined with reasonable accuracy with subcutaneous injection of the marker in a concentrated gelatin solution and collection of a single blood sample at the end of a 60-mill urine-collection period. Some researchers have suggested that the anesthetic agents by themselves can alter renal function.
Tubular Function
Tubular dysfunction in experimental animals can be assessed through relatively simple and inexpensive tests, such as those for the measurement of glucosuria, enzymuria, and osmolality. Some are sensitive enough to detect relatively small effects on the kidney after acute
administration of a nephrotoxicant, but caution must be exercised in predicting specific effects on transport processes or cell viability on the basis of the data obtained from these types of in vivo tests (Berndt, 1981). Some researchers (Daugaard et al., 1988; Dieperink et al., 1983) have used the renal clearance of lithium as a more subtle technique for evaluating renal damage that occurs during chronic studies; this noninasive method is applicable to humans (Thompson et al., 1984), as well as to animals. The loss of renal tubular function can also be assessed with test conditions that impose stresses on renal function, e.g., maximal urinary dilution or concentration or urinary acidification or alkalinization. Similarly, tests of maximal tubular reabsorption of glucose or maximal tubular secretion of p-aminohippurate (PAH) can be valuable in assessing tubular damage. They can also be applicable to humans and yield relatively sensitive markers of renal damage. However, these approaches require carefully controlled experimental studies and are not suitable for casual observations in the workplace.
Proteinuria
Proteinuria is the appearance of proteins in the urine after increase in the permeability of the glomerular membranes, reduction in tubular reabsorption of filtered proteins, shedding of specific constituents into urine as a consequence of cellular turnover or selective renal tubular damage, or a combination of the above. Glomerular or tubular damage can occur in the absence of a substantial reduction in GFR, so it has long been thought that the evaluation of proteinuria can be useful in detecting renal dysfunction at either the glomerular or tubular level. Although this topic is discussed in considerable detail in Chapter 4, a few comments concerning proteinuria. as a marker of renal dysfunction are incorporated here for the sake of completeness.
Although one can measure total protein in urine, it is a relatively insensitive assessment of renal damage. Total-protein measurement also offers no insights into whether one is assessing damage to glomerular membranes or to tubular membranes. A more rational approach is the use of electrophoretic separation of single proteins to provide a comprehensive approach to chemically induced renal dysfunction.
Proteins of relatively high molecular weight (over 45,000 daltons) usually are retained in the vascular compartment by the various glomerular membranes. Those membranes also serve as charge discriminators and tend to retain negatively charged proteins in the plasma compartment as well. Proteins of low molecular weight pass the glomerular barrier with various degrees of efficiency and are later (more or less efficiently) taken up by proximal tubular cells. Indeed, the reabsorption of proteins that pass the glomerular membranes is very efficient; even slight decreases in tubular fractional reabsorption due to tissue damage increases the excretion of relatively low-molecular-weight proteins.
Electrophoretic patterns of urinary proteins can reveal glomerular damage, tubular damage, or both. Careful assessment of electrophoretic patterns can reveal selective glomerular damage (loss of glomerular polyanion) or unselective damage (glomerular hyperfiltration). Studies in experimental animals must be undertaken cautiously because in some species large variations in urinary protein excretion can lead to incorrect conclusions. For example, sex-, age-, and diet-related changes that occur in mate rats are not related to glomerular damage (Neuhaus et al., 1981). Young male rats can show "tubular" proteinuria, whereas aging rats show "glomerular" proteinuria; the proteinuria in the first instance is essentially physiologic, and that in the second is attributable to spontaneous nephropathy, which can be controlled in part by reducing dietary protein.
Tamm-Horsfall Protein
Excretion of Tamm-Horsfall protein (discussed in detail in Chapter 4) is increased after damage to the distal part of the nephron and is decreased when the renal mass is reduced.
Enzymuria
Several investigators have used enzymuria as a marker of nephrotoxicity, but Dubach et al. (1989) have indicated that none of the enzymes studied experimentally satisfies all the criteria of a nephrotoxic response. Because renal enzymes are not distributed uniformly along the nephron, it might be possible to localize renal damage within the nephron on the basis of the pattern of enzymuria. The site selectivity of single enzymes is questionable. Other factors that complicate the use of enzymuria as a marker of renal dysfunction have been suggested. For example, early renal changes induced by chemicals might be less selective, in which case the predictive value of enzyme markers would be compromised. Many procedures for analyzing urinary enzymes are poor and, rather than pinpointing specific nephron sites, might give rise to nonspecific patterns that are difficult to interpret. Most urinary enzymes are stable only over a narrow pH range, and their activity in urine could be affected by inhibitors, some of which can alter urinary pH (Price, 1982). Thus, enzymuria studies in experimental animals must be carried out under very carefully controlled experimental conditions. Contamination of urine with food or microorganisms must be minimized, and urine must be collected in vessels that are then stored in ice (Berlyne, 1984). (See Chapter 4)
Monoclonal Antibodies
Other potential markers of renal damage are immunoreactive tissue constituents that are released into urine because of increase in cellular turnover or cell death. Those constituents can be detected immunochemically, and monoclonal antibodies have been produced
against both rat (Tokoff-Rubin, 1986) and human (Mutti, 1989; Mutti et al., 1985) brush-border antigens. The selectivity of these markers for identifying specific nephron segments is still debated, although experimental results suggest that they can be useful. The earlier studies suggested that the BB-50 brush-border antigen was also localized in peritubular capillaries; Mutti et al. (1988) identified a monoclonal antibody that reacted with an antigen that was peculiar to the brush border—the so-called brush-border antigen. Monoclonal antibodies to the S3 segment of the nephron, where alkaline phosphatase is, also have been produced (Verpooten et al., 1989). They might prove useful as markers of the effects of chemicals, such as mercury, that act selectively on the straight part of the proximal tubule.
Bladder Toxicity
Xenobiotics
On the basis of results of long-term carcinogenicity studies of 358 xenobiotics (Barrett and Huff, 1991), the bladder is among the 10 most prevalent sites of cancer development in rodents. According to histopathologic findings after chronic exposure, 16 xenobiotics (4%) caused bladder tumors in at least one sex of either rats or mice. Histologic evaluation of rat bladders after various doses and durations of exposure to 4-butyl-(4-hydroxybutylnitrosamine) (BBN) and N-(4-[5-nitrofuryl]-2-thiazolyl) formamide (FANFT) demonstrated a series of changes in structure that are good models of the changes noted in human bladder cancer. A considerable amount of information based on those models and the results obtained with 2-acetylaminofluorene (2-AAF) is available on the potential of xenobiotics to influence the development of bladder cancer (Ito et al., 1989; Soloway and Hardeman, 1990; Staffa and Mehlman, 1980).
It should be noted, however, that there is not complete concordance across species, even for the genotoxic bladder carcinogens. That fact, by itself, makes extrapolation to humans difficult. The text that follows should be viewed in this light. For example, BBN produces tumors at a lower rate in mice than in rats, and at a lower rate in hamsters than in mice, and guinea pigs do not develop bladder tumors after exposure to BBN (Hirose et al., 1976). Furthermore, in the long-term carcinogenicity studies, 10 chemicals produced bladder tumors in rats, but only six were associated with tumors in mice (Barrett and Huff, 1991). It is also noteworthy that the first xenobiotic (or group of xenobiotics) shown to induce bladder cancer in humans, the aromatic amines (see Rubber et al., 1985, for discussion), readily induced bladder cancer in dogs but did not induce bladder tumors in rats until massive doses were given chronically by oral gavage (Hicks et al., 1982). In fact, it has been stated that the failure of the rat bladder to respond to the aromatic amines was one factor that led to the use of the maximum tol-
erated dose (MTD) in rodent bioassays (Wiseburger, 1992).
The capacity of chemicals and of various physical injuries to induce hyperplasia in rat bladders has been measured by histopathologic means (Ito et al., 1989) and on the basis of increased DNA replication and increased fresh and dry organ weight (Anderson, 1991; Cohen and Ellwein, 1991; Ito et al., 1989). When those techniques have been applied after the same treatments, they yielded similar results and appeared to be equally valid ways to measure cell division in the bladder. The use of the techniques in humans is questionable because they depend on using the isolated organ.
It has been reported that one can ascertain the potential of a chemical to induce bladder damage by determining its ability to enhance concanavalin A's agglutination of bladder cells from rats treated with the chemical (Kakizoe et al., 1981). That technique is reportedly capable of distinguishing between complete carcinogens and tumor promoters (R.L. Anderson, Procter and Gamble, unpublished material, 1987). Complete carcinogens increase cell agglutination directly. Tumor promoters do not produce the response when given alone, but they can sustain the response in animals that were first exposed to a known initiator carcinogen.
Extracellular Calcium
Studies with nitrilotriacetate (NTA), a nongenotoxic compound that causes bladder tumors in rats but not in mice, have demonstrated that high doses of this metal-chelating chemical cause an increase in urinary calcium and a coincident decrease in bladder-tissue calcium in rats (Anderson and Alden, 1989). That state is accompanied by the presence of crystalline calcium-sodium NTA in collected urine. Uncomplexed NTA in urine extracts calcium from the urothelial extracellular pool more rapidly than it can be replenished from the circulation. The removal of extracellular calcium reduces cell-cell contact in the urothelium and results in increased cell loss and increased urothelial replication. If this process is continued chronically, it can result in urothelial tumors (at a low rate). Only one other rat-bladder carcinogen, terephthalic acid, which forms calcium terephthalate crystals in urine, is known to cause bladder tumors by the mechanism demonstrated for NTA (Chin et al., 1981). Attempts to demonstrate a broader base for this mechanism with other treatments known to induce bladder tumors have not been successful (Anderson, 1991).
Zinc
There are few data to support the notion that tissue concentration of zinc is related to the development of bladder cancer. What data are available are the result of a single study with short-term exposure (four weeks) to BBN that demonstrated that the treatment caused increased bladder-tissue weight and tissue zinc without a change in any other mineral (Anderson et al., 1986a). It should
be noted, however, that exposure to a carcinogenic dose of sodium saccharin increased the tissue concentration not only of zinc, but also of several other minerals (Anderson, 1985; Schoenig and Anderson, 1985). Also, physical injury to the bladder of sufficient magnitude to induce tumors failed to show that urothelial proliferation is always accompanied by increased tissue zinc (Anderson, 1991).
Monovalent-Cation Salts and Urinary pH
Mineral analyses of bladders from rats fed various doses of sodium saccharin (NaSacc) showed that NaSacc caused a dose-dependent increase in the concentration of several minerals in the bladders of male but not female rats; note also that NaSacc is far more carcinogenic to the male than the female rat (Schoenig and Anderson, 1985). It has been proposed that tumor-promoting effects of several sodium salts in the bladder might result from high urinary sodium, which increases sodium transport into urothelial cells and induces increased cell replication (Ito and Fukushima, 1989).
Studies with NaSacc and several other monovalent-cation salts that have been shown to induce bladder tumors in male rats have led to the hypothesis that increased urinary pH and monovalent-cation concentration can cause bladder epithelial hyperplasia and even induce bladder tumors (Cohen et al., 1991b; Ito and Fukushima, 1989). Although potassium salts have the same response, divalent-cation salts do not induce the effects, at least in short-term experiments (Anderson et al., 1988; Cohen et al., 1991b; Hasegawa and Cohen, 1986; Ito and Fukushima, 1989).
NaSacc might act as a tumor promoter when ingested chronically by male rates after initiation with FANFT (Cohen et al., 1991b). The effect has not been noted in other species. For example, after initiation with 2-AAF, mice do not develop an increased number of bladder tumors in response to chronic ingestion of doses of NaSacc that act as potent promoters in rats (Fredrick et al., 1989).
Overall, the experimental evidence suggests that the male rat bladder is particularly sensitive to tumor development when heavily exposed to monovalentcation salts of a variety of acids. The effect has been noted especially in studies in which the rats were exposed to an initiating dose of a known carcinogen before exposure to the salt being studied. The results show good concordance with the pathologic sequence noted in human cancer development, but the mechanism by which the tumors are induced is controversial.
Mechanical Distention
In contrast with the idea that monovalent-salt-induced bladder toxicity results from high urinary monovalent-cation excretion and increased urinary pH, an alternative mechanism has been suggested to ac-
count for the bladder tumorigenicity associated with, for example, NaSacc. High NaSacc ingestion and the loss of urine concentrating capacity that accompanies the reduction in functional renal tissue in aging male rats result in an increase in urinary volume. The increase in urinary volume leads to an increased demand for tissue growth. The relationship between increased urinary volume and demand for tissue growth suggests that chronic diuresis can be causally related to bladder carcinogenicity (Anderson, 1991). Acute, sub-chronic, and chronic studies demonstrate that, at least in male rats, the bladder responds to increases in urinary volume with tissue proliferation and urothelial hyperplasia and even with bladder tumorigenesis if the treatment results in continued diuresis. The bladder-distention model is further supported by studies showing that bladder distention caused by an injection of water into the bladder or by intraperitoneal injections of water or saline causes a wave of increased cell replication in the urothelium that is associated with diuresis (Herbertson et al., 1982; Koo et al., 1979; Martin, 1962).
However, the failure of furosemide-induced diuresis to promote bladder tumors in rats after initiation with BBN and the lack of data showing increased bladder tumors in the spontaneously diabetic rat make the diuresis model tenuous (Shibata et al., 1989). It has been reported that patients with bladder tumors have reduced fluid intake, less-frequent micturition, and increased urine concentration—findings that are inconsistent with the diuresis model (Braver et al., 1987). Results of epidemiologic studies on this point are conflicting.
Growth Factors
Urine is an important component in bladder tumorigenesis, at least in experimental animals. Yara et al. (1989) fractionated urine and showed that the promoting activity is attributable to epidermal growth factor.
Treatment
The development of highly predictive models of bladder carcinogenesis in rodents has been useful in evaluating the potential for treatments for bladder tumors. With the FANFT mouse model, good concordance with clinical experience has been observed for both systemic and intravesical treatments for bladder cancer (Soloway and Hardeman, 1990). It should be noted, however, that at least two agents used clinically—adriamycin and mitomycin C—demonstrate tumor-promoting activity when instilled intravesically in rats after initiation with BBN (Ito and Fukushima, 1989).
Renal-Tumor Formation
The mechanism of renal-tumor formation associated with chronic high-dose ingestion of the nongenotoxic
agent NTA provides an approach to ascertaining the relevance of animal results to human risk associated with exposure (Anderson and Alden, 1989). NTA is a metal-chelating agent that has been used in laundry detergents in Canada for many years with continuous monitoring of its concentration in drinking water, which affords an opportunity to define human exposure (Anderson et al., 1985). The mean drinking-water concentration of NTA after several years of use was 2.5 μg/L. Thus, a 70-kg person consuming 2L of the average water each day would ingest ≈0.07 μg of NTA per kilogram of body weight per day (3.7 × 10-4 μmol/kg per day).
Results of several chronic-ingestion studies of NTA (Anderson et al., 1985) showed that ingestion rates greater than 1.35 μmole/kg per day were associated with renal proximal tubular-cell carcinomas. Extensive studies comparing biochemical and histologic changes induced by established carcinogenic doses of NTA demonstrated that carcinogenesis depended on the chronic delivery of a dose of NTA that induces cellular proliferation in the proximal tubules (Anderson and Alden, 1989). The biochemical studies demonstrated that the induction of renal tubular-cell proliferation depended on increased delivery to and accumulation of zinc in the renal tubular cells. Zinc delivery was accomplished through the formation of a zinc-NTA complex in the blood, which was cleared by the kidney, which reabsorbed and accumulated the zinc but not the NTA. The formation of sufficient zinc-NTA in the blood to increase zinc delivery to the renal tubules showed a distinct threshold with respect to blood NTA concentration. The threshold for sufficient zinc delivery coincided with the threshold for induction of histologic changes in the renal tubules and with a no-effect concentration in chronic-exposure studies. In the case of NTA, the threshold dose of NTA (≈q00 μmole/kg per day) is more than 107 times the mean human exposure from drinking water in Canada after many years of use of NTA in laundry detergents.
TECHNIQUES USED IN RISK ASSESSMENT
Animal Studies
Renal function and nephrotoxicity in experimental animals can be studied at various levels of tissue structure and organization (Table 5-1), whereas in humans, studies of renal function are likely to rely on noninvasive studies whenever possible. Under certain circumstances, tissues obtained by biopsy may be available for analysis, but this is not likely to be a routine part of most studies. The first stage of any investigation on the nephrotoxicity of a xenobiotic should be in vivo studies. In the absence of knowledge about the potential toxicity of a chemical or the potential risk to human injury associated with a chemical, the first goals of studies in experimental animals are to determine whether toxicity occurs, and if so, whether it is organ-specific. If target-
TABLE 5-1 Experimental Model Systems for Study of Renal Metabolism, Renal Function, and Nephrotoxicity
|
Model System |
Advantages |
Limitations |
Common Uses and Relation to Whole Animal |
|
Whole animal |
1. Intact organ structure 2. No possibility of in vitro artifacts 3. Ability to study interorgan metabolism 4. Study renal function |
1. Minimal control over incubation conditions 2. Expensive 3. Interanimal variability 4. Inability to distinguish intrarenal and extrarenal effects 5. Precise knowledge of exposure condition and site of action unavailable |
Target-organ specificity studies; interorgan metabolism studies |
|
Isolated perfused kidney |
1. Structurally identical with in vivo kidney 2. Study renal function 3. Can distinguish intrarenal and extrarenal effects 4. Somewhat better-defined incubation conditions |
1. Short-term use (up to 2 h) 2. Expensive 3. Interanimal variability 4. Incomplete control over conditions |
Ideal for studies of renal function on whole-tissue level without extrarenal factors |
|
Kidney slices |
1. Maintenance of renal-tissue structure 2. Ease of preparation 3. Improved definition of incubation conditions |
1. Short-term use (up to 2 h) 2. Limited access to brushborder membrane 3. Poor cellular oxygenation 4. Presence of many cell types |
Can be useful for studies of renal transport and metabolism; less potential for isolation artifacts vs. other in vitro systems because more of renal structure is maintained |
|
Model System |
Advantages |
Limitations |
Common Uses and Relation to Whole Animal |
|
Isolated single nephron |
1. Intact tubular structure 2. Precise definition of incubation condition 3. Ability to separate nephron cell types 4. High degree of cell-type homogencity 5. Limited damage during isolation |
1. Short-term use (up to 2 h) 2. Complicated microdissection techniques required 3. Low yield |
Most useful for electrophysiologic studies because of maintenance of cell-to-cell contacts within a specific nephron region |
|
Isolated tubules |
1. Intact tubular structure 2. Relative ease of preparation 3. Precise definition of incubation conditions 4. Perform several manipulations with paired controls |
1. Short-term use (up to 4 h) 2. Often limited access to brush-border membrane 3. Possible damage during isolation if enzymatic or chemical method used |
Metabolism, toxicity, and transport easily measured under controlled conditions; good model in vivo if functional integrity (e.g., open lumen) is maintained; short-term model |
|
Freshly isolated renal cells |
1. Bidirectional exposure 2. Relative ease of preparation 3. Precise definition of incubation conditions 4. Ability to separate specific nephron cell types 5. Perform several manipulations with paired controls |
1. Short-term use (up to 4 h) 2. Possible damage during isolation if enzymatic or chemical method used 3. Loss of polarization |
Metabolism, toxicity, and transport easily measured under controlled conditions; good model in vivo if cell polarity is not relevant; short-term model |
|
Model System |
Advantages |
Limitations |
Common Uses and Relation to Whole Animal |
|
Renal-cell culture lines |
1. Long-term use (weeks to months) 2. Precise definition of incubation conditions 3. Easily obtained and subcultured |
1. Dedifferentiation 2. Often ill-defined origin |
Can be useful model if phenotype of interest is maintained; long-term (>>24 h) processes can be studied in very reproducible system |
|
Primary renal-cell cultures |
1. Closely related to in vivo cells 2. Precise definition of incubation conditions 3. Longer-term viability (up to 2 weeks) 4. Ability to separate specific nephron cell types 5. Maintenance of polarity |
1. Difficult to maintain 2. Dedifferentiation can occur 3. Limited lifetime |
Potentially best model if phenotype is maintained; long-term (several hours to several days) processes can be studied |
organ specificity is found, more detailed studies, both in vivo and in vitro, can then be performed to elucidate mechanisms of toxicity and potential protective strategies.
Noninvasive or nondestructive studies of organ function can be used to demonstrate target-organ specificity. These studies generally involve measurement of enzyme activities in plasma or serum or measurement of urinary, plasma, or serum concentrations of metabolites or waste products that are secreted or excreted by the tissue of interest. Such measurements can be used to assess organ function without removal of tissues. Histopathologic techniques can then be applied to determine target-organ specificity of injury and to determine the site within the tissue at which injury occurs. In vivo measurements are important as first steps in assessment of xenobiotic-induced injury and as adjuncts to in vitro studies to validate in vitro techniques and to permit correlations between in vitro and in vivo measurements. Although in vivo techniques allow the study of organ function in intact animals, their use for routine screening of chemical toxicity or for more-detailed mechanistic studies is severely limited (Table 5-1), and detailed mechanistic information might not be available from them. The development of in vitro methods permits extended maintenance of cellular function outside an organism so that other types of measurements can be applied for the assessment of toxicity.
Whole-Kidney Preparations
A first step toward development of methods to study nephrotoxicity permits some control over exposure conditions. That can be accomplished by the use of an isolated perfused kidney. The advantages of this method are that intact tubular structure is maintained and that some control of exposure conditions is possible. Substantial advances in development of perfusion buffers that closely simulate renal arterial plasma have been made in recent years (Maack, 1986); these advances have enabled investigators to maintain tissue viability for longer periods. Examples of measurements that are possible with the isolated perfused kidney are those of renal clearance and metabolism. Release of renal enzymes, which is commonly measured in vivo to assess nephrotoxicity, can be conveniently determined without complications of extrarenal sequestration or metabolism. The principal advantage of the method is that the maintenance of intact tubular structure requires that limits be placed on incubation and exposure conditions; this limits the information that can be obtained. As with in vivo investigations, a single animal serves as a single experimental manipulation, so studies with the isolated perfused kidney require a large number of animals and are expensive. In addition, the target site in the kidney for a given toxic or pathologic condition might not be easily discernible.
Micropuncture techniques can be
used to examine responses in specific regions of the nephron (Burg and Knepper, 1986; Quamme and Dirks, 1986). This method has been used extensively in the study of nephrotoxicity. Information derived from these studies has been fundamental in the understanding of the consequences of both acute and chronic injury to the kidneys. It has the great advantage of allowing measurements to be made with the kidney in its proper environment but suffers from some of the limitations of time and expense as occurs with the isolated perfused kidney (Table 5-1).
In Vitro Studies
In vitro models for studies of renal function generally use physical means or digestive enzymes to separate cellular material from specific regions of the kidney. To obtain detailed information on biochemical mechanisms of action of toxic chemicals or on pathologic states, it is necessary to use living material from specific nephron regions. Physiologic and biochemical differences among mammals in the various kidney regions and nephron cell populations have important toxicologic implications and must therefore be considered in risk assessment of potentially nephrotoxic chemicals. The ability to localize nephrotoxic responses to specific cell populations is an important step in understanding biochemical mechanisms of action. Tissue slices, isolated nephron segments, isolated tubule fragments, and isolated cell suspensions can be obtained from the renal cortex or medulla. The degree to which nephron heterogeneity is taken into account determines the type of model that is required for any investigation.
Various experimental approaches can be used to separate cells from different nephron regions (Table 5-2) (Schlondorff, 1986). Because each cell population has distinct structural characteristics, visual separation with the aid of a dissecting microscope is possible. This allows preparation of nearly homogeneous material (Wilson et al., 1987). The low yield of material and the length of time required for separation this way generally make biochemical studies difficult. Biochemical or physiochemical properties of individual cell populations have been used to achieve purification and separation. Differences in cell-surface properties are useful in this regard. For example, the high glycoprotein content of the proximal tubular brush-border membrane gives proximal tubular cells a high density of negative charge, so electrophoretic techniques have been used to purify proximal tubules and isolated proximal tubular cells (Kreisberg et al., 1977a). Although high purity can often be obtained with electrophoresis, the technique requires expensive equipment, and the yield of material is often low.
A common method to separate a mixture of cell populations into its component cell types is density-gradient centrifugation. This method is relatively easy to use and rapid, so long pro-
TABLE 5-2 Procedures for Separation of Cells or Tubules from Specific Nephron Regions
|
Separation Method |
Advantages |
Limitations |
|
Microdissection |
1. Extremely high purity |
1. Very low yield 2. Difficult to perform 3. Time consuming |
|
Microdissection and immunologic reactivity |
1. Extremely high purity 2. Cell type specificity |
1. Very low yield 2. Difficult to perform 3. Time-consuming |
|
Electrophoresis |
1. High purity |
1. Very low yield 2. Requires special equipment 3. Time-consuming |
|
Density-gradient centrifugation (e.g., sucrose, Ficoll, Percoll) |
1. Easy to perform 2. High yield 3. Relatively rapid |
1. Enrichment rather than absolute purification |
cedures with complicated equipment or tedious steps are not necessary, and cell viability is generally not sacrificed. One limitation of separation methods based on cell density is that enrichment, rather than absolute purification, is obtained because cell density is not a discrete property; rather, a given cell population generally contains cells with a range of densities.
Either tubule fragments or isolated cells can be used as starting material from which to obtain enriched cell populations. Both have several advantages, including the ability to pair the materials with control samples from a single animal to perform several manipulations and replications and the ability to specify, and control incubation conditions. In comparing tubule fragments with isolated cells and in choosing a model system, several points should be considered (Table 5-1) (Lash, 1989). First, the lumens in many isolated-tubule preparations do not remain open during incubation; this can be problematic if transport or enzymatic processes on the brush-border membrane are of interest, and it restricts access of nutrients or toxicants to the intracellular milieu and thereby alters cellular response to exposure. Second, when tubules are fragmented during the isolation procedure—whether by mechanical, chemical, or enzymatic means—tubule length might become nonuniform and thereby introduce additional variability into the model. Isolated cell preparations, in contrast, generally yield more uniform materials; preparation methods are adjusted so that predominantly single cells in suspension, rather than multicellular aggregates or tubules, are found in the final material. Third, a given tubule frag-
ment might contain cellular material derived from more than one nephron region, depending on how connective tissue is digested during the initial isolation procedure; this limitation could substantially diminish the effectiveness of later density-gradient centrifugation steps that are designed to yield nearly homogeneous material. With suspensions of isolated cells, in contrast, single cells are the starting material for any further purification steps.
Isolated proximal tubule fragments have been used extensively to characterize the basic transport and energetic properties of this nephron segment (Gullans et al., 1984a,b; Brazy et al., 1984). These studies demonstrated the role of plasma membrane transport and other energy-requiring processes in determining the rates of cellular oxygen consumption. Because the in vitro model contains segments of intact epithelium, one can study such processes as transepithelial transport and membrane potential, provided that lumens remain open during the course of experiments. Additional studies with proximal tubule fragments that are of more toxicologic interest have focused on metabolic and mitochondrial inhibitors (Aleo and Schnellmann, 1992a; Dickman and Mandel, 1990; Gullans et al., 1982; Weinberg et al., 1990) and on anoxia (Almeida et al., 1992; Jacobs et al., 1991; Portila et al., 1992). Additional studies on cytotoxic effects of specific nephrotoxicants, such as cisplatin (Brady et al., 1990) and mercuric chloride (Zalups et al., 1993) have also been performed.
Ficoll and Percoll density-gradient centrifugation procedures have been used to separate proximal and distal tubular epithelial cells from rat renal cortex (Cojocel et al., 1983; Gesek et al., 1987; Scholer and Edelman, 1979; Vinay et al., 1981). Ficoll density-gradient centrifugation has been used to enrich proximal tubular cells from rat renal cortex (Kreisberg et al., 1977b), and Percoll centrifugation has been used to separate isolated cells from rat proximal and distal tubules (Lash and Tokarz, 1989). Both Ficoll and Percoll form density gradients spontaneously when a centrifugal field is applied. Ficoll is a nonionic, synthetic polymer of sucrose, is supplied as a powder, and must be placed in solution to be used. Percoll consists of colloidal, polyvinyl-pyrolidine-coated silica beads and is supplied as a liquid. Because of its chemical properties and because it is already in liquid form, procedures using Percoll are generally easier to perform and have more uniform density gradients than those using Ficoll (Pertoft and Laurent, 1982). Both methods are applicable to other regions of the nephron: Chamberlin et al. (1984) used Percoll to obtain an enriched preparation of medullary thick ascending-limb tubules from rabbit kidney, and Eveloff et al. (1980) used Ficoll to obtain an enriched preparation of single medullary thick ascending-limb cells from rabbit kidney as well.
Two types of approaches can be used with Percoll: The use of discontinuous and continuous gradients. Although the discontinuous density gradient might
yield a more complete separation of discrete cell populations, continuous gradients are much easier to prepare and are more reproducible. The Percoll method with a continuous, spontaneously generated density gradient is relatively simple to perform and yields highly enriched preparations of single proximal tubular cells (over 97% homogeneity) and distal tubular cells (over 88% homogeneity) (Lash, 1992, 1993; Lash and Tokarz, 1989).
Uses and Limitations of Freshly Isolated Renal Cells
Freshly isolated renal cells, prepared by collagenase perfusion and then separated into discrete cell populations by Percoll density-gradient centrifugation, have proved to constitute an extremely useful in vitro system for biochemical toxicology studies (Lash, 1990, 1992, 1993; Lash and Tokarz, 1989, 1990; Lash and Woods, 1991; Lash and Zalups, 1992; Lash et al., 1993). Validation of these cells as in vitro models that reflect accurately the biochemical, physiologic, toxicologic and pathologic events that occur in the proximal tubular and distal tubular segments in vivo is by measurements of several biochemical properties, respiratory characteristics, and drug-metabolizing enzymes. Examples of biochemical properties that can be measured include facilitated diffusion and active transport processes for organic ions, glutathione (GSH) metabolism, adenine and pyridine nucleotide status, and intracellular calcium distribution. Measurement of cellular respiration assesses the ability of the cells to integrate metabolic pathways and to regulate energy metabolism.
Freshly isolated renal cells are excellent models for studying acute mechanisms of chemical injury. The cells are particularly useful for studying the enzymology of xenobiotic bioactivation. For example, cysteine S-conjugates of various halogenated alkanes and alkenes are potent and specific nephrotoxicants that target the proximal tubular region of the nephron. Recent work in freshly isolated renal cortical cells demonstrated the central role of the cysteine conjugate beta-lyase in the generation of reactive thiol-containing metabolites that are ultimately responsible for the nephrotoxicity observed in vivo (Jones et al., 1986; Lash and Anders, 1986; Lash et al., 1986a). That similar enzymatic activation pathways occur in human renal tissue (Lash et al., 1990) suggests that similar mechanisms of nephrotoxicity operate. Studies in isolated rat renal cells can thereby provide information on human risk of nephrotoxicity associated with halogenated hydrocarbons that are metabolized by this pathway.
A major advantage of the Percoll density-gradient centrifugation method is that it allows study of biochemical processes and responses in specific regions of the nephron. It allows investigation of the hypothesis that each nephron cell population exhibits its own characteristic susceptibility to chemical injury. To understand how the various differences in each cell populations contribute to the toxic response, two strategies can
be used. One obvious strategy is to study the mechanism of action of a known nephrotoxic chemical that has a specific target cell in a purified preparation of that target cell. Such studies, however, do not address directly the issue of nephron heterogeneity and the associated toxicologic implications. A second strategy that does address that issue directly is to expose the different cell populations to cytotoxic chemicals or pathologic conditions that will produce injury by a specific chemical or biochemical mechanism but that do not themselves have specific renal target cells (Lash, 1990; Lash and Tokarz, 1990; Lash and Woods, 1991; Lash et al., 1993). The strategy allows analysis of the inherent susceptibility of isolated cells to such processes as oxidative stress, covalent-adduct formation, cellular-energy depletion, and oxygen deprivation. The contribution of such processes as GSH metabolism, mitochondrial function, drug metabolism, and active transport to nephrotoxicity can then be studied, and the approach can also help to increase our basic understanding of why chemicals are toxic and how cells respond to xenobiotics.
The period over which lactate dehydrogenase release can be a reliable measure of cellular viability in freshly isolated cells is generally limited to 4 hours. Beyond that period, isolated cells generally do not maintain adequate functional integrity to serve as valid controls for toxicologic studies. Similarly, many of the other measures that are used to assess cellular function can be valid measures of cellular integrity for only short periods. For most of the types of responses that occur as a consequence of acute exposure to chemical toxicants, the survival time is adequate to observe markers of exposure or to observe changes that can provide information about risk and biochemical mechanisms of action—information that is most useful if clinically observed toxicity is to be treated or prevented.
One potential limitation in the use of single cells in suspension that must be considered is the physical loss of membrane polarity that exists in these cells. Because the physical separation of the luminal and basal-lateral membranes by the epithelial tight junctions no longer exists, one cannot measure transepithelial fluxes or other types of processes that depend on an intact epithelium, as can be done with the isolated perfused kidney or, provided that lumens remain open, with isolated tubule fragments. However, for many toxicologic studies, the critical issue is not the physical separation of the membrane surfaces but the retention of enzymatic and transport activities on these membranes when the individual epithelial cells are placed into suspension. Ample evidence from studies with isolated proximal tubular cells from rats (Hagen et al., 1988; Jones et al., 1979; Lash and Anders, 1989; Lash and Jones, 1985; Lash and Tokarz, 1989, 1990) support the conclusion that these cells are metabolically competent and contain transport activities and other membrane-associated enzymatic activities at levels that are comparable with those in both in vivo and various in vitro model sys-
tems. One can often functionally distinguish processes that occur on the two membrane surfaces by the use of selective inhibitors. For example, transport of glucose across brush-border and basal-lateral plasma membranes can be readily distinguished by use of either phlorizin or phloretin to inhibit brush-border or basal-lateral transport, respectively. In the case of transport of glutathione across renal proximal tubular plasma membranes, the two transport processes can be readily distinguished from each other by virtue of their distinct kinetic and energetic properties and differential substrate specificity (Lash et al., 1988). That is not to minimize, however, the functional importance of plasma-membrane polarization in an intact epithelium. Many processes, as stated above, require retention of the physiologic cell-cell contacts to allow measurement to occur. Depending on the type of biochemical or physiologic process being examined, either single cells in suspension or tubule fragments might be the more appropriate in vitro model system.
Renal-Cell Cultures as Models of Chronic Injury and Carcinogenesis
Limitation of maintenance of viability of isolated tubules and isolated renal cells to, at most, 4 hours restricts investigations to processes that occur on a scale of minutes to hours and necessitates the development of other types of model systems to study processes that occur on a scale of hours to days. For example, mutagenesis, induction of renal tumors, and alterations in expression of some genes are common responses to many chemical toxicants, including carcinogens. Those processes change, for the most part, on a scale of hours to days; even longer-term models might be necessary to obtain a complete picture of the regulation of the processes.
A biologic model system needs to be developed, therefore, to meet two critical requirements. The first requirement is that the model consist of material whose renal-cell type of origin can be unambiguously defined; the second is that the model be maintained in a viable state for at least about 7–10 days. An excellent model system is primary cell culture that uses cellular material previously enriched in a particular renal-cell population. Although several renal-cell lines are available for such long-term in vitro studies (Table 5-3), their use raises many questions. Normal in vivo characteristics of the tissue must be adequately reflected and expressed in the culture models if they are to be useful indicators of exposure or useful tools for studying mechanisms of chronic toxicity; otherwise, no reliable conclusions concerning cell-type-specific responses or in vivo relevance can be drawn. A major problem with the existing renal-cell lines and with immortalized cells in general is that they dedifferentiate in culture. Therefore, cultured cells can express some properties of the original tissue but often do not express many other characteristics that are essential for the model to reflect in vivo cellular
TABLE 5-3 Some Immortalized Renal-Cell Culture Lines
|
Presumed Cell Population of Origin |
Cell Line |
Species |
|
Glomerulus |
SGE1 |
Wistar rat |
|
Proximal tubule |
LLC-PK1 |
Hampshire pig |
|
|
NRK52E |
Rat |
|
|
OK |
American opossum |
|
|
JTC-12 |
Cynomolgus monkey |
|
|
RK-L |
Sprague-Dawley rat |
|
Medullary thick ascending limb |
GRB-MAL |
Rabbit |
|
|
M-m TAL-lc |
Mouse |
|
Distal tubule and collecting duct |
A6 |
African clawed toad |
|
|
MDCK |
Cocker spaniel |
|
Unclear |
BSC-1 |
Monkey |
function accurately. Another major deficiency is that cell lines from several nephron segments are not available from a single species but are derived from several species. Ambiguity can arise in studies that use these various cells. Interspecies differences in biochemical properties and susceptibilities to chemical injury, however minor, can make it difficult to obtain a clear understanding of cell-type-specific mechanisms of injury with established cell lines.
Primary cell culture is by no means simple or straightforward. Many pitfalls are inherent in its performance, so many investigators use cell-culture lines to mimic cellular function in a given cell type or tissue. Apart from the problems and limitations, primary culture of specific renal epithelial-cell populations is an ideal approach for biochemical toxicology studies involving chronic or long-term exposures or expression of genetic or developmental processes. The three principal challenges to successful primary culture of renal epithelial cells are to obtain starting material of a high-enough purity derived from a specific nephron-cell population, to maintain differentiated function during the period of culture, and to prevent fibroblast overgrowth.
To validate a primary epithelial cell-culture procedure, several criteria must be satisfied. First, the cells must express key markers identified with the cell population of origin, thereby indicating expression of differentiated function; and expression of differentiated function must be reasonably well maintained during the course of culture. Second, the primary cultures must maintain expression of epithelial markers and must not
be overtaken in the culture by fibroblasts. Third, the cells must grow logarithmically long enough (at least about 5-7 days). Taub and colleagues (Aleo et al., 1989; Chung et al., 1982; Taub et al., 1989) have pioneered primary cell culture of rabbit proximal tubules, and several groups (Boogaard et al., 1990; Elliget and Trump, 1991; Lash, 1994; Smith et al., 1986; Tokarz and Lash, 1993) have recently developed procedures for primary culture of rat proximal-tubular cells for use in toxicity studies. In using cell cultures to study mechanisms of chemically induced toxicity, it is important to consider that placement of cells in culture can alter their inherent susceptibility to chemical toxicants. Hence, it is critical to validate in vitro cell-culture models so that their general susceptibility to chemicals or pathologic conditions, such as anoxia, are known. With that foundation, the models can be used as investigative tools.
One of the key markers of differentiated function in renal proximal tubular cells is the maintenance of high activity of mitochondrial oxidative phosphorylation and low glycolytic activity. Two studies (Aleo and Schnellmann, 1992b; Dickman and Mandel, 1989) reported that diminished oxygenation of stationary cell cultures produces the often-documented shift of cellular energy metabolism to glycolytic from mitochondrial ATP generation. That is a critical point for maintenance of a long-term cell-culture model. Although use of a shaking system for cell cultures is one means of improving oxygenation and hence maintenance of cellular energetics (Aleo and Schnellmann, 1992b; Dickman and Mandel, 1989), other investigators have used changes in glucose content of growth media (Blais et al., 1992) or a more complete addition of hormonal supplements to growth media to improve cellular energetics (Lash 1994; Tokarz and Lash, 1993).
Although most of the studies with primary renal-cell culture have used animal tissue as starting material, recent advances have been made in the use of human renal tissue as starting material for primary culture of proximal tubular cells (Chen et al., 1990b; Detrisac et al., 1984; Trifillis et al., 1985). Such methods will enable direct comparison between animal and human in vitro models.
Because primary cell cultures can be maintained for up to 10 days, two types of experimental designs can be used in toxicologic investigations: acute and chronic (Table 5-4). The cell-culture model could be particularly useful for assessing carcinogenic potency of chemical toxicants, and this would make it an excellent tool for risk assessment.
In Vitro Models of Nonrenal Urinary Epithelia
In vitro models for study of toxic and pathologic processes in regions of the urinary tract besides the kidneys have recently been developed. Because most of these studies have focused on growth, development, and neoplastic transformation, cell cultures derived from either
TABLE 5-4 Experimental Protocols Used with Primary Cell Cultures for Toxicologic Investigations
|
Characteristic |
Acute Model |
Chronic Model |
|
Dose |
Low-to-high |
Single or multiple low |
|
Exposure/Time |
< 24 h |
1–10 days |
|
Examples of processes measured |
Intermediary metabolism Enzyme activities Respiration Active transport Metabolite concentrations (e.g., ATP, glutathione) Lactate dehydrogenase release or trypan blue up-take |
Gene expression Protein, DNA, and RNA synthesis Mutagenesis DNA repair Processes measured in acute model |
bladder or ureter epithelium, rather than freshly isolated cells, have been used.
Reznikoff and colleagues (1983) cultured normal human uroepithelial cells from tissue explants consisting of transitional epithelial cells from ureter. In addition to fetal bovine serum (at 7%, by volume) hormone and growth factor supplements were included to optimize growth and differentiation. Culture of uroepithelial. cells from experimental animals actually lagged behind culture of cells from humans. Johnson et al. (1985) developed a successful primary culture and later a serial cultivation process for bladder epithelial cells from normal rats. Fetal bovine serum and hormonal supplements were used for the primary culture, but completely defined medium (i.e., with no serum) was used for serial cultures derived from the primary culture. Johnson et al. (1985) noted that optimal culture conditions differ between rat and human uroepithelial cells although structure and biochemical and physiologic functions are very similar.
Acute cytotoxicity of bladder carcinogens—such as biphenyls, nitrofurans, and 3-methylcholanthrene—has been examined in cultured normal human uroepithelial cells (Reznikoff et al., 1986). Reduction in cell number during culture was used as the index of toxicity. The prevalence of urinary carcinogenesis has stimulated interest in use of the uroepithelial-cell culture models for study of growth regulation and neoplastic transformation. Most published studies in which these cell cultures are used have focused on processes related to tumorigenesis and growth regulation. The studies have included investigation of effects of polyamine-synthesis inhibition on growth (Messing et al., 1988), effects of bacterial endotoxicants on cell survival and growth (Wille et al., 1992), role of activation of the EJ/ras oncogene in neoplastic transformation (Pratt et
al., 1992), characterization of chromosomal deletions induced by bladder carcinogens in SV40-immortalized human uroepithelial cells (Meisner et al., 1988; Reznikoff et al., 1988; Wu et al., 1991), and characterization of second-messenger mechanisms and the carcinogenic process in SV40-immortalized human uroepithelial cells (Jacob et al., 1991).
Chopra and colleagues (Chowdhury et al., 1989) have developed cell culture methods for prostatic epithelial cells from mouse. Cells were derived from the ventral prostate of normal adult mice. Primary cultures and serial propagation were performed in serum-free, hormonally defined media to optimize differentiation. The cells grew and exhibited tissue-specific markers, including prostatic acid phosphatase activity and prostate-specific antigen.
The issue of markers of exposure and susceptibility, which is a major focus of this report, has not been addressed directly in the in vitro systems. Tissue samples, such as those used as starting material for human uroepithelial-cell cultures (Jacob et al., 1991; Meisner et al., 1988; Messing et al., 1988; Pratt et al., 1992; Reznikoff et al., 1983, 1986, 1988; Wu et al., 1991), can be readily obtained and analyzed for transformation. Cytogenetic analysis should reveal the presence of chromosomal changes that indicate exposure to a mutagenic or carcinogenic agent. For prostatic epithelial cells, examination of tissue-specific markers as described by Chowdhury et al. (1989) might be a useful means of detecting exposure of the cells to toxic agents.
IMPROVED RISK-ASSESSMENT EXTRAPOLATION
Biologic markers are the key to improving risk-assessment extrapolation. Both qualitatively and quantitatively, biologic markers are crucial in determining the best available basis for and approach to cross-species, low-dose, and route-to-route extrapolation.
Relevance of Test Systems
The major qualitative question facing a risk assessment concerns the relevance of an experimental system or animal model to the human situation. Simply stated, one can address this concern by examining the sequence of markers that occurs in humans—the sequence that is associated with exposure to a toxicant and that indicates progression to a disease state. If that sequence or a portion of it is observed in the test system, that system can be regarded as relevant for human risk estimation. Or examination of the markers, particularly markers of susceptibility, associated with disease progression in an animal model might indicate substantial similarities or dissimilarities to humans and thus help to determine relevance. In the case of renal tumors mediated by alpha2u-globulin (see Chapter 6), for example, the rat model might not be appropriate for assessing the risk of human renal tumors associated with some chemicals. Other examples of similarities and dissimilarities are discussed below. It is imperative that the relevance of proposed test sys-
tems be established to enhance the scientific basis of risk assessment.
Metabolism and Kinetics
The concept of metabolism as a means by which chemicals are biotransformed to reactive and toxic species is central to an understanding of chemically induced nephrotoxicity. Mammalian kidneys have numerous enzymatic activities that can activate a diverse array of chemicals. An important concept is that many activation and detoxification pathways are present simultaneously and therefore compete for the same substrates. The balance between competing pathways is often the primary determinant of the ultimate biologic response. Knowledge of the biochemical regulation of the pathways and of the interorgan and intrarenal distribution of the various enzymes involved is necessary for correlation of in vitro data with the in vivo situation and of data from experimental animals with human risk assessment and development of markers of human exposure.
The importance of bioactivation in determining toxicity is not a new idea, but it is critical to understand the prevalence of enzymatic activation reactions in chemical-induced toxicity (Miller and Miller, 1985). Many toxic or carcinogenic chemicals do not produce their effects directly but must be metabolized by cellular enzymes to generate reactive, electrophilic intermediates. The metabolites are responsible for the interactions with cellular components that lead to toxicity. Much of the target-organ specificity of many toxic chemicals is due to the tissue-specific distribution of activation pathways. Other important factors in determining risk are the tissue-specific patterns in types and concentrations of protective molecules, such as glutathione (GSH) and alpha-tocopherol; in activity of detoxification enzymes, such as catalase and GSH peroxidase; and in types of and activities of transport systems that provide access to intracellular sites.
When the relative roles of all those factors have been assessed in an experimental species, one must consider how differences in one or more of them will alter susceptibility to injury. This point is central to realistic assessments of human risk because differences between humans and laboratory animals and between several laboratory animal species in activation and detoxification enzymes in specific tissues have been documented. As a consequence of qualitative and quantitative species differences in drug-metabolizing enzymes and transport activities, patterns observed and conclusions reached in one species might not be applicable to another laboratory animal species or, more important, to humans.
Study of nephrotoxicants is complicated further by the pharmacokinetics and interorgan pathways involved in their disposition. Several chemicals are initially metabolized by the liver or other tissues and then, by enterohepatic and renal-hepatic pathways, reach the kidneys where they are processed further and cause nephrotoxicity. As de-
scribed in Chapter 2, the kidneys are a frequent target of chemical toxicants because of high blood flow that reaches them; the large numbers, high activities, and overlapping substrate specificities of membrane transport systems; the high basal metabolic requirements; and the presence in renal epithelial cells of several activation enzymes, some of which are peculiar to kidneys (Anders, 1980, 1989; Commandeur and Verneulen, 1990; Jones et al., 1980; Lash et al., 1988; Rush and Hook, 1986; Rush et al., 1984).
The mammalian kidney is very active in numerous pathways of drug metabolism, and renal metabolism plays a quantitatively important role in overall metabolism of a large number and variety of chemicals (Table 3-1). The kidneys are sometimes overlooked as important sites of drug metabolism for several reasons: the liver has a quantitatively large role in metabolizing a huge number of xenobiotics, the kidneys make up only 1–2% of total body weight and so are thought not to contribute importantly to metabolism, and there is pronounced heterogeneity in the distribution of enzymes in the cell populations that constitute the nephron, so detection of enzyme activities in a tissue homogenate or in a mixed population of renal cells can be difficult if the enzymes are localized to a discrete cell population. For example, activities of some isozymic forms of cytochrome P-450 in proximal tubular cells from the rat kidney are comparable with those in the rat liver (Commandeur and Verneulen, 1990; Jones et al., 1980). Some phase II enzymes, particularly those in the GSH conjugation pathway, are found at very high activities in the renal proximal tubule but are nearly absent in other regions of the nephron.
Species and strain differences need to be evaluated for each chemical or class of chemicals. It needs to be determined whether the biochemical or physiologic responses obtained in test species will be the same as those in humans. Differences in isozymic forms or the presence or absence of metabolic pathways can have effects that vary from chemical to chemical. In some cases, the differences can produce entirely different pharmacokinetics and hence different biologic effects; in other cases, the species-dependent differences might produce only quantitative differences but not yield different overall biologic effects. Differences that are noted between responses in humans and in animal species do not necessarily invalidate an animal model, nor do they imply that an appropriate animal model is unavailable for a specific toxicant. Rather, the differences highlight that we do not have much mechanistic information about many urinary toxicants and that we have not yet developed an appropriate animal model. Species differences and resulting difficulties in extrapolating to humans also highlight the need for additional research.
The mercapturic acid pathway (see Chapter 3) and associated activation and detoxification pathways illustrate many of the above principles. Although the GSH S-transferases, which catalyze the initial reaction in this pathway, are
common, the succeeding enzymatic reactions show considerable variability. The variability occurs within a given species, with respect to specificity and activity, and occurs between species for a given tissue. The tissue distribution of γ-glutamyltransferase, which is the first enzyme in the pathways of GSH S-conjugate metabolism, is the primary determinant of the pattern of interorgan metabolism (Lash et al., 1988). This initial, hydrolytic reaction determines which metabolite reaches the target organ. Whether the GSH S-conjugate is metabolized to the cysteine S-conjugate or to the N-acetylcysteine S-conjugate determines how actively the protoxicant is transported into renal cells for further metabolism to a reactive intermediate.
Because most biochemical studies on the metabolism and transport of GSH, GSH S-conjugates, and related metabolites have used rats, the view that has evolved from numerous investigations is based on the extremely high γ-glutamyltransferase activity of rat renal proximal-tubular brush-border membranes and the nearly complete absence of the enzyme in rat hepatic canalicular membranes (Lash et al., 1988). Liver and biliary epithelium of humans and other mammals, such as rabbits and guinea pigs, contain substantial γ-glutamyltransferase activity (Ballatori et al., 1988; Hinchman and Ballatori, 1990). In species that have higher activities of hepatic γ-glutamyltransferase, the liver will make a larger contribution and the kidneys a correspondingly smaller contribution to interorgan metabolism of GSH and GSH S-conjugates. For reactive and therefore toxic electrophiles that are metabolized by GSH conjugation, it is the final step in the pathway that converts the metabolite to a highly polar N-acetylcysteine conjugate that is readily excreted in urine. λ-Glutamyltransferase also shows marked species differences in velocity and substrate specificity and is absent in guinea pigs. Data on metabolite distribution and interorgan metabolism of GSH S-conjugates cannot be directly extrapolated from laboratory animals to humans without consideration of those potential differences. Therefore, although pathways studied in rats or other laboratory animals will occur in all mammals, the relative importance of each pathway will differ considerably from species to species.
Mechanism of Action
Not as much is known about the mechanisms of toxicity as is known about metabolism and kinetics. Furthermore, the relevance of some findings from animal studies to prediction of human risk is controversial because it is not clear in some cases that the mechanism or mode of action by which toxicity is produced in the animals emulates what occurs in humans. Nevertheless, in recent times, more sophisticated scientific data have provided useful information for postulating mechanisms of toxicity in test animals for many chemicals that are potentially toxic to humans.
Understanding mechanisms of toxic-
ity in animals can lead to new methods for estimation of human risk, although exactly how to interpret scientific advances is not always clear and so how to incorporate the information into risk assessment has not found general agreement. It is evident from the mechanistic data, however, that different risk-assessment methods might be used to model human risk associated with exposure to different chemicals. Progress in the elucidation of underlying mechanisms of toxicity indicates possible high-dose phenomena), whereas comparative biology suggests differences among species. In some cases, markers in animals might not be relevant to humans; in other cases, only indirect extrapolation from animal studies may be inappropriate. For example, there is considerable discussion among scientists regarding chemicals that induce cancer in rodent bioassays but do not exhibit classical genotoxicity and regarding the role of cell proliferation in the development of cancer, particularly for these ''nongenotoxic'' chemicals (Ames and Gold, 1990; Cohen and Ellwein, 1991). Whether events related to cell proliferation induced by high doses of a specific chemical are limiting in carcinogenesis and whether these events are likely to occur in humans is important in predicting the likelihood of carcinogenesis in humans exposed to low doses of the chemical. Most important, if cell proliferation, particularly in response to toxicity, is integrally involved in the induction of tumors, then only the dosages and mechanisms that cause toxicity will produce a proliferative response and result in tumor formation.
Quantitative Issues
The qualitative issues discussed above are related primarily to the hazard-identification component of risk assessment. Secondarily, consideration of those issues focuses attention on the experimental results suitable for quantitative extrapolation. Traditionally, quantitative risk assessment that has involved dose-response modeling (almost all of which has until recently been cancer risk assessment) has relied on extrapolation of the relationship between administered dose and observable disease outcome (e.g., cancer incidence). Lately, with the consideration of physiologically based pharmacokinetic models and biologically based dose-response models, that has begun to change. The role of biologic markers is fundamental to the progress that has been made and will be crucial to further progress.
Consider the simplified flowchart of classes of biologic markers shown in Figure 5-1. The figure shows the sequence of markers paralleling the processes leading from exposure to clinical disease and, more important, indicates the relationships between the markers. The pictorial representation can be translated into semiquantitative form as follows:
DI = f1(E, s1),
DE = f2(DI, s2),
BE = f3(DE, s3),
A = f4(BE, s4), and
C = f5(A, s5),
where E, DI, DE, BE, A, and C are exposure magnitude, internal dose, biologi-
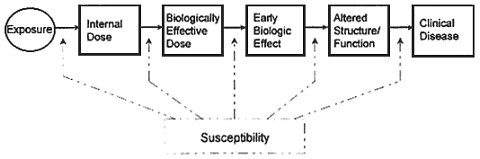
Figure 5-1 Simplified flow chart of classes of biologic markers (indicated by boxes). Solid lines indicate progression, if it occurs, to the next class of marker. Dashed lines indicate that individual susceptibility influences the rates of progression, as do other variables. Biologic markers represent a continuum of changes, and the classification of change might not always be distinct. Source: Adapted from Committee on Biological Markers of the National Research Council, 1987.
ically effective dose, early effective dose, early biologic effect, altered structure or function, and clinical disease, respectively. The functions relating the markers, fi (i = 1, ..., 5) are represented simply but might be complex systems of equations (e.g., a pharmacokinetic model). In this context, the markers of susceptibility, si (i = 1, ..., 5), which might include biologic and nonbiologic components, can be considered indicators of all the factors that can modify the relationships among the markers of exposure and effect. In most cases, it is more appropriate to consider the probability of clinical disease (and perhaps of altered structure or function, of early biologic effect, or of the dose markers) to be functions of the other variables—e.g., P(C) = f5(A, s5), where P(C) is the probability of clinical disease—but the important features of this discussion are not altered in either case.
An example is provided by the development of bladder tumors as a result of exposure to MOCA. In that case, the markers can be defined as follows:
DI: absorbed MOCA,
DE: concentrations of MOCA in urine,
BE: DNA adducts in bladder epithelial cells,
A: increase in red blood cells in urine, and
C: bladder cancer.
The traditional estimation of the risk of bladder cancer in humans would have relied on extrapolating from animals a relationship of the form
P(C) = g(DI),
where g(DI) is some function of administered dose expressed in terms like mg/kg/day or milligrams per kilogram per day or milligrams per square meter per
day. It is assumed here that the conversion from exposure magnitude (e.g., atmospheric concentration to which the animals were exposed) to internal dose, DI, is appropriate, although in practice the estimation of an absorbed dose has typically been rather crude. The function g(DI) might be the multistage model routinely used in cancer risk assessment (Anderson and the Carcinogen Assessment Group of the U.S. Environmental Protection Agency, 1983; Crump, 1984) fitted to the observed cancer results in the animal test species.
If we refer to the relationships between the markers (the functions f1 to f5,) we see that the overall relationship P(C) = g(DI) ought to be representing the marker-to-marker relationships from an internal-dose marker to a marker of clinical disease. In fact, it will be noted that
P(C) = g(DI)
= f5(f4(f3(f2(DI, s2), s3), s4), s5).
This convoluted form indicates why traditional risk-assessment extrapolations are problematic: the relatively simple functional relationship extrapolated from animals to humans should be representing a variety of biologically important processes. Moreover, given the variety of processes and differences among species with respect to markers of susceptibility, it is unlikely that the overall relationship is the same in humans as in animals.
The utility of an approach that considers biologic markers of effective dose, early biologic effect, and altered structure or function is related to our ability to model the relationships among those markers. The fundamental assumption of extrapolation—i.e., that relationships observed and modeled in one setting hold in another setting—can be applied at the more specific level of those marker-to-marker relationships. It might be possible to rely on extrapolation from experimental systems to humans for only a subset of those relationships.
In the case of MOCA, suppose that the observation of occupationally exposed people has provided sufficient information to derive a direct human model linking exposure and urinary MOCA concentrations. A physiologically based pharmacokinetic (PBPK) model derived from human data would serve that purpose; human PBPK models have been developed for other workplace contaminants, such as trichloroethylene (Allen and Fisher, 1993). The human PBPK model would provide the relationships represented by f1 and f2 in the above scheme.
Suppose also the availability of human data that relate the extent of hematuria with the probability of developing bladder cancer. Those data would not necessarily need to be specific to MOCA. This is another advantage of biologic markers in risk assessment: the relationship between two markers, especially markers of effect, need not be chemical-specific, so a wider base of information can be used to conduct an assessment.
If the links between exposure and
urinary concentration and between hematuria and bladder-cancer risk are established from human data, then the only relationships that need to be extrapolated from experimental systems are those between urinary concentration and adduct formation and between adduct formation and the degree of hematuria. Animal-based test systems—in vivo, ex vivo, or in vitro—can be used to elucidate those relationships. They can then be extrapolated to humans on the assumption that the relationships observed in the test systems are the same as in humans. That is the fundamental assumption of risk assessment, but in this context it is a much more particular and specific level than is typical in risk assessment. Because that is the case—i.e., because the extrapolations involve fewer variables and less "gap" between cause and effect—there is a greater chance of appropriately accounting for the modifying factors of susceptibility (e.g., species differences in DNA repair) and therefore a greater likelihood that the assumption is justified.
Even when human data are less direct than in the example just cited, the application of the extrapolation assumption to specific links in the chain leading from exposure to disease will improve the quality of risk-assessment estimates. The links in that chain can often be studied with experimental techniques or systems. Results of the studies can then be incorporated at the appropriate point, allowing even greater consideration of species differences or other differences that can be represented as differences in susceptibility. As mentioned above, the ability to separate the chemical-specific from the chemical-neutral relationships facilitates the incorporation of a larger base of data. The way to improve risk assessment is to give it a firmer scientific foundation. The use of biologic markers to break the disease process into manageable research pieces is a way to do that.
In risk-assessment practice, there will be a need to compromise between the ideal (all the markers one could want and a full understanding and representation of the sequence of events) and the real (less than complete information and less than perfect representation). The application of the basic assumption of extrapolation might not be at the level that one thinks is best; it might have to be at a level as close as possible to the best, given the data at hand. That introduces additional uncertainty, which can be reduced as additional research is conducted to provide more fundamental data, to understand more clearly how cause-effect relationships can be represented, or to elucidate the role of modifying factors.
For assessing risk, any model should account for individual variability in response and health or environmental status. Differences in those factors will alter susceptibility to potential chemical-induced injury.
SUMMARY
Extrapolation from animal models is a common and necessary component of risk assessment for humans. To im-
prove the validity of such extrapolations, a better understanding of the relationship between these markers and disease is needed. In most cases, the scientific basis for assuming that animals are good surrogates for humans, and therefore a suitable basis for extrapolation to humans, is overwhelming. It is reasonable to use animal models for extrapolation to humans unless specific information on specific chemicals indicates otherwise. Identification of chemical hazards should include assimilation and evaluation of all relevant information, including appraisal of physical and chemical properties and structure-activity relationships, which can often provide important indications of potential toxicity. Difficulties in diagnosing renal injury and predicting its health consequences are considerable, primarily because the kidneys can undergo substantial chemically induced injury without any clinical manifestation, and subtle injury can be negligible because of the considerable functional reserve of the kidneys. Standard diagnostic criteria are needed that are sensitive enough to serve as markers of renal damage in the presence of renal functional reserve.
Only whole-animal studies or observations in humans can provide information on the operation of multiple cells, tissues, and organs under the influence of complicated feedback mechanisms. Animals are necessary in the study of chemically induced toxicity, because studies that involve modulation of cellular responses and tissue sampling cannot be performed in humans.
The first stage of any investigation of the nephrotoxicity of a xenobiotic should be in vivo studies. In the absence of any knowledge about potential toxicity and target-organ specificity, the first step should be to determine whether toxicity occurs and the tissue distribution of the toxic response. More detailed studies, both in vivo and in a variety of in vitro models, can then be pursued to elucidate modes of chemical action, specific mechanisms of toxicity, and potential protective or preventive strategies. Animal models developed as surrogates for humans in the study of renal and urinary function should conform to some general principles: the animal model should be reproducible within and among laboratories, the model should be specific to the part of the urinary tract under consideration, the model should be sensitive enough to differentiate normal from abnormal changes or functions, and the model should be able to measure alterations in renal function caused by exogenous agents.
A variety of experimental model systems are available for study of renal metabolism, renal function, and nephrotoxicity. They range from whole-animal studies to those in the isolated perfused kidney, kidney slices, isolated nephron segments, isolated tubule fragments, and isolated renal cells. Each model has advantages and limitations that must be taken into account when developing conclusions and extrapolating animal data to human risk assessment. In vitro models of nonrenal urinary tract epithelia have also been developed and applied primarily toward examination of carci-
nogenesis. Development of markers of exposure and susceptibility has not been addressed directly with such nonrenal models and should be pursued for better extrapolation of data for risk assessment.
The importance of enzymatic activation of toxic chemicals is central to an understanding of chemically induced renal injury. Species and strain differences in amounts and tissue distribution of various enzymes can be critical in determining the ultimate toxic response. Consequently, patterns observed and conclusions reached in one species might not apply to another species. We recommend that species and strain differences in disposition and metabolism be evaluated for each chemical or class of chemicals. For assessing risk, any experimental model should account for individual variability in response and in health and environmental status. Differences in those factors will alter susceptibility to potentially toxic chemicals.






































