6
The Role of Extinction in Evolution
David M. Raup
The extinction of species is not normally considered an important element of Neodarwinian theory, in contrast to the opposite phenomenon, speciation. This is surprising in view of the special importance Darwin attached to extinction, and because the number of species extinctions in the history of life is almost the same as the number of originations; present-day biodiversity is the result of a trivial surplus of originations, cumulated over millions of years. For an evolutionary biologist to ignore extinction is probably as foolhardy as for a demographer to ignore mortality. The past decade has seen a resurgence of interest in extinction, yet research on the topic is still at a reconnaissance level, and our present understanding of its role in evolution is weak.
Charles Darwin on Extinction
In the Origin (1859), Darwin made his view of extinction, and its role in evolution, quite clear. He saw four essential features.
|
(i) |
Extinctions of species have occurred gradually and continuously throughout the history of life. … species and groups of species gradually disappear, one after another, first from one spot, then from another, and finally from the world. (Darwin, 1859, pp. 317–318) |
David M. Raup is professor of geophysical sciences at the University of Chicago, Chicago, Illinois.
-
… the complete extinction of the species of a group is generally a slower process than their production: if the appearance and disappearance be represented … by a vertical line of varying thickness the line is found to taper more gradually at its upper end, which marks the progress of extermination. … (p. 218)
-
(ii)
Sudden disappearances of many species, now called mass extinctions, did not actually occur. Although the Cretaceous–Tertiary (K–T) event was well known in Darwin's day (Lyell, 1833, p. 328), Darwin was convinced that sudden disappearances of species from the fossil record were due solely to unrecognized gaps in the temporal record.
With respect to the apparently sudden extermination of whole families or orders, as of Trilobites at the close of the palaeozoic period [Permian mass extinction] and of Ammonites at the close of the secondary period [K–T mass extinction], we must remember what has already been said on the probable wide intervals of time between our consecutive formations; and in these intervals there may have been much slower extermination. (Darwin, 1859, pp. 321–322)
Like his geologist colleague Charles Lyell, Darwin was contemptuous of those who thought extinctions were caused by great catastrophes.
… so profound is our ignorance, and so high our presumption, that we marvel when we hear of the extinction of an organic being; and as we do not see the cause, we invoke cataclysms to desolate the world, or invent laws on the duration of the forms of life! (p. 73)
-
(iii)
Species extinction is usually, though not always, caused by the failure of a species in competition with other species. That is, causes of extinction are generally biological, not physical.
The inhabitants of each successive period in the world's history have beaten their predecessors in the race for life, and are, insofar, higher in the scale of nature. … (p. 345)
If … the eocene inhabitants … were put into competition with the existing inhabitants, … the eocene fauna or flora would certainly be beaten and exterminated; as would a secondary [Mesozoic] fauna by an eocene, and a palaeozoic fauna by a secondary fauna. (p. 337)
… each new variety, and ultimately each new species, is produced and maintained by having some advantage over those with which it comes into competition; and the consequent extinction of the less-favoured forms almost inevitably follows. (p. 320)
-
(iv)
The extinction of species (and larger groups) is closely tied to the process of natural selection and is thus a major component of progressive evolution. In some passages of the Origin, Darwin seems to have seen extinction as part of natural selection; in others, as an inevitable outcome.
… extinction and natural selection … go hand in hand. (p. 172)
The extinction of species and of whole groups of species, which has played so
- conspicuous a part in the history of the organic world, almost inevitably follows on the principle of natural selection; for old forms will be supplanted by new and improved forms. (p. 475)
Thus, as it seems to me, the manner in which single species and whole groups of species become extinct accords well with the theory of natural selection. (p. 322)
In his final summary of the Origin (pp. 489–490), Darwin listed the fundamental components (''laws") of the evolutionary process: reproduction, inheritance, variability, struggle for life, and natural selection , with its "consequences" divergence of character and the extinction of less-improved forms. Despite Darwin's obvious concern for the role of extinction, the word does not appear in the index to the Origin, nor have biologists paid much attention to the phenomenon until the past decade. Mayr (1964) published an expanded and modernized index to the Origin, but even this contains only a small fraction of the possible citations to the word extinction. For some reason or reasons not entirely clear, extinction largely dropped out of the consciousness of evolutionary biologists and paleobiologists. Only with the advent of vigorous controversy (Alvarez et al., 1980) over the causes of the K–T event, and with the development of concern for presently endangered species, has the role of extinction been confronted in modern terms.
George Gaylord Simpson on Extinction
In Tempo and Mode (1944), Simpson detailed what he considered to be the most important determinants of evolution. These were (Chapter II) variability, mutation rate, character of mutations, generation length, population size, and natural selection. But missing from this chapter is any indication that extinction plays an important role in evolution. To be sure, Chapter II includes occasional mention of specific extinctions, but not as significant drivers of evolution. For example, Simpson suggests that mammals with long generation times (equated with large body size) suffered greater extinction in the latest Pleistocene because natural selection could not operate quickly enough for adaptation to changing climatic conditions. But the implications of this are not developed, and Simpson clearly did not share Darwin's view that extinction is a vital part of the evolutionary process. Elsewhere in Tempo and Mode, however, Simpson noted that major extinctions provide opportunities (space, ecological niches, etc.) for later diversification by the survivors.
In sharp contrast to Darwin's view, Simpson saw interspecies competition as only rarely the cause of extinction of species or larger groups. He thought replacement of one group by another was generally passive.
In the history of life it is a striking fact that major changes in the taxonomic groups occupying various ecological positions do not, as a rule, result from direct competition of the groups concerned in each case and the survival of the fittest, as most students would assume a priori. On the contrary, the usual sequence is for one dominant group to die out, leaving the zone empty, before the other group becomes abundant. … (Simpson, 1944, p. 212; emphasis added)
As examples of passive replacement, Simpson lists the extinction of the ichthyosaurs millions of years before being replaced by cetaceans, the gap between the extinction of the pterodactyls and diversification of bats, and the fact that dinosaurs died out before the radiation of large terrestrial mammals. In the latter case, he notes the not uncommon observation that the successor group (diverse mammals) had been in existence, albeit not thriving, through much of the dinosaur reign.
Simpson has relatively little to say about whether extinction is sudden or gradual. Mass extinctions are acknowledged, but he follows Darwin in arguing that gaps, saltations, and other abrupt changes in the fossil record should not be taken at face value.
Probably there is always a considerable period of time corresponding with the gap in morphology, taxonomy, and phylogeny. It is impossible to prove that there are no exceptions to this generalization, so that there is some danger that it may represent the statement of an a priori postulate rather than evidence for the postulate; but I believe that this is a valid deduction from the facts. (p. 111)
Taken as a whole, Simpson's treatment of extinction is very different from Darwin's: they differ in their perceptions of the causes of extinction (but not the rates) and, above all, in the role of extinction in evolution.
Despite the foregoing, Simpson made important and lasting contributions to the study of extinction in the fossil record through his use of survivorship analysis above the species level. His now-classic comparison of the slopes of survivorship curves for bivalve mollusks and carnivorous mammals set the stage for development of the technique by Van Valen (1973) and many others. Much of our synoptic knowledge of extinction today relies on an expanded use of these techniques.
The Record of Extinction
The known fossil record contains roughly a quarter of a million species, most of which are extinct. Although fossils of the earliest forms are important to our knowledge of the history of life, the fossil record is dominated numerically by the remains of multicellular organisms from the last 600 million years (Myr). Fossil species are grouped into about 35,000 genera and 4000 families. About one-quarter of the families are still living.
Although the fossil record is ample for statistical purposes, it contains a very small fraction of all the species that have ever lived. Estimates of that fraction range from <1% to a few percent, depending on the organisms being considered and assumptions about past biodiversity and turnover rates. The probability of fossilization is strongly influenced by many biological and physical factors. Marine animals with hard skeletons are strongly favored, and as a result, the fossil record is dominated by these groups (e.g., mollusks, brachiopods, reef corals). Even for these organisms, however, biases in preservation abound.
The dinosaur fossil record illustrates some of the more extreme sampling problems. According to a review by Dodson (1990), 336 of the named species of dinosaur are taxonomically valid. Of these, 50% are known only from a single specimen, and about 80% are based on incomplete skeletons. The 336 species are grouped into 285 genera, and of these, 72% have been found only in the rock formation where they were first discovered, and 78% have been found in only one country. These numbers are astonishing if viewed as if the data were complete. The species/genus ratio being barely above unity is undoubtedly due to incomplete sampling, as is the apparent biogeographic restriction.
Incomplete sampling also influences our estimates of extinction rates. Lack of fossilization inevitably shortens the apparent life span of species, and this may explain why durations of dinosaur species are far shorter than is typical of other, better preserved organisms. On the other hand, short-lived, localized species have a low probability of appearing in the fossil record at all. The net effect of these biases is that statistical estimates of mean duration are almost certainly exaggerated. That is, the fossil record is biased in favor of successful species—successful in the sense of surviving for a long time and being ecologically and geographically widespread. Thus, analysis of past extinctions must operate in a sampling regime very different from that of present-day biodiversity.
Figure 1 shows the frequency distribution of recorded life spans of 17,500 genera of fossil marine animals. The distribution is highly skewed, with the mean (28 Myr) being the result of many short durations combining with a few very long ones. Survivorship analysis of the genus data indicates a mean species duration of 4 Myr (Raup, 1991a), although as indicated above, this is probably a high estimate because of the dominance of successful species in the sample. Regardless of the uncertainties, however, species and genus residence times on earth are very short on geologic time scales. The longest-lived genus in Figure 1 (160 Myr) lasted only about 5% of the history of life.
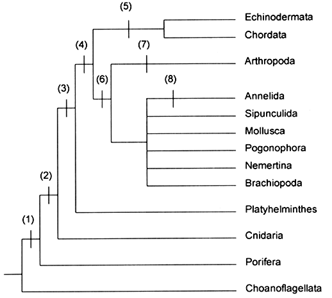
Figure 1 Life spans of about 17,500 extinct genera of marine animals (vertebrate, invertebrate, and microfossil) tabulated from data compiled by Sepkoski (1989).
Episodes of Extinction
It is conventional to divide extinctions into two distinct kinds: background and mass extinction. The term "mass extinction" is most commonly reserved for the so-called "Big Five" events: short intervals in which 75–95% of existing species were eliminated (Table 1). The K–T
TABLE 1 Comparison of species extinction levels for the Big Five mass extinctions
|
Extinction episode |
Age, Myr before present |
Percent extinction |
|
Cretaceous (K–T) |
65 |
76 |
|
Triassic |
208 |
76 |
|
Permian |
245 |
96 |
|
Devonian |
367 |
82 |
|
Ordovician |
439 |
85 |
|
Extinction data are from Jablonski (1991). |
||
event, mentioned earlier, is one of the Big Five, but not the largest. Although the Big Five were important events, their combined species kill amounted to only about 4% of all extinctions in the past 600 Myr (Raup, 1993). The mass/background dichotomy is unfortunate because it implies two modes of extinction, yet there is no evidence for a discontinuity between them. Figure 2 shows variation in percent species kill in 1-Myr intervals for the past 600 Myr. The events called mass extinctions are concentrated in the right-hand tail, but there is no break between this tail and the main distribution. The data appear to produce a single, highly skewed distribution. Thus, segregating mass extinction from background has no more meaning than distinguishing hurricanes from other tropical cyclonic storms on the basis of some arbitrary wind speed (64-knot sustained surface winds). Continued use of the mass/background dichotomy serves only to hide interesting structure in extinction data.
Figure 3 shows a cumulative distribution of extinction frequency, the so-called "kill curve" for species of the past 600 Myr. The format is one used commonly to analyze severe storms, floods, earthquakes, and other natural phenomena where the larger the event, the rarer it is. The kill curve gives the average time interval (mean waiting time) between an extinction event and the next one of equal or greater magnitude. An "event" is defined as the species kill occurring in an arbitrarily short interval. Thus, 10% (or more) of the standing crop of species goes
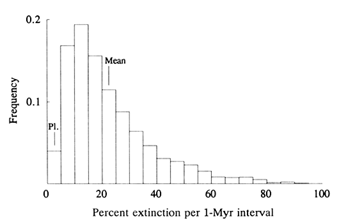
Figure 2 Variation in intensity of extinction for 1-Myr intervals during the past 600 Myr, based on the species kill curve (Raup, 1991a). "Pl." refers to extinction intensity of the Pleistocene glacial epoch. Mass extinctions occupy the right-hand tail of the distribution. The mean extinction rate, 25% extinction per 1 Myr, is the approximate reciprocal of the mean species duration (4 Myr).
Figure 3 Kill curve (heavy line) for the past 600 Myr (Raup, 1991a). Waiting time is the average interval between events of a given extinction intensity. Thus, for example, a short episode of extinction which kills 30% of standing species diversity occurs on average every 10 Myr ("10-million-year event"), with no implication of uniform periodicity in the spacing of events. Light curves bound the uncertainty in placement of the kill curve from fossil data (Raup, 1992).
extinct, on average, every 1 Myr, 30% every 10 Myr, and 65% every 100 Myr (Raup, 1991a). The 100-Myr events include the Big Five mass extinctions.
Without further analysis, one could assert that the kill curve is a natural result of chance coincidence of independent events. That is, pure chance might produce an episode of nearly simultaneous extinctions if we wait long enough. This is emphatically not the case. For a
random model that assumes that all species extinctions are independent of one other, the probability of a 10% extinction every 1 Myr (on average) is vanishingly small (Raup, 1991a). In fact, a kill curve based on this model could not be plotted at the scale of Figure 3: the curve would be indistinguishable from the horizontal axis. The only available conclusion is that extinctions are nonrandomly clustered in time, and this implies strongly that the K-T extinctions, for example, had a common cause.
Some of the clustering of extinction may be due to the removal of one or a few species that are crucial to the existence of other species. Or clusters may be due to destruction of one important ecosystem or habitat. However, for the larger events, at least, the extinctions are far more pervasive. At the end of the Cretaceous, high levels of species extinction (>50%) are found in all geographic areas and involve organisms as different as burrowing mollusks, planktonic microorganisms, land plants, and dinosaurs. This suggests, among other things, that the big mass extinctions cannot be explained by Darwin's species interactions unless one is willing to postulate an incredible degree of connectedness in the biosphere.
A striking effect of the typical mass extinction is its aftermath. For as long as 5–10 Myr, fossil faunas and floras are impoverished and are often dominated by only one or two species. The longest such interval followed the late Permian extinction (the largest of the Big Five): many major phyla and classes, known to have survived from later occurrences, are absent from the early Triassic assemblages. And about a third of the Triassic is characterized by what has been called the "coal gap," an interval where no coal deposits have been found—either of temperate or of tropical origin (A. M. Ziegler, University of Chicago, pers. comm., 1993).
When full diversity does return, it often has a strikingly different character. A classic example is the history of marine reefs. Reef communities have been wiped out several times in the past 600 Myr, coinciding in four cases with Big Five events. Each time reefs reappear, the principal framework organisms have changed, switching back and forth between calcareous algae, sponges, bryozoans, rudist mollusks, and various corals (Sheehan, 1985; Copper, 1988). The contemporary term "coral reef" describes only the current occupants of that adaptive zone.
Selectivity
Darwin argued that all extinction is selective: species not able to compete with other species die out. In one of the passages quoted above
(p. 337), Darwin expressed confidence that if ancient species could be re-created today and put in competition with their modern counterparts, the old species would be "beaten and exterminated." This is definitely not the current view, and major research programs are now being devoted to determining the extent, if any, of selectivity in past extinctions. A common (though by no means proven) view is that the victims of extinction are in no way different from the survivors, except for the fact of their extinction. Simpson (1944) was clearly moving in this direction when he suggested in Tempo and Mode that the mammals were the lucky recipients of space vacated by the dinosaurs.
Taxonomic Selectivity. Much of current extinction research attempts to identify taxonomic selectivity. Do some taxonomic groups suffer significantly more species extinction in an extinction episode than other groups? These studies can take advantage of the availability of taxonomic data bases, such as those compiled by Sepkoski for marine genera (Sepkoski, 1989) and families (Sepkoski, 1992), and thus have the benefit of large samples. The approach carries the tacit assumption that genealogical relatedness implies similarity of physiology, ecology, or other attributes that determine susceptibility to extinction.
Taxonomic selectivity has been documented, but the effect is generally quite small and requires massive samples for confirmation. For example, when extinction rates for several taxonomic groups are compared with the mean for all groups, about 10% differ from the mean at a 0.05 significance level, whereas 5% would be expected by chance. Similarly, about 2% of the tests are significant at the 0.01 level. Thus, taxonomic selectivity is present but minor.
Occasionally, pronounced taxonomic selectivity has been found. The dinosaur extinction is such a case. In the latest Cretaceous of western North America, Clemens (1986) tabulated 117 genera of fossil mammals, amphibians, reptiles, and fish; 50 of these (43%) died out at or near the end of the Cretaceous, including all 22 dinosaur genera. The null hypothesis that all genera shared the same probability of extinction (0.43) can be rejected easily, and this demonstrates a clear bias against dinosaur survival. But such cases are relatively rare. In the same data set, for example, 8 of 24 mammalian genera died out (33%), but because of the small sample size, it is impossible to demonstrate that this extinction rate is significantly lower than the mean for all groups.
Small sample sizes have plagued many studies of selectivity, giving rise to generalizations that are widely accepted but not supportable statistically. For example, it is often claimed that the amphibians survived the K-T event with little difficulty. In the Clemens data set, only one-third of the amphibian genera went extinct (equal to the
mammalian rate), but the sample size (12 genera) is, again, much too small for statistical significance.
Selectivity for Specific Traits. Somewhat more success has been achieved by focusing directly on aspects of physiology, behavior, habitat, and biogeography. Jablonski (1986a) showed, for example, that marine mollusks with planktonic larvae survive longer during "background" times than those which develop directly from the egg. This result is reasonable because species with a planktonic stage have greater dispersal capabilities and can attain wider geographic distributions, and thus are more likely to survive stresses that eliminate species in small areas. Whereas Jablonski has confirmed that widespread species are significantly less likely to go extinct during most geologic intervals, he has also shown that this protection breaks down at times of severe mass extinction, when broad geographic range at the genus level becomes important (Jablonski, 1986b).
It is commonly thought that tropical organisms suffer more extinction than those in higher latitudes. Although this is supported by anecdotal data for several extinction events, a recent study based on a global data base (3514 occurrences of 340 genera) found no recognizable geographic pattern in extinction of bivalve mollusks at the end of the Cretaceous, once reef-dwelling rudists were omitted (Raup and Jablonski, 1993). Approximately 50% of all genera died out, regardless of geographic position. Unpublished follow-up studies show a lack of habitat selectivity for bivalves and gastropods of the Gulf Coast during the same interval (D. Jablonski, University of Chicago, pers. comm., 1993). It may be that extinction is selective when overall extinction rates are low (so-called background extinction), but not when rates are high.
Large body size is thought to increase the risk of extinction. Indeed, many apparently good examples exist, including the now-extinct dinosaurs, ammonites, eurypterids, mammoths and mastodons, and rudist clams (LaBarbera, 1986). In the terrestrial realm, at least, decreased survival can be related easily to body size through demographic considerations (small populations, large home range, low birth rates, etc.). But the issue is clouded by the lack of rigorously controlled statistical analysis and by the fact that in several cases (e.g., eurypterids and ammonites), the largest species did not exist late in the group's range.
Species Selection. A special case of selective extinction involves differential origination and extinction of species in an evolving clade. In theory, species carrying a favored trait should survive longer and thus have greater opportunity for speciation than less well-adapted species.
The expected result is an increase in frequency of species carrying the favored trait. The species-selection idea is close to Darwin's view of selective extinction among closely related species. Unfortunately, well-documented cases of species selection are few. Also, there has been vigorous disagreement among evolutionary biologists and paleobiologists on whether species selection could ever be an important force in evolution. On the one hand, it is argued that species selection can alter frequencies of alternative traits, even eliminating some, but cannot be responsible for complex adaptations such as eyes and limbs (Levinton et al., 1986). The counterargument is that adaptations, originating at the population level by natural selection, may be sustained, even "cloned," by species selection, thus making possible further improvements in the trait by natural selection operating within the descendent species. By the latter scenario, species selection plays a useful and possibly indispensable part in the evolution of those adaptations (including complex organs) that require more time than is available during the life span of a single species. Again, it must be emphasized that there are too few authenticated cases of species selection to build a strong case for or against its role in evolution.
Units of Selectivity. To the extent that selective extinction occurs, it may operate at several alternative hierarchical levels. In the dinosaur extinction, all species of two major orders (Saurischia and Ornithischia) were eliminated. Presumably these species had something in common that made them all susceptible to the environmental stresses of the terminal Cretaceous. Selectivity at this high level is the only way by which highly diverse groups of species (classes or orders) have a measurable probability of being eliminated completely. It can be shown easily that if the determinants of extinction were at the species level, independent of membership in a larger group, diverse clades would never die out (Raup, 1981). But the fossil record contains ample instances of large, successful clades going extinct, either gradually (e.g., trilobites) or suddenly (e.g., ammonites).
Summary. Extinction is evidently selective at certain times and places but the effects tend to be subtle and require careful analysis of large data bases. Darwin's contention that all extinction is selective cannot be sustained, although this may reflect only our inability to recognize complex patterns in an imperfect fossil record.
Causes of Extinction
A remarkable feature of the history of life is that so many successful species have died out. Many of the extinctions recorded in the fossil record are of species or large groups of species that were ecologically tolerant and occurred in great numbers in all parts of the world. If these extinctions were caused by slow declines over long periods of time, as Darwin thought, they might be explicable in terms of the cumulative effect of very slight deficiencies or disadvantages. But it is becoming increasingly clear that successful species often die out quickly. This is best documented for the K-T mass extinction because of the extensive field work inspired by the controversy over the cause of that event. Several important biologic groups, including the ammonites and dinosaurs, now appear to have existed at full diversity right up to the K-T boundary (Ward, 1990; Sheehan et al., 1991).
For a species to survive for several million years, as many do, it must be well adapted to the physical and biological stresses normal in its environment. Tree species, for example, that can withstand, or even benefit from, forest fires have presumably evolved this ability because forest fires are common in their environment. It may well be that most species have evolved ways of surviving anything that their environment can throw at them, as long as the stress occurs frequently enough for natural selection to operate. This implies, in turn, that likely causes of extinction of successful species are to be found among stresses that are not experienced on time scales short enough for natural selection to act.
The recent Pleistocene glaciation produced very few complete extinctions of species. To be sure, extinction rates during the last deglaciation were high among large mammals and some bird groups but overall, global data (including marine organisms) show the Pleistocene to be on the left-hand tail of the distribution in Figure 2. A reasonable explanation is that although the glaciation was associated with marked shifts in climatic regimes, most species were already equipped to cope with the changes by natural physiological tolerance, by having populations in refugia, or by having the ability to migrate to more favorable areas. This appears to be especially true in the marine realm (Clarke, 1993).
In view of the foregoing, recent hypotheses for extinction caused by the catastrophic effects of extremely rare physical events (e.g., asteroid or comet impact, global volcanism) have great appeal.
The Role of Extinction
Despite many uncertainties, we can formulate a reasonable statement of the probable role of extinction, containing the following elements.
-
(i)
Extinction of a widespread species, or a widespread group of species, requires an environmental shock (physical or biological) which is not normally encountered during the geological life spans of such species or groups, and the shock must be applied rapidly enough over a broad geographic area to prevent adaptation by natural selection or escape by migration. If the most effective extinction mechanisms are beyond the experience of the victims, a high degree of apparent randomness should be expected. Survivors are most likely to be those organisms which are fortuitously preadapted to an ''unexpected" stress (Raup, 1991b).
-
(ii)
The most intense episodes of extinction, like the Big Five, produce major restructuring of the biosphere. Three-quarters, or more, of the standing diversity is removed, and diversification of the surviving lineages yields a global biosphere very different from that before the extinctions. Previously successful clades are lost, and unlikely survivors expand. Although the extinction does not, by itself, make a creative contribution to the evolution of complex structures such as wings or limbs, it may be decisive in sustaining or eliminating such structures.
The pterosaurs died out in the latest Cretaceous, and reptiles never again achieved powered flight. Did this foster the Tertiary radiation of bats? What further adaptations might pterosaurs have evolved had they survived? Seen in this light, the major extinctions have a profound influence on the future course of evolution, sometimes constructive and sometimes destructive.
-
(iii)
At lower levels of extinction intensity, Darwin-style selectivity may be relatively common, but except for a few spectacular cases, including Jablonski's studies of the effects of larval development and geographic range (Jablonski, 1986a), we do not have enough hard evidence to claim that low-level extinction has anything approaching the importance given it by Darwin. Further studies in this area, under the rubric of species selection, are sorely needed.
Summary
The extinction of species is not normally considered an important element of neodarwinian theory, in contrast to the opposite phenomenon, speciation. This is surprising in view of the special importance Darwin attached to extinction, and because the number of species extinctions in the history of life is almost the same as the number of originations; present-day biodiversity is the result of a trivial surplus of originations, cumulated over millions of years. For an evolutionary biologist to ignore extinction is probably as foolhardy as for a demographer to ignore mortality. The past decade has seen a resurgence of
interest in extinction, yet research on the topic is still at a reconnaissance level, and our present understanding of its role in evolution is weak. Despite uncertainties, extinction probably contains three important elements. (i) For geographically widespread species, extinction is likely only if the killing stress is one so rare as to be beyond the experience of the species, and thus outside the reach of natural selection. (ii) The largest mass extinctions produce major restructuring of the biosphere wherein some successful groups are eliminated, allowing previously minor groups to expand and diversify. (iii) Except for a few cases, there is little evidence that extinction is selective in the positive sense argued by Darwin. It has generally been impossible to predict, before the fact, which species will be victims of an extinction event.
I thank David Jablonski for many helpful discussions and for his helpful comments on the manuscript. This research was supported by National Aeronautics and Space Administration Grants NAGW-1508 and NAGW-1527.
References
Alvarez, L. W., Alvarez, W., Asaro, F. & Michel, H. V. (1980) Extraterrestrial cause for the Cretaceous-Tertiary extinction. Science 205, 1095–1108.
Clarke, A. (1993) Temperature and extinction in the sea: a physiologist's view. Paleobiology 19, 499–518.
Clemens, W. A. (1986) Evolution of the vertebrate fauna during the Cretaceous-Tertiary transition, in Dynamics of Extinction, ed. Elliot, D. K. (Wiley-Interscience, New York), pp. 63–85.
Copper, P. (1988) Ecological succession in Phanerozoic reef ecosystemns: Is it real? Palaios 3, 136–152.
Darwin, C. (1859) On the Origin of Species (Murray, London).
Dodson, P. (1990) Counting dinosaurs: How many kinds were there? Proc. Natl. Acad. Sci. USA 87, 7608–7612.
Jablonski, D. (1986a) Causes and consequences of mass extinctions: a comparative approach, in Dynamics of Extinction, ed. Elliot, D. K. (Wiley-Interscience, New York), pp. 183–229.
Jablonski, D. (1986b) Mass and background extinctions: the alternation of macroevolutionary regimens. Science 231, 129–133.
Jablonski, D. (1991) Extinctions: a paleontological perspective. Science 253, 754–757.
LaBarbera, M. (1986) The evolution and ecology of body size, in Patterns and Processes in the History of Life, eds. Raup, D. M. & Jablonski, D. (Springer-Verlag, Berlin), pp. 69–98.
Levinton, J. S. et al. (1986) Organismic evolution: the interaction of microevolutionary and macroevolutionary processes, in Patterns and Processes in the History of Life, eds. Raup, D. M. & Jablonski, D. (Springer-Verlag, Berlin), pp. 167–182. [other authors: Bandel, K., Charlesworth, B., Muller, G., Nagl, W., Runnegar, B., Selander, R. K., Stearns, S. C., Turner, J. R. G., Urbanek, A. J. & Valentine, J. W.]
Lyell, C. (1833) Principles of Geology (Murray, London), Vol. 3.
Mayr, E. (1964) Index to Facsimile Edition of Darwin's Origin of Species (Harvard Univ. Press, Cambridge, MA), pp. 497–513.
Raup, D. M. (1981) Extinction: bad genes or bad luck? Acta Geol. Hispan. 16, 25–33.
Raup, D. M. (1991a) A kill curve for Phanerozoic marine species. Paleobiology 17, 37–48.
Raup, D. M. (1991b) Extinction: Bad Genes or Bad Luck? (Norton, New York).
Raup, D. M. (1992) Large-body impact and extinction in the Phanerozoic. Paleobiology 18, 80–88.
Raup, D. M. (1993) Extinction from a paleontological perspective. Eur. Rev. 1, 207–216.
Raup, D. M. & Jablonski, D. (1993) Geography of end-Cretaceous marine bivalve extinctions. Science 260, 971–973.
Sepkoski, J. J., Jr. (1989) Periodicity in extinction and the problem of catastrophism in the history of life. J. Geol. Soc. London 146, 7–19.
Sepkoski, J. J., Jr. (1992) A compendium of fossil marine families. Milwaukee Contrib. Biol. Geol. 83, 1–156.
Sheehan, P. M. (1985) Reefs are not so different—they follow the evolutionary pattern of level-bottom communities. Geology 13, 46–49.
Sheehan, P. M., Fastovsky, D. E., Hoffmann, R. G., Berghaus, C. B. & Gabriel, D. L. (1991) Sudden extinction of the Dinosaurs: Latest Cretaceous, Upper Great Plains, U.S.A. Science 254, 835–839.
Simpson, G. G. (1944) Tempo and Mode in Evolution (Columbia Univ. Press, New York).
Van Valen, L. (1973) A new evolutionary law. Evol. Theory 1, 1–30.
Ward, P. D. (1990) A review of Maastrichtian ammonite ranges. Geol. Soc. Am. Spec. Pap. 247, 519–530.
















