The Chemistry of Sex Attraction
WENDELL L. ROELOFS
Mate attraction involves multiple sensory modalities with almost as many variations as there are numbers of species. It broadly includes pheromones of single-celled organisms to those involved in uniting mates over distances of several kilometers, elaborate plumages and songs in birds, croaks and chucks in frogs, electrical discharges in fish, etc. In insects, a pattern of light flashes from a firefly or of chirps from a cricket are conspicuous signals used to attract mates, but the most common form of long-distant sex attraction in insects (and many other animal species) involves invisible odors (chemicals!). One interesting exception to invisibility is when a visible aerosol of pheromone is produced to attract a mate (Figure 1).
The phenomenon of virgin female moths attracting large numbers of male moths was noted long before it was understood that a trail of chemicals could be so effective over great distances. In 1882 J. A. Lintner, the first state entomologist in New York, described (1) a spectacle of 50 large male Promethea moths being attracted to a female moth placed in his office window, which in turn attracted a large crowd of people on the sidewalk. He not only acknowledged the role of chemicals in this process and the existence of ''smell organs" but also foresaw the potential use of these chemicals for insect control. He writes (1):
Wendell Roelofs is Liberty Hyde Bailey Professor of Insect Biochemistry at Cornell University, Geneva, New York.
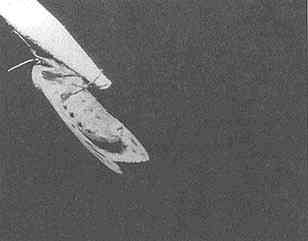
FIGURE 1 Female arctiid moth in a wind tunnel. Pheromone emission as a visible stream of liquid droplets is shown. Photo by S. Krasnoff.
It is the existence of these organs that many of the Families of Lepidoptera, especially among the moths, owe the continuance of the species. They operate in the association of the sexes, with such an irrestible and far-reaching force as to exceed our conception and to be marvelous to our eyes. . . . Can not chemistry come to the aid of the economic entomologist in furnishing at moderate cost, the odorous substances needed? Is the imitation of some of the more powerful animal secretions impracticable?
This statement sums up much of the research that has been conducted on the chemistry of sex attraction in insects over the past few decades. Research efforts have focused on the overt chemical signals used in the mating process, with the driving force and financial backing for much of the research due to the potential for use of synthetic pheromone chemicals in pest control programs. This effort has resulted in the identification of sex attractants for >1600 insect species from >90 families in nine orders, with an emphasis on Lepidoptera (2). The variety and complexity of chemical structures (>300 reported structures) observed among the various insect orders attest to the insect's amazing ability to sequester and synthesize unique structures and blends, mainly composed of acetogenins and mevalogenins. These essential signaling chemicals of the "sex attractant" communication system are interesting but represent only the more obvious chemicals involved in complex systems of synchronous emitters and receivers. Rather than review all data on this enormous
topic, this paper will discuss some of the chemistry involved in various aspects of the sex pheromone communication system in insects.
OVERT CHEMICAL SIGNALS
The sex pheromone communication system basically involves the release of specific chemicals from a pheromone producer (emitter), the transmission of these chemicals in the environment to a receiver, and the processing of these signals to mediate appropriate behavioral responses in the receiver. The chemicals transmitted downwind have been the most obvious targets for characterization. The code was first broken with the publication in 1959 (3) of the sex pheromone for the domesticated silkworm Bombyx mori after extraction of a half million female silkworm pheromone glands and 30 years of classical chemical analyses. The pheromone was found to be (E10, Z12)-hexadecadien-1-ol, which was called bombykol. This work showed that there was nothing magical about the communication system, and chemists around the world were "attracted" to this area of research on insect pheromones.
Rapid Identification of Pheromone Components with the Electroantennogram
Research on the isolation and identification of various chemical signals used for mate attraction in insects has progressed through stages of improving technologies [e.g., capillary gas/liquid chromatography (GLC), airborne collectors, flight-tunnel bioassays, and powerful instrumental analyses on microsamples]. One of the most sensitive instruments used throughout the past two decades has been the electroantennogram (EAG) apparatus, which utilizes the male's antenna as a finely tuned detector for active materials. Schneider (4, 5) was the pioneer for setting up the EAG to carry out electrophysiological experiments on olfaction in insects. The sensitivity and specificity of the male's antenna to its own pheromone components made it a powerful tool in assaying for pheromone components and in predicting their structures (6). GLC retention times on nonpolar and polar columns of active compounds in crude female gland extracts could be determined quickly by collecting the GLC effluent in a separate capillary tube each minute and puffing air through each tube and across an isolated male antenna in the EAG setup. Depolarization of the isolated antenna in response to each puff is amplified and recorded or observed with an oscilloscope. The amplitude of response to active compounds was found to correlate to the frequency of generated nerve impulses, and thus, pheromone components in the extract elicited responses with the highest amplitudes.
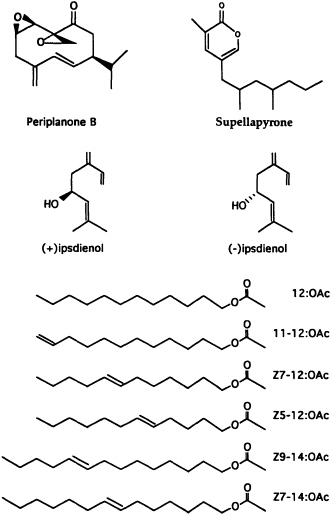
FIGURE 2 Pheromone structures of the American cockroach (periplanone B), the brownbanded cockroach (supellapyrone), bark beetles (ipsdienol enantiomers), and the cabbage looper moth (six acetates).
The EAG technique as a bioassay tool for active fractions has been used in the identification of sex pheromones of many insect species in several orders and remains as a key factor in the identification of pheromones. In the recent identification of the brownbanded cockroach pheromone supellapyrone (7) (Figure 2), the EAG technique was used throughout the entire process of isolating and purifying active material from 12,000
female roaches. Additionally, the EAG technique was used on many moth species to predict the positions and configuration of double bonds in long-chain fatty acetates, aldehydes, and alcohols. GLC retention time data was obtained to determine the carbon-chain length and functional group, and then every monounsaturated compound from the suggested library (e.g., monounsaturated 12-carbon acetates) was puffed across the antenna to determine relative response amplitudes. In many cases it was easy to determine that the main pheromone component was (Z8)- or (Z9)or (E10)-dodecenyl acetate, etc. In the first use of the EAG for pheromone identification, the highly pursued structure of the codling moth sex pheromone was determined in a short time with only a few moths (8). The GLC retention time data indicated a conjugated 12-carbon alcohol, and responses from the library of monounsaturated 12-carbon alcohols showed that the antenna responded quite specifically to the (E8), the (E9), and the (E10) isomers in that series. These data indicated an (E8, E10) conjugated system. Synthesis and EAG analyses of all four 8,10 geometrical isomers showed that the E,E isomer produced the greatest antennal responses of all test compounds. Behavioral studies proved it to be a very potent sex attractant for this species, and it was later isolated from the female pheromone gland.
Complex Structures vs. Specific Blends
A mating communication signal wafting through a "noisy" chemical background in the environment must be specific for the receiver. One answer to this problem is to make a chemical that is so complex that it is not duplicated in nature and so imparts great species specificity. Single complex chemicals indeed have been found that elicit the full repertoire of behavioral responses at very low biologically meaningful concentrations. The cockroach sex pheromones (Figure 2) periplanone B for the American cockroach Periplaneta americana (9) and supellapyrone for the brownbanded cockroach Supella longipalpa (7) are examples of such chemicals that alone can attract conspecific males from a distance with the release of only hundreds of molecules.
Although a number of species utilize complex chemicals containing chiral centers and may, in fact, produce only one stereoisomer, the behavior of the responding male in some species is not affected by the presence of the other isomers and a racemic blend is as active as the pure isomer. However, other cases have been found in which a distinct chemical is found to be used by an insect species, but it is also part of the mating communication system of other related species. Species specificity then can be effected by utilizing a specific blend of the enantiomers (10). An example of this is with the two bark beetle species that utilize
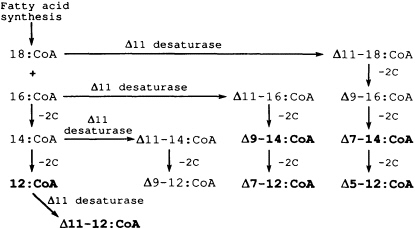
FIGURE 3 Pheromone biosynthetic pathways commonly used in moth sex pheromone glands to produce precursors for specific blends of acetates, alcohols, or aldehydes. Cascades of precursors are produced by combinations of unique Δ11-desaturases and limited chain-shortening steps. The six precursors for the cabbage looper blend (Figure 2) are in boldface type.
ipsdienol. The western species Ips pini produces only the (-)-enantiomer, whereas the eastern species Ips pini pini utilizes a 65% (+)/35% (-) mixture (11) (Figure 2).
In contrast to pheromones that involve single complex compounds, many moth species have been found to utilize a specific blend of relatively simple fatty acid-derived compounds. It appears that the evolution of a unique enzyme, Δ11-desaturase, used in combination with 2-carbon chain-shortening reactions (Figure 3) has allowed moth species to produce a variety of unsaturated acetates, aldehydes, and alcohols that can be combined in almost unlimited blends to impart species specificity. For example, biosynthetic precursors for the six-component pheromone blend of acetates for the cabbage looper moth (12) (Figure 2) can be determined easily from the cascade of acyl intermediates produced by the Δ11-desaturase and chain-shortening reactions (Figure 3).
SEX PHEROMONE PRODUCTION
Key Biosynthetic Enzymes
The overt chemical signals are only part of the chemistry involved in the overall communication system. The production of each pheromone component is dependent on specifically evolved chemical reactions
taking place in special chemical factories (secretory cells) in the producing insect. Key to the biosynthesis are enzymes that either convert exogenous materials (such as oleic acid) into pheromone precursors or produce unique compounds from mevalonic acid or acetate. As shown above with the cabbage looper moth, a unique ∆11-desaturase is found in lepidopteran sex pheromone glands to produce a whole variety of precursors in combination with chain-shortening reactions, followed by the action of reductases and acetyltransferases to produce the acetate pheromone components.
The enzymes themselves are only part of the chemistry involved at this stage. A more complete understanding of the biosynthesis includes studies on the structure of the enzymes, their cofactors, and the corresponding mRNA and gene. By using the ∆11-desaturase as an example, it probably functions similarly to the stearoyl-CoA desaturase in animals, which has been found to be the terminal component of the electron transport system of endoplasmic reticulum that utilizes cytoplasmic NADH and molecular oxygen to effect ∆9-desaturation of CoA esters of long-chain fatty acids (13). Research with liver microsomes has shown that there are three enzyme components of this sequence: a NADHcytochrome b5 reductase (a flavoprotein), cytochrome b5 (a hemoprotein), and the ∆9-desaturase (cyanide-sensitive factor). All three enzyme components are oriented with their catalytic domains on the cytoplasmic side of the endoplasmic reticulum, but since the cytochrome enzymes are solubilized more readily than is the desaturase, it was suggested that the desaturase protein is essentially buried in the microsomal membrane with just a small portion of it containing the active center exposed to the cytoplasm.
Stearoyl-CoA desaturase from rat liver has been purified and identified to be a protein with 358 amino acids and a molecular weight of 41,400 (14). The mRNA for this desaturase was found to be 4900 bases long, although a 1074-base open reading frame was found to code for a protein consisting of 358 amino acids of which 62% are hydrophobic. Two stearoyl-CoA desaturases have been characterized from mouse adipose tissue (15, 16) with 92 and 86% homology to that of the rat, and one from yeast (17) shows 36% identity and 60% similarity to the rat desaturase. The rat ∆9-desaturase is able to functionally replace the yeast ∆9-desaturase. Research on the characterization of the unique ∆11-desaturase in moth pheromone glands is based on the tenet that the specialized desaturases in the pheromone glands are structurally and functionally related to the common ∆9-desaturase that performs a general function in cellular lipid metabolism of animals and fungi. It must be determined whether the two desaturases are derived from independent transcription units or whether they are produced from the same transcription unit by
a developmentally regulated alternative splicing mechanism. Data on the ∆11-desaturase used in the biosynthesis of sex pheromone components will be important in addressing questions on how this enzyme evolved and what role this process played in speciation.
Hormonal Control
Another aspect of the sex pheromone communication system concerns the endogenous signals that control pheromone production and release from the emitting insect. A number of hormones have been found to be involved in the control of pheromone production in various insect species (18). Juvenile hormone was found to induce vitellogenesis and sex pheromone production in some cockroach and beetle species. However, ecdysteroids were found to be involved in regulating reproductive processes, including vitellogenin synthesis, in dipteran species.
In moths, it was discovered in Helicoverpa zea that a peptide produced in the subesophageal ganglion portion of the brain complex regulates pheromone production in female moths (19). This factor has been purified and characterized in three species, Helicoverpa zea (20), Bombyx mori (21, 22), and Lymantria dispar (23). They are all a 33- or 34-amino acid peptide (named pheromone biosynthesis activating neuropeptide, PBAN) and have in common an amidated C-terminal 5-amino acid sequence (FXPRLamide), which is the minimum peptide fragment required for pheromontropic activity. In the redbanded leafroller moth, it was shown that PBAN from the brain stimulates the release of a different peptide from the bursae copulatrix that is used to stimulate pheromone production in the pheromone gland found at the posterior tip of the abdomen (24).
The chemistry involved in the mode of action of PBAN is complex and still under investigation, but present studies have shown that PBAN can interact directly with the pheromone gland and apparently stimulates pheromone biosynthesis by activating a plasma membrane calcium channel that regulates production of cAMP (25) (Figure 4). Other enzymes and effectors are likely involved in the signal transduction but remain uncharacterized. Several studies have been carried out to determine which enzyme(s) in the pheromone biosynthetic pathway is (are) controlled by PBAN. It apparently affects a step in or prior to fatty acid synthesis in several species (26, 27), perhaps involving acetyl-CoA carboxylase, although reduction of fatty acids to alcohols (reductase) was found to be controlled by PBAN in some other species (28, 29).
Studies of PBAN at the molecular level have shown that PBAN is synthesized as part of a larger precursor from which it is derived by posttranslational processing (30, 31). The two PBAN genes identified to date encode not only the PBAN peptide but also four additional neu-
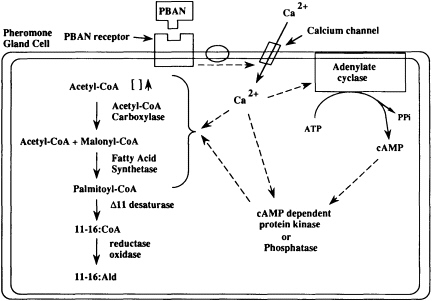
FIGURE 4 Proposed model for the interaction of PBAN with a receptor on the sex pheromone gland of a corn earworm female, and the resulting stimulation of acetyl-CoA carboxylase of the pheromone biosynthetic pathway.
ropeptides having the common C-terminal pentapeptide motif (FXPRLamide) that is needed for pheromonotropic activity and is common with insect pyrokinin and myotropin peptide families. It is not clear if all five peptides are involved in the control of pheromone production, but one of them has been identified as the egg diapause hormone in Bombyx mori (32).
One additional factor that comes into play in the overall chemistry of the communication system relates to chemical signals from host plants that can override the photoperiodic control of phermone production. With the corn earworm, it was found that a volatile chemical signal from corn silk, probably ethylene, was required by wild insects for stimulation of pheromone production (33). This signal probably interacts with controls on the photoperiodic release of PBAN.
PHEROMONE PERCEPTION
Peripheral Detection
Insects can detect just a few hundred molecules of their sex pheromone in the environment with their highly efficient antennae, which can contain
several hundred thousand receptor cells. A more detailed description of this highly efficient olfactory system will be presented by Hildebrand (34). The chemistry of perception, however, is just starting to be unraveled and still presents some interesting challenges. At the antennal level, this includes pheromone binding, transport to specific receptor sites, catabolism, and the process of signal transduction.
Chemistry responsible for the great specificity of insect pheromone receptors for single compounds is still unknown, but research has shown the involvement of special binding proteins in the receiving insect's antennae for pheromone transportation and sensillar enzymes for catabolism (35). Evidence also is accumulating that the chemical signals are transduced into intracellular second messenger responses via G-proteincoupled reaction cascades (36), involving phospholipase C, inositol trisphosphate, and the calcium channel. Additional data suggests that the cGMP cascade is involved secondarily in adaptation and fine tuning of the sensitivity of receptor cells (37, 38).
Plume Structure
The chemistry of signal transduction is important in the overall communication system, and a key factor in this process relative to obtaining an appropriate behavioral response is the frequency at which the pheromone components interact with the receptor cells. This frequency depends to a great extent on the release rate of the signal chemicals and the plume structure that carries them to the receiving insect. With moths, it has been found that the pheromone chemicals do not actually attract a mate by means of a chemical gradient, but rather the correct blend and abundance of pheromone molecules in a fluctuating plume will turn on two programs, optomotor anemotaxis and self-steered counterturning, in flying male moths and mediate flight upwind to the odor source (39).
Detection of pheromone, thus, turns on a visually guided motor program that produces upwind surges by the male moth, and when the signal is lost, the male stops upwind flight and the counterturning program causes it to cast sideways back and forth until the chemical signals are once again detected. The fluctuating plume structure is an integral part of the signal since the antennal receptors need intermittent stimulation to sustain upwind flight. The male moth will not fly upwind with continuous stimulation from a uniform plume, and high concentrations of pheromone that bombard the antennal receptors with plume filaments at a high frequency will cause the male to undergo arrestment of upwind flight. Thus, the patterning of the pheromone is as important as the composition.
Behavioral Thresholds
The processing of pheromone quality continues in the central nervous system (see ref. 34), involving the integration of odor input with visual stimuli and finally with messages sent to motor neurons for the appropriate behavioral responses. However, the chemistry involved in the brain is more complex than the processing of signals. Typically, the receiving insect becomes ''receptive" to the sex pheromones at a specific time when the emitting insect is "calling." Although photoperiod cues and temperature are important in the diel periodicity of this response, it has been shown that biogenic amines also are involved (40).
Initially, it was found that injections of octopamine greatly enhanced a male's sensitivity to the chemical signal and that serotonin expanded the period of time in the scotophase during which males responded to pheromone. Recently, studies involving analyses of biogenic amines in various brain tissues showed that there was a significant correlation between decreased levels of octopamine in the brain in scotophase and the probability of a male to respond to low doses of pheromone in the flight tunnel (41). Decreased levels of octopamine also were correlated with increased levels of N-acetyloctopamine, suggesting that octopamine was being utilized in these tissues during that time period. Thus, processing and integration of the chemical signal in the central nervous system involves not only the neural pathways but also the endogenous influence of octopamine as it exerts a modulatory effect on the male's sensitivity to the signal.
Brain Black Box
Beyond the chemistry of the communication system, as described above, is an unknown and speculative factor that operates in the brain of male moths. It is a factor that seems to play a role in determining what is an appropriate signal for that male and what is not. The primary evidence for this factor comes from research with the European corn borer moth. Data show that a sex-linked gene(s) determines whether the male responds to the 97:3 Z/E pheromone blend of one race or to the 1:99 Z/E pheromone blend of another race (42, 43). The product of this gene(s) appears to function in the central nervous system and is not linked with the autosomal factor whose product effects different electrophysiological responses to pheromone components in antennal olfactory cells or with the autosomal factor controlling pheromone blend composition. These genetic studies involving various crosses and backcrosses of E- and Z-pheromone parents show that male moths will respond behaviorally as determined by the sex-linked factor, regardless of what antennal type they possess.
Additionally, an undescribed brain factor could be involved in the evolution of pheromone blends as input from particular pheromone components is switched from being "good" to one that elicits an antagonistic response from the male. Some evidence for this is when highly specific antennal receptors are found in the male's olfactory hairs that respond to a behaviorally antagonistic compound, and the special biosynthetic precursors for this component still are found in the female sex pheromone gland as evidence that at one time the component could have been part of the pheromone blend. An example is with the European corn borer. The male antennae have olfactory cells for the E11- and Z11-14:OAc pheromone components, but also exhibit one specialized cell that responds only to Z9-14:OAc—a compound that is antagonistic to the behavioral responses to pheromone (44). Also, the corn borer female pheromone gland contains large amounts of Z11-16:acid, which is a precursor for Z9-14:OAc, a compound that apparently no longer is produced or has a function in the pheromone system.
Many pheromone systems are finely tuned to a specific blend of components. Although some of the specificity can be effected by blend-specific sensory cells and other attributes of the neural system, there are still many questions related to the perception of pheromone blends. Included are questions not only on how the male changes its response to pheromone components produced by the female over time and how antagonists evolved but also on how a male can respond to a wider range of blend ratios under different environmental conditions (e.g., higher temperatures), how a male handles redundant pheromone signals, and how males might have changed their behavioral thresholds to higher or lower ones over time and in allopatry.
The chemistry of sex attraction, thus, presents challenges not only among the diverse animal species but also within each communication system. Some of the chemistry has been defined, as described above, but knowledge of the chemistry in a few insect species pales in view of the vast number of frontiers yet to be attacked in other animal species. Similarities will, no doubt, be found in the communication systems of other animals, but the diversity of nature predicts that there are still many surprises to be discovered by tomorrow's chemists.
SUMMARY
The chemical communication system used to attract mates involves not only the overt chemical signals but also indirectly a great deal of chemistry in the emitter and receiver. As an example, in emitting female moths, this includes enzymes (and cofactors, mRNA, genes) of the pheromone biosynthetic pathways, hormones (and genes) involved in
controlling pheromone production, receptors and second messengers for the hormones, and host plant cues that control release of the hormone. In receiving male moths, this includes the chemistry of pheromone transportation in antennal olfactory hairs (binding proteins and sensillar esterases) and the chemistry of signal transduction, which includes specific dendritic pheromone receptors and a rapid inositol triphosphate second messenger signal. A fluctuating plume structure is an integral part of the signal since the antennal receptors need intermittent stimulation to sustain upwind flight. Input from the hundreds of thousands of sensory cells is processed and integrated with other modalities in the central nervous system, but many unknown factors modulate the information before it is fed to motor neurons for behavioral responses. An unknown brain control center for pheromone perception is discussed relative to data from behavioral-threshold studies showing modulation by biogenic amines, such as octopamine and serotonin, from genetic studies on pheromone discrimination, and from behavioral and electrophysiological studies with behavioral antagonists.
I thank Drs. Russell Jurenka and Charles Linn for their assistance in preparing this manuscript and for their role, along with many other excellent scientists in my laboratory, for conducting much of the research discussed in this paper. Recent research has been sponsored by National Science Foundation Grants IBN-9108743 and IBN-9017793, National Institutes of Health Grant AI-32498, and a grant from the Cornell Center for Advanced Technology in Biotechnology.
REFERENCES
1. Lintner, J. A. (1882) West. N. Y. Hortic. Soc. Proc. 27, 52-66.
2. Mayer, M. S. & McLaughlin, J. R. (1991) Handbook of Insect Pheromones and Sex Attractants (CRC, Boca Raton, FL).
3. Butenandt, A., Beckman, R., Stamm, D. & Hecker, E. (1959) Z. Naturforsch. B 14, 283-284.
4. Schneider, D. (1969) Science 163, 1031-1037.
5. Schneider, D. (1992) Naturwissenschaften 79, 241-250.
6. Roelofs, W. L. (1984) in Techniques in Pheromone Research, eds. Hummel, H. E. & Miller, T. A. (Springer, New York), pp. 131-159.
7. Charlton, R. E., Webster, F. X., Zhang, A., Schal, C., Liang, D., Sreng, I. & Roelofs, W. L. (1993) Proc. Natl. Acad. Sci. USA 90, 10202-10205.
8. Roelofs, W. L., Comeau, A., Hill, A. & Milicevic, G. (1971) Science 174, 297-299.
9. Persoons, C. J., Verwiel, P. E. J., Ritter, F. J., Talman, E., Nooyen, P. E. J. & Nooen, W. J. (1976) Tetrahedron Lett. 24, 2055-2058.
10. Silverstein, R. M. (1979) in Chemical Ecology: Odour Communication in Animals, ed. Ritter, F. J. (Elsevier/North-Holland, Amsterdam), pp. 133-146.
11. Lanier, G. N., Classon, A., Stewart, T., Piston, J. J. & Silverstein, R. M. (1980) J. Chem. Ecol. 6, 677-687.
12. Bjostad, L. B., Wolf, W. A. & Roelofs, W. L. (1987) in Pheromone Biochemistry, eds. Prestwich, G. D. & Blomquist, G. J. (Academic, New York), pp. 77-120.
13. Jeffcoat, R. (1979) Essays Biochem. 15, 1-36.
14. Thiede, M. A., Ozols, J. & Strittmatter, P. (1986) J. Biol. Chem. 261, 13230-13235.
15. Ntambi, J. M., Buhrow, S. A., Kaestner, K. H., Christy, R. J., Sibley, E., Kelly, T. J., Jr., & Lane, M. D. (1988) J. Biol. Chem. 263, 17291-17300.
16. Kaestner, K. H., Ntambi, J. M., Kelly, J., T. J. & Lane, M. D. (1989) J. Biol. Chem. 264, 14755-14761.
17. Stukey, J. E., McDonough, V. M. & Martin, C. E. (1989) J. Biol. Chem. 264, 16537-16544.
18. Prestwich, G. D. & Blomquist, G. J. (1987) Pheromone Biochemistry (Academic, New York).
19. Raina, A. K. & Klun, J. A. (1984) Science 225, 531-533.
20. Raina, A. K., Jaffe, H., Kempe, T. G., Keim, P., Blacher, R. W., Fales, H. M., Riley, C. T., Klun, J. A., Ridgway, R. L. & Hayes, D. K. (1989) Science 244, 796-798.
21. Kitamura, A., Nagasawa, H., Kataoka, H., Inoue, T., Matsumoto, S., Ando, T. & Suzuki, A. (1989) Biochem. Biophys. Res. Commun. 163 , 520-526.
22. Kitamura, A., Nagasawa, H., Kataoka, H., Ando, T. & Suzuki, A. (1990) Agric. Biol. Chem. Tokyo 54, 2495-2497.
23. Masler, E. P., Raina, A. K., Wagner, R. M. & Kochansky, J. P. (1994) Insect Biochem. Mol. Biol. 24, 829-836.
24. Jurenka, R. A., Fabriás, G. & Roelofs, W. L. (1991) Insect Biochem. 21, 81-89.
25. Jurenka, R. A. & Roelofs, W. L. (1993) in Insect Lipids: Chemistry, Biochemistry and Biology, eds. Stanley-Samuelson, D. W. & Nelson, D. R. (Univ. of Nebraska Press, Lincoln), pp. 353-388.
26. Jurenka, R. A., Jacquin, E. & Roelofs, W. L. (1991) Arch. Insect Biochem. Physiol. 17, 81-91.
27. Jacquin, E., Jurenka, R. A., Ljungberg, H., Nagnan, P., Löfstedt, C., Descoins, C. & Roelofs, W. L. (1993) Insect Biochem. Mol. Bol. 24, 203-211.
28. Martinez, T., Fabriás, G. & Camps, F. (1990) J. Biol. Chem. 265 , 1381-1387.
29. Arima, R., Takahara, K., Kadoshima, T., Nagasawa, H., Kitamura, A. & Suzuki, A. (1991) Appl. Ent. Zool. 26, 137-147.
30. Kawano, T., Kataoka, H., Nagasawa, H., Isogai, A. & Suzuki, A. (1992) Biochem. Biophys. Res. Commun. 189, 221-226.
31. Ma, P. W. K., Knipple, D. C. & Roelofs, W. L. (1994) Proc. Natl. Acad. Sci. USA 91, 6506-6510.
32. Sato, Y., Nakazawa, Y., Menjo, N., Imai, K., Komiya, T., Saito, H., Shin, M., Ikeda, M., Sakakibara, K., Isobe, M. & Yamashita, O. (1992) Proc. Jpn. Acad. Ser. B 68, 75-79.
33. Raina, A. K., Kingan, T. G. & Mattoo, A. K. (1992) Science 255 , 592-594.
34. Hildebrand, J. G. (1995) Proc. Natl. Acad. Sci. USA 92, 67-74.
35. Vogt, R. G. (1991) in Pheromone Biochemistry, eds. Prestwich, G. D. & Blomquist, G. J. (Academic, New York), pp. 385-431.
36. Stengl, M., Hatt, H. & Breer, H. (1992) Annu. Rev. Physiol. 54, 665-681.
37. Boekhoff, I., Seifert, E., Göggerle, S., Lindemann, M., Krüger, B.-W. & Breer, H. (1993) Insect Biochem. Mol. Biol. 23, 757-762.
38. Kaissling, K.-E. & Boekhoff, I. (1993) in Sensory Systems of Arthropods, eds. Wiese, K., Gribakin, F. G., Popov, A. V. & Renninger, G. (Birkhaeuser, Basel), pp. 489-502.
39. Vickers, N. J. & Baker, T. C. (1994) Proc. Natl. Acad. Sci. USA 91, 5756-5760.
40. Roelofs, W. L. & Linn, C. E., Jr. (1987) J. Cell Biochem. Suppl. 10, 54.
41. Linn, C. E., Jr., Campbell, M. G., Poole, K. R. & Roelofs, W. L. (1994) Comp. Biochem. Physiol. 1086, 87-98.
42. Roelofs, W. L., Glover, T. J., Tang, X.-H., Sreng, I., Robbins, P., Eckenrode, C. J., Lofstedt, C., Hansson, B. S. & Bengtsson, B. O. (1987) Proc. Natl. Acad. Sci. USA 84, 7585-7589.
43. Glover, T. J., Campbell, M. G., Robbins, P. S. & Roelofs, W. L. (1990) Arch. Insect Biochem. Physiol. 15, 67-77.
44. Glover, T. J., Perez, N. & Roelofs, W. L. (1989) J. Chem. Ecol. 15, 863-873.
















