Page 23
Chapter 3
X-Ray Computed Tomography
3.1 Introduction
3.1.1 History
Computed tomography (CT) was the first non-invasive radiological method allowing the generation of tomographic images of every part of the human body without superimposition of adjacent structures. Even more importantly, a contrast resolution could be achieved that for the first time in radiology permitted the differentiation of soft tissue inside the highly attenuating skull. The technological improvements in the first 20 years of CT can best be characterized by a comparison of typical scan parameters from 1972 and 1994, as shown in Table 3.1.
The greatest progress has been made in reducing scan times and in improving spatial resolution. Further increasing the contrast resolution will require an increase in the patient dose, since the efficiency of the detector systems is already close to its theoretical limits. An improvement in the transaxial or planar spatial resolution is sensible only if the resolution as measured along the scanner axis (i.e., the slice thickness) is improved simultaneously. With today's typical scan parameters there is a discrepancy between the planar resolution, which is less than 1 mm, and the slice thickness, which customarily is several millimeters. With the recent introduction of spiral CT (section 3.3), x-ray CT, like other imaging modalities, has started to move from a slice-by-slice to a volume imaging method, with more isotropic spatial resolution in all three dimensions allowing a better
Page 24
Table 3.1 Technological Improvements in the First 20 Years of Computed Tomography
| ||||||||||||||||||||||||
three-dimensional representation of anatomical structures.
3.1.2 Principle of Operation
The attenuation of monochromatic x-rays in homogeneous objects is governed by
![]()
where I is the x-ray intensity behind the object, I 0 is the x-ray intensity without the object, x is the length of the x-ray path through the object, and a is the linear attenuation coefficient of the material for the x-ray energy employed.
For inhomogeneous objects like the human body, the attenuation of x-rays consequently can be described by
![]()
This model is explored further in section 14.1.1.
In CT scanners the x-ray attenuation according to equation 3.2 is measured along a variety of lines within a plane perpendicular to the long axis of the patient with the goal of reconstructing a map of the attenuation coefficients a for this plane. The resulting attenuation coefficients are usually expressed with reference to water:
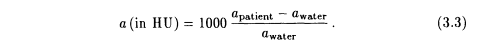
Page 25
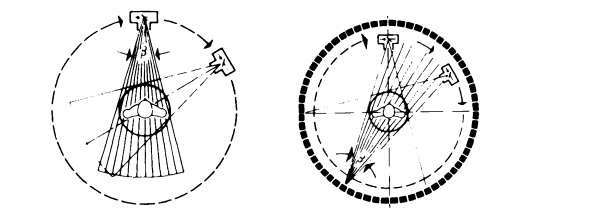
Figure 3.1. Scanning principles of computed tomography systems. Left: Fan beam systems employ a multicellular detector system rotating about the patient together with the x-ray tube. Right: Ring detector based systems have a fixed detector ring, operate with a fan-shaped x-ray beam, and require only a rotational movement of the x-ray tube or the x-ray focal spot in the case of electron beam type x-ray generation. (Reprinted with permission from E. Krestel, ed., Imaging Systems for Medical Diagnosis, Siemens Aktiengesellschaft, Berlin and Munich, 1990. Copyright Publicis MCD Werbeagentur GmbH (Siemens AG), 1990.)
Equations 3.1 and 3.2 ignore the fact that the spectrum of the x-rays entering the patient is generally non-monochromatic. It turns out that the resulting ''beam hardening artifacts" can be corrected to a sufficient degree by a simple nonlinear correction to the measured line integrals.1
Figure 3.1 shows the two principal measurement geometries used in today's CT scanners. In fan beam systems (Fig. 3.1, left) a multicellular linear detector array and the x-ray tube are mounted on the same carrier, which rotates around the patient. A fan-shaped x-ray beam is utilized to successively measure the x-ray attenuation across the patient for the required number of projection angles. The image then can be reconstructed either after rearranging the central projections into parallel projections with parallel beam reconstruction techniques, or more directly by using the central projections with a modified fan beam reconstruction algorithm. A variant of the fan beam system employs a number of x-ray tubes and detector arrays rotating around the patient with the x-ray focal spots shining through the gaps be-
1 Beam hardening is the effect of different attenuations of the different parts of the x-ray spectrum when the x-ray passes through an object.
Page 26
tween the detector arrays. This configuration allows for a shorter exposure time since the system has to be rotated only through a small angle rather than 180° or more in order to obtain a complete data set for image reconstruction. However, because of the very high cost involved and because of the improved speed of slip ring or electron beam based systems, this variant of the conventional fan beam system has not gained wide distribution.
Ring detector based systems (Fig. 3.1, right) use a complete stationary ring of detector elements surrounding the patient port and move only the x-ray tube—or, in the case of electron beam systems, the focal spot—around the patient. The measurement and reconstruction geometry also employs a fan of x-rays through the patient in the same way as with fan beam systems. However, particularly in electron beam based systems, it is often difficult to move the x-ray focal spot in the same plane that is occupied by the ring detector. In this case the focal spot has to follow orbit in a plane adjacent to the detector system, which represents an imperfection in the measurement geometry and can lead to typical artifacts (see also section 3.2.3).
3.2 Present Status of CT Instrumentation and Technology
3.2.1 X-Ray Tubes
The dramatic increase in the performance of conventional-i.e., non-electron-beam based-CT scanners over the past 20 years has required significant improvements in the technology of the x-ray tubes used. Today, typically, a metal (tungsten-rhenium-molybdenum) target is brazed to a thick graphite block for improved heat dissipation. Target diameters of up to 20 cm are used with rotational speeds of up to 10,000 rpm. Typically the target is operated at a temperature of 1200°C and the area around the focal spot reaches over 2500°C. The maximum heat dissipation of these tubes can reach up to 10 kW. Still, the major limitation in the use of CT scanners imposed by the x-ray tube is the maximum permissible sustained x-ray power. A further increase will require major developments in target cooling (e.g., liquid cooled-rotating targets), bearing systems, and generation of the electron beam.
3.2.2 Detector Systems
Figure 3.2 gives an overview of the two types of detector systems employed with CT. Both types of systems are usually arranged in linear arrays with up
Page 27
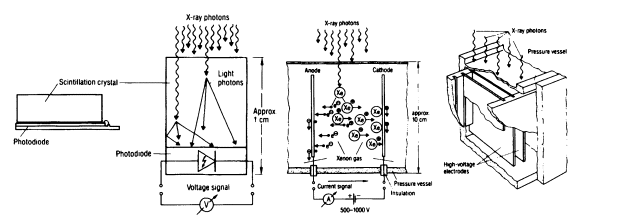
Figure 3.2. CT detectors. Left: Scintillation crystal with photo-sensitive semiconductor detector. Right: Rare-gas ionization chamber detector. (Reprinted with permission from E. Krestel, ed., Imaging Systems for Medical Diagnosis, Siemens Aktiengesellschaft, Berlin and Munich, 1990. Copyright Publicis MCD Werbeagentur GmbH (Siemens AG), 1990.)
to about 1,000 detector elements or as rings with even more elements in the case of systems with a geometry as shown on the right in Figure 3.1. At least one commercial system uses two adjacent rows of detector elements in order to scan two slices simultaneously. For scintillation based detectors the most commonly used scintillator materials have been cesium iodide (CsI) and cadmium tungstate, as well as ceramic scintillators employing rare-earth oxides and oxysulfides. A particularly critical parameter for CT detector scintillators is their afterglow behavior. Since CT data acquisition systems do not use pulse counting as in most nuclear applications, but rather integrate the detector current, the afterglow leads to measurement errors near tissue/air and tissue/bone interfaces. Data correction schemes have been developed to at least compensate for first-order effects related to the detector afterglow. Compared to the rare-gas ionization detector, the scintillation detector usually has a lower geometric efficiency (ratio of active versus ineffective frontal area), which is, however, generally more than compensated for by the latter's higher quantum efficiency.2 In fan beam systems a collimator is usually employed in front of the scintillator elements to reduce the effects of scat-
2The quantum efficiency is a measure of detector (material) sensitivity related to its ability to interact with a certain fraction of the incoming x-ray or y-ray quanta. The quantum efficiency is related to the linear attenuation coefficient.
Page 28
tered x-rays. In gas detector arrays the chamber walls effectively serve the same purpose by acting as a collimator aligned with the position of the x-ray focal spot. This self-collimating effect of gas detectors, on the other hand, excludes their application in ring detector based systems, since the focal spot of the x-ray tube moves relative to the detector elements.
3.2.3 Image Artifacts
Image imperfections or artifacts observed in CT systems can be classified according to their origin:
1. Physics-related causes
· Quantum noise: Physically unavoidable statistical fluctuations in the x-ray beam intensity will show up as signal-dependent noise in the projection data. A reduction of the noise level requires an increase in x-ray intensity or data acquisition time, along with an increase in patient dose. The signal dependency of the quantum noise leads to typical radial noise structures in the reconstructed images.
· X-ray scattering by the patient: In fan beam type systems it is possible to use detectors that are sufficiently collimating to reduce the effect of scatter adequately. In detector ring based systems, however, mathematical scatter models have to be used to correct for this effect.
· Beam hardening: Simple empirical corrections are usually sufficient to compensate for this effect for all diagnostic purposes. In particular situations such as when the amount of bone in a particular slice is very large, residual beam hardening effects (e.g., the dark bands typically seen between the petrosal bones in head scans) may show up.
· Nonlinear partial volume effects: As a consequence of the exponential, thus nonlinear, attenuation of radiation when high-contrast structures are immersed only partially in the imaged slice, the measured data become inconsistent with the mathematical model used for reconstruction. Although correction methods have been devised, they have not been widely accepted owing to the associated increase in expense, measurement time, and complexity. The simplest and most effective way to reduce nonlinear partial volume effects is to reduce the slice thickness.
Page 29
2. Patient-related effects
· Movement of the patient or of individual organs: Typical motion artifacts will show up as stripes or groups of stripes and streaks. The intensity will depend on the contrast of the moving structures. Although in the images these artifacts can usually be easily identified as motion related, the combination of several artifacts can make it considerably more difficult to interpret the images. The most effective way to reduce motion artifacts, of course, is to speed up the data acquisition time accordingly.
· Metallic foreign objects: Due to the strong attenuation of metal (for example, in dental fillings), the dynamic range of the detector and data acquisition system is often not large enough to accurately accommodate the measurement of the projection data. To make things worse, in such a situation nonlinear partial volume effects usually amplify inconsistencies in the projection data. As a consequence the images show streaking artifacts originating from the metallic objects.
3. System-related causes
· Insufficient calibration of detector sensitivity
· Time-, temperature-, or irradiation-history-dependent drift in detector sensitivity
· Inadequacies in the reconstruction algorithm
· Non-uniform scanning motion
· Fluctuations in x-ray tube voltage
All of the above errors will show up in the reconstructed images with different typical patterns that vary further with the scanning geometry employed. For example, detector-related problems in fan beam systems usually manifest themselves as ring-shaped artifacts, whereas in systems based on stationary ring detectors they will have the character of streaks or stripes.
3.2.4 Quantitative CT
Although in the overwhelming majority of all examinations the resulting images are inspected only visually and qualitatively by the radiologist providing the diagnosis, a few special applications require absolute evaluation of the attenuation coefficients. These quantitative CT applications have a fairly
Page 30
long tradition, particularly in the case of bone mineral density measurements for diagnosis and therapeutic followup of osteoporosis. Here the measured attenuation coefficients or "Hounsfield numbers" are usually compared with simultaneous measurements made on calibrated, calcium-containing inserts. By using inserts with calcium content similar to that of the vertebra to be investigated, patient-dependent beam hardening effects can be compensated for, at least to a first-order approximation. A further refinement requires measuring attenuation data with two different x-ray spectra by changing the x-ray tube voltage. By using these so-called dual-energy scans it is possible to distinguish the individual contributions of calcium and waterlike tissue to the total measured attenuation coefficient, and thus better determine the bone mineral content of the vertebra.
There are a number of less customary quantitative CT procedures such as measurement of tissue perfusion with dynamic CT or xenon CT.3 Measurement of lung density represented an area of great interest very early on but did not become feasible in practice until spirometric (related to a measurement of the respiratory cycle) control of the scan and suitable automated evaluation methods were available. Apart from documenting findings objectively, as is frequently necessary in occupational diseases, the primary hope is to be able to detect diseases sensitively and early by selective analysis of different areas, e.g., by automated evaluation of the peripheral parts of the lungs.
3.2.5 Requirements for High-Speed CT
Most routine applications of CT ask for the best possible image acquired within the shortest possible time. The requirement for short acquisition times comes both from the necessity to effectively freeze the motion of organs like the heart and from the desire to cover large areas of the patient in the shortest possible time to improve patient comfort and patient throughput. To avoid the relatively greater effects of quantum noise associated with shorter scan times, the most critical and technically most demanding requirement is improvement to both instantaneous and sustained x-ray power from the tube or electron beam system.
3Dynamic CT is the acquisition of sequential images from the same anatomical area. Xenon CT measures the attenuation of certain tissue before and after the inhalation of xenon gas, which is dissolved in the blood stream, and thus allows one to quantitate brain perfusion.
Page 31
3.3 Spiral CT
With the introduction of slip ring based scanners in the mid-1980s, continuous-volume data acquisitions became possible. Such imaging is achieved by moving the patient slowly through the gantry while continuously acquiring data. The resulting helical movement of the x-ray tube focal spot relative to the patient allows rapid volumetric data acquisition over large areas of the body. The data interpolation schemes developed for spiral CT permit the reconstruction of images with arbitrary position and spacing, including overlapping reconstruction for improved three-dimensional displays. Further, the improvement in rapidly covering larger volumes in spiral CT allows a better and more comfortable use of breath-holding techniques for improved imaging of internal organs that are susceptible to respiratory motion. Thus spiral CT, in particular, has improved the assessment of pulmonary nodules, has significantly increased the quality of three-dimensional surface-rendering type image representations, and-with the use of contrast agents-for the first time has allowed the display of vascular structures with previously unachievable quality.
The main limitation in spiral CT image quality today still is due to the available sustained power from the x-ray tube. While 40 kW are available typically for single scans, the power has to be reduced for spiral scans of 30 seconds and more to avoid overloading of the tube. As a result of the associated increase in quantum noise, soft tissue images frequently have to be reconstructed with smoothing convolution kernels. On the other hand, because of the high contrast of bone structures, the full spatial resolution can be made available for orthopedic examinations employing thin slice width settings. Blurring of the images in the direction of the patient's movement through the gantry can be minimized by appropriate choices for the table motion, and by reconstructing images for every 180° of scanner rotation rather than using the customary 360º scan coverage. Depending on the specific imaging situation, table feed values of one to two times the slice thickness per 360º of scanner rotation are being used today.
Even from the limited clinical experience with spiral CT available today, it is fairly obvious that this scanning mode will become the standard technique by the year 2000. Single-slice scanning will then be the exception. Required technical improvements will have to come in the available sustained x-ray power, as well as in the advances in computation time expected through increased processor performance. Another important impact of future technological developments will come from the use of multislice de-
Page 32
tectors or planar detector arrays extending over a larger solid angle, thus allowing better use of the available x-ray power and covering larger volumes within a given time. Such detectors are considered a most important future development. Depending on the technology employed, these detectors possibly will also allow improvement of the spatial resolution in the scanner axis direction.
3.4 Electron Beam Techniques
With conventional techniques, scan times cannot be shortened arbitrarily, because considerable masses have to be accelerated and moved, and because higher continuous x-ray power is difficult to achieve with the current rotating anode tubes. A technical alternative to overcome these problems and facilitate subsecond scanning is the use of electron beam techniques to replace conventional x-ray tubes. With electron beam systems the patient is surrounded by stationary anode rings inside a cone-shaped vacuum chamber. An electron beam is accelerated to about 130 or 140 keV and electromagnetically focused and steered onto the stationary anode rings, which can be cooled directly with an appropriate cooling agent. The electron beam can be rapidly scanned over the target rings, allowing scan times of 50 to 100 ms per slice. Using EKG triggering—synchronizing the data acquisition with the patient's heart beat by deriving a trigger from an electrocardiogram (EKG) signal—these short scan times allow the acquisition of unblurred images at a specific cardiac phase. Respiratory gating can be similarly used for longer studies, including the diagnosis of the great vessels. Switching between multiple target rings combined with conventional table feed permits fast and efficient volume acquisition.
The most promising and fastest-growing application of ultrafast electron beam based CT systems is coronary artery screening for early detection and quantitation of the calcification associated with arteriosclerosis. This procedure requires no contrast agents. Customarily, 20 to 40 3-mm-thick EKG-triggered slices are acquired at a rate of one at every heartbeat to cover a 6- to 12-cm volume through the coronary arteries. In chest imaging, electron beam CT allows covering the entire volume of the lungs within one breath-holding interval. This technique enables more accurate diagnosis of such conditions as pulmonary nodules, pulmonary embolism, and diffuse pulmonary emphysema than can be obtained from plain chest x-rays or slower CT techniques.
Page 33
For the current generation of electron beam CT systems, image reconstruction still requires significantly more time than does image acquisition. Further research into faster filtered backprojection implementations or the increased use of Fourier domain-based reconstruction methods, along with an increase in fairly inexpensive processor speed, should help to close this obvious gap in the near future. Similarly, as with conventional CT scanners, the development of advanced detector array technologies will improve the spatial sampling of electron beam type systems. The spatial separation of the target rings and detectors along the scanner axis leads to "cone beam" errors that can show up as rib shadows. Volumetric imaging and reconstruction techniques are being developed to overcome this problem.
3.5 Data Handling and Display Techniques
Improvements in data acquisition speed and spatial resolution in both conventional and electron beam CT inevitably will lead to a vast increase in the amount of data collected and the number of images reconstructed. Data reduction techniques have to be developed to allow physicians to extract and display the information that is relevant out of the vast amount of three-dimensional volume data. A fairly straightforward method is to reduce a large number of transverse, tomographic slices into a few three-dimensional views of the structures and organ boundaries considered important. A critical prerequisite for generating three-dimensional views quickly and efficiently is the development of supervised semiautomatic segmentation techniques to routinely identify organ boundaries for the three-dimensional representation. The resulting three-dimensional models are used increasingly in surgical planning and, even more directly, in CT-guided interventional techniques. Particularly for surgery that follows complex trauma, deals with congenital abnormalities, or reconstructs after tumor resection, accurate three-dimensional models of the anatomy are providing the detailed information required for accurate surgical planning as well as serving as a reference during the surgery itself. A particular application of three-dimensional volume techniques in CT that recently attracted a large amount of interest is the assessment of vascular structures with CT angiography. Three-dimensional volume images are acquired using a spiral CT scan synchronously triggered with contrast agent injection. For data reduction and display, two methods have been applied so far. A surface-rendering technique based on threshold type segmentation of the images is used predominantly for the display
Page 34
of complex vascular structures involving large vessels such as in the thorax. Alternatively, maximum intensity projection-a volume-rendering technique that generates projection images from the highest intensity pixels along the projection rays-of the image data allows a good visualization of smaller vascularity as well as vascular wall calcifications. Both of these projection techniques allow the retrospective generation of views at arbitrary angles, a capability that is not possible with conventional angiography. Applications of CT angiography established to date range from the diagnosis of renal artery stenosis to examinations of complex vascular abnormalities and aortic aneurysms.
3.6 Research Opportunities
To summarize, the following areas of scientific or technological development appear to have the greatest potential for enhancing the applications and clinical usefulness of x-ray CT:
· Increased instantaneous and, even more importantly, sustained x-ray power capabilities in both conventional x-ray tubes and electron beam systems;
· New and innovative technologies for the required high-intensity x-ray source, beyond those currently available;
· Two-dimensional detector arrays encompassing a larger solid angle and allowing improved spatial resolution along the scanner axis, including the associated high-throughput data acquisition electronics;
· Decrease in image reconstruction times, for example, through higher-performance low-cost processors and the increased use of Fourier domain based reconstruction algorithms;
· Mathematical means for utilizing the information contained in scattered photons;
· Effective means for the correction of patient motion-related artifacts;
· Better and easier-to-use three-dimensional data reduction and visualization techniques;
· Monochromatic x-ray generators, a major challenge; and
Page 35
· Detectors that provide information on angles of incidence and high energy resolution, perhaps requiring new ideas from physicists and mathematical scientists.
3.7 Suggested Reading
1. Boyd, D., Couch, J., Napel, S., Peschmann, K., and Rand, R., Ultrafast cine-CT for cardiac imaging, Am. J. Cardiac Imaging 1 (1987), 175-185.
2. Brundage, B., Detrano, R., and Wong, N., Ultrafast computed tomography: Imaging of coronary calcium in atherosclerosis, Am. J. Cardiac Imaging 6 (1992), 340-345.
3. Kalender, W.A., Quo vadis CT? CT in the year 2000, Electromedica 61 (1993), 30-39.
4. Kalender, W.A., and Polacin, A., Physical performance characteristics of spiral CT scanning, Med. Phys. 18 (1991), 910-915.
5. Krestel, E. (ed.), Imaging Systems for Medical Diagnosis, Siemens Aktiengesellschaft, Berlin and Munich, 1990.
6. Polacin, A., Kalender, W.A., and Marchal, G., Evaluation of section sensitivity profiles and image noise in spiral CT, Radiology 185 (1992), 29-35.
7. Rubin, G., Dake, M., Napel, S., McDonnell, C., and Jeffrey, B., Threedimensional spiral CT angiography of the abdomen: Initial clinical experience, Radiology 186 (1993), 147-152.
8. Schäfer, C., Prokop, M., Nischelsky, J., Reimer, P., Bonk, K., and Galanski, M., Vascular imaging with spiral CT, in Advances in CT II, R. Felix and M. Langer, eds., Springer-Verlag, Berlin, Heidelberg (1992), 109-115.
9. Schiemann, T., Bomans, M., Tiede, U., Hohne, K.H., Interactive 3Dsegmentation, Proc. SPIE 1808 (1992), 376-383.
10. Stanford, W., and Rumberger, J.A. (eds.), Ultrafast Computed Tomography in Cardiac Imaging: Principles and Practice, Futura Publishing Co., Inc., Mount Kisco, N.Y., 1992.














