New Knowledge for Health
Baruch S. Blumberg, M.D., Ph.D.
Fox Chase Distinguished Scientist, Fox Chase Cancer Center
The assignment given to me is to discuss new knowledge related to health in the 21st century. This is a very broad topic and I have decided to consider one aspect of it, genetic susceptibility to disease and the maintenance of good health.
The genome project is on schedule and it is likely that all, or nearly all, of the human genome will have been identified and sequenced early in the next millennium. Francis C. Collins, director of the National Center for Human Genome Research, has estimated its completion in 2002 or 2003. Many genes that "cause" inherited diseases have been identified. In addition, a large number of susceptibility genes are already known and many more will be identified in the future. Susceptibility genes affect the probability of incurring an illness and are often associated with exposure to an agent in the environment. For example, susceptibility genes for several cancers have been identified; BRCA 1 and BRCA 2 for cancer of the breast, AT for cancer of the breast and other cancers, p53 for a large variety of cancers, and many others.
There are also genetic effects on exogenous and environmental agents that affect the host and are potentially pathogenic. For example, genes have been identified that control the production of enzymes that detoxify carcinogenic or other disease-causing agents. These enzyme systems are often polymorphic, that is, they are controlled by a series of alleles segregating at the same locus, which can impart different levels of activity. Some of the alleles may produce levels of enzyme that detoxify the exogenous agents, while other alleles will control enzymes that are null or are deficient in their ability to detoxify. Hence different individuals will respond very differently to the same level of exposure. Those with increased inherited susceptibility, that is, who have inherited the defective detoxifying enzyme, are at greater risk of becoming ill after exposure to the exogenous agent than those who have inherited an
alternate allele with adequate levels of enzyme. This information can be used to discover exogenous agents that increase risk for a particular disease. The allele that determines the lesser-detoxifying enzyme would occur in higher frequency in those diseases for which the exogenous agent in question increases risk. This technique could focus attention on the elimination of these dangerous elements from the environment. I will provide an example of this phenomenon later in this chapter.
The rapid progress of the Human Genome Project opens the possibility of the identification of potential disease in clinically normal individuals, and its prevention before it becomes symptomatic or life threatening. This raises the prospect of living a disease-free life, until the inevitable demands of the aging process lead to death at an advanced age. Yet this happy goal is achievable only if the pace and sophistication of research are increased.
The notion of preventive medicine at a personal level is in some respects antithetical to the traditional social contract between a physician trained in the Western tradition and his or her patient. The contract reads, in essence: "If you become sick, then I am available for help. If you are not ill, that is someone else's responsibility." It will be interesting to see how this doctor and "nonpatient" relation develops in the coming decades.
I would like to digress a moment from the discussion of prevention and talk briefly about therapeutic medicine. The science and art of medicine are dependent on accurate diagnosis. The physician attempts to sort patients into diagnostic categories or "bins." Once classified, the patient is assigned a prescribed pattern of therapy. Diagnosis will become more specific as the genetic and other etiologies of more and more symptom complexes become known. Instead of treating a patient as a member of a category, each patient will have his or her unique diagnosis and treatment. Oddly, this pattern is similar to the style of some non-Western indigenous medical systems in which individualized treatment is prescribed for each patient.
Currently, there are person-to-person differences in response to treatment, such as age and gender, that are known prior to the initiation of treatment and that influence treatment. With increased knowledge of the genome, more genes that influence response to drugs will be discovered and understood. The science of "pharmacogenetics" will make it possible to design therapy based on prior knowledge of the response expected.
Application of this genetic knowledge has and will raise many ethical questions, which can only be alluded to in this presentation. The organizers of the Genome Project have, wisely, included a study of actual and potential ethical problems as an integral part of the research project. This may be the first organized effort incorporated in a major research program to identify potential ethical issues before, or at the same time, that they arise. It can be said to be an anti-Frankenstein's monster program; a disaster-prevention strategy.
A goal of a program of identifying susceptibility genes would be to advise people at risk of the hazards, in order to mitigate them. The geneticist would also want to advise people who do not have the susceptibility genes that they
are at a lower risk. However, this raises problems. To identify the presence of susceptibility requires the development of appropriate genetic probes that identify the mutations. The genomes are often large, there may be many mutations, some of them rare, and the distribution of the mutations may vary greatly from population to population. Therefore, if one or a small number of genetic tests are used and found to be negative, the individual may still be at risk. Assuring the individual to the contrary could raise medical, public health, legal and ethical problems.
Another ethical problem arises when susceptibility is detected, but there is no available intervention. Does a person want to know of an unhappy fate if prevention or cure is not possible? There are potential questions related to health and life insurance. Insurance companies may want to know who is at genetic risk in order to either exclude them from coverage and shift their care to other organizations, or to charge a higher premium. However, many of the traits we are discussing are genetic polymorphisms, which implies that there are both positive and negative values. Whereas a gene may increase risk for some diseases, it could concur advantage in other circumstances. Polymorphisms are common and any individual may carry a complex combination of advantageous and disadvantageous genes. If he or she is to be penalized for one trait, then fairness requires a search for other traits that may compensate for the disadvantage. If there is a polymorphic locus segregating in a community, then some individuals would be at greater than average risk, and others at less than average risk. Would they be offered a lower premium?
What I believe will happen is that the principle of mutuality, on which insurance premiums are often based, will prevail. A uniform fee will be charged on the understanding that the risk in a large population will even out from person to person and some intermediate risk estimate will lead to an overall economy.
Single-gene therapy has, so far, been limited in its results. This, in large part, is a consequence of technical problems, that is, the delivery of the genes into the host, duration of effect of the introduced gene, and so on. Yet, when these difficulties are surmounted others are likely to remain. Only a relatively small number of diseases are a consequence of single-gene effects, and even in these the repair or replacement of the affected gene may not cure. It is important to keep in mind that genetic control of characteristics and diseases, that is, the phenotype, is complex. Diseases and characteristic are often polygenic, meaning that several genes act on the phenotype. Genes are often pleomorphic, a single gene may have multiple effects. As we have already discussed, many genes are polymorphic, that is, there may be minor sequence differences at the same locus which have profound differences on the resultant protein and on pathology and physiology. Further, posttranslational events can affect function, tropism, and the quality of the protein produced by the gene. In most cases disease is a consequence of complex interactions of many genes and exogenous agents in the environment. Often it will be more expedient to intervene to change the exposure to an environmental agent than to change the genes. I will illustrate this point later.
We are frequently reminded that we live in an information age, and that the storing, distribution, and management of data are as important as its collection. This will be particularly true as knowledge of the complexity of the genome and its protein products increases. Continuing development of software to manage these data will be required before it can be fully utilized. Progress in medical research and applications will be dependent on the establishment of intellectually independent groups operating within a medical or research environment, so that people who are expert in the design and use of the data systems and also thoroughly conversant with biology will be available. As more and more scientists grow up with computers, it will be possible to find these talents within a single person. (Film lovers with an eclectic taste may recall that Buckaroo Banzai was such an individual.) More likely it will require close cooperation between scientists skilled in one or the other of the disciplines.
There will also be an increased need to construct mathematical models of biological systems. As more and more complex systems are considered, multiple genes, multiple products, multiple external agents all interacting will be chaotic if not organized into an understandable model which can then be subjected to experimental and observational testing. The current attempts to understand "complexity" in a wide variety of disciplines is a wholesome move in this direction.
It will be possible to "mine" data bases. This metaphor carries many implications; for example, the probing of data bases to obtain information that one, a priori, did not know was needed. An additional application is likely to come in storing data on individual human genetic "fingerprints" that carries with it ethical and legal problems. It is likely that prediction on the basis of an individual's genome will be far more complex than at appears at present when the identification of all of the genes is incomplete. Yet, there will be a large amount of predictability incorporated in an individual's genome. How will individuals react to knowing their unique fate?
The past few decades have made it apparent that we can expect new plagues and the return of old ones. When cholera first emerged from Asia into Western Europe, it traveled at the speed of a man on a horse or in a sailing ship. Infectious agents, using the vector of jet aircraft, travel at nearly the speed of sound. As the world becomes more integrated, economically, politically, and culturally, we become more exposed to infectious agents from foreign parts. Strains of bacteria, viruses, and other pathogens which, in the past had remained within geographic bounds, now travel as widely and as rapidly as their hosts. That includes pathogens that have developed resistance to antibiotics and vaccines.
In order to effect some measure of protective control over these agents, it will become necessary to establish surveillance stations, often in hitherto remote areas, to determine when new strains develop. Ideally, it would be better to know about the emergence of new pathogenic strains before they cause an epidemic. New strains can be identified by the determination of their gene sequences. Very detailed epidemiology and transmission histories will be
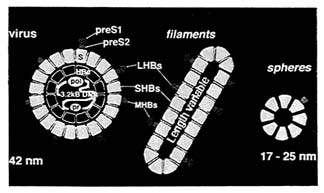
Figure 1 The three particles associated with hepatitis B virus. The large particle contains DNA and polymerase, is infectious, and can replicate. The smaller particles contain only the surface antigen. The locations of preS1 and preS2, Large, Medium, and Small surface antigen proteins (HBs) are indicated.
feasible, since a unique strain from the individual who was first infected could be traced through contacts. It may be possible to determine exactly who transmitted an infection to another.
The management of this vast amount of data and the comparison of sequences to establish homologies and differences, polymorphisms, mutations, interacting genes, and other complex phenomena, will require very sophisticated data base management. New technical applications will be needed to facilitate the transmission of this information between the scene of the mutation to the scientific centers where these data can be collated and decisions on interventions made.
I have raised these issues in a general manner, but I would now like to provide concrete examples from the research with the hepatitis B virus (HBV).
The human hepatitis viruses, of which at least seven (designated HAV to HGV) are now known, are among the most important pathogens that invade humans. Each of them is a different virus, and many come from completely different families and genera, but the clinical outcomes and means of transmission are also similar for many of them. My illustration will apply to HBV, but the same principles may apply to other hepatitis viruses and other diseases.
The three particles of HBV are shown in Figure 1. The large particle is infectious and replicates. The small sphere and the rod do not have DNA and contain only surface antigen. There are about 300 million carriers of HBV in the world. Many of these people carry infectious and replicating virus, and a portion of them are at high risk of developing life-shortening chronic liver disease and hepatocellular cancer (HCC). HCC is one of the most common cancers in the world, particularly in Asia and in sub-Saharan Africa, where there are a very large number of HBV carriers. Fortunately, an effective vaccine, invented in our laboratory in 1969, has been available for more than a decade, and it has been used very effectively in national universal childhood vaccination programs. This has markedly decreased the number of carriers
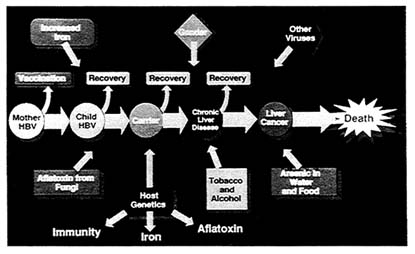
Figure 2 Diagram illustrating the possible courses of chronic HBV infection and factors that influence progression. See the text for further explanation.
among the younger age groups (i.e., up to about 10 years of age) in several places, including Taiwan, Japan, and Korea, but the number of adult carriers who were unprotected in their youth are a major medical burden and will remain so for many decades. Research on treatments of carriers of the virus and those afflicted with the disease is very encouraging, but it is important to increase our understanding of the complexity of infection and pathogenesis.
Figure 2 demonstrates the complexity of the factors that bear on infection and disease. It illustrates the course of chronic hepatitis B infection from the time of transmission in early life until the development of chronic liver disease and hepatocellular carcinoma. In Asia, in particular, children are often infected from their mothers, and this increases the probability of becoming a chronic carrier. Only a portion of those chronically infected proceed to disease and a shortened life span. What are the factors that affect the speed and direction that the infection will take? The diagram illustrates some of these, arranged in no particular order on the periphery of the diagram, and shows the decision nodes where one or another factor, or several, will impel toward ongoing disease or to a different outcome.
Males are more likely to become carriers than are females, and male carriers are more likely to remain carriers and develop chronic liver disease and cancer. There appears to be a genetic predisposition to the carrier state, and the development of hepatocellular carcinoma. This may be related to the genetic control of the human immune system, to the control of other factors, or to as-yet undiscovered genes that directly affect the probability of chronicity.
Environmental factors include elevated total body iron stores, which increases the probability of cancer in those who are chronically infected with HBV. Iron store levels depend in part on dietary iron intake. Elevated levels of arsenic in potable water also appear to increase the probability of HCC.
Tobacco and alcohol use, which are behavioral characteristics, also appear to affect the course of the disease. HBV interacts with other viruses, including the other hepatitis viruses, with HIV, and with malarial infection; all of these can affect the course of the infection.
The detailed interaction of exogenous agents and genes can be illustrated by the impact of aflatoxin, a potent carcinogen elaborated by species of the fungus Aspergilus, which can infect improperly stored grains and other foodstuffs. In a study in China (Table 1), it was shown that exposure to aflatoxin and chronic infection with HBV has a multiplicative effect on the risk for HCC. Individuals neither infected with HBV nor exposed to aflatoxin were assigned a risk ratio of 1. Those who had increased levels of aflatoxin but were not infected with HBV had a significantly increased risk, as did those infected with HBV but without increased aflatoxin levels. However, those exposed to both agents had a 60-fold increase in their risk!
Even with this increased risk, there were some people who did not succumb. Additional information is now available that can explain this, in part. McGlynn and her colleagues reported the effect of genes segregating at two different human gene loci that produced protein enzymes that detoxify aflatoxin. Each of these gene systems are polymorphic, and an individual, depending on the genes he or she has inherited, will or will not effectively detoxify aflatoxin. Exposure to aflatoxin also appears to be associated with a mutation in the p53 system that can affect carcinogenesis. Hence, the individuals who have the alleles that produce the less effective detoxifying enzymes will be more likely to have the mutation in p53 and DNA damage, and will be at increased risk for HCC (Figure 3).
A great deal is known about genetic and environmental factors and their interactions that lead to cancer of the liver. Knowledge of the genetics has helped immeasurably to understand these phenomenon, but intervention for prevention, and possibly treatment as well is based not on changing the host's genome, but in intervening to protect against environmental factors. A large percentage of HCC can be prevented by infant vaccination against HBV, and this is now in common use. The contamination of food by Aspergilus can be prevented by appropriate storage methods; iron storage can be controlled by avoiding supplemental iron and other measure, and the levels of arsenic in water can also be controlled. It is likely that many other cancers and other diseases will yield to similar practicable methods.
A disease-free life is a heady promise, but it is already a blessing that many enjoy, and one can hope that by continuing research, it can in the future be extended to many others.
Table 1. Risk Ratios (95% CI) for Hepatocellular Carcinoma Associated with and without Chronic Infection with Hepatitis B Virus (HBsAg) and with Urinary Levels of Aflatoxin in a Population from Shanghai
|
|
Aflatoxin |
|
|
HBsAg |
Negative |
Positive |
|
Negative |
1.0 |
3.4 (1.1, 10.0) |
|
Positive |
7.3 (2.2, 24.4) |
59.4 (16.6, 212.0) |
|
SOURCE: Adapted from Qian et al., Cancer Epidemiol. Biomarkers Prevent, 3:3–10, 1994. |
||

Figure 3 Features of the relation between hepatocellular carcinoma and enzymatic detoxification of aflatoxin. SOURCE: Adapted from McGlynn et al., Proc. Natl. Acad. Sci. USA, 92, 2384–7, 1995.
New Knowledge for Health: Response
Michael M. E. Johns, M.D.
Vice President for Medical Affairs, Professor of Otolaryngology– Head and Neck Surgery, and Dean of the Faculty, Johns Hopkins University School of Medicine
I thought that I would start with a summary, but in that summary I am going to raise more questions than give answers. Perhaps my message is quite simply that we as professionals must find the answers to these and to other questions that science will raise for us as members of the global community.
In his chapter Dr. Blumberg has covered a number of areas and has suggested important issues that undoubtedly will preoccupy us for many years to come. Perhaps what stands out most in this discussion is the extraordinary new levels of knowledge to which we shall soon have access and the extraordinary levels of wisdom to which we must aspire in order to make the most of what we will know.
We will attain new levels of understanding because we will be generating unprecedented amounts of knowledge and because we will have enormously expanded computational data capabilities. Any scientist contributing to the genome database will collect billions of bits and bytes of information about the molecular basis of the genome. New advances in computer science and computational biology will provide us with the capacity to uncover links, patterns, and associations that we never would have suspected.
For instance, we are just beginning to see the possibility of whole new areas of knowledge based on new discoveries of evolutionary and functional relationships. An example is the discovery that vision and hormone action involve similar signal transduction. This is a tremendously powerful insight. In short, it is difficult to overestimate how much we will know about our genome in 10–20 years and the impact that this knowledge will have on our capacities to cure and prevent disease.
No doubt, much more biology will be done on the computer. Computational biology, structural biology, and combinatorial analysis are developing powerful new software and mathematical techniques that can determine the structure of drugs and mimic actual molecular interactions. All of these will grow in importance.
Also of increasing importance will be the need for a larger cadre of clinical investigators who can sort out the efficacy of hundreds of new drugs that will be developed with increasing speed using these new capabilities.
What is easy to underestimate is the impact that this knowledge will have on the organization of medicine, on our roles as physicians, and on the ways in which we allocate health care resources in our society. Knowledge, after all, is power. Biologically at least, we are going to have a lot of both new knowledge and new power.
One very important question is, ''Who is going to control all of this knowledge and the power to employ it?'' Right now, there is a growing trend to have much of the traditional physician and patient control over the employment of medical knowledge restrained by certain types of delivery organizations and financial considerations. This is just an initial skirmish. What may prove to be a much bigger battle is the control of knowledge produced by biomedical science and medicine over the next half-century.
First, following Dr. Blumberg's lead, let us look at a few ways in which our knowledge will affect us. Curative gene therapy aimed at somatic cells in general probably will have only a modest impact on medicine and on the organization of health care delivery as we know it. However, it will raise new issues about societal costs and the ethics of rationing care. Therapies aimed at germinal cells will be far more controversial and have far larger implications for the organization of medicine, societal values, and health care costs. It is easier to see how the former therapies will play out than it is to see how the latter will.
At present, we know how to make decisions and choices about treatments for individuals—at least we think we do. In fact, that is what we do best. On a case-by-case basis, patients and physicians still basically make treatment decisions, even if that has been made more difficult by the types of third-party interventions and new cost-based considerations that recently have challenged and changed the calculus.
To the extent that genetic interventions begin to occur at the somatic cell level with individuals, the basic physician–patient relationship should not be altered significantly. However, the ability to intervene at the molecular level in somatic cell disease processes likely will generate considerable controversy at the socioeconomic and ethical levels.
For instance, let us assume that we arrive at a point where we have developed our capacities in what can be called molecular epidemiology and can predict susceptibility to lung cancer in each of the individuals in a population (e.g., of the seventh grade at your local middle school) with a fair degree of certainty. We will know the environmental risk factors, with cigarette smoking perhaps the most important of many. If this knowledge is
then transmitted on a systematic basis to these students so that each is made fully aware of the risk factors over a period of years, what are the ethical or economic implications for individuals who disregard the known risk and at age 50, after almost 40 years of smoking, are diagnosed with lung cancer?
The first obvious—or at least likely—private insurance industry response might be to link insurance premiums to individual risk factors and personal compliance. If you know the risks and the risks are high, and if you take the risks and smoke, for example, then your premiums are high. If you comply with a low-risk regimen where you do not smoke, your premiums are lower. It is certainly possible to foresee an insurance industry assembling a wide variety of such risk factor profiles and crafting premiums to reflect genetic factors and environmental compliance. This would be the ultimate in risk-adjustment factors.
This new genetic knowledge is going to raise many issues about treatment decisions, the allocation and distribution of health information, privacy, and much more. What if society's inclination were to link health care spending in some way to personal compliance? We already have this argument going on between cigarette manufacturers and those who claim injury from these products.
Suppose these middle-schoolers reach age 50, are poor or disabled, and therefore are insured through public programs. This, of course, assumes that such programs still exist at that point. What if a high public priority is to limit health care spending and one important part of that policy in this new age of molecular medicine is to hold individuals more accountable for certain behaviors that substantially increase health costs? What if public policy wants to preclude the lung replacement surgery that is available, although at substantial cost, and to limit total spending on the lung conditions of those with relevant risk factors who did smoke or who work in an environment identified as high risk?
One can imagine any number of scenarios in which this type of issue could arise. What is more difficult to imagine are satisfactory solutions. The implications of interventions in germinal cells are even more far-reaching.
So let's move on to some implications beyond all of this. Most important in my mind is the need to realize that our new knowledge will bring with it both new power and new responsibilities. Not only we, but also the public, will have much more information and many more choices—often very difficult and expensive choices. Some in medicine may think that the new issues, especially the ethical, economic, and social ones, are not the concern of biomedical scientists and physicians. You might say that we developed the science, and someone else must figure out its broader implications.
My main concern with the future of knowledge in health care is that we will not cede "jurisdiction," to use an important concept from the legal lexicon, for any of these issues. This forum illustrates the need to understand these issues as part of the evolving and growing purview of the medical profession. Knowledge is power. As medicine becomes more powerful, our professionalism must become more developed.
Actually, I prefer to say that as our knowledge and our capabilities become more powerful, our professionalism must become more profound. That is one reason why at Johns Hopkins we have introduced a four-year required course of study about the role of physicians in society, in which our students study the implications of new knowledge and evolving technologies. Future generations of physicians and scientists are going to have a more profound understanding of the knowledge and capabilities they possess and an ability to play a leading role in defining and controlling the application of this knowledge. Otherwise, medicine will be reduced to the level of a craft, physicians will lose their status as professionals, and society will lose a great deal more.
I have jotted down a few of the implications of this new knowledge for our academic health centers, and they are all in the form of questions relating to this new science and the potential that we see coming from it and its applications. Fundamentally, we need to ask ourselves what kind of students we will select. What kind of faculty will we recruit for this new era? How do we organize for the future in terms of departmental structure? Some of that has been discussed for the clinical side. We must ask the same questions about the scientific side. What kind of facilities do we need? Will we need any? Will there be virtual universities, and will everybody work at home? How do we integrate where we are going into the larger university? How does this new information become part of our daily lives? Will anybody be there in line to pay for it?
There are many strong forces acting in the medical profession at the moment that would happily see us become more like employees and line workers, technicians, or skilled crafts people. The nationwide skirmish going on right now about who makes the call in the application of health care knowledge may presage larger battles to come. There is only one way we can address this, and that is not individually, but as a profession.












