Page 74
4—
Processing Technologies
Expansion of biobased industrial production in the United States will require an overall scale-up of manufacturing capabilities, di versification of processing technologies, and reduction of costs. The development of efficient ''biorefineries" that integrate production of numerous biobased products would help reduce costs and allow biobased products to compete more effectively with petroleum-based products. The development of new or improved low-cost processing technologies will largely determine which biobased products become available. Currently, certain processing technologies are well established while others show promise but will require additional refinement or research before they come into practical use. U.S. production of low-cost agricultural materials and experience with prototype biorefineries position the nation well to capitalize on such processing improvements.
The market prices of large-scale (commodity) biobased industrial products will depend on two primary factors: (1) the cost of the biobased raw material from which a product is made and (2) the cost of processing technology to convert the raw material into the desired biobased product. The industries for producing chemicals and fuels from petroleum are characterized by high raw material costs relative to processing costs, while in the analogous biobased industries processing costs dominate. Reducing the costs of producing commodity biobased industrial products such as chemical intermediates derived from fermentation will depend most strongly on reducing the costs of processing technology, the focus of this chapter.
Page 75
The Biorefinery Concept
Today's petroleum refineries generate numerous products efficiently and at a very large scale from crude oil, an inexpensive raw material. The development of comparable biorefineries will be essential to make many biobased products competitive with their fossil-based equivalents. Each biorefinery would process essentially all of its biobased feedstock into multiple value-added products. The product types would include not only those manufactured by petroleum refineries but also many others that petroleum refineries cannot produce. Some examples of known and potential biorefinery products are:
• fermentation feedstocks (starch, dextrose, sucrose, cellulose, hemicellulose, molasses);
• food products (oil, starch, sweeteners);
• nonfood industrial products (loose fill packing material, paper sizes, textiles sizes, adhesives);
• chemical intermediates (lactic, acetic, citric, and succinic acids);
• fuels (ethanol, acetone, butanol);
• solvents (ethanol, acetone, butanol, esters);
• industrial enzymes; and
• biodegradable plastic resins.
Existing U.S. Prototypes
Prototypes of highly integrated biorefineries already exist in the United States. Plants that currently process agricultural and forestry materials into value-added products include corn wet and dry milling plants, soybean processing plants, wheat mills, and paper mills. The wheat, soybean, and corn operations are highly efficient and process over 95 percent of incoming feedstocks into value-added products. In some places, industrial complexes centered around corn wet milling use a single feedstock, corn, to produce a variety of products. Similar refinery complexes could be developed around corn dry mills or fermentation ethanol plants. Today's paper mills, wood products plants, and sugar beet refineries are partially integrated systems. Several pulp and paper mills produce pulp, paper, lignin byproducts, and ethanol while recycling waste paper—all on a single site with leftovers being dewatered and burned to produce steam and electricity. They have the potential to become more fully integrated by further processing, thereby enhancing the value to consumers of all their outputs. Waste paper and municipal sludge are examples of feedstocks around which biorefineries might one day be built, although this development has not yet occurred.
Page 76
Corn Wet Milling
Corn wet mills used 11 percent of the U.S. corn harvest in 1992 (worth $2.6 billion on the grain market), made $7.0 billion of products, and employed almost 10,000 people (Agricultural Census 1994). Wet milling of corn yields corn oil, corn fiber, and corn starch (Figure 4-1). The starch products of the U.S. corn wet milling industry are fuel alcohol (31 percent), high-fructose corn syrup (36 percent), starch (16 percent), and dextrose (17 percent). Although a greater volume of the starch goes to non-food uses, food uses (excluding syrups) are more significant in terms of dollars per pound. Corn wet milling also generates other products (sometimes called "coproducts")—gluten feed, gluten meal, oil, and steepwater (Hacking, 1986).
Most corn fed into a wet mill goes to the primary products (starch and oil), and the balance goes to other products; the relative proportion depends on the corn's initial moisture content (Figure 4-1). High-quality No. 2 yellow dent corn currently is the preferred feedstock for wet mills
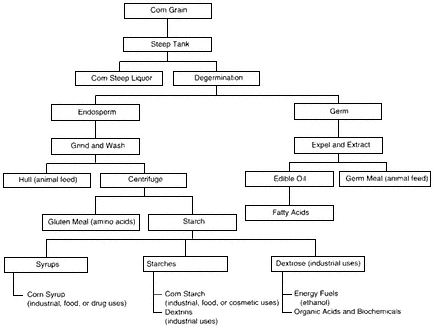
Figure 4-1
Corn processing and fermentation chemicals.
Page 77
because it results in the highest yield of starch and oil relative to the lower-value coproducts. North American corn gives the best yields in comparison to European, African, and Asian corn varieties. Latin America could produce corn of equal quality to North American corn.
Wastes generated by corn wet milling are relatively benign and readily treated onsite. However, these wastes also represent potential fermentation feedstocks for generating additional value-added products.
Corn wet mills could comprise the front end of an industrial complex that produces food, specialty chemicals, industrial products, fuels, and pharmaceuticals. Such an expanded biorefinery would provide cleaner and more economical processes for producing existing products, new intermediates for manufacturing new products, an expanded stable market for wet millers, and an expanded market for corn farmers.
The Archer Daniels Midland (ADM) complex in Decatur, Illinois, is the prototype for such an expanded biorefinery. There, a large corn wet-milling plant and a steam and electric cogeneration station form the nucleus for several other plants. The wet mill is the source of materials for plants that produce industrial enzymes, lactic and citric acids, amino acids, and ethanol. The enzymes are then used to convert starch to lower-molecular-weight products, principally various maltodextrins and syrups (i.e., in liquefaction). The lactic and citric acids are used in processed foods, detergents, and polymers. The amino acids are used as feed and food supplements and, in the case of phenylalanine, to make aspartame. The ethanol is used as a fuel or an industrial solvent.
The United States is well positioned to develop biobased industries according to the above model of a biorefinery complex having corn wet milling as its nucleus. The current U.S. corn wet-milling industry and U.S. production of preferred corn feedstocks could provide the springboard. In addition, certain corn hybrids under development may yield higher levels of starch. This could lower feedstock costs and decrease byproduct production if these hybrids were more widely planted and used for industrial purposes. In the 1996 to 1997 marketing year, about 7 percent of U.S. corn grain was processed into industrial products (e.g., fuel alcohol; refer to Table 4-1); the remainder was used for food and animal feed.
Soybean Processing
The United States is the largest grower and processor of soybeans in the world. Soy processing yields 80 percent products (i.e., soybean oil, soy protein, lecithin, and soy protein hydrolyzate) and 20 percent defatted soy meal coproduct (see Figure 4.2; Szarant, 1987). Most of this defatted meal
Page 78
| ||||||||||||||||||||||||||||||||||||||||||||||||
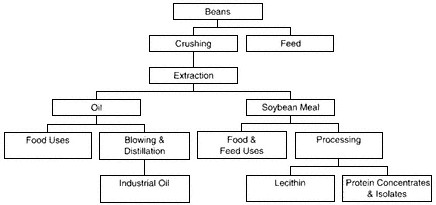
Figure 4-2
Soybean processing.
Page 79
ends up as animal feed. The U.S. industry is internationally competitive and stable. Moreover, all of the wastes from soy processing plants are relatively benign biological materials that can be readily degraded in conventional waste treatment plants and are usually processed onsite.
As with corn wet milling, soy processing plants could serve as the front end of an industrial complex that produces food, specialty chemicals, fuels, and pharmaceuticals. North American soybean growers are highly efficient and outperform international competitors, except where the competition is supported by local tariffs. American and Canadian soybean production and processing technologies are modern and very advanced. As with corn, such characteristics position the United States well for developing industrial biorefineries centered on soy processing. Such expansion would, however, require new uses for soy protein, since existing protein markets in animal feeds are saturated and could not absorb additional production without depressing prices. In the future, genetic engineering techniques may be used to alter the soy proteins, leading to expanded uses and increased value of industrial processed soybeans.
Comparison of Biorefineries to Petroleum Refineries
The comparison of biorefineries to petroleum refineries in Table 4-2 suggests that biorefineries offer a number of potential advantages because they rely on sustainable, domestically produced raw materials.
The development and expansion of biorefineries could yield great benefits to the public at large (see also Chapter 1). Such biorefineries could produce significant amounts of valuable products from domestic renewable resources and consequently reduce national dependence on fossil feedstocks. This change could also reduce the level of pollutants generated by industrial production while still providing goods at prices and qualities comparable to those derived from nonrenewable resources. Further research is necessary to examine environmental and energy impacts at all phases of manufacturing. Switching industries to the efficient use of renewable feedstocks through biorefineries will take time and require considerable technical and financial effort. The quickest pathway would be to build on the existing corn wet mills. Cargill and ADM already have several corn wet-milling facilities that approach being integrated biorefineries. Lessons learned from these operations and from refining fossil feedstocks could help in upgrading other large grain mills and soybean processors to biorefinery status. With additional effort, paper, sugar, and wood products manufacturers could be brought online. In the future, biorefineries could use other feedstocks such as municipal sludge and mixed waste paper, crop residues, or dedicated lignocellulosic crops such as poplar or switchgrass.
Page 80
| |||||||||||||||||||||||||||
Lessons from Petroleum Refinery Experience
Several lessons from fossil feedstock refineries might prove helpful in the future development of biorefineries and should be incorporated into strategic planning for the industry. These lessons include the following:
• refineries invariably produce more and more products from the same feedstock over time, thereby diversifying outputs;
• refineries are flexible and can shift outputs in response to change
• processes in refineries improve incrementally over time; and
• process improvement invariably makes the cost of raw material the dominant factor in overall system economics.
Developers and analysts of biorefineries can use the above criteria to measure progress toward fully developed biorefinery systems. The eco-
Page 81
nomic and technical performance of relatively new biorefinery systems will virtually always improve over time.
Processes for Converting Raw Materials to Biobased Products
Biorefineries of the future will use technologies based on thermal, mechanical, chemical, and biological processes to derive industrial products from renewable resources. Conversion of raw materials—such as wood, other lignocellulosic biomass, vegetable crops, starch-producing crops, and oil seeds—to final end products often will require a combination of processes.
This section identifies the technologies that might be used by future biobased industries. A distinction is made here between "proven" and "potential" technologies. The former covers technologies that have been evaluated at a large enough scale to ensure the process is technically practicable in a commercial plant. ''Potential" technologies, in contrast, are generally those that have been evaluated at laboratory scale and are sufficiently attractive to be of potential commercial interest.
Lignocellulose Fractionation Pretreatment: A Key Step
Biological conversion of carbohydrates by fermentation and enzymatic processes is perhaps the most flexible method of converting renewable resources into industrial products. The carbohydrates for most fermentation-derived products currently come from corn starch. Without new carbohydrate sources, the cost and availability of starch ultimately would determine the scale of biobased industries based on carbohydrates. Lignocellulosic materials could potentially provide a new, much larger (by at least two orders of magnitude), and less expensive carbohydrate source for biobased industries. Realization of this potential will depend on the development of inexpensive and effective processing technology to fractionate and convert lignocellulosics to fermentable sugars (Lynd, 1996). Despite past investigations of many processes, none has yet enabled growth of a large-scale bioconversion industry based on lignocellulosics (McMillan, 1994). The necessary technical advances present a formidable problem but one that is appropriately a high research and development priority for the nascent biobased products industry.
Utilization of all three fractions of the lignocellulose— the lignin (15 to 30 percent), the cellulose (35 to 50 percent), and the hemicellulose (20 to 40 percent)—is desirable from a "no waste" engineering design maxim, just as utilization of the oil, protein, and fiber of corn and the steepwater of the wet-milling process is essential in a starch-based industry. Thus, pro-
Page 82
cesses discussed below include those for lignocellulose fractionation and those for processing the component fractions.
Proven
Removing the lignin from lignocellulose is one possible step toward obtaining carbohydrate for further bioconversion. Lignin makes up about 15 to 30 percent by weight of lignocellulosics such as wood and the woody parts of annual plants. Because paper production from this raw material requires removal of most lignin, the pulp and paper industries have well-developed processes for this purpose. Chemical wood pulping produces as a byproduct more than 50 million metric tons of lignin a year in the United States. The most common process in the United States for removing lignin is kraft pulping, which involves cooking wood chips with a mixture of sodium hydroxide and sodium sulfide to partially depolymerize and solubilize the lignin. The resulting kraft pulp has a value six to eight times that of the original wood raw material. Most of the byproduct kraft lignin is burned to provide fuel for pulp and paper mills and to recover and regenerate the inorganic pulping reagents. The most modern mills actually produce more energy than they consume, reducing the need for fossil fuels. About 0.1 percent of kraft lignin produced in the United States (45,000 tons) is diverted for use as a chemical resource, and more could be diverted if its value in the production of chemicals, building materials, and other products exceeded its value as a fuel.
The other major chemical pulping process, used more in Europe and elsewhere outside the United States, involves cooking wood with sulfite salts to convert the lignin to water-soluble ligninsulfonate. Although some such mills still operate in the United States, mills must absorb high costs to recover waste materials that were previously disposed of in streams and waterways. Unlike the kraft process that degrades carbohydrates that are inadvertently solubilized during pulping, sulfite pulping produces spent liquor that contains nondegraded fermentable monomeric and oligomeric sugars. Some mills in the United States and Europe ferment sugars in the spent liquor to produce ethanol.
Acid hydrolysis of wood is an old technology developed extensively during and following World War II. It involves using dilute or concentrated sulfuric acid to effect hydrolysis at temperatures between 120° and 170°C. The solid residue resulting from hydrolysis contains some of the cellulose and most of the initial lignin. Notable processes in the published literature that have not, however, been adopted in wide-scale commercial practice include the "Hokkaido Process" (Bungay, 1981), the "Berguis Rheinau Process" (Bungay, 1981), and continuous acid hydrolysis. Hydrochloric acid has also been used as a lignocellulose pretreat-
Page 83
ment, generally in concentrations of 35 to 50 percent and at temperatures between 100°C and 170°C. O'Neil (1978) has reviewed variations in the hydrochloric acid hydrolysis process.
Potential
Lignin is an important coproduct with sugars resulting from the hydrolysis of lignocellulosics. The overall economics of biorefineries that use lignocellulosics as raw materials would be improved by finding uses for lignin and avoiding lignin disposal costs. At a minimum, lignin can be burned to produce electricity and steam for supplying the biorefinery, with excess electricity exported to the grid. Higher-value uses may be possible, however.
Kraft lignin and ligninsulfonate have a wide variety of relatively low-value applications as polymers. Attempts by pulp and paper research laboratories to develop additional markets for byproduct lignins have failed. Similarly, although low-molecular-weight compounds, particularly phenols, can readily be produced from kraft lignin, they have not been produced in a commercially competitive manner. However, a recent study by the U.S. Department of Energy (DOE) concluded that phenolics and anthraquinones could be produced competitively from lignin by pyrolysis of lignocellulosics (Bozell and Landucci, 1993). The study further concluded that production of benzene, tolulene, and xylenes from lignin by pyrolysis also looked promising.
Although carbohydrates comprise about 70 percent by dry weight of lignocellulosic plant material, most are in a form that cannot be readily fermented. Without pretreatment—chemical, mechanical, biological, or combined—carbohydrate yield following hydrolysis will be low, making processes based on fermenting these sugars less economical. Purdue University researchers undertook a major effort in the late 1970s using various solvents to dissolve and swell lignocellulose (e.g., Ladisch et al., 1978). The key to their approach was using a solvent that could permeate the lignocellulose and disorganize its crystalline structure. This change increases lignocellulose reactivity and makes it more amenable to subsequent hydrolysis. One method causes the dissolution and swelling of lignocellulose using ethylenediamine (25 to 30 percent) in water plus cadmium oxide (4.5 to 5.2 percent) at room temperature. The treatment makes the lignocellulose highly susceptible to enzymatic hydrolysis and enables nearly 100 percent of the theoretical glucose yields. This type of process is in experimental stages and has not yet been adopted by industry.
Many solvents have been shown to extract the lignin effectively from lignocellulose, including organic solvents such as butanol and inorganic extractants such as ammonia. The use of solvents to delignify wood and
Page 84
make the lignocellulosic biomass more susceptible to biological processing is often called the "organosolv" process (Holtzapple and Humphrey, 1984). Much work has been done on this process, and a demonstration pulp mill based on the process (Repap) currently operates in Canada. Avgerinos and Wang (1983) also have reported the use of selective solvents for delignification of lignocellulose.
A novel pretreatment for lignocellulosic biomass, known as the ammonia fiber explosion (AFEX) process, has been developed for various feedstocks, including alfalfa hay, bagasse, wheat straw, switchgrass, and coastal bermudagrass (Holtzapple et al., 1991; de la Rosa et al., 1994). This technology uses liquid ammonia at elevated pressures and temperatures (between 50°C and 90°C) to permeate the lignocellulosic biomass (Dale and Moreira, 1982). The treated material is then decompressed to achieve an "explosion" of the biomass, yielding material that is much more susceptible to microbiological and enzymatic degradation. The AFEX process followed by treatment with cellulases and hemicellulases gives high sugar yields (approximately 90 percent of theoretical estimates) from various raw materials using low enzyme levels (about 5 IU per gram of dry biomass; Holtzapple et al., 1991; de la Rosa et al., 1994). More than 99 percent of the ammonia in the AFEX process can be recovered and reused. In comparison to the many pretreatment technologies studied over the past 80 years, the AFEX process appears to be attractive from technical, environmental, and economic perspectives, but it is too early in the development stage to make accurate judgments on its potential commercialization.
Another relatively new and promising lignocellulose pretreatment involves using liquid water under pressure at temperatures over 200°C (Van Walsum et al., 1996). While data on this approach are still incomplete, the possibility of using liquid water as a pretreatment chemical has obvious advantages. Pressurization of wood with steam to 45 to 52 atmospheres with subsequent decompression shatters the structure of lignocellulose (Bungay, 1981). This method has been shown to be effective with various biomass materials and forms the basis of several processes to disintegrate the lignocellulosic biomass (Carrasco et al., 1994). The technology has undergone pilot-scale development but is not yet used in industrial processes.
Conversion of lignocellulosic waste materials is an active area of research. The conventional technology for converting cellulose from waste paper to glucose is a stirred-tank batch reactor with cellulase enzymes; however, the enzyme hydrolysis takes over 40 hours to complete and the enzyme costs are prohibitive, at least at the high enzyme levels typically used (>20 IU per gram of dry biomass; Scott et al., 1994). An advanced alternative reactor technology couples two types of continuous bioreactors
Page 85
in a stirred-tank attrition bioreactor containing cellulase enzymes, followed by a fixed-bed reactor with immobilized cellobiase (cellobiose hydrolase) entrapped in gel beads. Under experimental conditions, this approach achieved essentially complete hydrolysis of waste paper in 25 hours (Scott et al., 1994). Since waste paper is already substantially pretreated, this process may not prove as effective for untreated lignocellulosic substrates such as aspen or poplar. A roadblock to lignocellulose conversion is the high cost of cellulase enzymes and lack of proven alternatives (Rooney, 1998).
Thermal, Chemical, and Mechanical Processes
Certain thermal, chemical, and mechanical processes for the conversion of renewable resources into chemicals and fuels have been practiced for many years (for further details see Bungay, 1981, and Elliott et al., 1991). Others have been experimentally or theoretically demonstrated but are not yet in commercial use. In general, the role of mechanical processes is to prepare various feedstocks for subsequent conversion by other types of processes. This section describes proven and potential thermal, chemical, and mechanical processes. It does not cover pulping technologies because pulp cost is generally too high for most uses in biobased products.
Proven
Direct combustion of wood for cooking and heating has been practiced for centuries. Industrial-scale generation of steam that, in turn, can be used to produce electricity frequently relies on combustion of wood biomass and municipal solid wastes. The biomass power industry in the United States is expected to grow from 31 billion kilowatt hours in 1990 to 59 billion kilowatt hours in 2010. This is approximately 1 percent of the total generation of electricity in the United States (EIA, 1997), including conventional (e.g., hydroelectric) and renewable resources.
Biomass from renewable resources often must be mechanically densified before it provides an effective combustion fuel. Densification of woody biomass, for example, generally involves pelting, cubing, briquetting, extrusion, and roller compression. A number of densifiers are commercially available for industrial operations (Bungay, 1981).
Mechanical and thermal processes, such as distillation, are involved in the refining of pine tree exudates into industrial chemicals (e.g., turpentine, oleoresins, gum spirits, pinenes, camphene, abietic acid). The exudates, or "naval gum stores," are byproducts of the paper pulp and lumber industries, and most naval gum currently comes from processing
Page 86
of kraft pulp. The naval gum industry, although not large, is economically viable in combination with the lumber and paper industries.
Pyrolysis is a well-established thermochemical technology using "destructive distillation" to convert biomass into useful chemicals and fuels (see Elliott et al., 1991, for review). High temperature and limited air—sometimes in the presence of a catalyst—yield primarily hydrocarbon liquids (equaling 50 to 70 percent of inlet raw material), gases, and char. Major product yields following liquid pyrolysis can vary from 25 to 70 percent of inlet raw materials.
Commercial production of methanol has been accomplished by the destructive distillation of wood. Despite earlier optimistic projections, today no large-scale methanol manufacturing plant relies on destructive distillation (Hokanson and Katzen, 1978; Stinson, 1979). Most methanol is produced by the lower-cost method of chemically oxidizing natural gas (methane).
The production of furfural (the chemical furaldehyde) and its derivatives is another well-established technology (McKillip and Sherman, 1979). The major raw materials are hemicelluloses from annual crops. Although any agricultural product containing high-pentose-content hemicelluloses could be used in theory, only a few (corn cobs, oat hulls, rice hulls, and bagasse) are available in tonnage quantities within economic hauling distance of furfural production plants. Furfural production involves loading raw materials into a digester containing a strong inorganic acid. The hemicelluloses are hydrolyzed to pentoses, and these pentoses are then cyclodehydrated to furfural. High-pressure steam provides the necessary temperature, and the furfural is steam distilled. Various subsequent chemical modifications can produce furan derivatives. The current market for furfural is small; however, furfural was the sole source of adipic acid and hexamethylenediamine in the early days of nylon 66 and may have renewed potential in the future.
Well-developed chemical processes exist for deriving cellulose esters from natural cellulose. Cellulose acetate is the major cellulose ester currently in commercial production. The production process uses sulfuric acid as a catalyst with acetic anhydride in an acetic acid solvent. Cellulose acetate production declined from 392,000 tons in 1979 to 321,000 tons in 1993, mostly because other organic polymers have partially replaced cellulose acetate.
Cellulose films can be made by dissolution of very clean cellulose preparations in solvents. This process has been used for several decades to produce cellophane, the basis for camera films and hot dog casings. However, cellophane at present is not competitively priced for many applications, largely because the dissolution process for cellulose xanthate requires carbon disulfide, an expensive solvent that poses environmental
Page 87
hazards. New solvent-based processes could increase the competitiveness of cellulose films and enable replacement of petrochemical-derived plastics in applications such as packaging. Less expensive dissolution processes also could make rayon and other cellulose polymers more competitively priced. Two companies from the United Kingdom and the Netherlands recently agreed to take this approach and formed a joint venture to develop and produce a solvent-spun cellulose textile yarn.
Gasification is an established technology involving reaction of carbonaceous materials with steam and oxygen to produce a mixture of carbon monoxide, hydrogen, carbon dioxide, methane, tar, and char. Numerous different reactors have been used for gasification, and coal is usually the raw material (Bliss and Blake, 1977). Only limited commercial development of gasification using renewable raw materials, such as wood, to produce useful products has occurred, mainly because of the poor economics.
Fractionation of protein-rich renewable resources (i.e., forages and grasses) is a thermal/mechanical process that has been proposed for producing animal feeds. Pirie and co-workers reported the first experiments producing "leaf protein" feeds in the 1940s (Pirie, 1971). Despite much subsequent research, a viable industry has not yet emerged. This failure is due at least in part to economic considerations. The existing leaf protein processes do not increase the value of the fibrous portion of the forage or grass (i.e., the cellulose, hemicellulose, and lignin) that constitutes over 80 percent of the plant material's dry weight. The economics of protein extraction might be more favorable if the fibrous portion's value could be enhanced—for example, by fermenting the carbohydrates to fuels and chemicals.
Potential
Liquefaction of wood is a theoretically possible technology that produces synthetic fuels having properties similar to petroleum and natural gas. However, past work on synthetic fuel technologies has focused primarily on coal as the raw material, and wood liquefaction differs from that of coal. The wood's lignocellulosic biomass cannot be catalytically hydrogenated to produce liquid fuels because liquefaction of cellulose requires removal of oxygen as well as addition of hydrogen. In several feasibility and pilot studies, researchers have catalytically removed oxygen from lignocellulosic biomass and then used liquefaction and catalytic hydrogenation to produce liquid fuels (Bliss and Blake, 1977; Elliot and Walkup, 1977). More recent studies using extrusion-fed reactors have obtained 80 to 100 percent of the theoretical oil yield from wood flour (White and Wolf, 1988). Other liquefaction approaches (by solvolysis,
Page 88
catalysis, steam/water, or high pressure) have generally given lower oil yields, typically 40 to 60 percent (Bain, 1993).
Another possible approach builds on earlier protein fractionation work to coproduce protein and ethanol (Dale, 1983; de la Rosa et al., 1994). The process combines protein extraction from dry forages with hydrolysis of the extracted fiber to fermentable sugars. Researchers pretreated coastal bermuda grass with the ammonia fiber explosion process (AFEX) and then extracted more than 80 percent of the protein with dilute calcium hydroxide. Hydrolysis of the resulting extracted fiber yielded more than 90 percent of the theoretical sugar yield, which was subsequently fermented to ethanol (de la Rosa et al., 1994). Coproducing protein and fuels could help reduce the perceived or real conflicts inherent in allocating renewable resources to the two important human needs: food and fuel.
Biological Processes
Biological processes often play a role in the pretreatment and conversion of renewable resources into industrial products. For example, starch resulting from corn wet milling is enzymatically hydrolyzed to glucose syrup that is then fermented to ethanol and other products. In bioprocesses, fermentation and enzymatic reactions carry out most classes of reactions used in the chemical industry, including oxidations, reductions, hydrolysis, esterifications, and halogenations. Unlike thermal and chemical processes, bioprocesses occur under mild reaction conditions, usually result in stereospecific conversions, and produce a smaller number of generally nontoxic byproducts (see Box 4-1). One drawback is that bioprocesses typically yield dilute aqueous product streams, requiring subsequent thermal, chemical, or mechanical processing for separation and purification. Other barriers to economic competitiveness include feedstock cost, product yield, and high capital costs.
Proven
The fermentation industry produces alcohol, organic acids, polymers, antibiotics, amino acids, enzymes, and other products. The most common production technology is the conventional stirred-tank batch fermentor. However, continuous stirred-tank reactors with process control can enhance the economics of fermentation by allowing higher volumetric productivity. Immobilized cell reactors can provide a higher productivity in the fermentation broth than do stirred-tank reactors, but difficulties can arise in ensuring that adequate carbohydrate is transferred to the fermenting cells ("mass transfer") or in removing heat produced by the biological reactions.
Page 89
|
Fuel ethanol production involves fermentation of glucose syrup by the yeast Saccharomyces cerevisiae in large-scale, high-capacity fermentors. The technology has evolved from a batch-scale system to the cascade system now found in almost all large U.S. plants (Hacking, 1986). The cascade system approximates a continuous operation by continuous flow through a series of fermentors.
Page 90
The major organic acids produced by fermentation are citric and acetic acid for food industry uses. Other organic acids, primarily gluconic and lactic acids, are produced in smaller quantities. Production of citric acid worldwide relies on either the yeast Candida lipolytica or the fungus Aspergillus niger. Today, citric acid production usually involves submerged aerobic fermentations using glucose or molasses as the carbon source (USDA, 1994). Over the past 30 years, mutation and selection programs have yielded superior production strains of the yeast and fungus that can produce high concentrations of citric acid with few or no undesirable byproducts, such as isocitric acid.
Research on acetic acid production has focused for more than 50 years on homoacetogenic bacterium, such as Clostridium thermoaceticum. ("Homoacetogenic" refers to organisms that ferment a variety of substrates to primarily acetic acid.) The bacteria can convert glucose, xylose, and some other pentoses almost completely to acetate, which is formed by neutralization of acetic acid with dolomite. The energy crisis of 1972 stimulated general interest in the complex metabolism of homoacetogenic bacteria, and the U.S. Department of Transportation's Federal Highway Administration has supported recent related research. Calcium magnesium acetate is important as an organic, biodegradable, noncorrosive deicing salt and as an additive to coal-fired combustion units for control of sulfur emissions.
The biopolymer xanthan gum is produced by large-volume fermentation of molasses or corn syrup using the aerobic bacterium Xanthamonas campestris. This polysaccharide has uses in the food, paper, and oil field industries because of its high viscosity at low concentrations. The high viscosity also, however, makes heat and mass transfer problematic, especially in large-scale fermentors.
Large-capacity fermentors, typically stirred tanks, are used in the production of amino acids for food enhancers and animal feed supplements. The Japanese fermentation industry has dominated worldwide production of amino acids for the past 20 years and during this period has used random mutation and selection programs to develop high-producing microbe strains for many amino acids (e.g., glutamic acid, phenylalanine and lysine). ADM has recently begun to challenge dominance of the amino acid industry by Japanese companies (see Box 4-2). This example illustrates how the combination of low-cost raw materials and advanced technologies potentially makes U.S. companies competitive, even in industries historically dominated by foreign companies.
Microbial fermentation can produce enzymes for a variety of commercial processes and products, including production of corn sweeteners, laundry detergents and digestive aids, cheese and beverages, leather and textiles, and medicine and clinical diagnostics. Major industrial uses of
Page 91
|
enzymes occur in the liquefaction of starch to produce maltodextrins, corn syrup, high-fructose corn syrup, and laundry detergents, and as catalysts in the manufacture of other commercial products. Enzymes are used to catalyze reactions of commercial interest where chemical catalysts fail or perform poorly. Enzymes are widely used in the food, chemical, and pharmaceutical industries because of their unusually high catalytic activity, selective action, ability to function under mild conditions, frequent low cost, biodegradability, and high product yields.
Enzyme-catalyzed processes can be accomplished in both stirred-tank reactors and immobilized enzyme reactors. The corn-ethanol industry relies on enzymatic digestion of starch in stirred-tank reactors to produce glucose. Two processes are involved: digestion by amylases in a lique-
Page 92
faction process and digestion by amyloglucosidases in a saccharification process. The amylases now used for liquefaction are thermostable; some come from genetically engineered microorganisms. Ongoing research is examining ways to improve the liquefaction and saccharification steps by using immobilized enzyme reactors (e.g., Zanin et al., 1994; Gerhartz, 1990). Immobilized-enzyme reactor technology contributed to the greatest success in enzymatic processing to date—the commercial production of high-fructose corn syrup from glucose using the enzyme glucose isomerase (Klibanov, 1983). The example of high-fructose corn syrup demonstrates how advanced bioreactor concepts derived from research and development can be commercialized (see Box 4-3).
Several other significant examples exist of commercial enzymatic processing (Klibanov, 1983). Penicillin G and V catalyzed by penicillin acylase produces the 6-aminopenicillanic acid used in the synthesis of semi-synthetic penicillin. The production of L-malic acid, an acidulant used in beverages, involves conversion of fumaric acid by fumarase. A number of different L-amino acids used in food and feed supplements are produced enzymatically. Finally, acrylonitrile catalyzed by nitrile hydratase yields acrylamide, a chemical used to make polymers.
In recent years new opportunities for enzymatic processing have arisen from changing market conditions and growing environmental concerns. For example, in the pulp and paper industry, enzymes can help reduce chlorine use in the bleaching process. The kraft process is the major pulping process in the United States. It produces cellulose pulp that is brown because it contains residual modified lignin. The lignin is whitened by oxidative degradation and washed using chlorine and chlorine dioxide. However, environmental concerns have begun to reduce chlorine use. Work in the late 1980s showed that hemicellulases, primarily xylanases, could facilitate chlorine bleaching and thereby reduce bleaching chemical requirements and the production of chlorinated waste products. The hemicellulases are thought to "uncover" lignin shielded by residual hemicelluloses. Scandinavian and Canadian mills now use xylanases at a commercial scale, and other mills are examining this approach (Daneault et al., 1994), along, with other technologies. Oxidoreductase enzymes (e.g., laccase) also have potential value in these applications. Nonchlorine bleaching agents, such as ozone, oxygen/alkali, and peroxide, are gradually replacing chlorine, and xylanases will probably prove useful in combination with these chlorine substitutes (Allison and Clark, 1994).
The pulp and paper industry has begun to adopt several other new enzyme applications. Pitch reduction by mechanical pulping with lipases is now used commercially in Japan (Fujita et al., 1992). In France, cellulase/hemicellulase mixtures are used to improve drainage of water from pulp in paper making. Cellulase/hemicellulase mixtures are also effec-
Page 93
|
tive in deinking office waste paper (Jeffries et al., 1994), and this method is also likely to come into commercial use.
Potential
Funding by government agencies (DOE, USDA, National Science Foundation) from 1975 through 1985 supported fundamental research in biological technologies for ethanol production (NRC, 1992). A significant focus of this research was improved cell suspension and immobilized-cell reactor designs (Maiorella et al., 1981 and 1984; Bailey and Ollis, 1986). One promising approach resulting from this work provides an alternative
Page 94
to cell suspension bioreactors—a fluidized bed using microorganisms at high concentrations immobilized in either cross-linked industrial grade carrageenan, cross-linked modified bone gel, or other porous materials (Scott et al., 1994; Davison and Scott, 1988). A fluidized-bed approach using the bacterium Zymomonas mobilis with industrial feed materials has a volumetric productivity at least 10 times greater than conventional (batch reactor) technologies (Webb et al., 1995). Ethanol product concentrations of over 7.5 percent (w/v—weight of ethanol per volume of solution) can be maintained at an overall volumetric productivity of more than 60 grams ethanol per liter per hour.
Another advanced bioreactor concept, simultaneous saccharification and fermentation (SSF) reactors, can produce ethanol from mixed waste papers, agricultural wastes, and pulp and paper mill wastes (Emert and Katzen, 1980, Katzen and Fowler, 1994). The SSF reactor combines enzymatic and fermentation steps in one process unit using novel recombinant strains of bacteria. In-house production of cellulase enzymes (to further reduce costs) will be used in a planned application of this technology to convert waste paper in Florida (Katzen and Fowler, 1994).
A great deal of interest centers on use of the bacterium Clostridium acetobutylicum to produce acetone and butanol from starch, especially if the fermentation can be performed on a continuous scale. This fermentation has been practiced for more than a century at a batch scale, but the low-cost availability of petrochemicals has limited commercial production of bulk quantities by fermentation. As a group, these bacteria can grow on a wide variety of compounds, including waste streams that otherwise present disposal problems, such as cheese whey, fruit pulp, and partially degraded hemicelluloses. The fermentation produces large volumes of carbon dioxide and hydrogen as byproducts, and efficient recovery of these gases increases the process's value. Another "byproduct" is the bacterial biomass, a potential source of animal feed.
Genetic engineering and improved continuous production technology potentially could make fermentation by clostridia economically competitive with petrochemical sources. However, these advances pose significant challenges. Engineering the bacteria to produce a single end product from fermentation of carbon-rich compounds, for example, is made difficult by the inherent complexity of carbon flow in the clostridia. Carbon enters these bacterial cells in many forms. Simple and complex carbohydrates are readily used, and carbon is channeled by intermediary metabolism from pyruvate through acetyl-CoA to ethanol, acetate, acetone, butyrate, and butanol. All of these end products are produced to some extent, but some species may make predominantly one or more of them. The relative production of certain products depends on growing conditions such as carbon source, pH, and temperature. For instance, high glucose
Page 95
concentrations cause high solvent production and low acid production. Conversely, cells growing under conditions of limited carbon produce only acids because they do not enter the solvent production stage.
Additional problems arise because the clostridia and other microorganisms that make desirable products come from extreme environments. These "extremophiles" not only survive but actually thrive under conditions such as high temperatures (>100°C), high salinity, acidic or alkaline solutions, or where the atmosphere lacks oxygen (Gilmour, 1990). The bacteria have evolved special enzymes and metabolic strategies that allow growth in hostile conditions. They may survive low oxygen levels, for example, by channeling or adjusting the way they convert various intracellular metabolites. Consequently, attempting to direct carbon flow to desired products often fails to achieve the intended result.
Experimental attempts to drive carbon into one biosynthetic pathway over another by inducing overexpression of an enzyme may not succeed because the availability of cofactors and substrates becomes limiting. In theory, genetic engineering approaches could overcome this problem by directly modifying amounts of cofactors and substrates. In addition, requirements for growing clostridia under extreme environmental conditions and dealing with production of flammable hydrogen gas as a byproduct present technical challenges in process design and operation. Another approach has been to clone genes from clostridia for solvent production into other bacteria such as Escherichia coli or Bacillus. This strategy has had only limited success so far because these organisms, unlike clostridia, have not evolved to tolerate high levels of solvent during growth.
Needed Developments in Processing Technology
The establishment of large new biorefineries will require feedstocks of satisfactory price, quality, and quantity that are consistently and reliably available for processing. Sufficient numbers of trained individuals must be available to design, build, and operate these refineries, and economical and effective processes for converting raw materials into value-added products must be known or reasonably well developed. This section identifies key areas of processing technology where focused research and development efforts would have the greatest impact.
Upstream Processes
Renewable natural resources, including lignocellulosic biomass, will provide the raw materials for future biobased industries. Developing economical processes for pretreating this biomass will be essential to make
Page 96
sufficient carbohydrate polymers available for subsequent biological conversions. An efficient approach to this task would be to test, at a demonstration scale, the most promising pretreatment technologies developed over the past 10 to 15 years in order to assess their large-scale technical and economic feasibility.
Extensive research has examined enzymatic degradation of cellulose and hemicellulose into fermentable sugars (NRC, 1992). Improvements in pretreatment technology (to enhance enzyme accessibility) and enzyme hydrolysis would lower the economic barriers to commercial lignocellulose conversion. Developments that reduce enzyme costs and increase the efficiency of xylose fermentation would also improve the overall economics. A particularly attractive possibility is to combine, in a single microorganism, the ability to produce extracellular hydrolytic enzymes that degrade cellulose and hernicelluloses with fermentation machinery and thereby directly convert biomass to useful products.
Bioprocesses
Advances in bioprocess engineering will play a key role in enabling the development of biorefineries. Advanced bioreactor concepts, especially for high-volume products, might allow significant increases in productivity and thus reduce capital and operating costs. The goals for future research on fermentation operations should include:
• combining the biological and physical operations of sugar production, fermentation, and product recovery in fewer vessels and fewer microorganisms to reduce capital costs and inhibition by microbial products, thereby increasing rates, yields, and selectivities.
• improving bioreactors for heat, momentum, and mass transfer for viscous, non-Newtonian fermentation broths and solid-liquid broths;
• developing new methods for monitoring biological processes, such as discrete sensors using microfabrication, real-time monitoring, and digital imaging of bioreactors; and
• developing new concepts in process control, such as the application of expert systems, artificial intelligence, neural networks, and principal component analysis.
The development and improvement of biological conversion processes will depend on continuing research to elucidate fundamental biological principles, combined with engineering analyses to improve biobased product yield (raw material conversion), selectivity (reduction of
Page 97
byproducts), and productivity (enhanced rate of production). Future work on molecular biology, genetics, and microbial physiology should include:
• identification of genetic changes that enable use of multiple substrates;
• analysis of biochemical pathways combined with directed mutagenesis (deletion and addition of enzymes) to reduce production of undesirable byproducts; and
• analysis of processes or steps in biochemical pathways that limit formation of desired products and identification of genetic changes to alleviate these limitations.
Microbiological Systems
Microbiological systems, particularly those involving anaerobic bacteria, are essentially multicatalytic reactors that direct the microbial metabolism toward the production of useful biobased products. Effective control of the extracellular environment by engineers would allow optimum product synthesis. Such control, however, will require a good understanding of the complex intracellular biochemical reactions involved in cellular metabolism and physiology. Several groups of bacteria in particular—the anaerobic bacteria, the Archei bacteria, and the extremophiles—provide a vast potential of host organisms for the production of chemicals and fuels from renewable resources, yet they have received relatively little research investment in comparison to aerobic microorganisms. Capturing their potential will require both research on fundamental aspects of microbial metabolism and physiology as well as tools for assessing and manipulating such processes. For example, one important goal is to identify strategies for either improving biosynthesis of products in bacteria that normally produce them or moving the relevant genes from these bacteria into new hosts. In either case, a greater understanding of the basic biology of biosynthesis and regulation of these compounds will be necessary. Future research on microbial metabolism, nutrition, and physiology should include:
• Development of basic tools to assess metabolic pathways for microorganisms such as intracellular measurements of metabolic intermediates.
• Analyses of biochemical pathways that integrate these basic intracellular measurements. Such analyses will provide fundamental understanding of the microbial metabolism and physiology necessary to focus metabolic engineering manipulations on enhancing organisms' overall productivity.
Page 98
• Identification of microorganism strains that can produce products for important biobased markets (see also Chapter 3), including organisms that can ferment 5-carbon and 6-carbon sugars to ethanol, organisms that can efficiently use sugar syrups from lignocellulose to produce other target chemicals (e.g., butanol and acetone), and organisms that can use ethanol and other oxygenated intermediates as a substrate to produce target chemicals.
• Understanding the interactions between the environment and the metabolism and physiology of microorganisms, particularly unusual microorganisms such as Archei bacteria and extreme thermophiles. Such understanding will enable manipulation of bio-reactor environments to mimic the natural environments in which these organisms have evolved.
• Studies of the fundamental principles of microbial physiology that affect regulation of metabolic pathways. Such information may make it possible to enhance product synthesis and secretion by overcoming biochemical regulatory constraints and metabolic flow bottlenecks.
• Basic research on principles of intermediate microbial metabolism to gain a better understanding of how concentrations of substrate or product can inhibit rates of product formation. Such understanding will aid in engineering bioreactor control to enhance the rate and conversion of raw materials into useful products.
• Basic research on cultures containing multiple microorganisms, particularly those in which the microorganisms have symbiotic relationships that increase growth and product formation. Advanced analytical tools are needed to assess the metabolism and physiology of these mixed-culture systems and the intricate relationships among mixed-culture populations.
Enzymes
The scope of current industrial applications of enzymes is limited by certain drawbacks of enzymes as practical catalysts. Such drawbacks include the insufficient operational stability of many enzymes, their real or perceived requirement for an aqueous reaction medium, and their often very narrow substrate specificity (i.e., enzymes frequently catalyze a particular reaction with a given substance but not an analog that is of commercial interest). Recent scientific advances in enzymology and molecular biology promise to eliminate, or at least alleviate, these limitations and significantly expand the opportunities for enzymatic processing.
For example, genetic engineering now makes it possible to replace
Page 99
any amino acid residue in an enzyme with any other amino acid virtually at will. Since most properties of enzymes are determined by their amino acid sequence, such replacement should in principle bring about a desired change in enzyme performance, provided that the structure-function relationship is understood. This type of ''protein engineering" has successfully enhanced the stability of enzymes against heat and oxidizing agents as well as broadened their substrate specificity (Knowles, 1987). However, the greatest impacts of protein engineering may well lie ahead.
Other major developments have made it clear that many enzymes can work as catalysts not only in aqueous reaction media but also in organic solvents to efficiently convert water-insoluble substances and catalyze new reactions (Klibanov, 1990). Also, the solvent can control enzyme properties, such as selectivity of action (Wescott and Klibanov, 1994). An industrial example of this nonaqueous bioprocessing is lipase-catalyzed conversion of palm oil to cocoa butter substitutes developed by the Unilever Company in Europe (Macray, 1985). It may even be possible to create artificial enzymes with tailor-made properties—for example, by generating "catalytic antibodies" (Schultz et al., 1990). Such advances should further increase the versatility of enzymes and the applicability of enzymatic catalysis to a broader range of industrial processes.
Professor Stephen Withers of the University of British Columbia has altered, by genetic engineering, the active site of a xylosidase so that it can no longer catalyze the hydrolysis of xylosidic bonds, its normal reaction, but only their synthesis (Withers et al., 1996). In the new enzyme the normal glycosyl-enzyme intermediate is intercepted with a sugar acceptor, rather than with water, which gives hydrolysis, forming a new glycosidic bond. This initial step in reversing enzyme activity gives hope that it can be done with other enzymes, but extensive fundamental research to elucidate the precise structure and mechanism of the catalytic sites of the enzymes in question must precede such work.
To capitalize on and expand recent developments in enzyme processing, future research in this area should:
• further explore catalysis by enzymes from thermophiles and other extremophiles;
• elucidate the mechanisms that allow enzymes isolated from extremophiles to remain stable in hostile environments (such mechanisms might eventually be incorporated into stabilization strategies for industrial enzymes from nonextremophiles);
• broaden the substrate specificity of enzymes by means of site-directed mutagenesis;
• identify enzymes that can produce products for important bio-based markets (see also Chapter 3), such as enzymes that can use
Page 100
ethanol and other oxygenated intermediates as a substrate to produce target chemicals;
• enhance and develop rational control of enzyme performance in organic solvents;
• investigate catalytic antibodies and nonprotein enzyme models to carry out industrially relevant conversions of renewable feedstocks; and
• improve inexpensive large-scale production of industrially useful enzymes.
Downstream Processes
A major weakness of fermentation processes in the production of large-volume, low-value chemicals is that the products are in dilute levels in aqueous effluent streams. In the production of organic solvents, this low product concentration reflects the microbe's low tolerance for these solvents. For organic acids the protonated species of the acids adversely affect the biosynthetic enzymes; neutralization of the acid is often required during fermentation.
Conventional unit operations—such as distillation, adsorption, liquid-liquid extraction, pervaporation, ion exchange, crystallization, and membrane separation—have been used for downstream purification and separation of fermentation-derived products. However, these separation technologies were developed for other systems, usually petroleum refining, and applying them to fermentation systems in a cost-effective manner will require much research. New concepts and methods must be developed to address the unique problems of fermentation products in aqueous solution, as must databases of product properties and ways to combine the processes into low-cost separation systems.
One example of a novel and environmentally benign solution was reported by Ladisch and Dyck (1979) in the replacement of benzene or pentane for removing the water in the ethanol-water azeotrope. They used corn as a readily available absorbent for removing the water in the 95 percent ethanol distillate. This approach has been applied only to starch-based ethanol fermentation due to the availability of corn as the raw material. It has been adopted in commercial-scale operations (NRC, 1992). A second novel example, in separation of organic salts produced by fermentation, is the use of electrodialysis combined with bipolar membranes to regenerate the alkaline agent for neutralization and simultaneously produce free acid. This approach avoids creation of undesirable byproduct (Bozell and Landucci, 1993).
New concepts need to be developed and past separation technologies should also be reexamined to reduce costs in downstream processes.
Page 101
These initiatives should focus on engineering and biological principles as well as combinations of both to improve product purification. New concepts through engineering might use the following approaches:
• supercritical fluid extraction;
• selectively permeable membranes, such as water retentive but solute permeable membranes;
• combination and integration of different separation principles, such as solvent extraction with membrane permeation;
• nonequilibrium or rate-governed processes;
• biomimiary separation technology that capitalizes on the ability of some biological molecules to selectively bind others.
Improved understanding of the inherent abilities of microorganisms—for example, of how solvents and acids impart their toxicity on producing organisms—could provide new ways to reduce downstream processing costs. Yeasts such as Saccharomyces cerevisiae can produce ethanol in excess of 150 grams per liter, and bacteria such as Acetobacter suboxydans can produce acetic acid (as free acid) in excess of 200 grams per liter. However, the mechanisms and molecular bases for these organisms' ability to tolerate and produce alcohol and acids at these high concentrations is not well understood. Exploration of such biological principles on a more fundamental basis could help improve the commercial viability of fermentation production processes.
Summary
The development of biorefineries will be a key approach to integrating food, feed, chemical, and fuel production in the future. Prototypes of the biorefinery already exist in corn, soybean, and wheat processing plants. These prototype biorefineries process 95 percent of incoming feedstocks into value-added products. Currently 18 to 19 percent of U.S. corn is industrially processed in prototype biorefineries. Pulp mills also represent a biorefinery prototype. Over the near term, stand alone biorefineries will be built around existing wet corn mills. Other near-term opportunities include sugar and wood product manufacturing plants as biorefineries. Future opportunities for biorefineries will be processing plants that efficiently convert plant lignocellulosics to liquid fuels and chemicals based on yet-to-be-proven, high-volume, low-cost processes. Biorefineries offer a number of potential advantages over petroleum refineries, including domestic raw materials, lower environmental impacts, and potentially greater sustainability.
Thermal, mechanical, chemical, and biological processes all play a
Page 102
role in the conversion of biological raw materials into industrial products. Some processes have been proven and used in the past but are not in wide use today. Examples of thermal, mechanical, and chemical processes include liquefaction, destructive distillation to methanol, fast pyrolysis of wood, biomass desiccation, protein fractionation, and chemicals from pentose sugars (e.g., furfural).
Biological processes also show great potential. The fermentation industry currently produces alcohol, organic acids, polymers, antibiotics, enzymes, and amino acids. Advances are occurring in reactor design and the selection and genetic modification of microorganisms. Modern tools of molecular biology are being used to improve yield by increasing product concentration and minimizing undesired byproducts. The major U.S. effort to compete internationally in the amino acid industry provides an example of such advances. Poly(hydroxybutyrate), a biopolymer, is produced by microorganisms and is being introduced into high-value niche markets. Enzymes are being used, for example, in the corn-ethanol industry to convert starch to glucose. Possibly the greatest success to date of enzymatic processing is the use of immobilized glucose isomerase to produce high-fructose corn syrup as a sugar substitute.
The development of lignocellulose treatments will be key to unlocking a major sugar source for biological conversion into industrial products. A number of new potential technologies exist, such as clean fractionation, AFEX, liquid hot water, and perhaps others. For example, the high-fructose corn syrup industry has demonstrated that immobilized enzyme processing and chromatographic separation of biomass can be efficient and economical on a large scale. More research is needed, however, to demonstrate the commercial feasibility of these approaches and to identify new processes.
Other research challenges lie ahead in the development of processing technologies. Advances are needed in the engineering principles for fermentation operations. Biological and engineering approaches should be combined to improve product yield, selectivity, productivity, and product purification. Finally, expansion of our fundamental knowledge of microbial physiology, biochemistry, and genetics will be essential in the development of improved biological processes.
For large-scale biobased products, the dominant factors influencing market share will be the cost of the starting raw material and the cost of processing technology to convert the raw material to the desired biobased products. Further reductions in the price of the biobased raw materials will help process economics but not as much as will reducing the costs of processing technologies. Very low-cost processing technologies must be developed if biobased industrial products are to penetrate commodity markets.





























