E-ANNEX 1Exposures to Miner Cohorts: Review of Estimates for the Studies
Colorado Plateau Uranium Miners
Introduction
Uranium mining in the Colorado Plateau expanded rapidly in the post-World War II period to include more than 200 mines by 1950 (see Time Line E-1). The start of an industry and the boom times did not lead to orderly administration and record keeping, c.f. Czechoslovakia and Ontario below. Moreover, some of the miners who worked in the mines during the post-war uranium boom had previously worked the same ore bodies for radium and vanadium without any accounting of exposure to radon progeny. Most of the early mines were small and depended on natural ventilation so that ambient temperature change was the driving force for exchange of the mines' air with outside air. Until 1967, mining operations were regulated only by the states where mining was taking place, even though all ore was sold to the Atomic Energy Commission. There was no requirement in place for measurement of exposure and there was not a federal standard for exposure to radon progeny. Consequently adequate ventilation practices were not uniformly introduced from the outset and the extent of radon measurement was initially quite limited. As a result, estimates of cumulative exposures to uranium miners on the Colorado Plateau were largely based on various estimation procedures rather than direct measurements relating to a particular mine shaft or even the mine where a given worker was exposed.
The history of radon exposures to the miners was described by Holaday (1969) and the approaches followed by the U.S. Public Health Service for esti-

mating exposures of individual participants in the epidemiologic study of Colorado uranium miners are described in National Institute for Occupational Safety and Health-National Institute for Environmental Sciences Joint Monograph No. 1 (Lundin and others 1971). A 1968 report of the Federal Radiation Council addressed the accuracy of the exposure estimates. SENES Consultants Limited of Ontario, Canada, has prepared a report entitled "Preliminary feasibility study into the re-evaluation of exposure data for the Colorado Plateau uranium miner cohort study" (SENES 1995). This report provides an extensive description of the calculation of the WLM values for the epidemiologic study and gives insights into the sources of variability and error in the estimates.
Estimation of WLM
The following description is taken largely from the 1971 monograph authored by Lundin and colleagues. The U.S. Public Health Service began surveying for radon in uranium mines in 1949. In 1950 they were joined by the Colorado State Department of Health and in 1951 by the U.S. Bureau of Mines for mines on Indian reservations. Coverage was far from complete; 1949 "a few measurements," 1950 "relatively few mines," 1951 "but again coverage was incomplete," (Lundin and others 1971). By 1952 an effort was made to survey all operating mines and radon progeny were sampled in 157 mines. This sampling may have examined most of the larger mines, but government records
indicate that over 450 mines shipped ore in 1951. Mining companies introduced radon surveys in 1956 and the state programs continued through 1960. Both company and state-sampling efforts were made in work areas for information purposes, not for control purposes, and "are considered to be representative of the areas in the mines in which miners were exposed" (Lundin and others 1971). This early data base is of primary importance in considering the adequacy and precision of miner's exposure estimates as utilized in epidemiology assessments of risks due to radon since a large portion of the cumulative exposure occurred in the 1950's.
By 1960, exposure levels had dropped precipitously in anticipation of Colorado's adoption of a 10 WL shutdown level in 1961. However, regulatory control probably reduced the validity of the measurements in mines for epidemiologic purposes. As outlined in Joint Monograph No. 1, the most complete description of the Colorado Plateau miner data (Lundin and others 1971) "Most radon daughter measurements available from Colorado, Utah, and Wyoming after 1960 were made by mine inspectors who measured air samples primarily for control purposes." This may have led to bias in the estimated exposures. As noted by Lundin and others (Lundin and others 1971), "Proportionately more measurements were made in sections of mines having high levels which tended to yield radon-progeny values greater than would have been obtained by sampling all work areas with equal frequency." In addition more measurements were concentrated in mines having high levels of radon. The U.S.P.H.S. investigators who assembled the data base for estimating cumulative exposures chose to exclude company measurements made after 1960 on the grounds that they might have been "minimized to avoid regulatory action." The aim was clearly ''to assure a consistent direction of bias, that is, over estimation of radon daughter levels" (Lundin and others 1971).
Even though the number of radon-progeny measurements increased during the 1960's, the number per mine increased only slowly from about six in 1960 to almost 12 in 1968 (Figure E Annex 1-1). Measurements of radon progeny in a particular mine were never extensive and, more importantly, were not made on even a once per year basis in the majority of mines. Only 341 miners, about 10% in the Colorado Plateau miner cohort, had their exposure assignments based on measured radon-progeny concentration. For the majority of the miners, information on measured levels was combined with estimates made using a variety of methods as described by Lundin and others (1971).
Many of the uranium miners were also employed as hardrock miners or previous to 1950 some had mined the same ore bodies, where uranium was found, for radium, vanadium etc., particularly in the Urivan Mineral Belt in Colorado. In the epidemiologic study, hardrock miners were assigned an exposure level of 1 WL for mining that occurred before 1935, 0.5 WL for 1935 through 1939, and 0.3 WL for later years (Lundin and others 1971). No information is given as to the basis of these estimates but a statement is included in Joint Monograph No. 1
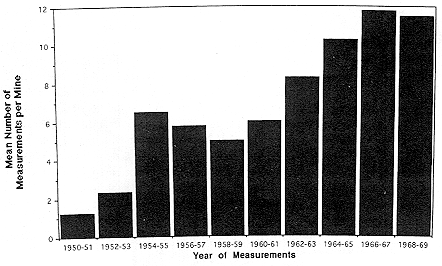
FIGURE E ANNEX 1-1 Frequency of radon-progeny measurements on the Colorado Plateau in two-year intervals 1950–1969. Source: Presentation to the committee of analysis of the data tapes for the Colorado Plateau miners by Duncan Thomas and Dan Stram, September 1995.
(Lundin and others 1971) which indicates these estimates were thought to have been too high and that the average exposure was less.
A re-evaluation of a sample of the Colorado Plateau cohort for exposure during hardrock mining is described in Monograph 1. This reassessment indicates that the tabulation of hard rock mining duration was subject to error and that misclassification of exposure was fairly common for that portion of a cohort member's work experience. For example, for a sample of 101 cases and 202 controls, misclassification was only about 10% for cumulative exposures of less than 20 WLM but 50% or more at higher levels. Nevertheless, hard rock mining may be a relatively unimportant source of exposure compared to the mining of uranium-bearing ores for which exposure levels were often much higher than 1 WLM.
Because relatively few mines were initially monitored for radon or radon progeny, exposure estimates in uranium mines that occurred before 1951 were referred to as "guesstimates" in Joint Monograph No. 1 (Lundin and others 1971). According to that report, "guesstimates" were made on the basis of knowledge concerning ore bodies, ventilation practices, emission rates from different types of ores, and such radon or radon progeny measurements as were performed in 1951 and 1952.
For mining that occurred after 1950, three other methods were used to estimate exposure levels. By far the most common was a process called area average estimation. This consisted of using the available, albeit often sparse, measured values to estimate concentrations in a given locality to obtain an "area average." In order to reduce sampling variability for these area averages it was required that three or more mines and ten or more samples had to be available for a locality in a year, otherwise the locality was assigned the average for the district in which it was located (Lundin and others 1971). If sufficient data for a district were not available, a state average was used or, in a few cases for which state data were insufficient, data for the state of Colorado were used. The degree to which area estimates were used to obtain exposure estimates is not often appreciated. Area estimates account for most of the exposure assignments throughout the study period of the Colorado Plateau cohort (Figure E Annex 1-2) Monograph 1 implies that when an individual mine was thought to differ appreciably from others in the same locality due to its ore quality or mining practices, guesstimation was substituted for an area average.
To complete gaps in the measurements in calculating individual WLM estimates, a system of extrapolation, interpolation, and expert judgment was used to estimate the exposure in mines monitored less frequently than once a year. For mines with actual measurements at least once every five years, working-level estimates were obtained by interpolation, that is, averaging the measured values
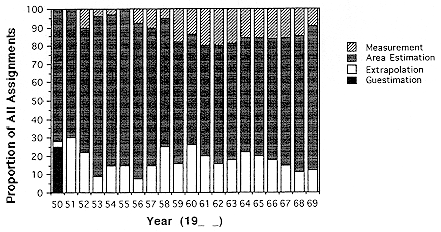
FIGURE E ANNEX 1-2 Bases for the assignment of exposure estimates by calendar year 1950–1969. Source: Presentation to the committee of analysis of the data tapes for the Colorado Plateau miners by Duncan Thomas and Dan Stram, September 1995.
(Lundin and others 1971). Approximately 20% of the exposure assignments were made using this method (Figure E Annex 1-2). An assessment of this extrapolation procedure described in Monograph No. 1 indicates that it tended to overestimate exposures in the early years of mining but became more valid in the 1960s as information from more frequent measurements became available.
For mines with yearly monitoring information available, the measured concentration was used to assign a worker's cumulative exposure in a given year. Table IV-3 in the BEIR IV Report (NRC 1988) indicates that the number of measurements per year per mine surveyed was usually between 10 and 20 after 1959 so that the measured values provide a reasonably stable estimate of the average working levels in those areas monitored. However, the average number per mine was somewhat less, 8–9 as illustrated in Figure E Annex 1-1. Although nearly 43,000 measurements were obtained (Lundin and others 1971), there were about 2,500 mines and measured concentrations were not a frequent method of exposure assignment. Figure E Annex 1-2 indicates that from 1959 to 1969 only 10–20% of the exposure assignments in a given year were based on direct measurement of radon progeny concentration and that even fewer were made on such direct information prior to 1959, when exposure levels were, on the whole, much higher.
Assessment of Errors in the WLM Estimates
A comparison of exposure estimates in relation to calendar year is given in Figure E Annex 1-3 for each assignment method. Except for 1950, estimates based on the extrapolation procedures are in reasonable agreement with those based on direct measurement while area average estimates tend to be somewhat greater than obtained by other methods. This may in part be due to measurements having been made more frequently in large mines having more employees and because of larger capital investment in better ventilation.
The degree of variation in exposures among workers in a given mine was not well characterized. Before 1960 mechanical ventilation was not commonly used and a near equilibrium between radon and progeny was probably the rule under conditions of convective ventilation as indicated by the early data described by Holaday (1969). There appears to be no information on aerosol size distribution or even the unattached fraction in early mines. Even though diesel power was not common, compressed air or electricity was used to operate equipment including ore cars; dust was plentiful from drilling and hauling operations so that it is likely that the unattached fraction was low.
An extensive study of air quality in nine uranium mines was carried out by the AEC Health and Safety Laboratory (HASL), now the DoD Environmental Monitoring Laboratory, in 1967–1968. Mines were selected by the U.S. Bureau of Mines to represent a cross section of the uranium mining industry (Breslin and others 1969). This investigation was in response to the concerns expressed at the
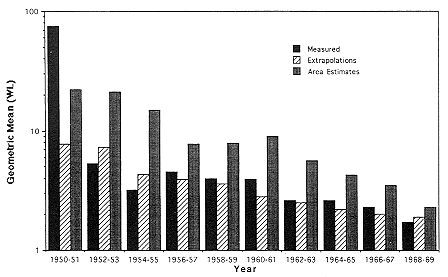
FIGURE E ANNEX 1-3 Comparison of mean WL estimation by various methods in two year intervals 1950–1969. Source: Presentation to the committee of analysis of the data tapes for the Colorado Plateau miners by Duncan Thomas and Dan Stram, September 1995.
Joint Committee on Atomic Energy hearings in 1967 in which the validity of exposure and early risk estimates of increased lung-cancer in miners were questioned. A particular point in question was "the extreme variation of atmospheric characteristics within a mine and among mines"; the HASL study was directed at exploring this question (Breslin and others 1969).
The nine mines studied ranged in size from having two to 112 workers. Ore production varied from 150 to 11,000 tons per month. Mechanical ventilation rates varied from 5,600 to 100,000 cu. ft. per minute. Given this range of conditions, atmospheric conditions were surprisingly uniform, giving some credence to the validity of the estimation methods described above. In most of the mines the variation in radon-progeny concentration at different times and locations was only occasionally as large as a factor of two and 80% of the time had a coefficient of variation of 30% or less. The average WL ratio (pCi progeny to pCi radon) averaged 0.23 with a geometric standard deviation of 1.6 and showed limited variation with the absolute level of radon progeny. Equilibrium values F were also in a narrow range: about two-thirds were between 0.20 and 0.30; mode 0.25. Polonium-214 was most often at 16% of the equilibrium value, range 0.09 to 0.49.
Simultaneous measurements of radon progeny were made at various locations in stopes (mining chambers) and in drifts (tunnels). While drifts showed
greater variation than stopes, as indicated in Figure E Annex 1-4, the report's authors indicated that sampling location was not critical within a radius of 10 to 20 feet of the miners' location and that breathing-zone sampling was unnecessary. Similarly, the HASL study indicated that differences between various mining operations, for example, drilling, mucking, etc., had little effect on the measured working level (Figure E Annex 1-5). No measurements were taken immediately after blasting but such areas would not have been occupied because of other safety considerations. While the HASL study does indicate that mine-wide averaging probably provides a useful measure of worker exposure in the mines studies, this is probably less accurate for the high exposures which occurred before the introduction of mechanical ventilation in U.S. uranium mines.
The recent report from SENES Consultants Limited provides additional relevant information. Tables for several mines demonstrate substantial variation in WL values within a mine during a single visit by an inspector, typically one day. For example, U.S. Bureau of Mines data for one Utah mine in 1968 showed variation from 0.4 to 5.4 WL across the mine (Table-E Annex 1-1).
The Public Health Service investigators used self-reported mining history as the basis for estimating time spent underground in specific mines. This information was collected both retrospectively and prospectively during the annual miner censuses. The possibility of error in these histories has been acknowledged. The SENES report provides a series of case descriptions documenting inconsistencies in these histories and gives a compilation of exposure estimates for 78 miners for whom exposures have been calculated for both the epidemiologic study and for other purposes. Substantial variation is evident in these
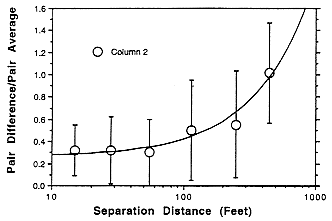
FIGURE E ANNEX 1-4 Variation of radon concentration with distance in ventilated uranium mine drifts on the Colorado Plateau (Breslin and others 1969).
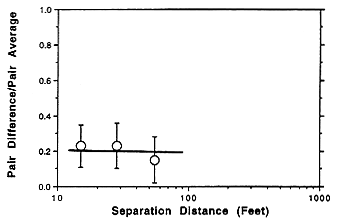
FIGURE E ANNEX 1-5 Variation of radon concentration with distance in ventilated uranium mine stopes on the Colorado Plateau (Breslin and others 1969).
estimates, largely reflecting various discrepancies in the alternative work histories used for the purpose of estimating the exposure.
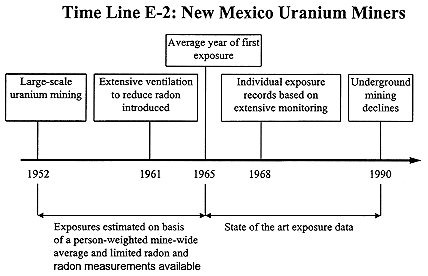
New Mexico Uranium Miners
Large-scale uranium mining began in the early 1950s (see Time Line E-2) with the opening of the Jackpile mine, an open-pit mine. By the late 1950s, a number of large mines were operating at Ambrosia Lake and the Churchrock mining district became active in the late 1970s. The industry continued operating into the early 1990s, longer than in other U.S. locations, so that miners working after 1968 have individual exposure records (work location estimates and estimates of exposure) for this period of employment. These were calculated based on area measurements and work locations. For the most part, post-1968 employment was in very large industrial operations with state of the art ventilation. Mean annual exposures in 1968 were about 3.8 WLM and declined to 1.2 WLM or less by 1972 (Samet and others 1986b). Earlier exposures were not estimated as accurately, although the State Health Department and the State Mine Inspector had implemented active measurement programs by the late 1950s. The state implemented a progressively more stringent series of shut-down concentrations. As for the Colorado Plateau miners (see above), median annual exposures were considerably larger during the earlier years of the industry, about 30 WLM in the 1960's. Some members of the New Mexico cohort, who had also mined in the Colorado Plateau, had annual exposures as high as 300 WLM or more (Samet and others 1991).
Investigators directed substantial effort at tracing employment histories for the purpose of estimating the cumulative exposures for those employed before exposure estimates were individualized (Samet and others 1991). The miners' underground employment and exposures in specific mines were traced by examining company personnel records and self-reported work histories taken at the time of periodic medical examinations. Estimated exposures for miners who had worked underground on the Colorado Plateau were supplied by the USPHS (Lundin and others 1971; Samet and others 1991). Contributions to the total mean exposure from various information sources are listed in Table E Annex 1-2 (Samet and others 1991).
With the notable exception of those members of the work force employed on the Colorado Plateau, this cohort probably has maintained the most extensively documented exposure estimates. In this regard, it should be noted that the state of New Mexico had more extensive and more frequent monitoring for radon then was common elsewhere in the early 1950's when exposures were very high (Lundin and others 1971). From 1957 to 1967 exposure estimates are based on 20,086 measurements taken during 1,886 visits. Most annual exposures were relatively low during this period, mean 4-5 WLM per year, so that this cohort has a large sub-cohort of miners exposed at low rates and relatively low cumulative exposures.
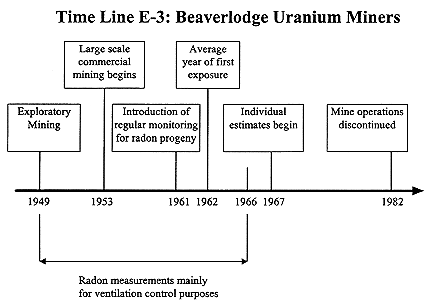
Beaverlodge Uranium Miners
The BEIR-IV report also includes a description of the exposure estimates for this cohort (NRC 1988). Exploratory uranium mining at Beaverlodge,
TABLE E ANNEX 1-1 U.S. Bureau of Mines February 1968 survey at North Alice Mine, Utaha
|
No. of Men |
Location, Operation |
Estimated Average Full Shift Exposure to Radon Daughtersb (WLc) |
|
2 men, night shift |
416 NE from 360 NW; mining |
0.5 |
|
2 men, day shift |
236 from 325 S; mining |
1.7 |
|
1 man night shift |
|
|
|
1 man day shift |
240 W incline station and hoist |
1.0 |
|
1 man night shift |
|
|
|
1 man day shift |
248 SE from 225 N; mining |
5.4 |
|
2 men, night shift |
242 S from 190 W; mining |
3.6 |
|
1 man day shift |
|
|
|
1 man night shift |
240 W incline to main incline; tramming |
2.2 |
|
2 men, night shift |
100 S area; mining |
0.6 |
|
2 men, day shift |
147 N from 130 E; mining |
2.8 |
|
3 men, day shift |
|
|
|
2 men, night shift |
128 S from 145 E; mining |
1.5 |
|
1 man day shift |
|
|
|
1 man night shift |
main incline; trip rider |
0.4 |
|
1 man day shift |
all areas; electrician |
1.4 |
|
2 men |
all areas; mechanics |
1.6 |
|
5 men |
all areas; shift bosses |
1.4 |
|
3 men |
all areas; staff |
1.0 |
|
3 men |
all areas; bratticemen |
2.6 |
|
a This table is from a February 1968 report on a Radiation Survey prepared by U.S. Bureau of Mines, obtained from SENES 1995. b Average Levels are estimated from information gained by questioning the miners about where there time is spent and weighing the radon daughter concentrations in each place by the time spent in that place. c NIOSH database: 1967 WL is 1.3, based on 39 measurements; 1968 WL is 3.3, based on 120 measurements. |
||
Saskatchewan started in 1949 and commercial production began with a greatly expanded labor force in 1953 (see Time Line E-3). Radon monitoring was carried out in 1954 and 1956 but only sporadically until the end of 1961. A number of radon-progeny measurements were also made at this time but monitoring was mostly for radon and viewed as a check on ventilation rather than as a tool for exposure control. Nevertheless, the frequency of radon-progeny measurements increased and by 1961 exposure records were maintained for all full-time underground employees. These records listed each worker's occupancy time at each work place on a daily basis. In 1970 worker's exposure records were estimated retrospectively to 1 November 1966 and in 1971 part-time underground workers were included in the exposure assessment (SENES 1989).
TABLE E ANNEX 1-2
|
Source of Information on Underground Employment |
Contributions To Mean Cumulative Exposure |
|
Work outside New Mexico |
24.9 WLM |
|
New Mexico Employment records |
59.5 WLM |
|
Self reported work histories |
11.8 WLM |
|
Company individual records (1967 and later) |
10.0 WLM |
|
Other (1967 and later) |
5.2 WLM |
Two assessments of lung-cancer risk observed in Beaverlodge miners have been made by Howe and colleagues (Howe and others 1986; Howe and Stager 1996) using two related but differing exposure estimates. The first of these estimates was prepared by Frost (1983) who, observing a wide dispersal in the recorded concentrations in a given year, assigned the median of this quasi lognormal distribution as the best measure of exposure. Although, it was possible to assign work locations for service personnel, for miners, mine-wide medians were used in the cohort study reported by Howe and colleagues (1986).
A reassessment of the Beaverlodge exposure estimates was carried by SENES Consultants, Ltd. at the direction of the Atomic Energy Control Board (SENES 1989, 1991). This included a painstaking reconstruction of mining activity and its correlation with exposure information for the Beaverlodge mining complex. The revised exposure estimates were used by Howe as the basis for a recent case-
control analysis of lung-cancer mortality (Howe and Stager 1996), which used cases and controls from a previous analysis that used the original exposure estimates (L'Abbe and others 1991). In the new analysis, averages rather than medians of the individual measurement data were used to estimate exposure levels in a given location. There was also a systematic consideration of the locations where mining took place at a given period of time and, in many cases, individual miners could be assigned to a given mine face, as recorded in bonus-pay information, removing in some measure the radon error inherent in using mine-wide averages.
The effects of these changes is illustrative of what an improved exposure assessment can accomplish. The exposure estimates for each of the miners in the case-control study were compared to those used in the original cohort and case-control studies. In general, the more recent exposure estimates were considerably higher than the original estimates, the mean exposure increasing from 50.6 WLM to 81.3 WLM. There is evidence that the new estimates reduced exposure misclassification. Table E Annex 1-3 compares the cumulative exposure estimates used in the original cohort study to the newer estimates for the case-control study.
Because of the wide intervals of grouped exposures, most workers remained in the same exposure category even though their estimated exposures were on the average considerable larger in the revised exposure estimates. However, there was a decrease in the number of workers receiving low exposures and a corresponding increase at higher levels. For example, the number of workers in the 200+ WLM group increased from 10 to 15 (Table E Annex 1-3). Because there is less misclassification of estimated exposures, the slope of the regression of risk on cumulative exposure is increased even though the estimated exposures increased. The original estimate of the excess relative risk from the case-control was an excess relative risk of 2.70% per 100 WLM while the revised exposure assessment was 3.25% (Howe and Stager 1996).
TABLE E ANNEX 1-3 Number of miners in each exposure category
|
Cumulative Exposure WLM |
Cohort Study |
Case-Control Study |
|
0 |
43 |
42 |
|
1–24 |
90 |
80 |
|
25–49 |
15 |
18 |
|
50–99 |
15 |
17 |
|
100–199 |
18 |
19 |
|
200+ |
10 |
15 |
Ontario Uranium Miners
Uranium mining in Ontario, Canada, started in 1953, somewhat later then in the United States and was conducted in relatively few mines in comparison to the United States (see Time Line E-4). A 12 WLM annual limit was adopted in 1954 with a concomitant decrease in annual exposure thereafter. Radon measurement and radon control programs were instituted within two years of the start of mining. Except for exposures occurring before 1958, exposure estimates are largely based on actual measurements (SENES 1989). However, some Ontario uranium miners had worked earlier as gold miners and were exposed to both radon progeny and arsenic in those operations. These miners had an estimated average cumulative exposure 2 WLM due to gold mining (Kusiak and others 1991) compared to an average of 30 WLM in uranium mines (Kusiak and others 1993). Even for those with gold mining experience, the approximated exposures from gold mining are only a minor portion of the total exposure.
The BEIR IV report provides a complete description of the exposure estimates for their cohort (NRC 1988). The radon-progeny measurement program was extensive: 131,000 measurements in 15 mines. Exposures were estimated using different methods for 1967 and earlier years and for 1968 and later years for which WLM estimates made by the companies were used. For 1957–1967, WLM were calculated by combining WL data with work histories. Two separate sets of estimates were derived for these years: the ''standard" or lower WL values were
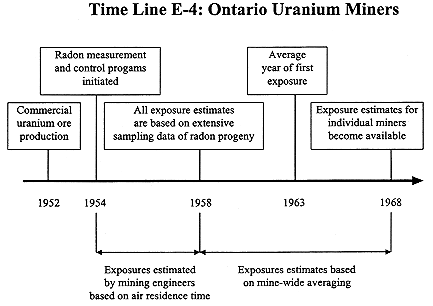
the averages of the four quarterly averages or three four-month averages for a particular year while the "special" or upper WL values were a weighted average of the four highest quarterly measurements or the three highest four-month measurements in headings, stoops, raises, and travel ways. The differences between these two sets of values varied by mine and by year, with the special values being up to four times as high as the standard values. The investigators considered that the true exposures were bounded by the two sets of values. For its analysis, the BEIR IV committee used the WLM values based on the standard WL values. Some estimation of exposures for the earliest years of the industry, before 1954, required extrapolation from measured values, taking into account such factors as ventilation. These years included the highest exposures and consequently 22 percent of the total WLM accumulated by the cohort was based on extrapolation of measured values.
Port Radium Uranium Miners
The approach for exposure estimation for the Port Radium miners is well documented in a 1996 report by SENES Consultants Limited (1996b). Underground uranium and/or pitchblende mining at Port Radium started in 1932 and continued, with a two-year interruption, until 1960 (see Time Line E-5). Because records of employment before 1940 were not available, exposures occurring before that date have not been accounted for (Howe and others 1987, SENES
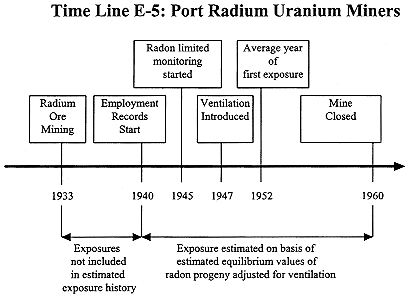
1989). Exposures occurring after 1939 have been estimated on the basis of rather sparse monitoring data for radon. Between 1945 and 1957, 261 radon measurements were made in seven years of this period, with from nine to 71 samples in an individual year. A few radon-progeny measurements were made and only three paired measurements of radon and progeny were obtained.
Absent information on concentrations of radon progeny, the equilibrium between radon and its progeny was estimated on the basis of knowledge of mine operations and by analogy from the Beaverlodge mine and the radon concentrations were then converted to concentrations of progeny. Although ventilation was introduced in 1947, it apparently was used in limited fashion during the winter season because of the cold. Consequently, the approach to estimating the WL values considered seasonal variation in equilibrium factor. The SENES report provides detailed documentation of the assumptions made in estimating the WL values from the radon measurements.
Reported radon concentrations were extremely variable ranging from 50–300,000 pCiL-1 and it is thought that before ventilation was introduced, some exposures could have been as high as 1,000 WLM per year (SENES 1989). Unfortunately, such large annual exposures could not be assigned to the involved workers unless the exposures took place after 1940 because of the missing work-history information before 1940.
The potential limitations of the exposure data were acknowledged in the initial report on the findings of the epidemiologic study of Port Radium miners (Howe and others 1987). SENES Consultants Limited (1996b) have recently re-estimated exposures to radon progeny for 171 miners included in a case-control study (see Table E Annex 1-4). For the 171 miners, employment histories were reconstructed and used with revised WL estimates to calculate WLM. Substan
TABLE E ANNEX 1-4 Summary of differences between the SENES reevaluation of miner exposures and epidemiology exposures used by Howe and others (1987). Data from 171 minersa
|
Months Worked |
Months Worked |
WLM |
|
Mean difference |
-1.64b |
-5.2d |
|
Maximum |
66.93 |
2908 |
|
75th percentile |
0.26 |
69 |
|
Median |
-0.23c |
0d |
|
25th percentile |
-1.97 |
-12 |
|
Inter quartile range |
2.24 |
80 |
|
Minimum |
-38.04 |
2348 |
|
a Based on SENES 1989, table 4.1. b Mean difference significantly different at 5% level. c Median difference significantly different from 0 at 1% level. d Median difference and mean difference not significantly different from 0. |
||
tial differences were found in individual estimates, although the mean WLM values were comparable for the two sets of estimates. There were large differences in employment duration for some of the men and changes in estimated exposure as large as 2900 WLM were found. The report comments on key sources of uncertainty in the exposure estimates for the Port Radium miners: incomplete employment histories for other employment; lack of employment information for years before 1940 when exposures were extremely high; and the numerous assumptions made in calculating the WL values from the radon measurements. Finally, the report also indicates that Port Radium ores contained "significant concentrations of various elements including for example arsenic, nickel, and cobalt."
Czechoslovakia Uranium Miners
A cohort of miners in Czechoslovakia, who started uranium mining between 1948 and 1957, has been described extensively in the literature by Sevc and Placek (1976), Sevc and others (1988), Kunz and others (1978, 1979), and more recently by Tomásek and others (1993, 1994a,b). This cohort is often designated as group S by the Czech authors (see Time Line E-6). Compared to miner studies in other countries, exposure information for group S is among the most extensive. Measurement of radon and other potentially hazardous materials had become routine in Czech mines before 1948 so that estimation was not necessary for periods of employment during which radon measurements were not made. Even so, exposure estimates for radon-progeny exposures prior to 1961 are based on radon concentrations as concentrations of progeny were not measured.
These radon measurements were, however, extensive, the annual number varying from 100 to 700 per shaft (Sevc 1993). Estimates of WLM for years before 1961, during which radon measurements only were made, were based on F values taken during periods of ventilation failure which occurred in 1969 and 1973. These data indicated an F value of 0.86 for the earliest years 1948–1952. From 1961–1969 the mean number of annual radon-progeny measurements recorded per shaft was 952, about 3 per work day.
In 1953, natural ventilation was augmented by mechanical means and F values decreased to an estimated average value of 0.55 in 1953–1959 and to 0.36 thereafter. Czech investigators estimate the coefficient of variation in converting radon levels to working levels as 28%. The fraction of unattached plutonium-214 has been estimated by Czech investigators as 0.1 (Hamilton and others 1990).
Estimated exposures of these miners have recently been reevaluated (Tomásek and others 1994a,b). The principal change appears to have been a more thorough investigation of workers' employment histories to take account of prior mining experience and the assignment of exposure for each month based on the particular shaft in which the miner worked. In any event, the newest account given below is more complete than those published previously. Additional background can be found in a report of a WHO-sponsored trip to Czechoslovakia made in 1988 by L.D. Hamilton, L.W. Swent, and D. B. Chambers (published by SENES Consultants Limited—see Hamilton and others 1990).
"During 1949–1963 about 39,000 measurements of radon gas were made in the 19 mine shafts in Jachymov and Horoni Slakov in which the men were employed. Some men also worked, particularly after the closure of most shafts at Jachymov and Horoni Slakov in 1963, at other Czechoslovak uranium mines, and substantial numbers of measurements were also made in these mines. An initial review of exposure estimates used in previous reports found a considerable number of errors, and for some miners a part of their employment histories had not been taken into account. Therefore, exposure estimates have been completely revised for the present analysis, based on a review of all available information. The radon-gas measurements were converted into estimates in terms of working levels using equilibrium factors based on radon-progeny measurements made after 1960, and on data collected during two accidents in the uranium mines at Zadni Chodov in 1969 and at Pribram in 1973, when mechanical ventilation was stopped for at least a month. An estimate of each man's exposure in each month in terms of working months (WLM) was calculated from the time he spent in each mine shaft in conjunction with the year- and shaft-specific WL estimates. Men worked 6 days per week with 1-month of holiday each year. For most men underground work was assumed to last 8 hours per day, but for geologists, safety and ventilation technicians, and emergency workers, it was estimated that 70% of working time was spent underground, while for managers 50% was estimated. About 300 men were involved in exploratory work, which was normally carried out in shallow shafts near the surface. Explicit radon measurements are not
available for this work, but exposures are thought to have been low, and are estimated at 3.3 WLM per year" (Tomásek and others 1994a).
With regard to the magnitude of exposure error, it is of interest that less than 5% of the new exposure estimates differed by as much as 50% from those used in previous analyses of these data. The mean cumulative exposure in the new evaluation is 219 WLM compared with 227 WLM in the older work (Sevc 1993). Presumably, random error as well as systematic biases were reduced in the reevaluation. Using a simple model in which the estimated relative risk is linearly related to the cumulative exposure yields a relative risk of 0.37%/WLM (95% C.I. = 0.18–0.55) with the old exposure estimates. With the new ones the estimated relative risk is 0.61%/WLM (95% C.I. = 0.29–0.8) (Tomásek and others 1994), providing evidence that the dose-response was flattened by errors in the original exposure estimates.
French Uranium Miners
Uranium mining in France started in 1946 with exploratory operations that continued through 1948 when extensive commercial operations commenced (Tirmarche and others 1984, 1993) (see Time Line E-7). The first reported radon sampling occurred in 1953 when 40 measurements were taken, an average of 10 per mine. Large-scale radon monitoring began in 1956 when forced ventilation was introduced. Exposures prior to this date have been estimated retrospectively by an expert group which considered mine characteristics and type and duration of work. For this early period, before forced ventilation, exposures were relatively high and varied substantially between individuals. The estimated median annual exposure from 1947 through 1955 was 11 WLM and varied for the 3rd quartile up to 55 WLM per year.
Exposures declined rapidly after forced ventilation was introduced, median exposures averaging about 3 WLM per year from 1956 to 1975 with a further decline to 1 WLM by the early 1980's. About half of the French miners started their underground employment before 1956 but most of their person years of exposure occurred after monitoring became comprehensive, ventilation improved, and exposures were relatively low. Nevertheless, for a large number of the French miners, a major portion of their cumulative exposure was based on estimations by experts for the period when ventilation was poor and routine monitoring lacking. After sufficient monitoring data became available, worker-exposure assessments were individualized to some extent by considering the type and location of work performed. Recently, personal dosimeters, using track-etch dosimeters, have provided direct information on individual miners. This has allowed a comparison of exposure based on area monitoring in 1982 with direct personal measurements in 1983. The comparison indicated that annual exposures based on area monitoring and work locations were, on the average, underestimated by almost 30% (Bernhard and others 1984; Piechowski and others 1981). Con-
sidering the year-to-year variation in the true levels and miner location, as well as the accuracy of the personal dosimeters, the reported difference may not be indicative of a significant bias in the exposure estimates.
Radium Hill Uranium Miners
The Radium Hill mine began operations in Australia in 1952 and radon monitoring began two years later (see Time Line E-8). In estimating exposures, exposure levels in the prior years were assumed to be the same as in early 1954 (Woodward and others 1991). A total of 56 samples were collected by 1 April 1955. Early radon concentrations were low (estimated 1.8 WL) even before forced ventilation was introduced and declined substantially thereafter—range 0. 10–0.55 WL. Apparently only radon concentrations were measured; WL concentrations were estimated by means of a calculated equilibrium factor based on ventilation rates and air volumes at various locations but such methods do not account for plateout and recirculation of progeny.
Enough radon measurements were made to allow exposure estimates by job category for work after 1 April 1955. The estimated exposures for workers show an exponential distribution with a median exposure of 3 WLM; a mean of 7 WLM and a 3rd quartile limit of 7.4 WLM. A few heavily exposed workers received about 80 WLM (Woodward and others 1991).
Chinese Tin Miners
Exposure assessments of Chinese miners employed by the Yunnan Tin Corporation are largely retrospective as no measurements of either radon or radon progeny were made prior to 1972 when mechanical ventilation was introduced (see Time Line E-9). Exposure estimates for two periods prior to 1972 reflect changes in the mining industry that occurred after nationalization in 1949. Before nationalization, mining was conducted in small mines with back hauling performed manually, often by children (Xuan and others 1993). To estimate exposures under these conditions, 117 measurements were made in 13 local mine pits that had been in operation before the large-scale expansion of the tin mines that started in 1953.
Exposure estimates for miners employed between 1953 and 1972 were based on 413 measurements obtained in the 1990's by recreating conditions in tunnels and galleries in original areas or in similarly configured areas in nearby mines that used techniques similar to those in the index year (Xuan and others 1993). Evidently, there was little change in radon progeny concentration in the larger, post 1953 mines. The reported average mean WL before 1950 was 2.3 ± 0.8 and 2.2 ± 1.2 thereafter. Mechanical ventilation was introduced rather slowly with priority given to new tunnels. Working levels decreased moderately in 1971–1975 to 1.7 ± 1.1, to 1.2 ± 0.8 in 1980 and 0.9 ± 0.3 in 1985 (Xiang-Zhen and others 1993). Exposure estimates from experience since 1972 have been based on over 26,000 measurements of radon progeny.
Little information is available on other characteristics of the occupational environment. Equilibrium factors, F, were measured in 1977–1978 and found to vary from 20% to 84% with a mean of 72% in "small pit operations" and of 62% in "larger tunnels" (Lubin and others 1990b). Evidently the mean of 72% refers to conditions prior to 1953 and the mean of 62% to the expanded operations. In any event the mines were very dusty by contemporary standards. Airborne dust was first measured in the 1950's and maximum levels were between 20 and 192 mg/m3. Wet drilling was introduced in the late 1950's and became widespread in 1964 when dust levels fell to about 6.2 mg/m3 (Xiang-Zhen and others 1993). Given these levels of dust, it is probable that equilibrium levels remained rather high throughout and that the unattached fraction was small.
For the epidemiological studies, workers were assumed to be exposed to radon progeny seven hours per day. For exposures occurring after 1972, estimates of exposures were adjusted by the worker's job title to take account of those exposed intermittently (Lubin and others 1990b). Exposure to arsenic in airborne dust was also accounted for in these studies and shows a large decrease over time from 0.4 mg/m3 in the mid 1950's to .01 mg/m3 in 1985. The radon exposure estimates for the Yunnan tin miners are not very well documented but, given the apparently uniform level of exposure throughout the period of miner employment, about a factor of 2, the estimates may be less subject to errors in estimation then for those uranium mines where exposure levels varied over time by factors of ten or more due to changes in ventilation practices. Arsenic exposure, on the other hand, did decrease appreciably as wet drilling became standard practice.
Newfoundland Fluorspar Miners
Underground mining for fluorspar started in 1936 in Newfoundland, Canada, and continued for more than three decades before monitoring for radon and radon progeny was initiated in 1960 (see Time Line E-10). Radon levels were found to be highly variable, range 0–190 WL (Morrison and others 1988). Ventilation was immediately introduced and in 1960 levels declined to an average of 0.5 WL (1960–1967) and then to an average of 0.17 WL (1969–1978) (Morrison and others 1988). Therefore, an average worker for the entire period of radon control would have accumulated about 30 WL as opposed to an estimated average exposure for all cohort members of 382.8 WL.
Exposure estimates for epidemiological purposes have been developed by Dory and Cockill (1984) and are described in SENES 1989. Exposure estimates for the period before 1961 were based on maps of the various mines, reports by mine inspectors, and workers' recollections. Apparently experience gained when the mines were monitored was also taken into account as well as the entry of water, the source of the radon, into the mines. Eventually a computational model was developed to simulate the annual radon progeny concentration in each mine so as to yield "average workplace concentration for high, medium, and low areas" (SENES 1989). For epidemiological purposes, workers have on the basis of their jobs been assigned to approximate areas of radon concentration, high, medium, etc. for a particular year and cumulative exposures estimated on this basis.
For exposures occurring after 1960, worker job records and monitoring data have been used to assign individual exposure estimates. As noted above, this period of employment is likely to be relatively unimportant for risk estimation because of the relatively small exposures. It is impossible to estimate the accu-
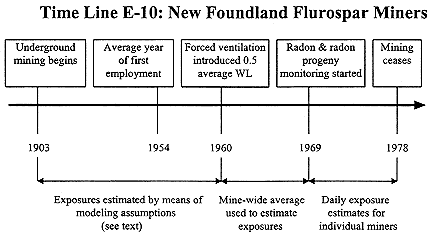
racy of the exposure estimates for before 1960. Radon concentrations in mine water varied form 300 to 1300 pCiL-1 in 1960 (Report of the Royal Commission 1969). Under such conditions, actual exposures must have been highly variable and in spite of the thorough assessment performed by Corkhill and Dory, the estimated cumulative exposures are guesstimates c.f. Colorado Plateau miners.
Swedish Iron Miners
The BEIR IV report provides an extensive description of exposure estimates for this cohort (NRC 1988). Additional description can be found in a report submitted to the BEIR IV committee, ''Comments to the U.S. Mine Safety and Health Administration for the American Mining Congress", prepared by L.W. Swent and D.B. Chambers. This report describes a visit to the mine by Swent and Chambers and their discussion with mine personnel. Much of the material is described in the 1989 report by SENES Consultants Limited (1989).
Exposure estimates for the Swedish iron miners are primarily retrospective. The cohort includes those who started work earlier but for the cohort as a whole the average year of first exposure was 1934 (see Time Line E-11). The first extensive measurements of radon in these mines were not made until 1968; radon-progeny measurements were initiated somewhat later. Most of the mine radon came from water seepage and there is limited evidence that this source was relatively constant in strength from 1915 to 1972. Comparison of radon measurements in water taken in 1915 with data from 1972 and 1975 indicated constant groundwater concentration of radon. Exposures have been estimated using the assumption that levels of radon progeny were constant until forced ventilation was introduced in 1972 (Radford and St. Clair Renard 1984). This assumption
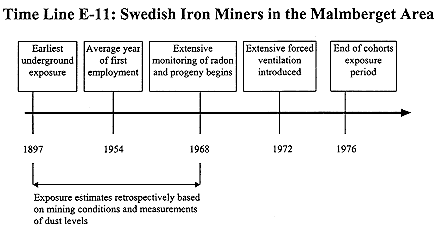
was supported by consideration of the pattern of natural convection and by data on quartz dust concentrations that extended to the 1930s. On the basis of their visit, Swent and Chambers have questioned the assumption of stable ventilation and suggest that the estimates of exposure were low by a factor of two or more due to air recirculation and changing ventilation conditions as the mines became deeper (SENES 1989). Because there are no measurement data for years before 1968, it is a matter of speculation as to how much exposures varied with time. It is likely, however, that the estimated error of the exposure estimated initially made by Radford and St.Clair Renard (1984), about 30%, was unduly optimistic.
E-ANNEX 2Workshop on Uncertainty in Estimating Exposures to Radon Progeny in Studies of Underground Miners
INTRODUCTION
The epidemiologic studies of underground miners have been the principal basis for estimating the risk of indoor radon (NCRP 1984; NRC 1988; USEPA 1992c).
To estimate exposure to radon progeny, information is needed on the concentrations to which the miners have been exposed as well as the time spent at these concentrations. There are now 12 studies that include estimates of exposure to radon progeny (Lubin and others 1995b; Darby and others 1995). The exposures received by the miners in these studies began as long ago as the end of the nineteenth century in the case of the Malmberget iron miners (Radford and St. Clair Renard 1984) and are continuing for some of the more contemporary groups, such as the Chinese tin miners (Qiao and others 1989) and the French uranium miners (Tirmarche and others 1993). The information available for estimating exposures varied among the cohorts and even within the cohorts by time period. The measurements tended to be more sparse during the initial years of mining operations, the same years during which exposures were generally highest. In some of the studies (Chinese tin miners, Czechoslovakian uranium miners, Colorado Plateau uranium miners, Ontario uranium miners, and Radium Hill uranium miners, and French uranium miners) concentrations of radon progeny were not measured in the early years of operations and it was necessary to estimate WL based on radon measurements, assuming a value for the equilibrium of radon with its progeny (Lubin and others 1994a). Information was also used on mining practices and measurements were made in the Chinese tin mines based on
re-created mine conditions of earlier years. Personnel records were generally used to document time underground, although the detail of the information available also varied among studies. Gaps in the information available for estimating exposures of the underground miners in these epidemiologic studies are an acknowledged source of exposure misclassification with attendant implications for uncertainty in the risk estimates derived from these studies.
The consequences for risk estimation of the errors in exposure estimates have been of concern to the BEIR VI committee. The committee recognizes that exposure misclassification is inevitable in the epidemiologic studies of miners. However, techniques are becoming available to account for these errors in estimating exposure-response relationships (Thomas and others 1994). Statistical approaches to measurement error, with the specific application to the underground miner studies, was a topic of a one-half day workshop held in 1994 by the BEIR VI committee.
To further address the issue of measurement error and its consequences of risk estimation, the BEIR VI committee convened a second workshop on January 23 and 24, 1995. The workshop was designed to bring together geologists and mining and ventilation engineers who had worked on exposure issues in the underground mines with statisticians and epidemiologists who are now contending with the measurement-error problem. The workshop's goal was to obtain additional documentation on approaches followed to assess exposures and to obtain historical insights that might lead to better quantification of the errors in the exposure estimates. The committee also invited statisticians engaged in investigation of approaches for correcting for the effect of measurement error on risk estimates.
The workshop participants selected were appropriate for these objectives. William Chenoweth, now a consulting geologist in Grand Junction, Colorado, worked for many years for the Atomic Energy Commission. He has written extensively on the history of the various uranium mining districts in the United States. James Cleveland, an engineer, worked for Kerr-McGee Corporation in New Mexico for many years, directing ventilation and safety for the Ambrosia Lake operations of Kerr McGee, by far the largest uranium producer in the Grants Mineral Belt. Andreas George, from the Environmental Measurement Laboratory of the U.S. Department of Energy, made measurements of the attached and unattached fractions of radon progeny during the late 1960s and early 1970s; these data represent the only published, historical information on the distribution of progeny between attached and unattached fractions. Douglas Chambers from SENES Consultants Limited in Ottawa, Canada, has had long-standing interest in the assessment of exposures to radon progeny and the consequences of error for risk estimation. Neal Nelson from the U.S. Environmental Protection Agency, also a participant, has followed the exposure assessment issue closely for many years.
The practical experience of these participants was complemented by the statistical expertise of Dan Stram and Duncan Thomas, both from the University
of Southern California. Stram and Thomas have been applying new statistical methods for errors-in-variables to data from the Colorado Plateau miners. Their models make adjustments to risk estimates for measurement error. The workshop was also attended by BEIR VI committee members and Jonathan Samet, the committee's chair, presented descriptive analyses of exposure data for the New Mexico cohort of uranium miners.
The workshop provided valuable and not-well-documented information concerning the U.S. and Canadian uranium mining industries. Consequently, the committee decided to publish portions of the workshop proceedings as an appendix to its report. We include the historical material presented and not the discussion of ongoing work on measurement error by Stram and Thomas.
Workshop Introduction
The workshop began with a welcome and introductions by Jonathan Samet and Evan Douple, the study director. Ethel Gilbert, the workshop chair, reviewed the goals of the workshop and called attention to questions to be considered throughout the workshop. Roger McClellan reminded participants to focus on "uncertainties," not just on "errors." Philip Hopke asked that participants try to recall details regarding the underground mining procedures, especially environmental conditions and the methods used for measuring radon and radon progeny. He asked participants to review those details that have not been previously published or documented. These committee members provided a reminder that the committee needs to characterize the uncertainties that affect different levels of exposure received by the miners.
William Chenoweth (Grand Junction, Colorado)
Chenoweth joined the Atomic Energy Commission (AEC) in 1953 as a geologist. His work initially involved the vanadium mines of the Colorado Plateau. Vanadium mining was initiated in the mid-1930s; he noted that the mines were not ventilated. He then reviewed the uranium mining operations across the years 1947–1981; approximately two-thirds of the mines were in the Colorado Plateau region. The peaks of uranium mining production were in the 1960's and the 1980's on the Colorado Plateau (Moab, Monticello, and Uravan areas and the Grants Mineral Belt). The peak number of mines in operation was around 750, a total reached in the mid-1950s. The AEC stopped buying ore in 1962. However, it continued to purchase concentrates (yellowcake) from the mills through 1970.
Most uranium ore was mined from sandstone. Mining involves drilling, blasting, and mucking, the extraction of the ore. The needs for mining include compressed air and water; powder, blasting caps, and fuses; and a shovel or machine to load the ore. Typically blasting was done at night and the miners would return the next morning without ventilation operative in the mine. In the
early mines, ventilation was used primarily to reduce blasting fumes. Chenoweth explained that the typical cut was made with a burn of multiple fuses. The miners used wheelbarrows in remote areas and the larger mines used compressed-air locomotives. There was no ventilation when the 1952 measurements were made. There were incentives for miners to be productive since the AEC needed the uranium.
Chenoweth showed the structure of a larger Grants mine with a central ventilation shaft. He also showed a map of the Uravan area near the Colorado-Utah border. Brenner asked whether some of the vanadium miners beginning in 1936 were also uranium miners later. The answer was "yes." The same mines were mined back in the 1920's for radium; there was a radium industry from 1910 through 1923. Douglas Chambers stressed that many of the miners worked their own mines on weekends and others were vanadium miners before they worked in uranium mines. Krewski asked whether there were houses in the area. The answer was "no," as the mines tended to be in very remote locations with just a few ranches nearby.
The mines were generally 400 to 800 feet below the surface, although some were as much as 2000 feet deep. Chenoweth provided definitions of various mining terms: a "stope," the site of mining, is an area at the end of a "drift," or passage; a "raise" is a passage up between drifts whereas a "winze" is a passage down between drifts. A map of one early mine showed that there was no ventilation as the rooms were interconnected and the open rooms had randomly placed pillars. The Deremo mine was the largest that Union Carbide mined in the Uravan area. Pictures taken by the U.S. Bureau of Mines in 1953–1955 do not show ventilation; one photo showed a fan that was not in use. Chenoweth surmised that it may have been used for venting blasting powder. Between 1948 and 1956, smaller mines probably had less ventilation. Ventilation rates are affected by temperature and recirculation of radon progeny could have been a problem.
There were various types of operations [U.S. Vanadium (USV) contractors, large mining companies like Walter Duncan, Climax Uranium, Kerr-McGee, Union Carbide, small mining companies, Vanadium Corporation of American (VCA) mines, VCA leasors, small independent operators, and one- or two-man operations]. Some of these mines probably had adequate ventilation, that is, about 500 cubic ft per minute, enough to keep oxygen concentration at about 20%. Early miners did not live in the mines but typically lived in boarding houses; they worked 12-hour shifts and worked 30 days and then spent four or five days in town.
Chenoweth presented a summary of the key events in the history of uranium mine ventilation. Diesel-powered trucks and loaders were introduced in the 1960s. Respirators were issued, but they were not necessarily worn. A Bureau of Mines report (IC07908) states that the Climax Uranium Co. Was the first to use rubber-tired diesel equipment in their mines in the Uranan area in the early 1950s.
Union Carbide adopted extensive air sampling in 1957; it began using diesel in 1958. A shut-down level of 10 WL was adopted in 1958. The U.S. Department of Labor proposed a 3.6 WLM exposure limit in 1969.
Samet asked whether there is any information on arsenic in these mines. Chenoweth explained that a few years ago the Bureau of Land Management asked for a sampling for trace metals in mine dumps. Arsenic was a constituent of some uranium ores. With respect to the question of how many total miners were employed, Chenoweth pointed out that 780 Native American miners were included in the Public Health Service's epidemiologic study, but the Navajo Tribe has registered approximately 2,500 former miners through its Office of Navajo Uranium Workers. There were about 75 miners in 1948 and 5,000 in 1960. Frank Lundin had advised Neal Nelson that 15,000 miners were identified in the Public Health Service surveys, beyond those included in the epidemiologic study (U.S. Bureau of Mines). The current number of miners worldwide is uncertain but underground uranium mining is no longer carried out in the U.S. Some of the current sites of active uranium mining include Canada, China, and Russia. Uranium production is down substantially since the early 1980s and there is now an oversupply of uranium. Countries producing uranium in 1995 were:
|
• Canada |
32% (of world total) |
|
• Australia |
11% |
|
•Niger |
9% |
|
•USA |
7% |
|
•Russia |
6% |
|
• Uzbekistan |
6% |
|
• Kazakstan |
6% |
|
• Namibia |
9% |
|
• South Africa |
4% |
|
• France |
3% |
|
• China |
2% |
|
• Gabon |
2% |
|
• Other |
6% |
|
(From the International Atomic Energy Agency) |
|
James Cleveland (Edmond, Oklahoma)
From 1960 until 1985, Cleveland was with Kerr-McGee. He provided a historical perspective on uranium mining in New Mexico. Uranium mining in New Mexico began with small mines on the Navajo reservation, which operated from the 1948 to 1968. Most Kerr-McGee mines on the reservation were small, operated by less than 20 people and usually by only two or three. The Shiprock uranium mill operated by Kerr-McGee was closed in 1963. The Grants mineral belt, the principal site of uranium mining in the state, stretches 200 miles from the Arizona border to the city of Albuquerque; it is about 25 to 50 miles wide. The Ambrosia Lake and the Jackpill-Paguate districts were the first large-scale production mining areas in New Mexico. In 1980, Grants-area mines employed 4,500 to 5,000 people.
Cleveland described a four-day meeting in 1961 between Archer and Wagoner, from the Public Health Service, and the industry. A warning concerning the
health effects of radon and radon progeny was presented. Three aspects of exposure control were addressed: 1) instrumentation for making measurements, then lacking and very temperamental; 2) ventilation; and 3) record-keeping.
-
Instrumentation for measurements initially involved off-site equipment. Evacuated flasks were opened underground and then shipped to the Massachusetts Institute of Technology or elsewhere for measurement of radon. The Kusnetz method was then developed for measuring radon progeny. The Working Level (WL) unit of concentration and the Working Level Month (WLM) unit of exposure were established by the U.S. Public Health Service. A joint survey with state health department officials and mine inspectors found high levels of radon. The findings were discussed with the industry in 1961. The Junod instrument was used universally until 1967 to measure WLs, but this instrument was very sensitive to environmental conditions. Gas- and battery-powered air samplers were used. The samples were frequently four-hours old before the measurements were made. A probe was then developed for alpha counting and used underground. One could then measure for 40 to 90 minutes using the Kuznetz method. Next, Eberline Instruments developed an instant WL meter which could provide readings after a 2.5 minute count. Several groups developed other instruments for measuring radon. Area monitors were then designed that would give warnings. Cleveland estimated accuracy of the early instruments as plus/minus about 50%.
A discussion followed about the accuracy of the measurements. Andreas George indicated that the uncertainties associated with the measurements were in the range of 50%. Duncan Thomas asked what was reported--the highs, the lows or an average? Douglas Chambers indicated that if there is a bias it is probably small, but with a substantial uncertainty. Lubin asked a series of questions about the measurements: Where were the readings taken? The answer was "in the worker's area in a stope." Within a room the concentration tended to not vary substantially if the air was not very mobile. How much difference was there between measurements? The answer was the variation depended on where the miners were working, and was possibly up to 4 WLM. In the Public Health Service study, Archer assembled all of the measurements and then averaged them for a particular mine. That average was then multiplied by the number of months worked in the mine to obtain WLM.
-
Ventilation was increased in 1962 in most of the mines and the mining companies started to develop and maintain records. New Mexico adopted a 10 WL cease and desist level. Up to that time, ventilation had been natural or used gas-powered fans. The large mines in Ambrosia Lake had 30-inch diameter fans. The major purpose of the fans was to remove powder smoke which contained nitrogen oxides. Simple ventilation was used before 1961. Specifications called for 500 cubic feet per man per minute and, if diesel was used, greater ventilation was called for. State officials had the authority to shut-down mines if ventilation
-
was not adequate. Eventually, large-diameter vent holes, up to 72-inch diameter, were installed to increase ventilation. The mines needed parallel ventilation to deliver fresh air to all parts of the mines. Secondary protection measures, such as filters, respiratory protection (a simple two-canister and air supply unit, or a self-supply unit, depending on levels), and electrostatic precipitators, were proscribed and the latter were used in isolated instances for short periods of time.
-
Records of measurements taken once or twice a quarter were initiated by the companies in 1962. The records used the worker's time in travel, time in the stope, and in other locations, and assigned WLM values to each worker. In 1972, the work week was assumed to include 40 hours. When levels were particularly high, measurements were sometimes taken more than once per week. Total WL-hours for the day were then expanded to the work in a week and the exposure was developed for the month. From 1967 on, records became quite complete. Before 1962, however, records were generally of poor quality. Exposures were grossly underestimated in the early days of the industry, when the work week was longer than 40 hours. Ambrosia Lake mines worked a 48-hour week until 1966.
Hopke asked whether there was smoking underground. Smoking was banned in Ambrosia Lake in about 1975. Standards for exposure to radon progeny were not in place until 1972 when MESA adopted a 4 WLM standard.
Andreas George (New York, New York)
George addressed sources of variation in the measurements made in mines. Sometimes measurements were not made in proximity to the miners. George provided tables showing some typical measurement values and the range of concentrations measured over three days in various locations in the mines (Note: the mines were not named). In the table (Table E Annex 2-1), the arrow indicates either downcast or upcast ventilation by its direction. The WL values relate to specific locations in the mines but not to individual miners. The measurements were made as often as every 30 minutes. The variation in WL values within a stope was generally not large. Ziemer asked whether there were WL values for samples taken at similar locations but at different times of the year. The answer was "no."
Methods were developed for measuring the unattached fraction and applied in the mining environment. George recalled that measurements were made in four different mines for one week. Polonium-218 was measured to determine the uncombined fraction, which was almost related to the unattached fraction (less than 0.2 %). The uncombined fraction tends to be inversely related to particle concentrations. In the Beaverlodge uranium mines, diesel was used infrequently and the vehicles were primarily powered by electricity. Blasting dust remained in the mine for long time intervals. Ventilation ducts were occasionally damaged during blasting.
TABLE E ANNEX 2-1
|
Date |
Mine |
Location |
Activity |
T(°F) |
|
9/26–28/67 |
A |
1-stope |
none (dry) |
57 |
|
9/26–28/67 |
A |
2-stope |
track laying |
50 |
|
9/26–28/67 |
A |
3-drift |
blasting — ore hauling |
50 |
|
9/29–10/2/67 |
B |
1-drift |
none (wet) |
52 |
|
9/29–10/2/67 |
B |
2-stope |
slushing/mucking |
53 |
|
9/29–10/2/67 |
B |
3-stope |
drilling/slushing |
53 |
|
10/3–4/67 |
C |
1-stope |
mucking/slushing (wet) |
52 |
|
10/3–4/67 |
C |
2-stope |
blasting/slushing |
52 |
|
10/3–4/67 |
C |
3-drift |
none |
51 |
|
11/2/67 |
D |
1-drift |
mucking ore (dry) |
57 |
|
11/2–4/67 |
D |
2-drift |
drilling/charging |
55 |
|
11/3–4/67 |
D |
3-drift |
hauling ore |
55 |
|
11/2–4/67 |
D |
4-stope |
drilling/mucking |
58 |
|
11/6–8/67 |
E |
1-drift |
drilling/slushing (dry) |
50 |
|
11/6–8/67 |
E |
2-drift |
hauling ore |
50 |
|
11/6–8/67 |
E |
3-drift |
drilling/charging/slushing |
50 |
|
11/10–15/67 |
F |
1-stope |
drilling/mucking (wet) |
55 |
|
11 |
F |
2-stope |
drilling/mucking (wet) |
60 |
|
11/10, 11/13/67 |
F |
3-stope |
drilling/mucking (wet) |
59 |
|
11 |
F |
4-stope |
drilling/mucking (wet) |
59 |
|
11/14, 11/15/67 |
F |
5-drift |
slushing/mucking (wet) |
49 |
|
11/10, 11/13/67 |
F |
6-stope |
drilling/mucking (wet) |
53 |
|
11/15/67 |
F |
7-drift |
drilling/hauling (wet) |
60 |
|
1/24–26/68 |
G |
1-drift |
drilling/mucking/hauling |
63 |
|
1/24–26/68 |
G |
2-drift |
drilling/mucking/hauling |
53 |
|
1/24, 1/25/68 |
G |
3-heading |
mucking (dry) |
67 |
|
1/26/68 |
G |
4-heading |
drilling/charging |
69 |
|
2/1–2/68 |
H |
1-drift |
drilling/slushing |
34 |
|
1/30–31/68 |
H |
2-drift |
slushing/mucking |
39 |
|
1/30, 2/1/68 |
H |
3-drift |
drilling/slushing/hauling |
45 |
|
2/1–2/68 |
H |
4-drift |
drilling/slushing |
44 |
|
2/2/68 |
H |
5-drift |
none |
40 |
|
1/30, 3/1/68 |
H |
6-stope |
drilling/slushing/mucking |
42 |
|
2/6, 7/68 |
I |
1-drift |
near shaft/ore hauling |
55 |
|
2/5/68 |
I |
2-drift |
blasting/slushing |
55 |
|
2/5–7/68 |
I |
3-heading |
drilling/slushing |
62 |
|
2/5–7/68 |
I |
4-cross-cut |
|
60 |
|
|
RH(%) |
(Ft3/min) |
Ventilation (pCiL-1) |
Radon WL |
Mines |
|
9/26–28/67 |
79 |
5,500 |
ND |
0.67–2.8 |
Beaver |
|
9/26–28/67 |
93 |
2,200–4,400 |
ND |
1.4–4.5 |
Mesa, |
|
9/26–28/67 |
93 |
4,500–5,900 |
ND |
1.9–4.5 |
Colorado |
|
9/29–10/2/67 |
94 |
22,000–34,000 |
ND |
0.95–1.4 |
Beaver |
|
9/29–10/2/67 |
95 |
200 |
ND |
2.1–2.4 |
Mesa, |
|
9/29–10/2/67 |
97 |
1,000 |
ND |
2.1–2.3 |
Colorado |
|
10/3–4/67 |
96 |
2,000 |
ND |
5.0–5.5 |
Uravan, |
|
10/3–4/67 |
96 |
3,500 |
ND |
3.8–4.1 |
Colorado |
|
10/3–4/67 |
97 |
13,000 |
ND |
(3.8) |
|
|
11/2-67 |
47 |
27,000 |
(410) |
(0.69) |
Uravan, |
|
11/2–4/67 |
78 |
900 |
190–380 |
0.66–1.13 |
Colorado |
|
11/2–4/67 |
73 |
3,000 |
(260) |
(1.1) |
|
|
11/2–4/67 |
71 |
1,000 |
410–1000 |
0.41–0.78 |
|
|
11/6–8/67 |
81 |
500 |
460–1000 |
1.27–3.10 |
|
|
11/6–8/67 |
63 |
3,000 |
ND |
1.15–2.0 |
|
|
11/6–8/67 |
82 |
3,000 (on/off) |
180–270 |
0.36–0.67 |
|
|
11/10–15/67 |
57 |
1,500–3,000 |
(430) |
0.35–2.13 |
Uranum, |
|
11 |
84 |
none |
(490) |
(1.28) |
Colorado |
|
11/10, 11/13/67 |
95 |
none |
(360) |
(2.36) |
|
|
11 |
92 |
none |
(340) |
(1.74) |
|
|
11/14, 11/15/67 |
66 |
fresh air 5,000–9,000 |
88–110 |
0.22–0.27 |
|
|
11/10, 11/13/67 |
92 |
7,000–9,000 |
180–220 |
0.42–0.46 |
|
|
11/15/67 |
94 |
ND |
(540) |
(1.28) |
|
|
1/24–26/68 |
81 |
14,000 |
380–420 |
0.8–1.0 |
Ambrosia Lake, NM |
|
1/24–26/68 |
80 |
8,000 |
1350–1790 |
3.1–5.10 |
|
|
1/24–1/25/68 |
90 |
(convection) |
1900–2300 |
(1.70) |
|
|
1/26/68 |
92 |
ND |
(680) |
|
|
|
2/1-2/68 |
57 |
26,000–29,000 |
330–370 |
0.26–0.43 |
Ambrosia Lake, NM |
|
1/30-31/68 |
66 |
4,000 |
830–1010 |
1.04–1.08 |
|
|
1/30, 2/1/68 |
71 |
3,000–6,000 |
780–1150 |
1.4–2.1 |
|
|
2/1-2/68 |
60 |
6,000 |
770–920 |
1.28–1.60 |
|
|
2/2/68 |
62 |
15,000 (exhaust) |
(870) |
(2.13) |
|
|
1/30, 3/1/68 |
67 |
3,000 |
670–960 |
1.10–1.37 |
|
|
2/6, 7/68 |
84 |
(fresh air) 66,000–72,000 |
84–190 |
1.19–0.23 |
Ambrosia Lake, NM |
|
2/5/68 |
84 |
3,000 |
(160) |
(0.26) |
|
|
2/5-7/68 |
94 |
3,000–5,000 |
640–900 |
2.2–2.7 |
|
|
2/5-7/68 |
95 |
1,000–2,000 |
1020–1440 |
3.0–3.8 |
|
Measurements were made using diffusion batteries developed in his (George's) laboratory. A bimodal distribution of alpha activity was found in some mines, similar to more recent information from homes. The 5–6 nm size was critical because this size is relevant for the bronchial deposition. Classical, bimodal, or unimodal (diesel mines) distributions were obtained for activity-weighted sizes. Five nm is the critical size for the tracheal deposition. NCRP 1978 gives a number of about 1.7, which should be compared to 4 in the publication ''Summary of dose conversion factors from reanalysis of New Mexico uranium mine particle-size data." Most of the progeny are attached to the larger particles while the smaller particles are deposited in the lung. It is realistic to assume two-fold variation in measurements from mine to mine.
Douglas Chambers (Ottawa, Canada)
Chambers discussed the characteristics of several mines, beginning with the Newfoundland fluorspar mines. The Black Duck Mine opened in 1933 and there was not forced ventilation until the 1950s. For the Newfoundland fluorspar mines, uncertainties are quite high for data before 1967, perhaps as high as 300-fold. The miners smoked heavily and it was a very dusty environment. Chambers was not aware of other contaminants in the mines, such as arsenic. The reconstruction of exposures for these mines has been difficult and the approaches used have been as much as can reasonably be done.
The original client for uranium from the Ontario mines was the U.S. AEC. Two uranium mines were operational in Ontario in 1955; in 1958 there were 15 mines. Between 1955 and 1981, 131,000 radon-daughter measurements were made over 141 mine-years of operation, averaging 929 measurements per mine-year. Two sets of exposures were calculated, "standard" and "special" (see 1988 NRC BEIR IV Report for a description of these two sets of exposures). Exposure to aluminum powder was used in an attempt to prevent silicosis. The Canadian report on health and safety in mines is a potential source of information (Canadian Task Force on the Periodic Health Examination 1990).
A comparison was made between past and present exposure conditions. Pre-1958 conditions included radon progeny at 0.3 to 1.4 WL and mineral dust at approximately 1 to 9 mg/m3 and for 1990 conditions at 0.05 to 0.3 WL and 0.05 to 1 mg/m3, respectively. The lung-cancer experience of Ontario uranium miners with and without gold-mining history was provided. The observed to expected ratio was greater for those with gold-mining experience.
Chambers also provided information concerning the Port Radium uranium mine. In the Port Radium mine, the ventilation was frequently turned off in the winter months. Arsenic may have been 6–7% of the pitchblende ore. Exposures to radon progeny were estimated based on retrospective reconstruction. The ore grade was high and yielded high WLs (50–100 WL). The original estimates were
done by Frost with Eldorado Nuclear. Chambers indicated that he has all of the records for the Port Radium mine.
Chambers also discussed the Beaverlodge mines. There has recently been a reassessment of exposures for some of the miners in the epidemiologic cohort of Beaverlodge mines. He noted that at Beaverlodge, while the person running the drill would be expected to get the highest dose, this was not necessarily the case. In 1990, the Atomic Energy Control Board asked if exposures could be reconstructed from the records. The Schwartzwalter Mine in Colorado is similar to the Beaverlodge Mines.
Chambers then discussed the estimation of WLM for Beaverlodge miners. Exposure was estimated by year and type of workplace. Men tended to migrate from one mine to another in the Beaverlodge area and this was not accounted for in the exposure reconstructions. There were six or seven mine areas and nine work-type categories. The reconstructed exposures were compared to the original estimates used in the epidemiologic report of Howe and colleagues. The correlation was strong, although the original exposure estimates tended to be less than the revised exposure estimates. Means rather than medians of individual measurements were used in the new estimates. A summary of observations in the cohort was provided. Many of the miners lived in homes built on uranium-containing foundations. A positive correlation was found between WLMs and konimeter data for particle counts. The category of a miner at first work was a factor (miners needed previous work experience).
Finally, Chambers provided some remarks concerning the Colorado Plateau study. In the Colorado mines, exposures were deliberately overestimated (see Lundin and others 1971). For other hardrock mining, exposures at concentrations of 1.0, 0.5, or 0.3 WL were assigned. A crude trace of work histories of 29 people was presented from Archer, 1966. The uncertainties were related to the location of the miners and their work histories. Studies in the past have not sufficiently considered the uncertainties in the exposures. The exposures after 1969 were probably not that significant, particularly in comparison to earlier years.
Jonathan Samet (Baltimore, Maryland)
Samet began by recommending a book, Uranium Frenzy by Raye C. Ringholz (University of New Mexico Press), for a review of the early history of the uranium-mining industry. He reviewed the measurements made in the New Mexico mines. Measurements were first made in the 1950s and by 1961–1967, WL measurements were being made routinely by the State Mine Inspector, the State Health Department, and the industry. He showed examples of data sheets for the years through 1967. Person-weighted totals of the individual measurements were made. These were referred to as Total Mine Indexes; the data sheets also included measurements by the type of area. Samet showed the scatter in the
data and the substantial variation in the measurements made within individual visits. Jim Cleveland mentioned that inspections by the State Agencies were unannounced. Attention was not paid to ventilation if the inspection results were satisfactory. Exposures in the Colorado Plateau were grossly underestimated by small companies.
Discussion Of Exposure Estimates
Samet reminded the participants that time-dependent errors affect the exposure estimates with implications for the committee's modeling. Hopke commented that there is ample qualitative information on errors, but it is not clear what should be done quantitatively. Brenner indicated that the uncertainty appears to be more of a problem in the small mines. Can mine size be incorporated on the basis of the total number of miners? Gilbert mentioned that the committee may want to develop a questionnaire. Can we remove certain cohorts for which the uncertainty was largest? Can we rank cohorts? Can we obtain additional data? Chambers mentioned that the presence of other factors in the dust should be considered. Lubin mentioned that data on arsenic are available for the China and Ontario cohorts. The Swedish study has some information on silica and the Czech data have some additional information as well. Chambers mentioned that he had inquired about this issue (other contaminants) in the mines that he visited. Do higher levels of exposure to radon progeny entail higher exposures to other agents? Has there been consideration of parallel analyses of entire cohorts versus the group with exposure to uranium alone. Ziemer asked "How consistent are the ore forms"? There appears to be a difference from location to location. Bill Chenoweth has a report (PP-320) describing measurements made by the U.S. Geological Survey of trace metals in different mines. There are data available from Union Carbide in Grand Junction from retired ventilation engineers and state records (by engineers such as Vern Bishop, Bob Beverly, and Ben Kilgore). Umetro Minerals Corp. is now doing restoration. Lubin asked—How common was it that workers worked at their own mines on weekends and holidays? Hopke asked—What do we mean by "other hardrock"? Gold and silver mines were common, as opposed to vanadium (or copper mines). The Port Radium mine was reopened as a silver mine (Chambers). The participants were reminded that AMSA keeps records on other metals in mines (Nelson).
What fraction of mines used diesel? Jim Cleveland answered that Kerr-McGee was almost all electric until the last few years; United Nuclear was mostly diesel. The Ambrosia Lake mines were mostly diesel. Was the use of diesel always accompanied by better ventilation? Yes. Good ventilation was needed for the diesel. Diesel was used in the '60's. Typically the miners blasted at noon and at 8:00 pm. They could not leave the lunchroom until 30 minutes after blasting. There was no diesel used until 1958 on the Colorado Plateau. By 1971, diesel use was as much as 90% and reached 100% by 1980. What were other sources of
particles in the air of the mines? Drilling and diesel were the major causes. The drilling did not fracture sandstone sand grains and the rock was very wet (20% moisture) at Ambrosia Lake. Slushing created some dust. Colorado mines were mostly dry mines with substantial dust. Mucking, pushing ore out the haulway, dropping it down chutes, and like activities, all created dust. In the late 1970s and early 1980s, gravel was hauled in for building good roadways. Was the mineralogy about the same throughout the Colorado mines? No, the mines differed in vanadium and other metals.
Appendix F
Exposures Other Than Radon in Underground Mines
OVERVIEW
Underground miners are exposed to a number of agents, in addition to radon progeny which may adversely affect the lung. Several of these agents are known or suspect carcinogens (arsenic, diesel exhaust, and silica), and some may cause airways inflammation (blasting fumes and diesel exhaust). Silica exposure causes silicosis and several investigations have assessed modification of the effect of radon progeny by the presence of this fibrotic disorder.
These exposures of miners, in addition to radon progeny, are a source of uncertainty in extending risk estimates based on the epidemiologic studies of miners to the general population. Inflammatory changes in the epithelium might non-specifically affect the risk of lung-cancer from radon progeny and the additional exposure to other carcinogens might alter the risk of radon progeny as well. These other exposures were considered in the BEIR IV report (NRC 1988) and subsequently in the radon dose panel report (NRC 1991).
In this appendix, we update the earlier reviews for exposure to arsenic, silica, and diesel exhaust. Information on exposures of the miners to the agents is limited and only a few studies provide human information on arsenic and silica. None of the studies have direct information on exposure to diesel exhaust. The limited data available on these exposures are summarized by cohort in appendix D. Use of diesel engines in U.S. mines is described in the workshop summary that is part of appendix E annex 2. The more general topic of interactions between agents is addressed in appendix C in considering the combined effect of cigarette smoking and radon.
Arsenic
Although evidence in experimental animal studies of the carcinogenicity of arsenic is limited, there is substantial evidence that inorganic arsenic is a carcinogen in humans (Blot and Fraumeni 1994; IARC 1987). Neubauer (1947) reports that arsenic was suspect as being carcinogenic as early as 1879 as a result of high lung-cancer rates in German miners (Bates and others 1992; Furst 1983). The ingestion of arsenic in drinking water and in pharmaceuticals has been associated with a number of disease outcomes, such as liver angiosarcoma and meningioma, and cancers of the skin, bladder, kidney, and colon, as well as black-foot disease (IARC 1987). Studies have also clearly shown that inhaled arsenic (arsenic trioxide) is a human lung carcinogen (IARC 1987). The principal concern for this committee is the role of exposure to airborne arsenic in mine dusts as a primary risk factor for lung-cancer, and how its presence might affect the evaluation of the relationship between radon-progeny exposure and lung-cancer.
Occupational studies have been the main source of data on the effects of exposure to arsenic and risk of lung-cancer. These studies have included workers manufacturing and using arsenical-containing pesticides (Hill and Faning 1948; Roth 1957; Ott and others 1974; Mabuchi and others 1985), smelter workers and underground miners (for summaries, see Blot and Fraumeni 1994 and IARC 1987). Although the majority of occupational studies of arsenic exposure have been conducted in smelter workers, an increased risk of lung-cancer with arsenic exposure has been observed in several studies of miner populations (Taylor and others 1989; Kusiak and others 1993; Xuan and others 1993; Enterline and others 1987; Simonato and others 1994). However, among the studies of miners, only the investigations of Chinese tin miners (Xuan and others 1993) and Ontario uranium miners (Kusiak and others 1993) have included a quantitative evaluation of arsenic and of the joint association of arsenic and radon-progeny exposure.
Although studies have consistently shown an increasing risk of lung-cancer with greater cumulative exposure to arsenic, there have been few detailed analyses of the shape of the dose-response curve for arsenic exposure. The analysis by Enterline and others (1995) and a meta-analysis of published studies (Hertz-Picciotto and Smith 1993) suggested a curvilinear relationship with a decrease in the excess relative risk per unit exposure as exposure increases, that is, the exposure-response curve was concave from below. Analyses of the Ontario miners (Kusiak and others 1993) and Chinese miners (Lubin, communication to the committee) showed a similar concave relationship, even after adjustment for radon-progeny exposure.
The distribution of histological types of lung-cancer in arsenic-exposed populations has not been extensively studied. There have been several small investigations, with little consistency in their finding. Based on 25 cases, Newman and others (1976) reported a higher proportion of poorly differentiated epidemiod carcinoma, while Wichs and others (1981) studied 42 smelter workers and 42
matched controls and found an excess of adenocarcinomas. In contrast, in a larger study of 93 lung-cancer cases highly exposed to arsenic and 136 referent lung-cancer cases, Pershagen and others (1987) found no variation in the histological distribution of lung-cancer cases when data were classified by a measure of arsenic exposure. The distributions of histological type in underground miners have been reported, but are potentially confounded by smoking and radon-progeny exposure.
Mathematical models, based on the Armitage-Doll multistage theory for carcinogenesis (Armitage and Doll 1961), were applied to data on lung-cancer from two studies of copper-smelter workers in Tacoma (Mazumdar and others 1989) and in Montana (Brown and Chu 1983). Both analyses drew similar conclusions, namely, arsenic exposure acts primarily as a late-stage carcinogen, but that the possibility of an early-stage effect cannot be ruled out. However, one limitation of both analyses was the inability to directly incorporate cigarette-smoking into the modeling, a factor which is thought to act as both an early-and late-stage carcinogen.
In the miner pooled analysis by Lubin and others (1994a), adjustment for arsenic exposure reduced the ERR/WLM in the Chinese miners from 0.61% to 0.16%. Interpretation of the reductions is hampered by the high correlation coefficient, 0.48, between cumulative radon-progeny exposure and arsenic exposure among jointly exposed miners. This suggests that the best estimate of the ERR/WLM for the radon progeny exposure-lung-cancer relationship lies between 0.0061 and 0.0016. In the Ontario data, adjustment for arsenic exposure reduced the ERR/WLM from 0.0093 to 0.0084. The correlation coefficient between radon-progeny exposure and arsenic exposure was 0.02. After adjustment for arsenic exposure as a primary risk factor, the ERR/WLM did not vary significantly with level of arsenic exposure in either study (Lubin and others 1994a). This pattern is consistent with a multiplicative association between radon-progeny exposure and arsenic exposure. However, interpretation of these results is hampered by differences in definition of the arsenic-exposure measure, which was percent arsenic in rock multiplied by duration of exposure for the Ontario study and duration of arsenic exposures (mgm-3y) for the China study. The evidence appears to suggest a greater than additive (synergistic) association for the combined relative risks for cigarette use and airborne arsenic exposure (Hertz-Picciotto and others 1992). In miner populations, the joint association of the three factors, radon progeny, arsenic, and smoking, has not been evaluated.
Silica
Silica, a ubiquitous exposure in many types of underground mining, is of particular interest in that it not only causes silicosis but also has been identified as a suspect human carcinogen by the International Agency for Research on Cancer (IARC 1987). In classifying crystalline silica as carcinogenic, IARC indicated
that evidence of silica was sufficient in animals while limited in humans. For a detailed review of silica and lung-cancer, see Goldsmith and Samet (1994). Abelson (1991) has identified silica in mines as one of the key factors contributing to uncertainty in the use of radon-associated lung-cancers for miners to estimate population risks for radon. Silica might modify the risk of radon directly as an additional carcinogenic exposure or indirectly by causing fibrosis and airways damage.
With regard to this possible indirect mechanism, there have been several studies on respiratory disease patients that suggest a significant association between obstructive lung function and lung-cancer (Davis 1976; Skillrud and others 1986; Tockman and others 1987). Similar findings have been reported for pneumoconiotic workers by Harber and others (1986) and by Carta and others (1991). Carta and others (1994) suggest that "airways obstruction may be an independent risk factor for bronchogenic carcinoma." Accordingly, they studied the lung-cancer mortality in relation to airways obstruction among Sardinian metal miners exposed to silica and low levels of radon progeny. In one of the two mines studied, the quartz concentration in the respirable dust was between 0.2% and 2.0% while the radon exposures averaged 0.07 Jm-3 (0.13 WL) with the maximum cumulative exposure in the 0.28 to 0.42 Jhm-3 (80–120 WLM) range. In the second mine, the silica levels were much greater, ranging from 6.5% to 29%, while the radon levels were lower than in the first mine. The cohort included some 1,741 miners and a total of 25,842.5 person-years of exposure. Lung function tests, chest radiographs, and smoking histories were available for all subjects entering the cohort. A total of seventeen subjects from the first mine and seven from the second died of lung-cancer. The standardized mortality ratio (SMR) for lung-cancer was higher for the first mine. Furthermore, among miners with initial spirometric airways obstruction, those in the first mine showed the highest risk. Carta and others concluded that crystalline silica as such does not affect lung-cancer mortality. They further suggest that impaired pulmonary function may be an independent predictor of lung-cancer and may be an important risk factor because of enhancement of residence times for inhaled carcinogens.
An important investigation that considered silica dust and silicosis as risk factors for lung-cancer in underground miners was reported by Radford and St. Clair Renard (1984). They conducted a case-control study of silicosis in Swedish iron miners involving 50 lung-cancer cases in deceased miners and 100 controls matched on age, year mining began, and duration of time mining. Both the severity of silicosis and the frequency of radiographic evidence of silicosis were comparable for the cases and the controls, indicating no effect of this disease on lung-cancer risk.
Epidemiological evidence of increased lung-cancer risk in silicotic patients has been reported by Koskela and others (1990) as well as by Chiyotani and others (1990) and Merlo and others (1990). However, there have been a number of studies that present conflicting results on lung-cancer risks for workers with
and without silicosis exposed to dust that contained silica. These studies include: Hessel and others (1990), Meijers and others (1990), Ng and others (1990), Ahlman and others (1991), Amandus and Costello (1991), Carta and others (1991), Chen and others (1991), Chia and others (1991), Hnizdo and Sluis-Cremer (1991), Kusiak and others (1991), and McLaughlin and others (1992). Generally these studies demonstrate no clear dose-response relationship for silica exposure even though an overall association between lung-cancer and the presence of silicosis was observed in some of the studies.
Samet and coworkers (1994) conducted a case-control study in the cohort of underground uranium miners in New Mexico to assess the presence of radiographic silicosis as a risk factor for lung-cancer. This is one of the cohorts included in the pooled data set. The presence of silicosis as determined by chest radiographs taken at or near the beginning of employment was determined for 65 lung-cancer cases and 216 controls. Data on the individual exposures to silica were not available, but there are data available that demonstrate the presence of silica in mines in the region of the study. Also, silicosis is well documented in underground uranium miners in the southwestern states. The study showed that the presence of silicosis was not associated with lung-cancer risk after adjustment was made for cumulative exposure to radon. These investigators recognized that the findings were limited by the small number of subjects, but they were able to conclude nonetheless that there was a lack of association of silicosis with lung-cancer. They stated that "silica exposure should not be regarded as a major uncertainty in extrapolating radon risk estimates from miners to the general population."
Finkelstein (1995) examined the presence of radiographic silicosis as a lung-cancer risk-factor in miners from the Ontario Silicosis Surveillance Database. In contrast to the findings of Samet and others (1994), he found that silicosis was a highly significant risk factor for lung-cancer. Accordingly, he concluded that the radon lung-cancer risk decreased if an adjustment for the presence of silicosis was made. However, Archer (1996) has criticized Finkelstein's conclusion on the basis that early lung-cancer is very difficult to discern from radiographs of individuals whose lungs contain fibrotic abnormalities. Archer states that it is likely that at the time they were admitted into the study the silicotics in Finkelstein's cohort had more undetected cancers than did the controls. Archer also criticized Finkelstein's assumption that radon exposures for the nonuranium miners was zero.
Recently, Enderle and Friedrich (1995) published a review of the exposure conditions and the health consequences for the East German uranium miners in the Saxony and Thuringia regions. They point out that in the 1946 to 1955 period working conditions were extremely poor and the miners were exposed not only to radon progeny, but also to very high dust levels, and to toxic chemicals or elements including arsenic and crystalline silica. They offer no direct evidence relating silica and lung-cancer for these miners, but they do cite a study by Melhorn (1992) that reports a high rate of bronchial carcinoma occurring in
miners with known silicosis. They also cite the work by Tockman and Samet (1994) who describe silicosis as a risk factor for lung-cancer.
Goldsmith and coworkers (1995) have also shown that, in addition to having increased mortality from nonmalignant respiratory diseases and from tuberculosis, silicotics have a significantly elevated risk of death from cancers of the trachea, bronchus, and lung.
Diesel Engine Exhaust and Fumes
Exposure to diesel is also relevant to extrapolation of risks from miners to the population. Some uranium-mining operations used diesel engine-powered equipment resulting in the exposure of miners to diesel exhaust. As will be discussed below, the diesel soot particles are readily respirable. They are carbonaceous particles and have associated hydrocarbons some of which are mutagenic and also carcinogenic. This raises the potential for the diesel soot to be carcinogenic, and further raises the possibility that diesel exhaust may induce lung-cancer. In turn, this raises the possibility for diesel exhaust to be a confounding factor in evaluating the lung-cancer risks of exposure to radon.
In this section, the evidence is reviewed for diesel exhaust causing lung-cancer. This is followed by a discussion of the possible role of diesel exhaust as a causative factor in lung-cancers observed in uranium miners.
The diesel engine, patented by Rudolph Diesel in 1892, has found wide use in commerce, including use in mining operations and in railroad locomotives. The dieselization of railroads occurred principally after World War II, reached its midpoint in 1952, and by 1959, approximately 95% of the locomotives in the United States were diesel powered (U.S. Department of Labor 1972).
Concern for health effects of exposure to diesel exhaust has existed for some time. This concern relates to the readily inhalable size of diesel soot particles, 0.1 to 0.5 µ (Cheng and others 1984), giving concern for the development of lung-cancer. This concern is heightened by an awareness that a significant portion, typically 10 to 15%, of the diesel soot particles by weight consist of organic compounds readily extractable by organic solvents (Johnson 1988). The extracted material includes many polycyclic aromatic hydrocarbons including many that are mutagenic and some that are carcinogenic (Schuetzle and Jensen 1985; Schuetzle and Lewtas 1986). Kotin and others (1955) demonstrated that organic solvent extracts of diesel soot were carcinogenic when painted on mouse skin.
The prospect for increased use of diesel engines in light-duty vehicles in the late 1970s increased concern for the cancer risks of inhalation exposure to diesel soot. This concern stimulated the conduct of epidemiological investigations, bioassays in laboratory animals, and a wide range of mechanistic studies at all levels of biological organization from cells to populations of mammals.
The epidemiological studies have been recently reviewed by Cohen and Higgins (1995) and Nauss and others (1995) in a special report prepared by the
Health Effects Institute (1995). Two figures from that report provide a summary of the currently available data on lung-cancer risks evaluated in railroad workers (Figure F-1), and truck drivers (Figure F-2). From these figures, it is clear that the relative risk of lung-cancer measured in the various studies is only elevated significantly if at all in a few studies. A major confounder in these studies, as is usually the case, is cigarette-smoking which is a dominant causative factor in lung-cancer. This is illustrated by considering Table F-1 taken from Garshick and others (1987). The slightly elevated lung-cancer risk (odds ratio = 1.41, 95% CE = 1.06, 1.88) contrasts sharply with the substantial risk measured for cigarette-smoking. Cigarette-smoking risk increased with amount of cigarette smoking and age to an odds ratio of 9.14, 59% CE = 6.11, 13.70 for cases age greater than 65 years and >50 pack-years of cigarette smoking. Crump and others (1991) reanalyzed the data used by Garshick and others (1987) as well as additional data on the same population and was unable to discern an exposure-related increase in lung-cancer risk.
In the late 1980s, results of a number of well-conducted laboratory animal bioassays of diesel exhaust became available. These results have been extensively reviewed (Mauderly 1992; Health Effects Institute 1995; McClellan 1987). The results, summarized in Figure F-3 taken from the HEI report (1995), clearly indicate that long-term high-level exposure to diesel exhaust increases an excess of lung-cancer in rats. Mice and Syrian hamsters similarly exposed have yielded negative or equivocal results. An excellent example of this contrasting result is apparent from the studies of rats (increased lung-cancer) and mice (no increase in
FIGURE F-1 Lung cancer and exposure to diesel exhaust in railroad workers. ![]() = RR adjusted for cigarette-smoking;
= RR adjusted for cigarette-smoking; ![]() = RR not adjusted for cigarette-smoking. For two studies (adapted from Nauss and The HEI Diesel Working Group, 1995).
= RR not adjusted for cigarette-smoking. For two studies (adapted from Nauss and The HEI Diesel Working Group, 1995).
FIGURE F-2 Lung-cancer and exposure to diesel exhaust in truck drivers. ![]() = RR adjusted for cigarette smoking;
= RR adjusted for cigarette smoking; ![]() = RR not adjusted for cigarette smoking. For the study by Williams (William and others 1977), CLs were not reported and could not be calculated. For the Steenland study (Steenland and others 1992), the data were gathered from the union reports of long-haul truckers; for the 1988 Boffetta study (Boffetta and others 1988), the data were self-reported by diesel truck drivers; and for the Siemiatycki study (Siemiatycki 1991), they were self-reported by heavy-duty truck drivers (personal communication).
= RR not adjusted for cigarette smoking. For the study by Williams (William and others 1977), CLs were not reported and could not be calculated. For the Steenland study (Steenland and others 1992), the data were gathered from the union reports of long-haul truckers; for the 1988 Boffetta study (Boffetta and others 1988), the data were self-reported by diesel truck drivers; and for the Siemiatycki study (Siemiatycki 1991), they were self-reported by heavy-duty truck drivers (personal communication).
lung-cancer) reported by Mauderly and others (1996) to diesel exhaust from the same source.
As the significance of the diesel exhaust rat lung-cancer findings was discussed, it was noted that chronic inhalation exposure of other particulate materials (lacking in capability to directly damage DNA) also caused an increase in lung-cancer in rats (Vostal 1986). This raised questions as to the mechanisms by which diesel exhaust and these other materials might be acting. It was speculated that the effects of these materials might be related to their ability, when inhaled at high concentrations, to overload lung-clearance mechanisms and cause chronic inflammation and, ultimately, lung-cancer (Vostal 1986; Morrow 1988; McClellan 1990).
To test this hypothesis, studies were conducted in which rats were chronically exposed to carbon black particles, which were relatively devoid of mutagenic organic compounds. Two major laboratories found that carbon black had about the same effectiveness as diesel exhaust in producing lung-cancer in rats. Recently, Driscoll and others (1996) and Oberdörster (1996) have shown that exposure to high concentrations of carbon black produced persistent pulmonary inflammation, and an increase in mutations in lung epithelial cells. These results provide a plausible mechanism for the pathogenesis of the particle-induced lung-cancer in rats. This is illustrated schematically in Figure F-4.
TABLE F-1 Regression results using diesel exhaust exposure as a single continuous variable (diesel-years) adjusted for cigarette-smoking and asbestos exposure
|
Exposure Category |
Odds Coefficient |
Ratio |
95% Cl |
p Values |
|
Case age = 64 |
|
|
|
|
|
Diesel-years |
0.01719 |
1.41a |
1.06, 1.88 |
0.02 |
|
Asbestos, Y/N |
0.18111 |
1.20 |
0.87, 9.65 |
0.27 |
|
= 50 pack-yearsb |
1.19196 |
3.29 |
1.57, 6.93 |
<0.01 |
|
>50 pack-yearsb |
1.73606 |
5.68 |
2.73, 11.80 |
<0.01 |
|
Pack-years missingb |
1.37975 |
3.97 |
1.86, 8.51 |
<0.01 |
|
Case age = 65 |
|
|
|
|
|
Diesel-years |
-0.00461 |
0.91a |
0.71, 1.17 |
0.47 |
|
Asbestos, Y/N |
-0.01807 |
0.98 |
0.81, 1.20 |
0.86 |
|
= 50 pack-yearsb |
1.47641 |
4.38 |
2.90, 6.60 |
<0.01 |
|
>50 pack-yearsb |
2.21321 |
9.14 |
6.11, 13.70 |
<0.01 |
|
Pack-years missingb |
1.35379 |
3.87 |
2.56, 5.84 |
<0.01 |
|
a Calculated on the basis of 20 years of exposure. b Reference category of zero pack-years (never-smokers). From Garshick and others (1987). |
||||
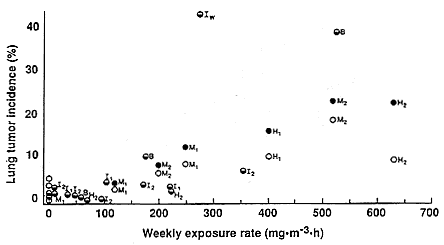
FIGURE F-3 The relation between rat lung tumor incidence and exposure rates for diesel exhaust particulate matter. Data point code is: B = Brightwell and others 1989; H1 = Heinrich and others 1995; I1 = Ishinishi and others 1986; (exhaust from 1.8-L engine); I2 Ishinishi and others 1986 (exhaust from 11-L engine); Iw = Iwai and others 1986; M1 = Mauderly and others 1987; M2 = Mauderly and others 1994. ![]() = Includes lesions identified by the investigator as ''benign squamous tumors";
= Includes lesions identified by the investigator as ''benign squamous tumors"; ![]() = excludes these lesions.
= excludes these lesions.
The recent cancer results from rats exposed to diesel exhaust raises questions as to the appropriateness of their use for defining the carcinogenic risk of diesel exhaust to humans (McClellan 1996). Thus, the human lung-cancer risk of diesel exhaust exposure should be based exclusively on the epidemiological data reviewed earlier.
With the above information as background, the potential interaction between radon and diesel exhaust can be considered. In the absence of either epidemiological studies of radon-exposed individuals that have included characterization of diesel exhaust exposure as a confounder or laboratory animal studies with controlled exposure to radon and diesel exhaust, it is only possible to speculate on the potential combined effects of radon and diesel exhaust. Some insight may be gained by considering the data in Table F-1 from the study of railroad workers exposed to diesel exhaust. The results of this study can be interpreted as identifying diesel exhaust as a potential low-potency carcinogen. The characterization of diesel exhaust as a low-potency carcinogen is made with reference to cigarette-smoking that was substantially more potent than diesel exhaust depending on the extent of smoking. It is unlikely that the uranium miners had smoking histories substantially different from the railroad workers. Compared to diesel exhaust, radon exposure may also be classed as a high-potency risk factor. It can be argued that in an exposure environment involving the two high-potency risk factors, radon and cigarette-smoking, the addition of a low-potency risk factor, diesel exhaust, would be unlikely to affect the combined risk from the two high-potency risk factors.
Muscat and Wynder (1995) conducted a case-control study to determine the effects of exposure to diesel engine exhaust and fumes. The subjects were truck drivers, mine workers, firefighters, and railroad workers, and included 235 male hospital patients with laryngeal cancer. These investigators showed that diesel engine exhaust is unrelated to laryngeal cancer risk. They offered no suggestion that lung-cancer would be directly related to diesel fume exposures.
Mycotoxins
Recently there has been speculation about the possible role of mycotoxins in the production of lung-cancers. In a letter to the editor of Lancet, Venitt and Biggs (1994) suggested that exposure of uranium miners to mycotoxins such as sterigmatocystin could account for the mutations in p53 at codon 249 that had been reported by Taylor and coworkers (1994). Taylor and others had suggested that the codon 249 mutation may be a marker for radon-induced lung-cancer, but Venitt and Biggs point out that the gross damage caused by a particles would be expected to produce gross damage to the DNA rather than a precise mutation at a specific codon. Likewise, Hei and others (1994b) suggest that although a point mutation could be induced by a particles, complete loss of the p53 gene would be more likely. Bartsch and others (1995) also assert that a radon-induced hotspot
mutation would be surprising since one would expect mainly random DNA strand breaks. They screened for the presence of the codon 249 mutation in lung-cancers from the Saxony, Germany uranium miners and found that none of the 50 lung tumors analyzed showed the hotspot mutation. Lo and others (1995) also raised the possibility that the results found by Taylor and colleagues could be related to mycotoxins.
Hypotheses concerning a possible role of mycotoxins are presently speculative and not supported by any observation data. Information on exposures is completely lacking.
Sram and coworkers (1993) have reported that Czech uranium miners are exposed to chemical mutagens as well as radon. They found molds in throat swabs from 27% of the miners studied as compared to only 5% in controls. Various mycotoxins were found in the swabs, including sterigmatocystin, a bisfuranoid mycotoxin that is structurally related to aflatoxins. Sterigmatocystin is reported by Gopalakrishnan and others (1992) to be a potent carcinogen and mutagen that produces squamous carcinomas and adenocarcinomas in animal lungs.
SUMMARY
Exposures other than radon progeny sustained by underground miners could plausibly modify the lung-cancer risk associated with exposure to radon progeny. The relevant data for assessing such modification in the miner cohorts are scant. Uncontrolled arsenic exposure may be a source of positive bias, as shown for example in the Chinese tin miners. The role of silica has not been directly assessed; the scant epidemiologic evidence indicates that the presence of silicosis is not a strong modification of the risk of radon. Diesel exhaust, present in some of the more recent miners, was also probably not a strong modifier of the risk of radon progeny.
Appendix G
Epidemiologic Studies in the Indoor Environment
This appendix examines the epidemiologic evidence of an association between indoor radon-progeny exposure and lung-cancer. Although data from indoor-radon studies are not yet sufficient to develop a general risk-assessment model or to estimate precisely the magnitude of risk posed by radon in houses, the data do support a small increase in lung-cancer risk due to indoor radon exposure and are consistent both with the extrapolation of lung-cancer risk using miner-based models and with relative risks among miners with cumulative exposures similar to exposures that might be experienced by long-term residents in houses that exceed the Environmental Protection Agency (EPA) action level. However, there are sufficient uncertainties in current epidemiologic studies that the residential data alone do not conclusively support a definable excess lung-cancer risk associated with radon-progeny exposure.
Ecologic studies and analytic case-control studies are the 2 types of epidemiologic studies that have considered the issue. In an ecologic study, regional rates of lung-cancer are related to a measure of regional radon concentration. The measures of radon concentration are regional mean radon concentrations obtained from direct measurement in a small number of houses and purported correlates of indoor-radon concentration, such as geologic formations and housing characteristics. In an analytic case-control study, data are obtained directly from lung-cancer cases and controls, or their surrogates, through personal interviews. Radon-progeny exposure is estimated for each person and is based on either direct data from indoor-radon measurement or surrogate measures, such as housing type.
The committee concludes that only analytic case-control studies that rely on
direct measurement of radon in houses are useful for evaluating the risk of lung-cancer posed by indoor-radon exposure. In contrast, because of the inability to control for confounding at the level of the individual, limitations in the use of a few radon measurements to represent exposures for an entire region, and the large risk associated with cigarette-smoking (an excess lung-cancer risk of 1,000–2,000%, with an estimated 20–30% for indoor radon-progeny exposure), the committee believes that ecologic studies of indoor-radon exposure and lung-cancer are essentially noninformative and shed little light on the association of indoor radon-progeny exposure and lung-cancer.
In this appendix, we review the sources of exposure to radon progeny in the general population and the epidemiologic studies of indoor exposure, and we consider the results of the epidemiologic studies and their design limitations.
SOURCES OF ENVIRONMENTAL RADON EXPOSURE AMONG NONMINERS
The principal source of radon-progeny exposure in buildings is emanation from soil and rock below ground. In a few special situations, well water or building materials can contribute substantially but they make relatively small contributions to the overall dose (NCRP 1984).
The most-complete survey of radon concentrations in U.S. dwellings, the National Residential Radon Study (NRRS), was performed by EPA in 125 counties in 50 states (Marcinowski and others 1994). The arithmetic mean radon concentration was 46.3 Bqm-3 (geometric standard deviation, 3.11); the EPA action level is 148 Bqm-3. Figure 1–4 from Marcinowski and others (1994) shows that single-family homes have a slightly higher radon concentration (arithmetic mean 54.0 Bqm-3; geometric standard deviation, 2.97) than all dwellings, whereas multi-family units (defined as attached single-family dwellings, townhouses, apartments, duplexes, and condominiums) have a markedly lower mean radon concentration (arithmetic mean, 24.1 Bqm-3; geometric standard deviation, 3.23). In the survey, 6.1% of houses exceeded the EPA action level; this confirms earlier estimates, which were based on smaller studies compiled by Nero and others (1986), but it is much lower than estimates from commercial test vendors of 19% (Cohen and others 1984; Cohen and Gromicko 1988) and 23% (Alter and Oswald 1987). Presumably, the latter higher estimates result from a selection bias of homeowners who suspect they have a radon problem and not from biased measurement. Also in agreement with the earlier findings of Nero and others (1986), the NRRS found that the distribution of radon concentrations in residences could be satisfactorily represented by a lognormal distribution.
The NRRS also confirmed earlier studies that indicated that basements have higher average radon concentrations than higher floors. Their data showed that average radon concentrations in first-floor rooms were about 40% of those in basements, average second-floor room concentrations were about 90% of those
of first floors, and average third-floor or higher room concentrations were about 84% of those of second floors.
There is no evidence that average radon concentrations in U.S. dwellings are significantly different from those in other nontropical countries. The most-comprehensive international survey was compiled in the 1988 UN Scientific Committee on Effects of Atomic Radiation (UNSCEAR) report (UNSCEAR 1988), which analyzed worldwide radon surveys. The population-weighted arithmetic mean in temperate and high-latitude countries was estimated to be about 50 Bqm-3, which was combined with a "guesstimate" of about 20 Bqm-3 in tropical countries to yield a worldwide population-weighted arithmetic mean of about 40 Bqm-3.
Within a given dwelling or building, the radon concentration is determined essentially by the ratio (Scott 1992) of the average radon concentration in soil gas near foundation openings (Csoil) to the airflow resistance of soil around the house foundation (Isoil). That ratio is often called the soil-radon potential (SRP), and much effort has been devoted to its characterization. Csoil depends on depth, soil radium concentration, and water content (Rogers and Nielson 1991), and it is not well correlated with radium or uranium concentrations in the underlying bedrock (the poor correlation makes SRP predictions based on gross geologic considerations rather inaccurate). Isoil depends essentially on the basement dimensions and the soil permeability to air, which varies widely from 10 to 18 m2 for well-graded gravel, from 10 to 11 m2 for sand and gravel, and from 10 to 15 m2 for clay (Tanner 1990).
The various methods that have been used for estimating SRP have been reviewed by Yokel (1989). However, as the number of screening measurements of radon concentrations in homes has increased dramatically in the last decade, the need for predictive methods has correspondingly decreased, and radon-prone areas can be identified simply by analyzing the home measurements (Scott 1992, 1993). For example, the International Commission on Radiological Protection (ICRP 1993) has proposed that areas in which more than 1% of buildings have radon concentrations that are more than 10 times the national average might be designated as radon-prone.
In contrast with earlier expectations (for example, Rundo and others 1979; Cohen and Gromicko 1988), it now appears that house design and weatherproofing (both determinants of the rate of exchange of indoor with outdoor air) are not strong determinants of domestic radon concentration. For example, in a study of 2,000 British homes, Gunby and others (1993) found that house design, building-material type, and amount of weatherproofing together accounted for less than 5% of the observed variation in radon concentration; the implication is that, on the average, SRP is the dominant determinant.
The occupancy factor of schools and workplaces is about 2/7 that of homes (ICRP 1993). Thus, radon in schools and workplaces is likely to be an important contributor to the overall dose. ICRP (1993) has suggested that action levels in these buildings should be the reciprocal of that fraction, or 7/2, as high as in
dwellings (and should be weighted by the relevant dose-conversion coefficients, if applicable), but this suggestion has not been implemented.
Relatively few studies of radon concentrations in the workplace have been undertaken (Cohen and others 1984; Saccomanno and others 1986; Turk and others 1986; Westin and others 1991). In general, concentrations have been lower than those in local dwellings, presumably because of the larger number of floors and the greater ventilation rates in workplaces (Nero and others 1988).
Radon concentrations in schools are of particular interest because of the possible variation in radon susceptibility of children, compared with adults (Probart 1989). During 1990 and 1991, EPA undertook a randomized national survey of radon concentrations in U.S. public schools (the National Schools Radon Survey, or NSRS, EPA 1993). Of a random sample consisting of 927 public schools, about 19% had at least 1 classroom with a radon concentration above 148 Bqm-3, and 2.7% of all schoolrooms had concentrations above 148 Bqm-3. The NSRS, however, is not useful for estimating the contribution of schools to total radon-progeny exposure of children, teachers, and others from time in schools, in that measurements were conducted continuously, including on weekends and vacations, and thus failed to account for the intermittent occupancy of schools by students, teachers, and others. Typically, heating, ventilating, and air-conditioning systems would not be in use when children and teachers were away from school, but the increased concentrations during those times would not contribute to personal exposure.
ECOLOGIC STUDIES
In ecologic studies, data are considered at the group level, rather than the individual level, as in other epidemiologic studies (Morgenstern 1995). Ecologic studies typically use existing information, such as vital-statistics data, and are therefore relatively easy to perform. Ecologic studies have been useful for generating hypothesis about environmental exposures and disease but have been used less to characterize risks. Because recognized limitations in data and approximations of the form of the regression model, ecologic analyses are not generally useful for confirmatory purposes, such as risk estimation and hypothesis-testing. Piantadosi and others (1988) present several examples from a national health and nutrition survey that showed that ecologic regression coefficients based on aggregated data are larger and smaller than regression coefficients based on individual data and have opposite signs. In the case of radon risk, limitations of ecologic studies are particularly serious because of the presence of smoking, which constitutes an overarching risk factor for lung-cancer; as noted above, cigarette-smoking causes a 1,000–2,000% excess risk of lung-cancer.
Soon after the potential hazard of indoor radon was first identified, a number of ecologic studies were performed and reported. The findings of these studies were mixed in their support of the hypothesized lung-cancer risk associated with
indoor radon (Stidley and Samet 1993). One particularly large on-going study of lung-cancer mortality by county in the United States, reported in several papers by Cohen (1990, 1995) and Cohen and Colditz (1990), even shows an unanticipated inverse association between lung-cancer mortality by county and estimated average radon exposure of residents of the counties. Stidley and Samet (1993) reviewed the ecologic approach to indoor radon and lung-cancer and considered 15 studies published as of 1992. This chapter extends their review and considers the general utility of information from ecologic studies.
The Ecologic Study Design
In ecologic studies, the relation between exposure (radon) and disease (lung-cancer) is assessed by examining the association between a measure of disease occurrence (generally the age-adjusted lung-cancer mortality) in a group of people, usually those residing in a defined geographic unit, and the extent of exposure estimated for the group. Ecologic studies have proved feasible for developing hypotheses for further testing in studies at the individual level. For example, an ecologic association between breast-cancer mortality in a number of western countries and estimates of average fat consumption led to the hypothesis that higher intakes of fat increase breast-cancer risk (Armstrong and Doll 1975). Ecologic studies may also be useful if there is general homogeneity of exposure in a population. Typically, the ecologic design has not been used to assess risks associated with exposures at the individual level.
Morgenstern (1995) has classified ecologic studies by method of exposure measurement and method of grouping. Studies are termed exploratory if the primary exposure of interest is not measured and the data are analyzed to identify patterns that could lead to more-specific hypotheses. Analytic studies incorporate the exposure of interest; studies of radon have been of this type. With regard to the method of grouping, the groups in a study may come from multiple locations (multiple-group design), from multiple periods (time-trend design), or from multiple locations and periods (mixed design). The multiple-group design has been the principal ecologic approach to the study of indoor radon and lung-cancer; in the application of this approach to indoor radon, lung-cancer mortality is compared across geographic groups assumed to have a range of associated exposures to indoor radon. Stidley and Samet (1993) further classified the studies of indoor radon and lung-cancer according to the primary analytic approach—comparison of disease rates in different groups classified by radon exposure or regression of disease rates on a continuous estimate of radon exposure for the group.
Ecologic Studies of Radon and Lung-Cancer
In their 1993 publication, Stidley and Samet (1993) summarized 15 ecologic studies on lung-cancer and residential radon exposure. Through 1995, 4 addi-
tional studies were reported, including a study of lung-cancer mortality in U.S. counties by Cohen (1995). Details of those studies are provided in Tables G-1 through G-4: their approach to estimation of radon exposure in Table G-1, their outcome variables and controlled covariates in Table G-2, their handling of smoking in Table G-3, and their findings in Table G-4. Following the approach of Stidley and Samet (1993), we have broadly grouped the studies as "comparison" or "regression" studies on the basis of the primary analytic approach for assessing the effect of the radon exposure measure. In the comparison studies, disease rates and mortality are compared in 2 or more groups; in the regression studies, the outcome measure is modeled as a function of exposure.
Diverse approaches have been used to estimate the exposures of the groups (Table G-1). In the comparison studies, exposure rankings have been assigned to the groups on the basis of geology, measurements, or other factors. In most of the regression studies, data on indoor radon concentrations from population-based surveys or from less formally developed samples were used to assign quantitative exposures to geographic units. Background gamma radiation and radon concentration in well water were also used as surrogates.
The outcome measure in the studies was either the age-adjusted incidence or mortality from lung-cancer (Table G-2). The extent to which other factors were considered in the analyses was variable. Analyses were done separately by sex, with adjustment for sex, or with restriction to one sex. Socioeconomic factors and urbanization were incorporated in some studies.
A number of the studies included measures of smoking by the members of the analytic groups (Table G-3); these measures were based on cigarette-sales information and smoking surveys.
The finding of the studies vary widely, from positive and statistically significant associations between radon-exposure measures and lung-cancer rates to negative and statistically significant associations (Table G-4). A number of studies showed no evidence of association. The studies reported by Cohen have been particularly prominent because of the large number of U.S. counties included in the analyses and the strong negative association between estimated county-average radon exposure and lung-cancer mortality. We have cited two, representative reports based on Cohen's analyses, including the most recent report (1995).
In the most-recent report by Cohen (1995), data from 1,601 counties, representing most of the U.S. population, were used. Radon exposures were assigned to the counties by combining data from 3 sources: measurements made by the University of Pittsburgh, measurements made by EPA, and measurements compiled by individual states. Potential confounding by smoking was addressed by extending 1985 data on statewide prevalence to the county level with adjustment for the degree of urbanization of the county. The potential for confounding by sociodemographic factors or their correlates was explored by stratification on levels of 54 variables. Confounding by geography was assessed by stratification,
TABLE G-1 Characteristics of radon-exposure measures in 19 ecologic studies of lung-cancer and indoor radon
|
Study |
Exposure Measure |
|
Comparison studies |
|
|
Archer (1987) |
Proportion of county covered by Reading Prong granites |
|
Bean and others (1982a,b) |
Radium concentration in municipal well-water supply |
|
Dousset and Jammet (1985) |
Two regions differing by a factor of 3–4 in indoor radon concentrations |
|
Fleischer (1981) |
Proximity of phosphate mines, deposits, or processing plants |
|
Fleischer (1986) |
Proportion of county within Reading Prong |
|
Forastiere and others (1985) |
Characteristics of soil |
|
Hofmann and others (1985) |
Adjacent areas varying by radon and thoron concentrations |
|
Vonstille and Sacarello (1990) |
Indoor radon |
|
Ennemoser and others (1994) |
Indoor radon |
|
Neuberger and others (1994) |
Indoor radon |
|
Regression studies |
|
|
Cohen (1993) |
Indoor radon concentration |
|
Study |
Group measure |
Exposure grouping and number of groups |
|
Comparison studies Archer (1987) |
Location |
Reading Prong (7 counties) Border (9 counties) Control (17 counties) |
|
Bean and others (1982a,b) |
Averages based on average of 9 measurements per town taken between 1958 and 1979 |
0–74 Bqm-3 (2–5 pCiL-1) (10 towns) 74–185 Bqm-3 (2–5 pCiL-1) (9 towns) >>185 Bqm-3 (2–5 pCiL-1) (9 towns) |
|
Dousset and Jammet (1985) |
Location and radon |
Limousin (high) Poitou-Charentes (control) |
|
Fleischer (1981) |
Location |
Counties with phosphate mines (25), deposits, or processing plants (total of 316 counties) |
|
Fleischer (1986) |
Location |
Mostly within (3 counties) Less than half within (10 counties) Adjacent counties (138 counties adjacent to counties with mines) |
|
Forastiere and others (1985) |
Lithology |
Volcanic (27 municipalities) Nonvolcanic (4 municipalities) (Total population for both groups <200,000) |
|
Hofmann and others (1990) |
Location, radon, and thoron |
High background (0.38 WLM/yr) (764,696 person-yr) Control (0.16 WLM/yr) (777,482 person/yr) |
|
Vonstille and Sacarello (1990) |
Averages for U.S. Geological Survey quadrangles based on statewide survey of 6,500 homes commissioned in 1985 |
High (99 Zip-code areas) Low (1,983 Zip-code areas) None (918 Zip-code areas) commissioned in 1985 |
|
Ennemoser and others (1994) |
Location |
Austria Alp area (mean, 4,121 Bqm-3 in 178 homes), remainder of Tyrol |
|
Neuberger and others (1994) |
Location |
«8pCiL-1, 8–10 pCiL-1, >>10 pCiL-1 |
|
Regression studies Cohen (1993) |
Geometric means based on several surveys (some nonrandom) of homes: 39,000 measurements in living areas and 29,000 measurements in basements |
411 countries |
|
Study |
Exposure Measure |
|
Edling and others (1982) |
Background gamma radiation |
|
Haynes(1988) |
Indoor radon concentration |
|
Hess and others (1983) |
Radon concentration in well water |
|
Letourneau and others (1983) |
Indoor radon concentration |
|
Ruosteenoja (1991) |
Average annual indoor radon concentration |
|
Stranden (1987) |
Indoor radon concentration |
|
Magnus and others (1994) |
Indoor radon concentration |
|
Cohen (1995) |
Indoor radon concentration |
|
Source: Samet and Stidley 1993. |
|
|
Study |
Group measure |
Exposure grouping and number of groups |
|
Edling and others (1982) |
Averages based on county random sample of 1,500 homes; published in 1987 |
24 counties |
|
Haynes (1988) |
Averages based on county survey of 2,309 homes; published in 1987 |
55 counties |
|
Hess and others (1983) |
Averages weighted by proportion of rock type; survey of 2,000 wells |
16 counties |
|
Letourneau and others (1983) |
Geometric means from survey of 14,000 homes conducted in summers of 1978–1980 |
18 cities (total population about 11,000,000) |
|
Ruosteenoja (1991) |
Geometric means based on nonrandom survey of average of 120 homes per municipality conducted by end of 1985 |
18 municipalities (total population of 59,000 males in 1980) |
|
Stranden (1987) |
Averages based on nonrandom sample of 20 houses in each municipality during heating season, 2 locations per house |
75 municipalities |
|
Magnus and others (1994) |
Average based on national sample of 7,500 homes |
427 municipalities |
|
Cohen (1995) |
Average based on 3 data sets |
1,601 counties |
TABLE G-2 Outcome and controlled variables in 19 ecologic studies of lung-cancer and indoor radon
|
Study |
Outcome variable |
Controlled variable |
Comments on controlled variables |
|
Comparison studies |
|||
|
Archer (1987) |
Lung-cancer mortality, 1950–1979 |
Age |
Rates standardized to 1970 U.S. census population. |
|
|
|
Sex |
Both sexs combined; analyses by sex gave similar results. |
|
|
|
Ethnicity |
Analysis restricted to Caucasians. |
|
|
|
Socioeconomic |
Groups ''similar." |
|
|
|
Urbanization |
Counties with large cities omitted; groups "similar." |
|
|
|
Population growth |
No adjustment, but rates differed for groups. |
|
Bean and others (1982a,b) |
Lung-cancer incidence, 1969–1978 (1972 excluded) |
Age |
Rates standardized to 1970 Iowa age distribution. |
|
|
|
Sex |
Analyses done by sex. |
|
|
|
Smoking |
Not included in model, but groups checked for similarity. |
|
|
|
Socioeconomic |
Included in regression model. |
|
|
|
Urbanization |
Included towns had 1970 population of 1,000–10,000; towns categorized by size. |
|
|
|
Water characteristics |
Used as exclusion criteria or included in regression model. |
|
Dousset and Jammet (1985) |
Lung-cancer mortality, 1968–1975 |
Age |
Rates standardized to 1968 population. |
|
|
|
Sex |
Analyses done by sex. |
|
|
|
Smoking |
Similar average tobacco consumption. |
|
Fleischer (1981) |
Lung-cancer incidence, 1950–1969 |
Age |
Rates standardized to the 1960 U.S. population. |
|
|
|
Sex |
Analyses done by sex. |
|
|
|
Ethnicity |
Main analysis restricted to Caucasians. |
|
|
|
Urbanization |
Analysis included stratification by population size. |
|
Study |
Outcome variable |
Controlled variable |
Comments on controlled variables |
|
Fleischer (1986) |
Lung-cancer incidence, 1950–1969 |
Age |
Rates standardized to the 1960 U.S. population. |
|
|
|
Sex |
Analyses done by sex. |
|
|
|
Ethnicity |
Analysis restricted to Caucasians. |
|
Forastiere and others (1985) |
Lung-cancer mortality, 1969–1978 |
Age |
Analyses done by age groups or used age-adjusted rates; only those 35–74 yr old included. |
|
|
|
Sex |
Analyses done by sex or used sex-adjusted rates. |
|
|
|
Smoking |
Stratified by per capita yearly cigarette sales. |
|
|
|
Urbanization |
Largest town excluded; remaining municipalities stratified by population size. |
|
Hofmann and others (1985) |
Lung-cancer mortality, 1970–1983 |
Age |
Age-adjusted rates. |
|
|
|
Sex |
Sex-adjusted rates. |
|
|
|
Smoking |
Neighboring groups; assumed similar; women did not smoke. |
|
|
|
Socioeconomic |
Neighboring groups; assumed similar. |
|
|
|
Urbanization |
Both groups rural. |
|
|
|
Mobility |
Stable populations. |
|
Vonstille and Sacarello (1990) |
Percentage of serious illnesses that were malignant neoplasms |
Age |
Age-adjusted to 1985 Florida population. |
|
|
|
Sex |
Analyses done by sex. |
|
|
|
Socioeconomic and mobility |
Limited one analyses to the lower class in an attempt to reduce effect of mobility. |
|
Ennemoser and others (1994) |
Lung-cancer mortality, 1970–1991 |
Age |
Age-adjusted rates. |
|
|
|
Sex |
Sex-adjusted rates |
|
Neuberger and others (1994) |
Lung-cancer incidence, 1973–1990 |
Age |
Age-adjusted rates. |
|
|
|
Sex |
Women only |
|
Regression studies |
|||
|
Cohen (1993) |
Lung-cancer mortality, 1950–1969 |
Age |
Age-adjusted rates. |
|
|
|
Sex |
Analyses done by sex. |
|
|
|
Ethnicity |
Analysis restricted to Caucasians. |
|
Study |
Outcome variable |
Controlled variable |
Comments on controlled variables |
|
|
|
Smoking |
Included in regression model. |
|
|
|
Socioeconomic |
Several variables included in regression models. |
|
|
|
Urbanization |
Several variables included in regression models. |
|
|
|
Mobility |
In some analyses, radon exposures were adjusted to account for mobility; Blacks omitted to reduce effect of mobility. |
|
Edling and others (1982) |
Lung-cancer mortality, 1969–1978 |
Age |
Rates standardized to 1974 Swedish population>>40 yr; restricted to people>>40 yr. |
|
|
|
Sex |
Analyses done by sex. |
|
Haynes (1988) |
Standardized mortality ratios for lung-cancer mortality, 1980–1983 |
Age |
Age adjustment attempted through standardized rate ratios. |
|
|
|
Sex |
Analyses done by sex. |
|
|
|
Smoking |
Included in regression model. |
|
|
|
Socioeconomic |
Included in regression model. |
|
|
|
Urbanization |
Population density included in regression model. |
|
|
|
Diet |
Vitamin A consumption included in regression model. |
|
Hess and others (1983) |
Lung-cancer mortality, 1950–1969 |
Age |
Age-adjusted rates from National Cancer Institute. |
|
|
|
Sex |
Analyses done by sex. |
|
|
|
Smoking |
Concluded that smoking did not account for observed differences. |
|
|
|
Urbanization |
Concluded that population density did not account for observed differences. |
|
|
|
Mobility and growth |
Acknowledged that mobility would diminish an effect, but observed that state population has been stable in 1900s. |
|
Study |
Outcome variable |
Controlled variable |
Comments on controlled variables |
|
Letourneau and others (1983) |
Lung-cancer mortality, 1966–1979 |
Age |
Rates age-adjusted to 1971 Canadian population; restricted to those 45–79 yr old. |
|
|
|
Sex |
Analyses done by sex. |
|
|
|
Smoking |
Included in regression model. |
|
|
|
Socioeconomic |
Correlated with rates. |
|
|
|
Mobility |
Study restricted to people>>45 yr old to restrict effect of mobility, but mobility still high. |
|
Ruosteenoja (1991) |
Lung-cancer incidence, 1973–1982 |
Age |
Rates age-adjusted to world standard population. |
|
|
|
Sex |
Study restricted to males. |
|
|
|
Smoking |
Included in regression model. |
|
|
|
Urbanization |
All groups rural. |
|
|
|
Mobility |
Stable population. |
|
Stranden (1987) |
Lung-cancer incidence, 1966–1985 |
Age |
Age-adjusted rates. |
|
|
|
Sex |
Analyses done by sex. |
|
|
|
Smoking |
Included in regression model. |
|
|
|
Urbanization |
Oslo, Norway excluded. |
|
|
|
House characteristics |
Examined, but not controlled. |
|
Magnus and others (1994) |
Lung-cancer incidence, 1979–1988 |
Age |
Age-adjusted rates. |
|
|
|
Sex |
Analyses done by sex. |
|
|
|
Smoking |
Included in regression model. |
|
|
|
Asbestos |
Included in regression model. |
|
Cohen (1995) |
Lung-cancer mortality, 1970–1979 |
Age |
Age-adjusted rates. |
|
|
|
Sex |
Sex-specific analyses. |
|
|
|
Smoking |
Included in regression model. |
|
|
|
Socioeconomic |
Included in regression model. |
|
Source: Samet and Stidley 1993. |
|||
TABLE G-3 Adjustments for cigarette-smoking in 19 ecologic studies of lung-cancer and indoor radon
|
Study |
Adjustment for cigarette-smoking |
|
Comparison studies |
|
|
Archer (1987) |
No adjustment, but concluded that average smoking behavior should not differ significantly among groups. |
|
Bean and others (1982a,b) |
By examining lung-cancer rates in neighboring towns, they concluded that neighboring towns were similar to each other with respect to smoking behavior; from analysis of controls in National Collaborative Case-Control Study, they concluded that smoking rates were lower in counties with study town with high radium concentration in water than in counties with "low"-radium town. |
|
Dousset and Jammet (1985) |
No adjustment, because groups did not differ in average tobacco consumption. |
|
Fleischer (1981) |
No adjustment, but noted that average smoking rates differ only slightly among states. |
|
Fleischer (1986) |
No adjustment. |
|
Forastiere and others (11985) |
Stratified by per capita yearly cigarette sales from 1971 survey (thus, used current smoking behavior). |
|
Hofmann and others (16) |
No adjustment, because groups were assumed to be similar and women generally did not smoke. |
|
Vonstille and Sacarello (1990) |
No adjustment. |
|
Ennemoser and others (1994) |
No adjustment. |
|
Neuberger and others (1994) |
Smoking as of 1960 estimated from data from case-control study |
|
Study |
Adjustment for cigarette-smoking |
|
Regression studies |
|
|
Cohen (1993) |
Average state cigarette sales and information from state tax collections were included in regression model restricted to state averages. |
|
Edling and others (1982) |
No adjustment. |
|
Haynes (1988) |
Regression model included average weekly household expenditure on cigarettes for 1961–1963; information obtained from 1962 Ministry of Labor report, from about 20 yr before lung-cancer deaths. |
|
Hess and others (1983) |
No variable included in regression model, but concluded that smoking did not account observed differences in lung-cancer mortality rates. |
|
Letourneau and others (1983) |
Regression model included percentage of people>>45 yr old who were current smokers or ex-smokers; information obtained from Canadian Labour Force Surveys in 1977, 1979, and 1981, so current smoking behavior was considered. |
|
Ruosteenoja (1991) |
Regression model included percentage of smokers; information obtained from recent smoking survey of men 19–70-yr old in each municipality. |
|
Stranden (1987) |
Regression model included average number of cigarettes smoked/d; information obtained from a 1964–1965 study, 1–21 yr before considered lung-cancer cases. |
|
Magnus and others (1994) |
Smoking data from 1964–1965 mailed survey. |
|
Cohen (1995) |
1985 smoking survey by state, adjusted for time trend. |
|
Source: Samet and Stidley 1993. |
|
TABLE G-4 Findings of 19 ecologic studies of lung-cancer and indoor radon
|
Study |
Location of study |
Findings |
|
Comparison studies |
||
|
Archer (1987) |
Reading Prong and |
Increase in lung-cancer mortality for counties containing Reading Prong (p < 0.01), with increase from annual rate of 23.9 per 100,000 (95% CIa, 23.4–24.4) for control to 31.3 per 100,000 (95% CI, 30.5–32.1) for Reading Prong counties (rate ratio of 1.3b) |
|
Bean and others (1982a,b) |
Towns in Iowa |
Lung-cancer incidence for males increased with increasing average radium concentration in water (p<.002); relative risk of 1.68 for males with exposure >185 Bqm-3 (5 pCiL-1) to those with exposure «74 Bqm-3 (2 pCiL-1). Relative risk of 1.45 for females not statistically significant. |
|
Dousset and Jammet (1985) |
Two regions in France |
No difference in lung-cancer mortality between 2 exposure groups (rate ratios, 0.97 for males and 1.00 for females).b |
|
Fleischer (1981) |
U.S. counties |
More counties than expected with high lung-cancer rates in group with phosphate deposits or processing plants under assumption of no association between phosphate and lung-cancer rates (p < 0.0001). |
|
Fleischer (1986) |
Reading Prong counties |
More counties than expected with high lung-cancer rates in group mostly within Reading Prong under hypothesis of no geographic association with lung-cancer rates (p = 0.017 for males and p = 0.038 for females). |
|
Forastiere and others (1985) |
Towns in central Italy |
Nonsignificant increase in lung-cancer mortality in volcanic area over nonvolcanic area; standardized rate ratio of 1.22 for males (p = 0.22) with 95% CI, 0.89–1.68 and standardized rate ratio of 1.24 for females (p = 0.37) with 95% CI, 1.77–1.98.b |
|
Hofmann and others (1985) |
Two adjacent areas in China |
No association between lung-cancer mortality and radon exposure; 2.7 deaths per 100,000 in high-exposure group and 2.9 per 100,000 in control (rate ratio, 0.93).b |
|
Vonstille and Sacarello (1990) |
Florida |
No difference in percentage of total serious illnesses that were malignant neoplasms among 3 exposure groups, with 3.6%, 5.4%, and 3.9% for males in no-exposure, low-exposure, and high-exposure groups, respectively; percentage for females were lower and with no difference. |
|
Ennemoser and others (1994) |
Tyrol, Austria |
SMR for lung-cancer increased for high-radon region vs. Tyrol total; SMR = 6.2; 95% confidence interval, 4.4–8.4. |
|
Neuberger and others (1994) |
Counties in Iowa |
Effect of radon with smoking, radon, and histologic type. |
|
Study |
Location of study |
Findings |
|
Regression studies |
||
|
Cohen (1993) |
US counties |
Negative association between lung-cancer mortality and average indoor radon concentration were as follows: -0.45 (37 Bqm-3)-1 (1 pCiL-1) per 00,000 (95% CI, -0.57 to -0.33) for females; -3.38 (95% CI, -4.03 to -2.73) for males.b |
|
Edling and others (1982) |
Counties in Sweden |
Positive association between lung-cancer mortality and average background gamma-radiation exposure; correlation, 0.46 (p = 0.12) for males and 0.55 (p = 0.03) for females. |
|
Haynes (1988) |
Counties in Great Britain |
Negative association between lung-cancer mortality and average indoor radon concentration; partial correlation, -0.20 (p < 0.01) for males and -0. 16 (p < 0.01) for females after adjustment for population density, social class, smoking, and diet. |
|
Hess and others (1983) |
Counties in Maine |
Positive association between lung-cancer mortality and average radon concentration in water; correlation 0.65 (p < 0.01) for females and 0.46 (p < 0.10) for males. |
|
Letourneau and others (1983) |
Cities in Canada |
No significant association between lung-cancer mortality and average indoor radon exposure; correlation -0.34 for males and 0.13 for females; after adjustment for smoking, estimates of ß1E in Model IIa were -2.7 (95% CI, -12. to -7.5) and 0.9 (95% CI, -1.4 to -3.2) for males and females, respectively. |
|
Ruosteenoja (1991) |
Municipalities in Finland |
No significant association between lung-cancer incidence and average indoor radon concentration; adjusted for smoking, relative risk, 1.08 for 100 Bqm-3 (95% CI, 0.92–1.27); weighted correlation, 0.36 (p = 0.14). |
|
Stranden (1987) |
Cities in Norway |
Positive association between lung-cancer incidence and average radon exposure; 95% CI for lifetime relative risk, 0.001–0.003 (Bqm-3)-1 radon. |
|
Magnus and others (1994) |
Municipalities in Norway |
No overall association with radon; significant increase for small-cell carcinoma in women. |
|
Cohen (1995) |
U.S. counties |
Negative association between lung-cancer mortality and average indoor radon concentration; smoking-adjusted coefficients, -7.3 per pCiL-1 per 100,000 for men and -8.3 per pCiL-1 per 100,000 for women. |
|
a CI = confidence interval/ b Some numeric results were calculated from information provided in the articles. Source: Samet and Stidley 1993. |
||
and the sensitivity of the findings to outliers was examined. There was a strong negative association between 1970–1979 lung-cancer mortality and the county-average radon concentrations; the association could not be explained by confounding. In interpreting this finding, Cohen proposes that the negative association implies failure of the linear, nonthreshold theory for carcinogenesis from inhaled radon decay products.
Limitations of the Ecologic Design for Investigating Indoor Radon and Lung Cancer
Methodologic limitations of the ecologic design have received extensive treatment in recent publications in the epidemiologic literature (Greenland and Morgenstern 1989; Brenner and others 1992; Greenland and Robins 1994a; Morgenstern 1995). Morganstern's (1995) review provides a framework for considering the limitations of ecologic studies of indoor radon and lung-cancer. He notes that the goal of an epidemiologic study might be to draw biologic inferences about individual risks or ecologic inferences about group rates. In the ecologic studies of radon and lung-cancer, the goal is to make inferences about the radon-associated lung-cancer risk of individuals, so there is a potential for cross-level bias as observations made at the group level (such as, the county level) are extended to individuals (for example, the county residents). The extension of quantitative risk estimates from ecologic studies to the individual level is also problematic. Estimated risks depend strongly on the choice of model form (Morgenstern 1995). Control of confounding might be accomplished by regression modeling (which includes stratification) or standardization, as typically done for age. However, in the context of ecologic studies, regression modeling for control of confounding might be unsuccessful unless a series of conditions are met with regard to associations among predictors and disease rates (Greenland and others 1989; Morgenstern 1995). Standardization for control of confounding might be unsuccessful unless all predictors are mutually standardized for the same confounders—a condition that requires data misspecification (Brenner and others 1992; Morgenstern 1995). Effect modification, that is, interaction effects—further complicates interpretation of ecologic estimates of risk.
Ecologic bias has long been known to be a principal limitation of the ecologic study design. This bias refers to the difference between associations at the group and individual levels (Morgenstern 1995). Ecologic bias has been given quantitative definition (Greenland and others 1989). Greenland and Morgenstern (1989) have described sources of ecologic bias in using linear regression to estimate the exposure effect; these sources include biases acting within a group, confounding by group, and effect modification by group. Other forms of bias can affect ecologic studies, including inadequate control of confounding, model
misspecification, and misclassification. Morgenstern (1995) also lists a lack of adequate data, temporal ambiguity of the exposure-disease relationship, collinearity of predictor variables within groups, and migration across groups.
In their 1993 review, Stidley and Samet (1993) specifically addressed limitations of ecologic studies of indoor radon and lung-cancer, covering measurement error and model misspecification. Each of the 15 studies was reviewed for 14 potential limitations in those 3 broad categories. All studies were found to have multiple limitations.
Stidley and Samet noted 5 sources of measurement error: use of current exposure to represent the biologically relevant period of past exposure, the inherent error of the measurement devices, use of an indirect measure of indoor concentrations as an index of indoor radon exposure, use of sample rather than total-population information, and estimation of individual exposure by a group indicator, the ecologic fallacy.
Within a region, radon concentrations for houses are extremely variable (Piantadosi and others 1988) and estimates of regional mean concentrations are usually derived from relatively few measurements. Radon concentrations are derived from measurements in houses, which are occupied an average of 60–70% of the time; the remaining 30–40% of people's time spent in other houses, in workplaces, outside, and so on. Time spent in unmeasured areas increases exposure uncertainty. And current measurements might not accurately reflect radon exposures of individuals over the last 30 yr or more. The effects of exposure errors can bias results in many ways; trends can increase or decrease or even reverse direction.
With regard to the fourth source of measurement error (use of sample data), Stidley and Samet showed that there are substantial probabilities of misclassifying counties or other geographic units as to exposure if only a few measurements are available. In an expanded analysis, Stidley and Samet (1994) assessed the impact of measurement error on the estimated effect of radon and on the standard error of the regression coefficient describing the ecologic relationship between radon and lung-cancer risk. They found that the effect of radon and the standard error of the effect estimate were underestimated because of measurement error; the degree of bias was greater for smaller samples. The underestimation of the standard error would tend to overstate significance levels for tests on regression coefficients.
Model misspecification refers to a biologically incorrect formulation of the relationship between radon exposure and lung-cancer risk. Possible errors in model specification include omission of confounders (such as, age and smoking) or effect modifiers (such as smoking), the use of inappropriate functional forms (such as linear rather than loglinear increase in risk posed by an exposure if the latter were correct), and the use of an incorrect form of the model (such as an additive model for the joint association of radon exposure and smoking rather than a supra-additive model). Effect modification at the individual level is an intractable problem at the ecologic level (Stidley and Samet 1993, 1994).
Miner data clearly indicate that the relationship of lung-cancer occurrence in a person, to cumulative exposure, is not simply linear, and that the joint relationship of radon and smoking is not additive. The ''best" models indicate that the regression relationship depends on cumulative radon progeny exposure and on attained age, time since exposure, and exposure rate, although at the concentrations of radon found in homes exposure rate might be of less importance. In addition, effects of smoking are greater than additive. Thus, for analyses of aggregated data, the model for age-specific rates is not a simple linear regression in exposure and smoking. Ecologic regressions typically fit linear models to age-adjusted rates and to estimates of radon concentration and smoking. The dependence of the radon-progeny exposure effects on the various factors implies that comparisons can be made only among regions that have the same population profile for age and past radon exposures, including exposure rates, or these factors must be age-standardized to a common population. Furthermore, regions must have a similar joint distribution for radon-progeny exposure and smoking.
When the exposure-response relationship is linear, there are no group-level effects, and regressor variables are measured without error, population cumulative exposure can be used to obtain an unbiased estimate of the exposure-response parameter. However, that simplification, which theoretically might arise when the true model for individuals is linear, does not apply for radon-progeny exposure and lung-cancer. The complexity of the risk model at the individual level (exposure-response effects with age-specific, time-since-exposure and exposure-rate variations and multiplicative or submultiplicative effects of smoking) does not lend itself to a simply linear approximation for aggregated data and guarantees that a linear model for age-adjusted disease rates is misspecified.
Statistical power of published reports was considered in the 1993 review of Stidley and Samet (1993). They found power to be limited for the reported studies; given the expected magnitude of effect of radon on lung-cancer risk at typical indoor concentrations, inadequate statistical power can lead to the incorrect conclusion that there is no association.
Stidley and Samet (1994) and Greenland and Robins (1994a) have further considered limitations of ecologic studies of radon and lung-cancer. Using simulation, Stidley and Samet (1994) assessed the sensitivity of the ecologic design to confounding by cigarette-smoking. The average estimate of the effect of radon was negative when the correlation between radon and smoking was between -0.17 and -1.00 (Figure G-1). In an additional series of simulations, they explored the consequences of model misspecification, assessing the findings of a simple linear-regression model when the underlying model is nonlinear. They showed that age-dependent risks and smoking-specific risks can be incorrectly estimated by simple regression methods.
Greenland and Robins (1994a) considered biases that affect ecologic studies, using a number of examples based on investigating radon and lung-cancer. They provide an informative example based on a multiplicative relationship between
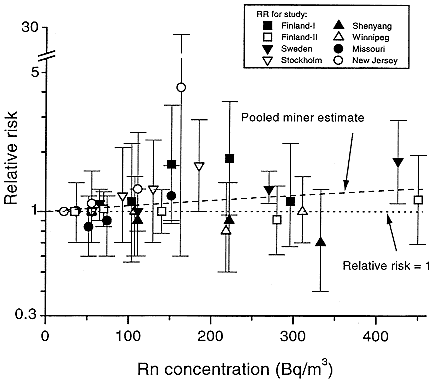
FIGURE G-1 Relative risks from 8 lung-cancer case-control studies of indoor radon. Dashed line, extrapolation of risk from miners (Lubin and others 1994); dotted line; relative risk of 1.
lung-cancer and radon level and smoking that mimics the negative exposure-response results of Cohen (1995). The example is based on the following: lung-cancer occurrence is positively associated with radon concentration and cigarette-smoking rate, with the relationship linear in radon level and jointly multiplicative in radon concentration and cigarettes smoked per day; proportions of never-smokers, 1-pack/d smokers and 2-pack/d smokers vary by region; smoking rates vary by region (smoking rate is higher in regions with lower proportions of ever-smokers), so regional mean smoking rate is independent of region; and radon concentration is uniform within region but varies by region and is negatively correlated with the proportion of ever-smokers. Even though the "true" relationship specifies an increasing risk with radon concentration, and the ecologic regression of lung-cancer rates on mean regional smoking rate shows a positive exposure-response relationship, the regression of lung-cancer rates on regional radon concentration shows a negative exposure-response relationship.
The example does not prove that confounding from smoking is causing the negative regression in Cohen's analysis, but it shows that results of an ecologic regression can be affected by a risk factor that is confounding at the level of the individual, but not at the level of region.
That example and others in the same paper make it clear that conditions for confounding at the individual and ecologic levels are distinct and that regression methods might not fully control confounding. Greenland and Robbins also note misconceptions concerning ecologic regression: the incorrect assumption that a linear model should approximate the true model because of Taylor's theorem; failure to recognize that nonlinearities at the individual level can lead to ecologic bias; an incorrect belief that important departures from linearity in the individual-level model will be detected by a test of fit of the ecologic linear model; an incorrect belief that having a large number of analytic groups, such as, regions, will ensure a random relationship between exposure and covariates; and an incorrect assumption that for ecologic bias to be present, region must be a confounder on the individual level with control of other factors.
Cohen (1994), in responding to Greenland and Robins, dismissed those limitations as not applicable to ecologic studies of radon and lung-cancer. He argued that his ecologic study has at least 4 advantages over an individual study: a large number of data points, the small degree of uncertainty affecting county mortality, the availability of "good" data on many potential confounding factors, and the size and diversity of the population being studied. However, Cohen's response did not specifically address the inherent limitations of ecologic studies and ecologic regression, as detailed by Stidley and Samet (1993) and Greenland and Robins (1994a). Greenland and Robins (1994b), in responding to Cohen, found little merit in his responses and disagreed with a principal assertion of his: that the ecologic fallacy does not affect a test of "linear-nonthreshold theory."
Piantadosi (1994), commenting on the exchange, suggested that Cohen's findings do "more to discredit the analysis than the theory." He elaborated: "The result of Cohen's analysis will seem biologically implausible to many investigators although it is probably theoretically possible at the individual level. Many epidemiologists will likely attribute the discrepancy between theory and result more to deficiencies in ecologic analyses than to failure of the dose-response theory ... Most of us would not be willing to discard a useful theory on the basis of such a test." Like Greenland and Robbins, Piantadosi is concerned by the limitations of ecologic analyses and the inability to determine whether bias is present and to estimate the direction and magnitude of its effect.
Uncontrolled confounding by smoking remains an explanation for the negative association between radon and lung-cancer reported by Cohen. Stidley and Samet (1994) noted that there might be confounding in Cohen's analysis reflecting the higher concentrations of radon in western states, where smoking tends to be lower than elsewhere in the country. Gilbert (1994) further noted that other
smoking-related cancers are also negatively associated with radon concentration in Cohen's data, possibly providing further evidence of confounding.
Conclusions
Although a number of ecologic studies have been published since the BEIR IV report, the present committee did not find the new evidence to be informative concerning the risks posed by radon. The finding of a statistically significant negative association between radon and lung-cancer in Cohen's analysis of lung-cancer mortality in the United States was considered to have resulted from inherent limitations of the ecologic method. That analysis has been widely cited as weighing against any risk of lung-cancer at typical indoor radon concentrations (White and others 1992; Marcinowski and others 1994). The finding was considered to be an inappropriate basis for concluding that indoor radon is not a potential cause of lung-cancer. We also note that the case-control studies reported to date, although limited in statistical power, have not yielded evidence of a negative association between exposure to radon progeny and lung-cancer risk. The ecologic studies were also not considered to be an appropriate basis for quantitatively estimating lung-cancer risk associated with radon exposure. Ecologic regression coefficients can be biased, and extensive individual-level data are available for estimating risk.
CASE-CONTROL STUDIES
The most-direct evidence of health consequences of radon-progeny exposure in homes is offered by case-control studies, in which characteristics of lung-cancer patients are compared with those of control subjects who do not have the disease. After age, smoking, and other factors are accounted for, if residential radon causes lung-cancer, it would be expected that the mean of a measure of exposure of cases would exceed the mean of controls, given proper assessment of statistical sampling variation. Exposure measures are usually based on a surrogate thought to be correlated with exposure, such as type of home construction, or on a more-direct correlate, such as measured radon in current and past homes.
Although straightforward in principle, case-control studies of residential radon are burdened with several limitations. These are discussed later in this chapter and include in particular an inability to measure radon in current and all past homes and thereby create an accurate measure of exposure, the lack of an estimate of radon exposure outside the home, and the relatively small relative risk (RR) that is expected even for long-term residents of higher-radon homes, which are not common. A small RR implies that mean exposures of cases and controls differ by only a small amount, thus limiting study power. The detection of an excess risk of lung-cancer is potentially complicated also by an inability to
control completely for other lung-cancer risk factors, particularly cigarette-smoking, which has an RR of 10–20.
In this section, we review case-control studies, first those which use surrogate measures of exposure and then those in which direct measurements of radon concentration in homes were used.
Studies Using Surrogate Measures of Exposure
Many of the earliest studies of the effects of residential radon exposure relied on surrogate measures of exposure, such as housing style, for example, the presence or absence of a basement, the type of construction materials, or the characteristics of the local geology. Investigators often supplemented their observational data on houses with direct measurements of radon concentration to validate their "exposure" classifications. Table G-5 adapted from Samet (1989), lists the principal studies that used surrogate measures as the primary source of exposure classification. In those studies, measured concentrations were generally positively correlated with housing characteristics thought to be related to high indoor radon concentrations. For example, in several studies conducted in Sweden, radon measurements were related with their classification scheme whereby wood houses without basements on normal ground were classified as low-radon houses; wood houses on alum shale (known to have high radiation emanation rates), stone houses with basements, and stone houses without basements on alum shale were classified as high-radon houses; and the remainder were classified as moderate-radon houses. But housing type was not always directly related to radon concentration. In the Stockholm County study by Svensson and others (1989), which was supplemented by direct radon measurements in houses and reported by Pershagen and others (1992), measured houses with ground contact classified by "type of ground" had geometric means of 99, 108 and 153 Bqm-3 for low, moderate and high categories, respectively.
Results of the studies varied, but, the overall pattern of RRs suggests a positive association between the surrogate measure of radon concentration and lung-cancer risk, with an RR for the high-radon houses about twice that for the low-radon houses. When data were available, results were not materially affected by controlling for smoking. Because the links between the radon potential of a house and actual radon concentration and between radon concentration and individual exposure are uncertain, it is difficult to interpret the RRs in relation to extrapolations from miners or to studies in which radon concentrations in houses were measured.
The agreement among the studies that used surrogate measures complicates their interpretability, in that the results appear more consistent than do results of studies in which indoor radon was measured directly. Classification of indoor radon concentrations based on housing type or building materials might be expected to be less accurate and to have greater random and systematic errors in
TABLE G-5 Epidemiologic studies of residential redon-progeny exposure and lung-cancer with surrogate measures of exposure
|
Location: Reference |
Study Design |
Subjects |
Exposure |
Resultsa |
|
Southern Sweden: Axelson and others 1979 |
Case-control |
37 cases deceased in 1965–1977 and 178 controls deceased at same time as cases, excluding cancers; rural residents only |
Residence type: wood without basement, "mixed," or stone with basement |
RR=1.8 [95% CI (1.0,3.2)] for stone and mixed, compared with wood |
|
Oeland, Sweden: Edling and others 1984 |
Case-control |
23 cases deceased in 1960–1978 and 202 deceased controls |
Residence type: wood without basement on normal ground, "mixed," or wood on alum shale, stone with basement and stone without basement on alum shale; 1 mo measurements in some homes |
RRs of 1.2 [90% CI (0.5,3.1)] and 4.3 [90% CI (1.7,10.6)] for intermediate-and high- vs. low-exposure categories; p for trend <0.01. |
|
Southern Sweden: Edling and others 1984 |
Case-control |
23 cases and 202 controls |
Measurement with a-sensitive film |
RR increased for hightest- vs. lowest- exposure categories. |
|
Northern Sweden: Pershagen and others 1984 |
Case-control (matched pairs) |
15 never-smoker and 15 ever-smoker case-control pairs |
Construction characteristics |
Estimated mean exposure significantly higher for smoking cases than controls; exposure not different for nonsmokers. |
|
Sweden: Pershagen and others 1984 |
Case-control (matched pairs) |
11 never-smoker and 12 ever-smoker case-control pairs |
Construction characteristics |
Estimated mean exposures similar for cases and controls, regardless of smoking status. |
|
Location: Reference |
Study Design |
Subjects |
Exposure |
Resultsa |
|
Northern Sweden: Damber and Larsson 1987 |
Case-control |
589 male cases, 582 deceased controls, and 453 living controls |
Residence type: wood or nonwood |
RR not increased with or without smoking adjustment; RR increased for those never employed in non-lung-cancer-related occupations. |
|
Stockholm, Sweden: Svensson and others 1987 |
Case-control |
292 female cases diagnosed in 1972–1980 and 584 controls, resident in Stockholm for at least 28 of prior 30 yr |
Geology and living near ground level; grab-sample measurements in some homes |
RR=2.2 [95% CI (1.2,4.0)]; exposure-response trend not significant. |
|
Southern Sweden: Axelson and others 1988 |
Case-control |
177 cases deceased 1960–1981 and who lived in same house at least 30 yr before death and 677 controls deceased in same years of noncancer causes |
Residence type: wood without basement on normal ground, "mixed," or wood on alum shale, stone with basement and stone without basement on alum shale; 2 mo measurements in some homes |
RR=1.8 [90% CI (1.0,3.3)] for nonsmokers and light smokers in rural areas; no association for smokers in rural areas or for urban residents. |
|
Location: Reference |
Study Design |
Subjects |
Exposure |
Resultsa |
|
Stockholm County, Swedenb: Svensson and others, 1989 |
Case-control |
187 female cases in 1983–1985, 160 "hospital" controls with suspect lung-cancer found not to have the disease, and 177 population-based controls |
Geology and living near ground level; 2-w radon measurements during the heating season in a sample of homes |
RRs of 1.8 [95% CI (1.2,2.9)] and 1.7 [95% CI (0.9,3.3)] for intermediate-and high- vs. low-exposure categories; p for trend 0.03. Slight variation of risk by cell type or smoking status; steeper RR trend at highest ages. |
|
Washington County, Maryland, U.S.A.: Simpson and Comstock 1983 |
Cohort |
298 cases over 12-yr period |
Housing characteristics |
No association of incidence with housing characteristics. |
|
a Parentheses provide 95% confidence interval for RR. b Initial study, which was later expanded to include indoor radon measurements and reported by Pershagen and others 1992. Source: adapted from Samet (1989). |
||||
exposure assessment; as a result, there would likely be greater attenuation of the observed association and greater variability in the outcomes of the independent studies. However, one should not necessarily conclude that studies using surrogate measures will be more misclassified than those using actual measurements, because their error structures differ fundamentally. There is a possibility that such might have some advantages. The ideal study would include both measurements and surrogates, and both should be included in an analysis incorporating measurement errors.
Results of these studies should therefore be interpreted cautiously. In addition, several of the studies had few lung-cancer cases, that often precluded subgroup analyses, which would permit evaluation of both internal consistency and consistency among studies.
Studies with Direct Measurements of Indoor Radon
Potentially, the most important source of direct information on the consequences of exposure to indoor radon is epidemiologic studies in which long-term measurements of radon concentration were carried out. Several such studies have been done, and they are described below. All studies used a case-control design, in which estimates of radon or radon-progeny exposure of lung-cancer subjects are compared with estimates of exposure of controls selected from the same target population as the cases taking account of other factors that might influence the comparison—such as age, smoking status, and sex—are accounted for.
Case-control studies that incorporate direct measurement of indoor radon concentrations have several advantages over ecologic studies and over case-control studies that use surrogate exposure measures. Such case-control studies must be viewed as generally having greater validity for the identification and ultimately the quantification of an excess risk of lung-cancer. In contrast with an ecologic study, a case-control study offers a well-defined target population, and outcome status is assessed unambiguously. Direct, long-term measurement of radon in houses permits estimation of exposures specific to individuals, thereby reducing exposure errors, compared with ecologic studies and studies that define exposure in terms of house type. Direct measurement data permit the reconstruction of historical exposure profiles and the evaluation of biologically plausible exposure periods. With direct measurement data, it is possible to evaluate the consequences of missing data and the effects of various imputation approaches. In addition, measurements from track-etch devices are generally more comparable across countries than are crude classifications by house type and so allow more valid comparison among studies from different countries. Thus, results of ecologic studies are considered noninformative, results of case-control studies that use surrogate exposure measures are provocative, and case-control studies with direct, long-term radon
measurement offer the best opportunity for identifying an excess risk of lung-cancer associated with indoor radon.
New Jersey Case-Control Study of Females
Study Subjects
The radon component of this study was an add-on to a continuing lung-cancer case-control study of females in New Jersey (Schoenberg and others 1990; Klotz and others 1993). The original case group consisted of 1,306 female residents with histologically confirmed lung-cancer diagnosed from August 1982 through September 1983 (see Table G-6). They were identified from hospital pathology records and from the New Jersey State Cancer Registry and death-certificate files. Data were collected on 994 cases (76%) from 532 in-person interviews and 462 next-of-kin interviews.
Controls were selected from New Jersey drivers-license file, on those under age 65 and from Health Care Financing Administration file, on those 65 and over. For cases with next-of-kin respondents, controls were selected at random from death certificates, excluding deaths from respiratory disease. Controls were individually matched to cases by race, age, and, for deceased cases, closest date of death. A total of 1,449 controls were identified, and interview data were obtained on 995 women (69%).
Houses to be measured were defined in the study in 2 phases (Table G-7). In phase I, a single "index" residence per subject was chosen in which the subject lived for at least 10 yr in the period 10–30 yr before diagnosis or selection. In phase II, the residence criteria were broadened, to add subjects to the radon component of the study and houses for subjects selected in phase I. The eligibility period for the index residence was extended to cover the period 5–30 yr before diagnosis, and the study enrolled all houses in which a subject resided for 4 yr or more in the 6 New Jersey counties with high average radon concentrations, or for 7 yr or more in the rest of the state. Twelve subjects were excluded because their eligible residences represented less than 9 yr of coverage in the exposure-time window. Of the 994 cases and 995 controls with completed interviews in the original study, 661 cases (66%) and 667 controls (67%) had residences that were eligible under the expanded phase II criteria.
Data Collection
Subjects or surrogate respondents were interviewed by trained interviewers. Study subjects provided 53% of the interviews, spouses 17%, and other next-of-kin 29%. Information was obtained on lifetime smoking history, smoking by other household members, lifetime residential and occupational histories, and consumption of food high in vitamin A. Information on specific addresses of past residences was collected several years after the original interview through telephone contacts. During eligible residencies, information was obtained on house characteristics, including heat circulation and modifications to the structure or to heating and ventilation.
Methods of Radon Measurement
Long-term a-track detectors were deployed
TABLE G-6 Summary of results of New Jersey female case-control study
|
Factor |
Comment |
|
Principal references |
Schoenberg and others 1990, 1992. |
|
Design |
Case-control study in females. |
|
Study subjects |
Cases: Cases were selected from 1,306 histologically confirmed lung-cancers in females diagnosed in August 1982 through September 1983 throughout the state. In original study, 994 women were interviewed: 532 subjects and 462 next-of-kin. Cases for the radon analysis were further restricted by measurement protocol Phases I and II. |
|
|
Controls: For living cases, controls were selected randomly from New Jersey driver's license files (age < 65 yr) or Health Care Financing Administration files (age = 65 yr). For next-of-kin cases, controls were selected randomly from death certificates that did not mention respiratory disease. Controls were matched by race, age, and, for deceased cases, date of death. |
|
|
Subjects in radon study: Phase I included subjects who had lived in a single residence = 10 yr in the period 10–30 yr before diagnosis or selection; phase II included subjects who had lived in one or more residences in the period 5–30 yr before. Phase II added subjects to phase I and added houses. Subjects restricted to those with 9 yr of coverage. Totals of 480 cases and 442 controls were included. |
|
Lung-cancer histology |
480 cases: squamous 25.8%; small cell 29.8%; adenocarcinoma 21.9%; other 22.5%. |
|
Rn measurement protocol |
Measurements: 1-yr a track in living area (76%), 1-yr a track in basement (5.4%), basement and upstairs charcoal canister (6.5%), upstairs charcoal canister (1.4%). Canister below minimal detectable concentrations assigned MDC concentration. Apartments above the 2nd floor assigned 0.4 pCiL-1. Regressions linked basement and canister measurements to long-term values for living areas. |
|
|
Missing: Under phase II eligibility, 74% of cases and 72% of controls had measurements. |
|
Rn measurements |
Mean: Cases, 0.7 pCiL-1; controls 0.7 pCiL-1. |
|
|
Median: Cases, 0.5 pCiL-1; controls 0.5 CiL-1. |
|
Rn-exposure estimation |
Exposure-time window: 5–30 yr before the date of case diagnosis or control selection. |
|
|
Coverage: Median 22 yr for cases and for controls; 35% of subjects fully covered. |
|
|
Imputation for gaps: None for TWA radon exposure; for cumulative exposure, 0.6 pCiL-1 (22 Bqm-3) was assigned for missing intervals. |
|
Results |
Overall: For categories <1, 1–1.9, 2–3.9, and = 4 pCiL-1, RRs were 1.0, 1.2, 1.1, and 8.7, with p value for 1-sided test of linear trend 0.04. Only 5 cases and 1 control in highest category. |
|
Factor |
Comment |
|
|
Histology: Increasing RRs with pCiL-1 only for ''other" cell types; no trend with other histologic types (Lubin and others 1994). |
|
|
Smoking: For RR with pCiL-1 no trend in never-smokers, increasing RRs for <15 and 15–24 cigarettes/d, and decreasing trend for =25 cigarettes/d. |
TABLE G-7 Distribution of 944 cases and 955 controls in original New Jersey lung-cancer case-control study by status in radon substudy for Phase I only and Phase I and II
|
|
No. (%) Subjects |
|||
|
|
Phase I only |
|
Phase II only |
|
|
Status |
Cases |
Controls |
Cases |
Controls |
|
Included in radon studya |
433 (44) |
402 (40) |
480 (48) |
442 (44) |
|
No address-specific informationb |
140 (14) |
126 (13) |
168 (17) |
152 (15) |
|
No address met residence criterionc |
253 (25) |
256 (26) |
165 (17) |
176 (18) |
|
No radon testing at index addressd |
168 (17) |
211 (21) |
181 (18) |
255 (23) |
|
a Index residence(s) tested for radon with a-track detectors or charcoal canisters. If index residence was apartments above the 2nd floor, radon exposure assumed to be < 1 pCiL-1. Seven cases and 5 controls with complete measurements in phase I or II were excluded because they represented 8 yr or less of 25 yr exposure history. b Respondent refused further contact after initial interview, respondent lost to follow-up, respondent refused address-specific information, or inadequate address-specific information. c Subject did not meet residence criterion for inclusion in phase I (phase I and phase II). d Index residence demolished, refusal by current resident, or no contact with current resident. Source: Schoenberg and others 1992. |
||||
for 1-yr. In each dwelling, 1 detector was placed in the living area, usually the master bedroom, and another in the lowest habitable level, usually the basement. In addition, 4-d screening measurements were made with the house closed, during the heating season, with charcoal canister detectors. The screening measurements were used primarily as a backup if long-term measurements could not be completed and to identify homes that required immediate mitigation. The radon concentration used for the house was based on the nonbasement primary-living-area a-track measurement when available (76% of houses). When it was unavailable, the nonbasement radon concentrations were estimated from other measurements in descending order of priority: basement a-track (5.4%), basement charcoal canister with upstairs canister (6.5%), and upstairs charcoal canister (1.4%). The estimates were derived from regression equations based on complete sets of measurements which also took into account the heating system (forced air versus
other). Canister readings below the minimal detectable concentration (MDC) were assigned the MDC value. Apartments above the 2nd floor (10.6%) were assigned a value of 14.8 Bqm-3 (0.40 pCiL-1). Usable measurements were obtained for 480 cases and 422 controls or, 74% and 72%, respectively, of those eligible under the Phase II criteria.
For analysis, 2 measures of exposure were developed. The time-weighted average (TWA) radon concentration was the mean concentration for all houses measured weighted by the years of residence in the exposure-time window of 5–30 yr. Cumulative radon exposure was computed as the product of residence time within the 5–30 yr and measured radon concentration. Within the 5–30 yr, unmeasured houses were assigned a radon concentration of 22.2 Bqm-3 (0.6 pCiL-1), the median concentration of all phase I control houses.
Results
On the basis of phase II data, the mean residence time within the 5–30 yr was 22 yr for cases and for controls; 35% of the subjects had radon measurements for all their homes in the exposure period. The houses in the New Jersey study had the lowest radon concentration of any of the current case-control studies; most of the TWA concentrations were below 37 Bqm-3 (1 pCiL-1). The median radon concentration was 18.5 Bqm-3 (0.5 pCiL-1) and was the same for cases and controls.
Table G-8 shows the RRs for categories of TWA radon concentration adjusted for cigarettes per day, cessation of smoking, age, occupation, type of respondent, and interaction of type of respondent with cigarettes per day (Schoenberg and others 1992). RRs were flat and increased only in the highest category, 148 Bqm-3 (4.0 pCiL-1), which included 5 cases and 1 control. The p-value for linear trend was significant at p = 0.05 but was based on a 1-sided, rather than the traditional 2-sided, test of the null hypothesis. For this study, it should be pointed out that 90% Cls were used for RR rather than the more
TABLE G-8 Distribution of cases and controls and adjusted odds ratios a (OR) and confidence intervals (CI) by time weighted average (TWA) radon concentration for the New Jersey case-control study of females
|
|
TWA Rn concentration, pCiL-1 |
|||||
|
|
<1 |
1–1.9 |
2–3.9 |
=4 |
Total |
P for trend |
|
No. Cases |
384 |
72 |
19 |
5 |
480 |
— |
|
No. Controls |
360 |
69 |
12 |
1 |
442 |
— |
|
ORa |
1.0 |
1.2 |
1.1 |
8.7 |
— |
0.05b |
|
90% CI |
|
(0.8,1.7) |
(0.6,2.3) |
(1.3,57.8) |
— |
— |
|
a Adjusted for lifetime average cigarettes/d, years since smoking cessation, age, occupation, respondent type, and interaction between respondent type and cigarettes/d. b One-sided test of linear trend Source: Schoenberg and others 1992. |
||||||
conventional 95% Cls. Results for cumulative radon exposure were similar to those for TWA radon concentration (Schoenberg and others 1992).
Shenyang China Case-Control Study of Females
Study Subjects
Like the New Jersey study, the radon component of this study was an add-on to an existing lung-cancer case-control study of woman in Shenyang China (Xu and others 1989; Blot and others 1990). Potential cases were female residents of Shenyang, who were 30–69 yr old and were listed in the Shenyang Cancer Registry with primary lung-cancer in September 1985 to September 1987 (Table G-9). In the full study, 75% of the diagnoses for the female lung-cancer cases were based on pathologic or cytologic material; histologic information was available on 73% of all female cases (Xu and others 1989).
A population-based, age-matched control group of women was selected from the Shenyang general population by using the system of area administrative units and neighborhood population lists. Controls were randomly selected in 5-year age groups to reflect the age distribution of the cases.
The radon component of the study was initiated 6 months after the start of the original study and, because of budgetary reasons, ended before completion of case acquisition in the full study. A total of 397 cases and 391 control subjects had detectors placed in their houses, representing 95% and 99% of eligible cases and controls, respectively.
Data Collection
Trained nurses sought personal interviews with the subjects, except for those who were too ill or deceased. Participation rates were 95% for cases and 97% for controls. For most patients, the time between diagnosis and interview was less than 1 month. A structured questionnaire was used in an interview to inquire about smoking by the subject and other household members, occupation, prior medical conditions, residential history, and housing characteristics, such as indoor air pollution. A time-weighted index of lifetime air-pollution exposure was determined from housing characteristics, including type of heating, fuel used for cooking, and whether cooking facilities were in a separate kitchen or combined with living room or bedroom (Xu and others 1989).
Methods of Radon Measurement
Two a-track detectors were placed for 1 yr in the current residence of each case and control; 1 detector was usually in the living room and 1 in the bedroom. Nearly all homes were single-story buildings. For persons who lived in the current house less than 5 years, a prior Shenyang residence was tested, provided that it was accessible and the subject had lived there at least 5 years. Detectors were collected for 308 cases (78%) and 356 controls (91%).
The maximum of the 2 measurements were used in analysis. Among the paired measurements, the correlation was 0.52, 77% were within 74 Bqm-3 (2 pCiL-1), and 78% of the ratios of the 2 measurements were less than a factor of 2.
Results
Among the subjects, the median number of reported residences was 3, the median residence time in the last home was 24 yr, and 76% lived in the
TABLE G-9 Summary of results for Shenyang, China female case-control study
|
Factor |
Comment |
|
Principal reference |
Blot and others 1990. |
|
Design |
Case-control study of females. |
|
Study subjects |
Cases: Cases included all female residents of Shenyang, China, aged 30–69 yr with primary lung-cancer diagnosed in September 1985 to September 1987 and listed in the Shenyang Cancer Registry. All case diagnoses were reviewed. |
|
|
Controls: Controls were randomly selected in 5-yr age groups from the general population. |
|
|
Subjects in radon study: For the radon component, ascertainment was delayed 6 mo but included all subjects. A total of 308 cases and 356 controls had radon measurements. |
|
Lung-cancer histology |
308 cases: squamous, 23.4%; small cell, 12.7%; adenocarcinoma, 30.8%; other or unknown 31.1%. |
|
Rn measurement protocol |
Measurements: 1-yr a-track detectors in living room and in bedroom of current home. For those who had lived for < 5 yr in the current home, 1-yr a-track detectors were placed in the previous residence if it was in Shenyang and accessible and subject had lived there 5 yr. |
|
|
Missing: Among those eligible, 79% of cases and 91% of controls had measurements. |
|
Rn measurements |
Median: Cases, 2.8 pCiL-1; controls, 2.9 pCiL-1. |
|
Rn-exposure estimation |
Exposure-time window: 5–30 yr before case diagnosis or control selection. |
|
|
Coverage: Median residence in last home was 24 yr, and 76% lived in measured home = 10 yr. |
|
|
Imputation for gaps: None; analyzed only measured radon concentration. |
|
Results |
Overall: For categories <2, 2–3.9, 4–7.9, and =8 pCiL-1, RRs were 1.0, 0.9, 0.9 and 0.7 and 1.0, 0.7, 1.2, and 0.7 when analyses were restricted to subjects who lived =25 yr in their last residence. |
|
|
Histology: RRs for small cell for pCiL-1 categories were 1.0, 1.2, 1.7, and 1.4, but with nonsignificant trend. |
|
|
Smoking: Little evidence of a trend in RRs with pCiL-1 in any smoking category. |
|
|
Subgroup analyses: RR patterns were the same within levels of an index of indoor air pollution or after adjustment. |
TABLE G-10 Distribution of cases and controls and adjusted odds ratiosa (OR) and confidence intervals (CI) by radon concentration in Shenyang, China, case-control study of females
measured home for 10 or more. On the average, subjects lived 66% of their adult lives in the measured home (Blot and others 1990). Using the maximum of the 2 radon measurements, the medians were 103.6 Bqm-3 (2.8 pCiL-1) for cases and 107.3 Bqm-3 (2.9 pCiL-1) for controls.
Categories of radon concentration ranged from < 74 Bqm-3 (2 pCiL-1) to 296 Bqm-3 (8.0 pCiL-1) (Table G-10). The RR for lung-cancer adjusted for age, education, smoking status, and an index of indoor air pollution showed no increase with increasing radon concentration.
In the Shenyang data, cigarette-smoking and indoor air pollution were found to be significant risk factors for lung-cancer for males and females (Xu and others 1989). Among females, 55% of cases and 35% of controls smoked cigarettes. The risk was over 9 times as high in women who smoked more than 1 pack/d for at least 40 yr, as in never-smokers. For females in the radon component of the study, the RR pattern with radon concentration was similar, that is, it showed no increase in never-smokers, light smokers and heavy smokers.
Indoor air pollution was found to increase lung-cancer risk by a factor of 2–3, depending on the variable analyzed. The greatest risks were associated with the use of a coal-burning kang (a brick bed under which heated smoke is passed through pipes before venting to the outside through a chimney or other opening) or cooking in the same room as the sleeping quarters.
An air-pollution index was developed to incorporate the type of heating for the home, the type of cooking fuel, and whether the kitchen and the bedroom were the same room. For females, no positive association was found with radon concentration for low or high categories of the indoor air-pollution index (Blot and others 1990).
Stockholm, Sweden Case-Control Study of Females
Study Subjects
The methods of the Swedish investigation have been described (Pershagen and others 1992) and are summarized in Table G-11. Women
suspected of having lung-cancer on admission in 1983–1985 to the 3 clinical departments of pulmonary medicine and the only department of thoracic surgery in Stockholm County were interviewed. Those later diagnosed as having lung-cancer (210) were classified as cases.
Two control groups were selected. Hospital controls consisted of women suspected of having lung-cancer who were later found not to have it (191), and population-based controls (209) were obtained from Stockholm County population registers. Results were reported for both control groups combined.
Data Collection
Subjects were interviewed by physicians using a structured questionnaire. For cases and hospital controls, information was obtained on admission. Population controls were interviewed in visits or by telephone. Information
TABLE G-11 Summary of results of Stockholm female case-control study
|
Factor |
Comment |
|
Principal reference |
Pershagen and others 1992. |
|
Design |
Case-control study of females. |
|
Study subjects |
Cases: Cases (210) included females admitted to the 3 pulmonary departments and the 1 thoracic-medicine department in Stockholm County in September 1983 to December 1985. |
|
|
Controls: Two controls were selected: "hospital" controls (191) included females suspected to have had lung-cancer but found not to, and population controls (209) selected randomly from County population registers. |
|
|
Subjects in radon study: For the radon analysis, 31 women (5%) could not be measured, leaving 201 cases and 378 controls with radon measurements. |
|
Lung-cancer histology |
201 cases: squamous, 26.9%; small cell, 25.4%; adenocarcinoma, 34.4%; other 13.4%. |
|
Rn measurement protocol |
Measurements: 1-yr a-track detectors in living room and in bedroom (85.1 %) or thermoluminescence detector (TLD) for 1 wk in living room followed by 1 wk in bedroom (14.9%) in all homes occupied 2 yr or more since 1945. TLD values were then adjusted empirically to link with a-track measurements. |
|
|
Missing: 2,118 homes fulfilled criteria for measurement; a-track detectors retrieved from 1,339 homes (63%) and TLD from 234 homes (11%). |
|
Rn measurements |
Median: Cases, 3.1 pCiL-1; controls, 2.9 pCiL-1 |
|
|
Mean: Cases, 3.6 pCiL-1; controls, 3.7 pCiL-1 (Lubin and others 1994). |
|
Rn-exposure estimation |
Exposure-time window: From 1945 to 5 yr before interview. |
|
|
Coverage: 26.3 yr and 25.3 yr of residence corresponding to 78% and 77% of the time window. |
|
|
Imputation for gaps: None for TWA radon concentration; unclear for cumulative exposure-some analyses set missing to zero, and some replaced missing with estimates based on housing characteristics. |
was obtained on smoking, exposure to environmental tobacco smoke, occupational history, and consumption of foods rich in vitamins A and C. Also obtained was a history of all residences in which the subject lived for 2 yr or more since birth or arrival in Sweden. The residential history included information on type of house, building material, and year of construction. Data from parish registries on past residences were used to verify and supplement the residential histories.
Methods of Radon Measurement
Measurements were sought for all dwellings where the subject resided for 2 years or more between 1945 and the end of the observation period, 1983–1985. For cases and hospital controls, the end of the exposure observation period was 5 years before the date of the study interview; for the population controls, it was 5 years before the interview of the corresponding case. Of the 2,118 residences so identified, no measurements could be made in 27.4%—in 11.2% because the house no longer existed, in 4.4% because the house was abroad, in 3.2% because the current owner refused, and in 8.6% for various other reasons (Pershagen and others 1992).
Year-long radon-concentration measurements were made in 1,339 dwellings with 2 a-track detectors: one in the living room and the other in the bedroom. In 234 dwellings (15%), measurements were made for 2 weeks during the winter with thermoluminescence detectors (TLDs) designed by the Swedish Institute of Radiation Protection. A TLD was placed in the living room for 1 week, then moved to the bedroom for another week. The 2 methods gave readings that had correlations above 0.8, although the TLD values were higher on the average, reflecting decreased ventilation in the colder months and the greater likelihood of their placement in areas of high-radon ground emanation (Svennson and others 1989). For analyses, TLD values were adjusted empirically to reflect a-track detector concentrations (Svensson and others 1988). The radon concentration assigned to a house was either the mean of the 2 a-track readings or the adjusted TLD reading.
Two measures of exposure were developed for analysis. The TWA radon concentration was the mean concentration for all houses measured weighted by the years of residency in the exposure window from 1945 to 5 years before enrollment. Cumulative radon exposure was computed as the product of residency time in the exposure window and measured radon concentration. The handling of missing measurements in the calculation of cumulative radon exposure was unclear. It appears that missing measurements were sometime set to zero and that "in certain analyses, missing radon measurements were replaced by estimates based on dwellings actually measured and information from the interview questionnaire on type of house, building material, and year of construction" (Pershagen and others 1992). Specific details were not provided.
Results
For cases and controls, the mean times covered by measurement data were 26.3 years and 25.3 years and represented 78% and 77% of the relevant period, respectively. For the subjects, median radon concentrations were 114.70 Bqm-3 (3.1 pCiL-1) for cases and 107.3 Bqm-3 (2.9 pCiL-1) for controls.
Results show a significant increase in RRs with increasing TWA radon concentration (Table G-12). The p value for trend was 0.05. As indicated by Pershagen and others and expanded on in the pooled analysis by Lubin and others (1994a), the significance of the test for trend depended on the cut points and on the quantitative value used. The p value of 0.05 computed by Pershagen and others (1992) used the median for each category as the quantitative trend variable, whereas Lubin and others computed the p value as 0.46 by using the continuous value for radon concentration. The former approach minimized the impact of extreme values; the latter approach eliminates the arbitrariness of categorization. The trend of increasing RR was reduced when adjusted for occupancy or when exposure 15 years and more before was given half the weight in line with results of miner studies. These differences highlight the need to interpret the Stockholm results with caution.
Because of the small number of cases, the trends in the RR with level of radon concentration were probably statistically homogeneous by histological type, although no formal assessment was done. However, the gradient of increase appeared greater for squamous cell and small cell carcinomas (Pershagen and others 1992).
Similarly, there was no statistical evaluation of the joint association of radon concentration and smoking status; however, the trend appeared slightly greater in never-smokers than in ever-smokers.
TABLE G-12 Distribution of cases and controls and adjusted odds ratiosa (OR) and confidence intervals (CI) by radon concentration for the Stockholm case-control study of females
|
|
Radon concentration (pCiL-1) |
|||||
|
|
< 2 |
2–2.9 |
3.0–4.0 |
= 4.1 |
Total |
P for trend |
|
Cases |
43 |
59 |
38 |
61 |
201 |
|
|
Controls |
89 |
113 |
76 |
100 |
378 |
|
|
OR |
1.0 |
1.2 |
1.3 |
1.7 |
|
0.05b |
|
95% CI |
|
(0.7,2.1) |
(0.7,2.3) |
(1.0,2.9) |
|
|
|
ORc |
1.0 |
1.5 |
1.6 |
1.5 |
|
0.19 |
|
95% CI |
|
(1.0,2.4) |
(0.9,2.7) |
(0.6,3,4) |
|
|
|
ORd |
1.0 |
1.4 |
1.2 |
1.3 |
|
0.65 |
|
95% CI |
|
(0.9,2.3) |
(0.7,2.1) |
(0.6,3.1) |
|
|
|
a ORs adjusted for age, smoking, and municipality of residence. b For test of linear trend using category means, P = 0.05; using continuous exposure, P 0.46. c Exposure adjusted for occupancy. d Exposure adjusted for BEIR IV weighting, exposures 5–15 yr before given full weight, exposures =15 yr before given 0.5 weight Source: Pershagen and others 1992. |
||||||
Swedish National Case-Control Study
Study Subjects
This study, the largest to date, relied on various national data files for the identification of subjects for the study (Pershagen and others 1994). The study is summarized in Table G-13. The study base was defined as all subjects 35–74 years old who had lived in any of 109 municipalities in Sweden at some time from January 1980 through December 1984 and who had been living in Sweden on January 1, 1947. The municipalities were selected to include areas suspected of having homes with high and low radon concentrations on the basis of measurement data or geologic and other information. Municipalities with mining activities and the large cities of Stockholm, Göteborg and Malmö were not included.
Using Swedish Cancer Registry files, cases included subjects diagnosed with primary lung-cancer in 1980-1984. All 650 women and a radon sample of 850 men (about 40% of all men with lung-cancer) were identified. After excluding those not in the study base, a total of 1,360 cases were enrolled 586 females and 774 males.
Two control groups were defined by using the population registers of Statistics Sweden. One control series consisted of a radon sample of women frequency-matched in 5-year age categories and calendar year of residence to the case group and included 1,424 subjects—730 women and 694 men. A second control group was selected by matching on age and calendar year and on vital status. Deceased controls were ascertained from the Swedish Cause of Death Registry, excluding subjects who had died of smoking-related diseases (cancer of the mouth, esophagus, liver, pancreas, larynx, or uterine cervix or bladder; ischemic heart disease; aortic aneurysm; cirrhosis of the liver; chronic bronchitis and emphysema; gastric ulcer; violent causes; or intoxication). In the second control group, there were a total of 1,423 subjects—650 women and 773 men.
At the time of selection on December 31, 1986, about 90% of the cases and of the second control group had died; about 9% of subjects in the first control group had died.
Data Collection
All subjects or their next of kin were mailed a standardized questionnaire. Information was collected on smoking habits of the subject, and their spouses and parents and on lifetime occupational and residential histories since 1947. Residential history included information on type of house, building material, heating system, and time spent at home. For incomplete questionnaires or nonrespondents, telephone interviews were attempted. Data from parish registries on past residences were used to supplement residential histories from questionnaires.
Methods of Radon Measurement
Measurements were sought for all dwellings where a subject resided for 2 years or more between 1947 and 3 years before the end of the observation period, defined as year of diagnosis for the case and calendar year of selection for the controls. Of a total of 13,392 residences,
TABLE G-13 Summary of results of Swedish national case-control study
|
Factor |
Comment |
|
Principal reference |
Pershagen and others 1994. |
|
Design |
Case-control study of females and males. |
|
Study subjects |
Cases: A total of 1,500 subjects 35–74 yr old with primary lung-cancer diagnosed in January 1980 to December 1984 were selected from the Swedish Cancer Registry, including all 650 females and 850 males. After various exclusions, 586 females and 774 males remained. |
|
|
Controls: Two controls were selected: 1 control group (730 females and 694 males) derived from a randomly selected sample from population registers, frequency matched on age to the cases; and 1 control group (650 females and 773 males) similarly selected and matched by vital status against the Swedish Cause of Death Registry. |
|
|
Subjects in radon study: For the radon analysis, measurements were not obtained on 27.4% of the homes. A total of 1,281 cases and 2,576 controls were included. |
|
Lung-cancer histology |
1,281 cases: squamous, 33.1%; small cell, 23.1%; adenocarcinoma, 26.9%; other or unknown 16.8%. |
|
Rn measurement protocol |
Measurements: 3-mo a-track detectors in living room and in bedroom in all homes occupied 2 or more years since 1947. |
|
|
Missing: 12,394 homes fulfilled criteria for measurement; a-track detectors were retrieved from 8,992 homes (73%). |
|
Rn measurements |
Median: 1.5 pCiL-1. |
|
Rn-exposure estimation |
Exposure-time window: From 1947 to 3 yr before end of follow-up diagnosis for cases or matched date of selection for controls. |
|
|
Coverage: 23.5 yr and 23.0 yr of residence corresponding to 72% and 71% of the exposure-time window. |
|
|
Imputation for gaps: None for TWA radon concentration; unclear for cumulative exposure—some analyses set missing to median concentration, and some replaced missing with estimates based on housing characteristics. |
|
Results |
Overall: For categories < 1.4, 1.4–2.1, 2.2–3.8, 3.8–10.8, and = 10.8 pCiL-1, RRs were 1.0, 1.2, 1.0, 1.3, and 1.8, with a P value for test of trend < 0.05. |
|
|
Histology: RR trends showed no difference by cell type. |
|
|
Smoking: No difference in RR trend greatest by smoking status, in contrast with the authors' view. |
|
|
Subgroup analyses: RR trend occurred only for subgroup that reportedly did not sleep near an open window; no trend was observed in those who sleep near an open window. |
addresses could not be identified for 7.5%. Of the remaining 12,394 residences, 27.4% could not be measured, usually because they no longer existed or because they were used only as summer houses (Pershagen and others 1994). In all, 73% of identified homes (8,992) were measured.
Three-month radon measurements were made during the heating season—from October 1 to April 30—with a-track detectors, which were processed by the Swedish Radiation Protection Institute. Two detectors were used: one in the living room and the other in the bedroom. For analyses, the mean of the 2 values was assigned to the residence. The authors estimate that the winter measurements might be 10–20% higher than yearly values, although the basis for this estimate is not provided.
Cumulative radon exposure since 1947 was estimated by multiplying the measured radon concentration concentration and the length of residency in each home. For each subject, TWA radon concentration was calculated by dividing cumulative radon exposure by the total residential time covered by radon measurements. Missing measurement time was not included; in effect the concentration and duration during those times were zero. For some analyses, imputation of missing measurement data was accomplished by replacing the missing data with the median radon concentration for all subjects or values adjusted to reflect residential characteristics (Pershagen and others 1994).
Results.
Results were presented for males and females combined. There were a totals of 1,281 cases and 2,576 controls.
Radon measurements covered 23.5 years and 23.0 years of the exposure-time for cases and controls, representing 72% and 71 % of the intended period, respectively. For individuals, the median TWA radon concentration was 55.5 Bqm-3 (1.5 pCiL-1).
Results show a significant increase in RR with increasing radon concentration (Table G-14). The RR patterns appeared similar by cell type. For the 5 categories shown in Table G-14 RRs were: 1.0, 1.2, 1.3, 1.5, and 1.7 for squa
TABLE G-14 Distribution of cases and controls (males and females combined) and adjusted odds ratiosa (OR) and confidence intervals (CI) by radon concentration for the Swedish national case-control study
mous cell carcinomas; 1.0, 0.9 1.1, 1.2, and 2.8 for small cell carcinomas; and 1.0, 1.1, 1.0, 1.4, and 2.3 for adenocarcinomas. Differences in RRs for the highest category could have arisen by chance, in as much as the category included only 11, 15, and 12 squamous, small cell, and adenocarcinoma cases, respectively.
RR patterns were also similar by smoking status. For categories of radon concentration shown in Table G-14, RRs were: 1.0, 1.1, 1.0, 1.5, and 1.2 for never-smokers; 1.0, 0.9, 1.2, 1.7, and 0.4 for ex-smokers; 1.0, 1.0, 1.0, 1.2, and 4.0 for current smokers consuming fewer than 10 cigarettes/d; and 1.0 0.9, 0.9, 1.2, and 2.6 for current smokers consuming at least 10 cigarettes/d. RRs in the highest radon concentration category were based on 5, 1, 12, and 16 lung-cancer cases for never-smokers, ex-smokers, and current smokers of fewer than 10 and at least 10 cigarettes/d, respectively.
Pershagen and others found that the RR trend with radon concentration increased for those who reportedly sleep with their bedroom windows closed, but the trend disappeared for subjects who reported sleeping next to an open window. Those patterns of risk are difficult to interpret. Sleeping next to an open window is not itself a risk factor for lung-cancer. Furthermore, the radon concentration of a bedroom with an open window will be reflected in a reduced radon measurement. Subjects or next of kin were interviewed about sleeping practices many years after disease occurrence. The relationship between the radon measurement, the current practice of sleeping with an open window, and whether the case or control subject slept with an open window at the time of enrollment is uncertain, particularly in homes that no longer were occupied by the subjects or their spouses. Effects of errors in exposure estimation might also play a role in the observed RR patterns. Control data suggest that in this age group about 70% of the population sleep with closed windows. Among subjects who sleep with an open window, measurements in homes, particularly former homes, are more likely to occur with owners who sleep with closed windows, thereby adding to error in exposure estimation and obscuring exposure-response effects. Among subjects who sleep with windows closed, measurements in former homes are more likely to occur with closed windows; however, for owners who sleep with open windows, there is a systematic (nondifferential) under estimation of exposure, a condition that can induce an increase in the trend of the exposure-response relation (Dosemeci and others 1990). No data on sleeping next to an open window were obtained at the time of the radon measurement. Further conclusions regarding sleeping with an open window are problematic, in that other studies have not considered the issue.
Winnipeg Case-Control Study
Study Subjects
This study, summarized in Table G-15, was a case-control study of lung-cancer in males and females in Winnipeg, Canada. In Létourneau
TABLE G-15 Summary of results of Winnipeg, Canada, case-control study
|
Factor |
Comment |
|
Principal reference |
Létourneau and others 1994. |
|
Design |
Case-control study of males and females. |
|
Study subjects |
Cases: Cases included all residents of Winnipeg, Canada 35–80 yr old with histologically confirmed, primary lung-cancer diagnosed in September 1983 to September 1990 and listed with the provincial cancer-incidence registry. |
|
|
Controls: Controls were randomly selected from the Winnipeg telephone directory and individually matched on age within 5 yr and sex. |
|
|
Subjects in radon study: A total of 759 pairs were assembled. After exclusion for misdiagnosis or improper control selection, 738 case-control pairs were enrolled. 257 cases and 78 controls had proxy interviews. |
|
Lung-cancer histology |
738 cases: squamous, 31.4%; small cell, 15.9%; adenocarcinoma, 32.9%; other 19.8%. |
|
Rn measurement protocol |
Measurements: Two sequential 6-mo a-track detectors in the bedroom and two in the basement of up to 3 homes in the Winnipeg metropolitan area. For apartments, only bedroom measurements were made. Yearly values were taken as the mean of the 2 measurements. |
|
|
Missing: Subjects had a mean of 5 homes in the Winnipeg area; attempts were made to measure 3 homes. 7,318 homes were eligible, and 4,448 were measured (61%). |
|
Rn measurements |
Mean: For bedrooms: cases, 3.1 pCiL-1; controls, 3.4 pCiL-1. For basements: cases, 5.1 pCiL-1; controls, 5.6 pCiL-1. |
|
Rn-exposure estimation |
Exposure-time window: Two windows defined: 5–30 yr and 5–15 yr before date of case diagnosis or control selection. |
|
|
Coverage: About 67% of 5 to 30 yr window and 80% of 5 to 15 yr window. |
|
|
Imputation for gaps: For cumulative exposure, used mean concentration for Winnipeg (3.3 in the living area and 5.3 in the basement). |
|
Results |
Overall: For categories (estimated from cumulative exposure) <1.9, 1.9–3.9, 3.9–7.8, and = 7.8 pCiL-1, RRs were 1.0, 1.0, 0.8, and 1.0 for the 5 to 30 yr window and 1.0, 1.0, 0.8, and 1.0 for the 5 to 15 yr window. |
|
|
Histology: RRs similar and show no increased risk with exposure by cell type. |
|
|
Smoking: Smoking patterns were used only for adjustment; no evaluation of effect modification of radon RRs was conducted. |
|
|
Subgroup analyses: Data on occupational exposures were used only for adjustment; no evaluation of effect modification of radon RR was conducted. |
and others (1994), cases were histologically confirmed primary lung-cancer cases diagnosed between the ages of 35 and 80 years and, listed in the provincial cancer-incidence registry maintained by the Manitoba Cancer Treatment and Research Foundation for 1983–1990. All patients were residing in Winnipeg at the time of diagnosis. Controls were individually matched to cases on age within 5 years and on sex and were identified through the Winnipeg telephone directory.
A total of 759 matched pairs were initially identified. When cases that did not have primary lung-cancer or had improperly matched controls were excluded, a total of 738 pairs remained for analysis.
Data Collection
Information from in-person interviews was collected on demographic characteristics, education, and smoking practices and on detailed residential history. The questionnaire also incorporated a detailed occupational history, including information on specific job exposures.
Methods of Radon Measurement
There was a mean of 9 homes per subject, of which 5 were in the Winnipeg metropolitan area. It was not clear whether these means reflect lifetime residency or residencies in an exposure-time window. Radon was measured in 3 of these homes, although it was not clear precisely what criteria were used to select homes. The authors identified a total of 7,745 homes to be measured. This number was reduced for homes that had been occupied for less than 1 year (6%), for refusals (11%), for homes that no longer existed, were commercial institutions, or could not be located (24%), or where the dosimeter was lost or damaged (2%). Radon measurements were obtained for 4,448 homes (57%).
Year-long monitoring of the bedroom and, if there was one, the basement was undertaken. The basement was selected to provide the maximal possible residential exposure. Year-long monitoring was achieved through the sequential placement of two 6 mo detectors.
The detectors were developed and calibrated in house by laboratories of the Bureau of Radiation and Medical Devices in the Department of National Health and Welfare. Although the in-house calibration might affect comparisons with other studies in overall mean radon concentration, it should have no effect on the evaluation of trends in the exposure-response relation.
Two exposure windows were defined: 5–30 yr and 5–15 yr before the date of enrollment in the study. For estimation of cumulative radon exposure, imputation of missing measurements used the mean concentration in living areas [122.10 Bqm-3 (3.3 pCiL-1)] or in basements [196.10 Bqm-3 (5.3 pCiL-1)].
Results
Available radon measurements covered about 67% and 80% of the exposure windows of 5–30 yr and 5–15 yr, respectively. For cases and controls mean radon concentrations were 114.7 Bqm-3 (3.1 pCiL-1) and 125.8 Bqm-3 (3.4 pCiL-1) for bedrooms and 188.7 Bqm-3 (5.1 pCiL-1) and 207.2 Bqm-3 (5.6 pCiL-1) for basements, respectively.
Preliminary analysis revealed that cases were significantly less educated than controls and somewhat less likely to be born in Canada, although almost
TABLE G-16 Distribution of cases and controls and adjusted odds ratiosa (OR) and confidence intervals (CI) by radon concentration in Winnipeg, Canada, case-control study (concentration level estimated from cumulative exposure)
80% of the subjects were born in Canada. All analyses were adjusted for education and smoking status; age and sex were adjusted through the study matching. Table G-16 shows that there was no trend in the RRs with increasing TWA radon concentration in the bedrooms. Similar results hold for basement measurements. The results were similar when cases were restricted by histologic type.
Missouri Case-Control Study of Nonsmoking Females
Study Subjects
The Missouri study was a population-based case-control study of white non-smoking woman, defined as lifelong never-smokers or former smokers who ceased 15 yr or more before interview (Alavanja and others 1994). The study is summarized in Table G-17. Among former smokers, the median time since smoking cessation was 24 yr. Cases were women 30–84 yr old with primary lung-cancer who were reported to the Missouri Cancer Registry from June 1, 1986, to June 1, 1991. After exclusion of ineligible cases, interviews were completed on 618 cases. Radon measurements were obtained for 538 cases (83%); measurements were not obtained for 80 cases because of refusal, homes that were out of state or destroyed, or other reasons. Although all cases were confirmed when diagnosis was reported to the registry, a separate panel of experts was established to review available slides; 409 of the 538 cases (76%) were reviewed.
A population-based control sample of white nonsmoking women was randomly selected by using Missouri state driver's license files (age 30–64 yr) or files of the Health Care Financing Administration (age 65–84 yr). The controls were selected to match the age distribution of cases in 5-yr categories. Of the 1,527 controls who satisfied enrollment criteria, 1,402 (92%) agreed to an interview and 1,183 (77%) had at least one valid year-long a-x97;-track measurement.
Data Collection
An initial telephone questionnaire was used to screen eligible subjects. If a subject agreed to participate, a telephone-interview survey
TABLE G-17 Summary of results of Missouri case-control study of female never-smokers
|
Factor |
Comment |
|
Principal reference |
Alavanja and others 1994. |
|
Design |
Case-control study of female never-smokers and long-term former smokers. |
|
Study subjects |
Cases: 618 women 30–84 yr old with primary lung-cancer listed with the Missouri Cancer Registry an June 1, 1986, to June 1, 1991, who never smoked or were long-term former smokers. |
|
|
Controls: Population-based controls (1,527) selected from state drivers-license files or files of the Health Care Finance Administration, frequency matched by age. |
|
|
Subjects in radon study: After refusals and other exclusions, 538 cases (87%) and 1,183 controls (78%) had at least 1 home in the 5–30 yr before enrollment measured for radon. |
|
Lung-cancer histology |
A histologic review, separate from registry notification, was conducted. From 409 cases, there were 262 adenocarcinomas (53.5%); other cell types were not reported. |
|
Rn measurement protocol |
Measurements: 1-yr a-x97;-track detectors in kitchen and in bedroom in all homes in Missouri occupied 1 yr or more from 5–30 yr before date of enrollment. |
|
|
Missing: Radon measurements available for 74% of identified dwellings. |
|
Rn measurements |
Mean and median: Case and control values were the same, 1.8 pCiL-1 (mean) and 1.4 pCiL-1 (median). About 7% had homes above 4 pCiL-1. |
|
Rn-exposure estimation |
Exposure-time window: 5–30 yr before to case incidence or control interview. |
|
|
Coverage: Mean 20 yr of residence corresponding to 78% of the exposure-time window. |
|
|
Imputation for gaps: None for TWA radon concentration; missing values for cases and controls replaced with means all cases and controls, respectively. |
|
Results |
Overall: For quintile categories < 0.8, 0.8–1.2, 1.2–1.7, 1.7–2.5, and = 2.5 pCiL-1, RRs were 1.0, 1.0, 0.8, 0.9, and 1.2, P value for trend, 0.99 with continuous value for test and 0.19 with category means. |
|
|
Histology: RR showed a suggestive trend with adenocarcinoma cell type; P value for trend was 0.31 with continuous and 0.04 with categoric radon values. |
|
|
Smoking: No difference in RR trend with radon concentration for never-smokers or former smokers. |
|
|
Subgroup analyses: Suggestive RR trend (P = 0.06) for data restricted to in-person interview. Since measured quantity, reason for differences uncertain. |
obtained information on demographic factors, occupational history, lifetime passive smoking, previous active smoking, diet, and previous diseases, and a detailed residential history.
Methods of Radon Measurement
For each subject, radon was measured in all homes in the state of Missouri occupied for at least 1 yr during the 30 yr before enrollment. One year-long a-track detector was placed in the bedroom and one in the kitchen. Every 3 mo, subject's homes were checked to see whether the dosimeters were still in place. Quality control procedures—blind inclusion of blank and spiked dosimeters and duplicate detectors—suggested excellent validity in the measurement protocol. A small subsample of 3-mo winter measurements had a mean value twice the year-long readings.
The time-weighted radon concentration was computed for each subject by using all available measurement data; gaps in the exposure window were ignored. Cumulative radon exposure was estimated for an exposure window of 5–30 yr before lung-cancer incidence for cases and before interview for controls. Missing values for cases or controls were set to the mean radon concentration data for cases or controls, respectively.
Questionnaire data revealed that the subject occupancy factor was 84%. No special adjustment for occupancy was carried out.
Results
An average of about 20 yr of occupancy in the exposure period of 5–30 yr was covered by measurement data, 78% of the residency time. Mean and median radon concentrations were the same for cases and for controls: 66.6 Bqm-3 (1.8 pCiL-1) and 51.8 Bqm-3 (1.4 pCiL-1), respectively.
RR results were presented by categories defined by quintiles; the mean for the highest radon-concentration category was 151.7 Bqm-3 (4.1 pCiL-1). Table G-18 shows no increase in age-adjusted RR with increasing radon concentration. The RRs were adjusted only for age, but the pattern was unaffected by further adjustment of RRs for previous smoking, pack-years of smoking, previous lung disease, education, or intake of saturated fat.
TABLE G-18 Distribution of cases and controls (males and females combined) and adjusted odds ratiosa (OR) and confidence intervals (CI) by quintiles of radon concentration in Missouri case-control study of female never-smokers
There was no RR trend with exposure within age categories or smoking status. Limiting cases to the 262 women with an adenocarcinoma cell type resulted in a suggestive RR trend. RRs for the 5 radon-concentration categories of Table G-18 were 1.0, 1.4, 1.1, 1.2, and 1.7; the p value for the test of linear trend was 0.04 when mean values for each category were used and 0.31 when continuous radon concentration.
Finnish Case-Control Study (Finland I)
Study Subjects
A population-based case-control study (denoted Finland I) of lung-cancer in men was conducted in southern Finland in 19 municipalities. As of 1980, about 65,000 males lived in these areas (Ruosteenoja 1991). The study is summarized in Table G-19. Cases consisted of lung-cancers diagnosed in men in the designated municipalities in 1980–1985. In the period 1980–1982, cases were identified from the Finnish Cancer Registry; the more-recent cases were accrued directly from the records of hospital that diagnose and treat lung-cancer. A total of 291 cases were available for study.
Controls were a random sample of all men in the Finnish Population Registry files who were living in the designed area on January 1, 1980. Controls were frequency-matched to the age profile of the cases. Controls were then sent a mail questionnaire to obtain information on tobacco use. From the returned questionnaires (91%), controls were further selected to match the smoking proportions of the cases; 10% never-smokers, 10% ex-smokers who quit before 1979, and 80% current smokers or recent ex-smokers. A total of 495 controls were enrolled into the study: 50 never-smokers, 50 ex-smokers, and 395 current smokers.
Data Collection
In-person interviews were conducted for cases and controls or, if they were deceased, with their next of kin. Information was collected on residential history, house type, smoking, education, and occupation.
Methods of Radon Measurements
The Finnish Centre for Radiation and Nuclear Safety conducted measurements of indoor radon concentrations in all dwellings occupied for 1 yr or more since 1950; a-track detectors were placed for 2 mo in the winter between November 1, 1986 and April 30, 1987.
For analysis, an exposure window was defined as the 25-yr period from 1950 to 1975. For homes that could not be measured, a regression equation developed by Mäkeläinen and others (1987) that accounted for housing type and other factors was applied to estimate radon concentration. For dwellings higher than the ground floor, radon concentration was assigned the value 51.8 Bqm-3 (1.4 pCiL-1). Two radon measures were calculated: the TWA radon concentration based on available measurements and a TWA radon concentration for the entire 25-yr period based on measured and estimated concentrations.
Results
A total of 238 cases and 434 controls were available for analysis after exclusion for nonresponse to interview or inability to locate the subject. At the time of interview, 88% of cases and 21% of controls were deceased.
TABLE G-19 Summary of results of the Finnish case-control study
|
Factor |
Comment (1991) |
|
Principal references |
Ruosteenoja 1991, Ruosteenoja and others 1996. |
|
Design |
Case-control study of males. |
|
Study subjects |
Cases: 238 males with primary lung-cancer diagnosed in 19 municipalities in Finland 1980–1985. For 1980–82 cases obtained from the Finnish Cancer Registry; from 1983–1985 cases from records of treatment hospitals. |
|
|
Controls: Population-based sample of mean living in 19 municipalities on January 1, 1980, frequency matched by age category. With information on smoking from a mail questionnaire, a random sample of 10 never-smokers, 10 ex-smokers and 395 current smokers was selected to serve as controls. |
|
Lung-cancer histology |
238 cases: 91 (38.2%) squamous cell, 61 (25.6%) small cell, 18 (7.6%) adenocarcinomas, and 68 (28.6%) other or unknown. |
|
Rn measurement protocol |
Measurements: 2-mo a-track detectors in the living room or bedroom in all homes occupied 1 yr or more 1950–1975. |
|
|
Missing: Radon measurements available for 50% of identified dwellings; and at least 1 measurement available for 76% of subjects. |
|
Rn measurements |
Mean or median: Not provided, but quintile cut-points indicate that 40% and 20% have concentrations above 4.7 and 7.4 pCiL-3, respectively. |
|
Rn-exposure estimation |
Exposure-time window: 25 yr between 195 to 75 or 5-10 y prior to case incidence or control interview. |
|
|
Coverage: Mean 20 yr of residence corresponding to 78% of the exposure-time window. |
|
|
Imputation for gaps: For TWA radon concentration, missing values were estimated on basis of regression of housing type, municipality and other factors. |
|
Results |
Overall: For quintile categories < 2.2, 2.2–3.4, 3.4–4.7, 4.7–7.4, and = 7.4 pCiL-1, RRs were 1.0, 1.1, 1.7, 1.9, and 1.1; and the P value for trend was not significant. |
|
|
Smoking: Little effect of adjustment in pattern of RRs with radon concentration. |
Subjects resided in a total of 1,393 homes during the 1950–1975 period and indoor radon measurements were conducted in 696 homes (50%). Radon measurements in at least one house were obtained for 171 cases (72%) and 342 controls (79%); in this subgroup, the mean residency time covered by measurement data was about 20 yr or about 80% of the exposure window.
It was not entirely clear, but there seemed to be little difference in results between TWA radon-concentration measures. The distribution of cases and controls by category concentration was not provided by the author; however,
RRs, adjusted for age and smoking, for 5 categories based on quintiles were 1.0, 1.1, 1.7, 1.9, and 1.1 (Ruosteenoja 1991).
Finnish Case-Control Study (Finland II)
Study Subjects
Subjects for this case-control study, denoted Finland II, were selected from a subset of records of the Finnish Population Registry on persons living in the same single-family house (called the index dwelling) from January 1, 1967, or earlier to the end of 1985 (Auvinen and others 1996). Cases eligible for the study consisted of all persons with lung-cancer diagnosed from January 1, 1986, to March 31, 1992, that were listed with the Finnish Cancer Registry. A total of 1,973 cases were identified. One control for each case was selected, matched by birth year and sex. The control had to be alive at the time of the diagnosis of the case. The investigators selected additional controls when possible, and a total of 2,885 controls were identified. There were 1,644 (83%) deceased cases and 326 (11%) deceased controls. The study is summarized in Table G-20.
Data Collection
In September 1992, a mail questionnaire was sent to subjects or their next of kin. Information was obtained on residential history, smoking habits and occupational exposures. Information was also obtained on the daily number of hours spent indoors in the 1960s and 1970s. Response rates for the mail questionnaire were 55% for cases and 54% for controls.
Methods of Radon Measurement
One-year measurements with track-etch devices were undertaken for all subjects for their index dwelling. In the winter of 1992–1993, residents were mailed a detector and instructed to place it in a bedroom or living room. The detectors were returned the next winter. Houses were excluded for a number of reasons, the most common being the building of new houses on the same locations as index dwellings, uncertain dates of construction, measurements of less than 150 days, uninhabitation, and extensive renovation. About 20% of houses were excluded for those reasons.
The authors estimated the relative precision of the 1-yr measurements as ±20% for concentrations below 50 Bqm-3 and ±15% for concentrations above 400 Bqm-3, with a systematic bias of less than 10%.
Results
After exclusions for incomplete questionnaires and missing radon measurements, data were available on 1,055 cases and 1,544 controls. The study had originally been designed as an individually matched study, so results were presented only for analyses restricted to 517 case-control pairs. However, the authors state that unmatched results based on all available data were similar to the results from the matched analysis.
The mean radon concentrations were 103 Bqm-3 for cases and 96 Bqm-3 for controls; the median was 67 for both groups. The median occupancy times were similar, 11.5 h/d for cases and 11.7 h/d for controls.
Median residence times in the index house were 37 yr for cases and 35 yr for
TABLE G-20 Summary of results of Finland-II case-control study
|
Factor |
Comment (1991) |
|
Principal reference |
Auvinen and others 1996. |
|
Design |
Case-control study of males and females. |
|
Study-subjects |
Subjects were selected from the Finnish Population Registry of persons living in the same single-family house from January 1, 1967, or earlier until the end of 1985. |
|
|
Cases: All lung-cancers diagnosed from January 1, 1986, to March 31, 1992, listed with the Finnish Cancer Registry; 1,973 cases were identified. |
|
|
Controls: For each case, subject was matched by birth year and sex and alive at the time of the diagnosis of the case. Additional controls were selected when possible, and a total of 2,885 controls were identified. For the matched analysis, 517 pairs were available. |
|
Lung-cancer histology |
Histologic or cytologic confirmation was available on 92% of cases. In the final series, the distribution was 36% squamous, 14% small cell, 13% adenocarcinoma, 9% other, and 28% undefined. Sex distribution was not provided. |
|
Rn measurement protocol |
Measurements: One a-track detector was mailed to each subject with instructions for it to be placed in the bedroom or the living room. |
|
|
Missing: About 20% of houses (subjects) were excluded due because of missing information or other problems with the index dwelling. |
|
Rn measurements |
Mean: Cases, 103 Bqm-3; and controls, 96 Bqm-3. |
|
|
Median: 67 Bqm-3 for cases and controls. |
|
Rn-exposure estimation |
Exposure-time window: Defined by study design; medians for years in the index house, 38 yr for cases and 35 yr for controls; median occupancy times, 11.5 hr/d for cases and 11.7 hr/d for controls. |
|
Results |
Overall: For categories < 50, 50–99, 100–199, 200–399, and = 400 Bqm-3, adjusted RRs (and 95% CIs) were 1.0, 1.03 (0.8–1.3), 1.00 (0.8–1.3), 0.91 (0.6–1.4), and 1.14 (0.7–1.9). RRs similar when radon levels weighted by occupancy. |
|
|
Histology: RR similar by cell type. |
|
|
Smoking: RR patterns were similar within smoking categories. |
controls, and 70% of cases and 76% of controls lived more than 30 yr in the index house. Although the design indicated a minimum of 19 yr of residency in the index house, registry information appears not to reflect actual residency. A total of 26 cases (5%) and 36 controls (7%) had less than 16 yr residency in the index house.
For indoor-radon categories of less than 50, 50–99, 100–199, 200–399, and = 400 Bqm-3, RRs (and 95% confidence intervals) adjusted for age, sex, and smoking were 1.0, 1.03 (0.8–1.3), 1.00 (0.8–1.3), 0.91 (0.6–1.4), and 1.14 (0.7–
1.9). RRs were similar when radon concentrations were weighted by occupancy or when adjusted for occupational asbestos exposure.
RRs were also similar when data were analyzed by histologic type of lung-cancer.
Israeli Case-Control Study
Study Subjects
This was a small hospital-based case-control study at the Rambam Medical Center in Israel, summarized in Table G-21. Subjects were consecutive patients with primary lung-cancer seen in the oncology ward in 1985-
TABLE G-21 Summary of results of Israeli case-control study
|
Factor |
Comment (1991) |
|
Principal reference |
Biberman and others 1993. |
|
Design |
Hospital-based case-control study. |
|
Study subjects |
Cases: Two case groups defined on the basis of consecutive patients with primary lung-cancer at an oncology ward of the Rambam Medical Center in 1985–1989:35 cases with small-cell carcinoma (SCC) (including ever-smokers and never-smokers) and 26 cases with non-small-cell carcinoma (16 adenocarcinomas) who were never-smokers (NS). Cases must have live in Israel for at least 10 yr before diagnosis. |
|
|
Controls: Patients without lung-cancer matched by sex and 5-yr age group who were admitted to the same hospital immediately after case admission and lived in Israel 10 yr or more. |
|
|
Subjects in radon study: After exclusions and refusals, 52 cases and 43 controls were eligible; however, only 35 matched pairs (20 SCC pairs and 15 NS pairs) were available for analysis. |
|
Rn measurement protocol |
Measurements: a-track detectors placed for an average of 9 mo from June or July 1990 through April 1991. |
|
Rn measurements |
Median: For cases and controls, 1.09 and 0.9 pCiL-1 for SCC pairs and 0.9 and 1.07 pCiL-1 for NS pairs, respectively. Differences were not statistically significant. Overall mean concentration was 1.0 pCiL-1. |
|
Rn-exposure estimation |
Exposure-time window: None defined; measurement in current house only. |
|
|
Coverage: 28 (80%) cases and 19 (54%) controls lived 20 yr or more in measured house; 15 (43%) cases and 13 (37%) controls lived 30 yr or more in measured house. |
|
Results |
Overall: No significant differences in median radon concentrations between cases and controls. RR for = 1 pCiL-1 compared with <1 pCiL-1 was 1.5 with 90% CI (0.4,5.4) for SCC pairs and 0.5 with 90% CI (0.1,2.2) for NS pairs. |
1989 (Biberman and others 1993). Two case groups were defined: 35 patients with small cell carcinoma (denoted the SCC group), including ever-smokers and never-smokers, and 26 patients with non-small cell carcinoma who were never-smokers (denoted the NS group and including 16 patients with adenocarcinoma). All subjects had to have lived in Israel 10 yr or more before diagnosis.
Controls were matched by sex and age group within 5 yr from admissions to the same hospital immediately after case admission. Controls were also limited to those who had lived 10 yr or more in Israel before admission.
A total of 52 cases and 43 controls were eligible; however, after refusals or an inability to obtain radon measurements, a total of 35 pairs were available for analysis (20 SCC pairs and 15 NS pairs).
Data Collection
Personal interviews with subjects or next of kin yielded information on residential, occupational, and smoking histories.
Methods of Radon Measurement
One a-track detector was placed in the bedroom of each subject in June-July 1990 and collected starting in April 1991. The detectors remained in place for a mean of 9 mo (Biberman and others 1993). Of the 70 dwellings, 33 (47%) were single-story or ground-floor units of multi-unit apartments. The geometric mean was 37 Bqm-3 (1.0 pCiL-1), and the range was 7.4-262.7 Bqm-3 (0.2-7.1 pCiL-1). Seven measurements were at or above 74 Bqm-3 (2 pCiL-1).
Results
In the SCC group, 17 subjects were males and 3 females; 19 subjects and 14 controls were ever-smokers. In the NS group, 4 subjects were males and 11 females; no subjects were smokers, and 2 controls were ever-smokers.
There was no significant difference in radon concentration between cases and controls for the SCC or NS groups. For cases and controls, median concentrations were 40.33 and 33.3 Bqm-3 (1.09 and 0.9 pCiL-1) for the SCC pairs and 32.93 to 39.59 Bqm-3 (0.89 and 1.07 pCiL-1) for the NS pairs. After adjustment for pack-years of cigarette use, the RR for 37 Bqm-3 (1.0 pCiL-1) compared with 37 Bqm-3 (1.0 pCiL-1) was 1.5 with a 90% CI of 0.4-5.4 for the SCC pairs and 0.5 with a 90% CI of 0.1–2.2 for the NS pairs. RRs were significantly increased for long-term residency on a ground floor for both case groups, but the interpretation of this result is clouded by other possible case-control differences for which no adjustment could be made.
Port Hope Case-Control Study
One of the earliest case-control studies to estimate indoor radon from direct measurements is summarized in Table G-22. This study was conducted in Port Hope, a town of about 10,000 residents on the north shore of Lake Ontario. In 1932, mining operations included processing of ore and the recovery of radium; after 1939, operations shifted to the production of uranium. Disposal of residue from the operations occurred on the plant site and in other designated areas. In 1953, modification of the operations resulted in use of demolition rubble and
TABLE G-22 Summary of results for the Port Hope case-control study
|
Factor |
Comment (1991) |
|
Principal reference |
Lees and others 1987. |
|
Design |
Case-control study of males and females living in Port Hope, Ontario. |
|
Study subjects |
Cases: 27 lung-cancer cases diagnosed in 1969–1979, in persons who lived 7 yr or more in Port Hope, and were never employed at the uranium-refining plant. |
|
|
Controls: 49 subjects matched on sex and date of birth, who lived 7 yr or more in Port Hope, with at least 1 of these years during the 7-yr period before the date of diagnosis of the matched case. One dead and 1 live control were matched to each deceased case, and 2 live controls were matched to each live case. |
|
Lung-cancer histology |
Cell types included 11 squamous cell carcinomas, 6 adenocarcinoma, and 11 unknown. |
|
Rn progeny protocol |
Measurements: Precise protocol was not provided, but apparently measurements were of WL. |
|
WL measurements |
Mean or median: Not provided. |
|
Rn progeny exposure estimation |
Exposure-time window: Estimates of exposure based on all estimation homes occupied in Port Hope since 1933. Residences outside Port Hope area were ignored. Exposure estimates in WLM and adjusted by a background exposure of 0.229 WLM/yr. On basis of WLM distributions, it was estimated that for cases and controls mean WLMs were 2.7 and 0.5, including 33% and 49% with ''zero" WLM exposure (below estimated background exposure), respectively; among exposed, means were 4.1 and 1.0 WLM. |
|
Results |
Overall RR of 1.55 with 95% CI (0.6,4.1) and with adjustment for smoking RR of 2.36 with 95% CI (0.8,7.1). |
reclaimed building materials throughout the town for various construction purposes (Lees and others 1987).
Study Subjects
Subjects were defined as persons who died of lung-cancer in 1969–1979 and who lived in Port Hope for 7 yr or more before the year of diagnosis. Cases were identified through the Provincial Cancer Registry and by contacting local physicians.
For each case, 2 controls, matched on sex and year of birth, were selected from among persons who had lived in Port Hope for 7 yr or more, with at least 1 yr during the 7-yr period before the date of diagnosis of the matched case.
Persons were excluded if they had worked in the uranium-refining plant or if they did not meet residency requirements. After exclusions, 27 lung-cancer cases and 57 matched controls were studied.
Data Collection
Data were collected by personal interviews with subjects or next of kin, including information on residential, occupational, and smoking histories.
Methods of Radon Measurement
The protocol used to measure radon or radon progeny in homes was not given in Lees and others (1987). However, the authors indicate that in 1976 a "complete survey of the town was undertaken to delineate radiation contaminated areas and measure radon levels." It was not stated what was measured, but for the analysis exposure of each subject was estimated in cumulative Jhm-3 (WLM), on the basis of all houses occupied in Port Hope from 1933. Estimated values were adjusted to exclude background exposures, on the basis of an estimate 0.0008015 Jhm-3/yr (0.229 WLM/yr) and the assumption that a "worker" and a "nonworker" spend 60% and 85% of the year, respectively, inside the house.
Results
After adjustment of exposures, 67% of cases and 51% of controls had "nonzero" exposures. Among the exposed, mean exposures were 0.01435 and 0.0035 Jhm-3 (4.1 and 1.0 WLM) for cases and controls, respectively. The matched RR estimate for exposure was 1.55 (95% CI, 0.6–4.1) without adjustment for smoking status and 2.36 (95% CI, 0.8–7.1) with adjustment for smoking status.
Case-Control Studies of Indoor Radon in Progress
Extrapolations from miner studies suggest that lung-cancer risk posed by indoor radon exposure may be a potentially important public-health problem. There have been substantial interest in obtaining direct evidence of harmful effects of indoor radon to validate miner-based extrapolations and to identify an upper bound of the risk. Few studies have been published, and difficult design issues remain in the conduct of the studies (Lubin and others 1990a, 1995c; Stidley and Samet 1993). To expand the base of information on the consequences of indoor radon exposure, several case-control studies are under way. Table G-23 lists these studies, which total some 13,000 lung-cancer cases.
SUMMARY OF STUDIES OF LUNG CANCER AND INDOOR RADON
In this section, we provide an overall perspective from the various studies of residential radon, including results of a meta-analysis of current indoor-radon studies.
Ecologic Studies
Ecologic studies are limited by the inability to estimate relevant exposures, by the presence of an extremely strong risk factor for lung-cancer (cigarette-smoking), and by the intrinsic confounding arising from regression analyses that use summary data and model misspecification We conclude that ecologic studies are noninformative for estimating risks posed by exposure to indoor radon or for evaluating a potential threshold exposure below which radon-progeny exposure would not be harmful.
TABLE G-23 Summary of continuing studies of residential radon and lung-cancer risk
|
Country |
Location |
Cases |
Controls |
Estimated completion date |
Comments |
|
European studies: |
|||||
|
Belgium-France |
Ardennes-Eiffel |
1,200 |
— |
1996 |
— |
|
France |
Brittany |
600 |
1,200 |
1996 |
— |
|
Germany |
Western |
2,500 |
|
1996 |
— |
|
|
Eastern |
1,500 |
|
1997 |
— |
|
|
Tyrol |
250 |
|
1997 |
— |
|
Sweden |
|
480 |
|
1998 |
— |
|
United Kingdom |
Cornwall and Devon |
986 |
|
1997 |
— |
|
North American studies: |
|||||
|
Canada |
— |
800 |
|
1998 |
Includes only never-smoking subjects |
|
United States |
Connecticut |
960 |
|
1995 |
Jointly conducted with Utah |
|
|
Iowa |
450 |
|
1998 |
Includes subjects with at least 20 yr in current house |
|
|
Missouri |
700 |
|
1996 |
Extension of previous study, but includes ever-smoking and never-smoking women |
|
|
New Jersey |
787 |
|
1995 |
|
|
|
Utah |
600 |
|
1997 |
Jointly conducted with Connecticut |
|
Other: |
|||||
|
China |
Gansu Province |
900 |
1,800 |
1997 |
About 50% of population live in homes built below ground level |
Case-Control Studies
Qualitative Summary of Results
Results of epidemiologic studies of indoor radon concentration and lung-cancer that used surrogate measures were generally consistent with increased risk at higher exposures. Their interpretation is complicated by the inability to link the surrogate measure directly to an estimate of exposure to radon progeny for the study participants. Thus, although the results of these studies are an important step in establishing a link between residential radon and lung-cancer risk, their direct relevance in assessing either the risk posed by indoor exposure or the validity of miner-based risk extrapolations is limited. The most relevant epidemiologic studies of lung-cancer are those which used in-home measurements of radon to estimate exposure, in that direct measurements provide the most accurate estimates of exposure. Eight major case-control studies have been reported that included direct radon measurements, along with a pooled analysis of 3 of the studies and a meta-analysis of the major studies.
Table G-24 summarizes the sizes of the various studies, the radon concentrations and overall results. The highest radon concentrations were found in the Finland's-I study [mean, 210.9 Bqm-3 (5.7 pCiL-1 )], and the next highest in the Stockholm study [mean, 129.5 Bqm-3 (3.5 pCiL-1)] and Winnipeg study (mean, 118.4 and 199.8 Bqm-3 (3.2 and 5.4 pCiL-1) in the living room and basement, respectively). Intermeditate radon concentrations were measured in the Swedish national study [mean, 107.3 Bqm-3 (2.9 pCiL-1)], the Finland-II study [mean, 99.9 Bqm-3 (2.7 pCiL-1)], and the Shenyang study [median, 85.1 Bqm-3 (2.3 pCiL-1)]; and the lowest concentrations were measured in the New Jersey study [median, 22.2 Bqm-3 (0.6 pCiL-1)]. The relationship of the measurement information for both the Swedish national study and the Finland-I study relative to the other studies is uncertain, inasmuch as radon was measured in winter, with detectors placed for 3 and 2 mo, respectively.
Comparisons of results from subgroup analyses provide an additional framework for evaluating consistency among studies. Variations of risk patterns within subgroups and inconsistencies between studies compel a cautious interpretation of results. Three studies—Shenyang, Winnipeg, and Finland-II—found no association with exposure overall and after intense subgroup analysis. Results of the other studies offer mixed support for a positive association. In Finland-I, RRs exceeded 1.0 for all radon categories, but there was no significant trend with increasing radon concentration and the highest category had a low RR. In New Jersey, there was a significant linear trend, but RRs for radon categories, of less than 1.0, 1.0–1.9, 2.0–3.9, and at least 4.0 pCiL-1. were, 1.2, 1.2, 1.3, and 8.7, indicating that the trend was strongly influenced by the highest category, which included 5 cases and 1 controls. In Stockholm, there was a significant trend with radon concentration; however, the trend was affected by occupancy or when
TABLE G-24 Summary of results from case-control studies of residential radon exposure
|
Study |
Cases |
Controls |
Rn levl- pCiL-1 (med/mean) |
Comment |
|
Finland-I |
238 |
415 |
40% > 4.7 20% > 7.4 |
Results show only a modest suggestion of an overall trend with increasing radon level, but all RRs exceeded 1. |
|
Finland-II |
517 |
517 |
cases, 103 Bqm-3 (mean); controls, 96 Bqm-3 (mean) |
Results show no overall trend. Residential occupancy less than 12 h/d. |
|
Israel |
35 |
35 |
1.0 (mean) |
Study has few cases; radon concentrations are very low; no conclusions can be drawn. |
|
Missouri |
538 |
1,183 |
cases, 1.8 (mean); controls, 1.8 (mean) |
Results show no trend with increasing radon level; suggestive trends were found when analyses restricted to adenocarcinoma cases or in-person interviews. |
|
New Jersey |
480 |
442 |
cases, 0.5 (med) controls, 0.5 (med) |
Significant exposure-response trend, but mean exposures very low and results influenced strongly by highest exposure category with 5 cases and 1 control. |
|
Port Hope |
27 |
49 |
Nonsignificant excess relative risk with or without adjustment for smoking. |
|
|
Shenyang |
308 |
356 |
cases, 2.8 (med); controls, 2.9 (med) |
Results show no increasing RR with increasing radon level, overall and within categories of indoor air pollution. |
|
Stockholm |
201 |
378 |
cases, 3.1 (med); controls, 2.9 (med) |
Results suggest positive increase, but cautious interpretation indicated because trend depends on cut-points and disappears after adjustment for occupancy or with BEIR IV weighting. |
exposures more than 15 yr before were given half the weight. Furthermore, it was found that the p value for the test of trend differed when continuous Bqm-3 (pCiL-1) was used as the quantitative value in the test statistic, as opposed to category-specific means. The Swedish national study offered the clearest pattern of increasing RR trend with radon concentration.
Subgroup analyses revealed inconsistencies within and between studies. In New Jersey, there was no trend in the RRs with radon concentration among never-smokers, a positive trend in light smokers (under 25 cigarettes/d), and a negative trend in heavy smokers (at least 25 cigarettes/d); in Stockholm the trend was observed only in never-smokers and in heavy smokers (at least 20 cigarettes/ d); and in the Swedish national study trends, were the same for never-smokers, ex-smokers, and current smokers. In New Jersey, the RR trend was steepest when the case group was restricted to large cell carcinomas (a relatively rare histologic type); in Stockholm, trends were most apparent with small and squamous cell carcinoma; and in Sweden, there was no difference by histologic type. By way of comparison, in miners, there is suggestive evidence that radon-progeny exposure might be more closely associated with small cell carcinoma (Land and others 1993; Yao and others 1994) and adenocarcinoma (Yao and others 1994).
Quantitative Summary Based on Pooled Analysis of Pooled Data from 3 Studies
Results from studies of indoor radon and lung-cancer are quantitatively summarized by either pooling data (Chekoway 1991; Friedenreich 1993) or con-
ducting meta-analysis (Greenland 1987; Thacker 1988). In the former approach, original, primary data from multiple studies are combined and analyzed jointly. In the latter approach, only data from published papers are used (Glass 1976); that is, the study is the unit of analysis (Greenland 1987). Both approaches have well-known limitations due to differences among the studies in design, type and method of data collection, source population, quality-control procedures, information on important confounding variables, and time (Friedenreich 1993; Thacker 1988). Meta-analysis have added burdens associated with the need to rely on information that is available only in the published papers; that limits flexibility to assess the exposure of interest, adjust for important confounders, and evaluate subtle effects (Greenland 1984; Oakes 1990; Petitti 1994; Shapiro 1994).
An analysis of pooled primary data from residential case-control studies in New Jersey, Shenyang, and Stockholm—including almost 1,000 cases—concluded that these 3 studies were consistent with each other and that any differences among them could have arisen by chance (Lubin and others 1994b). The study-specific estimates of RR and 95% CIs at 150 per Bqm-3 based on fitted linear excess-RR models were 1.7 (0.8–3.8), 0.9 (0.0–1.2), and 1.2 (0.8–2.4), respectively. The combined exposure-response relationship showed no trend, with a pooled RR estimate of 1.0 with 95% CI (0.8–1.3) at 150 per Bqm-3. Results suggest that RRs were consistent with no effect of exposure; however, results were also consistent with extrapolations from miners.
Quantitative Summary Based on Meta-Analysis of 8 Studies
A recent meta-analysis involved the 8 studies that had enrolled 200 or more lung-cancer cases is listed in Table G-24 (and shown in figure G-1) (Lubin and Boice 1997). Overall, 4,263 lung-cancer cases and 6,612 controls contributed to the meta-analysis. Figure G-1 suggests that RRs from indoor studies are consistent with the extrapolation based on miner studies, but also that RRs are quite variable. The Cls for the individual RRs are large, suggesting that results are also consistent with no effect of radon concentration. However, more of the RRs exceeded 1.0 than were less than 1.0, and there was a general tendency for higher RRs with higher radon concentrations.
Lubin and Boice (1997) obtained RR estimates and 95% Cls for categories of radon concentration in Bqm-3 from published results and carried out weighted linear-regression analyses of the natural logarithm of the RR estimates using inverse variances as weights (Draper and Smith 1966). For each study, a loglinear RR model that passed through the quantitative value for the baseline category was fitted. For exposure at concentration x, the regression model was
log[RR(x;x0)] = ß (x - x0), (1)
where x0 was the exposure for the referent category and ß the unknown exposure-response parameter. Model (1) was fitted to each study, and an esti-
mate of ßi, denoted ßi, was obtained. A summary estimate for ß1,...,ß8 was obtained with the same 2-step approach used by the committee in its analysis of miners.
Except for the Finland-I study, loglinear models provided good fits to the RR from the individual studies (Figure G-2), and there were no significant deviations from linearity. The study-specific values for the exponential of the estimates in units of 150 Bqm-3, that is, exp(ßi×150), are shown in Table G-25. The fitted RRs at 150 Bqm -3 ranged from 0.8 to 1.8. A test of homogeneity of the estimates was rejected (p < 0.001). The fitted RR at 150 Bqm-3 was exp(0.0009×150)= 1.14 with 95% CI of (1.0–1.3).
The baseline categories for the RRs differed for the various studies. RRs for each category and for each study were adjusted to a baseline concentration of "zero" radon (that is, ambient concentrations) by multiplying each RR by exp(ßix0i), where x0i was the concentration for the baseline category and ßi the estimate for the ith study. With the adjusted RRs, 5 categories of radon concentration were created on the basis of quintiles, less than 55.4, 55.5–88.7, 88.8–142.2, 142.3–250.8, and at least 250.9 Bqm-3. Estimates of RR and 95% CIs for the 5 categories were 1.0, 1.05 (0.9–1.2), 1.05 (0.9–1.2), 1.25 (1.0–1.5), and 1.20 (1.0–1.4). Those RRs were in turn adjusted to a zero baseline by multiplying by exp(ßx0), where x0 was the mean concentration for the lowest radon category, 34.2 Bqm-3, and ß the parameter estimate. Figure 3-2 presented earlier in chapter 3 shows the adjusted RRs (solid squares). The figure also shows that the summary loglinear model, log[RR(x)]= 0.009x, provided a good fit to the data.
Mean cumulative exposure in the miner studies was over 20 times greater than living 30 yr in an average US house at the mean concentration of 46 Bqm-3. With data from the < 0.175 Jhm-3 (< 50 WLM) restricted analysis of miners (see page 3–15), RRs in miners were compared with RRs from the residential studies. A correspondence between exposures for miners in Jhm-3 (WLM) and radon concentrations in homes in Bqm-3 was made assuming 30 yr of exposure, standard residential occupancy assumptions [living for 1 yr in a house at 37 Bqm-3 and assuming 70% occupancy, and 0.4 equilibrium factor is approximately equal to 0.00014 Jhm-3 (0.4 WLM) of exposure], and a 1.0 K factor. For example, a miner exposed to 0.0875 Jhm-3 (25 WLM) was assumed to have about the same exposure as a person living 30 yr in a house with a radon concentration of 220 Bqm-3 [= 37 × 25 WLM/(30 yr × 0.14 × 1.0)]. For miners, RRs were calculated for 0, 1–9, 10–19, 20–29, 30–39, and 40–49 WLM. Figure 3-1 shows RRs and 95% CIs from the miner data (open squares).
The estimate for RR from a loglinear model fitted to the miner RRs under 0. 175 Jhm-3 (50 WLM) was 1.13 at 150 Bqm-3 with a 95 % CI of 1.0–1.2, essentially the same as the 1.14 (1.0, 1.3) estimate from the meta-analysis of residential studies. Thus, RRs for miner exposures under 0.175 Jhm-3 (50 WLM) were similar to extrapolations with the miner-based risk model (Figure G-1), devel-
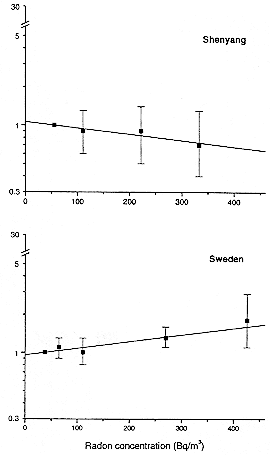
oped from data with generally higher exposures, and similar to the RRs from the indoor studies.
Results from the indoor case-control studies do not provide direct information on lifetime risks posed by radon exposure. The excess risk of 14% at 150 Bqm-3 corresponds to only 30 years of exposure in a house at a constant radon concentration and hence does not reflect the risk of lung-cancer following lifetime exposure, where the estimated excess lifetime relative risk at 150 Bqm-3 based on the miner models is 40 to 50% (Table 3-6). Estimated relative risks from indoor studies and from miner-based models reflect a 30-year exposure period at 148 Bqm-3 and not lifetime exposures at this level. Thus, if exposures outside this 30-year period influence lung-cancer risk, as suggested by the miner data, then the 14% excess relative risk at 148 Bqm-3 from indoor studies is a
TABLE G-25 Estimates of relative risk (RR) at 150 Bqm-3 and the 95% confidence interval (CI) each study and for all studies combined
|
Study |
RRa |
95% CI |
Reported in original paperb |
|
Finland-Ic |
1.30 |
(1.09,1.55) |
N.A. |
|
Finland-II |
1.01 |
(0.94,1.09) |
1.02 |
|
New Jersey |
1.83 |
(1.15,2.90) |
1.77 |
|
Shenyang |
0.84 |
(0.78,0.91) |
0.92d |
|
Winnipeg |
0.96 |
(0.86,1.08) |
0.97 |
|
Stockholm |
1.83 |
(1.34,2.50) |
1.79 |
|
Sweden |
1.20 |
(1.13,1.27) |
1.15 |
|
Missouri |
1.12 |
(0.92,1.36) |
N.A. |
|
Combinede |
1.14 |
(1.01,1.30) |
— |
|
a Values shown are estimated RR at 150 Bqm-3 that is, exp(ß × 150), where ß was obtained from a weighted linear regression fitting the model log(RR) = ß(x - x0), where x0 is the quantitative value for the lowest radon category and x is the category-specific radon level. b RR at 150 Bqm-3, on the basis of computed from exposure-response relationship provided in original reports. Exposure-response data not available (N.A.) in Finland-I and Missouri studies. c For Finland-I, there was a significant departure from linearity (P = 0.03). Estimated RR for 150 Bqm-3 with linear-quadratic model was 1.71. d Taken from results in pooled analysis (Lubin and others 1994a). e Combined estimate and confidence interval based on random-effects model. Fixed effects estimate was 1.11 with 95% CI (1.07,1.15). |
|||
biased estimate of the lifetime relative risk at this concentration and therefore cannot be used to estimate attributable risks for a population.
In the meta-analysis, study-specific exposure-response estimates differed significantly. In an attempt to explain the differences, values for overall mean radon level, percentage of exposure interval covered by radon-measurement data, mean number of homes per subject, mean number of measured homes per subject, percent of cases who smoked, percentage of eligible subjects included in the radon analysis, percentage of homes with year-long radon measurements, percentage living subjects, and percentage female subjects were obtained for each study. None of those variables, individually or jointly, explained the heterogeneity in the study-specific exposure-response estimates.
For the meta-analysis an influence analysis, in which summary estimates were computed on the basis of 7 of 8 studies, indicated that the overall estimates change very little when any single study is omitted.
In summary, there was a significant exposure-response relationship in the meta-analysis by Lubin and Boice (1997) with an estimated RR at 150 Bqm-3 of 1.14, and results were generally confirmatory of miner-based extrapolations of risk and with RR among the least-exposed miners. However, meta-analysis are known to have numerous limitations, including an inability to explore adequately the consistency of results within and between studies and to control for poten-
tially important confounding factors. Nonetheless, the results are consistent with a small effect on lung-cancer associated with exposure to indoor radon progeny.
Finally, the results of the ecologic analysis by Cohen (1995) can be compared with the results of the meta-analysis. In his analysis, Cohen fitted a linear model that resulted in a declining linear excess-RR trend of 0.002/Bqm-3. Figure 3-2 compares the ecologic regression line with RRs from residential studies and from miner studies. It is clear that the negative exposure-response relationship is contradicted by both the miner data and the data from the indoor radon studies.
DESIGN LIMITATIONS OF INDOOR-RADON STUDIES
If miner studies are so unequivocal in showing the carcinogenicity of radon, why are results from current studies of indoor radon, particularly those which include measurements of indoor radon, variable and relatively inconclusive? Case-control studies of lung-cancer and indoor radon are, of course, limited by factors that affect any epidemiologic study as summarized in Table G-26 (Lubin and others 1990a). Inadequate design elements might result in reduced power for a study to detect a significant effect and in biased or confounded estimates. However, studies of lung-cancer and indoor radon have unique features that place additional burdens on the accurate assessment of the effects of exposure and the attainment of sufficient study power (Table G-27):
-
The use of miner-based extrapolations provides uncertain estimates of the size of the RR in homes, although expected RRs are very small—an RR in the range of 1.1–1.3 for a 25-yr exposure at 148 Bqm-3 (4 pCiL -1). For a case-control study, this implies that the distribution of exposures for cases is very similar to the distribution of exposures for controls. As a consequence, substantial numbers of subjects are needed to establish a significant difference in the distributions and
TABLE G-26 Potential limiting factors of case-control studies of indoor radon and lung-cancer
|
Error in estimation of radon exposure |
|
Errors in estimation of tobacco use and other potential confounders |
|
Data missing because of nonresponse |
|
To interview |
|
To radon measurement |
|
From use of surrogate responders |
|
Misclassification of disease |
|
Identification of appropriate target population for selection of controls |
|
Inappropriate design assumptions for accurate assessment of sample size and power: |
|
Incorrect specification of dose-response |
|
Incorrect specification of true exposure distribution |
|
Failure to consider consequences of residential mobility |
|
Failure to consider effects of random error in exposure assessment |
TABLE G-27 Sources of error in estimation of cumulative indoor radon progeny exposure
|
Errors related to measurement of radon: |
|
Counting error for a-track device |
|
Possible effects of airborne contaminants |
|
Measurement at fixed location in room |
|
Measurements limited to 1 or 2 rooms only |
|
Diurnal and seasonal variation |
|
Use of contemporary measurements to characterize past levels |
|
Sources of errors in duration of exposure: |
|
Variation in occupancy over time |
|
Imprecision of estimate of occupancy time |
|
Radon exposures occurring outside home |
|
Measurement gaps for homes within exposure period |
|
Exposure as duration times mean exposure rate as an approximation of time-integrated exposure rate |
|
Conversion of radon concentration to WLa |
|
a WL denotes working levels, the unit of radon progeny measured in studies of underground mines. Conversion of radon to radon progeny is needed to estimate risk based on miner models. |
-
to estimate effects precisely. In addition, because the distribution of radon concentrations is skewed, few homes exceed 148 Bqm-3 (4 pCiL-1); in the United States, only 5–7% of homes are estimated to exceed this level (EPA 1991; Marcinowski and others 1994).
-
Subjects usually live in many homes during their lifetimes, thereby narrowing the range of exposures in the target population and reducing study power. The consequences of residential mobility can be demonstrated with an extreme example: If every member of the population moved every day and radon levels in homes were statistically independent, the total exposure of each subject after, say, 25 yr would be about 25 times the mean exposure rate; thus, there would be little or no exposure variation in the population, and this would complicate the detection of any risk.
-
The use of contemporary measurements in current and past homes results in exposures estimated with great imprecision. Unless those formidable limitations can be addressed—with new measurement technologies, with studies in low-mobility or high-exposure populations, or with the pooling of data—it is uncertain whether definitive results can ever be achieved.
EFFECTS OF ERROR, MOBILITY, AND MISSING DATA ON INDOOR-RADON STUDIES
The pattern of lung-cancer risk in miners suggests that exposures in the preceding 5–30 yr are the most relevant for estimating radon-associated lung-
cancer risk (NRC 1988; Lubin and others 1994). That permits exposure-assessment efforts in residential studies to focus on more-recent years, which is fortunate because it is often impossible to locate residences and measure radon concentrations for homes in which subjects lived previously. However, the extent to which exposures before to the defined exposure-time window contribute to lung-cancer risk, then omitting them by design adds imprecision to the exposure estimates (Lubin and others 1990).
There is an important distinction between error in a measurement device and error in assessment of individual exposure. The most-common area dosimeter used in epidemiologic studies is the a-track detector; radon concentration is determined by counting the number of etched tracks made on plastic film by alpha particles, and the number of tracks is proportional to concentration (Alter and Fleischer 1981; Lovett 1969). Both the counting and measurement processes are subject to error, which has been estimated to be about 15–25% (Létourneau and others 1994; Yeager and others 1991). The error in the measurement process defines the absolute lower bound of the accuracy of any exposure assessment based on a-track devices.
Many other factors contribute to error in the estimation of personal exposure (Table G-27). Total exposure to radon progeny is the sum of exposures received in all environments, including the home, the workplace, and outdoors. a-track detectors are usually left in place for several months to a year. In some studies, concentration measurements from short-term devices (3–7 days) supplemented those from long-term devices that might have been lost or unusable. Residential radon concentrations vary daily and seasonally (Swedjemark 1985), and short-term measurements or single-season measurements might not provide an accurate characterization of year-long radon levels.
For analysis, radon exposure in a defined time window is often computed as the time-weighted average concentration (TWA) or cumulative exposure in Bqm-3-yr (or pCiL-1-yr). For TWA, gaps in the measurement data for previous homes are often ignored when there is more than 1 residence. That can induce bias. Suppose that 1 subject lived for 30 yr in a home measured at 150 Bqm-3 and a second subject lived for 15 yr in a home measured at the same level and 15 yr in an unmeasured home. If one ignores the missing data from the unmeasured home, each subject would have a computed TWA of 150 Bqm-3. However, because of regression toward the mean, the TWA for the latter subject is likely an overestimate. If coverage of the exposure-time window is related to case status, ignoring measurement gaps is potentially biasing. To minimize such biases, an imputation procedure for missing data with adjustment for the variance estimates of parameters would be the preferred approach (Weinberg and others 1996).
In indoor-radon studies, only current radon levels can be measured, and they might not accurately reflect those of 15–30 yr earlier because of tightening of homes for energy conservation or other modifications (Kendall and others 1994). In addition, measurements are typically made only in a few rooms of a house.
Simulation Studies
The effects of errors in exposure, residential mobility, and the inability to measure radon concentrations in all homes in the exposure-time window were illustrated in a series of simulation studies by Lubin and others (1995), expanding earlier analyses of Lubin and others (1990).
Steps in the simulation of data are shown in Table G-28. Case-control studies with M = 700 cases and N = 700 controls were selected from independently generated populations of 10,000 persons, with an overall lung-cancer rate of 10%. Initially, it was assumed that each person lived in only 1 house. The lognormal distribution of US radon concentrations was used, with a geometric mean (GM) of 24.8 Bqm-3 and a geometric standard deviation (GSD) of 3.11 (Marcinowski and others 1994).
A multiplicative error (U) was assumed; In(U) was assumed to be normally distributed with a mean of 0 and a variance of t2. Measurement error was specified by exp(t) as 1.0 (no error), 1.50, 2.0, and 3.0, which roughly correspond to exposure errors of zero, ±50%, ±100%, and ±200%, respectively. For example, exp(t) = 2.0 implies that a true exposure of 0.07 Jhm-3 (20 WLM) will be estimated as 0.035–0.14 Jhm-3 (10–40 WLM). For comparison with current case-control studies, radon progeny exposures were rescaled on the basis 25 yr of exposure to radon concentration, and RRs were computed by categories of Bqm-3.
Simulations were also conducted to illustrate the effects of error, mobility, and missing radon-measurement data from past residences (Lubin and others 1995). Table G-29 provides an empirical calculation of study power for case-control studies with 700 cases and 700 controls and with 2,000 cases and 2,000
TABLE G-28 Steps in simulation study conducted by Lubin and others (1995)
|
1. For each individual, generate a true radon concentration by randomly sampling from a lognormal distribution with geometric mean 24.8 Bqm-3 and GSD 3.11. Convert to radon progeny exposure in WLM on the basis of 25 yr of exposurea (X), multiplying by (0.18/37)25yr, where the first factor represents the conversion under standard assumptions of 1 yr residence in a house at 1 Bqm-3 to exposure in WLM/yr.a 2. Compute the probability of lung-cancer, on the basis of P(D = 1|X) = ea(1+bX){1+ea(1+bX)}-1, with a and b specified. Randomly sample from a uniform 0-1 distribution to determine disease status D. 3. Include a multiplicative error, U, by randomly sampling from a lognormal distribution, where ln(U) is normal with mean 0 and variance t2, and create an observed exposure Z = XU. 4. Repeat steps 1–3 10,000 times to generate a population. 5. Select M disease cases and N controls, categorize radon concentration, and compute RRs and test statistics, using continuous radon concentration as the quantitative variable. 6. Repeat steps 1–5 to generate each simulated case-control data set. |
|
a Initial assumption was 25 yr of occupancy in a single house. This assumption was later relaxed. |
TABLE G-29 The percentage of times P value for score test of no linear trend in relative risk with exposure is less than 0.05, based on 1,000 simulated case-control studiesa
|
|
Number of homes occupied in 5 to 30 yr exposure windowb |
|||||
|
|
1 |
2 |
|
3 |
|
|
|
Error distributionc: exp(t) |
Percent coverage of exposure-time window |
|||||
|
100% |
100% |
50% |
100% |
67% |
33% |
|
|
|
Study size: 700 cases and 700 controls |
|||||
|
1.0 |
45.0 |
30.6 |
17.1 |
28.2 |
24.2 |
11.5 |
|
1.5 |
41.4 |
26.6 |
16.6 |
24.9 |
16.4 |
10.8 |
|
2.0 |
29.4 |
18.8 |
12.7 |
9.4 |
9.2 |
6.8 |
|
3.0 |
17.1 |
11.8 |
7.2 |
6.4 |
8.6 |
5.8 |
|
|
Study size: 2,000 cases and 2,000 controls |
|||||
|
1.0 |
89.8 |
73.6 |
46.0 |
60.8 |
52.6 |
32.1 |
|
1.5 |
84.8 |
66.8 |
44.1 |
55.1 |
42.1 |
29.4 |
|
2.0 |
71.0 |
54.0 |
35.6 |
34.4 |
26.6 |
23.5 |
|
3.0 |
40.8 |
32.9 |
22.0 |
23.6 |
14.6 |
13.0 |
|
a Risk is based on a 0.10 background rate of lung-cancer and an excess relative risk of 0.015 per working level month. Exposure is based on 25 yr of residence and a lognormal radon concentration distribution with geometric mean 24.8 Bqm-3 and geometric standard deviation 3.11. b For multiple homes, it is assumed that equal numbers of years are spent in each home. Thus, for 2 homes, 50% indicates that 12.5 yr of the exposure-time window was covered by radon-measurement data, for 3 homes, 33% and 67% indicate that 8.3 and 16.7 yr of the exposure window were covered by measurement data, respectively. c The multiplicative error distribution is assumed to be lognormal, with the logarithm of the error having mean 0 and variance t2. The row with exp(t) = 1 shows results when exposure is measured without error. |
||||||
controls, that is, the percentage of 1,000 simulated studies that rejected the null hypothesis of a radon effect. The table shows that a study with 700 cases and 700 controls, in which all subjects lived in a single residence for 30 yr and exposure is measured without error, has a power of 0.45 of rejecting a hypothesis test of no exposure effect when the true trend is 0.015/WLM. With 2,000 cases and 2,000 controls, the study has a power of 0.90. The table also illustrates the marked decline in power with increasing exposure error and mobility and with decreasing coverage of the exposure-time window.
Sample Sizes for Case-Control Studies
Table G-30 shows the number of cases required for a study designed to have 90% power to reject a null hypothesis based on a 2-sided 0.05-level test if the alternative, b = 0.015 WLM, were true (Lubin and others 1995). This table updates the sample sizes provided in Lubin and others (1990a). With typical
TABLE G-30 The effects of measurement error, exp(t), and mobility on sample sizea (entries are number of lung-cancer cases required)
|
|
Mobility pattern |
||
|
Exp(t)b |
1 × 25 yr |
2 × 12.5 yr |
3 × 8.3 yr |
|
|
Control-to-case ratio = 1:1 |
||
|
1.0 |
2,033 |
2,447 |
3,408 |
|
1.5 |
2,521 |
3,292 |
4,879 |
|
2.0 |
3,716 |
5,365 |
8,484 |
|
3.0 |
8,429 |
13,530 |
22,694 |
|
|
Control-to-case ratio 2:1 |
||
|
1.0 |
1,488 |
1,810 |
2,530 |
|
1.5 |
1,846 |
2,437 |
3,626 |
|
2.0 |
2,724 |
3,974 |
6,311 |
|
3.0 |
6,183 |
10,034 |
16,895 |
|
a Study required to have 90% power to reject a trend in radon exposure, b = 0, when the true trend is b = 0.015, using a 2-sided 0.05-level test. Exposure based on 25 yr of exposure and occupancy in 1, 2, or 3 houses. b It is assumed that error is multiplicative and the logarithm of the error is normally distributed with mean 0 and variance t2. |
|||
mobility and with exp(t) about 1.5–3.0, about 5,000–18,000 lung-cancer cases and an equal number of controls would be needed, or about 4,000–13,000 cases and twice the number of controls. Those numbers should be interpreted cautiously, perhaps as a lower bound, because calculations do not account for unmeasured houses and adjustment of other risk factors.
CONCLUSIONS
Accurate exposure estimation is essential for any study of lung-cancer and indoor radon. Estimating past exposures is a formidable task, and a present-day measurement, even if made for an entire year, might not accurately reflect radon concentrations of 30 yr ago or earlier. Exposure assessment is further burdened by subject mobility, which decreases the range of exposures in a population and thereby decreases study power. Mobility also creates the potential for gaps in the reconstruction of exposure histories because of an inability to measure all previous houses. When reasonable assumptions are made about measurement errors, mobility and gaps in the exposure-time window, any calculated dose-response relationship will probably be consistent with no exposure effect unless there are substantial numbers of cases and controls. On the basis of those observations, the committee concludes that the seeming inconsistency among case-control studies to date is in large part an inherent consequence of errors in dosimetry and residential mobility.
In recent years, new statistical techniques have been developed for analyses of case-control data that attempt to take errors in exposure assessment into account. The validity of the techniques and a resulting ''corrected" risk estimate require direct information on the form of the error distribution. Thus, the estimation of the true effects of residential radon exposure could be enhanced by the collection of data that allows an evaluation of exposure errors.
The ability to estimate lung-cancer risk from indoor-radon studies is much more complex than the simple computer simulations, which were based only on measurement errors, considered only simple residential mobility patterns, and were defined by an ideal situation: occupancy known exactly, no variation in the equilibrium of radon with its decay products, and no misspecification of disease status. In reality, radon studies suffer from further uncertainties arising from a variable relationship between exposure and dose and from the potential confounding and potentiating effect of tobacco smoke, both active and passive, and the possible presence of other factors, such as indoor air quality or occupational exposures, which may also be measured only imprecisely.
Computer simulations document that small predicted levels of risk and misspecification of radon exposures contribute to the mixed results of current radon case-control studies. The public is perplexed by the seemingly conflicting results and uncertain as to the existence of an adverse effect. Although many of the newer studies are larger than published studies, the marked reduction in study power from (random) errors in exposure suggests that results from the newer studies might also be mixed (Samet 1994).
In the long term, several steps can help to address problems caused by extensive exposure-assessment errors. The most obvious is improvement in estimating exposures, which can be accomplished by selection of a stable target population so that the potential for gaps in exposure measurements is minimized or by using of improved technology for the measurement of radon concentrations.
The power of an indoor-radon study to detect an excess risk could also be enhanced by targeting special populations, such as a population with high exposures, a broad range of exposures, and low residential mobility.
Case-control studies of residential radon are limited by the generally low dose of alpha energy delivered to the lung, which reflects the low radon concentrations to which most of us are exposed every day. The anticipated excess risk is small and not readily measured, because of errors that affect estimation of exposure. On the basis of results of the simulations, the committee concludes the following:
-
Because of error intrinsic in the measurement and estimation of prior radon exposures, a single study cannot be expected to provide sufficiently precise lung-cancer risk estimates.
-
The inconsistencies in existing indoor radon concentrations are not surprising and are probably the consequence of mobility and errors in dosimetry related to missing radon measurements and poor exposure assessment.
-
With increased residential mobility and errors in dosimetry, it is virtually impossible practically to distinguish between studies for which an underlying radon effect is assumed and studies for which no radon risk exists.
-
Investigators are encouraged to include procedures to estimate the distribution of exposure errors in the design of indoor-radon studies, and analyses of studies should adjust for exposure errors.
-
Additional studies of the consequences of missing exposure data within the exposure-time window, of ignoring the roughly 30% of time exposed to nonhome sources of radon progeny, and of incomplete coverage of radon concentrations in prior homes are needed.
-
Improved technologies are needed for the accurate estimation of prior radon-progeny exposure.
Combining data from prior and current studies should be encouraged. However, even with a large sample size a clear picture of lung-cancer risk posed by residential radon exposure might not arise, because the substantial influence of errors in radon-exposure assessment.
References
Aaltonen LA, Peltomaki P, Leach FS, Sistonen P, Pylkkanen L, Mecklin JP, Jarvinen H, Powell SM, Jen J, Hamilton SR, Petersen GM, Kinzler KW, Vogelstein B, de la Chapelle A. 1991. Clues to the pathogenesis of familial colorectal cancer. Science 260:812–816.
Abelson PH. 1990. Uncertainties about health effects of radon (editorial). Science 250:353.
Abelson PH. 1991. Mineral dusts and radon in uranium mines (editorial). Science 254:777.
Adamson IYR. 1985. Cellular kinetics of the lung. Pp. 289–317 in Toxicology of Inhaled Materials, H.P. Witschi and J.P. Brain, eds. Berlin: Springer-Verlag.
Agnew JE. 1984. Physical properties and mechanisms of deposition of aerosol. P. 49 in Aerosols and the Lung: Clinical and Experimental Aspects, S.W. Clarke and D. Pavia, eds. Boston: Butterworths.
Ahlberg J, Ahlbom A, Lipping H, Norell S, Osterblom L. 1981. Cancer among professional drivers: A problem-oriented register-based study. [Swed.] Lakartidningen 78:1545–1546.
Ahlman K, Koskela RS, Kuikka P, Koponene M, Annanmaki M. 1991. Mortality among sulfide ore miners. Am J Ind Med 19:603–617.
Alavanja MCR, Brownson RC, Lubin JH, Brown C, Berger C, Boice JD, Jr. 1994. Residential radon exposure and lung-cancer among nonsmoking women. J Natl Cancer Inst. 86:1829–1837.
Albering HJ, Engelen JJ, Koulischer L, Welle IJ, Kleinjans JC. 1994. Indoor radon, an extrapulmonary genetic risk? Lancet 344:750–751.
Albrecht E and Kaul A. 1967. Continuous registration of 222Rn in air varying with time. In Assessment of Airborne Radioactivity in Nuclear Operations, Vienna: International Atomic Energy Agency.
Alexandrie AK, Sundberg MI, Seidegard J, Tornling G, Rannug A. 1994. Genetic susceptibility to lung-cancer with special emphasis on CYP1A1 and GSTM1: A study on host factors in relation to age at onset, gender and histological cancer types. Carcinogenesis 15(9):1785–1790.
Alter HW and Fleischer RL. 1981. Passive integrating radon monitor for environmental monitoring. Health Phys 40:693–700.
Alter HW and Oswald RA. 1987. Nationwide distribution of indoor radon measurements: A preliminary data base . J Air Pollution Control Assoc 37:227–231.
Amandus H and Costal J. 1991. Silicosis and lung-cancer in U.S. metal miners. Arch Environ Health 46:82–89.
Amstad P, Reddel RR, Pfeifer R, Malan-Shibley L, Mark GE, Harris CC. 1988. Neoplastic transformation of a human bronchial epithelial cell line by a recombinant retrovirus encoding viral Harvey ras. Mol Carcinog 1: 151–60.
Amundson SA, Chen JD, Okinaka RT. 1996. Alpha particle mutagenesis of human lymphoblastoid cell lines. Int J Radiat Biol 70(2):219–226.
Anderson RE, Hill RB, Key CR. 1989. The sensitivity and specificity of clinical diagnosis during five decades. JAMA 261:1610–1617.
Anonymous. 1993. Cigarette smoking-attributable mortality and years of potential life lost in United States, 1990. MMAWR 42(33):645–649.
Anttila SA, Hirvonen K, Husgafvel-Pursiainen A, Karjalainen AT, Nurminen H, Vainio H. 1994. Combined effect of CYP1A1 inducibility and GSTM1 polymorphism on histological type of lung-cancer. Carcinogenesis 15:1133–1135.
Archer VE. 1987. Association of lung-cancer mortality with precambrian granite. Arch Environ Health 42:87–91.
Archer VE. 1996. Radon, silicosis, and lung-cancer. Health Phys 70:268.
Armitage P and Doll R. 1961. Stochastic models for carcinogenesis. Pp. 19–38 in Vol. 4 of the Proceedings of the Fourth Berkeley Symposium on Mathematical Statistics and Probability, J. Neyman, ed. Berkeley and Los Angeles: Univ Calif Press.
Armstrong B and Doll R. 1975. Environmental factors and cancer incidence and mortality in different countries, with special reference to dietary practices. Int J Cancer 15:617–631.
Arnold A, Cossman J, Bakshi A, Jaffe ES, Waldmann TA, Korsmeyer SJ. 1993. Immunoglobulin gene rearrangements as unique colonal markers in human lymphoid neoplasms. N Engl J Med 309:1593–1599.
Arnstein A. 1913. On the so-called "Schneeberg Lung-Cancer." Verhandl di deutsch path Gesellsch 16:332–342.
Atencio EM. 1994. Epithelial cell kinetics of the upper respiratory tract of Wistar rats following radon exposure. Thesis, Masters of Science in Environmental Science, Washington State University.
Auvinen A, Mäkeläinen I, Hakama M, Castrén O, Pukkala E, Reisbacka H, Rytömaa T. In press. Indoor radon and the risk of lung-cancer: A nested case-control study in Finland. J Natl Cancer Inst
Axelson O, Edling C, Kling H. 1979. Lung-cancer and residency — a case-referent study on the possible impact of exposure to radon and its daughters in dwellings. Scand J Work Environ Health 5:10–15.
Axelson O, Andersson K, Desai G, Fagerlund I, Jansson B, Karlsson C, Wingren G. 1988. Indoor radon exposure and active and passive smoking in relation to the occurrence of lung-cancer. Scand J Work Environ Health 14:286–292.
Baird SJS, Cohen JT, Graham JD, Shlyakhter AI, Evans JS. 1996. Noncancer risk assessment: A probabilistic alternative to current practice. Human and Ecological Risk Assessment 2:79–102.
Bale WF. 1980. Memorandum to the files, March 14, 1951: Hazards associated with radon and thoron. Health Phys 38:1062–1066.
Bao CY, Ma AH, Evans HH, Horng MF, Mencl J, Hui TE, Sedwick WD. 1995. Molecular analysis of hypoxanthine phosphoribosyl transferase gene deletions induced by a- and X-radiation in human lymphoblastoid cells. Mutat Res 326:1–15.
Bao S, Harwood PW, Chrisler WB, Groch KM, Brooks AL. 1997. Comparative clastogenic sensitivity of respiratory tract cells to gamma rays. Radiat Res 148:90–97.
Barendsen GW. 1985. Do fast neutrons at low dose rate enhance cell transformation in vitro? A basic problem of microdosimetry and interpretation. Int J Radiat Biol 47:731–734.
Barendsen GW, Coot CJ, Van Kersen GR, Bewley DK, Field SB, Parnell CJ . 1966. The effect of oxygen on impairment of the proliferative capacity of human cells in culture by ionising radiations of different LET. Int J Radiat Biol 10:317–327.
Bartlett S, Krewski D, Wang Y, Zielinski JM. 1993. Evaluation of error rates in large scale computerized record linkage studies. Survey Methodology 19:3–12.
Bartlett S, Richardson GM, Krewski D, Rai SN, Fyfe M. 1996. Characterizing uncertainty in risk assessment — Conclusions drawn from a workshop. Human and Ecological Risk Assessment 2:217–227.
Bartsch H, Hollstein M, Mustonen R, Schmidt J, Spiethoff A, Wesch H, Wiethege T, Muller KM. 1995. Screening for putative radon-specific p53 mutation hotspot in German uranium miners. Lancet 346(8967):121.
Bates MN, Smith AH, Hopenhayn-Rich C. 1992. Arsenic ingestion and internal cancers: A review. Am J Epid 135:462–476.
Battista G, Belli A, Carboncini F, Comba P, Levante G, Sartorelli P, Strambi F, Valentini F, Axelson O. 1988. Mortality among pyrite miners with low-level exposure to radon daughters. Scand J Work Environ Health 14:280–285.
Bauchinger M, Schmid E, Braselmann H, Kalka U. 1994. Chromosome aberrations in peripheral lymphocytes from occupants of houses with elevated indoor radon concentrations. Mutat Res 310:135–142.
Bauchinger M, Braselmann H, Kulka U, IIuber R, Georgiadau-Schumacher V. 1996. Quantification of FISH-painted chromosome aberrations after domestic random exposure. Int J Radiat Biol 70:657–663.
Bauer FW and Robbins SL. 1972. An autopsy study of cancer patients. I. Accuracy of the clinical diagnosis (1955 to 1965) Boston City Hospital. JAMA 221:1471–4.
Baverstock, KF. 1990. Radon and leukaemia (Letter). Lancet 335:1337–1338.
Bean JA, Isacson P, Hausler, Jr. WJ, Kohler J. 1982a. Drinking water and cancer incidence in Iowa. I. Trends and incidence by source of drinking water and size municipality. Am J Epidemiol 116(6):912–23.
Bean JA, Isacson P, Hahne RMA, Kohler J. 1982b. Drinking water and cancer incidence in Iowa. II. Radioactivity in drinking water. Am J Epidemiol 116(6):924–32.
Becker, KH, Reineking A, Scheibel HG, Porstendörfer J. 1984. Radon daughter activity size distributions. Radiat Prot Dosimet 7:147–150.
Bedford JS and Goodhead DT. 1989. Breakage of human interphase chromosomes by alpha-particles and X-rays. Int J Radiat Biol 55:211–216.
Beebe GW, Ishida I, Jablon S. 1962. Studies of the mortality of A-bomb survivors. I. Plan of study and mortality in the medical subsample (Selection 1), 1950–1958. Radiat Res 16:253–280.
Bender MA, Awa AA, Brooks AL, Evans HJ, Groer PG, Littlefield LG, Pereira C, Preston RJ, Wachholz BW. 1988. Current status of cytogenetic procedures to detect and quantify previous exposures to radiation. Mutat Res 196:103–159.
Bernhard S, Pineau JF, Rannou A, Zettwoog P. 1984. 1983: One year of individual dosimetry in French miners. Occupational Radiation Safety in Mining, Vol.2 Proceedings of the International Conference, Oct 14–18, Toronto. H. Stocker, Ed. 526. Canadian Nuclear Association.
Biberman R, Lusky A, Schlesinger T, Margaloit M, Neeman E, Modan B. 1993. Increased risk for small call lung-cancer following residential exposure to low-dose radon: A pilot study. Arch Environ Health 48:209–12.
Bigu J. 1985. Theoretical models for determining 222Rn and 220Rn progeny levels in Canadian underground U mines—a comparison with experimental data. Health Phys 48:371–399.
Birchall A and James AC. 1994. Uncertainty analysis of the effective dose per unit exposure from radon progeny and implications for ICRP risk-weighting factors. Radiat Prot Dosimet 53:133–140.
Bishop JM. 1983. Cellular oncogene retroviruses. Ann Rev Biochem 52:301–354.
Bishop JM and Varmus HE. 1984. Functions and origins of retroviral transforming genes. Pp. 990–1108 in RNA Tumor Viruses: Molecular Biology of Tumor Viruses, 2nd ed., R. Weiss, N. Teich, H. Varmus, J. Coffin, eds. Cold Spring Harbor: Cold Spring Harbor Laboratory.
Bishop VJ. 1971. Control of radon daughters in the Colorado Plateau mines. Presented at the Mining Environmental Conference. University of Missouri-Rolla, October.
Bisson M, Collier CG, Poncy JL, Taya A, Morlier JP, Strong J, Baker S, Monchaux G, Fitsch P. 1994. Biological dosimetry in different compartments of the respiratory tract after inhalation of radon and its daughters. Radiat Prot Dosimet 56:89–92.
Blocher D. 1988. DNA double strand break repair determines the RBE of alpha particles. Int J Radiat Biol 54:761–771.
Blot WJ and Fraumeni JF, Jr. 1994. Arsenic and lung-cancer. Pp. 207–218 in The Epidemiology of lung-cancer, J. Samet, ed. New York: Marcell Dekker.
Blot WJ, Xu Z-Y, Boice, JD Jr., Zhao D-Z, Stone BJ, Sun J, Jing L-B, Fraumeni JF, Jr. 1990. Indoor radon and Lung-Cancer in China. J Natl Cancer Inst 82:1025–30.
Bodmer WF, Bishop T, Karran P. 1994. Genetic steps in colorectal cancer. Nat Genet 6:217–219.
Boffetta P, Stellman SD, Garfinkel L. 1988. Diesel exhaust exposure and mortality among males in the american cancer society prospective study. Am J Ind Med 14:403–415.
Boffetta P, Harris RE, Wynder. 1990. Case-control study of occupational exposure to diesel exhaust and lung-cancer risk. Am J Ind Med 17:577–591.
Bogen KT. 1994. A note on compounded conservatism. Risk Anal 14:379–381.
Bogen KT. 1995. Methods to approximate joint uncertainty and variabitity in risk. Risk Anal 15:411–419.
Boivin J. 1995. Smoking, treatment for Hodgkin's Disease, and subsequent lung-cancer risk. J Natl Cancer Inst 87:1502–3.
Boothman DA, Davis TW, Sahijdak WM. 1994 Enhanced expression of thymidine kinase in human cells following ionizing radiation. Int J Radiat Oncol Biol Phys 30:391–398.
Borek C, Ong A, Mason H. 1987. Distinctive transforming genes in X-ray — Transformed mammalian cells. Proc Natl Acad Sci USA 84:794–798.
Boring CC, Squires TS, Tong T. 1993. Cancer statistics. CA Cancer J Clin 43:7–26.
Bos JL. 1990. Ras oncogenes in human cancer: A review [published erratum appears in Cancer Res 50:1352. 1990] Cancer Res 49:4682–4689.
Bowie SHU. 1990. Radon and leukaemia (Letter). Lancet 335:1336.
Braby LA. 1992. Microbeam studies of the sensitivity of structures within living cells. Scanning Microscopy 6(1):167–174.
Brain JD and Valberg PA. 1974. Models of lung retention based on ICRP task group report. Arch Environ Health 28(1): 1–11.
Brain JD and Valberg PA. 1979. Deposition of aerosols in the respiratory tract. Am Rev Respir Dis 120:1325–1373.
Brenner DJ. 1989. The effectiveness of single alpha particles. Pp. 477–480 in Low Dose Radiation: Biological Bases of Risk Assessment, J. Lancashire, ed. London and New York: Taylor and Francis.
Brenner DJ. 1992. Radon: current challenges in cellular radiobiology. Int J Radiat Biol 61:3–13.
Brenner DJ. 1994. The significance of dose rate in assessing the hazards of domestic radon exposure. Health Phys 67:76–79.
Brenner DJ and Hall EJ. 1990. The inverse dose-rate effect for oncogenic transformation by neutrons and charged particles. A plausible interpretation consistent with published data . Int J Radiat Biol 58: 745–758.
Brenner DJ and Hall EJ. 1992. Radiation induced oncogenic transformation: The interplay between dose, dose protraction, and radiation quality. Adv Radiat Biol 16: 859–885.
Brenner DJ and Sachs RK. 1994. Chromosomal "fingerprints" of prior exposure to densely ionizing radiation. Radiat Res 140:123–142.
Brenner DJ and Sachs RK. 1996. Comments on "Comment on the ratio of chromosome-type dicentric interchanges to centric rings for track-clustered compared with random breaks" by Savage and Papworth. Radiat Res 146(2):241–242.
Brenner DJ and Ward JF. 1992. Constraints on energy deposition and target size of multiply damaged sites associated with DNA double-strand breaks. Int J Radiat Biol 61:737–748.
Brenner DJ, Hall EJ, Randers-Pehrson G, Miller RC. 1993. Mechanistic considerations on the dose-rate/LET dependence of oncogenic transformation by ionizing radiations. Radiat Res 133:365–369.
Brenner DJ, Miller RC, Huang Y, Hall EJ. 1995. The biological effectiveness of radon-progeny alpha particles III Quality factors. Radiat Res 142:61–69.
Brenner DJ, Hahnfeldt P, Amundson SA, Sachs RK. 1996. Interpretation of inverse dose-rate effects for mutagenesis by sparsely ionizing radiation. Int J Radiat Biol 70(4):447–458.
Brenner H, Savitz DA, Jockel KH, Greenland S. 1992. The effects of nondifferential exposure misclassification in ecological studies. Am J Epidemiol 135(1):85–95.
Breslin AJ, George AC, Weinstein MS. 1969. Investigation of the radiological characteristics of uranium mine atmospheres. HASL-220. Health and Safety Laboratory. New York, NY: U.S. Atomic Energy Commission.
Breslow NE and Clayton DG. 1993. Approximate Inference in Generalized Liner Mixed Models. J Am Stat Assoc 88:9–25.
Breslow NE and Day NE. 1987. Statistical Methods in Cancer Research. Volume II. The Design and Analysis of Cohort Studies. Lyon: International Agency for Research on Cancer.
Breslow NE and Storer BE. 1985. General relative risk functions for case-control studies. Am J Epidemiol 122(1):149–62.
Breslow NE, Lubin JH, Marek P, Langholz B. 1983. Multiplicative models and the analysis of cohort studies. J Am Stat Assoc 78:1–12.
Bridges BA, Cole J, Arlett CF, Green MHL, Waugh APW, Beare D, Henshaw DL, Last RD. 1991. Possible association between mutant frequency in peripheral lymphocytes and domestic radon concentrations. Lancet 337:1187–1189.
Brightwell J, Fouillet X, Cassano-Zoppi AL, Bernstein D, Crawley F, Duchosal F, Gatz R, Percel S, Pfeifer H. 1989. Tumors of the respiratory tract in rats and hamsters following chronic inhalation of engine exhaust emissions. J Appl Toxicol 9:23–31.
Brodeur GM, Seeger RC, Schwab M, Varmus HE, Bishop JM. 1984. Amplification of N-myc in untreated human neuroblastomas correlates with advanced disease stage. Science 224:1121–1124.
Brooks AL. 1975. Chromosome damage in liver cells from low dose rate alpha, beta and gamma irradiation: derivation of RBE. Science 190:1090–1092.
Brooks AL. 1980. Low dose and low dose-rate effects on cytogenetics. Pp. 263–276 in Radiation Biology in Cancer Research, R.E. Meyn and H.R. Withers, eds. New York: Raven Press.
Brooks AL. 1996. Biodosimetry and molecular signatures (Cellular Changes) In: Radiation Risk Assessment, Statistical methodology and mechanisms, American Statistical Association 1996 Conference on Radiation and Health, June 23–27. Vail, Colorado.
Brooks AL, Benjamin SA, Jones RK, McClellan RO. 1982. Interaction of 144CE and partial-hepatectomy in the production of liver neoplasms in the Chinese Hamster. Rad Res 91:573–588.
Brooks AL, Newton GJ, Shyr L-J, Seiler FA, Scott BR. 1990a. The combined effects of alpha-particles and X-rays on cell killing and micronuclei induction in lung epithelial cells. Int J Radiat Biol 58:799–811.
Brooks AL, Rithidech K, Johnson NF, Thomassen DG, Newton GJ. 1990b. Evaluating chromosome damage to estimate dose to tracheal epithelial cells. Pp. 601–614 in Part 2 of the Twenty-Ninth Hanford Life Sciences Symposium, Indoor Radon and Lung-Cancer: Reality or Myth? Richland: Hanford Press.
Brooks AL, Khan MA, Duncan A, Buschbom RL, Jostes RF, Cross FT. 1994. Effectiveness of radon relative to acute 60Co gamma-rays for induction of micronuclei in vitro and in vivo. Int J Radiat Biol 66:801–808.
Brooks AL, Miick R, Buschbom RL, Murphy MK, Khan MA. 1995. The role of dose rate on the induction of micronuclei in deep-lung fibroblasts in vivo after exposure to cobalt-60 gamma rays. Radiat Res 144:114–118.
Brooks AL, McDonald KE, Mitchell C, Culp DS, Lloyd A, Johnson NF, Kitchin RM. 1996. The combined genotoxic effects of radiation and occupational pollutants. Appl Occup Environ Hyg 11(4):414–416.
Brooks AL, Bao S, Harwood PW, Wood BH, Chrisler WB, Khan MA, Gies RA, Cross FT. 1997. Induction of micronuclei in respiratory tract following radon inhalation. Int J Radiat Biol 72(5):485–495.
Brown CC and Chu KC. 1983. Implications of the multistage theory of carcinogenesis applied to occupational arsenic exposure. J Natl Cancer Inst 70:455–463.
Burch PRJ and Chesters MS. 1986. Neoplastic transformation of cells in vitro at low and high dose rates of fission neutrons: an interpretation. Int J Radiat Biol 49:495–500.
Burchall A and James AC. 1994. Uncertainty analysis of the effective dose per unit exposure from radon progeny and implications for ICRP risk-weighting factors. Radiat Prot Dosimet 53(1–4):133–40.
Burmaster DE and Anderson PD. 1994. Principles of good practice for the use of Monte Carlo techniques in human health and ecological risk assessments. Risk Anal 14:477–481.
Burmaster DE and Thompson KM. 1998. Fitting second-order parametric distributions to data using maximum likelihood estimation. Human and Ecological Risk Assessment 4(2):319–339.
Burmaster DE and Wilson AM. 1996. An introduction to second-order random variables in human health risk assessment. Human and Ecological Risk Assessment 2(4):892–919.
Burnett RT, Ross WH, Krewski D. 1995. Nonlinear random effects regression models. Environmentrics 6:85–99.
Burns DM. 1994. Tobacco smoking. In Epidemiology of Lung-Cancer, JM Samet, ed. New York: Marcel Dekker, Inc.
Burns PB and Swanson GM. 1991. The occupational cancer incidence surveillance study (ociss): risk of lung-cancer by usual occupation and industry in the Detroit metropolitan area. Am J Ind Med 19:655–671.
Busigin A, Van der Vooren AW, Babcock JC, Phillips CR. 1981. The nature of unattached 218Po (RaA) particles. Health Phys 40:333–343.
Butland BK, Muirhead CR, Draper GJ. 1990. Radon and leukaemia (letter). Lancet 335:1338–1339.
Butterworth BE and Goldsworthy TL. 1991. The role of cell proliferation in multistage carcinogenesis. Proc Soc Exp Biol Med 198:683–7.
Canadian Task Force on the Periodic Health Examination. 1990. Periodic health examination, 1990 update: 3. Interventions to prevent lung-cancer other than smoking cessation. Can Med Assoc J 143:269–272.
Cao JM, Wells RL, Elkind MM. 1992. Enhanced sensitivity to neoplastic transformation by 137Cs gamma-rays of cells in the G2-/M-phase age interval. Int J Radiat Biol 62:191–199.
Cao JM, Wells RL, Elkind MM. 1993. Neoplastic transformation of C3H mouse embryo cells, 10T1/2: cell cycle dependence for 50 kV X-rays and UV-B light. Int J Radiat Biol 64:83–92.
Caporaso N, DeBaun MR, Rothman N. 1995. Lung-cancer and CYP2D6 (the debrisoquine polymorphism): sources of heterogeneity in the proposed association. Pharmacogenetics 5:S129–34.
Cardis E, Gilbert ES, Carpenter L, Howe G, Kato I, Armstrong BK, Beral V, Cowper G, Douglas A, Fix J, Fry SA, Kaldor J, Lave C, Salmon L, Smith PG, Voelz GL, Wiggs LD. 1995. Effects of low doses and low dose rates of external ionizing radiation: Cancer mortality among nuclear industry workers in three countries. Radiat Res 142:117–32.
Carrano AV and Heddle JA. 1973. The fate of chromosome aberrations. J Theor Biol 38:289–304.
Carta P, Cocco PL, Casula D. 1991. Mortality from lung-cancer among Sardinian patients with silicosis. Br J Ind Med 48:122–129.
Carta P, Cocco P, Picchiri, G. 1994. Lung-cancer mortality and airways obstruction among metal miners exposed to silica and low levels of radon daughters. Am J Ind Med 25:489–506.
Cartwright BG and Shirk EK. 1978. A nuclear-track-recording polymer of unique sensitivity and resolution. Nucl Instrum Meth 153:457–460.
Cavanee WK. 1989. Tumor progression stage; specific losses of heterozygosity. Int Symp Princess Takamatsu Cancer Res Fund 20:33–42.
Cavenee WK, White RL. 1995. The genetic basis of cancer. Sci Am 272:72–79.
Cavanee WK, Hansen MF, Nordenskjold M, Kock E, Maumenee I, Squire JA, Phillips RA, Gallie BL. 1985. Genetic origin of mutations predisposing to retinoblastoma. Science 228:501–503.
CDC (Centers for Disease Control). 1995. Morbidity and Mortality Weekly Report, Atlanta, GA — Cigarette smoking among adults — United States 1993. JAMA 273(5):369–370.
Chameaud J, Perraud R, Chretien J, Masse R, Lafuma J. 1982. Lung carcinogenesis during in vivo cigarette smoking and radon daughter exposure in rats. Recent Results Cancer Res 82:11–20.
Chameaud J, Masse R, Lafuma L. 1984. Influence of radon daughter exposure at low doses on occurrence of lung-cancer in rats. Radiat Prot Dosimet 7:385–391.
Chang WP and Little JB. 1992. Persistently elevated frequency of spontaneous mutations in progeny of CHO clones surviving X-irradiation; association with delayed reproductive death phenotype. Mutat Res 270:191–199.
Charles M, Cox R, Goodhead DT, Wilson A. 1990. CEIR forum on the effects of high-LET radiation at low doses/dose rates. Int J Radiat Biol 58:859–885.
Charlton DC, Nikjoo H, Humm JL. 1989. Calculation of initial yields of single and double-strand breaks in cell nuclei from electrons, protons and alpha particles. Int J Radiat Biol 56:1–19.
Chaudhry MA, Jiang Q, Ricanati M, Horng MF, Evans HH. 1996. Characterization of multilocus lesions in human cells exposed to X radiation and radon. Radiat Res 145:31–38.
Checkoway H. 1991. Data pooling in occupational studies. J Occup Med 33:1257–1260.
Chen DJ, Strinste GF, Tokita N. 1984. The genotoxicity of a-particles in human embryonic skin fibroblasts. Radiat Res 100:321–327.
Chen DJ, Carpenter S, Hanks T. 1990. Mutagenic effects of alpha particles in normal human skin fibroblasts. Pp. 569–581 in Twenty-Ninth Hanford Life Sciences Symposium, Indoor Radon and Lung-cancer: Reality or Myth? Richland: Battelle Press.
Chen JQ, McLaughlin JK, Zhang J, Stone BJ, Jiamo L, Rongan C, Dosemeci M, Rexing SH, Wu Z, Hearl FJ, McCawley MA, Blot WJ. 1991. Mortality among dust-exposed Chinese mine and pottery workers in Tongji Medical University China and National Cancer Institute USA (eds): "Study of Silicosis and Lung-cancer for Dust Exposed Workers (Silica, Silicosis and Lung-cancer)." 1:1–12.
Cheng, YS and Yeh HC. 1980. Theory of a screen-type diffusion battery. J Aerosol Sci 11:313–320.
Cheng YS, Keating JA, Kanapilly GM. 1980. Theory and calibration of a screen-type diffusion battery. J Aerosol Sci 11:549–556.
Cheng YS, Yeh HC, Mauderly JL, Mokler BV. 1984. Characterization of diesel exhaust in a chronic inhalation study. Am Ind Hyg Assoc J 45:547–555.
Chia SE, Chia KS, Phoon WH, Lee HP. 1991. Silicosis and lung-cancer among Chinese granite workers. Scand J Work Environ Health 17:170–174.
Chiyotani K, Saito K, Okubo T, Takahashi K. 1990. Lung-cancer risk among pneumoconiosis patients in Japan, with special reference to silicotics. In: Simonato L, Fletcher AC, Saracci R, Thomas TL (eds): "Occupational Exposure to Silica and Cancer Risk," IARC Sci Pub 97:95–104. Lyon, France: IARC.
Chmelevsky D, Kellerer AM, Lafuma J, Morin M, Masse R. 1984. Comparison of the induction of pulmonary neoplasms in Sprague-Dawley rats by fission neutrons and radon daughters. Radiat Res 98:519–535.
Churg A. 1994. Lung-Cancer Cell Type and Occupational Exposure. In Epidemiology of Lung-Cancer, JM Samet, ed. New York: Marcel Dekker, Inc.
Clayson DB, Nera EA, Lok E. 1989. The potential for the use of cell proliferation studies in carcinogen risk assessment. Regulat Toxicol Pharmacol 9:284–295.
Clifton KH, Groch KM, Domann FE, Jr. 1991. Thyroid clonogen biology and carcinogenesis. Prog Clin Biol Res 369:173–183.
Cohen AJ and Higgins MWP. 1995. Health effects and diesel exhaust: Epidemiology. Pp. 251–292 in Diesel Exhaust: A Critical Analysis of Emissions, Exposure, and Health Effects. A Special Report of the Institute's Diesel Working Group. Cambridge, MA: Health Effects Institute.
Cohen BL. 1990. A test of the linear-no threshold theory of radiation carcinogenesis. Environ Res 53:193–220.
Cohen BL. 1993. Relationship between exposure to radon and various types of cancer. Health Phys 65:529–537.
Cohen BL. 1994. Invited Commentary: In defense of ecologic studies for testing a linear-no threshold theory. Am J Epidemiol 139:756–768.
Cohen BL. 1995. Test of the linear-no threshold theory of radiation carcinogenesis for inhaled radon decay products . Health Phys 68:157–174.
Cohen BL and Colditz GA. 1990. Tests of the linear no-threshold theory of radon induced lung-cancer. Environ Res 53:193–220.
Cohen BL and Colditz GA. 1995. Lung-cancer mortality and radon exposure: A test of the linear-no-threshold model of radiation carcinogenesis. In Radiation and Public Perception. Benefits and Risks. Washington, DC: American Chemical Society.
Cohen BL and Gromicko N. 1988. Variation of radon levels in U.S. homes with various factors. JAPCA 38(2):129–134.
Cohen BL, Kulwicki DR, Warner KR Jr, Grassi CL. 1984. Radon concentrations inside public and commercial buildings in the Pittsburgh area. Health Phys 47(3):399–405.
Cohen SM and Ellwein LB. 1990. Cell proliferation in carcinogenesis. Science 249:1007–1011.
Cohen SM, Garland EM, Ellwein LB. 1992. Cancer enhancement by cell proliferation. Prog Clin Biol Res 374:213–229.
Cole J, Green MHL, Bridges BA, Waugh APW, Beare DM, Henshaw D, Last R, Liu Y, Cortopassi G. 1996. Lack of evidence for an association between the frequency of mutants or translocations in circulating lymphocytes and exposure to radon gas in the home. Radiat Res 145:61–69.
Cole LA. 1993. Elements of Risk: The Politics of Radon. Washington, DC: AAAS Press.
COMARE (Committee on Medical Aspects of Radiation in the Environment). 1996. Fourth Report: The incidence of cancer and leukaemia in young people in the vicinity of the Sellafield site. West Cumbria: Further studies and an update of the situation since the publication of the report of the Black Advisory Group in 1984. Conference on Occupational Radiation Safety in Mining. (Stocker, H., ed.). Toronto, Canada: Canadian Nuclear Association, pp. 344–349.
Cooper JA, Jackson PO, Langford JC, Petersen MR, Stuart BO. 1973. Characteristics of attached radon-222 daughters under both laboratory and filed conditions with particular emphasis upon underground uranium mine environments. US Bureau of Mines Report H0220029. Battelle: Pacific Northwest Laboratories (Report No. BN-SA-299).
Coquerelle TM, Weibezahn KF, Lücke-Huhle C. 1987. Rejoining of double-strand breaks in normal human and ataxia-telangiectasia fibroblasts after exposure to 60Co ?-rays, 241Am a-particles or bleomycin. Int J Radiat Biol 51:209–218.
Corkill DA, Dory AB. 1984. A retrospective study of radon daughter concentrations in the work-place in the fluorospar mines of St. Lawrence, NFLD. Report No. INFO-0127. Ottawa: Atomic Energy Control Board.
Cornforth MN and Goodwin EH. 1991. The dose-dependent fragmentation of chromatin in human fibroblasts by 3.5-MeV alpha particles from 238Pu: Experimental and theoretical considerations pertaining to single-track effects. Radiat Res 127:64–74.
Court-Brown WM and Doll R. 1958. Expectation of life and mortality from cancer among British radiologists. Br Med J 2:181–190.
Cowell JK and Hogg A. 1992. Genetics and cytogenetics of retinoblastoma. Cancer Genet Cytogenet 64:1–11.
Cox R. 1982. A cellular description of the repair detect in ataxia-telangiectasia. In: Bridges BA, Harnden DG, eds. Ataxia Telangiectasia: A Cellular and Molecular Link Between Cancer, Neuropathology, and Immune Deficiency. New York, NY: Wiley Medical Publications.
Cox R. 1994a. Human cancer predisposition and the implications for radiological protection. Int J Radiat Biol 66:643–647.
Cox R. 1994b. Molecular mechanisms of radiation oncogenesis. Int J Radiat Biol 65:57–64.
Cox R and Masson WK. 1979. Mutation and inactivation of cultured mammalian cells exposed to beams of accelerated heavy ions. III. Human diploid fibroblasts. Int J Radiat Biol 36:149–160.
Cox R, Thacker J, Goodhead DT. 1977a. Inactivation and mutation of cultured mammalian cells by aluminium characteristic ultrasoft X-rays. II. Dose-responses of Chinese hamster and human diploid cells to aluminium X-rays and radiations of different LET. Int J Radiat Biol Relat Stud Phys Chem Med (6):561–576.
Cox R, Thacker J, Goodhead DT, Munson RJ. 1977b. Mutation and inactivation of mammalian cells by various ionizing radiations. Nature 267:425–427.
Cross FT. 1981. Experimental studies on lung carcinogenesis and their relationship to future research on radiation-induced lung-cancer in humans. Pp. 27–35 in The Future of Human Radiation Research, Gerber GB, Taylor DM, Cardis E, Thiessen JW, eds. (Section 2 Lung-Cancer), Report 22, British Institute of Radiology, London. Madison: Medical Physics Publishing.
Cross FT. 1992. A review of experimental animal radon health effects data. Pp. 476–481 in Radiation Research: A Twentieth-Century Perspective, Vol. II. JD Chapman, WC Dewey, GF Whitmore, eds. San Diego: Academic Press.
Cross FT. 1994a. Invited commentary: residential radon risks from the perspective of experimental animal studies. Am J Epidemiol 140:333–339.
Cross FT. 1994b. Evidence of cancer risk from experimental animal radon studies. Pp. 79–87 in Radiation and Public Perception. American Chemical Society.
Cross FT, Palmer RF, Busch RH, Filipy RE, Stuart BO. 1981. Development of lesions in Syrian golden hamsters following exposure to radon daughters and uranium ore dust. Health Phys 41:135–153.
Cross FT, Palmer RF, Dagle GE, Busch RH, Buschbom RL. 1984. Influence of radon daughter exposure rate, unattachement fraction, and disequilibrium on occurrence of lung tumors. Radiat Prot Dosimet 7:381–384.
Cross FT, Palmer RF, Busch RH, Dagle GE, Filipy RE. 1986. An overview of PNL radon experiments with reference to epidemiological data. Pp. 608–623 in Lifespan Radiation Effects Studies in Animals: What Can They Tell Us? CONF-830951, Office of Scientific and Technical Information, United States Department of Energy.
Cross FT, Dagle GE, Gies RA, Smith LG, Buschbom RL. 1995. Experimental animal studies of radon and cigarette smoke. Pp. 821–844 in Indoor Radon and Lung-Cancer: Reality or Myth? Part 2, Cross FT, ed.
Crump KS. 1994a. Limitations of biological models of carcinogenesis for low-dose extrapolation. Risk Anal 14:883–6.
Crump KS. 1994b. Use of mechanistic models to estimate low-dose cancer risks. Risk Anal 14:1033–8.
Crump KS, Lambert T, Chen C. 1991. Assessment of Risk from Exposure to Diesel Engine Emissions: Report to the U.S. Environmental Protection Agency for Contract No. 68-02-4601 (Work Assignment No. 182, July). Office of Health Assessment, U.S. Environmental Protection Agency, Washington, DC.
Curtis SB. 1989. A possible role of the inverse dose-rate effect in the radon exposure problem. Pp. 547–553 in Low Dose Radiation, Biological Bases of Risk Assessment, Baverstock KF, Stather JW, eds. London: Taylor and Francis.
Dakins ME, Toll JE, Small MJ. 1994. Risk-based environmental remediation: Decision framework and roles of uncertainty. Environ Toxicol Chem 13:1907–1915.
Dalla-Favera RS, Martinotti S, Gallo R, Erikson J, Croce C. 1983. Translocation and rearrangement of the c-myc oncogene locus in human undifferentiated B-cell lymphomas. Science 219:963–997.
Damber L and Larsson LG. 1987. Lung-cancer in males and type of dwelling. An epidemiologic pilot study. Acta Oncol 26:211–215.
Darby SC and Doll R. 1990. Radiation and exposure rate. Nature 344:824.
Darby S, Whitley E, Howe GR, Hutchings SJ, Kusiak RA, Lubin JH, Morrison HI, Timarche M, Tomásek L, Radford EP, Roscoe RJ, Samet JM, Yao SX. 1995. Radon exposure and cancers other than lung-cancer in underground miners: a collaborative analysis of 11 studies. J Natl Cancer Inst 87:378–384.
Dare WL, Lindblom RA, Soule JH. 1953. Uranium mining on the Colorado Plateau. Bureau of Mines Information Circular 7726, September.
Davidian M and Giltinan DM. 1995. Nonlinear Models for Repeated Measurement Data. New York: Chapman and Hall.
Davies CN, ed. 1967. Aerosol Science. New York: Academic Press.
Davies CN, 1980. An algebraical model for the deposition of aerosols in the human respiratory tract during steady breathing-Addendum. J Aerosol Sci 11:213–224.
Davis AL. 1976. Bronchogenic carcinoma in chronic obstructive pulmonary disease. JAMA 235:621–622.
de Lara CM, Jenner TJ, Townsend KMS, Marsden SJ, O'Neill P. 1995. The effect of dimethyl sulfoxide on the production of DNA double-strand breaks in V79-4 mammalian cells by alpha particles. Radiat Res 144:43–49.
Dennis JA and Dennis A. 1988. Neutron dose effect relationships at low doses. Radiat Environ Biophys 27:91–101.
Deshpande A, Goodwin EH, Bailey SM, Marrone BL, Lehnert BE. 1996. Alpha-particle-induced sister chromatid exchange in normal human lung fibroblasts: Evidence for an extranuclear target. Radiat Res 145:260–267.
Doll R and Peto R. 1978. Cigarette smoking and bronchial carcinoma: Dose and time relationships among regular smokers and life-long non-smokers. J Epidemiol Community Health 32:303–313.
Doll R and Peto R. 1981. The Causes of Cancer. Oxford: Oxford University Press.
Doll R, Gray R, Hafner B, Peto R. 1980. Mortality in relation to smoking: 22 years' observations on female British doctors. Br Med J 2:967–71.
Dosemeci M, Wacholder S, Lubin JH. 1990. Does nondifferential misclassification of exposure always bias a true effect towards the null value? Am J Epidemiol 132:746–8.
Dousset M and Jammet H. 1985. Comparison of cancer mortality in Limousin and Poitou-Charentes. Radioprotection 20:61–7.
Draper NR and Smith H. 1966. Applied Regression Analysis. New York: Wiley.
Driscoll KE, Maurer JK, Hassenbein D, Carter J, Janssen YMW, Mossman BT, Osier M,. Oberdörster G. 1994. Contribution of macrophage-derived cytokines and cytokine networks to mineral dust-induced lung inflammation. Pp. 177–189 in Toxic and Carcinogenic Effects of Solid Particles in the Respiratory Tract, U. Mohn, D. L. Dungworth, J. L. Mauderly and G. Oberdörster, eds. Washington: International Life Sciences Institute Press.
Driscoll KE, Carter JM, Howard BW, Hassenbein DG, Pepelko W, Baggs RB, Oberdörster G. 1996. Pulmonary, inflammatory, chemokine, and mutagenic responses in rats after subchronic inhalation of carbon black. Toxicol Appl Pharmacol 136:372–380.
DSMA Atcon Ltd. 1985. Elliot Lake Study: Factors affecting the uranium mine working environment prior to the introduction of current ventilation practices. A research report prepared for the Atomic Energy Control Board, Ottawa, Canada. March 4, 1985.
Dua SK, Hopke PK, Raunemaa T. 1995. Hygroscopic Growth of Indoor Aerosols. Aerosol Sci Technol 23:331–340.
Eatough JP and Henshaw DL. 1992. Radon and thoron associated dose to the basal layer of the skin. Phys Med Biol 37:955–967.
Eatough JP and Henshaw DL. 1995. The theoretical risk of nonmelanoma skin cancer from domestic radon exposure. J Rad Protect 15(1):45–51.
Economou P, Lechner JF, Samet JM. 1994. Familial and genetic factors in the pathogenesis of lung-cancer. In Epidemiology of Lung-Cancer, JM Samet, ed. New York: Marcel Dekker, Inc.
Edelmann KG and Burmaster DE. 1997. Are all distributions of risk with the same 95th percentile equally acceptable? Human and Ecological Risk Assessment 3:223–234.
Edling C, Comba P, Axelson O, Flodin U. 1982. Effects of low-dose radiation — a correlation study. Scand J Work Environ Health 8:59–64.
Edling C, Kling H, Axelson O. 1984. Radon in homes — a possible cause of lung-cancer. Scand J Work Environ Health 10:25–34.
Edling C, Wingren G, Axelson O. 1984. Radon daughter exposure in dwellings and lung-cancer. Pp. 29–34 In Indoor Air. Radon, Passive Smoking, Particulates and Housing Epidemiology, Vol. 2, Berglund B, Lindvall T, Sundell J, eds. Stockholm Council for Building Res.
Elenitoba-Johnson K, Medeiros LJ, Khorsand J, King TC. 1995. Lymphoma of the mucosa-associated lymphoid tissue of the lung A multifocal case of common clonal origin. Am J Clin Pathol 103:341–345.
Elkind MM. 1991. Enhanced neoplastic transformation due to protracted exposures of fission-spectrum neutrons: a biophysical model (Letter). Int J Radiat Biol 59:467–1475.
Elkind MM. 1994. Radon-induced cancer: A cell-based model of tumorigenesis due to protracted exposures. Int J Radiat Biol 66(5):649–653.
Emerit I, Oganesian N, Sarkisian T, Arutykuknykan R, Pogosian A, Asrian K, Levy A, Cernjavski L. 1995. Clastogenic Factors in the plasma of chernobyl accident recovery workers: Anti-clastogenic effect of Ginko Biloba extract. Radiat Res 144:198–205.
EML (Environmental Measurements Laboratory). 1990. EML Procedures Manual, 27th edition. US Department of Energy Report No. HASL-300. Chapter 2. New York, NY: Environmental Measurements Laboratory.
Enderle GJ, Friedrich K. 1995. East German uranium miners (Wismut) — exposure conditions and health consequences. Stem Cells 13:78–89.
Ennemoser O, Ambach W, Brunner P, Schneider P. 1994. Unusual high radon exposure in homes and lung-cancer. Lancet 344:127.
Enstrom JE. 1979. Rising lung-cancer mortality among nonsmokers. J Natl Cancer Inst 62:755–760.
Enterline PE, Marsh GM. 1983. Mortality among workers exposed to arsenic and other substances in a copper smelter. Pp. 226–244 in Arsenic: Industrial, Biomedical Environmental Perspectives. New York: Van Nostrand Reinhold Co.
Enterline PE, Marsh GM, Esmen NA, Henderson VL, Callahan CM, Paik M. 1987. Exposure to arsenic and respiratory cancer: A reanalysis. Am J Epidemiol 125:929–938.
Enterline PE, Day R., Marsh GM. 1995. Cancers related to exposure to arsenic at a copper smelter. Occup Environ. Med. 52:28–32.
Evans HH. 1991. Cellular and molecular effects of radon and other alpha particle emitters. Adv Mutat Res 3:28–52.
Evans HH. 1992. Relationship of the cellular and molecular effects of alpha-particle irradiation to radon-induced lung-cancer. Pp. 537–555 in Twenty-Ninth Hanford Life Sciences Symposium, Indoor Radon and Lung-Cancer: Reality or Myth? Richland: Battelle Press.
Evans HH, Mencl J, Bakale G, Rao PS, Jostes RF, Hui TE, Cross FT, Schwartz JL. 1993. Interlaboratory comparison of the effects of radon on L5178Y cells: Dose contribution of radon daughter association with cells. Radiat Res 136:49–56.
Evans HJ. 1993. Molecular genetic aspects of human cancers: The 1993 Frank Rose Lecture. Br J Cancer 68:10951–1060.
Fearon ER, Cho KR, Nigro JM, Kern SE, Simons JW, Ruppert JM, Hamilton SR, Preisinger AC, Thomas G, Kinzler KW, Vogelstein B. 1990. Identification of a chromosome 189 gene that is altered in colorrectal cancers. Science 247:49–56.
Fialkow PJ. 1976. Colonal origin of human tumors. Biochem Biophys Acta 458:283–321.
Fidler IJ and Talmadge JE. 1986. Evidence that intravenously derived murine pulmonary melanoma metastases can originate from the expansion of a single tumor cell. Cancer Res 46(10):5167–5171.
Finkelstein MM. 1995. Silicosis, radon, and lung-cancer risk in Ontario miners. Health Phys 69:396–399.
Finkelstein MM and Kusiak RA. 1995. Clinical Measures, Smoking, Radon Exposure and Lung-Cancer Risk Among Elliot Lake Uranium Miners. Ontario: Ontario Ministry of Labour.
Finley B and Paustenbach D. 1994. The benefits of probabilistic exposure assessment: 3 case studies involving contaminated air, water, and soil. Risk Anal 14:53–74.
Finley B, Proctor D, Scott P, Harrington N, Paustenbach D, Price P. 1994. Recommended distributions for exposure factors frequently used in health risk assessment. Risk Anal 14:533–554.
Fleischer RL. 1981. A possible association between lung-cancer and phosphate mining and processing. Health Phys 41:171–175.
Fleischer RL. 1986. A possible association between lung-cancer and a geological outcrop. Health Phys 50:823–827.
Folkard M, Prise KM, Vojnovic B, Newman HC, Roper MJ, Hollis KJ, Michael BD. 1995. Radiat Prot Dosimet 61:215–218.
Forastiere F, Valesini S, Arca' M, Magliola ME; Michelozzi P; Tasco C. 1985. Lung-cancer and natural radiation in an Italian province. Sci Total Environ 45:519–526.
Ford JR, Terzaghi-Howe M. 1992a. Basal cells are the progenitors of primary tracheal epithelial cell cultures. Exp Cell Res 198:69–77.
Ford JR, Terzaghi-Howe M. 1992b. Characteristics of magnetically separated rat tracheal epithelial cell populations. Am J Physiol 263 (Lung Cell. Mol. Physiol. 7): L568–L574.
Fox JC and McNally NJ. 1990. The rejoining of DNA double-strand breaks following irradiation with 238Pu a-particles: evidence for a fast component of repair as measured by neutral filter elution. Int J Radiat Biol 57:513–521.
Franko AJ, Sharplin J, Ward WF, Taylor J. 1996. Evidence for two patterns of inheritance of sensitivity to lung fibrosis in mice by radiation, one of which involves two genes. Radiat Res 146:68–74.
Fried BM. 1931. Primary carcinoma of the lung: Bronchiogenic cancer—a clinical and pathological study. Medicine X:373–188.
Friendenreich CM. 1993. Methods for pooled analyses of epidemiologic studies . Epidemiology 4:295–302.
Frost SE. 1983. Beaverlodge working level calculations, draft number 4. Eldorado Resources, Ltd., Ottawa. August 10.
Fry RJM, Powers-Risius PE, Alpen L, Ainsworth EJ. 1985. High-LET radiation carcinogenesis. Radiat Res 104:S188–195.
Fuchs, NA. 1964. The Mechanics of Aerosols. Elmsford: Pergamon Press.
Furst A. 1983. A new look at arsenic carcinogenesis. Pp. 151–164 in Arsenic—Industrial, Biomedical Environmental Perspectives. New York: Van Nostrand Reinhold Co.
Furuse T, Otsu H, Noda Y, Kobayashi S, Ohara H. 1992. Induction of liver tumors and lung tumors in C57BL/6J male mice irradiated with low doses of high LET radiations. In Proceedings of the International Conference on Low Dose Irradiation and Biological Defense Mechanisms, Kyoto, Japan, Sugahara T, Sagan LA, Aoyama T, eds. Amsterdam: Elsevier Science Publishers.
Garfinkel L. 1981. Time trends in lung-cancer mortality among nonsmokers and a note on passive smokers. J Natl Cancer Inst 66:1061–1066.
Garshick E, Schenker MB, Muñoz A, Segal M, Smith TJ, Woskie SR, Hammond SK, Speizer FE. 1987. A case-control study of lung-cancer and diesel exhaust exposure in railroad workers. Am Rev Respir Dis 135:1242–1248.
Garshick E, Schenker MB, Muñoz A, Segal M, Smith TJ, Woskie SR, Hammond SK, Speizer FE. 1988. A retrospective cohort study of lung-cancer and diesel exhaust exposure in railroad workers. Am Rev Respir Dis 137:820–825.
Geard CR. 1985. Charged particle cytogenetics: Effects of LET, fluence, and particle separation on chromosome aberrations. Radiat Res Suppl 8:S112–S121.
Geard CR, Brenner DJ, Randers-Pehrson G, Marino S. 1991. Single-particle irradiation of mammalian cells at the Radiological Research Accelerator Facility: Induction of chromosomal changes. Nucl Instr Meth B54:411–416.
George AC. 1972. Measurement of the uncombined fraction of radon daughters with wire screens . Health Phys 23:390–392.
George AC. 1976. Scintillation flasks for the determination of low level concentrations of radon. In Proceedings of Ninth Midyear Health Physics Symp. Denver.
George AC. 1977. A passive environmental radon monitor. In: Breslin, AJ, ed., Radon Workshop-February 1977, New York: Health and Safety Laboratory; HASL-325, 25–30.
George AC. 1984. Passive integrated measurement of indoor radon using activated carbon. Health Phys 46:867–872.
George AC. 1993. Measurement of airborne 222Rn daughters by filter collection and alpha radioactivity collection. IARC Sci Publ 109:173–179.
George AC and Breslin AJ. 1980. The Distribution of Ambient Radon and Radon Daughters in Residential Buildings in the New Jersey-New York Area, National Radiation Environment III, Vol. 2, CONF-780422. Technical Information Center, U.S. Department of Energy, p. 1272.
George AC and Weber T. 1990. An improved passive activated C collector for measuring environmental 222Rn in indoor air. Health Phys 58:583–589.
George AC, Hinchliffe L, Sladowski R. 1975. Size distribution of radon daughter particles in uranium mine atmospheres. Am Ind Hyg Assoc J 36:484–490.
George AC, Hinchliffe L, Sladowski R. 1977. Size distribution of radon daughter particles in uranium mine atmospheres. Report No. HASL-326.
George AC, Wilkening MH, Knutson EO, Sinclair D, Andrews L. 1984. Measurements of Radon and Radon Daughter Aerosols in Socorro, New Mexico, Aerosol Sci Technol. 3:277–281.
Gilbert ES. 1994. Smoking as an explanation for the negative relationship between exposure to radon and certain types of cancer. Health Phys 67:192.
Gilbert ES, Cross FT, Dagle GE. 1996. Analysis of lung tumor risks in rats exposed to radon. Radiat Res 145(3):350–360.
Gilliland FD and Samet JM. 1994. Lung-Cancer. London, England: Imperial Cancer Research Fund. 175 19. Trends in Cancer Incidence and Mortality.
Gilliland DG, Blanchard KL, Levy J, Perrin S, Bunn F. 1991. Clonality in myeloproliferative disorders: analysis by means of the polymerase chain reaction. Proc Natl Acad Sci USA 88:6848.
Glass GV. 1976. Primary, secondary, and meta-analysis of research. Educ Researcher 5:3–8.
Goddard MJ and Krewski D. 1995. The future of mechanistic research in risk assessment: where are we going and can we get there from here? Toxicology 102:53–70.
Goddard MJ, Krewski D, Zhu Y. 1994. Measuring carinogenic potency. Pp. 193–208 in Environmental Statistics, Assessment and Forecasting, C.R. Cothern and N.P. Ross, eds. Boca Raton: Lewis Publishers.
Goldsmith DF and Samet JM, editors. 1994. Silica exposure and pulmonary cancer. Epidemiology of Lung-Cancer. New York, New York: Marcel Dekker, Inc. 11:248–298.
Goldsmith DF, Beaumont JJ, Morrin LA, Schenker MB. 1995. Respiratory cancer and other chronic disease mortality among silicotics in California. Am J Ind Med 28:459–467.
Goldstein SD and Hopke PK. 1985. Environmental neutralization of polonium-218. Environ Sci Technol 19:146–150.
Goldsworthy TL, Morgan KT, Popp JA, Butterworth BE. 1991. Guidelines for measuring chemically induced cell proliferation in specific rodent target organs. Pp. 253–284 in Chemically Induced Cell Proliferation: Implications for Risk Assessment. Wiley-Liss, Inc.
Goodhead DT. 1988. Spatial and temporal distribution of energy. Health Phys 55:231–240.
Goodhead DT. 1994. Initial events in the cellular effects of ionizing radiations: clustered damage in DNA. Int J Radiat Biol 65:7–17.
Goodhead DT and Nikjoo H. 1989. Track structure analysis of ultrasolf X-rays compared to high- and low-LET radiations. Int J Radiat Biol 55:513–529.
Goodhead DT, Munson RJ, Thacker J, Cox R. 1980. Mutation and inactivation of cultured mammalian cells exposed to beams of accelerated heavy ions: IV. Biophysical interpretation. Int J Radiat Biol 37:135–167.
Goodhead DT, Bance DA, Stretch A, Wilkinson RE. 1991. A versatile plutonium-238 irradiator for radiobiological studies with alpha-particles. Int J Radiat Biol 59:195–210.
Goodwin EH and Cornforth MN. 1994. RBE: Mechanisms inferred from cytogenetics. Adv Space Res 14:249–255.
Gopalakrishnan S, Lui-X, Patel DJ. 1992. Solution structure of the covalent sterigmatocystin-DNA adduct. Biochemistry 31:10790–10801.
Gorgojo L and Little JB. 1989. Expression of lethal mutations in progeny of irradiated mammalian cells. Int J Radiat Biol 55:619–630.
Gray DJ and Windham ST. 1987. EERF standard operating procedures for radon-222 measurement using charcoal cannisters. U.S. Environmental Protection Agency Report EPA 520/5-87-005, Washington, D.C.
Gray RG, Lakuma J, Parish SE, Peto R. 1986. Lung tumors and radon inhalation in over 2,000 rats: approximate linearity across a wide range of doses and potentiation by tobacco smoke. In: Lifespan radiation effects studies in animals: what can they tell us? Pp. 592–607 in Proceedings of the 22nd annual Hanford Life Sciences Symposium (CONF-830951). Springfield: National Technical Information Service.
Greenland S. 1983. Tests for interaction in epidemiologic studies: a review and s study of power. Stat Med 2:243–251.
Greenland S. 1984. Bias methods for deriving standardized morbidity ratio and attributable fraction estimates. Stat Med 3:131–141.
Greenland S. 1987. Quantitative methods in the review of epidermiologic literature. Epidemiol Reviews 9:1–30.
Greenland S. 1993. Basic problems in interaction assessment. Environ Health Perspect 101:59–66.
Greenland S. 1994. Can meta-analysis be salvaged? Am J Epidemiol 140:783–787.
Greenland S and Morgenstern H. 1989. Ecological bias, confounding, and effect modification. Int J Epidemiol 18:269–274.
Greenland S and Robins J. 1994a. Accepting the limits of ecologic studies: Drs. Greenland and Robins reply to Drs. Piantadosi and Cohen. Am J Epidemiol 139:769–771.
Greenland S and Robins J. 1994b. Invited Commentary: Ecologic studies — biases, misconceptions, and counterexamples. Am J Epidemiol 139:747–760.
Griffin CS, Harvey AN, Savage JRKS. 1994. Chromatid damage induced by 238Pu alpha particles in G2 and S-phase Chinese hamster cells. Int J Radiat Biol 66:85–98.
Griffin CS, Marsden SJ, Stevens PL, Simpson P, Savage JRK. 1995. Frequencies of complex chromosome exchange aberrations induced by 238Pu alpha-particles and detected by fluorescence in situ hybridization using single chromosome-specific probes. Int J Radiat Biol 67:431–439.
Griffiths SD, Marsden SJ, Wright EG, Greaves MF, Goodhead DT. 1994. Lethality and mutagenesis of B lymphocyte progenitor cells following exposure to a-particles and X-rays. Int J Radiat Biol 66:197–205.
Groch KM, Khan MA, Brooks AL, Saffer JD. 1997. Differential lung-cancer response following inhaled radon by A/J and C57BL/6J mice. Int J Radiat Biol 71(3):301–308.
Guerrero I, Villasante A, Corces V, Pellicer A. 1984. Activation of a c-K-ras oncogene by somatic mutation in mouse lymphomas induced by gamma radiation. Science 225:1159–1162.
Gunby JA, Darby SC, Miles JC, Green BM, Cox DR. 1993. Factors affecting indoor radon concentration in the United Kingdom. Health Phys 64:2–12.
Haenszel W, Shimkin MB, Mantel N. 1958. A retrospective study of lung-cancer in women. J Natl Cancer Inst 21:825–842.
Hall EJ. 1987. Radiobiology for the Radiologist, 3rd Edition. Philadelphia: Lippincott.
Hall EJ and Freyer GA. 1991. The molecular biology of radiation carcinogenesis. Basic Life Sci 58:3–19; discussion 19–25.
Hall EJ and Hei TK. 1990. Modulating factors in the expression of radiation-induced oncogenic transformation. Environ Hlth Perspect 88:149–155.
Hall EJ, Geard CR, Brenner DJ. 1992. Risk of breast cancer in ataxia telangiectasia. N Engl J Med 326:1358–9.
Hall JM, Lee MK, Newman B, Morrow JE, Anderson LA, Huey B, King MC. 1990. Linkage of early-onset familial breast cancer to chromosome 17q21. Science 250:1684–1689.
Hall NEL and Wynder EL. 1984. Diesel exhaust exposure and lung-cancer: A case-control study. Environ Res 34:77–86.
Hamilton LD, Swent LW, Chambers DB. 1990. Visit to the Centre of Radiaion Hygiene, Institute of Hygiene and Epidemiology, Prague, Czechoslovakia. Trip report to Division of Environmental Health, World Health Organization, Geneva, Switzerland. December, SENES Consultants Limited, Ontario, Canada.
Hammon EC, Garfinkel L. 1968. Changes in cigarette smoking 1959–1965. Am J Public Health Nations Health 68(1):30–45.
Han A and Elkind MM. 1979. Transformation of mouse C3H/10T1/2 cells by single and fractionated doses of x-rays and fission-spectrum neutrons. Cancer Res 39:123–30.
Han A, Hill CK, Elkind MM. 1980. Repair of cell killing and neoplastic transformation at reduced dose rates of 60Co gamma rays. Cancer Res 40:3328–3332.
Harber P, Oren A, Moshenifar Z, Lew M. 1986: Obstructive airway disease as risk factor for asbestos-associated malignancy. J Occup Med 28:82–86.
Harley JH. 1952. Sampling and measurement of airborne daughter products of radon. Doctoral dissertation. Rensselaer Polytechnic Institute.
Harley JH. 1953. Sampling and measurement of airborne daughter products of radon. Nucleonics 11:12–15.
Harley JH. 1980. Sampling and measurement of airborne daughter products of radon. Reprinted in Health Phys 38:1067.
Harley NH. 1988. Radon daughter dosimetry in the rat tracheobronchial tree. Radiat Prot Dosimet 24:457–461.
Harley NH and Pasternack BS. 1972. Alpha absorption measurements applied to lung dose from radon daughters. Health Phys 23:771–782.
Harley NH and Pasternack BS. 1981. A model for predicting lung-cancer risks induced by environmental levels of radon daughters. Health Phys 40:307–316.
Harley NH and Pasternack BS. 1982. Environmental radon daughter alpha dose factors in a five-lobed human lung. Health Phys 42:789–799.
Harley NH and Robbins ES. 1992. 222Rn alpha dose to organs other than the lung. Radiat Prot Dosimet 45: 617–622.
Harley NH, Cohen BS, Robbins ES. 1996. The variability in radon decay product bronchial dose. Environmental International 22:S959–S964.
Harris CC and Hollestein M. 1993. Clinical implications of the p53 tumor-suppressor gene. N Engl J Med 329:1318–1327.
Harris CC, Reddel R, Pfeifer A, Iman D, McMenamin M, Trump BF, Weston A. 1991. Role of oncogenes and tumour suppressor genes in human lung carcinogenesis. IARC Sci Publ 105:294–304.
Harting FH and Hesse W. 1879. Der Lungenkrebs, die bergkrankheit in den schneeberger gruben. Viertel Gerichtl Med Oeff Sanitaetswes 31:102–132, 313–337.
Harvey GJ, Jr. 1977. Trackless mining at Union Carbide's operations in the Uravan District in southwestern Colorado and southeastern Utah. Presented at First Conference. Uranium Mining Technology. University of Nevada, Reno, April 24–29, 1977.
Harris D and Barlow K. 1996. Human interindividual variability in cancer risks — Technical and management challenges. Human and Ecological Risk Assessment 2:194–220.
Hattis D and Burmaster DE. 1994. Assessment of variability and uncertainty distributions for practical risk analysis. Risk Anal 14:713–730.
Hattis D and Silver K. 1994. Human interindividual variability — A major source of uncertainty in assessing risks for noncancer health effects. Risk Anal 14:421–432.
Hayes RB, Thomas T, Silverman DT, Vineis P, Blot WJ, Mason TJ, Pickle LW, Correa P, Fontham TH, Schoenberg JB. 1989. Lung-cancer in motor exhaust-related occupations. Am J Ind Med 16:685–895.
Haynes RM. 1988. The distribution of domestic radon concentrations and lung-cancer mortality in England and Wales. Radiat Prot Dosimet 25:93–96.
Heady JA, Kennaway EL. 1949. The increase in deaths attributed to cancer of the lung. Br J Cancer 111:311–321.
HASL (Health and Safety Laboratory). 1960. Experimental Environmental Survey of AEC Leased Uranium Mines. Report No. HASL-91. New York: U.S. Atomic Energy Agency, Health and Safety Laboratory.
HASL (Health and Safety Laboratory). 1969. Investigation of the radiological characteristics of uranium mine atmospheres. New York: U.S. Atomic Energy Commission.
HEI (Health Effects Institute). 1995. Diesel Exhaust: A Critical Analysis of Emissions, Exposure, and Health Effects. A Special Report of the Institute's Diesel Working Group, Health Effects Institute, Cambridge, MA, April 1995.
Hei TK, Bedford J, Waldren CA. 1994a. p53 mutation hotspot in radon-associated lung-cancer. Lancet 343:1158–1159
Hei TK, Piao CQ, Willey JC, Thomas S, Hall EJ. 1994b. Malignant transformation of human bronchial epithelial cells by radon-simulated alpha particles. Carcinogenesis 15:431–437.
Hei TK, Zhu LX, Waldren CA. 1994c. Molecular mechanisms of mutagenesis by radiation of different qualities. Pp 171–176 in Molecular Mechanisms in Radiation Mutagenesis and Carcinogenesis, Chadwick KH, Cox R, Leehouts HP, Thacker J, eds. Brussels: European Commission.
Heinrich U, Muhle H, Takenaka S, Ernst E, Furst R, Mohr U, Pott F, Stöber W. 1986. Chronic effects on the respiratory tract of hamsters, mice and rats after long-term inhalation of high concentrations of filtered and unfiltered diesel engine emissions. J Appl Toxicol 6:383–395.
Heinrich U, Furst R, Rittinghausen S, Creutzenberg O, Bellmann B, Koch W, Levsen K. 1995. Chronic inhalation exposure of wistar rats and two different strains of mice to diesel engine exhaust, carbon black, and titanium dioxide. Inhal Toxicol 7:533–556.
Henshaw DL, Eatough JP, Richardson RB. 1990. Radon as a causative factor in induction of myeloid leukemia and other cancers. Lancet 335(8696):1008–1012.
Henshaw DL, Eatough JP, Richardson RB. 1990. Radon and leukaemia (Letter). Lancet 335:1339.
Hertz-Picciotto I, Smith AH. 1993. Observations on the dose-response curve for arsenic exposure and lung-cancer. Scand J Environ Health 19(4):217–226.
Hertz-Picciotto I, Smith AH, Holtzman D, Lipsett M, Alexeeff G. 1992. Synergism between occupational arsenic exposure and smoking in the induction of lung-cancer. Epidemiology 3:23–31.
Hess CT, Weiffenbach CV, Norton SA. 1983. Environmental radon and cancer correlations in Maine. Health Phys 45:339–348.
Hessel PA, Sluis-Crerner GK, Hnizdo E. 1990. Silica exposure, silicosis, and lung-cancer: A necropsy study. Br J Ind Med 47:4–9.
Heyder J and Schuech G. 1983. Diffusional transport of nonspherical aerosol particles. Aerosol Sci Technol 2:41.
Heyder J, Gebhart J, Stahlhofen W, Stuck B. 1982. Biological variability of particle deposition in the human respiratory tract during controlled and spontaneous mouth-breathing. Ann Occup Hyg 26:137.
Heyder J, Gebhart J, Rudolf G, Schiller CF, Stahlhofen W. 1986. Deposition of particles in the human respiratory tract in the size range 0.0015–15 m. J Aerosol Sci 17:811.
Hickman AW, Jaramillo RJ, Lechner JF, Johnson NF. 1994. Alpha particle-induced p53 protein expression in a rat lung epithelial cell strain. Cancer Res 54:5797–5800.
Hill AB and Faning EL. 1948. Studies in the incidence of cancer a factory handling inorganic compounds of arsenic. I. Mortality experience in the factor. Br J Ind Med 5:1–6.
Hill CK, Han A, Elkind MM. 1984. Fission-spectrum neutrons at a low dose rate enhance neoplastic transformation in the linear, low dose region (0–10 cGy). Int J Radiat Biol 46:11–15.
Hill CK, Carnes BA, Han A, Elkind MM. 1985. Neoplastic transformation is enhanced by multiple low doses of fission-spectrum neutrons. Radiat Res 102:404–410.
Hill CK, Renan M, Buess E. 1991. Is neoplastic transformation by high-LET radiations dose rate dependent or cell cycle dependent? P. 344 in Proceedings of the International Congress on Radiation Research, JD Chapman, WC Dewey, and GF Whitmore, eds. San Diego: Academic Press.
Hinds WC. 1982. Aerosol Technology-Properties, Behavior, and Measurement of Airborne Particles. New York: Wiley-Interscience.
Hlatky L, Sachs RK, Hahnfeldt P. 1992. The ratio of dicentrics to centric rings produced in human lymphocytes by acute low-LET radiation. Radiat Res 129:304–308.
Hnizdo E and Sluis-Cremer GK. 1991. Silica exposure, silicosis, and lung-cancer: A mortality study of South African gold miners. Br J Ind Med 48:53–60.
Hodgkins PS, O'Neill P, Stevens D, Fairman MP. 1996. The severity of a a-particle induced DNA damage is revealed by exposure to cell-free extracts. Radiat Res 146:660–667.
Hodgson RD and Jones RD. 1990. Mortality of a cohort of tin miners 1941–86. Br J Ind Med 47:665–676.
Hoffman FO and Hammonds JS. 1994. Propagation of uncertainty in risk assessments: The need to distinguish between uncertainty due to lack of knowledge and uncertainty due to variability. Risk Anal 14:707–712.
Hofmann W. 1982. Dose calculations for the respiratory tract from inhaled natural radionuclides as a function of age, 11. Basal cell dose distributions and associated lung-cancer risk. Health Phys 43:31–44.
Hofmann W, Katz R, Zhang CX. 1985. Lung-cancer in a Chinese high background area - epidemiological results and theoretical interpretation. Sci Total Environ 45:527–534.
Holaday, DA. 1969. History of the exposure of miners to radon. Health Phys 16(5):547–552.
Holaday DA, Rushing DE, Coleman RD, Woolrich PF, Kusnetz HL. 1957. Control of radon and daughters in uranium mines and calculations on biological effects. PHS Publication No. 494. Washington, D.C. U.S. Government Printing Office.
Holley WR and Chatterjee A. 1996. Clusters of DNA damage induced by ionizing radiation: Formation of short DNA fragments. I. Theoretical modelling. Radiat Res 145:188–199.
Hollstein M, Sidransky D, Vogelsetein B, Harris CC. 1991. p53 mutations in human cancers. Science 253:49–53.
Hollstein M, Bartsch H, Wesch H, Kure EH, Mustonen R, Muhlbauer KR, Spiethoff A, Wegener K, Wiethege T, Muller KM. 1997. P53 gene mutation analysis in tumors of patients exposed to alpha-particles. Carcinogenesis 18(3):511–516.
Holub RF and Knutson EO. 1987. Measuring polonium-218 diffusion-coefficient spectra using multiple wire screens. Pp. 340–356 in Radon and Its Decay Products: Occurrence, Properties and Health Effects, Symposium Series 331 , Hopke, P.K., ed. Washington, DC: American Chemical Society.
Hoover HC and Hoover LH. 1950. Georgius Agricola De Re Metalallica. New York: Dover Publications, Inc.
Hopke PK. 1992. Some thoughts on the ''unattached" fraction of radon decay products. Health Phys 63:209–212.
Hopke PK, Ramamurthi, Knutson EO, Tu KW, Scofield P, Holub RF, Cheng YS, Su YF, Winklmayr W, Strong JC, Solomon S, Reineking A. 1992. The measurement of activity-weighted size distribution of radon progeny: methods and laboratory intercomparison studies. Health Phys 63(5):560–570.
Hopke PK, Montassier N, Wasiolek P. 1993. Evaluation of the effectiveness of several air cleaners for reducing the hazard from indoor radon progeny. Aerosol Sci Technol 19:268–278.
Hopke PK, Jensen B, Montassier N. 1994. Evaluation of several air cleaners for reducing indoor radon progeny. J Aerosol Sci 25:395–405.
Hopke PK, Jensen B, Li CS, Montassier N, Wasiolek P. 1995. Assessment of the exposure to and dose from radon decay products in normally occupied homes. Environ Sci Technol 29:1359–1364.
Hornung RW and Meinhardt TJ. 1987. Quantitative risk assessment of lung-cancer in U.S. uranium miners. Health Phys 52:417–430.
Hornung RW, Deddens J, Roscoe R. 1995. Modifiers of exposure-response estimates for lung-cancer among miners exposed to radon progeny. Environ Health Perspect 103(Supp 2):49–53.
Howe GR and RH Stager. 1996. Risk of lung-cancer mortality after exposure to radon decay products in the Beaverlodge cohort based on revised exposure estimates. Radiat Res 146:37–42.
Howe GR, Nair RC, Newcombe HB, Miller AB, Abbatt JD. 1986. Lung-cancer mortality (1950–80) in relation to radon daughter exposure in a cohort of workers at the Eldorado Beaverlodge Uranium Mine. J Natl Cancer Inst 77:357–362.
Howe GR, Nair RC, Newcombe HB, Miller AB, Burch JD, Abbat JD. 1987. Lung-cancer mortality (1950–1980) in relation to radon daughter exposure in a cohort of workers at the Eldorado Port Radium uranium mine: Possible modification by exposure rate. J Natl Cancer Inst 79:1255–1260.
Hui TE, James AC, Jostes RF, Schwartz JL, Swinth KL, Cross FT. 1993. Evaluation of an alpha probe detector for in vitro cellular dosimetry. Health Phys 64(6):647–652.
Hunter T. 1991. Cooperation between oncogenes. Cell 64:249–270.
IARC (International Agency for Research on Cancer). 1986. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Tobacco smoking.
IARC (International Agency for Research on Cancer). 1987. International Agency for Research Monography on the Evaluation of Carcinogenic Risks to Humans. Overall Evaluations of Carcinogenicity: An Updating of IARC Monographs Volumes 1 to 42, Supplement 7. Lyon, IARC.
IARC (International Agency for Research on Cancer). 1988. Man-made Mineral Fibres and Radon. Lyon, France: International Agency for Research on Cancer. p. 143. IARC Monographs on the Evaulation of Carcinogenic Risks to Humans.
ICRP (International Commission on Radiological Protection). 1987. Lung-cancer risk from indoor exposures to radon daughters. Oxford, England: Pergamon Press 50.
ICRP (International Commission on Radiological Protection). 1991. 1990 Recommendations of the International Commission on Radiation Protection, ICRP Publication 60, Ann. of ICRP 21.
ICRP (International Commission on Radiological Protection). 1993. Protection Against Radon-222 at Home and at Work. London, England: Pergamon Press 23(2):1–65. ICRP Publication 65.
ICRP (International Commission on Radiological Protection). 1994. Human Respiratory Tract Model for Radiological Protection. A Report of Committee 2 of the ICRP. ICRP Publication 66. Ann ICRP 24(1/4) (Oxford: Pergamon Press).
ICRU (International Commission on Radiation Units and Measurements). 1986. The Quality Factor in Radiation Protection. ICRU Report 40. Washington, DC: ICRU Publications.
Iliakis G. 1984. The mutagenicity of a-particles in Ehrlich ascites tumor cells. Radiat Res 99:52–58.
Ishinishi N, Kuwabara N, Nigase S, Suzuki T, Ishiwata S, Kohno T. 1986. Long-term inhalation studies on effects of exhaust from heavy and light duty diesel engines on F344 rats. In: Carcinogenic and Mutagenic Effects of Diesel Engine Exhaust. Eds. N Ishinishi, A Koizumi, RO, McClellan, and W Stöber, pp. 329–348. New York: Elsevier.
Ives JC, Buffler PA, Selwyn BJ, Hardy RJ, Decker M. 1988. Lung-cancer mortality among women employed in high-risk industries and occupations in Harris County, Texas, 1977–1980. Am J Epidemiol 127:65–74.
Iwai K, Udagawa T, Yamagishi M, Yamada H. 1986. Long-term inhalation studies of diesel exhaust on F344 SPF rats. Incidence of lung-cancer and lymphoma. In: Carcinogenic and Mutagenic Effects of Diesel Engine Exhaust. Eds. N Ishinishi, A Koizumi, RO McClellan, W Stöber, pp. 349–360. Amsterdam: Elsevier.
Jaberaboansari A, Dunn WC, Preston RJ, Mitra S, Waters LC. 1991. Mutations induced by ionizing radiation in a plasmid replicated in human cells. II. Sequence analysis of alpha-particle-induced point mutations. Radiat Res 127(2):202–210.
Jablon S, Tachikawa K, Belsky JL, Steer A. 1971. Cancer in Japanese exposed as children to atomic bombs. Lancet 1(7706):927–932.
Jacobi W. 1994. The History of the Radon Problem in Mines and Homes. Protection Against Radon-222 at Home and at Work. Pergamon Press; London, England.
Jacobi W and Eisfeld K. 1980. Dose to Tissues and Effective Dose Equivalent by Inhalation of Radon-222, Radon-220 and Their Short-lived Daughters, GSF Report S-626. Gesellschaft für Strahlen-und Umweltforschung, Munich-Neuherberg, West Germany.
Jacobi W, Henrichs K, Barclay D. 1992. Verura schungswarhscheinlichkeit von Lungenkrebs durch die berufliche Strahlenexposition von Uran-Bergarbeitern der WISMUT AG. GSF Research Center for Environment and Health, Neugerberg, Germany. GSF-Report S-14/92.
James AC. 1988. Lung dosimetry. Pp. 259–309 in Radon and Its Decay Products in Indoor Air, Nazaroff WW and Nero AV, eds. New York: Wiley Interscience.
James AC. 1992. Dosimetry of radon and thoron exposures: Implications for risks from indoor exposure. Pp. 167–198 in Indoor Radon and Lung-Cancer: Reality or Myth, Cross FT, ed. Columbus: Battelle Press.
James AC. 1994. Dosimetry of inhaled radon and thoron progeny. Pp. 161–180 in Integrally Radiation Dosimetry, Health Physics Society 1994 Summer School, Raabe OG, ed. Madison: Medical Physics Publishing.
James AC, Fisher DR, Hui TE, Cross FT, Durham JS, Gehr P, Egan MJ, Nixon W, Swift DL, Hopke PK. 1991. Dosimetry of Radon Progeny. In: Pacific Northwest Laboratory Annual Report for 1990 to the DOE Office of Energy Research, Pt. 1, pp 55–63. PNL-7600, Pacific Northwest Laboratory, Richland, Washington.
Jarvis NS, Birchall A, James AC, Bailey MR, Dorrian MD. 1993. LUDEP 1.0 Personal Computer Program for Calculating Internal Doses Using the New ICRP Respiratory Tract Model, NPRB-SR264, National Radiological Protection Board, Chilton, Didcot Oxon, England.
Jenner TJ, de Lara CM, O'Neill P, Stevens DL. 1993. Induction and rejoining of DNA double strand breaks in V79-4 mammalian cells following gamma-and alpha-irradiation. Int J Radiat Biol 64:265–273.
Jin Y, Yie TA, Carothers AM. 1995. Non-random deletions at the dihydrofolate reductase locus of Chinese hamster ovary cells induced by a-particles simulating radon. Carcinogenesis 16(8):1981–1991.
Johnson JH. 1988. Automotive emissions. In Air Pollution, The Automobile, and Public Health, Watson AY, Bates RR, Kennedy D, eds. Washington, DC: National Academy Press. pp. 39–75.
Johnson NF. 1995. Radiobiology of lung target cells. Radiat Prot Dosimet 60:327–330.
Johnson NF and Hubbs AF. 1990. Epithelial progenitor cells in the rat trachea. Am J Respir Cell Molec Biol 3:579–585
Johnson NF and Newton GJ. 1994. Estimation of the dose of radon progeny to the peripheral lung and the effect of exposure to radon progeny on the alveolar macrophage. Radiat Res 139:163–169.
Jones GDD, Milligan JR, Ward JF, Calabro-Jones PM, Aguilera JA. 1993. Yield of strand breaks as a function of scavenger concentration and LET for SV40 irradiated with He ions. Radiat Res 136:196–196.
Jostes RF, Hui TE, Cross FT. 1993. Use of the single-cell gel technique to support hit probability calculations in mammalian cells exposed to radon and radon progeny. Health Phys 64:675–679.
Jostes RF, Fleck EW, Morgan TL, Stiegler GL Cross FT. 1994. Southern blot and polymerase chain reaction exon analyses of HPRT-mutations induced by radon and radon progeny. Radiat Res 137:371–379.
Jostes RF. 1996. Genetic, cytogenetic and carcinogenic effects of radon: A review. Mutat Res 340:125–139.
Kadhim MA, Macdonald DA, Goodhead DT, Lorimore SA, Marsden SJ, Wright EG. 1992. Transmission of chromosomal instability after plutonium a-particle irradiation. Nature 355:738–740.
Kadhim MA, Lorimore SA, Hepburn MD, Goodhead DT, Buckle VJ, Wright EG. 1994. Alpha-particle-induced chromosomal instability in human bone marrow cells. Lancet 344:987–988.
Kadhim MA, Lorimore SA, Townsend KM, Goodhead DT, Buckle VJ, Wright EG. 1995. Radiation-induced genomic instability: delayed cytogenetic aberrations and apoptosis in primary human bone marrow cells. Int J Radiat Biol 67:287–293.
Kaldor JM, Day NE, Bell J, Clarke EA, Langmark F, Karjalainen S, Band P, Pedersen D, Choi W, Blair V, Henryamar M, Prior P, Assouline D, Pompekirn V, Cartwright RA, Koch M, Arslan A, Fraser P, Sutcliffe SB, Host H, Hakama M, Stovall M. 1992. Lung-cancer following Hodgkin's Disease: A case-control study. Int J Cancer 52:677–681.
Kasten MB, Onyuuekwere O, Sidransky D, Vogelstein B, Craig R. 1991. Participation of p53 protein in the cellular response to DNA damage. Cancer Res 51:6304–6311.
Kaur GP and Athwal RS. 1989. Complementation of a DNA repair defect in xeroderma pigmentosum cells by transfer of human chromosome 9. Proc Natl Acad Sci USA 86:8872–8876.
Keenan KP, Wilson TS, McDowell EM. 1983. Regeneration of hamster tracheal epithelium after mechanical injury. V. Histochemical immunocytochemical and ultrastructural studies. Vircros Arch [B] 43:213–240.
Kelly G, Stegelmeir BL, Hahn FF, 1995. p53 alterations in plutonium-induced F344 rat lung tumors. Radiat Res 142:263–269.
Kemp CJ, Wheldon T, Balmain A. 1994. p53 deficient mice are extremely susceptible to radiation-induced tumorigenesis. Nat Genet 8:66–69.
Kendall GM, Miles JCH, Cliff KD, Green BMR, Muirhead CR, Dixon DW, Lomas PR, Goodridge SM. 1994. Exposure to radon in UK dwellings. Publication No. NRPB-R272, National Radiological Protection Board, Chilton, Didcot, Oxfordshire, UK.
Kennedy AR and Little JB. 1984. Evidence that a second event in X-ray induced oncogenic transformation in vitro occurs during cellular proliferation. Radiat Res 99:228–248.
Khan MA, Cross FT, Jostes RF, Hui E, Morris JE, Brooks AL. 1994. Micronuclei induced by radon and its progeny in deep-lung fibroblasts of rats in vivo and in vitro. Radiat Res 139:53–59.
Khan MA, Cross FT, Buschbom RL, Brooks AL. 1995. Inhaled radon-induced genotoxicity in Wistar rat, Syrian hamster, and Chinese hamster deep-lung fibroblasts in vivo. Mutat Res 334:131–137.
Kihara M, Kihara M, Noda K. 1995. Risk of smoking for squamous and small cell carcinomas of the lung modulated by combinations of CYP1A1 and GSTM1 gene polymorphisms in a Japanese population. Carcinogenesis 16:2331–2336.
Kilburn KH. 1977. Clearance mechanisms in the respiratory tract. In Handbook of Physiology, Section 9, Reactions to Environmental Agents, Lee DHK, Falk HL, and Murphy SD, eds. Bethesda: American Physiological Society.
Kimbell JS, Gross EA, Joyner DR, Godo MN, Morgan KT. 1993. Application of computational fluid dynamics to regional dosimetry of inhaled chemicals in the upper respiratory tract of the rat. Tox Appl Pharm 121:253–263.
Klotz JB, Schoenberg JB, Wilcox HB. 1993. Relationship among short-and long-term radon measurements within dwellings: Influence of radon concentrations. Health Phys 65(4):367–374.
Knudson AG. 1971. Mutation and cancer: Statistical study of retinoblastoma. Proc, Natl Acad Sci USA 68:820–823.
Knutson EO and George AC. 1992. Reanalysis of data on particle size dsitributions of radon progeny in uranium mines. Pp. 149–164 in Indoor Radon and Lung-Cancer: Reality or Myth, Cross FT, ed. Columbus: Battelle Press.
Knutson EO, George AC, Knuth RH, Koh BR. 1984. Measurements of radon daughter particle size. Radiat Prot Dosimet 7:121–125.
Kodell RL, Krewski D, Zielinski JM. 1991. Additive and multiplicative relative risks in the two-stage clonal expansion model of carcinogenesis. Risk Anal 11:483–490.
Kopecky KJ, Nakashima E, Yamarnato T, Kato H. 1988. Lung-cancer, radiation, and smoking among A-bomb survivors, Hiroshima and Nagasaki. Radiation Effects Research Foundation. 13–86. p.1 RERF Technical Report Series.
Koskela RS, Klockars M, Jarvinen E, Rossi A, Kolari PJ. 1990. Cancer mortality of granite workers 1940–1985. In Simonato L, Fletcher AC, Saracci R, Thomas TL (eds): "Occupational Exposure to Silica and Cancer Risk." IARC Sci Publ no. 97:105–111. Lyon, France: IARC.
Kotin P, Falk HL, Thomas M. 1955. Aromatic hydrocarbons. III. Presence in the particulate phase of diesel-engine exhaust extracts. Arch Ind Health 11:113–120.
Krewski D, Goddard MJ, Zielinski JM. 1992. Dose-response realtionships in carcinogenesis. In: Mechanisms of Carcinogenesis in Risk Identification (H.Vainio, P.N. Magee, D.B. McGregor & A.J. McMichael, eds). IARC Scientific Publications No. 116, International Agency for Research on Cancer, Lyon, pp. 579–599.
Krolewski B, Little JB. 1989. Molecular analysis of DNA isolated from the different stages of X-ray-induced transformation. Vitro Mol Carcinog 2:27–33.
Kronenberg A and Little JB. 1989. Molecular characterization of thymidine kinase mutants of human cells induced by densely ionising radiation. Mutat Res 211:215–224.
Kronenberg A, Gauny S, Criddle K, Vannais D, Ueno A, Kraemer S, Waldren CA. 1995. Heavy ion mutagenesis: linear energy transfer effects and genetic linkage. Radiat Environ Biophys 34:73–78.
Kunz E, Sevc J, Placek V. 1978. Lung-cancer mortality in uranium miners. Health Phys 35:379–580.
Kunz E, Sevc J, Placek V, Horacek. 1979. Lung-cancer in man in relation to different time distributions of radiation exposure. Health Phys 36:699–706.
Kurosu K, Yumoto N, Mikata A, Taniguchi M, Kuriyama T. 1996. Monoclonality of B-cell lineage in primary pulmonary lymphoma demonstrated by immunoglobulin heavy chain gene sequence analysis of histologically non-definitive transbronchial biopsy specimens. J Pathol 178:316–22.
Kusiak RA, Springer J, Ritchie AC, Muller J. 1991. Carcinoma of the lung in Ontario gold miners: possible aetiological factors. Br J Ind Med 48:808–817.
Kusiak RA, Ritchie AC, Muller J, Springer J. 1993. Mortality for lung-cancer in Ontario uranium miners. Br J Ind Med 50:920–928.
Kysela BP, Arrand JE, Michael BD. 1993. Relative contributions of levels of initial damage and repair of double-strand breaks to the ionizing radiation-sensitive phenotype of the Chinese hamster cell mutant, XR-V15B. Part II. Neutrons. Int J Radiat Biol 64:531–538.
L'Abbe KAL, Howe GR, Burch JF, Miller AB, Abbatt J, Band P, Choi W, Du J, Feather J, Gallagher R, Hill G, Matthews B. 1991. Radon exposure, cigarette smoking, and other mining experience in the Beaverlodge uranium miners cohort. Health Phys 60:489–495.
Laird NM and Mosteller F. 1990. Some statistical methods for combining experimental results. International J Tech Asses in Hlth Care 6:5–30.
Land CE, Shimosato Y, Saccomanno G, Tokuoka S, Auerbach O, Tateishi R, Greenberg SD, Nambu S, Carter D, Akiba S, Keehn R, Madigan P, Mason TJ, Tokunaga M. 1993. Radiation-associated lung-cancer: A comparison of the histology of lung-cancers in uranium miners and survivors of the atomic bombings of Hiroshima and Nagasaki. Radiat Res 134:234–243.
Land H, Parada LF, Weinberg RA. 1983. Tumorigenic conversion of primary embryo fibroblasts requires at least two cooperating oncogenes. Nature 304:596–602.
Last JM. 1983. A Dictionary of Epidemiology. Second Edition. New York: Oxford University Press.
Lauer GR, Gang QT, Lubin JH, Jun-Yao L, Kan CS, Xiang YS, Jian CZ, Yi H, WanDe G, Blot WJ. 1993. Skeletal lead 210Pb levels in lung-cancer among radon-exposed tin miners in southern China. Health Phys 64:253–259.
Lavin MF, Bennett I, Ramsay J, Gardiner RA, Seymour GJ, Farrell A, Walsh M. 1994. Identification of a potentially radiosensitive subgroup among patients with breast cancer. J Natl Cancer Inst 86:1627–1634.
Lee JM, Abramson JLA, Kandel R, Donehower A, Bernstein A. 1994. Susceptibility to radiation-carcinogenesis and accumulation of chromosomal breakage in p53 deficient mice. Oncogene 9:3731–3736.
Lee WH, Bookstein R, Hong, Young LH, Shew, Lee EY-HP. 1987. Human retinoblastoma susceptibility gene: Cloning identification and sequence. Science 235:1394–1399.
Lee-Feldstein A. 1983: Arsenic and respiratory cancer in man: Follow-up of an occupational study. In Arsenic: Industrial, Biomedical Environmental Perspectives. Van Nostrand Reinhold Co., New York. pp 245–254.
Leenhouts HP, Chadwick KH. 1994. A two-mutation model of radiation carcinogenesis: application to lung tumors in rodents and implications for risk evaluation. J Radiol Prot 14:115–130.
Lees REM, Steele R, Roberts JH. 1987. A case-control study of lung-cancer relative to domestic radon exposure. Int J Epidemiol 16:7–12.
Létourneau EG, Mao Y, McGregor RG, Semenciw R, Smith MH, Wigle DT. 1983. Lung-cancer mortality and indoor radon concentrations in 18 Canadian cities. Epidemiology applied to health physics. Proceedings of a conference; 1983 Jan 10; Albuquerque, NM. Pp 470–483.
Létourneau EG, Krewski D, Choi NW, Goddard MJ, McGregor RG, Zielinski JM, Du J. 1994. Case-control study of residential radon and lung-cancer in Winnipeg, Manitoba, Canada. Am J Epidemiol 140:310–322.
Leupker RV and, Smith ML. 1978. Mortality in unionized truck drivers. JOM 20:677–682.
Levin ML. 1953. The occurrence of lung-cancer in man. Acta Unio International Contra Cancrum 9:531–541.
Li CS and Hopke PK. 1991. Characterization of radon decay products in a domestic environment. Indoor Air 1:539–561.
Li CS and Hopke PK. 1991. The efficacy of air cleaners in controlling indoor radon decay products. Health Phys 61:785–797.
Li CS and Hopke PK. 1993. Initial size and distributions and hygroscopicity of indoor combustion aerosol particles. Aerosol Sci Tech 19:305–316.
Li CS and Hopke PK. 1994. Hygroscopic growth of consumer spray products. Aerosol Sci 25:1342–1351.
Lippmann, M., D.B. Yeates, and R.E. Albert. 1980. Deposition, retention, and clearance of inhaled particles. Br J Ind Med 37:337.
Little JB. 1968. Delayed initiation of DNA synthesis in irradiated human diploid cells. Nature 218:1064–1065.
Little MP, Hawkins MM, Charles MW, Hildreth NG. 1992. Fitting the Armitage-Doll model to radiation-exposed cohorts and implications for population cancer risks. Radiat Res 132:207–221 (see also erratum in Radiat Res 137:124–128, 1994).
Little MP. 1995. Are two mutations sufficient to cause cancer? Some generalizations of the two-mutation model of carcinogenesis of Moolgavkar, Venzon, and Knudson, and of the multistage model of Armitage and Doll. Biometrics 51:1278–91.
Liu BYH and Pui DYH. 1975. On the performance of the electrical aerosol analyzer. J Aerosol Sci 6:249–264.
Lloyd DC, Edwards AA, Prosser JS, Bolton D, Corp MJ. 1984. Chromosome aberrations induced in human lymphocytes by D-T neutrons. Radiat Res 98:561–578.
Lloyd EL and Henning CB. 1981. Morphology of cells malignantly transformed by alpha particle irradiation. Scanning Electron Microscopy IV:87–92.
Lloyd EL, Gemmell MA, Henning CB, Zabransky BJ. 1979. Transformation of mammalian cells by alpha particles. Int J Radiat Biol 36:467–478.
Lo YM, Darby S, Noakes L, Whitley E, Silcocks PBS, Fleming KA, Bell JI. 1995. Screening for codon p53 mutation in lung-cancer associated with domestic radon exposure. Lancet 345:60.
Löbrich M, Rydberg B, Cooper PK. 1994. DNA double-strand breaks induced by high-energy neon and iron ions in human fibroblasts. II. Probing individual NotI fragments by hybridization. Radiat Res 139:143–151.
Löbrich M, Cooper PK, Rydberg B. 1996. Non-random distribution of DNA double strand breaks induced by particle irradiation. (submitted for publication).
Lorenz E. 1944. Radioactivity and lung-cancer: A critical review of lung-cancer in the miners of Schneeberg and Joachimsthal. J Natl Cancer Inst 5:1–15.
Lorimore SA, Goodhead DT, Wright EG. 1993. Inactivation of haemopoietic stem cells by slow a-particles. Int J Radiat Biol 63:655–660.
Lorimore SA, Goodhead DT, Wright EG. 1995. The effect of p53 status on the radiosensitivity of haemopoietic stem cells. Cell Death and Differentiation 2:233–234.
Loucas BD and Geard CR. 1994. Initial damage in human interphase chromosomes from alpha particles with linear energy transfers relevant to radon exposure. Radiat Res 139:9–14.
Lovett DB. 1969. Track etch detectors for alpha exposure estimates. Health Phys 16:623–628.
Lubin JH. 1994. Invited commentary: Lung-cancer and exposure to radon. Am J Epidemiol 140:323–332.
Lubin JH and Boice JD, Jr. 1989. Estimating radon-induced lung-cancer in the U.S. Health Phys 7:417–427.
Lubin JH and Boice JD, Jr. 1997. Lung-cancer risk from residential radon: meta-analysis of eight epidemiologic studies. J Natl Cancer Inst 89:49–57.
Lubin JH and Gaffey W. 1988. Relative risk model for assessing the joint effects of multiple factors. Am J Ind Med 13:149–167.
Lubin JH and Steindorf K. 1995. Cigarette use and the estimation of radon-attributable lung-cancer in the U.S. Radiat Res 141:79–85.
Lubin JH, Blot WJ, Berrino F, Gillis CR, Kunze M, Shamahl D, Visco G. 1984. Patterns of lung-cancer risk according to type of cigarette smoked. Int J Cancer 33:569–576.
Lubin JH, Samet JM, Weinberg C. 1990a. Design issues in epidemiologic studies of indoor exposure to Rn and risk of lung-cancer. Health Phys 59:807–817.
Lubin JH, You-Lin Qiao, Taylor PR, Schatzkin A, Bao-Lin Mayo, Jian-Yu Rao, Xiang-Zhen Xuan, Jun-Yao Li. 1990b. Quantitative evaluation of the radon and lung-cancer association in case-control study of Chinese tin miners. Cancer Res 50:174–180.
Lubin JH, Boice Jr. JD, Edling C, Hornung RW, Howe G, Kunz E, Kusiak RA, Morrison HI, Radford EP, Samet JM, Tirmarche M, Woodward A, Yao SX, Pierce DA. 1994a. Radon and Lung-cancer Risk: A Joint Analysis of 11 Underground Miners Studies. National Institutes of Health, National Cancer Institute. NIH Publication No. 94-3644. Washington, D.C. U.S. Department of Health and Human Services.
Lubin JH, Liang Z, Hrubec Z, Pershagen G, Schoenberg JB, Blot WJ, Klotz JB, Xu Z-Y, Boice JD Jr. 1994b. Radon exposure in residences and lung-cancer among women: Combined analysis of three studies. Cancer Causes Control 5:114–128.
Lubin JH, Boice JD, Jr., Edling C, Hornung RW, Howe G, Kunz E, Kusiak RA, Morrison HI, Radford EP, Samet JM, Tirmarche M, Woodward A, Yao SX. 1995a. Radon-exposed underground miners and inverse exposure-rate (protraction enhancement) effects. Health Phys 69:494–500.
Lubin JH, Boice JD, Jr., Edling C, Hornung RW, Howe GR, Kunz E, Kusiak RA, Morrison HI, Radford EP, Samet JM, Tirmarche M, Woodward A, Yao SX, Pierce DA. 1995b. Lung-cancer in radon-exposed miners and estimation of risk from indoor exposure. J Natl Cancer Inst 87:817–27.
Lubin JH, Boice JD Jr, Samet JM. 1995c. Errors in exposure assessment, statistical power, and the interpretation of residential radon studies. Radiat Res 144:329–341.
Lubin JH, Tomásek L, Edling C, Hornung RW, Howe G, Kunz E, Kusiak RA, Morrison HI, Radford EP, Samet JM, Timarche M, Woodward A, Yao, SX. 1997. Estimating lung-cancer mortality from residential radon using data for low exposures in miners. Radiat Res 147(2):126–134.
Lucas JN, Awa A, Straume T, Poggensee M, Kodama Y, Nakano M, Ohtaki K, Weier HU, Pinkel D, Gray D, Littlefield G. 1992. Rapid translocation frequency analysis in humans decades after exposure to ionizing radiation. Int J Radiat Biol 62:53–63.
Lucie NP. 1989. Radon exposure and leukemia. Lancet 2(8654):99–100.
Lucke-Huhle C, Comper W, Hieber L, Pech M. 1982. Comparative study of G2 delay and survival after 241Americium-alpha and 60cobalt-gamma irradiation. Radiat Environ Biophys 20:171–185.
Ludwig P and Lorenser S. 1924. Untersuchungen der grubenluft in den Schneeberger gruben auf den gehalt an radiumemanation. Z Physik 22:178–185.
Luebeck EG, Curtis SB, Cross FT, Moolgavkar SH. 1996. Two-stage model of radon-induced malignant lung tumors in rats: Effects of cell killing. Radiat Res 145(2):163–173.
Lundin FE, Wagoner JK, Archer VE. 1971. Radon Daughter Exposure and Respiratory Cancer Quantitative and Temporal Aspects. NIOSH-NIEHS Joint Monograph No. 1. U.S. Department of Health, Education and Welfare.
Lutze LH, Winegar RA, Jostes R, Cross FT, Cleaver JE. 1992. Radon-induced deletions in human cells: Role of nonhomologous strand rejoining. Cancer Res 52:5126–5129.
Lutze LH, Cleaver JE, Winegar RA. 1994. Factors affecting the frequency, size, and location of ionizing-radiation-induced deletions in human cells. In: Molecular Mechanisms in Radiation Mutagenesis and Carcinogenesis (Eds. K.H. Chadwick, R. Cox, H.P. Leenhouts and J. Thacker). European Commission EUR 15294:41–46.
Lyon JL, Gardner JW, West DW. 1980. Cancer in Utah: Risk by religion and place of residence. J Natl Cancer Inst 65:1063–1071.
Mabuchi K, Lilienfeld AM, Snell LM. 1985. Cancer and occupational exposure to arsenic: A study of pesticide workers. Prev Med 9:51–77.
Mabuchi K, Land CE, Akiba S. 1992. Radiation, smoking and lung-cancer. RERF Update 7–8.
Macklin MT. 1942. Has a real increase in lung-cancer been proved? Am J Ind Med 17:308–324.
Magnus K, Engeland A, Green BM, Haldorsen T, Muirhead CR, Strand T. 1994. Residential radon exposure and lung-cancer - An epidemiological study of Norwegian municipalities. Int J Cancer 58:1–7.
Mahaffey JA, Parkhurst MA, James AC, Cross FT, Alavanja MCR, Boice JD Jr., Ezrine S, Henderson P, Brownson RC. 1993. Estimating past exposure to indoor radon from household glass. Health Phys 64:381–391.
Maher EF and Laird NM. 1985. EM algorithm reconstruction of particle size distributions from diffusion battery data. J Aerosol Sci 16:557–570.
Maillie HD, Simon W, Greenspan BS, Watts RJ, Quinn BR. 1994. The influence of life table corrections for smokers and nonsmokers on the health effects of Radon using the BEIR IV method. Health Phys 686:615–620.
Maity A, McKenna WG, Muschel RJ. 1994 The molecular basis for cell cycle delays following ionizing radiation: A review. Radiother Oncol 31:1–13.
Mäkeläinen I, Voutilainen A, Castrén O. 1987. Uppskattning av radonhalten i småhus på basen av lokation och byggnadsdata. Nordiska Sällskapet för Strålskydd, mötet 26–28 Aug., Mariehamn.
Mapel DW, Samet JM, Coultas DB. 1996. Corticosteroids and the treatment of idiopathic pulmonary fibrosis. Past, present, and future. Chest 110(4):1058–1067.
Marchaux G, Morlier J-P, Morin M, Chameaud J, Lafuma J, Masse R. 1994. Carcinogenic and cocarcinogenic effects of radon and radon daughters in rats. Environ Health Perspect 102:64–73.
Marcinowski F, Lucas RM, Yeager WM. 1994. National and regional distributions of airborne radon concentrations in U.S. homes. Health Phys 66:699–706.
Marder BA, Morgan WF. 1993. Delayed chromosomal instability induced by DNA damage. Mol Cell Biol 13:6667–6677.
Martins MB, Sabatier L, Ricoul M, Pinton A, Dutrillaux B. 1993. Specific chromosome instability induced by heavy ions: a step towards transformation of human fibroblasts. Mutat Res 285:229–237.
Martland HS. 1931. Occurrence of malignancy in radioactive persons. Am J Cancer 15:2435–2516.
Martz DE, Falco RJ, Langner Jr. GH. 1990. Time-averaged exposures to 220Rn and 222Rn progeny in Colorado homes. Health Phys 58:705–713.
Mason TJ. 1994. The descriptive epidemiology of lung-cancer. Samet JM, Ed. Epidemiology of Lung-Cancer. Marcel Dekker, Inc. New York, New York.
Masse R and Cross FT. 1989. Risk considerations related to lung modeling. Health Phys 57(Suppl 1):283–9.
Mauderly JL. 1992. Diesel exhaust. Pp. 119–162 in Environmental Toxicants-Human Exposures and Their Health Effects, Lippmann M, ed. New York: Van Nostrand Reinhold.
Mauderly JL. 1993. Toxicological approaches to complex mixtures. Environ Health Perspect 101:155–165.
Mauderly JL. 1994. Contribution of inhalation bioassays to the assessment of human health risks from solid airborne particles. Pp. 355–365 in Toxic and Carcinogenic Effects of Solid Particles in the Respiratory Tract, Mohr U, Dungworth DL, Mauderly JL, and Oberdörster G, eds. Washington: International Life Sciences Institute Press.
Mauderly JL, Jones RK, Griffith WC, Henderson RF, McClellan RO. 1987. Diesel exhaust is a pulmonary carcinogen in rats exposed chronically by inhalation. Fundam Appl Toxicol 9:208–221.
Mauderly JL, Snipes MB, Barr EB, Belinsky SA, Bond JA, Brooks AL, Chang IY, Cheng YS, Gillett NA, Griffith WC, Henderson RG, Mitchell CF, Nikula KJ, Thomassen DG. 1994. Pulmonary toxicity of inhaled diesel exhaust and carbon black in chronically exposed rats. Part I: Neoplastic and nonneoplastic lung lesions. Res Rep Health Eff Inst 68:1–75.
Mauderly JL, Banas DA, Griffith WC, Hahn FF, Henderson RF, McClellan RO. 1996. Diesel exhaust is not a pulmonary carcinogen in CD-1 mice exposed under conditions carcinogenic to F344 rats. Fundam Appl Toxicol (in press).
May KR. 1945. The cascade impactor: An instrument for sampling coarse aerosols. J Sci Instr 22:187–195.
Mayer SA. 1973. Present regulations on control of radiation in uranium mines. U.S. Atomic Energy Commission.
Mazumdar S, Redmond CK, Enterline PE, Marsh GM, Costantino JP, Zhou SYJ, Patwardham RN. 1989. Multistage modeling of lung-cancer mortality among arsenic-exposed copper-smelter workers. Risk Anal 9:551–563.
McClellan RO. 1987. Health effects of exposure to diesel exhaust particles. Am Rev Pharmacol Toxicol 27:279–300.
McClellan RO. 1990. Particle overload in the lung: Approaches to improving our knowledge. J Aerosol Med (Suppl.) 3:S197–S207.
McClellan RO. 1996. Lung-cancer in rats from prolonged exposure to high concentrations of carbonaceous particles: Implications for human risk assessment. Inhal Toxicol 8(Suppl):193–226.
McDonald JW, Taylor JA, Watson MA, Saccomanno G, Devereux TR. 1995. p53 and K-ras in radon-associated lung adenocarcinoma. Cancer Epidemiol Biomarkers Prev 4(7):791–793.
McDowell EM and Trump BF. 1984. Histogenesis of preneoplastic lesions in tracheobronchial epithelium. Surv Synth Pathol Res 2:235–279.
McLaughlin JK, Chen J-Q, Dosemeci M, Chen R-A, Rexing SH, Wu Z, Hearl FJ, McCawley MA, Blot WJ. 1992. A nested case-control study of lung-cancer among silica exposed workers in China. Br J Ind Med 49:167–171.
Meijers JMM, Swaen GMH, Volovics A, Slangen JJM, Van Vliet K. 1990. Silica exposure and lung-cancer in ceramic workers: A case-control study. Int J Epidemiol 19:19–25.
Melhorn J, Selig R, Pabst R. 1992. Zum silikosegeschehen i3m Uranerzbergbau der DDR. In Kreutz R, Piekarski C (eds): Arbeitsmedizinische Aspekte...Stuttgart: Gentner Verlag 415–419.
Menck HR and Henderson BE. 1976. Occupational differences in rates of lung-cancer. JOM 18:797–801.
Mercer TT. 1981. Production of therapeutic aerosols; principles and techniques. Chest 80(Suppl.): 181–189.
Merlo F, Doria M, Fontana L, Ceppi M, Chesi E, Santi L. 1990. Mortality from specific causes among silicotic subjects: A historical prospective study. In Simonato L, Fletcher AC, Saracci R, Thomas TL (eds): "Occupational Exposure to Silica and Cancer Risk." IAARC Sci Publ no. 97:105–111. Lyon, France: IARC.
Metting NF and Little JB. 1995. Transient failure to dephosphorylate the cdc2-cyclin B1 complex accompanies radiation-induced G2-phase arrest in HeLa cells. Radiat Res 143:286–292.
Metting NF, Palayoor ST, Macklis RM, Atcher RW, Liber HL, Little JB. 1992. Induction of mutations by bismuth-212 a-particles at two genetic loci in human B-lymphoblasts. Radiat Res 132:339–345.
Mifune M, Sobue T, Arimoto HG, Komoto Y, Kundo S, Tanooka. H. 1992. Cancer mortality survey in a spa area (Misasa, Japan) with a high radon background. Japan J of Cancer Res 83:1–5.
Miki Y, Swensen J, Shattuck-Eidens D, Futreal PA, Larshman KI, Tavtigian S, Liu Q, Cochran C, Bennet LM, Ding W, Bell R, Rosenthral J, Hussey C, Tran T, McClure M, Frye C, Hattier T, Phelps R, Haugen-Strano A, Katcher H, Yakumo K, Gholami Z, Shaffer D, Stone S, Bayer S, Wray C , Bogden R, Dayananth P, Ward J, Tonin P, Narod S, Bristow PK, Norris FH, Helvering L, Morrison P, Rosteck P, Lai M, Barrett JC, Lewis C, Neuhausen S, Cannon-Albright L Goldgar D, Wiseman R, Kamb A, Skolnick MH. 1994. A strong candidate for the breast and ovarian cancer susceptibility gene BRCA1. Science 266:66–71.
Miller RC, Brenner DJ, Randers-Person G, Marino SA, Hall EJ. 1990. The effects of temporal distribution of dose on oncogenic transformation by neutrons and charged particles of intermediate LET. Radiat Res 124:S62–S68.
Miller RC, Geard CR, Geard MJ, Hall EH. 1992. Cell-cycle dependent radiation-induced transformation of C3H10T1/2 cells. Radiat Res 130:129–133.
Miller RC, Randers-Pehrson G, Hieber L, Marino SA, Richards M, Hall EJ. 1993. The inverse dose rate effect for oncogenic transformation by charged particles is LET dependent. Radiat Res 133:259–263.
Miller RC, Marino SA, Brenner DJ, Martin SG, Richards M, Randers-Pehrson G, Hall EJ. 1995. The biological effectiveness of radon-progeny alpha particles II Neoplastic transformation as a function of LET. Radiat Res 142:54–60.
Mole RH. 1990: Radon and leukaemia (Letter). Lancet 335:1336.
Moolgavkar SH. 1993. Cell proliferation and carcinogenesis models: general principles with illustrations from the rodent liver system. Environ Health Perspect 101(Suppl 5):91–94.
Moolgavkar SH. 1994. Biological models of carcinogenesis and quantitative cancer risk assessment. Risk Anal 14:879–882.
Moolgavkar SH and Luebeck EG. 1990. Two-event model for carcinogenesis: Biological, mathematical and statistical considerations. Risk Anal 10:323–341.
Moolgavkar SH and Leubeck EG. 1993. Two-mutation model for radiation carcinogenesis in humans and rodents. In New Frontiers in Cancer Causation. Proceedings of the Second International Conference on Theories of Carcinogenesis, Iversen OH, ed. Taylor & Francis.
Moolgavkar SH, Cross FT, Luebeck G, Dagle GE. 1990. A two-mutation model for radon-induced lung tumors in rats. Radiat Res 121:28–37.
Moolgavkar SH, Luebeck EG, Krewski D, Zielinski JM. 1993. Radon, cigarette smoke, and lung-cancer: A re-analysis of the Colorado plateau uranium miners' data. Epidemiology 4:204–217.
Morgan M, Henrion M, Small M. 1990. Uncertainty. New York: Cambridge University Press.
Morgan WF, Day JP, Kaplan MI, McGhee FM, Limoli CL. 1996. Genomic instability induced by ionizing radiation. Radiat Res 146:247–258.
Morgan WKC, Clague HW, Vinitski S. 1983. On paradigms, paradoxes, and particles. Lung 161:195.
Morgenstern H. 1995. Ecologic studies in epidemiology: Concepts, principles, and methods. Ann Rev Public Health 16:61–81.
Morken DA. 1973. The biological effects of radon on the lung. In: Nobel Gases, Edt. by R.E. Stanley, A.A. Moghissi, CONF-730915, U.S. Energy Development and Research Agency. National Environmental Research Center, Washington D.C., pp. 501–506.
Morlier J-P, Morin M, Monchaux G, Fritsch P, Chameaud J, Lafuma J, Masse R. 1994. Lung-cancer incidence after exposure of rats to low doses of radon: Influence of dose rate. Radiat Prot Dosimet 56:93–97.
Morrison HI, Semenciw RM, Mao Y, Wigle DT. 1988. Cancer mortality among a group of fluorspar miners exposed to radon progeny. Am J Epidemiol 128:1266–1275.
Morrow PE. 1988. Possible mechanisms to explain dust overloading of the lungs. Fundam Appl Toxicol 10:369–384.
Muirhead CR, Butland BK, Green BM, Draper GJ. 1991. Childhood leukemia and a natural radiation. Lancet 337:503–504.
Muller J, Wheeler WC, Gentleman JF, Suranji JF Kusick. R. 1983. Study of mortality of Ontario miners 1955-1957 Part 1. Toronto: Ontario Ministry of Labor.
Muller J, Wheeler WC, Gentleman JP, Suranyi G, Kusiak R. 1984. Study of mortality of Ontario miners. In: Proceedings of the International Conference on Occupational Radiation Safety in Mining, (Stocker, H., ed). Toronto, Canada: Canadian Nuclear Association, pp 335–343.
Murnane JP. 1995. Cell cycle regulation in response to DNA damage in mammalian cells: A historical perspective. Cancer and Metastasis Reviews 14:17–29.
Muscat JE, Wynder EL. 1995. Diesel exhaust, diesel fumes, and laryngeal cancer. Otolaryngol-Head Neck Surg 112:437–440.
Nagarkatti M, Nagarkatti PS, Brooks AL. 1996. Effect of radon on the immune system: Alterations in the cellularity and functions of T cells in lymphoid organs of mouse. J Toxicol Environ Health 47:535–545.
Nagasawa H and Little JB. 1992. Induction of sister chromatid exchanges by extremely low doses of alpha-particles. Cancer Res 52:6394–6396.
Nagasawa H, Robertson J, Little JB. 1990. Induction of chromosomal aberrations and sister chromatid exchanges by alpha particles in density-inhibited cultures of mouse 10T1/2 and 3T3 cells. Int J Radiat Biol 57:35–44.
NCRP (National Council of Radiation Protection and Measurements). 1984a. Exposures from the Uranium Series with Emphasis on Radon and Its Daughters. NCRP Report 77. Washington, DC: National Council on Radiation Protection and Measurements.
NCRP (National Council on Radiation Protection and Measurements). 1984b. Evaluation of occupational and environmental exposures to radon and radon daughters in the United States. NCRP Report 78. Washington, DC: National Council on Radiation Protection and Measurements.
NCRP (National Council on Radiation Protection and Measurements). 1988. Epidemiological studies of lung-cancer in underground miners. Proceedings of the Twenty-Fourth Annual Meeting of the National Council on Radiation Protection and Measurements. Bethesda, MD. Pp. 30–50.
NCRP (National Council on Radiation Protection and Measurements). 1990. The Relative Biological Effectiveness of Radiations of Different Quality. NCRP Report 104. Issued December 15, 1990. Bethesda, Maryland. National Council on Radiation Protection and Measurements.
NCRP (National Council on Radiation Protection and Measurements). 1991. Radon Exposure of the U.S. Population — Status of the Problem. Bethesda, Maryland: National Council on Radiation Protection and Measurements. p. 16.
NCRP (National Council on Radiation Protection and Measurements). 1993. Research Needs for Radiation Protection. NCRP Report 117. Washington, DC: National Council on Radiation Protection and Measurements.
NEA (Nuclear Energy Agency Group of Experts). 1983. Dosimetry Aspects of Exposure to Radon and Thoron Daughter Products. Paris: Organization for Economic Cooperation and Development.
NIOSH (National Institute for Occupational Safety and Health). 1987. Criteria for a Recommended Standard: Occupational Exposure to Radon Progeny in Underground Mines. Washington, DC. U.S. Government Printing Office.
NRC (National Research Council). 1980. Committee on the Biological Effects of Ionizing Radiations (BEIR III). The Effects on Populations of Exposure to Low Levels of Ionizing Radiation; Washington, D.C. National Academy Press.
NRC (National Research Council). 1988. Committee on the Biological Effects of Ionizing Radiations. Health risks of radon and other internally deposited alpha-emitters: BEIR IV; Washington, D.C. National Academy Press.
NRC (National Research Council). 1990. Committee on the Biological Effects of Ionozing Radiations. Health Effects of Exposure to Low Levels of Ionizing Radiation: BEIR V; Washington, D.C. National Academy Press.
NRC (National Research Council). 1991. Panel on Dosimetric Assumptions Affecting the Application of Radon Risk Estimates. Comparative Dosimetry of Radon in Mines and Homes . Washington, D.C. National Academy Press.
NRC (National Research Council). 1994a. Committee on Health Effects of Exposure to Radon (BEIR VI), and Commission on Life Sciences. Health Effects of Exposure to Radon: Time for Reassessment? Washington, D.C. National Academy Press.
NRC (National Research Council). 1994b. Committee on Risk Assessment of Hazardous Air Pollutants. Science and Judgment in Risk Assessment. National Academy Press, Washington DC.
Nauss KM, The HEI Diesel Working Group. 1995. Critical issues in assessing the carcinogenicity of diesel exhaust: A synthesis of current knowledge. Pp. 11–61 in Diesel Exhaust: A Critical Analysis of Emissions, Exposure, and Health Effects. Cambridge: Health Effects Institute.
Nelson JM, Brooks AL, Metting NF, Khan MA, Buschbom RL, Duncan A, Miick R, Braby LA. 1996. Clastogenic effects of defined numbers of 3.2 MeV alpha particles on individual CHO-K1 cells. Radiat Res 145:568–574.
Nero AV, Schwehr MB, Nazaroff WW, Revzan KL. 1986. Distribution of airborne radon-222 concentrations in U.S. homes. Science 234:992–997.
Nero AV, Nazaroff WW, Nero Jr AV, eds. 1988. Radon and its decay products. Pp. 1–53 in Indoor Air: An Overview. New York: John Wiley & Sons, Inc.
Nero AV, Gadgil AJ, Nazaroff WW, Revzan KL. 1990. Indoor radon and decay products: concentrations, causes, and control strategies. Report prepared for the U.S. Department of Energy, Office of Health and Environmental Research, Washington, D.C., November 1990. DOE/ER-0480P, NTIS, Springfield, VA.
Neuberger JS, Lynch CF, Kross BC, Field RW, Woolson RF. 1994. Residential radon exposure and lung-cancer: Evidence of an urban factor in Iowa. Health Phys 66:263–269.
Neuberger O. 1947. Arsenical cancer: A review. Br J Cancer 1: 192–251.
Neugat AI and Murray T. 1994. Increased risk of lung-cancer after breast cancer radiation therapy in cigarette smokers. Cancer 73:1615–20.
Newcomb EW, Steinberg JJ, Pellicer A. 1988. Ras oncogenes and phenotypic staging in N-methyl nitrosourea and g-irradiation-induced mymic lymphomas in C57BL/6J mice. Cancer Res 48:5514–5521.
Newman B, Austin MA, Lee M, King MC. 1988. Inheritance of human breast cancer: evidence for autosomal dominant transmission in high-risk families. Proc Natl Acad Sci USA 85(9):3044–3048.
Newman JA, Archer VE, Saccomanno G, Kuschner M, Auerbach O, Grondahl D, Wilson JC. 1976. Histologic types of bronchogenic carcinoma among members of copper mining and smelting communities. Ann NY Acad Sci 271:260–268.
Ng TP, Chan SL, Lee J. 1990. Mortality of a cohort of men in a silicosis register: Further evidence of an association with lung-cancer. Am J Ind Med 17:163–171.
Nikula KJ, Snipes MB, Barr EB, Griffith WC, Henderson RF, Mauderly JL. 1995. Comparative pulmonary toxicities and carcino-genicities of chronically inhaled exhaust and carbon black in F344 rats. Fundam Appl Toxicol 25:80–94.
Oakes M. 1990. On meta-analysis. Pp. 157–163 in Statistical Inference. Chestnut Hill, MA: Epidemiology Resources Inc.
Oberdörster G. 1996. Significance of particle parameters in the evaluation of exposure-dose-response relationships of inhaled particles. Inhal Toxicol 8(Suppl):73–89.
Ott MG, Holder BB, Gordon HL. 1974. Lung-cancer among pesticide workers exposed to inorganic arsenicals. Arch Environ Health 29:250–255.
Page S. 1993. EPA's strategy to reduce risk of radon. J Environ Health 56:27–36.
Palmer RF, Stuart BO, Filipy RE. 1973. Biological effects of daily inhalation of radon and its short-lived daughters in experimental animals. In: Nobel gases, Edt. by R.E. Stanley, A.A. Moghissi, CONF-730915. U.S. Energy Development and Research Agency, National Environmental Research Center, Washington D.C. Pp. 507–519.
Park MS, Hanks T, Jaberaboansari A, Chen DJ. 1995. Molecular analysis of gamma-ray-induced mutations at the hprt locus in primary human skin fibroblasts by multiplex polymerase chain reaction. Radiat Res 141(1):11–18.
Pathak S. 1990. Cytogenetic abnormalities in cancer: With special emphasis on tumor heterogeneity. Cancer and Metastases Reviews 8:299–318.
Pavia D. 1984. Lung mucociliary clearance. In Aerosols and the Lung: Clinical and Experimental Aspects, Clarke SW, and Pavia D, eds. London: Butterworths.
Peltomaki P, Aaltonen LA, Sistonen P, Pylkkanen L, Mecklin J-P, Jarvinen H, Green JS, Jass JR, Weber JL, Leach FS, Petersen GM, Hamilton SR, de la Chapelle A, Vogelstein B. 1993. Genetic mapping of a locus predisposing to human colorectal cancer. Science 260:810–812.
Percy C, Stanek E 3d, Gloeckler L. 1981. Accuracy of cancer death certificates and its effect on cancer mortality statistics. Am J Public Health 71(3):242–250.
Pershagen G, Damber L, Falk R. 1984. Exposure to radon in dwellings and lung-cancer: A pilot study. Pp. 29–34 in Indoor Air. Radon, Passive Smoking, Particulates and Housing Epidemiology, vol 2, Berglund B, Lindvall T, and Sundell J, eds. Stockholm Council for Building Res.
Pershagen G, Bergman F, Klominek J, Damber L, Wall S. 1987. Histological types of lung-cancer among smelter workers exposed to arsenic. Br J Ind Med 44:454–458.
Pershagen G, Liang Z-H, Hrubec Z, Svensson C, Boice JD Jr. 1992. Residential radon exposure and lung-cancer in Swedish women. Health Phys 63:179–186.
Pershagen G, Åkerblom G, Axelson O, Clavensjö B, Damber L, Desai G, Enflo A, Lagarde F, Mellander H, Svartengren M, Swedjermark GA. 1994. Residential radon exposure and lung-cancer in Sweden. N Engl J Med 330:159–164.
Petersen DD, Gonzalez FJ, Rapic V, Kozak CA, Lee JY, Jones JE, Nevert DW. 1989. Marked increases in hepatic NAD(P)H:oxidoreductase gene transcription and mRNA levels correlated with a mouse chromosome 7 deletion. Proc Natl Acad Sci USA 86(17):6699–6703.
Petersen GR, Gilbert ES, Buchanan JA, Stevens RG. 1990. A case-cohort study of lung-cancer, ionizing radiation, and tobacco smoking among males at the Hanford Site. Health Phys 58:3–11.
Petitti DB. 1994. Of babies and bathwater. Am J Epidemiol 140:779–782.
Peto J. 1990. Radon and the risks of cancer. Nature 345:389–390.
Peto R, Lopez AD, Boreham J, Thun M, Heath C Jr. 1992. Mortality from tobacco in developed countries: Indirect estimation from national vital statistics. Lancet 339:1268–1278.
Pfeifer AMA, Jones RT, Bowden PE, Mann D, Spillare E, Klien-Szanto AJP, Trump BF, Harris CC. 1991. Human bronchial epithelial cells transformed by c-raf-1 and c-myc protooncogenes induce multidifferentiated carcinomas in nude mice: A model for lung carcinogenesis. Cancer Res 51:3793–3801.
Piantadosi S. 1994. Invited commentary: Ecologic biases. Am J Epidemiol 139:761–764.
Piantadosi S, Byar D, Green S. 1988. The ecological fallacy. Am J Epidemiol 127:893–904.
Piao CQ, Hei TK. 1993. The biological effectiveness of radon daughter alpha particles I. Radon, cigarette smoke and oncogenic transformation. Carcinogenesis 14:497–501.
Piechowski JW, LeGac J, Brenot J, Nenot JC, Zettwoog P. 1981. Exposure to short-lived radon daughters: Comparison of individual and ambient monitoring in a French uranium mine. Radiation Hazards in Mining: control, measurement, and medical aspects. Golden Conference, Oct 4–9. M. Gomez, Editor. American Institute of Mining, Metallurgical and Petroleum Engineering, Inc. New York City, NY.
Pierce DA, Stram DO, Vaeth M. 1990. Allowing for random errors in radiation dose estimates for the atomic bomb survivor data. Rad Res 123:275–284.
Pirchan A, Sikl H. 1932. Cancer of the lung in the miners of Jachymov. Am J Cancer 16:681–722.
Porstendörfer J, Röbig G, Ahmed A. 1979. Experimental determination of the attachment coefficients of atoms and ions on monodisperse particles. J Aerosol Sci 10:21–28.
Prentice AG, Copplestone, JA. 1990. Radon and leukaemia (Letter). Lancet 335:1337.
Prentice RL, Yoshimoto Y, Mason MW. 1983. Relationship of cigarette smoking and radiation exposure to cancer mortality in Hiroshima and Nagasaki. J Natl Cancer Inst 70:611–622.
Preston DL, Lubin JH, Pierce DA, McConney ME. 1991. EPICURE: User's Guide, HiroSoft International. Corporation. 1463 E. Republican Ave. Suite 103, Seattle, WA 98112, USA.
Prise K. 1994. Use of radiation quality as a probe for DNA lesion complexity. Int J Radiat Biol 65:43–48.
Prise KM, Davies S, Michael BD. 1987. The relationship between radiation-induced DNA double-strand breaks and cell kill in hamster V79 fibroblasts irradiated with 250 kVp X-rays, 2.3 MeV neutrons or 238Pu a-particles. Int J Radiat Biol 52:893–902.
Probart CK. 1989. Issues related to radon in schools. J Sch Health 59(10):441–443.
Proctor RN. 1995. Cancer Wars. How Politics Shapes What We Know and Don't Know About Cancer. New York: Basic Books.
Puskin JS. 1992. An analysis of the uncertainties in estimates of radon-induced lung-cancer. Risk Anal 12:277–285.
Puskin JS, Nelson CB. 1989. EPA's perspective on risks from residential radon exposure. JAPCA 39:915–920.
Qiao YL, Taylor RP, Yao SX, Schatzkin A, Mao BL, Lubin JH, Rao JY, Li JY. 1989. The relation of radon exposure and tobacco use to lung-cancer among tin miners in Yunnan Province, China. Am J Ind Med 16:511–521.
Raabe OG. 1982. Deposition and clearance of inhaled aerosols. In Mechanisms in Respiratory Toxicology, Vol. I, Witschi H and Nettesheim P, eds. Boca Raton: CRC Press.
Radford EP and St. Clair Renard KG. 1984. Lung-cancer in Swedish iron miners exposed to low doses of radon daughters. N Engl J Med 310(23):1485–1494.
Rai SN and Krewski D. 1998. Uncertainty and variability analysis in multiplicatie risk models. Risk Anal 18:37–45.
Rai SN, Krewski D, Bartlett S. 1996. A general framework for the analysis of uncertainty and variability in risk assessment. Human and Ecological Risk Assessment 2(4):972–989.
Raju MR, Eisen Y, Carpenter S, Jarret K, Harvey WF. 1993. Radiobiology of alpha particles IV. Cell inactivation by alpha particles of energies 0.4-3.5 MeV. Radiat Res 133:289–96.
Ramamurthi M and Hopke PK. 1989. On improving the validity of wire screen unattached fraction daughter measurements. Health Phys 56:189–194.
Ramamurthi M and Hopke PK. 1991. An automated, semi-continuous system for measuring indoor radon progeny activity-weighted size distributions, dp: 0.5-500 nm. Aerosol Sci Technol 14:82–92.
Ramamurthi M., Strydom R, Hopke PK. 1990. Assessment of wire and tube penetration theories using a 218PoOx cluster aerosol. J Aerosol Sci 21:203–211.
Rannou A. 1987. Contribution a l'étude du risque lie a la presence du radon 220 et du radon 222 dans l'atmosphere das habitations. Rapport CEA-R-5378. Commissariat a l'Energie Atomic. Sarclay, France.
Rannou A, Mouden A, Renouard H, Kerlau G, Tymen G. 1988. An assessment of natural radiation exposure in granitic areas in the west of France. Radiat Prot Dosimet 24(1/4):327–331.
Raunio H, Husgafvel-Pursiainen K, Anttila S, Hietanen E, Hirvonen A, Pelkonen O. 1995. Diagnosis of polymorphisms in carcinogen-activating and inactivating enzymes and cancer susceptibility—a review. Gene 159:113–121.
Redpath JL and Sun C. 1990. Sensitivity of a human hybrid cell line (HeLa x skin fibroblast), to radiation-induced neoplastic transformation in G2, M, and mid-G1 phases of the cell cycle . Radiat Res 121:206–211.
Reineking A and Porstendörfer J. 1986. High-volume screen diffusion batteries and a-spectroscopy for measurement of the radon daughter activity size distributions in the environment. J Aerosol Sci 17:873–879.
Reineking A and Porstendörfer J. 1990. ''Unattached" fraction of short-lived Rn decay products in indoor and outdoor environments: An improved single-screen method and results. Health Phys 58(6):715–727.
Reineking A, Becker KH, Porstendörfer J. 1985. Measurements of the unattached fractions of radon daughters in houses. Sci Total Environ 45:261–270.
Reineking A, Becker KH, Porstendörfer J. 1988. Measurement of activity size distributions of the short-lived radon daughters in the indoor and outdoor environment. Radiat Prot Dosimet 24:245–250.
Reist PC. 1984. Introduction to Aerosol Science. New York: McMillan.
Report of the Royal Commission regarding radiation, compensation, and safety at the Fluorospar mines of St. Laurence, Newfoundland. 1969.
Rice AJ. 1956. Radiation concentrations in uranium mines and methods of control on the Colorado Plateau. Address to the Colorado Plateau Section, AIME, Grand Junction, Colorado, March 3, 1956 .
Richardson RB, Eatough JP, Henshaw DL. 1991. Dose to red bone marrow from natural radon and thoron exposure. Br J Radiol 64:608–824.
Risch HA, Howe GR, Jain M, Burch JD, Holowaty EJ, Miller AB. 1993. Are female smokers at higher risk for lung-cancer than male smokers? A case-control analysis by histologic type [see comments]. Am J Epidemiol 93:281–293.
Ritter MA, Cleaver JE, Tobias CA. 1977. High-LET radiations induce a large proportion of non-rejoining DNA strand breaks. Nature 266:653–655.
Robbins ES and Meyers OA. 1995. Cycling cells of human and dog tracheobronchial mucosa: Normal and repairing epithelia. Technology: Journal of the Franklin Institute 332A:35–42.
Roberts CJ and Goodhead DT. 1987. The effect of 238Pu a-particles on the mouse fibroblast cell line C3H 10T1/2: Characterization of source and RBE for cell survival. Int J Radiat Biol 52:871–882.
Rogers VC and Nielson KK. 1991. Correlations for predicting air permeabilities and 222Rn diffusion coefficients in soils. Health Phys 61(2):225–230.
Rogot E and Murray JL. 1980. Smoking and causes of death among U.S. veterans: 16 years of observation. Public Health Rep 95:213–222.
Rossi HH. 1991. Point mutations and radiation carcinogenesis. Radiat Res 128(1):115.
Rossi HH and Kellerer AM. 1986. The dose rate dependence of oncogenic transformation by neutrons may be due to variation of response during the cell cycle. Int J Radiat Biol 50:353–361.
Roth R. 1957. The sequelae of chronic arsenic poisoning in Moselle vinters. Germ Med Month 2:172–176.
Rothman KJ. 1986. Modern Epidemiology. Boston: Little, Brown and Company.
Rothman KJ, Greenland S, Walker A. 1980. Concepts of interaction. Am J Epidemiol 112:467–470.
Rowley R. 1998. Mammalian cell cycle responses to DNA damaging agents. In: DNA Damage and Repair: Vol. 2. DNA Repair in Higher Eucaryotes, Nickoloff, JA, Hoekstra, MF, eds. Totowa, NJ: Humana Press.
Ruffle B, Burmaster DE, Anderson PD, Gordon HD. 1994. Lognormal distributions for fish consumption by the general U.S. population. Risk Anal 14:395–404.
Rundo J, Markun F, Plondke NJ. 1979. Observation of high concentrations of radon in certain houses. Health Phys 36(6):729–730.
Ruosteenoja E. 1991. Indoor radon and risk of lung-cancer: An epidemiologic study in Finland. Doctoral Disseration, Department of Public Health, University of Tampere, Finnish Government Printing Centre, Helsinki.
Ruosteenoja E, Makelainen I, Rytomaa T, Hakulinen T, Hakama M. 1996. Radon and lung-cancer in Finland. Health Phys 71(2):185–189.
Rutter CM and Elashoff RM. 1994. Analysis of longitudinal data: Random coefficient regression modeling. Stat Med 13:1211–1231.
Ruzer LS, Nero AV, Harley NH. 1995. Assessment of lung deposition and breathing rate of underground miners in Tadjikistan. Radiat Prot Dosimet 58:261–268.
Rydberg B. 1966. Clusters of DNA damage induced by ionizing radiation: Formation of short DNA fragments. II. Experimental detection. Radiat Res 145:200–209.
Sabitier L, Dutrillaux B, Martins MB. 1992. Chromosomal instability. Nature 357:548.
Sabatier L, Lebeau J, Dutrillaux B. 1994. Chromosomal instability and alterations of telomeric repeats in irradiated human fibroblasts. Int J Radiat Biol 66(5):611–613.
Saccomanno GS, Yale C, Dixon W, Auerbach O, Huth GC. 1986. An epidemiological analysis of the relationship between exposure to Rn progeny, smoking and bronchogenic carcinoma in the U-mining population of the Colorado Plateau 1960–1980. Health Phys 50(5):605–618.
Saccomanno GS, Huth GC, Auerbach O, Kuschner M. 1988. Relationship of radioactive radon daughters and cigarette smoking in the genesis of lung-cancer in uranium miners. Cancer 62:1402–1408.
Saccomanno G, Auerbach O, Kuschner M, Harley NH, Michels RY, Anderson MW, Bechtel JJ. 1996. A comparison between the localization of lung tumors in uranium miners and in nonminers from 1947 to 1991. Cancer 77:1278–1283.
Sachs RK, Awa A, Kodama Y, Nakano M, Ohtaki K, Lucas JN. 1993. Ratios of radiation-produced chromosome aberrations as indicators of large-scale DNA geometry during interphase. Radiat Res 133:345–350.
Samet JM. 1988. Involuntary exposure to tobacco smoke. Ann Sports Med 4:1–15.
Samet JM. 1989. Radon and lung-cancer. J Natl Cancer Inst 81:745–757.
Samet JM. 1992. Diseases of uranium miners and other underground miners exposed to radon. In Environmental and Occupational Medicine, 2nd ed, Rom WN, ed. Boston: Little, Brown and Company.
Samet JM. 1994. Indoor radon and lung-cancer: Risky or not? (Editorial). J Natl Cancer Inst 86(24):1813–1814.
Samet JM. 1995. Lung-cancer. In: Cancer Prevention and Control, Greenwald P, Kramer BS, Weed DL, eds. New York: Marcel Dekker, Inc.
Samet JM, ed. (In Press). Changes in Cigarette Related Disease Risks and Their Implication for Prevention and Control. Bethesda, Maryland: U.S. Government Printing Office.
Samet JM and Spengler JD. 1991. Introduction. In: Indoor Air Pollution. A Health Perspective, Samet JM and Spengler JD, eds. Baltimore: Johns Hopkins University Press.
Samet JM, Humble CG, Pathak DR. 1986a. Personal and family history of respiratory disease and lung-cancer risk. Am Rev Respir Dis 134:466–470.
Samet JM, Morgan MV, Key MV, Pathak DR, Valdivia AA. 1986b. Studies of uranium miners in New Mexico. Proceedings of the International Conference in Health of Miners, Cincinnati, American Conference of Governmental Hygienists, RW Wheeler, ed. Pp. 351–355.
Samet JM, Wiggins CL, Humble CG, Pathak DR. 1988. Cigarette smoking and lung-cancer in New Mexico. Am Rev Resp Dis 88(5):1110–1113.
Samet JM, Pathak DR, Morgan MV, Marbury MC, Key CR, Valdivia AA. 1989. Radon progeny exposure and lung-cancer risk in New Mexico uranium miners: A case-control study. Health Phys 56:415–421.
Samet JM, Pathak DR, Morgan MV, Key CR, Valdivia AA, Lubin. JJ. 1991. Lung-cancer mortality and exposure to radon progeny in a cohort of New Mexico uranium miners. Health Phys 61:745–752.
Samet JM, Pathak DR, Morgan MV, Coultas DB, James DS, Hunt WC. 1994. Silicosis and lung-cancer risk in underground uranium miners. Health Phys 66:450–453.
(SAMMEC) Smoking-Attributable Mortality, Morbidity, and Economic Cost [computer program]. 1992. Shultz JM, Novotny TE, Rice DP. Vers. 2.1. Atlanta, GA: Centers for Disease Control and Prevention.
Sanders CL. and Lundgren DL. 1995. Pulmonary carcinogenesis in F 344 and Wistar rat after inhalation of plutonium dioxide. Radiat Res 144:206–214.
Sanders CL, Lauhala KE, McDonald KE. 1989. Tritiated thymidine-labeled bronchioloalveolar cells and radiation dose following inhalation of plutonium in rats. Exp Lung Res 15:755–769.
Sanford KK, Parshad R, Gantt R, Tarone RE, Jones GM, Price FM. 1989. Factors affecting and significance of G chromatid radiosensitivity in predisposition to cancer. Int J Radiat Biol 55:963–981.
Sankaranarayanan K and Chakraborty R. 1995. Cancer predisposition, radiosensitivity and the risk of radiation-induced cancers. I. Background. Radiat Res 143:121–143.
Savage JR. 1996. Insight into sites. Mutat Res 366(2):81–95.
Saxon PJ, Srivatsan ES, Stanbridge EJ. 1986. Introduction of normal human chromosome 11 via microcell transfer controls tumorigenic expression of HeLa cells. EMBO J:5:3461–3466.
Scheibel HG and Porstendörfer J. 1984. Penetration measurements in the ultrafine particle size range. J Aerosol Sci 15:549–556.
Schenker MB, Smith T, Muñoz A, Woskie S, Speizer FE. 1984. Diesel exposure and mortality among railway workers: result of a pilot study. Br J Ind Med 41:320–327.
Schery SD. 1990. Thoron in the environment. J AWWA 40:493–497.
Schery SD. 1992. Thoron and its progeny in the atmospheric environment. Chapter 10 of Gaseous Pollutants: Characterization and Cycling. J Wiley & Sons.
Scheuch G. 1991. Die Dispersion, Deposition, und Clearance von Aerosolpartikeln in den menschlkichen Atemwegen (Ph.D. thesis), J.W. Goethe-Universität. Frankfurt am Main, Germany.
Scheuch G, Kreyling W, Haas F, Stahlhofen W. 1993. Effect of settling velocity on particle recovery from human conducting airways after breath holding. J Aerosol Med 6(Suppl.) 47.
Scheutzle D and Jensen TE. 1985. Analysis of nitrated polycyclic aromatic hydrocarbons (nitro-PAH) by mass spectrometry. Pp. 121–167 in Nitrated Polycyclic Aromatic Hydrocarbons, White C, ed. Heidelberg: Huthig Verlag.
Scheutzle D and Lewtas J. 1986. Bioassay-directed chemical analysis in environmental research. Anal Chem 58:1060A–1075A.
Schlesinger RB. 1985. Clearance from the respiratory tract. Fund Appl Toxicol 5:435.
Schoenberg JB, Wilcox HB, Mason TJ, Bill J, Stemhagen A, Benhamou E, Benhamou S, Auquier A, Flamant R. 1989. Variation in smoking-related lung-cancer risk among New Jersey women. Changes in patterns of cigarette smoking and lung-cancer risk: Results of a case-control study. Am J Epidemiol 89(4):601–604.
Schoenberg JB, Klotz JB, Wilcox GP, Gil-del-Real MT, Stemhagen A, Mason TJ. 1990. Case-control study of residential radon and lung-cancer among New Jersey women. Cancer Res 50:6520–6524.
Schwab M, Alitalo K, Klempnauer KH, Varmus HE, Bishop JM, Gilbert F, Brodeur G, Goldstein M, Trent J. 1983. Amplified DNA with limited homology to myc cellular oncogene is shared by human neuroblastoma cell-lines and a neuroblastoma tumor. Nature 305:245–248.
Schwartz JL, Rotmensch J, Atcher RW, Jostes RF, Cross FT, Hui TE, Chen D, Carpenter S, Evans HH, Mencl J, Bakale G, Roe PS. 1992. Interlaboratory comparison of different alpha-particle and radon sources: cell survival and relative biological effectiveness. Health Phys 62:458–461.
Schwartz JL, Rotmensch J, Sun J, An J, Xu Z, Yu Y, Hsie A. 1994. Multiplex PCR reaction based deletion analysis of spontaneous, gamma-rays and alpha induced mutants of CHO-K1 cells. Mutagenesis 9:537–540.
Scott AG. 1992. Site characterization for radon supply potential: A progress review. Health Phys 62:422–428.
Scott AG. 1993. Comparison of criteria to define radon-prone areas (letter). Health Phys 64(4):435–436.
Scott D, Spreadborough AR, Jones LA, Roberts SA, Moore CJ. 1996. Chromosomal radiosensitivity in G2-phase lymphocytes as an indicator of cancer predisposition. Radiat Res 145:3–16.
Searle AG, Beechey CV, Green D, Humphreys ER. 1976. Cytogenetic effects of protracted exposures to alpha-particles from plutonium-239 and to gamma-rays from cobalt-60 compared in male mice. Mutat Res 41:297–310.
Selvanayagam CS, Davis CM, Cornforth MN, Ullrich RL. 1995. Latent expression of p53 mutations and radiation-induced mammary cancer. Cancer Res 55:3310–3317.
SENES Consultants, Limited 1989. Uncertainty in exposure of underground miners to radon daughters and the effect of uncertainty on risk estimates. Report to the Atomic Energy Control Board. Ottawa.
SENES Consultants, Limited 1991. Detailed reconstruction of radon daughter exposures of Eldorado Beaverlodge uranium mine employees. Report to the Atomic Energy Control Board. Ottawa.
SENES Consultants Limited. 1995. Preliminary feasibility study into the re-evaluation of exposure data for the Colorado Plateau uranium miner cohort study. A report prepared for the National Mining Association, Washington, D.C. Ontario Canada, November.
SENES Consultants Limited. 1996a. An algorithm for estimating radon decay product exposures from underground employment at the Eldorado Beaverlodge mine. A report prepared for Atomic Energy Control Board, Ottawa, Canada. Ontario, Canada, March.
SENES Consultants Limited. 1996b. A re-evaluation of radon decay product exposures to underground workers at the Port Radium mine. Report to Atomic Energy Control Board of Canada. Ontario, Canada.
Sevc JE and Placek V. 1976. Lung-cancer in uranium miners and long-term exposure to radon daughter products. Health Phys 30:433–437.
Sevc J, Kunz E, Tomásek L, Placek V, Horacek J. 1988. Cancer in man after exposure to Rn daughters. Health Phys 54:27–46.
Sevc J, Tomásek L, Kunz E, Placek V, Chemelevsky D, Barclay D, Kellerer AM. 1993. A survey of the Czechoslovak follow-up of lung-cancer mortality in uranium mines. Health Phys 64:355–369.
Seymour CB, Mothersill C, Alper T. 1986. High yields of lethal mutations in somatic mammalian cells that survive ionizing radiation. Int J Radiat Biol 50:167–179.
Shapiro S. 1994. Meta-analysis/shmeta-analysis. Am J Epidemiol 140:771–778.
Shiloh Y. 1995. Ataxia-telangiectasia: closer to unraveling the mystery. Eur J Human Genet 3:116–138.
Shimizu H, Morishita M, Mizuno K, Masuda T, Ogura Y, Santo M, Nishimura M, Kunishima K, Karasawa K, Nishiwaki K, Yamamoto M, Hisamichi S, Tominaga S. 1988. A case-control study of lung-cancer in nonsmoking women. Tohoku J Exp Med 154:389–397.
Short SR and Ptesonk EL. 1993. Respiratory health risks among nonmetal miners. Occup Med 8:57–70.
Shuin J, Billings PC, Lillehaug JR, Patierno SR, Roy-Burman P, Landolph. JR. 1986. Enhanced expression of c-myc and decreased expression of c-fos protooncogenes in chemically and radiation transformed C3H/10T1/2 mouse embryo cell line. Cancer Res 46:5302–5311.
Siemiatycki J, ed. 1991. Risk Factors for Cancer in the Workplace. Boca Raton: CRC Press.
Simmonds JR, Robinson CA, Phipps AW, Muirhead CR, Fry FA. 1995. Risks of leukaemia and other cancers in Seascale from all sources of ionizing radiation exposure. Report NRPB-R276 HMSO, London.
Simmons JA, Cohn P, Min T. 1996. Survival and yields of chromosome aberrations in hamster and human lung cells irradiated by alpha particles. Radiat Res 145:174–180.
Simonato L, Moulin JJ, Javelaud B, Ferro G, Wild P, Winkelmann R, Saracci R. 1994. A retrospective mortality study of workers exposed to arsenic in a gold mine and refinery in France. Am J Ind Med 25:625–633
Simpson SD, Crosby EH, Yourt GR. 1954. A survey of radioactivity, dust, and ventilation at Eldorado Beaverlodge, Unpublished report, Eldorado Mining & Refining, Ltd., Ottawa, Ontario.
Simpson SG and Comstock GW. 1983. Lung-cancer and housing characteristics. Arch Environ Health 38:248–251.
Sinclair D, George AC, Knutson EO. 1977. Application of diffusion batteries to measurement of submicron radioactive aerosols. In: Airborne Radioactivity. La Grange Park, IL: American Nuclear Society. Pp. 103–114.
Skillrud DM, Offord KP, Miller RD. 1986. Higher risk of lung-cancer in chronic obstructive pulmonary disease. Ann Intern Med 105:503–507.
Small MJ. 1994. Invariably Uncertain about Variability? Try the Normal-Gamma Conjugate!, Presented at the 87th Annual Meeting & Exhibition of the Air & Waste Management Association, Cincinnati, Ohio, June 19–24.
Smith ML, Zhan Q, Bae I, Fornace AJ, Jr. 1994. Role of retinoblastoma. gene product in p53-mediated DNA damage response. Exp Cell Res 215:386–389.
Solli HM, Andersen A, Stranden E, Langard S. 1985. Cancer incidence among workers exposed to radon and thoron daughters at a niobium mine. Scand J Work Env Health 11(1):7–13.
Solomon S. 1989. Personal communication.
Sram RJ, Binkova B, Dobias L, Rossner P, Topinka J, Vesela D, Vesely D, Stejskalova J, Bavorova H, Rericha V. 1993. Monitoring genotoxic exposure in uranium miners. Environ Health Perspect 99:303–305.
Stahlhofen W. 1989. Human lung clearance following bolus inhalation of radioaerosols. Pp. 153–166 in Extrapolation of Dosimetric Relationships for Inhaled Particles and Gases. Washington: Academic Press.
Stahlhofen W, Gebhart J, Heyder J. 1980. Experimental determination of the regional deposition of aerosol particles in the human respiratory tract. Am Ind Hyg Assoc J 41:385.
Stahlhofen W, Gebhart J, Rudolf G, Scheuch G. 1986a. Measurement of lung clearance with pulses of radioactively-labelled aerosols. J Aerosol Sci 17:333–336.
Stahlhofen W, Gebhart J, Rudolf G, Scheuch G, Philipson K. 1986b. Clearance from the human airways of particles of different sizes deposited from inhaled aerosol boli. In: Aerosols: Formation and Reactivity, Second International Aerosol Conference, West Berlin, Germany, September 22–26, 1986, pp. 192–196, Pergamon Press, Oxford, U.K.
Stahlhofen W, Gebhart J, Rudolf G, Scheuch G. 1987a. Retention of radiolabelled Fe2O3-particles in human lungs. In: Deposition and Clearance of Aerosols in the Human Respiratory Tract, Second International Symposium, Salzburg, Austria, September 18–20, 1986, pp 123–128, (ed. Hofmann, W.) Facultas Universitätsverlag Ges.m.b.H. Vienna Austria.
Stahlhofen W, Gebhart J, Rudolf G, Scheuch G, Bailey MR. 1987b. Human lung clearance of inhaled radioactively labelled particles in horizontal and vertical position of the inhaling person. J Aerosol Sci 18:741–744.
Stahlhofen W, Koebrich R, Rudolf G, Scheuch G. 1990. Short-term and long-term clearance of particles from the upper human respiratory tract as function of particle size. J. Aerosol Sci. 21(Suppl. 1):S407–S410.
Stahlhofen W, Scheuch G Bailey MR. 1994. Measurement of the tracheobronchial clearance of particles after aerosol bolus inhalation. In: Inhaled Particles VII, Proceedings of an International Symposium on Inhaled Particles Organized by the British Occupational Hygiene Society, 16–22 September 1991. Dodgson J, McCallum RI, eds. Ann Occup Hyg 189.
Stanbridge EJ. 1976. Suppression of malignancy in human cells. Nature 260:17–20.
Stather JW, Dionian J, Brown J, Fell TP, Muirhead CR. 1986. The risks of leukaemia and other cancers in Seascale from radiation exposure. Addendum to Report R171. Report NRPB-R171. Addendum HMSO, London.
Stayner LT and Wegman DH. 1983. Smoking, occupation, and histopathology of lung-cancer: A case-control study with the use of the Third National Cancer Survey. J Natl Cancer Inst 70:421–426.
Steenland K and Goldsmith DF. 1995. Silica exposure and autoimmune disease. Am J Ind Med 28:603–608.
Steenland K, Silverman D, Zaebst D. 1992. Exposures to diesel exhaust in the trucking industry and possible relationships with lung-cancer. Am J Ind Med 21:887–890.
Steinhäusler F. 1996. Environmental 220Rn: A review. Environ Int 22(Suppl 1): S1111–1123.
Stenerlow B, Blomquist E, Grusell E, Hartman T, Carlsson J. 1996. Rejoining of DNA double-strand breaks induced by accelerated nitrogen ions. Int J Radiat Biol 70:413–420.
Stewart BW. 1994. Mechanisms of apoptosis: integration of genetic, biochemical, and cellular indicators. J Natl Cancer Inst 86(17):1286–1296.
Stidley CA and Samet JM. 1993. A review of ecological studies of lung-cancer and indoor radon. Health Phys 65:234–251.
Stidley CA and Samet JM. 1994. Assessment of ecologic regression in the study of lung-cancer and indoor radon. Am J Epidemiol 139:312–322.
Stranden E. 1987. Radon-222 in Norwegian dwellings. In Radon and Its Decay Products: Occurrence, Properties, and Health Effects. Washington: American Chemical Society.
Strong JC. 1988. The size of attached and unattached radon daughters in room air. J Aerosol Sci 19:1327–1330.
Strong JC. 1989. Design of the NRPB activity size measurement system and results, presented at the Workshop on "Unattached" Fraction Measurements, University of Illinois, Urbana, IL, April 1989.
Stuart BO. 1984. Deposition and clearance of inhaled particles. Environ Health Perspect 55:369.
Stuart BO, Palmer RF, Fillip RE, Gaven J. 1978. Inhaled radon daughters and uranium ore dust in rodents, pp. 3.70–3.72. In: Pacific Northwest Laboratory Annual Report for 1977 to the DOE assistant secretary for environment, PNL-2500, Pt. 1, JNTIS, Springfield, VA.
Svensson C, Eklund G, Pershagen G. 1987. Indoor exposure to radon from the ground and bronchial cancer in women. Int Arch Occup Environ Health 59:123–131.
Svensson C, Pershagen G, Hrubec Z. 1988. A comparative study on different methods of measuring Rn concentrations in homes. Health Phys 55(6):895–902.
Svensson C, Pershagen G, Klominek J. 1989. Lung-cancer in women and type of dwelling in relation to radon exposure. Cancer Res 49:1861–1865.
Swartout HO and Webster RG. 1940. To what degree are mortality statistics dependable? Am J Public Health 30:811–815.
Swedjemark GA. 1985. Radon and its decay products in housing. Estimation of the radon daughter exposure to the Swedish population and methods for evaluation of the uncertainties in annual averages. Doctoral Dissertation, Department of Radiation Physics, University of Stockholm.
Swift M, Morrell D, Cromartie E, Chamberlin AR, Skolnick MH, Bishop, DT. 1986. The incidence and gene frequency of ataxia-telangiectasia in the United States. Am J Hum Genet 39:573–583.
Swift M, Morrell D, Massey RB, Chase CL. 1991. Incidence of cancer in 161 families affected by ataxia-telangiectasia. N Engl J Med 325:1831–1836.
Swift DL, Montassier N, Hopke PK, Karpen-Hayes K, Cheng YS, Su YF, Yeh HC, Strong JC. 1992. Inspiratory deposition of ultrafine particles in human nasal replicate casts. J Aerosol Sci 23:65–72.
Takeshima Y, Seyama T, Bennett WP, Akiyama M, Tokuoka S, Inai K, Mabuchi K, Land CE, Harris CC. 1993. p53 mutations in lung-cancers from non-smoking atomic-bomb survivors. Lancet 342:1520–1521.
Taya A., Morgan A, Baker ST, Humphreys JA, Bisson M, Collier CG. 1994. Changes in the rat lung after exposure to radon and its progeny: effects on incorporation of bromodeoxyuridine in epithelial cells and on the incidence of nuclear aberrations in alveolar macrophages. Radiat Res 139:170–177.
Taylor JA, Watson MA, Devereux TR, Michels RY, Saccomanno G, Anderson M. 1994. p53 muitation hotspot in radon-associated lung-cancer. Lancet 343:86–87.
Taylor PR, Qiao YL, Schatzkin A, Yao SX, Lubin JH, Mao BL, Rao JY, McAdams M, Xuan XZ, Li JY. 1989. The relation of arsenic exposure to lung-cancer among tin miners in Yunnan Province, China. Br J Ind Med 46:881–886.
Thacker J. 1986. The nature of mutants induced by ionising radiation in cultured hamster cells. III. Molecular characterization of HPRT-deficient mutants induced by gamma-rays or alpha-particles showing that the majority have deletions of all or part of the hprt gene . Mutat Res 160:267–275.
Thacker J. 1988. Meta-analysis. A quantitative approach to research integration. JAMA 88(11):1685–1689.
Thacker J. 1994. The study of responses the model DNA breaks induced by restriction endonucleases in cells and cell-free systems: Achievements and difficulties. Int J Radiat Biol 66:591–596.
Thacker J. 1995. Molecular mechanisms of radiation mutagenesis in normal and radiosensitive human cells. In: Proceedings of the 10th International Congress on Radiation Research, Wiirzburg, 27 August – 1 September 1995.
Thacker J, Stretch A, Stephens MA. 1979. Mutation and inactivation of cultured mammalian cells exposed to beams of accelerated heavy ions. II. Chinese hamster cells. Int J Radiat Biol 36:37–148.
Thacker J, Stretch A, Goodhead DT. 1982. The mutagenicity of a-particles from plutonium-238. Radiat Res 92:343–352.
Thacker SB. 1988. Meta-analysis: A quantitative approach to research integration. JAMA 259:1685–1689.
Thomas DC. 1981. General relative risk models for survival time and matched case-control studies . Biometrics 37:673–686.
Thomas DC. 1988. Models for exposure-time-response relationships with applications to cancer epidemiology. Annu Rev Public Health 9:451–482.
Thomas DC and McNeill KG. 1982. Risk Estimates for the Health Effects of Alpha Radiation. Info-0081. Ottawa: Atomic Energy Control Board.
Thomas DC, McNeill KG, Dougherty C. 1985. Estimates of lifetime lung-cancer risks resulting from Rn progeny exposures. Health Phys 49:825–846.
Thomas D, Stram D, Dwyer J. 1993. Exposure measurement error: Influence on exposure-disease relationships and methods of correction. Ann Rev Public Health 14:69–93.
Thomas D, Pogoda J, Langholz B, Mack W. 1994. Temporal modifiers of the radon-smoking interaction. Health Phys 66:257–262.
Thomassen DG, Seiler FA, Shyr LJ, Griffith WC. 1990. Alpha-particles induce preneoplastic transformation of rat tracheal epithelial cells in culture. Int J Radiat Biol 57:395–405.
Thomassen DG, Newton GJ, Guilmette RA, Johnson, NF. 1992. A biodosimetric approach for estimating radiation dose to the respiratory epithelium from inhaled radon progeny. Radiat Prot Dosimet 38:65–71.
Thompson CB. 1995. Apoptosis in the pathogenesis and treatment of disease. Science 267(5203):1456–1462.
Thun MJ, Day-Lally C, Myers DG, Calle EE, Flanders WD, Zhu B, Namboodiri MM, Health CW. 1997. Trends in tobacco smoking and mortality from cigarette use in Cancer Prevention Studies 1 (1959 through 1965) and II (1982 through 1988). In Changes in Cigarette-Related Disease Risks and Their Implication for Prevention and Control, Shopland DR, Burns DM, Garfinkel L, Samet JM, eds. National Cancer Institute. Monograph 8: Smoking and Tobacco Control. NIH Publication no. 97–4213.
Tirmarche M, Brenot J, Piechowski J, Chameaud J, Paradel J. 1984. The Present State of an Epidemiological Study of Uranium Miners in France. Pp. 344–349 in Proceedings of the International Conference on Occupational Radiation Safety in Mining , Vol 1, Stocker H, ed. Toronto: Canadian Nuclear Association.
Tirmarche M, Raphalen A, Allin F, Chameaud J, Bredon P. 1993. Mortality of a cohort of French uranium miners exposure to relatively low radon concentrations. Br J Cancer 67:1090–1097.
Tockman MS and Samet JM, eds. 1994. Other host factors and lung-cancer susceptibility. Pp. 397–412 in Epidemiology of Lung-cancer. New York: Marcel Dekker, Inc.
Tockman MS, Anthonisen NR, Wright EC, Donithan MG. 1987. Airway obstruction and risk for lung-cancer. Ann Intern Med 106:512–518.
Tomásek L and Darby SC. 1995. Recent results from the study of West Bohemian uranium miners exposed to radon and its progeny. Environ Health Perspect 103(Suppl 2):55–57.
Tomásek L, Darby SC, Swerdlow AJ, Placek V, Kunz E. 1993. Radon exposure and cancers other than lung-cancer among uranium miners in West Bohemia . Lancet 341:919–923.
Tomásek L, Darby SC, Fearn T, Swerdlow AJ, Placek V, Kunz E. 1994a. Patterns of lung-cancer mortality among uranium miners in West Bohemia with varying rates of exposure to radon and its progeny. Radiat Res 137:251–261.
Tomásek L, Swerdlow AJ, Darby SC, Plcek V, Kunz E. 1994b. Mortality in uranium miners in West Bohemia: A long term cohort study. Occup Environ Med 41:308–315.
Trump BF, McDowell EM, Harris CC. 1984. Chemical carcinogenesis in the tracheobronchial epithelium. Environ Health Perspect 55:77–84.
Tu KW and Knutson EO. 1988a. Indoor radon progeny particle size distribution measurements made with two different methods. Radiat Prot Dosimet 24:251–255.
Tu KW and Knutson EO. 1988b. Indoor outdoor aerosol measurements for two residential buildings in New Jersey. Aerosol Sci Technol 9:71–82.
Twomey S. 1975. Comparison of constrained linear inversion and an iterative nonlinear algorithm applied to the indirect estimation of particle size distributions. J Comp Phys 18:188–200.
Ullrich RL. 1983. Tumor induction in BALB/c female mice after fission neutron or γ irradiation. Radiat Res 93:506–515.
Ullrich RL, Jernigan MC, Cosgrove GE, Satterfield LC, Bowles ND, Storer JB. 1976. The influence of dose and dose rate on the incidence of neoplastic disease in RFM mice after neutron irradiation. Radiat Res 68:115–131.
UNSCEAR (United Nations Scientific Committee on the Effects of Atomic Radiation). 1988. Sources, Effects and Risks of Ionizing Radiation. Report of the United Nations Scientific Committee on the Effects of Atomic Radiation. New York: United Nations.
UNSCEAR (United Nations Scientific Committee on the Effects of Atomic Radiation). 1994. Sources and Effects of Ionizing Radiation. United Nations, New York.
U.S. Department of Health Education and Welfare. 1964. Smoking and Health. Report of the Advisory Committee to the Surgeon General. Washington, DC: U.S. Government Printing Office.
USDHHS (U.S. Department of Health and Human Services). 1985. A report of the Surgeon General: The health consequences of smoking — Cancer and chronic lung disease in the workplace. Washington, DC: U.S. Government Printing Office .
USDHHS (U.S. Department of Health and Human Services). 1988. A report of the Surgeon General: The health consequences of smoking: Nicotine addiction. Washington, DC: U.S. Government Printing Office.
USDHHS (U.S. Department of Health and Human Services). 1989. A report of the Surgeon General: Reducing the Health Consequences of Smoking. 25 Years of Progress. Washington, DC: U.S. Government Printing Office.
USDHHS (U.S. Department of Health and Human Services). 1990. A report of the Surgeon General: The health benefits of smoking cessation. Washington, DC: U.S. Government Printing Office.
USDHHS (U.S. Department of Health and Human Services). 1991. Strategies to control tobacco use in the United States: A blueprint for public health action in the 1990's. Washington, DC: U.S. Government Printing Office.
USDHHS (U.S. Department of Health and Human Services). 1995. SEER Cancer Statistics Review 1973–1990. National Institutes of Health 93-2789.
USDHHS (U.S. Department of Health and Human Services). 1996. Public Health Service, and National Cancer Institute (NCI). Burns, D.M., Garfinkel, L., and Samet, J.M. editors. Changes in Cigarette-Related Disease Risks and Their Implication for Prevention and Control. Bethesda, Maryland: U.S. Government Printing Office. 1996; 6 (In Press). Smoking and Tobacco Control Monograph.
U.S. Department of Labor, Bureau of Labor Statistics. 1972. Railroad Technology and Manpower in the 1970's. USGPO, Washington, D.C.
USEPA (U.S. Environmental Protection Agency). 1991. EPA's National Residential Radon Survey Preliminary Results. U.S. Government Printing Office, Washington, D.C.
USEPA (U.S. Environmental Protection Agency). 1992a. Respiratory health effects of passive smoking: Lung-cancer and other disorders. EPA/600/006F. U.S. Government Printing Office, Washington, D.C.
USEPA (U.S. Environmental Protection Agency). 1992b. Technical Support Document for the 1992 Citizen's Guide to Radon. 1992b; EPA-400-R-92-011. U.S. Government Printing Office, Washington, D.C.
USEPA (U.S. Environmental Protection Agency). 1992c. A Citizen's Guide to Radon. The Guide to Protecting Yourself and Your Family from Radon. 2nd edition. U.S. Government Printing Office, Washington, DC.
USEPA (U.S. Environmental Protection Agency). 1993. National School Radon Survey: Report to Congress. Washington, DC: Environmental Protection Agency.
U.S. Surgeon General. 1989. Reducing the Heath Consequences of Smoking. 25 Years of Progress. U.S. Government Printing Office, Washington, D.C.
Vahakangas KH, Samet JM, Metcalf RA, Welsh JA, Bennett WP, Lane DP, Harris CC. 1992. Mutations of p53 and ras genes in radon-associated lung-cancer from uranium miners. Lancet 339:576–580.
Van Leeuwen FE, Klokman WJ, Stovall M, Hagenbeek A, van-den-Belt-Dusebout AW, Noyon R, Boice JD Jr, Burgers JM, Somers R. 1995. Roles of radiotherapy and smoking in lung-cancer following Hodgkin's disease. J Natl Cancer Inst 87:1530–1537.
Venitt S and Biggs PJ. 1994. Radon, mycotoxins, p53, and uranium mining. Lancet 343:795.
Vogel F. 1979. Genetics of retinoblastoma. Hum Genet 52:1–54.
Vogelstein B. 1990. A deadly inheritance. Nature 348:681.
Vonstille WT and Sacarello HLA. 1990. Radon and cancer: Florida study finds no evidence of increased risk. J Environ Health 53:25–28.
Vostal JJ. 1986. Factors limiting the evidence for chemical carcinogenicity of diesel emissions in long-term inhalation experiments. In Carcinogenic and Mutagenic Effects of Diesel Engine Exhaust, eds. N. Ishinishi, A. Koizumi, R. O. McClellan and W. Stöber, pp. 381–396, Proceedings of the International Satellite Symposium on Toxicological Effects of Emissions from Diesel Engines held in Tsukuba Science City, Japan, July 26–28, 1986. Amsterdam: Elsevier.
Wainscoat JS and Fey MF. 1990. Assessment of clonality in human tumors: A review. Cancer Res 50:1355–1360.
Wang Y, Krewski D, Lubin JH, and Zielinski JM. 1995. Meta-analysis of multiple cohorts of underground miners exposed to radon. Pp. 21–28 in Proceeding of Statistics Canada Symposium 95: From Data to Information—Methods and Systems. Ottawa: Statistics Canada.
Ward JF. 1985. Biochemistry of DNA lesions. Radiat Res 104:S103–S111.
Ward JF. 1988. DNA damage produced by ionizing radiation in mammalian cells: identities, mechanisms of formation and repairability. Progress in Nucleic Acids and Molecular Biology 35:95–125.
Ward JF. 1994. The complexity of DNA damage: relevance to biological consequences. Int J Radiat Biol 66:427–432.
Wasiolek PT, Hopke PK, James AC. 1992. Assessment of exposure to radon decay products in realistic living conditions. J Exposure Anal Environ Epidemiol 2:309–322.
Waxweiler RJ, Stringer W, Wagoner JK, Jones J, Falk H, Carter C. 1976. Neoplastic risk among workers exposed to vinyl chloride. Ann NY Acad Sci (5NM) 271:40–48.
Wei L, Sha Y, Tao Z, He W, Chen D, Yuan Y. 1990. Epidemiological investigation in high background radiation areas of Yangjiang, China. Sohrabi M, Ahmed U, Durrani SA, eds. Proc Int Conf On High Levels of Natural Radioactivity.
Weinberg CR, Moledor ES, Umbach DM, Sandler D: Submitted. Imputation for exposure histories with gaps.
Westin JB, Cramer Z, Richter ED, Shani J, Ne'eman E, Elyakim O, Tal Y. 1991. Radon in a self-selected sample of Isreali homes, schools, and workplaces. Public Health Rev 19(1–4):199–203.
White E. 1994. p53, guardian of Rb. Nature 371:21–22.
White SB, Bergston JW, Alexander BV, Rodman NF, Phillips JL. 1992. Indoor radon 222Rn concentrations in a probability sample of 43,000 houses across 30 states. Health Phys 62:41–50.
Whitehead A and Whitehead J. 1991. A general parametric approach to the meta-analysis of randomized clinical trials. Stat Med 10:1665–1677.
Whittemore AS and McMillan A. 1983. Lung-cancer mortality among U.S. uranium miners: A reappraisal. J Natl Cancer Inst 71:489–499.
Wicks M, Archer V, Auerbach O, Kuschner M. 1981. Arsenic exposure in a copper smelter as related to histological type of lung-cancer. Am J Ind Med 2:25–31.
Wiggins CL, Becker TM. 1993. Racial and ethnic patterns of mortality in New Mexico. Albuquerque: University of New Mexico Press.
Williams RR, Stegens NL, Goldsmith JR. 1977. Associations of cancer site and type with occupation and industry from the third national cancer survey interview. J Natl Cancer Inst 59:1147–1185.
Wolff SP. 1991. Leukaemia risks and radon. Nature 352:288.
Wolff S, Afzal V, Wiencke JK, Olivieri G, Michaeli A. 1988. Human lymphocytes exposed to low doses of ionizing radiations become refractory to high doses of radiation as well as to chemical mutagens that induce double-strand breaks in DNA. Int J Radiat Biol 53:39–48.
Wolff S, Jostes RF, Cross FT, Hui TE, Afzal V, Wiencke JK. 1991. Adaptive response of human lymphocytes for the repair of radon-induced chromosomal damage. Mutat Res 250:299–306.
Woodward AD, Roder AJ, McMichael P, Crouch, Mylvaganam A. 1991. Radon daughter exposures at the Radium Hill Uranium Mine and lung-cancer rates among former workers, 1952–87. Cancer Causes and Control 2:213–220.
Wooster R, Heuhausen SL, Mangion J, Quirk Y, Ford D, Collins N, Nguyen K, Seal S, Tran T, Averill D, Fields P, Marshall G, Narod S, Lenoir GM, Lynch H, Feunteun J, Devilee P, Cornelisse CJ, Menko FH, Daly PA, Ormiston W, McManus R, Pye C, Lewis CM, Cannon-Albright LA, Peto J, Ponder BAJ, Skolnick MH, Easton DF, Goldgar DE, Stratton MR. 1994. Localization of a breast cancer susceptibility gene, BRCA2, to Chromosome 13q12-13. Science 265:2088–2090.
Wright HA, Magee JL, Hamm RN, Chatterjee A, Turner JE, Klots CE. 1985. Calculations of physical and chemical reactions produced in irradiated water containing DNA. Radiat Prot Dosimet 13:133–136.
Wu AH, Henderson BE, Pike MC, Yu MC. 1985. Smoking and other risk factors for lung-cancer in women. J Natl Cancer Inst 85(4):747–751.
Wu-Williams AH and Samet JM. 1994. Lung-cancer and cigarette smoking. In Epidemiology of Lung-Cancer, Samet JM, ed. New York: Marcel Dekker, Inc.
Wynder E and Graham EA. 1950. Tobacco smoking as a possible etiologic factor in bronchiogenic carcinoma. A study of six hundred and eighty-four proved cases. JAMA 143:329–346.
Wynder EL, Bross IJ, Cornfield J. 1956. Lung-cancer in women — a study of environmental factors. N Engl J Med 255:1111–1121.
Wynder EL, Mabuchi K, Beattie EJ. 1970. The epidemiology of lung-cancer: Recent trends. J Am Med Assoc 213:2221–2228.
Xiang-Zhen X, Lubin JH, Jun-Yao L, Li-Fen Y, Qing-Sheng L, Lan Y, Jian-Zhang W, Blot WJ. 1993. A cohort study in southern China of tin miners exposed to Radon and radon decay products. Health Phys 64:120–131.
Xu Z-Y, Blot WJ, Xiao H-P, Wu A, Feng Y-P, Stone BJ, Sun J, Ershow AG, Henderson BE, Fraumeni JF Jr. 1989. Smoking, air pollution and the high rates of lung-cancer in Shenyang, China. J Natl Cancer Inst 81:1800–1806.
Xuan XZ, Lubin JH, Li JY, Blot WJ. 1993. A cohort study in southern China of workers exposed to radon and radon decay products. Health Phys 64:120–131.
Yalow RS. 1995. Radiation and Public Perception. In Radiation and Public Perception. Benefits and Risks. Washington: American Chemical Society.
Yao SX, Lubin JH, Qiao YL, Boice JD, Jr., Li JY, Cai SK, Zhang FM, Blot WJ. 1994. Exposure to radon progeny, tobacco use and lung-cancer in a case-control study in southern China. Radiat Res 138:326–336.
Yeager WM, Lucas RM, Daum KA, Sensintaffar E, Poppell S, Feldt L, Clarkin M. 1991. A performance evaluation study of three types of a-track detector radon monitors. Health Phys 60:507–515.
Yeh HC, Cheng YS, Orman MM. 1982. Evaluation of various types of wire screens as diffusion battery cells, J Colloid Interface Sci 86:12–16.
You M, Wang Y, Stoner G, You L, Maronpot R, Reynolds SH, Anderson M. 1992. Parental bias of Ki-ras oncogenes detected in lung tumors from mouse hybrids. Proc Natl Acad Sci USA 89:5804–5808.
Zeger S, Liang KY, Albert PS. 1988. Models for longitudinal data: A general estimating equation approach. Biometrics 44:1049–1060.
Zhu LX, Waldren CA, Vannias D, Hei TK. 1996. Cellular and molecular analysis of mutagenesis induced by charged particles of defined linear energy transfer. Radiat Res 145(3):251–259.
Zielinski JM and Krewski D. 1990. Application of the two-stage model clonal expansion model in characterizing the joint effect of exposure to two carcinogens. Pp. 846–880 in Indoor Radon and Lung-Cancer: Reality or Myth? 29th Hanford Symposium on Health and Environment, Columbus, Ohio. Health and Environment. Columbus: Battle Press.
Glossary
A
Absolute risk.
An expression of excess risk based on the assumption that the excess risk from radiation exposure adds to the underlying (baseline) risk by an increment dependent on dose but independent of the underlying natural risk.
Absorbed dose.
The mean energy imparted by ionizing radiation to an irradiated medium per unit mass. Units: gray (Gy), rad. 1 Gy = 100 rad.
Action level.
A concentration of radon in air that the Environmental Protection Agency recommends should be the maximum concentration in homes; i.e., concentrations above this level should be reduced or mitigated to this level. In 1998, EPA recommends an action level of 148 Bqm-3 or 4 pCiL-1.
Activity.
The amount of radionuclide radioactivity defined as the mean number of decays per unit time. Units: becquerel (Bq), curie (Ci). 1 Bq = 2.7 × 10-11 Ci.
Activity Median Aerodynamic Diameter (AMAD).
The diameter of a unit-density sphere with the same terminal settling velocity in air as that of the aerosol particle whose activity is the median for the environment.
Additive effects.
Equal to the sum of effects from two agents when acting alone.
Aerosol.
Solid or liquid particles that are dispersed in a gaseous medium, are able to remain suspended for a relatively long time, and that have a high surface area to volume ratio.
Alpha particle.
Two neutrons and two protons bound as a single particle that is emitted from the nucleus of certain radioactive isotopes in the process of decay or disintegration. Is positively charged and indistinguishable from a helium atom nucleus.
Apoptosis.
Programmed cell death. The cell death is characterized by a distinctive fragmentation of DNA which is regulated by cellular functions.
Attributable risk (AR).
The estimated burden of a disease (such as lung-cancer) that could, in theory, be prevented if all exposures to a particular causative agent (such as radon) were eliminated.
B
Background radiation.
The amount of radiation to which a member of the population is exposed from natural sources, such as terrestrial radiation due to naturally occurring radionuclides in the soil, cosmic radiation originating in outer space, and naturally occurring radionuclides deposited in the human body.
Baseline rate.
The cancer incidence observed in a population in the absence of the specific agent being studied; the baseline rate includes cancers from a number of other causes, such as smoking, background radiation, etc.
Becquerel (Bq).
SI unit of activity. (see Units). 1 Bq = 1 disintegration per second.
BEIR IV.
Refers to the report of the fourth National Research Council Committee on Biological Effects of Ionizing Radiation; the report was published in 1988.
Bias.
Factors that influence the outcome of data collection such as causing certain measurements to have a greater chance of being included than others.
Bronchial morphometry.
Characterization of the cellular and anatomical structure of the bronchial region of the lung.
C
Cancer.
A malignant tumor of potentially unlimited growth, capable of invading surrounding tissue or spreading to other parts of the body by metastasis.
Carcinogen.
An agent that is believed to be able to cause cancer. Ionizing radiations are physical carcinogens; there are also chemical and biologic carcinogens and biologic carcinogens may be external (e.g., viruses) or internal (genetic defects).
Carcinoma.
A malignant tumor (cancer) of epithelial origin.
Case-control study.
An epidemiologic study in which people with disease and a similarly composed control group are compared in terms of exposures to a putative causative agent.
Cell Culture.
The growing of cells in vitro (in a glass or plastic container, or in suspension) in such a manner that the cells are no longer organized into tissues.
Cohort study.
An epidemiologic study in which groups of people (the cohort) are identified with respect to the presence of, or absence of, exposure to a disease-causing agent and in which the outcomes in terms of disease rates are compared; also called a follow-up study.
Competing risks.
Other causes of death which affect the outcome of the risk being studied. Persons dying from other causes are not recorded at risk of dying from the factor in question.
Confidence limits or intervals.
A measure of the reliability of a risk estimate. A 90% confidence interval means that 9 times out of 10 the estimated risk would be within the specified interval.
Constant relative risk (CRR).
A risk model which assumes that, after a certain time, the ratio of the risk at a specific dose to the risk in the absence of the dose does not change with time.
Curie (Ci).
A unit of activity equal to 3.7 × 1010 disintegrations/s or 3.7 × 1010 becquerels. (see Units).
D
DNA.
Deoxyribonucleic acid; the genetic material of cells.
Deletions.
Type of mutation in which sections of DNA are removed; can refer to the removal of a single base or several bases.
Dose.
The quantity of energy or chemical agent delivered to a specific tissue following exposure. (see Absorbed dose).
Dose-distribution factor.
A factor which accounts for modification of the dose effectiveness in cases in which the radionuclide distribution and the resultant dose are non-uniform.
Dose-effect (dose-response) model.
A mathematical formulation and description of the way the effect (or biological response) depends on the dose.
Dose equivalent.
A quantity that expresses, for the purposes of radiation protection and control, an assumed equal biological effectiveness of a given absorbed dose on a common scale for all kinds of ionizing radiation. SI unit is the Sievert (see Units). 1 Sv = 100 rem.
Dose rate.
The quantity of absorbed dose delivered per unit time.
Dose Rate Effectiveness Factor (DREF).
A factor by which the effect caused by a specific type of radiation changes at low (protracted or fractionated delivery of dose) as compared to high (or acute) dose rates.
Dosimetric model.
A method for estimating risk based on the use of physical models for doses to target cells and the use of results from epidemiologic studies of exposures to humans from other types of radiations.
E
Ecologic study.
A method of epidemiologic analysis in which, for radon, regional rates of lung-cancer are related to the measure of regional radon concentrations.
Effective attributable risk (EAR).
The reduced attributable risk such as the fraction of total lung-cancer deaths that would be eliminated by implementing a radon-mitigation scenario.
Electron volt (eV).
A unit of energy = 1.6 × 10-12 ergs or 1.6 × 10-19 J; 1 eV is equivalent to the energy gained by an electron in passing through a potential difference of 1 V; 1 keV-1,000 eV; 1 MeV - 1,000,000 eV.
Empirical model.
A model that is derived from measurements in populations as opposed to a theoretical model.
Epidemiology.
The study of the determinants of the frequency of disease in humans. The two main types of epidemiological studies of chronic disease are cohort (or follow-up) studies and case-control (or retrospective) studies.
Equilibrium fraction.
In equilibrium, the radioactivity from the parent nuclides and progeny nuclides are equal.
Etiology.
The science or description of cause(s) of disease.
Ever-smokers.
People who smoked cigarettes for a period of time, regardless of whether they have stopped smoking.
Excess relative risk (ERR).
A model that describes the risk imposed by exposures as a multiplicative increment to the excess disease risk above the background rate of disease.
Exposure.
The condition of having contact with a physical or chemical agent.
Exposure-age-concentration model.
Risk model based on the average radon concentration.
Exposure-age-duration model.
Risk model based on the duration of exposure to radon.
F
Fibrosis.
Damage to normal tissue which results in a modification of tissue structure but which is not cancer.
Fractionation.
The delivery of a given dose of radiation as several smaller doses, separated by intervals of time.
G
Gamma radiation.
Also gamma rays; short wavelength electromagnetic radiation of nuclear origin, similar to x rays but usually of higher energy (100 keV to several MeV).
Geometric mean.
The geometric mean of a set of positive numbers is the exponential of the arithmetic mean of their logarithms. The geometric mean of a lognormal distribution is the exponential of the mean of the associated normal distribution.
Geometric standard deviation (GSD).
The geometric standard deviation of a lognormal distribution is the exponential of the standard deviation of the associated normal distribution.
Gray (Gy).
SI unit of absorbed dose (see Units). 1 Gy = 100 rad or deposition of 1 joule per Kg.
H
Half-life, biologic.
Time required for the body to eliminate half of an administered dose of any substance by regular processes of elimination; it is approximately the same for both stable and radioactive isotopes of a particular element.
Half-life, radioactive.
Time required for a radioactive substance to lose 50% of its activity by decay.
Histologic types.
Pathologists have identified 4 principal lung-cancers based upon microscopic analysis of cellular characteristics: squamous cell carcinoma, adenocarcinoma, small cell carcinoma, and large cell carcinoma.
I
Incidence.
Or incidence rate; the rate of occurrence of a disease within a specified period of time, often expressed as a number of cases per 100,000 individuals per year.
Inhalation.
To draw air into the lungs by breathing; considered an exposure route for radon and radon progeny.
In utero.
In the womb, i.e., before birth.
Inverse dose-rate effect.
An effect in which, for a given exposure, the probability of effect increases as the dose rate is lowered.
In vitro.
Refers to cell culture conditions in glass or plastic containers as opposed to in vivo, in the living individual.
In vivo.
In the living organism.
Ionizing radiation.
Radiation sufficiently energetic to dislodge electrons from an atom thereby causing an ion pair. Ionizing radiation includes x and gamma radiation, electrons (beta radiation), alpha particles (helium nuclei), and heavier charged atomic nuclei. Neutrons ionize indirectly by first colliding with components of atomic nuclei.
Isotopes.
Nuclides that have the same number of protons in their nuclei, and hence the same atomic number, but that differ in the number of neutrons, and therefore in the mass number; chemical properties of isotopes of a particular element are almost identical.
K
K factor.
A dimensionless parameter in the risk model that characterizes the comparative doses to lung cells for exposures in homes compared to similar exposures in mines.
L
Latent period.
The period of time between exposure and expression of the disease. After exposure to a dose of radiation, there is a delay of typically several years (the latent period) before any cancers are observed.
Life table.
A table showing the number of persons who, of a given number born or living at a specified age, live to attain successive higher ages, together with the numbers who die in each interval.
Lifetime relative risk (LRR).
The relative increment in lung-cancer risk resulting from exposure to an agent such as radon.
Linear energy transfer (LET).
Average amount of energy lost per unit track length.
Low LET radiations.
Light, charged particles such as electrons or x rays and gamma rays that produce sparse ionizing events far apart on the scale of a cellular nucleus.
High LET radiations.
Heavy, charged particles such as protons and alpha particles that produce dense ionizing events close together on the scale of a cellular nucleus.
Linear (L) model or relationship.
Also, linear dose-effect relationship; expresses the effect (e.g., mutation or cancer) as a proportional (linear) function of the dose.
Linear-quadratic (LQ) model.
Also, linear-quadratic dose-effect relationship; expresses the effect (e.g., mutation or cancer) as a function of two components, one directly proportional to the dose (linear term) and one proportional to the square of the dose (quadratic term). The linear term will predominate at lower doses, the quadratic term at higher doses.
Lognormal distribution.
When the logarithms of a randomly distributed quantity have a normal (Gaussian) distribution.
M
Mechanistic basis.
An explanation derived from a knowledge of the individual stages leading to an effect.
Meta-analysis.
A method for analyzing epidemiologic data based on grouping or pooling information obtained from several studies.
Mitigation.
The act of reducing radon concentrations in homes.
Model.
A schematic description of a system, theory, or phenomenon that accounts for its known or inferred properties and may be used for further study of its characteristics.
Monte Carlo Calculation.
The method for evaluation of a probability distribution by means of random sampling.
Mortality (rate).
The rate to which people die from a disease, e.g., a specific type of cancer, often expressed as deaths per 100,000 per year.
Multiplicative interaction model (MIM).
The assumption that the relative risk (the relative excess risk plus one) resulting from the exposure to two risk factors is the product of the relative risks from the two factors taken separately.
N
Neoplasm.
Any new and abnormal growth, such as a tumor; neoplastic disease refers to any disease that forms tumors, whether malignant or benign.
Never-smokers.
People who have not smoked cigarettes.
Nonstochastic.
Describes effects whose severity is a function of dose; for these, a threshold may occur; some examples of somatic effects believed to be nonstochastic are cataract induction, nonmalignant damage to skin, hematological deficiencies, and impairment of fertility.
Normal distribution.
Referring to the so-called ''bell-shaped curve" of randomly distributed quantities; also referred to as a "Gaussian distribution."
Nuclide.
A species of atom characterized by the constitution of its nucleus, which is specified by its atomic mass and atomic number (Z), or by its number of protons (Z), number of neutrons (N), and energy content.
O
Oncogenes.
Genes which encode the potential for cancer.
P
Person-gray.
Unit of population exposure obtained by summing individual dose-equivalent values for all people in the exposed population. Thus, the number of person-grays by 1 person exposed to 1 Gy is equal to that contributed by 100,000 people each exposed to 10 µGy.
Person-years-at-risk (PYAR).
The number of persons exposed times the numbers of years after exposure minus some lag period during which the dose is assumed to be unexpressed (minimum latent period).
Pooled analysis.
When data from more than one study is combined for evaluation.
Potential Alpha Energy Concentration (PAEC).
The concentration of potential alpha energy from radon progeny suspended in a volume of air. PAEC is measured in quantities of Jm-3 or Working Levels (1 WL = 2.08 x 10-5 Jm-3)
Prevalence.
The number of cases of a disease in existence at a given time per unit of population, usually 100,000 persons.
Probability of causation.
A number that expresses the probability that a given cancer, in a specific tissue, has been caused by a previous exposure to a carcinogenic agent, such as radiation.
Progeny.
The decay products resulting after a series of radioactive decay. Progeny can also be radioactive, and the chain continues until a stable nuclide is formed.
Projection model.
A mathematical model that simultaneously describes the excess cancer risk at different levels of some factor such as dose, time after exposure, or baseline level of risk, in terms of a parametric function of that factor. It becomes a projection model when data in a particular range of observations is used to assign values to the parameters in order to estimate (or project) excess risk for factor values outside that range.
Promoter.
An agent which is not by itself carcinogenic, but which can amplify the effect of a true carcinogen by increasing the probability of late-stage cellular changes needed to complete the carcinogenic process.
Protraction.
The spreading out of a radiation dose over time by continuous delivery at a lower dose rate.
Pulmonary interstitium.
The spacing between the linings in the structure of the lung.
Q
Quadratic-dose model.
A model which assumes that the excess risk is proportional to the square of the dose.
Quality factor (Q).
An LET-dependent factor by which the absorbed doses are multiplied to obtain (for radiation protection purposes) a quantity which corresponds more closely to the degree of the biological effect produced by x or low-energy gamma rays. Dose in Gy × Q = Dose equivalent in Sv.
R
Rad.
A unit of absorbed dose equal to 100 ergs of energy absorbed per gram of tissue. Replaced by the gray in SI units. 100 rad = 1 Gy. (see Units).
Radiation.
Energy emitted in the form of waves or particles by radioactive atoms as a result of radioactive decay.
Radioactivity.
The property of nuclide decay in which particles or gamma radiations are usually emitted.
Artificial radioactivity.
Man-made radioactivity produced by fission, fusion, particle bombardment, or electromagnetic irradiation.
Natural radioactivity.
The property of radioactivity exhibited by more than 50 naturally occurring radionuclides.
Radiogenic.
Caused by radiation.
Radioisotope.
A radioactive atomic species of an element with the same atomic number and usually identical chemical properties.
Radionuclide.
A radioactive species of an atom characterized by the constitution of its nucleus.
Radon.
A naturally occurring radioactive gas produced from uranium; decays to form radon progeny.
Radon progeny.
The radioactive products formed following the radioactive decay of radon; radionuclides which when inhaled can expose living cells to their emitted alpha particles.
Relative risk.
An expression of excess risk relative to the underlying (baseline) risk; if the excess equals the baseline risk the relative risk is 2.
Relative biological effectiveness (RBE).
An adjustment factor used to qualify an absorbed dose to account for its relative potential to do damage in biologic tissues. RBE is standardized to effects caused by x rays of a standard energy.
Rem.
(rad equivalent mammal); unit of dose equivalent. The unit of dose equivalent or "rem" is numerically equal to the absorbed dose in "rad" multiplied by the "quality factor" (see Quality factor), "relative biological effectiveness" (see Relative biological effectiveness), the distribution factor, and any other necessary modifying factor. 1 Rem = 0.01 Sievert.
Respiratory epithelium.
The cells lining the lung surfaces.
Retinoblastoma.
An eye tumor that is an example of an inherited malignant tumor with a dominant autosomal gene inheritance pattern.
Risk.
A chance of injury, loss, or detriment. A measure of the deleterious effects that may be expected as the result of an action or inaction.
Risk assessment.
The process by which the risks associated with an action or inaction are identified and quantified.
Risk coefficient.
The increase in the annual incidence or mortality rate per unit dose: (1) absolute risk coefficient is the observed minus the expected number of cases per person year at risk for a unit dose; (2) relative-risk coefficient is the fractional increase in the baseline incidence or mortality rate for a unit dose.
Risk estimate.
The number of cases (or deaths) that are projected to occur in a specified exposed population per unit dose for a specified exposure regime and expression period: number of cases per person-gray or, for radon, the number of cases per person cumulative working-level-month.
S
SI units.
The International Systems of Units as defined by the General Conference of Weights and Measures in 1960. These units are generally based on meter/kilogram/second units, with special quantities for radiation including the becquerel, gray, and sievert.
Sievert.
The SI unit of radiation dose equivalent. It is equal to dose in gray times a quality factor or times other modifying factors; for example, a distribution factor; 1 sievert (Sv) equals 100 rem. (see Units).
Specific Activity.
Total activity of a given nuclide per gram of a compound, element, or radioactive nuclide.
Specific energy.
The actual energy per unit mass deposited per unit volume in a
given event. This is a stochastic quantity as opposed to the average value over a larger number of instances (i.e., the absorbed dose).
Squamous cell carcinoma.
A cancer composed of cells that are scaly or platelike.
Standard mortality ratio (SMR).
Standard mortality ratio is the ratio of the disease or accident mortality rate in a certain specific population compared with that in a standard population. The ratio is based on 100 for the standard so that an SMR of 200 means that the test population has twice the mortality from that particular cause of death.
Stochastic.
Random events leading to effects whose probability of occurrence in an exposed population (rather than severity in an affected individual) is a direct function of dose; these effects are commonly regarded as having no threshold; hereditary effects are regarded as being stochastic; some somatic effects, especially carcinogenesis, are regarded as being stochastic.
Submultiplicative effects.
Effects less than the anticipated effect if the joint effect were the product of the risks from the two agents individually (but more than if the joint effect were the sum of the individual risks).
Suppressor gene.
A gene which can suppress another gene such as an oncogene. Changes in suppressor genes can lead to expression by genes such as oncogenes.
Synergistic.
An increased effectiveness resulting from an interaction between two agents so that the total effect is greater than the sum of the effects of the two agents acting alone.
T
Target cells.
Cells in a tissue that have been determined to be the key cells in which changes occur in order to produce an endpoint such as cancer.
Threshold hypothesis.
The assumption that no radiation injury occurs below a specified dose.
Time-since-exposure (TSE) model.
A model in which the risk is not constant but varies with the time after exposure.
Transfection.
The introduction of DNA into a host cell.
Transformed cells.
Tissue-culture cells changed from growing in an orderly pattern exhibiting contact inhibition to growing in a pattern more like that of cancer cells.
U
Uncertainty.
The range of values within which the true value is estimated to lie. It is a best estimate of possible inaccuracy due to both random and systemic errors.
Random Errors.
Errors that vary in a nonreproducible way around a limiting mean. These errors can be treated statistically by use of the laws of probability.
Systemic Errors.
Errors that are reproducible and tend to bias a result in one direction. Their causes can be assigned, at least in principle, and they can have constant and variable components. Generally, these errors cannot be treated statistically.
|
Units |
|
|
Unitsa |
Conversion Factors |
|
Becquerel (SI) |
1 disintegration/s = 2.7 × 10-11 CI |
|
Curie |
3.7 × 1010 disintegrations/s = 3.7 × 1010 Bq |
|
Gray (SI) |
1 J/kg - 100 rad |
|
Rad |
100 erg/g; also 0.01 Gy |
|
Rem |
0.01 Sievert |
|
Sievert (SI) |
100 Rem |
|
a International Units are designated SI. |
|
UNSCEAR.
United Nations Scientific Committee on the Effects of Atomic Radiation publishes periodic reports on sources and effects of ionizing radiation.
V
Variability.
The variation of a property or a quantity among members of a population. Such variation is inherent in nature and is often assumed to be random. However, it can sometimes be represented by a frequency distribution.
W
Working levels (WL).
Any combination of the short-lived progeny of radon in 1 liter of air, under ambient temperature and pressure, that results in the ultimate emission of 1.3 × 105 MeV of alpha particle energy. This is approximately the total amount of energy released over a long period of time by the short-lived progeny in equilibrium with 100 pCi of radon. 1 WL = 2.08 × 10-5 Jm-3
Working level months (WLM).
A cumulative exposure equivalent to one working level for a working month (170 hours). 1 WLM = 2 × 10-5 Jhm -3 × 170 h = 3.5 × 10-3 Jhm-3.
X
x radiation.
Also x rays; penetrating electromagnetic radiation, usually produced by bombarding a metallic target with fast electrons in a high vacuum.
Xeroderma pigmentosum (XP).
An inherited disease in which skin cells are highly susceptible to cancer; XP cells have a defect in DNA repair which results in the accumulation of DNA damage after ultraviolet irradiation which apparently accounts for the development of cancer.
Committee Biographies
Jonathan M. Samet, M.D., M.S., (Chairman), is presently professor and chairman of the Department of Epidemiology of the Johns Hopkins University School of Hygiene and Public Health. Dr. Samet received a Bachelor's degree in Chemistry and Physics from Harvard College, an M.D. degree from the University of Rochester School of Medicine and Dentistry, and a Master of Science in epidemiology from the Harvard School of Public Health. He is trained as a clinician in the specialty of internal medicine and in the subspecialty of pulmonary diseases. From 1978 through 1994, he was a member of the Department of Medicine at The University of New Mexico School of Medicine and he is presently co-director of the Risk Sciences and Public Policy Institute at the Johns Hopkins University School of Hygiene and Public Health. His research has addressed the effects of environmental agents on human health, including risks of lung-cancer and other diseases in uranium miners. He was a member of the BEIR IV Committee and chair of the Panel on Dosimetric Assumptions Affecting the Application of Radon Risk Estimates of the National Research Council. He has served on the Science Advisory Board for the U.S. Environmental Protection Agency. He has been a member of the Commission on Life Sciences and the Board on Radiation Effects Research and was elected to the Institute of Medicine in 1997.
David Brenner, Ph.D., is a professor of Radiation Oncology and Public Health at Columbia University, New York. He earned a M.Sc. from St. Bartholomews' Hospital, University of London, in Radiation Physics, a Ph.D. from the University of Surrey in Physics, and has been awarded an honorary D.Sc. degree by Oxford University. He is a radiation biophysicist focusing on mechanisms of
induction of DNA and chromosomal damage by different radiations. His research also involves modeling and analyzing low dose epidemiological studies, as well as the analysis and design of fractionation schedules for radiotherapy. He has authored books on Radiation, Risk and Remedy and Making the Radiation Therapy Decision as well as publishing around 120 papers in the peer-reviewed literature. He has served on various EPA, NAS, IAEA, and NCRP committees. He is a past recipient of the Radiation Research Society Annual Research Award, and the Robert D. Moseley Annual Award from the National Council on Radiation Protection and Measurements.
Antone L. Brooks, Ph.D., is currently working as a senior research scientist at Washington State University Tri-Cities. Formerly he was the Cell and Molecular Biology Group head at Lovelace Inhalation Toxicology Research Institute and then was the manager of the Cellular and Mammalian Biology section at Battelle Pacific Northwest Laboratories. He has conducted extensive research on the health effects of internally deposited radioactive materials with special emphasis on the induction of chromosome aberrations and cancer from low-dose and dose-rate exposures. His research has used cellular and molecular changes to relate exposure to dose and to determine the role of early cellular changes during the process of cancer induction. He has conducted research using radon in experimental animals and cellular systems and using biodosimetry, has defined the relationships that exist between exposure, dose, and alpha-particle traversals. A major goal of this research is to link mechanistic cellular and molecular biology studies to human-health risk.
William H. Ellett, Ph.D., is retired and lives in Crofton, Maryland. He was formerly a senior staff officer in the Board on Radiation Effects Research at the National Academy of Sciences. Dr. Ellett was study director for the BEIR IV and V Studies of the National Research Council.
Philip K. Hopke, Ph.D., is the R.A Plane Professor of Chemistry at Clarkson University. He holds a joint appointment in the Department of Civil and Environmental Engineering and was recently appointed as the Dean of the Graduate School. Dr. Hopke has been studying radon-progeny behavior since 1975 and has made major contributions to the understanding of the neutralization, deposition, and radiolytic formation of particles. His group has made assessments of radon decay-product exposure in normally occupied homes and the incremental effects of showering with radon-laden water. He has been a member of several National Research Council committees assessing the risk of exposure to hazardous air pollutants. He previously served as one of the principal scientists in the U.S. Department of Energy's Radon Research Program and was the General Chair of the Sixth International Symposium on the Natural Radiation Environment.
Ethel S. Gilbert, Ph.D., spent several years as a biostatistician and senior staff scientist at Battelle, Pacific Northwest Laboratories, but recently joined the Radiation Epidemiology Branch of the National Cancer Institute as a special expert. Her research has focused on epidemiologic studies of nuclear workers, which has included combined analyses of national and international data and the development of statistical methods for examining the relationship of health effects and low-level chronic radiation exposures. She has also analyzed data on experimental animals exposed to radon and to inhaled plutonium. Dr. Gilbert was a member of the working group responsible for revising the health effects model in the U.S. Nuclear Regulatory Commission's Reactor Safety Study, where she provided and updated a model for estimating cancer risks. Dr. Gilbert is a fellow of the American Statistical Association and a member of the National Council on Radiation Protection and Measurements.
Dudley T. Goodhead, Ph.D., heads the Radiation and Genome Stability Unit of the Medical Research Council (MRC) in the United Kingdom and he is an honorary professor of Brunel University. He was formerly deputy director of the MRC Radiobiology Unit. Dr. Goodhead earned his D.Phil. from the University of Oxford in high-energy physics and subsequently held academic appointments at the Universities of California, London, and Natal. His own research has been predominately in the field of the biophysics of radiation effects. Dr. Goodhead has served on the Task Group on the Biological Effectiveness of Neutrons of the Committee on Interagency Radiation Research and Policy Coordination, the Committee on Medical Aspects of Radiation in the Environment (UK), and other national and international committees involved with the biological effects of radiation. He has served as consultant to the United Nations Scientific Committee on the Effects of Atomic Radiation, the International Atomic Energy Agency, and the General Accounting Office of the U.S. Congress. He is Councillor for Physics of the International Association of Radiation Research and has served on the editorial boards of the Radiation Research Journal and the International Journal of Radiation Biology.
Eric J. Hall, Ph.D., is the Higgins Professor of Radiation Biophysics, professor of Radiology and Radiation Oncology, and director of the Center for Radiological Research. Dr. Hall received his M.A., D. Phil., and D.Sc. degrees in radiation biology from Oxford University. He is past-president of the Radiation Research Society and currently president-elect of the International Association of Radiation Research and Secretary of the American Radium Society. He is Senior editor for biology of the International Journal of Radiation Oncology Biology Physics. Dr. Hall has received the Gold Medal of both the Radiological Society of North America (1992) and the American Society of Therapeutic Radiology and Oncology (1993), the Janeway Medal of the American Radium Society as well as the Failla Medal of the Radiation Research Society (1991). Dr. Hall is a
member of the National Council for Radiation Protection and served on the BEIR V Committee.
Daniel Krewski, Ph.D., M.H.A., is the director of Risk Management, Health Canada, professor of epidemiology and biostatistics in the Department of Epidemiology and Community Medicine at the University of Ottawa, and adjunct research professor of statistics from Carleton University. Dr. Krewski earned his M.S. and Ph.D. in mathematics and statistics from Carleton University, and his M.H.A. from the University of Ottawa. He is associate editor of Risk Analysis, the Journal of Epidemiology and Biostatistics, and Regulatory Toxicology and Pharmacology. He is currently a member of the NRC Board on Environmental Studies and Toxicology and its Committee on Toxicology, and he recently chaired the NRC's Colloquium on Scientific Advances and the Future in Toxicologic Risk Assessment.
Jay H. Lubin, Ph.D., is a mathematical statistician in the Division of Cancer Epidemiology and Genetics at the U.S. National Cancer Institute. He holds a B.A. and a M.A. in mathematics from the University of California, Los Angeles, and a Ph.D. in Biostatistics from the University of Washington, Seattle. He has been a member of the Regional Advisory Board of the Biometrics Society and a recipient of the U.S. Public Health Service Special Recognition Award. He is currently associate editor of Biometrics. In 1994, Dr. Lubin was elected a Fellow of the American Statistical Association. His statistical research interests are the design and analysis of epidemiology studies. Dr. Lubin has conducted extensive epidemiology research into the etiology of cancer, particularly in the areas of occupational exposures and radiation, and of lung-cancer and exposure to radon and radon progeny.
Roger O. McClellan, D.V.M., serves as president of the Chemical Industry Institute of Toxicology located in Research Triangle Park, NC, a position he has held since 1988. He is well known for his work in the related fields of toxicology and risk assessment, especially concerning the potential human risks of airborne materials. McClellan has previously served as president of the Society of Toxicology and American Association for Aerosol Research. He is a Fellow of the Society for Risk Analysis and an elected member of the Institute of Medicine of the National Academy of Sciences. He has served in an advisory role to many public and private organizations including Chairmanship of the U.S. Environmental Protection Agency Clean Air Scientific Advisory Committee and the National Research Council Committee on Toxicology. McClellan is a strong advocate of the need to integrate data from epidemiological, controlled clinical, laboratory animal, and cell studies to assess human health risks of occupational or environmental exposures to chemicals.
Paul L. Ziemer, Ph.D., is professor and head of the School of Health Sciences, Purdue University, a position he has held since 1983. From August, 1990, to January, 1993, Dr. Ziemer took a leave of absence from Purdue to serve as the Assistant Secretary of Energy for Environment, Safety, and Health at the U.S. Department of Energy during the Bush Administration. He has held several positions at Purdue relating to health sciences, health physics, and bionucleonics. As head of the School of Health Sciences, Dr. Ziemer is administratively responsible for teaching and research programs in industrial hygiene, health physics, medical physics, environmental health, and medical technology. Dr. Ziemer has served as a health physicist at Oak Ridge National Laboratory, as a Radiological Physics Fellow at Vanderbilt University, and as a physicist at the U.S. Naval Research Laboratory in Washington, D.C. He is a Certified Health Physicist and has been national president of the Health Physics Society, president of the American Academy of Health Physics, and a member of the American Board of Health Physics.
Index
A
Absolute risk, 43-44, 64, 74, 75, 90, 134-135, 137, 164, 201, 226-228, 301
Action levels
EPA, 3, 13, 19, 24, 31, 94, 356, 357
Adenocarcinomas, 33, 239, 249, 250, 251, 346, 355, 386, 390, 392, 396, 398, 399, 402-410 (passim), 414, 415
Aerosol characteristics, general, 199, 203, 208, 214, 223
size distribution, 31, 180-181, 183-184, 311
smoking-generated, 252
Age factors, 31, 71, 82, 83, 88, 89, 102, 103, 104, 110, 137, 140, 141, 144, 167, 210, 212, 213, 255, 274
attained age, 8, 74, 75-76, 80-84 (passim), 110, 114, 134-139 (passim), 144, 147, 149, 151, 257-272 (passim), 275, 280, 281, 285, 286, 287, 376
diesel fumes as carcinogen, 352
ecologic studies, 360, 361, 366-360, 376
exposure-age-concentration, 8, 11, 12, 14, 15, 18, 25-26, 80-82, 84-87, 92-99 (passim), 105-109, 113, 115, 147-149, 151-154, 173, 175
exposure-age-duration, 8, 11, 12, 14, 15, 18, 25-26, 80-87, 92-99 (passim), 105, 106-109, 113, 147-149, 151-154, 174, 175
first exposure, age at, 134, 136, 138, 139, 140, 165, 254, 255, 256-275, (passim), 280, 282, 284, 285-286, 290, 292, 315, 317, 319, 322, 325
gender factors and, 12, 232, 233
inverse dose rate effect, 60, 61
lifetime relative risk, 110-112, 134-135, 167, 171
relative risk/excess relative risk, 8, 25, 81, 84, 87, 88, 90, 93, 110, 114, 134-135, 136, 138, 147, 151-154, 164-165, 280, 284
smoking, 95-96, 143, 226, 232, 233-234, 238, 239, 240, 371
see also Children;
Infants
Airways and airway diseases, 47, 50, 205, 208
American Cancer Society, 238, 239
diesel fumes as carcinogen, 351-354
extrapolation to human effects, 63, 243
genetic susceptibility, 38, 42, 43-44, 68, 200-201
mycotoxins as carcinogens, 355
nonmalignant respiratory diseases, 117
silica as carcinogen, 347
smoking-radon synergism, 7, 22, 62-63, 225, 242-243
Arsenic, 31, 32, 102, 114, 162, 254, 274, 289, 292, 335, 340, 344, 345-346
Asbestos, 25, 208, 262, 352, 369, 408
Atomic-bomb survivors, 7, 25, 27, 41, 63, 70, 72, 90, 135, 249, 251
Australia, Radium Hill, 7, 8, 122, 129, 138, 151, 255, 261, 268, 271, 286, 325, 326.
see also Multi-country data and analyses
Australian Radiation Laboratory, 186
B
Background exposure, 8, 10, 11, 92, 121, 136, 166, 236, 256, 361, 411
Basal cells, 36, 47, 48, 52, 57, 60, 118, 203, 259
Basements, houses with, 190, 357, 358, 363, 380, 381, 382, 386, 387, 399, 400, 401, 413, 415
BEIR IV report, 4, 5, 8, 23, 25, 62, 117-118, 120, 121, 124, 127, 133, 137, 138, 254, 255, 311, 315, 329, 379
BEIR VI model compared to, 8, 10, 11, 35, 69, 74, 76, 82, 91-95, 97, 110-113, 115, 136, 137, 251-252, 280, 281, 284, 285, 291, 332
smoking-radon synergism, 10, 94-95, 225, 231, 241-242, 251-252, 245, 250
BEIR V report, 158
Bias, see Uncertainty
Biologic processes, general
biologically-motivated models, 7, 27, 63-66, 70-71, 72, 140-143
exposure rate and carcinogenesis, 46, 49, 50, 58-62, 63, 66, 67, 142
two-stage clonal expansion, 27, 70, 71, 140, 141-143, 261
breathing rate, 176, 203, 204, 205, 210, 219, 220, 223
lung-cancer, 20-35 (passim), 36, 49-56
relative biological effectiveness, 25, 54, 55, 63-64, 118, 199
see also Animal studies;
Carcinogenesis and carcinogens;
Cells and cellular processes;
Molecular processes
Bladder cancer, 40, 123, 345, 395
Blasting fumes, 119, 194, 333-334, 337, 338, 342, 344
Blood
leukemias, 18, 38, 117, 119-127 (passim), 223
Bone and skeletal system
leukemias, 18, 38, 117, 119-127 (passim), 223
surfaces, 118
Breasts
cancer, 40, 42, 118, 127, 165, 249, 250, 360
doses to, 118
Breathing rate, 176, 203, 204, 205, 210, 219, 220, 223
Bronchi, 35, 37, 46, 47, 48, 52, 91, 118, 203, 207, 208, 209, 213, 236, 339, 347, 348-349, 395
Brownian diffusion, 203, 204, 205
Buccal cavity cancer, 121
Building materials, 22, 28, 127, 130, 177, 358, 380-382
Bureau of Mines, 311-313, 316, 334, 335
C
Canada
indoor studies, 398-401, 409-411, 412, 413, 415
miner cohorts, 8, 110, 121, 130, 136, 162, 222, 245, 254, 261-262, 263-267, 280, 286, 289, 283, 300-301, 315-322, 335, 337, 340-341, 348
see also Multi-country data and analyses
Carcinogenesis and carcinogens, general, 18, 36-68, 226
biologically-motivated risk assessment, 7, 27, 64, 70-71, 72, 140-143, 261
exposure, general, 46, 49, 50, 58-62, 63, 66, 67, 142
genetic susceptibility, 5, 10, 33, 37, 38-39, 41-44, 54, 64, 66, 68, 105
animal studies, 38, 42, 43-44, 68, 200-201
oncogenes, 33, 37, 38-39, 64, 66
monoclonal origin of cancer, 6, 37, 67, 88, 141
mycotoxins, 354-355
other than radon, 5, 22, 28, 31, 32-33, 102, 114, 162, 344-355;
see also specific substances
signatures, 7, 33-34, 68, 250-251
see also Animal studies;
Cells and cellular processes;
Molecular processes;
Risk assessments;
Smoking;
other specific carcinogens and carcinomas
Case-control studies, 5, 30, 73, 317-318, 356-357, 379-422, 427-428
BEIR VI models, methodology, 7, 28, 72, 78, 84, 85, 88, 114, 130, 250, 266
cohort studies, nesting of case-control studies, 78, 130, 250, 266
congenital abnormalities, 127, 128
diesel fumes as carcinogen, 354
direct measurement studies, 384-411
methodology, 7, 28, 72, 78, 84, 85, 88, 114
sampling issues, 426-427
simulation studies, 425-426
smoking synergism, 244-245, 250, 300-301
surrogate measurements, 244, 356, 361, 380-384, 413
uncertainties, 7, 30, 128, 159, 379-380, 384, 422, 423, 425-426
Cells and cellular processes, 5, 22, 28, 31, 34-35, 37, 44-60, 65-68, 88, 143, 199-200
basal cells, 36, 47, 48, 52, 57, 60, 118, 203, 259
biologically-motivated risk assessment, 7, 27, 64, 70-71, 72, 140-143
two-stage clonal expansion, 27, 70, 71, 140, 141-143, 261
cycle-cell effects, 44-45
diesel fumes as carcinogen, 349, 351, 353
in vitro studies, 6, 37, 39, 47, 57, 63-64, 67-68, 124
in vivo studies, 6, 47, 63-64, 67, 200, 241-243
monoclonal origin of cancer, 6, 37, 67, 88, 141
repair, 6, 30, 33, 34, 37, 44, 47, 50, 53, 60, 65, 66, 68, 134
proliferation, 45-46, 60, 64, 66, 68, 142, 143, 200, 245
secretory cells, bronchial, 35, 47, 48, 52, 118, 208
secretory cells, tracheal, 47-48
target cells, 34, 35, 41, 48-49, 57
see also Epithelial cells
Children, 3, 9, 10, 41, 89, 91, 104, 119, 126, 137, 210, 212, 213, 254, 256, 286
schools, 359
China
indoor studies, 389-391, 412, 413, 414, 416
miner cohort, 8, 77-78, 122, 130, 162, 222, 245, 247, 248, 254, 256-257, 286, 289, 295, 326, 331-332, 335, 355
see also Multi-country data and analyses
dose-response relationship, 54-55, 66, 199
genomic instability, 41
suppressor genes, 39-40
Cigarettes, see Smoking
Cohort studies, general, 141, 238
atomic-bomb survivors, 7, 25, 27, 41, 63, 70, 72, 90, 135, 249, 251
case-control data, nesting, 78, 130, 250, 266
methodological issues, 28, 71, 73, 79-80, 92, 122, 129, 144, 145, 147, 149, 152, 227-228, 254-275 (passim)
smokers, 227-228, 238-239, 249-251
see also Miner studies;
Multi-country data and analyses;
Pooled analyses;
specific countries
Colorado Plateau, miner cohort, 7, 76, 77, 78, 83, 110, 120, 130, 131, 132, 136, 137, 141, 142, 152, 194, 245, 247, 248, 259-261, 271, 283, 288-289, 292-293, 295, 299, 300, 306-314, 315, 338-339, 343.
see also Multi-country data and analyses
Commission of the European Communities, 116
Committee on the Biological Effects of Ionizing Radiation, see BEIR IV report
Compagnie General des Matieres Nucleaires, 62, 242
Constant relative risk models, 3, 13
Construction materials, see Building materials
Copper mining and smelting, 261, 342, 346
Czech Republic, miner cohort, 7-8, 78, 121, 130, 136, 162, 256, 258-259, 283, 286, 288-289, 300, 322-324, 342, 355.
see also Multi-country data and analyses
D
Decay, radioactive, general, 1, 4, 20-22, 177-180, 196, 210, 211, 222-223
Demographic factors, general, 16, 79, 361, 366-369, 374, 397, 400, 403
uncertainty, 16, 100, 101, 157, 158, 167
see also Age factors;
Gender factors;
Vital statistics
Department of Defense, 311
Department of Energy, 116
Descriptive models, see Empirical risk models
Diesel fumes and exhaust, 22, 32, 102, 119, 162, 274, 311, 334, 337, 342, 343, 344, 349-354, 355
cellular processes, 349, 351, 353
smoking synergies, 350, 352-354
Diffusional factors, see Brownian diffusion
Diseases and disorders, not lung-cancer, 18, 117-128
airways and airway diseases, 47, 50, 205, 208
malignancies, 117, 119, 120-127, 165, 347-349;
see specific types of cancers
bladder cancer, 40, 123, 345, 395
breast, 40, 42, 127, 165, 249, 250, 360
bronchial, 37, 39, 46, 47, 347
indoor studies, 119, 124, 125, 126, 127, 360
leukemias, 18, 38, 117, 119-127 (passim), 223
pancreatic cancer, 122, 123, 223, 395
pooled analyses, 94, 122, 127-128
reproductive outcomes, 117-118, 127
respiratory, nonmalignant, 117, 119-120, 233;
see also Fibrosis/fibroblasts;
Silica and silicosis;
Tuberculosis
see also specific diseases
DNA, 1, 33, 34, 40, 44, 50, 168
mycotoxins, 354-355
Dose and dosimetry, 5, 7, 25, 27, 50, 63-64, 70, 72-73, 88, 120, 135, 202-203, 293
atomic-bomb survivors, 7, 25, 27, 41, 63, 70, 72, 90, 135, 249, 251
biodosimetry, 198-202
breasts, 118
comparative, 176-223
K factor, 9, 10, 88, 91, 105-109, 144, 157, 165-166, 170-172, 175, 177, 210-219, 223, 284, 417
cycle-cell effects, 44-45
marrow, 126
gender factors in comparative dosimetry, 201, 210, 212, 213
organs other than lungs, 118-119
relative biological effectiveness, 25, 54, 55, 63-64, 118, 199
uncertainty, 16, 17, 27, 70, 73, 165-166, 177, 222-223
see Exposure
Dose-effect (dose-response) models, 6, 16, 18, 22, 199-200, 225, 226
carcinogenesis, general, 37-38, 44
chromosomes, 54-55
indoor measurement techniques, 130, 133, 180-189, 359
larynx cancer, 121
low doses of high-LET, 37, 65-67
linear model/relationship, 2, 5, 9, 66, 88, 293, 376
nonthreshold, 9, 34, 66, 99, 378, 374
linear-quadratic model/relationship, 133
miner studies, 2, 22, 114, 251, 255, 293, 296, 297, 324-325, 340
respiratory cells at risk, 46-48, 50
silica, 348
see also Inverse dose/exposure rate effect
Dust, 1, 22, 143, 274, 292, 327, 337, 343-343
see also Blasting fumes;
Silica and silicosis
E
Ecologic studies, 356, 359-379, 411
classification of, 360
diseases other than lung-cancer, 18, 124, 125, 126-127
gender factors, 361, 366-369, 372, 373
methodology, 359-361, 362-373, 374-379, 411
smoking, 357, 361, 366-371, 373, 376-379
biologically-based models and, 7, 27, 64, 70-71, 72, 140-143, 261
Environmental Protection Agency, vii-viii
National Residential Radon Survey, 11, 106, 115, 357-358
National Schools Radon Survey, 359
presence of indoor radon, 28, 190, 357, 359, 361
action levels, 3, 13, 24, 19, 31, 94, 356, 357
smoking synergism, 252
EPICURE program, 138, 147, 148
Epithelial cells, 6, 22, 41, 89, 199, 201, 209, 351
bronchial, 35, 47, 50, 57, 60, 63, 66, 118, 208, 209
tracheal, 47-48
secretory, 35, 47-48, 52, 118, 208
Equations
absolute risk, 226-227
airborne potential alpha energy concentration, 179
dosimetric factors, 177
exposure, 110, 142, 177, 178, 298
lung-cancer mortality/rate, 74
population attributable risk (AR), 169-171, 229-231
random-effects, 145, 146-147, 148
relative risk, 8, 75, 81, 137, 139-140, 144, 148, 168, 227, 298, 416-417
excess relative risk, 134, 135
synergistic/multiplicative risk, 169, 227, 228-229, 230-231
uncertainty, 105
Errors of measurement, 16, 168, 229, 424
case-control studies, exposure, 7, 30, 128, 159, 379, 422, 425-429
ecologic studies, 374-379
exposure, general, 13, 28, 162, 429
individual risk, 106
miner studies,
exposure, 16, 17, 80, 81, 100, 101-102, 103, 114, 115, 160-162, 290, 291-293, 295-305, 307-343
vital status data, 16, 100, 101, 133, 159-160
surrogate measurements, 380, 384, 413
see also Sampling;
Uncertainty
European Commission, see Commission of the European Communities
Excess relative risk, 7, 8, 25-26, 74, 75, 81, 84, 85, 88, 93, 95-96, 114, 129, 134, 136, 139, 147-155, 245, 250, 252
age factors, 8, 90, 114, 136, 151-154, 164-165, 280
BEIR VI models, 74, 81, 84, 85, 88, 93, 95-96, 114, 147-155, 283
exposure, 8, 91, 96, 114, 139, 152, 154, 280, 281, 282, 286, 289, 300-303, 346
Exposure
background exposure, 8, 10, 11, 92, 121, 136, 166, 236, 253, 361, 411
BEIR VI models, 8, 11, 16-17, 33, 74-84, 95-96, 110-113, 275-290 (passim)
cell proliferation and, 46
congenital abnormalities, 127
cumulative, 14, 16, 32, 34, 58, 60, 70, 71, 74, 75, 76, 82, 100, 103, 114, 123, 126, 129, 134, 135, 136, 138, 140-141, 143, 144, 148, 163, 170, 257-269 (passim), 271-272, 280, 283, 284-285, 302, 303
defined, 76
diseases other than lung-cancer, 18, 124, 126
dose relations, 9, 16, 20, 22, 28, 30, 71, 91, 100, 157, 176, 177-180, 202, 225
equations, 110, 142, 177, 178, 298
exposure distribution model, 9, 11, 19, 32, 35, 91, 93, 100, 101, 169, 190, 202, 230-231, 270, 281, 317, 422
uncertainty, 16, 100, 101, 105-106, 158
exposure rate, 9-10, 16, 28, 30, 81, 82, 110, 149, 287-289, 298, 303, 376
carcinogenesis and, biologic models, 46, 49, 50, 58-62, 63, 66, 67, 142
extrapolation from high to low, general, 6, 32, 67, 88, 103, 161-162, 284
inverse dose/exposure rate effect, 7, 9-10, 33, 58-62, 66, 67, 74, 84, 143, 274, 288, 305
low-level and risk, 2, 5, 6, 9-10, 19, 26, 27, 28, 31-32, 58-62, 67, 76, 88, 116, 284
excess, 8, 91, 96, 114, 139, 152, 154, 280, 281, 282, 289, 301
uncertainties, 16, 31, 100, 102, 103, 161, 162
exposure-response models, general, 16, 18, 22, 34, 37, 44, 71, 74-76, 80-81, 88, 91, 103, 133, 136, 137, 139, 140, 142, 149, 157, 159, 163-165, 292, 293, 296-299, 305, 332
first exposure, age at, 134, 136, 138, 139, 140, 165, 254, 255, 256-275, (passim), 280, 282, 284, 285-286, 290, 292, 315, 317, 319, 322, 325
indoor atmospheres, 2, 27-28, 124, 126, 130, 244-245, 356-429
linear models, 9, 32, 66, 74, 75, 84, 88, 99, 133, 136, 292, 293, 303, 376
nonthreshold, 6, 9, 32, 34-35, 66, 99, 116, 374, 378
miner studies, 1-2, 30, 74, 79, 100, 101-102, 120, 130, 191-196, 220, 222-223, 255-290 (passim), 291-355, 376
non-radon carcinogens, viii, 5, 22, 28, 31, 32-33, 102, 114, 162, 274-275, 344-355;
see also specific substances
quadratic dose models, 54, 66, 133
relative risk models, 84, 144, 236, 280, 283, 284, 298, 301-302, 303, 380
excess, 8, 91, 96, 114, 139, 152, 154, 280, 281, 282, 286, 289, 300 -303, 346
uncertainties, 16, 17, 28, 30, 31, 89-90, 100-106 (passim), 157, 158, 160-165, 284, 295-343
see also Dose and dosimetry;
Low-LET radiation;
High-LET radiation;
Standards;
Time factors
Exposure-age-concentration, 8, 11, 12, 14, 15, 18, 25-26, 80-87, 92-99 (passim), 113, 115, 147-149, 151-154
uncertainties, 105-109, 173, 175
Exposure-age-duration, 8, 11, 12, 14, 15, 18, 25-26, 80-87, 92-99 (passim), 105, 113, 147-149, 151-154
uncertainties, 106-109, 174, 175
F
Fibrosis/fibroblasts, 18, 38, 39, 44, 47, 53, 64, 117, 120, 128, 199-201, 289, 344.
see also Silica and silicosis
Fluorspar, miner studies (Newfoundland), 8, 77, 83, 102, 121, 138, 148, 152, 154, 156, 222, 247, 255, 262-263, 328, 340.
see also Multi-country data and analyses
Foreign countries, see International activities and perspectives;
Multi-country data and analyses;
specific countries
Formulas, see Equations
France, 7, 62, 78, 130, 162, 242, 269, 286, 324-325, 412.
see also Multi-country data and analyses
Fungi, see Mycotoxins
G
Gallbladder cancers, 121, 123, 127
Gamma radiation, 27, 39, 50, 64, 70, 177, 196, 200, 361, 364, 372
atomic-bomb survivors, 7, 25, 27, 41, 63, 70, 72, 90, 135, 249, 251
cell-cycle effects, 44
BEIR VI model, 31, 35, 87, 88, 91, 93, 100, 164, 167
comparative dosimetry, 201, 210, 212, 213
ecologic studies, 361, 366-369, 372, 373
lifetime relative risk, 11, 110-113, 167, 281
malignancies, not lung-cancer, 127
breast dose/cancer, 40, 42, 118, 127, 165, 249, 250, 360
miner studies, 382-394 (passim)
extrapolation to indoor assessment, 2-3, 6-7, 9, 10, 87, 88, 89, 90, 91, 100, 104, 135, 157
population attributable risk (AR), 13, 14, 15, 93, 106-108, 175
synergies,
multiplicative synergies, non-tobacco substances, 104
smoking, 3, 12, 87, 90, 97, 164, 215, 232, 234-235, 238, 239
signatures, 7, 33-34, 68, 250-251
suppressor genes, 39-40
susceptibility, 5, 10, 33, 37, 38-39, 41-44, 54, 64, 66, 68, 105
animal studies, 38, 42, 43-44, 68, 200-201
individual-level risk, 41-44, 68, 76
oncogenes, 33, 37, 38-39, 64, 66
smoking and, 42-43
see also Cells and cellular processes;
Chromosomes;
DNA;
Molecular processes
Glioblastomas, 40
Gold miners, 144, 261, 262, 319, 340, 342
Gravitational settling/displacement, particles, 203, 204, 205
H
High-LET radiation, 6, 49-51, 53-56
cell-cycle effects, 44-45
extrapolations from high to low, 37, 57, 64, 65-67, 65-66, 67
leukemia, 119
Historical perspectives, 20-23, 25-27
diesel fumes, 349-351
miner exposure, 1-2, 20, 193-194, 306-355 (passim)
smoking and lung-cancer, 231-236, 238, 252
Home exposure, see Indoor radiation exposure
I
Individual-level risk, 8, 34, 228, 266, 281, 287, 293-294, 301, 400, 405, 416
dosimetric factors, 157, 199, 205, 210
ecologic studies, 359, 360, 374, 375, 378, 379, 380
exposure estimates, 92, 161, 162, 314-325 (passim), 328, 337, 341, 348, 354, 375, 376, 380, 424, 425
lifetime relative risk and, 11, 167
lung morphometry, 205
uncertainty, 101, 105, 154, 161, 162, 165, 167, 168, 169, 171, 303, 304, 357, 424
genetic susceptibility, 41-44, 68, 76
synergistic effects, 10, 154, 156, 162, 190, 348, 354, 376
variability vs, 156, 157, 158, 168
see also Case-control studies
Indoor radiation exposure, 1, 4-5, 22, 31, 60, 73, 157, 163, 180-191, 214-222, 356-429
basements, 190, 357, 358, 363, 380, 381, 382, 386, 387, 399, 400, 401, 413, 415
bedrooms, 130, 387, 389, 390-409 (passim)
BEIR VI models, 5, 7-19, 28-29, 84, 85-100, 103-104, 114, 116
case-control studies, 5, 84, 85, 114, 379-422
cell-cycle effects, 44-45
ecologic studies, 356, 359-479, 411
diseases other than lung-cancer, 18, 124, 125, 126-127
gender factors, 361, 366-369, 372, 373
smoking, 357, 361, 366-371, 373, 376-379
living rooms, 389, 390, 392, 393, 396, 397, 405, 407, 413
malignancies, other than lung-cancer, 119, 124, 125, 126, 127, 360
miner exposure to, 166
National Residential Radon Survey, 11, 106, 115, 357-358
pooled analyses, 415-422
smoking in studies of, 232, 253, 357, 361, 366-371, 373, 376-379, 380, 381, 387-391, 394, 396, 399, 402, 403, 405, 409, 414, 415
sources and presence of, 22, 28, 106
building materials, 22, 28, 127, 130, 177, 358, 380-382
soil and rock, vii, 1, 28, 177, 357, 358, 362
surrogate measurements, 356, 361, 380-384, 413
uncertainty, 31, 106-109, 357, 360, 422-429
Inertial forces, particles, 203, 204-205
International activities and perspectives, 7-8, 20, 22, 30, 335, 358-359
standards, action levels, 31, 358
see also Multi-country data and analyses;
specific countries
International Agency for Research on Cancer, 346-347
International Commission on Radiological Protection, 25, 63, 75, 135, 137, 203, 207, 210, 212, 219, 220, 252, 358
Iron miners, see Sweden, miner cohort
Inverse dose/exposure rate effect, 7, 9-10, 33, 58-62, 66, 67, 74, 84, 143, 274, 288, 305
J
Japan, 127
atomic-bomb survivors, 7, 25, 27, 41, 63, 70, 72, 90, 135, 249, 251
K
Kidney doses, 118
L
in vitro studies, 6, 37, 39, 47, 57, 63-64, 67-68, 124
in vivo studies, 6, 47, 63-64, 67, 200, 241-243
see also Animal studies
Large cell carcinomas, 33, 239, 415
Leukemias, 18, 38, 117, 119-127 (passim), 223
Lifetime relative risk, 11, 12, 17, 63, 69, 85, 92, 115, 157, 162, 165, 170, 223, 281, 283, 421
age factors, 16, 110-112, 134-135, 167, 171
BEIR VI models, 11, 13, 17, 85, 92, 110-113, 157, 162, 281, 283
gender factors, 11, 16, 90, 110-113, 164, 167, 281
individual-level risk and, 11, 115, 167
smoking, 11, 12, 16, 90, 157, 281, 283
uncertainties, 16, 93, 100, 101, 103, 157, 167, 170-171
Linear energy transfer (LET), 199.
see also Low-LET radiation;
High-LET
radiation
Liver
doses, 118
Low-LET radiation, 6, 25, 49-51, 54-56, 70, 72, 159, 200, 201, 293
age at first exposure, 285
atomic-bomb survivors, 7, 25, 27, 41, 63, 70, 72, 90, 135, 249, 251
doses to organs other than lung, 119, 124
genetic susceptibility and, 42
Lymphocytes, 55, 118, 124, 126, 199
M
Measurement and measurement techniques
FISH-translocation, 126
indoor measurements, 130, 133, 180-189, 384-411 (passim), 424
surrogate, 356, 361, 380-384, 413
miner studies, 292, 296, 336-337, 340
personal dosimeters, 293, 324, 325
surrogate measurements, 244, 292, 296, 356, 361, 380-384, 413
see also Dose and dosimetry;
Equations;
Error of measurement;
Morphometry;
Uncertainty
Men, see Gender factors
Meta-analyses, see Pooled analyses
Mitigation measures, see Risk management
Miner studies, viii, 1-2, 28, 30, 114, 129, 291-355
BEIR VI risk assessment models, 7-8, 28, 76-85, 101-103, 110, 114, 116, 275-290
blasting fumes, 119, 194, 333-334, 337, 338, 342, 344
design and characteristics, 28, 71, 73, 76, 79-84, 92, 122, 129, 131-154, 227-228, 254-275 (passim), 291-355 (passim)
diseases other than lung-cancer, 18, 119-124
dose-effect (dose-response) models, 2, 22, 114, 251, 255, 293, 296, 297, 324-325, 340
excess relative risk, 110, 129, 134, 135, 136, 139, 147-155, 162, 303
exposure, 1-2, 30, 74, 79, 100, 101-102, 120, 130, 191-196, 220, 222-223, 255-290 (passim), 291-355, 376
gender factors, 382-394 (passim)
extrapolation to indoor assessment, 2-3, 6-7, 9, 10, 87, 88, 89, 90, 91, 100, 104, 135, 157
historical perspectives, 1-2, 20, 193-194, 306-355 (passim)
home exposure to radon, miners, 166
inverse dose rate effect, 58, 60, 61, 66-67, 305
leukemia, 119
Public Health Service, 259, 295, 306-307, 308, 313, 315, 335, 336
smoking, 2, 18, 30, 47, 79, 90-91, 103, 104, 114, 141, 142, 144, 150-154, 225, 243, 245-248, 274-275, 250-251, 254-255, 281, 283, 337
uncertainties, 16-18, 24, 30, 31, 81, 101-103, 109, 154-175 (passim), 291-294, 306, 307, 295-343
extrapolation to home exposures, 2-3, 4, 6-7, 9, 16-18, 31-32, 57, 73, 81-82, 84-85, 88-91, 119-120, 158, 176, 260, 274, 283, 349, 356, 422-423
diseases other than lung-cancer, 18, 119-120
heterogeneity issues, 79-80, 114, 145, 147, 159, 161, 168, 245, 249, 281, 285, 305
sampling variability, 16, 100, 101, 159, 313, 335
workshop, 307-343
see also Errors of measurement
ventilation, 180, 194, 219, 273, 292, 294, 305, 308, 311, 312, 314-317 (passim), 320-342 (passim)
see also Multi-country data and analyses;
Pooled analyses;
specific countries and minerals
Models, see Animal studies; Equations; Risk assessments
Molecular processes, 5, 22, 28, 31, 37, 44, 54
alpha particle damage, signatures, 7, 33-34, 68, 250-251
breast cancer, 42
see also Genetics
Monoclonal origin of cancer, 6, 37, 67, 88, 141
Monte Carlo calculations, 101, 106, 164, 168, 172, 175
Morphometry, 213
respiratory tract, 22, 35, 53, 205-209, 226
Multi-country data and analyses, 77, 83, 102, 131-132, 148, 152-156, 245-248, 271- 290, 414.
see also Pooled analyses
Mycotoxins, 354-355
N
National Cancer Institute, 138-140, 255, 256, 275, 280-283, 284, 288-290
National Council on Radiation Protection and Measurements, 25, 54, 74, 134-135, 209, 251-252
National Institute for Environmental Health Sciences, 295, 307
National Institute for Occupational Safety and Health, 295, 296, 307
National Radiation Protection Board (U.K.)186
National Residential Radon Survey, 11, 106, 115, 357-358
National Schools Radon Survey, 359
Neutron radiation, 73
atomic-bomb survivors, 7, 25, 27, 41, 63, 70, 72, 90, 135, 249, 251
New Mexico, miner cohort, 7, 120, 130, 137, 162, 194, 195, 245, 262-263, 280, 286, 294, 299, 314-315, 335-342.
see also Multi-country data and analyses
Nose and nasal cavity, 127, 201, 205, 207
O
Oncogenes, 33, 37, 38-39, 64, 66
P
Pancreatic cancer, 122, 123, 223, 395
Particle size, 31, 46, 91, 180-185, 190, 193-194, 196, 197-198, 202-203, 205, 208, 252, 340
Pharynx
particle disposition, 205
Pneumoconiosis, 120
Poisson methods, 57, 71, 110, 142, 144, 147, 149, 249, 290
Pooled analyses, 135-136, 138, 163, 255, 256, 270-290, 294-295, 301-303
BEIR committee approach, general, 5, 18, 19, 25, 27, 28, 74, 75, 78-80, 94, 110, 114, 143-154, 275-290
heterogeneity issues, 79-80, 114, 145, 147, 159, 161, 168, 245, 249, 281, 285, 305
indoor studies, 415-422
malignancies, other than lung-cancer, 94, 122, 127-128
Population attributable risk (AR), 11, 13-15, 93-100, 106-108, 156, 157, 175, 229
BEIR VI model, 11, 13-15, 17, 85, 86, 92, 93-100, 102, 106-109, 115, 128, 136, 157
gender factors, 13, 14, 15, 93, 106-108, 175
risk management and, 14, 97-100
smoking, 94-97
uncertainty, 105-109, 157, 168-175
see also Exposure-age-concentration model;
Exposure-age-duration model
Prospective studies
uncertainty, 133
Protection Against Radon-222 at Home and at Work, 25
Public Health Service, miner studies, 259, 295, 306-307, 308, 313, 315, 335, 336
Pulmonary fibrosis, 18
Q
Quadratic dose models, 54, 66, 133
R
Random-effects models, 79, 81, 130, 145-151, 421
Recommended Standard for Occupational Exposure to Radon Progeny in Underground Mines, 296
Regulatory measures, see Risk management;
Standards
Relative biological effectiveness, 25, 54, 55, 63-64, 118, 199
Relative risk, 3, 74, 76, 81, 84, 85, 138-139, 144, 227, 377
age factors, 8, 25, 81, 84, 87, 88, 90, 93, 110, 114, 134-135, 136, 138, 147, 151-154, 164-165, 280, 284
BEIR VI models, 74, 76, 81, 84, 85, 144, 156, 157
excess relative risk, 74, 81, 84, 85, 88, 93, 95-96, 114, 147-155, 283
lifetime relative risk (LRR), 11, 13, 17, 85, 92, 110-113, 157, 162, 281, 283
constant relative risk, 3, 13, 17, 18-19, 109
equations, 8, 75, 81, 137, 139-140, 144, 148, 168, 227, 298, 416-417
excess relative risk, 134, 135
exposure in, 84, 144, 236, 280, 283, 284, 298, 301-302, 303, 380
excess relative risk, 8, 91, 96, 114, 139, 152, 154, 280, 281, 282, 286, 289, 300-303, 346
indoor case-control studies, 379-382, 388-391, 394, 397-398, 401-403, 405-409, 413-423
uncertainties, 17, 18-19, 156, 157, 168, 422-423
see also Excess relative risk;
Lifetime relative risk
Reproductive outcomes, 117-118, 127
Residential exposure, see Indoor radiation exposure
Respiratory tract, general
anatomy/morphometry, 22, 35, 63, 205-209
breathing rate, 176, 203, 204, 205, 210, 219, 223
diseases, other than lung-cancer, 18, 117, 119-120, 233;
see also Fibrosis/ fibroblasts;
Silica and silicosis
dosimetry, 31, 180, 198-202, 205-209
nose and nasal cavity, 127, 201, 205, 207
see also Bronchi;
Cells and cellular processes;
Epithelial cells;
Larynx;
Trachea
Risk assessments, 4-5, 8-18, 69-116, 129-154
absolute risk, 43-44, 64, 74, 75, 90, 134-135, 137, 164, 201, 226-228, 301
BEIR VI models, 5, 7-19, 34-35, 69, 72-74, 76-116, 130, 143-154, 275-291
BEIR IV compared to, 8, 10, 11, 35, 69, 74, 76, 82, 91-95, 97, 110-113, 115, 136, 137, 251-252, 280, 281, 284, 285, 291, 332
excess relative risk, 74, 81, 84, 85, 88, 93, 95-96, 114, 147-155, 283
exposure-age-concentration, 8, 11, 12, 14, 15, 18, 25-26, 81-82, 84-87, 92-99 (passim), 113, 115, 147-149, 151-154
uncertainty, 105-109, 173, 175
exposure-age-duration, 8, 11, 12, 14, 15, 18, 25-26, 81-87, 92-99 (passim), 113, 147-149, 151-154
uncertainty, 105, 106-109, 174, 175
gender factors, 31, 35, 87, 88, 91, 93, 100, 164, 167
indoor/general population, 5, 7-19, 28-29, 84, 85-100, 103-104, 114, 116
lifetime relative risk (LRR), 11, 13, 17, 85, 92, 110-113, 157, 162, 281, 283
methodology, general, 2-3, 7-8, 23, 25, 27, 28-31, 76-84
miners, 7-8, 28, 76-85, 101-103, 110, 114, 116, 275-290
onon-radon carcinogens, 102, 289;
see also ''smoking" infra
population attributable risk (AR), 11, 13-15, 17, 85, 86, 92, 93-100, 102, 106-109, 115, 128, 136, 157
rationale, 34, 43, 69, 70, 72-74
relative risk, 74, 76, 81, 84, 85, 144, 156, 157
smoking, 10, 15, 18, 79, 88, 89, 90-91, 103, 104, 114, 144, 150-154, 281, 283
biologically-motivated models, 7, 27, 64, 70-71, 72, 140-143
exposure rate and carcinogenesis, 46, 49, 50, 58-62, 63, 66, 67, 142
two-stage clonal expansion, 27, 70, 71, 140, 141-143, 261
exposure-age-concentration, 8, 11, 12, 14, 15, 18, 25-26, 80-87, 92-99 (passim), 105-109, 113, 115, 147-149, 151-154, 173, 175
exposure-age-duration, 8, 11, 12, 14, 15, 18, 25-26, 80-87, 92-99 (passim), 105, 106-109, 113, 147-149, 151-154, 174, 175
International Commission on Radiological Protection, 25, 63, 75, 135, 137, 203, 207, 210, 212, 219, 220, 252, 358
mechanistic factors, 5-7, 36-68, 88
miner studies, extrapolation to home, 2-3, 4, 6-7, 9-10, 57, 91, 128, 176-223, 283
National Cancer Institute, 138-140, 255, 256 275, 280-283, 284, 288-290
previous, 57, 70-76, 130, 133-154, 251-252
BEIR IV, 8, 10, 11, 35, 69, 74, 76, 82, 91-95, 97, 110-113, 115, 136, 137, 251-252, 280, 281, 284, 285, 291, 332
International Commission on Radiological Protection, 25, 63, 75, 135, 137, 203, 207, 210, 212, 219, 220, 252, 358
National Cancer Institute, 138-140, 255, 256 275, 280-283, 284, 288-289
random-effects models, 79, 81, 130, 145, 147-151, 421
synergistic effects,
definitional issues, 226-229
non-tobacco substances, viii, 5, 22, 28, 31, 32-33, 102, 114, 162, 344-355
smoking, 2-3, 7, 10, 22, 24, 33, 62, 71, 73, 90-92, 103, 114, 130, 152, 225
see also Case-control studies;
Ecologic studies;
Empirical risk models;
Excess relative risk;
Individual-level risk;
Lifetime relative risk;
Population attributable risk;
Relative risk;
Uncertainty
Risk management, 19, 22, 24, 28-29, 97-100, 337
population attributable risks and, 14, 97-100
see also Action levels;
Standards;
Ventilation
S
Salivary gland cancer, 121, 127
Sampling
indoor studies, 130, 133, 180-189, 244-245, 375, 426-427, 429
miner studies, variability, 16, 100, 101, 159, 313, 335
Scandinavia, 22.
see also Finland;
Sweden
Secretory cells and processes
bronchial, 35, 47, 48, 52, 118, 208
tracheal, 47-48
SEER program, 239-240
Sex differences, see Gender factors
Silica and silicosis, 31, 32, 102, 117, 119-120, 162, 264, 274, 289, 340, 342, 344, 346-349, 355
Sister chromatid exchange, 48-49
Skin, 37, 42, 47, 118-119, 123, 124, 128, 259, 345, 349
Small cell carcinomas, 33, 40, 47, 239, 249, 250-251, 373, 386, 390, 392, 396, 398, 399, 405, 407, 415
Smoking, 2, 5, 10, 15, 22, 23, 24, 31, 88, 89, 190-191, 210, 214-219, 224-253
aerosol characteristics, 252
age factors, 95-96, 143, 226, 232, 233-234, 238, 239, 240, 371
animal studies, 7, 22, 62-63, 225, 242-243
BEIR IV models, 10, 94-95, 225, 231, 241-242, 251-252, 245, 250
BEIR VI models, 10, 15, 18, 79, 88, 89, 90-91, 103, 104, 114, 144, 150-154, 281, 283
cohort studies, general, 227-228, 238-239, 249-251;
see also Miner studies
cumulative exposure, 243, 247, 248
diesel fumes and, 350, 352-354
ecologic studies, 357, 361, 366-371, 373, 376-379
ever smokers, 3, 12, 32, 35, 47, 87, 91, 93, 95-98, 104, 115, 135, 152, 155, 156, 157, 159, 165, 225, 226, 228, 239-241, 252, 281, 283, 381, 394, 405, 415
gender factors, 3, 12, 87, 90, 97, 164, 215, 232, 234-235, 238, 239
genetic susceptibility to lung-cancer and, 42-43
historic trends, 231-236, 238, 252
indoor radon exposure studies, 232, 253, 357, 361, 366-371, 373, 376-379, 380, 381, 387-391, 394, 396, 399, 402, 403, 405, 409, 414, 415
lifetime relative risk, 11, 12, 16, 90, 157, 281, 283
miner studies, 2, 18, 30, 47, 79, 90-91, 103, 104, 114, 141, 142, 144, 150-154, 245-248, 274-275, 281, 283, 337
malignancies, other than lung-cancer, 127
never-smokers, 3, 12, 16, 19, 32, 35, 87, 91, 93, 95-98, 104, 115, 124, 135, 152, 155, 156, 157, 159, 165, 225, 226, 228, 232-233, 236, 238-241, 248, 250, 252, 281, 283, 382, 394, 415
particle characteristics, 180
passive, 183, 252, 254-255, 252
population attributable risk, 94-97
synergistic effects, 2-3, 7, 10, 22, 24, 33, 62, 71, 73, 90-92, 103, 129-130, 135, 136-137, 152, 154, 224-232, 242-245, 248, 274-275, 225-254, 281, 344, 351-354, 376
animal studies, 7, 22, 62-63, 225, 242-243
BEIR IV report, 10, 94-95, 225, 231, 241-242, 251-252, 245, 250
case-control studies, 244-245, 250, 300-301
EPA, 252
gender factors, 3, 12, 87, 90, 97, 164, 215, 232, 234-235, 238, 239
uncertainties, 2-3, 7, 10, 12, 17, 19, 22, 24, 28, 32, 33, 35, 42-43, 62, 71, 87, 89, 90-92, 97, 103, 104, 154, 159, 163, 165, 225, 232, 234-235, 238, 239, 346
Soil and rock, indoor impacts of radon-contaminated, 1, 28, 177, 357, 358, 362
Sputum cytology, 37
Squamous cell carcinomas, 33, 239, 250, 251, 355, 386, 390, 392, 396, 398, 399, 405, 415
Standards
action levels,
EPA, 3, 13, 19, 24, 31, 94, 356, 357
BEIR recommendations, other, 115, 128
historical perspectives, 306, 308
state, 308
Stochastic processes, 168
alpha particles, 88
carcinogenesis, 140
Stomach cancer, 122
Surgeon General, 225
case-control studies, 244, 356, 361, 380-384, 413
indoor exposure, 356, 361, 380-384, 413
Sweden
indoor studies, 380, 391-398, 412, 413-415, 416
miner cohort, 8, 77, 83, 110, 131, 136, 256, 262, 286, 329-330, 342
T
Target cells, 34, 35, 41, 48-49, 57
Temperature factors
smoking, 180
Temporal factors, see Time factors
mines, 222-223
Time factors, 25, 80-85 (passim), 284, 342
duration of exposure, 8, 27, 60, 61, 82, 84, 85, 88-89, 92, 110, 114, 120, 134, 140-141, 144-145, 147, 161, 178, 200, 257-282 (passim), 287-289, 292-293, 303
indoor exposures, 359, 385, 388, 390, 397, 402, 404, 405, 424, 427
malignancies, other than lung-cancer, 122
non-malignant lung disease, 120
see also Exposure-age-duration models
inverse dose/exposure rate effect, 7, 9-10, 33, 58-62, 66, 67, 74, 84, 143, 274, 288, 305
schools, 359
time since exposure, 8, 81, 82, 83, 96, 102, 103, 110, 134, 135, 138, 140, 148, 150, 163-164, 275, 280, 284, 286-287, 298, 299, 376.
see also Age factors
Tin miners, 8, 77, 121-122, 130, 131, 247, 248, 256-257, 285, 326.
see also China, miner cohort
Tobacco, see Smoking
Trachea, 46, 47-48, 50, 199, 201, 202, 205, 208, 340, 349
Two-stage models, 27, 70, 71, 79, 81, 82, 130, 140, 141-143, 145, 147-152 (passim), 156, 245, 261
U
Uncertainty, 5, 6, 16-19, 23-24, 31-34, 70, 115, 130, 133, 154-175, 283, 284, 422-427
case-control studies, 7, 30, 128, 159, 379-380, 384, 422, 423, 425-426
confidents limits/intervals, 19, 30, 101, 108-109, 114, 115, 121, 123, 127, 156, 159, 350, 388, 391, 394, 397, 401, 403, 407, 421
constant relative risk, 17, 18-19, 109
demographic factors, general, 16, 100, 101, 157, 158, 167
diagnostic/vital status errors, 16, 100, 101, 133, 160, 167, 233-236, 237, 253
dosimetry, 16, 17, 27, 70, 73, 165-166, 177, 222-223
ecologic studies, 359, 374-379, 384
exposure, 16, 17, 28, 30, 31, 89-90, 100-106 (passim), 157, 158, 160-165, 284, 295-343
exposure-age-concentration model, 105-109, 173, 175
exposure-age-duration model, 106-109, 174, 175
genes, suppressor, 39-40
genetic susceptibility, 5, 10, 33, 37, 38-39, 41-44, 54, 64, 66, 68, 105
animal studies, 38, 42, 43-44, 68, 200-201
oncogenes, 33, 37, 38-39, 64, 66
individual-level risk, 41-44, 68, 76
smoking and, 42-43
individual-level risks, 101, 105, 154, 161, 162, 165, 167, 168, 169, 171, 303, 304, 357, 424
genetic susceptibility, 41-44, 68, 76
synergistic effects, 10, 154, 156, 162, 190, 348, 354, 376
variability vs uncertainty, 156, 157, 158, 168
lifetime attributable risk, 93, 157, 170-171
lifetime relative risk, 16, 93, 100, 101, 103, 157, 167, 170-171
miner studies, 16-18, 24, 25, 30, 31, 75, 81, 101-103, 109, 154-175 (passim), 291-294, 306, 307, 295-343
extrapolation to home exposures, 2-3, 4, 6-7, 9, 16-18, 31-32, 57, 73, 81-82, 84-85, 88-91, 158, 176, 260, 274, 283, 349, 356, 422-423
diseases other than lung-cancer, 18, 119-120
heterogeneity issues, 79-80, 114, 145, 147, 159, 161, 168, 245, 249, 281, 285, 305
sampling variability, 16, 100, 101, 159, 313, 335
workshop, 306-343
Monte Carlo calculations, 101, 106, 164, 168, 172, 175
qualitative, 16, 100-104, 154-167
quantitative, 16, 43, 100, 104-110, 156, 162, 167-169
parameters, general, 16, 17, 78, 79, 100, 101-103, 106, 108, 109, 156-162, 168, 172
population attributable risk (AR), 105-109, 157, 168-175
prospective studies, 133
relative risk, 17, 18-19, 156, 157, 168, 422-423
sampling variability, miner studies, 16, 100, 101, 159, 313, 335
synergistic effects, 2-3, 7, 10, 12, 17, 19, 22, 24, 28, 32, 33, 35, 42-43, 62, 71, 87, 89, 90-92, 97, 103, 104, 154, 159, 163, 165, 225, 232, 234-235, 238, 239, 346
gender factors, non-tobacco substances, other, 104
smoking, 3, 12, 87, 90, 97, 164, 215, 232, 234-235, 238, 239
non-tobacco substances, viii, 5, 22, 28, 31, 32-33, 102, 344-355
smoking, 2-3, 7, 10, 12, 17, 19, 22, 24, 28, 32, 33, 35, 42-43, 62, 71, 87, 89, 90-92, 97, 103, 104, 154, 159, 163, 165, 225, 232, 234-235, 238, 239, 346
variability vs, 156, 157, 158-159, 168
vital statistics, use of, 16, 133, 159-160
see also Error of measurement;
Gender factors
Underground mining, see Miner studies
United Kingdom, 119, 121, 122, 124, 125, 126, 141, 142-143, 358, 412
Uranium, general, 1, 4, 56, 47, 77, 127, 130, 131, 132, 141-143, 177, 289, 293, 296, 300-301, 311, 335, 358
mycotoxins, 354-355
non-malignant respiratory diseases, 18, 117, 120
see various cohorts (e.g., Australia, Canada, Colorado Plateau, etc.)
Uranium Frenzy, 341
Urban areas, 361, 366-369, 391-394, 397-401
V
Vanadium, 194, 306, 308, 333, 334, 342, 343
Ventilation
mines, 180, 194, 219, 273, 292, 294, 305, 308, 311, 312, 314-317 (passim), 320-342 (passim)
Vital statistics, 231-236, 280, 359
population attributable risk using, 13
uncertainty and, 16, 133, 159-160
W
Water
arsenic-containing, 345
radon-containing, 28, 198, 293, 328, 329, 357, 358, 361, 362, 364, 366
Women, see Gender factors
X
Xeroderma pigmentosum, 42
There was a problem loading page 501.
There was a problem loading page 502.
There was a problem loading page 503.
There was a problem loading page 504.
There was a problem loading page 505.
There was a problem loading page 506.
There was a problem loading page 507.
There was a problem loading page 508.
There was a problem loading page 509.
There was a problem loading page 510.
There was a problem loading page 511.
There was a problem loading page 512.
There was a problem loading page 513.
There was a problem loading page 514.
There was a problem loading page 515.
There was a problem loading page 516.
There was a problem loading page 517.
There was a problem loading page 518.
There was a problem loading page 519.
There was a problem loading page 520.
There was a problem loading page 521.
There was a problem loading page 522.
There was a problem loading page 523.
There was a problem loading page 524.
There was a problem loading page 525.
There was a problem loading page 526.
There was a problem loading page 527.
There was a problem loading page 528.
There was a problem loading page 529.
There was a problem loading page 530.
There was a problem loading page 531.
There was a problem loading page 532.
There was a problem loading page 533.
There was a problem loading page 534.
There was a problem loading page 535.
There was a problem loading page 536.
There was a problem loading page 537.
There was a problem loading page 538.
There was a problem loading page 539.
There was a problem loading page 540.
There was a problem loading page 541.
There was a problem loading page 542.
There was a problem loading page 543.
There was a problem loading page 544.
There was a problem loading page 545.
There was a problem loading page 546.
There was a problem loading page 547.
There was a problem loading page 548.
There was a problem loading page 549.
There was a problem loading page 550.
There was a problem loading page 551.
There was a problem loading page 552.
There was a problem loading page 553.
There was a problem loading page 554.
There was a problem loading page 555.
There was a problem loading page 556.
There was a problem loading page 557.
There was a problem loading page 558.
There was a problem loading page 559.
There was a problem loading page 560.
There was a problem loading page 561.
There was a problem loading page 562.
There was a problem loading page 563.
There was a problem loading page 564.
There was a problem loading page 565.
There was a problem loading page 566.
There was a problem loading page 567.
There was a problem loading page 568.
There was a problem loading page 569.
There was a problem loading page 570.
There was a problem loading page 571.
There was a problem loading page 572.
There was a problem loading page 573.
There was a problem loading page 574.
There was a problem loading page 575.
There was a problem loading page 576.
There was a problem loading page 577.