Chapter 1
Life-Cycle Thinking for Wood and Paper Products
Bernard Yaros
Scott Paper Company
Life-cycle analysis examines the physical behavior of industrial systems. The fate of raw materials is traced from their extraction from the earth to their final disposal. All materials, including packaging materials, must be accounted for. Therefore, even when paper and board products do not form the central materials flow, it is impossible to analyze systems in which paper, board, and wood products are not involved. It is therefore important to have a logical and consistent method for life-cycle analysis for these products; this chapter discusses factors to consider.
When the idea of life-cycle analysis was developed in the early 1970s, it emphasized the use of energy and raw materials in inorganic systems; and the analyses gave little, if any, mention of emissions to air or water. The starting boundary for such systems was defined as the point at which materials are taken from the earth (through mining or oil and gas extraction, for example), and because minerals, oil, and gas are inert, their ultimate disposal to a landfill meant that any effects after landfilling could be ignored.
When paper and board products were incorporated into life-cycle analyses of industrial systems, their treatment was essentially ad hoc. The starting boundary usually was placed at the felling of the timber, and, as with inorganic systems, the finishing boundary was placed at the point at which the paper was placed in a landfill. There was relatively little energy recovery from the incineration of paper, so this, too, posed few perceived problems. By treating wood products in this way, the carbon source did not enter into the analysis, and the fate of the carbon after disposal was similarly excluded from the system.
The handling of wood feedstock (the energy content of the wood) was, however, more cavalier. Wood and wood products clearly contain bound-in energy.
They can be burned and the energy reclaimed. Some analysts included it in the same way as they did the feedstock energy associated with synthetic polymers. Others omitted it. Several arguments were advanced for omitting this component of energy, but most owed more to value judgments and interpretation than to science. They ranged from the simple sentiment that "omitting makes the numbers lower," through the more sophisticated "the energy is free and should not be included," to the pseudoeconomic "wood is not a commercial fuel and so should not be treated as though it were one."
The problem with omitting wood feedstock from calculations is that it leads to the absurd conclusion that some processing steps are net producers of energy. For example, if paper in municipal waste is incinerated and energy is recovered, then the incinerator is a net producer of energy from an energy-free feed.
Today, however, life-cycle analyses are done with an increasing degree of sophistication, and many of the systems involve not only wood but other biologic products. Furthermore, the system boundaries are being pushed back to the initial planting of the tree as a seedling (in some cases even nursery operations are included, although some seedlings occur naturally) through to the effect of any products after they have been landfilled. It is therefore essential to take a consistent approach to analyzing all biologic systems and to account for all the potential burdens that could influence the impact of their products.
Wood and Paper
Wood is a complex structure consisting of four main components: cellulose, hemi-cellulose, lignin, and extractives. The relative proportions of these components vary from one species to another, but Table 1-1 shows typical percentages.
Cellulose is a carbohydrate polymer with an empirical formula (C6H10 O5)x. Note that hydrogen and oxygen are present in the same proportions as in water—hence cellulose is a carbohydrate. Hemi-cellulose is a polymer mixture of carbohydrates, principally glucose, mannose, and galactose (isomers of C6H12O6) and xylose and arabinose (isomers of C5H10O5). Lignin is an extremely complex polymer structure with repeat units typically having the empirical formula C178H200O70. The extractives are low-molecular-weight compounds, principally
TABLE 1-1 Typical composition of wood
|
Component |
Weight % |
|
Cellulose |
45 |
|
Hemi-cellulose |
25–35 |
|
Lignin |
21–25 |
|
Extractives |
2–8 |
terpenes, phenols, fatty acids, and, in softwoods, resin acids; they are called extractives because they can be readily extracted with neutral solvents.
For paper making, the best fibers are those that consist of cellulose with substantial quantifies of hemi-cellulose, but with all of the lignin and extractives removed. Paper and board substrates can therefore be regarded as carbohydrates that contain 45 percent carbon by weight.
The Carbon Cycle
A tree absorbs atmospheric CO2, water, and sunlight for conversion to the materials we call wood and bark. After felling, the wood is treated to liberate cellulose fibers and remove lignin and extractives. In the course of this process, most of the waste products—and increasingly, the bark—are burned to generate some or all of the steam needed in the process. The fibers are then converted to paper, which, in an integrated process will again use some of the energy derived from waste wood products. The result of burning the paper products is that some of the CO2 that was fated when the tree was growing is liberated to the atmosphere. After use, the consumer can either burn the paper, liberating more of the fixed CO2, or send the waste for disposal with other rubbish. At final disposal, the waste could be incinerated, liberating the remaining fixed CO2. It could be sent to a landfill, where it decomposes aerobically, to liberate CO2, or anaerobically, to liberate hydrocarbons, which, after decomposition in the atmosphere are converted to CO2. The sequence is shown in Figure 1-1.
If trees are continuously replaced as they are felled, all of the carbon in the system is recycled eventually; that is, carbon is a constantly cycling burden. Within the system there is a time mismatch. A tree might take 50–100 years to reach the felling stage. Its conversion into paper can occur within a matter of months from felling, and its use and disposal also could be measured in months. If, after disposal, the waste products are hydrocarbons, the rate of decomposition in the atmosphere is unknown, but it probably is tens if not hundreds of years. If,
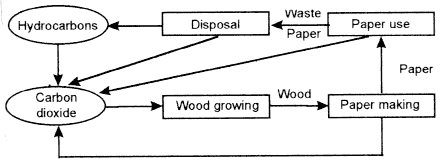
Figure 1-1 Materials flows in the wood and paper products system.
however, the system is operated so that the rate of replacement of timber mass by planting and growing at least matches the rate of extraction, then the system would exhibit a carbon equilibrium apart from any hydrocarbon emissions; this is discussed later. This equilibrium system, on a global scale, will be relatively insensitive to the rate of extraction, provided that the rate of extraction is small compared with the total mass of standing timber.
Starting Boundary
Whether the consumption of CO2 by a tree as it grows should be included in a life-cycle analysis depends on where the starting boundary is drawn. If it is placed at the point of felling the tree, then the absorption of CO2 would be omitted. If however, the boundary is placed at the point at which the tree is planted, then CO2 absorption must be included. In a true life-cycle, the starting point would most obviously be placed at the point of planting, in which case the inputs to the system will be CO2, water, and any other nutrients; the wood is simply an internal flow within the system boundary.
To be consistent, if the emission of CO2 during the processing of wood products is recorded, the corresponding input of CO2 to the system must be considered at the growing stage. Otherwise, the simple carbon balance will be violated; that is, the system would be acting as a generator of carbon atoms,
Energy Balance
Only a small fraction of the sun's energy falling on a tree is absorbed and used. Nevertheless, what is absorbed is used to fix the carbon, which manifests itself as the energy content of the wood. This feedstock energy depends on the type of tree and whether the wood is oven dried or air dried. Typical ranges are shown in Table 1-2.
Feedstock energy must be included in energy calculations. It is, however, important to keep its contribution to the overall total energy separate from other contributions—in the same way that electrical energy is kept separate from the contribution of fossil fuels. It is also important to recognize that, within the
TABLE 1-2 Typical ranges for wood calorific values
|
Wood type |
Drying method |
Calorific value (MJ/kg) |
|
Softwood |
Air dried |
14–18 |
|
|
Oven dried |
17–23 |
|
Hardwood |
Air dried |
13–14 |
|
|
Oven dried |
17–19 |
paper-processing sequence, some of the feedstock will be converted to fuel—as when waste products of pulping are used to generate steam.
The inclusion of wood feedstock energy as a separate contribution to the overall energy total is particularly important when examining integrated pulp and paper plants and when judging the efficacy of paper-recycling processes. By including wood feedstock in calculations for integrated pulp and paper plants, total energy might not be reduced compared with nonintegrated plants; indeed, it might actually be increased because of the lower conversion efficiencies of woodfueled boilers. The critical factor is not total energy but the amount of fossil fuel energy that is put into the system. When judged in terms of fossil fuel input, integrated plants usually show significant benefits because fossil fuels are displaced by wood products.
In paper-recycling processes, the option to use waste wood products does not occur on any significant scale. There is occasionally some burning of sludges to generate steam, but the calorific value of such sludges is low. All the energy input to the system must be fossil fuel based. Thus, even if the total energy for the production of recycled paper is less than that for the production of virgin paper, the increased use of fossil fuels in recycling processes usually means that virgin production should be favored because of its lower use of fossil fuels.
Much of the controversy over whether wood feedstock should be included in calculations centers on the total energy of the system. Energy should not be reported as a single value; the contributions of different fuels and feedstocks are far more important than is the total. When judging the efficiency of a process it is the quality of the energy used rather than the amount of energy used that is important. Renewable wood energy is preferable to nonrenewable fossil fuel energy.
Paper in Landfill
The precise fate of paper deposited in a landfill is not fixed. Depending on the nature of the landfill, the paper could decompose entirely in a relatively short time or it could remain intact for a long period. This depends on such factors as temperature, acidity, the presence of bacteria and nutrients, the composition of the waste, and the form of the paper—shredded paper would decompose faster than would an intact telephone book.
The basic decomposition reaction for cellulose is well known:
C6H10O5 + H2O = 3CH4 + 3CO2 (1)
Only half of the carbon present in paper will result in methane formation during decomposition. Typically, carbon constitutes 45 percent of the mass of paper. Thus, the carbon content of 1 kg of paper will be 0.45 kg, and that giving rise to methane, assuming 100 percent decomposition, will be 0.225 kg. The mass of methane produced will be 0.30 kg and the corresponding mass of the coproduct CO2 will be 0.83 kg. This will not necessarily be emitted to air; if a
landfill emits significant quantities of methane, the gas usually would be collected for use as fuel.
Interest in this form of decomposition arises because of the potential greater global warming effects of methane compared with CO2. We can gain some insight about this effect by using a global warming potential of 11 for methane (based an a 100-year time horizon). With 100 percent decomposition, as in Equation 1 above, the CO2 equivalent of the gases emitted is 4.11. If all of the carbon is emitted as CO2—as would occur with incineration—the CO2 equivalent is 1.66; that is, about half of the decomposition value. Clearly, incineration is preferable to decomposition, and given the potential for energy recovery at incineration, there is a strong case to be made for burning paper products.
Summary
When dealing with wood products in life-cycle inventories, there are six points to apprehend:
- Carbon dioxide absorption during tree growing should be included in the analysis.
- Carbon dioxide emissions from wood product incineration must be included in the analysis.
- The feedstock energy of wood inputs must be included in calculations.
- When wood feedstock is used as a fuel in intermediate processes, the change must be calculated.
- Energy must be reported in sufficient detail so that the contribution of wood fuel and feedstocks can be identified; reporting energy as a single number is meaningless.
- Quantification of methane emissions from landfilled paper is still imprecise, but if it is included, at the least, the yield, measured in terms of CO2 equivalents, will be increased by a factor of 2.5 compared with the CO2 emitted during complete incineration.
The primary aim of life-cycle inventories and life-cycle analyses is to provide the data needed to inform decision making. They are complementary to economic, social, and political considerations but do not replace them. When used improperly, they are useless—as are all decision-making tools. But when used properly and intelligently, they provide insights about the working of industrial systems that are unavailable elsewhere.






