12
Doubly Labeled Water for Energy Expenditure
James P. DeLany1
INTRODUCTION
The doubly labeled water (DLW) technique was developed as a method for measurement of free-living energy expenditure in animals (Lifson and McClintock, 1966). The method has since been validated in many animal species (for summary see Roberts, 1989; Schoeller, 1988). Twenty years after the initial report of the DLW method (Lifson et al., 1955), the economic feasibility of using the method in humans was considered (Lifson et al., 1975). The obstacle to applying the method to humans was the cost of the large quantities of oxygen-18 labeled water (H218O) needed to attain an adequate isotopic enrichment for calculation of valid elimination rates. This obstacle was overcome by improvements in gas isotope ratio mass spectrometers and a decrease in the cost of H218O. Another 7 years passed before the DLW method eventually was validated against the intake balance (I/B) method in humans (Schoeller and van Santen, 1982). The method has since been validated against indirect calorimetry
and I/B in many different subject populations by several laboratories, with a demonstrated precision (coefficient of variation) of 2 to 8 percent (Schoeller, 1988).
THEORY OF DOUBLY LABELED WATER
The DLW method is based on the premise that after a dose of doubly labeled water, 2H218O, the two isotopes equilibrate with total body water (TBW) and then are eliminated differentially from the body (Figure 12-1). Deuterium (2H) leaves the body as water, while 18O leaves as water (H2O) and carbon dioxide (CO2). Therefore, CO2 production can be calculated by subtracting 2H elimination from 18O elimination. The DLW method is an ideal method for military nutrition studies because it is an accurate field technique for determination of free-living energy expenditure. There are no requirements for subject compliance (except giving urine and/or saliva samples) such as filling out logs; there is no fear of equipment breakage; and it can be used to validate other field techniques. The initial validations of DLW were conducted under sedentary conditions, while military studies generally have involved soldiers undergoing strenuous activity. In a study of subjects under sedentary conditions (1.4 × sleeping metabolic rate) and in subjects undergoing heavy bicycle ergometer workouts (2.6 × metabolic rate), the DLW method was shown to be accurate compared to indirect calorimetry (Westerterp et al., 1988). The method
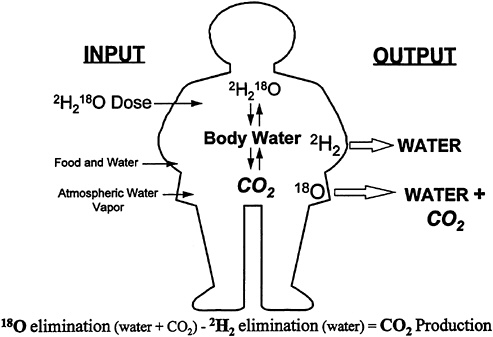
FIGURE 12-1 Theory of doubly labeled water method.
has since been validated in soldiers under various conditions (DeLany et al., 1989; Forbes-Ewan et al., 1989; Hoyt et al., 1991, 1994).
In addition to providing measurement of integrated energy expenditure, the DLW method provides simultaneous measures of TBW (from which body composition can be calculated) and water turnover, key measurements for many military nutrition studies. Hydration status is perhaps more critical to the sustained performance of soldiers than energy balance, and water requirements have been studied during many field studies. Use of 2H elimination for measurement of water turnover was initially validated in animal studies (Lifson and McClintock, 1966). It was pointed out that water intake can be overestimated unless corrections are made for water turnover in the lungs (Figure 12-1) and fractionation (preferential loss of one isotope due to different physical properties) of 2H. Water intake by 2H elimination also was investigated in the first validation of DLW in humans (Schoeller and van Santen, 1982). The isotope method was within 1.3 ± 6.7 percent (coefficient of variation) of the measured water intake, although the range was from -6.3 percent to +8.0 percent. Data compiled from analysis of 77 male soldiers in a variety of field settings were used to compare water intake measured by deuterium oxide (2H2O) elimination with logbook self-reports with canteens as a unit of consumption (Jones et al., 1993). Water intake by the 2H2O elimination method agreed very well with that obtained by the logbook method (3,750 ± 1,120 vs. 3,810 ± 1,450 g/dl).
Doubly Labeled Water Protocol
A typical DLW protocol is depicted in Figure 12-2. The study begins with the collection of baseline urine and/or saliva samples, followed by oral administration of the 2H218O dose. Saliva samples are obtained 2 to 4 hours after the dose for calculation of dilution spaces (Schoeller et al., 1980). A urine sample is collected the following morning for measurement of initial enrichment. Urine samples then are collected at the end of the period for measurement of final isotopic enrichment. The length of time a DLW study can be carried out depends

FIGURE 12-2 Typical doubly labeled water protocol. TBW, total body water.
on the turnover of the two isotopes, which is dependent on H2O and CO2 output. For studies in typical adults, the optimal metabolic period is 4 to 21 days (Schoeller, 1988), with military nutrition studies generally limited to the shorter time periods. If TBW is expected to change over the metabolic period, deuterium oxide will need to be readministered at the end of the study for a final TBW measurement.
Since the DLW dose and the analyses are so expensive, it is imperative that enough specimens are obtained to have backup samples at the beginning and end of the period of interest. Problems with specimens, such as inability to obtain a specimen, loss of a specimen, or contamination of a specimen, can occur in the field or in the lab. The protocol depicted in Figure 12-2 illustrates the minimum number of specimens to be collected for a DLW study. This protocol provides backup samples for the initial and final time points. If there is a problem with the 24-h urine, the 4-h saliva specimen can be used as the initial time point. If the final sample is bad, the urine from the previous day can be used as the final enrichment for elimination rate calculations. The 18O isotope abundances are measured on a gas-inlet isotope ratio mass spectrometer with a CO2-water equilibration device (DeLany et al., 1989). Briefly, urine and saliva samples are equilibrated with CO2 at a constant temperature in a shaking water bath for at least 12 hours. The CO2 then is purified cryogenically under vacuum before introduction into the mass spectrometer. The hydrogen isotope abundances usually are measured on a gas-inlet isotope ratio mass spectrometer, after microdistillation and zinc (or uranium) reduction (DeLany et al., 1989).
The 2H and 18O isotope elimination rates (kH and kO) can be calculated by the two-point method using the initial (i) isotopic enrichment and the final (f) enrichments: k = (ln enrichmentf-ln enrichmenti)/Δ t, where ln is the natural log, enrichment is the enrichment above baseline, and Δt is the number of days between the initial and final sample. CO2 production has been calculated according to Schoeller (1988):
(Equation 12-1)
where N is the TBW calculated from the 18O enrichment in the 4-h saliva (or average of initial and final 4-h saliva samples if TBW is expected to change), and rH2Of is the rate of fractionated evaporative water loss, which is estimated to be 1.05N(1.01kO -1.04kH).
Two-Point versus Multipoint Sampling
There has been some debate about the number of time points needed to obtain accurate measures of isotope elimination rates. As originally developed for experiments in animals and in the first human validations, elimination rates were calculated from an initial and a final time point, which has become known as the two-point method (although more than two points are often collected). For the
multipoint DLW protocol, samples are obtained throughout the period, and elimination rates are calculated by regression analyses. The rationale of the multiple-point method is that by using more time points to calculate elimination rates, sample-to-sample variation (analytical error) is averaged out, and a more precise measure of energy expenditure might be obtained. Although multiple samples may provide an advantage with regard to increased precision, there are several disadvantages to this method. The obvious problems with obtaining multiple samples are that it intrudes more into the subject's daily routine, may interfere with the habitual daily energy expenditure, and increases the laboratory work load. The less obvious and major concern with the multiple-sample method is that it does not give an exact average over the metabolic period if there are systematic variations in the water (Lifson et al., 1955; Nagy and Costa, 1980) or CO2 flux (Lifson et al., 1955; Speakman and Racey, 1986). However, in contrast to earlier comments by other investigators (Coward, 1988; James et al., 1988), the two-point method gives the arithmetically correct energy expenditure. This is because the two-point method gives an exact average of elimination rates over time even in the face of systematic variations, while the regression models do not (Coward, 1990; Nagy and Costa, 1980; Schoeller and Coward, 1990; Speakman and Racey, 1986; Welle, 1990). Variations in energy expenditure and water turnover often occur during military nutrition studies, either due to the nature of a field training exercise or due to environmental factors.
Two U.S. Army Research Institute of Environmental Medicine studies demonstrate the advantage of the two-point method over the multiple-point method in military studies. A multipoint sampling strategy was employed that would incorporate the best aspects of the two-point with those of the multipoint protocol. Samples were analyzed from the first 2 and the last 3 days of the metabolic period for calculation of elimination rates by linear regression. This is similar to the optimal sampling design suggested to minimize error (Cole and Coward, 1992). Results were compared with those obtained by the two-point method using isotope enrichments measured on samples from the first and last days. In a high-altitude study (Edwards et al., 1991) in which energy expenditure and water turnover were relatively constant throughout the study, energy expenditure by the two-point method (3,550 ± 610 kcal/d) was nearly identical to that obtained using the multipoint method (3,565 ± 675 kcal/d). This study demonstrated that energy expenditure could be measured accurately with only two time points, eliminating the need for extra sample collection and laboratory analyses. A summary of the available data on repeat DLW measures of energy expenditure also showed that there was no improvement in accuracy or precision for the multipoint method over the two-point method (Cole and Coward, 1992). The variance of repeat measures in two studies using the two-point method (7.4% and 7.3%) was similar to that observed in two studies using the multipoint method (7.4% and 6.7%).
A second study demonstrated the major potential disadvantage of the multipoint methods. During a cold-weather study, there was a significant change in
energy expenditure and water turnover towards the end of the metabolic period due to environmental extremes (DeLany et al., 1991). During the second to the last day of the metabolic period, the soldiers moved and set up a defensive position, underwent live-fire exercises, and moved back to camp (February 12–13). During the previous day and the day following the live-fire exercises, the soldiers ''stood down" (remained inactive) the entire day because of helicopter support and extreme cold weather. Therefore, H2O and CO2 elimination were very different over these 3 days. Deviations from a linear isotope elimination were apparent from a close examination of a plot of the log of 2H enrichment (18O data were similar) versus time (Figure 12-3). Examination of regression coefficients that were 0.99 or better suggested a linear response. However, this physiologic variation (as opposed to analytical error) introduced an error in calculation of isotope elimination by the regression method, giving an energy expenditure that was significantly (p < 0.01) lower than that obtained using the two-point method (5,015 ± 510 vs. 5,140 ± 630 kcal/d). Therefore, as has been described previously, the two-point and regression methods are both accurate when there are no deviations in energy expenditure and water turnover during the measurement period (assuming isotope enrichments are measured accurately). However, when there are systematic variations during the period, the multipoint methods using simple linear regression do not give true measures of elimination rates.
The Importance of Isotope Dilution Space Ratios
Another recent debate has centered on the relationship between the two isotope dilution spaces and TBW. When the original equation for humans was developed (Equation 12-1 above), there were few available data on the relation
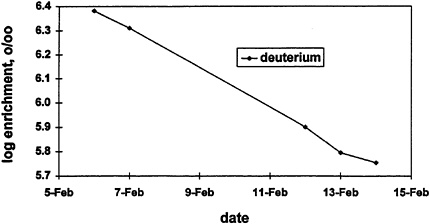
FIGURE 12-3 Deviation in isotope elimination due to physiologic variation. 0\00, per mille derivations in heavy isotope ratio relative to standard mean ocean water (smow).
ship between the two isotope dilution spaces. The ratio between the 2H and 18O dilution spaces (ND/NO) in 23 subjects was 1.033, which was rounded to three significant digits to give a ratio of 1.03 (Schoeller et al., 1986). The ratio between NO and TBW based on available animal data, and the theoretical estimate of the nonaqueous exchangeable oxygen pool was estimated to be 1.007, which was rounded to 1.01 (Schoeller et al., 1980). This gave a ratio of 1.04 for ND/TBW. In a recent review of available data, a new ratio of 1.0427 was recommended (Speakman et al., 1993). The following year, two independent investigators reviewed the available literature and came to a different conclusion (Coward et al., 1994; Racette et al., 1994). These investigators noted that differences in dilution space ratios reported in the literature appeared to be method dependent. Labs using the method of direct addition of biological fluid (urine or saliva) to the zinc reagent for reduction to hydrogen gas obtained a higher ND/NO ratio than those using uranium reduction or zinc reduction after distillation of the sample. A study of the effect of sample matrix (i.e., compounds in urine) in biological fluids demonstrated inaccuracies in the measurement of 2H enrichment with the direct addition technique (Ritz et al., 1994). For a urine sample with an average solids concentration of 40 g/kg, the 2H enrichment was underestimated by 2.4 percent, causing an overestimation in the calculated dilution space (Ritz et al., 1994). Based on these observations, the new recommended ND/NO ratio is 1.034 (Coward et al., 1994; Racette et al., 1994), and individual factors for ND/TBW and NO/TBW are 1.041 and 1.007. These modifications are made in Equation 12-1 to give Equation 12-2 below:
(Equation 12-2)
The details are the same as for Equation 12-1 above, except the new factors also are used in the calculation of rH2Of to give 1.05N(1.007kO -1.041kH).
This may seem like a small modification to Equation 12-1, but small differences in dilution space ratios introduce significant changes in the calculation of rCO2. A 1 percent difference in the ND/NO ratio (e.g., 1.04 vs. 1.03) can introduce a 4 percent error in the calculation of CO2 production and energy expenditure during a typical DLW study in soldiers. Errors would be even higher in studies where energy expenditure is lower. When the modified equation (Equation 12-2) was used to recalculate existing validation data, the accuracy of the DLW method improved from an overestimate of 3.3 ± 8.9 percent (when using the original Equation 12-1) to an energy expenditure that agreed within 1.2 ± 8.5 percent of that obtained by indirect calorimetry. However, when the higher proposed ratio was used, energy expenditure was underestimated by 3.1 ± 9.9 percent (Racette et al., 1994).
PARTICULAR CONCERNS FOR MILITARY NUTRITION DLW STUDIES
Additional concerns when applying the DLW method to military nutrition studies are changes in baseline isotope during the study, the possible need for a higher dose of isotope and the need for administration of multiple doses of DLW to obtain energy expenditure during a long training study. For typical DLW studies, the baseline isotope abundance is measured before administration of the DLW and is assumed to remain constant throughout the study. The problem of baseline isotope shifts occurs in military studies because the soldiers often are transported to a different geographical location to carry out a field-training study. This poses a problem because 2H and 18O are naturally occurring isotopes, and variations in the isotopic enrichment of water occur based on geographical location (Dansgaard, 1964). When the soldiers move to the location of a field-training study, drinking water with a different isotopic abundance will mix with TBW (Figure 12-1) and appear as a change in elimination rates, leading to errors in the measurement of energy expenditure. This phenomenon has been observed in patients receiving parenteral nutrition, in infants changing parenteral formulas or switching to oral formula, and in soldiers during field-training studies (DeLany et al., 1989; Hoyt et al., 1991, 1994; Jones et al., 1988; Schoeller et al., 1986). Therefore, in military nutrition studies employing DLW, a control group not receiving isotope must also be studied to correct for background shifts in isotope.
Another concern in military nutrition studies is the possible requirement for higher DLW dose due to high water turnover and high energy expenditure that occur during field-training exercises. For example, during one phase of a Ranger training course (Moore et al., 1992), water turnover during the first 3 days was over 9 liters/d, while energy expenditure was 5,350 ± 770 kcal/d. The high water turnover and energy expenditure combined to flush out the isotope, with over half of the administered dose eliminated in the first 4 days (Figure 12-4). This posed a problem because there also was interest in ascertaining energy expenditure over the next 11 days. However, there was only enough isotope remaining to measure energy expenditure with any degree of accuracy over the next 6 days. A high water turnover had been anticipated, and a higher dose than usual was given. However, with such high turnover rates (even higher than anticipated), giving a higher DLW dose is not very effective. The only way to obtain DLW data for this entire period of interest would be to administer a second dose of 2H218O on day 10, or perhaps even on day 4 (Figure 12-4).
A multiple-dosing (administration of a dose of DLW) strategy was used in an earlier study to obtain data on energy expenditure during a 28-d field-training exercise (DeLany et al., 1989). Multiple doses have also been used in Ranger training studies to obtain energy expenditure during much of the 63-d course (Moore et al., 1992). In the Ranger studies, multiple dosings have been administered and samples have been collected at various times throughout a dosing pe-
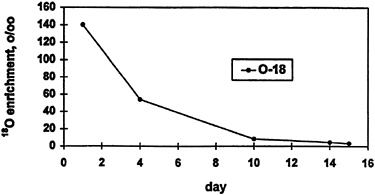
FIGURE 12-4 Washout of isotope due to high water turnover. 0/00, ratio per mille derivations in heavy isotope ratio relative to standard mean ocean water (smow).
riod to obtain data from multiple phases within a period (Figure 12-5). The days between the arrows indicate the training phases covered by four separate DLW dosings, and the points indicate the energy expenditure for a typical subject for each period. Note that in the first and third period, two measurements of energy expenditure were obtained, corresponding to two separate portions of that phase of the training course. This was accomplished by obtaining a specimen in the middle of the period as well as at the end of the period. The data presented in Figure 12-4 correspond to Period 3 in Figure 12-5. The day 4 sample (Figure 12-4) served as the final time point for the first 3 days of the study, as well as the initial time point for measurement of energy expenditure over the next 6 days. Using this strategy, it was possible to calculate energy expenditure during two distinct portions of the training program.
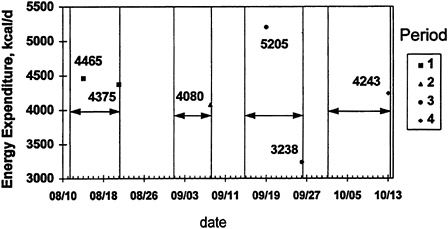
FIGURE 12-5 Multiple administrations of doubly labeled water doses to obtain energy expenditure during much of a Ranger training course.
Drawbacks of the DLW Method
The major drawbacks of the DLW method are cost of the 18O-labeled water and the cost and technical difficulties associated with the isotope analyses. The need for an expensive isotope ratio mass spectrometer and sample preparation systems limits the number of laboratories that can carry out the method on-site. In addition, the technical difficulties of measuring the isotope enrichments with the required accuracy are formidable. High accuracy in isotope measurements is required because energy expenditure is calculated based on a small difference between the two isotope elimination rates. The primary difficulties arise from fractionation of the isotopes during sample preparation and measurement due to the different physical characteristics of the heavy isotopes compared with the major isotope. Furthermore, sample matrix can interfere with isotope measurements (as discussed above). However, there are several labs throughout the world that have considerable experience with the method and have overcome the analytical difficulties.
Price and availability of the 18O-labeled water have been drawbacks of the DLW method. In early 1990, the price of a gram of H218O (10 g of a 10% solution) had risen to $36, with a dosing for a typical 70-kg man costing $400. Due to increased demand and production problems over the next couple of years, the price of H218O rose to over $63/g, with delivery times often exceeding 12 months. This price increase brought the cost of dosing a single subject to at least $700. By early 1995, the shortage was overcome, with the price dropping to $35/g and delivery within a few weeks, bringing the cost of dosing a typical 70-kg subject down to $385. The increased availability of H218O at a lower price will allow more widespread use of the DLW method. In addition, it will allow an entire training course to be covered with DLW measurements, with no gaps, as has been done in the past (see Figure 12-5).
AUTHOR'S CONCLUSIONS AND RECOMMENDATIONS
The doubly labeled water method is an ideal tool for military nutrition research. It is a true field technique for accurate measurement of free-living energy expenditure of soldiers undergoing training exercises. The only intrusion into the soldier's daily routine is collection of urine and saliva samples at the beginning and end of the study. The method also provides simultaneous measures of TBW, body composition, and water turnover. When applying the DLW method to military nutrition research, the following recommendations are made:
-
The two-point method should be utilized to calculate elimination rates, with backup specimens collected for the initial and final time points.
-
A control group should be included to correct for baseline isotope shifts due to changes in food and water supply.
-
Careful planning is required so that the optimum dosing and specimen collection protocol are utilized based on (1) estimated water turnover and energy expenditure, and (2) maximization of information gained from study.
-
When calculating energy expenditure, use Equation 12-2, which incorporates new dilution space factors based on the latest available data.
-
Multiple DLW dosings can be used to obtain data on energy expenditure for long training exercises.
-
The decrease in cost of the H218O dose should allow for adequate dosings to cover an entire training study, with no gaps in energy expenditure data.
REFERENCES
Cole, T.J., and W.A. Coward 1992 Precision and accuracy of doubly labeled water energy expenditure by multipoint and two-point methods. Am. J. Physiol. 263 (26):E965–E973.
Coward, A. 1990 The doubly labeled water method for measuring energy expenditure. Technical recommendations for use in humans. Calculation of pool sizes and flux rates. Pp. 48–68 in NAHRES-4, A Consensus Report by the IDECG Working Group, A.M. Prentice, ed. Vienna: IAEA.
Coward, W.A. 1988 The doubly labeled water (2H218O) method: Principles and practice. Proc. Nutr. Soc. 47:209–218.
Coward, W.A., P. Ritz, and T.J. Cole 1994 Revision of calculations in the doubly labeled water method for measurement of energy expenditure in humans. Am. J. Physiol. 267 (30):E805–E807.
Dansgaard, W. 1964 Stable isotopes in precipitation. Tellus 16:436–468.
DeLany, J.P., D.A. Schoeller, R.W. Hoyt, E.W. Askew, and M.A. Sharp 1989 Field use of D218O to measure energy expenditure of soldiers at different energy intakes. J. Appl. Physiol. 67(5):1922–1929.
DeLany, J.P., R.J. Moore, and R.W. Hoyt 1991 Use of doubly labeled water for measurement of energy of soldiers during training exercises in a cold environment and at high altitude [abstract]. Int. J. Obes. 15:48.
Edwards, J.S.A., E.W. Askew, N. King, C.S. Fulco, R.W. Hoyt, and J.P. DeLany 1991 An assessment of the nutritional intake and energy expenditure of unacclimatized U.S. Army soldiers living and working at high altitude. Technical Report No. T10-91. Natick, Mass.: U.S. Army Research Institute of Environmental Medicine .
Forbes-Ewan, C.H., B.L.L. Morrissey, G.C. Gregg, and D.R. Waters 1989 Use of doubly labeled water technique in soldiers training for jungle warfare. J. Appl. Physiol. 67(1):14–18.
Hoyt, R.W., T.E. Jones, T.P. Stein, G.W. McAninch, H.R. Lieberman, E.W. Askew, and A. Cymerman 1991 Doubly labeled water measurement of human energy expenditure during strenuous exercise. J. Appl. Physiol. 71(1):16–22.
Hoyt, R.W., T.E. Jones, C.J. Baker-Fulco, D.A. Schoeller, R.B. Schoene, R.S. Schwartz, E.W. Askew, and A. Cymerman 1994 Doubly labeled water measurement of human energy expenditure during exercise at high altitude. Am. J. Physiol. 266:R966–R971.
James, W.P.T., J.P. Haggarty, and B.A. McGaw 1988 Recent progress in studies on energy expenditure: Are the new methods providing answers to old questions? Proc. Nutr. Soc. 47:195–208.
Jones, P.J.H., A. Winthrop, D.A. Schoeller, R.M. Filler, P.R. Swyer, J. Smith, and T. Heim 1988 Evaluation of doubly labeled water for measuring energy expenditure during changing nutrition. Am. J. Clin. Nutr. 47:799–804.
Jones, T.E., R.W. Hoyt, J.P. DeLany, R.L. Hesslink, and E.W. Askew 1993 A comparison of two methods of measuring water intake of soldiers in the field [abstract]. FASEB J. 7:A610.
Lifson, N., and R. McClintock 1966 Theory of use of turnover rates of body water for measuring energy and material balance. J. Theor. Biol. 12:46–74.
Lifson, N., G.B. Gordon, and R. McClintock 1955 Measurement of total carbon dioxide production by means of D2O18. J. Appl. Physiol. 7:705–710.
Lifson, N., W.S. Little, D.G. Levitt, and R.M. Henderson 1975 D218O method for CO2 output in small mammals and economic feasibility in man. J. Appl. Physiol. 39(4):657–664.
Moore, R.J., K.E. Friedl. T.R. Kramer, L.E. Martinez-Lopez, R.W. Hoyt, R.E. Tulley, J.P. DeLany, E.W. Askew, and J.A. Vogel 1992 Changes in soldier nutritional status and immune function during the Ranger training course. Technical Report No. T13-92. Natick, Mass.: U.S. Army Research Institute of Environmental Medicine.
Nagy, K.A., and D.P. Costa 1980 Water flux in animals: Analysis of potential errors in the tritiated water method. Am. J. Physiol. 238:R454–R465.
Racette, S.B., D.A. Schoeller, A.H. Luke, K. Shay, J. Hnilicka, and R.F. Kushner 1994 Relative dilution spaces of 2H- and 18O-labeled water in humans. Am. J. Physiol. 267 (30):E585–E590.
Ritz, P., P.G. Johnson, and W.A. Coward 1994 Measurements of 2H and 18O in body water: Analytical considerations and physiological implications. Br. J. Nutr. 72:3–12.
Roberts, S.B. 1989 Use of doubly labeled water method for measurement of energy expenditure, total body water, water intake, and metabolizable energy intake in humans and small animals. Can. J. Physiol. Pharmacol. 67:1190–1198.
Schoeller, D.A. 1988 Measurement of energy expenditure in free-living humans by using doubly labeled water. J. Nutr. 188:1278–1289.
Schoeller, D.A., and A. Coward 1990 The doubly-labeled water method for measuring energy expenditure. Technical recommendations for use in humans. Practical consequences of deviations from the isotope elimination model. Pp. 166–190 in NAHRES-4, A Consensus Report by the IDECG Working Group, A.M. Prentice, ed. Vienna: IAEA.
Schoeller, D.A., and E. van Santen 1982 Measurement of energy expenditure in humans by doubly labeled water method. J. Appl. Physiol. 53(4):955–959.
Schoeller, D.A., E. van Santen, D.W. Peterson, W. Dietz, J. Jaspan, and P.D. Klein 1980 Total body water measurement in humans with 18O and 2H labeled water. Am. J. Clin. Nutr. 33:2686–2693.
Schoeller, D.A., R.F. Kushner, and P.J.H. Jones 1986 Validation of doubly labeled water for measuring energy expenditure during parenteral nutrition. Am. J. Clin. Nutr. 44:291–298.
Speakman, J.R., and P.A. Racey 1986 Measurement of CO2 production by the doubly labeled water technique. J. Appl. Physiol. 61:1200–1202.
Speakman, J.R., K.S. Nair, and M.I. Goran 1993 Revised equations for calculating CO2 production from doubly labeled water in humans. Am. J. Physiol. 264 (27):E912–E917.
Welle, S. 1990 Two-point vs. multipoint sample collection for the analysis of energy expenditure by use of the doubly labeled water method. Am. J. Clin. Nutr. 52:1134–1138.
Westerterp, K.R., F. Brounds, W.H.M. Saris, and F.T. Hoor 1988 Comparison of doubly labeled water with respirometry at low-and high-activity levels. J. Appl. Physiol. 65(1):53–56.
DISCUSSION
JOHANNA DWYER: How long is long term with multiple doses?
JAMES DeLANY: We attempted to cover a 2-mo time period. We did not quite make it. We would have needed at least six to eight dosings, maybe seven, to cover the whole Ranger training study.
JOHANNA DWYER: But it is within the realm of possibility?
JAMES DeLANY: Yes. But that was all the isotope Reed Hoyt had.
DENNIS BIER: I, like most of the other people in the room, I would guess, are enamored with this method for its advantages, but I still have a problem with the two-dose approach. You showed us several lines which clearly were not linear; I mean, they had breakpoints. In one case, it was because the subjects had eaten more, as I remember. With the last line, you had the last two points 1 day apart, and there was a change in the slope of the curve.
JAMES DeLANY: That was actually from the same study.
DENNIS BIER: Two points will always define a straight line, so you are okay there, but then when you have these other lines, there may be breakpoints because there is a change in energy expenditure. If that is the case, if they break in short periods of time, then you no longer have the steady state circumstance you need to make the calculation because you are not far enough into the decay pe-
riod of the water. Or depending on what point you have, if you are taking only two of them, you can change the slope of that line, depending on where you are. You showed us lines at several points.
Finally, I want to comment on just the simple matter of when people collect samples. I can imagine a situation in the field where a guy is supposed to collect a sample at noon. But then noon passes and next it's "Oh, 12:20 or 1:00, I forgot," and he puts it in the noon bottle. That happens to us all the time. It may not happen to you.
JAMES DeLANY: Oh, I am sure it does. Okay, let us start with your first point.
DENNIS BIER: Could you just go back to one slide for an example, just about the second or the third from the end?
(Slide)
Depending on which one of those last three points you pick, or the last two points …
JAMES DeLANY: The last two points, right.
This is the same study that I showed you where on this day, the subjects were on a 12-mi march, and on this day they were stuck in the tent. The advantage of the two-point method is it gives the true cumulative elimination. If this was appropriately modeled with a modeling technique, you would get the correct answer, but no one does that.
This is actually a lower-energy expenditure day. Doubly labeled water does not give you a day-by-day breakdown; it is integrated, so you measure it over 7 to 10 days and then divide by 7, and you get an average. Tut that does not mean that the energy expenditure was 6,000 calories a day every day during this study.
For instance, on this day I am sure it was 2,500 or less; they were stuck in the tent sleeping all day. The two-point method, though, will give you the true elimination over that time period.
DENNIS BIER: Which two points did you pick?
JAMES DeLANY: The beginning and end. Between this day and this day, the average energy expenditure would be 100 kcals more than this final day. I do not know, I am just pulling a number out of the hat. But we were interested in the whole time period.
ROBERT WOLFE: Since you made the case so strongly for the two-point method, the other thing I would add that there is a potential advantage to collecting multiple points. It is that you use the jackknife technique to examine bad data points. For example, if you have seven data points, you calculate energy expenditure using all of the data points. Then you recalculate energy expenditure, sequentially eliminating each data point. You end up with a series of values that can be used to calculate a standard deviation of the energy expenditure determination in that individual.
That has some practical advantages in two senses. One, if you get an outlier that is way off, you can present the standard deviation beyond which you will not accept a value. Where we found that to be particularly useful was when we were studying the energy expenditure of the U.S. swimmers prior to the Olympics. It turned out they were using much more energy than we had anticipated.
So by doing that, you can work backwards. In other words, if we had used the original whole period, that enrichment was so low and the variability was so much that the standard deviation for each individual was very large. We just kept removing the last time points from regression analysis until we reached the point where we had an acceptable standard deviation when we did this technique of dropping one point and using the remainder.
It is kind of a different issue than Denny and you were just arguing, but it gives you at least some sort of a statistical basis to decide whether an outlier is one that should be discarded and, also, the point at which you would say, again, in an objective way, whether the rest of the data points are giving us more variability than information.
DAVID SCHNAKENBERG: In the first study you did for us up in the mountains of Vermont, we were interested in water consumption, and you used this methodology to do water flux and estimate of water consumption over that entire period. You did not mention that. Is that at all feasible to do?
JAMES DeLANY: In fact, in many of the studies, particularly when the 18O was a problem, we did a lot of studies with turnover of total body water. In some of the altitude studies, we were interested just in water. I meant to bring that up, but you are right, we do measure water flux and have done some studies with canteen measurement of water intake versus the isotope and got very good agreement.
DAVID SCHNAKENBERG: What do you feel the reliability of that method is for estimating actual fluid consumption over extended periods?
JAMES DeLANY: It does not appear to be as good as energy expenditure, but some of the data that Tanya Jones has been compiling, some of the data that we have collected regarding the use of isotope versus the canteen measurements, look really good, with a correlation of maybe 0.85 between the measured intake and the isotope measurement of water turnover.
JOAN CONWAY: Do you need doubly labeled water to do the water flux?
JAMES DeLANY: No, just deuterium.
DAVID SCHNAKENBERG: It [water flux] is a by-product of going through all the calculations of the DLW method.
JAMES DeLANY: Yes, you get it because you calculate water turnover in the DLW method, yes.
















