18
Use of Isolated-Cell and Metabolic Techniques Applied to Vitamin Transport and Disposition
Donald B. McCormick1
INTRODUCTION
Performance capability centrally involves the ability to acquire and utilize nutrients at the cellular level and to make such metabolic adjustments as are needed to sustain competent functioning for a specified period of time. There are well-documented changes in needs for calories, water, and replenishable electrolytes relating to alterations in exercise and in ambient temperature. The metabolic conversion of macronutrients to supply the cell's energy needs depends upon micronutrients, which are essential in the enzymatic steps involved. Yet knowledge on shifts in micronutrient intake that may be needed to optimize cellular function with changing performance is largely missing. It is known, for example, that greater utilization of carbohydrate increases the demand for thiamin, but the span of time and level of adjustment of the latter to maximize efficiency of energy utilization from the former is not fully
quantitated. Even less information is available on the levels of other water-soluble vitamins that may be desirable for performance enhancement. Certainly essential roles for vitamin B6 in protein utilization and for riboflavin in general oxidative metabolism can be appreciated on the basis of known biochemical reactions, but the amounts of these and other vitamins that optimize performance with no compromise of long-term health in humans are not certain.
The gaps in information on the impact of stress or exertion on the ultimate disposition of natural derivatives of vitamins are especially great. There are few if any data on the efficiency of digestion of coenzymes within the tissues consumed as food in the face of shifting physical demands. Nothing is known about such effects on the release and recovery of vitamins from glycosides, esters, and peptides, which could account for a significant fraction of potential vitaminic material in many foods. The relationship of performance capability to micronutrient bioavailability has yet to be adequately researched.
What is known is that the metabolic pathways utilized in the molecular dissolution of nutrient compounds by E. coli and the human are generally similar, and among mammals even semiquantitative comparisons are usually valid. This is fortunate since it allows one to answer some questions at the cellular level with material obtained from animals (e.g., rats) that cannot ethically be accessed from humans. Cells that are freshly isolated after appropriate collagenase perfusion in situ of organs such as liver or kidney maintain viability for sufficient time in modified, oxygenated buffer to permit meaningful measurements of nutrient uptake and utilization. This technique allows separation of the cellular component in the overall fate of a nutrient that must undergo systemic absorption, circulatory transport, and organ dissemination. Moreover, there is no risk of cell transformation, which often occurs with the repeated transfers necessary in cell tissue culture techniques.
Considerable information has been gained regarding the mechanisms by which liver and kidney cells import and subsequently assimilate water-soluble vitamins such as riboflavin, B6, biotin, and C (Bowman et al., 1989; McCormick and Zhang, 1993; Rose et al., 1986). Coupled with the extensive work of this laboratory on the metabolism of these vitamins and the cofactor lipoic acid (Chastain and McCormick, 1991; McCormick, 1975, 1979, 1989; McCormick and Wright, 1970), these investigators were poised to compare the means by which cells take in and differentially utilize certain natural derivatives. To illustrate this, isolated-cell and metabolic techniques recently have been used to augment understanding of the bioavailability of the glucosides of pyridoxine (Zhang et al., 1993) and of riboflavin (Joseph and McCormick, 1995).
RESULTS WITH VITAMIN GLUCOSIDES
The occurrence of natural glucosides of both vitamins B2 (riboflavin) and B6 has been known for some time, but their roles were less clear. The extent to
which these glucosides represent forms used for storage, transport, and release of the vitamins only now is becoming evident.
Pyridoxine-5'-ß-D-Glucoside
The isolation of a ''bound" form of B6 from rice bran led to its identification as 5'-O-(ß-D-glucopyranosyl)pyridoxine (Yasumoto et al., 1977). The structure of this compound is shown in Figure 18-1. Only plant foods have been found to contain this compound, which can account for up to half of the total B6 (Leklem, 1994). It may represent a storage form for higher plants, but it clearly is a less bioavailable form of B6 for mammals. Rats absorb but poorly metabolize and largely excrete pyridoxine ß-glucoside in urine (Ink et al., 1986; Trumbo and Gregory, 1988). There is apparently an inverse correlation between the percentage of glucosylated B6 in selected foods and the bioavailability of this vitamin in humans (Kabir et al., 1983).
Since pyridoxine ß-glucoside actually retards the utilization of pyridoxine in rats (Gilbert and Gregory, 1992), the possibility that the ß-glucoside could inhibit uptake of pyridoxine by freshly isolated rat hepatocytes was examined first (Zhang et al., 1993). As shown by the data in Figure 18-2, the unlabeled glucoside competitively inhibits uptake of [3H]pyridoxine presented to the cells. The inhibitory constant (Ki) obtained from the plot was approximately 1.4 µM Pyridoxine ß-glucoside does not inhibit uptake of glucose, however.
The ß-glucoside of pyridoxine not only competes for the same plasma membrane transporter that has been shown to be relatively specific for the B6 moiety in both liver (Kozik and McCormick, 1984) and proximal tubular cells of the kidney (Bowman and McCormick, 1989), but can be taken up in a manner reflecting saturation kinetics, as illustrated in Figure 18-3. An apparent transport constant (Kt = 13.8 µM) and a maximal velocity Vmax = 82 pmol/106 cells/min can be compared to values reported earlier for pyridoxine entry into hepatocytes, with Kt = 6.3 µM and Vmax = 28 pmol/106 cells/min (Kozik and McCormick, 1984; Zhang and McCormick, 1992).
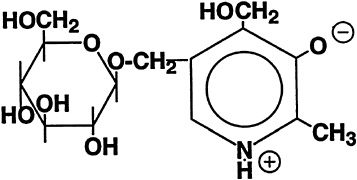
FIGURE 18-1 Structure of pyridoxine-5'-ß-D-glucoside.
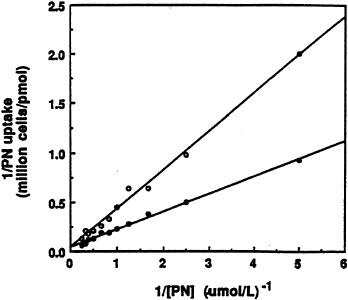
FIGURE 18-2 Double-reciprocal plots of the initial uptake rate of [3H]pyridoxine (PN) versus substrate concentration in the absence (•) and presence (o) of 2.0 µM unlabeled ß-D-glucoside. SOURCE: Zhang et al. (1993) © J. Nutr. (123:85–89), American Society for Nutritional Sciences.
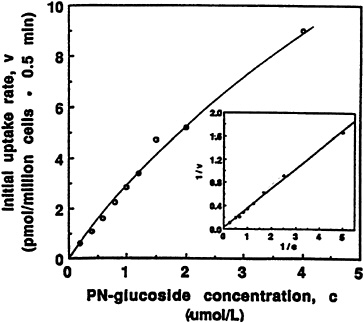
FIGURE 18-3 Dependence of initial rate of [3H]pyridoxine-5'-ß-D-glucoside (PN-glucoside) uptake (0.5 minutes) on concentration. The insert presents a double-reciprocal transformation of the same curve with the straight line obtained by regression analysis. SOURCE: Zhang et al. (1993) © J. Nutr. (123:85–89), American Society for Nutritional Sciences.
The effect of incubation time on the transport of pyridoxine and its ß-glucoside into liver cells is shown in Figure 18-4. Though the amount of transported pyridoxine increased significantly throughout the hour during which uptake was measured, the ß-glucoside reached an earlier limit near 20 percent that of the free vitamin. This result is very similar to the transport of pyridoxine and its a-glucoside into rabbit erythrocytes reported by others (Kawai et al., 1972).
The intracellular distribution of transported pyridoxine ß-glucoside was examined by incubating cells with the radioactive compound, then extracting and analyzing the cellular contents by chromatography. Pyridoxal 5'-phosphate (PLP) is the metabolite that has the greatest coenzyme function, ultimately arising after release of pyridoxine by a broad-specificity ß-glucosidase (Trumbo et al., 1990), which must act to unfetter the 5'-hydroxymethyl group that becomes phosphorylated prior to oxidation of the 4'-hydroxymethyl function. As shown by the results in Table 18-1, the amount of PLP formed from the cellular metabolism of the ß-glucoside was much less (only 0.2%) than that formed from pyridoxine. The formation of PLP also is much lower from the ß-glucoside than from free vitamin in the presence of homogenate or supernatant solution enriched with the cytosolic kinase and oxidase that convert free vitamin B6 to PLP. Hence, the rate-limiting step in metabolic utilization of pyridoxine ß-glucoside is the hydrolytic release of the vitamin.
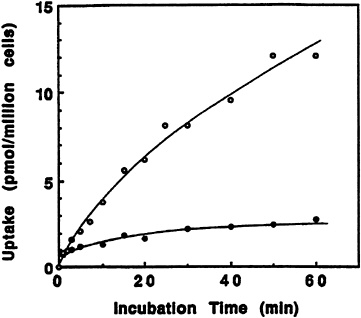
FIGURE 18-4 Effect of incubation time on the transport of pyridoxine (PN) and PN-glucoside. Isolated rat liver cells were incubated with 0.5 µM [3H]pyridoxine (o) or 3H-labeled ß-D-glucoside (•). SOURCE: Zhang et al. (1993) © J. Nutr. (123:85–89), American Society for Nutritional Sciences.
TABLE 18-1 Results of 50-minute Uptake, Cellular Metabolism, or Enzymatic Treatments of Pyridoxine or its ß-Glucoside
Overall, it is clear that pyridoxine ß-glucoside is less available as a source of vitamin B6 for mammals because it enters cells less well than the free vitamin and is limited considerably in its conversion to functional coenzyme.
Riboflavin-5'-a-D-Glucoside
The formation of riboflavin-5'-a-D-glucoside was first noted to occur when riboflavin was incubated with homogenates or aqueous extracts from acetone powders of rat liver (Whitby, 1952). The structure of this a-glucoside is shown in Figure 18-5. The compound also was found in the urine of rats after oral administration of [2-14C]riboflavin (Ohkawa et al., 1983). Hence, it became of interest to study disposition of the riboflavin a-glucoside with regard to its transport into and metabolic utilization by liver cells. Also since no bioavailability
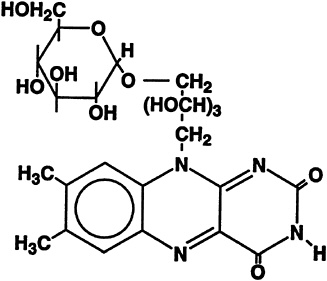
FIGURE 18-5 Structure of riboflavin-5'-a-D-glucoside.
studies had been done on this glucoside, its ability to replace riboflavin in the diets of young, growing rats was determined (Joseph and McCormick, 1995).
Uptake of [3H] riboflavin by freshly isolated rat hepatocytes is not affected by its unlabeled α-glucoside. This is true with comparable physiological concentrations over a period of time, as seen in Figure 18-6.
Because the solubility of riboflavin α-glucoside is much greater than that of riboflavin, it was suggested that this may be a natural derivative of importance in the transport of the relatively insoluble vitamin (Whitby, 1952). Results of a study on riboflavin α-glucoside uptake, shown in Figure 18-7, demonstrate that initial uptake of the glucoside is higher than that of riboflavin (Joseph and McCormick, 1995). However, the accumulation of α-glucoside after an hour was significantly lower.
As riboflavin α-glucoside does not inhibit uptake of riboflavin, the former probably enters the cell by an alternate route, bypassing the riboflavin transporter that has been shown to exhibit relative specificity for the flavin structure in both liver (Aw et al., 1983) and proximal tubular cells of the kidney (Bowers-Komro and McCormick, 1987). Also, the glucose transporter is not involved since uptake of riboflavin α-glucoside is not affected by glucose even when concentrations of the latter are considerably higher (Joseph and McCormick, 1995).
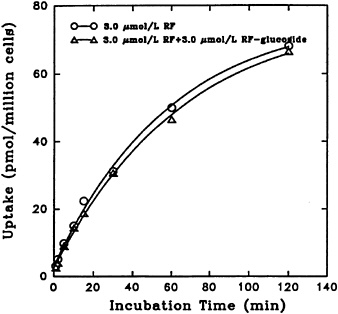
FIGURE 18-6 Time course of [3H]riboflavin (RF) uptake in the absence (o–o) and presence (Δ–Δ) of unlabeled α-D-glucoside. SOURCE: Joseph and McCormick (1995) © J. Nutr. (125:2194–2198), American Society for Nutritional Sciences.
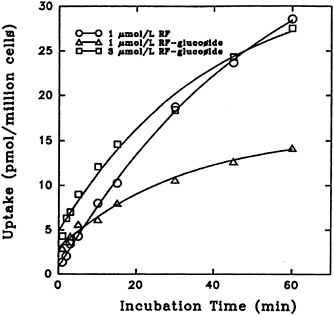
FIGURE 18-7 Effect of incubation time on the transport of [3H]riboflavin (RF) and 3H-labeled a-D-glucoside into isolated liver cells. SOURCE: Joseph and McCormick (1995) © J. Nutr. (125:2194–2198), American Society for Nutritional Sciences.
The effect of riboflavin a-glucoside concentration on its uptake into liver cells is shown in Figure 18-8. The process reflects saturation kinetics and exhibits an apparent Kt of 83.4 µM and a Vmax of 207 pmol/106 cells/min. The uptake was significantly affected by temperature, as expected for a facilitated transport. Decreasing sodium in the incubation mixture by replacement with other cations did not affect uptake. Hence, as previously reported for riboflavin entry into liver cells (Aw et al., 1983), sodium-dependent active transport does not seem to be involved.
The metabolic conversion of riboflavin a-glucoside upon entry can be seen by representative data presented in Table 18-2. By the end of 45 minutes, approximately 45 percent of the a-glucoside had been metabolized as compared with 25 percent of riboflavin. Hydrolysis is presumably mediated by nonspecific a-glucosidases known to occur in mammalian tissues (Yamamoto et al., 1990). This is supported by the observation that rat liver a-glucosidases catalyze formation as well as hydrolysis of a-glucoside derivatives of several xenobiotics (Kamimura et al., 1992). The vitamin, whether initially free or hydrolytically released, is converted by flavokinase to flavin mononucleotide (FMN) and by FAD synthetase to flavin adenine dinucleotide (FAD). These flavoenzymes account for most of the difference (between the initial and final concentrations of riboflavin), since only traces of other catabolites arise from enzymatic actions on riboflavin (Chastain and McCormick, 1991).
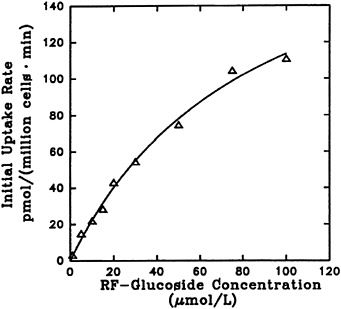
FIGURE 18-8 Dependence of initial rate of [3H]riboflavin 5'-a-D-glucoside uptake (1 minute) on its concentration. SOURCE: Joseph and McCormick (1995) © J. Nutr. (125:2194–2198), American Society for Nutritional Sciences.
When the effect of replacing riboflavin with its a-glucoside in the diet of weanling rats was examined (Joseph and McCormick, 1995), it was clear that the a-glucoside is at least as effective as riboflavin on an equivalent basis. Near-maximal growth rate was achieved with either 43 µg of riboflavin a-glucoside per 15 g of diet or 30 µg of the vitamin per 15 g of diet. It is probable that most of the ingested a-glucoside is hydrolyzed even before it reaches the liver since intestines are a rich source of a-glucosidases (Yamamoto et al., 1990).
Overall, it is clear that riboflavin a-glucoside is a very water-soluble form of the vitamin, which releases the vitamin after facile transport that does not involve the riboflavin transporter in the plasma membrane of cells. In fact this
TABLE 18-2 Flavins Present within Cells after a 45-minute Incubation with [3H]Riboflavin or its [3H]a-Glucoside
|
Initial Compound (1 µM) |
Riboflavin (% initial compound) |
a-Glucoside |
FMN |
FAD |
|
Riboflavin |
75 |
— |
16 |
4 |
|
Riboflavin a-glucoside |
32 |
55 |
9 |
2 |
|
NOTE: FMN, flavin mononucleotide; FAD, flavin adenine dinucleotide. |
||||
compound would provide an alternate way of delivering the vitamin, should there be a fault in the conventional transport system.
AUTHOR'S CONCLUSIONS AND RECOMMENDATIONS
The requirements for micronutrients as related to performance capability are arguably less well understood than requirements for calorie-yielding macronutrients. In particular, the changes in requirements that probably attend stressful conditions have not been assessed adequately for most of the water-soluble vitamins. Yet the means to acquire such information is at hand. Diverse techniques can be applied, but one that extends to animal models with the inherent potential for greater ranges of testing than ethically allowed with humans, is the isolated-cell technique. It is quite easy to project the type of experimentation outlined above with the disposition of natural glucosides of two vitamins in rat liver cells, to the more complete range of vitamins and derivatives that occurs within foods, using liver and other tissues from rats and other animals subjected to varying degrees of exertion or other stress. For example, exercising rats under controlled conditions followed by removal of cells for testing would allow information to be obtained on whether or not the increased physical demands affected uptake or metabolism of a vitamin or other essential nutrient. At present it is not clear, a priori, what could even be expected for some cases. Indications for work at the cellular level could then help focus what experiments might be appropriate using techniques that are often expensive, but aimed at the whole organism and, when possible, humans.
REFERENCES
Aw, T.Y., D.P. Jones, and D.B. McCormick. 1983 Uptake of riboflavin by isolated rat liver cells. J. Nutr. 113:1249–1254
Bowers-Komro, D.M., and D.B. McCormick 1987 Riboflavin uptake by isolated rat kidney cells. Pp. 449–453 in Flavins and Flavoproteins, D.E. Edmondson and D.B. McCormick, eds. New York: Walter de Gruyter.
Bowman, B.B., and D.B. McCormick 1989 Pyridoxine uptake by rat renal proximal tubular cells. J. Nutr. 119:745–749.
Bowman, B.B., D.B. McCormick, and I.H. Rosenberg 1989 Epithelial transport of water-soluble vitamins. Annu. Rev. Nutr. 9:187–199.
Chastain, J.L., and D.B. McCormick 1991 Flavin metabolites. Pp. 195–200 in Chemistry and Biochemistry of Flavoenzymes, vol. 1, F. Müller, ed. Boca Raton, Fla.: CRC Press.
Gilbert, J.A., and J.F. Gregory, III 1992 Pyridoxine-5'-ß-D-glucoside affects the metabolic utilization of pyridoxine in rats. J. Nutr. 122:1029–1035.
Ink, S.L., J.F. Gregory, and D.B. Sartain 1986 Determination of pyridoxine ß-glucoside bioavailability using intrinsic and extrinsic labeling in the rat. J. Agric. Food Chem. 34:857–862.
Joseph, T., and D.B. McCormick 1995 Uptake and metabolism of riboflavin-5'-a-D-glucoside by rat and isolated liver cells . J. Nutr. 125:2194–2198.
Kabir, H., J.E. Leklem, and L.T. Miller 1983 Relationship of the glycosylated vitamin B6 content of foods to the vitamin B6 bioavailability in humans. Nutr. Rep. Int. 28:709–715.
Kamimura, H., H. Ogata, and H. Takahara 1992 a-Glucoside formation of xenobiotics by rat liver a-glucosidases. Drug Metab. Disp. 20:309–315.
Kawai, T., H. Yamada, and K. Ogata 1972 Studies on transglucosidation to vitamin B6 by microorganisms. VII. Transport of pyridoxine glucoside into rabbit erythrocytes. J. Vitaminol. 18:183–188.
Kozik, A., and D.B. McCormick 1984 Mechanism of pyridoxine uptake by isolated rat liver cells. Arch. Biochem. Biophys. 229:187–193.
Leklem, J.E. 1994 Vitamin B6. Pp. 383–394 in Modern Nutrition in Health and Disease, 8th ed., vol. 1, M.A. Shils, J.A. Olson, and M. Shike, eds. Philadelphia: Lea and Febiger.
McCormick, D.B. 1975 Metabolism of riboflavin. Pp. 153–198 in Riboflavin, R.R. Rivlin, ed. New York: Plenum Press.
1979 Some aspects of the metabolism of sulfur-containing heterocyclic cofactors: Lipoic acid, biotin, and 8a-(S-L-cysteinyl)-riboflavin. Pp. 423–434 in Natural Sulfur Compounds, D. Cavallini, G.E. Gaull, and V. Zappia, eds. New York: Plenum Press.
1989 Two interconnected B vitamins: Riboflavin and pyridoxine. Physiol. Rev. 69:1170–1197.
McCormick, D.B., and L.D. Wright 1970 The metabolism of biotin and its analogues. Pp. 81–110 in Comprehensive Biochemistry, vol. 21, Metabolism of Vitamins and Trace Elements, M. Florkin and E.H. Stortz, eds. Amsterdam: Elsevier.
McCormick, D.B., and Z. Zhang 1993 Cellular assimilation of water-soluble vitamins in the mammal: Riboflavin, B6, biotin, and C. Proc. Soc. Exp. Biol. Med. 203:265–270.
Ohkawa, H., N. Ohishi, and K. Yagi 1983 Occurrence of riboflavinyl glucoside in rat urine. J. Nutr. Sci. Vitaminol. 29:515–522.
Rose, R.C., D.B. McCormick, T-K. Li, L. Lumeng, J.G. Haddad, Jr., and R. Spector 1986 Transport and metabolism of vitamins. Fed. Proc. 45:30–39.
Trumbo, P.R., and J.F. Gregory, III 1988 Metabolic utilization of pyridoxine-ß-glucoside in rats: Influence of vitamin B-6 status and route of administration. J. Nutr. 118:1336–1342.
Trumbo, P.R., M.A. Banks, and J.F. Gregory, III 1990 Hydrolysis of pyridoxine-5'-ß-D-glucoside by a broad-specificity ß-glucosidease from mammalian tissues. Proc. Soc. Exp. Biol. Med. 195:240–246.
Whitby, L.G. 1952 Riboflavinyl glucoside: A new derivative of riboflavin. Biochem. J. 50:433–438.
Yamamoto, I., N. Muto, E. Nagato, T. Nakamura, and Y. Suzuki 1990 Formation of a stable L-ascorbic acid a-glucoside by mammalian a-glucosideasecatalyzed transglucosylation. Biochem. Biophys. Acta 1035:44–50.
Yasumoto, K., H. Tsuji, and H. Mitsuda 1977 Isolation from rice bran of a bound form of vitamin B6 and its identification as 5'-O-(ß-D-glucopyranosyl)-pyridoxine. Agric. Biol. Chem. 41:1061–1067.
Zhang, Z., and D.B. McCormick 1992 Uptake and metabolism of N-(4'-pyridoxal)amines by isolated rat liver cells. Arch. Biochem. Biophys. 294:394–397.
Zhang, Z., J.F. Gregory III, and D.B. McCormick 1993 Pyridoxine-5'-ß-D-glucoside competitively inhibits uptake of vitamin B6 into isolated rat liver cells. J. Nutr. 123:85–89.
DISCUSSION
JOHN VANDERVEEN: Do you know if the traditional procedures for measuring nutrients in foods measure the glycosides as well? They are microbiological, I think, in both cases.
DONALD McCORMICK: Right. the answer to your question is "not uniformly." The results vary because the ability to measure glycosides depends on the nonspecific glucosidases that may or may not release it, and their presence varies from one microorganism to another. Some assays specifically do not do well; for instance, the streptococcal assays for B6 apparently do not have glucosidases that can release the B6 . So no, it is no good for assessing that.
It varies, so what you are dealing with, with any biological assay, including microbiological assays, are variations on competence for releasing bound forms, meaning that there is an error in that technique, too.












