Page 107
2
Atmospheric Chemistry Research Entering the Twenty-First
Century1
Summary
Atmospheric chemistry came of age during the latter half of the twentieth century. Through the application of modem analytical and computational techniques, scientists were able to elucidate the critical role the atmosphere plays as the "connective tissue" for life on Earth. In the process, another, more disturbing insight was uncovered: the activities of an increasingly populous and technological human society are changing the composition of the atmosphere on local to regional to global scales. Experience has shown that air pollution on local and regional scales can be environmentally and economically destructive. The consequences of chemical change on a global scale have yet to be fully assessed, but the potential for catastrophic effects exists.
1 Report of the Committee on Atmospheric Chemistry: W.L. Chameides (Chair), Georgia Institute of Technology; J.G. Anderson, Harvard University; M.A. Carroll, University of Michigan, Ann Arbor; J.M. Hales, ENVAIR; D.J. Hofmann, NOAA Aeronomy Laboratory; B.J. Huebert, University of Hawaii; J.A. Logan, Harvard University; A.R. Ravishankara, NOAA Aeronomy Laboratory; D. Schimel, University Corporation for Atmospheric Research; and M.A. Tolbert, University of Colorado, Boulder. The group gratefully acknowledges contributions from C. Ennis, NOAA Aeronomy Laboratory; D. Fahey, NOAA Aeronomy Laboratory; F. Fehsenfeld, NOAA Aeronomy Laboratory; I. Fung. University of Victoria, British Columbia; E.A. Holland, National Center for Atmospheric Research; D. Jacob. Harvard University; C.E. Kolb, Aerodyne Research, Inc.; H. Levy, II, NOAA Goddard Fluid Dynamics Laboratory; S. Liu, Georgia Institute of Technology; P. Reich, University of Minnesota; P. Samson. University of Michigan; and P. Tans, NOAA Aeronomy Laboratory.
Page 108
|
Box II.2.1 Environmentally Important Atmospheric Species |
|
|
These species are scientifically interesting and important to human health and welfare because of their radiative (e.g., climate changing) and/or chemical properties. They include the following: |
|
|
• Stratospheric ozone |
|
|
• Greenhouse gases |
|
|
• Photochemical oxidants |
|
|
• Atmospheric aerosols |
|
|
• Toxics and nutrients |
|
|
Documenting the changing concentrations and distribution of these species, elucidating the processes that control their concentrations, and assessing their impacts on important environmental and ecological parameters will define the principal challenges for atmospheric chemistry in the coming decades. |
|
The scientific questions facing atmospheric chemistry entering the twenty-first century are intellectually profound but are also of vital social and economic importance. They relate to atmospheric constituents that are fundamentally important to our environment: stratospheric ozone, greenhouse gases, ozone and photochemical oxidants in the lower atmosphere, atmospheric aerosols or particulate matter, and toxics and nutrients (see Box II.2.1). It is perhaps a measure of the strides made in recent decades, that the issues of atmospheric chemistry are familiar to the general public, policy makers, and scientists alike. Continued progress in the twenty-first century will require an ambitious, but judicious, commitment of financial, technological, and human resources to document the changing composition of the atmosphere and elucidate the causes and potential consequences of these changes.
Major Scientific Questions and Challenge
The principal focus for atmospheric chemistry research entering the twenty-first century will be the "Environmentally Important Atmospheric Species"— species that, by virtue of their radiative and/or chemical properties, affect climate, key ecosystems, and living organisms (including humans). From an intellectual point of view, these species are interesting because they influence the life support system of our planet. From a societal point of view, they are also of central importance because they directly impact human health and welfare.
Page 109
The challenge for atmospheric chemistry research in the coming decades follows:
Development and application of the tools and scientific infrastructure necessary to document and predict the concentrations and effects of Environmentally Important Atmospheric Species on a wide variety of spatial and temporal scales.
To meet this challenge, atmospheric chemistry research should be formulated around three fundamental questions:
1. What are the shorter-term periodic and longer-term secular trends in the concentrations of Environmentally Important Atmospheric Species on local to global scales? What are the causes of these trends?
2. How will the concentrations of these species change in the future? What are the most effective and feasible policy options for managing these changes?
3. What will be the totality of environmental effects of present and future trends in the concentrations of these species?
Overarching Research Challenges
The scientific strategy for atmospheric chemistry emerges logically from the application of these fundamental scientific questions to each of the Environmentally Important Atmospheric Species. It is a strategy that endeavors to continuously improve our understanding of the underlying chemical, physical, and ecological processes that control the concentrations of these species, while providing timely and relevant input to decision makers. Toward these ends, the scientific research strategy in atmospheric chemistry must include the following:
• Document the chemical climatology and meteorology of the atmosphere, particularly their variability and long-term trends, through the development and maintenance of diverse and interrelated arrays of monitoring networks.
• Develop and evaluate predictive tools and models of atmospheric chemistry through a synthesis of information gathered from process-oriented field studies, laboratory experiments, and other observational efforts; their representation in mathematical/numerical algorithms; and the testing of these algorithms in well-posed model-evaluation field experiments.
• Provide assessments of the efficacy of environmental management activities through the gathering and interpretation of relevant air quality data.
• Be holistic and integrated in the study of the Environmentally Important Atmospheric Species and of the chemical, physical, and ecological interactions that couple them together.
Page 110
Disciplinary Research Challenges
The disciplinary challenges listed below focus on the specific, key scientific issues facing the atmospheric chemistry community in the twenty-first century:
• Stratospheric Ozone Challenges: Document the distributions, variability, and trends of stratospheric ozone and the key species that control its catalytic destruction; elucidate the coupling between chemistry, dynamics, and radiation in the stratosphere and upper troposphere.
• Greenhouse Gas Challenges: Elucidate the processes that control the abundances, variabilities, and long-term trends of atmospheric CO2 (carbon dioxide), CH4 (methane), N2O (nitrous oxide), and upper-tropospheric and lower-stratospheric O3 (ozone) and water vapor; and expand global monitoring networks to include upper-tropospheric and lower-stratospheric O3 and water vapor.
• Photochemical Oxidant Challenges: Develop the observational and computational tools and strategies needed by decision makers to effectively manage ozone pollution; elucidate the processes that control, and the interrelations that exist between, the ozone precursor species, tropospheric ozone, and the oxidizing capacity of the atmosphere.
• Atmospheric Aerosol Challenges: Document the chemical, physical, and radiative properties of atmospheric aerosols, their spatial extent, and long-term trends; elucidate the chemical and physical processes responsible for determining the size, concentration, and chemical characteristics of atmospheric aerosols.
• Toxics and Nutrients Challenges: Document the rates of chemical exchange between the atmosphere and key ecosystems of economic and environmental import; elucidate the extent to which interactions between the atmosphere and biosphere are influenced by changing concentrations and deposition of harmful and beneficial compounds.
Infrastructural Initiatives
The following infrastructural initiatives provide the resources and capabilities recommended to accomplish the disciplinary challenges:
• Global Observing System: deployment of an observing system for moderately lived species to complement ongoing networks and measurement platforms focusing on long-lived species and stratospheric ozone.
• Ecosystem Exposure Systems: deployment of monitoring networks capable of assessing ecosystem exposure to primary and secondary toxics and nutrients.
• Surface Exchange Measurement Systems: development and deployment of measurement systems capable of quantifying chemical exchange between the atmosphere and key biological or ecosystems.
Page 111
• Environmental Management Systems: demonstration and assessment of the feasibility of operational "chemical meteorology" as a prognostic tool for environmental managers and regulators.
• Instrument Development and Technology Transfer: development of programs and facilities to support the evaluation of new atmospheric chemical instruments and their transfer to the scientific, regulatory, and private sector communities.
• Fundamental Condensed Phase and Heterogeneous Chemistry: development and maintenance of laboratory facilities focused on condensed phase and heterogeneous chemical processes relevant to the atmosphere.
Expected Benefits and Contribution to National Well-Being
The scientific questions to be addressed by the atmospheric chemistry research community entering the twenty-first century are central to our understanding of the chemical and physical environment in which we human beings must reside. For this reason, the science of atmospheric chemistry is highly relevant to the future development and economic vitality of our society. Today, the changing chemistry of our atmosphere on local, regional, and global scales is an observational fact. These changes are impacting human health and placing economically and environmentally important resources and ecosystems at risk. At the same time, air quality management activities in the United States cost tens of billions of dollars annually. Research in atmospheric chemistry and the resulting improvements in our predictive capabilities will help us to maximize the environmental and economic benefits gained from these sizable investments in air quality management, while also teaching us how to minimize the deleterious effects of human activity on the chemical and physical environment.
Introduction and Overview
As the world stands on the threshold of a new millennium, the atmospheric chemistry community stands at the portal of a new era of scientific research. During the latter half of the twentieth century, the discipline of atmospheric chemistry came of age. Scientific study revealed the crucial role that the chemistry of the atmosphere plays in the life support system of the planet, acting as a "connective tissue" by which organisms of the biosphere interact and exchange materials and energy. It also uncovered a more disturbing insight: the activities of an increasingly populous and technological human society are changing the composition of the atmosphere on local, regional, and even global scales. Experience has shown that air pollution on local and regional scales can be environmentally and economically destructive. The consequences of chemical change on a global scale could be even more damaging. Thus, the scientific questions
Page 112
facing atmospheric chemistry are not only intellectually challenging but also of vital social and economic importance.
The challenge for atmospheric chemistry as it enters the twenty-first century will be to build on the discoveries of the twentieth century by maintaining its scientific vitality and rigor while also making the results of its scientific and technological advances available to influence the nation's and the world's social and economic development. In this Disciplinary Assessment, we discuss the strategy that will be necessary to address the major scientific issues of the discipline while also providing decision makers with the information and tools they require to manage and maintain environmental and economic vitality. We begin our discussion with a statement of the mission for atmospheric chemistry research entering the twenty-first century.
The Mission
Development and application of the tools and scientific infrastructure necessary to document and predict the concentrations and effects of Environmentally Important Atmospheric Species on a wide variety of spatial and temporal scales.
In identifying the mission for atmospheric chemistry research entering the twenty-first century, we have adopted three basic premises:
1. The financial and human resources available for research and development in the coming decades will be limited.
2. The activities of an increasingly populous and technological society have and will continue to perturb critical environmental factors that affect the natural resources on which our society relies.
3. Unraveling the mechanisms that couple the chemistry of the atmosphere to the life support system of the planet represents one of the major intellectual and technological challenges of the coming decades.
Premises 1 and 2 relate to the resource- and policy-relevant issues that must be considered in defining the mission for atmospheric chemistry research, whereas premise 3 focuses on the intellectual or curiosity-based raison d'etre for the discipline. The prospect of limited resources for research and development indicated in premise 1 demands that a rigorous prioritization be applied to any contemporary research program, so that the most pressing scientific issues can be addressed in the allocation of public resources to the scientific community. Premise 2 suggests that priority should be placed on developing a scientifically robust, predictive, and systematic understanding of the Earth system, its chemical environment, and the relationships between the economic and technological growth of the world's nations and the environmental vitality and natural resources on which they depend.
The development of a research program often requires compromises between
Page 113
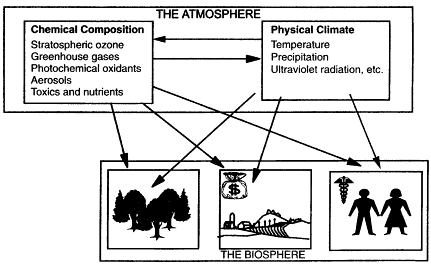
Figure II.2.1
Environmentally Important Atmospheric Species are atmospheric
constituents that affect human health and welfare and thus are
central to a policy-relevant research program in atmospheric
chemistry. Because these species drive the interaction between the
atmosphere and the life support system of the planet, they are also
central to a curiosity-based research program in atmospheric
chemistry. Environmentally Important Atmospheric Species include
greenhouse gases (e.g., CO2, CH4, N2O),
aerosols, and stratospheric ozone—species that, because of
their radiative properties, affect the climate and other physical
characteristics of our environment. They also include the
photochemical oxidants and tropospheric ozone, acid aerosols, and a
wide variety of toxic and nutritive substances that, because of
their chemical properties, affect humans and ecosystems of economic
and environmental importance when they come in direct contact with
them. Although the radiatively important species' effects are more
commonly felt on a global scale, the effects of the ecologically
important species are most often experienced on local-to-regional
scales. Nevertheless, despite the varying scales of their radiative
and chemical effects, research has revealed that the atmospheric
cycles of these species are coupled together through complex
photochemical and dynamical interactions. Unraveling these complex
interactions represents one of the major challenges of atmospheric
chemistry research in the coming decades.
the priorities dictated by policy-relevant issues and those dictated by more theoretical interests. However, in the case of atmospheric chemistry research we find a strong resonance between the two. Atmospheric chemistry research in the coming decades should be focused on documenting and predicting the concentrations and effects of the chemical constituents that most directly affect the physical and biological environment and, by extension, human health and welfare. We refer to these species, here, in the most generic sense, as the Environmentally Important Atmospheric Species that, by virtue of their radiative and/or chemical properties, directly affect living systems and key environmental parameters (see Figure II.2.1).
Page 114
For atmospheric chemistry to make significant scientific advances in the coming decades, however, its research focus on these Environmentally Important Atmospheric Species must go well beyond simple observation and documentation of chemical content and change, to a rigorous investigation of the underlying chemical, physical, and ecological processes that determine the atmospheric concentrations of these species. It is, after all, only through understanding these processes that a genuine appreciation of the atmosphere and its relationship to the Earth system can be fostered, and a reliable predictive capability can be achieved and made available for the development of effective public policy. Additionally, because the atmosphere and the stresses placed on it are continually changing, a significant portion of the resources made available for atmospheric chemistry research in the coming decades must be used to develop an enduring research infrastructure that can inform decision makers in an open, effective, and responsive fashion.
The mission for atmospheric chemistry research in the coming decades must therefore combine a focus on the Environmentally Important Atmospheric Species with a commitment to the development of a comprehensive, long-term research capability and technological infrastructure. Hence, the mission of atmospheric chemistry in the coming decades:
Development and application of the tools and scientific infrastructure necessary to document and predict the concentrations and effects of Environmentally Important Atmospheric Species on a wide variety of spatial and temporal scales.
In the following sections, we consider how to accomplish this mission most effectively by first considering what is now known about the atmosphere and then identifying the key unresolved scientific questions surrounding a number of Environmentally Important Atmospheric Species and the research challenges that grow from these questions.
Insights of the Twentieth Century
By grappling with a number of critical, but largely unforeseen environmental problems in recent decades, scientists have gained fundamental new insights about the atmospheric chemical system.
The study of atmospheric chemistry as a quantitative, scientific discipline can be traced to the eighteenth century when world-renowned chemists such as Joseph Priestley, Antoine-Laurent Lavoisier, and Henry Cavendish undertook the investigation of the chemical components of the atmosphere (Farber, 1961; Weeks and Leicester, 1968). It was largely through their efforts, as well as those of a number of prominent chemists and physicists who succeeded them in the nine-
Page 115
|
TABLE II.2.1 Important Trace Species of the Atmospherea |
||
|
Species |
Concentration (Mole Fraction) |
Principal Sources |
|
Methane (CH4) |
1.6 × 10-6 |
Biogenic |
|
Carbon Monoxide (CO) |
(0.5 - 2) × 10-7 |
Photochemical, Anthropogenic |
|
Ozone (O3) |
10-8 - 10-6 |
Photochemical |
|
Reactive Nitrogen (NOy) |
10-11 - 10-6 |
Lightning, Anthropogenic |
|
Ammonia (NH3) |
10-11 - 10-9 |
Biogenic |
|
Particulate Nitrate (NO3-) |
10-12 - 10-8 |
Photochemical, Anthropogenic |
|
Particulate Ammonium (NH4-) |
10-11 - 10-8 |
Photochemical, Anthropogenic |
|
Nitrous Oxide (N2O) |
3 × 10-7 |
Biogenic, Anthropogenic |
|
Hydrogen (H2) |
5 × 10-7 |
Biogenic, Photochemical |
|
Hydroxyl (OH) |
10-13 - 10-11 |
Photochemical |
|
Peroxyl (HO2) |
10-13 - 10-11 |
Photochemical |
|
Hydrogen Peroxide (H2O2) |
10-10 - 10-8 |
Photochemical |
|
Formaldehyde (H2CO) |
10-10 - 10-9 |
Photochemical |
|
Sulfur Dioxide (SO2) |
10-11 - 10-9 |
Anthropogenic, Volcanic |
|
Dimethylsulfide (CH3CCH3) |
10-11 - 10-10 |
Biogenic |
|
Carbon Disulfide (CS2) |
10-11 - 10-10 |
Anthropogenic, Biogenic |
|
Carbonyl Sulfide (OCS) |
10-10 |
Anthropogenic, Biogenic |
|
Particulate Sulfate (SO4-) |
10-11 - 10-8 |
Anthropogenic, Photochemical |
|
a After Chameides and Davis (1982). |
||
teenth century, that the identity and concentration of the major components of the atmosphere (i.e., nitrogen, oxygen, water, carbon dioxide, and the rare gases) were established.
In the late nineteenth and early twentieth centuries, atmospheric chemists shifted their focus from identifying the major atmospheric constituents to consideration of the trace constituents, that is, the gaseous and aerosol atmospheric species having concentrations of less than a few parts per million per volume of air (i.e., ppmv). The application of modem chemical analytical techniques revealed the atmosphere to be a reservoir of a myriad of trace species, whose presence can be attributed to a complex array of geological, biological, chemical, and in many cases, anthropogenic processes (see Table II.2.1). Moreover, these trace species were found to have a disproportionately large impact on our environment. In some instances, they adversely affect plant and animal life because of their toxic properties; in other instances, they benefit these or other organisms because of their nutritive properties; in still other instances, they affect the physical climate because of their radiative properties.
The latter half of the twentieth century has seen another major shift in atmospheric chemistry as scientists attempt to grapple with a number of potentially critical environmental problems, including stratospheric ozone depletion, urban
Page 116
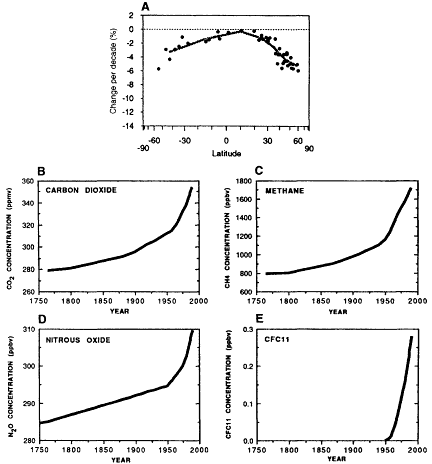
Figure II.2.2
From an atmospheric chemistry point of view, global change is an observational fact
not a theoretical possibility. (A) Average change per decade in the total atmospheric
ozone column as a function of latitude based on recent Dobson station measurements
(after WMO, 1995). (B-E) Average global concentrations of CO2, CH4,
N2O, and CFC-11 since the mid-1700s (after IPCC, 1990).
photochemical smog, and rising concentrations of ''greenhouse gases'' (NRC, 1984). In the process, a new, policy-relevant research paradigm for atmospheric chemistry has developed that has profoundly altered its role in society. More importantly, the insights gained from the study of these environmental crises have irrevocably changed our understanding of the atmospheric chemical system in which we as a species must reside. The major aspects of these new insights are outlined below.
Page 117
The Chemical State of the Atmosphere Has Changed in the Past and Is Continuing to Change
Observations have shown irrefutably that the chemistry of the atmosphere is changing on local, regional, and global scales; indeed, from a chemical point of view, global change is an observational fact not a theoretical possibility. The annual appearance of the Antarctic ozone hole provides striking evidence of the atmosphere's vulnerability to chemical perturbation. Although smaller in magnitude, the depletion of stratospheric ozone in the temperate latitudes over the past decade is perhaps equally disturbing (Figure II.2.2A). Moreover, present-day measurements coupled with analyses of ancient air trapped in ice cores provide a record of dramatic, global increases in the concentrations of a number of long-lived greenhouse gases such as carbon dioxide, methane, nitrous oxide, various chlorofluorocarbons (CFCs), and other halocarbons (Figure II.2.2B-E). Although secular trends in shorter-lived species are more difficult to document, a strong case can be made that the abundances of tropospheric ozone and sulfate and carbonaceous aerosols have also increased significantly in the Northern Hemisphere during the past century (NRC, 1993).
Humans Are a Significant Driving Force in Global Chemical Change
Many of the recent changes in atmospheric composition can be traced to anthropogenic causes. A classic example is that of atmospheric CO2, whose increasing rates of production from the burning of fossil fuels and biomass closely mimic its rising atmospheric abundance since the Industrial Revolution (see Figure II.2.3). In other examples, the forcing from anthropogenic activities is less obvious, largely arising when the photochemical oxidation and degradation processes of anthropogenic emissions lead to the production of secondary products that perturb important environmental parameters. Examples of these indirect perturbations include the release of chlorofluorocarbons that cause stratospheric ozone depletion, the emission of sulfur oxides that result in increasing concentrations of radiatively important and health-damaging sulfate aerosols, and the emissions of nitrogen oxides and volatile organic compounds that lead to the production of tropospheric ozone and other photochemical oxidants.
Chemical Emissions into the Atmosphere Can Have Long-Term Environmental Consequences That May Be Difficult to Reverse
Because of the long time scales associated with many of the processes that affect atmospheric composition, the chemicals we put into the atmosphere and the environmental effects they engender can persist for decades or even centuries. A prime example is the long-term impact of anthropogenic CFCs on stratospheric ozone.
Page 118
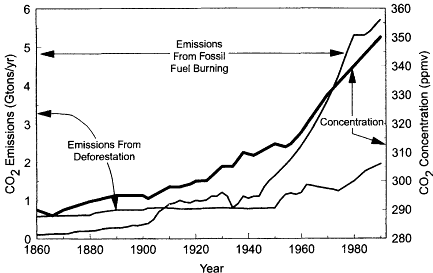
Figure II.2.3
Correlation between anthropogenic CO2
emissions and atmospheric CO2
concentrations. Left axis:
Global annual CO2emissions from fossil fuel
burning and deforestation. Right axis:
Annual average atmospheric CO2 concentration,
inferred from analyses of air trapped in ice cores and by direct
measurements. Increasing concentrations of CO2
and other greenhouse
gases may be triggering a perturbation of global climate that includes
increasing surface temperatures and alterations in rainfall patterns and
intensities. Source: Figure prepared from data in IPCC, 1995.
During the formation of the ozone hole over Antarctica each austral spring, a major fraction (currently about two-thirds) of the protective column of ozone disappears. Continuous measurements of the ozone column over Haley Bay at 76°S indicate that the ozone hole first began to form in the mid-1970s and has returned each year since, with varying but essentially increasing intensity (see Figure II.2.4A). An international program of research in the 1980s established that the ozone hole is caused by a unique combination of dynamical and anthropogenically driven photochemical processes over the Antarctic. The dynamical processes give rise to a wintertime polar vortex and the concomitant formation of polar stratospheric clouds (PSCs). The photochemical processes, driven largely by chlorine compounds from the photochemical degradation of anthropogenically produced CFCs, give rise to heterogeneous chemical reactions on the PSCs that cause a rapid and dramatic depletion of stratospheric ozone. In large part as a result of these findings, international protocols were implemented in the early 1990s to reduce and ultimately ban CFCs. These protocols should ultimately bring about the demise of this troubling phenomenon. Unfortunately, because of the long lifetime of CFCs in the atmosphere, it is projected that the ozone hole
Page 119
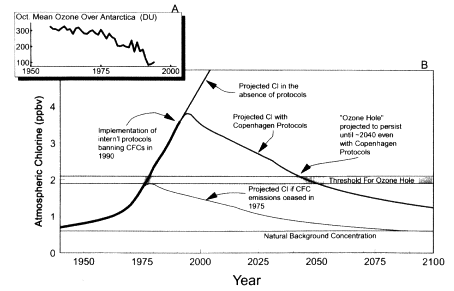
Figure II.2.4
History and projected evolution of atmospheric chlorine and the Antarctic ozone
hole. (A) October mean ozone column measured over Halley Bay, Antarctica (76°S)
from 1957 to 1993 in Dobson Units (DU). (B) Atmospheric concentration of chlorine
as a function of year; heavy solid line indicates observed and inferred concentrations
from 1940 to the present; thin solid lines indicate projected concentrations under the
Copenhagen Protocols if no protocols are implemented and if CFCs had been banned
in 1975. Also shown are the estimated natural background chlorine concentration and
the estimated threshold chlorine concentration needed to produce an ozone hole. Note
that actual chlorine concentrations first attained the threshold concentration in 1975.
Source: Figure prepared from data in WMO, 1995.
will continue to appear each austral spring for the next 40 to 50 years (Figure II.2.4B). Interestingly, if CFCs had been banned in 1975, one year after the connection between these compounds and stratospheric ozone depletion was first uncovered (Cicerone et al., 1974; Crutzen, 1974; Molina and Rowland, 1974), it is likely that the occurrence of the ozone hole would have been averted.
The Chemistry and Dynamics of the Atmosphere Interact with Each Other and with the Biosphere Through Complex, Nonlinear Mechanisms
The chemical, dynamical, and biological processes that shape atmospheric composition interact in complex ways and can give rise to unexpected but important phenomena. For this reason, effective management of the environment requires a comprehensive and quantitative understanding of all these processes. A prime example of the importance of understanding the complexities of the
Page 120
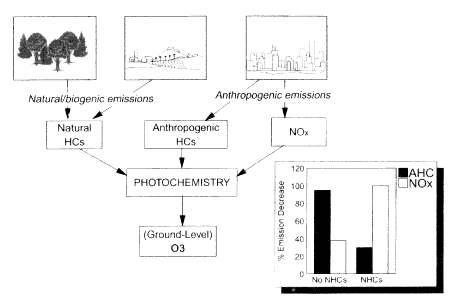
Figure II.2.5
Interaction of natural and anthropogenically emitted hydrocarbons (HCs) and nitrogen
oxides (NOx) in the production of ozone (O3) and photochemical smog. Inset: Estimated
percentage decrease in anthropogenic HC and NOx emissions required to bring Atlanta,
Georgia, into attainment of the National Ambient Air Quality Standard for ozone for a
selected air pollution episode in the summer of 1990 based on model calculations that
neglect natural hydrocarbons (labeled No NHCs) and model calculations that include natural
hydrocarbons (labeled NHCs). Without the inclusion of natural HCs the calculations
indicate that emission decreases in anthropogenic HCs (AHCs) would most effectively
reduce ozone pollution. However, inclusion of the effects of natural HC emission leads to
the opposite conclusions, namely, that a NOx-based strategy is most effective. Source:
Cardelino and Chameides, 1995.
atmospheric chemical system can be found in the nation's attempts to control photochemical smog.
Photochemical smog, with its high concentrations of ground-level ozone and other oxidants, is most severe during hot, stagnant weather conditions (NRC, 1991). It is generated by a complex series of photochemical reactions involving the oxidation of hydrocarbons in the presence of nitrogen oxides and sunlight (see Figure II.2.5). Thus, the control of photochemical smog can, in principle, be accomplished by decreasing emissions of hydrocarbons or nitrogen oxides or both. In practice, however, the problem has proved much more difficult to solve, as described below.
Despite the implementation of increasingly tighter controls on (mostly hydrocarbon) emissions in the United States since the 1970s, photochemical smog remains a major environmental problem in many of the nation's cities (EPA,
Page 121
1995). Much of this difficulty can be traced to the complexities of the chemical system. The rate of ozone production is a nonlinear function of the mixture of hydrocarbons and nitrogen oxides in the atmosphere. Depending on the relative concentrations of these species, ozone production rates can be sensitive to hydrocarbons and insensitive to nitrogen oxides (in which case a hydrocarbon control strategy would be most effective) or vice versa. Another difficulty arises from the potential for significant natural hydrocarbon emissions from trees and other vegetation. Because these natural emissions cannot be controlled, a strategy based on decreasing anthropogenic hydrocarbon emissions may not be effective even when ozone production in the area is most sensitive to hydrocarbons (inset in Figure II.2.5). It now appears that the lack of progress in reducing photo-chemical smog in many of the more sylvan urban areas of the United States can, in part, be traced to a failure to account for the effect of the natural hydrocarbon emissions (Chameides et al., 1988; NRC, 1991).
Linkages Between Atmospheric Species and Their Chemical Cycles Are Complex and Pervasive
The chemical constituents of the atmosphere are not processed independently of each other. They are instead linked through a complex array of chemical and physical processes. As a result of these linkages, a perturbation of one component of the atmospheric chemical system can lead to significant, nonlinear effects that ripple through the other components of the system and, in some cases, to feedbacks that can either amplify or damp the original perturbation. Figure II.2.6 provides a schematic illustration of the kinds of interactions and feedbacks that can occur.
Disciplinary Research Challenges
The successes of atmospheric chemistry research over the past decades have raised a number of intriguing scientific questions about the workings of the Earth system.
The identification and quantification of secular trends in the trace composition of the atmosphere, and the elucidation of complex mechanisms that link atmospheric species to each other and to atmospheric dynamics, are testaments to the vitality and growing technological capabilities of the research programs focusing on atmospheric chemistry and global change. However, while the successes of atmospheric chemistry research over the past decades have answered important scientific questions, they have raised many intriguing new questions that are at the core of understanding the inner workings of the Earth system and must be answered to cope effectively with society's ubiquitous and growing environmental problems. In their most fundamental form, these are the outstanding questions:
Page 122
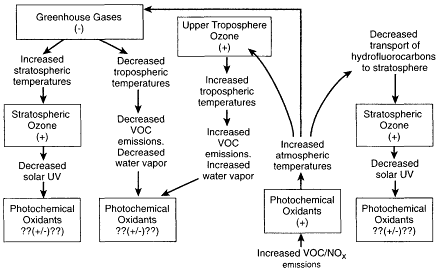
Figure II.2.6
Conceptual diagram illustrating potential interactions and feedbacks between
photochemical oxidants, greenhouse gases, and stratospheric ozone that might
ensue following an initial increase in the emissions of ozone precursor species,
that is, volatile organic compounds (VOCs) and nitrogen oxides (NOx). Note: +and
- indicate effect of the preceding change on the relevant species' concentration.
1. What are the causes of the variability and secular trends in the concentrations of Environmentally Important Atmospheric Species?
2. How will the concentrations of these species change in the future? What are the most effective and feasible policy options for managing these changes?
3. What are the environmental effects of present and future trends in the concentrations of these species?
These three questions form the basic framework and focus for atmospheric chemistry research in the coming decades. However, because each of the Environmentally Important Atmospheric Species has unique chemical properties and spatial and temporal variabilities, the application of these fundamental questions will require a specific and unique research strategy for each species. The main elements of these strategies are outlined below and then discussed in greater detail.
Stratospheric Ozone
Although great strides have been made in our understanding of stratospheric ozone depletion, major uncertainties remain. Thus, we must remain diligent in our focus on the stratosphere.
Page 123
Observations have now documented a significant depletion in the levels of ozone in the stratosphere. Epidemiological data showing the role of stratospheric ozone in protecting life from the harmful effects of ultraviolet radiation, along with the scientific evidence linking the depletion of stratospheric ozone to the emissions of anthropogenic chlorofluorocarbons and other ozone-depleting substances, has led to the Montreal Protocol and its subsequent amendments that require a phaseout of CFCs and similar compounds that can deplete stratospheric ozone. Although great strides clearly have been made in our understanding of stratospheric O3 and its depletion, major uncertainties remain. For example, current models of stratospheric chemistry significantly underpredict the magnitude of ozone depletion observed in recent decades over the midlatitudes during winter and spring (see Figure II.2.7).
If there is one basic lesson to be learned from our experience with the discovery of the ozone hole it is that in the face of scientific uncertainty, we must remain diligent in our focus on the stratosphere. Thus, a continued commitment to stratospheric ozone research in the coming decades is essential. This commitment should focus on two major research challenges: (1) Continued monitoring of the spatial distribution of stratospheric ozone and the key species responsible for its catalytic destruction in order to document the evolving nature of the stratosphere; and (2) elucidation of the coupling between chemistry, dynamics, and radiation in the stratosphere and upper troposphere in order to develop a more reliable predictive capability for stratospheric ozone.
Trends in Atmospheric Greenhouse Gases
A reliable prediction of future climatic trends demands first and foremost a reliable prediction of future trends in concentrations of the greenhouse gases. However, there are major gaps in our understanding of the processes that control the concentrations of these gases.
Greenhouse gases act as atmospheric thermal insulators; by absorbing infrared radiation from the Earth's surface and reradiating a portion of this radiation back to the surface, they enhance the so-called greenhouse effect that warms the Earth's surface above the warming caused by directly incident solar radiation. These gases include primary greenhouse gases emitted directly into the atmosphere (e.g., CO2, CH4, N2O, CFCs) and secondary greenhouse gases photo-chemically produced within the atmosphere (e.g., O3). Water vapor (H2O), the most important of the greenhouse gases, is both emitted into the atmosphere (via evapotranspiration) and produced photochemically in the stratosphere by the oxidation of CH4. Ozone is unique in that it not only acts as a greenhouse gas (by absorbing infrared radiation) but also is an important absorber of solar radiation.
As noted earlier, the observed increases in concentrations of CO2, CH4, N2O, and halocarbons provide one of the clearest manifestations of atmospheric global
Page 124
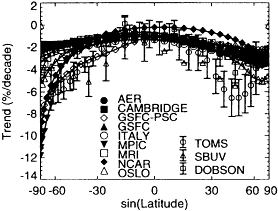
A Dobson Total Ozone Trend - Dec-Jan-Feb
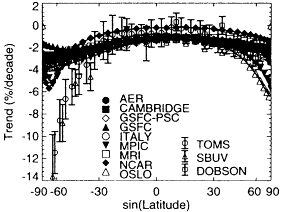
B Dobson Total Ozone Trend - Jun-Jul-Aug
Figure II.2.7
Total ozone column change from 1980 to 1990 inferred
from TOMS, SBUV, and Dobson measurement systems
compared with predicted trends from nine different atmospheric
models. (A) Data and model calculations for December-
January-February. (B) Data and model calculations for
June-July-August. Note the deviations between predictions
and measurements in the midlatitudes of each of the
hemispheres. The inability of present-day models to fully
explain the levels of ozone depletion seen in the atmosphere
indicates the need to remain diligent in monitoring the
evolution of the stratosphere under declining burdens of
CFCs and to continue to investigate the stratosphere to
develop a more reliable predictive capability. Note: SBUV =
Solar Backscatter Ultraviolet Spectrometer; TOMS = Total
Ozone Mapping Spectrometer. Source: After WMO, 1995.
Page 125
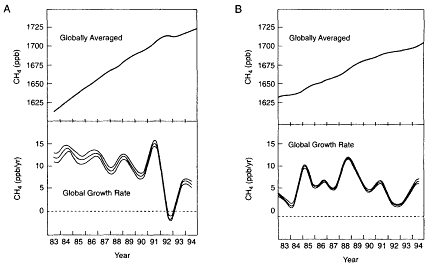
Figure II.2.8
Globally averaged concentration (top panel) and annual concentration change
or trend (bottom panel) in (A) CH4 and (B) CO2 during the 1980s and 1990s. The
causes of the large year-to-year fluctuations in the CH4 and CO2 trends, especially
during the early 1990s, are largely unknown and suggest that major gaps exist in
our understanding of the processes that control these species' concentrations
on an annual time scale. Source: After P. Tans, private communication, 1996.
change (Figure II.2.2B-E). These increasing concentrations are, in many cases, driven by human activity and related to energy use, industrialization, land use change, and/or agriculture. It is likely that increases in these gases are responsible, at least in part, for the global warming that has occurred in this century, and the warming might have been even greater were it not for the offsetting effects of aerosols such as sulfates (see below).
A reliable prediction of future climatic trends demands first and foremost a reliable prediction of the trend in concentrations of greenhouse gases over time. However, the dearth of data on present-day trends in upper-tropospheric and lower-stratospheric O3 and H2O makes projections of future trends for these species highly problematic. Moreover, the seemingly erratic and unexplained variations in the concentrations of many greenhouse gases in the early and mid-1990s (see Figure II.2.8) have exposed major gaps in our understanding of the processes that control the concentrations of these species.
In order to develop more reliable predictions of greenhouse gas trends and concentrations, it is imperative that we (1) maintain global monitoring for long-lived greenhouse gases, while expanding the monitoring capability for upper-tropospheric and lower-stratospheric O3 and H2O in order to document the changing radiative character of the atmosphere; and (2) elucidate the processes
Page 126
that control the concentrations and variability of CO2, CH4, N2O, and upper-tropospheric and lower-stratospheric O3 and H2O.
Tropospheric Photochemical Oxidants
The critical scientific issues surrounding photochemical oxidants range from the formation of urban air pollution to the role of tropospheric ozone in global climate change.
Photochemical oxidants are highly reactive compounds produced by photo-chemical reactions (Haagen-Smit, 1952). These compounds control the oxidizing capacity of the atmosphere and thus the atmospheric residence times and abundances of a host of other Environmentally Important Atmospheric Species (Figure II.2.6). Moreover, because of their reactivity with living tissue, photo-chemical oxidants can have deleterious effects on human health and ecosystems (LeFohn, 1992).
The most abundant photochemical oxidant is ozone. Paradoxically, whereas ozone in the stratosphere protects living organisms from the harmful effects of solar ultraviolet radiation, ozone in the lower atmosphere can have adverse effects on plants, animals, and human health. As described below, the critical scientific issues surrounding photochemical oxidants range from more fully understanding the processes involved in the development of urban and rural ozone pollution to elucidating the role of changing concentrations of tropospheric ozone in global environmental change.
In the United States, increasingly tighter emission controls have been implemented since the late 1970s to reduce ozone concentrations in the nation's urban centers and thereby protect human health. Measures now being implemented under the Clean Air Act Amendments of 1990 to address this problem are estimated to eventually cost the nation more than $10 billion annually. Despite these measures (see Figure II.2.9), 77 cities are still cited by the U.S. Environmental Protection Agency (U.S. EPA) for failure to comply with the ozone National Ambient Air Quality Standard (NAAQS) designed to protect human health (EPA, 1995). In addition, there is growing concern that enhanced O3 concentrations in the rural areas of the United States are harming agricultural and forest ecosystems of economic and environmental import (NRC, 1991).
On regional and global scales, data are sparse but suggest that significant changes in tropospheric ozone concentration may also be occurring, at least in some regions, and that these changes are distinctly different from those occurring in cities in the United States. Retrospective analyses of data gathered in the nineteenth century suggest that tropospheric O3 concentrations over the European continent (and perhaps even the Northern Hemisphere, as a whole) may have increased by a factor of two or more this century (see Figure II.2.10). Contempo-
Page 127
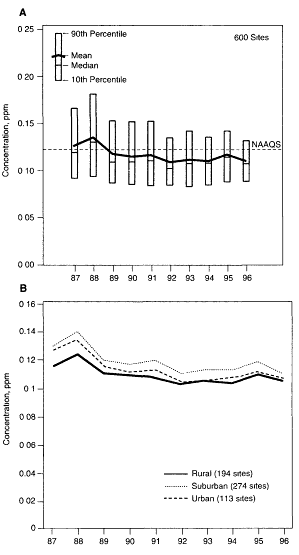
Figure II.2.9
Average trend in annual maximum one-hour ozone concentration
measured at urban and suburban sites in the United States and
recorded in the Aerometric Information Retrieval System. Data
indicate a modest (˜6 percent) decrease from 1986 to 1995 in both
overall air quality (II. 2.9 A) and in its components (II.2.9B). This
modest decrease occurred in the face of sizable investments made
in pollution control over this period, on the one hand, and significant
economic expansion, on the other. Source: After U.S. Environmental
Protection Agency, 1995.
Page 128
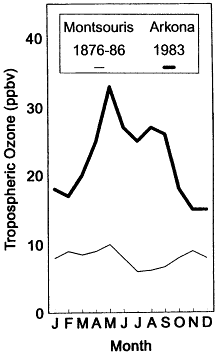
Figure II.2.10
Average O3 concentration as a function of month measured at
high altitude sites in Europe. Lighter solid line represents
concentration inferred from a retrospective analysis of data
collected in the late 1800s. Heavier solid line represents data
collected in 1983 using modern instrumentation. Source:
After Volz-Thomas and Kley, 1988..
rary data collected at remote surface sites over two decades indicate a more complex pattern, with increasing O3 levels in some regions of the troposphere, little or no change in other regions, and decreasing O3 levels in still others (see Figure II.2.11).
The reasons ozone is increasing in some regions of the globe and decreasing in others have yet to be fully explained. Moreover, the climatic and ecological impacts of these changes are poorly understood. Perhaps most importantly from the viewpoint of global change, the long-term trend in the overall oxidizing capacity of the atmosphere implied by these changing ozone concentrations is not known. As delineated in Figure II.2.6, a change in the overall oxidizing capacity of the atmosphere could have effects that ripple throughout the atmosphere's chemical system, ultimately affecting greenhouse gas and stratospheric ozone concentrations, as well as those of the photochemical oxidants.
Two research challenges emerge from this discussion: It is recommended (1) that the observational and computational tools and strategies needed by decision makers to devise more effective urban- and regional-scale ozone pollution abatement strategies and test their efficacy once implemented be developed; and (2) that the processes that control and the interrelations that exist between the ozone precursor species, tropospheric ozone, and the oxidizing capacity of the atmo-
Page 129
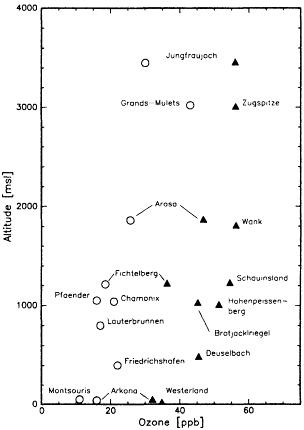
Figure II.2.11
Historical (circles) and recent (triangles) surface ozone
concentrations from different locations in Europe as a
function of altitude. Source: After Staehlin et al., 1994.
sphere be elucidated in order to develop a more robust predictive capability and better understand the consequences of perturbation of the cycle of photochemical oxidants in the atmosphere. The research strategy for addressing these challenges is presented later.
Chemical and Physical Properties of Atmospheric Aerosols
Aerosols play an important role in climate change, stratospheric ozone depletion, and air quality issues. However, large uncertainties in the chemical, physical, and radiative properties of atmospheric aerosols render quantitative assessment of their effects problematic.
Page 130
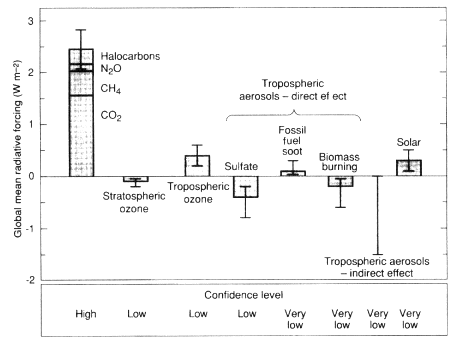
Figure II.2.12
Estimates of globally averaged radiative forcing due to changes in greenhouse gases
and aerosols from the preindustrial era to the present and changes in solar variability
since 1850, with uncertainties in estimates indicated by vertical lines. Although of ''low''
and "very low" confidence, calculated cooling from the direct and indirect aerosol effect
may be large enough to have significantly offset warming from greenhouse gases.
Source: After IPCC, 1996.
Atmospheric aerosols consist of solid and liquid particles suspended in the atmosphere. They include primary aerosols that arise from direct particulate emissions and secondary aerosols that arise from chemical reactions involving gaseous precursors. Aerosols have the potential to play a critical role in shaping our environment. They affect climate and stratospheric ozone concentrations and may pose a significant health threat in urban and industrial centers. However, large uncertainties in our knowledge of the chemical and physical properties of atmospheric aerosols, the processes that cause their formation, and the processes by which they influence cloud formation and radiative transfer of energy render quantitative assessments of their effects and society's influence on them problematic.
Atmospheric aerosols can affect climate in two ways: (1) directly, by which aerosols, depending on their properties, either cool the Earth by reflecting a portion of incoming solar radiation back to space or warm the Earth by absorbing radiant energy; and (2) indirectly, by which aerosols cool the Earth's surface by
Page 131
increasing the reflectivity of clouds (Charlson et al., 1992; IPCC, 1995; Ramaswamy et al., 1995). Because of increases in anthropogenic emissions of compounds that lead to the formation of aerosols in the atmosphere (mostly sulfur oxides and the combustion products of biomass burning), atmospheric aerosol loadings have undoubtedly increased since the Industrial Revolution, and this in turn has likely caused a climatic cooling (cf. Charlson et al., 1991). Calculations suggest that this cooling may have been large enough to offset much of the warming from increasing greenhouse gas concentrations over parts of the Northern Hemisphere (see Figure II.2.12; IPCC, 1996). However, the uncertainties in these calculations are significant because the sources, physio-chemical properties, and radiative effects of atmospheric aerosols remain poorly characterized.
Because atmospheric aerosols provide sites for heterogeneous reactions between gases, liquids, and solids, they can have a major impact on the gaseous- as well as the condensed-phase composition of the atmosphere. In the stratosphere, for instance, aerosols in the form of polar stratospheric clouds within the polar vortex, as well as sulfate aerosols from volcanic eruptions, play a pivotal role in the catalytic destruction of ozone by facilitating the conversion of chlorine compounds from relatively unreactive to reactive forms (Solomon et al., 1986; 1993). Similar processes also affect the chemistry of the troposphere. Tropospheric aerosols affect the rate of growth and the properties of clouds. In turn, chemical reactions on the surface and interior of cloud droplets affect the chemistry of the aerosols that result after cloud droplets evaporate. However, large gaps remain in our mechanistic understanding of these processes. As a result, aerosols are currently treated in atmospheric models through relatively crude parameterizations whose application to reliable forecasting of future trends is questionable.
On more local and regional scales, atmospheric aerosols inhibit visibility and can have a deleterious effect on human health (American Thoracic Society, 1996a,b). In fact, there is a growing body of epidemiological data suggesting that aerosols may cause a significant increase in human mortality and morbidity at concentrations significantly below the current National Ambient Air Quality Standard set by the U.S. EPA for particulate matter with diameters less than 10 µm (PM 10). As a result, the EPA has promulgated a new, more stringent NAAQS for fine particles, that is, particulate matter with diameters less than 2.5 µm (PM 2.5). Here again, however, the specific chemical and physical properties of aerosols that give rise to deleterious health effects at such low concentrations remain poorly defined.
The research challenges for atmospheric aerosols are thus to (1) document the chemical and physical properties of atmospheric aerosols to provide more accurate assessments of the effects of aerosols on climate, stratospheric ozone, tropospheric oxidation, human health and welfare, and ecosystem functioning; and (2) elucidate the chemical and physical processes responsible for determining the size, concentration, and chemical characteristics of atmospheric aerosols in order to develop a more robust predictive capability.
Page 132
Effects of Toxics and Nutrients on Biospheric Function
We know far too little about the atmospheric transport and deposition of toxics and nutrients, and their interactions with biota, to quantify their present or future effects on natural ecosystems.
The study of toxics and nutrients strives to (1) quantify the rates at which biologically important trace species are transferred from the atmosphere to terrestrial and marine ecosystems through dry and wet deposition; (2) document the net effect on metabolic function within the biosphere of the multiple stresses and benefits brought on by this deposition; and (3) elucidate the biochemical and biophysical mechanisms by which these effects are brought about. The importance of these processes to the long-term viability of our economically and environmentally important ecosystems is significant. The examples are numerous. There is evidence that forests are being damaged by ozone exposure (LeFohn, 1992), as well as the deposition of a variety of atmospheric pollutants (Godbold et al., 1988; Aber et al., 1989; Schulze, 1989; Van Dijk et al., 1990). Important fisheries, such as the Chesapeake Bay and the Great Lakes, are being decimated by the runoff of pesticides and nutrients from agricultural activities. Acid rain has contributed to the eutrophication of waters that once supported fisheries and recreation.
Although we are beginning to understand the more acute effects of atmospheric toxicity and overfertilization on natural ecosystems, we know far too little about the transport and deposition of toxics and nutrients and their interaction with biota to assess the extent of these problems or to predict new ones that might arise. From this brief discussion, two research challenges emerge: (1) document the rates of chemical exchange between the atmosphere and key ecosystems of economic and environmental import to provide more quantitative estimates of atmospheric chemical impacts on the biosphere and biospheric emission rates to the atmosphere; and (2) elucidate the extent to which interactions between the atmosphere and biosphere are influenced by changing concentrations and deposition of harmful and beneficial compounds to better assess the long-term anthropogenic influences on the coupled atmosphere-biosphere system.
Overarching Research Challenges
Four overarching research challenges emerge from the many disciplinary challenges.
The discussions in the preceding sections have identified a series of disciplinary research challenges aimed at addressing the major scientific issues concerning the Environmentally Important Atmospheric Species. These challenges are summarized in Box II.2.2.
Page 133
|
Box II.2.2 Summary of the Disciplinary Research Challenges for Atmospheric Chemistry* |
|
• Stratospheric Ozone Challenges: Document the concentrations and distribution of stratospheric ozone and the key species that control its catalytic destruction; elucidate the coupling between chemistry, dynamics, and radiation in the stratosphere and upper troposphere. |
|
• Greenhouse Gas Challenges: Elucidate the processes that control the abundances and variabilities of atmospheric CO2, CH4, N2O, and upper-tropospheric and lower-statospheric O3 and water vapor, expand global monitoring networks to include upper-tropospheric and lower-stratospheric O3 and water vapor. |
|
• Photochemical Oxidant Challenges: Develop the observational and computational tools and strategies needed by decision makers to effectively manage ozone pollution; elucidate the processes that control and the interrelations that exist between the ozone precursor species, tropospheric ozone, and the oxidizing capacity of the atmosphere. |
|
• Atmospheric Aerosol Challenges: Document the chemical and physicla properties of atmospheric aerosois; elucidate the chemical and physical processes responsible for determining the size, concentration, and chemical characteristics of atmospheric aerosols. |
|
• Toxics and Nutrients Challengers: Document the rates of chemical exchange between the atmosphere and key ecosystems of economic and environmental import; elucidate the extent to which interactions between the atmosphere and biosphere are influenced by changing concentrations and deposition of harmful and beneficial compounds. |
|
* The disciplinary research challenges focus on the efforts and activities needed to address key scientific issues associated with each of the Environmentally Important Atmospheric Species. |
Inspection of Box II.2.2 indicates that the individual disciplinary challenges can be organized into three Overarching Research Challenges related to documenting the chemistry of the atmosphere, developing predictive capabilities through the elucidation of mechanisms and processes, and supporting environmental management activities. Below, each of these challenges is discussed, along with a fourth related to the interactions between each of the Environmentally Important Atmospheric Species.
Overarching Research Challenge 1: Document the Chemical Climatology of the Atmosphere
The first priority of atmospheric chemistry research must be to establish the present chemical climatology of the atmosphere by documenting the spatial dis-
Page 134
tributions, temporal trends, and variability of the Environmentally Important Atmospheric Species and of the species that affect their formation and removal. This will require a commitment to long-term and careful measurements of atmospheric species at the appropriate spatial and temporal scales. Networks and observing systems have already been implemented to monitor the global distributions of many of the key long-lived species of the atmosphere and of stratospheric ozone. However, systems for monitoring the shorter-lived species, whose distributions tend to be more variable in time and space, are woefully inadequate. Therefore new initiatives will be required to (1) develop reliable and cost-effective instrumentation capable of monitoring the concentrations of these species; and (2) develop and implement monitoring networks with adequate spatial and temporal resolution to document their concentrations and trends. At the same time, resources must be provided to maintain the current monitoring capability for long-lived species.
Overarching Research Challenge 2: Develop Reliable Predictive Tools and Models
The fact that the chemistry of the atmosphere is changing today and that these changes could have significant societal impacts points to a critical need for predictive models that can (1) forecast future trends and responses to new anthropogenic and natural forcings, and (2) provide insight into the probable consequences of remedial actions and policies to reverse or slow undesirable trends. Unfortunately, our predictive capabilities in atmospheric chemistry are presently inadequate for this task because of major gaps in our knowledge of the chemical, physical, and biological processes and mechanisms that control the concentrations of the Environmentally Important Atmospheric Species. In order to develop sufficiently robust predictive models of the chemistry of the atmosphere and adequately evaluate the performance of these models, it is necessary that we commit adequate resources to relevant field studies designed to elucidate process and mechanisms.
Overarching Research Challenge 3: Support and Assess the Efficacy of Environmental Management
The Clean Air Act Amendments of 1990 (CAAA-90) address a variety of environmental issues related to atmospheric chemistry, including urban ozone pollution, acid rain, air toxics, and stratospheric ozone depletion. Although the environmental problems that the CAAA-90 address pose significant economic as well as environmental costs to the nation, management of these problems will also be quite costly. It is estimated, for example, that the incremental costs of full implementation of the CAAA-90 will amount to about $25 billion per year (J. Bachman, personal communication, 1996). Given the importance of the environ-
Page 135
mental problems being addressed by the CAAA-90 and the high costs associated with addressing them, it is imperative that atmospheric chemistry research be structured to support the regulatory activities called for in these amendments (and subsequent environmental legislation). This support should be focused on providing the tools and data required to assess the efficacy of the regulatory actions that have been implemented and to refine and improve these actions.
Overarching Research Challenge 4: Develop a Holistic Research Strategy
Thus far, our discussions have focused on the activities needed to address the uncertainties associated with each of the Environmentally Important Atmospheric Species. However, these species and the processes that cause their appearance, transport, transformation, and ultimate removal from the atmosphere operate within a highly coupled and interactive system of chemical and physical processes. Research programs that attempt to address individual environmental problems by focusing on only a single aspect or component of the system have the potential therefore to produce incomplete or misleading results. It is imperative that atmospheric chemistry research recognizes the complexities inherent in the atmospheric chemical system and undertake research aimed at elucidating and quantifying these interconnections. To accomplish this, atmospheric chemists will have to interact increasingly with scientists focused on related disciplines such as climate, cloud physics, terrestrial ecology, ocean sciences, and meteorology, as well as the social sciences and medicine.
Infrastructural Initiatives
The problems addressed by the field of atmospheric chemistry are critically linked to global economic vitality and environmental health. Seen in this light, the price for new initiatives that develop the infrastructure in atmospheric chemistry research, and thus strengthen its ability to address these problems, is relatively modest.
The scientific and technical issues confronting atmospheric chemistry in the twenty-first century are complex and challenging. However, recent and anticipated advances in instrumentation, aeronautical systems, and computational facilities promise to greatly expand the ability of atmospheric chemists to meet the challenges for atmospheric chemistry research outlined in the previous section. However, the field of atmospheric chemistry cannot be expected to undertake these new challenges and absorb these new technologies without significant investments to strengthen its research capabilities and infrastructure. In this section, six Infrastructural Initiatives in atmospheric chemistry are proposed that
Page 136
together would provide firm undergirding for meeting the research challenges outlined above.
Before turning to a discussion of the Infrastructural Initiatives, it is relevant to note that the resources being made available for research in the United States are increasingly limited, and for this reason, the implementation of any new research initiatives cannot be undertaken lightly. However, the problems confronted by atmospheric chemistry are critical to the continuing economic vitality and environmental health of our nation and to the sustainable development of the world. Seen in this light, the costs of the new initiatives in atmospheric chemistry needed to address these problems are relatively modest.
Infrastructural Initiative 1: Deployment of an Observing System for Moderately Lived Species
The need for long-term measurements of atmospheric species has been a recurring theme in our discussions. To meet this critical need, a new observing system must be designed and deployed that is able to establish the regional and global distributions and temporal trends in moderately lived species of environmental import and the chemical and physical properties of atmospheric aerosols.
As illustrated in Figure II.2.13, the residence or lifetimes of the trace species in the atmosphere are quite variable, ranging from centuries or more for long-lived species to seconds or less for short-lived species. Moreover, as Figure II.2.13 illustrates, the spatial and temporal variabilities of species are inversely related to their lifetimes. Long-lived species tend to be relatively uniformly mixed throughout the troposphere; shorter-lived species exhibit much more variability. Because of the uniformity in the distributions of long-lived species, a network consisting of a few dozen surface sampling sites strategically situated at various latitudes and making concentration measurements on the order of once a day has been adequate to characterize the primary distribution features and temporal trend of the long-lived gases. The situation for the shorter-lived gases, however, is quite different. As species' lifetimes become shorter and their distributions become more temporally and spatially variable, the task of designing a monitoring network becomes more complex. Greater numbers of sampling sites are required to ensure adequate spatial coverage, and increasing sampling frequencies are required to ensure proper measurement of short-term temporal variability. Unfortunately, the sparse nature of present observing networks greatly limits our understanding of the trends and distributions of these shorter-lived species.
The development of a network for moderately lived gases represents a significant challenge that will require instrumentation development and careful network design and implementation. Moreover, because of the important role of upper-tropospheric species such as ozone and water vapor, it is especially critical that the global system be configured to monitor the concentrations of key species in the upper troposphere and lower stratosphere as well as at the surface. This can
Page 137
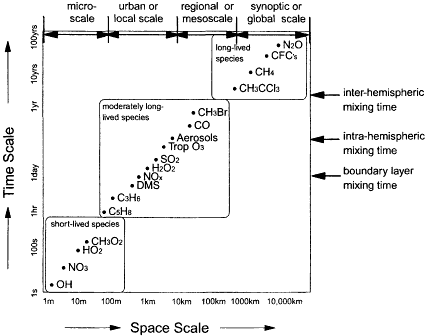
Figure II.2.13
Spatial and temporal scales of variability of a number of key constituents in the
atmosphere. NOTE: C3H6 = propene; C5H8
= isoprene; CH3Br = methyl bromide;
CH3CCl3 = methyl chloroform; CH3O2 = methyl peroxy radical; DMS = dimethyl
sulfide; H2O2 = hydrogen peroxide; NO3
= nitrogen trioxide; OH = hydroxyl radical;
SO2
= sulfur dioxide; Trop = tropospheric.
best be accomplished by taking advantage of existing platforms not yet used by the atmospheric chemistry community while also bringing into operation new observing platforms uniquely capable of collecting data at remote sites over extended periods of time.
Infrastructural Initiative 2: Deployment of Exposure Assessment Networks
Current monitoring networks have largely been designed to characterize the chemical climatology of the atmosphere or to monitor compliance with specific environmental regulations; they are not well suited to examining interactions between the biosphere and the atmosphere or to characterizing the exposure of ecosystems to toxics and nutrients. A new infrastructural initiative that is specifically aimed at assessing exposures of targeted populations and biomes through the deployment of exposure assessment networks is therefore critically needed.
Page 138
Infrastructural Initiative 3: Development of Surface Exchange Measurement Systems
Surface exchange is a fundamental component of the cycling of elements through the atmosphere. Not only does it help determine the concentrations of atmospheric species by controlling their rates of emission into and removal from the atmosphere, it also characterizes the interactions between the biosphere and ocean and the atmosphere. Thus, the accurate characterization and elucidation of surface exchange rates is highly relevant to Overarching Research Challenges 2 and 4, discussed earlier. Unfortunately, the rates of surface exchange and the mechanisms that control these processes are not well understood; as a result, these processes are treated in a highly parameterized fashion in atmospheric models. To aid in the development of predictive models and to better understand biospheric-atmospheric interactions, a new initiative is needed that focuses on the development and evaluation of measurement platforms designed to quantify the surface emission and deposition rates of Environmentally Important Atmospheric Species at a wide variety of spatial scales.
Infrastructural Initiative 4: Demonstration of an Operational Chemical Meteorology System
The standard approach in atmospheric chemistry research generally means that months or even years go by between initial observations and the analysis of these observations and model simulations. Such an approach slows the development and evaluation of predictive models and limits the utility of models to the policy-making community. By contrast the meteorological community has found that the use of real time data to continuously generate short-term and long-term predictions using three- and four-dimensional data assimilation modes has greatly aided scientific understanding and the development and evaluation of modeling systems, while also increasing the relevancy of the science to society and decision makers. The atmospheric chemistry discipline has now reached a level of maturity where the adoption of this so-called meteorological approach would be feasible. It is therefore proposed that a project be initiated to demonstrate the concept of an operational "chemical meteorology" system, involving the daily prediction of selected air quality parameters (e.g., oxidant and acid aerosol concentrations, visibility, ozone column densities) on local and regional scales through the real-time integration of meteorological and chemical observing systems, data analysis schemes, and statistical and/or mechanistic predictive air quality and atmospheric chemical transport models. Such an initiative would support efforts to meet Overarching Research Challenges 2 and 3, discussed earlier.
Page 139
Infrastructural Initiative 5: Instrument Development and Technology Transfer Program
One of the significant successes of the atmospheric chemistry discipline in recent decades has been the application of modern chemical analytical techniques to the measurement of atmospheric trace species under field conditions with research-grade instrumentation. However, the evaluation of these instrumentations, the development of standards and protocols for them, their dissemination to the general research community, and their ultimate transfer to the private and regulatory sectors have proved more problematic. To facilitate this process and thereby aid in the development of global sampling systems and data bases (Overarching Research Challenges 1 and 2) and in the support of environmental management activities (Overarching Research Challenge 3), it is proposed that a program be initiated to evaluate new instrumentation and aid in its transfer to the scientific, regulatory, and private-sector communities through the development of suitable standards and protocols.
Infrastructural Initiative 6: Condensed-Phase and Heterogeneous Chemical Process Laboratories
The atmosphere is an environment of extreme and varying temperatures, pressures, humidities, and fluxes of solar radiation. These conditions give rise to a myriad of gas-phase, condensed-phase, and heterogeneous chemical processes that ultimately determine the current and future composition of Earth's atmosphere. Historically, characterization of these chemical processes has depended critically on state-of-the-science laboratory measurements. Because the study of homogeneous gas-phase reactions is a relatively mature field with well-defined methodologies, the gas-phase chemistry of many important atmospheric species is reasonably well understood. In contrast, the study of heterogeneous reactions and the methodologies needed to study these processes are in their infancy. As a result, very little is known about the surfaces involved in atmospheric heterogeneous reactions or about how reactions on these surfaces occur. The growing appreciation of the importance of condensed-phase and heterogeneous reactions in the stratosphere and troposphere, along with the realization of the significant climatic and health impacts of atmospheric particulate matter, indicates that we must improve our basic knowledge of multiphase chemical processes. Toward this end, we propose an infrastructural initiative aimed at developing and maintaining laboratory facilities that focus on elucidating fundamental condensed-phase and heterogeneous chemistry.
Page 140
Conclusion
A carefully designed research strategy will yield significant advances in our understanding of atmospheric chemistry and our ability to manage many of the important environmental problems that confront society.
The challenges facing the atmospheric chemistry community for the next century are imposing. The array of chemical species that must be measured and ultimately understood is large. The concentrations at which these species require quantification often tax, and in some cases exceed, modern analytical chemical techniques. The fact that many of these measurements must be made outside the laboratory, in an uncontrolled environment, makes the task all the more difficult. Equally challenging is the variety of spatial and temporal scales at which the problems must be addressed (see, for example, Figure II.2.13). To make progress in understanding the most pressing and scientifically important issues in atmospheric chemistry, while also providing timely and relevant information for decision makers and regulators, we have proposed a matrix of four Overarching Disciplinary Challenges and six Infrastructural Initiatives. These are summarized in Table II.2.2.
Although the scientific and technical issues of the twenty-first century are complex and challenging, new technological advances in instrumentation, aeronautical systems, and computational facilities promise to greatly expand our ability to address these issues comprehensively. By capitalizing on these advances and working closely with scientists from related disciplines, a carefully designed research strategy should yield significant advances in our understanding of atmospheric chemistry and our ability to manage many of the important environmental problems that confront society.
The Environmentally Important
Atmospheric Species:
Scientific Questions and Research Strategies
Stratospheric Ozone
In the past couple of decades, column ozone abundance has decreased substantially over major portions of the globe, and the scientific evidence overwhelmingly points to anthropogenically produced chlorofluorocarbons and other halogenated compounds as the main cause of this ozone loss (WMO, 1995). The consequences of this reduction are serious; hence, steps have been taken by the international community to reverse the loss. As the stratosphere slowly recovers to what will hopefully be conditions similar to those of the pre-CFC era, the atmospheric chemistry community must continue to monitor this change and improve its basic understanding of the stratosphere.
In formulating a strategy for studying the stratosphere, we have identified
Page 141
|
TABLE II.2.2 Overarching Disciplinary Challenges and Infrastructural Initiatives |
||||
|
Overarching Disciplinary Challenge |
||||
|
Infrastructural Initiative |
1. Chemical Climatology |
2. Development of Predictive Models |
3. Support of Environmental Management |
4. Holistic Research Approach |
|
1. Observing system for moderately lived species |
X |
X |
X |
X |
|
2. Exposure assessment networks |
X |
X |
X |
|
|
3. Surface exchange measurement systems |
X |
X |
X |
|
|
4. Operational chemical meteorology |
X |
X |
||
|
5. Instrumentation and technology transfer |
X |
X |
X |
|
|
6. Condensed-phase and heterogeneous chemistry |
X |
X |
X |
X |
Page 142
five basic scientific questions that we believe will motivate research on stratospheric ozone in the coming decades:
1. Will the evolution of the Antarctic stratospheric ozone hole proceed as expected with a short period (˜5 years) of continued increasing intensity, followed by a much longer period (˜50 years) of recovery to normal conditions?
2. What will be the evolution of midlatitude ozone depletion, and can we develop models to simulate this evolution correctly?
3. What is the role of the tropical region of the stratosphere in global ozone change?
4. What are the interactions between stratospheric ozone depletion and climate change?
5. What are the consequences of current and future perturbations, such as emissions from aircraft and volcanic eruptions, for stratospheric ozone concentrations?
The essential research activities that will be required to address these questions are outlined in Box II.2.3.
Monitoring the Distribution of Stratospheric Ozone
The centerpiece of the strategy for research on stratospheric ozone depletion must be uninterrupted observations of the total concentration and vertical distribution of ozone made with high temporal resolution and accuracy using a combination of intercalibrated instruments on space-based and ground-based platforms as well as small balloon sondes. The critical nature of this research need cannot be overemphasized in light of the recent decline in our capabilities to monitor stratospheric ozone distributions from space.
At the very least, continuous measurements of stratospheric ozone will document the extent of the ozone loss and its expected recovery as stratospheric chlorine concentrations decrease. This will provide a critical gauge of the sufficiency of (or compliance with) international treaties devised to halt ozone depletions.
Perhaps even more importantly, continuous measurements of stratospheric ozone will provide rapid warning of any unanticipated trends in stratospheric ozone. The relationships between stratospheric ozone, anthropogenic activities, and various naturally occurring agents of chemical change are very complex and, as yet, not fully understood. As the stratospheric chemical composition evolves under changing chlorine and bromine loadings, the effects of perturbations—both natural and anthropogenic—may vary in unexpected ways. For example, we now know that volcanic eruptions into a chlorine-rich stratosphere can have a profoundly different effect on stratospheric ozone than they most likely had in the pre-CFC era (Brasseur and Granier, 1992; Solomon et al, 1993). Similar phe-
Page 143
|
Box II.2.3 Recommended Research Tasks for Stratospheric Ozone Research |
|
|
1. Monitor stratosphenic ozone to |
|
|
• observe unanticipated changes, |
|
|
• confirm that remediation policies are effective, and |
|
|
• provide short-term UV-B radiation forecasts. |
|
|
2. Examine the distribution of radical species to |
|
|
• map the loss rate of ozone, and |
|
|
• refine our ability to predict ozone loss. |
|
|
3. Map the distribution of tracer species to |
|
|
• identify mechanisms for tropospheric and stratospheric transport mechanisms, and |
|
|
• refine our ability to predict winds and temperatures. |
|
|
4. Characterize critical chemical processes to |
|
|
• provide data for chemical models, |
|
|
• predict chemical reactivities, and |
|
|
• examine the effects of heterogeneous reactions. |
|
nomena may very likely apply to anthropogenic perturbations such as those arising from certain aircraft emissions (see for example, Bekki and Pyle, 1993).
Extending Stratospheric Measurements of Critical Gas- and Condensed-Phase Species
Although monitoring the distribution of stratospheric ozone will be the central activity for stratospheric ozone research in the coming decades, the need to understand the evolving nature of stratospheric chemistry in the face of changing trace gas concentrations gives rise to an additional important research activity: mapping the distribution and variability of the species that determine the magnitude of ozone depletion and characterizing the complex of chemical reactions among these species. The total rate of stratospheric ozone destruction is governed by several catalytic loss processes whose individual rates are limited by the abundances of specific free-radical species such as hydroxyl radical (OH), hydroperoxyl radical (HO2), chlorine monoxide (ClO), bromine monoxide (BrO), nitric oxide (NO), and nitrogen dioxide (NO2), as well as atomic oxygen. A fundamental scientific question central to our understanding of the chemistry of ozone depletion is, What rate-limiting steps actually dominate ozone loss as a function of altitude, latitude, and season? Of equal importance, How do the rates and relative roles of each of these loss processes change as hydrogen-, chlorine-, bromine-, and nitrogen-containing species are added to or removed from the
Page 144
Figure II.2.14
Schematic illustrations of ozone loss rate as a function of NOx concentration.
Because of the coupling that exists between radical families, the response of the
total ozone loss rate to changes in NOx is nonlinear. For example, at the low NOx
concentrations observed during the Stratospheric Photochemistry, Aerosols and
Dynamics Expedition (SPADE), the ozone loss rate was found to be inversely
correlated with NOx. Source: Wennberg et al., 1994. Reprinted with permission
of the American Association for the Advancement of Science.
stratosphere and as coevolving changes in temperature, aerosol surface area, water vapor, and so forth, occur?
Clearly, answering these questions requires mapping the relevant radical concentrations in the upper troposphere and the stratosphere. However, to be able to define the full spectra of stratospheric ozone responses to the myriad of current and possible future perturbations of the stratosphere (e.g., volcanic injections of sulfur; aerosols, nitrogen oxides, and water vapor from subsonic and supersonic aircraft; emissions of bromine and chlorine compounds; temperature changes resulting from ozone depletion and greenhouse gas forcings), it is essential that we map the specific rates of change of rate-limiting reactions with respect to the variables on which the concentrations depend, as well as the radical concentrations themselves. Figure II.2.14 schematically illustrates the diagnostic power of this dual approach. In this case, the loss rate of ozone from the catalytic cycles driven by HOx, halogen, and NOx radicals is illustrated as a function of increasing NOx concentrations, with the total ozone loss given by the sum of the individual contributions. Because of the couplings that exist between NOx and other radical families, the response of the ozone loss to a change in NOx concentrations turns out to be a complex function that depends on the individual gradients in
Page 145
each of the rate-limiting steps with respect to NOx. Because of the nature of these gradients, the total ozone loss exhibits a minimum at an intermediate NOx concentration, as shown in the figure. Similar effects can be expected to determine the dependence of ozone destruction cycles on other key radical species.
Since the relationships between critical chemical species that control ozone loss change over various spatial and temporal scales, it is important to extend measurements of these critical species to the full range of relevant conditions. This will require the development of new instruments and new measurement platforms capable of covering the relevant regions of the stratosphere, with an emphasis on measurements that characterize the variations of each of the rate-limiting radicals and its catalytic ozone loss rate with respect to each of the other critical species.
Elucidating the Coupling Between Chemistry, Dynamics, and Radiation
The stratosphere is a coupled photochemical, dynamical, and radiative system in which ozone is exported from the high-altitude tropics to mid- and high latitudes along surfaces defined by constant mixing ratios of tracers such as N2O and CH4. The coherence of these tracer surfaces as a vertical coordinate is revealed by the tight relations between them and other reactive species; this dramatic feature has led to important insights into the dynamics of the stratosphere and the response of stratospheric ozone to chemical and physical perturbations. Air enters the stratosphere primarily in the cold inner tropics through a process that desiccates the air and confines the upwelling flow to the middle stratosphere, as shown in Figure II.2.15.
This exchange of air between the upward-moving tropical air mass and the stratospheric midlatitudes apparently occurs on time scales of a few months and varies with season. However, this exchange is largely uncharacterized and is a potential source of uncertainty in our ability to predict the response of the stratosphere to perturbations.
Polar regimes are characterized by rapid cooling in the fall, with subsidence of many kilometers associated with the establishment of a strong polar jet. This in turn restricts mixing with the midlatitudes, thus tending to isolate the polar stratosphere from the rest of the stratosphere during the winter months, especially in the Southern Hemisphere. This isolation of the wintertime polar stratosphere plays a significant role in the annual, dynamical cycle of the stratosphere and is an essential element in the formation of the Antarctic ozone hole.
To quantify the mechanisms for polar and midlatitude ozone loss, we must understand the patterns of air exchange between the stratosphere and troposphere, and between the tropical, extratropical, and polar stratosphere. This gives rise to another critical research activity that endeavors to elucidate the coupling between chemistry, dynamics, and radiation in the stratosphere. Because of the tight relationships between air mass origin and tracer concentrations, this can best be
Page 146
Figure II.2.15
Dynamical aspects of stratospheric-tropospheric exchange and intrastratospheric
transport. Tropopause is shown by the thick line. Thin lines are isentropic (constant
potential temperature) surfaces labeled in kelvin. Heavily shaded region is the lowermost
stratosphere, in which isentropic surfaces span the tropopause and exchange occurs by
tropopause folding. The region above the 380 K isentropic surface lies entirely in the
stratosphere. Light shading area denotes wave-induced forcing (the extratropical ''pump'').
Wavy double-headed horizontal arrows denote meridional transport by eddy motions,
which include tropical upper-tropospheric troughs and their cutoff cyclones, as well as
their midlatitude counterparts. Broad vertical arrows show transport by global-scale
circulation, which consists of tropical upwelling and extratropical downwelling. This large-
scale circulation is the primary contribution to exchange across the isentropic surfaces that
lie entirely in the stratosphere. Source: Holton et al., 1995. Reprinted with permission of the
American Geophysical Union.
accomplished with spatially resolved (0.1 km), highly accurate, in situ observations of the phase relationships between CO2 and H2O; measurements of the seasonal changes in the relationships between N2O, CH4, SF6 (sulfur hexafluoride), CFC-11, CFC-12, O3, and NOy; and determination of the age of the air mass in which the measurements are made. Such measurements can be carried out using ground-based instruments, piloted-reconnaissance and robotic aircraft, and small satellites because of the short time and low cost with which they can be
Page 147
deployed. Trajectory and three-dimensional models are required to interpret these observations.
Because of the sharp concentration gradients, the region from the upper troposphere to 30 km must rely heavily on in situ observations. At higher altitudes where vertical and horizontal gradients begin to soften, small satellites will be very useful. The altitude overlap of these two observational strategies must be enhanced, because it is essential to obtain these coupled observation sets with seasonal (about one-month) frequency for a number of annual cycles.
Quantification and Characterization of Critical Gas-Phase and Heterogeneous Mechanisms
Predicting future changes in stratospheric ozone concentrations will require a more thorough understanding of the fundamental processes, both chemical and physical, that govern the balances between formation, transport, and destruction of ozone in the stratosphere. The chemical transformations that take place in the stratosphere either in the gas phase or on condensed matter ultimately determine the composition of this region. The quantification and characterization of these processes through laboratory experiments (and in some cases computational techniques) provide the essential building blocks for interpreting, simulating, and predicting stratospheric processes. Continued development and application of laboratory experiments and computational techniques to the study of the relevant chemical processes of the stratosphere is thus an important component of any research strategy. Especially critical will be the investigation of heterogeneous processes, and of the chemical and physical processes of particle nucleation and growth, where our understanding lags far behind that of gas-phase homogeneous reactions.
Atmospheric Greenhouse Gases
Greenhouse gases absorb infrared radiation from the Earth's surface and reradiate a portion back to the surface. In so doing, they act as thermal insulators, warming the Earth's surface through the so-called greenhouse effect. Predicting future climate changes in response to secular trends in greenhouse gases requires not only an accurate knowledge of how the major greenhouse gases evolve in time, but also an understanding of the processes that control the production and removal rates of these gases in the atmosphere. These gases include the primary greenhouse gases emitted directly into the atmosphere (e.g., CO2, CH4, N2O, CFCs) and the secondary greenhouse gases produced within the atmosphere by photochemical processes (i.e., O3). Water vapor, the most important of the greenhouse gases, is both emitted into the atmosphere (via evapotranspiration) and produced photochemically in the stratosphere by the oxidation of CH4. Ozone is unique in that it not only acts as a greenhouse gas (by absorbing infrared radiation) but also is an important absorber of solar radiation.
Page 148
The observed increases in the concentrations of CO2, CH4, N2O, and CFCs (see Figure II.2.2) provide one of the clearest manifestations of global change in the atmosphere. Historic trends in H2O and O3 have yet to be quantitatively characterized. However, limited data suggest that background, surface-level ozone concentrations may have increased by a factor of two or more in this century (see Figure II.2.10), whereas stratospheric ozone concentrations have decreased over the past 20 years (see Figure II.2.2). In addition there are preliminary indications that stratospheric H2O concentrations are currently on the rise.
For the most part, the changing concentrations of atmospheric greenhouse gases appear to be driven by human activities such as energy use, industrialization, land use change, and agriculture (IPCC, 1995). The fact that these anthropogenic drivers of global change are not likely to abate in the coming decades without significant political and economic intervention has given the scientific debate over greenhouse gases and their climatic impact a sense of urgency. Moreover, although significant advances in our understanding of the sources and sinks of greenhouse gases have been attained in recent years, we do not as yet have a reliable predictive capability for evolution of their concentrations in the future. In the absence of such a predictive capability, our ability to predict climatic trends—and hence to formulate effective long-term policy responses—remains limited.
In this section, we outline a basic research strategy for studying atmospheric greenhouse gases. This strategy is motivated by one central question:
What will be the trends in concentrations of greenhouse gases during the twenty-first century?
From this motivating question, we have identified several critical scientific questions for investigation:
1. How do the natural carbon and nitrogen cycles control the amounts of CO2, CH4, and N2O in the atmosphere. How are these cycles perturbed by human activities? More specifically:
• What are the regional sources and sinks of CO2 other than fossil fuel burning?
• How large are the individual CH4 and N2O sources?
• How might future climate changes affect these sources?
• What causes year-to-year changes in the trends of greenhouse gases?
2. Are the Montreal Protocol and its successor agreements effective in mitigating the climatic warming from CFCs and hydrochlofluorocarbons (HCFCs)? Which new halogenated compounds may affect climate in the future?
3. What are the trends in O3 in the troposphere and stratosphere, and what are their causes?
Page 149
|
Box II.2.4 Recommended Research Tasks for Atmospheric Greenhouse Gases |
|
|
These must address the following: |
|
|
1. The primary greenhouse gases emitted directly into the atmosphere, which include |
|
|
• the biogenic greenhouse gases (CO2, CH4 N2O) that are closely coupled to ecosystem and biospheric processes, and |
|
|
• the halogenated greenhouse gases (CFCs, HCFCs, SF6) whose sources are dominated by anthropogenic activities. |
|
|
2. Ozone (O3): this compound is a secondary greenhouse gas produced in the atmosphere by photochemical processes. |
|
|
3. Water vapor (H2O): this compound is emitted into the atmosphere at the Earth's surface by evapotranspiration, but is also produced photochemically in the stratosphere as a result of the photochemical oxidation of CH4. |
|
4. What are the trends in water vapor in the upper troposphere and lower stratosphere, and what are the reasons for these trends?
The essential research activities that will be required to address these questions are outlined in Box II.2.4 and below. These activities are organized into three categories: (1) research related to the primary greenhouse gases that are emitted directly into the atmosphere; (2) research related to ozone, whose source is entirely photochemical; and (3) research activities related to H2O, whose sources are both surface emissions and photochemical. In general, these research activities focus first on enhancements of key strategies currently under way and then progress to new strategies that will require technology development and, in many cases, the investment of new resources. It should be noted that many of the research activities described here are relevant to other key issues in atmospheric chemistry highlighted in this Disciplinary Assessment. For example, investigations of the distributions and surface exchange rates of N2O, CH4, and the halogenated compounds, as well as O3, are clearly relevant to the study of stratospheric ozone and photochemical oxidants. Conversely, the research activities described under the other key atmospheric chemistry issues discussed in this Disciplinary Assessment are relevant to understanding the greenhouse gases discussed here. For example, phytotoxics and nutrients can strongly influence the structure and functioning of ecosystems, changes in which can significantly influence the balance of CO2, N2O, O3, and H2O in the atmosphere.
Page 150
Primary Greenhouse Gases
The primary greenhouse gases can be divided roughly into two categories: (1) the greenhouse gases whose sources and sinks are closely linked to biospheric processes (i.e., CO2, CH4, and N2O), and (2) halogenated compounds (e.g., CFCs, HCFC, SF6) that are entirely anthropogenically produced (NRC, 1993; IPCC, 1995). However, some halogenated compounds such as CH3Cl (methyl chloride) and CH3Br (methyl bromide) have important interactions with the biosphere and thus fall into both of the categories listed above. Recommended research tasks are listed in Box II.2.5 and discussed below.
The most robust large-scale signature of sources and sinks of the greenhouse gases and their time dependence will be variations in the mixing ratios of CO2, CH4, N2O; in their isotopic ratios; and in O2, which aids in distinguishing between terrestrial and
|
Box II.2.5 Recommended Research Tasks for Primary Greenhouse Gases |
|
|
1. Maintain current monitoring networks to |
|
|
• document trends and |
|
|
• elucidate source-sink signatures. |
|
|
2. Monitor vertical profiles to elucidate mesoscale variations in sources and sinks. |
|
|
3. Conduct flux measurements to |
|
|
• elucidate biological mechanisms, and |
|
|
• establish relationships between climatic variations and surface exchange rates. |
|
|
4. Conduct large-scale gas exchange studies to establish methodology for extrapolating small-scale flux information to the meso- and global scales. |
|
|
5. Monitor gas concentrations near source regions to |
|
|
• better characterize regional-scale source strengths, and |
|
|
• develop methodology for monitoring compliance with international emission agreements. |
|
|
6. Improve ocean flux measurements to |
|
|
• better characterize air-sea exchnage rates, and |
|
|
• provide tighter constraints on atmospheric budgets and process models. |
|
|
7. Devise new measurement systems for space-based, robotic aircraft and other innovative platforms capable of extended duty times on station to |
|
|
• provide global flux information, |
|
|
• provide long-term data from remote and inaccessible regions, and |
|
|
• provide technology for verifying compliance with international emissions agreements. |
|
Page 151
marine sinks of CO2. It is therefore essential that the current global networks for monitoring these parameters be maintained. For the data to be useful, high accuracy and precision will be required because the pertinent geochemical information is derived from small spatial and temporal variations.
Predicting the future concentrations of greenhouse gas requires, in part, an understanding of how the exchange rates of these gases with the biosphere behave as a function of season, soil moisture, and so forth, and of how these exchange rates will respond to local, regional, and global climate change. The effects of regional-scale climate variations on greenhouse gas emissions are not adequately captured by the present surface network. In addition, measurements from land surface sites are often difficult to interpret because of the effects of local sources and sinks. In contrast, vertical profiles over the continents can help us to infer important flux information for the biogenic greenhouse gases on the scale of 2,000 to 3,000 km. A key advantage of profiles is that they are less sensitive to details of vertical mixing. The sampling should be carried out throughout the year and should be of sufficient spatial density. The appropriate spatial scale for such measurements is suggested by the current distribution of ecosystems and the spatial extent of major climate "anomalies" such as the drought in the United States in 1988. For the North American continent, this would probably require several dozen sites. Consideration of the expected signal-to-noise ratio suggests that a feasible and economical strategy for the near future would be the collection of automated flask air samples aboard light aircraft at twice-weekly intervals (Tans et al., 1996). The advantage of such a system is that samples could be analyzed for multiple species and for isotopic ratios with tightly controlled calibration. Seasonal, and perhaps monthly, mass fluxes over the continent could be determined from such a system; these could be related to variations in climate and analyzed in the context of three-dimensional climate and chemical transport models. In addition, they would provide a large-scale analogue of the smaller-scale flux measurements described below. Larger-scale studies are also required as described below to extrapolate such subcontinental-scale flux studies to larger hemispheric and global scales.
Predicting future greenhouse gas concentrations requires understanding how the surface exchange rates of these gases will respond to local, regional, and global climate variations. For the biogenic greenhouse gases, this will require surface flux measurements, in concert with hydrological and climatic observations, over a variety of biomes and climate regimes for a multiyear period (see, for example, Baldocchi et al., 1996). These measurements will prove critical to the establishment of empirical relationships between climatic conditions and biospheric emis-
Page 152
sion and uptake rates, as well as the biological mechanisms that are responsible for these relationships.
Although multiyear flux measurements provide insight into the mechanisms responsible for gas exchange on scales of a few to perhaps hundreds of meters, larger-scale studies are needed to establish the methodology and the validity of extrapolating this flux information to regional and global scales. These studies, such as the one planned for Brazil in 1998, represent a natural progression from the mesoscale studies carried out in the 1980s and early 1990s and will require ground experiments, aircraft campaigns, and significant meteorology and transport modeling efforts. These experiments would be greatly enhanced by the availability of advanced flux measurement technology, drone aircraft, and improved instrumentation.
In principle, estimates of the continental source of a trace gas can be obtained from measurements at a surface site that is subject to intermittent pollution events from a nearby continent (Prather, 1985, 1988). For example, a pollution event measured on the west coast of Ireland is readily identified from trace gas concentration data; it appears as a temporary enhancement in the local concentrations of long-lived gases of anthropogenic origin. Prather was able to derive emission ratios of various halocarbons and N2O to CFCl3 (trichlorofluormethane) by measuring the covariance in their atmospheric concentrations. If the magnitude of emissions of a reference gas is known and the sources are co-located, absolute source strengths of the other gases can be derived. Ultimately, with a three-dimensional model that can reproduce synoptic-scale pollution events, it should be possible to infer the continental source strength of a gas without scaling to a reference gas (Prather et al., 1987).
Thus, the implementation of a monitoring network to document changing greenhouse gas concentrations downwind of major source regions would provide critical information about the magnitude of greenhouse gas emissions from these regions. Such information would prove highly valuable for constraining atmospheric budgets for these gases and could ultimately represent a means of verifying compliance with any future international emissions agreements. However, the problem of defining the source strength for biogenic greenhouse gases using this method is somewhat more challenging than for the CFCs whose emissions are essentially constant through the year. Because the sources of biogenic gases vary with season, their background concentrations also vary seasonally. Thus, continuous measurements at both upwind and downwind sites will be required. The challenge will be to develop algorithms that can reliably unravel this information in a quantitative manner, and to define the background concentration and the excess over background caused by the advection of pollutants from a given source region. An atmospheric transport model based on observed winds would play a key role in analysis of the data.
Page 153
The oceanic flux of biogenic greenhouse gases represents an important component of the atmospheric global budgets for these gases; thus, we must be able to adequately characterize the process of air-sea gas exchange. Unfortunately, at present most air-sea exchange rates are not known to better than a factor of two and in some cases an order of magnitude. Direct measurements of fluxes over the oceans have not been successful in reducing this uncertainty because of shortcomings in current platforms and instrumentation. Conventional eddy correlation methods can in principle provide data of sufficient accuracy and precision, but they require fast-response and highly precise chemical detectors. For the long-lived trace gases, the concentration gradients between atmosphere and ocean that must be measured are a very small fraction of the total concentration; as result, these differences are often masked by much larger effects caused by the fluxes of water vapor and heat.
With recent technological advances, a variant of the eddy correlation method (the conditional sampling method, which accumulates samples from the upward-moving eddies in one container and samples from the downward eddies in another) is becoming a viable and promising approach (Businger and Oncley, 1990). After careful conditioning, the difference between the two containers can then be measured with conventional slow-response instruments. Application of this atmospheric measurement over the oceans would enable us to determine some of the factors driving the kinetics of air-sea exchange, whereas for other factors, time-resolved full eddy correlation measurements are required.
Because of their global coverage, satellites represent ideal platforms for measuring concentrations of greenhouse gases on the meso- and global scales. Global data bases gathered from such platforms would undoubtedly revolutionize our grasp of the biogeochemical cycles of greenhouse gases if they were carefully linked to direct observations of flux using strategically deployed ground-based and in situ observations. To serve public policy needs, this approach may ultimately provide an important means for verifying compliance with some forms of international agreements to manage greenhouse gas fluxes. Requirements for such systems, in terms of both accuracy (0.1 percent or better) and spatial resolution (0.5° or better with some vertically resolved information) are beyond the currently available technology. High-precision remote sensing devices thus have to be developed and tested with in situ measurements.
Pilotless or robotic aircraft represent another emerging technology that could transform the study of greenhouse gases and constitute an important component in any systematic, global approach to this problem. Such aircraft could provide a platform for making near-continuous flux measurements over remote and/or inaccessible areas of the globe, such as the oceans, tropical forests, tundra, and icepack. To take advantage of this platform, however, lightweight instrumenta-
Page 154
tion with fast response times and high accuracy must be developed. This instrumentation could be designed for either in situ measurements at low altitude within the boundary layer or remote sensing measurements from high altitude.
The need for model development to address the long-term variability of the biogenic greenhouse gases is severalfold. Improved global biological process models are critically needed in order to extrapolate from small-scale intensive studies to larger temporal and spatial scales. The quality of such models is presently limited by the short duration of the field measurements on which they are based. Once based on longer-term field studies such as those described above, these global models can then be driven by parameters such as temperature and moisture and in some cases by satellite observations of vegetation.
Improved atmospheric chemical transport models are needed to better elucidate the effects of changing surface sources and sinks on atmospheric concentrations. Although atmospheric models require improvement on all scales, particular attention must be paid to subgrid-scale transport processes, such as turbulent mixing of the atmospheric boundary layer, and both shallow and deep convective transport. At present, most models do not incorporate the effects of ecosystems on atmospheric dynamics (e.g., through evapotranspiration). Transport models that use assimilated winds also have to be improved and used in tropospheric applications to trace gas budgets, to incorporate the effects of interannual variability in transport; such models will be essential for regional flux studies. Ocean models are needed for the integration of sparse oceanic data, to attain an understanding of the biogeochemical cycles in the oceans and to provide regional estimates of the trace gas exchanges between the oceans and atmosphere. Ultimately, coupled biospheric, oceanic, and atmospheric models that allow for the proper feedbacks must be developed to reach the goal of predicting trends in biogenic greenhouse gases.
Ozone
In addition to its critical role as an absorber of ultraviolet radiation in the stratosphere and in the oxidant chemistry of the troposphere, ozone's infrared absorption (at 9.6 µm) makes it an effective greenhouse gas, especially in the upper troposphere and lower stratosphere (Lacis et al., 1990). Thus, in addition to its role as a greenhouse gas, we have to document the trends in ozone in the upper troposphere and lower stratosphere and elucidate the reasons for these trends. The research activities necessary to address the causes of the ozone trends are outlined later. Below and in Box II.2.6, we concentrate on the research activities designed to elucidate the ozone trend itself.
Page 155
|
Box II.2.6 Recommended Research Tasks for Ozone as a Greenhouse Gas |
|
1. Expand ground-based vertical ozonesonde program to document trends in the upper troposphere and lower stratosphere with high vertical resolution. |
|
2. Maintain and expand space-based, remotely sensed ozone observations to document global-scale trends in the upper troposphere and lower stratosphere. |
In situ measurements of the vertical distribution of O3 are critical to characterizing its long-term trend in the upper troposphere and lower stratosphere; these measurements provide high vertical resolution and offer an independent check on remote sensing data. Although an international ozonesonde program currently exists, the present collection of ozonesonde stations does not provide a coherent or an adequate program (see, for example, Logan, 1994). The stations do not use the same techniques, they do not all maintain adequate calibration programs, and the frequency of measurements is too low at several stations. Some are located in sufficiently polluted locations that the quality of the tropospheric data may be compromised. Most importantly, the number of sites maintained under the current program is simply too few to provide a reliable global picture. A large number of measurement sites is particularly critical for characterizing upper-tropospheric O3 because of its relatively short lifetime and heterogeneous distribution.
Thus, to bring the current ozonesonde network to an adequate level, many of the existing sonde sites will have to be upgraded. In addition, new sites are desirable, primarily over the tropical continents and oceans. It should be noted in this regard that the planned Network for Detection of Stratospheric Change (NDSC) will not be adequate for the detection of trends in the troposphere, because the measurements are biased toward obtaining stratospheric data and the number of sites is small (about five). The use of pilotless aircraft may provide an efficient solution for this problem.
Although sonde measurements provide data with high vertical resolution, space-based measurements provide true global coverage. For this reason, maintenance and enhancement of satellite measurements of the ozone vertical profile are also critical. Measurements from SAGE II (Stratospheric Aerosol and Gas Experiment II)
Page 156
have provided valuable information on the trends in ozone above 17 km since 1984, with the exception of the period after the Mt. Pinatubo eruption in June 1991, when data could not be obtained in the lower stratosphere (McCormick et al., 1992). Launch of a new instrument while SAGE II is operational would allow for overlap and lead to more reliable trends in the future. Gaps in measurements of ozone have been a serious problem.
Unfortunately, although it is extremely useful for inferring ozone trends in the stratosphere, SAGE II does not yield data on ozone in the upper troposphere. Analysis of the combined data from the Total Ozone Mapping Spectrometer (TOMS) and SAGE II has proven useful for inferring tropospheric ozone column concentrations (Fishman et al., 1990, 1991), but the lack of vertical resolution and gaps in the data record make this technique of limited use for tracking upper-tropospheric O3 trends. Non-U.S. satellites may help fill this void to some degree. On the other hand, new remote sensing techniques have been developed that could form the basis for space-based measurements of tropospheric O3 with 2 to 3 km resolution. Deployment of this instrumentation should be a high priority for the near future.
Water Vapor
As in the case of ozone, elucidating the role of changes in water vapor concentration in an enhanced greenhouse effect most critically requires characterization of the species' trend in the upper troposphere and lower stratosphere (IPCC, 1995). Strategies for accomplishing this are described below and in Box II.2.7.
As for O3, in situ measurements (using sondes) offer the advantage of high vertical resolution and provide a means to validate satellite data. Measurements over Boulder,
|
Box II.2.7 Recommended Research Tasks for H2O |
|
|
1. Implement lower-stratospheric sonde program to document trends in the lower stratosphere with high vertical resolution. |
|
|
2. Maintain space-based observations to document long-term, global-scale trends in the lower stratosphere. |
|
|
3. Develop and implement upper-tropospheric measurement techniques to |
|
|
• close the upper-tropospheric gap in the atmospheric data base for H2O, and |
|
|
• document long-term, global-scale trends in the upper troposphere. |
|
Page 157
Colorado (Oltmans and Hofmann, 1995) indicate that a carefully executed monitoring program, using relatively simple, inexpensive instrumentation should be capable of documenting the probable changes in the concentration of stratospheric H2O that may occur in the twenty-first century. Such measurements at about five global locations, such as NDSC sites (i.e., polar and midlatitudes in each hemisphere and in the tropics), would be adequate, provided these measurements are complemented by global-scale data from satellites.
Instruments such as SAGE II (McCormick et al., 1992), and the Halogen Occultation Experiment and the Microwave Limb Sounder on the Upper Atmosphere Research Satellite (UARS) have demonstrated that stratospheric water vapor can be measured from satellites with adequate precision to characterize temporal trends at some levels. Because of the global coverage that space-based platforms provide, continuous measurements using these techniques are critical to properly tracking the potential causes of global climate change.
Compared to those typically found in the lower and midtroposphere, H2O concentrations in the upper troposphere are extremely low (H2O concentrations from the surface to the tropopause typically decrease by about three orders of magnitude or more). As a result, the technologies used for routine weather soundings do not have the sensitivity to reliably monitor H2O in the upper troposphere. Moreover, current space-based platforms (e.g., SAGE II) are able to quantify upper-tropospheric H2O only when aerosol loadings are low (McCormick et al., 1993; Rind et al., 1993). For these reasons, new approaches must be developed for measuring H2O trends in the upper troposphere. Ideally these approaches would be amenable to remote sensing from small satellite platforms, thus affording a strategy for obtaining global coverage at a reasonable cost.
Photochemical Oxidants
Elevated concentrations of oxidants on urban and regional scales in the industrialized countries of the world are proving to be among the most intractable air quality problems (NRC, 1991). Thus, the goals of atmospheric chemistry research in the twenty-first century must include the development of a more complete understanding of the chemical processes occurring in the boundary layer and troposphere that determine the distribution and trends of the photo-chemical oxidants and their precursors on urban, regional, and global scales.
To achieve this goal, several critical scientific questions outlined below, must be addressed in the coming decades.
Page 158
1. What determines the ability of the atmosphere to cleanse itself of pollutants via free-radical oxidation, both now and in the coming decades? More specifically:
• To what extent does our current understanding explain simultaneous measured OH concentrations and the principal OH chemical production and loss processes?
• Can the oxidation of compounds or the appearance of their oxidation products be successfully used to infer concentrations of OH?
• To what extent do oxidants other than OH (O3, NO3, H2O2, halogen atoms, etc.) play significant roles in atmospheric chemistry?
• To what extent do changes in stratospheric ozone, climate, and/or cloud cover affect the oxidizing capacity of the lower atmosphere?
2. What determines the distribution of ozone in the troposphere and how will this distribution change in the coming decades? More specifically:
• What fraction of tropospheric O3 can be attributed to transport from the stratosphere, and how does this change with meteorology and season?
• What portion of O3 precursors is emitted from biogenic sources, and how will these emissions change with natural (e.g., meteorological variability) and human-induced (e.g., land use, climate change) perturbations?
• What is the contribution of urban pollution to rural and regional O3, and conversely, what is the impact of rural or regional O3 on urban pollution?
• How does meteorological variability affect the trends of O3 and/or its precursors?
• What are the major sources of the oxides of nitrogen in each region of the atmosphere over various geographic regions? What are the rates of emission of NOx from these sources?
• Which major reservoir and oxidizing species and which gas-phase and heterogeneous chemical processes are responsible for partitioning within the NOy family?
• Where and when is the production of O3 limited by the availability of volatile organic compounds (VOCs) or NOx?
• What are the trends in regional and local O3 precursors (NOx, VOCs, carbon monoxide)?
3. How can atmospheric models be improved to better represent current atmospheric oxidants and better predict the atmosphere's response to future levels of pollutants? More specifically:
• What laboratory research is required to provide sufficient understanding of the fundamental chemical processes (heterogeneous as well as gas phase) involved in tropospheric oxidant formation?
Page 159
• What atmospheric measurements are required, and with what precision and accuracy, to apply diagnostic and predictive models of tropospheric oxidant chemistry?
• What are the quantitative uncertainties associated with the estimates from diagnostic and predictive models of tropospheric oxidant chemistry?
• How can models of tropospheric oxidant chemistry be improved to incorporate direct and indirect effects of multiple, interacting forcing agents (e.g., climate change, stratospheric ozone depletion, anthropogenic perturbations)?
4. What will enable us to evaluate and improve our air quality management strategies for photochemical oxidants? More specifically:
• What design and implementation strategies will provide monitoring networks capable of determining if control measures for photochemical oxidants are having the intended impact?
• What design and implementation strategies will yield monitoring networks capable of determining, for a particular air quality problem, what portion of the problem is essentially irreducible (i.e., natural emissions of ozone precursors and stratospheric influx of ozone) and what portion of the ozone problem is potentially controllable (i.e., human-made precursor emissions)?
To successfully address these questions in the coming decades, it must be recognized that research on photochemical oxidants is truly ''data poor'' and "measurement limited." As a result, significant progress in this area will require a commitment to acquire high-quality, observational data sets that, collectively, are global in coverage but, individually, are of high enough spatial and temporal resolution to elucidate the important chemical and physical processes responsible for the production, transport, and removal of photochemical oxidants. To accomplish this, a research strategy that is both evolutionary and revolutionary will be required. The beginning of such a strategy focused on the management of urban-and regional-scale photochemical oxidant pollution in North America has recently been developed [see North American Research Strategy on Troposphere Ozone (NARSTO) and charter available from NARSTO Home Page at URL: http://narsto.owt.com/Narsto/]. The research strategy outlined below and in Box II.2.8 is similar in many respects to this previous work but also addresses longer-term and globally relevant issues.
Continue Development and Validation of Chemical Instrumentation
Instrument development and validation should aim at improving the sensitivity, specificity, and sampling rates of instruments needed to measure the compounds of interest throughout the atmosphere from the measurement platforms of choice (Albritton et al., 1990). The focus should be on (1) the development of
Page 160
|
Box II.2.8 Recommended Research Tasks for Photochemical Oxidants |
|
|
1. Continue development and validation of chemical instrumentation to |
|
|
• provide techniques for long-term monitoring; |
|
|
• provide continuous, fast-response techniques for flux divergence methods; |
|
|
• provide miniaturized techniques for airbome platforms; and |
|
|
• provide long-path spatially resolved techniques for making multidimensional measurements. |
|
|
2. Continue implementation of integrated field campaigns to |
|
|
• elucidate fundamental processes; |
|
|
• document key species' trends, sources, and sinks; and |
|
|
• evaluate air quality and chemical transport models. |
|
|
3. Carry out observation-based studies to |
|
|
• elucidate trends and distribution in short-lived radical species; |
|
|
• independently infer emission inventories; and |
|
|
. |
• infer ozone precursor relationships. |
|
4. Develop and deploy monitoring networks to |
|
|
• document the chemical climatology of photochemical oxidants, and |
|
|
• document the response of ozone to changes in precursor concentrations (e.g., as a result of emission controls). |
|
|
5. Develop analytical models and tools to support integrated assessments. |
|
simpler and more reliable instruments to be used in long-term monitoring; (2) the miniaturization of instruments to accommodate a wide array of measurements on airborne platforms; (3) the development of continuous, fast-response instruments to be used for flux measurements and airborne applications; and (4) the use of spatially resolved, long-path methods (e.g., Lidar) that can be operated from airborne and mobile platforms to determine distributions of compounds of interest over considerable distances.
Continue Implementation of Integrated Field Campaigns
Integrated field campaigns are undertaken to increase our understanding of fundamental atmospheric processes; elucidate the distributions, sources, and sinks of key species; and provide data for the evaluation of air quality and chemical transport models. Scientific guidance is required to carefully define how key uncertainties are going to be reduced and what key science questions will be addressed in a specific field campaign. Atmospheric chemistry and meteorology must be integrated in the planning and deployment of air quality measurements and monitoring. The questions that are presently before us will require multi-
Page 161
disciplinary teams that can address chemistry, transport, and ecosystem feedbacks. Modeling tools adequate to depict or simulate these processes must be available to guide the planning of measurements as well as the interpretation of results. Moreover, an adequate fleet of research aircraft must be available to the atmospheric sciences community in order to make these studies feasible.
Carry out Inferential Observation-Based Studies
Carefully designed observations of specific tracer compounds or suites of tracer compounds can be used in conjunction with diagnostic and/or observation-based models to independently infer (1) the long-term trends, seasonal variability, and regional distribution of short-lived free-radical species not amenable to continuous, spatially extensive monitoring; (2) urban-, regional-, and global-scale emission inventories of ozone precursors; and (3) the sensitivity of ozone and other photochemical oxidants to precursor compounds. It should be noted however that interpretation of field measurements will require a solid understanding of the fundamental mechanisms involved in related atmospheric processes.
Develop and Deploy Monitoring Networks
The development and deployment of monitoring networks are necessary to establish the chemical climatology of ozone, other photochemical oxidants, and their precursors. This climatology will help shorten the time required to unequivocally observe a response in ozone to changes in the concentration of its precursor compounds. These networks must include a meteorological component that captures the role of meteorology and dynamics in the redistribution of airborne chemicals. Moreover, a comprehensive chemical climatology for the photochemical oxidants must include data from the free troposphere as well the surface. It is thus likely that these networks will require the use of balloon sondes; robotic, pilotless aircraft; and space-based platforms, in conjunction with newly developed instrumentation based on small, lightweight, low-power technology.
Support Integrated Assessments
Integrated assessments draw from a wide range of scientific information and disciplines in order to provide more comprehensive guidance on scientific and technical matters to the decision-making community. A thorough understanding of the distributions and trends in photochemical oxidants and the processes that determine their production and removal is not yet in hand, and this seriously limits our ability to conduct a rigorous integrated assessment of global change (Logan, 1994; IPCC, 1995). The research strategy in atmospheric chemistry should support these assessments by providing analytical and modeling tools that can be readily applied to these integrated assessments.
Page 162
Atmospheric Aerosols
Atmospheric aerosols play a critical role in the chemistry and radiative transfer of the atmosphere. Minute amounts of particulate matter in the stratosphere, along with increased levels of anthropogenic chlorine, are responsible for the Antarctic ozone hole and probably for the less dramatic but nevertheless significant global-scale ozone depletion (WMO, 1995). Aerosols emitted by industrial activity and biomass burning are now believed to be responsible for partially masking the expected increase in surface temperature associated with greenhouse gas radiative forcing (IPCC, 1995; NRC, 1996a). Atmospheric aerosols also have important impacts on human health and materials degradation (American Thoracic Society, 1996a,b). Despite our recent advances in appreciating the importance of aerosols, our understanding of this critical class of atmospheric species is in its infancy. Why is this the case? An outstanding reason is the complex nature of aerosols and the forces they exert. Unlike the atmospheric gases, aerosols have an infinite number of sizes and a variable, mixed composition. We are not able to fully comprehend the impacts of aerosols now and are not in a position to make predictions about how these impacts will change in the future due to mankind's activities.
The important questions that must be addressed in the twenty-first century involve the effects of atmospheric aerosols on climate, atmospheric chemistry, and human health and well-being and in a fundamental form can be stated as follows:
1. What is the role of natural and anthropogenic aerosols in climate, and how will future changes in the levels of aerosol precursors affect this role?
2. How will future natural and anthropogenic aerosols impact stratospheric and tropospheric ozone and the oxidizing capacity of the atmosphere?
3. What is the role of atmospheric chemistry in changing the composition of aerosols that impact human health, the environment, visibility, and infrastructural materials?
To answer these questions, we must go far beyond our current state of knowledge of atmospheric aerosols. The essential elements of the research strategy that will be needed are outlined below and in Box II.2.9. A more detailed discussion of many aspects of this strategy can be found in Aerosol Radiative Forcing and Climate Change (NRC, 1996a).
Maintain and Expand Stratospheric Aerosol Measurement Capability
Limb scanning of solar extinction from satellites has been very successful in monitoring the global stratospheric sulfate layer and its spatial and temporal response to volcanic perturbations. When validated by in situ measurements of
Page 163
|
Box II.2.9 Recommended Research Tasks for Atmospheric Aerosois |
|
|
1. Maintain and expand stratospheric aerosol measurements to |
|
|
• document aerosol chemical effects on stratospheric ozone, and |
|
|
• monitor impact of volcanic injections. |
|
|
2. Develop new suite of tropospheric aerosol measurements to |
|
|
• document complex chemical and physical aerosol properties, and |
|
|
• expand remote sensing capability. |
|
|
3. Deploy monitoring networks to document spatial and temporal frends in aerosol characteristics and their impact on climate, human health, and so forth. |
|
|
4. Design and implement intensive field campaigns to better understand processes that control aerosol formation, transformation, transport, and loss. |
|
|
5. Develop and evaluate models to provide predictive capability. |
|
particle size distributions from balloons and stratospheric aircraft for validation, satellite multiwavelength extinction measurements have provided stratospheric aerosol particle surface areas with an accuracy adequate for heterogeneous chemical applications. New instruments with higher wavelength resolution, possibly deployed on small satellites, will be the main monitoring tool for this component in the twenty-first century.
Design and Implement New Suite of Measurement Technologies for Tropospheric Aerosols
The complexity of tropospheric aerosol presents a considerably more difficult problem. Past in situ measurements have focused on determining the size distribution or chemical composition of aerosols at specific locations. Several new techniques under development are probing the chemical composition of single aerosol particles. However, these are essentially point measurements that yield little information about spatial and temporal variability. Moreover, there are few methods for analyzing the composition of organic aerosols, which are emitted from biomass burning and industrial activity. Clearly, a new suite of in situ instrumentation is needed that can quantitatively document the complex chemical composition of tropospheric aerosols in regions of the globe that are of interest for atmospheric chemistry.
Current remote sensing technology allows the measurement of gross tropospheric aerosol parameters over large spatial regions, but features such as composition and a complete size distribution cannot be measured yet. Technologies
Page 164
such as scanning polarimeters in the visible and near infrared appear to hold promise because they are able to retrieve tropospheric aerosol scattering characteristics from measurements of multispectral radiance and polarization by resolving aerosols from clouds. Moreover, surface and airborne lidars can be used to map tropospheric aerosol backscatter and, combined with Raman scattering techniques, can provide limited information on aerosol characteristics. Preliminary measurements with nadir-viewing lidars from the space shuttle show promise for obtaining detailed gross features of the tropospheric aerosol on a global basis. However, adequate opportunities for deployment of such instruments do not presently exist and must be a priority for the twenty-first century.
Design and Deploy Networks to Document Aerosol Climatology
With the development of new instrumentation, monitoring networks can be deployed to document the spatial and temporal trends in key aerosol characteristics. These characteristics include aerosol number, size distribution, chemical composition, and radiative properties. Moreover these networks must be designed in such a way that they can address issues on varying spatial scales. For example, urban-scale monitoring networks are needed to uncover the characteristics of aerosols that lead to pulmonary health effects in humans; regional-scale networks are needed to better establish the relationships between aerosol precursor species and visibility; and global-scale networks are needed to better quantify the role of aerosols in climate change.
Design and Implement Intensive Field Programs
To be able to predict how future anthropogenic activities will affect aerosols, and their consequent impacts on climate, chemistry, the environment, and human health, we must go beyond an aerosol climatology to a deeper understanding of the processes that control aerosol formation, transformation, and removal. This will require the design and implementation of intensive field programs that bring together chemical and physical aerosol measurements and precursor gas studies utilizing surface, aircraft, and ship measurements. It is relevant to note in this regard two novel experimental strategies that have emerged for resolving some of the key questions concerning tropospheric aerosols and their effects [see, for example, the ACE-1 Science and Implementation Plan (IGAC, 1995)]. The first of these is the "closure experiment," in which an overdetermined set of variables is measured. A subset of the observations and the relevant theories are then used to predict the "closure variable," which is also measured independently. The result is a test of both measurements and theory, with an opportunity to evaluate the quality of our understanding in each experiment. With instrumentation now available, it is possible to perform closure experiments on aerosol number concentration (using a variety of sizing instruments), mass (based on measurements
Page 165
of relevant inorganic and organic species), radiative properties (using chemical composition, relative humidity, and Mie theory), and the integrated column effect of aerosols on short- and longwave radiation. Closure experiments on aerosol mass can help answer questions about chemical composition, since missing species will make closure impossible. Theories concerning the impact of aerosols on radiative forcing of climate can also be tested by local and column closure experiments. Most of the aerosol experiments planned for the next decade depend heavily on this strategy, since it offers a rigorous test of both measurements and the process models on which more comprehensive models depend.
The other new strategy is to observe the evolution of aerosols and their precursor gases in a Lagrangian reference frame. The idea of Lagrangian experiments is not new, and variations on this theme have been used from time to time. Recently, however, there has been considerable work on tagging airmasses with balloons and chemical tracers, so that aircraft carrying large suites of instruments can revisit the airmass over a period of days to observe changes with time (Huebert, 1993; Draxler and Hefter, 1989). Although these experiments cannot eliminate the effects of dispersion and vertical mixing on concentrations, with ample dynamical measurements they make it possible to sort out the chemical and physical processes that cause changes in aerosols. These processes include gas-to-particle conversion, chemical transformations, wet and dry deposition, entrainment of air from other strata, and mixing through the sides of the "airmass" (dispersion). Although these experiments tend to be complex and expensive (at least one ship and one or two aircraft are required), they offer the potential to test the aerosol models that presently exist or will be developed from future laboratory work and other process studies.
Develop Predictive Model Capability
The overall strategic goal for the twenty-first century should be development of a predictive model that can be used to calculate atmospheric temperature and chemical species concentration fields and, from this information, to derive aerosol formation rates, predict the chemical content and size distribution of the aerosol fields, and determine their concomitant influence on atmospheric radiation and the reflectivity and lifetime of clouds. Since current atmospheric models generally impose, rather than predict, aerosol distributions, it will be necessary to achieve significantly more sophistication in representing precursor gas and gas-particle kinetics, nucleation and agglomeration kinetics, and vapor-particle interactions in future models. One way to naturally stimulate the necessary improvements in aerosol modeling capabilities is to encourage the modeling community to participate directly in the planning, execution, and data analysis portions of the strategic field measurements programs described above.
Furthermore, predictive aerosol models will require currently unavailable quantitative mechanistic and kinetic input data describing a large number of
Page 166
heterogeneous growth, nucleation, agglomeration, and accommodation or evaporation processes. These quantitative input data will have to come from a vigorous laboratory program in heterogeneous kinetics and aerosol microphysics.
Toxics and Nutrients
The atmosphere and biosphere are fundamentally coupled through the exchange of gases and aerosols. Ecological systems, including economically important ones such as those dedicated to agriculture and forestry, can be profoundly impacted by the wet and dry deposition of both toxic and nutritive atmospheric substances (e.g., Ridley et al., 1977; Duce, 1986; Aber et al., 1989; Schulze, 1989; Van Dijk et al., 1990; Lindquist et al., 1991; Benjamin and Honeyman, 1992; Vitousek et al., 1993; Shannon and Voldner, 1995). Although many of the atmosphere's naturally occurring components can have toxic and/or nutritive effects on the biosphere, there are a myriad of toxic and nutritive substances in the atmosphere that are significantly influenced by anthropogenic activities. These include nutrients such as sulfur and nitrogen compounds; heavy metals such as mercury, cadmium, and lead; and toxic organic compounds such as pesticides, polychlorinated biphenyls (PCBs), plasticizers, dioxins, and furans.
Although we are beginning to be able to identify the more acute effects of atmospheric toxicity and overfertilization on key ecosystems, our understanding is far too limited for us to assess the current extent of these problems or to predict future ones. Overall, the motivating scientific questions for the study of toxics and nutrients are as follows:
1. How are interactions between the atmosphere and biosphere influenced by changing atmospheric concentrations and by the deposition of harmful and beneficial compounds?
2. What are the rates at which biologically important atmospheric trace species are transferred from the atmosphere to terrestrial and marine ecosystems through dry and wet deposition?
The essential elements of a research strategy to address these questions are outlined below and in Box II.2.10.
Develop and Evaluate Techniques for Measuring Deposition Fluxes
Many of the key questions about toxics and nutrients cannot yet be answered comprehensively because we lack the necessary methods for measuring deposition fluxes on the appropriate spatial and temporal scales. This problem is most severe in the case of dry deposition, where technologies for reliably measuring many of the most biologically important fluxes do not yet exist. Adequate support for development of the necessary techniques in this area is thus critical; relaxed eddy accumulation, eddy correlation, and gradient methods offer particular promise.
Page 167
|
Box II.2.10 Recommended Research Tasks for Toxics and Nutrients |
|
|
1. Develop and evaluate deposition flux measurement techniques to |
|
|
• provide new methods for measuring dry deposition rates, and |
|
|
• provide methods for obtaining more spatially comprehensive deposition data. |
|
|
2. Design and implement ecosystem exposure monitoring networks to develop long-term record of stresses and benefits to ecosystems of economic and/or environmental import. |
|
|
3. Carry out process-oriented field studies to |
|
|
• developed and evaluate deposition flux algorithms, |
|
|
• contribute to the development of coupled ecosystem-atmospheric chemistr model, and |
|
|
• provide tools for integrated assessments. |
|
In the case of wet deposition, reliable techniques have in principle been developed, but serious questions exist about sampling representativeness and contamination problems. The problem is most severe for measuring wet deposition fluxes over the ocean, where it is virtually impossible to collect uncontaminated rain samples from a buoy in midocean and samples from shipboard platforms are necessarily intermittent. Present marine deposition estimates, often the result of comparing model calculations with a very small suite of shipboard and island observations, are typically subject to uncertainties of factors of three or more (Duce et al., 1991). The development of new techniques that will allow for more representative determination of wet as well as dry deposition fluxes, perhaps from a low-flying airborne platform, must therefore also be considered a high priority.
In some cases, such as high-altitude forests and foggy regions, the deposition of cloud droplets may be the primary avenue by which toxics and nutrients are delivered to the Earth's surface (Vong et al., 1991). It is extremely difficult to measure such fluxes, because the droplets are so transient that their flux is easily altered by the presence of measuring devices. Thus, new methodologies should be developed to assess the importance of droplet deposition and allow reliable flux measurements.
Design and Implement Ecosystem Exposure Monitoring Networks
In the recent past, deposition monitoring networks have proved useful for assessing the ecological impacts of atmospheric deposition (e.g., Cooperative
Page 168
Programme for the Monitoring and Evaluation of Long Range Air Pollutants in Europe, National Crop Loss Assessment Network). However, these networks have been largely limited to monitoring the deposition of a specific chemical or class of compounds (e.g., acid deposition, ozone). For this reason, they have provided very limited information on the full suite of stresses and benefits experienced by an ecosystem from atmospheric deposition and, thus, on the long-term effects of this deposition. With the development of new deposition measurement techniques, it should be possible to design more comprehensive atmospheric deposition and exposure monitoring networks. Implementation of these networks for key ecosystems and biomes (e.g., at Long-Term Ecological Research sites) would provide a long-term record of atmospheric deposition; with co-located ecological monitoring, this record would no doubt prove useful in establishing causal relationships between atmospheric deposition and ecosystem vitality and succession.
Carry out Process-Oriented Field Studies for Algorithm Development and Evaluation
Even with reliable and fully evaluated deposition measurement techniques, it will never be possible to measure dry and wet fluxes for all species of interest over all ecosystems of interest, over all time. For this reason, process-oriented field studies, involving observations of fluxes under a carefully selected range of conditions, have to be undertaken in order to identify the factors that control such fluxes. With these factors identified, algorithms and parameterizations describing deposition fluxes can be developed, tested by further observations, and incorporated into regional and global atmospheric chemistry models, as well as integrated atmospheric-biospheric response models.






























































