3
Fishing and Marine Ecosystems
Introduction
Ecosystems are complex, linked, interactive systems in which organisms, habitats, and external forcing (e.g., ocean currents, weather) act together to shape communities and regulate population abundances. Fishing is a major activity that can selectively remove large portions of animal populations and also significantly alter trophic interactions. Fishing gear that drags the bottom at times, such as bottom-trawls, pots, and longlines, can alter marine habitats, especially benthic or reef habitats. Because fish populations fluctuate naturally, sometimes by orders of magnitude, separating the effects of fishing from natural biological and environmental variability is difficult.
In analyzing the effects of fishing and environmental factors on populations and ecosystems, the committee did not try to be exhaustive. Instead, enough information is provided to be representative. One difficulty is that fishing has been pursued for many years in many marine ecosystems, so that most "baselines" represent ecosystems that already are much affected (Pauly 1995, Jackson 1997). Controls are difficult to establish, and without them, the effects of fishing can be difficult to gauge (Roberts 1997).
Fishing alters the age and size structure of populations. A consequence of fishing is an altered demography. Older and larger fish often are removed first, and the remaining older cohorts experience more cumulative fishing (Baranov 1918), and the population becomes more and more dependent on small, newly recruited individuals to support the fishery. In many ecosystems, large piscivorous species were the initial targets of fishing. After the "fishing down" of those populations, the fisheries shifted to smaller species at lower trophic levels (Pauly et al. 1998). The net effect is a major change in community structure through altered trophic interactions. Not only do fish abundances and biomasses decline,
but the entire ecosystem structure can be changed. Fishing also can alter the genetic structure of fished populations. Although the immediate results of such genetic alteration might be hard to detect (Policansky 1993a), the large-scale changes that can occur in widespread fish species, especially those with discrete subpopulations like Pacific salmon, can substantially reduce the species' ability to recover from the effects of depletion due to fishing and other causes (Policansky and Magnuson 1998, NRC 1996b).
To understand the effects of fishing in an ecosystem context, the ecology of removed fish must be examined. Not surprisingly, the majority (81 percent by weight) of the 50 major species in marine catches (excluding cultured organisms) (FAO 1996a) are the small and abundant animals that are low on the trophic pyramid, eating phytoplankton (40 percent), zooplankton (38 percent), and algae (3 percent). These planktivores may have an important role in controlling plankton productivity and community structure. For example, it is hypothesized that overfishing, habitat changes, and changes in water quality associated with the removal of oysters from Chesapeake Bay (Rothschild et al. 1994, Lenihan and Peterson 1998) caused a cascade of events that may have promoted explosions of ctenophores (comb jellies) (Newell 1988). The decline of oysters may have contributed to increased phytoplankton biomass and concomitant decreases in sunlight penetration that led to declines of benthic seagrasses, which were important nursery areas for many species. Ulanowicz and Tuttle (1992) developed a network model which indicated that re-establishment of oysters and control of fishing mortality on them would increase benthic primary production, fish abundances, and zooplankton densities. In addition, oyster reefs provide physical structure and habitat for a variety of invertebrates and fish. Another example, that of removing herbivores from coral reefs, is discussed below.
Although most marine landings are of small planktivores, fishing and the removal of large species at high trophic levels that affect ecosystem structure and functioning (i.e., piscivores and large predators) can have major effects on ecosystems (sometimes called "top-down control") (Carpenter et al. 1985, Hixon and Carr 1997). It has been hypothesized and demonstrated in some systems that removal of predators can have impacts out of proportion to their abundances through a trophic cascade that affects not only prey of predators but also indirectly the lower trophic levels that constitute the food resource of the small fish and invertebrates that were eaten by predators (e.g., Paine 1980, Simenstad et al. 1978, Carpenter and Kitchell 1988, Parsons 1992). Marine assemblages dominated by sea otters (Box 3-1) are useful model systems for understanding and describing multispecies, multitrophic-level interactions. Three trophic levels—carnivorous otters, herbivorous urchins, and photosynthetic benthic algae—are clearly present. Because otters can control the density and size structure of the urchins and the urchins control the specific identity and therefore the productivity of the algae, the linkage between the species and trophic levels is strong. At high density, the otters instigate a trophic cascade (Paine 1980).
Trophic cascades are characterized by three features generalizable to other benthic and aquatic systems: high trophic-level species influence the assemblages' structure (top-down control), indirect effects two or more links distant from the primary one are biologically conspicuous, and alternative community states in which different species are abundant and ecologically dominant (Sutherland 1974, Hughes 1994) can persist. To the degree that these results from benthic assemblages can provide an analogy for the dynamic organization of nearshore and oceanic communities, they provide a cautionary note. Fishery biologists should anticipate community changes when high trophic-level populations are heavily exploited. This effect is not necessarily only a consequence of modern technology. For example, Aleut exploitation of sea otters appears to have changed the structure of nearshore marine communities as long 2,500 years ago (Simenstad et al. 1978).
In addition to the effects of directly removing animals and the effects on the ecosystem that this precipitates, fishing can physically affect the marine environment. The most prominent of such effects are destruction and disturbance of bottom communities and of bottom topography by trawls and dredges (Dayton et al. 1995, Auster et al. 1996). The advent of large-scale mariculture and the development of offshore facilities to support it also have the potential to alter marine ecosystems significantly.
Both long- and short-term environmental fluctuations have major effects on the abundances of marine organisms (e.g., Soutar and Isaacs 1974, Baumgartner et al. 1992). Although not caused by fishing, environmental effects are often hard to distinguish from those caused by fishing (e.g., Wooster 1983; Bakun 1993; Cushing 1982; Rothschild 1986, 1995) and can have profound consequences for management. For this reason, the following sections include a brief review of environmental effects as well as more detailed reviews of the effects of fishing.
Removal of Herbivorous Fishes from Coral Reef Ecosystems
One of the more dramatic ecological effects of removing herbivores, including fishes, from an ecosystem has been described by Hughes (1994), Jackson (1997), and others. Caribbean coral-reef ecosystems include herbivorous fishes and urchins (Diadema antillarum) as well as several species of coral. However, many of the herbivorous fishes and other animals had been removed by fishing long before any serious ecological study of coral reefs began in the 1960s, so the importance of their presence was not appreciated until a combination of recent events made matters clearer. Indeed, Jackson (1997) considered the study of reef herbivores since the 1950s to be analogous to studying termites and locusts in the Serengeti in the absence of wildebeest and elephants.
The first event in the chain that exposed the effects of chronic overfishing was Hurricane Allen, a category 5 hurricane that struck the Caribbean in 1980. It
|
BOX 3-1 Northern sea otters (Enhydra lutris) originally ranged across the Pacific rim from Hokkaido (Japan) to Baja California (Mexico). Their primary prey includes a broad range of invertebrates and fishes; because they inhabit nearshore environments, a wealth of ecological detail is available (Van Blaricom and Estes 1988). Exploitation for their pelts led to near extinction by 1911, when unregulated killing ceased. Preservation and more recently, restoration, rather than population sustainability have become the dominant themes of otter biology. During early phases of recovery, otter populations grew at a rate of about 15 percent per year, especially in the Aleutian Island chain, producing high population density on some islands in the same geographic region as islands that lack otters. Inter-island comparisons were central to the classic study of Estes and Palmisano (1974): otters as keystone species control the local biomass and, to a lesser degree, the abundance of sea urchins, which regulate benthic algal biomass and productivity. Duggins and colleagues have increased our confidence in these patterns of direct interactions by observations made before and after reinvasions demonstrating that in otter-dominated habitats, high kelp biomass and therefore the generation of detrital materials have also indirectly increased the growth rate of suspension-feeding invertebrates (Duggins 1980, Duggins et al. 1989). Otter populations increased generally until about 1990 throughout western Alaska and the Aleutian Islands; since then a decline amounting to 35 to 90 percent of the populations has occurred. Although the causes remain unknown, the consequences increase our confidence in the robustness of the classic otter-induced cascade. At Adak Island, where the otter population has declined from an estimated 5,000 to slightly more than 600 in 1997, urchins have increased about fivefold in number, and their maximum size has more than doubled. Conversely, kelp density has declined by a factor of 10 (James Estes, University of California, personal communication, 1997). It seems unlikely that aboriginal Aleut hunters maintained sustainable otter populations, based on cycles of apparent abundances and associated community changes identified in midden remains (Simenstad et al. 1978). The otter provides a textbook study of the (unresolved) difficulties that ecosystem managers may face, because legally mandated protection of otters conflicts with multiple-use concepts (Levin 1988). Otters consume and are capable of local decimation of sea-urchin populations (the exploitation of which is a growing multimillion-dollar industry), although in the process the otters indirectly facilitate kelp production (itself an important commercial resource). Kelp beds provide nursery grounds for nearshore fishes, and their detrital production may enhance the growth of abalone or other benthic invertebrates (e.g., Pismo clams). Otters consume these animal species, many of which are of recreational or commercial significance and clearly play a critical role in the structuring and organization of nearshore marine ecosystems. |
severely damaged coral reefs around Jamaica that had not seen a major hurricane for more than 40 years. For the next three years the corals recovered, but in 1983 a disease devastated the Diadema populations. Because both the major groups of herbivores were now absent—the fishes, reptiles, and mammals removed by exploitation and the urchins killed by disease—the recovery of corals stopped because they became overgrown by algae. The coral cover was reduced from a mean of 52 percent in 1977 to 3 percent in the early 1990s, and the cover of fleshy macroalgae increased from 4 to 92 percent. Although the events described by Hughes (1994) occurred in Jamaica, other areas of the Caribbean have been similarly affected, and much of the Caribbean has been subjected to overexploitation of herbivores for at least several hundred years (Jackson 1997). High nutrient loads that favor algal growth over coral growth also have adversely affected the region's marine ecosystems.
Fishing has devastated coral-reef ecosystems in the Pacific as well. Although illegal, dynamite is often used there. Johannes and Riepen (1995) reported on fishing with cyanide to catch live reef fishes in the Philippines, Malaysia, and Indonesia. Cyanide fishing is inefficient because many fish die in reef crevices and are not captured; of those captured, some die before reaching their destinations and are not used. The fishing technique also kills the reefs and nontarget species, so its effect on reef ecosystems is even greater than the loss of the fish.
Other Ecosystem Effects of Fishing in the Philippines
The Philippines were the first among Southeast Asian countries to develop a modern bottom-trawl fishery immediately after World War II. This fishery grew quickly, as did the pelagic fishery, which relied mainly on boats using light sources to attract fish into nets, and a coral reef fishery using a variety of mostly destructive methods, notably the persistent muro-ami technique of pounding reefs to scare fish into surrounding gillnets.
The Philippines consist of about 7,000 mostly high islands, surrounded by a relatively small continental-shelf area. The adjacent waters are extremely deep and infertile. The inherent limits to fishery catches implied by the small shelf area were until recently not recognized by government planners and development banks, which subsidized acquisition by the industrial sector of a fleet capacity about three times in excess of what was required to harvest the present marine catches of 1.9 million metric tons (t) per year (BFAR 1994).
The excess capacity in Philippine fisheries is particularly tragic in that it impoverishes the numerous small-scale fishers, who would be technically capable of catching and marketing most of the fish presently harvested by the overcapitalized industrial fleet. This impoverishment, further exacerbated by large numbers of landless farmers entering the small-scale fishery sector, leads to
what is now called "Malthusian" overfishing (Pauly et al. 1998), characterized by the widespread use of dynamite and cyanide as cheap ("entry-level") fishing gears.
The effects on coastal ecosystems of the combined fishing pressure of the industrial and small-scale sectors, fishing techniques that destroy reefs, and the downstream effects of nonsustainable agricultural and forestry practices have been devastating, with most of the fringing reefs surrounding the Philippine islands choking under silt and experiencing massive species changes. The slowly declining fish supply is now dominated by small, low-value species.
Bycatch, Discards, and Unobserved Fishing Mortality
Bycatch is the capture of nontarget species in directed fisheries. Discards are animals returned to the sea after being caught. Some bycatch is retained in most fisheries, but most of it is usually discarded and not reported in official landing statistics. Even some retained bycatch is not reported, and sometimes targeted species are discarded if they are too small. It is important to be clear about these terms (Alverson et al. 1994) because they are often used differently by different authors, and it is impossible to evaluate the importance of bycatch and discards if they are not clearly distinguished from each other.
Because a significant portion of the catch (discards) in many fisheries is not reported, the portion that is reported constitutes only a fraction of the actual catch. Discards make stock assessments and fishing mortalities difficult to estimate because they are usually unreported; even when they are, the reports usually lack specific information on the age and size of the animals discarded, which is needed for stock assessment. Thus, inferred patterns of exploitation are significantly underestimated. The situation is made worse by a variety of other unobserved sources of fishing mortality such as illegal fishing, underreporting, deaths of fish that escape from fishing gear, and ghost fishing. Recreational and subsistence fishing are also difficult to monitor and thus can represent a significant source of unobserved fishing mortality, especially for widely sought species such as bluefish (Pomatomus saltatrix), spotted seatrout (Cynoscion nebulosus), and summer flounder (Paralichthys dentatus) in the southern and eastern United States. The magnitude of discard mortality and unobserved fishing mortality could be important factors contributing to global overfishing and undesirable ecological changes.
Alverson et al. (1994) reviewed the literature on worldwide bycatch and discards and concluded that marine discards in the period 1988–1990 amounted to approximately 27 million t per year, roughly one-third as much as the total marine capture fisheries.1 There is great variation in bycatch associated with
various kinds of fisheries. By far the largest contributor to marine discards—nearly 10 million t per year—was shrimp trawling, especially in tropical regions. This biomass of discards represents approximately five times the biomass of the shrimp and includes more than 240 species, including the young of commercial species and adults of some species that mature at less than 10 cm in length (Alverson et al. 1994).
On the other hand, not all trawl fisheries have high bycatch rates. In the trawl fishery for northwest Atlantic silver hake (Merluccius bilinearis), bycatch is about 1 percent of landings by weight (Alverson et al. 1994). In some cases the very large catches of target species can result in large absolute bycatches and discards even when the bycatch rate is low. One example is the midwater-trawl fishery for walleye pollock (Theragra chalcogramma) in the Bering Sea and the Gulf of Alaska, where overall discards are reported to be approximately 6 percent of the landed weight and 0.5 percent of the landed number of animals, but discards in 1992 involved more than 130 species and exceeded 100 million animals, more than half of which were pollock. Aggregate discards in the Bering Sea and Gulf of Alaska bottom fisheries approach 1 billion animals each year (Alverson et al. 1994). Another example is the Gulf of Mexico shrimp-trawl fishery, which has resulted in the deaths of tens of thousands of sea turtles, but the bycatch rate is so low that many shrimpers do not catch even one turtle in the course of a year (NRC 1990).
In some fisheries the species in the bycatch are of special concern because they contribute to and aggravate an overfishing problem, involve the target species of other highly regulated fisheries (e.g., Pacific halibut [Hippoglossus stenolepis] caught in bottom-trawl or crab-pot fisheries in the region), or are threatened or endangered species (e.g., turtles caught in shrimp trawls). Bycatch mortality and the resulting discards can have a significant effect on a particular nontarget species or on a marine community. For example, the red snapper (Lutjanus campechanus) taken in the Gulf of Mexico shrimp fishery presents a convincing example of how bycatch losses can affect a valuable nontarget species. Young red snapper suffer heavy bycatch mortality in the trawl fishery for shrimp (Penaeus spp.) (Alverson et al. 1994). The discarded bycatch represents the single largest component of fishing mortality on the red snapper population.
Using present trawling gear and methods in combination with bycatch-reduction devices, prohibiting trawling in some places at some times, and limiting towing times would probably decrease landings and revenues from the shrimp fishery but increase landings and revenue to red snapper fishers. The societal tradeoffs involved are not clear, but studies suggest that if a substantial reduction in bycatch were achieved, a sustainable commercial red snapper fishery with landings at least three times higher than those now recorded would be possible (Goodyear 1985, 1995; Goodyear and Phares 1990). A 50 percent reduction in red snapper bycatch has been recommended by the Gulf of Mexico Regional
Fishery Management Council, but no regulatory action has yet been taken to achieve that goal.
Other well-known examples of bycatch problems have been or are being addressed by technological and operational changes in fishing. One example is the killing of dolphins in the yellowfin tuna fishery in the eastern tropical Pacific (NRC 1992a, MMC 1998). Formerly responsible for hundreds of thousands of dolphin deaths per year, the fishery now kills fewer than 3,000 dolphins annually.2 Another example is shrimp trawling, which was killing as many as 11,000 endangered sea turtles a year in the Gulf of Mexico and off the southeastern U.S. coast (NRC 1990), but the adoption of turtle-excluder devices has reduced that mortality. Many examples of bycatch and potential solutions to them were discussed at a recent workshop (Alaska Sea Grant 1996).
The ecological consequences of bycatch and discards are not well quantified for most marine ecosystems. As described in Chapter 5, more research is needed on those consequences. Nonetheless, there appears to be general agreement that bycatch and discards can and should be reduced (Alverson et al. 1994, Alaska Sea Grant 1996). Chapter 5 describes various current efforts to reduce them as well as suggestions for additional approaches.
Fishing and Large Marine Ecosystems
Georges Bank
Georges Bank—a shallow, productive, submarine plateau off New England and Nova Scotia—is the poster-child, so to speak, for the effects of overfishing. In addition to severe depletion of several populations of commercially valuable groundfish, the Georges Bank ecosystem and bottom habitat have suffered large impacts from fishing. The following account is adapted from Fogarty and Murawski (1998).
Georges Bank has supported commercial fishing for at least four centuries. Important finfish species included halibut and other flatfish species, especially yellowtail flounder (Pleuronectes ferrugineus), haddock, and cod. The two major impacts on the fishery were the arrival of the distant-water fleet in 1961 and the modernization and expansion of the domestic fleet after the establishment of extended U.S. jurisdiction over fisheries (the 200-mile limit) in 1977. Despite repeated warnings from scientists, the principal groundfish species have been severely depleted; Fogarty and Murawski (1998) reported that exploitation rates of cod and yellowtail flounder were 55 and 65 percent, respectively, of the
biomass in the early and mid-1990s;3 the recommended exploitation levels based on optimal sustained yield were 13 and 22 percent, respectively. Spawning-stock biomasses were also very low, about 10 percent or less of their values in the 1950s for haddock, cod, and yellowtail flounder.
The depleted groundfish populations have largely been replaced by populations of small elasmobranchs, mainly spiny dogfish (Squalus acanthias), smooth dogfish (Mustelis canis), and skates (Raja spp.). Other changes in the pelagic fishes included initial declines of mackerel and herring and concomitant increases in American sand lance (Ammodytes americanus), their prey. Recently, fishing pressure has resulted in declines in the elasmobranch populations—the species are mostly long lived and slow growing compared to many commercially important teleosts—and mackerel and herring populations have increased as well. This has been accompanied by a decline in sand lance populations.
This example makes the ecosystem consequences of fishing clear: the groundfish community first became dominated by small sharks (e.g., dogfish) and rays, and then overfishing reduced those populations. It is not yet known whether this ecosystem will recover to its preexploitation structure in the absence of fishing or whether it will attain some other composition.
The Bering Sea
Recent declines of many populations of marine mammals and birds that live in and near the Bering Sea, a semienclosed basin of the North Pacific Ocean between Alaska and Russia, have attracted attention and have been attributed by many to the effects of fishing. The National Research Council recently reviewed the information (NRC 1996a) and concluded that fishing probably has affected the ecosystem but in a more complicated fashion than simple overfishing and in combination with environmental changes. Documented changes include changes in abundances of many fish species and changes in the physical environment. There also is persuasive (although not conclusive) evidence that marine mammals and birds are declining because the juveniles are short of food.
The NRC report concluded that the changes in the Bering Sea ecosystem were probably caused by a combination of changes in the physical environment coupled with heavy exploitation of components of the system (whales and fishes). Many sperm and baleen whales were removed from the Bering Sea and adjacent waters in the 1950s, 1960s, and 1970s. Various flatfish species, Pacific Ocean perch, and herring were also heavily fished in that period, with resulting population declines. Many of those species feed heavily on zooplankton and thus compete with walleye pollock; others of those species prey on pollock. In the late
|
3 |
The exploitation rates are much lower following recent action by the New England Fishery Management Council (NEFMC; see NRC 1998b and the Northeast Multispecies Fishery Management Plan and amendments at NEFMC's web site, www1.shore.net/˜nefmc/. |
1970s the physical regime appears to have shifted as well, resulting in higher sea-surface temperatures and less ice cover than before, conditions that seem to favor pollock recruitment.
As a result, the ecosystem appears to have become more dominated by pollock than it was before. In recent years other predatory fishes—mostly flat-fishes—have increased as well. These predators might be responsible for the decline of species normally favored by marine mammals and birds, such as capelin (Mallotus villosus), Pacific sand lance (Ammodytes hexapterus), and squid (Berryteuthis sp. and Gonatus sp.) As a result of these changes, juvenile marine mammals and birds have been deprived of their preferred foods. Thus, fishing (including whaling) appears to have contributed to a significant change in the structure and functioning of this large marine ecosystem, although the total biomass removed by the current pollock fishery does not seem to be a major contributor to the problem.
Analogs to the Bering Sea
The complexity of marine ecosystems and the number of potential factors involved make it difficult to have great confidence in our understanding of the precise mechanisms that relate fishing to the populations of top predators. The NRC report (NRC 1996a) pointed out that, although there has been heavy fishing pressure in the North Sea, in the upwelling areas off South Africa and Namibia, and off Peru, there have not been clear effects on the populations of pinnipeds.
Nonetheless, it seems likely that continued removal of large portions of various trophic levels from marine ecosystems will affect ecosystem structure and functioning. One issue that needs resolution is the effects on marine ecosystems of populations of marine mammals as they recover from very heavy exploitation. Baleen whales eat zooplankton and thus compete for food with many commercially important fish species; toothed whales eat fish and squid and thus compete directly with humans for food. The recovery of whale populations is one of many examples where different policy goals (i.e., protection of marine mammals, catching fish for food) have the potential to conflict.
The Barents Sea
The Barents Sea, off the extreme northwestern coast of Russia and the extreme northern part of Norway, contains heavily exploited populations of cod (Gadus morhua), capelin (Mallotus villosus), and herring (Clupea harengus) as well as marine mammals (whales and seals). Collapses of fish populations, crises in the fisheries, and a destabilization of the ecosystem occurred during the 1980s, a consequence of overfishing in the Norwegian and Barents seas.
The mature stock of Atlanto-Scandian herring is fished primarily in the Norwegian Sea, but its young use coastal regions of the Barents Sea as a nursery,
where herring is an important prey of cod. Capelin is an opportunist species that is also an important prey of cod. All of the species experience good recruitment when temperatures are above average, which occurs during periodic events when substantial Atlantic water flows into the Barents Sea (Skjoldal et al., 1992; Hamre, 1994).
Atlanto-Scandian herring were overfished in the Norwegian Sea and areas outside the Barents Sea. The herring stock collapsed, and its recruitment failed during the 1960s and 1970s. The scarcity of herring, itself a major predator on larval and small juvenile capelin, favored recruitment and growth of the capelin population. Despite heavy fishing, the capelin population declined only slowly during the 1970s when temperatures were below normal. Capelin grew slowly and matured at relatively old ages, minimizing massive postspawning dieoffs. The cod population, which sustained continuous heavy fishing, declined from the mid-1970s through the early 1980s.
Paradoxically, Atlantic inflow events in the early 1980s triggered the ecological crisis. Herring pre-recruits (fish still too small to be targets of fishing) returned in modest numbers to the Barents Sea, not in high enough abundance to satisfy the predation demand of cod but numerous enough to cause recruitment failures of capelin during the mid-1980s. Higher temperatures and fast growth of previously recruited capelin during this period brought them to maturity; they spawned and died. As a result of this loss of mature capelin and the herring-induced recruitment failure, the capelin population collapsed. The strong year classes of cod in the warmer environment had insufficient capelin or herring prey to satisfy demands for growth; their condition deteriorated, cannibalism became common, and the biomass of cod declined (Laevastu 1993). The fisheries for cod and capelin were in crisis.
The capelin fishery was placed under moratorium from 1987 to 1990. The opportunist capelin made a strong recovery in the early 1990s, perhaps associated with warming of the ocean (Everett et al. 1996), but the populations collapsed again in the mid-1990s (Institute of Marine Research, Bergen 1995). The Barents Sea ecosystem remains in stress.
The relatively simple community structure of the Barents Sea ecosystem is easily destabilized. Even in ''simple" systems, interactions among species and the role of environment are complex and initially unpredictable. Herring, which apparently play a pivotal role in controlling Barents Sea dynamics, were over-fished in the adjacent Norwegian Sea. That spatially removed intervention, combined with complex changes in ocean climate and plankton dynamics (Skjoldal et al., 1992), set the stage for the collapse of Barents Sea fisheries.
The Southern Ocean
The Southern Ocean—the seas off Antarctica south of approximately 50 to 60 degrees—is substantially isolated from other oceans by the Antarctic Convergence or South Polar Front (Kock 1992). It is noted for the presence of the world's heaviest seabirds (penguins), large populations of krill (planktonic crustaceans, mainly Euphausia superba ), formerly large populations of whales, and some commercially valuable fish species. Most of the fishes are relatively slow growing and thus cannot support high exploitation rates (Kock 1992). Indeed, Kock estimated that the total sustainable catch of all finfish from the Southern Ocean is no more than 100,000 t per year, about 0.1 percent of the world catch. As mentioned in Chapter 2, the marbled rockcod (Notothenia rossi) has been severely overexploited, with 400,000 t being taken in 1969–1970 alone. Recent production has been on the order of a few thousand metric tons per year. The Patagonian toothfish (Dissostichus eleginoides) also appears vulnerable to overexploitation, which has probably already occurred. It has become popular in restaurants, where it is known as Chilean sea bass. Catch rates and population estimates of this fish are poorly known (Kock 1992, Albemus 1997), but Kock reported that the some D. eleginoides populations were significantly overfished.
Although ecosystem effects of overexploitation have probably occurred in the Southern Ocean, they have unfortunately not been well studied (Kock 1992). Despite the general lack of information, it is known that the main prey species for larger marine vertebrates in the ecosystem are krill, and they have been studied extensively (Miller and Hampton 1989). They appear to be larger, longer lived, and slower growing than most marine plankton. Biomass estimates vary, but the standing stock is probably at least several hundred million metric tons in the summer (Miller and Hampton 1989). Calculations by Bengston (1984), Laws (1985), and Yamanaka (1983) and a detailed review by Miller and Hampton (1989) suggest that current consumption of krill by predators in the Southern Ocean exceeds 200 million t per year, of which perhaps 40 million t is due to baleen whales. Baleen whale populations in the Southern Ocean consumed perhaps 190 million t per year before exploitation. Thus, there is a "krill surplus" of about 150 million t per year (Miller and Hampton 1989). This is not currently being exploited by fisheries (krill fisheries are only around 100,000 t/year [David Agnew, Imperial College, London, personal communication, 1997] and finfish landings are very low, as mentioned above). It would be of great interest to know how the "surplus" is being consumed in the ecosystem and how recovery of whale populations would affect the ecosystem. As Miller and Hampton (1989) pointed out, it is not really a surplus; the term refers only to krill that is no longer consumed by whales. Some of the "surplus" may be recycled through decomposition pathways; most of it probably is eaten by other predators. Thus, the removal of whales has led to increases in other predator populations. It would
also be of interest to know how recovery of whale populations in other areas with significant fisheries, such as the Bering Sea, might affect the ecosystem.
Several features of the Southern Ocean ecosystem make it a good system in which to study the ecological effects of fishing. The ecosystem has short food chains and thus a great abundance of top predators, such as seabirds, whales, and seals (Center for Law and Social Policy and The Oceanic Society 1980). It has received a great deal of attention and has experienced relatively little exploitation to date (with the obvious exception of the great whale fisheries). Drawbacks include its large size and lack of detailed fishery statistics.
Deep-Sea Fisheries
The deep ocean (more than 1,000 m) has an enormous volume, comprising more than 88 percent of the world's ocean (Merrett and Haedrich 1997). It contains many species of fishes and invertebrates, some of which have large populations (Merrett and Haedrich 1997, Haedrich 1997). As technology and fishing power have improved and as some populations of commercially important species have become less abundant in shallower waters, pressures have increased to fish deeper waters. Because most midwater fishes that live at great depths are small, much attention has been given to the larger demersal species, the best known of which is the orange roughy (Hoplostethus atlanticus) (Merrett and Haedrich 1997).
Merrett and Haedrich (1997) took an ecosystem approach to evaluating the potential of the deep sea to provide a significant yield of fish. They acknowledged that information on many aspects of deep-sea fishes and fisheries is sparse, but they concluded that the available information suggests that no large-scale fishery for deep-sea fishes is possible. Several factors support this conclusion. First, the energy available to deep-sea ecosystems is much less than that available for shallower marine ecosystems. Second, many deep-sea fishes have watery muscles, and as much as 90 percent of their biomass is water. As a result, although they can be quite large, many deep-sea fishes yield very little edible protein. Third, and perhaps most important, the growth rates of most deep-sea fishes are low, so they become big enough to catch profitably only at ages of 30 years or more. This means that only very low exploitation rates are sustainable; indeed, Merrett and Haedrich provided many examples of deep-sea fisheries that have substantially and rapidly reduced the biomass of the target species.
Finally, Merrett and Haedrich examined the community structure of various marine ecosystems and noted that many shallow ecosystems have several top predators other than human fishers—for example, marine mammals, large sharks, and large birds. In such ecosystems, humans are competitors with those predators. Indeed, their activities seem to affect the abundance of those predators (e.g., NRC 1996a). But in deep-sea ecosystems, such predators do not seem to be present in any abundance. In Merrett and Haedrich's words (1997 p. 226), "the
implication is that the addition of a higher predator, i.e., the fishery, may not be possible."
The above conclusion does not imply that no sustainable deep-sea fisheries are possible. A few localized fisheries have been conducted for many decades, apparently sustainably, such as the Madeira fishery for black scabbardfish (Aphanopus carbo) (Merrett and Haedrich 1997). But technological improvements and economic considerations such as the need to recoup capital investments have generally led recent large-scale deep-sea fisheries to deplete their target species. Management of such fisheries is made especially difficult by the lack of information, the lack of institutional authority when the fish are outside all countries' jurisdictions, and the very long generation times of most deep-sea fishes. Those factors make it even harder than usual to evaluate the effects and effectiveness of management.
Effects of Fishing on Benthic Ecosystems
In addition to catching fish and other target species, fishing can affect ecosystems in other ways, most significantly through mechanical changes to the bottom caused by dragging fishing gear across it. Auster et al. (1996) described the effects of mobile bottom-fishing gear on the seafloor communities of the Gulf of Maine. At Swans Island, a conservation area that had been closed since 1983 was compared in 1993 with adjacent sites outside the area. At Jeffreys Bank, surveys made in 1987 were compared with surveys in 1993, after a new type of mobile bottom-fishing gear had allowed fishing on the rock-strewn bottom. Stellwagen Bank, a heavily fished area, was observed in 1993 and 1994. At all sites fished, significant and large reductions in various components of the benthic community had occurred. In some cases the changes (and losses of animals) were observed in the paths of scallop dredges and trawls; at Jeffreys Bank, the previously protected areas (because of the large rocks) showed large losses of benthic communities after the new gear allowed fishing there.
Auster et al. (1996) and Dayton et al. (1995) reviewed literature showing significant effects of bottom-fishing gear, including reduction of habitat complexity and destruction of physical refuges for animals (including biotic structures such as the tubes of tube-worms). They concluded that those processes directly reduced the diversity and productivity of many benthic communities and indirectly could affect such processes as recruitment, growth, and reproduction of many species, including commercially important ones. It seems likely that large areas of continental-shelf waters worldwide have been affected by bottom fishing, and some deepwater areas (up to 1,000m) could have been affected as well, depending on the gear rigging and type and the substrate. Lenihan and Peterson (1998) described the degradation of oyster reefs caused by oyster-dredging in Chesapeake Bay and North Carolina's Neuse River, and showed that the physical degradation interacts with water quality to cause the observed pattern of oyster
mortality. They concluded that interactions among environmental disturbances imply a need for integrative ecosystem-based approaches to restoration of estuaries, and the following sections emphasize that their conclusion applies to coastal marine areas in general.
Mariculture
Mariculture, or marine aquaculture, is the farming of fish, molluscs, crustaceans, and plants. It has a very long history but has grown rapidly in recent decades (NRC 1992b, FAO 1997a, 1997c, Anderson 1997, Chamberlain 1997, Lu 1997). It has often been controversial, and many U.S. state and federal laws express various policies toward it, some cautionary (NRC 1992b). We consider it here only briefly. The major and closely related issues relevant to this study are the potential of mariculture to supplement marine capture fisheries and its environmental effects.
Mariculture (excluding plants) produced 6.7 million t in 1995, about 7 percent of the world's marine fish production (FAO 1997a). (Freshwater aquaculture, also excluding plants, produced 14.6 million t in 1995.) In some cases, however, mariculture comes closer to dominating: by 1994, farm-raised shrimp production totaled 891,000 t, almost half the world's production of 1.9 million t of wild-caught shrimp (Chamberlain 1997). Farmed salmon production exceeded 550,000 t in 1995 (Anderson 1997). By 1994 it exceeded U.S. (including Alaska) commercial landings for the first time (Johnson 1995). Cultured organisms usually are higher-valued species than the average of wild-caught species (FAO 1997a), so their total value is a greater fraction of the total value of world fisheries than would be suggested by comparing total biomasses. What does the future hold? The NRC (1992b) suggested that if mariculture continued to grow at the same rate as in the recent past, it would produce 33 million t by the year 2000 and could effectively supplement a commercial fishery yield of 100 million t. Others also have given high priority to expanding mariculture (e.g., FAO 1995b).
Many difficulties that accompany the promotion and growth of mariculture have been described (e.g., NRC 1992b, Anderson 1997, Chamberlain 1997), including environmental pollution, spread of disease, introduction of unwanted exotic species, and social and economic factors. A major environmental problem is the use of coastal habitat for shrimp farming, mainly in Asian countries, which results in the loss of native coastal ecosystems, especially mangroves (Chamberlain 1997).
One important question not addressed until recently is the degree to which mariculture depends on ecosystem subsidies from elsewhere (Folke et al. 1998). In other words, mariculture—with its production of wastes that must be dissipated by local ecosystems, and its demand for food that must be created by primary and secondary production in more distant, usually marine, ecosystems (Folke and Kautsky 1992)—does not necessarily represent a more efficient use of ecosystem services than marine capture fisheries. To the degree and in the
circumstances where it does, it does have potential for supplementing marine capture fishing. It is certain that terrestrial agriculture is more efficient at producing carbohydrates than natural terrestrial production, but even in terrestrial agriculture, local natural production of carbohydrate through photosynthesis is heavily subsidized by the use of fossil fuel to produce fertilizers, pesticides, and power machinery. That subsidy represents production of distant ecosystems, in this case distant in time. We do not have all the information at present to calculate and compare productivity of mariculture with marine capture fisheries, but Folke and Kautsky (1992) and Folke et al. (1998) provided some insights.
Folke and colleagues estimated that fish farming in cages uses the production of marine ecosystem areas between 20,000 and 50,000 times as large as the cages for food and areas 100 to 200 times as large for the dissipation of wastes. Folke and Kautsky (1989) calculated that intensive systems are much more demanding of ecosystem services than extensive ones and require approximately the same amount of production from natural ecosystems as capture fisheries. For example, mussel long-line rearing—an extensive rather than intensive system—needs a support area of only 20 times the area of the mussel farm. Folke and Kautsky argued that intensive mariculture is not a substitute for capture fisheries, and the production of 33 million t from mariculture may not be sustainably compatible with the current production of marine capture fisheries.
Despite these problems, aquaculture does have the potential to reduce some adverse consequences of capture fisheries. For example, as mentioned above, Alverson et al. (1994) estimated that 9.5 million t—more than one-third—of the world's discarded bycatch resulted from shrimp trawling, although some shrimp are trawled to provide broodstock for the shrimp farms and some shrimp farms have serious adverse effects on local coastal (e.g., mangrove) environments. Whatever other ecological and socioeconomic consequences the culture of shrimp might have, to the degree that it reduces demand for wild-caught shrimp it is likely to reduce that bycatch. However, adverse environmental effects can include genetic and food-web consequences of genetically modified or nonnative organisms that escape (e.g., NRC 1996b); degradation or destruction of wetland and mangrove habitats to provide space for mariculture facilities; contamination of surface- and groundwater by fish wastes, pesticides, antibiotics, and other drugs; generation of red tides and related phenomena; and overfishing of wild populations to provide broodstocks for mariculture farms, in addition to the effects described by the NRC (1992b).
Clearly, a simple generalization about mariculture is not possible. Mariculture is practiced in many different ways, some of which have fewer environmental effects than others. In some cases, in situ culture of suspension-feeding organisms such as oysters can help remediate the effects of fishing by helping to restore an ecological function to the ecosystem (Lenihan and Peterson 1998). Nonetheless, it seems safe to say that the potential for present practices to supplement marine capture fisheries should be carefully evaluated wherever it is being used or proposed.
Environmental Change and Variability
Marine ecosystems respond quite differently to environmental changes over various time scales, and they are different enough from terrestrial ecosystems that much of our understanding of terrestrial ecosystems does not apply to marine ones (Steele 1985, 1991a, 1991b, 1991c, 1996, 1998). Oceanic environmental changes occur diurnally (tides), over periods of several days (storms), over periods of months to years (upwelling, eddies, warm-core rings [Hofmann and Powell 1998]), over several years (El Niño-Southern Oscillation, North Atlantic Oscillation, variations in sea-ice cover in the Barents and Bering seas and Southern Ocean), and up to a century or more (the conveyor-belt, a large-scale ocean-circulation system involving all major oceans from arctic to Antarctic latitudes [Broecker 1991]). In addition, human activity other than fishing has affected the marine environment and marine and anadromous fishes at a variety of time and space scales.
Natural4 environmental changes have been implicated in several examples of changes in marine ecosystems, some of them described earlier in this chapter (e.g., the Bering Sea, the Barents Sea). Indeed, large fluctuations in population densities (or at least distributions) of marine fishes have been documented from periods long before fishing could have been a factor, most notably the very large fluctuations in the densities of scales of hake, anchovy, and sardine in sediment cores off California over the past 2,000 years (Soutar and Isaacs 1974) (Figure 3-1). Because they are so widespread and have been widely reviewed elsewhere (e.g., Wooster 1983, Wooster and Fluharty 1985, Laevastu 1993, Everett et al. 1996), only a few examples of environmentally related fluctuations in fish populations are provided here, both human caused and natural. We caution that it is often difficult to disentangle the effects of environmental changes from those of fishing on fish populations; often they are both important factors. The recent study by Polovina and Haight (in press) of spiny lobsters in a protected and an unprotected area in Hawaii is an excellent example of how experimental or observational controls are needed to prevent the effects of fishing and environmental changes from being confounded.
Salmon in the Pacific Northwest
Salmon populations and fisheries in the Pacific Northwest have declined severely over the last century (NRC 1996b). Salmon have disappeared from 40 percent of their historical breeding ranges in the continental United States and continue to decline despite a public and private investment of more than $1 billion in the last decade for protection and enhancement of salmon populations. The frequency of populations experiencing the greatest difficulty increases in a
southward trend from British Columbia to California. Species that spend extended periods as juveniles in freshwater are generally extinct, endangered, or threatened over a greater percentage of their ranges than species with abbreviated freshwater residence. In many cases, populations that have not declined in numbers are now composed mainly or entirely of hatchery fish, which tend to replace and otherwise threaten the gene pool of remaining wild salmon populations (NRC 1996b and references therein).
The salmon's life cycle begins in clear cold streams where salmon hatch, extends to the ocean where they grow, and returns again to natal streams where they spawn. Salmon habitats are degraded by agriculture, dams, forestry, grazing, industrialization, and urbanization, causing fish populations to decline. A recent NRC study (NRC 1996b) concluded that a strategy is needed to protect ecosystems and encourage the natural regeneration of lost salmon habitat.
Salmon productivity in the Pacific Northwest appears to be correlated with large-scale environmental changes in ocean circulation, temperature, and chemistry (NRC 1996b). These changes appear to be related to the ones that affect productivity in the Gulf of Alaska and Bering Sea and out of phase with them (NRC 1996a, 1996b, and references therein). Thus, when ocean conditions favor salmon productivity off California, Oregon, Washington, and southern British Columbia, they appear to be less favorable than average off northern British Columbia and Alaska, and vice versa. The interactions among fishing, human-caused environmental degradation in the streams where salmon spawn, and natural fluctuations in ocean climate are difficult to disentangle and complicate salmon management. Yet failure to consider natural fluctuations reduces the chances that management will be effective (NRC 1996b).
Upwelling Systems and ENSO Events
Upwelling systems have been reviewed by Everett et al. (1996). They are affected by larger-scale ocean-climate changes, such as El Niño-Southern Oscillation (ENSO) events, and changes in upwelling patterns change the distribution and abundance of many pelagic species (Bakun 1993). Upwelling is most common off west coasts of continents, as surface currents that flow towards the equator along the coasts are deflected away from them by the earth's rotation (Coriolis force). The water that flows away from the coasts as a result is replaced by colder, nutrient-rich water from below, and this upwelling supports abundant phytoplankton and hence fish production. ENSO events reduce the strength of the coastal currents, and hence the upwelling: as a result, coastal oceans off the west coasts of continents experience much warmer water than normal with fewer nutrients, less ecosystem productivity, and a variety of warm-water species that often are predators of or competitors with commercially important species. These ENSO-induced changes in upwelling patterns affect populations of small pelagic species such as sardines, pilchards, and anchovies off the coasts of Japan, California,
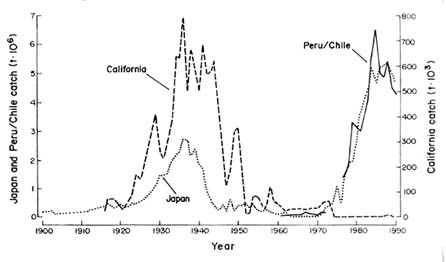
FIGURE 3-1 Record of catches of sardines in Chile and Japan, showing fluctuations, probably influenced by environmental fluctuations. Source: Redrawn from Lluch-Belda et al. (1989).
Chile, and Peru. Similar changes have been observed off west Africa (Bakun 1993).
ENSO events can have dramatic effects on fish populations. The 1972 event, combined with heavy fishing pressure, led to a major collapse of the Peruvian anchovy (Arntz 1986, Muck 1989, Sharp and McLain 1995). ENSO events, along with multiple impacts on their freshwater ecosystems, have also affected salmon fisheries off the Pacific coast of North America, depressing productivity and catches (Pearcy 1992, NRC 1996b). A strong ENSO event occurred in 1997–1998, and information on its effects on marine ecosystems and fish populations will increase our understanding.
Eutrophication
Eutrophication is a common and widespread human impact on the coastal ocean and in semienclosed seas, such as the Baltic and Mediterranean seas, often associated with other forms of pollution (Laevastu 1993). In estuaries and coastal areas, eutrophication and pollution can lead to algal blooms, including toxic dinoflagellates. Often, oxygen is depleted from benthic waters and fish kills result. However, eutrophication can increase productivity at various trophic levels, including fish. Examples of increased fish productivity attributed to eutrophication include the Baltic, where fish production increased substantially, partly because of eutrophication and partly because of reduced marine-mammal populations (Elmgren 1989, Nehring 1991). In the Mediterranean, increased
pelagic fish production (e.g., anchovies) has been attributed to eutrophication as well (Caddy and Griffiths 1990).
Multiple Impacts on Ecosystems
This chapter has described the effects of fishing and environmental fluctuations as well as the effects of some other human activities on ecosystems. Because the impacts do not act alone, we discuss here three ecosystems—Chesapeake Bay, the Laurentian Great Lakes, and San Francisco Bay—that have been subjected to numerous significant human impacts and environmental changes. Although the Great Lakes make up a freshwater system and Chesapeake Bay and San Francisco Bay are semienclosed estuaries, some people have argued that their condition represents a preview of the fate of the larger, more open marine ecosystem discussed earlier in this chapter. Kerr and Ryder (1997), for example, described a history in which people continued to believe that the next larger ecosystem was so large that it could never suffer the damage of smaller, more contained ecosystems. The Laurentian Great Lakes are the largest freshwater ecosystem in the world, yet even they have been dramatically altered by human activities. Kerr and Ryder argued that coastal marine ecosystems, in particular those off Atlantic Canada, are next in line for similar changes; eventually the open oceans will be affected as well. Although San Francisco and Chesapeake bays are much smaller, they are connected closely to the open ocean, which affects them. Changes in the biota and physical environment of San Francisco Bay also have affected large areas of California's coast, as described later in this chapter.
This committee is unable to predict whether the events described in these three ecosystems are a preview of events to come in marine ecosystems in general, but it is clear that they are analogous at least for semienclosed and some other coastal systems. It is at least possible, unless significant changes are made in the way marine ecosystems are treated, that even open-ocean systems are at risk.
Chesapeake Bay
The Chesapeake Bay is the largest estuarine system in the United States, and its 166,000-km2 watershed has supported important fisheries for centuries. Major changes in the bay ecosystem include eutrophication (from increased nutrient inputs), increased turbidity, decreased seagrass growth, and hypoxia in summer months. Fishery yields have remained surprisingly constant, or even increased, over the past 50 years, but the diversity and value of the harvest have declined as catches of many valuable species, especially oysters and anadromous fishes, have dwindled (Miller et al. 1996). Sturgeon, American shad, and two species of river herrings have declined precipitously in abundance. Seasonal migrants to the bay
(e.g., bluefish and weakfish) also have declined, probably because of overfishing on the Atlantic Coast and within the bay. Catches of eastern oysters fell dramatically in the 1980s and 1990s, the victim of overfishing, habitat destruction, and parasitic diseases (Rothschild et al. 1994) and the interactions of those factors with altered water quality (Lenihan and Peterson 1998). In the summer of 1997, there were outbreaks in some Chesapeake Bay tributaries of a predatory form of the dinoflagellate Pfiesteria piscicida, which attacks fish. Because of fears that the dinoflagellate might also harm humans, a section of the Pocomoke River, a Chesapeake Bay tributary in southern Maryland and Virginia, was temporarily closed (Pesticide and Toxic Chemical News 1997). The reasons for the dinoflagellate's transformation to a toxic phase are not known, but most of the fish kills have occurred in estuaries with elevated concentrations of nitrogen and phosphorus (Burkholder 1998).
Not all species have declined in the bay. Striped bass populations collapsed in the 1970s (Richkus et al., 1992) but were placed under a five-year fishing moratorium and then recovered spectacularly in the 1990s (Leffler, 1993), a reminder that single-species management, if pursued vigorously, can be successful for some species. Spanish mackerel populations have increased coastwide; they have become much more common in the bay over the past five years than before. Other populations have proved remarkably resistant to damage from heavy exploitation by commercial and recreational fishers, most notably blue crabs (the dominant species in terms of harvest value) and menhaden (the dominant finfish species in terms of weight of catch), although the status of both species is being carefully monitored at present.
The Chesapeake Bay situation provides important examples of the need for an integrated ecosystem-based approach to management (Lenihan and Peterson 1998) as well as attempts to implement ecosystem management. The Chesapeake Bay Program (CBP) is the largest and best coordinated effort to restore a major coastal ecosystem in the United States. The goal of this federal-state program is to plan and implement total ecosystem management (EPA 1995a), with its highest priority being restoration of the bay's living resources (EPA 1995b). One objective of the CBP is to reduce controllable nitrogen and phosphorus inputs to the bay by 40 percent. If this objective is achieved, hydrodynamic and ecosystem models indicate that summer hypoxia would decline, benthic and deepwater habitats could be reclaimed, and seagrasses might be restored (Magnien 1987), returning the system to a more desirable state.
Management and rebuilding of living resources have been identified as goals by which the CBP's success will be judged. Fishery management plans for major exploited species have been developed jointly by the states in the watershed. To date, these plans are based primarily on single species, although some include aggregates of species, each treated individually. It is generally recognized that the plans are deficient with respect to habitat requirements. Although the plans may meet CBP goals for restoration of living resources, they fall short of meeting
the program's call for ecosystem management. Multispecies management is now being explored. Whether multispecies management and ecosystem management are only distant goals or are imminently achievable are questions that the CBP will face as it attempts to reconcile the demands of multiple users with conditions required to restore a diverse, productive, and sustainable ecosystem.
The Laurentian Great Lakes
Great Lakes fisheries and the fish communities on which they are based have changed dramatically in the past 150 years. These large lakes contain one-fifth of the surface fresh water on the earth; their drainage basins are heavily developed and contain large portions of the human populations of the United States and Canada. Changes in fish communities have resulted from invasions and stocking of exotic species; overfishing; pollution; and loss or damage of habitat, especially in bays, tributaries, and shallower basins. The usefulness of fish as human food has been reduced by bioaccumulation of anthropogenic toxic substances; those chemicals have probably affected a variety of fish populations directly, especially salmonids (e.g., Mac and Gilbertson 1990, Mac et al. 1993). Yet important commercial (e.g., lake whitefish [Coregonus clupeaformis], Ebener 1997) and recreational fisheries persist or have recovered. Indeed, recreational fisheries have become very important in the Great Lakes, involving both introduced species such as Pacific salmon (Oncorhynchus spp.) and native species such as walleye (Stizostedion vitreum) (Knight 1997, Lichtkoppler 1997) and lake trout (Salvelinus namaycush) (Schreiner and Schram 1997). The management paradigm for the Great Lakes has been to take an ecosystem approach to rehabilitation of the ecological systems on which the fisheries depend (Francis et al. 1979). Even though the problems have been catastrophic at times, such as the elimination or near-elimination of fish species (Kerr and Ryder 1997), there is some positive sentiment among fishery managers, because of some successes in reversing or coping with a number of serious problems and because of the important recreational fisheries.
Sea lamprey (Petromyzon marinus), alewife (Alosa pseudoharengus), and rainbow smelt (Osmerus mordax) have invaded the Great Lakes in the past, severely affecting the native pelagic fish species. A combination of biocides to kill larval sea lamprey, fisheries for alewife and rainbow smelt, and artificial propagation of lake trout and Pacific salmon have been used to rehabilitate native populations and provide sport fisheries. Problems continue in terms of fish health (disease) and stocking at levels that overexploit the prey base, and the assemblage of fish species in the Great Lakes today is enormously different from the preindustrial assemblage. In particular, species of Pacific salmon, sea lamprey, and rainbow smelt are common, and some species have been exterminated (Becker 1983, Kerr and Ryder 1997).
Sources of input to the lakes of toxic and carcinogenic compounds have been
much reduced and in many ways the Great Lakes ecosystems are recovering. Mercury is no longer at levels that are dangerous in Lake St. Clair. Dichlorodiphenyltrichloroethane (DDT) was banned, as were polychlorinated biphenyls (PCBs), and by 1995 both had declined in all of the lakes to concentrations below which regulations require action (''action levels"). Reduced input of phosphorus was achieved from metropolitan and industrial point sources and the use of low-phosphate detergents. Phosphorus levels have declined to action levels and water quality has improved in the past 30 years. Nitrates and nitrites seem to be increasing, however, and will be more difficult to control. Eutrophication has resulted in increased algal blooms, especially in Lakes Erie and Ontario.
Perhaps the greatest problem in the Great Lakes today is the rate of arrival of undesirable exotic species, primarily through transportation in ships' ballast water, but also through accidental introductions caused by other human activities and some deliberate introductions. Among recent invaders of concern are the zebra mussel (Dreissena polymorpha), the European river ruffe (Acerina cernua), and a predaceous water flea (Bythrotrepes cederstroemi). According to Mills et al. (1993), 139 species of nonindigenous plants and animals have been introduced into the Great Lakes; at least 13 of those species have had significant ecological effects. Future invasions might be reduced by revised methods of handling ballast water (NRC 1996d), but these and other exotic species probably will spread throughout the Laurentian Great Lakes and beyond.
The Laurentian Great Lakes are a primary example of the dependence of fisheries on the entire land-water ecosystem and the direct and indirect impacts that humans have had on fisheries. Problems will continue to develop and persist. The region has a better chance of dealing with these issues and problems as they develop because of the coordinating roles played by the Great Lakes Fishery Commission and the International Joint Commission on Water Quality. However, Kerr and Ryder (1997) argued that the Great Lakes experience is an ominous portent for Canada's Atlantic Ocean fisheries, which will follow the same pathways and will be harder to remediate than Great Lakes fisheries.
San Francisco Bay
The San Francisco Bay estuary is the largest such body on the Pacific Coast of the United States. Including its delta, it encompasses more than 4,100 km2. Before 1950, the estuary contained 1,400 km2 of freshwater marshes and 800 km2 of salt marshes. The bay estuary drains 40 percent of California (Nichols et al. 1986). Runoff from the western slopes of the Sierra Nevada Mountains flows into the San Joaquin and Sacramento rivers and then into the San Francisco Bay and the Pacific Ocean. The estuary supports more than 120 species of fish and is critical habitat for migratory waterfowl (California Fish and Game Department 1998).
The estuary has undergone massive alterations through time in its physical
and biological properties. More than 95 percent of the historic tidal marshes have been leveed and filled. These alterations have significantly reduced most native fish and wildlife populations. Most of the major rivers and streams that flow into the estuary have been dammed for flood control, power generation, and water supply. These structures, plus diversion canals, have reduced the inflow into the estuary by 40 percent, thereby altering flow and sedimentation patterns and water temperatures and blocking migration pathways for salmon and steelhead and altering their spawning habitats (Nichols et al. 1986).
Hydraulic mining in the foothills of the Sierra Nevada during the gold rush, especially between 1856 and 1887, resulted in massive inputs of sediments to San Francisco Bay. Starting in about 1950 with dam construction, some of these sediments were gradually lost, but there has still been a net sediment gain over the last century in the northern portions of the bay (San Pablo Bay) of nearly 400 million m3. New input of sediments is now quite reduced because of upstream dams, and the waters are clearer than before (USGS 1998).
There continue to be large inputs of organic and inorganic chemicals into the bay. Toxic trace-metal accumulations accelerated during the 1950s. Some high accumulations of silver, cadmium, lead, and selenium are found at certain sites in the bay, which receives effluents from 46 wastewater-treatment plants and the discharges of 65 large industries. Approximately 40,000 tons of at least 65 contaminants accumulate in the bay each year. The sediments have been the repository of many organochlorine compounds, some of which bioaccumulate in the livers of striped bass and may be one cause of their declining populations (Pereira et al. 1994).
Sewage treatment plants discharge about 60 tons of nitrogen into the bay every day. Despite this input of fertilizer, the bay has not become more eutrophic because the very large populations of filter-feeding invertebrates keep phytoplankton from building up. Formerly, when ammonium nitrogen was not removed from treated sewage water, the south bay, with its poor circulation, became anoxic and fish died. Advanced sewage treatment has alleviated this problem and reduced the input of toxic metals (Cloern and Jassby 1995, Cloern 1996). Superimposed on this massive alteration of the San Francisco Bay estuary by human action are patterns of periodic major perturbations of the operation of the bay ecosystem in response to wide interannual variations in rainfall that affect salinity gradients. These perturbations are particularly evident during El Niño events (Peterson et al. 1995).
The Invaders
San Francisco Bay has been, and continues to be, considerably altered by invasive species. In recent years a new marine introduction has occurred about every 14 weeks. Many of these introductions have large impacts on the bay ecosystem. The Asian clam Potamocorbula amurensis first became established
in 1986. In two years it became commonest clam throughout the northern part of San Francisco Bay (Carlton et al. 1990). It has reached very high population densities and altered the water chemistry of the bay and hence has affected many dependent organisms. In 1989, the green crab (Carcinus maenas) invaded the bay and is now spreading throughout California's coastal waters, where its voracious appetite threatens the shellfish and crab industries in the coastal regions. At present, there are 164 known introduced species of plants, invertebrates, and fish in San Francisco Bay, many of which have displaced native species (Cohen and Carlton 1998).
The invasive species are causing direct changes in the food webs of economically important species as well as altering the physical nature of the bay as noted above. For example, the major food item for fish, including juvenile striped bass (itself an introduced species), is the zooplanktonic mysid Neomysis mercedis. Neomysis is being displaced by introduced Acanthomysis.
Changing Nature of San Francisco Bay Fisheries
As might be expected from the ecosystem disruptions noted above, fisheries in the bay have had a tumultuous history (Box 5-4). Commercial fisheries for salmon, sturgeon, sardines, flatfishes, crabs, and shrimp were established soon after the start of the gold rush to support the rapidly growing human populations. These fisheries, and others that developed later, especially for striped bass, annually provided millions of pounds of protein until changes in the estuary severely reduced them. Loss of the fisheries can be attributed to a variety of causes, including overfishing, changes in water quality, and reductions in freshwater input through the bay delta.
Shellfish
The native oyster Ostrea lurida was intensively harvested beginning in the 1850s and gradually declined in abundance. Larger oyster species were imported and cultivated, first from the Pacific Northwest, and subsequently from the east coast (Crassostrea virginica). In 1899 more than 1,100 t of oyster meat was produced in the bay. The increasing pollution in the bay resulted in a decline of production by 50 percent by 1908 and by 1921 no more attempts at cultivating oysters were made. The bay was, however, used for holding imported stock until 1939, when the industry closed. Oyster culture has been moved to cleaner bays elsewhere in California (Leet et al. 1992).
Crustaceans
Bay shrimp, an aggregate of four species with Crangon franciscorum being the primary one, have been harvested commercially from San Francisco Bay
since the early 1860s. By the 1890s, annual landings exceeded 2,300 t. Bycatch problems related to shrimping have led to fishing restrictions. Today the catch is down considerably from the peak years, and the shrimp is now sold mainly for bait (Leet et al. 1992).
Dungeness crab (Cancer magister) used to be commercially important in San Francisco Bay. Bay-area landings reached more than 3,500 t in 1956–1957 but have been less than 450 t during most years since. The fishing fleet in the bay area has been halved since 1957. The reasons for the collapse are not precisely known but probably include bay pollution, predation by nemertean worms, and changes in ocean temperature (Leet et al. 1992).
Striped Bass
A total of 432 striped bass (Morone saxatilis) from the Navesink River in New Jersey were deliberately introduced by the U.S. Bureau of Commercial Fisheries to San Francisco Bay in 1879 and 1882, along with American eels (Anguilla americanus), American lobsters (Homarus americanus), and other species (Lampman 1946). Although the lobsters and eels did not become established, the striped bass did, soon supporting major commercial and recreational fisheries. By 1899 more than 540 t of bass were commercially harvested (Skinner 1962). Striped bass have become established from approximately Monterey Bay, California, to Coos Bay, Oregon, with occasional fish being found as far south as northern Baja California, Mexico, and as far north as southern British Columbia, Canada (Hart 1973, Moyle 1976). The growing competition between recreational and commercial fisheries resulted in the banning of commercial fishing in 1935. Despite a gradual decline in striped bass populations, the sports fishery was still valued at $45 million in 1985. The populations in San Francisco Bay have continued to decline (California Fish and Game Department 1997), probably because of many factors, including water diversion, a changing food web caused by invasive species, and pollutants, although knowledgeable anglers continue to catch fish.
White Sturgeon
The commercial fishery for the longed-lived white sturgeon (Acipenser transmontanus) was quickly exhausted. During 1887, more than 680 t was caught. The catch dropped to 136 t by 1895, and the fishery was closed in 1917. Freshwater flow into the bay is apparently important for recruitment in this species as well (Leet et al. 1992).
Pacific Herring
Never a favored fish commercially in this region, Pacific herring (Clupea
pallasi) has seen large fluctuations in population sizes in recent years. Its bay population was halved during the 1983–1984 El Niño event. At present, as discussed elsewhere in this report, an interesting and important industry of herring roe-on-kelp has developed in San Francisco Bay (Box 5-4, Leet et al. 1992).
Endangered Fish Species
Several fishes are endangered in the San Francisco Bay estuary. Populations of the delta smelt (Hypomesus transpacificus), found only in the estuary, plummeted in the 1980s. It was listed as threatened in 1991. The southernmost spawning populations of chinook salmon (Oncorhynchus tshawytscha) occur in the estuary. The winter runs of this species were listed as endangered in 1990. Management of these threatened species, within a complex ecosystem, were in part the motivation for the joint California-federal program noted below (Leet et al. 1992).
A New Start
As a result of management conflicts in protecting endangered species, meeting water-quality standards, and providing irrigation water through the Central Valley Project Improvement Act, a landmark framework agreement was formalized in 1994 to establish a state-federal cooperative agreement (CALFED) to provide solutions to the San Francisco Bay delta-estuary problems. In the long term this agreement would address the health of fish and wildlife as well as water-supply reliability and quality. An ecosystem-restoration plan is part of the program, which would work on "unscreened water diversions, waste discharges and water pollution prevention, fishery impacts due to harvest and poaching, land derived salts, exotic species, fish barriers, channel alterations, loss of riparian wetlands and other causes of estuarine habitat degradation"—in essence, an ecosystem approach to balancing various demands on the estuary. In addition to the federal and state agencies, a 30-citizen advisory group representing agriculture, environmental organizations, urban considerations, business, and fishing will be involved in fulfilling the goals of the framework agreement. This complex decision process is well justified to address this exceedingly complex management issue.
Conclusions
Fishing has had substantial ecosystem effects in most estuarine, coastal, semienclosed, and continental-shelf marine waters. It is likely that these effects are larger than the data suggest, for two reasons. First, in many cases the largest effects occurred before the ecosystems were carefully studied. Second, information is often lacking, even for well-studied systems. In addition, it is likely that in
some systems the ecosystem effects of fishing have not fully developed or worked their way through the ecosystem. Although there is little information on ecosystem effects of fishing in the open ocean, those ecosystems that have been affected by fishing—estuarine, coastal, semienclosed, and continental-shelf ecosystems—provide most of the world's fishery products and many other services.
In addition to fishing, anthropogenic impacts and environmental changes continue to occur and are important. Anthropogenic impacts, which include contamination with toxic and other chemicals, habitat alteration and destruction, and introduction of exotic species, can be identified and in some cases managed. Most natural environmental changes cannot be managed in the traditional sense. Indeed, many are not even predictable in a precise way. However, they are becoming better understood in a general way and must be taken into account in any sensible management program.




























