3
Toxicokinetics of Military Fuels
It is not possible to fully describe the toxicokinetics of the individual volatile hydrocarbons that are present in JP-5, JP-8, and diesel fuel marine (DFM). However, toxicoldnetic data are available for the most toxic chemicals in each of the major classes of these fuels. For example, the n-paraffin group includes n-hexane, a human neurotoxicant. In the branched paraffin group, the chemical 2,2,4-trimethylpentane is thought to be responsible for the development of kidney-specific nephropathy and carcinogenicity in gasoline-exposed male rats. The aromatic group contains benzene (a human hematotoxicant and leukemogen) as well as toluene and the o-, m-, and p-xylenes. Finally, methoxyethanol, an important deicing additive in fuels, is a known developmental and reproductive toxicant. Thus, one of the major challenges in describing the behavior of the various hydrocarbons in these fuels is predicting the effect of other present hydrocarbons on the toxicokinetics of each highly toxic chemical.
GENERAL DETERMINANTS OF HYDROCARBON TOXICOKINETICS
Because of the great diversity in the biochemical and physical properties of the numerous volatile organic chemicals (VOCs) found in JP-5, JP-8, and DFM, it is useful to examine, in a general way, the factors important for uptake, distribution, metabolism, and elimination of hydrocarbons. Dispositional determinants include, but are not limited to, those of a physiological nature, such as alveolar ventilation, cardiac output, blood flow to organs, and organ volumes. Chemical determinants of disposition include partition coefficients, which describe the distribution of the VOC between blood and air or between blood and tissue at equilibrium. Metabolic determinants, including the capacity for metabolism and the enzyme affinity for the hydrocarbon, are critical in regulating the disposition of VOCs. Binding of the hydrocarbon or metabolite to tissue macromolecules might also be important because, in some cases, binding to a critical macromolecule determines toxicity. Determinants for metabolism or binding can be modified by the exposure history of an individual, resulting in either inhibition or induction of metabolism or binding. Alternatively, toxicoldnetic determinants can be modified by simultaneous exposure to other hydrocarbons. Because JP-5, JP-8, and DFM are complex mixtures of VOCs, it is most likely that this final factor, simultaneous exposure to other VOCs, is the most important determinant in understanding hydrocarbon disposition following inhalation of these mixtures.
Partitioning of VOCs between biological media is a key chemical factor in describing and predicting disposition and tissue dosimetry of hydrocarbons. Partition coefficients are a measure of the affinity of the VOC for one medium compared with another at equilibrium. Partition coefficients for VOCs are often measured in vitro by using methods such as the vial-equilibration method (Gargas et al., 1989). Partition coefficients for some of
the most highly toxic components of the three fuels are presented in Table 3-1. Partition coefficients are a useful indicator of the tissues in the body that will achieve the highest concentration of the chemical following exposure. For example, for most VOC hydrocarbons, the fat/air and fat/blood partition coefficients are large compared with partition coefficients for other tissues. Experimental studies have demonstrated that fat typically contains the highest concentrations of these chemicals following exposure. Similarly, the blood/air partition coefficient is a critical determinant in the uptake of VOCs into the arterial blood. At equilibrium, assuming no clearance of the chemical from the blood except by exhalation, the arterial blood concentration of a VOC is the product of the inhaled concentration and the blood/air partition coefficient. The net effect of this relationship is that chemicals with a high affinity or solubility in blood (a large blood/air partition coefficient) will be absorbed to a greater extent than chemicals with a low solubility.
TABLE 3-1 Partition Coefficients for Some Volatile Hydrocarbons Found in JP-5, JP-8, and DFMa
The fat compartment is important in accumulating hydrocarbons during exposure and in temporary sequestering of VOCs after exposure. After exposure, the net afflux of chemicals is from the fatty tissue to the blood. Thus, chemicals become available for distribution by systemic circulation to such organs as the liver, which can metabolize them. For hydrocarbons with large fat/air partition coefficients, metabolism might be quantitatively more important after exposure than during exposure. Thus, removal from the exposure atmosphere does not necessarily end systemic exposure to those hydrocarbons.
Metabolic determinants, such as Vmax (maximum metabolic capacity) and KM (affinity of the enzyme for a substrate), are critical in describing tissue dosimetry of toxic chemical species following exposure to hydrocarbons that are metabolized. Metabolism can be a significant clearance mechanism; yet for many VOCs, metabolic transformation of the parent results in compounds with increased toxicity. For many carcinogenic chemicals, such as benzene and trimethylpentane, for neurotoxicants, such as n-hexane, and for developmental toxicants, such as methoxyethanol, metabolic activation is critical for toxicity.
The first step in the metabolism of most VOCs is oxidation of the lipophilic chemical to an oxidized or more-water-soluble chemical. The cytochrome-P-450-dependent monooxygenase system is central to the oxidative metabolism of many chemicals. Cytochrome-P-450-dependent reactions cover a broad spectrum of chemicals, but the major oxidative reactions catalyzed by this enzyme are aliphatic hydroxylation, aromatic hydroxylation, epoxidation, n-dealkylation, o-dealkylation, deamination, sulfoxidation, and n-oxidation. Alcohol and aldehyde dehydrogenases are also important in the metabolism of many organic alcohols. Methoxyethanol, the glycol ether added to fuels as a deicer, is an excellent substrate for these enzymes. Oxidation is often followed by a phase II reaction, which conjugates the oxidized hydrocarbon to a water-soluble substrate, such as sulfate or glucuronic acid.
TOXICOKINETICS OF SPECIFIC MILITARY FUEL HYDROCARBONS
Based on either large concentrations in the fuel or documented adverse health effects in humans and experimental animals, some of the important components of JP-5, JP-8, and DFM are benzene, n-hexane, toluene, xylene, trimethylpentane, and methoxyethanol. Extensive research and numerous reviews of the toxicokinetics of each of those chemicals have been published (see Andrews and Snyder, 1991). Only the key features in the toxicokinetics and metabolism of those chemicals will be reviewed in this report.
Benzene
The major toxic effect of benzene is hematopoietic toxicity. That effect is specific to benzene within the class of simple aromatic hydrocarbons. Chronic exposure of humans to benzene is associated with blood disorders, such as aplastic anemia and leukemia. To understand the mechanisms of benzene toxicity, it is essential to study its disposition. Parke and Williams (1953) were among the first to suggest that a metabolite of benzene is responsible for its toxicity. Using 14C-labeled benzene, they identified phenol, catechol, hydroquinone, and 1,2,4-trihydroxybenzene as ethereal sulfates and glucuronide conjugates in the urine of benzene-treated animals. Other metabolites were l-phenyl mercapturic acid and trans,trans-muconic acid. A substantial portion of inhaled benzene is exhaled unchanged or stored temporarily in the fat of animals and humans.
The dosimetry of benzene and its metabolites in the target tissue of bone marrow depends on the balance between activation processes, such as cytochrome-P-450-mediated enzymatic oxidation, and deactivation processes, such as conjugation and excretion. Enzymes implicated in the metabolic activation of benzene
and its metabolites include cytochrome P-450 monooxygenases and myeloperoxidase. The multiplicity of benzene's metabolic pathways provides opportunities for modulation of benzene metabolism either by competition with other VOCs for the available enzyme sites, by induction or inhibition of the oxidation or conjugation enzymes, or by direct competition with its own metabolites. Several studies have demonstrated that modification of benzene biotransformation results in alterations in benzene toxicity (Andrews et al., 1977; Gad-El-Karim et al., 1984).
Alkylbenzenes
The alkylbenzenes are single-ring aromatic hydrocarbons containing one or more aliphatic side chains. Of the alkylbenzenes, toluene (methylbenzene) and the three xylenes (o-, m-, and p-xylenes) are found in the highest percentages in military fuels. The acute toxicity of inhaled alkylbenzenes is depression of the central nervous system (CNS). The mechanism of action of the alkylbenzenes under conditions of acute exposure resembles that of general anesthetics. The major metabolic pathways of the alkylbenzenes form metabolites that have a low order of toxicity and are readily excreted. Thus, toluene is oxidized at the methyl group and a series of oxidations lead to formation of benzoic acid, which is conjugated with glycine to form hippuric acid, which is then excreted. Hippuric acids are also metabolites of xylene. There is no evidence at the present time to indicate that these metabolic pathways can lead to formation of toxic-reactive intermediates and subsequent toxic or mutagenic effects (Andrews and Snyder, 1991).
Methoxyethanol
Ethylene glycol monomethyl ether (methoxyethanol) can be
metabolized by liver alcohol and aldehyde dehydrogenases to form methoxyacetic acid, which is the major metabolite. Methoxyethanol can also be metabolized by the cytochrome-P-450-dependent o-demethylase to form ethylene glycol. Methoxyacetic acid is the metabolite responsible for teratogenic effects of methoxyethanol. It has been shown to produce the same toxic effects of methoxyethanol on the testes of male rats. Therefore, the formation of the ethylene glycol metabolite through o-demethylation can be viewed as a detoxification pathway, as can conjugation of methoxyacetic acid with glycine. Conversely, the oxidation to the acid metabolite via the liver dehydrogenases can be viewed as a toxification reaction because the acid metabolite is responsible for methoxyethanol toxicity (Miller et al., 1982).
n-Hexane
Excessive exposure to n-hexane can result in peripheral neuropathies, which are best described as sensory-motor or motor polyneuropathies. The initial symptoms are symmetric sensory numbness and paresthesias of distal portions of the extremities. n-Hexane is biotransformed to 2-hexanol and further to 2,5-hexanediol by cytochrome-P-450 mixed-function oxidases via omega-1 oxidation. 2,5-Hexanediol can be further oxidized to 2,5-hexanedione, the neurotoxic metabolite of n-hexane in humans. Through the omega-1 oxidation reaction, 2,5-hexanone can also be biotransformed to 2,5-hexanedione. Identification of 2,5-hexanedione as the major neurotoxic metabolite of n-hexane and 2-hexanone occurred soon after its identification as a urinary metabolite. 2,5-Hexanedione has been found to produce polyneuropathy indistinguishable from that of n-hexane and 2-hexanone in experimental animals and is many times more potent than either parent compound (Andrews and Snyder, 1991).
Trimethylpentane
Exposure to unleaded gasoline has been shown to cause a low incidence of kidney tumors in male rats. Upon acute exposure, there is a toxic response, which has been referred to as protein-droplet nephropathy characterized by an excessive accumulation of protein droplets in lysosomes of kidney proximal tubule epithelial cells. The chemical-induced protein droplets that accumulate in male rat kidney proximal tubule cells have been shown to contain the low-molecular-weight protein, α2u-globulin. α2u-Globulin is synthesized and secreted at a rapid rate from the liver into the blood of male, but not female, rats. Approximately 50% of the protein filtered by the kidneys is absorbed by the proximal tubule cells. When the α2u-globulin is bound to certain chemicals, the rate of its hydrolysis in the kidney is decreased. For example, both protein-droplet accumulation and increased cell proliferation, specifically localized in the P2 segment of the proximal tubules, have been observed in male rats exposed to 2,2,4-trimethylpentane, a component of jet fuels and unleaded gasoline. Trimethylpentane undergoes oxidative metabolism leading to trimethyl branched pentanols, pentanoic acids, and hydroxypentanoic acids. 2,2,4-Trimethylpentanol is the major metabolite present in male rat kidney but is absent in the female rat kidney. 2,4,4-Trimethyl-2-pentanol is the metabolite of trimethylpentane that binds to α2u-globulin (Borghoff et al., 1990). Formation of a 2,4,4-trimethyl-2-pentanol–α2u-globulin complex renders the α2u-globulin less accessible to lysosomal degradation.
Metabolic activation of trimethylpentane is an important element in the development of nephrotoxicity. For example, modulation of cytochrome-P-450 metabolism also modulates the in vivo handling of trimethylpentane, suggesting that trimethylpentane undergoes cytochrome-P-450-mediated metabolism in the male rat. Therefore, the selected retention of the hydrocarbon-protein complex in the kidney of male rats results from the pro
duction of a bioactivated or reactive metabolite of trimethylpentane. That phenomenon does not occur in humans.
Chemical Mixtures
The behavior of chemicals in mixtures can differ greatly from that observed when compounds are tested as pure chemicals. For example, enzymatic metabolism of one component of the chemical mixture can be increased when a second component in that mixture induces the enzyme responsible for its metabolism. Alcohol is an inducer of cytochrome P-450, and a number of studies have shown that exposure to alcohol can increase the metabolism of a number of hydrocarbons (Ryan et al., 1986). In contrast, if fuel components are substrates for the same enzyme, each chemical can inhibit or reduce the metabolism of the other. If a metabolite of the chemical is the toxic species rather than the chemical itself, the interaction can result in decreased toxicity. Benzenetoluene mixtures are examples of that type of interaction (Andrews et al., 1977). Toluene inhibits the metabolism of benzene (Purcell et al., 1990) and reduces toxic effects such as bonemarrow and blood-cell damage. Because many of the hydrocarbons in fuels are substrates for the same cytochrome-P-450 enzymes, there are numerous opportunities for those types of chemical interactions.
Extrapolation issues of particular concern for chemical interactions include in-vitro-to-in-vivo, high-to-low-dose, and animal-to-human extrapolation. In the case of in-vitro-to-in-vivo extrapolations, many chemical interactions can be tested rapidly and with fewer animals when in vitro systems are used. However, interactions found in vitro might not be relevant to in vivo situations. For example, although chemicals can induce metabolizing enzymes in vitro, an increased amount of metabolism will not always be observed in vivo. Chemical interactions might occur at high-exposure concentrations when enzymes are saturated.
However, those interactions might not be significant at low-exposure concentrations when metabolizing enzymes are not operating at their full capacity. Most chemical interactions are demonstrated in animal studies; however, specific mechanisms of action important in laboratory animals might not be relevant to or operating in humans.
SIMULATED TISSUE CONCENTRATIONS AFTER EXPOSURE TO COMPONENTS OF MILITARY FUELS
To aid the subcommittee in understanding the relationships between vapor concentrations and the accumulation of fuel components in tissue and blood, physiologically based toxicokinetic models were used to simulate the uptake and metabolism of two components of the fuel vapors, toluene and n-hexane. Those chemicals were chosen because they represent the range of volatilities of VOCs (see Table 3-1). n-Hexane is a low-solubility aliphatic VOC, and toluene is a soluble aromatic VOC. The simulations are reported in Appendix A. The results indicate that CNS effects are most likely to be related to the short-term-exposure-limit (STEL) type of exposure and that other toxic effects are more closely related to the PEL type of exposure.
It is not clear whether the potential toxic effects, such as the neurobehavioral effects noted in the human epidemiology studies of Knave et al. (1978) (see Chapter 7), of exposure to fuel vapors are due to the peak concentrations of fuel components in blood and nerve tissue or to a total exposure as measured by an area-under-the-concentration-time curve (AUC). The anesthetic effects are not related to the total anesthetic but to the rate of delivery to the CNS. Those having high partition coefficients require more anesthetic to saturate the blood and are slower at producing anesthesia. For anesthesia, the peak blood concentra
tion is the key to induction. The duration of anesthesia relates to maintaining an anesthetic concentration over time. When the CNS and blood concentrations fall below the minimum concentrations needed for anesthesia, the patient wakes up. In the case of high-partition-coefficient gases, there might be considerable amounts of residual anesthetic remaining in the patient that are not fully exhaled for several days. To gain some insight into the effect of the exposure profile and the potential for accumulation of fuel components in the tissues of individuals repeatedly exposed to fuels in the workplace at the current recommended maximum levels, the simulations were conducted. Physiological and biochemical parameters and equations used in the simulations are given in the model code in Appendix A.
The first simulation was of 10 daily, 15-min exposures to the binary mixture of n-hexane at 900 mg/m3 and toluene at 900 mg/m3 for a combined exposure of 1,800 mg/m3, which is the current recommended STEL. The time courses for simulated concentrations of n-hexane and toluene in the blood and fat of a 70-kg man during and after these simulated exposures are shown Figure 3-1 Figure 3-2 Figure 3-3 through Figure 3-4. Simulated concentrations of both n-hexane and toluene rise rapidly in blood on initiation of exposure and decline rapidly in a biphasic pattern on cessation of the 15-min exposure. Of special note is the fact that the blood concentrations of both chemicals just before the start of the next day's exposure continue to rise with repeated exposure, suggesting that all the chemical is not cleared from the body before the start of a new day's exposure (Figure 3-1).
The accumulation of n-hexane and toluene after repeated exposure is even more apparent in fat (Figure 3-2). Peak fat concentrations of both chemicals are two times higher after 10 days of exposure than after the first day. The model also predicts that fat concentrations continue to rise for a short time even after the end of the 15-min exposure. The continued rise is the result of the higher solubility of toluene and n-hexane in fat compared with blood; the higher solubility in fat causes the chemicals to partition
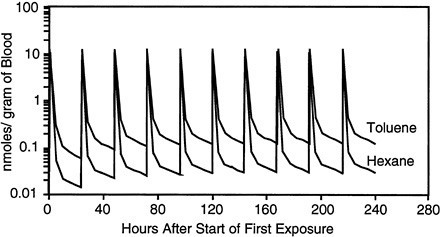
FIGURE 3-1 Predicted blood concentrations after repeated exposure to n-hexane or toluene. Simulated exposures were for 15 min per day for 10 days to binary mixtures of each chemical at 900 mg/m3. The lines represent the simulated time course for each chemical in blood.

FIGURE 3-2 Predicted fat concentrations after repeated exposure to n-hexane or toluene. Simulated exposures were for 15 min per day for 10 days to binary mixtures of each chemical at 900 mg/m3. The lines represent the simulated time course for each chemical in fat.
into the fat from the blood shortly after exposure. Even when exposure concentrations are identical, concentrations of toluene in fat and blood are higher than those of n-hexane because toluene is more soluble than n-hexane in body tissues and fluids (see Table 3-1).
Simulations of repeated exposure to the binary mixture for 8 hr at the lower time-weighted-average (TWA) concentration produced qualitatively similar results to those obtained after simulation of a STEL exposure (Figure 3-3 and Figure 3-4). The TWA simulations of simultaneous exposure to 150 mg/m3 of toluene and 150 mg/m3 of n-hexane were for 8 hr per day for 10 days. As noted in Figure 3-3, steady-state blood concentrations of n-hexane, the less-soluble organic vapor, were achieved during the 8-hr period. That was not the case for toluene, the more-soluble organic vapor. Blood concentrations of toluene did not reach steady-state even after 8 hr of simulated exposure. Similar to what was predicted in the shorter simulations, blood concentrations of each chemical continued to rise with repeated exposure, again suggesting the possibility for some accumulation of these VOCs in the body.
Simulations of the concentration of the VOC compounds in the fat predicted that the concentration would rise steadily during the 8-hr exposure. Sixteen hours after the 8-hr exposure and before the start of the next exposure, the concentrations of both chemicals in fat declined; however, there was still a substantial amount of the chemicals stored in the fat at the start of the next exposure. The net result was similar to that predicted in the STEL exposure scenarios—both VOCs accumulated in the fat over the 10-day simulated exposure period.
Because military fuels are a mixture of hundreds of VOCs and not just a simple binary mixture of an aliphatic and aromatic hydrocarbon, the concentration of any single VOC in tissues will be much lower than the concentrations obtained in the model simulations of two representative VOCs. Thus, the magnitude of the concentrations obtained in the model simulations cannot be used to predict tissue concentrations resulting directly from exposure
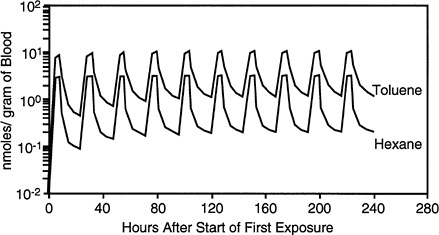
FIGURE 3-3 Predicted blood concentrations after repeated exposure to n-hexane or toluene. Simulated exposures were for 8 hr per day for 10 days to binary mixtures of each chemical at 150 mg/m3. The lines represent the simulated time course for each chemical in blood.
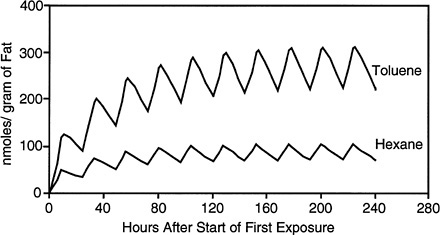
FIGURE 3-4 Predicted fat concentrations after repeated exposure to n-hexane or toluene. Simulated exposures were for 8 hr per day for 10 days to binary mixtures of each chemical at 150 mg/m3. The lines represent the simulated time course for each chemical in fat.
to military fuels. However, the rate of clearance of the VOCs from the tissues of exposed people will most likely be within the range observed for n-hexane and toluene. Thus, it is reasonable to assume that, on repeated exposure to these fuels, fuel components will accumulate in the fatty tissues of exposed individuals and that the body burden of the chemicals will continue to rise over an extended exposure period.
The simulations also attempted to address the effect of the exposure pattern (short-term high-concentration (STEL) exposures vs. long-term low-concentration (TWA) exposures) on exposure metrics, such as the peak blood or tissue concentration of the chemical. Figure 3-5 Figure 3-6 Figure 3-7 through Figure 3-8 compare simulations for the blood and fat concentrations of toluene and n-hexane achieved after repeated exposures using STEL and TWA exposure profiles. The most interesting and potentially significant model predictions are the different peak concentrations of n-hexane and toluene

FIGURE 3-5 Predicted blood concentrations after repeated exposure to n-hexane. Simulated exposures were for 8 hr per day for 10 days to binary mixtures of n-hexane and toluene at 150 mg/m3 (TWA) or for 15 min per day for 10 days to binary mixtures of each chemical at 900 mg/m3 (STEL).
achieved in blood (Figure 3-5 and Figure 3-6) and fat (Figure 3-7 and Figure 3-8) after a STEL exposure and a TWA exposure. As noted in Figure 3-5 and Figure 3-6, higher blood concentrations of both n-hexane and toluene are predicted following STEL exposures compared with TWA exposures. The exact opposite exposure pattern is noted for fat (Figure 3-7 and Figure 3-8). The model predicts that both the peak and AUC dose in fat will be greater for the TWA exposure than for the STEL exposure.
Although Knave et al. (1978) did not evaluate directly whether the acute effects of solvent exposure in the Swedish Air Force personnel were observed in conjunction with work assignments for which fuel-vapor measurements were particularly high, they believed that peak exposures were more important than the TWA for acute symptoms of hydrocarbon exposures. This observation supports the possibility that STEL is a more important exposure determinant and that nerve tissue has a kinetic profile similar to
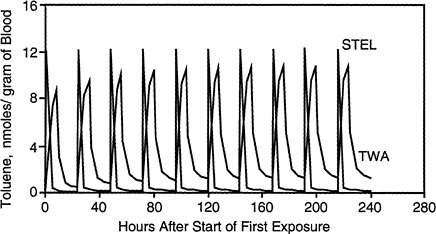
FIGURE 3-6 Predicted blood concentrations after repeated exposure to toluene. Simulated exposures were for 8 hr per day for 10 days to binary mixtures of n-hexane and toluene at 150 mg/m3 (TWA) or for 15 min per day for 10 days to binary mixtures of each chemical at 900 mg/m3 (STEL).
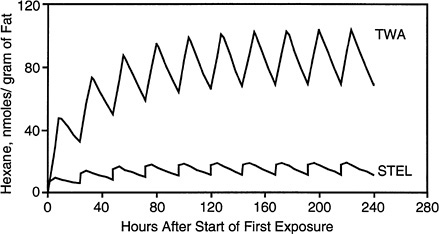
FIGURE 3-7 Predicted fat concentrations after repeated exposure to n-hexane. Simulated exposures were for 8 hr per day for 10 days to binary mixtures of n-hexane and toluene at 150 mg/m3 (TWA) or for 15 min per day for 10 days to binary mixtures of each chemical at 900 mg/m3 (STEL).
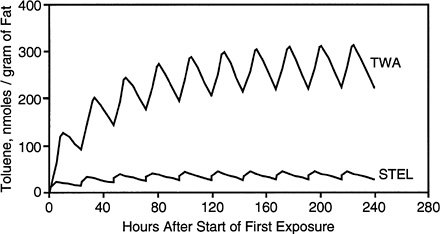
FIGURE 3-8 Predicted fat concentrations after repeated exposure to toluene. Simulated exposures were for 8 hr per day for 10 days to binary mixtures of n-hexane and toluene at 150 mg/m3 (TWA) or for 15 min per day for 10 days to binary mixtures of each chemical at 900 mg/m3 (STEL).
blood. If that is the case, as indicated in Figure 3-5 and Figure 3-6, the greater the partitioning of the individual fuel component into blood and tissues, the lower the differential in the peak blood concentrations due to a STEL or TWA exposure. For example, the differential is much greater for n-hexane, a VOC with limited solubility, than for toluene, a VOC with greater solubility. These important issues can be resolved only with additional experimental data.


















