







































Below is the uncorrected machine-read text of this chapter, intended to provide our own search engines and external engines with highly rich, chapter-representative searchable text of each book. Because it is UNCORRECTED material, please consider the following text as a useful but insufficient proxy for the authoritative book pages.
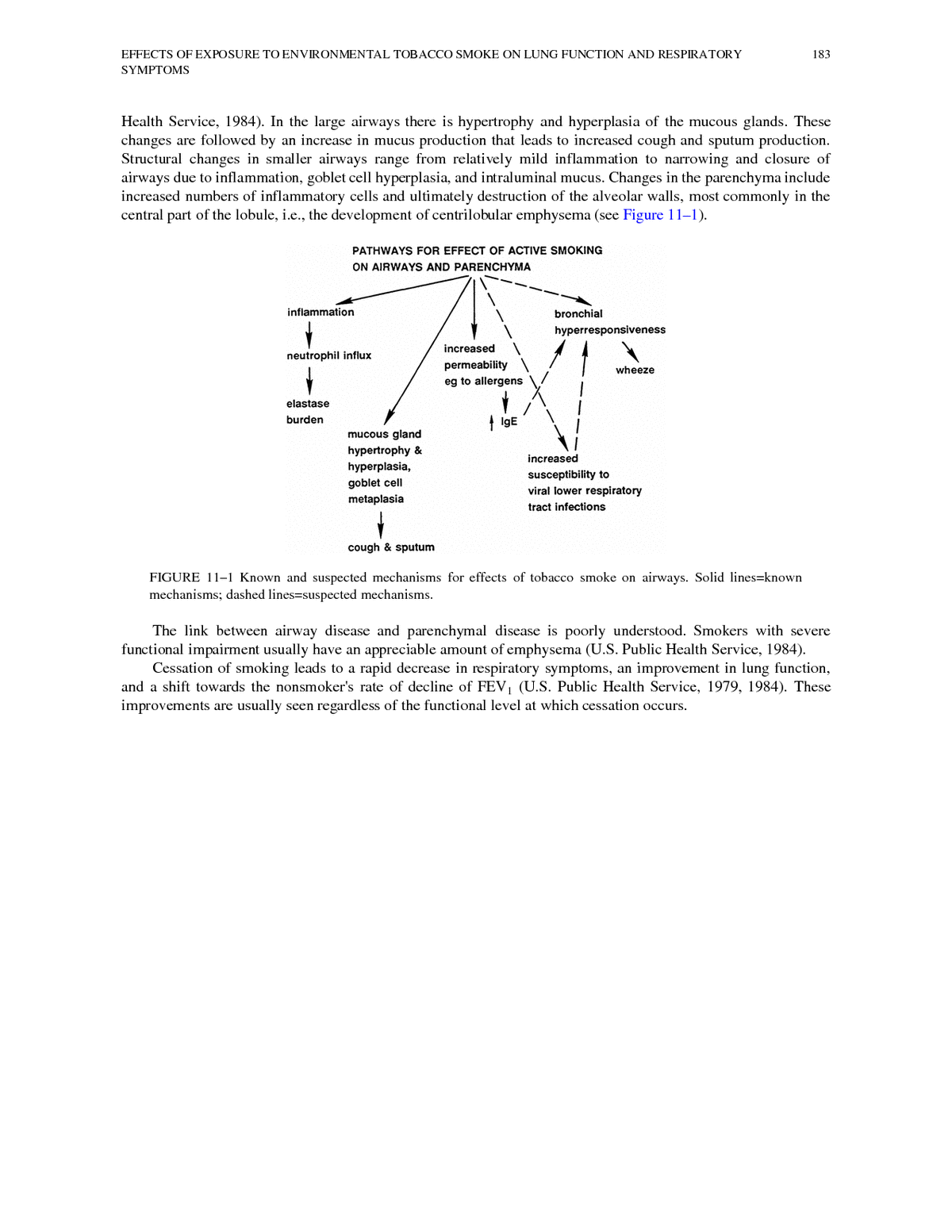
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 182 SYMPTOMS 11 Effects of Exposure to Environmental Tobacco Smoke on Lung Function and Respiratory Symptoms This chapter discusses epidemiologic studies of nonsmokers exposed to tobacco product smoke that have evaluated lung function or respiratory symptoms, most of which have evaluated children. The effects of active cigarette smoking are briefly reviewed to recount the reasons why certain aspects of lung function have been studied in nonsmokers. The plausibility of finding similar effects in nonsmokers exposed to ETS is discussed and the studies found in the literature are assessed. LUNG FUNCTION AND SYMPTOMS IN ACTIVE SMOKERS Cross-sectional studies of smokers have demonstrated that smokers, compared with nonsmokers, have (1) an increased prevalence of chronic cough, chronic sputum production, and wheezing and (2) decreased lung function (see U.S. Public Health Service, 1984, for an extensive review). The effects of smoking on both respiratory symptoms and lung function may be seen within a few years of the onset of regular smoking (U.S. Public Health Service, 1979, 1984; Woolcock et al., 1984). Longitudinal studies have demonstrated that the mean rate of decline with age of the 1-second forced expiratory volume (FEV1) is greater in smokers than in nonsmokers. In some smokers, the rate of decline of FEV1 is rapid, leading to clinically important chronic airflow obstruction. The structural changes associated with active cigarette smoking are seen in both the conducting airways and the pulmonary parenchyma (for a more detailed description, see U.S. Public
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 183 SYMPTOMS Health Service, 1984). In the large airways there is hypertrophy and hyperplasia of the mucous glands. These changes are followed by an increase in mucus production that leads to increased cough and sputum production. Structural changes in smaller airways range from relatively mild inflammation to narrowing and closure of airways due to inflammation, goblet cell hyperplasia, and intraluminal mucus. Changes in the parenchyma include increased numbers of inflammatory cells and ultimately destruction of the alveolar walls, most commonly in the central part of the lobule, i.e., the development of centrilobular emphysema (see Figure 11â1). FIGURE 11â1 Known and suspected mechanisms for effects of tobacco smoke on airways. Solid lines=known mechanisms; dashed lines=suspected mechanisms. The link between airway disease and parenchymal disease is poorly understood. Smokers with severe functional impairment usually have an appreciable amount of emphysema (U.S. Public Health Service, 1984). Cessation of smoking leads to a rapid decrease in respiratory symptoms, an improvement in lung function, and a shift towards the nonsmoker's rate of decline of FEV1 (U.S. Public Health Service, 1979, 1984). These improvements are usually seen regardless of the functional level at which cessation occurs.
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 184 SYMPTOMS Population-based studies of adults have generally shown a strong dose-response relationship between FEV1 with dose measured either in terms of years smoked, the number of cigarettes per day, or the integrated dose, i.e., pack-years (U.S. Public Health Service, 1984). It is worthwhile noting, however, that in two major studies (Burrows et al., 1977a; Beck et al., 1981) the active smoking dose accounted for only about 15% of the variation of FEV1 even after age and height adjustment. Most of the variance could be attributed to the naturally occurring large variability in pulmonary function. Another reason the active smoking dose did not explain much of the variance is that the number of cigarettes an individual smokes cannot readily be translated into the dose of smoke that is delivered into the airways and parenchyma. Many factors, such as puff volume and lung volume at which inhalation starts, clearance rates, and airway geometry of the lungs of exposed individuals, will influence the dose and the distribution of the smoke within the lungs. Variability in individual susceptibility to the effects of chemicals deposited in the lung has been demonstrated in studies of animals (Evans et al. 1971, 1975, 1978). PLAUSIBILITY FOR AN EFFECT DUE TO PASSIVE SMOKING The dose of cigarette smoke delivered to the lungs of nonsmokers exposed to ETS is both qualitatively and quantitatively different from mainstream smoke, being a small fraction of that delivered to the lungs of an active smoker (see discussions in Chapter 7). Exposure to constituents of tobacco smoke may begin in utero and continue throughout childhood through ETS exposure. During these periods, the lung is undergoing both growth and remodeling. Therefore, the lung of the fetus and young child may be particularly susceptible to environmental insults. Despite qualitative differences between mainstream smoke, sidestream smoke, and ETS, it has been customary to assume that exposure to ETS approximates a low-dose exposure to tobacco smoke. The ability to measure responses to low doses depends on the shape of the dose-response curves, the sensitivity and specificity of the measurement tools available, and whether there is a threshold of exposure below which there is no response in any individual.
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 185 SYMPTOMS The assumed shape of the dose-response curve determines what kinds of effects would be expected and the estimates of the probability of detecting them. If the dose-response curve were linear with a shallow slope, or a slope concave to the dose axis, the response at low doses might be so small that it would be difficult to detect. In such a situation, only the very susceptible portion of the population might have detectable effects. It is likely that there is a distribution of susceptibility to the effects of ETS within the population, such that there will be some persons who will respond at low doses and some persons for whom many years of heavy exposure may be needed to cause the same symptoms or change in lung function (Cockcroft et al., 1983). If individuals who are most susceptible to the irritating effects of cigarette smoke on the lower respiratory tract do not start to smoke or, having started, soon quit as smokers, then a population of nonsmokers would be more likely to include the most susceptible individuals than a population of smokers. The existence of different subpopulations introduces an additional complication to the extrapolation from high-dose exposure in active smokers to the low-dose exposures of nonsmokers. In addition, it is likely that the development of respiratory disease or symptoms, lung function level, and rate of decline reflect the cumulative burden of many environmental exposures and other insults, such as respiratory infections (Purvis and Ehrlich, 1963) to the lung. Furthermore, it might be hypothesized that the cumulative burden may interact with the individual's genetically determined susceptibility. METHODOLOGIC CONSIDERATIONS FOR EPIDEMIOLOGIC STUDIES A recent report of the National Research Council (1985) is devoted to methodologic issues of epidemiology and air pollution. In this section, many of the problems are reviewed briefly. Study Design and Analysis Chronic pulmonary effects of ETS have been the subject of several recent reviews (Lee, 1982; Weiss et al., 1983; Surgeon General, 1984; Guyatt and Newhouse, 1985; Taylor et al., 1985) and symposium or workshop reports (U.S. Public Health Service,
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 186 SYMPTOMS 1983; Gammage and Kaye, 1984; Rylander, 1984). Many of the studies reported in these reviews had not been originally designed to study chronic pulmonary effects of ETS exposure. Instead, these data sets were reanalyzed to address the question of the pulmonary effects of ETS. This use of these studies suggests the need for caution when interpreting their results. Several analytic approaches were used in the reported studies. Independent risk factors, such as age and sex, usually need to be taken into account, but this was not always done. Several statistical approaches, such as stratification or regression analysis, are used to take into account the effects of potentially confounding variables. For most of the potentially confounding variables, researchers do not agree on the nature of the roles of the variables as confounders and, hence, on the appropriate ways to introduce these variables into the data analyses. Assessing Exposure Interpretation of epidemiological studies is hampered by the existence of factors that interact with and modify the response to exposure and by confounding factors that are associated with the same symptom complex as exposure to ETS, such as coughing, production of sputum, and wheezing (see Table 11â1). These variables must be assessed and accounted for in the statistical analyses where possible. Unreported active smoking could lead to a large bias. Underreporting of smoking is likely in studies of older children, particularly when parents answer questionnaires for their children. Children who have parents who smoke are themselves more likely to smoke. Therefore, because active smoking is likely to have a considerably greater impact on respiratory symptoms and lung function than exposure to ETS, misclassification of the children who smoke will tend to overestimate the effect of exposure to ETS. For blue collar males, occupational exposure can also be important and may interact with both direct cigarette smoke and ETS. Many pulmonary toxicants can exist in the workplace. Furthermore, ETS exposure can occur in the workplace. Similarly, comparison of inner-city-dwelling persons with less urban, or sub-urban, controls can lead to biases.
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 187 SYMPTOMS TABLE 11â1 Potentially Confounding and Effect Modifying Factors in Epidemiologic Studies of Exposure to Environmental Tobacco Smoke Unreported active smoking Tobacco products Marijuana Clove cigarettes Developmental factors Maternal smoking during pregnancy Factors related to outdoor environment Outdoor temperature, humidity Respirable and nonrespirable particulates, e.g., fugitive dust Pollens and other allergens Factors related to indoor environment Crowding Number and age of siblings Total number of people/animals in dwelling unit Total number of smokers in dwelling unit Household conditions Frequency of air exchanges Temperature and humidity Use and condition of air conditioning units Conditions of child care facilities Unvented combustion products from heating/cooking stoves Respirable and nonrespirable particulates, e.g., wood smokes Pollens, molds, mites Allergens and infectious organisms Formaldehyde Factors related to work/hobbies Work/hobby-related exposure to gases, fumes, particulates Miscellaneous factors Annoyance response to tobacco smoking Reporting biases Assessing Respiratory Variables Methods commonly used to assess the effect of passive smoking on the respiratory system, such as respiratory symptom questionnaires and measurement of lung function, may lead to some error. The problems associated with the respiratory symptom questionnaires include: ⢠Different questionnaires are used in studies. Differences in how the questions are asked can sometimes lead to large differences in answers. For instance, asking âAre you a smoker?â may elicit a âNoâ response from an exsmoker whereas the question âHave you ever smoked?â would be answered âYesâ.
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 188 SYMPTOMS ⢠Some studies use a self-administered questionnaire, whereas other studies use a trained interviewer. Trained interviewers can determine whether the subject understands the questionnaire and can follow a prescribed set of probing questions that may help to resolve the specific nature of not-well-described symptoms. ⢠Some studies have parents complete the questionnaires for the children, whereas other studies have the child answer the questionnaire. For older children, parents may not be aware of active smoking by the child and exposures to ETS in environments outside the home. ⢠Questionnaires necessarily involve some subjective elements that are prone to recall bias. For example, a smoker who is symptomatic may be more likely to report the same symptom in his/her child (Schenker et al., 1983; Ferris et al., 1985). Many tests are prone to measurement error, which tends to obscure differences between groups of subjects. For example, it may be necessary to repeat lung function measurements for a given individual and to average results to get a reliable estimate. Lung function tests are often not sensitive to the structural and functional changes associated with lung disease (Drill and Thomas, 1980). CROSS-SECTIONAL STUDIES In the following sections, selected cross-sectional studies of respiratory symptoms, lung function, and respiratory infections and longitudinal studies of lung functions are reviewed. The studies reviewed here are larger studies in which attempts have been made to standardize assessments and many of the data-gathering techniques, including interviews. Studies of Respiratory Symptoms in Children Almost all of the cross-sectional studies that have compared children of parents who smoke with the children of parents who do not smoke have reported increased prevalences of respiratory symptoms, usually cough, sputum, or wheezing, in the children of smoking parents. Some studies, including some that have not found a statistically significant increase in the prevalence of respiratory symptoms in ETS exposed children, have demonstrated
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 189 SYMPTOMS an increase in respiratory symptom prevalence with an increasing number of parents or other adults who smoke in the home (see below). Three problems are especially important for studies of respiratory symptoms in children, i.e., underreported active smoking on the part of children, recall bias leading to overreporting of symptoms by parents, and the confounding variables of infections in parents. All three may lead to overestimation of symptom prevalences among children of smokers. Recall bias would occur if parents who have respiratory symptoms are more likely to report those symptoms in the children. (The possiblity also exists that parents with these symptoms would look upon them as so commonplace as not to be worthy of mention). Parents who are smokers are also more likely to have more respiratory symptoms and respiratory infections. Respiratory infections (and, as a consequence, symptoms) among children of smokers may be the result of direct transmission of infectious agents from the parent or may be caused by inflammation and irritation of lung tissues due to ETS exposure and consequent increase in susceptibility to infection. It has been observed that parents, especially mothers, who have a history of severe respiratory illness report higher rates of respiratory symptoms in their children (Schenker et al., 1983; Ferris et al., 1985). Various ways of dealing with these potential sources of bias have been proposed. Restricting the study or analysis to children below age 8 is likely to eliminate bias due to underreporting of children who currently smoke. It is more difficult to handle the overreporting of symptoms in children when the parents have respiratory symptoms. An additional problem for interpretation of parental reports of respiratory symptoms was noted by Schenker et al. (1983). In their study, children whose questionnaires were completed by fathers had significantly fewer symptoms reported than children with mother-completed questionnaires. There was no comparison of questionnaires completed separately by both mother and father for the same child. Because the rates for symptoms as reported by the mother were similar to what was found in other studies and because the fathers reported significantly fewer symptoms, the investigators suggested that fathers underreported symptoms in their children.
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 190 SYMPTOMS Table 11â2 reviews several selected cross-sectional studies of respiratory symptoms in children and adults. Lebowitz and Burrows (1976), reporting on children in the Tucson Epidemiologic Study of Obstructive Lung Disease, emphasized the need for controlling for parental symptoms. They reported that children had a higher prevalence of respiratory symptoms if they lived in households with adults with the same symptom, regardless of the family smoking habits. When the presence of symptoms in the adults was taken into account by partitioning households based on presence or absence of adult symptoms(s), the odds ratio that remained was greater than unity but was no longer statistically significant [Mantel-Haenszel odds ratio for all respiratory symptoms calculated from data presented is 1.35 (95% confidence limits of 0.91 to 1.98)]. Most symptoms were reported more frequently for children in currently smoking families. Ferris et al. (1985) have argued that correcting for parental symptoms represents an overcorrection for respiratory symptoms in children since it also corrects for the parents' smoking habits. In the Harvard Air Pollution Respiratory Health Studies (Six-Cities Study) of 10,106 white children aged 6â9 years, the variable indicating whether the parent had a history of bronchitis, emphysema, or asthma was found to be a highly significant independent risk factor for cough and wheeze and a history of respiratory illness among children (Figure 11â2). Children whose parents had a positive history had 72â155% higher symptom and illness rates than children whose parents had no history of these illnesses. Adjustment for parental respiratory history reduced the size of the estimated effects of maternal smoking on respiratory symptoms and illnesses by 20 to 30%, but the associations remained statistically significant for most of the outcome symptom and respiratory illness variables (odds ratios of 1.23 and 1.28, respectively). In both the Lebowitz and Ferris studies, adjustment for parental symptoms or respiratory illness decreased the strength of the apparent association between exposure to ETS and respiratory symptoms, but did not eliminate it. This finding leads to the reasonable conclusion that the exposures typical of ETS are sufficient to cause respiratory symptoms in some children. The increases in frequency of cough were 20 to 50%, and as high as 90%, when there were smoking parents. The increases in frequency of wheezing were more variable, which may indicate the difficulty in
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 191 SYMPTOMS TABLE 11â2 Effects of Passive Smoking on Respiratory Symptoms: Selected Cross-sectional Studies Involving Children/ Adolescents Study Source of Subjects Exposure Findings Comments Subjects Assessment Colley, Aylesbury, 1,328 boys, Self- 1. CloseSuggested cross- 1974 UK; seven 270 girls; ages administered association ofinfection may be public schools; 6â14 questionnaire 1971 from parents child coughimportant ause; used different and parentquestion from winter U.S. studies morning phlegm 2. Prevalence of cough, 15.6% no smokers, 22.2% both parents smoke (ns) Lebowitz Tucson, Ariz.; 1,655 Self- 1. Prevalence ofLess than 15 and stratified households; administered cough inyears old Burrows, cluster random Anglo-white; NHLBI assumed to be 1976 sample of 1,252 children questionnaire young nonsmokers; households; <16, from children children, 7.8%concluded 1972â1973 2,516 children >15; otherwise no smokers,familial >15 from parents 10.4% aggregation smokers important, potential (p<0.05) confounder 2. Significance gone when parental symptoms considered Schilling et Survey of 816 children in Respiratory 1. No effect ofTried to account al., 1977 towns in 376 families; Symptom parental for active Connecticut 607 children Questionnaire, and South <16, administered by smoking onsmoking in children Carolina 109 children interviewer children's >15 cough or wheeze 2. Prevalence of wheeze in young children related to parental wheeze (p<0.01) Bland et al., Derbyshire, 2,847 boys, Self- Prevalence of cough, 16% Effects of child's 1958 UK; 48 2,988 girls; administered no smokers, 19% one and parent's secondary 12 years old questionnaire by smoker, 23.5% two smoking schools; 1974 child smokers (p<0.01) independently analyzed.
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 192 SYMPTOMS Study Source of Subjects Subjects Exposure Findings Comments Assessment Tager et al., East Boston, Mass.; 444 children; NHLBI No increase in Controlled for 1979 random sample in ages 5â9 years questionnaire respiratory family size schools; 1975â1977 administered by illness with interviewer; if age parental <10, parent smoking answered Weiss et al., See Tager et al., 650 children; See Tager et al., Persistent See Tager et al., 1980 1979 ages 5â9 years 1979 wheeze, 1% no 1979 smokers, 6.8% one smoker, 11.8% two smokers (p<0.02) Dodge, 1982 Three towns in 558 children; Self-administered Child's wheeze Arizona; survey of ages 8â10 by parents (p<0.05), schools; 1978â1979 years sputum (p<0.05), and cough (p<0.01) related to parental smoking Schenker et al., Pennsylvania; 4,071 Self-administered Trend with Not adjusted for 1983 survey of schools children; ages by parents number of parental 5â14 smoking parents symptoms not significant although found for any to influence no. symptoms symptoms reported Ware et al., Six U.S. cities; 10,106 Self-administered 20â35% Multiple logistic 1984 different regions children; ages by parents increased risk of regression with survey of schools; 6â13 all respiratory gas cooking as 1974â1979 illness and other predictor symptoms with maternal smoking
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 193 SYMPTOMS assessing this symptom. Furthermore, there appears to be a dose-response relationship between exposure and the likelihood of the child's developing respiratory symptoms or a respiratory illness. In the Harvard Study, a significant dose-response relationship was reported; the more mothers who smoked, the greater the risk of respiratory symptoms and illnesses among their children. FIGURE 11â2 Relative odds of respiratory illness or symptoms versus average daily cigarette smoking by the child's mother. Reference value is zero cigarettes per day. From Ferris et al. (1985). Studies of Lung Function in Children A more quantitative measure of the impact of ETS on the lung is obtained by measures of lung function. Many of the studies that have examined the relationship between passive smoking and lung function have been cross-sectional.
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 194 SYMPTOMS Most studies have examined the effect of exposure to parental smoking rather than ETS exposure outside the immediate family. It is assumed that children are less likely than adults to be exposed to occupational irritants. The cumulative burden of respiratory It is often difficult (but not impossible) to measure lung function insults is, therefore, likely to be smaller in children than in adults. in young children and also hard to dissect out the relative contribution of ETS and that of natural variation and the effect of respiratory infections to pulmonary damage. A majority of the studies (reviewed in Table 11â3) has shown a small decrease (up to 0.5% FEV1 per year) in rate of increase in lung function associated with normal growth in children living with one or more parents who smoke compared with those living with nonsmoking parents (Table 11â3 and Figure 11â3). These differences have usually been statistically significant. Although the mean effect is small, there are individuals in each study who have large decrements in growth of lung function. Some studies have found a dose-response relationship with the number of smokers in the home or the amount smoked (Hasselblad et al., 1981). Ware (1984) shows (see Figure 11â4) a highly significant negative association between maternal smoking level and FEV1 at both the baseline and follow-up examinations. For a child of a mother who smoked one pack of cigarettes per day compared with a child of a nonsmoking mother, the FEV1 was 0.7±0.2% lower at the baseline examination and 0.8±0.2% lower at the follow-up examination 1 year later. This amounts to a 10- to 20-ml difference for a child with an FEV1 between 1.5 and 2.5 L. In most studies, only the maternal effect was statistically significant. This may be because mothers usually spend more time with their young children than fathers. A study carried out in Shanghai in the People's Republic of China reported a clear paternal effect. Chen and Li (1986), in a cross-sectional study of 303 boys and 268 girls aged 8â16, found that the number of cigarettes smoked by fathers was linearly related to a decrease in FEV1 and FEF25â75%, the average forced expiratory flow during the middle half of the period of expiration. None of the mothers in this study were smokers; therefore, there was no maternal effect in that population. Differences in father's smoking status accounted for 0.5% of the variation among individuals in FEV1 and 1.2% of the variation in FEF25â75%.
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 195 SYMPTOMS TABLE 11â3 Effects of Passive Smoking on Pulmonary Function: Selected Cross-sectional Studies Involving Children/ Adolescents Study Source of Subjects Exposure Findings Comments Subjects Assessment Lebowitz et al., Tucson, Ariz.; 1,655 Self-administered No relationship Less than 15- 1982 stratified cluster households; NHLBI of FEV1 with year-olds random sample Anglo-white; questionnaire from parental assumed to be of households; 1,252 children children >15; smoking when nonsmokers; 1972â1973 <16, otherwise from household concluded 2,516 children parents aggregation of familial >15 body mass taken aggregation into account important, potential confounder Schilling et al., Survey of towns 816 children in Respiratory MEF 50% lower Tried to account 1977 in Connecticut 376 families; Symptom in younger for active and South 607 children Questionnaire, children with smoking in Carolina <16, administered by maternal children 209 children >15 interviewer smoking (p<0.05); FEV1 , PEF not significant Tager et al., East Boston, 444 children; NHLBI Lower z-scores Controlled for 1979 Mass.; random ages 5â9 years questionnaire for FEF25â75% in family size sample in administered by children with schools; 1975â interviewer; if age smoking parents 1977 <10, parent answered Weiss et al., See Tager et al., 650 children; See Tager et al., Lower z-scores See Tager et al., 1980 1979 ages 5â9 years 1979 for FEF25â75% 1979; also with maternal controlled for smoking wheeze in child (p<0.005); FVC, FEV1 not significant Hasselblad et CHESS study, 16,689 children; Self-administered FEV0.75 dose- No information al., 1981 seven cities; ages 5â13 years by parent (usually response on child's survey of mother) relationship with smoking; small schools; 1970â mother's effect of maternal 1973 smoking smoking (0.1% of variance)
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 196 SYMPTOMS Study Source of Subjects Exposure Findings Comments Subjects Assessment Dodge, 1982 Three towns in 558 children; ages Self-administered No effect of Lung function Arizona; 8â10 years by parents parental smoking tests not survey of on any standardized schools; 1978â parameters, 1979 cough (p<0.01) related to parental smoking Ware et al., Six U.S. cities; 10,106 children; Self-administered FEV1 Multiple logistic 1984 different ages 6â13 by parents significantly regression with regions; survey negative; FVC gas cooking as of schools; positive relation other predictor 1974â1979 to maternal smoking Chen and Li, Shanghai, 571 children; ages Self-administered Paternal lifetime No effect of 1986 PRC, survey of 8â16 years questionnaire by smoking related maternal two schools; parents to z-scores of smoking, 1984 FEV1, MMEF, probably due to and FEF62.6â87.5% low prevalence of female smokers in PRC Tashkin et Los Angeles 971 nonsmoking, Modified NHLBI Inconsistent Effect in older al., 1984 County survey nonasthmatic questionnaire effect of maternal girls probably due of four areas in children, ages 7â administered by smoking in to unreported city; 1973 17 interviewer younger boys and smoking by child older girls Ferris et al., See Ware et al., See Ware et al., See Ware et al., Significant effect See Ware et al., 1985 1984 1984 1984 of parental 1984 smoking on FEV1
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 197 SYMPTOMS FIGURE 11â3 Mean percent lung function, by parental smoking, of nonsmoking males and females, ages 10â19, 1962â1965, from Tecumseh, Michigan. Burchfiel et al. (1986). The most important contributors to variation in lung function among children are size-related factors such as sex, age, and height. These account for about 50â60% of the variation (Comroe et al., 1962). It is not possible to determine whether ETS is directly causing the decreased lung function observed in children of smoking parents or if an increased infection rate in these children (see below) is responsible for the decrease. The annual small decrease in FEV1, which is related to exposure to ETS, is unlikely to be clinically significant. However, the effect may be important in two respects. First, the existence of statistically significant differences related to parental smoking leads to the conclusion that there are pathophysiologic effects of exposure to ETS in the lungs of the growing child. It may be an in utero effect, an effect on the growing and remodeling lung, or both. Second, it raises the question of whether the child who is adversely affected by parental smoking
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 198 SYMPTOMS may be at an increased risk for the development of chronic airflow obstruction in adult life. An accelerated decline in lung function could increase the risk of chronic pulmonary disease (Samet et al., 1983). FIGURE 11â4 Mean of pulmonary function residual (±1 SD) by mothers' reported daily cigarette smoking, compared with children whose mothers have never smoked. Squares represent the first examination (n=7,112) and triangles represent the second examination (n=6,278). From Ware et al. (1984). Studies of Lung Function in Adults White and Froeb (1980) studied 800 nonsmoking, middle-aged subjects, out of a total population size of 2,100, and found a small statistically significant decrease (8%) in FEF25â75% in both men and women who were nonsmokers exposed to ETS. The reported reduction in FEF25â75% for ETS exposed nonsmokers was almost identical to that of the smokers of 1â10 cigarettes per day. This raises questions about their findings. This study may suffer from problems of selection bias in the allocation of subjects to categories and the absence of any exsmokers (Adlkofer, et al., 1980; Aviado, 1980; Huber, 1980; Lee, 1982).
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 199 SYMPTOMS A cross-sectional study from France (Kauffmann et al., 1985) supports the conclusions that exposure to ETS may have an effect on lung function in nonsmoking adults. The French Cooperative Study surveyed more than 7,800 adult residents of seven cities in France in 1975 and found 1,675 were true nonsmokers. In men and women over 40, nonsmokers of either sex who had a spouse who smoked had a significantly lower FEF25â75% than those living with a nonsmoker. These differences were not explained by social class, educational level, air pollution, or family size. Among the women, there was also a significant difference in FEV1 and a dose effect was seen with the amount smoked by their husbands. These differences, only apparent in persons over 40, were small and were uncovered only following detailed examination of the data after the population had been stratified by age. Two other cross-sectional studies involving adult women have found an effect of exposure to ETS on lung function. In a study of 220 married women aged 25 to 69 years from five U.S. cities, Kauffmann and coworkers (1986) reported that standardized residuals for FEV1 and FEV1/FVC* for the group identified as passive smokers were intermediate between the results of nonsmokers and current smokers. In a study of 163 nonsmoking women living in a rural area of the Netherlands, Brunekreef and coworkers (1985) found that those exposed to ETS tended to have slightly lower mean values for all of the lung function variables measured. These differences reached statistical significance for peak flow and FEF25â75% in the 40- to 60-year-olds. The numbers in each of their groups were small. No information was given on possible childhood exposures to cigarette smoke of the women studied. Kentner and coworkers (1984), in a study of 1,351 white collar workers (941 men and 410 women) in northern Bavaria, and Comstock et al. (1981), a study that included 1,724 adults residents of Washington County, Maryland, examined the potential effects of ETS. In these studies, information was collected from subjects using questionnaires and the subjects were then classified as never smoked, exsmokers, and current smokers. The Kentner et al. study evaluated home and workplace exposures, whereas the Comstock et al. study evaluated only home exposures. In the Kentner et al. (1984) study, an additional classification was made for other smokers, representing those who were cigar and pipe smokers. These * FVC is the forced vital capacity.
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 200 SYMPTOMS investigators found no significant reductions in lung function with ETS exposure. In view of the large number of factors that affect lung function, it is not surprising that it is difficult to document the extent to which a single type of exposure affects lung function. The lungs of adults have been subjected to many environmental exposures and potential insults over a lifetime, making it unlikely that a specific effect could be isolated. The variability in lung function due to differences of the other factors tends to obscure effects of a single variable. In addition, results in adults should be evaluated for possible misclassification of exsmokers or occasional smokers as nonsmokers, as well as possible confounding by occupational exposures to other pollutants or to ETS. LONGITUDINAL STUDIES OF LUNG FUNCTION IN CHILDREN AND ADULTS An important unanswered question is whether exposure to ETS affects the way the lungs grow and develop during childhood. Respiratory symptoms, by themselves, may have little clinical significance but would be important if associated with a change in the rate of lung growth and development or the development of pulmonary pathology at older ages. There is evidence from two cohort studies (Table 11â4) that parental smoking may affect the rate of lung growth during childhood. Tager and coworkers (1983), who have followed 1,156 elementary school children in East Boston, Massachusetts, over a 7-year period, reported that maternal smoking was associated with a reduced rate of annual increase in FEV1 and FEF25â75%. There was a reported 3â5% decrease in expected lung growth over the 7-year period. Burchfiel and coworkers (1986) examined pulmonary function in 3,482 children in Tecumseh, Michigan. Children 0 to 19 years old were followed for 15 years, during which time questionnaire information was collected from both parents. FEV1 and FVC values were significantly lower by 5% in male nonsmokers 10 to 19 years of age whose parents were current smokers. The Harvard Air Pollution Respiratory Health Studies (Ferris et al., 1985; Berkey et al., 1986) (Figure 11â5) show a relatively smaller effect than that reported by Tager and coworkers (1983). The Harvard study included 7,834 children between the ages of
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 201 SYMPTOMS TABLE 11â4 Effects of Passive Smoking on Pulmonary Function: Selected Longitudinal Studies Involving Children/ Adolescents Study Source of Subjects Exposure Findings Comments Subjects Assessment Tager et al., East Boston, 1,156 children NHLBI 1-year change in Tried to account 1983 Mass.; random from 404 questionnaire FEV1 reduced in for child's sample in families; ages completed by smoking families smoking; change schools; 1975â 5â9 years parents (p<0.02); 9% scores corrected 1981 decrease over 2 for age, sex, years, 7% decrease height, first FEV1 over 5 years Ferris et al., Six U.S. cities; 8,380 white Self-administered Growth rate in Assumed 1985 different children; ages by parents FEV1 reduced with children did not regions; survey 5â19 years; 6 maternal smoking smoke; controlled of schools; annual visits (p<0.02), dose for city and SES 1974â1981 related Berkey et al., Same as Ferris 7,867 white Same as Ferris et al., Maternal and Corrected for 1986 et al., 1985 children ages 1985 paternal smoking parental 6â10 years not significantly education related to FEV1 growth rate; however, number of cigarettes smoked by mother significant effect (p<0.05)
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 202 SYMPTOMS 6 and 10 years who were followed over a 5-year period. Children whose mothers smoked one pack of cigarettes per day had FEV1 levels, at age 8, that were 0.81% lower than children with nonsmoking mothers. Growth rates for FEV1 were approximately 0.17% per year lower. For a child aged 8 years with an FEV1 of 1.62 L, this corresponds to a deficit in rate of growth of FEV1 of approximately 3 ml per annum and a deficit of 13 ml by age 8. In contrast to the lower FEV1 seen in children whose mothers smoked, higher levels for FVC were observed in children with smoking mothers compared with children whose mothers did not smoke. For example, average FVC at age 8 for a child whose mother smoked one pack per day, was 0.33% higher than a child with a nonsmoking mother. On the other hand, the growth rate for FVC was 0.17% lower for a child with a smoking mother. This would be equivalent to a 2.8 percent decrease in pulmonary development throughout childhood and implies a decrease in the development of pulmonary function in children of smoking parents. In view of the effects that climatic conditions can have on housing characteristics, and subsequent ventilation rates, it would be advantageous to conduct longitudinal studies in regions of the United States other than the Northeast. In any future studies, great care should be taken, as it was in the two cohort studies, to account for potential confounding variables in the analyses, such as socioeconomic status and gas cooking. Another aspect that deserves more attention in future studies is the effect on children's pulmonary function when parents stop smoking. THE EFFECT OF PASSIVE SMOKING ON RESPIRATORY INFECTIONS There is now strong evidence that bronchitis, pneumonia, and other lower-respiratory-tract illnesses occur more frequently (at least during the first year of life) in children who have one or more parents who smoke (see Table 11â5). Evidence that this increased frequency of acute respiratory infections continues into later childhood is less convincing, although the evidence from both cross-sectional studies and cohort studies shows such a trend. Harlap and Davies (1974) followed a cohort of 10,672 infants born in Israel between 1965 and 1968. Admissions to the hospital during the first year of life were recorded. Information about maternal smoking was obtained during the pregnancy only. Infants
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 203 SYMPTOMS with major congenital malformations and those dying before their first birthday were excluded from the study. For the total population studied, there were 25.4 admissions per 100 babies under 1 year of age. The infants of mothers who smoked had a 27.5% greater hospital admission rate for pneumonia and bronchitis than children of nonsmoking mothers. A dose-response relationship was also found between the amount of maternal smoking and admissions to hospital for pneumonia and bronchitis. FIGURE 11â5 Calculation of growth rate and level of ln(FEV1) for an individual child. The residuals in the upper panel, i.e., the difference between observed and predicted ln(FEV1), were regressed on age in the lower panel. From Berkey et al. (1986). Colley (1974; Leeder et al., 1976) carried out a similar study in London. The study involved a birth cohort of 2,205 infants born between 1963 and 1965. In this group of children, the incidence of pneumonia and bronchitis in the first year of life was associated with the parents' smoking habits. This was true whether or not the parent has respiratory symptoms. The incidence was lowest for children of nonsmoking parents, highest in families where both
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 204 SYMPTOMS TABLE 11â5 Childhood Respiratory Tract Illness and Passive Smoking Study Source of Subjects Exposure Findings Comments Subjects Assessment and Health Information Harlap and Birth cohort; All infants in Antenatal 1. SignificantlyInformation Davies, West cohort of interview of more admissionsabout mother's 1974 Jerusalem; 10,672 mothers 1965â1969 admitted to for bronchitis orsmoking obtained hospital in pneumonia, prenatally, not Jerusalem especially inconcurrent with winter, in infantschild's whose mothersadmission; no information smoke about father's 2. Dose-responsesmoking for number ofobtained cigarettes by mother and excess of bronchitis and pneumonia Colley, Birth cohort; 2,205 infants Annual follow-up 1. Incidence ofMost important 1974; Harrow, UK; by health visitors pneumonia anddeterminant of Leeder et 1963â1965 for 5 yr; al., 1976 questionnaire bronchitis inrespiratory illness was administered by first yearbronchitis or trained health associated withpneumonia in visitor parental sibling; analysis smoking: not controlled for number of incidence lowestsiblings with both nonsmokers, highest with both smokers, intermediate with one smoker 2. Associations inconsistent after 1 yr 3. In first year of life, ETS exposure doubled risk for pneumonia/ bronchitis
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 205 SYMPTOMS Rantakallio, Birth cohort; 1,821 Smoking Significant increase in Only maternal 1978 Northern exposed, determined in hospitalization for smoking Finland; 1966 1,821 interview respiratory illness evaluated; unexposed; during categories ages 0â5 pregnancy based on smoking during pregnancy Said et al., Cohort; France; 3,920 Self- Increase in tonsillectomy Smoking by 1978 1975â1976 children; ages administered and/or adenoidectomy parents may 10â20 by children not have coincided or preceded operations Fergusson et Birth cohort; 1,265 infants Follow-up by 1. Lower Analysis al., 1981 Christchurch, structured respiratory controlled for New Zealand; interviews with maternal age, 1977 mother at birth, illness education, 4 mo, 1, 2, and significantly family size, 3 yr; diaries related tofamily living kept by mother's conditions mothers on smoking in first child's history of medical year of life, care; check equivocal in with hospital second and records absent in third 2. No effect of paternal smoking 3. Linear dose- response between maternal smoking and incidence of lower respiratory infections Pedreira, 1985 Birth cohort 1,144 infants Interview with 1. Tracheitis andNo adjustment from practice of followed for 1 mother at first bronchitis made for four yr after birth well baby exam potentially pediatricians in carried out by significantly confounding suburb of doctor; all related tovariables; Washington, subsequent maternal relatively D.C.; 1976â office visits in smoking affluent area 1981 first year of life 2. No dose- and low for lower maternal respiratory response smoking rate tract infection relationship (19%) 3. Bronchiolitis not related to parental smoking
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 206 SYMPTOMS Study Source of Subjects Exposure Findings Comments Subjects Assessment and Health Information Speizer et al., Six U.S. cities; 8,120 children; Questionnaire Parental smoking Recall bias a 1980 1974â1979 ages 6â10 completed by and sex of child potential problem parents related to because children respiratory aged 6â10 at time disease before age of survey 2 Dutau et al., Survey in 892 children; ages Questionnaire Significant Pointed out 1981 south of 0â6 seen by administered to relationship importance of France; 1979â pediatrician or parents between annual day care centers 1980 admitted to incidence of lower and nursery hospital respiratory schools in infections and increasing rates total number of of lower cigarettes smoked respiratory inside home infections and difficulty of adjustment for this
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 207 SYMPTOMS parents smoked, and intermediate where one parent smoked. This effect was not seen consistently over age 1. A third birth-cohort study, involving 1,265 children in New Zealand, was reported by Fergusson et al. (1981). They studied the children from birth to age 3 years and found an increase in both bronchitis/pneumonia and lower respiratory illness during the first year in children whose mothers smoked. During the second year, the relationship between maternal smoking and lower respiratory illness was equivocal. The relationship disappeared by the third year. There was no effect observed of paternal smoking on the incidence of lower respiratory illness. Using logistic regression, they found that the rates of lower respiratory illness were related to maternal smoking. For each five cigarettes smoked per day by the mother, there was an increase of 2.5â3.5 lower respiratory âeventsâ per 100 children at risk. Adjustment for maternal age, education, family size, and family living conditions did not change the relationship. Rantakallio (1978) studied the effect of maternal smoking during pregnancy on morbidity and mortality of children to age 5 based on 12,068 births. Smoking status on the mother was only available from antenatal interview. Perinatal mortality was not higher among children of smokers, however, postneonatal mortality (between 28 days and 5 years) was significantly increased. Children of smokers were hospitalized for respiratory illness significantly more often than children of nonsmokers and the average duration of hospitalization was longer among children of smokers. Two case-control studies evaluated smaller groups of children hospitalized for respiratory infection and nonhospitalized controls. Pullan and Hay (1982) studied 130 children who were hospitalized with a documented respiratory syncytial virus (RSV) infection in infancy and 111 controls. They found that children hospitalized with documented RSV infections were more likely to have mothers who smoked and that the children had an excess of wheeze and asthma and lower levels of pulmonary function, which persisted to age 10. Sims et al. (1978) also suggested that cigarette smoking by parents during a baby's first year of life is associated with an increased risk of RSV infections. Speizer et al. (1980) studied approximately 8,000 children, aged 6â10 years, from six communities in the United States as part of a prospective study of the health effects of air pollution (Harvard Air Pollution Respiratory Health Studies). Parental smoking and
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 208 SYMPTOMS sex of the child was associated with respiratory disease before age 2, after other variables had been taken into account. Children from households with gas cooking also gave a history of more frequent respiratory illness before age 2 than children from households with electric cooking. Dutau et al. (1981) studied 892 children under age 6 in the south of France who were seen by a pediatrician or hospitalized for various reasons. They found a significant correlation between the annual incidence of pulmonary infections and the total number of cigarettes smoked inside the house. Pedreira et al. (1985) followed all newborns (1,144 infants) seen by a group of pediatricians for a first well- baby examination between 1976 and 1981. They found that tracheitis and bronchitis occurred significantly more frequently (89% and 44%, respectively) in infants whose parents smoked and that maternal smoking imposed greater risks upon the infants than paternal smoking. One study looked at the frequency of tonsillectomies and/or adenoidectomies in children (Said et al., 1978). They found the frequency was significantly increased among children with smoking parents. However, the smoking status reported for the parent may have been current smoking status, even though the operations had occurred 5 to 15 years previously. All the studies that have examined the incidence of respiratory illnesses in children under the age of 1 year have shown a positive association between such illnesses and exposure to ETS. The association is very unlikely to have arisen by chance. It may represent a direct association between ETS exposure and disease (a causal explanation) and/or an indirect one (noncausal) arising because children living in homes of smokers are at risk of such diseases for other reasons. Some of the studies have examined the possibility that the association is indirect by allowing for confounding factorsâsuch as social class, parental respiratory illnesses and birthweightâand have concluded that such factors do not explain the results. This argues, therefore, in favor of the causal explanation. Such an explanation is supported by the evidence of a dose-response relationship specific for respiratory disease (Tables 11â6 and 11â7). Also, the mother's smoking is more likely to affect the infant than the father's smoking, since the proximity of mother and child is closer during the child's first year when the effect is more marked and consistent than later in childhood (see Fergusson et al., 1981). This also supports a causal, rather than
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 209 SYMPTOMS an indirect, explanation. Therefore, the evidence indicates that smoking in the home does increase the incidence of respiratory illness in infants. The mechanism for this increase is less certain. It could represent a direct effect of ETS on the respiratory tract of the infant or it could be due to such infants' being exposed to more parental respiratory infections as a result of their parents' smoking. Either way, smoking in the home appears to increase the rate of respiratory illness in young children. WHEN DO PULMONARY EFFECTS OF PASSIVE SMOKING OCCUR? The weight of evidence is that there are clearly observable effects of ETS on the respiratory system. These effects include an increase in the incidence of acute respiratory infections in early infancy; increased prevalance of cough, sputum production, and wheezing; and a decrease both in lung function measured at an instant in time and in the growth of lung function. The finding of differences in symptom prevalence, respiratory infection rates, and lung function among children exposed and not exposed to ETS is often interpreted as evidence of a chronic effect of ETS on the airways. This is probably true, and it is unlikely that ETS is not an upper- and lower-respiratory- tract irritant in children. The possibility that there is an effect of maternal smoking in utero as well must be considered. Evidence of an in utero effect in pregnant rats exposed to whole tobacco smoke has been reported by Collins et al. (1985). These investigators reported that pregnant rats exposed to smoke daily from day 5 to day 20 of gestation, when compared with control rats, showed reduced lung volume at term and saccules that were reduced in number and increased in size. The internal surface area of the lung was decreased. The relevance of this study to maternal smoking during pregnancy in humans is not yet clear and deserves further investigation. Other factors that may alter the time when ETS effects during childhood include the relative immaturity of the immunologic system and the growth and remodeling that are occurring in the immature lung. The infant lung differs in a number of important ways from the adult lung: (1) T-lymphocyte and macrophage function are not fully developed at birth, (2) there is increased susceptibility to infection as a result of comparatively immature
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 210 SYMPTOMS TABLE 11â6 Experimental Studies of Acute ETS Exposures for Asthmatic Patients Study Population Exposure Findings Comments Shephard et al., Fourteen patients Room: 14.6 m3 Changes in pulmonary Patients on medication; 1979a from the Gage Time: 2 h function slight; slight associated chronic Research Institute Cig.: 7 decrease in total lung bronchitis or pulmonary (nine male, five CO: 24 ppm capacity (helium emphysema in some female); mean age mixing, p<0.02) patients; four patients 37 years claimed smoke sensitivity Dahms et al., 1981 Ten patients from Room: 30 m3 Linear decrease in Patients on medication St. Louis Univ. Time: 1 h pulmonary function with restricted use of Hospital Allergy Cig.: n.g. over time in patients; bronchodilators 4 h prior Clinic; ages 16â39; CO: 15â20 ppm FEV1 decreased 21.4%; to test; five patients and 10 controls, ages FEF25â75%, 19.2%; five controls complained 24â53 FVC, 20%; no change of irritation to ETS in controls Knight and Breslin, Six patients (4M, Details not given Significant decrease in No correlation of 1985 2F); mean age 25.5 3/6 subjects; PC20FEV1 decreased function with yr significantly decreased chest symptoms with histamine Wiedemann et al., Nine patients with Room: 4.25 m3 No change in Patients off medication; 1986 near normal lung Time: 1 h expiratory flow rates; six patients with history function; ages 19â Cig.: n.g. small decrease in of reaction to ETS 30 CO: 40â50 ppm bronchial reactivity; PD20FEV1 increased from 0.25 to 0.79 with methylcholine Abbreviations: n.g.=not given.
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 211 SYMPTOMS lung defenses, (3) the internal diameter of the small airways is extremely small and vulnerable to obstruction, and (4) the newborn child has its full complement of airways at birth but only a small proportion of the alveoli. During childhood the airways grow in internal diameter, and the alveoli both multiply and increase in size. TABLE 11â7 Admission Rates in the First Year of Life for Bronchitis and Pneumonia per 100 Infants by Maternal Smoking and Number of Cigarettes Smoked Daily (Number of Infants in Parentheses) Nonsmokers Never Smoked Former Smokers Smokers Total (Cigarettes per day) (8,900) (786) 1â10 11â20 21+ (10,672) (747) (179) (60) 9.6 7.8 10.8 16.2 31.7 9.8 NOTE: Differences among three categories of smoker p<0.001. SOURCE: Harlap and Davies (1974). The question of the timing of the effect of ETS on the growing and developing lung remains to be elucidated. If the effect is in utero, the question of how this carries over into infancy and childhood must be addressed. Likewise, the carryover effects of increased incidence of respiratory infections in infancy must be determined. In this regard, there is already some information relating early childhood respiratory illness to subsequent respiratory symptoms and impaired lung function later in childhood (Woolcock et al., 1984; McConnochie, 1985). Evidence is also accumulating that respiratory infections in early childhood are related to an accelerated decline of FEV1 in adult life (Burrows et al., 1977b; Lebowitz and Burrow, 1976). If this is so, and if exposure to ETS increases susceptibility to acute respiratory infections in infancy, ETS may have a carryover effect into adult life. From the evidence to date, it appears that the effects of exposure to ETS may start in utero by altering the growth pattern of the fetal lung. In infancy, exposure to ETS may increase susceptilibity to viral respiratory infections that in turn may have a
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 212 SYMPTOMS carryover effect into later childhood and adult life. Direct effects of ETS as an airway irritant are also likely, although the dose by itself may be insufficient except for the most susceptible individuals to cause symptoms and/or functional impairment. It is unlikely that exposure to ETS can cause much emphysema. As one of the many pulmonary insults, however, ETS may add to the total burden of environmental factors that become sufficient to cause chronic airway or parenchymal disease. STUDIES OF ACUTE PULMONARY EFFECTS Several studies have examined acute responses to ETS. Because asthmatics may be hypersensitive to exposures to noxious agents, a number of studies have also searched for acute effects of exposure to ETS among asthmatic populations. Other studies have been conducted on normal healthy adults. Normal Subjects Pimm et al. (1978) compared various physiologic responses of nonsmokers to either room air or room air plus machine-generated cigarette smoke. Each smoke exposure consisted of combustion of four cigarettes to produce an extremely polluted room with high levels of carbon monoxide (24 ppm) and particles (greater than 4 mg/m3). Pulmonary function tests, nitrogen washout curves, blood carboxyhemoglobin levels, and heart rates were measured before, during, and after a 2-hour exposure. A few statistically significant differences between smoke and ambient air exposure days were found. The differences were small and were considered by the investigators to be of questionable importance. Subjective complaints were common in this and other acute cigarette smoke exposure studies, particularly eye irritation and cough. CO and suspended particles are thought to be less important than the phenols, aldehydes, and organic acids in producing this symptomatology (Hinds and First, 1975). Shephard et al., (1979b) utilized a protocol similar to Pimm et al. (1978) but under conditions of intermittent moderate exercise (increasing the respiratory volume per minute 2.5 times). Moderate and heavy ETS exposures were considered, associated with CO concentrations of 20 and 31 ppm, respectively. Neither exercise
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 213 SYMPTOMS nor exposure level significantly influenced symptomatology. Small decrements (3â4%) in FVC, FEV1, Vmax50%, and Vmax25% (the volumes of air expired during the first half of the period of forced expiration or first quarter of the period, respectively) were noted in response to smoke exposures; however, static lung volumes were unaffected. Eye irritation and odor complaints were very common. One subject complained of wheezing and chest tightness, although his pulmonary function was not significantly impaired. Subjective symptom scores were higher overall for the higher smoke exposure (13.8 versus 10.3 points/subject at the lower exposure). A few subjects reported cough, nasal discharge, or stuffiness and throat irritation. TABLE 11â8 Pneumonia and Bronchitis by Parents Smoking in First Year of Follow-up, Annual Incidence per 100 Children (Number of Infants in Parentheses) Both Nonsmokers Both or One Exsmokers or Smoking Habits Changed One Smoker Both Smokers All 7.8 9.2 11.4 17.6 11.5 (372) (675) (552) (478) (2,077) SOURCE: Colley et al. (1974). Asthmatic Subjects A number of studies have examined acute pulmonary responses of asthmatic patients to exposure to ETS (Table 11â8). However, the mechanisms for bronchoconstriction among asthmatics differ. Therefore, the comparison between study populations and between individuals within studies is difficult. Shephard et al. (1979a) examined asthmatic persons to determine whether their response to ETS exceeded that of normal subjects in a previous study. The subjects (9 men and 5 women; average age, 37 years) were exposed for 2 hours to machine-generated smoke (CO, 24 ppm). None of the patients had current respiratory infections, but some may have had associated chronic bronchitis or pulmonary emphysema. No significant alterations in dynamic lung volumes (FEV1, Vmax50%, and Vmax25%) were detected when the asthmatics' responses to ambient air and cigarette smoke were
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 214 SYMPTOMS compared. A small, but significant, decrease in total lung capacity (TLC) was noted, although preexposure TLC was slightly higher than that on the same exposure day (96.5% and 103.5% relative to ambient air TLC, respectively). The lack of measurable change was interesting in light of a reported history of exacerbation with exposure to ETS by four subjects. Acute symptomatic responses during the experimental study were similar to those seen in the investigators' previous study of normal individuals; however, more complaints of tightness in the chest (43% of subjects) and wheezing (36%) were made by asthmatic subjects. It was concluded that asthmatics did not have unusual measurable responsiveness to ETS exposure in this study. The findings of Dahms et al. (1981) contrast with those of Shephard et al. (1979a). The exposure in this study was less intense, i.e., 1 hour at CO levels of 15â20 ppm. The patients were 16 to 39 years old, had mild impairment, and were on medication, except for the restriction that no bronchodilators might be used within 4 hours previous to the test. Five of the patients reported specific complaints when exposed to ETS. When compared with control subjects, asthmatics showed significant pulmonary function changes following 1 hour of smoke exposure. FVC decreased 20% and FEV1 declined 21.4% in the asthmatic subjects. These decreases are very large compared with the other studies. Based on a 0.40% increase in blood carboxyhemoglobin, the environmental CO concentration was calculated to be between 15 and 20 ppmâcompared with approximately 24 ppm in the Shephard et al. (1979a) studies. Reasons for the discrepancy between the Dahms and Shephard studies results are not clear, nor do Dahms et al. (1981) cite or discuss the earlier Shephard et al. (1979a) findings. Knight and Breslin (1985) evaluated six nonsmoking patients. The details of the subject population and exposure conditions were not specified. They measured a mean fall in FEV1 of 11% following exposure to ETS. Using a histamine inhalation test, they found that the provocative concentration (or dose) that produced a 20% fall in FEV1 (PC20FEV1 or PD20FEV1) decreased following exposure to ETS. This indicates an increased bronchial reactivity to histamine. The authors hypothesized that the airways may be primed to react more vigorously to other triggers. Wiedemann et al. (1986) evaluated nine asthmatic individuals (aged 19 to 30 years) with normal or nearly normal lung function
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 215 SYMPTOMS for both lung function and airway reactivity following exposure to ETS. Six patients reported a history of reaction to ETS. These subjects, all of whom were off medication, were exposed for 1 hour (CO between 40 and 50 ppm). Their carboxyhemoglobin levels increased an average of 0.86% (p<0.001), FVC decreased 2% (p<0.01), and FEV1 declined 1% (not statistically significant). Airway reactivity was assessed using a methylcholine challenge test. The PD20FEV1 increased from 0.25±0.22 on the day before exposure to 0.79±1.13 postexposure (p<0.05), indicating a decrease in airway reactivity following exposure. The magnitude of this decrease was small, and the clinical meaning of the change is uncertain. There are a number of possible reasons for the apparent inconsistency among these studies, not the least of which is small sample sizes. The subjects have not been characterized fully. As noted by the authors, the stability of patients and mechanisms of bronchoconstriction differ among subjects. For instance, patients were included in several of these studies, regardless of whether they were hypersensitive on the methylcholine challenge test. Further, some studies were performed on medicated patients. None of the studies could be performed blind to the presence of ETS. Therefore, the authors could not exclude the possibility that pulmonary function changes could be emotionally related to cigarette smoke exposure, especially in those patients who reported previous histories of adverse response to ETS exposure. There are several issues that are unresolved by these studies. For instance, what proportion of a clearly defined population of asthmatics do react to ETS? If the patients are selected according to methylcholine or histamine responsiveness, criteria should be given for the extent of responsiveness, since it is a continuum. To address the issue of degrees of sensitivity, the appropriate case-control or cross-over studies, with carefully selected populations, need to be done. Mechanisms of Response The mechanisms responsible for eye irritation and rhinitis, as well as possible changes in airway size, are almost entirely unknown. They could represent irritant effects from gases such as oxides of nitrogen, acrolein, ammonia, and other reactive constituents. Lundberg et al. (1983) reported that throat irritation
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 216 SYMPTOMS and local edema may be due to vapor-phase components that stimulate substance P release from local capsaicin- sensitive afferent neurons in the airway mucosa. It is also possible that an allergic mechanism could be involved. Several authors have described allergic reactions to cigarette smoke (see, for example, Zussmann, 1970). Cutaneous hypersensitivity to tobacco antigens has been described in clinical settings (Becker et al., 1976). Constituents of tobacco smoke have also been shown to be immunogenic in laboratory animals (Becker et al., 1979; Gleich and Welsh, 1979). During the last 10 years, Becker and colleagues (1979, 1981; Becker and Dubin, 1977) have isolated a tobacco glycoprotein both from cured tobacco leaves as well as from cigarette smoke condensate. Animals that were previously sensitized to this antigen had both pulmonary and cardiovascular changes when challenged (Levi et al., 1982). However, the role, if any, of this antigen, as well as other antigens that may be present in tobacco smoke, in the pathogenesis of cardiopulmonary disease in active smokers, let alone nonsmokers exposed to ETS, remains controversial. SUMMARY AND RECOMMENDATIONS There have been many studies of respiratory effects of exposure to ETS to children. In view of the weight of the scientific evidence that ETS exposure in children increases the frequency of pulmonary symptoms and respiratory infection, it is prudent to eliminate smoking and resultant ETS from the environments of small children. What Is Known 1. Children of parents who smoke compared with the children of parents who do not smoke show increased prevalences of respiratory symptoms, usually cough, sputum, and wheezing. The odds ratios from the larger studies, adjusted for the presence of parental symptoms, were 1.2 to 1.8, depending on the symptoms. These findings imply that ETS exposures cause respiratory symptoms in some children. 2. Estimates of the magnitude of the effect of parental smoking on FEV1 function of children range from zero to approximately 0.5% decrease per year. This small effect is unlikely by itself to be clinically significant. However, it may reflect pathophysiologic
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 217 SYMPTOMS effects of exposure to ETS in the lungs of the growing child and, as such, may be a factor in the development of chronic airflow obstruction in later life. 3. Bronchitis, pneumonia, and other lower-respiratory-tract illnesses occur up to twice as often during the first year of life in children who have one or more parents who smoke than in children of nonsmokers. What Scientific Information Is Missing 1. ETS exposure during childhood may influence the development of airway hyperresponsiveness in adult life. Research is needed to address this issue. To evaluate the timing of physiologic changes during development may require animal studies. 2. Future cross-sectional studies of ETS exposure and lung function in adults need to be designed to control for other factors that may affect lung function. 3. Little information is available from long-term longitudinal studies of the effect of exposure to ETS by nonsmokers on lung function in either children or adults. Studies need to be carried out in areas with different climates and characteristics of housing over long enough periods of time to assess the effects of changing smoking patterns. Animal studies may also be required to address these longitudinal questions. Intervention studies, in which parents stop smoking in the presence of children, should be done to assess the reversibility of these effects. 4. The pathophysiologic mechanism of increased susceptibility to viral infections in very young children exposed to ETS has not been clarified. 5. The extent to which normal and asthmatic adults are affected by short-term exposures to ETS needs to be studied further. 6. The few studies of the effect of short-term ETS exposure of asthmatic patients and of nonasthmatics are not consistent. This may be because they have not been conducted under adequate control and have examined persons with considerable variability in the severity of asthmatic disease and airway responsiveness. Future studies should carefully define the populations when addressing issues of frequency of reaction to ETS and should be done separately on hyperresponsive and nonhyperresponsive patients when addressing issues of severity of reaction to ETS.
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 218 SYMPTOMS 7. Studies of other patients with obstructive lung disorders, such as cystic fibrotic and alpha-1-antitrypsin patients, need to be done. Future studies need to identify susceptible subpopulations, if they exist, who are unusually vulnerable to the acute effects of ETS exposure. 8. There is no consensus on how to deal with data on parental respiratory symptoms. Investigations should report on rates of childhood illness/symptoms using analyses that are both adjusted and unadjusted for parental symptoms. 9. There is need for information on changes in pulmonary function between the end of the peak growth period and adult life to assess the possible reversibility of effects. REFERENCES Adlkofer, F., G.Scherer, and H.Weimann. Small-airways dysfunction in passive smokers. N. Engl. J. Med. 303:392, 1980. Aviado, D.M. Small-airway dysfunction in passive smokers. N. Engl. J. Med. 303:393, 1980. Beck, G.J., C.A.Doyle, and E.N.Schachter. Smoking and lung function. Am. Rev. Respir. Dis. 123:149â155, 1981. Becker, C.G., T.Dubin, and H.P.Wiedemann. Hypersensitivity to tobacco antigen. Proc. Natl. Acad. Sci. USA 73:1712â1716, 1976. Becker, C.G., and T.Dubin. Activation of factor XII by tobacco glycoprotein. J. Exp. Med. 146:457â467, 1977. Becker, C.G., R.Levy, and J.Zavecz. Induction of IgE antibodies to antigen isolated from tobacco leaves and from cigarette smoke condensate. Am. J. Pathol. 96:249â254, 1979. Becker, C.G., N.Van Hamont, and M.Wagner. Tobacco, cocoa, coffee, and ragweed: Cross-reacting allergens that activate factor-XII- dependent pathways. Blood 58:861â867, 1981. Berkey, C.S., J.H.Ware, D.W.Dockery, B.G.Ferris, Jr., F.E.Speiger. Indoor air pollution and pulmonary function growth in preadolescent children. Am. J. Epidemiol. 123:250â260, 1986. Bland, M., B.R.Bewley, V.Pollard, and M.H.Banks. Effect of children's and parents' smoking on respiratory symptoms. Arch. Dis. Child. 53:100â105, 1978. Brunekreef, B., P.Fischer, B.Remijn, R.Van der Lende, J.Schouten and P.Quanjer. Indoor air pollution and its effect on pulmonary function of adult non-smoking women. III. Passive smoking and pulmonary function. Int. J. Epidemiol. 14:227â230, 1985. Burchfiel, C.M., M.W.Higgins, J.B.Keller, W.J.Butler, W.F.Howatt, and I.T.T.Higgins. Passive smoking, respiratory symptoms and pulmonary function: A longitudinal study in children. Am. Rev. Respir. Dis. 133:A157, 1986.
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 219 SYMPTOMS Burrows, B., R.J.Knudson, M.G.Cline, and M.D.Lebowitz. Quantitative relations between cigarette smoking and ventilatory function. Am. Rev. Respir. Dis. 115:195â205, 1977a. Burrows, B., R.J.Knudson, and M.D.Lebowitz. The relationship of childhood respiratory illness to adult obstructive airway disease. Am. Rev. Respir. Dis. 115:751â760, 1977b. Chen, Y., and W.X.Li. The effect of passive smoking on children's pulmonary function in Shanghai. Am. J. Public Health 76:515â518, 1986. Chen, Y., W.Li, and S.Yu. Influence of passive smoking on admissions for respiratory illness in early childhood. Br. Med. J. 293:303â306, 1986. Cockcroft, D.W., B.A.Berscheid, and K.Y.Murdock. Unimodal distribution of bronchial responsiveness to inhaled histamine in a random human population. Chest 83:751â754, 1983. Colley, J.R.T. Respiratory symptoms in children and parental smoking and phlegm production. Br. Med. J. 2:201â204, 1974. Colley, J.R.T., W.W.Holland, and R.T.Corkhill. Influence of passive smoking an parental phlegm on pneumonia and bronchitis in early childhood. Lancet 2:1031â1034, 1974. Collins, M.H., A.C.Moessinger, J.Kleinerman, J.Bassi, P.Rosso, A.M. Collins, L.S.James, and W.A.Blanc. Fetal lung hypoplasia associated with material smoking: Morphometric analysis. Pediatr. Res. 19:408â412, 1985. Comroe, J.H., R.E.Forster II, A.B.Dubois, W.A.Briscoe, and E.Carlsen. The Lung: Clinical Physiology and Pulmonary Function Tests. Chicago: Year Book Medical Publ., Inc., 1962. pp. 323â364. Comstock, G.W., M.B.Meyer, K.J.Helsing, and M.S.Tockman. Respiratory effects on household exposures to tobacco smoke and gas cooking. Am. Rev. Respir. Dis. 124:143â148, 1981. Dahms, T.E., J.F.Bolin, and R.G.Slavin. Passive smoking: Effects on bronchial asthma. Chest 80:530â534, 1981. Dodge, R. The effects of indoor pollution on Arizona children. Arch. Environ. Health 37:151â155, 1982. Drill, S., and R.Thomas. Evaluation of Short-Term Bioassays to Predict Functional Impairment. Virginia: The MITRE Corp., 1980. Dutau, G., C.Enjaume, M.Petrus, P.Darcos, P Demeurisse, and P. Rochiccioli. Enquete epidemiologue sur la tabagisme passif des enfants de 0 a 6 ans. Arch. Fr. Pediatr. 38:721â725, 1981. Evans, M.J., W.Mayr, R.F.Bils, and C.G.Loosli. Effects of ozone on cell renewal in pulmonary alveoli of aging mice. Arch. Environ. Med. 22:450â453, 1971. Evans, M.J., L.J.Cabral, R.J.Stephens, and G.Freeman, 1975. Transformation of alveolar type 2 cells to type 1 cells following exposure to NO2. Exp. Mol. Pathol. 22:142â150, 1975. Evans, M.J., N.P.Dekker, L.J.Cabral-Anderson, and G.Freeman. Quantitation of damage to the alveolar epithelium by means of type 2 cell proliferation. Am. Rev. Respir. Dis. 118:787â790, 1978. Fergusson, D.M., L.J.Horwood, F.T.Shannon, and B.Taylor. Parental smoking and lower respiratory illness in the first three years of life. J. Epidemiol. Comm. Health 35:180â184, 1981.
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 220 SYMPTOMS Ferris, B.G., Jr., J.H. Ware, C.S.Berkey, D.W.Dockery, A.Spiro III, and F.E.Speizer. Effects of passive smoking on health of children. Environ. Health Perspect. 62:289â295, 1985. Gammage, R.B. and S.V.Kaye. Indoor air and human health, pp. 195â200. Proceedings of the Seventh Life Sciences Symposium, Knoxville, Tennessee, Oct. 29â31, 1984. 430 pp. Gleich, G.J., and P.W.Welsh. Immunochemical and physicochemical properties of tobacco extracts. Am. Rev. Respir. Dis. 120:995â1001, 1979. Guyatt, G.H., and M.T.Newhouse. Are active and passive smoking harmful? Determining causation. Chest 88:445â451, 1985. Harlap, S., and A.M.Davies. Infant admissions to hospital and maternal smoking. Lancet 1:529â532, 1974. Hasselblad, V., C.G.Humble, M.G.Graham, and H.S.Anderson. Indoor environmental determinants of lung function in children. Am. Rev. Respir. Dis. 123:479â485, 1981. Hinds, W.C., and M.W. First. Concentrations of nicotine and tobacco smoke in public places. N. Engl. J. Med. 292:844â845, 1975. Huber, G.L. Small-airways dysfunction in passive smokers. N. Engl. J. Med. 303:392, 1980. Kauffmann, F. Selection bias of PiMZ subjects. Am. Rev. Respir. Dis. 131:800â801, 1985. Kauffmann, F., D.W.Dockery, F.E.Speizer and B.G.Ferris, Jr. Respiratory symptoms and lung function in women with passive and active smoking. Am. Rev. Respir. Dis. 133:A157, 1986. Kentner, M., G.Triebig, and D.Weltle. The influence of passive smoking on pulmonary functionâA study of 1,351 office workers. Prev. Med. 13:656â659, 1984. Knight, A., and A.B.Breslin. Passive cigarette smoking and patients with asthma. Med. J. Aust. 4:194â195, 1985. Lebowitz, M.D., and B.Burrows. Respiratory symptoms related to smoking habits of family adults. Chest 69:48â50, 1976. Lebowitz, M.D., D.B.Armet, and R.Knudson. The effect of passive smoking on pulmonary function in children. Environ. Int. 8:371â373, 1982. Lee, P.N. Passive smoking. Food Chem. Toxicol. 20:223â229, 1982. Leeder, S.R., R.Corkhill, L.M.Irwig, W.W.Holland and J.R.T.Colley. Influence of family factors on the incidence of lower respiratory illness during the first year of life. Br. J. Prev. Soc. Med. 30:203â212, 1976. Levi, R., J.H.Zavecz, J.A.Burke, and C.G.Becker. Cardiac and pulmonary anaphylaxis in guinea pigs and rabbits induced by glycoprotein isolated from tobacco leaves and cigarette smoke condensate. Am. J. Pathol. 106:318â325, 1982. Lundberg, J.M., C.R.Martling, A.Saria, K.Folkers, and S.Rosell. Cigarette smoke-induced airway oedema due to activation of capsaicin- sensitive vagal afferents and substance P release. Neuroscience 10:1361â1368, 1983. McConnochie, K.M., and K.J.Roghmann. Predicting clinically significant lower respiratory tract illness in childhood following mild bronchiolitis. Am. J. Dis. Child. 139:625â631, 1985. McConnochie, K.M., and K.J.Roghmann. Parental smoking, presence of older sibling, and family history of asthma increase risk of bronchiolitis. Am. J. Dis. Child. 140:806â812, 1986.
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 221 SYMPTOMS National Research Council, Committee on the Epidemiology of Air Pollutants. Epidemiology and Air Pollution. Washington, D.C.: National Academy Press, 1985. 224 pp. Pedreira, F.A., V.L.Guandolo, E.J.Feroli, G.W.Mella, and I.P.Weiss. Involuntary smoking and incidence of respiratory illness during the first year of life. Pediatrics 75:594â597, 1985. Pimm P.E., F.Silverman, and R.J.Shephard. Physiological effects of acute passive exposure to cigarette smoke. Arch. Environ. Health 33:201â213, 1978. Purvis, M.R., and R.Ehrlich. Effects of atmospheric pollutants on susceptibility to respiratory infection. II. Effect of nitrogen dioxide. J. Infect. Dis. 113:72â76, 1963. Pullan, C.R., and E.N.Hey. Wheezing, asthma, and pulmonary dysfunction 10 years after infection with respiratory syncytial virus in infancy. Br. Med. J. 284:1665â1669, 1982. Rantakallio, P. Relationship of maternal smoking to morbidity and mortality of the child up to the age of five. Acta Paediatr. Scand. 67:621â 631, 1978. Rylander, R. Environmental tobacco smoke and lung cancer. Eur. J. Respir. Dis. 133(Suppl.):127â133, 1984. Said, G., J.Zalokar, J.Lellouch, and E.Patois. Parental smoking related to ademoidectomy and tonsillectomy in children. J. Epidemiol. Comm. Health 32:97â101, 1978. Schenker, M.B., J.M.Samet, and F.E.Speizer. Risk factors for childhood respiratory disease. The effect of host factors and home environmental exposures. Am. Respir. Dis. 128:1038â1043, 1983. Schilling, R.S.F., A.D.Letai, S.L.Hui, G.J.Beck, J.B.Schoenberg, and A. Bouhuys. Lung function, respiratory disease, and smoking in families. Am. J. Epidemiol. 106:274â283, 1977. Shephard, R.F., R.Collins, and F.Silverman. âPassiveâ exposure of asthmatic subjects to cigarette smoke. Environ. Res. 20:392â402, 1979a. Shephard, R.J., R.Collins, and F.Silverman. Responses of exercising subjects to acute âpassiveâ cigarette smoke exposure. Environ. Res. 19:279â291, 1979b. Sims, D.G., M.A.Downham, P.S.Gardner, J.K.Webb, and D.Weightman. Study of 8-year old children with a history of respiratory syncytial virus bronchiolitis in infancy. Br. J. Med. 1:11â14, 1978. Speizer, F.E., B.Ferris, Y.M.Bishop, and J.Spengler. Respiratory disease rates and pulmonary function in children associated with NO2 exposure. Am. Rev. Respir. Dis. 121:3â10, 1980. Tager, I.B., S.T.Weiss, A.Munoz, B.Rosner, and F.E.Speizer. Longitudinal study of the effects of maternal smoking on pulmonary function in children. N. Engl. J. Med. 309:699â703, 1983. Tager, I.B., S.T.Weiss, B.Rosner, and F.E.Speizer. Effect of parental cigarette smoking on the pulmonary function of children. Am. J. Epidemiol. 110:15â26, 1979. Tashkin, D.P., V.A.Clark, M.Simmons, C.Reems, A.H.Coulson, L.B. Bourque, J.W.Sayre, R.Detels, and S.Rokaw. The UCLA population studies of chronic obstructive respiratory disease. Am. Rev. Respir. Dis. 129:891â897, 1984.
EFFECTS OF EXPOSURE TO ENVIRONMENTAL TOBACCO SMOKE ON LUNG FUNCTION AND RESPIRATORY 222 SYMPTOMS Taylor, L.D., S.D.Greenberg, and P.A.Buffler. Health effects of indoor passive smoking. Tex. Med. 81:35â41, 1985. U.S. Public Health Service. Smoking and Health. A Report of the Surgeon General. DHEW (PHS) Publ. No. 79â50066. Rockville, Maryland: U.S. Department of Health and Human Services, Public Health Service, Office on Smoking and Health, 1979. U.S. Public Health Service. The Health Consequences of Smoking: Cardiovascular Disease. A Report of the Surgeon General. DHHS(PHS) Publ. No. 84â50204. Rockville, Maryland: U.S. Department of Health and Human Services, Public Health Service, Office on Smoking and Health, 1983. 384 pp. U.S. Public Health Service. The Health Consequences of Smoking: Chronic Obstructive Lung Disease. A Report of the Surgeon General. DHHS (PHS) Publ. No. 84â50205. Rockville, Maryland: U.S. Department of Health and Human Services, Public Health Service, Office on Smoking and Health, 1984. 545 pp. Ware, J.H., D.W.Dockery, A.Spiro III, F.E.Speizer, and B.G.Ferris, Jr. Passive smoking, gas cooking, and respiratory health of children living in six cities. Am. Rev. Respir. Dis. 129:366â374, 1984. Weiss, S.T., I.B.Tager, F.E.Speizer, and B.Rosner. Persistent wheeze: Its relation to respiratory illness, cigarette smoking, and level of pulmonary function in a population sample of children. Am. Rev. Respir. Dis. 122:697â707, 1980. Weiss, S.T., I.B.Tager, M.Schenker, and F.E.Speizer. The health effects of involuntary smoking. Am. Rev. Respir. Dis. 128:933â942, 1983. White, J.R., and H.F.Froeb Small-airways dysfunction in nonsmokers chronically exposed to tobacco smoke. N. Engl. J. Med. 302:720â723, 1980. Wiedemann, H.P., D.A.Mahler, J.Loke, J.A.Virgulto, P.Snyder, and R.A. Matthay. Acute effects of passive smoking on lung function and airway reactivity in asthma subjects. Chest 89:180â185, 1986. Woolcock, A.J., J.K.Peat, S.R.Leeder, and C.R.B.Blackburn. The development of lung function in Sydney children: Effects of respiratory illness and smoking. A ten-year study. Eur. J. Respir. Dis. 65(Suppl. 132):1â137, 1984. Zussman, B.M. Tobacco sensitivity in the allergic patient. Ann. Allergy 28:371â377, 1970.
