6
MARIJUANA AND CANCER
Approximately 30 percent of all Americans will develop cancer in their lifetimes. Although two-thirds will eventually die as a result, many will live with cancer for years beforehand. For this reason, researchers not only seek medicines to prevent and cure the disease but also drugs to make life more comfortable for people with cancer.
Is marijuana such a medicine? Several patients and their relatives —many of whom had no prior experience with the drug—have claimed that it is. They include this woman, an author of a 1992 medical marijuana proposal that served as the basis for California 's Proposition 215 (see Chapter 11). At the time she was a member of the California Senior Legislature, an elected body that represents the interests of older Californians. Although she herself has never used the drug, she was convinced to take action by her husband's experience, which she described to the IOM team:
He started chemo. He was ill. He was sicker from the chemo than he was from the cancer, because he wasn't even aware how bad the cancer was. It was not only in the lung; it was in the liver and pancreas. He was given three months.
The oncologist agreed [that] he could use marijuana. I had to do the back alley bit to get some. The first I got wasn't that effective. When I mentioned it to someone, I got a better grade [of mari-
juana]. Two puffs and my husband would go for chemotherapy with a smile and come home happy. He didn't [need to smoke it] again until the next day.
This man died of his cancer but, according to his wife, using marijuana —a drug he would never have tried otherwise—made his last months bearable.
People with cancer who use marijuana say that it benefits them in several ways: by quelling nausea, suppressing vomiting, increasing appetite, relieving pain, and soothing anxiety. Clinical studies indicate that marijuana does none of these things as well as the best medications available, but marijuana has the apparent advantage of treating several symptoms simultaneously. Medicines based on certain chemicals in marijuana could also be used to complement standard medications or to treat patients for whom such therapies have failed.
Considerable clinical evidence indicates that marijuana could yield a variety of useful medicines, especially for nausea, vomiting, and appetite stimulation. THC, in the form of Marinol (dronabinol), has already been used for more than a decade to treat these symptoms in cancer patients and for several years in AIDS patients as well. But other cannabinoids, or combinations of cannabinoids, may prove to be more effective than THC alone. If so, any pharmaceuticals that result from such discoveries could benefit people with AIDS as well as those living with cancer.
CHEMOTHERAPY-INDUCED NAUSEA AND VOMITING
Nausea and vomiting occur when one of several sensory centers, which are located in the brain and the digestive tract, becomes stimulated (see Figure 6.1). It is possible to become nauseous without vomiting or to vomit without feeling nauseous beforehand. Vomiting (also called emesis) involves a complex co-ordination of the digestive tract, respiratory muscles, and posture. Because all of these actions can be readily measured, scientists have been able to reconstruct the chain of physiological events that lead to vomiting.
Conversely, little is known about the actual mechanisms that trigger nausea, which appears to result from brain activity alone.
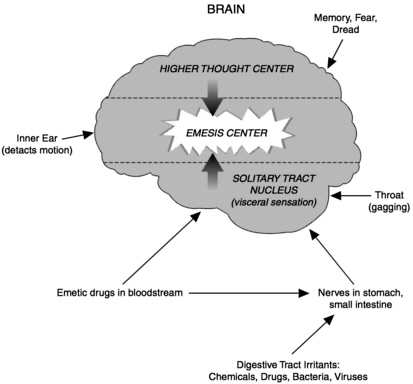
FIGURE 6.1 Emesis pathways. Signals travel to the brain's emesis center, which triggers vomiting, through a variety of routes. Each of these pathways represents a potential site of action for anti-vomiting medications. (Adapted from Bruton LL The Pharmacological Basis of Therapeutics, 9th edition. Hardman et al., eds. 1996, p. 929. New York: McGraw-Hill.)
Since nausea lacks any observable action, researchers studying its origins rely on patients' subjective descriptions of their own feelings. As a result of these limitations, most clinical research aimed at relieving the side effects of chemotherapy focuses on the ability of candidate compounds to prevent or curtail vomiting.
Although researchers do not completely understand how chemotherapy agents cause vomiting, they suspect that the drugs or their digestive byproducts stimulate receptors in key sensory cells. Some agents, including cisplatin, cause nearly every patient to vomit repeatedly; others, such as methotrexate, produce this
effect in a small minority of chemotherapy patients. Vomiting may begin within a few minutes of treatment, as is the case with the drug mustine, or up to an hour after chemotherapy, as occurs with cisplatin. Most clinical trials of antiemetics—medicines that prevent vomiting—tend to be conducted on patients being treated with cisplatin, because drugs that decrease vomiting following cisplatin treatment are likely to work at least as well as other chemotherapy agents.
Researchers have tested several cannabinoids for their ability to suppress vomiting, including two forms of THC (delta-9 and the less abundant delta-8-THC). Two synthetic cannabinoids (nabilone and levonantradol) that activate the same receptors as THC have also been examined as potential antiemetics. All four compounds have proven mildly effective in preventing vomiting following cancer chemotherapy, as will be described. Two additional clinical studies, also to be discussed, provide evidence that, to a limited extent, smoking marijuana helps suppress chemotherapy-induced emesis.
In clinical comparisons THC tended to reduce chemotherapy-induced vomiting better than a placebo. But few trials have used the same chemotherapy agent among all patients, and some contain substantial flaws. For example, one trial tested THC's effectiveness in patients who received methothrexate—a drug that only occasionally causes vomiting. 1 Some experiments compared the efficacy of THC with prochlorperazine (Compazine), one of the most effective antiemetics available in the 1980s, and found that they were similar. With the advent of more effective medications, such as ondansetron (Zofran) and granisetron (Kytril), both serotonin antagonists, these results carry little weight. Even when administered together, THC and prochlorperazine failed to stop vomiting in two-thirds of patients.2
In one particularly well designed study, researchers compared THC with metoclopramide (sold in the United States under various brand names, including Clopra, Maxolon, Octamide PFS, Reclomide, and Reglan), an effective and widely used antiemetic. None of the patients in this study had previously received chemotherapy, so there was no danger that they would vomit simply because they had become conditioned to do so—a reaction that often occurs in people who have undergone several rounds of
chemotherapy. Every patient in this study received the same dose of cisplatin; participants were also randomly assigned to receive either THC or metoclopramide. Seventy-three percent of the patients who received THC vomited at least twice following chemotherapy, compared with only 27 percent of the patients who received metoclopramide. 3
Several additional but less rigorous studies reached similar conclusions: that THC reduces vomiting following chemotherapy, but is not particularly effective in doing so. Nevertheless, the U.S. Food and Drug Administration has approved the drug, in the form of Marinol, for use when chemotherapy-induced nausea and vomiting are not relieved by other antiemetic medications.
Participants in clinical trials of THC have reported several unpleasant side effects, including dry mouth, low blood pressure, sedation, and mood changes. Patients who had no prior experience with marijuana or related drugs were more likely to report psychological discomfort after taking it than those who had tried marijuana previously. On the other hand, advocates of marijuana use for medical purposes maintain that, when such patients receive prior guidance on marijuana's effects, they rarely experience adverse psychological reactions upon using the drug for the first time. Although this claim has not been objectively tested, it may apply equally to the effects of THC, the main psychoactive component in marijuana.
In some clinical trials of THC for antiemesis, patients who underwent the most dramatic mood changes tended to vomit least; other trials found no correlation between THC's psychoactive and antiemetic effects. If they are linked, however, it may be possible to separate the two effects by creating synthetic analogs of the THC molecule. Researchers have found that 11-OH-THC—a breakdown product of THC that forms in the body—is a weaker antiemetic than THC but causes stronger psychological reactions. Perhaps, then, scientists could make additional chemical alterations to the THC molecule to create a chemical analog that controls vomiting better and is less psychoactive than THC.
In fact, such a compound may already exist naturally. Delta-8-THC is a less potent variant of delta-9-THC, the primary psychoactive ingredient in marijuana. In a study of eight children, ages three to 13, delta-8-THC was found to completely block their
chemotherapy-induced vomiting. The only side effect reported was irritability in the two youngest children (ages three and one-half and four years).4
Of the existing chemical analogs of THC, two have been tested in chemotherapy trials.5 Nabilone (marketed in the United Kingdom as Cesamet) and levonantradol, neither of which is approved for sale in the United States, fared similarly to THC in these studies. Both were found to be somewhat effective in preventing vomiting following chemotherapy but not as effective as other antiemetics already on the market.
Although many medical marijuana users claim that smoked marijuana controls nausea and vomiting better than oral THC, no rigorous studies that support this contention have yet been published. In a study that directly compared smoked marijuana with THC, researchers found that both prevented vomiting to a similar degree. Only one in four people in this study of 20 patients achieved complete control of chemotherapy-induced vomiting with either drug.6 Each underwent chemotherapy twice during the trial. During one session, patients smoked a marijuana cigarette and swallowed a placebo pill; at the other session they smoked a placebo cigarette and took a pill containing THC. Patients received the experimental treatments in random order, so approximately half tried marijuana before THC, while the others tried the drugs in the opposite sequence. When asked which form of treatment they preferred, 35 percent of the patients said they favored THC pills, 20 percent chose marijuana, and 45 percent had no preference.
Another preliminary study tested smoked marijuana in cancer patients who were not helped by conventional antiemetic drugs; however, serotonin antagonists—currently considered the most effective antiemetics—were not yet available in 1988 when this study was conducted. 7 Nearly 80 percent of the 56 participants rated marijuana as “moderately effective” or “highly effective,” compared with other antiemetics they had previously used. Since this group of patients varied greatly in terms of their chemotheraputic regimen as well as with regard to their prior experience with marijuana, these results must be considered approximate at best.
Nevertheless, it does make sense that inhaling THC in the
form of smoked marijuana would prevent vomiting better than swallowing a pill. If vomiting were severe or began immediately after chemotherapy, oral THC could not stay down long enough to take effect. Smoking also allows patients to take only the drug they want, one puff at a time, thus reducing their risk of unwanted side effects. But the long-term harms of smoking outweigh its benefits for all but the terminally ill, the IOM team concluded. Instead, they recommended the development and testing of a rapidonset method of delivering THC, such as an inhaler. Similar devices are now used to administer medicine for asthma and other respiratory disorders and are being developed to deliver pain medication.
MALNUTRITION
Wasting and appetite loss affect most cancer patients. At best these conditions diminish quality of life; at worst they hasten death. Depending on the type of cancer, 50 to 80 percent of patients will develop cachexia, a disproportionate loss of lean body tissue. Cachexia occurs most often during the final stages of advanced pancreatic, lung, and prostate cancers. Proteins called cytokines, produced by the immune system in response to the tumor, appear to stimulate this wasting process.
Cachexia also occurs as a result of HIV infection (see Chapter 5), and both cancer and AIDS patients currently receive similar treatments for the condition. Standard therapies for cachexia include intravenous or tube feeding as well as treatment with megestrol acetate (Megace), an appetite stimulant. If the latter causes patients to gain weight, however, it is mostly in the form of fat—not the lean tissue they would have lost through cachexia.
Marijuana is renowned for its ability to stimulate the appetite, otherwise known as “having the munchies.” This effect is due in large part to the action of THC, which has been confirmed in several studies.8 For example, cancer patients who took THC in the form of dronabinol tended to experience a slowing of weight loss and an increase in appetite.9 A study of AIDS patients, however, indicated that megestrol acetate stimulated weight gain more effectively than THC; when used in combination, the two drugs failed to augment each other's effects.10
Both megestrol acetate and dronabinol produce troublesome side effects in some patients. The former can cause hyperglycemia and hypertension; the latter can cause dizziness and lethargy. Because of these drawbacks, medical researchers are pursuing better treatments for cachexia. One promising class of compounds includes agents that can block the actions of the cytokines that promote wasting. Some patients might benefit from a combination therapy consisting of a cytokine blocker along with THC, to stimulate appetite and also, perhaps, to reduce nausea, pain (see Chapter 4), and anxiety.
HARDLY A MAGIC BULLET
Taken as a whole, clinical studies on cannabinoids and cancer pain have reached conclusions similar to those of comparable studies on nausea and malnutrition: marijuana-based treatments, while somewhat effective, underperform conventional medications and cause numerous side effects. The main advantage of cannabinoids lies in their potential to relieve several symptoms at once, but this versatility may come at the price of diminished potency.
For example, powerful opiate medications appear to relieve debilitating pain more effectively than cannabinoids. However, since they appear to reinforce the effects of opiates, cannabinoids may be useful as an adjunct to the stronger drugs. Patients who achieve the same relief with lower doses of opiates should also experience fewer narcotic side effects, such as constipation, drowsiness, and slowed breathing. Moreover, cannabinoids may counteract another common side effect of narcotics—nausea.
Nevertheless, most chemotherapy patients probably would not choose marijuana or THC as an antiemetic. Compared with the highly effective agents currently available, marijuana-based versions appear to offer most people only modest relief. In addition, many patients in clinical studies—in contrast to accounts of several patients who spoke at the IOM's public sessions—have found the side effects of marijuana to be intolerable. In particular, patients who have never smoked marijuana tend to react adversely to the drug's mood-altering properties.
But for the small proportion of patients who respond poorly
to conventional antiemetics, cannabinoids may be a useful alternative. And since they appear to suppress nausea and vomiting through different mechanisms than do other antiemetic compounds, cannabinoids may be able to boost the efficacy of superior medications. For these reasons the IOM study team recommended that researchers test the combined effects of cannabinoids and other antiemetics in suppressing nausea and vomiting in patients who respond poorly to standard treatments.
As described earlier, the IOM team also recommended the development of a rapid-onset drug delivery system that could provide the benefits of inhaling cannabinoids without the harmful effects of smoking. In the meantime for the small minority of cancer patients who have found that only smoking marijuana relieves their chemotherapy-induced vomiting, the IOM team concluded that the harmful effects of doing so for a limited time (i.e., during the course of chemotherapy treatment) might be outweighed by the antiemetic benefits. Such patients, the team suggested, should be evaluated on a case-by-case basis. Those who meet the following conditions could then be provided with marijuana for use under close medical supervision:
-
Documented evidence confirms that all approved medications have failed to provide relief.
-
There is reasonable expectation that the patient's symptoms could be relieved by inhaling cannabinoids.
-
Patients are treated under medical supervision and their treatment is assessed for effectiveness.
-
All such treatments are overseen by an institutional review board, such as is required for all federally funded research involving human subjects. (Institutional review boards consist of scientists with expertise in the areas being researched but who are not involved in the specific study being evaluated. The review board approves studies only after determining that the research will not violate the rights and welfare of human participants.)
For the terminally ill, the dangers of smoking are irrelevant. From a strictly medical standpoint—social consequences notwithstanding —there is no reason to deny marijuana to a dying per-
son. But this step should be seen for what it is—a last resort. Clinical evidence shows that existing treatments for pain, nausea, and malnutrition outperform marijuana in the vast majority of patients. To substitute marijuana for a more effective drug is to practice bad medicine.
NOTES
1. Chang AE, Shiling DJ, Stillman RC, et al. 1979. “Delta-9-tetrahydrocannabinol as an antiemetic in patients receiving high-dose methotrexate: A prospective, randomized evaluation.” Annals of Internal Medicine 91:819-824.
2. Frytak S, Moertel CF, O'Fallon J, et al. 1979. “Delta-9-tetrahydrocannabinol as an antiemetic in patients treated with cancer chemotherapy: A double comparison with prochlorperazine and a placebo.” Annals of Internal Medicine 91:825-830.
3. Gralla RJ, Tyson LB, Borden LB, et al. 1984. “Antiemetic therapy: A review of recent studies and a report of a random assignment trial comparing metoclopramide with delta-9-tetrahydrocannabinol. ” Cancer Treatment Reports 68:163-172.
4. Abrahamov A, Abrahamov A, Mechoulam R. 1995. “An efficient new cannabinoid antiemetic in pediatric oncology.” Life Sciences 56:2097-2102.
5. Steele N, Gralla RJ, Braun DW Jr. 1980. “Double-blind comparison of the antiemetic effects of nabilone and prochlorperazine on chemotherapy-induced emesis.” Cancer Treatment Report 64:219-224; Tyson LB, Gralla RJ, Clark RA, et al. 1985. “Phase I trial of levonantradol in chemotherapy-induced emesis.” American Journal of Clinical Oncology 8:528-532.
6. Levitt M, Faiman C, Hawks R, et al. 1984. “Randomized double-blind comparison of delta-9-THC and marijuana as chemotherapy antiemetics.” Proceedings of the American Society for Clinical Oncology 3:91.
7. Vinciguerra V, Moore T, Brennan E. 1988. “Inhalation of marijuana as an antiemetic for cancer chemotherapy. ” New York State Journal of Medicine 88:525-527.
8. Gorter R. 1991. “Management of anorexia-cachexia associated with cancer and HIV infection. ” Oncology (Suppl.) 5:13-17; Beal JE, Olson RLL, Morales JO, Bellman P, Yangco B, Lefkowitz L, Plasse TF, Shepard KV. 1995. “Dronabinol as a treatment for anorexia associated with weight loss in patients with AIDS.” Journal of Pain and Symptom Management 10:89-97; Beal JE, Olson R, Lefkowitz L, Laubenstein L, Bellman P, Yangco B, Morales JO, Murphy R, Powderly W, Plasse TF, Mosdell KW, Shepard KV. 1997. “Long-term efficiency and safety of dronabinol for acquired immunodeficiency syndrome-associated anorexia.” Journal of Pain Management 14:7-14.
9. Plasse TF, Gorter RW, Krasnow SH, Maontague L, Shepard KV, Wadleigh RG. 1991. “Recent clinical experience with dronabinol.” Pharmacology, Biochemistry, and Behavior 40:665-670.
10. Timpone JG, Wright DJ, Li N, Egorin MJ, Enama ME, Mayers J, Galetto G. 1997. “The safety and pharmacokinetics of single-agent and combination therapy with megestrol acetate and dronabinol for the treatment of HIV wasting syndrome.” The DATRI004 Study Group. AIDS Research and Human Retroviruses 13:305-315.











